WO1997041138A2 - Esculetin derivatives - Google Patents
Esculetin derivatives Download PDFInfo
- Publication number
- WO1997041138A2 WO1997041138A2 PCT/GB1997/001202 GB9701202W WO9741138A2 WO 1997041138 A2 WO1997041138 A2 WO 1997041138A2 GB 9701202 W GB9701202 W GB 9701202W WO 9741138 A2 WO9741138 A2 WO 9741138A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- general formula
- compound
- group
- alkyl
- cyclohexenoesculetin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C[C@]1(*2O)O*2O*(COc(cc(c(C(C)=C2C)c3)OC2=O)c3O)*1O Chemical compound C[C@]1(*2O)O*2O*(COc(cc(c(C(C)=C2C)c3)OC2=O)c3O)*1O 0.000 description 3
- LXXKMMYOKFJCDZ-UHFFFAOYSA-N COc(cc(C(CCCC1)=C1C(O1)=O)c1c1)c1OC Chemical compound COc(cc(C(CCCC1)=C1C(O1)=O)c1c1)c1OC LXXKMMYOKFJCDZ-UHFFFAOYSA-N 0.000 description 1
- YXFVVABEGXRONW-UHFFFAOYSA-N Cc1ccccc1 Chemical compound Cc1ccccc1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D311/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings
- C07D311/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D311/04—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring
- C07D311/06—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring with oxygen or sulfur atoms directly attached in position 2
- C07D311/08—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring with oxygen or sulfur atoms directly attached in position 2 not hydrogenated in the hetero ring
- C07D311/14—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring with oxygen or sulfur atoms directly attached in position 2 not hydrogenated in the hetero ring substituted in position 6 and unsubstituted in position 7
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/655—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms
- C07F9/6552—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms the oxygen atom being part of a six-membered ring
- C07F9/65522—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms the oxygen atom being part of a six-membered ring condensed with carbocyclic rings or carbocyclic ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H17/00—Compounds containing heterocyclic radicals directly attached to hetero atoms of saccharide radicals
- C07H17/04—Heterocyclic radicals containing only oxygen as ring hetero atoms
- C07H17/06—Benzopyran radicals
- C07H17/065—Benzo[b]pyrans
- C07H17/075—Benzo[b]pyran-2-ones
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/02—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving viable microorganisms
- C12Q1/04—Determining presence or kind of microorganism; Use of selective media for testing antibiotics or bacteriocides; Compositions containing a chemical indicator therefor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2334/00—O-linked chromogens for determinations of hydrolase enzymes, e.g. glycosidases, phosphatases, esterases
- C12Q2334/20—Coumarin derivatives
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S435/00—Chemistry: molecular biology and microbiology
- Y10S435/975—Kit
Definitions
- the present invention relates to novel esculetin derivatives and their application as substrates for the specific detection of micro-organisms .
- Esculetin is the aglycone of esculin and of cichoriin and can obtained by hydrolysis of these molecules. Esculetin has the following chemical formula:
- esculetin is 6, 7-dihydroxy-2H- l-benzopyran-2-one (also known as 6, 7-dihydroxycoumarin or cichorigenin) .
- glycoside esculin (6, 7-dihydroxycoumarin-6-glucoside or 6- ( ⁇ -D-glucopyranosyloxy) -7-hydroxy-2H-l-benzopyran-2- one) .
- Synthetic substrates are also available for detection of /3-D-glucosidase to yield either chromogenic or fluorescent compounds upon hydrolysis (Manafi et al Microbiol . Reviews 55 335-348 (1991)).
- the fluorogenic compound 4-methylumbelliferyl-/3-D-glucoside has been widely used although it has several disadvantages when incorporated into an agar medium. These include the fact that the recognition of colonies can only be performed under the presence of long wave ultraviolet light and also the glucose released from the substrate by hydrolysis, may be utilised by the organisms to produce acid. This causes a reduction in the fluorescence produced by 4-methylumbelliferone due to the predominance of the undissociated form of the molecule at low pH.
- the released umbelliferone has a tendency to diffuse through the agar, creating difficulties distinguishing individual colonies producing the target enzyme.
- Other substrates commonly employed include /S-D-glucoside derivatives of nitrophenol (Trepeta et al Antonie van Leewenhoek 53 273-277 (1987)) .
- widespread diffusion is again a severely limiting factor when incorporating such substrates into solid media (Manafi et al Microbiol . Reviews 55 335-348 (1991)) .
- / 3-galactosidase is also an important diagnostic marker in microbial identification. It is perhaps the most widely studied of all microbial enzymes and its presence has long been recognised as a valuable taxonomic marker. This is particularly true in the bacterial family Enterobacteriaceae where assay of /3-galactosidase has been used for many years for the differentiation of non- lactose fermenting species from slow or late lactose fermenters (James, A. L. , In Chemical Methods in Prokaryotic Systematics, 471-492, ed. Goodfellow and O'Donnell, Wiley & Sons (1994)). Numerous substrates are available for the detection of / 3-galactosidase, the most common being ortho-nitrophenyl / ⁇ -D-galactoside (ONPG) which releases yellow o-nitrophenol upon hydrolysis
- OPG ortho-nitrophenyl / ⁇ -D-galactoside
- Fluorogenic substrates have also been used utilising labels such as resorufin, fluorescein and 4- methylumbelliferone (Manafi et al Microbiol . Reviews 55 335-348 (1991) , Plovins et al Applied and Environmental Microbiol . 60 4638-4641 (1994)).
- j ⁇ -galactosidase assay An important application of the j ⁇ -galactosidase assay is the detection of "coliforms" in water and food samples. This has led to the development of membrane filtration techniques which incorporate a suitable substrate for the direct detection of ⁇ -galactosidase (Brenner et al Applied and Environmental Microbiol . 59 3534-3544 (1993) , Ceneci et al Microbios 76 47-54 (19 )) . The most widely used substrate for this purpose is 4-methylumbelliferone /3-D-galactoside.
- this substrate was used in a rapid assay which allowed detection of as few as 1 faecal coliform per 100ml in six hours (Berg et al Applied and Environmental Microbiol . 54 2118-2122 (1988)) .
- the limitations of this substrate are that the released 4-methylumbelliferone readily diffuses across the filter and the fluorescence produced can only be visualised under ultra-violet light.
- chromogenic compounds Due to the limitations of these substrates for identification of / 3-glucosidase and ⁇ -galactosidase, chromogenic compounds have been employed which yield insoluble products upon hydrolysis. Such substrates provide the advantage that the released chromogen remains localised around the bacterial colony without diffusing through the medium (Kodaka et al J. Clinical Microbiol . 33 199-201 (1995)) .
