WO1993006142A1 - Copolymerisate von hydroxyalkylvinylethern und verwendung von homo- und copolymerisaten von hydroxyalkylvinylethern in wasch- und reinigungsmitteln - Google Patents
Copolymerisate von hydroxyalkylvinylethern und verwendung von homo- und copolymerisaten von hydroxyalkylvinylethern in wasch- und reinigungsmitteln Download PDFInfo
- Publication number
- WO1993006142A1 WO1993006142A1 PCT/EP1992/002041 EP9202041W WO9306142A1 WO 1993006142 A1 WO1993006142 A1 WO 1993006142A1 EP 9202041 W EP9202041 W EP 9202041W WO 9306142 A1 WO9306142 A1 WO 9306142A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- copolymers
- monomers
- vinyl ether
- weight
- ether
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F216/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical
- C08F216/12—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical by an ether radical
- C08F216/14—Monomers containing only one unsaturated aliphatic radical
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/37—Polymers
- C11D3/3703—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C11D3/3711—Polyacetal carboxylates
Definitions
- the invention relates to copolymers of hydroxyalkyl vinyl ethers, addition products of C 2 to C 4 alkylene oxides with hydroxyalkyl vinyl ether and / or polytetrahydrofuran vinyl ether and optionally other copolymerizable monomers and
- copolymers mentioned and the use of cationically or free-radically polymerized hydroxyalkyl vinyl ethers which contain the monomers (a) or (b) in copolymerized form as sole monomers or in a mixture with other copolymerizable monomers, as a viscosity depressant for surfactants and as an additive to detergents and cleaning agents.
- Catalyst uses 0.001 to 5 wt .-% of a practically anhydrous inorganic acid.
- Suitable catalysts are, for example, aluminum chloride, iron chloride, sulfuric acid, hydrochloric acid and boron trifluoride. From US-B-26 407 the radical copolymerization of
- Divinyl compounds such as divinyl ether, divinyl dimethyl silane or divinyl sulfone with monoethylenically unsaturated monomers are known, e.g. Vinyl acetate, maleic anhydride or dimethyl fumarate.
- Suitable catalysts are peroxy compounds, such as di-tert-butyl peroxide, hydrogen peroxide, alkyl hydroperoxides and diacyl peroxides. Ziegler catalysts are also suitable for initiating the polymerization.
- the polymers obtainable in this way form fibers and films and can be used to produce coatings or packaging materials. They are also suitable as lubricants and lubricant additives, adhesives and as crosslinking agents for epoxy resins.
- EP-A-0 359 055 discloses detergent formulations which contain 0.5 to 65% by weight of a surface-active agent and 1 to 80% by weight of a builder, which consists of a polymer which is obtained by radical copolymerization of divinyl ether and maleic anhydride in a molar ratio of 1: 2 and subsequent hydrolysis of the anhydride groups and neutralization is available.
- the copolymers are used in coating and molding compositions to improve the flow or as anti-foaming agents.
- the copolymerization of monovinyl ethers of glycols with vinyl esters of saturated fatty acids having a carbon chain with up to 6 carbon atoms in the presence of peroxides is known from US Pat. No. 3,131,162.
- the copolymers can be vulcanized to rubber-like materials with the addition of conventional vulcanizing aids.
- the object of the present invention is to provide new substances. Another object is to improve the primary and secondary washing action of detergents and to lower the viscosity of aqueous surfactant solutions and detergent slurries.
- copolymers of hydroxyalkyl vinyl ethers which can be obtained by cationically or radically initiated copolymerization of (a) 99 to 1% by weight of hydroxyalkyl vinyl ethers,
- Suitable monomers of group (a) are all hydroxyalkyl vinyl ethers which are derived from compounds having at least 2 OH groups in which the hydrogen atom of an OH group has been replaced by the vinyl group. These compounds can be characterized, for example, using the following general formula:
- R C 2 -C 20 -alkylene, phenylene, substituted with OH groups or with ester groups C 3 - to C 20 alkylene or phenylene groups.
- Individual compounds of this type are, for example, hydroxyethyl vinyl ether, 3-hydroxypropyl vinyl ether, 2-hydroxypropyl vinyl ether, 2-hydroxy-2-methyl-propyl vinyl ether, 2-hydroxybutyl vinyl ether, 3-hydroxybutyl vinyl ether, 4-hydroxybutyl vinyl ether, 2-hydroxy-2-ethylethyl vinyl ether, 3- Hydroxy ⁇
- the following can be used as sachcharides, for example: glucose, methyl glucoside, butyl glucoside, sucrose, palatinose, palatinit, lactose, maltose, fructose, leucrose, panose, dextrins, isomaltose, trehalurose, raffinose, panose.
- sugar acids that can be used are: galacturonic acid, galactonic acid, galactic acid, galactaric acid, gluconic acid.
- vinyl ethers preference is given to using the isomeric hydroxybutyl vinyl ethers, in particular 4-hydroxybutyl-1-vinyl ether and
- the monomers of group (a) make up 99 to 1, preferably 90 to 10% by weight of the copolymers.
- Suitable alkylene oxides are ethylene oxide, propylene oxide, 1,2-butylene oxide, 2,3-butylene oxide and isobutylene oxide.
- Alkylene oxides are added to the hydroxyalkyl vinyl ethers in order to obtain the monomers of component (b).
- Compounds of this type are known.
- the monomers of component (b) can also be prepared by vinylation of polyalkylene glycols.
- Individual monomers of component (b) are for example the addition products which are obtainable by adding 3 mol of ethylene oxide to 1 mol of 3-hydroxypropyl vinyl ether, 4 mol of butylene oxide to 1 mol of 2-hydroxypropyl vinyl ether, 10 mol of ethylene oxide to 1 mol of 2-hydroxy-2-methyl-propyl vinyl ether, 3 mol of ethylene oxide 1 mole of 4-hydroxybutyl vinyl ether, 7 moles
- Suitable monomers of component (b) are also suitable for
- Ethylene propylene glycol monovinyl ether diethylene glycol propylene glycol monovinyl ether.
- block copolymers of alkylene oxides come in
- composition (b) suitable addition products that are obtainable by addition of propylene oxide and then ethylene oxide and optionally butylene oxide to compounds of formula I.
- suitable addition products that are obtainable by addition of propylene oxide and then ethylene oxide and optionally butylene oxide to compounds of formula I.
- polytetrahydrofuran monovinyl ethers which can be obtained by vinylation of polytetrahydrofurans with the molecular weights from 250 to 4,500.
- reaction products of alkylene oxides with monovinyl ethers of polyhydroxy compounds can be used, for example reaction products of 1 mol of glycerol monovinyl ether with 25 mol of ethylene oxide and 1 mol of erythritol monovinyl ether 7 moles of propylene oxide, 1 mole of sorbitol monovinyl ether with 3 moles of butylene oxide, 1 mole of tartaric acid diethyl ester monovinyl ether with 11 moles of ethylene oxide, 1 mole of bis (hydroxymethyl) propionic acid ethyl ester with 25 moles of propylene oxide and 1 mole of glycosemonovinyl ether with 50 moles of ethylene oxide.
- the monomers Preferably used as the monomers
- the alkylene oxides are generally added to 1 mol of hydroxyalkyl vinyl ether in amounts of 1 to 100, preferably 2 to 60, mol. However, even larger amounts of alkylene oxides can be added to the compounds of the formula I to prepare the copolymers according to the invention.
