WO1995008542A1 - Farnesyltransferase inhibitor - Google Patents
Farnesyltransferase inhibitor Download PDFInfo
- Publication number
- WO1995008542A1 WO1995008542A1 PCT/JP1994/001543 JP9401543W WO9508542A1 WO 1995008542 A1 WO1995008542 A1 WO 1995008542A1 JP 9401543 W JP9401543 W JP 9401543W WO 9508542 A1 WO9508542 A1 WO 9508542A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- same
- different
- represent
- derivative according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/08—Bridged systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/54—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/02—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings
- C07D241/06—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having one or two double bonds between ring members or between ring members and non-ring members
- C07D241/08—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having one or two double bonds between ring members or between ring members and non-ring members with oxygen atoms directly attached to ring carbon atoms
Definitions
- the present invention relates to a pharmacophorase inhibitor having antitumor activity and useful as an antitumor agent, comprising a piperazinedione derivative as an active ingredient.
- the ras oncogene is point-mutated in many human tumor tissues and is detected in an active form that can transform normal cells.
- Expression of the transformation activity of the ras oncogene product requires binding to the cell membrane by farnesylation of a cysteine residue in the C-terminal region in addition to the amino acid mutation at positions 12, 13, or 61 . This reaction is catalyzed by pharmacophorase. Therefore, the funesyltransferase inhibitor is expected to suppress the function of the ras oncogene product, and is expected to have an antitumor effect.
- Glyotoxin and acetyldariotoxin are known as pharmacophorase inhibitors having a piperazinedione skeleton [J. Antibiotics, 45, 1802 (1992)].
- piperazinedione derivatives of the present invention have, for example, a platelet activating factor antagonistic activity (JP-A-61-233675), an antibacterial activity (Ger. Offen. 2029306) and the like.
- JP-A-61-233675 a platelet activating factor antagonistic activity
- an antibacterial activity Ger. Offen. 2029306
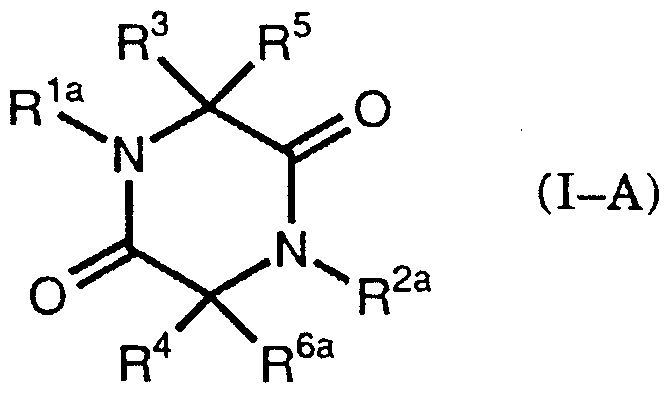
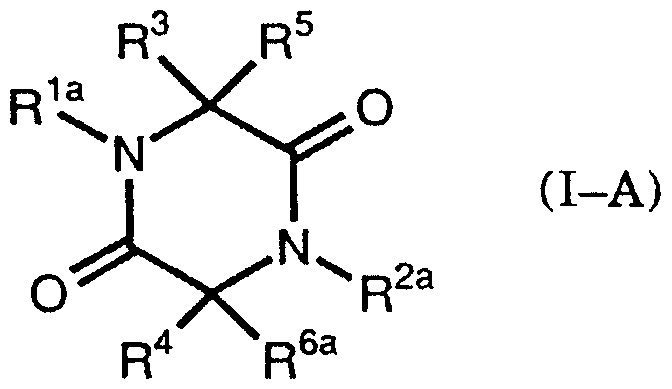
- the present invention provides a compound of the formula (I)
- R ′ and R a are the same or different and represent lower alkyl, lower alkoxyalkyl, substituted or unsubstituted aryl or aralkyl
- R : i and R are the same or different and are mercapto, lower alkanoylthio, aroylthio
- R 3 and R 1 together represent disulfide
- R ′ and R ′ are the same or different and are hydrogen, lower alkyl, lower alkoxy.
- the present invention provides a method for preventing or treating a disease state caused by the action of pharmacinyltransferase comprising administering an effective amount of a piperazinedione derivative represented by the above formula (I), and a method for treating a tumor. It relates to a prevention or treatment method.
- the present invention provides a compound of the above formula (I) for the manufacture of a pharmacological composition useful for the prevention or treatment of a disease state caused by the action of pharmacinyltransferase and the prevention or treatment of a tumor. It relates to the use of the represented perazinedione derivatives.
- a compound represented by the formula (I-A) which has a pharmacokinase inhibitory activity, an antibacterial activity and an antitumor activity.
- R′a represents a substituted or unsubstituted aryl, represents a lower alkyl
- R ′′ represents a lower alkyl, a lower alkoxyalkyl, a hydroxyalkyl, a lower alkanoyloxyalkyl, an aryloxy.
- alkyl represents ⁇ Lal kill O carboxymethyl al kills or Ararukiru, R: l, and piperazine dione derivative represented by R 5 is as defined above
- R ′ b and R 2b are the same or different and each represents an aralkyl, and R : ′ and R ′ have the same meanings as described above, and a compound represented by the formula (I-C)
- R and R are the same or different and each represents an arylothio, a lower alkoxy group, propyloxy or aryloxycarbonylthio, and R 1 and R 2 have the same meanings as described above.
- a perazinedone derivative is provided.
- the lower alkyl may be a straight-chain or branched C1-C4 group.
- methyl, methyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, hexyl and the like can be mentioned.
- aryl include phenyl, naphthyl and the like.
- the aryl portion of alkoxyalkyl and aralkyloxyalkyl has the same meaning as the above.
- substituent of the substituted aryl include 1 to 3 substituents such as halogen, lower alkyl, hydroxy, and lower alkoxy.
- Halogen means each atom of iodine, bromine, chlorine and fluorine, and the lower alkyl and the alkyl moiety of the lower alkyne have the same meaning as the lower alkyl.
- Compound I can be produced by a known method [Tetrahedron, 2045 (1981)] or a method analogous thereto.
- R represents lower alkyl, aryl, lower alkoxy or aryloxy, and R ′ and R 2 have the same meanings as above.
- lower alkyl, aryl, lower alkoxy and aryloxy each have the same meaning as defined above.
