WO2001068713A2 - Polysaccharide - based superabsorbent film - Google Patents
Polysaccharide - based superabsorbent film Download PDFInfo
- Publication number
- WO2001068713A2 WO2001068713A2 PCT/EP2001/003863 EP0103863W WO0168713A2 WO 2001068713 A2 WO2001068713 A2 WO 2001068713A2 EP 0103863 W EP0103863 W EP 0103863W WO 0168713 A2 WO0168713 A2 WO 0168713A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polysaccharide
- superabsorbent
- peg
- polyalkyleneglycol
- cellulose
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B11/00—Preparation of cellulose ethers
- C08B11/20—Post-etherification treatments of chemical or physical type, e.g. mixed etherification in two steps, including purification
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/28—Polysaccharides or their derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/60—Liquid-swellable gel-forming materials, e.g. super-absorbents
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B37/00—Preparation of polysaccharides not provided for in groups C08B1/00 - C08B35/00; Derivatives thereof
Definitions
- the present invention relates to flexible superabsorbent films based on poly- saccharides such as cellulose and derivatives thereof, and to a process for producing such films.
- Superabsorbent materials for use in hygiene products which arc based on polysaccharides such as cellulose and starch, have recently become widely known in the art, for example in WO 98/27117.
- the absorbing capacity of such materials can be increased by crosslinking the polymers, e.g. by using epichlorohydrin, diglycidyl ethers, divinyl sulphone or other commonly known crosslinkers capable of reacting with hydroxyl groups, or by using carboxylated polysaccharides and crosslinkers capable of reacting with carboxyl groups, such as divalent metals.
- there is a demand for thinner absorbent products which implies that superabsorbent materials have to be found which have further increased absorbing capacity and have increased flexibility.
- WO 97/191 16 describes superabsorbent acrylic polymers which are crosslinked by polymerisation of acrylic acid in the presence of a combination of irimethylolpropane triacrylate or triallylamine, polyethyleneglycol mono(meth)acrylate monoallyl ether and polyethyleneglycol mono(meth)acrylate monoalkyl ether.
- WO 97/31971 discloses similar, foamed superabsorbent acrylic polymers which are crosslinked e.g. with trimethylolpropane triacrylate, to which internal or external plasticisers (e.g. glycerol or acrylic esters) may added to increase flexibility of the foam.
- internal or external plasticisers e.g. glycerol or acrylic esters
- the polysaccharides to be used according to the present invention are in particular ⁇ -glucans like starch, amylose and amylopectin, ⁇ -glucans like cellulose, galactomannans like guar gum (guaran) and locust bean gum, glucomannans including e.g.
- xanlhan gum fructans, (arabino)xylans and galactans, as well as derivatives such as carboxymethyl, alkyl, hydroxy- ethyl and hydroxypropyl derivatives of such polysaccharides.
- Cellulose and cellulose derivatives are preferred for practical reasons.
- Combinations of such polysaccharides, or combinations with other polymers such as polyacrylates, polyvinyl alcohol etc. can also be used.
- the chain length of the polysaccharides is important, although there is no critical minimum for the molecular weight. In general, polysaccharides having a molecular weight of more than 25,000 are preferred.
- the polysaccharides to be used according to the present invention may also be carboxymethylated or carboxyethylated, especially in the case of cellulose.
- Other carboxy- alkylated polysaccharides include the half esters obtained from cyclic anhydrides such as succinic and maleic anhydride, and addition products of maleic half esters to which sulphite has been added.
- the degree of carboxyalkylation is preferably between 0 and 1.5, in particular between 0.1 and 1.0 carboxyalkyl groups per monosaccharide unit.
- the carboxyl derivatives may be in their acid or in salt form.
- carboxylated polysaccharides such as CMC (carboxymethyl cellulose) and hydroxyalkylated polysaccharides (e.g. hydroxyethyl cellulose, HEC) are especially useful, whether as mixtures of different derivatives (e.g. HEC and CMC, or HEC and carboxymethyl starch, or HEC and methyl cellulose) or as multiply derivatised single compounds (e.g. sodium carboxymethyl-hydroxyethyl cellulose, CMHEC) [0008]
- the polyalkyleneglycols to be used as spacers may for example be polyethyleneglycol (PEG), polypropyleneglycol (PPG) and the like.
