WO2001091748A2 - Methods for prevention of ulcers and improving physiological performance - Google Patents
Methods for prevention of ulcers and improving physiological performance Download PDFInfo
- Publication number
- WO2001091748A2 WO2001091748A2 PCT/EP2001/005788 EP0105788W WO0191748A2 WO 2001091748 A2 WO2001091748 A2 WO 2001091748A2 EP 0105788 W EP0105788 W EP 0105788W WO 0191748 A2 WO0191748 A2 WO 0191748A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- mammal
- proton pump
- pump inhibitor
- ulcers
- formulation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- GUXJXWKCUUWCLX-UHFFFAOYSA-N CC1=NCCO1 Chemical compound CC1=NCCO1 GUXJXWKCUUWCLX-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/4184—1,3-Diazoles condensed with carbocyclic rings, e.g. benzimidazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/4709—Non-condensed quinolines and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
Definitions
- the present invention provides a method for the prevention of ulcers in animals, including horses, dogs and humans.
- this paste is to be administered orally once a day for 4 weeks at the dosage of 1.8 mg omeprazole/lb body weight (4 mg/kg). Thereafter, recurrence of gastric ulcers can be prevented if the treatment is continued for at least an additional 4 weeks at a lower dose of 0.9 mg/lb (2 mg/kg). ; ⁇ '
- PPI Proton pump inhibitors
- the PPI omeprazole is disclosed in US patent No. 4 255 432.
- Pharmaceutical compositions containing proton pump inhibitors are also disclosed in the PCT Patent Application WO 96/31213 and US Patent No. 5708017 which discloses a stable, ready-to-use oral paste composition of proton pump inhibitor, such as, for example, omeprazole. , -
- the present invention provides a method for the prevention of gastric ulcers, such as gastrointestinal ulcers in a mammal, e.g. a domestic mammal, a farm mammal, a companion mammal, a game or sport mammal, such as horses, dogs, humans, and other mammals that may be susceptible to such ulcers.
- the method comprises administering e.g., periodically, an effective amount of a proton pump inhibitor to the mammal.
- the administration can be prior to or during a stressful event. It can also be a single treatment or administration over one or two days. This administering can be of amounts used in animals for the treatment of ulcers, although lower or higher doses can also be employed.
- This prevention of gastric ulcers is in mammals prior to the occurrence of a gastric ulcer condition and is in contrast to treatment or prevention of reoccurrence in an animal that has already had a gastric ulcer condition.
- the invention also provides the use of a proton pump inhibitor, preferably omeprazole, for the preparation of a formulation for the prevention of gastric ulcers in mammals, and/or improving physiological responses in a mammal.
- a proton pump inhibitor preferably omeprazole
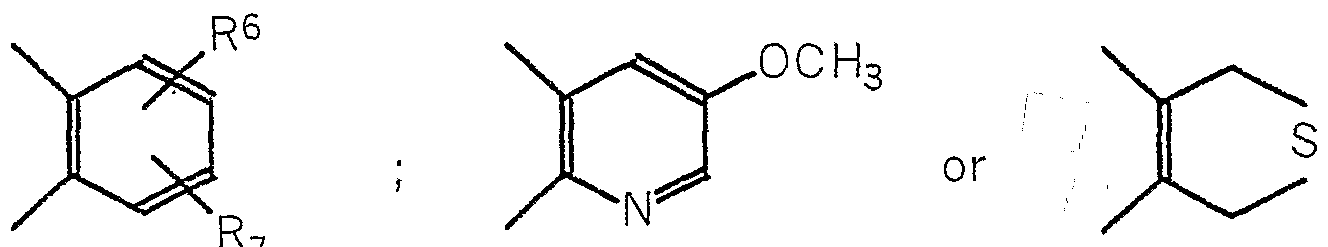
- the proton pump inhibitors used in the present invention can include compounds of the general formula :
- R 1 and R 3 are independently selected from hydrogen, lower alkyl, lower alkoxy and halogen
- R 2 is selected from hydrogen, lower alkyl, lower alkoxy-lower alkoxy, lower fluoralkoxy and
- R 4 and R 5 are independently selected from lower alkyl, A is
- R 6 and R 7 are independently selrected from h lower alkyl, lower alkoxy, lower fluoroalkoxy, lower fluoroalkyl, halogen,
- R 8 is lower alkyl or lower alkoxy.