- Compound Ia is obtained by reacting compound II with a halogenating agent such as N-bromosuccinic acid imide or bromine in the presence of a catalyst such as perbenzoic acid or the like in an inert solvent such as carbon tetrachloride, chloroform, or dichloromethane. After that, it can be obtained by reacting with a thioacetate such as potassium thioacetate or sodium thioacetate.
- the reaction temperature is preferably from 0 to 150 ° C, and the reaction time is usually from 30 minutes to 5 hours.
- the reaction temperature is preferably from 0 to 100 ° C, and the reaction time is usually from 30 minutes to 10 hours.
- Compound lb can be obtained by treating compound Ia with an acid such as hydrogen chloride gas in a solvent such as methanol or ethanol.
- the reaction temperature is preferably from 0 to 150 ° C, and the reaction time is usually from 10 minutes to 5 hours.
- Compound Ic can be obtained by oxidizing compound Ib with 1 to 10 equivalents of an oxidizing agent such as iodine in an inert solvent such as dichloromethane, chloroform, and tetrahydrofuran.
- an oxidizing agent such as iodine
- an inert solvent such as dichloromethane, chloroform, and tetrahydrofuran.
- the reaction temperature is preferably ⁇ 30 to 100 ° C., and the reaction time is usually 10 minutes to 5 hours.
- Compound Id comprises compound Ib and an acylating agent such as an acid chloride, an acid bromide or an acid anhydride, or a chloroformate such as an aryl chloroformate or an alkyl chloroformate. It can be obtained by reacting in a solvent such as chloroform, dichloromethane, acetonitrile, dimethylformamide or the like in the presence of a base such as pyridine, triethylamine or diisopropylamine.
- the reaction temperature is preferably ⁇ 30 to 100 ° C., and the reaction time is usually 10 minutes to 5 hours.
- compounds Ie, R : i, and IT are both mercapto and R1 ; other than hydrogen, where R : 'and R are combined to form a disulfide and R (; is a group other than hydrogen.
- a compound I f, wherein R : i and IT are both lower alkanolthio, arylothio, lower alkoxycarbonylthio or aryloxycarbonylthio, and a compound I g wherein R ′′ is a group other than hydrogen, It can be produced by the following reaction steps.
- Compound IIIa and compound IIIb are prepared by converting compound Ib with paraanisaldehyde in the presence of a boron trifluoride getyl ether complex, Lewis acid such as titanium tetrachloride, dichloromethane, chloroform, tetrahydrofuran, It can be obtained by reacting in an inert solvent such as ether.
- the reaction temperature is preferably -80 to 50 ° C, and the reaction time is usually 10 minutes to 10 hours.
- Compound IIIa and compound IIb can be separated and purified by methods used in ordinary organic synthesis, for example, recrystallization, various types of chromatography, and the like.
- Compound IV is obtained by combining compound IIIa with a reagent such as chloromethylbenzyl ether, chloromethylmethylether, methyl iodide, or benzyl bromide in the presence of a base such as phenyllithium, butyllithium, or lithium diisopropylamide in tetrahydrofuran.
- a reagent such as chloromethylbenzyl ether, chloromethylmethylether, methyl iodide, or benzyl bromide in the presence of a base such as phenyllithium, butyllithium, or lithium diisopropylamide in tetrahydrofuran.
- a base such as phenyllithium, butyllithium, or lithium diisopropylamide in tetrahydrofuran.
- the reaction temperature is preferably from -100 to 50 ° C
- Compound IV b of one or both of R 5 and R “are human Dorokishimechiru Among the compounds IV, the compounds IV a one or both of R 5 and R (i is Benjiruoki Shimechiru Among the compounds IV, dichloromethane Can be obtained by treating with a Lewis acid such as boron trichloride or boron tribromide in an inert solvent such as chloroform, tetrahydrofuran, ether, etc.
- the reaction temperature is -100 to 50 ° C.
- the reaction time is usually from 10 minutes to 10 hours.
- Compound Ie is obtained by oxidizing compound IV to sulfoxide in a solvent such as chloroform, dichloromethane, tetrahydrofuran, or ether with 1 to 2 equivalents of an oxidizing agent such as perbenzoic acid, peracetic acid, or hydrogen peroxide. Then, it can be obtained by treating with an acid such as perchloric acid, hydrochloric acid, or hydrobromic acid.
- a solvent such as chloroform, dichloromethane, tetrahydrofuran, or ether
- an oxidizing agent such as perbenzoic acid, peracetic acid, or hydrogen peroxide.
- an acid such as perchloric acid, hydrochloric acid, or hydrobromic acid.
- the reaction The temperature is preferably ⁇ 100 to 50 ° C., and the reaction time is usually 10 minutes to 5 hours.
- the reaction temperature is preferably 0 to 50 ° C, and the reaction time is usually 1 to 5 hours.
- Compound Ig is obtained by reducing compound Ie to dithiol with a reducing agent such as sodium borohydride in a solvent such as methanol or ethanol to obtain compound If.
- a reducing agent such as sodium borohydride
- a solvent such as methanol or ethanol
- the reaction temperature is preferably ⁇ 100 to 50 ° C., and the reaction time is usually 10 minutes to 2 hours.
- the hydroxyl group may be simultaneously acylated.
- the isolation and purification of the product in the above production method can be carried out by appropriately combining methods used in ordinary organic synthesis, for example, filtration, extraction, washing, drying, concentration, crystallization, various types of chromatography, and the like.
- Compound I and its pharmaceutically acceptable salts may exist in the form of adducts with water or various solvents, and these adducts are also used as the therapeutic agent of the present invention.
- Table 1 shows the structures of typical examples of Compound I.
- Compound 1 The physical properties of Compound I are shown below.
- Compound 1-6 and Compound I-9 Compound 1-21 are novel compounds and are shown in Examples.
- JP-A-61-233675 describes production methods and further detailed physical properties.
- German Patent Publication (Ger. Offen. 2029306) describes production methods and further detailed physical properties.
- the production method of compound 1-5 is described in Journal of Chemical, Society, Chemical, Communication (J. Chem. Soc., Chem. Commun.), 810 (1983).
- the production methods and more detailed physical properties of compound 1-3 are shown in Reference Examples.
- Table 2 shows the structures and compound numbers of the compounds in Reference Examples.
