WO2003089413A1 - Pharmacia corporation - Google Patents
Pharmacia corporation Download PDFInfo
- Publication number
- WO2003089413A1 WO2003089413A1 PCT/US2003/011551 US0311551W WO03089413A1 WO 2003089413 A1 WO2003089413 A1 WO 2003089413A1 US 0311551 W US0311551 W US 0311551W WO 03089413 A1 WO03089413 A1 WO 03089413A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- salt
- bipiperidine
- carboxylate
- propionylphenyl
- amino
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/36—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D211/56—Nitrogen atoms
- C07D211/58—Nitrogen atoms attached in position 4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- the present invention relates to manufacture of camptothecin derivatives therapeutically useful as anti-cancer drugs, in particular the camptothecin derivative irinotecan and salts thereof. More particularly, the invention relates to a novel intermediate and to a process for preparing a camptothecin derivative via that intermediate.
- U.S. Patent No. 6,121,451 to Henegar & Sih discloses a process for preparing the antineoplastic drug irinotecan, also known as 7-ethyl- 10-hydroxycamptothecin 10-[l,4'-bipiperidine]-r-carboxylate or CPT-11 free base.
- a compound therein identified as 14CPT is first reacted with l-(4- hydroxy-2-arninophenyl)-l-propanone (II) to form an intermediate compound (III), which is then reacted with 4-piperidinopiperidinecarbamyl chloride (IV) to produce CPT-11 free base (V), as shown schematically below.
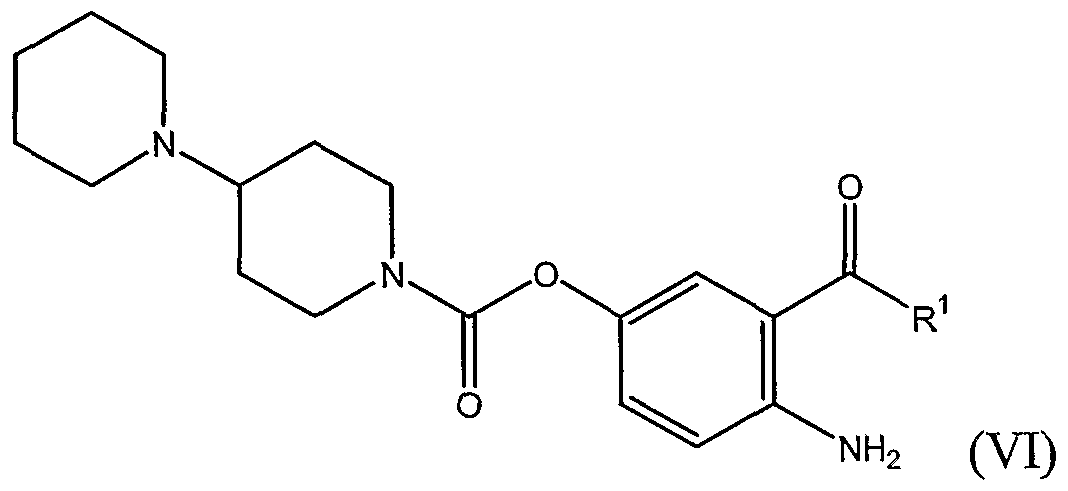
- R 1 is hydrogen, an alkyl, aralkyl, hydroxymethyl, carboxymethyl, acyloxymethyl or trialkylsilyl group, or a group -CH 2 NR 3 R 4 where N is a linking nitrogen atom and where R 3 and R 4 are as defined hereinbelow.
- N is a linking nitrogen atom and where R 3 and R 4 are as defined hereinbelow.
- salts of the compounds of formula (VI) with pharmaceutically acceptable anions are:
- R 3 and R 4 are independently selected from hydrogen and alkyl, alkenyl, hydroxyalkyl and alkoxyalkyl groups;
- R 3 is hydrogen or an alkyl, alkenyl, hydroxyalkyl or alkoxyalkyl group
- R 4 is -COR 5 where R 5 is hydrogen or an alkyl, alkenyl, hydroxyalkyl or alkoxyalkyl group
- R 1 is as defined above and R 2 is hydrogen or an alkyl group, preferably hydrogen.
- R 1 is an ethyl group and R 2 is hydrogen.
- the novel compound of formula (VI) is 4-amino-3-propionylphenyl- l,4'-bipiperidine-r-carboxylate, which is a useful intermediate in a process to prepare irinotecan (V) and salts thereof, for example the hydrochloride salt CPT-11.
- a process comprising a step of reacting 4-amino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate with the compound of formula (I) above to form irinotecan.
- a process for preparing a compound of formula (VI).
- This process can be illustrated with respect to the compound of formula (VI) where R 1 is an ethyl group, i.e., 4-amino-3-propionylphenyl- l,4'-bipiperidine-r-carboxylate.
- the process comprises a step of reacting l-(4- hydroxy-2-aminophenyl)-l-propanone (II) with 4-piperidinopiperidinecarbamyl chloride (IV) to form 4-arnino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate.
- an irinotecan or CPT-11 product prepared by a process using 4-amino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate will contain at least a detectable amount of that compound.
- a pharmaceutical composition comprising a therapeutically effective amount of irinotecan and/or a salt thereof and at least a detectable amount of 4-amino-3- propionylphenyl- 1 ,4-bipiperidine- 1 -carboxylate and/or a salt thereof.
- 4-amino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate is useful as an analytical marker for a product of (a) a process involving that compound as a reagent or (b) a process involving as reagents l-(4-hydroxy-2-aminophenyl)-l-propanone and 4-piperidinopiperidinecarbamyl chloride under circumstances permitting these reagents to react to form 4-amino-3-propionylphenyl-l,4'-bipiperidine- -carboxylate.
- R 1 in a compound of formula (VI) of the present invention is hydrogen, an alkyl, aralkyl, hydroxymethyl, carboxymethyl, acyloxymethyl or trialkylsilyl, e.g., trimethylsilyl, group, or a group -CH 2 NR 3 R 4 where N is a linking nitrogen atom and where (a) R 3 and R 4 are independently selected from hydrogen and alkyl, alkenyl, hydroxyalkyl and alkoxyalkyl groups; (b) R 3 is hydrogen or an alkyl, alkenyl, hydroxyalkyl or alkoxyalkyl group, and R 4 is -COR 5 where R 5 is hydrogen or an alkyl, alkenyl, hydroxyalkyl or alkoxyalkyl group; or (c) R 3 and R 4 taken together with the linking nitrogen atom form a saturated 3- to 7-member heterocyclic group.
- Alkyl, alkenyl, aralkyl, acyl and alkoxy groups herein, unless otherwise defined, have 1-30, preferably 1-18, more preferably 1-6, carbon atoms.
- R 1 is a ⁇ alkyl, most preferably an ethyl, group.
- Compounds of formula (VI) above exist in free base form and in various pharmaceutically acceptable salt forms, which are embodiments of the present invention.
- compositions of formula (VI) include without restriction salts of the following acids: hydrochloric, hydrobromic, sulfuric, methanesulfonic, phosphoric, nitric, benzoic, citric, tartaric, fumaric and maleic acids, and mono- and dicarboxylic acids of formula CH 3 -(CH 2 ) n -COOH and HOOC-(CH 2 ) n -COOH where n is 0 to 4, for example malonic acid.
- the hydrochloride salt is particularly preferred.
- a process comprising a step of reacting 4-amino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate (the compound of formula (VI) where R 1 is ethyl) or a salt thereof with 14CPT ((4S)-4-ethyl-7,8-dihydro-4- hydroxy-(lH)-pyrano[3,4-flindolizine-3,6,10(4H)-trione), the compound of formula (I) above, to form irinotecan or the corresponding salt thereof.
- 4-amino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate the compound of formula (VI) where R 1 is ethyl
- 14CPT ((4S)-4-ethyl-7,8-dihydro-4- hydroxy-(lH)-pyrano[3,4-flindolizine-3,6,10(4H)-trione)
- This reaction can illustratively be carried out by heating 4-amino-3- propionylphenyl-l,4'-bipiperidine- l'-carboxylate and 14CPT together in a suitable solvent, for example acetic acid.
- a suitable solvent for example acetic acid.
- the product of the reaction can then be purified by a suitable chromatographic method and isolated, for example by crystallization from a suitable solvent medium.