- Examples of these for the detection of /3-glucosidase include, indoxyl /3-D-glucoside and 5- bromo-4-chloro-3-indolyl /3-D-glucoside and examples for the detection of /3-galactosidase include galactosides of indoxyl and its halogenated derivatives such as 5-bromo- 4-chloro-3-indolyl /3-D-galactoside (X-gal) (Kodaka et al J. Clinical Microbiol . 33 199-201 (1995)) .
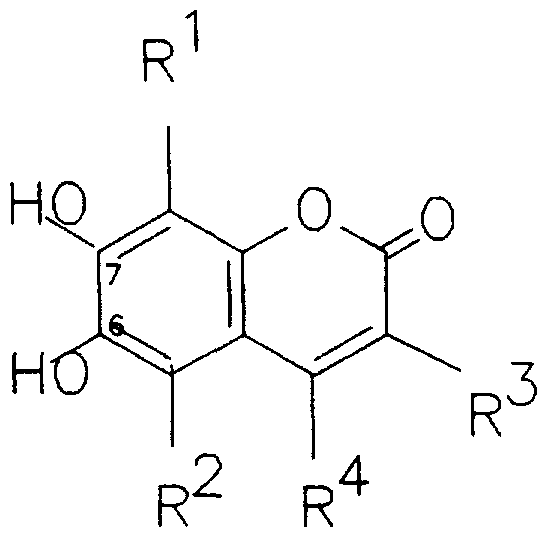
- the present invention relates to certain novel 6- or 7- substituted derivatives of esculetin (6,7- dihydroxycoumarin) which are microbially hydrolysable to fluorescent, iron-chelating esculetin moieties which have a low tendency to diffuse through agar or other aqueous environments without suffering from all the disadvantages referred to above.
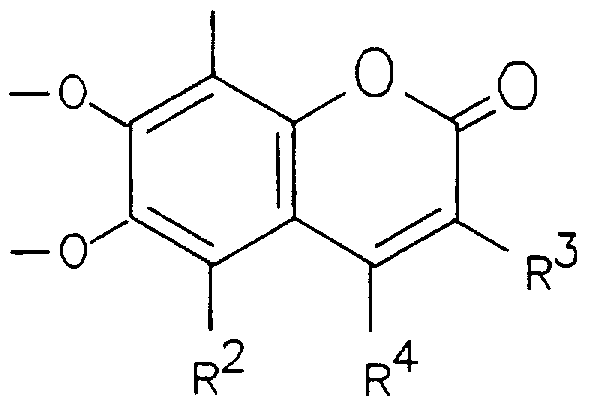
- each of R 1 and R 2 independently represents a hydrogen or a halogen atom or another group which does not interfere with subsequent iron chelation; each of R 3 and R 4 independently represents a hydrogen atom or a (C ⁇ Cg)alkyl or (C 6 or C 10 )aryl (C x -C 8 )alkyl or an optionally modified carboxyl-bearing group of the general formula -CH 2 (CH 2 ) n C0X, where n is a number from 0 to 3 and X represents a hydroxyl group or another hydrophillic group, and, R 3 may alternatively represent an acyl group of the general formula -COR, in which R represent a (C x - C 8 )alkyl, (C 6 or C 10 )aryl (C 1 -C 8 )alkyl or (C 5 -C 8 )cycloalkyl group, provided that R 3 and R 4 between them contain at least three carbon atoms; or R 3 and R 4
- halogen or its abbreviation "halo" means fluoro, chloro, bromo or iodo.
- atom or group which does not interfere with iron chelation refers to the fact that one of the principle means of detection if aglycones of general formula I is by chelation by means of hydroxyl groups at the 6 and 7 positions of the coumarin ring system. Groups which do not interfere with this chelation may be substituted at R 1 and/or R 2 . Examples include hydrogen, hydroxyl, halogen or (C- ⁇ Cg)alkyl.
- the halogen atom may be a fluorine atom or a chlorine atom and the lower alkyl group may be methyl, ethyl, propyl, butyl or benzyl.
- (C 1 -C 8 )alkyl refers to straight chain or branched chain hydrocarbon groups having from one to eight carbon atoms. Illustrative of such alkyl groups are methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, neopentyl, hexyl, heptyl and octyl. From one to four carbon atoms may be preferred.
- (C ⁇ C-, ⁇ )alkyl refers to straight chain or branched chain hydrocarbon groups having from one to ten carbon atoms. Illustrative of such alkyl groups are methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, neopentyl, hexyl, heptyl, octyl, nonyl and decyl. From one to six carbon atoms may be preferred.
- (C 6 or C 10 )aryl includes phenyl and naphthyl.
- (C 5 -C 8 )cycloalkyl refers to an alicyclic group having from 5 to 8 carbon atoms. Illustrative of such cycloalkyl groups are cyclopentyl and cyclohexyl.
- (C 5 -C 8 )cycloalkene ring refers to an alicyclic ring having from 5 to 8 atoms and having in addition one or more double bonds.
- Illustrative of such cycloalkenyl groups are cyclopentenyl, cyclohexenyl, cycloheptenyl and cyclooctenyl.
- suitable salt refers to a salt prepared by contacting a compound of formula I with an acid or base whose counterpart ion does not interfere with the intended use of the compound.
- Examples include the sodium salt or magnesium salt of a phosphate derivative or the salt formed from a primary, secondary or tertiary amine where the compound of general formula I is a carboxylic acid.
- An example of a primary amine salt can be the cyclohexylammonium salt, a suitable secondary amine salt may be the piperidine salt and a tertiary amine salt may be the triethylamine salt .
- Preferred compounds of general formula I include those in which, independently or in any compatible combination:
- R 1 is chlorine or, preferably hydrogen;
- R 2 is chlorine or, preferably hydrogen;
- R 3 is (C 1 -C 4 ) alkyl, particularly butyl, or benzyl;
- R 4 is (C 1 -C 4 )alkyl; or, -CH 2 (CH 2 ) n COX, where n is a number from 0 to 3 and X represents a hydroxyl group or one of the following hydrophillic groups, namely: -NHCH 2 CONHCH 2 C0 2 H -NHCH 2 C0NHCH 2 C0NHCH 2 C0 2 H -NHCHCH 2 CONH 2
- R 3 and R 4 together with the carbon atoms to which they are attached form a (C 5 -C 8 ) cycloalkene ring, preferably a cyclopentenyl or cyclohexenyl ring;
- R 3 is -CH 2 (CH 2 ) n C0X, where n is a number from 0 to 3, then the group X is as previously defined,
- the enzymatically cleavable group represented by Y or Z is an ⁇ ;- or, preferably, /S-linked sugar residue such as /3-D-glucose, /3-D-galactose, /3-D-xylose, /3-D-glucuronic acid or N-acetyl-/3-D-glucosamine, or a phosphate or a carboxylate (R 5 COO-) group, where R 5 represents a ⁇ C ⁇ - C 10 ) alkyl group.
