WO1994002584A1 - Sensor devices - Google Patents
Sensor devices Download PDFInfo
- Publication number
- WO1994002584A1 WO1994002584A1 PCT/GB1993/001565 GB9301565W WO9402584A1 WO 1994002584 A1 WO1994002584 A1 WO 1994002584A1 GB 9301565 W GB9301565 W GB 9301565W WO 9402584 A1 WO9402584 A1 WO 9402584A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sensor device
- polyvinyl chloride
- membrane
- polyaryl sulphone
- sulphone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/001—Enzyme electrodes
- C12Q1/002—Electrode membranes
Definitions
- This invention relates to sensor devices such as are used in the determination of a component or components which may be present in a fluid sample, such as a physiological fluid (e.g. blood) or other fluids of biological origin (e.g. fruit), process fluids or effluents.
- a fluid sample such as a physiological fluid (e.g. blood) or other fluids of biological origin (e.g. fruit), process fluids or effluents.
- the forms of construction have much in common with each other, and mainly differ in the nature of the membrane or media within it or combined with it in some way.
- Some forms of sensor rely on the components used to make the membrane, while others rely on the mode of fabrication of the membrane, selecting its physical properties (for example its porosity) or treatments given to it, as these factors can control its effectiveness and selectivity in use, or to the conditions under which the sensor is used.
- sensors incorporate an enzyme, which converts one substrate compound or analyte into another which may then be more easily measured.
- an enzyme which converts one substrate compound or analyte into another which may then be more easily measured.
- oxidase enzymes which generate hydrogen peroxide — a substance which can be measured very conveniently and very accurately by electrolytic methods, especially ampere-metrically.
- European Patent No. 216577 (of ICI PLC) which specifies an enzyme electrode sensor with membrane barrier of low (>5%) porosity.
- the known sensors utilising various membrane materials, are very valuable in analytical techniques and the analysis of biological fluids (e.g. blood) for the presence of substances considered critical for medical reasons — e.g. glucose and other materials which may be oxidisable or reducable, or have toxic properties.
- substances considered critical for medical reasons e.g. glucose and other materials which may be oxidisable or reducable, or have toxic properties.
- membranes which are permeable to glucose are usually also permeable to other compounds which interfere with the analytical determination of the glucose.
- Membrane materials vary in the degree of permeability or selectivity towards various species which may be present in samples to be examined, so no membrane material is universally appropriate and the choice depends partly upon the particular application for which it is to be used.
- a sensor it is common to require a sensor to be highly selective or, at least, to be very efficient at excluding compounds which can interfere with the accuracy of the detection and determination of a desired chemical species, for example at an electrode surface. It is also desirable to have a form of sensor which can be used with accuracy at higher concentrations of glucose than are practicable with the conventional sensors and study media.
- an improved sensor device comprising means for detecting components present in fluid samples and providing an output representative of the content of said component, comprising the said detecting means and a membrane barrier between the detecting means and the sample to be analysed, characterised in that the membrane barrier is composed of a combination comprising at least one polyvinyl chloride and at least one polyaryl sulphone.
- a method for determining a component (an analyte) in a fluid sample which comprises contacting the sample with a sensor device as defined above. This is done especially by bringing the sample into contact with the membrane and measuring the response of the measuring means to the desired component (especially glucose) which has diffused through the membrane.
- a sensor device as defined above.
- the desired component especially glucose
- PVC polyvinyl chloride
- PS polyaryl sulphone
- the sensor device comprises a detecting means in contact with an electrolyte medium and both are enclosed by a membrane of mixed PVC and PS polymers which provides an interface for contact with a sample to be analysed.
- the polyvinyl chloride may be any polymer of vinyl chloride, as for example those made and available commercially,
- the molecular weight of the PVC is relatively non-critical to the present invention; most commercial grades can be used satisfactorily, and the grade most appropriate for any particular use can be determined readily by simple trial.
- a typical and convenient molecular weight is in the range 10,000 to 200,000 but products outside this range and mixtures of different molecular weight materials may be used if desired.
- the polyaryl sulphone (PS) may be a sulphonated or an un- sulphonated material , or a mixture of two or more thereof.
- a product comprising mainly one structure fitting the description "polyaryl sulphone" or it may be a mixture of such components having different structure or molecular configuration characteristics. Conveniently, it may be a commercially available product.
- the polyaryl sulphone may be, for example, any of those compounds disclosed and more fully described in European Patent No. 225094, referred to therein as "sulphonated or un-sulphonated polyaryl sulphones" and, in abbreviated form, as "PAS.” These are described as polymers containing repeating units of the general formula -(-Ar - Y-)- wherein Ar represents a divalent aromatic radical and optionally, but preferably, at least some of the Ar groups are sulphonated, and Y represents - S0 2 -.
- the group Ar is preferably a group containing at least two aromatic rings fused together or linked together by a direct bond or linked together by an aliphatic group, an oxygen atom or sulphur atom or a sulphone group.
- the two types of polymer component (PVC and PS) should be, as far as is reasonably practicable, miscible with each other. So, it is preferred that the membranes made from them for the purposes of this invention are made by mixing the components thoroughly and intimately.
- the membrane material i.e. the mixture of two polymers PVC and PS
- the membrane material may be made into membranes by any known or conventional method. Most conveniently, this can be done by solution-casting techniques, using solvents to dissolve the polymers and then spreading the solution on a plate or flat surface and allowing the solvent to evaporate, leaving the polymers deposited as a film on the flat surface — from which it can then be removed for use.
- the solvents which may be used for this vary, and any of those known in the art (individually or as mixtures) may be used.
- the polymer composition of PVC and PS also comprises a plasticiser.
- a plasticiser may be any compound which has the property of plasticising the components polymer or polymers, especially the PVC, and there is a wide variety of commercially available products to choose from.
- An example of a preferred plasticiser is isopropyl myristate (IPM) .
- the proportion of the plasticiser may vary considerably, and the optimum amount for any particular purpose can be determined by simple trial. In general, the proportion may be calculated as that appropriate for plasticising the amount of the PVC component used, and the proportion required in relation to the combined PVC and PS diminishes as the proportion of PVC in the mixture decreases.
- plasticiser When a plasticiser is used, it may be incorporated in any known manner, but most conveniently by inclusion in the solution used for solution-casting — either by adding it as such to the final solution of the mixed polymers or by adding it in solution in one or other of the solutions of the component polymers or dissolved in a separate solvent.
- the detecting means is most conveniently of one of an electrochemical nature, but other types (e.g. spectrophotometric or optical detecting systems) may be used if desired.
- the detecting means will usually comprise an electrode system and a liquid or gel phase electrolyte-containing medium.
- the electrolyte will be aqueous (i.e. aqueous or aqueous-based) but the use of non-aqueous electrolyte media (for example organic-based media) is not excluded.
- the active electrode may be any of those known in the art, for example a metal electrode, but especial ly a platinum anode.
- Thi s i s most conven ient l y made in combi nat ion w i th a si lver /si lver chloride counter-electrode, as for example in the so-cal led Clark electrode , which comprises a platinum electrode surrounded by a silver/silver chloride ring.
- the sensor device of our invention can have a single membrane or, if desired, multiple layers of membrane material. When multiple layers of membrane are used, these may be the same or different, but we prefer that the outermost membrane, i.e. the one which furthest from the active electrode and is contacted with the sample under examination, is made of the membrane composition (mixed PVC and PS polymers) defined according to this invention. This arrangement gives the greatest contribution to determining the access of the components present in the sample to the active electrode surface.
- the inner membrane layer or layers may be made of a wide variety of materials, as is known in the art. We prefer, however, that such an inner membrane is made of a porous film of polycarbonate. Alternatives include membranes (e.g. dialysis membranes) made of cellulose or cellulose derivatives.
