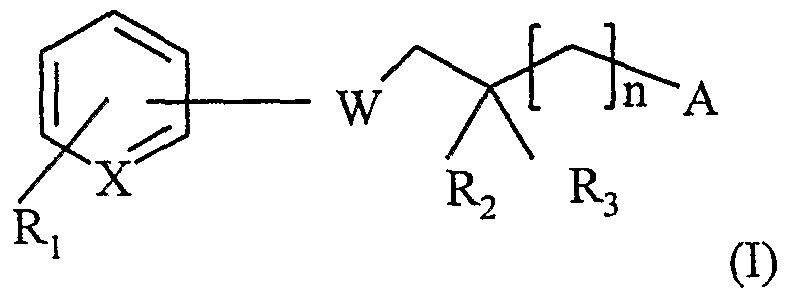WO2003106425A2 - Phenyl- et pyridyl-diazaheterocycles ayant une activite modulatrice du tnf - Google Patents
Phenyl- et pyridyl-diazaheterocycles ayant une activite modulatrice du tnf Download PDFInfo
- Publication number
- WO2003106425A2 WO2003106425A2 PCT/FR2003/001813 FR0301813W WO03106425A2 WO 2003106425 A2 WO2003106425 A2 WO 2003106425A2 FR 0301813 W FR0301813 W FR 0301813W WO 03106425 A2 WO03106425 A2 WO 03106425A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- group
- solvates
- compound according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/04—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to the ring carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/06—Antigout agents, e.g. antihyperuricemic or uricosuric agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
- A61P31/06—Antibacterial agents for tuberculosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/02—Antiprotozoals, e.g. for leishmaniasis, trichomoniasis, toxoplasmosis
- A61P33/06—Antimalarials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/02—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with only hydrogen atoms or radicals containing only carbon and hydrogen atoms, directly attached to carbon atoms of the nitrogen-containing ring; Alkylene-bis-isoquinolines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/02—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings
- C07D241/06—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having one or two double bonds between ring members or between ring members and non-ring members
- C07D241/08—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having one or two double bonds between ring members or between ring members and non-ring members with oxygen atoms directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
Definitions
- the present invention relates to new phenyl- and pyridyldiazaheterocycles having TNF modulating activity, the pharmaceutical compositions containing them and a process for their preparation.
- US3, 188,313 describes piperazines substituted by an indolyl-alkyl radical showing activity on the central nervous system, on the cardiovascular system and on the muscular and bone systems.
- WO01 / 29026 describes certain tetrahydro-pyridines, substituted by a quinolinyl-alkyl or isoquinolyl-alkyl radical with activity on the modulation of TNF-alpha (from the English Tumor Necrosis Factor).
- TNF-alpha is a cytokine which has recently aroused interest as a mediator of immunity, inflammation, cell proliferation, fibrosis etc. This mediator is abundantly present in inflamed synovial tissue and plays an important role in the pathogenesis of autoimmunity
- diazaheterocycles carrying a quinolinyl-alkyl or isoquinolyl-alkyl radical have a potent activity with respect to the modulation of TNF-alpha.
- the present invention relates, according to one of its aspects, to diazaheterocycles of formula (I):
- X represents N or CH
- Ri represents a hydrogen or halogen atom or a group CF 3 ;
- R 2 and R 3 independently represent a hydrogen atom or a methyl group; n is 0 or 1;
- W represents a diazoheterocycle of formula (a) to (d)
- (e) (f) or 4 represents a hydrogen or halogen atom, a (C ⁇ -C) alkyl group, a CF 3 group, an ino group, mono (C ⁇ - C 4 ) alkylamino, di (C ⁇ -C) alkylamino;
- R 5 represents a hydrogen or halogen atom, a group
- (C ⁇ -C) alkyl denotes a monovalent radical of a saturated C ⁇ -C 4 straight chain or branched hydrocarbon.
- halogen denotes an atom chosen from chlorine, bromine, iodine and fluorine.
- the quinoline and isoquinoline rings can be attached to the rest of the molecule of formula (I) by any of the carbon atoms at positions 6 or 7.
- Des Preferred compounds of formula (I) are those where n is zero.
- Other preferred compounds are those where R 2 and R 3 are each a hydrogen atom.
- Ri is a group CF 3 .
- Other preferred compounds are those where Ri is a fluorine or chlorine atom.
- the compounds of formula (I) can exist as N-oxide derivatives.
- the compounds of formula (I) can in particular carry one or two N-oxide groups on the diazoheterocycles (a) to (d) and / or an N-oxide group on the quinoline or the isoquinoline of the group A.
- the above three nitrogen can all be oxidized, compounds bearing one or two N-oxide groups, one on the diazoheterocycle and the other on quinoline or isoquinoline, are preferred.
- the salts of the compounds of formula (I) according to the present invention also include the addition salts with pharmaceutically acceptable inorganic or organic acids such as the hydrochloride, the hydrobromide, the sulfate, the hydrogen sulfate, the dihydrogen phosphate, the citrate, maleate, tartrate, fumarate, gluconate, methanesulfonate, 2-naphthalenesulfonate, etc., than addition salts which allow proper separation or crystallization of the compounds of formula (I), such as picrate, oxalate or addition salts with optically active acids, for example camphosulfonic acids and mandelic or substituted mandelic acids.
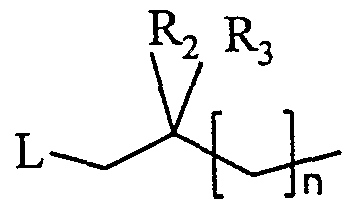
- the compounds of formula (I) can be synthesized by a process which provides for a condensation / reduction reaction starting from a compound of formula (II): in which X, W and Ri are as defined above, with an aldehyde of formula (III):
- the condensation reaction is normally carried out by mixing the starting compounds (II) and (IV) in an inert organic solvent, according to conventional methods.
- inert organic solvent a solvent which does not interfere with the reaction.
- solvents are, for example, alcohols such as methanol, ethanol, isopropanol or butanol.
- leaving group L it is possible, for example, to use a chlorine or bromine atom or else a mesyloxy group (CH 3 -SO -O-).
- the reaction is carried out at a temperature between -10 ° C. and the reflux temperature of the reaction mixture, the reflux temperature being preferred.
- the reaction can be suitably carried out in the presence of a proton acceptor, for example an alkali carbonate or a tertiary amine such as triethylamine.
- a proton acceptor for example an alkali carbonate or a tertiary amine such as triethylamine.
- the reaction is normally completed after a few hours, normally 1 to 6 hours are sufficient to complete the condensation.
- the desired compound of formula (I) is isolated according to conventional techniques in the form of the free base or one of its salts.
- the free base can be transformed into one of its salts by simple salification in an organic solvent such as an alcohol, preferably ethanol or isopropanol, an ether such as 1,2-dimethoxyethane, ethyl acetate , acetone or a hydrocarbon such as hexane.
- the starting compounds of formula (II) containing a diazotized nucleus (a) or (d) are known or they can be prepared in a similar manner to the known compounds.
- the starting compounds of formula (II) in which the diazotized nucleus is (b) or (c) and X is N are also known or else they can be prepared in a similar manner to the known compounds, as described for example in J. Med . Chem., 1998, 41, 674-681.
- X and CH can be prepared by reaction of an optionally substituted bromo-benzene with the nucleus (b) or (c), the nitrogen not having to take part in the reaction being suitably protected beforehand. Examples of such a reaction are given in the experimental part.
- W represents a group of formula (b) or (c) above are new and represent another aspect of the present invention.
- the compounds of formulas (III) and (IV) are known and can be prepared in a similar manner to the known compounds, for example as described in WO01 / 29026.
- the compounds of formula (I) carrying an N-oxide group on the nitrogen atom of the quinoline or of the isoquinoline can be prepared from the N-oxide derivatives of the compounds of formula (III).
- the compounds of formula (I) carrying an N-oxide group on the nitrogen atoms of the rings (a) to (d) can be prepared by oxidation of the corresponding compounds of formula (I).
- the compound of formula (I) as obtained by the above syntheses is subjected to an oxidation reaction according to conventional methods, for example to a reaction with m-chloro-perbenzoic acid in a suitable solvent and isolated according to the usual techniques well known to those skilled in the art.
- the compounds of the invention have advantageous properties with respect to the inhibition of TNF- ⁇ .
- LPS lipopolysaccharide
- test products are administered orally to groups of 5 female Balb / c mice (Charles River, France) aged 7 to 8 weeks.