- hydrophilic or hydrophobic spacers may also be used, as long as they are flexible, i.e. contain no or only a few double bonds or cyclic structures; examples are polyalkylene (as in decamethylene diisocyanate), polyhydroxyalkylene, polyalkylene succinate, polylactide, etc, with chain lengths from about 9 to about 750 chain atoms.
- the chain length of the polyalkyleneglycols may vary from e.g. 3 units (MW about 150 Da) up to e.g. 250 (MW about 11,000). Molecular weights from about 1000 to about 8000 are preferred.
- the relative amount of polyalkyleneglycol with respect to the polysaccharide may vary from about 1/200 to about 1/1, especially from about 1/50 to about 1/1.5 (weight ratios), depending on the required thickness and the required flexibility of the product.
- Such groups may be directly attached to the polyalkyleneglycol, e.g.
- (sulphonate) esters or through alkylene or phenylene groups.
- Particularly advantageous is the coupling product of a polyalkyleneglycol with divinyl sulphone on either side of the polyglycol.
- Other terminal crosslinkers include (activated) halomethyl, activated hydroxymethyl, activated formyl, epoxy, isocyanate, and the like.
- Examples of such coupling agents are maleic anhydride, dichloroacetone, l,3-dichloro-2-propanol, dimethylolurea, dimethylol- imidazolidone, diepoxides such as bisepoxybutane or bis(glycidyl ether), epichloro- hydrin, diisocyanates, bis(2-hydroxyethyl) sulphone, formaldehyde, glyoxal.
- the weight ratio between terminal crosslinker (such as divinyl sulphone) and spacer (such as polyalkylene glycol) can be between about 1/1 to about 100/1, especially between about 1.5/1 and 30/1.
- the weight ratio between crosslinker and polysaccharide may vary from e.g. 1/1 to 1/50, especially form 1/1.5 to 1/10.
- the production of the superabsorbent films according to the invention can be divided in three steps: (1) mixing of reactants and other compounds, (2) reaction and washing stage, and (3) desiccation.
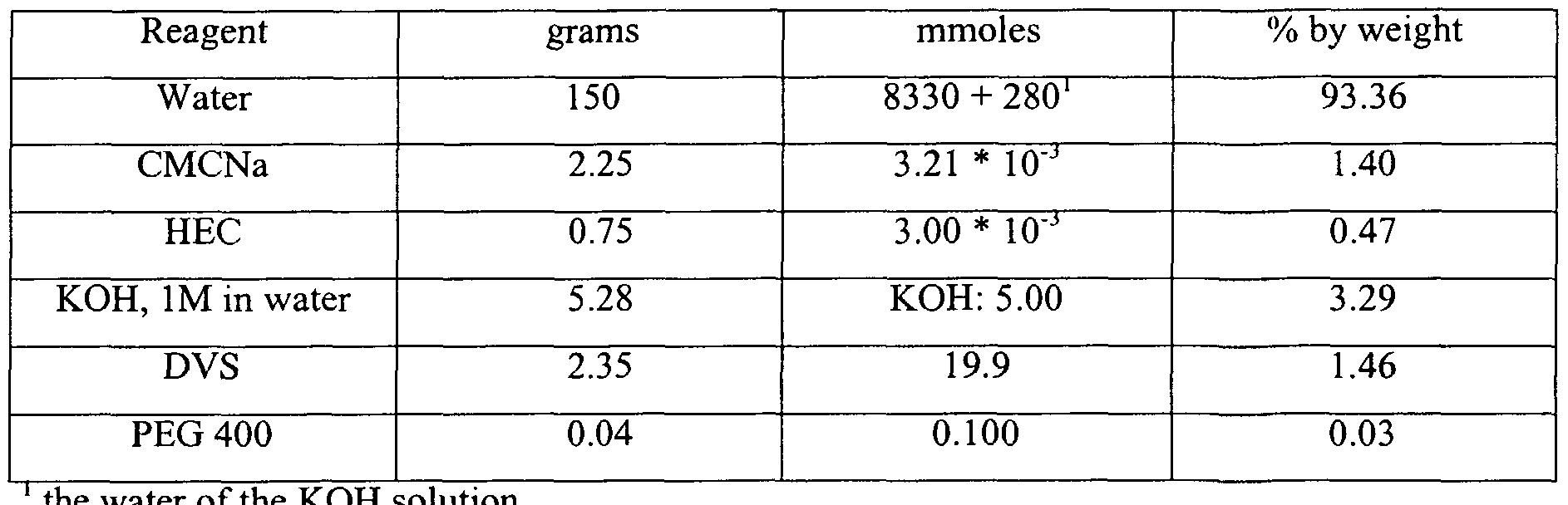
- the components involved in the reaction can be divided in different classes: (a) components of the base structure of the network, i.e. the polysaccharides, e.g. carboxymethyl cellulose sodium salt (CMCNa) and/or hydroxyethyl cellulose (HEC); (b) crosslinkers, e.g.