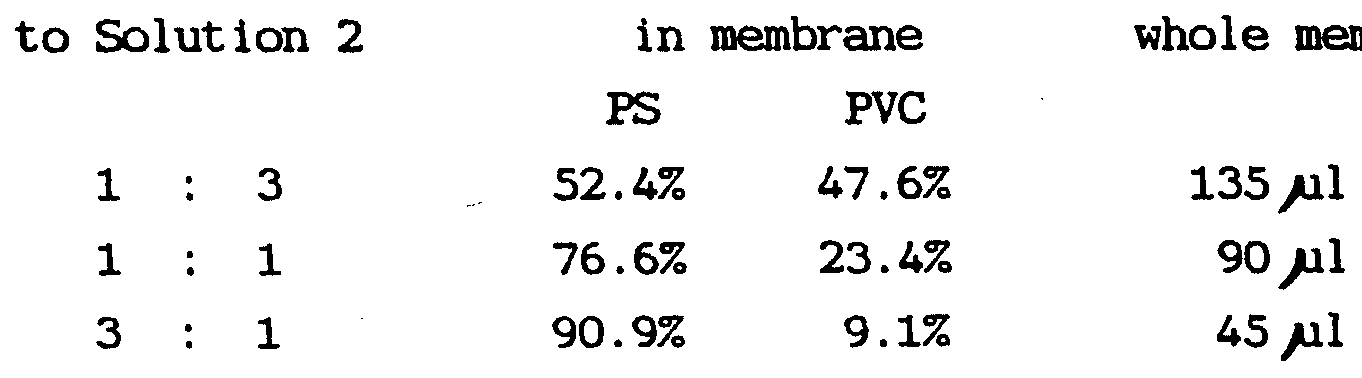
- the proportion of the two polymer components (PVC and PS) used to form the membrane may vary, and the proportion chosen for use in any particular case will depend upon such factors as the degree of selectivity desired and, of course, cost and ease of making it.
- the most advantageous proportion is when the two components are in approximately 3:1 (PS:PVC) or 75/25, as the permeability to glucose at that ratio can be as much as 10 or even 100 times as great as it is for membranes made of the individual components (PVC or PS alone) .
- the exact degree of enhancement of glucose permeability depends, of course, on the precise details and conditions involved, and this figure of up to 100 times will not necessarily be attained in every possible circumstance.
- the thickness of the membranes can be of the order already used conventionally in the art, but may be varied as found most appropriate having regard for the particular mixed polymer composition being used and the conditions under which it is to be used. Thus a convenient thickness is in the range 10 to 40 am, though larger or smaller thicknesses can be used if desired.
- the senor is in the conventional electrode cells. These can be polarised at the conventional potentials appropriate for the determination procedure, and using conventional media. Thus, a polarisation at +650 v in the usual phosphate/chloride buffer is usually most convenient except when the pH is raised from 7.4 to 13 (as discussed below) and the buffer and the polarisation may be altered to suit the high pH used.
- Glucose sensors can be made which directly oxidise glucose at the electrode (anode) without any enzymatic action or the step of generation of hydrogen peroxide. This is very similar to other electro-analytical procedures, in which a component (e.g. paracetamol) is oxidised.
- a component e.g. paracetamol
- the mixed membrane composition PS/PVC has this property, and screens out such compounds as paracetamol, ascorbate and urate while being permeable to glucose.
- an enzyme is present and in the other it is not present.
- the alternative form, without an enzyme, preferably has the construction sequence:-
- a high pH is necessary to render electroactive the glucose diffusing through the membrane.
- high pH we mean a pH of at least 10, and preferably in the range 12 to 14.
- a good and convenient pH is approximately 13.
- the components are mainly the conventional ones, and the many variants known in the art may be used.
- the permeability of the PVC/PS membrane is most marked with glucose, and this is seen as the basis for the most important and surprising effect and its most important utility.
- the effect is not limited solely and absolutely to glucose, and other sugars can diffuse through it and be detected at high pH, like glucose, at an electrode, and so be detected and determined by means of the sensors and. methods of the present invention.
- PVC and PS (and mixtures of PVC and PS) all have the ability to retain a stable high pH environment below the membrane, and so are very suitable for this form.
- glucose concentrations are far beyond clinical glucose levels, but indicates the range of applicability and other uses, for example in the food and brewing industries.
- the usefulness can be seen when one realises that the glucose concentrations to which the invention is applicable are of the order of up to 10% wei ht/volume, or even higher.
- the advantage of the use of a high pH is that it enables glucose to be measured without oxygen dependence — i.e. there is no need to generate hydrogen peroxide in order to get a signal , and as there is no need to produce hydrogen peroxide there is then no need for either an enzyme or oxygen.
- the sensors of the present invention there may be mentioned measurements in oxygen-deprived tissues (e.g. in tumours), in fruit and fruit products, in sugar manufacture, and in industrial fields for study or monitoring of process solutions or other media, or of industrial effluents.
- our sensors can be applied to the study of fruit juices or to fruit as such, for it is not necessary to isolate the fruit juice and contact with a cut surface or by insertion into fruit, to secure a simple juice/sensor contact, can be quite sufficient for use in study and evaluation.
- PVC polyvinyl chloride
- IPM isopropyl myristate
- the films or membranes made as described above were used as the membrane component of a sensor cell comprising a platinum anode and a surrounding silver/silver chloride ring in a Clark electrode assembly, and were found to show high permeability in favour of glucose when contacted with aqueous solutions containing glucose and various contaminants, e.g. paracetamol.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Analytical Chemistry (AREA)
- Biotechnology (AREA)
- Physics & Mathematics (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Immunology (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Investigating Or Analysing Biological Materials (AREA)
- Push-Button Switches (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Measuring Fluid Pressure (AREA)
- Measuring Pulse, Heart Rate, Blood Pressure Or Blood Flow (AREA)
- Saccharide Compounds (AREA)
- Glass Compositions (AREA)
- Measurement Of The Respiration, Hearing Ability, Form, And Blood Characteristics Of Living Organisms (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU47158/93A AU4715893A (en) | 1992-07-28 | 1993-07-23 | Sensor devices |
| JP6504313A JPH08502348A (en) | 1992-07-28 | 1993-07-23 | Sensor device |
| DE69304921T DE69304921T2 (en) | 1992-07-28 | 1993-07-23 | SENSOR DEVICES |
| US08/374,739 US5547561A (en) | 1992-07-28 | 1993-07-23 | Sensor devices and method of using same |
| EP93917907A EP0652941B1 (en) | 1992-07-28 | 1993-07-23 | Sensor devices |
| GR960403482T GR3022050T3 (en) | 1992-07-28 | 1996-12-16 | Sensor devices. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB929215971A GB9215971D0 (en) | 1992-07-28 | 1992-07-28 | Sensor devices |
| GB9215971.4 | 1992-07-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1994002584A1 true WO1994002584A1 (en) | 1994-02-03 |
Family
ID=10719391
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB1993/001565 Ceased WO1994002584A1 (en) | 1992-07-28 | 1993-07-23 | Sensor devices |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US5547561A (en) |
| EP (1) | EP0652941B1 (en) |
| JP (1) | JPH08502348A (en) |
| AT (1) | ATE143050T1 (en) |
| AU (1) | AU4715893A (en) |
| CA (1) | CA2140444A1 (en) |
| DE (1) | DE69304921T2 (en) |
| DK (1) | DK0652941T3 (en) |
| ES (1) | ES2093448T3 (en) |
| GB (1) | GB9215971D0 (en) |
| GR (1) | GR3022050T3 (en) |
| WO (1) | WO1994002584A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997017607A1 (en) * | 1995-11-08 | 1997-05-15 | The Victoria University Of Manchester | Membrane for chemical and biosensors |
| AT404992B (en) * | 1997-04-17 | 1999-04-26 | Avl List Gmbh | SENSOR FOR DETERMINING AN ENZYME SUBSTRATE |
| DE102006020131B4 (en) * | 2006-05-02 | 2012-04-26 | Jinping Liu | Nano- and microstructured biosensor and process for its preparation |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6613379B2 (en) * | 2001-05-08 | 2003-09-02 | Isense Corp. | Implantable analyte sensor |
| US7678567B2 (en) * | 2004-07-26 | 2010-03-16 | Kabushiki Kaisha Toshiba | Optical biosensor |
| US20070021734A1 (en) * | 2005-07-15 | 2007-01-25 | Sai Bhavaraju | Bioelectro-osmotic engine fluid delivery device |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0216577A2 (en) * | 1985-09-16 | 1987-04-01 | Imperial Chemical Industries Plc | Sensor |
| EP0225094A2 (en) * | 1985-11-28 | 1987-06-10 | Imperial Chemical Industries Plc | Membrane |
| EP0503943A1 (en) * | 1991-03-14 | 1992-09-16 | The Victoria University Of Manchester | Sensor devices |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5627643A (en) * | 1979-08-14 | 1981-03-18 | Toshiba Corp | Electrochemical measuring device |
| US4678698A (en) * | 1983-04-12 | 1987-07-07 | Minnesota Mining And Manufacturing Company | Contact lens cleaning article |
-
1992
- 1992-07-28 GB GB929215971A patent/GB9215971D0/en active Pending
-
1993
- 1993-07-23 JP JP6504313A patent/JPH08502348A/en active Pending
- 1993-07-23 ES ES93917907T patent/ES2093448T3/en not_active Expired - Lifetime
- 1993-07-23 DK DK93917907.3T patent/DK0652941T3/da active
- 1993-07-23 AT AT93917907T patent/ATE143050T1/en not_active IP Right Cessation
- 1993-07-23 WO PCT/GB1993/001565 patent/WO1994002584A1/en not_active Ceased
- 1993-07-23 AU AU47158/93A patent/AU4715893A/en not_active Abandoned
- 1993-07-23 CA CA002140444A patent/CA2140444A1/en not_active Abandoned
- 1993-07-23 DE DE69304921T patent/DE69304921T2/en not_active Expired - Fee Related
- 1993-07-23 EP EP93917907A patent/EP0652941B1/en not_active Expired - Lifetime
- 1993-07-23 US US08/374,739 patent/US5547561A/en not_active Expired - Fee Related
-
1996
- 1996-12-16 GR GR960403482T patent/GR3022050T3/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0216577A2 (en) * | 1985-09-16 | 1987-04-01 | Imperial Chemical Industries Plc | Sensor |
| EP0225094A2 (en) * | 1985-11-28 | 1987-06-10 | Imperial Chemical Industries Plc | Membrane |
| EP0503943A1 (en) * | 1991-03-14 | 1992-09-16 | The Victoria University Of Manchester | Sensor devices |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997017607A1 (en) * | 1995-11-08 | 1997-05-15 | The Victoria University Of Manchester | Membrane for chemical and biosensors |
| AU716185B2 (en) * | 1995-11-08 | 2000-02-24 | Victoria University Of Manchester, The | Membrane for chemical and biosensors |
| US6706532B2 (en) | 1995-11-08 | 2004-03-16 | The Victoria University Of Manchester | Membrane for chemical and biosensors |
| AT404992B (en) * | 1997-04-17 | 1999-04-26 | Avl List Gmbh | SENSOR FOR DETERMINING AN ENZYME SUBSTRATE |
| DE102006020131B4 (en) * | 2006-05-02 | 2012-04-26 | Jinping Liu | Nano- and microstructured biosensor and process for its preparation |
Also Published As
| Publication number | Publication date |
|---|---|
| AU4715893A (en) | 1994-02-14 |
| US5547561A (en) | 1996-08-20 |
| EP0652941B1 (en) | 1996-09-18 |
| CA2140444A1 (en) | 1994-02-03 |
| DK0652941T3 (en) | 1997-02-24 |
| DE69304921T2 (en) | 1997-04-10 |
| ES2093448T3 (en) | 1996-12-16 |
| GB9215971D0 (en) | 1992-09-09 |
| EP0652941A1 (en) | 1995-05-17 |
| GR3022050T3 (en) | 1997-03-31 |
| ATE143050T1 (en) | 1996-10-15 |
| DE69304921D1 (en) | 1996-10-24 |
| JPH08502348A (en) | 1996-03-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0204468B1 (en) | Membrane | |
| EP0216577B1 (en) | Sensor | |
| Mizutani et al. | Amperometric l-lactate-sensing electrode based on a polyion complex layer containing lactate oxidase. Application to serum and milk samples | |
| Emr et al. | Use of polymer films in amperometric biosensors | |
| AU595260B2 (en) | Membrane | |
| US6033866A (en) | Highly sensitive amperometric bi-mediator-based glucose biosensor | |
| Palleschi et al. | Ideal hydrogen peroxide-based glucose sensor | |
| EP0859954B1 (en) | Biosensors comprising a membrane | |
| EP0652942A1 (en) | DETECTION DEVICES. | |
| EP0652941B1 (en) | Sensor devices | |
| KR20130012059A (en) | Biosensor desiccant system having enhanced measurement performance | |
| Reddy et al. | Ion exchanger modified PVC membranes—selectivity studies and response amplification of oxalate and lactate enzyme electrodes | |
| Santoni et al. | Enzyme electrode for glucose determination in whole blood | |
| EP0916092A1 (en) | Analytical method and apparatus | |
| WO2001013103A1 (en) | Analytical apparatus and method | |
| WO2001006245A1 (en) | Polymeric compositions and sensor devices | |
| Buch-Rasmussen | Determination of d-glucose in undiluted whole blood using chemically modified electrodes and segmented sample injection in a flow system | |
| JPS59228160A (en) | Amylase analyzing method |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AT AU BB BG BR BY CA CH CZ DE DK ES FI GB HU JP KP KR KZ LK LU MG MN MW NL NO NZ PL PT RO RU SD SE SK UA US VN |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2140444 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1993917907 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08374739 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 1993917907 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1993917907 Country of ref document: EP |