- Sugar residues, particularly those derived from glucose and galactose are the most preferred compounds.
- the aglycone moiety of general formula I is a substituted esculetin.
- Y is a glucose moiety and Z is hydrogen (i.e. when the esculetin ring system is substituted at the 7- position) the compounds of general formula I are cichoriins.
- Z is a glucose moiety (i.e. when the esculetin ring system is substituted at the 6- position) the compounds of general formula I are esculins.
- Cichoriins, and analogues of cichoriins when the sugar residues other than glucose are substituted at the 7-hydroxyl group position, are preferred.
- each of Y or Z may independently represent an (C x - C 10 )alkylcarbonyl group.
- Preferred esters include the octanoate and butyrate esters. Ester derivatives are useful where the microbial enzyme is an esterase.
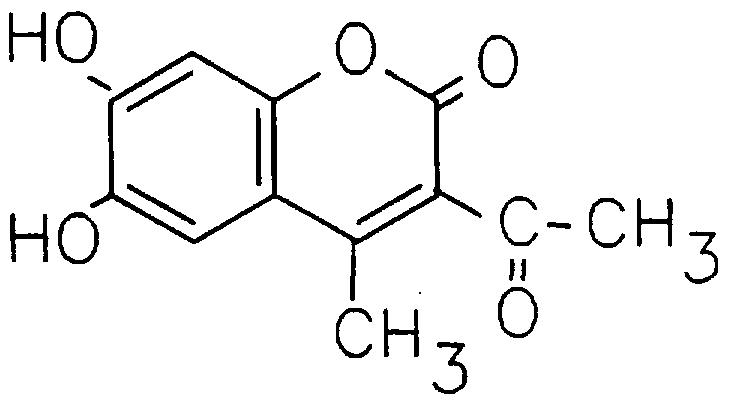
- Preferred compounds of general formula I are:
- a process for the preparation of the compounds defined in general formula I comprising derivatising an optionally protected compound of general formula II at position 6 or 7, and optionally thereafter converting the compound of general formula I so formed into another compound of general formula I.
- Derivatising the compound of general formula II to form a compound of general formula I includes glycosylation, phosphorylation and esterification.
- the glycoside derivative of the compounds of general formula II may be formed by treating the compound with a glycoside under suitable conditions. Glycosylation of an esculetin derivative occurs at the 7-position on the coumarin ring unless this position is blocked in which case glycosylation may take place at the 6-position.
- An example of a suitable blocking group is a benzylic halide, such as benzyl chloride, benzyl bromide, benzyl iodide, or other group such as benzyl cyanide (Greene Protective Groups in Organic Synthesis, publ. Wiley- Interscience 97-99 (1981)). Glycosylation is then followed by debenzylation by suitable treatment.
- the hydroxyl groups of the glycoside be protected by addition of a removable protector group such as an acetyl moiety.
- a suitable protected glycoside compound is the tetraacetyl form of the /3-glucoside or /3-galactoside.
- the tetraacetyl derivative may be derived from the corresponding ⁇ - acetobromohexose.
- Deprotection of the hydroxyl groups may be undertaken at any particular time, but in practice this will be the final step in the formation of the desired compound. Deprotection may comprise treatment with a solution of sodium methoxide in methanol where the protector group is an acetyl moiety.
- Conditions for glycosylation may be chosen by the person skilled in the art without undue experimentation. However, suitable conditions may comprise treating the protected glycoside of the esculetin in a solution of potassium hydroxide in aqueous acetone, followed by deprotection.
- Organic esters may be formed from the appropriate 6,7- dihydroxycoumarin carboxylic acid of general formula II.
- the dihydroxycoumarin carboxylic acid may be optionally first acetylated to protect the 6- and 7-hydroxyl groups and can then converted to the acid chloride by addition of thionyl chloride.
- the mono-, di-, or tri-aminoacyl can be formed by conjugation of the acid chloride with the appropriate aminoacyl compound.
- the aminoacyl conjugate may be esterified by treatment with a (C x - C 10 )alkyl carbonylhalide in the presence of pyridine.
- suitable alkyl carbonylhalides include, octanoyl chloride or butyryl chloride.
- Formation of phosphate ester derivatives may be achieved by treating the esculetin derivative with phosphorous chloride (P0C1 3 ) in the presence of pyridine (or other appropriate monophosphorylating agent) .
- a compound of general formula I can optionally be modified to another compound of general formula I.
- the glycoside may be modified in si tu ; e.g. a glucose residue may be modified to a glucuronic acid residue with oxygen in the presence of platinum catalyst and carbon.
- other glycosides may be oxidised to their 6- carboxyl derivatives (glycuronic acids) .
- 7- substituted substrates e.g. / 3-glucosides of general formula I where Y is glucose, can by appropriate reactions at the vacant phenolic group (6-hydroxyl) yield other substrates e.g.
- the compounds of general formula II may be prepared by treating an appropriate linear or cyclic ⁇ -ketoester (substituted or unsubstituted) of general formula III with a compound of general formula IV in a Pechmann condensation.
- each of R 6 and R 7 independently represent an acetyl (CH 3 CO-) or an acyl group of the formula X(CH 2 ) n CO-, where n is a number from 0 to 6 and X represents:
- R 7 represents hydrogen, a (C ⁇ C g )alkyl, (C 5 - C 8 )cycloalkyl, a (C 6 or C 10 )aryl (C- ⁇ Cg)alkyl, or a (C 6 or C 10 )aryl (C 1 -C 8 )alkyl aroyl group; or,
- R 6 and R 7 together form a cyclic ketone -(CH 2 ) n CO-, where n is a number between 4 and 7; and,
- R 8 represents (C 1 -C 8 )alkyl, phenyl or methylphenyl.
- each R 9 independently represents hydrogen or (C j -Cg)alkyl-CO; R 1 and R 2 are as described previously.
- the preferred compounds of general formula IV are 1,2,4- trihydroxybenzene derivatives
- 1,2,4-trihydroxybenzene is relatively unstable and the preferred starting material is therefore 1,2,4-triacetohydroxybenzene.