- Test Example 1 Inhibitory activity of pharmacosyltransferase
- FTase For pharmacophorase transferase (FTase), the extract obtained by crushing bovine brain is subjected to DEAE-Sephacel (Pharmacia) column chromatography, and the active fraction is concentrated by ultrafiltration and dialyzed. 20mM Tr i s-HCl P H8 . 0, 50mM NaCl, 20mM ZnC, using ImM Jichiosurei Torr (DTT), a material obtained by 0. 2 mM off We methylpropenylmethyl sulfonyl Rufuruorai de (PMSF)] as a crude enzyme solution. The activity was measured using the enzyme obtained by the above-mentioned method and FTase [; !
- the anticellular activity was measured using BALB 3T3 cells transformed with the oncogene H-ras (hereinafter referred to as BALB 3T3 / H-ras cells).
- BALB 3T3 was adjusted to 3.0 x 10 1 / ml with DME medium (manufactured by Nissi) and medium containing 10% fetal bovine serum (hereinafter referred to as medium A) in each well of a 96-well microtiter plate. / H-ras cells were dispensed in 0.1 ml portions. After culturing the plate in a carbon dioxide gas incubator at 37 ° C for 20 hours, 0.1 ml of a sample (test compound) appropriately diluted with medium A was added thereto.
- DME medium manufactured by Nissi
- medium A medium fetal bovine serum
- the antibacterial activity was measured using a medium (pH 7) prepared by dissolving 3 g of pact-tripton (manufactured by Difco), 3 g of meat extract, 1 g of yeast extract, 1 g of glucose, and 16 g of agar in 1 L of water. It was measured by the method.
- the antimicrobial activity of Compound I is expressed as the minimum inhibitory concentration (MIC). The results are shown in Table 4. Table 4
- Test Example 4 Antitumor effect in K-BALB mouse syngeneic tumor line
- the ratio was calculated by calculating the ratio (T / C) of the tumor volume (T) of the test compound administration group to the tumor volume (C) of the non-administration group.
- Table 5 shows the results.
- BALB / c mice also survived when 1,15 mg / kg of compound 1 was intraperitoneally administered once daily for 5 days to BALB / c mice.
- Compound I or a pharmacologically acceptable salt thereof is administered orally or almost parenterally as it is or as various pharmaceutical compositions.
- dosage form of such a pharmaceutical composition include tablets, pills, powders, granules, capsules, suppositories, injections, and drops.
- Carriers used in pharmaceutical compositions include, for example, water, distilled water for injection, physiological saline, glucose, fructose, sucrose, mannite, lactose, starch, corn starch, cellulose, methylcellulose, carboxy Examples include methylcellulose, hydroxypropylcellulose, alginic acid, talc, sodium citrate, calcium carbonate, calcium hydrogen phosphate, magnesium stearate, urea, silicone resin, sorbitan fatty acid esters, glycerin fatty acid esters, and the like. It is appropriately selected according to the type of the preparation.
- the dose and frequency of administration depend on the desired therapeutic effect, administration method, duration of treatment, age, Depending on body weight, etc., oral or parenteral (eg, injection, infusion, rectal administration by suppository, skin patch, etc.), usually 0.01 to 2 mg / kg per adult per day, depending on administration method.
- oral or parenteral eg, injection, infusion, rectal administration by suppository, skin patch, etc.
- ordinary post-treatment means the following post-reaction treatment.
- a 10% hydroxypropylcellulose solution was added to the above mixture and kneaded.
- the kneaded mixture was granulated with an extruder and granulator equipped with a 1.0 basket and then magnesium stearate was added to form granules for tableting.
- the compound 1-in one formulation (nOmg) was prepared in a conventional manner. Tablets of 8 ⁇ diameter containing 1 mg of 1 were prepared.
- a 10% hydroxypropylcellulose solution was added to the above mixture and kneaded. After granulating in the same manner as in Example 15, magnesium stearate was added to obtain a capsule containing 50 mg of compound 1-1 in one capsule (170 mg) by a conventional method.
- Compound 1-1 (10 g) was dissolved in soybean oil (100 g), and the resulting solution was injected into a capsule in a conventional manner to prepare a soft carbose containing 10 mg of Compound 1 per capsule. .
- Example 15 In the same manner as in Example 15, a tablet having a diameter of 8 mm containing 100 mg of Compound 12 in one formulation (nOmg) was prepared.
- Example 15 In the same manner as in Example 15, a tablet having an 8-diameter diameter containing 100 mg of Compound 13 in one preparation (170 mg) was prepared.
- Example 15 In the same manner as in Example 15, a tablet having a diameter of 8 mm containing 100 mg of compound 1-15 in one preparation (170 mg) was prepared.
- Example 16 In the same manner as in Example 16, a capsule preparation containing 50 mg of compound I-15 in one capsule (170 mg) was prepared.
- Example 17 In the same manner as in Example 17, a soft capsule preparation containing 10 mg of compound 1-15 per capsule was prepared.
- Example 15 In the same manner as in Example 15, a tablet of 8 mm in diameter containing 100 mg of compound 1-17 in one preparation (170 mg) was prepared.
