WO2002102889A1 - Rubber composition, crosslinkable rubber composition and crosslinked articles - Google Patents
Rubber composition, crosslinkable rubber composition and crosslinked articles Download PDFInfo
- Publication number
- WO2002102889A1 WO2002102889A1 PCT/JP2002/005777 JP0205777W WO02102889A1 WO 2002102889 A1 WO2002102889 A1 WO 2002102889A1 JP 0205777 W JP0205777 W JP 0205777W WO 02102889 A1 WO02102889 A1 WO 02102889A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- rubber
- gen
- rubber composition
- based polymer
- block
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/18—Homopolymers or copolymers of hydrocarbons having four or more carbon atoms
- C08L23/20—Homopolymers or copolymers of hydrocarbons having four or more carbon atoms having four to nine carbon atoms
- C08L23/22—Copolymers of isobutene; Butyl rubber; Homopolymers or copolymers of other iso-olefins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L9/00—Compositions of homopolymers or copolymers of conjugated diene hydrocarbons
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08C—TREATMENT OR CHEMICAL MODIFICATION OF RUBBERS
- C08C1/00—Treatment of rubber latex
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F297/00—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer
- C08F297/02—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L21/00—Compositions of unspecified rubbers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/16—Ethylene-propylene or ethylene-propylene-diene copolymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/26—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers modified by chemical after-treatment
- C08L23/28—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers modified by chemical after-treatment by reaction with halogens or halogen-containing compounds
- C08L23/283—Iso-olefin halogenated homopolymers or copolymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L53/005—Modified block copolymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L7/00—Compositions of natural rubber
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2312/00—Crosslinking
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L53/02—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers of vinyl-aromatic monomers and conjugated dienes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L53/02—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers of vinyl-aromatic monomers and conjugated dienes
- C08L53/025—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers of vinyl-aromatic monomers and conjugated dienes modified
Definitions
- the present invention relates to a novel rubber composition, a crosslinkable rubber composition, and a crosslinked product obtained by crosslinking the same, which can be suitably used for tire applications, industrial member applications, and the like.
- nitrile rubber Z polyvinyl chloride is known to have improved tensile strength, oil resistance, etc. as compared to nitrile rubber alone, and a polybutadiene rubber Z syndiotactic 1, It is known that a blend of 2-butadiene rubber has improved tear strength and crack growth resistance as compared to a polybutadiene rubber alone (for example, The Japan Rubber Association, Vol. 72, pp. 593-598 (1) 999) See).
- gen-based rubbers such as natural rubber (NR), polyisoprene rubber (IR), polybutene rubber (BR), and styrene-butadiene copolymer rubber (SBR) have many primary carbons in their primary structures.
- NR natural rubber
- IR polyisoprene rubber
- BR polybutene rubber
- SBR styrene-butadiene copolymer rubber
- the rubber composition obtained has a problem in that mechanical properties such as flexural properties and tensile properties are impaired as a result of insufficient adhesion at the interface of the mixed rubber phase in the obtained rubber composition.
- an object of the present invention is to provide a rubber composition, a crosslinkable rubber composition and a rubber composition which are excellent in tensile properties and can improve bending properties when different types of rubbers of a gen-based rubber and a highly saturated rubber are mixed.
- An object of the present invention is to provide a cross-linked product obtained by cross-linking
- the present inventors have conducted intensive studies to achieve the above object, and found that when a rubber composition is produced by kneading a gen-based rubber and a highly saturated rubber, a gen-based polymer block and a hydrogenated gen-based polymer block are used.
- a block copolymer having a specific primary structure By adding a block copolymer having a specific primary structure, the miscibility, dispersibility, and interfacial adhesion of the gen-based rubber and the highly saturated rubber are improved, and excellent tensile properties, A rubber composition having physical properties such as bending performance can be obtained.
- a cross-linkable rubber composition obtained by further adding a cross-linking agent to the rubber composition and a cross-linked product obtained by cross-linking the rubber composition have the same physical properties.
- the inventors have found that the present invention has been completed. Disclosure of the invention
- a rubber composition comprising a block copolymer having a primary structure of any one of the above, wherein the block copolymer has a total mass of 100 parts by mass of the gen-based rubber (1) and the highly saturated rubber (2).
- a rubber composition comprising 0.1 to 25 parts by mass of the polymer (3);
- the term “gen-based rubber” means a polymer having a rubber-like property and mainly comprising a conjugated diene compound such as butadiene and isoprene.