- CMCNa carboxymethyl cellulose sodium salt
- HEC hydroxyethyl cellulose
- step (2) the reactants are allowed to react for a sufficient time to allow the production of a crosslinked gel.
- the polyalkylene glycol and the reagent introducing the terminal double bonds are reacted first, followed by reaction with the polysaccharide, preferably in the presence of an alkaline catalyst.
- the crosslinking reaction can be performed at varying temperatures e.g. from about 5°C to about 40°C, for about 1 hour to about 2 days, preferably form 5-24 hours.
- the superabsorbent products according to the invention are flexible films with thicknesses between 10 and 500 ⁇ m and having absorption capacities between about 15 and 30 g of synthetic urine (300 mM urea, 60 mM KC1, 130 mM NaCl, 2.0 mM CaSO 4 , 3.5 mM MgSO 4 , 29 mM KH 2 PO 4 , 5.3 mM Na 2 HPO 4 , 1 mg/1 Triton X-100 in deionised water) per g of product. They can be used in absorbent articles, such as diapers, incontinence guards, sanitary napkins, and the like. They can also be used in tissue papers including kitchen towels, napkins, industrial wipes and the like. Examples
- the reaction mixture was spread on a teflon sheet with a Gardner knife in order to obtain a film with a controlled thickness.
- the film was allowed to crosslink at ambient temperature for between 5 and 24 hours (best results after 10-14 hours). Higher temperatures did not increase the crosslinking rate, and resulted in decreased viscosity. A thin, partially swollen gel film was obtained. [0014] From this point on, two different procedures were followed. According to the first procedure, the teflon sheet with the partly swollen film was then put in a jar containing distilled water. As soon as the film started to swell further, the teflon sheet was removed.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- General Health & Medical Sciences (AREA)
- Hematology (AREA)
- Veterinary Medicine (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Biochemistry (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Molecular Biology (AREA)
- Dispersion Chemistry (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Absorbent Articles And Supports Therefor (AREA)
- Laminated Bodies (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
- Other Resins Obtained By Reactions Not Involving Carbon-To-Carbon Unsaturated Bonds (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MXPA02009064A MXPA02009064A (en) | 2000-03-16 | 2001-03-09 | Polysaccharide - based superabsorbent film. |
| DE60113392T DE60113392T2 (en) | 2000-03-16 | 2001-03-09 | SUPER ABSORBENT FILM BASED ON POLYSACCHARIDES |
| AU2001268964A AU2001268964A1 (en) | 2000-03-16 | 2001-03-09 | Polysaccharide - based superabsorbent film |
| US10/221,739 US7071327B2 (en) | 2000-03-16 | 2001-03-09 | Polysaccharide-based superabsorbent film |
| AT01947216T ATE304554T1 (en) | 2000-03-16 | 2001-03-09 | SUPERABSORBENT FILM BASED ON POLYSACCHARIDES |
| JP2001567803A JP2004500469A (en) | 2000-03-16 | 2001-03-09 | Superabsorbent film based on polysaccharide |
| EP01947216A EP1268557B1 (en) | 2000-03-16 | 2001-03-09 | Polysaccharide - based superabsorbent film |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP00200965A EP1134232A1 (en) | 2000-03-16 | 2000-03-16 | Polysaccharide-based superabsorbent film |
| EP00200965.2 | 2000-03-16 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2001068713A2 true WO2001068713A2 (en) | 2001-09-20 |
| WO2001068713A3 WO2001068713A3 (en) | 2001-12-20 |
Family
ID=8171213
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2001/003863 Ceased WO2001068713A2 (en) | 2000-03-16 | 2001-03-09 | Polysaccharide - based superabsorbent film |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US7071327B2 (en) |
| EP (2) | EP1134232A1 (en) |
| JP (1) | JP2004500469A (en) |
| AT (1) | ATE304554T1 (en) |
| AU (1) | AU2001268964A1 (en) |
| DE (1) | DE60113392T2 (en) |
| DK (1) | DK1268557T3 (en) |
| MX (1) | MXPA02009064A (en) |
| PL (1) | PL357834A1 (en) |
| WO (1) | WO2001068713A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7427643B2 (en) * | 2003-03-21 | 2008-09-23 | Xylophane Aktiebolag | Polymeric film or coating comprising hemicellulose |
| EP2067793B2 (en) † | 2007-11-27 | 2014-05-21 | Innventia AB | Utilization of a wood hydrolysate |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2351253A1 (en) | 2000-11-10 | 2002-05-10 | Groupe Lysac Inc./Lysac Group Inc. | Crosslinked polysaccharide, obtained by crosslinking with substituted polyethylene glycol, as superabsorbent |
| SE0201081D0 (en) * | 2002-04-10 | 2002-04-10 | Sca Hygiene Prod Ab | Absorbent articles with improved discretion |
| ITTO20040918A1 (en) * | 2004-12-29 | 2005-03-29 | Luigi Ambrosio | POLYMERIC HYDROGEL BIODEGRADABLE ABSORBERS AND PROCEDURE FOR THEIR PREPARATION |
| WO2009022358A1 (en) * | 2007-08-10 | 2009-02-19 | Luigi Ambrosio | Superabsorbent polymer hydro gels and a method of preparing thereof |
| US9345809B2 (en) * | 2007-11-28 | 2016-05-24 | Fziomed, Inc. | Carboxymethylcellulose polyethylene glycol compositions for medical uses |
| US20130338354A1 (en) | 2010-08-30 | 2013-12-19 | Ann-Christine Albertsson | Renewable superabsorbents |
| EP2717685B1 (en) | 2011-06-07 | 2018-08-22 | Gelesis LLC | Method for producing hydrogels |
| GB2502972B (en) | 2012-06-11 | 2014-07-16 | Bioquell Uk Ltd | Aseptic processing workstation |
| MX386595B (en) | 2014-06-20 | 2025-03-19 | Gelesis Llc | METHODS FOR THE TREATMENT OF OVERWEIGHT OR OBESITY. |
| CN107250164B (en) | 2015-01-29 | 2023-01-31 | 吉莱斯公司 | Method for making hydrogels combining high elastic modulus and absorbency |
| CA3022090A1 (en) | 2016-04-25 | 2017-11-02 | Gelesis, Llc. | Method for treating constipation |
| BR112019020724A2 (en) * | 2017-04-05 | 2020-05-12 | Gelesis, Llc | IMPROVED SUPERABSORVENT MATERIALS AND PRODUCTION METHODS OF THE SAME |
| EP4467599A3 (en) * | 2017-12-14 | 2025-04-23 | Nutrition & Biosciences USA 4, Inc. | Alpha-1,3-glucan graft copolymers |
| US12128144B2 (en) | 2018-04-02 | 2024-10-29 | Polygreen Ltd | Process for the production of biodegradable superabsorbent polymer with high absorbency under load based on styrene maleic acid copolymers and biopolymer |
| EP3773388A4 (en) | 2018-04-02 | 2022-01-19 | Polygreen Ltd. | LIQUID POLYMER SOLUTION FOR THE TREATMENT OF NON-WOVEN STRIPS |
| WO2020102227A1 (en) | 2018-11-13 | 2020-05-22 | Green Polymers Ltd. | Polymeric composition for use as soil conditioner with improved water absorbency during watering of the agricultural crops |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5414135A (en) * | 1991-12-30 | 1995-05-09 | Sterling Winthrop Inc. | Vinyl sulfone coupling of polyoxyalkylenes to proteins |
| US5573994A (en) * | 1994-05-13 | 1996-11-12 | University Of Cincinnati | Superabsorbent foams, and method for producing the same |
| DE19543368C2 (en) * | 1995-11-21 | 1998-11-26 | Stockhausen Chem Fab Gmbh | Water-absorbing polymers with improved properties, processes for their production and their use |
-
2000
- 2000-03-16 EP EP00200965A patent/EP1134232A1/en not_active Withdrawn
-
2001
- 2001-03-09 WO PCT/EP2001/003863 patent/WO2001068713A2/en not_active Ceased
- 2001-03-09 DE DE60113392T patent/DE60113392T2/en not_active Expired - Lifetime
- 2001-03-09 MX MXPA02009064A patent/MXPA02009064A/en active IP Right Grant
- 2001-03-09 JP JP2001567803A patent/JP2004500469A/en active Pending
- 2001-03-09 EP EP01947216A patent/EP1268557B1/en not_active Expired - Lifetime
- 2001-03-09 DK DK01947216T patent/DK1268557T3/en active
- 2001-03-09 AU AU2001268964A patent/AU2001268964A1/en not_active Abandoned
- 2001-03-09 US US10/221,739 patent/US7071327B2/en not_active Expired - Lifetime
- 2001-03-09 AT AT01947216T patent/ATE304554T1/en not_active IP Right Cessation