- ADVANTAGE OF THE INVENTION according to this invention, it has a piperazine dione skeleton, and can provide a pharmaco-transferase inhibitor useful as a pharmaceutical.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/436,458 US5728830A (en) | 1993-09-22 | 1994-09-20 | Farnesyltransferase inhibitor |
| EP94927069A EP0670314A4 (en) | 1993-09-22 | 1994-09-20 | FARNESYL TRANSFERASE INHIBITORS. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP5/236179 | 1993-09-22 | ||
| JP23617993 | 1993-09-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1995008542A1 true WO1995008542A1 (en) | 1995-03-30 |
Family
ID=16996945
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP1994/001543 Ceased WO1995008542A1 (en) | 1993-09-22 | 1994-09-20 | Farnesyltransferase inhibitor |
Country Status (3)
| Country | Link |
|---|---|
| US (2) | US5728830A (ja) |
| EP (1) | EP0670314A4 (ja) |
| WO (1) | WO1995008542A1 (ja) |
Cited By (63)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6387903B1 (en) | 1997-08-27 | 2002-05-14 | Merck & Co., Inc. | Inhibitors of prenyl-protein transferase |
| WO2006123182A2 (en) | 2005-05-17 | 2006-11-23 | Merck Sharp & Dohme Limited | Cyclohexyl sulphones for treatment of cancer |
| WO2007093827A1 (en) | 2006-02-15 | 2007-08-23 | Istituto Di Ricerche Di Biologia Molecolare P. Angeletti Spa | Thiophene and thiazole substituted trifluoroethanone derivatives as histone deacetylase (hdac) inhibitors |
| WO2008106692A1 (en) | 2007-03-01 | 2008-09-04 | Novartis Vaccines And Diagnostics, Inc. | Pim kinase inhibitors and methods of their use |
| WO2008144062A1 (en) | 2007-05-21 | 2008-11-27 | Novartis Ag | Csf-1r inhibitors, compositions, and methods of use |
| WO2009002495A1 (en) | 2007-06-27 | 2008-12-31 | Merck & Co., Inc. | 4-carboxybenzylamino derivatives as histone deacetylase inhibitors |
| US7482367B2 (en) | 2005-08-30 | 2009-01-27 | Novartis Vaccines And Diagnostics, Inc. | Substituted benzimidazoles and methods of their use |
| US7553854B2 (en) | 2006-04-19 | 2009-06-30 | Novartis Vaccines And Diagnostics, Inc. | 6-O-substituted benzoxazole and benzothiazole compounds and methods of inhibiting CSF-1R signaling |
| WO2010114780A1 (en) | 2009-04-01 | 2010-10-07 | Merck Sharp & Dohme Corp. | Inhibitors of akt activity |
| WO2011046771A1 (en) | 2009-10-14 | 2011-04-21 | Schering Corporation | SUBSTITUTED PIPERIDINES THAT INCREASE p53 ACTIVITY AND THE USES THEREOF |
| EP2336120A1 (en) | 2007-01-10 | 2011-06-22 | Istituto di ricerche di Biologia Molecolare P. Angeletti S.R.L. | Combinations containing amide substituted indazoles as poly(ADP-ribose)polymerase (PARP) inhibitors |
| WO2011115725A2 (en) | 2010-03-16 | 2011-09-22 | Dana-Farber Cancer Institute, Inc. | Indazole compounds and their uses |
| WO2011163330A1 (en) | 2010-06-24 | 2011-12-29 | Merck Sharp & Dohme Corp. | Novel heterocyclic compounds as erk inhibitors |
| WO2012018754A2 (en) | 2010-08-02 | 2012-02-09 | Merck Sharp & Dohme Corp. | RNA INTERFERENCE MEDIATED INHIBITION OF CATENIN (CADHERIN-ASSOCIATED PROTEIN), BETA 1 (CTNNB1) GENE EXPRESSION USING SHORT INTERFERING NUCLEIC ACID (siNA) |
| WO2012024170A2 (en) | 2010-08-17 | 2012-02-23 | Merck Sharp & Dohme Corp. | RNA INTERFERENCE MEDIATED INHIBITION OF HEPATITIS B VIRUS (HBV) GENE EXPRESSION USING SHORT INTERFERING NUCLEIC ACID (siNA) |
| WO2012027236A1 (en) | 2010-08-23 | 2012-03-01 | Schering Corporation | NOVEL PYRAZOLO[1,5-a]PYRIMIDINE DERIVATIVES AS mTOR INHIBITORS |
| WO2012030685A2 (en) | 2010-09-01 | 2012-03-08 | Schering Corporation | Indazole derivatives useful as erk inhibitors |
| WO2012036997A1 (en) | 2010-09-16 | 2012-03-22 | Schering Corporation | Fused pyrazole derivatives as novel erk inhibitors |
| WO2012058210A1 (en) | 2010-10-29 | 2012-05-03 | Merck Sharp & Dohme Corp. | RNA INTERFERENCE MEDIATED INHIBITION OF GENE EXPRESSION USING SHORT INTERFERING NUCLEIC ACIDS (siNA) |
| WO2012087772A1 (en) | 2010-12-21 | 2012-06-28 | Schering Corporation | Indazole derivatives useful as erk inhibitors |
| WO2012145471A1 (en) | 2011-04-21 | 2012-10-26 | Merck Sharp & Dohme Corp. | Insulin-like growth factor-1 receptor inhibitors |
| WO2013063214A1 (en) | 2011-10-27 | 2013-05-02 | Merck Sharp & Dohme Corp. | Novel compounds that are erk inhibitors |
| WO2013165816A2 (en) | 2012-05-02 | 2013-11-07 | Merck Sharp & Dohme Corp. | SHORT INTERFERING NUCLEIC ACID (siNA) COMPOSITIONS |
| EP2698157A1 (en) | 2006-09-22 | 2014-02-19 | Merck Sharp & Dohme Corp. | Method of treatment using fatty acid synthesis inhibitors |
| WO2014052563A2 (en) | 2012-09-28 | 2014-04-03 | Merck Sharp & Dohme Corp. | Novel compounds that are erk inhibitors |
| WO2014085216A1 (en) | 2012-11-28 | 2014-06-05 | Merck Sharp & Dohme Corp. | Compositions and methods for treating cancer |
| WO2014100065A1 (en) | 2012-12-20 | 2014-06-26 | Merck Sharp & Dohme Corp. | Substituted imidazopyridines as hdm2 inhibitors |
| US8765747B2 (en) | 2009-06-12 | 2014-07-01 | Dana-Farber Cancer Institute, Inc. | Fused 2-aminothiazole compounds |
| WO2014120748A1 (en) | 2013-01-30 | 2014-08-07 | Merck Sharp & Dohme Corp. | 2,6,7,8 substituted purines as hdm2 inhibitors |
| WO2015034925A1 (en) | 2013-09-03 | 2015-03-12 | Moderna Therapeutics, Inc. | Circular polynucleotides |
| JP2015526516A (ja) * | 2012-08-29 | 2015-09-10 | ユニヴァーシティー オブ サザン カリフォルニア | 低酸素誘導性転写因子複合体の活性を阻害するための組成物および方法、ならびに腫瘍の治療のためのその使用 |