- the gen-based rubber (1) include natural rubber (NR), polyisoprene rubber (IR), polybutadiene rubber (BR), and styrene-butadiene copolymer rubber (SBR).
- “highly saturated rubber” is mainly composed of olefin units represented by ethylene, propylene, and isobutylene units, and has an olefinic carbon-carbon double bond (unsaturated bond) in its skeleton. ) Means a polymer having a rubber-like property in which the unit having is 20% by mass or less.
- Examples of the highly saturated rubber (2) include ethylene-propylene copolymer rubber (EPR), ethylene-propylene-copolymer rubber (EPDM), butyl rubber (IIR), chlorinated butyl rubber, and brominated butyl rubber.
- the block copolymer (3) used in the present invention is composed of a gen-based polymer block A and a hydrogenated gen-based polymer block B, and
- X represents an integer of 1 or more.
- Lock A has excellent compatibility with the gen-based rubber (1) and is mainly composed of conjugated gen compounds such as butadiene and isoprene, and other aromatic compounds such as vinyl aromatic compounds such as styrene. It is a polymer block which may contain a polymerizable compound.
- the gen-based polymer block A include polyisoprene, polybutadiene, butadiene-isoprene random copolymer, styrene-butadiene random copolymer, and styrene-isoprene random copolymer.
- the hydrogenated gen-based polymer block B has excellent compatibility with the highly saturated rubber (2), and is mainly composed of a conjugated gen compound such as butadiene and isoprene, and is composed of vinyl such as styrene.
- This is a polymer block obtained by hydrogenating a polymer block which may contain another polymerizable compound such as an aromatic compound.
- the hydrogenated diene polymer block B include hydrogenated polybutadiene, hydrogenated styrene-butadiene random copolymer, hydrogenated butadiene-isoprene random copolymer, hydrogenated polyisoprene, hydrogenated polybutadiene, and the like. be able to.
- Types of the gen-based polymer block A and the hydrogenated gen-based polymer block B constituting the block copolymer (3), the styrene content when each block contains styrene, and the bonding mode of the conjugated gen unit (1,4-one bond unit, 1,2-bond unit, 3,4-bond unit) and their abundance ratio depend on the type of gen-based rubber (1) and highly saturated rubber (2) used.
- a suitable block can be appropriately selected from the viewpoint of compatibility.
- polyisoprene is used as the gen-based polymer block A of the block copolymer (3), preferably 1 of the total isoprene units.
- 4 Polyisoprene with an amount of one bond unit in the range of 50% or more and 99.5% or less, and styrene-butene diene copolymer rubber (SBR) as the gen-based rubber (1)
- SBR styrene-butene diene copolymer rubber
- a styrene-butadiene random copolymer is used as the gen-based polymer block A of the block copolymer (3). It can be suitably used.
- the hydrogenated polymer block B of the block copolymer (3) is used.
- Hydrogenated polybutadiene preferably hydrogenated polybutadiene obtained by hydrogenating polybutadiene having an amount of 1,4-bond units in all butadiene units in the range of 10% or more and 90% or less. Can be.
- the olefinic carbon-carbon double bond (based on the gen unit in the hydrogenated gen-based polymer block B of the block copolymer (3))
- the unit having an unsaturated bond is preferably 20% or less based on all conjugated gen units constituting the hydrogenated gen-based polymer block B.
- the olefinic carbon-carbon double bond based on the conjugated gen unit in the gen-based polymer block A of the block copolymer (3) is used.
- the unit having (unsaturated bond) is preferably at least 50 mol%, more preferably at least 70 mol%, based on all conjugated gen units constituting the gen-based polymer block A. .
- the number average molecular weight (Mn) of the block copolymer (3) used in the present invention is not particularly limited, but is usually preferably in the range of 10,000 to 100,000, and more preferably in the range of 20,000 to 500,000. preferable.
- the value of X in the formula representing the primary structure of the block copolymer (3) is an integer of 1 or more.
- the value of X is appropriately set according to the number average molecular weight (Mn) of the gen-based polymer block A and the hydrogenated gen-based polymer block B so as to satisfy the preferable number-average molecular weight (Mn) described above. It is usually in the range of 1 to 10, preferably in the range of 1 to 5, and more preferably in the range of 1 to 3.
- the block copolymer (3) used in the present invention has a molecular weight distribution represented by a ratio of weight average molecular weight (Mw) to Z number average molecular weight (Mn) in the range of 1.0 to 1.5. It is preferred that The number-average molecular weight and weight-average molecular weight used here are measured in terms of polystyrene by gel permeation chromatography (GPC). Are the number average molecular weight and weight average molecular weight.
- the block copolymer (3) used in the present invention has a functional group such as a hydroxyl group, a carboxyl group, an amino group, or an epoxy group at the main chain terminal or a side chain within a range that does not impair the effects of the present invention. You may have. Furthermore, the block copolymer (3) used in the present invention has a partial structure derived from a coupling agent such as 1,2-dibromoethane, ge- ine tetrachloride, tin tetrachloride in the molecular main chain. Is also good.
- a coupling agent such as 1,2-dibromoethane, ge- ine tetrachloride, tin tetrachloride in the molecular main chain. Is also good.
- the method for synthesizing the block copolymer (3) is not particularly limited. For example, after separately synthesizing a gen-based polymer block A having a functional group at the terminal end and a hydrogenated gen-based polymer block B, It can be produced by performing a coupling reaction between such functional groups. For example, a gen-based polymer block A having a hydroxyl group at a terminal is subjected to anion polymerization of a conjugated gen compound (and, if necessary, a vinyl aromatic compound such as styrene) by a conventionally known method.
- a hydrogenated gen-based polymer block B having a terminal hydroxyl group is converted to a conjugated gen compound (and, if necessary, a pinyl aromatic compound such as styrene) by anion polymerization using a conventionally known method. Then, when the anion polymerization is stopped, ethylene oxide is added, and then the obtained polymer is synthesized by hydrogenation, followed by coupling reaction of both with diisocyanate.
- a gen-based polymer block which is a precursor of the gen-based polymer block A and a hydrogenated gen-based polymer block B, is formed by successive anion polymerization, followed by a hydrogenation (hydrogenation) reaction. Can also be produced.
- the polymerization temperature may be in the range of 100 to 110, under an inert gas such as dry argon or nitrogen.
- the polymerization time is within the range of 0.01 to 200 hours.
- the polymerization initiator is an alkali metal such as sodium metal or lithium metal; an organic alkali metal such as methyl lithium, ethyl lithium, n-butyl lithium, or s-butyl lithium.
- a gen-based polymer block that is a precursor of a gen-based polymer block A and a hydrogenated gen-based polymer block B is formed, respectively.
- a conjugated diene compound (and, if necessary, a vinyl aromatic compound such as styrene) may be sequentially added to carry out anion polymerization.
- one or two or more solvents selected from solvents that can be used in ordinary anion polymerization for example, hexane, cyclohexane, benzene, and toluene can be used as appropriate.
- a butadiene unit and an isoprene unit are used.
- getyl ether, tetrahydrofuran, ethylene glycol methyl ether can be used to control the ratio of bonding modes (1,2-bond, 1,4-single bond, 3,4-bond, etc.)
- Additives such as ethers such as triethylamine, N, N, ⁇ ′, N′—amines such as tetramethylethylenediamine may be added.
- a hydrogenated (hydrogenated) reaction is performed on the gen-based polymer obtained by the above-described anion polymerization to form a hydrogenated gen-based polymer block ⁇ ⁇ ⁇ ⁇ , and a block copolymer (3) is obtained.
- the hydrogenation reaction is carried out according to a known method.
- a supported catalyst comprising a metal such as Pt, Pd, Ru, Rh, and Ni supported on a carrier such as carbon, alumina, or diatomaceous earth; Raney nickel; Ziegler catalysts comprising a combination of a metal compound and an organoaluminum compound or an organolithium compound; hydrogenation catalysts such as a meta-opening catalyst comprising a combination of an organotitanium compound and an organoaluminum compound or an organolithium compound; By hydrogenating the gen-based polymer in a solvent inert to a hydrogenation reaction such as hexane or cyclohexane in the presence of
- the gen-based polymer is used.
- triethylamine, N, N, ⁇ ' , ⁇ '-tetramethylethylenediamine Hydrogenation can be performed by adding amines such as acetyl ether and tetrahydrofuran.
- the content of each block in the block copolymer (3) used in the present invention is not particularly limited. However, from the viewpoint of the effect of improving the dispersion characteristics and the effect of improving the adhesion at the interface of the rubber phase, the gen-based polymer is used.
- the content of the coalesced block A (the sum of a plurality of gen-based polymer blocks A when the block copolymer (3) is present in the block copolymer (3)) is in the range of 20 to 80% by mass before hydrogenation.
- the content of the hydrogenated gen-based polymer block B (when a plurality of hydrogenated gen-based polymer blocks B are present in the block copolymer (3), the sum thereof is preferable). ) Is preferably in the range of 80 to 20% by mass before hydrogenation.
- the reaction pressure, reaction temperature and reaction time of the hydrogenation reaction are not particularly limited, but usually, the hydrogen pressure is in the range of 0.01 to 20 MPa, the reaction temperature is in the range of 10 to 250, and the reaction time is Is used in the range of 0.1 to 200 hours.
- the method for obtaining the block copolymer (3) from the obtained reaction mixture is not particularly limited.
- the reaction mixture containing the block copolymer (3) is converted to a poor mixture such as methanol.
- the block copolymer (3) can be obtained by coagulating by contacting with a solvent, removing the coagulated material, pre-drying it, and drying it under heating or reduced pressure.
- the rubber composition of the present invention contains 0.1 to 25 parts by mass of the block copolymer (3) based on 100 parts by mass of the total weight of the gen-based rubber (1) and the highly saturated rubber (2). It is characterized by the following.
- the content of the block copolymer (3) is in the range of 0.5 to 20 parts by mass based on 100 parts by mass of the total mass of the gen-based rubber (1) and the highly saturated rubber (2). It is more preferably in the range of 1 to 15 parts by mass.
- the amount of the block copolymer (3) is less than 0.1 part by mass or more than 25 parts by mass with respect to 100 parts by mass of the total mass of the gen-based rubber (1) and the highly saturated rubber (2). In both cases, the effect of improving the dispersion characteristics and the effect of improving the adhesion at the interface are poor.
- the rubber composition of the present invention is produced by applying a commonly used kneading method. It is obtained by kneading predetermined amounts of the gen-based rubber (1), the highly saturated rubber (2) and the block copolymer (3) using, for example, a Brabender, Banbury mixer, roll kneader or the like.
- a reinforcing agent usually added for the purpose of reinforcing the rubber composition for example, carbon black / silica can be further added as long as the characteristics of the compounding composition are not impaired.
- crosslinkable rubber composition of the present invention will be described.
- the cross-linking agent that can be contained in such a cross-linkable rubber composition those usually used for rubber cross-linking can be used without any particular limitation. Examples thereof include sulfur, morpholine disulfide, and alkylphenol disulfide.
- Sulfur cross-linking agents such as chloride; cyclohexanone peroxide, methyl acetate acetate peroxide, tert-butyl butoxy benzoate, tert-butyl vinyl benzoate, benzoyl peroxide, lauroyl peroxide
- Organic peroxide crosslinking agents such as amide, dicumyl peroxide, di-tert-butyl peroxide, and 1,3-bis (tert-butylvinyl isopropyl) benzene.
- the amount of the crosslinking agent to be used is usually 0.05 to 10 parts by mass with respect to 100 parts by mass of the total amount of the diene rubber (1), the highly saturated rubber (2) and the block copolymer (3). Part by mass, more preferably 0.1 to 5 parts by mass.
- the crosslinkable rubber composition of the present invention may further contain a crosslinking accelerator or a crosslinking assistant, if necessary.
- the crosslinking accelerator and the crosslinking assistant are not particularly limited, and can be appropriately selected and used according to the crosslinking agent used.
- the crosslinking accelerator include thiuram-based accelerators such as tetramethylthiuram monosulfide, tetramethylthiuram disulfide, and tetraethylthiuram disulfide; 2-mercaptobenzothiazole, dibenzothiazyl disulfide, and the like; N-cyclohexyl-2-benzothiazylsulfenamide, N-tert-butyl-12-benzothiazylsulfenamide, N-oxydiethylene-12-benzothiazole Sulfenamide-based accelerators such as azolylsulfenamide and the like can be mentioned. These crosslinking accelerators may be used in combination of two or more.
- crosslinking aid examples include metal oxides such as zinc oxide and magnesium oxide; metal hydroxides such as calcium hydroxide; metal carbonates such as zinc carbonate and basic zinc carbonate; fatty acids such as stearic acid and oleic acid. Metal salts of fatty acids such as zinc stearate and magnesium stearate; ethylene dimethacrylate, diaryl phthalate, N, Nm-phenylene maleimide, triaryl isocyanurate, trimethylolpropane trimethacrylate, and the like. Can be These crosslinking aids may be used in combination of two or more.
- the crosslinkable rubber composition of the present invention may further contain various additives such as various fillers, antioxidants, fillers, plasticizers, and softeners, as long as the performance is not impaired. Can also.
- the crosslinkable rubber composition of the present invention can be produced by applying a commonly used kneading method, wherein a predetermined amount of the gen-based rubber (1), the highly saturated rubber (2) and the block copolymer (3) is determined. It can be obtained by adding a crosslinking agent, and if necessary, a crosslinking accelerator, a crosslinking assistant, and the like. In addition, a crosslinked product can be obtained by subjecting such a crosslinkable rubber composition to crosslinking by, for example, a press molding machine or mold crosslinking.
- the rubber composition of the present invention is excellent in miscibility and has improved adhesion at the interface of the rubber phase, it has excellent tensile properties and improved bending properties. Since the rubber composition of the present invention maintains its properties even when vulcanized rubber is further crosslinked, it can be suitably used for applications such as tires, industrial belts, and industrial parts such as industrial rubber hoses. Can be.
- the mixture was poured into 23700 g of methanol to coagulate the polymer.
- the coagulated product was collected by filtration, and further dried under reduced pressure at 80 for 12 hours to obtain 575 g of the product.
- Natural rubber (NR; RSS # 1, ribbed smoked sheet), ethylene-propylene-gene copolymer rubber (EPDM; manufactured by Mitsui Petrochemical Co., Ltd., trade name: EPT 4045, ethylidene norbornene type, iodine
- the EB (1) obtained by the method of Reference Example 24) and Reference Example 1 is kneaded at a mixing ratio (mass ratio) shown in Table 1 using a Brabender at 50 at 100 rpm for 5 minutes.
- a rubber composition was obtained by pressing the obtained rubber composition under conditions of 100 t and 1 minute to prepare a dumbbell-shaped No. 5 type test piece having a thickness of 2 mm, which was JISK-6251.
- a 100% modulus, breaking strength and breaking elongation were measured by performing a tensile test according to the method.
- a part of the obtained rubber composition was cut with a freezing microtome, stained with osmium tetroxide (only the natural rubber part was stained), and then observed with a scanning electron microscope (SEM). The dispersion state of the rubber component was observed, and the average dispersed particle size of EPDM was determined. Table 1 shows the results.
- a rubber composition consisting only of NR and EPDM was prepared in the same manner as in Example 1 without adding IR- ⁇ (1), and a tensile test and observation by SEM were performed in the same manner as in Example 1. Table 1 shows the results.
- E PDM “E PT 4045” manufactured by Mitsui Petrochemical Co., Ltd.
- the average dispersed particle size of EPDM was 1.5 m in Comparative Example 1. On the other hand, in Example 1, the value was 0.5 / m.
- IR-EB (1) a kneader having a relatively small torque and a low mechanical dispersing ability like Brabender was used. However, the dispersed particle diameter could be reduced.
- Example 1 Comparing the tensile test results, it can be seen that the rubber composition obtained in Example 1 has improved elongation due to the addition of IR-EB (1), and hardly decreases other mechanical properties.
- NR (RS S # 1, ribbed smoked sheet), EPDM (manufactured by Mitsui Petrochemical Co., Ltd., trade name: EPT 4045, ethylidene norbornene type, iodine value 24), IR obtained by the method of Reference Example 1 — EB (1), carbon black, zinc white and stearic acid were kneaded at a mixing ratio (mass ratio) shown in Table 2 with a Banbury mixer at 100 t: for 4 minutes.
- a mixing ratio mass ratio
- sulfur and a crosslinking accelerator [trade name “Noxeller CZ” (N-cyclohexyl 2-benzothiazylsulfenamide), manufactured by Ouchi Shinkosha Co., Ltd.]
- an antioxidant [product The name "Nocrack 8100 NAJ (N-isopropyl-N'—Fenilou p-Fenylenediamine), manufactured by Ouchi Shinkosha] was mixed with an open roll at the compounding ratio shown in Table 2, and at 150, Crosslinking was performed by pressing for 10 minutes to obtain a crosslinked rubber sheet having a thickness of 2 mm.
- the hardness of the obtained crosslinked rubber sheet was measured with Type A durometer overnight in accordance with JISK-6250.
- a dumbbell-shaped No. 5 test piece was prepared from the obtained crosslinked rubber sheet by punching and subjected to a tensile test in accordance with JISK-6251, with a 100% modulus, a 300% modulus, a breaking strength, and a break. The elongation was measured.
- a test piece was prepared from the obtained crosslinked rubber sheet in accordance with JIS-6260, and a 2 mm notch was made in the center of the test piece to form a flex crack according to JIS K-6260.
- test was performed, and the number of flexures until the crack grew to a length of 8 mm was evaluated by the average value of two samples. Furthermore, a test piece was prepared from the obtained crosslinked rubber sheet in accordance with JISK-6260, and a bending crack generation test was performed. The surface condition of the test piece at 80,000 bending times was determined according to the series described in JIS.
- Example 2 The same operation as in Example 2 was carried out except that I R -EB (1) was not added, to obtain a crosslinked rubber sheet.
- Table 2 summarizes the results of the hardness, tensile test, flex crack growth test, and flex crack initiation test of the obtained crosslinked rubber sheet.
- EP DM Mitsui Petrochemical Co., Ltd. “EP 4045 J
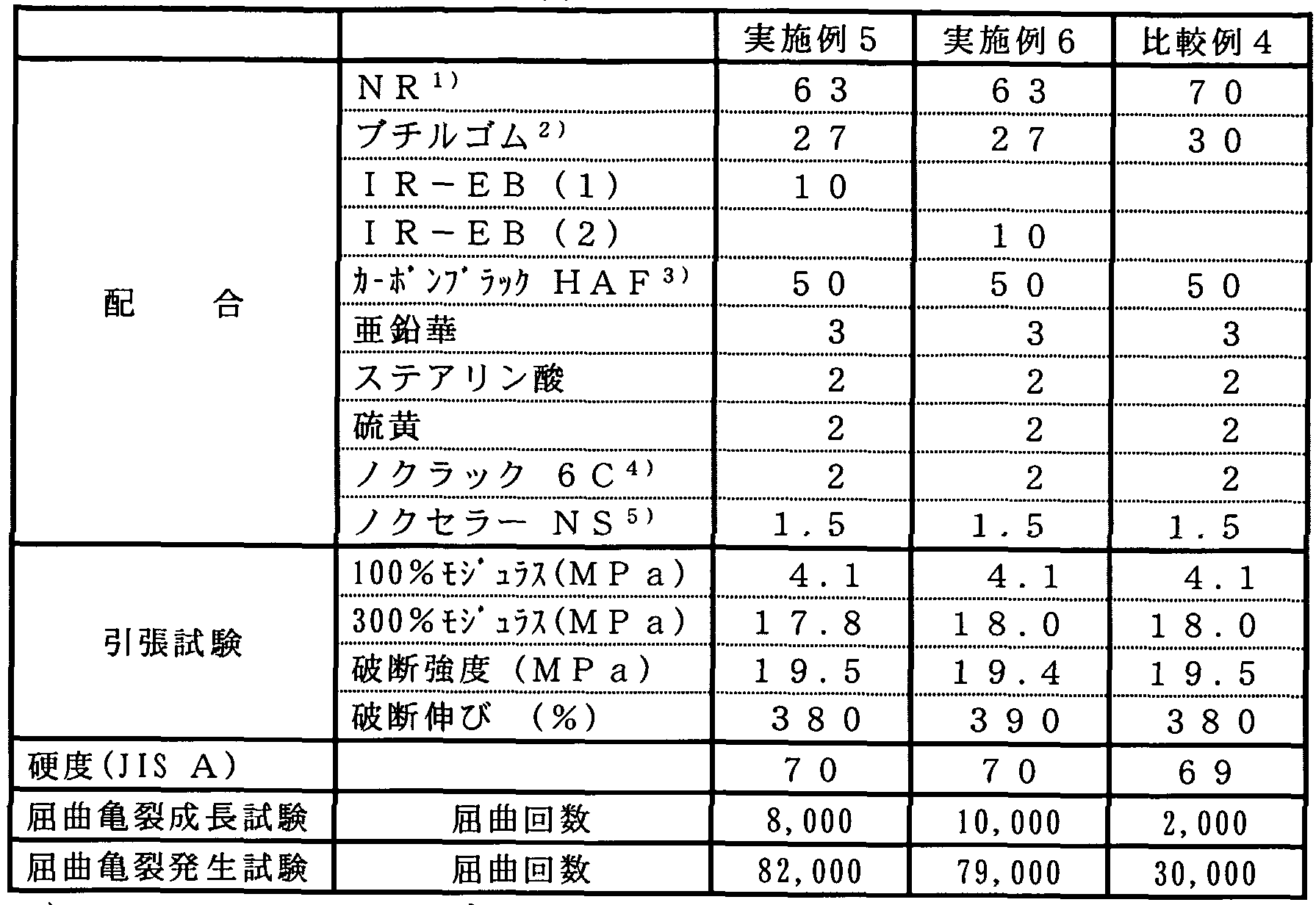
- a rubber composition consisting only of NR and butyl rubber was prepared in the same manner as in Examples 3 and 4, and observed by SEM in the same manner as in Examples 3 and 4. Table 3 shows the results.
- the average dispersed particle diameter of butyl rubber was 1.0 m in Comparative Example 3, 0.3 im in Example 3, and 0.2 jum in Example 4, and I R- By adding EB (1) or IR-EB (2), the dispersed particle diameter could be reduced.
- NR ribbed smoked sheet
- butyl rubber manufactured by Exxon Chemical Company, trade name "EXXP RO 3745", bromide of p-methylstyrene copolymerized butyl rubber
- IR-EB (1) or IR-EB (2), force pump rack, zinc white and stearic acid were kneaded at a ratio shown in Table 4 with a Banbury mixer at 75 for 5 minutes.
- a test piece was prepared in accordance with K-6260 and subjected to a bending crack generation test.
- the class described in JIS was class 3 (the crack length was 0.5 mm or more and 1.0 mm even with one " ⁇ hole") ) was evaluated by the average value of the number of samples of 2.
- Table 4 summarizes the results of the hardness, tensile test, flex crack growth test, and flex crack initiation test.
- Crosslinked rubber sheets were obtained by performing the same operations as in Examples 5 and 6 except that IR-EB (1) or IR-EB (2) was not added.
- the hardness, tensile test, flex crack growth test and flex crack initiation test of the obtained crosslinked rubber sheet were performed in the same manner as in Examples 5 and 6. The results are summarized in Table 4.
- the miscibility between the gen-based rubber and the highly saturated rubber that is, the rubber composition which is improved in the dispersibility and the interfacial adhesion, is excellent in the tensile properties and the bending properties, and the A rubber composition and a crosslinked product can be obtained.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP02733458A EP1403312B1 (en) | 2001-06-14 | 2002-06-11 | Rubber composition, crosslinkable rubber composition and crosslinked articles |
| KR1020037016289A KR100585310B1 (ko) | 2001-06-14 | 2002-06-11 | 고무 조성물, 가교성 고무 조성물 및 가교물 |
| US10/479,985 US20040167281A1 (en) | 2001-06-14 | 2002-06-11 | Rubber composition, crosslinkable rubber composition and crosslinked articles |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2001179998 | 2001-06-14 | ||
| JP2001-179998 | 2001-06-14 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2002102889A1 true WO2002102889A1 (en) | 2002-12-27 |
Family
ID=19020483
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2002/005777 Ceased WO2002102889A1 (en) | 2001-06-14 | 2002-06-11 | Rubber composition, crosslinkable rubber composition and crosslinked articles |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20040167281A1 (ja) |
| EP (1) | EP1403312B1 (ja) |
| KR (1) | KR100585310B1 (ja) |
| WO (1) | WO2002102889A1 (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7909194B2 (en) * | 2005-07-01 | 2011-03-22 | Exxonmobil Chemical Patents Inc. | Thermoplastic vulcanizates and sealing devices made therewith |
| JP6546570B2 (ja) * | 2016-08-31 | 2019-07-17 | 住友理工株式会社 | 防振ゴム組成物および防振ゴム |
| CN109160966A (zh) * | 2018-07-06 | 2019-01-08 | 杭州师范大学 | 一种硅藻土负载茂金属催化剂及其制备方法与应用 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08109288A (ja) * | 1994-08-19 | 1996-04-30 | Japan Synthetic Rubber Co Ltd | ポリオレフィン系樹脂組成物 |
| JPH11335498A (ja) * | 1998-05-26 | 1999-12-07 | Jsr Corp | 熱可塑性エラストマー組成物 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4250273A (en) * | 1977-06-13 | 1981-02-10 | The Firestone Tire & Rubber Company | Thermoplastic elastomer blends |
| US5187236A (en) * | 1990-01-16 | 1993-02-16 | Mobil Oil Corporation | Solid block and random elastomeric copolymers |
| CN1124970A (zh) * | 1993-04-19 | 1996-06-19 | 埃克森化学专利公司 | 含有异烯烃和烷基苯乙烯的接枝聚合物的相容化弹性体共混物 |
| US5571865A (en) * | 1994-05-16 | 1996-11-05 | Kuraray Co., Ltd. | Thermoplastic polymer composition |
| US6184307B1 (en) * | 1998-01-30 | 2001-02-06 | Shell Oil Company | Minimum residence time hydrogenation process for polyisoprene-polybutadiene block copolymers |
-
2002
- 2002-06-11 WO PCT/JP2002/005777 patent/WO2002102889A1/ja not_active Ceased
- 2002-06-11 US US10/479,985 patent/US20040167281A1/en not_active Abandoned
- 2002-06-11 EP EP02733458A patent/EP1403312B1/en not_active Expired - Lifetime
- 2002-06-11 KR KR1020037016289A patent/KR100585310B1/ko not_active Expired - Lifetime
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08109288A (ja) * | 1994-08-19 | 1996-04-30 | Japan Synthetic Rubber Co Ltd | ポリオレフィン系樹脂組成物 |
| JPH11335498A (ja) * | 1998-05-26 | 1999-12-07 | Jsr Corp | 熱可塑性エラストマー組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1403312A4 * |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1403312B1 (en) | 2012-04-25 |
| KR100585310B1 (ko) | 2006-06-01 |
| EP1403312A1 (en) | 2004-03-31 |
| KR20040012926A (ko) | 2004-02-11 |
| EP1403312A4 (en) | 2005-05-11 |
| US20040167281A1 (en) | 2004-08-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20100152368A1 (en) | Rubber composition and crosslinked object | |
| WO2005097887A1 (en) | Thermoplastic elastomeric material comprising a vulcanized rubber in a subdivided form | |
| JP2003253051A (ja) | ゴム組成物及びそれを用いたタイヤ | |
| WO2004000931A1 (ja) | タイヤ用ゴム組成物及びこれを用いたタイヤ | |
| CN1142966C (zh) | 橡胶组合物 | |
| EP3625291A1 (en) | Vulcanizable composition and moldable thermoplastic elastomer product therefrom | |
| KR102654433B1 (ko) | 고무 조성물 | |
| US20060189720A1 (en) | Rubber composition, crosslinkable rubber compositions, and crosslinked articles | |
| WO2002102889A1 (en) | Rubber composition, crosslinkable rubber composition and crosslinked articles | |
| JP4067151B2 (ja) | タイヤ用ゴム組成物 | |
| US4433107A (en) | Polyisoprene rubber compositions | |
| US6653409B2 (en) | Radical-modified polymer and polymer composition containing the same | |
| JP2004075882A (ja) | ウエットグリップ性の改良されたゴム組成物 | |
| JPH0373584B2 (ja) | ||
| JP3917012B2 (ja) | ゴム組成物、架橋性ゴム組成物および架橋物 | |
| JPH0615647B2 (ja) | ゴム組成物 | |
| JP3999117B2 (ja) | ゴム組成物、架橋性ゴム組成物および架橋物 | |
| JP2001247722A (ja) | ゴム組成物 | |
| EP4353493A1 (en) | Rubber composition for tire, tread rubber, and tire | |
| JP2004359934A (ja) | フラーレンに結合した重合体を含むゴム組成物 | |
| EP1432760A1 (en) | Covulcanisation of polymers | |
| JP2003128726A (ja) | ラジカル変性ポリマー及びそれを含むポリマー組成物 | |
| JP6852985B2 (ja) | ゴム組成物 | |
| JP2002212363A (ja) | タイヤインナーライナー用ゴム組成物 | |
| WO2003054079A1 (en) | Rubber composition, crosslinkable rubber composition and crosslinked rubber |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2002733458 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020037016289 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10479985 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 2002733458 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |