WO2003095462A2 - Platinum complexes containing chemically modified bile acids, having antitumor activity - Google Patents
Platinum complexes containing chemically modified bile acids, having antitumor activity Download PDFInfo
- Publication number
- WO2003095462A2 WO2003095462A2 PCT/EP2003/004829 EP0304829W WO03095462A2 WO 2003095462 A2 WO2003095462 A2 WO 2003095462A2 EP 0304829 W EP0304829 W EP 0304829W WO 03095462 A2 WO03095462 A2 WO 03095462A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- acid
- antitumor activity
- platinum
- platinum complexes
- bile acids
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CNC(*)(C(C(O)=O)O)O Chemical compound CNC(*)(C(C(O)=O)O)O 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J51/00—Normal steroids with unmodified cyclopenta(a)hydrophenanthrene skeleton not provided for in groups C07J1/00 - C07J43/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- the present invention relates to platinum (II) complexes having antitumor activity, in particular platinum (II) complexes in which the ligands are represented by ammonia and a ketal at the 3- position with a dihydroxy bicarboxylic acid.
- the present invention relates to platinum (II) complexes of formula (1): in which n is 0 or 1 and A is C or the CH-CH group.
- Preferred embodiments of the invention are the complexes in which: n is 0 and A is the CH-CH group (compound la); n is 1 and A is C (compound lb).
- the salt (2) is prepared through a synthesis which comprises the following steps:
- a mixture of dehydrocholic acid (3) and tartaric acid (4) is esterified by heating under reflux in a low molecular weight ROH alcohol selected from methanol, ethanol, propanol, isopropanol, preferably ethanol, in the presence of a sulfonic acid R ⁇ OsH, preferably methanesulfonic acid
- the ester mixture from the previous step is saponified by treatment with an alkali hydroxide BOH, wherein B is sodium or potassium, preferably sodium, in a water - alcohol mixture, in which the alcohol is a low molecular weight ROH alcohol selected from methanol, ethanol, propanol, isopropanol, preferably methanol, in which the alcohol - water ratio ranges from 3 / 1 to 1 / 1, preferably 2 / 1, according to Scheme 3.
- BOH alkali hydroxide
- B is sodium or potassium, preferably sodium
- a water - alcohol mixture in which the alcohol is a low molecular weight ROH alcohol selected from methanol, ethanol, propanol, isopropanol, preferably methanol, in which the alcohol - water ratio ranges from 3 / 1 to 1 / 1, preferably 2 / 1, according to Scheme 3.
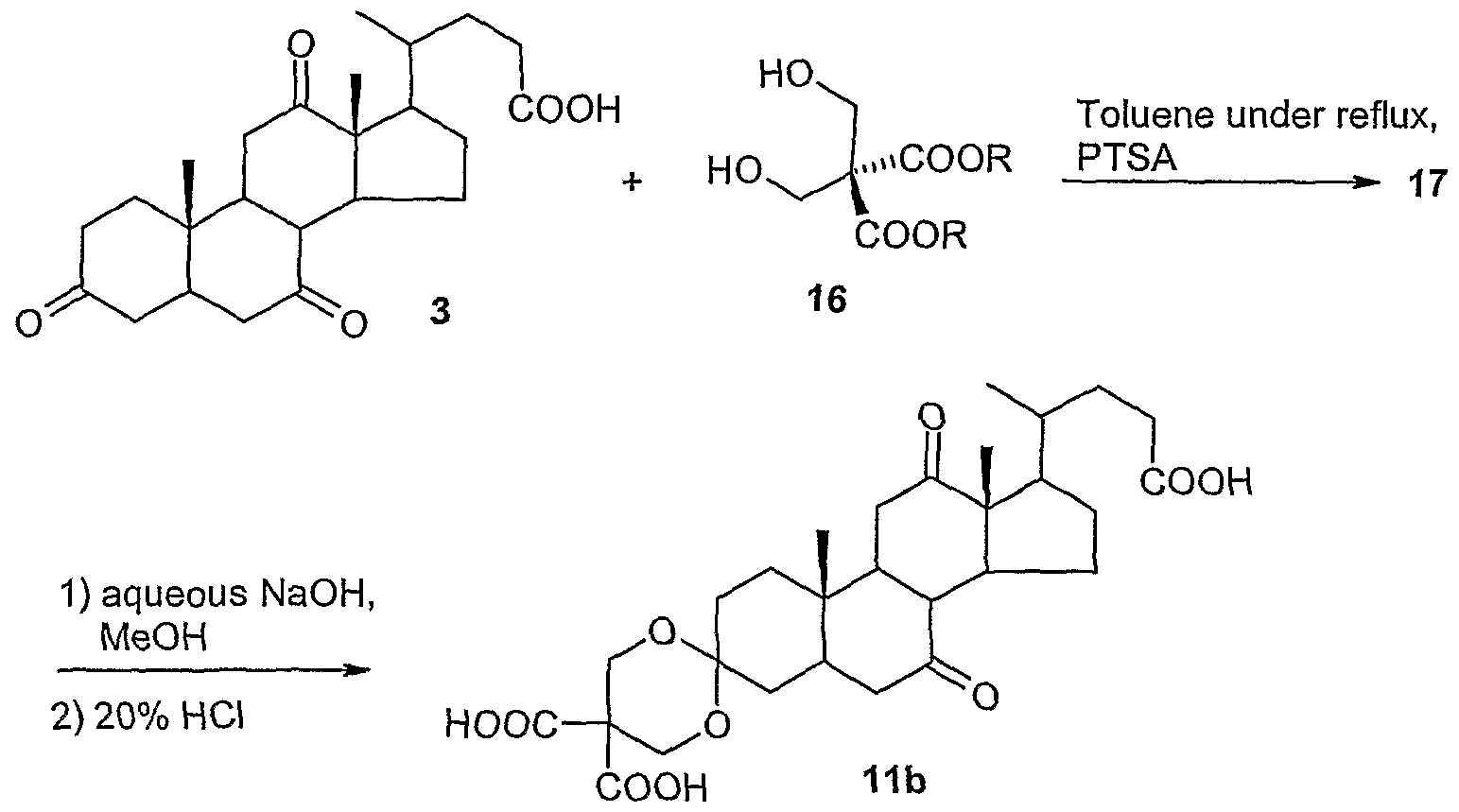
- Dehydrocholic acid (3) can be obtained by adjusting pH to 5 with an acid selected from, for example, hydrochloric acid, sulfuric acid, methanesulfonic acid and paratoluenesulfonic acid, preferably 20% sulfuric acid, followed by extraction with an organic solvent, preferably ethyl acetate; pH is then further lowered to to approx. 3 to extract the triacid (11a), whereas adjusting pH to values below 2 provides a mixture consisting of diketals (12) and (13).
- an acid selected from, for example, hydrochloric acid, sulfuric acid, methanesulfonic acid and paratoluenesulfonic acid, preferably 20% sulfuric acid, followed by extraction with an organic solvent, preferably ethyl acetate; pH is then further lowered to to approx. 3 to extract the triacid (11a), whereas adjusting pH to values below 2 provides a mixture consisting of diketals (12) and (13).
- aquocomplex (14) is prepared from cis-platinum according to what described in literature.
- Dehydrocholic acid is reacted with a bis(hydroxymethyl) malonic acid ester (16), in which R has the meanings defined above, in toluene under reflux in the presence of a sulfonic acid, preferably para-toluenesulfonic acid, to give the corresponding ketal of formula (17).
- a sulfonic acid preferably para-toluenesulfonic acid
- Hydrolysis with sodium hydroxide in methanol and subsequent treatment with 20% hydrochloric acid yield the triacid of formula (lib).
- Compounds (1) when administered to humans or animals bearing tumors which can be treated with cisplatin or are resistant to cisplatin, are capable of inducing the regression of said tumors.
- Compounds (1) can be used for the treatment of those pathological conditions for which cisplatin is used, in particular for the treatment of tumors [Douple et al. Cisplatin Current Status and Developments, Ed. A.W. Prestayk et al., Academic Press, 125 (1980; Douple et al. Platinum Metals Res., 29; 118
- the present invention also relates to pharmaceutical compositions containing a therapeutically effective amount of a compound of formula (1) in mixture with conventional carriers and excipients.
- the effective dose of compounds (1) will be determined by the expert physician according to conventional methods. The relationship between the dosages used for animals of various species and size and those for the humans (based on mg/m 2 body area) is described by Freirech et al., Quantitative Comparison of Toxicity of Anticancer Agents in Mouse, Rat, Hamster, Dog, Monkey and Man Cancer Chemother. Rep, 50, n. 4, 219-244 (1986).
- the patient will receive doses of the complex ranging from 0.1 to 200 mg/kg body weight, with a dosage regimen which will vary depending on a number of factors, well known to the skilled clinician, and the treatment regimen will vary depending on the type of tumor to treat and the conditions of the patient.
- Compounds (1) can be administered through the oral, parenteral, topical or intratumoral routes.
- compositions for the parenteral administration comprise saline sterile solutions or suspensions or sterile powders for the extemporaneous preparation of solutions or suspensions.
- the pharmaceutical compositions for the parenteral administration also comprise oily preparations for the intramuscular or intraperitoneal administration.
- compositions for the oral administration comprise, for example, syrups or similar liquid forms, as well as solid forms such as tablets, capsules and the like.
- compositions according to the present invention are prepared following conventional methods, such as those reported in Remington's Pharmaceutical Sciences Handbook, XVII Ed., Mack Pub., N.Y., U.S.A.
- Compounds (1) may be administered together with one or more agents which enhance their antitumor activity or which alleviate the side effects accompanying platinum complexes therapy, for example together with reduced glutathione, as disclosed in GB 2.174.905 and in US 4.871.528.
- Complexes (1) can also be advantageously administered together with other platinum complexes having antitumor activity, therefore a further object of the present invention are pharmaceutical compositions containing compounds (1) in combination with a platinum complex with antitumor activity.
- the present invention further relates to the use of compounds (1) for the preparation of pharmaceutical compositions for the treatment of mammals affected with tumors which can be treated with cis-platinum or are resistant to cis-platinum.
- the invention is illustrated in greater detail by the following examples.
- EXAMPLE 1 SYNTHESIS OF CHOLIC ACID AND L(+)TARTARIC ACID 3-KETAL
- the white solid from the previous step is taken up into 150 ml of toluene, added with 0.4 ml (0.1 mols) of methanesulfonic acid, then refluxed in a Dean Stark apparatus for 10 hours.
- the aqueous phase is added with a further 100 ml of ethyl acetate and adjusted to pH 2.5 - 3 with 20% H 2 S ⁇ 4 ; the triacid precipitates and is immediately extracted in the organic phase. The latter is separated and dried; in a short time, crystallization of the triacid (usually as pale brown crystals) is observed.
- aqueous phase is treated with 20% with H S0 4 to pH 1 to obtain a precipitate mainly consisting of dehydrocholic acid 3,7-diketal and 3,12- diketal.
- Preparation Id Crystallization of triacid in ethyl acetate 20 g of the crude acid from the first crystallization are dissolved in 200 ml of ethyl acetate and 15 ml of water, in a 250 ml round-bottom flask. The resulting brown solution contains a residue of the same color, which is filtered off.
- the hot solution of the triacid disodium salt is added with the aquocomplex solution; a white-yellow flaky precipitate forms.
- the reaction mixture is kept under stirring in a bath thermostatized at 60°C for 10 hours.
- the precipitate is filtered with suction and left to dry in the air, or in a static dryer at a temperature of 100°C.
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Steroid Compounds (AREA)
Abstract
Description
Claims
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2003233308A AU2003233308B2 (en) | 2002-05-10 | 2003-05-08 | Platinum complexes containing chemically modified bile acids, having antitumor activity |
| SI200330401T SI1513863T1 (en) | 2002-05-10 | 2003-05-08 | Platinum complexes containing chemically modified bile acids, having antitumor activity |
| NZ537884A NZ537884A (en) | 2002-05-10 | 2003-05-08 | Platinum complexes containing chemically modified bile acids, having antitumor activity |
| CA002493704A CA2493704C (en) | 2002-05-10 | 2003-05-08 | Platinum complexes containing chemically modified bile acids, having antitumor activity |
| EA200500111A EA008221B1 (en) | 2002-05-10 | 2003-05-08 | Platinum complexes containing chemically modified bile acids, having antitumor activity |
| DE60307103T DE60307103T2 (en) | 2002-05-10 | 2003-05-08 | PLATINUM COMPLEXES CONTAINING CHEMICALLY MODIFIED GALLEN ACID WITH ANTITUMORACTIVITY |
| EP03727454A EP1513863B1 (en) | 2002-05-10 | 2003-05-08 | Platinum complexes containing chemically modified bile acids, having antitumor activity |
| HK05103675.1A HK1071144B (en) | 2002-05-10 | 2003-05-08 | Platinum complexes containing chemically modified bile acids, having antitumor activity |
| UAA200500951A UA78581C2 (en) | 2002-05-10 | 2003-08-05 | Platinum complexes containing bile acids derivatives having antitumor activity |
| IL166446A IL166446A (en) | 2002-05-10 | 2005-01-23 | Platinum complexes containing chemicaly modified bile acids having antitumor activity |
| NO20050509A NO20050509L (en) | 2002-05-10 | 2005-01-28 | Platinum complexes containing chemically modified bile acids that have antitumor activity |
| CY20061101096T CY1105123T1 (en) | 2002-05-10 | 2006-08-04 | WHITE GOLD COMPLEXES WHICH COMPOSE CHEMICALLY MODIFIED BILE ACIDS WHICH HAVE ANTI-CANCER ACTIVITY |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ITMI2002A000992 | 2002-05-10 | ||
| IT2002MI000992A ITMI20020992A1 (en) | 2002-05-10 | 2002-05-10 | PLATINUM COMPLEXES CONTAINING CHEMICALLY MODIFIED BILIARY ACIDS AND ANTI-CANCER ACTIVITIES |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2003095462A2 true WO2003095462A2 (en) | 2003-11-20 |
| WO2003095462A3 WO2003095462A3 (en) | 2005-01-13 |
Family
ID=11449868
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2003/004829 Ceased WO2003095462A2 (en) | 2002-05-10 | 2003-05-08 | Platinum complexes containing chemically modified bile acids, having antitumor activity |
Country Status (19)
| Country | Link |
|---|---|
| US (1) | US7348320B2 (en) |
| EP (1) | EP1513863B1 (en) |
| AT (1) | ATE334140T1 (en) |
| AU (1) | AU2003233308B2 (en) |
| CA (1) | CA2493704C (en) |
| CY (1) | CY1105123T1 (en) |
| DE (1) | DE60307103T2 (en) |
| DK (1) | DK1513863T3 (en) |
| EA (1) | EA008221B1 (en) |
| ES (1) | ES2270033T3 (en) |
| IL (1) | IL166446A (en) |
| IT (1) | ITMI20020992A1 (en) |
| NO (1) | NO20050509L (en) |
| NZ (1) | NZ537884A (en) |
| PL (1) | PL374345A1 (en) |
| PT (1) | PT1513863E (en) |
| UA (1) | UA78581C2 (en) |
| WO (1) | WO2003095462A2 (en) |
| ZA (1) | ZA200500727B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3509603B1 (en) * | 2016-09-07 | 2024-01-03 | Alyssum Therapeutics, Inc. | Immune memory induction by platinum based compounds |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105713047B (en) * | 2016-03-25 | 2018-05-08 | 东南大学 | One eka-platinium (II) complex and its preparation method and application |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2562134B2 (en) * | 1986-09-19 | 1996-12-11 | 喜徳 喜谷 | Novel platinum-steroid complex |
-
2002
- 2002-05-10 IT IT2002MI000992A patent/ITMI20020992A1/en unknown
-
2003
- 2003-05-08 DE DE60307103T patent/DE60307103T2/en not_active Expired - Lifetime
- 2003-05-08 NZ NZ537884A patent/NZ537884A/en active Application Filing
- 2003-05-08 AU AU2003233308A patent/AU2003233308B2/en not_active Expired - Fee Related
- 2003-05-08 DK DK03727454T patent/DK1513863T3/en active
- 2003-05-08 PT PT03727454T patent/PT1513863E/en unknown
- 2003-05-08 PL PL03374345A patent/PL374345A1/en unknown
- 2003-05-08 WO PCT/EP2003/004829 patent/WO2003095462A2/en not_active Ceased
- 2003-05-08 EP EP03727454A patent/EP1513863B1/en not_active Expired - Lifetime
- 2003-05-08 AT AT03727454T patent/ATE334140T1/en not_active IP Right Cessation
- 2003-05-08 EA EA200500111A patent/EA008221B1/en not_active IP Right Cessation
- 2003-05-08 ES ES03727454T patent/ES2270033T3/en not_active Expired - Lifetime
- 2003-05-08 CA CA002493704A patent/CA2493704C/en not_active Expired - Fee Related
- 2003-08-05 UA UAA200500951A patent/UA78581C2/en unknown
-
2004
- 2004-10-21 US US10/970,484 patent/US7348320B2/en not_active Expired - Fee Related
-
2005
- 2005-01-23 IL IL166446A patent/IL166446A/en not_active IP Right Cessation
- 2005-01-25 ZA ZA200500727A patent/ZA200500727B/en unknown
- 2005-01-28 NO NO20050509A patent/NO20050509L/en not_active Application Discontinuation
-
2006
- 2006-08-04 CY CY20061101096T patent/CY1105123T1/en unknown
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3509603B1 (en) * | 2016-09-07 | 2024-01-03 | Alyssum Therapeutics, Inc. | Immune memory induction by platinum based compounds |
Also Published As
| Publication number | Publication date |
|---|---|
| US20050113352A1 (en) | 2005-05-26 |
| EP1513863A2 (en) | 2005-03-16 |
| UA78581C2 (en) | 2007-04-10 |
| PT1513863E (en) | 2006-12-29 |
| EA008221B1 (en) | 2007-04-27 |
| EP1513863B1 (en) | 2006-07-26 |
| EA200500111A1 (en) | 2005-08-25 |
| HK1071144A1 (en) | 2005-07-08 |
| ES2270033T3 (en) | 2007-04-01 |
| ITMI20020992A1 (en) | 2003-11-10 |
| CY1105123T1 (en) | 2010-03-03 |
| PL374345A1 (en) | 2005-10-17 |
| WO2003095462A3 (en) | 2005-01-13 |
| AU2003233308B2 (en) | 2008-10-16 |
| ATE334140T1 (en) | 2006-08-15 |
| IL166446A (en) | 2008-04-13 |
| AU2003233308A1 (en) | 2003-11-11 |
| ITMI20020992A0 (en) | 2002-05-10 |
| ZA200500727B (en) | 2006-07-26 |
| DE60307103T2 (en) | 2006-11-23 |
| US7348320B2 (en) | 2008-03-25 |
| IL166446A0 (en) | 2006-01-15 |
| NZ537884A (en) | 2006-04-28 |
| DK1513863T3 (en) | 2006-11-13 |
| CA2493704C (en) | 2008-11-18 |
| DE60307103D1 (en) | 2006-09-07 |
| NO20050509L (en) | 2005-01-28 |
| CA2493704A1 (en) | 2003-11-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111171080B (en) | High-efficiency low-toxicity anticancer compound synthesized by autocatalysis in cells and living bodies and synthesis method thereof | |
| WO2008038965A1 (en) | 7alpha-aminosteroid derivatives or pharmaceutically acceptable salts thereof, preparation method thereof and composition for anticancer or antibiotics containing the same as an active ingredient | |
| EP0166366A2 (en) | Novel platinum complexes | |
| US6057303A (en) | Highly lipophilic Camptothecin derivatives | |
| EP1513863B1 (en) | Platinum complexes containing chemically modified bile acids, having antitumor activity | |
| CN101845075B (en) | 6-substituted-4-azepine-A-homo-3-oxosteroid compound and application thereof to preparing anti-tumor medicine | |
| JPWO1996026949A1 (en) | Novel platinum (IV) complex, its production method and anticancer drug containing the same | |
| CN101654456B (en) | Water-soluble derivants containing 10-hydroxycamptothecine and preparation method thereof | |
| HK1071144B (en) | Platinum complexes containing chemically modified bile acids, having antitumor activity | |
| US7205287B2 (en) | Platinum complexes having antitumor activity | |
| EP1896492B1 (en) | Platinum complexes with mononitrile-containing ligands | |
| CN113651868B (en) | Cisplatin-derived tetravalent platinum anti-cancer complex containing ursolic acid ligand as well as preparation method and application thereof | |
| CN112824425B (en) | A kind of 6-position alkenyl substituted cholic acid compound and its preparation method and application | |
| CN110759961B (en) | A class of ursolic acid indolequinone amide derivatives and their preparation method and application | |
| CN114907437B (en) | Androstanol derivative with anti-tumor activity and preparation method and application thereof | |
| CA1274529A (en) | Platinum(ii) complexes of 1,1-cyclobutane- dicarboxylate and process for their production | |
| AU2005318227A1 (en) | Stereoselective process and crystalline forms of a camptothecin | |
| CN118515719A (en) | Cordycepin-cisplatin coupling derivatives and preparation methods thereof and applications in the preparation of anti-tumor drugs | |
| CN112940069A (en) | 5 alpha-pregna-18, 20 beta-oxidized-3 alpha-alcohol and preparation method thereof | |
| JPH0753745B2 (en) | Cis-platinum (▲ II ▼) complex compound having phospholipid derivative | |
| KR20050020794A (en) | Platinum complexes having antitumor activity |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PH PL PT RO RU SC SD SE SG SK SL TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2003727454 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2493704 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005/00727 Country of ref document: ZA Ref document number: 537884 Country of ref document: NZ Ref document number: 200500727 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200500111 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003233308 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 374345 Country of ref document: PL |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003727454 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 537884 Country of ref document: NZ |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: JP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2003727454 Country of ref document: EP |