| WO2016020864A1 (en) | 2014-08-06 | 2016-02-11 | Novartis Ag | Protein kinase c inhibitors and methods of their use |
| US9758522B2 (en) | 2012-10-19 | 2017-09-12 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged small molecules as inducers of protein degradation |
| US9862688B2 (en) | 2014-04-23 | 2018-01-09 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged janus kinase inhibitors and uses thereof |
| WO2018058022A1 (en) | 2016-09-26 | 2018-03-29 | Merck Sharp & Dohme Corp. | Anti-cd27 antibodies |
| WO2018071283A1 (en) | 2016-10-12 | 2018-04-19 | Merck Sharp & Dohme Corp. | Kdm5 inhibitors |
| US10000483B2 (en) | 2012-10-19 | 2018-06-19 | Dana-Farber Cancer Institute, Inc. | Bone marrow on X chromosome kinase (BMX) inhibitors and uses thereof |
| US10017477B2 (en) | 2014-04-23 | 2018-07-10 | Dana-Farber Cancer Institute, Inc. | Janus kinase inhibitors and uses thereof |
| WO2018190719A2 (en) | 2017-04-13 | 2018-10-18 | Aduro Biotech Holdings, Europe B.V. | Anti-sirp alpha antibodies |
| US10112927B2 (en) | 2012-10-18 | 2018-10-30 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 (CDK7) |
| US10144730B2 (en) | 2011-11-17 | 2018-12-04 | Dana-Farber Cancer Institute, Inc. | Inhibitors of c-Jun-N-terminal kinase (JNK) |
| WO2019094312A1 (en) | 2017-11-08 | 2019-05-16 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| WO2019094311A1 (en) | 2017-11-08 | 2019-05-16 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| WO2019152642A1 (en) | 2018-02-01 | 2019-08-08 | Merck Sharp & Dohme Corp. | Anti-pd-1/lag3 bispecific antibodies |
| JP2019529409A (ja) * | 2016-09-15 | 2019-10-17 | シティ・オブ・ホープCity of Hope | ジチオetp誘導体 |
| US10550121B2 (en) | 2015-03-27 | 2020-02-04 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| WO2020033282A1 (en) | 2018-08-07 | 2020-02-13 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| WO2020033284A1 (en) | 2018-08-07 | 2020-02-13 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| WO2020033288A1 (en) | 2018-08-07 | 2020-02-13 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| US10702527B2 (en) | 2015-06-12 | 2020-07-07 | Dana-Farber Cancer Institute, Inc. | Combination therapy of transcription inhibitors and kinase inhibitors |
| US10870651B2 (en) | 2014-12-23 | 2020-12-22 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 (CDK7) |
| US10906889B2 (en) | 2013-10-18 | 2021-02-02 | Dana-Farber Cancer Institute, Inc. | Polycyclic inhibitors of cyclin-dependent kinase 7 (CDK7) |
| US11040957B2 (en) | 2013-10-18 | 2021-06-22 | Dana-Farber Cancer Institute, Inc. | Heteroaromatic compounds useful for the treatment of proliferative diseases |
| US11096950B2 (en) | 2006-11-01 | 2021-08-24 | Barbara Brooke Jennings | Compounds, methods, and treatments for abnormal signaling pathways for prenatal and postnatal development |
| US11142507B2 (en) | 2015-09-09 | 2021-10-12 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| US11826365B2 (en) | 2009-12-29 | 2023-11-28 | Dana-Farber Cancer Institute, Inc. | Type II raf kinase inhibitors |
| WO2024180169A1 (en) | 2023-03-02 | 2024-09-06 | Carcimun Biotech Gmbh | Means and methods for diagnosing cancer and/or an acute inflammatory disease |
| US12187701B2 (en) | 2018-06-25 | 2025-01-07 | Dana-Farber Cancer Institute, Inc. | Taire family kinase inhibitors and uses thereof |
| US12281126B2 (en) | 2018-12-28 | 2025-04-22 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 and uses thereof |
| US12441730B2 (en) | 2019-12-17 | 2025-10-14 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
| WO2026027944A1 (en) | 2024-07-30 | 2026-02-05 | Sairopa B.V. | Anti-sirp alpha antibody formulations and uses thereof |
| US12552826B2 (en) | 2018-08-07 | 2026-02-17 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
| US12583871B2 (en) | 2019-12-17 | 2026-03-24 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995000497A1 (en) * | 1993-06-18 | 1995-01-05 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| US5990112A (en) * | 1996-06-18 | 1999-11-23 | Affymax Technologies N.V. | Inhibitors of metalloproteases pharmaceutical compositions comprising same and methods of their use |
| CA2249604A1 (en) * | 1996-04-03 | 1997-10-09 | Theresa M. Williams | Inhibitors of farnesyl-protein transferase |
| CA2250587A1 (en) * | 1996-04-03 | 1997-10-09 | Christopher J. Dinsmore | Inhibitors of farnesyl-protein transferase |
| US5919785A (en) * | 1996-04-03 | 1999-07-06 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| US5859012A (en) * | 1996-04-03 | 1999-01-12 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| US5972942A (en) * | 1997-03-27 | 1999-10-26 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| GB9801231D0 (en) | 1997-06-05 | 1998-03-18 | Merck & Co Inc | A method of treating cancer |
| JP2002519428A (ja) | 1998-07-02 | 2002-07-02 | メルク エンド カムパニー インコーポレーテッド | プレニル−タンパク質トランスフェラーゼの阻害剤 |
| AU2001227755A1 (en) | 2000-01-12 | 2001-07-24 | Merck And Co., Inc. | Inhibitors of prenyl-protein transferase |
| WO2001077116A1 (en) | 2000-04-10 | 2001-10-18 | Merck & Co., Inc. | Inhibitors of prenyl-protein transferase |
| WO2018132791A1 (en) | 2017-01-16 | 2018-07-19 | The Regents Of The University Of California | Treatment of neurodegenerative conditions by disruption of rhes |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS4862773A (ja) * | 1971-11-24 | 1973-09-01 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3562253A (en) * | 1968-07-15 | 1971-02-09 | American Cyanamid Co | 1,4 - dialkyl - 2,5 - piperazinedione - 3,6 - dithioacetates,-dithiols,-disulfides and -tetrasulfides |
| FR2086041A1 (en) * | 1969-06-13 | 1971-12-31 | American Cyanamid Co | Piperazinedione derivs prepn |
| US4007190A (en) * | 1971-11-24 | 1977-02-08 | Merck & Co., Inc. | 3,6-Bis(2-piperidinyl)-2,5-piperazinedione compounds |
| US3883561A (en) * | 1972-10-16 | 1975-05-13 | Lilly Co Eli | Antibiotic lactones |
| JPS50116486A (ja) * | 1974-03-02 | 1975-09-11 | ||
| GB8507778D0 (en) * | 1985-03-26 | 1985-05-01 | Fujisawa Pharmaceutical Co | Piperazine compound |
| JPS63290868A (ja) * | 1987-05-22 | 1988-11-28 | Fujisawa Pharmaceut Co Ltd | ジケトピペラジン誘導体およびその塩類 |
-
1994
- 1994-09-20 EP EP94927069A patent/EP0670314A4/en not_active Ceased
- 1994-09-20 US US08/436,458 patent/US5728830A/en not_active Expired - Fee Related
- 1994-09-20 WO PCT/JP1994/001543 patent/WO1995008542A1/ja not_active Ceased
-
1997
- 1997-08-19 US US08/912,870 patent/US5925641A/en not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS4862773A (ja) * | 1971-11-24 | 1973-09-01 |
Cited By (88)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6387903B1 (en) | 1997-08-27 | 2002-05-14 | Merck & Co., Inc. | Inhibitors of prenyl-protein transferase |
| WO2006123182A2 (en) | 2005-05-17 | 2006-11-23 | Merck Sharp & Dohme Limited | Cyclohexyl sulphones for treatment of cancer |
| US7482367B2 (en) | 2005-08-30 | 2009-01-27 | Novartis Vaccines And Diagnostics, Inc. | Substituted benzimidazoles and methods of their use |
| WO2007093827A1 (en) | 2006-02-15 | 2007-08-23 | Istituto Di Ricerche Di Biologia Molecolare P. Angeletti Spa | Thiophene and thiazole substituted trifluoroethanone derivatives as histone deacetylase (hdac) inhibitors |
| US8710048B2 (en) | 2006-04-19 | 2014-04-29 | Novartis Ag | 6-O-substituted benzoxazole and benzothiazole compounds and methods of inhibiting CSF-1R signaling |
| US7553854B2 (en) | 2006-04-19 | 2009-06-30 | Novartis Vaccines And Diagnostics, Inc. | 6-O-substituted benzoxazole and benzothiazole compounds and methods of inhibiting CSF-1R signaling |
| US8173689B2 (en) | 2006-04-19 | 2012-05-08 | Novartis Ag | 6-O-substituted benzoxazole and benzothiazole compounds and methods of inhibiting CSF-1R signaling |
| EP2698157A1 (en) | 2006-09-22 | 2014-02-19 | Merck Sharp & Dohme Corp. | Method of treatment using fatty acid synthesis inhibitors |
| EP2946778A1 (en) | 2006-09-22 | 2015-11-25 | Merck Sharp & Dohme Corp. | Method of treatment using fatty acid synthesis inhibitors |
| US11096950B2 (en) | 2006-11-01 | 2021-08-24 | Barbara Brooke Jennings | Compounds, methods, and treatments for abnormal signaling pathways for prenatal and postnatal development |
| EP2336120A1 (en) | 2007-01-10 | 2011-06-22 | Istituto di ricerche di Biologia Molecolare P. Angeletti S.R.L. | Combinations containing amide substituted indazoles as poly(ADP-ribose)polymerase (PARP) inhibitors |
| EP2805945A1 (en) | 2007-01-10 | 2014-11-26 | MSD Italia S.r.l. | Amide substituted indazoles as poly(ADP-ribose)polymerase (PARP) inhibitors |
| WO2008106692A1 (en) | 2007-03-01 | 2008-09-04 | Novartis Vaccines And Diagnostics, Inc. | Pim kinase inhibitors and methods of their use |
| WO2008144062A1 (en) | 2007-05-21 | 2008-11-27 | Novartis Ag | Csf-1r inhibitors, compositions, and methods of use |
| EP3103791A1 (en) | 2007-06-27 | 2016-12-14 | Merck Sharp & Dohme Corp. | 4-carboxybenzylamino derivatives as histone deacetylase inhibitors |
| WO2009002495A1 (en) | 2007-06-27 | 2008-12-31 | Merck & Co., Inc. | 4-carboxybenzylamino derivatives as histone deacetylase inhibitors |
| WO2010114780A1 (en) | 2009-04-01 | 2010-10-07 | Merck Sharp & Dohme Corp. | Inhibitors of akt activity |
| US8765747B2 (en) | 2009-06-12 | 2014-07-01 | Dana-Farber Cancer Institute, Inc. | Fused 2-aminothiazole compounds |
| US9505784B2 (en) | 2009-06-12 | 2016-11-29 | Dana-Farber Cancer Institute, Inc. | Fused 2-aminothiazole compounds |
| WO2011046771A1 (en) | 2009-10-14 | 2011-04-21 | Schering Corporation | SUBSTITUTED PIPERIDINES THAT INCREASE p53 ACTIVITY AND THE USES THEREOF |
| US11826365B2 (en) | 2009-12-29 | 2023-11-28 | Dana-Farber Cancer Institute, Inc. | Type II raf kinase inhibitors |
| WO2011115725A2 (en) | 2010-03-16 | 2011-09-22 | Dana-Farber Cancer Institute, Inc. | Indazole compounds and their uses |
| US8987275B2 (en) | 2010-03-16 | 2015-03-24 | Dana-Farber Cancer Institute, Inc. | Indazole compounds and their uses |
| WO2011163330A1 (en) | 2010-06-24 | 2011-12-29 | Merck Sharp & Dohme Corp. | Novel heterocyclic compounds as erk inhibitors |
| WO2012018754A2 (en) | 2010-08-02 | 2012-02-09 | Merck Sharp & Dohme Corp. | RNA INTERFERENCE MEDIATED INHIBITION OF CATENIN (CADHERIN-ASSOCIATED PROTEIN), BETA 1 (CTNNB1) GENE EXPRESSION USING SHORT INTERFERING NUCLEIC ACID (siNA) |
| EP3330377A1 (en) | 2010-08-02 | 2018-06-06 | Sirna Therapeutics, Inc. | Rna interference mediated inhibition of catenin (cadherin-associated protein), beta 1 (ctnnb1) gene expression using short interfering nucleic acid (sina) |
| EP4079856A1 (en) | 2010-08-17 | 2022-10-26 | Sirna Therapeutics, Inc. | Rna interference mediated inhibition of hepatitis b virus (hbv) gene expression using short interfering nucleic acid (sina) |
| WO2012024170A2 (en) | 2010-08-17 | 2012-02-23 | Merck Sharp & Dohme Corp. | RNA INTERFERENCE MEDIATED INHIBITION OF HEPATITIS B VIRUS (HBV) GENE EXPRESSION USING SHORT INTERFERING NUCLEIC ACID (siNA) |
| WO2012027236A1 (en) | 2010-08-23 | 2012-03-01 | Schering Corporation | NOVEL PYRAZOLO[1,5-a]PYRIMIDINE DERIVATIVES AS mTOR INHIBITORS |
| WO2012030685A2 (en) | 2010-09-01 | 2012-03-08 | Schering Corporation | Indazole derivatives useful as erk inhibitors |
| WO2012036997A1 (en) | 2010-09-16 | 2012-03-22 | Schering Corporation | Fused pyrazole derivatives as novel erk inhibitors |
| EP3766975A1 (en) | 2010-10-29 | 2021-01-20 | Sirna Therapeutics, Inc. | Rna interference mediated inhibition of gene expression using short interfering nucleic acid (sina) |
| EP3327125A1 (en) | 2010-10-29 | 2018-05-30 | Sirna Therapeutics, Inc. | Rna interference mediated inhibition of gene expression using short interfering nucleic acids (sina) |
| WO2012058210A1 (en) | 2010-10-29 | 2012-05-03 | Merck Sharp & Dohme Corp. | RNA INTERFERENCE MEDIATED INHIBITION OF GENE EXPRESSION USING SHORT INTERFERING NUCLEIC ACIDS (siNA) |
| WO2012087772A1 (en) | 2010-12-21 | 2012-06-28 | Schering Corporation | Indazole derivatives useful as erk inhibitors |
| WO2012145471A1 (en) | 2011-04-21 | 2012-10-26 | Merck Sharp & Dohme Corp. | Insulin-like growth factor-1 receptor inhibitors |
| WO2013063214A1 (en) | 2011-10-27 | 2013-05-02 | Merck Sharp & Dohme Corp. | Novel compounds that are erk inhibitors |
| US10981903B2 (en) | 2011-11-17 | 2021-04-20 | Dana-Farber Cancer Institute, Inc. | Inhibitors of c-Jun-N-terminal kinase (JNK) |
| US10144730B2 (en) | 2011-11-17 | 2018-12-04 | Dana-Farber Cancer Institute, Inc. | Inhibitors of c-Jun-N-terminal kinase (JNK) |
| WO2013165816A2 (en) | 2012-05-02 | 2013-11-07 | Merck Sharp & Dohme Corp. | SHORT INTERFERING NUCLEIC ACID (siNA) COMPOSITIONS |
| EP3919620A1 (en) | 2012-05-02 | 2021-12-08 | Sirna Therapeutics, Inc. | Short interfering nucleic acid (sina) compositions |
| US9416145B2 (en) | 2012-08-29 | 2016-08-16 | University Of Southern California | Compositions and methods for inhibiting activity of hypoxia-inducible transcription factor complex and its use for treatment of tumors |
| JP2015526516A (ja) * | 2012-08-29 | 2015-09-10 | ユニヴァーシティー オブ サザン カリフォルニア | 低酸素誘導性転写因子複合体の活性を阻害するための組成物および方法、ならびに腫瘍の治療のためのその使用 |
| WO2014052563A2 (en) | 2012-09-28 | 2014-04-03 | Merck Sharp & Dohme Corp. | Novel compounds that are erk inhibitors |
| US10112927B2 (en) | 2012-10-18 | 2018-10-30 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 (CDK7) |
| US10787436B2 (en) | 2012-10-18 | 2020-09-29 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 (CDK7) |
| US9758522B2 (en) | 2012-10-19 | 2017-09-12 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged small molecules as inducers of protein degradation |
| US10000483B2 (en) | 2012-10-19 | 2018-06-19 | Dana-Farber Cancer Institute, Inc. | Bone marrow on X chromosome kinase (BMX) inhibitors and uses thereof |
| USRE48175E1 (en) | 2012-10-19 | 2020-08-25 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged small molecules as inducers of protein degradation |
| WO2014085216A1 (en) | 2012-11-28 | 2014-06-05 | Merck Sharp & Dohme Corp. | Compositions and methods for treating cancer |
| WO2014100065A1 (en) | 2012-12-20 | 2014-06-26 | Merck Sharp & Dohme Corp. | Substituted imidazopyridines as hdm2 inhibitors |
| WO2014120748A1 (en) | 2013-01-30 | 2014-08-07 | Merck Sharp & Dohme Corp. | 2,6,7,8 substituted purines as hdm2 inhibitors |
| WO2015034925A1 (en) | 2013-09-03 | 2015-03-12 | Moderna Therapeutics, Inc. | Circular polynucleotides |
| US11040957B2 (en) | 2013-10-18 | 2021-06-22 | Dana-Farber Cancer Institute, Inc. | Heteroaromatic compounds useful for the treatment of proliferative diseases |
| US10906889B2 (en) | 2013-10-18 | 2021-02-02 | Dana-Farber Cancer Institute, Inc. | Polycyclic inhibitors of cyclin-dependent kinase 7 (CDK7) |
| US10017477B2 (en) | 2014-04-23 | 2018-07-10 | Dana-Farber Cancer Institute, Inc. | Janus kinase inhibitors and uses thereof |
| US9862688B2 (en) | 2014-04-23 | 2018-01-09 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged janus kinase inhibitors and uses thereof |
| EP4552697A2 (en) | 2014-08-06 | 2025-05-14 | Novartis AG | Protein kinase c inhibitors and methods of their use |
| WO2016020864A1 (en) | 2014-08-06 | 2016-02-11 | Novartis Ag | Protein kinase c inhibitors and methods of their use |
| EP3514151A1 (en) | 2014-08-06 | 2019-07-24 | Novartis AG | Protein kinase c inhibitors and methods of their use |
| US10870651B2 (en) | 2014-12-23 | 2020-12-22 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 (CDK7) |
| US12168663B2 (en) | 2014-12-23 | 2024-12-17 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 (CDK7) |
| US11325910B2 (en) | 2015-03-27 | 2022-05-10 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| US10550121B2 (en) | 2015-03-27 | 2020-02-04 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| US12098154B2 (en) | 2015-03-27 | 2024-09-24 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| USRE50776E1 (en) | 2015-03-27 | 2026-02-03 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| US10702527B2 (en) | 2015-06-12 | 2020-07-07 | Dana-Farber Cancer Institute, Inc. | Combination therapy of transcription inhibitors and kinase inhibitors |
| US11142507B2 (en) | 2015-09-09 | 2021-10-12 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| JP2019529409A (ja) * | 2016-09-15 | 2019-10-17 | シティ・オブ・ホープCity of Hope | ジチオetp誘導体 |
| WO2018058022A1 (en) | 2016-09-26 | 2018-03-29 | Merck Sharp & Dohme Corp. | Anti-cd27 antibodies |
| WO2018071283A1 (en) | 2016-10-12 | 2018-04-19 | Merck Sharp & Dohme Corp. | Kdm5 inhibitors |
| WO2018190719A2 (en) | 2017-04-13 | 2018-10-18 | Aduro Biotech Holdings, Europe B.V. | Anti-sirp alpha antibodies |
| WO2019094311A1 (en) | 2017-11-08 | 2019-05-16 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| WO2019094312A1 (en) | 2017-11-08 | 2019-05-16 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| WO2019152642A1 (en) | 2018-02-01 | 2019-08-08 | Merck Sharp & Dohme Corp. | Anti-pd-1/lag3 bispecific antibodies |
| US12187701B2 (en) | 2018-06-25 | 2025-01-07 | Dana-Farber Cancer Institute, Inc. | Taire family kinase inhibitors and uses thereof |
| WO2020033288A1 (en) | 2018-08-07 | 2020-02-13 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| US11981701B2 (en) | 2018-08-07 | 2024-05-14 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
| WO2020033282A1 (en) | 2018-08-07 | 2020-02-13 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| US12173026B2 (en) | 2018-08-07 | 2024-12-24 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
| WO2020033284A1 (en) | 2018-08-07 | 2020-02-13 | Merck Sharp & Dohme Corp. | Prmt5 inhibitors |
| US11993602B2 (en) | 2018-08-07 | 2024-05-28 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
| US12552826B2 (en) | 2018-08-07 | 2026-02-17 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
| US12281126B2 (en) | 2018-12-28 | 2025-04-22 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 and uses thereof |
| US12441730B2 (en) | 2019-12-17 | 2025-10-14 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
| US12583871B2 (en) | 2019-12-17 | 2026-03-24 | Merck Sharp & Dohme Llc | PRMT5 inhibitors |
| WO2024180169A1 (en) | 2023-03-02 | 2024-09-06 | Carcimun Biotech Gmbh | Means and methods for diagnosing cancer and/or an acute inflammatory disease |
| WO2026027944A1 (en) | 2024-07-30 | 2026-02-05 | Sairopa B.V. | Anti-sirp alpha antibody formulations and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| US5925641A (en) | 1999-07-20 |
| US5728830A (en) | 1998-03-17 |
| EP0670314A4 (en) | 1996-04-10 |
| EP0670314A1 (en) | 1995-09-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO1995008542A1 (en) | Farnesyltransferase inhibitor | |
| US6350749B1 (en) | Aminoguanidine hydrazone derivatives, process for producing the same and drugs thereof | |
| FR2676446A1 (fr) | Nouveaux derives du thiopyranopyrrole, leur preparation et les compositions pharmaceutiques qui les contiennent. | |
| KR960011775B1 (ko) | 새로운 피롤 유도체, 이들의 제조방법 및 이들을 포함하는 약제학적 조성물 | |
| AU2002233706C1 (en) | Pyridopyrimidine or naphthyridine derivative | |
| RU2198885C2 (ru) | Производные трициклического триазолобензазепина, способы их получения, фармацевтическая композиция и способ лечения аллергических заболеваний, промежуточные соединения и способы их получения | |
| JPWO1995018130A1 (ja) | 3環性ベンゾアゼピンおよびベンゾチアゼピン誘導体 | |
| JPWO1999016770A1 (ja) | 三環性トリアゾロベンゾアゼピン誘導体およびその製造法並びに抗アレルギー剤 | |
| CN116600808A (zh) | 一类作为kras突变体g12c抑制剂的四氢萘啶类衍生物、其制备方法及其应用 | |
| JPH11509541A (ja) | 新規なガランタミン誘導体、その製造方法、その医薬としての用途及びそれを含有する医薬組成物 | |
| Remuzon et al. | Fluoronaphthyridines as antibacterial agents. 6. Synthesis and structure-activity relationships of new chiral 7-(1-, 3-, 4-, and 6-methyl-2, 5-diazabicyclo [2.2. 1] heptan-2-yl) naphthyridine analogs of 7-[(1R, 4R)-2, 5-diazabicyclo [2.2. 1] heptan-2-yl]-1-(1, 1-dimethylethyl)-6-fluoro-1, 4-dihydro-4-oxo-1, 8-naphthyridine-3-carboxylic acid. Influence of the configuration on blood pressure in dogs. A quinolone-class effect | |
| JP3539926B2 (ja) | 抗ウイルス性ピリミジンジオン誘導体及びそれらの製造方法 | |
| EP3932915A1 (en) | Acryloyl-containing nuclear transport regulator and uses thereof | |
| WO2000077003A1 (en) | Optically active pyrrolopyridazine compounds | |
| JP3050982B2 (ja) | 新規なトリアゾール誘導体およびその塩 | |
| WO2004067537A1 (ja) | トリアゾール誘導体およびそれを含んでなる抗真菌剤 | |
| HUP0204579A2 (hu) | Pirrolopiridazinszármazékok, ezeket tartalmazó gyógyszerkészítmények és alkalmazásuk | |
| WO2000041689A1 (en) | Telomerase inhibitors | |
| WO1999002527A1 (fr) | Derives de naphthyridine | |
| HU201082B (en) | Enolethers of 6-chloro-4-hydroxy-2-methyl-N-(2-pyridyl)-2H-thieno(2,3-e)-1,2-thiazin-3-carboxylic acid amide-1,1-dioxide, a process for their preparation and their use. | |
| EP3676260B1 (en) | Thiophene compounds, process for synthesis and use thereof for the treatment of autoimmune disorders | |
| IE903606A1 (en) | Chemical compounds | |
| JPWO1995008542A1 (ja) | ファルネシルトランスフェラーゼ阻害剤 | |
| FR2696179A1 (fr) | Dérivés de thioalkylthiocarbacéphalosporine, procédé pour leurs préparations et formulation pharmaceutique les contenant pour le traitement ou la prophylaxie des infections bactériennes. | |
| RU2838463C2 (ru) | Акрилсодержащие модуляторы ядерного транспорта и их применение |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08436458 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1994927069 Country of ref document: EP |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWP | Wipo information: published in national office |
Ref document number: 1994927069 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref country code: US Ref document number: 1997 912870 Date of ref document: 19970819 Kind code of ref document: A Format of ref document f/p: F |
|
| WWR | Wipo information: refused in national office |
Ref document number: 1994927069 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1994927069 Country of ref document: EP |