- 2001-03-09 PL PL01357834A patent/PL357834A1/en not_active Application Discontinuation
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7427643B2 (en) * | 2003-03-21 | 2008-09-23 | Xylophane Aktiebolag | Polymeric film or coating comprising hemicellulose |
| AU2004221959B2 (en) * | 2003-03-21 | 2009-03-26 | Seelution Ab | Polymeric film or coating comprising hemicellulose |
| EP2067793B2 (en) † | 2007-11-27 | 2014-05-21 | Innventia AB | Utilization of a wood hydrolysate |
Also Published As
| Publication number | Publication date |
|---|---|
| US7071327B2 (en) | 2006-07-04 |
| DK1268557T3 (en) | 2005-11-28 |
| DE60113392T2 (en) | 2006-04-27 |
| EP1134232A1 (en) | 2001-09-19 |
| US20030149263A1 (en) | 2003-08-07 |
| PL357834A1 (en) | 2004-07-26 |
| AU2001268964A1 (en) | 2001-09-24 |
| MXPA02009064A (en) | 2004-08-12 |
| DE60113392D1 (en) | 2005-10-20 |
| JP2004500469A (en) | 2004-01-08 |
| WO2001068713A3 (en) | 2001-12-20 |
| EP1268557B1 (en) | 2005-09-14 |
| ATE304554T1 (en) | 2005-09-15 |
| EP1268557A2 (en) | 2003-01-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1268557B1 (en) | Polysaccharide - based superabsorbent film | |
| US5780616A (en) | Cationic polymer | |
| EP0946598B1 (en) | Superabsorbent material and method for producing said material | |
| EP0815879B1 (en) | Bioabsorbable medical devices from oxidized polysaccharides | |
| EP3609507B1 (en) | Variable-size hydrophobically-modified polymers | |
| US7737214B2 (en) | Adhesion preventive material | |
| CN111440310A (en) | Polyethylene glycol derivative, preparation method thereof and polyethylene glycol hydrogel capable of rapidly generating crosslinking reaction | |
| CA2351253A1 (en) | Crosslinked polysaccharide, obtained by crosslinking with substituted polyethylene glycol, as superabsorbent | |
| JP2001520281A (en) | Substantially fiber-free cellulose ethers exhibiting improved water retention, methods of making and using the same | |
| KR102584470B1 (en) | Biodegradable superabsorbent polymer having the excellent absorption under load performance and method for preparing the same | |
| US9434787B2 (en) | Cellulose ethers having reactive anchor group, modified cellulose ethers obtainable therefrom and methods for the preparation thereof | |
| EP1119378A1 (en) | Polysaccharide based absorbent polymer material | |
| Hon | Chemical modification of cellulose | |
| CZ232493A3 (en) | Fibrous polysaccharides of cationic nature | |
| Fei et al. | High-toughness and biodegradable superabsorbent hydrogels based on dual functional crosslinkers | |
| JP4074905B2 (en) | Water-absorbing material, production method thereof and water-absorbing article | |
| CA2205026C (en) | Cationic polymer | |
| KR101856656B1 (en) | Preparation method of textile material of cellulose with high water absorption and superior morphostasis | |
| US5739304A (en) | Process for the preparation of water-soluble aminoalkyl derivatives of polysaccharides | |
| WO1999029352A1 (en) | Superabsorbent material and method for producing said material | |
| US20250032666A1 (en) | Hemostatic Materials Based on Aldehyde-Functional Polysaccharides | |
| JPS6129375B2 (en) | ||
| NO763135L (en) | ||
| JP2018535285A (en) | Use of cellulose ethers having 3-azido-hydroxyalkyl groups in water-insoluble adhesives | |
| JPH09124701A (en) | Polysaccharide derivative |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2001947216 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref country code: JP Ref document number: 2001 567803 Kind code of ref document: A Format of ref document f/p: F |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2002/009064 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10221739 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 2001947216 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2001947216 Country of ref document: EP |