- 1,2,4- triacetohydroxybenzene may be synthesised by Thiele- Winter acetylation of the corresponding benzoquinone compound.
- Preferred /3-ketoesters are: ethyl-2-n-butylacetoacetate ethyl-2-benzylacetoacetate, ethyl-2-cyclohexanone carboxylate, and ethyl-2-cyclopentanone carboxylate.
- the product of this step is a compound of general formula II as defined above.
- Other compounds mentioned are trivially synthesisable by methods known in the art.
- 6, 7-dihydroxycoumarin carboxylic acids within general formula II may be carried out by treating a suitable /3-ketoester of general formula III, where X is
- Preferred starting compounds include diethylacetone dicarboxylate, dimethyl 2-acetylglutarate or diethyl acetylglutarate. Where diethylacetone dicarboxylate is treated with 1,2, 4-triacetoxybenzene, the product is 6,7- dihydroxycoumarin-4-acetic acid, after hydrolysis.
- composition comprising a compound of general formula I and optionally a microbiologically acceptable diluent, excipient or substrate.
- the substrate may be a solid or semi-solid growth support medium or a liquid growth support medium.
- Agar is the traditional support medium used in microbiology for the growth of microorganisms. It is prepared by autoclaving the dehydrated agar in water with other components as necessary. The autoclaving process at temperatures above 100°C causes the agar to rehydrate to form a gelatinous liquid agar solution. The liquid agar formed is allowed to cool slightly and is then poured, while still liquid, into suitable containers such as culture plates or Petri dishes. The agar will gel to form a support medium upon further cooling.
- agar will depend on the strain of micro-organism to be grown but may be selected from the one of the following preparations: bacteriological agar, Columbia agar, noble agar, select agar, brain-heart infusion agar, LB agar or Lennox Broth agar, luria agar, yeast extract or tryptone digest .
- Other choices of medium are described in Sambrook et al Molecular Cloning, 2nd edition, A1-A3, Cold Spring Harbor Laboratory (1989) and in Difco Manual 10th edition, Difco Laboratories, Detroit, MI, USA.
- the compounds of the present invention may admixed with the dehydrated agar as separate components or prepared as combined particles as appropriate.
- the growth medium selected may also comprise one or more essential nutrient, mineral, vitamin and/or antibiotic to select, promote or assist the growth of a particular micro-organism or to assist in the identification or enumeration of a particular micro ⁇ organism.
- Antibiotics commonly used in micro-biological growth media include: ampicillin, chloramphenicol, D- cycloserine, gentamicin, kanamycin, nalidixic acid, rifampicin, spectinomycin, streptomycin and tetracycline.
- Essential nutrients include amino acids for which the micro-organism to be grown is growth deficient or a fermentable carbon source such as a carbohydrate. It may also be necessary to add certain minerals, such as for example magnesium, calcium, sodium, potassium or iron, in the form of an appropriate salt, or ammonium or phosphate salts.
- a solid support medium such as agar
- a suitable container comprises a base with an overhanging lid to permit oxygen to be supplied to aerobic bacteria when the plate is being incubated.
- the lid may be sealed to the base or the plate may be incubated in an anaerobic incubator.
- a method for the detection and/or identification of micro-organisms in a sample comprising the step of (a) growing a micro-organism isolated from the sample on a growth support medium containing a compound of general formula I and detecting the release of an identifiable chromogenic or fluorogenic marker following hydrolysis of the compound of general formula I.
- the microorganisms may also be present in a liquid growth support medium or a liquid sample to be analysed.
- the method according to the present invention can be used to detect both Gram negative and Gram positive bacteria.
- the method has application to, but is not limited to, the detection of the following genera: Enterococcus, Listeria, Streptococcus, Citrobacter, Enterobacter, Escherichia, Hafnia, Klebsiella, Proteus, Providencia, Salmonella, Serratia, Shigella and Yersinia .
- the fluorescent or, in conjunction with iron, coloured marker detected in the method is the result of the hydrolysis of the compound of general formula I which liberates the esculetin moiety. Hydrolysis of the compound may be characteristic of the presence of a micro-organism to be detected.
- micro-organisms Specific detection of micro-organisms is required in hospitals to assist in clinical diagnosis and effective assessment of an appropriate course of medical treatment.
- Common biological samples that a clinician will want analyse for their micro-organism content are saliva, urine, blood, faeces, the contents of the stomach or a biopsy sample.
- the food and drinks industries also require specific detection of micro-organisms in order to monitor and maintain product quality and safety.
- the water industry also needs to be able to monitor the presence and quantity of micro-organisms present in the water supply. Suitable samples for analysis in the method according to this aspect of the invention will therefore be an edible substance, water or other potable liquid.
- the method in its preferred form utilises the various novel esculetin derivatives as indicators of particular enzymatic activities by incorporation into agar media.
- colonies can be visualised and, provided that the media are appropriately selective, identified.
- the method adapts well to the use of cellulose nitrate membrane filters and thereby to colony counting.
- the compounds of the present invention may be present in the growth support medium at an effective concentration which may be of from 0.Img/ml to 2mg/ml, suitably of from 0.2mg/ml to lmg/ml and preferably of from 0.4mg/ml to 0.8mg/ml. It is preferred that a concentration of 0.5mg/ml be used when the growth support medium is Columbia agar.
- kits comprising a growth medium containing a compound of general formula I.
- the kit may additionally comprise a container such as a Petri or culture dish.
- 1,2,4-triacetoxybenzene (6.3g, 25mmol) and ethyl cyclohexanone-2-carboxylate (4.25g, 25mmol)with heated together in a small flask until a homogenous viscous liquid was obtained.
- the lowest temperature possible was employed.
- the hot liquid was cooled somewhat and a magnetic stirrer introduced. Cooling below 30°C caused solidification and therefore at this temperature 75% w/w sulphuric acid (35ml) cooled to 10°C was rapidly added to the stirred liquid. This lowered the temperature whilst maintaining homogeneity.
- the reaction was allowed to continue at ambient temperature for 6 hours, a white suspension forming after about 3 hours.
- the suspension was poured in a thin stream into rapidly stirred ice/water (300ml) and gave a suspension of cream- coloured solid. After stirring for 10 minutes, the solid was removed by vacuum filtration, washed with abundant water, and sucked dry, then air dried at 40-50°C. The crude material was crystallised from hot ethanol. On cooling, pale yellow needles separated. These were removed by vacuum filtration and air dried (5.2g (89%)) .
