WO2004055120A1 - Structurally modified silica - Google Patents
Structurally modified silica Download PDFInfo
- Publication number
- WO2004055120A1 WO2004055120A1 PCT/EP2003/012380 EP0312380W WO2004055120A1 WO 2004055120 A1 WO2004055120 A1 WO 2004055120A1 EP 0312380 W EP0312380 W EP 0312380W WO 2004055120 A1 WO2004055120 A1 WO 2004055120A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- silica
- water
- structurally
- coating agent
- waxes
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C1/00—Treatment of specific inorganic materials other than fibrous fillers; Preparation of carbon black
- C09C1/28—Compounds of silicon
- C09C1/30—Silicic acid
- C09C1/309—Combinations of treatments provided for in groups C09C1/3009 - C09C1/3081
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C1/00—Treatment of specific inorganic materials other than fibrous fillers; Preparation of carbon black
- C09C1/28—Compounds of silicon
- C09C1/30—Silicic acid
- C09C1/3072—Treatment with macro-molecular organic compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C1/00—Treatment of specific inorganic materials other than fibrous fillers; Preparation of carbon black
- C09C1/28—Compounds of silicon
- C09C1/30—Silicic acid
- C09C1/3081—Treatment with organo-silicon compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/42—Gloss-reducing agents
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/10—Solid density
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/11—Powder tap density
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/12—Surface area
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/19—Oil-absorption capacity, e.g. DBP values
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/29—Coated or structually defined flake, particle, cell, strand, strand portion, rod, filament, macroscopic fiber or mass thereof
- Y10T428/2982—Particulate matter [e.g., sphere, flake, etc.]
- Y10T428/2991—Coated
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/29—Coated or structually defined flake, particle, cell, strand, strand portion, rod, filament, macroscopic fiber or mass thereof
- Y10T428/2982—Particulate matter [e.g., sphere, flake, etc.]
- Y10T428/2991—Coated
- Y10T428/2993—Silicic or refractory material containing [e.g., tungsten oxide, glass, cement, etc.]
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/29—Coated or structually defined flake, particle, cell, strand, strand portion, rod, filament, macroscopic fiber or mass thereof
- Y10T428/2982—Particulate matter [e.g., sphere, flake, etc.]
- Y10T428/2991—Coated
- Y10T428/2998—Coated including synthetic resin or polymer
Definitions
- the invention provides a structurally coated silica, a process for its preparation, and its use.
- An aerogel-like structured silica is disclosed in DE 24 14 478. This silica is prepared by incorporating and uniformly distributing water in an air-dispersed pyrogenic silica and drying the powdery mixture obtained.
- This silica has the disadvantage that it has a strong tendency to settle out and is impossible or very difficult to redisperse.
- the document DE 15 ' ' 92 863 describes organically modified precipitation silicas which are coated, for example, with a wax and may be used as matting agents.
- the invention provides a structurally coated silica. It may have a carbon content of 1 to 30 wt.%.
- the silica according to the invention may have a BET surface area of 80 to 450 m 2 /g. It may have a compacted bulk density of 10 to 100 g/1.
- the ' DBP index may be 200 to 150.
- a 4 % strength aqueous suspension of the silica according to the invention may have a pH of 6 to 8.
- structurally coated means that the end product is more highly structured than the starting product and is also coated.
- the structurally coated, silica has a higher DBP index than the initial silica.
- the structurally coated silica according to the invention may be prepared by spraying and mixing a pyrogenic -silica with water and a coating agent in a suitable mixing vessel, then milling and subsequently drying.
- Any known pyrogenic silica may be used as the pyrogenic silica.
- pyrogenic silicas in accordance with Table 1 may be used.
- They are prepared by flame hydrolysis, when a vaporizable metal or metalloid compound, such as for example silicon tetrachloride, is burnt with hydrogen- and oxygen-containing gases in a flame.
- a vaporizable metal or metalloid compound such as for example silicon tetrachloride
- HCI content is a constituent of the
- V goods are supplied in 20 kg sacks
- Waxes and/or organically modified polysiloxanes in the form of aqueous dispersions or emulsions may be used as coating agents.
- Aqueous dispersions or emulsions of waxes for use according to the invention may contain the following waxes:
- PTFE waxes polytetrafluoroethylene with a molecular weight between 30,000 and 2,000,000 g/mol, in particular between 100,000 and 1,000,000 g/mol.
- Polypropylene homopolymer and copolymer waxes ; number average molecular weight 700 - 10,000 g/mol, with a dropping point of 80 - 160°C.
- Amide waxes prepared by reacting ammonia or ethylenediamine with saturated and unsaturated fatty acids.
- the fatty acids are, for example, stearic acid, tallow fatty acid, palmitic acid or erucic acid.
- Montan waxes including acid and ' ester waxes with a carboxylic acid carbon-chain length of C 22 - C 3 e.
- the ester waxes are reaction products of Montan wax with one or more polyhydric alcohols such as, for example, ethanediol, butane-1, 3-diol or propane-1, 2, 3-triol.
- Natural waxes such as, for example, carnauba wax or candelilla wax.
- Macro- or microcrystalline paraffins which are produced during the refining of petroleum.
- the dropping point of the paraffins is between 45 and 65 °C, that of the microcrystalline waxes is between 73 and 100 °C • Sorbitan esters of Montan alcohols.
- a homopolymer or copolymer o'f C 2 -C ⁇ 8 - ⁇ -olefins prepared using Ziegler metal catalysis and one or more other waxes as auxiliary agents, chosen from the group:
- the homopolymers or copolymers of C 2 -C ⁇ 8 - ⁇ -olefins prepared using Ziegler metallocene catalysis preferably have the following properties:
- Suitable polyolefin waxes are homopolymers of ethylene or propylene or copolymers of ethylene and propylene with each other or with one or more 1-olefins.
- Polysiloxanes for use according to the invention may be:
- a dispersion-stable substance with high macrovolumes and very low apparent density (bulk density) corresponding to a Kistler Aerogel, is produced from a pyrogenic silica by targeted loading with water and coating agent followed by drying.
- silicas with large surface areas and thus small primary particles are used.
- silicas with BET surface areas between 10 and 480, in particular 250 to 410 2 /g are used.
- Complete wetting of the primary particles can be achieved when 5 to 20, in particular 7 to 15 wt.% of water and coating agent are incorporated and uniformly distributed in the silica. Since the water which has been incorporated is to be dried out again, the smallest possible amount of water is aimed at, for economic reasons. However, the amount required depends to some extent on the type of incorporation procedure used.
- the build up of a structure in accordance with the invention can be greatly encouraged when basic compounds such as, for example, ammonia, sodium or potassium hydroxide, water-soluble amines, waterglass, etc. are added to the water and coating agent.
- basic compounds such as, for example, ammonia, sodium or potassium hydroxide, water-soluble amines, waterglass, etc.
- the amounts added are expediently chosen so that the pH in the water is adjusted to a value of 7 to 14, preferably 8 to 12, in particular 10 to 11.
- the alkalis used act as solution promoters for silica and bring about an increase in the macroporosity of the process products.
- free silica or hydrolytic silica and/or alkali-releasing substances can also be added to the water and coating agent.
- free silica produced for example by the acidification or ion exchange of silicate solutions or by hydrolytic decomposition of silicon-organic compounds, for example of tetramethyl silicate, also promotes the build up of a structure.
- a hydrolytic alkali and silica releasing substance is, for example, sodium methylsiliconate.
- Uniform distribution of the water and coating agent in the silica can take place by dripping or spraying these into/onto the silica which is being mixed by agitation with the silica at temperatures between 20 and 100, preferably 40 to 70, in particular 50 to €0°C. Mixing by agitation is expediently performed by stirring.
- Another variant for introducing the water comprises spraying the silica with water and coating agent in a fluidized stream of material, for example using a downpipe.
- the water to be incorporated with the coating agent can have a temperature between.20 and 100, preferably 50 to 100, in particular 90 to 100 °C.
- the build up of structure can also be promoted by short-term steaming of the loaded silica in a sealed space. Steaming , leads to especially good distribution of the water. It has proven advantageous here to steam the water-laden silica, before drying, in a sealed vessel for about 5 to 60, preferably 10 to 30, in particular about 20 minutes, at temperatures up to the boiling point of water, preferably 50 to 80, in particular about 60°C.
- Another possibility for improving the distribution of water and coating agent comprises milling the silica laden with water and coating agent, for example in pin mills or air-jet mills.
- the silica is then dried, wherein the preformed structure is probably fixed via the primary particles whose surface is covered with dissolved silica or coated with free silica.
- the type of drying is less critical.
- the prepared mixture of silica and coating agent which always appears to have the status of a dry powder, can be dried, for example, in tray, disc, B ⁇ ttner, continuous flow or microwave driers.
- the silica laden with water and coating agent -can however, also be simultaneously milled and dried in a steam or air-jet mill in order to .save a separate process step.
- silica according to the invention can be used as a matting agent in lacquers. It then has the following advantages:
- the silica according to the invention can be used in particular in polyurethane lacquers .
- Hydrophilic pyrogenic silica (Aerosil 300) with the following physico-chemical properties is used:
- a plough bar mixer is used as the mixing vessel.
- the coating agent is sprayed in at room temperature using a two-fluid nozzle.
- Coating agent A is known from EP 0 341 383 A2. It is a wax emulsion and is prepared as follows:
- the wax emulsion is prepared in an autoclave which can be heated with steam and is provided with a disperser.
- 4.8 parts by wt. of an alkyl polyglycol ether (Marlowet® GFW) at 100°C are first dissolved in 81.0 parts by wt. of water at about 100°C.
- 14.2 parts by wt. of low-pressure polyethylene wax are added .and the mixture is heated to 130°C.
- the disperser is switched on and the mixture is dispersed for 30 minutes. The temperature is held at between 130 and 140°C for this period. After switching off the disperser and cooling to about 110°C, the final emulsion is discharged.
- the polyethylene wax is characterized by the following physical characteristics : average molecular weight 1000 solidification point 100 - 104°C dropping point 110 - 117 °C density (g/cm 3 ) 0.93
- the emulsion is then adjusted to the required pH.
- Coating agent B is known from EP 0 341 383 A2. It is a wax emulsion and is prepared as follows:
- the wax emulsion is prepared in an autoclave which can be heated with steam and which is provided with a disperser.
- 4.8 parts by wt. of an alkyl polyglycol .ether (MarlowetO GFW) at 100°C are first dissolved in 81.0 parts by wt. of water at about 100°C.
- the polyethylene wax used is characterized by the following characteristics: average molecular weight 2700
- Coating agent C consists of 656.4 g of an aqueous polysiloxane emulsion which is diluted with 210 g of water and adjusted to the required pH.
- the polysiloxane emulsion has the following physico-chemical properties :
- Solubility in water miscible pH: 5.5 at 20°C in original state viscosity, dynamic: about 2,300 Pa.s at 25°C
- Coating agent D consists of 503.0 g of an aqueous alkyl ester / polydimethylsiloxane emulsion which is diluted with 116 g of water and adjusted to the required pH.
- the alkyl ester / polydimethylsiloxane emulsion has the following physico-chemical properties:
- Boiling point about 100 °C
- Viscosity, kinematic about 40 - 120 mm 2 /s at 20°C
- Coating agent E consists of 365.6 g of an aqueous emulsion of an ester wax and ⁇ anauba wax and 346 g of water. The mixture is adjusted to a pH of' 10.6.
- the emulsion of ester wax, carnauba wax, emulsifier and water has the following composition:
- the emulsion used has the following physico-chemical properties:
- the BET surface area is determined in accordance with DIN '66 131 using nitrogen.
- the compacted density is determined using a method based on DIN ISO 787/XI
- the compacted density (previously compacted volume) is given by the quotient of the weight and the volume of the powder after compacting in a compacting volumeter under fixed conditions . According to DIN ISO 787/XI the compacted density is given in g/cm 3 . Due to the very low compacted density of the oxides, however, we give the values in g/1. Furthermore, we do not perform drying and screening procedures nor do we repeat the compacting process.
- the weight of the sample in the measuring cylinder is determined accurately to 0.01 g.
- the measuring cylinder with the sample is inserted into the measuring cylinder holder in the compacting volumeter and rammed down 1250 times .
- the volume of the compacted oxide is read off accurately to
- the pH is determined in 4 % strength aqueous solution; in the case of hydrophobic oxides in water : methanol 1 : 1.
- the method for determining pH is based on DIN/ISO 787/IX:
- hydrophilic oxide 4 g are stirred with the aid of a dispensette in a 250 ml beaker with 96 g (96 ml) of water and then for five minutes with a magnetic stirrer (speed ca. 1000 min -1 ) with the pH electrode immersed therein.
- the lid is put on before cooling. A second drying is not performed.
- the result is recorded to one decimal place.
- the result is recorded to one decimal place.
- Plastic beaker 250 ml
- sample are weighed into a plastic beaker and placed in the compounding chamber. If instructed, the amount weighed out may differ from this (e.g. 8 or 20 g) .
- the DBP metering unit is switched on. As soon as the filling procedure is complete (display F) , the plastograph is ready for use. Measurement starts when the Start buttons are pressed simultaneously.
- the metering unit meters 4 ml DBP/min until the pre-set cutoff point (l €OO) is reached.
- the instrument switches off automatically. Only the consumption of DBP can be read on the display for the metering unit.
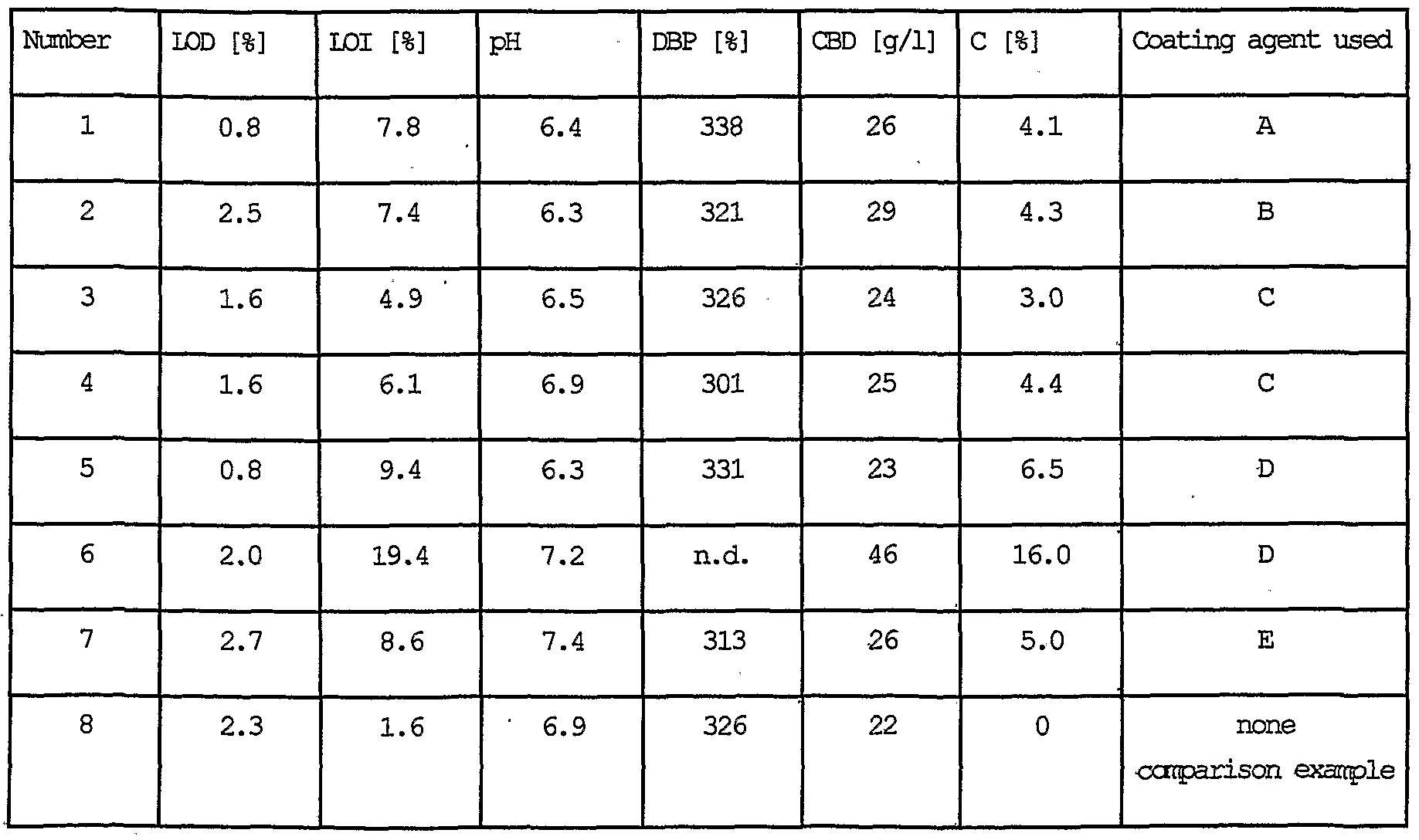
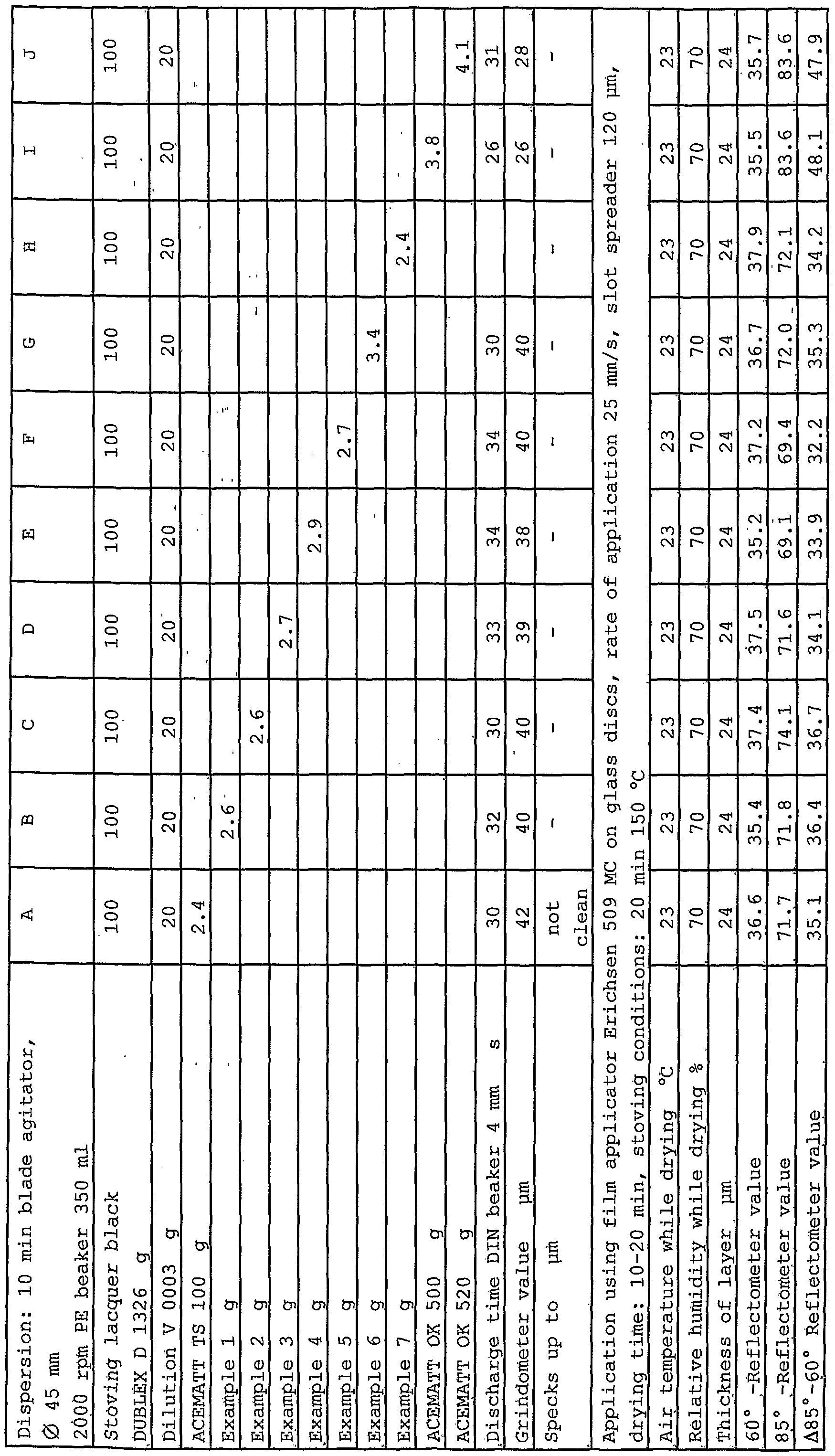
- Silicas according to the invention in accordance with examples 1 to 7 are tested in various lacquer systems. The results of these tests are given in Tables 5 to 8.
- Table 5 shows the effect of silicas according to the invention in accordance with examples 1 to 7 which are used in black stoving lacquer DUPLEX D 1326 and this is compared with the matting agent in accordance with DE 24 14 478.
- the stoving lacquer DUPLEX 1326 is a commercial product from DuPont Coatings, Austria.
- the basis of the binder is an alkyd/melamine resin. Characteristics: black pigmented alkyl/melamine ' resin stoving lacquer, high-gloss, original use: motor vehicle (HGV) mass production lacquer. It is used as a standard test system, in particular for testing the grindometer value and the matting efficiency.
- Table 6 shows the effect of silicas according to the invention in accordance with examples 1 to 7 which are used in a NC test lacquer for suspended particle tests and this is compared with the matting agent in accordance with DE 24 14 478.
- the NC test lacquer is used exclusively for testing the settling out behavior of matting agents. It has the following composition.
- Table 7 shows the effect of silicas according to the invention in accordance with examples 1 to 7 which are used in DD blue- streak lacquer P and this is compared with the matting agent in accordance with DE 24 14 478.
- the blue-streak lacquer is a 2-component polyurethane lacquer and is used in particular to test matting efficiency. It is used as a model for unpigmented wood and furniture lacquers which are difficult to provide with a matt finish.
- the direct effect of the matting agent on the degree of gloss (reflectometer value) is obtained by always weighing out the same amount of all the products.
- the blue-streak lacquer ha-s the following composition: Raw material Concentration Amount
- Table 8 shows the effect of silicas according to the invention in accordance with examples 1 to 7 in the standardized wax detachment test. This test is used to check whether all the coating agent applied to prevent settling out remains on the silica in the lacquer and cannot be removed. For this purpose, the coated silicas are subjected to storage in ethoxypropyl acetate at elevated temperature. If a product passes this test, it can be predicted with high probability that the result will apply in all lacquer systems .
- Amount of matting agent weighed out lg
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Paints Or Removers (AREA)
- Silicon Compounds (AREA)
- Pigments, Carbon Blacks, Or Wood Stains (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR0317395-0A BR0317395A (en) | 2002-12-18 | 2003-11-06 | Structurally modified silica |
| JP2004559676A JP4860928B2 (en) | 2002-12-18 | 2003-11-06 | Structurally modified silica |
| AU2003286153A AU2003286153A1 (en) | 2002-12-18 | 2003-11-06 | Structurally modified silica |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US43396302P | 2002-12-18 | 2002-12-18 | |
| US60/433,963 | 2002-12-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004055120A1 true WO2004055120A1 (en) | 2004-07-01 |
Family
ID=32595256
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2003/012380 Ceased WO2004055120A1 (en) | 2002-12-18 | 2003-11-06 | Structurally modified silica |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US7303624B2 (en) |
| JP (1) | JP4860928B2 (en) |
| KR (1) | KR100954073B1 (en) |
| CN (1) | CN100436548C (en) |
| AU (1) | AU2003286153A1 (en) |
| BR (1) | BR0317395A (en) |
| WO (1) | WO2004055120A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2301667A2 (en) | 2009-09-29 | 2011-03-30 | Evonik Degussa GmbH | Low pressure grinding method |
| US11999857B2 (en) | 2017-06-02 | 2024-06-04 | W.R. Grace & Co.-Conn. | Coated particles and methods of making and using the same |
| US12203006B2 (en) | 2017-08-03 | 2025-01-21 | W.R. Grace & Co.-Conn. | Silica-based matting agents and methods of making and using the same |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102004029074A1 (en) * | 2004-06-16 | 2005-12-29 | Degussa Ag | Paint formulation for improving the surface properties |
| DE102004029073A1 (en) * | 2004-06-16 | 2005-12-29 | Degussa Ag | Paint formulation with improved rheological properties |
| DE102005001408A1 (en) * | 2005-01-12 | 2006-07-20 | Degussa Ag | Pyrogenic silica powder |
| DE102005001414A1 (en) * | 2005-01-12 | 2006-07-20 | Degussa Ag | Pyrogenic silica powder |
| DE102005001409A1 (en) * | 2005-01-12 | 2006-07-20 | Degussa Ag | Pyrogenic silica powder and silicone sealant containing this powder |
| BRPI0617107A2 (en) * | 2005-08-25 | 2011-07-12 | Du Pont | modified nanoparticle and coating composition |
| DE102008000499A1 (en) * | 2008-03-04 | 2009-09-10 | Evonik Degussa Gmbh | Silica and epoxy resins |
| US20090258230A1 (en) * | 2008-04-11 | 2009-10-15 | Kobo Products, Inc. | Porous and/or hollow material containing uv attenuating nanoparticles, method of production and use |
| KR101047965B1 (en) * | 2009-06-11 | 2011-07-12 | 한국에너지기술연구원 | Airgel mat, manufacturing method and apparatus thereof |
| DE102009045104A1 (en) | 2009-09-29 | 2011-03-31 | Evonik Degussa Gmbh | Novel matting agents for UV coatings |
| DE102012211121A1 (en) | 2012-06-28 | 2014-01-02 | Evonik Industries Ag | Granular, functionalized silicic acid, process for their preparation and their use |
| CN104556068B (en) * | 2013-10-24 | 2017-05-17 | 中国石油化工股份有限公司 | Porous silica and preparation method thereof |
| CN104556067B (en) * | 2013-10-24 | 2017-05-17 | 中国石油化工股份有限公司 | Porous silica and synthesis method thereof |
| TWI481498B (en) * | 2013-12-27 | 2015-04-21 | Plastics Industry Dev Ct | Fruit and Vegetable fresh packing material and producing method thereof |
| WO2020092231A1 (en) | 2018-10-29 | 2020-05-07 | Leica Biosystems Richmond, Inc. | Ink composition and methods of use and making |
| US12448528B2 (en) | 2019-02-01 | 2025-10-21 | W.R. Grace & Co.-Conn. | Silica-based matting agents and methods of making and using the same |
| CN114585672B (en) | 2019-08-30 | 2023-10-31 | 格雷斯公司 | Silica-based matting formulations and methods of making and using the same |
| EP3954743A1 (en) | 2020-08-12 | 2022-02-16 | Evonik Operations GmbH | Use of silicon dioxide to improve the conductivity of coatings |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1110331A (en) * | 1964-05-06 | 1968-04-18 | Degussa | Process for the production of hydrophobic silica |
| DE4032619A1 (en) * | 1990-10-15 | 1992-04-16 | Grace Gmbh | New flattening agents |
| EP0808880A2 (en) * | 1996-04-26 | 1997-11-26 | Degussa Aktiengesellschaft | Silanized silicas |
| US5711797A (en) * | 1994-01-27 | 1998-01-27 | Degussa Aktiengesellschaft | Silanized silicic acids |
| EP1182233A2 (en) * | 2000-08-23 | 2002-02-27 | Degussa AG | Method of coating silicic acid with waxes |
| EP1199336A1 (en) * | 2000-10-21 | 2002-04-24 | Degussa AG | Functionalized, structure modified silicic acids |
| US20020077381A1 (en) * | 2000-10-21 | 2002-06-20 | Stephanie Frahn | Radiation-curing coating systems |
Family Cites Families (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2870108A (en) | 1954-05-06 | 1959-01-20 | Monsanto Chemicals | Novel silica products and silicone rubber containing same coated silica aerogel, silicone rubber reinforced therewith and method of making |
| US3037933A (en) | 1960-10-31 | 1962-06-05 | Gen Electric | Greases made by thickening silicone oils with silica and silica coated with octamethylcyclotetrasiloxane |
| DE2414478C3 (en) * | 1974-03-26 | 1978-07-13 | Deutsche Gold- Und Silber-Scheideanstalt Vormals Roessler, 6000 Frankfurt | Airgel-like structured silica and process for making same |
| JPS54101795A (en) * | 1978-01-30 | 1979-08-10 | Toyo Soda Mfg Co Ltd | Hydrophobic rendering method for oxide fine powder |
| JPS5915084B2 (en) * | 1979-07-03 | 1984-04-07 | 株式会社トクヤマ | Manufacturing method of modified silica |
| US4477607A (en) * | 1983-08-31 | 1984-10-16 | Loctite Corporation | Thixotropic cyanoacrylate compositions |
| DE3707226A1 (en) * | 1987-03-06 | 1988-09-15 | Wacker Chemie Gmbh | METHOD FOR PRODUCING HIGHLY DISPERSAL METAL OXIDE WITH AMMONIUM-FUNCTIONAL ORGANOPOLYSILOXANE MODIFIED SURFACE AS A POSITIVELY CONTROLLING CHARGING AGENT FOR TONER |
| US5021317A (en) * | 1987-10-28 | 1991-06-04 | Konica Corporation | Electrostatic latent image developer with toner particles surface treated with a polysiloxane having ammonium salt functional groups |
| DE3815670A1 (en) | 1988-05-07 | 1990-01-25 | Degussa | FINE-PARTICULATED SOFT SEEDS WITH HIGH STRUCTURE, METHOD FOR ITS MANUFACTURE AND USE |
| JPH0217932A (en) * | 1988-07-05 | 1990-01-22 | Nippon Chem Ind Co Ltd | Modified inorganic particle and preparation thereof |
| US5221337A (en) * | 1990-02-14 | 1993-06-22 | W. R. Grace & Co.-Conn. | SiO2 flatting agent, process for its production and its use |
| US5620773A (en) | 1990-02-21 | 1997-04-15 | Mcghan Nusil Corporation | Composition and method for texturing the surface of gloves |
| ES2033153T3 (en) * | 1990-07-19 | 1993-03-01 | Degussa Aktiengesellschaft | MODIFIED SILICON DIOXIDE ON THE SURFACE. |
| JP2646150B2 (en) * | 1990-08-27 | 1997-08-25 | 出光興産 株式会社 | Water repellent silica sol and method for producing the same |
| WO1993008236A1 (en) | 1991-10-25 | 1993-04-29 | Nusil Technology | Composition and method for texturing the surface of articles |
| CA2090793A1 (en) | 1992-10-09 | 1994-04-10 | Ronald Sinclair Nohr | Nonwoven webs having improved tensile strength characteristics |
| CN1052742C (en) * | 1993-02-12 | 2000-05-24 | 卡伯特公司 | Surface modified silica and method for thickening aqueous systems using same |
| DE4343818A1 (en) * | 1993-12-22 | 1995-06-29 | Rhein Chemie Rheinau Gmbh | External release agent |
| GB9409388D0 (en) | 1994-05-11 | 1994-06-29 | Unilever Plc | Silica products |
| US5959005A (en) | 1996-04-26 | 1999-09-28 | Degussa-Huls Aktiengesellschaft | Silanized silica |
| JPH10279829A (en) * | 1997-04-07 | 1998-10-20 | Central Glass Co Ltd | Water-repellent composite particle and water-repellent material prepared therefrom |
| DE19917228B4 (en) * | 1999-04-16 | 2006-02-02 | E.I. Du Pont De Nemours And Co., Wilmington | Matting and structurant-containing pastes and coating compositions containing them |
| EP1238021B1 (en) * | 1999-09-28 | 2007-03-14 | Cabot Corporation | Surface coating composition |
| JP2001158871A (en) * | 1999-12-02 | 2001-06-12 | Kao Corp | Surface modifier |
| EP1249475B1 (en) * | 1999-12-24 | 2005-03-16 | Nippon Aerosil Co., Ltd. | Surface-modified inorganic oxide powder, process for producing the same, and use thereof |
| DE10105750A1 (en) * | 2001-02-08 | 2002-10-10 | Degussa | Precipitated silicas with a narrow particle size distribution |
-
2003
- 2003-11-06 CN CNB2003801068124A patent/CN100436548C/en not_active Expired - Fee Related
- 2003-11-06 AU AU2003286153A patent/AU2003286153A1/en not_active Abandoned
- 2003-11-06 KR KR1020057011432A patent/KR100954073B1/en not_active Expired - Fee Related
- 2003-11-06 WO PCT/EP2003/012380 patent/WO2004055120A1/en not_active Ceased
- 2003-11-06 BR BR0317395-0A patent/BR0317395A/en not_active Application Discontinuation
- 2003-11-06 JP JP2004559676A patent/JP4860928B2/en not_active Expired - Fee Related
- 2003-12-11 US US10/732,238 patent/US7303624B2/en not_active Expired - Lifetime
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1110331A (en) * | 1964-05-06 | 1968-04-18 | Degussa | Process for the production of hydrophobic silica |
| DE4032619A1 (en) * | 1990-10-15 | 1992-04-16 | Grace Gmbh | New flattening agents |
| US5711797A (en) * | 1994-01-27 | 1998-01-27 | Degussa Aktiengesellschaft | Silanized silicic acids |
| EP0808880A2 (en) * | 1996-04-26 | 1997-11-26 | Degussa Aktiengesellschaft | Silanized silicas |
| EP1182233A2 (en) * | 2000-08-23 | 2002-02-27 | Degussa AG | Method of coating silicic acid with waxes |
| EP1199336A1 (en) * | 2000-10-21 | 2002-04-24 | Degussa AG | Functionalized, structure modified silicic acids |
| US20020077381A1 (en) * | 2000-10-21 | 2002-06-20 | Stephanie Frahn | Radiation-curing coating systems |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2301667A2 (en) | 2009-09-29 | 2011-03-30 | Evonik Degussa GmbH | Low pressure grinding method |
| DE102009045116A1 (en) | 2009-09-29 | 2011-03-31 | Evonik Degussa Gmbh | Niederdruckvermahlungsverfahren |
| US8864056B2 (en) | 2009-09-29 | 2014-10-21 | Evonik Degussa Gmbh | Low-pressure milling process |
| US11999857B2 (en) | 2017-06-02 | 2024-06-04 | W.R. Grace & Co.-Conn. | Coated particles and methods of making and using the same |
| US12203006B2 (en) | 2017-08-03 | 2025-01-21 | W.R. Grace & Co.-Conn. | Silica-based matting agents and methods of making and using the same |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2003286153A1 (en) | 2004-07-09 |
| JP4860928B2 (en) | 2012-01-25 |
| CN1729255A (en) | 2006-02-01 |
| JP2006521411A (en) | 2006-09-21 |
| CN100436548C (en) | 2008-11-26 |
| US20040127604A1 (en) | 2004-07-01 |
| US7303624B2 (en) | 2007-12-04 |
| KR100954073B1 (en) | 2010-04-23 |
| KR20050088315A (en) | 2005-09-05 |
| BR0317395A (en) | 2005-11-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7303624B2 (en) | Structurally coated silica | |
| US7816442B2 (en) | Surface-modified precipitated silicas | |
| AU2005202577B2 (en) | Surface-modified silica gels | |
| CN1747899B (en) | Surface-modified airgel-type structured silica | |
| US6899951B2 (en) | High-whiteness, non-heat-treated hydrophobic precipitated silica | |
| US9567229B2 (en) | Fumed silica | |
| US8552107B2 (en) | Fumed silanized silica | |
| US20120152151A1 (en) | Fumed silanized and ground silica | |
| EP2147059A1 (en) | Fumed silanized and ground silica | |
| US9624318B2 (en) | Matting agent for UV varnishes | |
| TW200530128A (en) | Hydrophilic precipitated silica for defoamer formulations | |
| KR101329381B1 (en) | Pyrogenic silicic acid manufactured in a small-scale production plant | |
| EP1431338B1 (en) | Texture-coated silica | |
| MXPA01008286A (en) | Method of coating silicic acid with waxes. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1275/CHENP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020057011432 Country of ref document: KR Ref document number: 2004559676 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20038A68124 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020057011432 Country of ref document: KR |
|
| ENP | Entry into the national phase |
Ref document number: PI0317395 Country of ref document: BR |
|
| 122 | Ep: pct application non-entry in european phase | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) |