- PPI examples include esomeprazole (nee: perprazole), rabeprazole, and IY-81 149 (distributed by Axican Pharma).
- the preferred proton pump inhibitor used in the present invention is the compound known as omeprazole.
- omeprazole is disclosed in EP 5129, lansoprazole in EP 174.716, pantoprazole in EP 166,287, leminoprazole in GB
- the preferred compound for horses and dogs is omeprazole.
- Omeprazole is also preferred for humans.
- the periodic treatment is preferably a daily treatment. However, a single treatment or administration of a PPI or over one or two days (e.g., once daily for one or two days), such as prior to or during a stressful event, is also envisioned.
- the effective amount of proton pump inhibitor is preferably of 0.1 to 8 mg per kilogram body weight. It is preferred to administer relatively low doses, preferably equal or more preferably, less than the usual doses for the treatment of ulcers in the animal.
- the dose is about 50% of the usual dose for the treatment in the animal.
- the compound is administered daily but this frequency can be lowered, e.g. to every other day or once every three days or once weekly, for compounds which persist for a long time in the organism.
- the treatment can include administration of the compound in the form of a formulation for controlled release and long lasting delivery, in which case the administration can be less frequent , for example, a weekly or a monthly administration.
- the duration of the treatment for preventing ulcers is preferably at least several days.
- the treatment for prevention is a continuous treatment, either for life or at least during the whole period where the animal is or is suspected to be under stress conditions or other conditions which may increase the risk of formation of ulcers.
- the treatment is conducted on animals having substantial risk of developing gastrointestinal ulcers.
- the treatment is more preferably administered during periods of stress, training, transportation, change in environment (weather changes, housing changes) or pregnancy.
- the inhibitor can be administered under any suitable formulation for delivery, preferably for oral delivery.
- Suitable oral formulations include oral solutions, oral suspensions, feed premix, pastes, gels, granules, tablets, capsules or boli.
- the proton pump inhibitor is provided for horses as a pharmaceutical composition for oral administration comprising a proton pump inhibitor, a thickening agent , a basifying agent, and a hydrophobic, oily liquid vehicle.
- these formulations make a paste for horses.
- Such formulations are disclosed in PCT Patent WO 96/31213 and US Patent N°5 708 017 which are hereby incorporated by reference.
- oral formulations can be made in the form of enteric dry particles mixed with a dry gelling agent.
- the following formulations are preferred for humans: oral solutions or suspensions, gels, tablets, capsules or powder.
- this method improves physiological responses in mammals such as horses, e.g. oxygen consumption and time to fatigue, and thus, the present invention envisions methods for improving oxygen consumption and/or time to fatigue comprising administering to a mammal a PPI as herein discussed, e.g., for prevention of ulcers (e.g. such as administration of PPI on continuous basis).
- a proton pump inhibitor preferably omeprazole
- omeprazole for the preparation of a formulation for the prevention of gastric ulcers in mammals, and/or for improving physiological responses in a mammal.
- the following exemplifies a comparative method showing the efficiency of a formulation according to the invention for the prevention of gastric ulcers in horses.
- Example 1 Preparation of an omeprazole-containing oral paste for horses.
- the paste for horses, containing 37% w/w omeprazole can be prepared according to US Patent N° 5 708 017. This paste is contained in adjustable-dose syringes for oral delivery. In this example, the paste is the paste for horses sold in the United States under the trademark GASTROGARD.
- Example 2 Feeding, environmental and training regime for race horses .
- Horses were acclimatised for 2 weeks to the treadmill, following which they entered a standardised ascending training program for a 10 weeks. Ulcers were be induced in half of the horses during the second week by alternately withholding feed on alternate days, In brief, food, but not water, was withheld for periods of approximately 24 hours on days 14, 16, 18, 20 and 22. In horses in which ulceration was induced, bedding was removed from the stalls during periods when feed was withheld.
- Alfalfa hay and grass hay were offered ad libitum. Ten kilograms oats was feed at 4 pm daily once the horses had entered their ascending exercise program.
- the horses entered a standardised exercise protocol.