- 1,2,4-triacetoxybenzene (6.3g, 25mmol) and ethyl diacetoacetate (4.3g, 25mmol) were heated together as described previously and treated with 75% sulphuric acid (35ml) . After 16 hours the crude product was isolated by pouring into ice/water. The air dried material was crystallised from hot ethanol with addition of hot water to the point of crystallisation. On cooling, pale yellow needles formed (3.5g, 60%) .
- 1,2,4-triacetoxybenzene (6.3g, 25mmol) and dimethyl 2- acetylglutarate (5.75g, 25mmol) were heated together and treated with 75% sulphuric acid (35ml) as described previously. After stirring for 20 hours, the crude material was isolated by pouring into ice/water and the product collected after washing with water. The product (ethyl 6, 7-dihydroxy-4-methyl coumarin 3-propionate) was crystallised from hot ethanol (4.1g, 56%) . The ester was hydrolysed by stirring with an aqueous solution of potassium hydroxide (3% w/v) comprising water
- Cognate preparation 6 , 7 -dihvdroxycoumarin-4 -acetic acid .
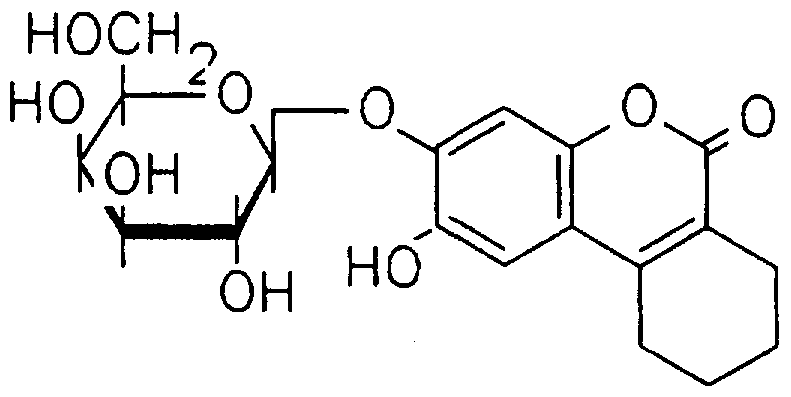
- 3,4-cyclohexenoesculetin (6,7 -dihydroxy- 3,4- cyclohesenocoumarin) (3.5g, 15mmol) was dissolved in an aqueous solution of potassium hydroxide; 16ml of 10% w/v KOH (29mmol) . With stirring, the esculetin rapidly dissolved. To this was added ⁇ -acetobromoglucose (6.25g, 15mmol) dissolved in acetone (18ml) . The solution was homogenous and became lighter in colour on stirring. After 18 hours at 10-15°C a yellow precipitate formed. This was removed by filtration to give a yellow cake (3.0g dryweight) .
- the filtrate was poured slowly into stirred ice-water (200ml) .
- the precipitate which formed was filtered onto a Buchner funnel, washed with water and dried to give a gummy mass. This was dissolved in dichloromethane (50ml) with warming. The slight precipitate which formed on cooling was removed. TLC showed this to be 3,4- cyclohexenoesculetin.
- the filtrate contained the tetraacetyl glycoside with a trace of the esculetin.
- the tetraacetyl compound (2.5g) was dissolved in methanol (50ml) and treated with sodium methoxide solution (20ml) the progress of deacetylation was followed by TLC until only baseline material (solvent - ethyl acetate/toluene, 3:1) remained.
- the methanolic solution was brought to pH 6.5 by addition of aliquots of Amberlite 1R120(H + ), analytical grade, washed after treatment with acid. The supernatant liquid was decanted from the resin and the latter washed with methanol (3x10ml) . Methanol was removed by rotary evaporation at 30-40°C and the residual solid crystallised from methanol/water (1.5g, 86%).
- 3-benzyl-4-methyl esculetin (Example 2) (4.25g, 15.6mmol) was dissolved in an aqueous solution of potassium hydroxide (16.5ml of 10% w/v KOH (29mmol) with stirring. A yellow solid, the potassium salt, soon separated. A solution of cc-acetobromoglucose (6.6g, 16mmol) dissolved in acetone (20ml) was added to the stirred solution. Further addition of acetone (3-4 ml) ensured homogeneity. The light brown solution was stirred overnight (16 hours) . The resulting paste was filtered with suction and the residue sucked dry, then air dried at room temperature.
- the tetraacetylglucoside was extracted into hot chloroform (3x30ml) and the resulting suspension filtered from a scanty precipitate.
- the chloroform solution was washed with cold dilute (3%) sodium carbonate solution (3x30ml) followed by water.
- the organic phase was dried using anhydrous magnesium sulphate and rotary evaporated yielding a white solid. This was dissolved in hot ethanol (100ml) and cooled to 0°C overnight. The crystals were removed by suction filtration and pressing. After drying in a vacuum over the product weighed 3.0g(32%) .
- Deacetylation of the tetraacetylglucoside (3.0g) using methanolic sodium methoxide was achieved as described in Example 5, and gave the /3-glucoside (1.8g, 81.4%) .
- 3,4-cyclohexenoesculetin (6.95g, 30mmol) was dissolved in an aqueous solution of potassium hydroxide (35ml of 10% w/v KOH (62.5 mmol) .
- ⁇ -acetobromogalactose (13.6g, 33mmol) was dissolved in acetone (36ml) and this solution was added to the stirred aqueous esculetin solution. Addition of a little more acetone was needed to give a homogenous solution.
- the reaction mixture was stirred overnight (10-15°C) . Precipitation commenced after 4 hours. The suspension was filtered after 16 hours. The residue ⁇ lg was shown to be mainly the unchanged esculetin, and was rejected.
- the filtrate was biphasic, consisting of a lower viscous phase and an upper aqueous layer.
- the lower phase after separation was poured slowly into well stirred ice/water (250ml) to give an abundant white precipitate. This was removed by suction filtration, washed with cold water and dried. The dry solid was dissolved in dichloromethane. TLC showed a large fast-running spot of the tetraacetyl galactoside and very little of the esculetin.
- the aqueous upper layer from the reaction mixture was concentrated by rotary evaporation at 30-40°C and poured into ice water.
- the product was much less than from the lower layer but still contained a substantial amount of the desired product.
- the precipitated solid was removed by suction filtration, dried and dissolved in dichloromethane.
- the oily tetraacetyl galactoside was deprotected by solution in methanol and addition of methanolic sodium methoxide (30ml of 1% solution) as described in Example 5. After evaporation of methanol, the galactoside formed an oil which rapidly solidified to a mass of white crystals (5.2g, 44.8%)
- acetylated methyl uronate was deprotected by solution in methanol and addition of one-fifth mol. ratio of methanolic sodium hydroxide (95% methanol/5% water) . After stirring for 5 hours, methanol was removed by rotary evaporation. Water (10ml) was added and the solution neutralised with 2M hydrochloric acid. Water was removed by rotary evaporation at 60°C and the residue suspended and stirred in acetone for 1 hour. The suspended glucuronide was removed by suction filtration and acetone removed by rotary evaporation to give an amorphous light brown powder (0.41g, 65%) .
- glycosides were synthesised although not isolated in a pure state.
- the oily acid chloride was directly dissolved in tetrahydrofuran (20ml) and introduced in a slow stream into a well stirred solution of glycylglycine (1.32g, lOmmol) dissolved in a mixture of water (20ml) and pyridine (2ml) to act as base. After 3 hours the solution was rotary evaporated to remove tetrahydrofuran and deprotected by addition of concentrated ammonia (5ml) . After 1 hour, deacetylation was complete and solvents were removed by rotary evaporation under reduced pressure at 50-60°C. The product was contaminated with excess glycylglycine and salts and was therefore recrystallised from methanol/water to give the product N(6,7-dihydroxycoumarin-4-acetyl)glycylglycine.
- the glycylglycine conjugate (1.68g, 5mmol) was dissolved in pyridine (10ml) and to the stirred solution was added octanoyl chloride (0.9g, 5.5mmol) the reaction mixture was stirred at room temperature for 2 hours and then poured into a mixture of ice/water (90ml) and concentrated hydrochloric acid (10ml) with efficient stirring.
- the gummy solid which separated was- dissolved in ethyl acetate and washed with concentrated sodium chloride solution.
- the organic phase was separated, dried (anhydrous sodium sulphate) and rotary evaporated to give a waxy solid of the octanoate ester of the hydrophillic esculetin.
- Example 11 Test procedures using compounds previously synthesised in Examples 1 to 10
- the medium was allowed to cool to 55°C before being poured in 20ml volumes.
- Agar containing 3,4,- cyclohexenoesculetin-j ⁇ -D-glucoside was prepared in an identical fashion except that this novel glucoside (CHE- /3-D-gluc) (0.5g) was substituted for esculin.
- Suspensions of these 10 strains were prepared as above at IO 8 organisms per ml and diluted in sterile distilled water by standard methods to produce suspensions of approximately 1 organism per ml. 3x100 ml volumes of each suspension were then filtered onto 3 cellulose nitrate membrane filters (Sartorius) using a standard filtration method. One of these filters was placed onto a CHE-/3-D-glucoside plate, the second onto an esculin plate and the third onto a Columbia agar plate without substrate. All plates were incubated at 37°C in air for exactly 18 hours, except for the plates inoculated with streptococci which were incubated in air supplemented with 5% carbon dioxide. After incubation plates were examined for the presence of black or brown colonies and colony counts were performed on the membrane filters.
- Figure 1 shows a strain of Listeria monocytogenes (NCTC 11994) on CHE-/3-D- glucoside medium and a traditional esculin-based selective agar.
- NCTC 11994 Listeria monocytogenes
- CHE- /3-D-glucoside would be to look for generation of fluorescence in the absence of iron salts .
- CHE-/3-D-glucoside is a highly useful substrate which produces a black non-diffusible product upon hydrolysis by /3-D-glucosidase in the presence of iron.
- the substrate appears to be non- inhibitory and its synthesis is relatively straightforward.
- Enterococcus casseliflavus 1 100 100 Enterococcus durans 2 100 100
- Enterococcus faecalis 83 100 100 Enterococcus gallinarum 3 100 100 Enterococcus raffinosus 5 100 100 Enterococcus faecium 56 100 100 Listeria ivanovii 2 100 100
- Listeria monocytogenes 10 100 100 Streptococcus agalactiae 7 0 0 Streptococcus bovis 10 100 100 Streptococcus dysgalactiae 2 0 0 Streptococcus mitis 4 0 0 Streptococcus mutan ⁇ 2 100 100 Streptococcus oralis 4 0 0 Streptococcus pneumoniae 4 0 0 Streptococcus pyogenes 2 100 100 Streptococcus sanguis 5 40 40
- Example 12 CHE-glucoside in blood agar
- Cyclohexenoesculetin-glucoside was incorporated into Blood agar bases for detection and identification of organisms producing haemolysis and glucosidase .
- CHE-glucoside Blood agar bases can be used to detect the presence of glucosidase producing anaerobic bacteria such as Bact . fragilis.
- CHE-glucoside incorporated into blood agar bases gives clear detection of glucosidase enzymes with no interference with haemolysis patterns. Using these two indicators and appropriate selective agents these important pathogens can be isolated and provisionally identified on primary culture plates.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Biotechnology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Immunology (AREA)
- Microbiology (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Toxicology (AREA)
- Biophysics (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Saccharide Compounds (AREA)
- Furan Compounds (AREA)
- Investigating Or Analysing Biological Materials (AREA)
- Manufacture And Refinement Of Metals (AREA)
- Pyrane Compounds (AREA)
- Apparatus Associated With Microorganisms And Enzymes (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
Abstract
Description
Claims
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| NZ332606A NZ332606A (en) | 1996-05-01 | 1997-05-01 | Esculetin derivatives substituted by a beta linked sugar residue or a phosphate group or a carboxylate group, their use as substrates for detecting microorganisms |
| AT97918287T ATE210674T1 (en) | 1996-05-01 | 1997-05-01 | ESCULETIN DERIVATIVES |
| KR1019980708651A KR100517757B1 (en) | 1996-05-01 | 1997-05-01 | Esculetin derivatives |
| DE69709092T DE69709092T2 (en) | 1996-05-01 | 1997-05-01 | ESCULETIN DERIVATIVES |
| DK97918287T DK0900230T3 (en) | 1996-05-01 | 1997-05-01 | Esculetin derivatives |
| CA002253506A CA2253506C (en) | 1996-05-01 | 1997-05-01 | Esculetin derivatives |
| AU26469/97A AU713108B2 (en) | 1996-05-01 | 1997-05-01 | Esculetin derivatives |
| EP97918287A EP0900230B1 (en) | 1996-05-01 | 1997-05-01 | Esculetin derivatives |
| JP53867697A JP4155599B2 (en) | 1996-05-01 | 1997-05-01 | Esculetin derivatives |
| NO19985070A NO313010B1 (en) | 1996-05-01 | 1998-10-30 | Eskuletin derivatives, method of preparation thereof, preparation, culture dish and kit, and method of detection and / or identification of a microorganism in a sample |
| US09/184,231 US6008008A (en) | 1996-05-01 | 1998-11-02 | Esculetin derivatives |
| US09/472,980 US6287798B1 (en) | 1996-05-01 | 1999-12-28 | Esculetin derivatives |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GBGB9609024.6A GB9609024D0 (en) | 1996-05-01 | 1996-05-01 | Compounds |
| GB9609024.6 | 1996-05-01 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/184,231 Continuation US6008008A (en) | 1996-05-01 | 1998-11-02 | Esculetin derivatives |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO1997041138A2 true WO1997041138A2 (en) | 1997-11-06 |
| WO1997041138A3 WO1997041138A3 (en) | 1997-12-11 |
Family
ID=10792941
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB1997/001202 Ceased WO1997041138A2 (en) | 1996-05-01 | 1997-05-01 | Esculetin derivatives |
Country Status (15)
| Country | Link |
|---|---|
| US (2) | US6008008A (en) |
| EP (1) | EP0900230B1 (en) |
| JP (1) | JP4155599B2 (en) |
| KR (1) | KR100517757B1 (en) |
| AT (1) | ATE210674T1 (en) |
| AU (1) | AU713108B2 (en) |
| CA (1) | CA2253506C (en) |
| DE (1) | DE69709092T2 (en) |
| DK (1) | DK0900230T3 (en) |
| ES (1) | ES2169859T3 (en) |
| GB (1) | GB9609024D0 (en) |
| NO (1) | NO313010B1 (en) |
| NZ (1) | NZ332606A (en) |
| PT (1) | PT900230E (en) |
| WO (1) | WO1997041138A2 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6368817B1 (en) | 1997-06-04 | 2002-04-09 | Newcastle Upon Tyne Hospitals National Health Service Trust | Identification of salmonella |
| US7029886B1 (en) | 1999-10-28 | 2006-04-18 | Bio Merieux | Enzymatic substrate, synthesis method and uses |
| US7538233B2 (en) | 2003-09-05 | 2009-05-26 | Aventis Pharmaceuticals Inc. | Coumarins as iNOS inhibitors |
| US8404460B2 (en) | 2008-01-21 | 2013-03-26 | Biomerieux | Method for detecting and/or identifying Clostridium difficile |
| WO2015155018A1 (en) * | 2014-04-11 | 2015-10-15 | Balel S.A. | Method for detecting and identifying clostridium difficile |
| EP3196274A1 (en) * | 2010-09-21 | 2017-07-26 | Etablissements J. Soufflet | Sulfonated coumarins, synthesis thereof, and uses thereof |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9609024D0 (en) * | 1996-05-01 | 1996-07-03 | Idg Uk Ltd | Compounds |
| US6350588B1 (en) * | 1999-07-20 | 2002-02-26 | Micrology Laboratories, Llc | Test media and quantitative or qualitative method for identification and differentiation of biological materials in a test sample |
| US7344854B2 (en) * | 1999-07-20 | 2008-03-18 | Micrology Laboratories, Llc | Test media for quantitative or qualitative identification and differentiation of general coliforms, E. coli, Aeromonas spp and Salmonella spp in a test sample |
| US7273719B2 (en) * | 1999-07-20 | 2007-09-25 | Micrology Laboratories, Llc | Test media for quantitative or qualitative identification and differentiation of general coliforms, E coli, Aeromonas spp and Salmonella spp materials in a test sample |
| FR2810676B1 (en) * | 2000-06-27 | 2003-03-07 | Alain Rambach | CULTURE MEDIUM FOR THE DETECTION AND / OR DISCRIMINATION OF VIBRIO BACTERIA AND METHOD OF IMPLEMENTING |
| US20070155701A1 (en) * | 2002-08-23 | 2007-07-05 | Alexandros Makriyannis | Keto cannabinoids with therapeutic indications |
| US7094528B2 (en) * | 2004-06-30 | 2006-08-22 | Kimberly-Clark Worldwide, Inc. | Magnetic enzyme detection techniques |
| US7906276B2 (en) * | 2004-06-30 | 2011-03-15 | Kimberly-Clark Worldwide, Inc. | Enzymatic detection techniques |
| US7504235B2 (en) | 2005-08-31 | 2009-03-17 | Kimberly-Clark Worldwide, Inc. | Enzyme detection technique |
| US8758989B2 (en) * | 2006-04-06 | 2014-06-24 | Kimberly-Clark Worldwide, Inc. | Enzymatic detection techniques |
| US7897360B2 (en) | 2006-12-15 | 2011-03-01 | Kimberly-Clark Worldwide, Inc. | Enzyme detection techniques |
| US20100111914A1 (en) | 2007-05-21 | 2010-05-06 | Yuanyuan Zhang | Stem cells from urine and methods for using the same |
| EP2146728A4 (en) * | 2007-05-21 | 2010-12-29 | Univ Wake Forest Health Sciences | Progenitor cells from urine and methods for using the same |
| EP2714923A4 (en) * | 2011-05-31 | 2015-07-08 | Univ Texas | METHODS AND COMPOSITIONS FOR THE DETECTION OF FUNCTIONAL DIFFICULT CLOSTRIDIUM TOXINS |
| GB201319768D0 (en) | 2013-11-08 | 2013-12-25 | Glycosynth Ltd | Naphthalene derived chromogenic enzyme substrates |
| EP3857220B1 (en) | 2018-09-24 | 2023-01-18 | 4GENE GmbH | Method, device and indicator for localizing damage by overheating |
| WO2020247723A1 (en) | 2019-06-06 | 2020-12-10 | Albert Einstein College Of Medicine | Rapid synthesis of metal nanoparticles |
| KR102664861B1 (en) * | 2024-01-17 | 2024-05-10 | 주식회사 바오젠 | New compound obtained through biorenovation or compositions containing the same for preventing or treating hair loss |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59192099A (en) * | 1983-04-12 | 1984-10-31 | Ajinomoto Co Inc | Measurement of number of microorganism |
| US5055594A (en) * | 1990-07-19 | 1991-10-08 | Becton, Dickinson And Company | Fluorogenic trypotophanase substrates |
| JPH06312925A (en) * | 1993-03-02 | 1994-11-08 | Kureha Chem Ind Co Ltd | Cartilage-protection agent and new esculetin derivative |
| WO1994024119A1 (en) * | 1993-04-13 | 1994-10-27 | Morinaga Milk Industry Co., Ltd. | Coumarin derivative and use thereof |
| JP2902318B2 (en) * | 1994-12-28 | 1999-06-07 | 呉羽化学工業株式会社 | Esculetin derivatives, their production method and matrix metalloprotease inhibitors |
| GB9609024D0 (en) * | 1996-05-01 | 1996-07-03 | Idg Uk Ltd | Compounds |
-
1996
- 1996-05-01 GB GBGB9609024.6A patent/GB9609024D0/en active Pending
-
1997
- 1997-05-01 DK DK97918287T patent/DK0900230T3/en active
- 1997-05-01 JP JP53867697A patent/JP4155599B2/en not_active Expired - Fee Related
- 1997-05-01 ES ES97918287T patent/ES2169859T3/en not_active Expired - Lifetime
- 1997-05-01 AU AU26469/97A patent/AU713108B2/en not_active Expired
- 1997-05-01 AT AT97918287T patent/ATE210674T1/en active
- 1997-05-01 EP EP97918287A patent/EP0900230B1/en not_active Expired - Lifetime
- 1997-05-01 CA CA002253506A patent/CA2253506C/en not_active Expired - Lifetime
- 1997-05-01 KR KR1019980708651A patent/KR100517757B1/en not_active Expired - Fee Related
- 1997-05-01 WO PCT/GB1997/001202 patent/WO1997041138A2/en not_active Ceased
- 1997-05-01 NZ NZ332606A patent/NZ332606A/en not_active IP Right Cessation
- 1997-05-01 PT PT97918287T patent/PT900230E/en unknown
- 1997-05-01 DE DE69709092T patent/DE69709092T2/en not_active Expired - Lifetime
-
1998
- 1998-10-30 NO NO19985070A patent/NO313010B1/en not_active IP Right Cessation
- 1998-11-02 US US09/184,231 patent/US6008008A/en not_active Expired - Lifetime
-
1999
- 1999-12-28 US US09/472,980 patent/US6287798B1/en not_active Expired - Lifetime
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6368817B1 (en) | 1997-06-04 | 2002-04-09 | Newcastle Upon Tyne Hospitals National Health Service Trust | Identification of salmonella |
| US7029886B1 (en) | 1999-10-28 | 2006-04-18 | Bio Merieux | Enzymatic substrate, synthesis method and uses |
| US7538233B2 (en) | 2003-09-05 | 2009-05-26 | Aventis Pharmaceuticals Inc. | Coumarins as iNOS inhibitors |
| US8404460B2 (en) | 2008-01-21 | 2013-03-26 | Biomerieux | Method for detecting and/or identifying Clostridium difficile |
| EP3196274A1 (en) * | 2010-09-21 | 2017-07-26 | Etablissements J. Soufflet | Sulfonated coumarins, synthesis thereof, and uses thereof |
| WO2015155018A1 (en) * | 2014-04-11 | 2015-10-15 | Balel S.A. | Method for detecting and identifying clostridium difficile |
| FR3019833A1 (en) * | 2014-04-11 | 2015-10-16 | Balel S A | METHOD FOR DETECTION AND IDENTIFICATION OF DIFFICULT CLOSTRIDIUM |
Also Published As
| Publication number | Publication date |
|---|---|
| NZ332606A (en) | 1999-10-28 |
| NO985070L (en) | 1998-10-30 |
| CA2253506C (en) | 2007-09-25 |
| AU713108B2 (en) | 1999-11-25 |
| WO1997041138A3 (en) | 1997-12-11 |
| EP0900230A2 (en) | 1999-03-10 |
| JP2000510457A (en) | 2000-08-15 |
| US6287798B1 (en) | 2001-09-11 |
| DK0900230T3 (en) | 2002-04-08 |
| NO313010B1 (en) | 2002-07-29 |
| KR20000065074A (en) | 2000-11-06 |
| AU2646997A (en) | 1997-11-19 |
| US6008008A (en) | 1999-12-28 |
| ATE210674T1 (en) | 2001-12-15 |
| CA2253506A1 (en) | 1997-11-06 |
| DE69709092T2 (en) | 2002-08-22 |
| JP4155599B2 (en) | 2008-09-24 |
| EP0900230B1 (en) | 2001-12-12 |
| PT900230E (en) | 2002-05-31 |
| DE69709092D1 (en) | 2002-01-24 |
| NO985070D0 (en) | 1998-10-30 |
| GB9609024D0 (en) | 1996-07-03 |
| ES2169859T3 (en) | 2002-07-16 |
| KR100517757B1 (en) | 2006-06-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0900230B1 (en) | Esculetin derivatives | |
| US5210022A (en) | Method test media and chromogenic compounds for identifying and differentiating general coliforms and Escherichia coli bacteria | |
| EP0961834B1 (en) | Identification of salmonella | |
| DE60224072T2 (en) | ENZYME ACTIVITY INDICATORS | |
| James et al. | Note: Cyclohexenoesculetin‐β‐D‐glucoside: a new substrate for the detection of bacterial β‐D‐glucosidase | |
| JP5751658B2 (en) | Enterohemorrhagic Escherichia coli O157, O26, O111 selective separation medium | |
| JP4638645B2 (en) | Use of a chromogenic substrate to detect the presence of enzyme activity | |
| US6130057A (en) | Method for differentiating microorganisms in a sample | |
| CN106432369B (en) | Synthetic method of the one kind based on Indophenols, the glucosides of 2- (benzothiazole -2 '-yl) phenol derivatives | |
| JP2001008679A (en) | E. coli O26 isolation medium | |
| EP1224196B1 (en) | Enzymatic substrate, synthesis method and uses | |
| JP2001128666A (en) | Chromogen indicating medium containing blood or hemin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GE GH HU IL IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK TJ TM TR TT UA UG US UZ VN YU AM AZ BY KG KZ MD RU TJ TM |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH KE LS MW SD SZ UG AT BE CH DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF |
|
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GE GH HU IL IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK TJ TM TR TT UA UG US UZ VN YU AM AZ BY KG KZ MD RU TJ TM |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH KE LS MW SD SZ UG AT BE CH DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1019980708651 Country of ref document: KR |
|
| ENP | Entry into the national phase |
Ref document number: 2253506 Country of ref document: CA Kind code of ref document: A Ref document number: 2253506 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 332606 Country of ref document: NZ Ref document number: 09184231 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1997918287 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1997918287 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 1019980708651 Country of ref document: KR |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1997918287 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1019980708651 Country of ref document: KR |