- the LPS is administered intravenously (10 ⁇ g / mouse).
- the blood of each animal is taken 1.5 hours after administration of the LPS.
- the samples are centrifuged, the plasma is recovered and frozen at -80 ° C.
- TNF- ⁇ is measured using commercial kits (R&D, Abingdon, UK).
- the compounds of. formula (I) and its salts or solvates may well be used in the treatment of diseases linked to immune and inflammatory disorders or as analgesics.
- the compounds of formula (I) can be used to treat atherosclerosis, autoimmune diseases, diseases which cause demyelination of neurons (such as multiple sclerosis), asthma, rheumatoid arthritis, fibrotic diseases , idiopathic pulmonary fibrosis, cystic fibrosis, glumerulonephritis, rheumatoid spondylitis, osteoarthritis, gout, bone and cartilage resorption, osteoporosis, Paget's disease, multiple myioma, uveoretinitis, septic shock, sepsis, endotoxin shock, graft versus host reaction, transplant rejection, adult respiratory distress syndrome, silicosis, asbestosis, pulmonary sarcoidosis, Crohn's disease
- the compounds of formula (I) and their pharmaceutically acceptable salts and solvates are preferably administered orally.
- the active principle can be administered in unit administration forms, in admixture with conventional pharmaceutical carriers, to animals and humans for the treatment of the abovementioned conditions.
- Suitable unit dosage forms include, for example, possibly scored tablets, capsules, powders, granules and oral solutions or suspensions.
- the main active ingredient is mixed with a pharmaceutical carrier such as gelatin, starch, lactose, magnesium stearate, talc, gum arabic or the like.
- the tablets can be coated with sucrose or other suitable materials or else they can be treated in such a way that they have a prolonged or delayed activity and that they continuously release a predetermined quantity of active principle.
- a preparation in capsules is obtained by mixing the active ingredient with a diluent and by pouring the mixture obtained into soft or hard capsules.
- a preparation in the form of a syrup or elixir may contain the active ingredient together with a sweetener, preferably calorie-free, methylparaben and propylparaben as antiseptics, as well as a flavoring agent and an appropriate color.
- Water dispersible powders or granules may contain the active ingredient in admixture with dispersing agents or wetting agents, or suspending agents, such as polyvinylpyrrolidone, as well as with sweeteners or correctors taste.
- the active principle can also be formulated in the form of microcapsules, optionally with one or more carriers or additives.
- the active principle can also be in the form of an inclusion complex in cyclodextrins, their ethers or their esters.
- the amount of active ingredient to be administered depends as always on the degree of progression of the disease as well as on the age and weight of the patient.
- the unit doses generally comprise from 0.001 to 100 mg, better still from 0.01 to 50 mg, preferably from 0.1 to 20 mg of active principle, advantageously from 0.5 to 10 mg.
- the present invention relates to a combination comprising a compound of formula (I) or one of its pharmaceutically acceptable salts or solvates, and at least one compound chosen from immunosuppressive agents, such as interferon beta -lb; adrenocorticotropic hormone; glucocorticoids such as prednisone or methylprednisolone; interleukin-1 inhibitors.
- immunosuppressive agents such as interferon beta -lb; adrenocorticotropic hormone; glucocorticoids such as prednisone or methylprednisolone; interleukin-1 inhibitors.
- the compounds of the invention can be combined with a compound chosen from roquinimex (1,2-dihydro-4-hydroxy-N, l-dimethyl l-2-oxo-3 -quinolinecarboxanilide), myloran (product from the company Autoimmune containing bovine myelin), the antegren (human monoclonal antibody from the companies Elan / Athena Neurosciences) the interferon beta-lb recombinant.
- roquinimex 1,2-dihydro-4-hydroxy-N, l-dimethyl l-2-oxo-3 -quinolinecarboxanilide
- myloran product from the company Autoimmune containing bovine myelin
- the antegren human monoclonal antibody from the companies Elan / Athena Neurosciences
- the interferon beta-lb recombinant the interferon beta-lb recombinant.
- the invention relates to a method of treatment of diseases linked to immune and inflammatory disorders.
- autoimmune diseases diseases that cause demyelination of neurons (such as multiple sclerosis), asthma, rheumatoid arthritis, fibrotic diseases, fibrosis idiopathic pulmonary, cystic fibrosis, glumerulonephritis, rheumatoid spondylitis, osteoarthritis, gout, bone and cartilage resorption, osteoporosis, Paget's disease, multiple myeloma, uveoretinitis, septic shock, septicemia, endotoxin shock, graft versus host reaction, transplant rejection, adult respiratory distress syndrome, silicosis, asbestosis, pulmonary sarcoidosis, Crohn's disease, ulcerative colitis, lateral sclerosis amyotrophic, Alzheimer's disease, Parkinson's disease, systemic lupus erythematosus, hemodynamic shock, ischemic pathologies (myocardi
- the compounds of Examples 3 to 9 are prepared by following the procedures described in Example 1.
- the compounds of Examples 10 to 13 are prepared by following the procedures described in Example 2.
Landscapes
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- General Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Physical Education & Sports Medicine (AREA)
- Rheumatology (AREA)
- Immunology (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Pulmonology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Pain & Pain Management (AREA)
- Tropical Medicine & Parasitology (AREA)
- Cardiology (AREA)
- Urology & Nephrology (AREA)
- Heart & Thoracic Surgery (AREA)
- Virology (AREA)
- Psychiatry (AREA)
- Psychology (AREA)
- Hospice & Palliative Care (AREA)
- Vascular Medicine (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Other In-Based Heterocyclic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/518,652 US20050209216A1 (en) | 2002-06-18 | 2003-06-16 | Phenyl-and pyridyl-diazaheterocycles having a tnf-modulating activity |
| EP03760042A EP1517896A2 (fr) | 2002-06-18 | 2003-06-16 | Phenyl- et pyridyl-diazaheterocycles ayant une activite modulatrice du tnf |
| JP2004513258A JP2005533790A (ja) | 2002-06-18 | 2003-06-16 | Tnf調整作用を有するフェニル−およびピリジル−ジアザ複素環 |
| AU2003263237A AU2003263237A1 (en) | 2002-06-18 | 2003-06-16 | Phenyl- and pyridyl-diazaheterocycles having a tnf-modulating activity |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0207507A FR2840896B1 (fr) | 2002-06-18 | 2002-06-18 | Phenyl-et pyridyl-piperazines, procede pour leur preparation et compositions pharmaceutiques les contenant |
| FR02/07507 | 2002-06-18 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2003106425A2 true WO2003106425A2 (fr) | 2003-12-24 |
| WO2003106425A3 WO2003106425A3 (fr) | 2004-04-01 |
Family
ID=29595360
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2003/001813 Ceased WO2003106425A2 (fr) | 2002-06-18 | 2003-06-16 | Phenyl- et pyridyl-diazaheterocycles ayant une activite modulatrice du tnf |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20050209216A1 (fr) |
| EP (1) | EP1517896A2 (fr) |
| JP (1) | JP2005533790A (fr) |
| AU (1) | AU2003263237A1 (fr) |
| FR (1) | FR2840896B1 (fr) |
| WO (1) | WO2003106425A2 (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1892238A4 (fr) * | 2005-06-14 | 2011-03-09 | Eisai R&D Man Co Ltd | Derive du benzene 1, 2-di(groupe cyclique)substitue |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB944443A (fr) * | 1959-09-25 | 1900-01-01 | ||
| US4544657A (en) * | 1983-05-19 | 1985-10-01 | Hoffmann-La Roche Inc. | Substituted isoquinolines |
| FR2736053B1 (fr) * | 1995-06-28 | 1997-09-19 | Sanofi Sa | Nouvelles 1-phenylalkyl-1,2,3,6-tetrahydropyridines |
| HK1046532B (zh) * | 1999-10-22 | 2005-03-18 | Sanofi-Aventis | 具有抑制tnf活性的苯基-和吡啶基-四氫吡啶 |
| FR2804430B1 (fr) * | 2000-01-28 | 2002-03-22 | Sanofi Synthelabo | Derives de 4-heteroaryl-1,4-diazabicyclo[3.2.2] nonane, leur preparation et leur application en therapeutique |
-
2002
- 2002-06-18 FR FR0207507A patent/FR2840896B1/fr not_active Expired - Fee Related
-
2003
- 2003-06-16 JP JP2004513258A patent/JP2005533790A/ja not_active Withdrawn
- 2003-06-16 US US10/518,652 patent/US20050209216A1/en not_active Abandoned
- 2003-06-16 EP EP03760042A patent/EP1517896A2/fr not_active Withdrawn
- 2003-06-16 WO PCT/FR2003/001813 patent/WO2003106425A2/fr not_active Ceased
- 2003-06-16 AU AU2003263237A patent/AU2003263237A1/en not_active Abandoned
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1892238A4 (fr) * | 2005-06-14 | 2011-03-09 | Eisai R&D Man Co Ltd | Derive du benzene 1, 2-di(groupe cyclique)substitue |
Also Published As
| Publication number | Publication date |
|---|---|
| FR2840896B1 (fr) | 2005-04-08 |
| WO2003106425A3 (fr) | 2004-04-01 |
| FR2840896A1 (fr) | 2003-12-19 |
| EP1517896A2 (fr) | 2005-03-30 |
| AU2003263237A8 (en) | 2003-12-31 |
| US20050209216A1 (en) | 2005-09-22 |
| JP2005533790A (ja) | 2005-11-10 |
| AU2003263237A1 (en) | 2003-12-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2004099176A1 (fr) | Derives de piperidinyl- et piperazinyl-alkylcarbamates, leurs procedes de preparation et leur application en therapeutique | |
| EP1720872A1 (fr) | Derives d'aryl- et d'heteroaryl-piperidinecarboxylates, leur preparation et leur application comme inhibiteurs de l'enzyme faah | |
| EP1708994B1 (fr) | Derives de type aryloxyalkylcarbamates, leur preparation et leur application en therapeutique | |
| EP1248785B1 (fr) | Tetrahydropyridines, procede pour leur preparation et compositions pharmaceutiques les contenant | |
| EP1214313B1 (fr) | Heteroaryloxypropanolamines comme agonistes du recepteur beta3-adrenergique | |
| EP1438291B1 (fr) | Aralkyl-tetrahydro-pyridines, leur preparation et compositions pharmaceutiques les contenant | |
| EP1226137B8 (fr) | Phenyl- et pyridyl-tetrahydro-pyridines possedant une activite inhibitrice de fnt | |
| EP1383762B1 (fr) | Tetrahydropyridyl-alkyl-heterocycles, procede pour leur preparation et compositions pharmaceutiques les contenants | |
| EP1383761B1 (fr) | Phenyl- et pyridyl-pipéridines avec une activité du tnf | |
| EP1517896A2 (fr) | Phenyl- et pyridyl-diazaheterocycles ayant une activite modulatrice du tnf | |
| EP0950049B1 (fr) | Diphenylalkyl-tetrahydropyridines, procede pour leur preparation et composition pharmaceutique les contenant | |
| EP1448550B1 (fr) | Tetrahydropyridyl-alkyl-heterocycles azotes avec une activite du tnf | |
| WO2000063170A1 (fr) | Derives de cyclobutene-3,4-dione en tant qu'inhibiteurs de la phosphodiesterase 5 | |
| EP0266246A1 (fr) | Dérivés d'imidazo[4,5-b]pyridinone-2, leur préparation et leur application en thérapeutique | |
| FR2810984A1 (fr) | Pyridyl-tetrahydropyridines, procede pour leur preparation et compositions pharmaceutiques les contenant | |
| FR2800071A1 (fr) | Tetrahydropyridines, procede pour leur preparation et compositions pharmaceutiques les contenant | |
| FR2823748A1 (fr) | Tetrahydropyridyl-alkyl-benzodiazines, procede pour leur preparation et compositions pharmaceutiques les contenant | |
| WO1998003485A1 (fr) | PHENOXYPROPANOLAMINES A ACTION ANTAGONISTE β3-ADRENERGIQUE | |
| EP0695747A1 (fr) | Dérivés N-oxydes de 1-(7-chloroquinoléin-4-yl)pyrazole-3-carboxamides substitués, procédé pour leur préparation et les compositions pharmaceutiques les contenant | |
| FR2823750A1 (fr) | Tetrahydropyridyl-alkyl-heterocycles, procede pour leur preparation et compositions pharmaceutiques les contenant | |
| CH408953A (fr) | Procédé de préparation de nouveaux carbazates substitués |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PH PL PT RO RU SC SD SE SG SK SL TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2003760042 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004513258 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10518652 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003760042 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2003760042 Country of ref document: EP |