- the addition products of 1 to 100 mol of ethylene oxide with 1 mol of hydroxybutyl vinyl ether are preferably suitable.
- the monomers (b) make up 1 to 99, preferably 10 to 90% by weight of the copolymers.
- Suitable monomers (c) are all other monomers which are copolymerizable with the monomers (a) and (b). Examples include monoethylenically unsaturated C 3 to C 8 carboxylic acids, such as acrylic acid, methacrylic acid, ethacrylic acid, maleic acid, fumaric acid, aconitic acid and itaconic acid.
- esters of the carboxylic acids mentioned with monohydric C 1 to C 18 alcohols or polyhydric C 2 to C 8 alcohols are suitable, for example the following esters may be mentioned: methyl acrylate, ethyl acrylate, tert-butyl acrylate, n-acrylic acid butyl ester, methyl acrylate, methyl methacrylate, methyl methacrylate, n-propyl methacrylate, isopropyl methacrylate, n-butyl methacrylate, isobutyl methacrylate, dimethyl methacrylate, dimethyl maleate, mono maleate maleate, maleate maleate n-butyl ester, maleic acid monoisodecyiester and maleic acid monoisononyl ester. So much for the above
- Comonomers can be used in copolymerization, e.g. Maleic anhydride, acrylic anhydride, methacrylic anhydride and itaconic anhydride.
- the amides are also suitable
- Carboxylic acids such as acrylamide and methacrylamide and nitriles of the carboxylic acids mentioned such as acrylonitrile and methacrylonitrile.
- Further comonomers of group (c) are vinyl esters, which are preferably derived from C 1 - to C 18 -saturated carboxylic acids, for example vinyl formate, vinyl acetate, vinyl propionate, vinyl butyrate, vinyl pivalate and vinyl stearate.
- alkyl vinyl ethers preferably those derived from C 1 to C 18 alcohols, such as methyl vinyl ether, ethyl vinyl ether, propyl vinyl ether, n-butyl vinyl ether, tert-butyl vinyl ether, sec-butyl vinyl ether , isobutyl vinyl ether, dodecyl vinyl ether, octadecyl vinyl ether and styrene.
- the monomers of group (c) which can also be used are the crosslinkers which are customarily used in the radical copolymerization
- crosslinkers contain at least two non-conjugated, ethylenically unsaturated double bonds.
- Suitable crosslinkers are, for example, N, N'-methylenebisacrylamide, polyethylene glycol diacrylates and polyethylene glycol dimethacrylates, which are each derived from polyethylene glycols with a molecular weight of 126 to 8500, trimethylolpropane triacrylate, trimethylolpropane trimethacrylate, and ethylene glycol diacrylate diacrylate diol acrylate, block diacrylate diacrylate diacrylate diacrylate, Propylene oxide, double or
- Methacrylic acid esterified polyhydric alcohols such as glycerol or pentaerythritol, triallylamine, tetraallylethylene diamine, divinylbenzene, diallyl phthalate, polyethylene glycol divinyl ether,
- Trimethylolpropane dially ether polyethylene glycol divinyl ether, butanediol divinyl ether, pentaerythritol triallyl ether and / or
- Water-soluble crosslinking agents are preferably used, for example N, N'-methylene-bisacrylamide, polyethylene glycol diacrylates, polyethylene glycol dimethacrylates, pentaerythritol triallyl ether and / or divinyl urea.
- Crosslinkers which are preferably used are divinyl ethers of C 2 -C 20 -diols, polyalkylene glycols and diphenols, such as, for example 1,2-ethylene glycol divinyl ether, 1,4-butanediol divinyl ether,
- crosslinking agents are used in the radical polymerization, the amounts used are 0.1 to 15, preferably 0.2 to 5,% by weight.
- Polytetrahydrofuran divinyl ethers with molecular weights of 250 to 4500 can also be used.
- the copolymerizable monomers (c) used are preferably monomers containing at least two vinyl ether groups. Divinyl ethers are preferred, it being possible to use all divinyl ethers, for example the divinyl ethers of C 2 -C 20 -alkylene diols or
- Divinyl ether of polyalkylene glycols with a molecular weight up to 35,000 of the polyalkylene glycol segment preference is given to using butanediol divinyl ether, in particular 1,4-butanediol divinyl ether, 1,6-hexanediol divinyl ether and 1,4-bis (hydroxymethyl) cyclohexyl divinyl ether.
- the amounts here are up to 20% by weight, based on the
- C 2 - bis is preferably used
- C 20 diols for example ethylene glycol, propylene glycol, 1,4-butanediol, 1,2-butanediol, 1,3-butanediol, 1,6-hexanediol and polyalkylene glycols with molecular weights up to 35,000, for example
- the copolymers can contain the monomers in statistical distribution or in the form of blocks. These monomers are present in the copolymerization, if used, in amounts of 0.1 to 20% by weight.
- Polyalkylene oxides can be formed in the alkoxylation of hydroxyalkyl vinyl ethers (monomers a).
- the monomers containing at least two vinyl ether groups and the compounds having at least two OH groups for example alkylene glycols, can be used together in order to obtain polymers with high molecular weights.
- this then requires that vinyl groups and OH groups in the monomer mixture be approximately equimolar
- the monomers (a), (b) and optionally (c) can be copolymerized both by free radicals and by cationic means.
- a C-C linkage occurs via the double bonds of the monomers with the formation of polymers.
- initiators can be soluble or insoluble in water.
- Water-soluble initiators are, for example, inorganic peroxides, such as potassium, sodium and ammonium peroxodisulfate and hydrogen peroxide.
- organic peroxides hydroperoxides, peracids, ketone peroxides, perketals and peresters, e.g. Methyl ethyl ketone hydroperoxide, cumene hydroperoxide, tert-butyl hydroperoxide,
- Redox systems are also suitable which contain a reducing component in addition to a peroxy compound.
- Suitable reducing components are, for example, cerium III and iron II salts, sodium sulfite, sodium bisulfite, sodium dithionite, ascorbic acid and sodium formaldehyde sulfoxylate.
- Suitable initiators are preferably selected by using compounds which form free radicals and which have one at the polymerization temperature chosen in each case
- Suitable initiators are also azo compounds such as
- Radical initiator or a mixture of such polymerization initiators.
- the amount of initiator used has a considerable influence on the molecular weight of the homopolymers and copolymers formed.
- the radical polymerization is preferably carried out in the presence of a diluent.
- a diluent can be solvents for the monomers and the polymers or only solvents for the monomers.
- Suitable solvents are, for example, ethers, such as diethyl ether, dibutyl ether, diisobutyl ether,
- the alkylene oxide units can be used in the reaction products
- reaction products are components of liquid detergents. Since the polymers are used in detergents, they are advantageously prepared in those described
- Detergents are used as diluents.
- Preferred solvents for the polymerization are, for example, reaction products of monovalent C 1 -C 28 -alipnatic
- Propylene oxide and / or butylene oxide for example the addition products of 3 to 11 moles of ethylene oxide to one mole of a C 13 / C 15 alcohol, addition products of 5 to 15 moles of ethylene oxide to 1 mole of nonylphenol, addition products of 7 to 11 moles of ethylene oxide and 3 to 5 Moles of propylene oxide to 1 mole of oleyl alcohol and addition products of 5 to 15 moles of ethylene oxide to 1 mole of stearyl alcohol or tallow fatty alcohol.
- ethylene oxide, propylene oxide and butylene oxide with polyhydric alcohols, for example glycol, diethylene glycol, triethylene glycol, tetraethylene glycol, propylene glycol, dipropylene glycol, block copolymers of ethylene oxide and propylene oxide, glycerol and oligoglycerols are also suitable. Particularly preferred from this group of
- Compounds are diethylene glycol and polyethylene glycols with degrees of condensation up to 25.
- C 1 to C 6 alcohols such as
- the polymerization of the monomers of groups a) to c) can optionally also be carried out in the presence of regulators.
- Suitable regulators are, for example, mercapto compounds, such as mercaptoethanol, mercaptopropanol, mercaptobutanol, mercaptoacetic acid, mercaptopropionic acid, butyl mercaptan and dodecyl mercaptan.
- allyl compounds such as allyl alcohol, aldehydes, such as formaldehyde, acetaldehyde, propionaldehyde, n-butyraldehyde and isobutyraldehyde,
- regulators are used in particular if larger amounts of crosslinking monomers of group c) are used in the polymerization. In such cases, the use of the regulators results in water-soluble polymers or polymers which can be easily dispersed in water.
- the copolymerization can also be carried out without a diluent.
- This process variant is particularly suitable if, as component (b) monomer, higher ethoxylated hydroxyalkyl vinyl ether is used, e.g. in reaction products of 4-hydroxybutyl vinyl ether with 3, 7, 11, 25 or 50 moles of ethylene oxide per mole of 4-hydroxybutyl vinyl ether and as a monomer of component (c) dimethyl maleate or diethyl maleate.
- the progress of the copolymerization can be determined by means of a decrease in the vinyl ether content in the reaction mixture
- Polymers are split in an aqueous acidic medium.
- the resulting acetaldehyde can easily be removed from the aqueous polymer solution using steam distillation. In this way it is possible to produce copolymers that are virtually free of residual monomers.
- the copolymers can be modified after the polymerization. For example, it is possible to use copolymers containing the monomers (a), (b) and, as monomer (c), an ester of an ethylenically unsaturated carboxylic acid in copolymerized form, by treatment with aqueous bases, e.g. Sodium hydroxide solution under ester hydrolysis to a carboxylate group-containing
- Modify copolymer Another possibility is to allow hydroxylamine, ammonia or amines to act in copolymerized form according to the invention in copolymerized form with ester groups, so that hydroxamic acids or carboxamides are obtained. A large number of subsequent reactions for modifying the copolymers according to the invention are possible here.
- a polymer main chain is obtained in which the monomer units are linked to one another via acetaldehyde acetals.
- Cationic copolymerization of the monomers of groups (a) and (b) can be initiated with the aid of all organic or inorganic acid-reacting substances.
- Suitable cationic initiators are, for example, oxalic acid, tartaric acid, adipic acid, succinic acid, succinic anhydride, citric acid, formic acid, acetic acid, propionic acid, malic acid, mono- or poly-halogenated carboxylic acids, such as trifluoroacetic acid or trichloroacetic acid, hydrogen chloride, hydrogen bromide, hydrogen chloride, phosphoric acid, sulfuric acid, sulfuric acid, hydrogen sulfide -Toluenesulfonic acid, boric acid, ascorbic acid, acidic aluminum oxide, aluminum sulfate, potassium aluminum sulfate, iron (II) sulfate, iron (III) sulfate, aluminum oxide, titanyl sulfate, iron III chloride, boron
- the initiators for the cationic polymerization are usually used in amounts of 0.001 to 20, preferably 0.01 to 1% by weight, based on the monomers of groups (a) and (b).
- the copolymerization is very exothermic. Depending on the activity of the initiator used, the reaction temperatures are between -20 and 250, preferably 0 to 200 ° C.
- the monomers (a) and (b) react in the presence of 0.1 to 5% by weight of p-toluenesulfonic acid at 40 ° C. in an exothermic reaction within 10 minutes. completely. If 0.2% by weight of oxalic acid is used to initiate the copolymerization, the copolymerization is within 90 ° C.
- the copolymerization usually produces colorless oils and resins that are very stable when using oxalic acid and tartaric acid as catalysts.
- the temperature resistance of the copolymers thus produced ranges up to 250 ° C.
- the cationic polymerization of the monomers (a) and (b) is preferably carried out in the absence of a solvent. However, it is also possible to work in the presence of inert diluents. This can be advantageous in certain cases because the heat of reaction can then be easily removed using the evaporating diluent. If a strong acid is used as the initiator, a solution of the initiator in a diluent is preferably used, for example a 0.005 to 10 wt. %, particularly preferably a 0.01 to 5% by weight solution of p-toluenesulfonic acid in toluene.
- Suitable diluents for cationic copolymerization are all diluents which do not have any functional groups which can react with vinyl ethers.
- Diluents which are readily available in anhydrous form and are not hygroscopic are preferably used.
- suitable diluents are ethyl acetate, diethylene glycol diethyl ether, ethoxyethyl acetate, butoxyethyl acetate, aliphatic hydrocarbons such as pentane, hexane, cyclohexane, n-heptane, n-octane and isooctane and
- aromatic hydrocarbons such as toluene, xylene, mesitylene, tetraiin and ansiol.
- Suitable solvents are also
- the copolymerization can be carried out, for example, as a one-pot reaction.
- the monomers of groups (a) and (b) and optionally (c) can be mixed in a reaction vessel with the exclusion of atmospheric moisture, mixed with initiator and heated to the required reaction temperature.
- the total amount of initiator is placed in the reaction vessel together with 10% of the monomer mixture of components (a), (b) and optionally (c) to be polymerized at 20 ° C. and the polymerization reaction is preferably started under an inert gas atmosphere by heating the reaction vessel in the reaction vessel submitted components. The mixture is stirred and also during the subsequent copolymerization.
- the remaining monomer mixture of the compounds of components (a), (b) and (c) is added continuously or batchwise to the extent that the heat of polymerization can be safely removed. If If oxalic acid, tartaric acid or citric acid is used as the initiator, temperatures of about 70 to about 160 ° C. are required to start the copolymerization of the monomers (a), (b) and optionally (c). The acids then dissolve in the monomer mixture to form a homogeneous solution and the polymerization begins.
- the heterogeneous reaction mixture is heated to a temperature in the range from 100 to 250 ° C. until the polymerization starts.
- the initiator is either separated off or deactivated. Deactivation of the catalyst
- protic solvents are subject to hydrolysis with molecular weight reduction.
- the reaction mixture is added after the copolymerization and
- reaction mixture optionally after cooling the reaction mixture with alkalis, preferably sodium hydrogen carbonate, sodium hydroxide solution,
- Potassium hydroxide solution sodium carbonate, potassium carbonate, ammonium carbonate, amines such as ammonia, methylamine, dimethylamine, trimethylamine, ethylamine, diethylamine, dibutylamine, tributylamine, ethanolamine, diethanolamine, triethanolamine, morpholine or salts from strong bases and weak acids, such as sodium acetate, sodium formate or potassium hydrogen carbonate .
- amines such as ammonia, methylamine, dimethylamine, trimethylamine, ethylamine, diethylamine, dibutylamine, tributylamine, ethanolamine, diethanolamine, triethanolamine, morpholine or salts from strong bases and weak acids, such as sodium acetate, sodium formate or potassium hydrogen carbonate .
- Calcium oxide, calcium hydroxide, basic aluminum oxide and basic ion exchangers are also suitable for initiators. Insoluble initiators can easily be removed by filtration.
- the copolymer After deactivation or separation of the initiator, the copolymer can be converted into a polymer solution or a dispersion by adding water.
- the copolymer solutions can be purified by subjecting them to steam distillation and volatile components
- Copolymers that have been subjected to steam distillation do not change color if they are subjected to an alkaline aftertreatment.
- the copolymers can be 99 to 1, preferably 90 to
- the monomers are preferably selected such that water-soluble or dispersible copolymers are formed.
- the solubility of the copolymers obtained in water can be influenced by varying the mixing ratios of monomers of group (b) and monomers of group (a). Depending on the ratio of the copolymerized monomers of groups (a) and (b) in the form of 1% aqueous solutions at a pH of 7.5, the copolymers have cloud points of 0 to 100, preferably 5 to 85 ° C. By increasing the molecular weight, the cloud point of the copolymers can be reduced with a constant ratio of the monomers of groups (a) and (b).
- the molecular weight of the copolymers can be determined using the
- Initiator amount can be influenced. An increasing amount of initiator leads to copolymers with lower molecular weight.
- the molecular weight of the copolymers can be influenced by the use of copolymerizable monomers of group (c).
- group (c) divinyl ethers and compounds containing at least two OH groups are suitable comonomers (c) which increase the molecular weight of the copolymers in the cationic copolymerization of (a) and (b).
- polyalkylene glycols are present as impurities in the preparation of the monomers (b) by alkoxylation of the monomers of group (a) and cannot be separated off easily, these mixtures of alkoxylated hydroxyalkyl vinyl ethers and polyalkylene glycols can be used in the copolymerization.
- Polyalkylene glycols act as diols in the polymerization of monomers of groups (a) and (b) to limit the molecular weight. In this case, one preferably adds one of the OH groups equimolar amount of vinyl ether groups in the form of div vonnyl ethers, so that copolymers with higher molecular weights are obtained.
- the divinyl ethers are used in a stoichiometric ratio to the polyalkylene glycols, they have a molecular weight limiting effect. If they are also used in the copolymerization, the divinyl ethers are used in amounts of up to 10% by weight.
- the copolymers of cationically polymerized monomers (a), (b) and optionally (c) are not stable in aqueous solutions at pH values below 7. Under these conditions, the copolymers hydrolytically cleave.
- copolymers which are prepared by cationic polymerization of the monomers (a) and (b) are in aqueous solution at a pH of 6.2 within 10 to 20 days, at a pH of 5.3 within Hydrolysed for 1 to 10 days, at a pH of 4.7 within 1 to 30 hours and at a pH of 2.5 within 1 to 90 minutes. Attention should therefore be paid to the sensitivity of the copolymers to acids during processing and storage.
- the copolymers are used as additives for detergents.
- the free-radically or cationically polymerized homopolymers of monomers (a) and (b) can also be used as detergent additives. Such homopolymers belong to the prior art.
- copolymers of hydroxyalkyl vinyl ether and other monomers copolymerizable therewith can be used as detergent additives.
- These copolymers contain at least 1% by weight of at least one hydroxyalkyl vinyl ether of component (a) and at least one monomer of component (c) mentioned above. Also come as an additive to detergents
- the monomers of components (b) are in these
- Copolymers contain at least 1 wt .-% in copolymerized form.
- the copolymers which contain either (a) or (b) as characteristic monomers can be prepared by cationic or by radical copolymerization. Some polymers of this type are known from the prior art cited above for other applications. Are of particular interest
- Copolymers which are preferably used and contain monomers of group (b) as a characteristic building block are, for example, copolymers of (1) the reaction product of 1 mole of 4-hydroxybutyl vinyl ether with 3 moles, 7 moles, 11 moles, 25 moles and 50 moles of ethylene oxide and
- maleic acid diethyl ester maleic acid dimethyl ester, acrylic acid, methacrylic acid, maleic anhydride, methyl (meth) acrylate, butyl (meth) acrylate, t-butyl acrylate or
- copolymers according to the invention and the cationically or radically polymerized hydroxyalkyl vinyl ethers which
- Monomers (a) or (b) contained as copolymerized monomers or in copolymerized form with other copolymerizable monomers have K values of 9 to 100, preferably 10 to 70 (determined according to H. Fikentscher in 1% solution in tetrahydrofuran at 25 ° C).
- Viscosity lowerers can be used for surfactants. At the
- Surfactant formulations with water as is customary in the field of commercial laundries or in the production of detergent formulations, often have a concentration range with a very high viscosity. This phenomenon is often troublesome when transporting the formulations in pipelines, metering with the aid of pumps and the dissolving speed in the wash liquor. If the viscosity is high, a great deal of mechanical energy must be expended to process the surfactants. In addition, the further dissolution process of the surfactants is associated with an increased expenditure of time due to the high viscosity of a partially diluted surfactant. The increase in
- Viscosity of surfactants when diluted with water occurs above all with nonionic surfactants, especially with polyalkoxylated compounds. If the copolymers described above are added to the surfactants in amounts of 0.1 to 50, preferably 0.5 to 10% by weight, the viscosity of such mixtures can be significantly reduced on dilution.
- the homopolymers and copolymers to be used according to the invention are preferably first dissolved or dispersed in the concentrated surfactant. The mixture is then diluted by adding water.
- the surfactant concentrate mixed with the polymers to be used according to the invention can also be introduced into an aqueous washing liquor. However, it is also possible to add a highly viscous, dilute aqueous surfactant solution to the polymers to be used according to the invention in order to subsequently lower the viscosity.
- copolymers according to the invention are added
- the detergents and cleaning agents contain essential ones
- the detergents and cleaning agents can be in the form of a powder or a liquid formulation.
- the copolymers to be used according to the invention are particularly suitable for the preparation of liquid detergent formulations.
- Suitable anionic surfactants are, for example, sodium alkylbenzenesulfonates, fatty alcohol sulfates and fatty alcohol polyglycol ether sulfates. Individual compounds of this type are, for example, C 8 bis
- anionic surfactants are sulfated fatty acid alkanolamides, fatty acid monoglycerides or reaction products of 1 to 4 moles of ethylene oxide with primary or secondary fatty alcohols or alkylphenols.
- anionic surfactants are sulfated fatty acid alkanolamides, fatty acid monoglycerides or reaction products of 1 to 4 moles of ethylene oxide with primary or secondary fatty alcohols or alkylphenols.
- Fatty acid esters or fatty acid amides of hydroxy or amino carboxylic acids or sulfonic acids such as, for example, the fatty acid sarcosides, glycolates, lactates, taurides or isothionates.
- the anionic surfactants can be in the form of sodium, potassium and Ammonium salts as well as soluble salts of organic bases, such as mono-, di- or triethanolamine or other substituted amines.
- the anionic surfactants also include the usual soaps, ie the alkali salts of the natural fatty acids.
- Nonionic surfactants e.g. Addition products of 3 to 40, preferably 4 to 20, moles of ethylene oxide and 1 mole of fatty alcohol, alkylphenol, fatty acid, fatty amine, fatty acid amide or alkanesulfonamide can be used.
- the addition products of 5 to 16 moles of ethylene oxide with coconut or tallow fatty alcohols, with oleyl alcohol or with synthetic alcohols with 8 to 18, preferably 12 to 18, carbon atoms and on are particularly important
- Mono- or dialkylphenols with 6 to 14 carbon atoms in the alkyl radicals are also of interest, in particular if they are used together with water-soluble nonionic or anionic surfactants.
- non-ionic surfactants that can be used are the water-soluble adducts of ethylene oxide with 20 to 250 ethylene glycol ether groups and 10 to 100 propylene glycol ether groups with polypropylene glycol ether, alkylene diaminopolypropylene glycol and alkyl polypropylene glycols with 1 to 10 carbon atoms in the alkyl chain, in which the polypropylene glycol ether chain functions as a hydrophobic residue.
- Nonionic surfactants of the amine oxide or sulfoxide type can also be used.
- the foaming power of the surfactants can be increased or decreased by combining suitable types of surfactants. A reduction can also be achieved by adding non-surfactant-like organic substances.
- the liquid, aqueous detergents contain 10 to 50% by weight of surfactants. You can contain an anionic or nonionic surfactant in the amount specified. However, it is also possible to use mixtures of anionic and nonionic surfactants. In such a case, the content of anionic surfactants in the liquid detergent is chosen from 10 to 30% by weight and the content of nonionic surfactants in the liquid detergent from 5 to 20% by weight, based on the total detergent formulation.
- the polymers can also be used in concentrated liquid detergents, compact detergents and zeolite-containing detergents. They are also used in detergents containing layered silicates. Liquid detergents contain as essential components the copolymers to be used according to the invention in amounts of 0.1 to 20, preferably 1 to 10% by weight and optionally water in amounts of 10 to 80.
- Liquid detergents may also contain other substances for modification. These include, for example, alcohols, such as ethanol, n-propanol and isopropanol. If used, these substances are used in amounts of 3 to 8% by weight, based on the total detergent formulation.
- the liquid detergents may also contain hydrotropes. This includes compounds such as 1,2-propanediol, cumene sulfonate and toluene sulfonate. If such compounds are used to modify the liquid detergent, their amount, based on the total weight of the liquid detergent, is 2 to 5% by weight. In many cases, addition of complexing agents has also proven to be advantageous for modification.
- Complexing agents are, for example, ethylenediaminetetraacetic acid, nitrilotriacetate and isoserinediacetic acid, and phosphonates, such as aminotrismethylenephosphonic acid, hydroxyethane diphosphonic acid, ethylenediaminetetramethylenephosphonic acid and their salts.
- the complexing agents are used in amounts of 0.1 to 10% by weight, based on the liquid detergent.
- the liquid detergents can also contain citrates, di- or triethanolamine, opacifiers, optical brighteners, enzymes, perfume oils and dyes. If they are used to modify the liquid detergents, these substances are present together in amounts of up to 5% by weight.
- the liquid detergents are preferably phosphate-free. However, they can also contain phosphates, e.g. Pentasodium triphosphate and / or
- Tetrakai iumpyrophosphate Tetrakai iumpyrophosphate. If phosphates are used, the proportion of phosphates in the total formulation of
- the liquid detergents can also contain Na aluminum silicate (Zeolite A). The amounts used are generally 10 to 30% by weight.
- the copolymers according to the invention have a homogenizing and stabilizing effect owing to their good dispersing properties.
- the liquid detergents described above have the advantage over the powder detergents that they are easy to dose and a very good one at lower washing temperatures
- Liquid detergents contain high levels of detergent substances, which remove dirt from the textile fabric at washing temperatures of 40 to 60 ° C.
- the dispersing properties of polymers could not previously be used in aqueous liquid detergents because of the high electrolyte concentrations in the detergents
- Secondary washing action is understood to mean the effects which arise from the deposition of the dirt detached from the fabric on the fabric in the wash liquor.
- the secondary washing effect can only take place after several washes, e.g. 3, 5, 10 or even 20 washes that become visible in one
- Phosphate-free detergent formulations especially concentrated, powdery compact detergents can contain zeolite and / or layered silicates in the form of crystalline or amorphous powdered hydrated soda water glasses in addition to the usual surfactant components as builders.
- Detergent and cleaning agent formulations usually contain surfactants and optionally builders. This information applies to both liquid and powder detergent formulations. Examples of that
- composition of detergent formulations that are common in Europe, the USA and Japan can be found, for example, in Chemical and Engn. News, Vol. 67, 35 (1989) tabulated and in Ullmann's Encyclopedia of Industrial Chemistry, Verlag Chemie, Weinheim 1983, 4th edition,
- the percentages in the examples are% by weight.
- the K values were determined according to H. Fikentscher, Zellulosechemie, Vol. 13, 58-64 and 71-74 (1932), and - unless otherwise stated - in 1% by weight solution in tetrahydrofuran at 25 ° C.
- PEG 300 polyethylene glycol with a molecular weight of 300 FG: solids content Detergents containing crystalline layered silicates are described in: Soaps, oils, fats, waxes, 116, 20 (1990), pp. 805-808. VEG: iodometrically detectable vinyl ether end groups in
- the reaction mixture is then allowed to cool to 20 ° C. and is neutralized by adding 28 g of solid, anhydrous sodium carbonate. Stirring at 20 ° C. for 5 hours gives a solution of the neutralized polymer in a mixture of water and polyethylene glycol. Small portions of undissolved sodium carbonate are filtered off through a filter cloth. The polymer content of the solution is 26%.
- the copolymer has a K value of 61 (measured in 1% strength aqueous solution at 25 ° C.).
- a copolymer is prepared by adding a template from a
- the solids content of the reaction solutions is given in Table 3.
- the copolymers no longer contain any detectable vinyl ether groups.
- the exothermic copolymerization begins, which leads to a rise in the temperature of the reaction mixture to 120 to 190 ° C. within 2 minutes.
- the reaction mixture is cooled to 20 ° C., 1 g of finely crushing sodium hydrogen carbonate is added in each case and it is suspended in the reaction mixture by stirring. Then 100 g of water are added in one portion and the volatile constituents are removed
- a monomer mixture of 15 g of 4-hydroxybutyl vinyl ether, 35 g of the addition product of 3 moles of ethylene oxide and 1 mole of 4-hydroxybutyl vinyl ether and 0.7 g of 1,4-butanediol divinyl ether was polymerized with the difference that 1/5 of the monomer mixture is heated together with oxalic acid to a temperature of 120 ° C. and, after the start of the polymerization, the remaining amount of monomer is metered in at such a rate that the temperature of the reaction mixture is kept at 120 ° C. during the polymerization.
- a copolymer with a K value of 35 is obtained.
- the cloud point of a 1% strength aqueous solution of the copolymer is 25 ° C.
- Example 29 is repeated with the exception that the copolymerization is carried out in the absence of 1,4-butanediol divinyl ether with 5 g of 4-hydroxybutyl vinyl ether and 45 g of the addition product of
- Example 29 According to the preparation instructions given in Example 29, the starting materials given in Table 8 are copolymerized cationically. 0.15 g of oxalic acid was used as the polymerization initiator in all cases. After the end of the copolymerization, 1 g of sodium hydrogen carbonate was added to neutralize the oxalic acid. As indicated in Example 29, a polymer solution is also produced in the preparation of the polymers 31-37 by adding 100 g of water in each case. The cloud points of 1% aqueous solutions and the K values of the copolymers are in
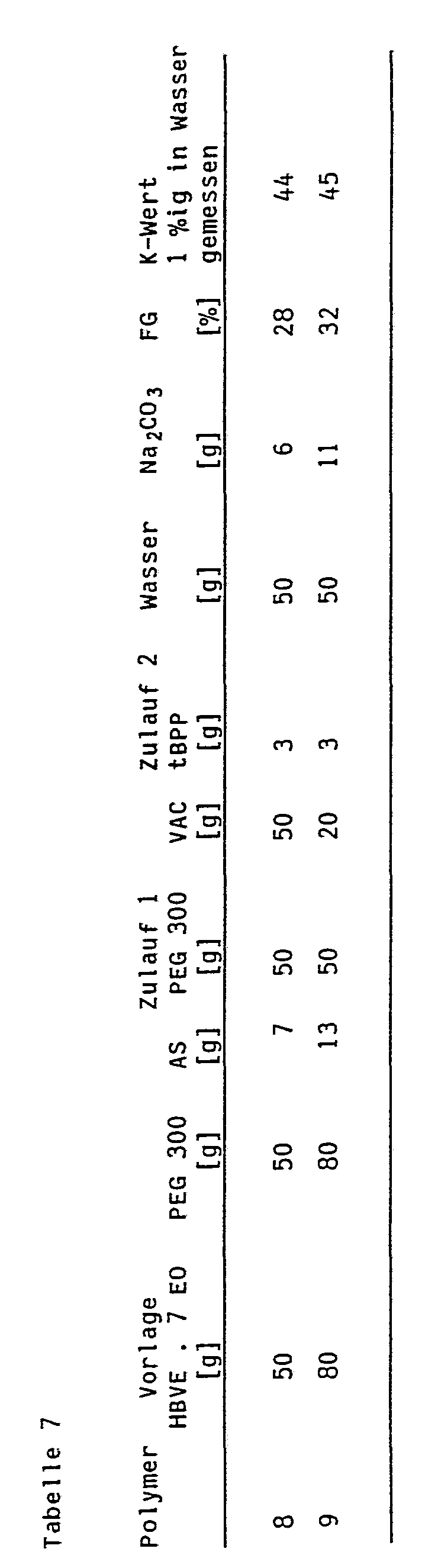
- Polymers 1 to 3 These polymers were obtained according to the general preparation instructions for Examples 1-5 with the amounts of starting materials shown in Table 6. The K values of the polymers are also given in Table 6.
- a copolymer is prepared by adding 106 g of the addition product of 7 mol of ethylene oxide to 1 mol of 4-hydroxybutyl vinyl ether in 106 g a nonionic surfactant (reaction product of 1 mol
- tert-butyl perpivalate is added.
- a copolymer having a K value of 14 is obtained.
- 100 g of water and 6 ml of 50% strength aqueous sodium hydroxide solution are then added to the copolymer, the ester groups of the copolymer being converted into carboxylate groups.
- the reaction mixture consists of 29% copolymer, 29% nonionic surfactant and 42% water.
- the reaction mixture consists of 24% copolymer, 24% nonionic surfactant and 52% water.
- a mixture is added as feed 1 to a template from 100 g of the addition product of 7 mol of ethylene oxide and 1 mol of hydroxybutyl vinyl ether and 100 g of polyethylene glycol with a molecular weight of 300
- Example 1-5 1/10 of a mixture consisting of 122 g of an addition product of 11 mol of ethylene oxide with 1 mol of methanol, 100 g of an addition product of 7 mol of ethylene oxide with 1 mol of 4-hydroxybutyl vinyl ether and 22 g of acrylic acid are introduced and admixed with 0.3 g of tert-butyl perpivalate.
- the polymerization is started by heating to 75 ° C. and the remaining monomer mixture is added so quickly that the temperature does not exceed 80 ° C. After stirring at 75 ° C. for a further hour, the vinyl ether content is determined to be 0.006 mol by means of iodine titration.
- the polymer is neutralized by adding a solution of 40 g of sodium carbonate decahydrate and 75 g of water. The resulting polymer solution is adjusted to a solids content of 55%. The non-neutralized polymer has a K value of 14. Polymer 15-17
- a surfactant-free polymer is produced by producing a mixture of an addition product of ethylene oxide with 4-hydroxybutyl vinyl ether and diethyl maleate and polymerizing this mixture at 75 ° C. using tert-butyl perpivalate. After adding 100 g of water to the polymer, water vapor is introduced into the solution and the polymer is then subjected to an alkaline aftertreatment at 20 ° C. by adding 25% aqueous sodium hydroxide solution.
- the following table shows the starting materials and quantities:
- Polymer 18-20 In the apparatus of Example 1-5, 1/10 of a mixture consisting of the addition product of ethylene oxide with 4-hydroxybutyl vinyl ether, polyethylene oxide with a molecular weight of 300 and acrylic acid is introduced at 75 ° C. and by adding 1/10 of the Initiator amount initiated for polymerization. The remaining monomer mixture is metered in so quickly that the temperature can be kept at 80.degree. After the polymerization is complete, the K value of the polymer is determined and the polymer is quantitatively neutralized by adding stoichiometric amounts of aqueous soda solution. Dilution with water results in a clear solution with the stated polymer contents:
- the copolymers obtained according to Examples 30, 32, 34 and 36 were examined for hydrolytic degradation. For this purpose, the decrease in viscosity of 5% solutions of the polymers in water at the corresponding pH values was monitored. An Ubbelohde viscometer was used as the measuring device. The measurements were carried out at 25 ° C. The following pH values were set: pH of 2.5: 1% aqueous acetic acid
- pH of 4.6 equimolar mixture of acetic acid and sodium acetate pH of 5.3: addition of 0.1% acetic acid
- pH of 6.2 addition of 0.1% acetic acid.
- the decrease in run-out time in the viscometer at 25 ° C was characterized by the half-life: T half and the end value of the run-up time (time for complete depolymerization): T end .
- Examples 38-42 A mixture of the anhydrous surfactant A (addition product of 7 mol of ethylene oxide and 1 mol of a C 13 / C 15 alcohol mixture) and the polymer 4 described above are prepared. The proportion of polymer 4 in the mixture is 5%. Table 10 shows the viscosities of the mixture of surfactant A and polymer 4 described at different water contents. As can be seen from this, polymer 4 acts as a viscosity lowering agent for the water-containing surfactant A.
- a 40% aqueous solution of surfactant A is with the in
- Surfactant A is diluted to 40% by weight by adding water and the viscosity of the mixture is measured. It is 744 mPas.
- the non-hydrolyzed polymer 4 ie the copolymer obtained by radical polymerization of the addition product of 7 moles of ethylene oxide with 1 mole of hydroxybutyl vinyl ether and diethyl maleate, is made up in the form of a 5% solution in anhydrous surfactant A by adding water to 40% by weight. diluted. The viscosity of this
- the polymers to be used according to the invention are important auxiliaries in the production of phosphate-reduced and phosphate-free washing and
- the slurry concentration in the crutcher can be increased to at least 80%. This means better economy through cheaper utilization of the spray tower and a saving in energy because less water has to be evaporated.
- the homogenizing and viscosity-lowering effect of the polymers to be used according to the invention is illustrated below by viscosity measurements on detergent formulations.
- a rheometer from Physika serves as the measuring device. Measuring system is a Z 3 DIN type.
- a slurry of 80% by weight detergent components and 20% by weight water is used as the detergent slurry.
- the polymers given in Table 13 in an amount of 2% by weight, based on the detergent formulation, are used as viscosity-reducing additives.
- the detergent for making the slurry is composed as follows:
- the whiteness differences on the individual fabrics are given before and after washing.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Detergent Compositions (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE59204523T DE59204523D1 (de) | 1991-09-13 | 1992-09-04 | Copolymerisate von hydroxyalkylvinylethern und verwendung von homo- und copolymerisaten von hydroxyalkylvinylethern in wasch- und reinigungsmitteln. |
| US08/185,971 US5576407A (en) | 1991-09-13 | 1992-09-04 | Copolymers of hydroxyalkyl vinyl ethers for use in detergents and cleaning agents |
| EP92918765A EP0603236B1 (de) | 1991-09-13 | 1992-09-04 | Copolymerisate von hydroxyalkylvinylethern und verwendung von homo- und copolymerisaten von hydroxyalkylvinylethern in wasch- und reinigungsmitteln |
| JP5505730A JPH06511021A (ja) | 1991-09-13 | 1992-09-04 | ヒドロキシアルキルビニルエーテルのコポリマー及び洗剤及び清浄剤中でのヒドロキシアルキルビニルエーテルのホモ−及びコポリマーの使用 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE4130428A DE4130428A1 (de) | 1991-09-13 | 1991-09-13 | Copolymerisate von hydroxyalkylvinylethern und verwendung von homo- und copolymerisaten von hydroxyalkylvinylethern in wasch- und reinigungsmitteln |
| DEP4130428.4 | 1991-09-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1993006142A1 true WO1993006142A1 (de) | 1993-04-01 |
Family
ID=6440490
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP1992/002041 Ceased WO1993006142A1 (de) | 1991-09-13 | 1992-09-04 | Copolymerisate von hydroxyalkylvinylethern und verwendung von homo- und copolymerisaten von hydroxyalkylvinylethern in wasch- und reinigungsmitteln |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US5576407A (de) |
| EP (1) | EP0603236B1 (de) |
| JP (1) | JPH06511021A (de) |
| CA (1) | CA2114847A1 (de) |
| DE (2) | DE4130428A1 (de) |
| ES (1) | ES2080514T3 (de) |
| WO (1) | WO1993006142A1 (de) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014079722A1 (en) | 2012-11-20 | 2014-05-30 | Basf Se | Process for the production of a polyvinylether |
| US9447212B2 (en) | 2013-03-08 | 2016-09-20 | The Yokohama Rubber Co., Ltd. | Production method for vinyl ether polymer |
| EP3318583A4 (de) * | 2015-07-03 | 2019-03-27 | Maruzen Petrochemical Co., Ltd. | Verbessertes verfahren zur herstellung eines hydroxygruppenhaltigen vinyletherpolymers |
| US10323107B2 (en) | 2013-10-30 | 2019-06-18 | Basf Se | Alkoxylates of S-vinylthioalkanols |
| WO2020084033A1 (de) | 2018-10-26 | 2020-04-30 | Basf Se | Hydrophob assoziierende copolymere für die tertiäre erdölförderung umfassend monomere mit propylenoxy-einheiten |
| WO2020084046A1 (de) | 2018-10-26 | 2020-04-30 | Basf Se | Verfahren zur herstellung von propylenoxy-haltigen hydrophob assoziierenden monomeren mittels dmc-katalyse |
| WO2024200585A1 (en) | 2023-03-29 | 2024-10-03 | Basf Se | Reduction of acetaldehyde content in vinylether-based copolymers |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE398643T1 (de) * | 2003-04-03 | 2008-07-15 | Basf Se | Gemische von (meth)acrylester von polyalkoxyliertem trimethylolpropan |
| CA2617645C (en) * | 2005-08-03 | 2014-09-09 | Merck Patent Gesellschaft Mit Beschraenkter Haftung | Hydrophilic crosslinked polymer |
| WO2011060293A1 (en) * | 2009-11-12 | 2011-05-19 | Ndsu Research Foundation | Polymers derived from plant oil |
| US9382352B2 (en) | 2009-11-12 | 2016-07-05 | Ndsu Research Foundation | Polymers derived from plant oil |
| JP5936184B2 (ja) * | 2012-02-14 | 2016-06-15 | 国立大学法人福井大学 | ヒドロキシル基含有ビニルエーテルのホモポリマー又はランダム共重合体の製造方法 |
| US8623151B2 (en) | 2012-03-23 | 2014-01-07 | Ecolab Usa Inc. | Terpolymer containing maleic acid, vinyl acetate, and alkyl acrylate monomers for aluminum protection |
| US8740993B2 (en) | 2012-03-23 | 2014-06-03 | Ecolab Usa Inc. | Method for reduced encrustation of textiles using a polymer comprising maleic acid, vinyl acetate, and alkyl acrylate |
| US9834626B2 (en) | 2013-01-15 | 2017-12-05 | Ndsu Research Foundation | Plant oil-based materials |
| EP2840122A1 (de) | 2013-08-21 | 2015-02-25 | Basf Se | Härtbare zusammensetzung auf der basis von hydroxylgruppen-enthaltendem polyvinylether |
| KR20170030538A (ko) * | 2014-07-09 | 2017-03-17 | 바스프 에스이 | 중합체의 제조를 위한 공단량체로서의 반응성 단량체 |
| WO2018153995A1 (en) * | 2017-02-22 | 2018-08-30 | Basf Se | Use of copolymers as binders for pelletizing metal containing ores |
| US10767006B2 (en) * | 2017-06-07 | 2020-09-08 | Medtronic, Inc. | Hydrogenation of tyrosine derived polyarylates |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1025146B (de) * | 1951-03-09 | 1958-02-27 | Rohm & Haas | Verfahren zur Herstellung von Polymerisaten und Mischpolymerisaten |
| US2981692A (en) * | 1957-02-04 | 1961-04-25 | Dow Chemical Co | Detergent compositions |
| US3131162A (en) * | 1960-11-03 | 1964-04-28 | Dow Chemical Co | Copolymer of monovinyl ether of a glycol and vinyl ester of acetic acid and vulcanizate thereof |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US26407A (en) * | 1859-12-13 | Alexander beckers | ||
| US2104004A (en) * | 1936-09-15 | 1938-01-04 | Ackerman William | Combined cultivator and weed cutter |
| US2828220A (en) * | 1955-03-21 | 1958-03-25 | Rohm & Haas | Method of coating leather with polymers containing hydroxyl groups and the resulting article |
| US3328468A (en) * | 1964-02-19 | 1967-06-27 | Dow Chemical Co | Method of preparing the homopolymer of the monovinyl ether of diethylene glycol |
| US4906397A (en) * | 1988-09-15 | 1990-03-06 | National Starch And Chemical Corporation | Detergent compositions utilizing divinyl ether polymers as builders and novel divinyl ether polymers |
| DE3901608A1 (de) * | 1989-01-20 | 1990-07-26 | Byk Chemie Gmbh | Beschichtungs- und formmassen enthaltend polymerisate von alkylvinylethern und deren verwendung als verlauf- oder antischaummittel |
-
1991
- 1991-09-13 DE DE4130428A patent/DE4130428A1/de not_active Withdrawn
-
1992
- 1992-09-04 JP JP5505730A patent/JPH06511021A/ja active Pending
- 1992-09-04 CA CA002114847A patent/CA2114847A1/en not_active Abandoned
- 1992-09-04 WO PCT/EP1992/002041 patent/WO1993006142A1/de not_active Ceased
- 1992-09-04 US US08/185,971 patent/US5576407A/en not_active Expired - Fee Related
- 1992-09-04 DE DE59204523T patent/DE59204523D1/de not_active Expired - Lifetime
- 1992-09-04 EP EP92918765A patent/EP0603236B1/de not_active Expired - Lifetime
- 1992-09-04 ES ES92918765T patent/ES2080514T3/es not_active Expired - Lifetime
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1025146B (de) * | 1951-03-09 | 1958-02-27 | Rohm & Haas | Verfahren zur Herstellung von Polymerisaten und Mischpolymerisaten |
| US2981692A (en) * | 1957-02-04 | 1961-04-25 | Dow Chemical Co | Detergent compositions |
| US3131162A (en) * | 1960-11-03 | 1964-04-28 | Dow Chemical Co | Copolymer of monovinyl ether of a glycol and vinyl ester of acetic acid and vulcanizate thereof |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014079722A1 (en) | 2012-11-20 | 2014-05-30 | Basf Se | Process for the production of a polyvinylether |
| US9447212B2 (en) | 2013-03-08 | 2016-09-20 | The Yokohama Rubber Co., Ltd. | Production method for vinyl ether polymer |
| US10323107B2 (en) | 2013-10-30 | 2019-06-18 | Basf Se | Alkoxylates of S-vinylthioalkanols |
| EP3318583A4 (de) * | 2015-07-03 | 2019-03-27 | Maruzen Petrochemical Co., Ltd. | Verbessertes verfahren zur herstellung eines hydroxygruppenhaltigen vinyletherpolymers |
| US10633469B2 (en) | 2015-07-03 | 2020-04-28 | Maruzen Petrochemical Co., Ltd. | Method for producing hydroxyl group-containing vinyl ether polymer |
| WO2020084033A1 (de) | 2018-10-26 | 2020-04-30 | Basf Se | Hydrophob assoziierende copolymere für die tertiäre erdölförderung umfassend monomere mit propylenoxy-einheiten |
| WO2020084046A1 (de) | 2018-10-26 | 2020-04-30 | Basf Se | Verfahren zur herstellung von propylenoxy-haltigen hydrophob assoziierenden monomeren mittels dmc-katalyse |
| WO2024200585A1 (en) | 2023-03-29 | 2024-10-03 | Basf Se | Reduction of acetaldehyde content in vinylether-based copolymers |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2114847A1 (en) | 1993-04-01 |
| EP0603236A1 (de) | 1994-06-29 |
| DE4130428A1 (de) | 1993-03-18 |
| US5576407A (en) | 1996-11-19 |
| JPH06511021A (ja) | 1994-12-08 |
| DE59204523D1 (de) | 1996-01-11 |
| ES2080514T3 (es) | 1996-02-01 |
| EP0603236B1 (de) | 1995-11-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0603236B1 (de) | Copolymerisate von hydroxyalkylvinylethern und verwendung von homo- und copolymerisaten von hydroxyalkylvinylethern in wasch- und reinigungsmitteln | |
| EP0451508B1 (de) | Verfahren zur Herstellung von Homo- und Copolymerisaten monoethylenisch ungesättigter Dicarbonsäuren und ihre Verwendung | |
| EP0471710B1 (de) | Copolymerisate, die polyalkylenoxid-blöcke enthaltende monomere einpolymerisiert enthalten, ihre herstellung und ihre verwendung | |
| EP0824553B1 (de) | Wasserlösliche copolymere und verfahren zu ihrer herstellung und ihre verwendung | |
| EP0441197B1 (de) | Propfcopolymerisate von Monosacchariden, Oligosacchariden, Polysacchariden und modifizierten Polysacchariden, Verfahren zu ihrer Herstellung und ihre Verwendung | |
| DE3926059C2 (de) | Phosphonomethylierte Polyvinylamine, Verfahren zu ihrer Herstellung und ihre Verwendung | |
| EP0368214B1 (de) | Verwendung von Copolymerisaten als Zusatz zu Flüssigwaschmitteln | |
| EP0548639B1 (de) | Die Verwendung von Polyacetalen auf Basis von Vinylethern und Dihydroxyverbindungen in Wasch- und Reinigungsmitteln | |
| DE3837013A1 (de) | Verwendung von partiell veresterten copolymerisaten in fluessigwaschmitteln | |
| EP0573463A1 (de) | Polyacetale, verfahren zu ihrer herstellung und ihre verwendung in wasch- und reinigungsmitteln. | |
| WO1995030731A1 (de) | Verwendung von pfropfpolymerisaten als zusatz zu wasch- und reinigungsmitteln | |
| EP0165452B1 (de) | Verfahren zur Herstellung von Mischpolymerisaten aus Vinylphosphonsäure und (Meth-)Acrylsäure in wässriger Lösung und ihre Verwendung | |
| DE112014006990T5 (de) | Sulfonatgruppenhaltiges polymer und verfahren zur herstellung hiervon | |
| EP0377448A2 (de) | Verwendung von Copolymerisaten aus 1,2-Di-alkoxyethylenen und monoethylenisch ungesättigten Dicarbonsäureanhydriden in Waschmitteln und Waschmittel, die diese Copolymerisate enthalten | |
| EP0848723B1 (de) | Pulverförmige, poröse, n-vinylimidazol-einheiten enthaltende polymere, verfahren zu ihrer herstellung und ihre verwendung | |
| DE3744592C2 (de) | Wasserlösliche Polymerisate enthaltende Geschirrspülmittel | |
| EP0541588B1 (de) | Verwendung von n-(alkyloxy-polyalkoxymethyl) carbonamid-gruppen aufweisenden polymerisaten als zusatz zu wasch- und reinigungsmitteln | |
| DE4128510A1 (de) | Phosphonomethylierte polyacrylamide, verfahren zu ihrer herstellung und ihre verwendung | |
| EP0469018A1 (de) | Copolymerisate auf basis von monoethylenisch ungesättigten dicarbonsäureanhydriden, diketen und monoethylenisch ungesättigten monocarbonsäuren, ihre herstellung und verwendung | |
| DE2910133A1 (de) | Gerueststoffsubstanzen fuer wasch- und reinigungsmittel, die aus einer mischung aus acrylsaeurecopolymerisaten und polymaleinsaeure bestehen | |
| EP0765352B1 (de) | Vinylformiateinheiten enthaltende copolymerisate, verfahren zu ihrer herstellung und ihre verwendung in wasch- und reinigungsmitteln |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): CA JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1992918765 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2114847 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08185971 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 1992918765 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1992918765 Country of ref document: EP |