- An illustrative example of a process of this embodiment of the invention is provided below in Example 2.
- novel compound 4-amino-3-propionylphenyl-l ,4'- bipiperidine-r-carboxylate or a salt thereof is a useful intermediate in a manufacturing process for irinotecan or a salt thereof.
- irinotecan or a salt thereof prepared by a process of the invention can be further processed to yield other camptothecin derivatives by methods known in the art.
- irinotecan prepared as described herein can be subjected to hydrolysis in an acid medium, for example in presence of hydrochloric acid, to yield 7-ethyl-10- hydroxycamptothecin.
- 4-arnino-3-propionylphenyl-l,4 , -bipiperidine-r-carboxylate comprising a step of reacting l-(4-hydroxy-2-aminophenyl)-l-propanone (II) with
- This reaction can illustratively be carried out by mixing known compounds (II) and (IV) together in a suitable solvent, for example pyridine.
- a suitable solvent for example pyridine.
- a solvent system comprising methylene chloride, tetrahydrofuran, acetonitrile or the like together with a suitable base such as triethylamine or dusopropylethylamine can be used as a medium for the reaction.
- the product of the reaction can then be subjected to isolation and purification steps, illustratively those described in Example 1 below, to yield 4-amino-
- a pharmaceutical composition or drug substance comprising (a) irinotecan and/or one or more pharmaceutically acceptable salts thereof in a therapeutically effective total amount, and (b) 4-amino-3- propionylphenyl-l,4'-bipiperidine- l'-carboxylate and/or one or more salts thereof in at least a detectable amount.
- a "therapeutically effective" amount of irinotecan or salt thereof is meant an amount useful as at least a single dosage amount for treatment of cancer.
- the drug substance comprises not more than about 5%, more preferably not more than about 2.5% and most preferably not more than about 1% by weight of 4-amino-3-propionylphenyl-l,4'-bipiperidine-r- carboxylate.
- the presence of 4-arnino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate in a drug substance of this embodiment is useful as an analytical marker, providing evidence, for example, that the irinotecan with which it occurs has been prepared by (a) a process involving 4-arr ⁇ ino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate as a reagent (e.g., the process described above and illustrated in Example 2 below) or (b) a process involving as reagents compounds (II) and (IV) under circumstances permitting these compounds to react to form 4-arnino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate.
- a process involving 4-arr ⁇ ino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate e.g., the process described above and illustrated in Example 2
- Example 1 4-A-mino-3-propionylphenyl-l .4'-bipiperidine-l '-carboxylate [0023] A solution of 3.26 g (19.6 mmol) 2-amino-5-hydroxypropiophenone and 6.97 g (26.1 mmol) 4-piperidinopiperidinecarbamoyl chloride hydrochloride in 35 mlpyridine was stirred at room temperature for 13 hours. The pyridine was then evaporated and 20 ml water and 120 ml ethyl acetate were added. The resulting organic phase was separated and dried over sodium sulfate.
- the sodium sulfate was removed by filtration and 10 g silica-60 was added to the filtrate, which was stirred to form a slurry.
- the slurry was loaded onto a column of 20 g silica-60 (230-400 mesh) and eluted with a 95:5 (v/v) methylene chloride/methanol mixture.
- the resulting product fractions were combined and evaporated to provide a residue, which was then redissolved in a mixture of 20 ml methylene chloride and 50 ml heptane.
- the resulting solution was evaporated to yield 6.22 g of4-amino-3-propionylphenyl-l,4'-bipiperidine-r-carboxylate as a yellow-green solid, melting point 132.0-133.5°C.
- the irinotecan produced as above (0.216 g) was dissolved in 2 ml water and 0.43 ml 1M HC1 and heated to 60°C to form a yellow solution. This solution was filtered hot over powdered activated carbon (DarcoTM G-60) (0.5 g). The filtrate was cooled and seeded with 5 mg irinotecan hydrochloride crystals and allowed to crystallize overnight. The solids were filtered, washed with water (2 x 1 ml), and suction dried under air to yield 0.123 g of irinotecan hydrochloride trihydrate as pale yellow crystals having a melting point of 259.8°C.
- DarcoTM G-60 powdered activated carbon
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Animal Behavior & Ethology (AREA)
- General Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Hydrogenated Pyridines (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MXPA04009426A MXPA04009426A (en) | 2002-04-17 | 2003-04-16 | Pharmacia corporation. |
| BR0309284-4A BR0309284A (en) | 2002-04-17 | 2003-04-16 | Compounds useful in the preparation of camptothecin derivatives |
| EP03721671A EP1501799A1 (en) | 2002-04-17 | 2003-04-16 | Compounds useful in preparing camptothecin derivatives |
| KR10-2004-7016524A KR20050009293A (en) | 2002-04-17 | 2003-04-16 | Compounds Useful in Preparing Camptothecin Derivatives |
| AU2003224973A AU2003224973A1 (en) | 2002-04-17 | 2003-04-16 | Compounds useful in preparing camptothecin derivatives |
| CA002482712A CA2482712A1 (en) | 2002-04-17 | 2003-04-16 | Compounds useful in preparing camptothecin derivatives |
| YUP-913/04A RS91304A (en) | 2002-04-17 | 2003-04-16 | Compounds useful in preparing campthothecin derivatives |
| JP2003586134A JP3839813B2 (en) | 2002-04-17 | 2003-04-16 | Compounds useful for the preparation of camptothecin derivatives |
| IL16431104A IL164311A0 (en) | 2002-04-17 | 2004-09-27 | Compounds useful in preparing coamptothecin derivatives |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US37372702P | 2002-04-17 | 2002-04-17 | |
| US60/373,727 | 2002-04-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2003089413A1 true WO2003089413A1 (en) | 2003-10-30 |
Family
ID=29251068
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2003/011551 Ceased WO2003089413A1 (en) | 2002-04-17 | 2003-04-16 | Pharmacia corporation |
Country Status (15)
| Country | Link |
|---|---|
| US (1) | US6723729B2 (en) |
| EP (1) | EP1501799A1 (en) |
| JP (1) | JP3839813B2 (en) |
| KR (1) | KR20050009293A (en) |
| CN (1) | CN1646491A (en) |
| AU (1) | AU2003224973A1 (en) |
| BR (1) | BR0309284A (en) |
| CA (1) | CA2482712A1 (en) |
| IL (1) | IL164311A0 (en) |
| MX (1) | MXPA04009426A (en) |
| PL (1) | PL371681A1 (en) |
| RS (1) | RS91304A (en) |
| RU (1) | RU2287527C2 (en) |
| WO (1) | WO2003089413A1 (en) |
| ZA (1) | ZA200407821B (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006016203A1 (en) * | 2004-08-09 | 2006-02-16 | Shilpa Medicare Limited | An improved process for the preparation of irinotecan hydrochloride trihydrate |
| WO2008035377A3 (en) * | 2006-09-18 | 2009-01-08 | Shilpa Medicare Ltd | Novel intermediates for the preparation of camptothecin analogues |
| US7662964B2 (en) | 2005-02-08 | 2010-02-16 | Fermion Oy | Process for producing [1,4′] bipiperidinyl-1′-carbonyl chloride or hydrochloride thereof |
| US7767813B2 (en) * | 2005-02-08 | 2010-08-03 | Fermion Oy | Preparation method |
| US7910737B2 (en) | 2005-02-07 | 2011-03-22 | Fermion Oy | Process for the manufacturing of 7-ethyl-10-hydroxy camptothecin |
| EP2399908A1 (en) * | 2009-11-18 | 2011-12-28 | Cadila Healthcare Limited | Polymorphic form of 7-ethyl-10[4-(1-piperidino)-1- piperidino]carbonyloxy- camptothecin and its use |
| WO2012032531A1 (en) * | 2010-09-06 | 2012-03-15 | Avra Laboratories Pvt. Ltd. | Process for the manufacture of irinotecan hydrochloride by total synthesis |
| JP5086804B2 (en) * | 2005-06-09 | 2012-11-28 | 株式会社ヤクルト本社 | Method for producing tricyclic ketone |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050197355A1 (en) * | 2004-03-02 | 2005-09-08 | Henegar Kevin E. | Compounds useful in preparing camptothecin derivatives |
| EP2881396A1 (en) * | 2013-12-03 | 2015-06-10 | Synbias Pharma AG | Method for the synthesis of irinotecan |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6121451A (en) * | 1995-04-07 | 2000-09-19 | Pharmacia & Upjohn Company | Intermediates and process for the manufacture of camptothecin derivatives (CPT-11) and related compounds |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0220601B1 (en) * | 1985-10-21 | 1991-12-11 | Daiichi Seiyaku Co., Ltd. | Pyranoindolizine derivatives and preparation process thereof |
| US5952354A (en) * | 1993-07-21 | 1999-09-14 | American Home Products Corporation | Tris carbamic acid esters: inhibitors of cholesterol absorption |
-
2003
- 2003-04-16 CA CA002482712A patent/CA2482712A1/en not_active Abandoned
- 2003-04-16 AU AU2003224973A patent/AU2003224973A1/en not_active Abandoned
- 2003-04-16 US US10/414,852 patent/US6723729B2/en not_active Expired - Fee Related
- 2003-04-16 JP JP2003586134A patent/JP3839813B2/en not_active Expired - Fee Related
- 2003-04-16 RS YUP-913/04A patent/RS91304A/en unknown
- 2003-04-16 KR KR10-2004-7016524A patent/KR20050009293A/en not_active Ceased
- 2003-04-16 EP EP03721671A patent/EP1501799A1/en not_active Withdrawn
- 2003-04-16 BR BR0309284-4A patent/BR0309284A/en not_active IP Right Cessation
- 2003-04-16 MX MXPA04009426A patent/MXPA04009426A/en unknown
- 2003-04-16 RU RU2004130493/04A patent/RU2287527C2/en not_active IP Right Cessation
- 2003-04-16 CN CNA038083353A patent/CN1646491A/en active Pending
- 2003-04-16 WO PCT/US2003/011551 patent/WO2003089413A1/en not_active Ceased
- 2003-04-16 PL PL03371681A patent/PL371681A1/en not_active Application Discontinuation
-
2004
- 2004-09-27 IL IL16431104A patent/IL164311A0/en unknown
- 2004-09-28 ZA ZA200407821A patent/ZA200407821B/en unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6121451A (en) * | 1995-04-07 | 2000-09-19 | Pharmacia & Upjohn Company | Intermediates and process for the manufacture of camptothecin derivatives (CPT-11) and related compounds |
Non-Patent Citations (1)
| Title |
|---|
| JOSIEN H ET AL: "A GENERAL SYNTHETIC APPROACH TO THE (20S)-CAMPTOTHECIN FAMILY OF ANTITUMOR AGENTS BY A REGIOCONTROLLED CASCADE RADICAL CYCLIZATION OF ARYL ISONITRILES", CHEMISTRY - A EUROPEAN JOURNAL, VCH PUBLISHERS, US, vol. 4, no. 1, 1998, pages 67 - 83, XP001015572, ISSN: 0947-6539 * |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006016203A1 (en) * | 2004-08-09 | 2006-02-16 | Shilpa Medicare Limited | An improved process for the preparation of irinotecan hydrochloride trihydrate |
| JP2008509211A (en) * | 2004-08-09 | 2008-03-27 | シルパ・メディケア・リミテッド | Improved process for the preparation of irinotecan hydrochloride trihydrate |
| US7910737B2 (en) | 2005-02-07 | 2011-03-22 | Fermion Oy | Process for the manufacturing of 7-ethyl-10-hydroxy camptothecin |
| US7662964B2 (en) | 2005-02-08 | 2010-02-16 | Fermion Oy | Process for producing [1,4′] bipiperidinyl-1′-carbonyl chloride or hydrochloride thereof |
| US7767813B2 (en) * | 2005-02-08 | 2010-08-03 | Fermion Oy | Preparation method |
| JP5086804B2 (en) * | 2005-06-09 | 2012-11-28 | 株式会社ヤクルト本社 | Method for producing tricyclic ketone |
| WO2008035377A3 (en) * | 2006-09-18 | 2009-01-08 | Shilpa Medicare Ltd | Novel intermediates for the preparation of camptothecin analogues |
| EP2399908A1 (en) * | 2009-11-18 | 2011-12-28 | Cadila Healthcare Limited | Polymorphic form of 7-ethyl-10[4-(1-piperidino)-1- piperidino]carbonyloxy- camptothecin and its use |
| WO2012032531A1 (en) * | 2010-09-06 | 2012-03-15 | Avra Laboratories Pvt. Ltd. | Process for the manufacture of irinotecan hydrochloride by total synthesis |
Also Published As
| Publication number | Publication date |
|---|---|
| RS91304A (en) | 2007-02-05 |
| ZA200407821B (en) | 2005-06-29 |
| BR0309284A (en) | 2005-02-22 |
| KR20050009293A (en) | 2005-01-24 |
| JP2005523324A (en) | 2005-08-04 |
| US6723729B2 (en) | 2004-04-20 |
| EP1501799A1 (en) | 2005-02-02 |
| JP3839813B2 (en) | 2006-11-01 |
| MXPA04009426A (en) | 2005-01-25 |
| US20040048832A1 (en) | 2004-03-11 |
| IL164311A0 (en) | 2005-12-18 |
| RU2287527C2 (en) | 2006-11-20 |
| CA2482712A1 (en) | 2003-10-30 |
| CN1646491A (en) | 2005-07-27 |
| PL371681A1 (en) | 2005-06-27 |
| AU2003224973A1 (en) | 2003-11-03 |
| RU2004130493A (en) | 2005-04-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Nesterova et al. | Synthesis and study the pharmacological activity of derivatives of 5-dimethylaminopyrano [3, 2-c] quinolin-2-ones | |
| US6723729B2 (en) | Compounds useful in preparing camptothecin derivatives | |
| WO2014135096A1 (en) | Ivacaftor preparation method and intermediate thereof | |
| CN115215921A (en) | A kind of preparation method of linker drug conjugate and intermediate thereof | |
| JPS63310882A (en) | Polyoxygenated labdane derivative and manufacture | |
| JP4094710B2 (en) | New camptothecin derivatives | |
| JPH0635458B2 (en) | Pyridonecarboxylic acid derivatives, their esters and their salts | |
| US20050197355A1 (en) | Compounds useful in preparing camptothecin derivatives | |
| JP2001064282A (en) | Imidazoline compound, its intermediate and production thereof and production of azepine compound and its salt | |
| JPH0536433B2 (en) | ||
| JPWO1995021163A1 (en) | Pyridonecarboxylic acid derivatives substituted with bicyclic amino groups, esters thereof, salts thereof, and bicyclic amines which are intermediates therefor | |
| EP0663914A1 (en) | Process for the preparation of 9-amino camptothecin | |
| CN113336703A (en) | Synthesis of 1,3,4, 5-tetrasubstituted 1H-pyrazole derivatives | |
| JPH02289563A (en) | Improved process for producing ortho-carboxypyridyl- and ortho-carboxyquinolylimidazolinones | |
| JP2886586B2 (en) | Novel guanidinobenzoic acid derivatives and their acid addition salts | |
| US9505792B2 (en) | Forms of cidofovir | |
| CN116867789B (en) | Preparation method of irinotecan derivative and intermediate thereof | |
| HK1075662A (en) | Compounds useful in preparing camptothecin derivatives | |
| WO2010068049A2 (en) | Process for preparing (r)-(+)-lansoprazole and intermediate used therein | |
| US6426417B1 (en) | Processes and intermediates useful to make antifolates | |
| JPH0522712B2 (en) | ||
| JPH09301975A (en) | Pyrroloquinoline derivative | |
| JP2006528671A (en) | Synthesis of substituted heterocyclic compounds. | |
| HK1023344A1 (en) | Pharmaceutically active tricyclic amines | |
| HK1023344B (en) | Pharmaceutically active tricyclic amines |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: P-913/04 Country of ref document: YU |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PH PL PT RO RU SC SD SE SG SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2003224973 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2887/DELNP/2004 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004/07821 Country of ref document: ZA Ref document number: PA/a/2004/009426 Country of ref document: MX Ref document number: 200407821 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003721671 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20038083353 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2482712 Country of ref document: CA Ref document number: 1020047016524 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003586134 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 2004130493 Country of ref document: RU Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020047016524 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003721671 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2003721671 Country of ref document: EP |