- the horses were initially walked (2m/s) for 4 minutes, then trotted (4m/s) for 3.5 minutes, after which they were galloped for a further 3 minutes at a speed such that they achieved a heart rate of 80% of their maximum heart rate (generally 10 to 11 m/s).
- the heart rate was monitored using telemetry equipment attached to their girths. Initially horses were run suing this protocol 3 times per week, and were run at a speed producing 50% of their heart rate max on the remaining 2 days. However, there was a reduction in the severity of the ulceration using this regime, and consequently horses were run at a speed producing 80%o of their heart rate max for 5 days per week.
- Moderate ulceration extensive superficial appearing lesions of deeper focal lesions with or without some mucosal proliferation along lesion margins and small amount of bleeding.
- Vo 2 and Vco 2 were determined from standard equations.
- the horses performed a standardised test consisting of walking at 2 m/s for 4 minutes, then trotting at 4 m/s for 3.5 minutes, and then galloping at approximately 15 m/s until "exhaustion " (defined as when the horse could no longer maintain the speed of the treadmill).
- Heart rate was determined by counting the number of QRS complexes on a 15-second trace.
- Blood lactate values was measured in blood taken from the 14-gauge jugular cartheter and placed in fluoride-oxalate tubes. The Vo 2 and Vco 2 and HR measurements are taken when they had reached a steady state.
- Ulcers were induced in all horses subjected to the alternate withholding of feed. Ulcers were maintained in the horses within the ulcer group, although during the period when horses were running at a lower intensity (ie at 50% heart rate max two times per week). However, when the
- lactate concentrations measured were consistent with the horses reaching their anaerobic threshold. Heart rate was not significantly altered in either group when the initial evaluation was compared to the final evaluation. There were no differences in the weight of the horses between groups.
- Horses submitted to the training protocol according to example 2 were divided into two groups of 14 horses each group. They were then handled and identically trained for 56 days in the trial according to example 2 to induce gastric ulcers.
- Group 1 received the horse paste formulation of example 1 at 4 mg/kg omeprazole while Group 2 received no medication. Within two weeks all of the non-treated horses developed moderate to severe ulcers that remained or worsened during the trial.
- horses in the omeprazole-treated group demonstrated physiological responses that were significantly better compared to the non-treated group. Oxygen consumption was substantially increased, as well as time to exhaustion and stride length.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Fodder In General (AREA)
- Medicines Containing Plant Substances (AREA)
Abstract
Description
Claims
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE60142515T DE60142515D1 (en) | 2000-05-30 | 2001-05-18 | COMPOSITIONS FOR PREVENTING GRAVES IN HORSES |
| EP01945171A EP1286669B1 (en) | 2000-05-30 | 2001-05-18 | Compositions for prevention of ulcers in horses |
| CA2408920A CA2408920C (en) | 2000-05-30 | 2001-05-18 | Methods for prevention of ulcers and improving physiological performance |
| NZ522450A NZ522450A (en) | 2000-05-30 | 2001-05-18 | Methods for prevention of ulcers |
| MXPA02011608A MXPA02011608A (en) | 2000-05-30 | 2001-05-18 | Methods for prevention of ulcers and improving physiological performance. |
| AU2001267465A AU2001267465B2 (en) | 2000-05-30 | 2001-05-18 | Methods for prevention of ulcers and improving physiological performance |
| AU6746501A AU6746501A (en) | 2000-05-30 | 2001-05-18 | Methods for prevention of ulcers and improving physiological performance |
| AT01945171T ATE473002T1 (en) | 2000-05-30 | 2001-05-18 | COMPOSITIONS FOR PREVENTING ULCERS IN HORSES |
| DK01945171.5T DK1286669T3 (en) | 2000-05-30 | 2001-05-18 | Preparations for the prevention of ulcers in horses |
| BR0111215-5A BR0111215A (en) | 2000-05-30 | 2001-05-18 | Methods for ulcer prevention and improvement of physiological performance |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US20787800P | 2000-05-30 | 2000-05-30 | |
| US60/207,878 | 2000-05-30 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2001091748A2 true WO2001091748A2 (en) | 2001-12-06 |
| WO2001091748A3 WO2001091748A3 (en) | 2002-04-18 |
Family
ID=22772351
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2001/005788 Ceased WO2001091748A2 (en) | 2000-05-30 | 2001-05-18 | Methods for prevention of ulcers and improving physiological performance |
Country Status (16)
| Country | Link |
|---|---|
| US (2) | US6939881B2 (en) |
| EP (1) | EP1286669B1 (en) |
| AR (1) | AR028641A1 (en) |
| AT (1) | ATE473002T1 (en) |
| AU (2) | AU2001267465B2 (en) |
| BR (1) | BR0111215A (en) |
| CA (1) | CA2408920C (en) |
| CY (1) | CY1110797T1 (en) |
| DE (1) | DE60142515D1 (en) |
| DK (1) | DK1286669T3 (en) |
| ES (1) | ES2345873T3 (en) |
| MX (1) | MXPA02011608A (en) |
| NZ (1) | NZ522450A (en) |
| PT (1) | PT1286669E (en) |
| WO (1) | WO2001091748A2 (en) |
| ZA (1) | ZA200209632B (en) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MXPA02011608A (en) * | 2000-05-30 | 2003-10-06 | Merial Ltd | Methods for prevention of ulcers and improving physiological performance. |
| US9387386B2 (en) * | 2003-07-31 | 2016-07-12 | First Principles, Inc. | Method and apparatus for improving performance |
| US20090197170A1 (en) * | 2008-01-31 | 2009-08-06 | Viavattine Joseph J | Maximization of active material to collector interfacial area |
| WO2014076092A1 (en) * | 2012-11-14 | 2014-05-22 | Boehringer Ingelheim Vetmedica Gmbh | A proton pump inhibitor for use in a method of treating dermatological diseases in canine |
| CA3033065A1 (en) * | 2016-08-11 | 2018-02-15 | Adamis Pharmaceuticals Corporation | Drug compositions |
| US11564910B2 (en) | 2017-12-08 | 2023-01-31 | Adamis Pharmaceuticals Corporation | Drug compositions |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4073929A (en) * | 1976-02-13 | 1978-02-14 | Merck & Co., Inc. | 3-(2-Substitutedbenzimidazolyl) alanines |
| US4743609A (en) * | 1985-02-12 | 1988-05-10 | Banyu Pharmaceutical Co., Ltd. | Indole derivatives having gastric and antisecretory and cytoprotective properties, and pharmaceutical preparations containing same |
| US5708017A (en) * | 1995-04-04 | 1998-01-13 | Merck & Co., Inc. | Stable, ready-to-use pharmaceutical paste composition containing proton pump inhibitors |
| EA004683B1 (en) * | 1999-02-23 | 2004-06-24 | Мерк Энд Ко., Инк. | Pharmaceutical composition containing omeprazole |
| MXPA02011608A (en) * | 2000-05-30 | 2003-10-06 | Merial Ltd | Methods for prevention of ulcers and improving physiological performance. |
-
2001
- 2001-05-18 MX MXPA02011608A patent/MXPA02011608A/en active IP Right Grant
- 2001-05-18 DK DK01945171.5T patent/DK1286669T3/en active
- 2001-05-18 WO PCT/EP2001/005788 patent/WO2001091748A2/en not_active Ceased
- 2001-05-18 DE DE60142515T patent/DE60142515D1/en not_active Expired - Lifetime
- 2001-05-18 AU AU2001267465A patent/AU2001267465B2/en not_active Expired
- 2001-05-18 EP EP01945171A patent/EP1286669B1/en not_active Expired - Lifetime
- 2001-05-18 NZ NZ522450A patent/NZ522450A/en not_active IP Right Cessation
- 2001-05-18 ES ES01945171T patent/ES2345873T3/en not_active Expired - Lifetime
- 2001-05-18 AT AT01945171T patent/ATE473002T1/en active
- 2001-05-18 PT PT01945171T patent/PT1286669E/en unknown
- 2001-05-18 CA CA2408920A patent/CA2408920C/en not_active Expired - Lifetime
- 2001-05-18 BR BR0111215-5A patent/BR0111215A/en not_active Application Discontinuation
- 2001-05-18 AU AU6746501A patent/AU6746501A/en active Pending
- 2001-05-29 US US09/867,285 patent/US6939881B2/en not_active Expired - Lifetime
- 2001-05-29 AR ARP010102548A patent/AR028641A1/en unknown
-
2002
- 2002-11-27 ZA ZA200209632A patent/ZA200209632B/en unknown
-
2005
- 2005-04-19 US US11/108,974 patent/US20050187216A1/en not_active Abandoned
-
2010
- 2010-09-16 CY CY20101100845T patent/CY1110797T1/en unknown
Non-Patent Citations (1)
| Title |
|---|
| None |
Also Published As
| Publication number | Publication date |
|---|---|
| CY1110797T1 (en) | 2015-06-10 |
| EP1286669A2 (en) | 2003-03-05 |
| US20050187216A1 (en) | 2005-08-25 |
| US20020065306A1 (en) | 2002-05-30 |
| US6939881B2 (en) | 2005-09-06 |
| AU6746501A (en) | 2001-12-11 |
| ZA200209632B (en) | 2004-04-23 |
| BR0111215A (en) | 2003-12-16 |
| EP1286669B1 (en) | 2010-07-07 |
| PT1286669E (en) | 2010-09-20 |
| NZ522450A (en) | 2004-07-30 |
| CA2408920C (en) | 2011-07-19 |
| DK1286669T3 (en) | 2010-08-09 |
| CA2408920A1 (en) | 2001-12-06 |
| AU2001267465B2 (en) | 2006-02-02 |
| WO2001091748A3 (en) | 2002-04-18 |
| ATE473002T1 (en) | 2010-07-15 |
| AR028641A1 (en) | 2003-05-21 |
| ES2345873T3 (en) | 2010-10-05 |
| MXPA02011608A (en) | 2003-10-06 |
| DE60142515D1 (en) | 2010-08-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4603631B2 (en) | Pharmaceutical composition containing a proton pump inhibitor | |
| Buchanan et al. | Treatment and prevention of equine gastric ulcer syndrome | |
| Videla et al. | New perspectives in equine gastric ulcer syndrome | |
| Vatistas et al. | Induction and maintenance of gastric ulceration in horses in simulated race training | |
| MacAllister et al. | Effects of omeprazole paste on healing of spontaneous gastric ulcers in horses and foals: a field trial | |
| BG107038A (en) | GENERAL PARASITIZED COMPOSITION | |
| EP1286669B1 (en) | Compositions for prevention of ulcers in horses | |
| AU2001267465A1 (en) | Methods for prevention of ulcers and improving physiological performance | |
| CN101426490B (en) | Compositions comprising alpha-ketoglutarate and their use for modulating muscle performance | |
| AU608672B2 (en) | Method for treating hoof thrush and hoof rot | |
| AU773696B2 (en) | Animal stereotypy | |
| JP2001526217A (en) | Novel use of local anesthetics for vascular headache | |
| WO2006026829A1 (en) | Stable paste composition of enteric coated acid labile active agent and use thereof | |
| Ralphs et al. | Ability of apomorphine and lithium chloride to create food aversions in cattle | |
| AU2023203385A1 (en) | Omeprazole based oral paste formulation having increased temperature stability and enhanced absorption | |
| JP2008535901A (en) | Treatment of human and animal parasitic infections | |
| FR2461495A1 (en) | Oblate, anthelmintic, prolonged release tablet for ruminants - releases active cpd. over 5-7 days and controls helminths in grazing animals without danger of toxicity | |
| AU2005100669A4 (en) | Enteric coated paste compositions and uses thereof | |
| WO2005000269A1 (en) | Pharmaceutical formulations comprising a proton pump inhibitor | |
| CN109793838A (en) | A kind of plaster preparation for preventing and treating Tibetan antelope stomatitis and preparation method thereof | |
| GB1583802A (en) | Use of moxnidazole for combating diseases in the veterinary field | |
| WO2001051064A1 (en) | Ph-regulators against oral stereotypies in equines | |
| JPH07103036B2 (en) | Method for preventing and treating nodular disease of fish | |
| HK1004658B (en) | Pharmaceutical composition containing proton pump inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2001945171 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 522450 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2408920 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2001267465 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2002/011608 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200209632 Country of ref document: ZA |
|
| WWP | Wipo information: published in national office |
Ref document number: 2001945171 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 522450 Country of ref document: NZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 522450 Country of ref document: NZ |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |