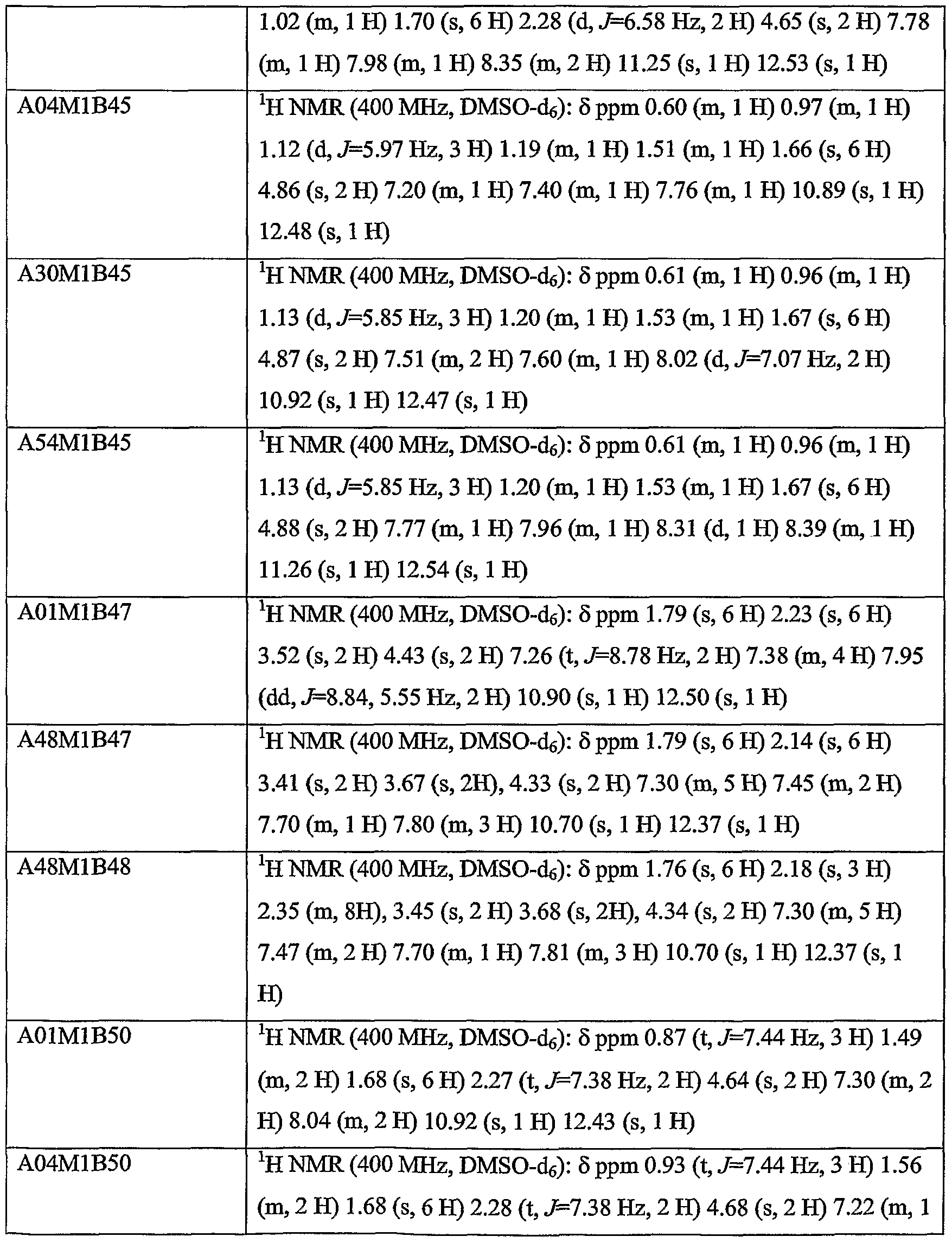
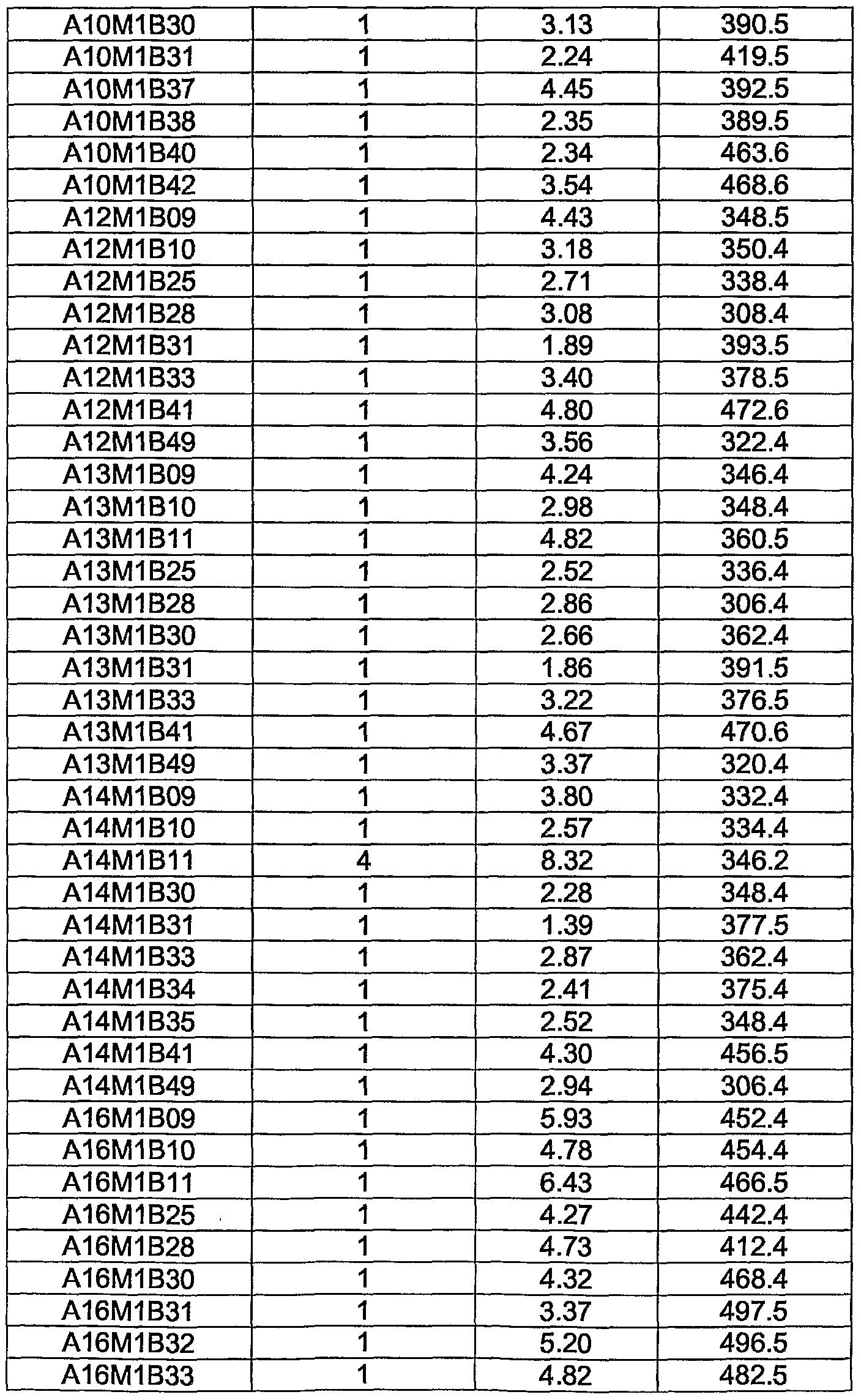
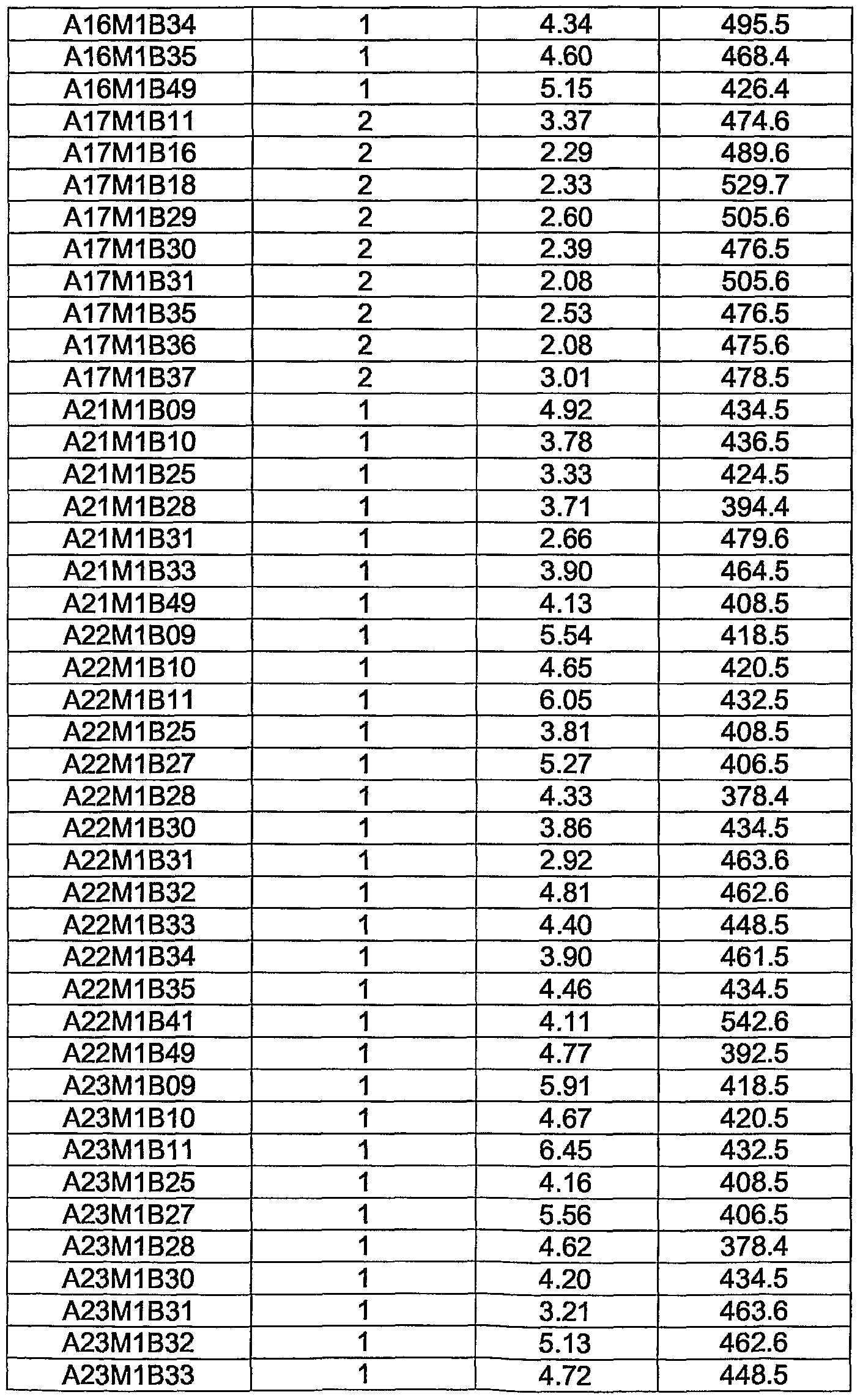
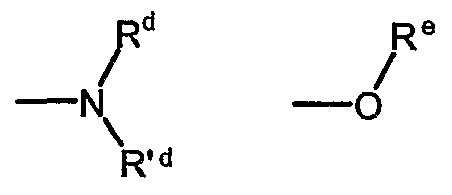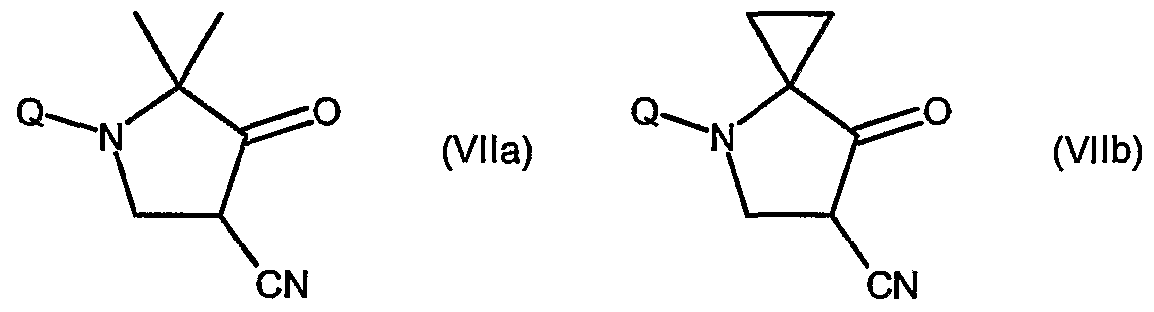WO2004056827A2 - Substituted pyrrolo-pyrazole derivatives as kinase inhibitors - Google Patents
Substituted pyrrolo-pyrazole derivatives as kinase inhibitors Download PDFInfo
- Publication number
- WO2004056827A2 WO2004056827A2 PCT/EP2003/050942 EP0350942W WO2004056827A2 WO 2004056827 A2 WO2004056827 A2 WO 2004056827A2 EP 0350942 W EP0350942 W EP 0350942W WO 2004056827 A2 WO2004056827 A2 WO 2004056827A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- group
- compound
- alkyl
- cycloalkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*1C(C(*)(*(C2)C(*)=O)NC)=C2C(**)=*1 Chemical compound C*1C(C(*)(*(C2)C(*)=O)NC)=C2C(**)=*1 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
- A61K31/4162—1,2-Diazoles condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4523—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems
- A61K31/454—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. pimozide, domperidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N5/00—Radiation therapy
- A61N5/10—X-ray therapy; Gamma-ray therapy; Particle-irradiation therapy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/08—Drugs for disorders of the urinary system of the prostate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/14—Drugs for dermatological disorders for baldness or alopecia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
Definitions
- the present invention relates to pyrrolo-pyrazole derivatives, to a process for their preparation, to pharmaceutical compositions comprising them, and to their use as therapeutic agents, particularly in the treatment of cancer and cell proliferation disorders. Discussion of the Background
- cytotoxic drugs such as, e.g., fluorouracil (5-FU), doxorubicin and camptothecins, damage DNA or affect cellular metabolic pathways and thus cause, in many cases, an indirect block of the cell cycle. Therefore, by producing an irreversible damage to both normal and tumor cells, these agents result in a significant toxicity and side-effects.
- the present inventors have now discovered that certain pyrazole compounds are endowed with cdk cyclin kinase inhibitory activity and are thus useful in therapy as antitumor agents and lack, in terms of both toxicity and side effects, the aforementioned drawbacks associated with currently available antitumor drugs.
- osteosarcoma osteosarcoma, xeroderma pigmentosum, keratoxanthoma, thyroid follicular cancer and
- the compounds of this invention may also be useful in the treatment of cancer, viral infections, prevention of AIDS development in HTV-infected individuals, autoimmune diseases and neurodegenerative disorders.
- the compounds of this invention may be useful in inhibiting tumor angiogenesis and metastasis, as well as in the treatment of organ transplant rejection and host versus graft disease.
- the compounds of the invention may also act as inhibitor of other protein kinases, e.g., protein kinase C in different isoforms, Met, PAK-4, PAK-5, ZC-1, STLK-2, DDR-2, Aurora 1, Aurora 2, Bub-1, PLK, Chkl, Chk2, HER2, rafl, MEKl, MAPK, EGF-R,
- protein kinases e.g., protein kinase C in different isoforms, Met, PAK-4, PAK-5, ZC-1, STLK-2, DDR-2, Aurora 1, Aurora 2, Bub-1, PLK, Chkl, Chk2, HER2, rafl, MEKl, MAPK, EGF-R,
- the compounds of the invention are also useful in the treatment and prevention of radiotherapy-induced or chemotherapy-induced alopecia.
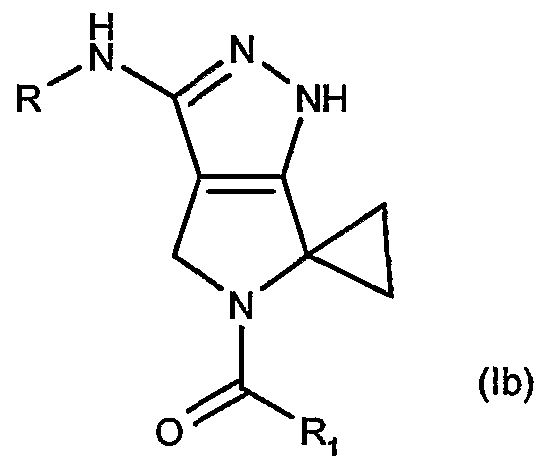
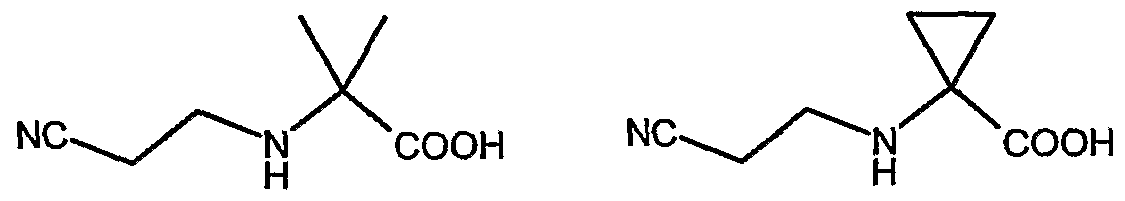
- R is a group -COR a , -CONHR or -CONR a R b wherein R a and R are, each independently, hydrogen or an optionally substituted group selected from straight or branched -C 6 alkyl, C 3 -Ce cycloalkyl, aryl, arylalkyl, heterocyclyl or heterocyclylalkyl or; together with the nitrogen atom to which they are bonded, R a and R may form an optionally substituted 5 or 6 membered heterocycle optionally containing one additional heteroatom or heteroatomic group selected among N, NH, O or S; Ri is selected from the group consisting of: a) straight or branched C 3 -C alkyl; b) cycloalkyl, cycloalkyl-alkyl or alkyl-cycloalkyl wherein the cycloalkyl moiety comprises any C3-C6 cycloalkyl group and wherein the alkyl moiety comprises any straight or branched
- Ri is selected from the group consisting of: a) straight or branched C 3 -C 4 alkyl;
- Prodrugs are any covalently bonded compounds, which release the active parent drug according to formula (I) in vivo.
- a metabolite of a compound of formula (I) is any compound into which this same compound of formula (I) is converted, in vivo, for instance upon administration to a mammal in need thereof.
- R is as defined in the general formula
- Ri is a group selected from tert-butyl, l-methyl-piperidyl-4-yl, 1- methyl-piperazinyl-4-yl, 2-(R,S)-tetrahydrofuranyl-2-yl, 2-(R)-tetrahydrofuranyl-2-yl or 2-
- step (d) of the process the compound of formula (Via) or (Vlb) is converted into the corresponding cyclic derivative of formula (Vila) or (Vllb) through reaction with sodium hydride.
- the reaction is carried out in the presence of a suitable solvent such as dioxane or tetrahydrofuran at refluxing temperature.
- step (e) of the process the compound of formula (VHa) or (VHb) is reacted with hydrazine hydrate, preferably with an excess of hydrazine monohydrated, for instance up to 10 equivalents, in the presence of a suitable solvent such as halogenated hydrocarbons, lower alcohols or admixtures thereof.
- the reaction is preferably carried out in the presence of ethanol, by adding hydrazine to a solution of the compound of formula (VHa) or (Vllb) and under stirring for a suitable time, for instance about 48 hours, at the temperature ranging from about 20°C to about 70°C.
- a suitable time for instance about 48 hours, at the temperature ranging from about 20°C to about 70°C.
- the above reaction is carried out also in the presence of glacial acetic acid.
- step (g.2) of the process the compound of formula ( Ca) or (LXb) is reacted with an isocyanate derivative of formula (XII), by operating substantially as set forth in step (g.l) of the process, except that the base may not be required.
- the compound of formula (Xla) or (Xlb) is thus suspended in a suitable solvent such as dichloromethane or dioxane, and treated with a concentrated solution of the selected acid.
- Step (i.l) is carried out with an acyl halide, preferably chloride, of formula (XV) in a suitable solvent such as dichloromethane and under basic conditions, for instance in the presence of a suitable amine such as diisopropylethylamine.
- a suitable solvent such as dichloromethane
- a suitable amine such as diisopropylethylamine
- step (i.3) is carried out with a carboxylic acid derivative of formula (XLX), in the presence of a suitable condensing agent such as, for instance, dicyclohexylcarbodiimide (DCC), l-elhyl-3-(3'-dimethylaminopropyl)carbodiimide (EDC) or 0-benzotriazolyl tetramethylisouronium tetrafluoroborate (TBTU), and by operating according to well-known methods for preparing carboxamido derivatives.
- a suitable condensing agent such as, for instance, dicyclohexylcarbodiimide (DCC), l-elhyl-3-(3'-dimethylaminopropyl)carbodiimide (EDC) or 0-benzotriazolyl tetramethylisouronium tetrafluoroborate (TBTU), and by operating according to well-known methods for preparing carboxamido derivatives
- step (i.4) of the process the compound of formula (XTVa) or (XlVb) is reacted with a suitable derivative of formula (XX) wherein Ri is a group of formula (lid) and R e is as set forth in formula (la) or (lb), so as to obtain the conesponding carbamate derivatives of formula (XVIa) or (XVIb).
- step (j) of the process the compound of formula (XVIa) or (XVIb) being obtained in any one of steps from (i.l) to (i.4) is reacted with a suitable base, for instance triethylamine, and in the presence of a suitable solvent such as methanol or ethanol so as to obtain the desired compound of formula (la) or (lb).
- a suitable base for instance triethylamine
- a suitable solvent such as methanol or ethanol
- the operative conditions are those widely known in the art and may comprise, for instance in the conversion of a carboxyester group into a carboxamide group, the reaction with ammonia or ammonium hydroxide in the presence of a suitable solvent such as a lower alcohol, dimethylformamide or mixtures thereof; preferably the reaction is carried out with ammonium hydroxide in a methanol/dimethylformamide mixture, at a temperature ranging from about 50°C to about 100°C.
- Analogous operative conditions apply in the preparation of N-substituted or N,N- disubstituted carboxamides wherein a suitable primary or secondary amine is used in place of ammonia or ammonium hydroxide.
- carboxyester groups may be converted into carboxylic acid derivatives through basic or acidic hydrolysis conditions, widely known in the art.
- compounds of formula (la) or (lb) bearing an amino function may be easily converted into the corresponding carboxamido or ureido derivatives.
- the compounds of formula (I) of the invention may be also prepared according to combinatorial chemistry techniques widely known in the art, by accomplishing the aforementioned reactions between the several intermediates in a serial manner and by working under solid-phase-synthesis (SPS) conditions.
- SPS solid-phase-synthesis
- the supported intermediate thus obtained may be then reacted according to step (h) and any one of steps (i.l), (i.2), (i.3) or (i.4) of the process, so as to obtain the conesponding compound of formula (la) or (lb) of the invention still supported on the polymeric resin.
- Subsequent resin cleavage for instance under basic or acidic conditions according to known methods, allows to obtain the desired compounds of formula (la) or (lb).
- R is a group -COR a , -CONHR a or -CONR a R b wherein R a and R b are, each independently, hydrogen or an optionally substituted group selected from straight or branched Ci-C ⁇ alkyl, C 3 -C 6 cycloalkyl, aryl, arylalkyl, heterocyclyl or heterocyclylalkyl or; together with the nitrogen atom to which they are bonded, R a and R may form an optionally substituted 5 or 6 membered heterocycle optionally containing one additional heteroatom or heteroatomic group selected among N, NH, O or S; Ri is selected from the group consisting of: a) straight or branched C 3 -C 4 alkyl; b) cycloalkyl, cycloalkyl-alkyl or alkyl-cycloalkyl wherein the cycloalkyl moiety comprises any C3-C6 cycloalkyl group and wherein the alkyl moiety comprises any
- R d , R' d and R e represent, the same or different and independently from each other, a hydrogen atom or a straight or branched Ci-Ce alkyl optionally substituted by one or more groups selected from hydroxy (-OH), aminocarbonyl (-CONH 2 ) or methylaminocarbonyl (-CONHCH 3 ); provided that in formula (la), when Ri is a group of formula (He) and one of R d or R' d is a hydrogen atom whilst the other of R d or R' d is ethyl or n-butyl, then R is other than -COR a with R a as 3-bromophenyl, benzyl, 4-tert-butylphenyl, 4-tert-butylphenyhnethyl, 4- fluorophenylmethyl, cyclopropyl or 2-naphthylmethyl; or a pharmaceutically acceptable salt thereof.
- R is a group -COR a , -CONHR or -CONR a R wherein R and R b are, each independently, hydrogen or an optionally substituted group selected from straight or branched Q-Cg alkyl, C3-C6 cycloalkyl, aryl, arylalkyl, heterocyclyl or heterocyclylalkyl or; together with the nitrogen atom to which they are bonded, R a and R may form an optionally substituted 5 or 6 membered heterocycle optionally containing one additional heteroatom or heteroatomic group selected among N, NH, O or S;
- Ri is selected from the group consisting of: a) straight or branched C 3 -C 4 alkyl;
- cycloalkyl cycloalkyl-alkyl or alkyl-cycloalkyl wherein the cycloalkyl moiety comprises any C3-C6 cycloalkyl group and wherein the alkyl moiety comprises any straight or branched C ⁇ -C alkyl group: c) 3-methylthienyl-2-yl; 2-thienyl; phenyl; 2,6-difluorophenyl; 4- (aminosulfonyl)phenyl; 4-(dimethylaminomethyl)phenyl; 4-(4-methylpiperazinyl)methyl- phenyl; d) a group of formula (Ila) or (lib) :
- R d , R' d and R e represent, the same or different and independently from each other, a hydrogen atom or a straight or branched Ci-C ⁇ alkyl optionally substituted by one or more groups selected from hydroxy (-OH), aminocarbonyl (-CONH 2 ) or methylaminocarbonyl (-CONHCH 3 ); or a pharmaceutically acceptable salt thereof.
- the compounds of formula (I) are active as protein kinase inhibitors and are therefore useful, for instance, to restrict the unregulated proliferation of tumor cells. In therapy, they may be used in the treatment of various tumors, such as those formerly reported, as well as in the treatment of other cell proliferative disorders such as psoriasis, vascular smooth cell proliferation associated with atherosclerosis and post-surgical stenosis and restenosis and in the treatment of Alzheimer's disease.
- the inhibiting activity of putative Cdk/Cyclin inhibitors and the potency of selected compounds was determined through a method of assay based on the use of the SPA technology (Amersham Pharmacia Biotech).
- the assay consists of the transfer of radioactivity labelled phosphate moiety by the kinase to a biotinylated substrate.
- the resulting 33P-labelled biotinylated product is allowed to bind to streptavidin-coated SPA beads (biotin capacity 130 pmol mg), and light emitted was measured in a scintillation counter.
- reaction was stopped by 100 ⁇ l PBS + 32 mM EDTA + 0.1% Triton X-100 + 500 ⁇ M ATP, containing 1 mg SPA beads. Then a volume of 110 ⁇ l is transfened to Optiplate.
- the selected compounds have been characterized on a panel of ser/threo kinases strictly related to cell cycle (Cdk2/Cyclin E, Cdkl/cyclin Bl, Cdk5/p25, Cdk4/Cyclin Dl), and also for specificity on MAPK, PKA, EGFR, IGF1-R, Aurora-2 and Akt.
- Inhibition assay of Cdk2/Cvclin E activity Kinase reaction 10 ⁇ M in house biotinylated histone HI (Sigma # H-5505) substrate, 30 ⁇ M ATP (0.3 microCi P 33 ⁇ -ATP), 4 ng GST-Cdk2/Cyclin E complex, inhibitor in a final volume of 30 ⁇ l buffer (TRIS HCl 10 mM pH 7.5, MgCl 2 10 mM, DTT 7.5 mM + 0.2 mg ml BSA) were added to each well of a 96 U bottom. After 60 min at r.t.
- reaction was stopped by 100 ⁇ l PBS + 32 mM EDTA + 0.1% Triton X-100 + 500 ⁇ M ATP, containing 1 mg SPA beads. Then a volume of 110 ⁇ l is transfe ⁇ ed to Optiplate. After 20 min.
- reaction was stopped by 100 ⁇ l PBS + 32 mM EDTA + 0.1% Triton X-100 + 500 ⁇ M ATP, containing 1 mg SPA beads. Then a volume of 110 ⁇ l is transfe ⁇ ed to Optiplate.
- the inhibition assay of Cdk5/p25 activity was performed according to the following protocol.
- SPA beads Then a volume of 110 ⁇ l is transfe ⁇ ed to Optiplate. After 20 min. incubation for substrate capture, lO ⁇ l 5M CsCl were added to allow statification of beads to the top of the plate and let stand 4 hours before radioactivity counting in the Top-Count instrument.
- Detection filters were allowed to dry at 37°C, then 100 ⁇ l/well scintillant were added and 33 P labeled Rb fragment was detected by radioactivity counting in the Top-Count instrument.
- IC50 determination see above Inhibition assay of EGFR activity Kinase reaction: 10 ⁇ M in house biotinylated MBP (Sigma # M-1891) substrate, 2 ⁇ M ATP (0.04 microCi P 33 ⁇ -ATP), 36 ng insect cell expressed GST-EGFR, inhibitor in a final volume of 30 ⁇ l buffer (Hepes 50 mM pH 7.5, MgCl 2 3 mM, MnCl 2 3 mM, DTT 1 mM, NaV0 3 3 ⁇ M + 0.2 mg/ml BSA) were added to each well of a 96 U bottom. After 20 min at r.t. incubation, reaction was stopped by 100 ⁇ l PBS + 32 mM EDTA + 0.1% Triton X-100
- the inhibition assay of IGF1-R activity was performed according to the following protocol.
- Kinase reaction 10 ⁇ M biotinylated MBP (Sigma cat. # M-1891) substrate, 0-20 ⁇ M inhibitor, 6 ⁇ M ATP, 1 microCi 33 P-ATP, and 22.5 ng GST-IGF1-R (pre-incubated for 30 min at room temperature with cold 60 ⁇ M cold ATP) in a final volume of 30 ⁇ l buffer (50 mM HEPES pH 7.9, 3 mM MnCl 2 , 1 mM DTT, 3 ⁇ M NaV0 3 ) were added to each well of a 96 U bottom well plate.
- Biotinylated peptide 4 repeats of LRRWSLG
- 10 ⁇ M ATP 0.5 uCi P 33 ⁇ -ATP
- 15 ng Aurora2, inhibitor in a final volume of 30 ⁇ l buffer (HEPES 50 mM pH 7.0, MgCl 2 10 mM, 1 mM DTT, 0.2 mg/ml BSA, 3 ⁇ M orthovanadate) were added to each well of a 96 U bottom well plate. After 30 minutes at room temperature incubation, reaction was stopped and biotinylated peptide captured by adding 100 ⁇ l of bead suspension.
- the inhibition assay of Cdc7/dbf4 activity was performed according to the following protocol.
- Biotin-MCM2 substrate is trans-phosphorylated by the Cdc7/Dbf4 complex in the presence of ATP traced with ⁇ 33 -ATP.
- the phosphorylated Biotin-MCM2 substrate is then captured by Streptavidin-coated SPA beads and the extent of phosphorylation evaluated by ⁇ counting.
- the inhibition assay of Cdc7/dbf4 activity was performed in 96 wells plate according to the following protocol. To each well of the plate were added:
- test compound (12 increasing concentrations in the nM to ⁇ M range to generate a dose-response curve) - lO ⁇ l of a mixture of cold ATP (lO ⁇ M final concentration) and radioactive ATP (1/2500 molar ratio with cold ATP) was then used to start the reaction which was allowed to take place at 37°C.
- the solvent for test compounds also contained 10% DMSO.
- novel compounds of the invention are unexpectedly endowed with a cdk inhibitory activity significantly higher than that of the structurally closest prior art compounds of WO 02/12242 and are thus particularly advantageous, in therapy, against proliferative disorders associated with an altered cell cycle dependent kinase activity.
- the compounds of the present invention can be administered either as single agents or, alternatively, in combination with known anticancer treatments such as radiation therapy or chemotherapy regimen in combination with cytostatic or cytotoxic agents, antibiotic-
- alkylating agents e.g., alkylating agents, antimetabolite agents, hormonal agents, immunological agents, interferon-type agents, cyclooxygenase inhibitors (e.g. COX-2 inhibitors), matrixmetalloprotease inhibitors, telomerase inhibitors, tyrosine kinase inhibitors, anti- growth factor receptor agents, anti-HER agents, anti-EGFR agents, anti-angiogenesis agents (e.g. angiogenesis inhibitors), farnesyl transferase inhibitors, ras-raf signal transduction pathway inhibitors, cell cycle inhibitors, other cdks inhibitors, tubulin binding agents, topoisomerase I inhibitors, topoisomerase II inhibitors, and the like.
- cyclooxygenase inhibitors e.g. COX-2 inhibitors
- matrixmetalloprotease inhibitors e.g. COX-2 inhibitors
- telomerase inhibitors e.g. telomerase inhibitors
- the compounds of formula (I) of the present invention suitable for administration to a mammal, e.g., to humans, can be administered by the usual routes and the dosage level depends upon the age, weight, conditions of the patient and administration route.
- a suitable dosage adopted for oral administration of a compound of formula (I) may range from about 10 to about 500 mg per dose, from 1 to 5 times daily.
- the compounds of the invention can be administered in a variety of dosage forms, e.g., orally, in the form tablets, capsules, sugar or film coated tablets, liquid solutions or suspensions; rectally in the form suppositories; parenterally, e.g., intramuscularly, or through intravenous and/or intrathecal and/or intraspinal injection or infusion.
- the present invention also includes pharmaceutical compositions comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof in association with a pharmaceutically acceptable excipient, which may be a carrier or a diluent.
- compositions containing the compounds of the invention are usually prepared following conventional methods and are administered in a suitable pharmaceutical form.
- the solid oral forms may contain, together with the active compound, diluents, e.g., lactose, dextrose saccharose, sucrose, cellulose, corn starch or potato starch; lubricants, e.g., silica, talc, stearic acid, magnesium or calcium stearate, and/or polyethylene glycols; binding agents, e.g., starches, arabic gum, gelatine
- diluents e.g., lactose, dextrose saccharose, sucrose, cellulose, corn starch or potato starch
- lubricants e.g., silica, talc, stearic acid, magnesium or calcium stearate, and/or polyethylene glycols
- binding agents e.g., starches, arabic gum, gelatine
- methylcellulose, carboxymethylcellulose or polyvinyl py ⁇ olidone e.g., starch, alginic acid, alginates or sodium starch glycolate; effervescing mixtures; dyestuffs; sweeteners; wetting agents such as lecithin, polysorbates, laurylsulphates; and, in general, non-toxic and pharmacologically inactive substances used in pharmaceutical formulations.
- disintegrating agents e.g., starch, alginic acid, alginates or sodium starch glycolate; effervescing mixtures; dyestuffs; sweeteners; wetting agents such as lecithin, polysorbates, laurylsulphates; and, in general, non-toxic and pharmacologically inactive substances used in pharmaceutical formulations.
- disintegrating agents e.g., starch, alginic acid, alginates or sodium starch glycolate
- effervescing mixtures e.g., dyestuffs; sweeten
- the liquid dispersions for oral administration may be, e.g., syrups, emulsions and suspensions.
- the syrups may contain, as carrier, saccharose or saccharose with glycerine and/or mannitol and sorbitol.
- the suspensions and the emulsions may contain, as examples of carriers, natural gum, agar, sodium alginate, pectin, methylcellulose, carboxymethylcellulose, or polyvinyl alcohol.
- the suspension or solutions for intramuscular injections may contain, together with the active compound, a pharmaceutically acceptable carrier, e.g., sterile water, olive oil, ethyl oleate, glycols, e.g., propylene glycol and, if desired, a suitable amount of lidocaine hydrochloride.
- the solutions for intravenous injections or infusions may contain, as a carrier, sterile water or preferably they may be in the form of sterile, aqueous, isotonic, saline solutions or they may contain propylene glycol as a carrier.
- the suppositories may contain, together with the active compound, a pharmaceutically acceptable carrier, e.g., cocoa butter, polyethylene glycol, a polyoxyethylene sorbitan fatty acid ester surfactant or lecithin.
- Each code identifies a single specific compound of formula (la) or (lb) and consists of three units A-M-B.
- A represents any substituent R [see formula (la) or (lb)] and is attached to the rest of the molecule through the -NH-group; each specific A group is represented and consecutively numbered in the following table I.
- B represents any substituent Ri [see formula (la) or (lb)] and is attached to the rest of the molecule through the carbonyl (CO) group; each specific B group is represented and consecutively numbered in the following table II.
- M refers to the central core of the divalent moiety which is substituted by groups A and B; in particular, M may vary from Ml or M2 as per the formulae below, each identifying the central core of a compound having formula (la) or (lb), respectively:
- the compound A06-M1-B01 of table III represents the compound of formula (la) having the central Ml core, being substituted by the group A06 and by the group BOl, in the positions indicated by the anows;
- the compound A04-M2-B08 of table V represents the compound of formula (lb) having the central M2 core, being substituted by the group A04 and by the group B08, in the positions indicated by the anows:
- Example 15 4-(4-Hydroxy-piperidm-l-yl)benzoic acid ethyl ester
- a mixture of 4-fluoro-benzoic acid ethyl ester (1.68 g, 10 mmoles), piperidin-4-ol (1.12 g, 11 mmoles) and anhydrous potassium carbonate (1.38 g, 10 mmoles) in DMSO (10 ml) was heated at 120°C for 6 hours. After cooling, the mixture was poured into water and ice (500 ml) and extracted with ethyl acetate. The organic layer was washed with water and brine, dried and evaporated. The residue was purified by flash chromatography on silica gel using hexane EtOAc (10/30) as eluent to give the title compound as a white solid (1.6 g, 64%).
- the HPLC equipment consisted of a Waters 2790 HPLC system equipped with a 996
- HPLC HPLC was carried out at 25°C at a flow rate of 1 ml/min using a RP18 Waters X Te ⁇ a (4,6 x 50 mm, 3.5 ⁇ m) column.
- Mobile phase A was ammonium acetate 5 mM buffer (pH 5.5 with acetic acid/acetonitrile 95:5), and
- Mobile phase B was H 2 0/acetonitrile (5:95); the gradient was from 10 to 90% B in 8 minutes then hold 90% B 2 minutes.
- the injection volume was 10 ⁇ l.
- the mass spectometer was operated in positive and in negative ion mode, the capillary voltage was set up at 2.5 KV; the source temperature was 120°C; cone was 10 V; full scan, mass range from 100 to 800 amu was set up.
- HPLC/MS Method 2 The HPLC equipment consisted of a Waters 2790 HPLC system equipped with a 996 Waters PDA detector and Micromass mod. ZQ single quadrupole mass spectrometer, equipped with an electrospray (ESI) ion source. Instrument control, data acquisition and data processing were providen by Millennium 4.0 and MassLynx 3.5 software.
- HPLC was carried out at 25°C at a flow rate of 1 ml/min using a RP18 Waters X Te ⁇ a (4,6 x 50 mm, 3.5 ⁇ m) column.
- Mobile phase A was ammonium acetate 5 mM buffer (pH 5.5 with acetic acid/acetonitrile 95:5), and Mobile phase B was H 2 0/acetonitrate (5:95); the gradient was from 10 to 90% B in 4 min then hold 90% B 1 minute.
- the injection volume was 10 ⁇ l.
- the mass spectometer was operated in positive and in negative ion mode, the capillary voltage was set up at 2.5 KV; the source temperature was 120°C; cone was 10 V; full scan, mass range from 100 to 800 amu was set up.
- Mass spectra were recorded on a Finnigan LCQ ion trap mass spectrometer using the electrospray (ESI) ionization technique with positive and negative ion detection.
- the mass spectrometer was directly connected to a SSP4000 HPLC system (Thermo Separation), equipped with an LcPal autosampler (CTC Analytics) and a UV 6000LP PDA detector (Thermo Separation). Instrument control, data acquisition and processing were performed by using Xcalibur 1.2 software.
- HPLC analysis were carried out at room temperature at a flow rate of 1 ml/min using an RP C18 Waters X-Te ⁇ a column (4.6x 50mm; 3.5 ⁇ m).
- Mobile phase A was ammonium acetate 5 mM buffer (pH 5.5 with acetic acid): acetonitrile 90:10
- Mobile phase B was ammonium acetate 5 mM buffer (pH 5.5 with acetic acid): acetonitrile 10:90; the gradient was from 0 to 100% B in 7 minutes then hold 100% B for 2 minutes before requilibration. Total LC time is 12 minutes.
- the injection volume was 1 O ⁇ l. UV Detection was performed between 215 and 400 nm.
- Ions were generated under the following conditions: ESI sprayer voltage 4.0 kV, heated capillary temperature 255°C, sheath gas nitrogen with a pressure of 5.0 Bar. A full scan detection mode (from 50 to 1000 amu) was used with an MS/MS analysis of the most intense ion (normalized collision energy: 35%). UV Detection: 215-400 nm. HPLC/MS Method 4
- the HPLC system used (Alliance 2790, with thermostated autosampler and divert valve LabPro, UV detector 2487 and satin interface, ZQ mass spectrometer with ESI interface) is a product of Waters Inc., Milford, Massachusetts.
- the chemiluminescent nitrogen detector (CLND) mod. 8060 is a product of ANTEK Instruments Inc., Houston, Texas.
- the Uquid handler Miniprep 75 is a product of Tecan Group Ltd., Maennedorf, Switzerland.
- the chromatographic conditions used were as follows: The flow rate was set at 1 mL/min.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Immunology (AREA)
- Epidemiology (AREA)
- Biomedical Technology (AREA)
- Oncology (AREA)
- Neurology (AREA)
- Dermatology (AREA)
- Neurosurgery (AREA)
- Virology (AREA)
- Communicable Diseases (AREA)
- Rheumatology (AREA)
- Urology & Nephrology (AREA)
- Radiology & Medical Imaging (AREA)
- Pathology (AREA)
- Physical Education & Sports Medicine (AREA)
- Psychiatry (AREA)
- Transplantation (AREA)
- Heart & Thoracic Surgery (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Hospice & Palliative Care (AREA)
- Pain & Pain Management (AREA)
- Cardiology (AREA)
- Molecular Biology (AREA)
- Pulmonology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
Claims
Priority Applications (27)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| UAA200507098A UA81790C2 (en) | 2002-12-19 | 2003-04-12 | Substituted derivatives of pyrolopyrazol as kinaze inhibitors |
| HR20050562A HRP20050562A2 (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| ES03799536.2T ES2605848T3 (en) | 2002-12-19 | 2003-12-04 | Pyrrolo-pyrazole derivatives substituted as kinase inhibitors |
| MXPA05006791A MXPA05006791A (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors. |
| BR0317455-7A BR0317455A (en) | 2002-12-19 | 2003-12-04 | Pyrrolpyrazole derivatives substituted as kinase inhibitors |
| KR1020057011433A KR101223826B1 (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| EA200501009A EA010596B1 (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| CN2003801064477A CN1726217B (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolopyrazole derivatives useful as kinase inhibitors |
| EP03799536.2A EP1575954B1 (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| HK06106083.9A HK1086256B (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| JP2004561484A JP4739761B2 (en) | 2002-12-19 | 2003-12-04 | Pyrrolo-pyrazole substituted derivatives as kinase inhibitors |
| AU2003300255A AU2003300255B9 (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| AP2005003343A AP2005003343A0 (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinaseinhibitors |
| YUP-2005/0462A RS20050462A (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| NZ540644A NZ540644A (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| MEP-524/08A MEP52408A (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| CA2508069A CA2508069C (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| US10/539,145 US7407971B2 (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| IS7857A IS7857A (en) | 2002-12-19 | 2005-05-23 | Installs pyrrolo-pyrazole derivatives that inhibit kinase |
| IL169019A IL169019A (en) | 2002-12-19 | 2005-06-06 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| NO20052805A NO333178B1 (en) | 2002-12-19 | 2005-06-09 | Substituted pyrrolo-pyrazole derivatives, pharmaceutical compositions comprising such, such compounds for use as a medicament and the use thereof in the manufacture of medicament having antitumor activity |
| TNP2005000163A TNSN05163A1 (en) | 2002-12-19 | 2005-06-17 | SUBSTITUTED PYRROLO-PYRAZOLE DERIVATIVES AS INHIBITORS OF KINASES |
| US12/181,772 US8557857B2 (en) | 2002-12-19 | 2008-07-29 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| IL215617A IL215617A0 (en) | 2002-12-19 | 2011-10-06 | Substituted pyrrolo-pyrazole derivatives and process for their preparation |
| US13/614,499 US8481585B2 (en) | 2002-12-19 | 2012-09-13 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| US13/614,545 US8557845B2 (en) | 2002-12-19 | 2012-09-13 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| US14/010,819 US8785448B2 (en) | 2002-12-19 | 2013-08-27 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US43495202P | 2002-12-19 | 2002-12-19 | |
| US60/434,952 | 2002-12-19 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10539145 A-371-Of-International | 2003-12-04 | ||
| US12/181,772 Continuation US8557857B2 (en) | 2002-12-19 | 2008-07-29 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2004056827A2 true WO2004056827A2 (en) | 2004-07-08 |
| WO2004056827A3 WO2004056827A3 (en) | 2004-10-28 |
Family
ID=32682128
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2003/050942 Ceased WO2004056827A2 (en) | 2002-12-19 | 2003-12-04 | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
Country Status (33)
| Country | Link |
|---|---|
| US (5) | US7407971B2 (en) |
| EP (2) | EP2266987B1 (en) |
| JP (2) | JP4739761B2 (en) |
| KR (2) | KR20120038465A (en) |
| CN (1) | CN1726217B (en) |
| AP (1) | AP2005003343A0 (en) |
| AR (1) | AR042525A1 (en) |
| AU (1) | AU2003300255B9 (en) |
| BR (1) | BR0317455A (en) |
| CA (1) | CA2508069C (en) |
| CR (2) | CR7880A (en) |
| CU (1) | CU23550B7 (en) |
| EA (1) | EA010596B1 (en) |
| EC (1) | ECSP055864A (en) |
| ES (2) | ES2635191T3 (en) |
| GE (1) | GEP20084435B (en) |
| HR (1) | HRP20050562A2 (en) |
| IL (2) | IL169019A (en) |
| IS (1) | IS7857A (en) |
| MA (1) | MA27569A1 (en) |
| ME (1) | MEP52408A (en) |
| MX (1) | MXPA05006791A (en) |
| MY (1) | MY141196A (en) |
| NO (1) | NO333178B1 (en) |
| NZ (1) | NZ540644A (en) |
| OA (1) | OA13015A (en) |
| PL (1) | PL377285A1 (en) |
| RS (1) | RS20050462A (en) |
| TN (1) | TNSN05163A1 (en) |
| TW (1) | TWI327470B (en) |
| UA (1) | UA81790C2 (en) |
| WO (1) | WO2004056827A2 (en) |
| ZA (1) | ZA200504298B (en) |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006072831A1 (en) | 2005-01-10 | 2006-07-13 | Pfizer Inc. | Pyrrolopyrazoles, potent kinase inhibitors |
| WO2007099171A2 (en) | 2006-03-03 | 2007-09-07 | Nerviano Medical Sciences S.R.L. | Bicyclo-pyrazoles active as kinase inhibitors |
| WO2007072153A3 (en) * | 2005-12-21 | 2007-10-25 | Pfizer Prod Inc | Carbonylamino pyrrolopyrazoles, potent kinase inhibitors |
| WO2008043745A1 (en) * | 2006-10-11 | 2008-04-17 | Nerviano Medical Sciences S.R.L. | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| WO2008096260A1 (en) * | 2007-02-07 | 2008-08-14 | Pfizer Inc. | 3-amino-pyrrolo[3,4-c] pyrazole- 5 (1h, 4h, 6h) carbaldehyde derivatives as pkc inhibitors |
| US7468376B2 (en) | 2003-02-27 | 2008-12-23 | Palau Pharma, S.A. | Pyrazolopyridine derivates |
| US7550598B2 (en) | 2004-08-18 | 2009-06-23 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| US7572914B2 (en) | 2003-12-19 | 2009-08-11 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| US7612200B2 (en) | 2004-12-07 | 2009-11-03 | Locus Pharmaceuticals, Inc. | Inhibitors of protein kinases |
| US7652135B2 (en) | 2003-09-23 | 2010-01-26 | Vertex Pharmaceuticals Incorporated | Compositions useful as inhibitors of protein kinases |
| US7713973B2 (en) | 2004-10-15 | 2010-05-11 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| US7741479B2 (en) | 2004-12-07 | 2010-06-22 | Locus Pharmaceuticals, Inc. | Urea inhibitors of MAP kinases |
| CN101346382B (en) * | 2005-12-21 | 2011-07-06 | 辉瑞产品公司 | Carbonylamino pyrrolopyrazoles, potent kinase inhibitors |
| AU2008237660B2 (en) * | 2007-04-12 | 2011-12-22 | Pfizer Inc. | 3-amido-pyrrolo [3, 4-C] pyrazole-5 (1H, 4H, 6H) carbaldehyde derivatives as inhibitors of protein kinase C |
| US8119655B2 (en) | 2005-10-07 | 2012-02-21 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| US8278450B2 (en) | 2007-04-18 | 2012-10-02 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| CN101646673B (en) * | 2007-02-07 | 2014-06-25 | 辉瑞大药厂 | 3-Amino-pyrrolo[3,4-C]pyrazole-5(1H,4H,6H)carbaldehyde derivatives as PKC inhibitors |
| KR20170077168A (en) | 2014-10-31 | 2017-07-05 | 우베 고산 가부시키가이샤 | Substituted dihydropyrrolopyrazole compound |
| KR20180018661A (en) | 2015-06-15 | 2018-02-21 | 우베 고산 가부시키가이샤 | Substituted dihydropyrrolopyrazole derivatives |
| WO2020100944A1 (en) | 2018-11-14 | 2020-05-22 | 宇部興産株式会社 | Dihydropyrrolopyrazole derivative |
Families Citing this family (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2349762T3 (en) * | 2000-08-10 | 2011-01-11 | Pfizer Italia S.R.L. | BICYCLE-PYRAZOLES ACTIVE AS KINASE INHIBITORS, PROCEDURES FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS THAT INCLUDE THEM. |
| UA81790C2 (en) | 2002-12-19 | 2008-02-11 | Фармация Италия С.П.А. | Substituted derivatives of pyrolopyrazol as kinaze inhibitors |
| MXPA05009719A (en) * | 2003-03-11 | 2005-10-18 | Pharmacia Italia Spa | Bicyclo-pyrazole derivatives active as kinase inhibitors, process for their preparation and pharmaceutical compositions comprising them. |
| CA2559282A1 (en) * | 2004-03-18 | 2005-09-29 | Brigham And Women's Hospital, Inc. | Methods for the treatment of synucleinopathies |
| US20060106060A1 (en) * | 2004-03-18 | 2006-05-18 | The Brigham And Women's Hospital, Inc. | Methods for the treatment of synucleinopathies (Lansbury) |
| US20050272722A1 (en) * | 2004-03-18 | 2005-12-08 | The Brigham And Women's Hospital, Inc. | Methods for the treatment of synucleinopathies |
| US20070293539A1 (en) * | 2004-03-18 | 2007-12-20 | Lansbury Peter T | Methods for the treatment of synucleinopathies |
| CA2559285A1 (en) * | 2004-03-18 | 2005-09-29 | Brigham And Women's Hospital, Inc. | Methods for the treatment of synucleinopathies |
| WO2009151683A2 (en) * | 2008-03-12 | 2009-12-17 | Link Medicine Corporation | Quinolinone farnesyl transferase inhibitors for the treatment of synucleinopathies and other indications |
| US20110060005A1 (en) * | 2008-11-13 | 2011-03-10 | Link Medicine Corporation | Treatment of mitochondrial disorders using a farnesyl transferase inhibitor |
| JP2012508768A (en) * | 2008-11-13 | 2012-04-12 | リンク・メディスン・コーポレーション | Azaquinolinone derivatives and uses thereof |
| US20100331363A1 (en) * | 2008-11-13 | 2010-12-30 | Link Medicine Corporation | Treatment of mitochondrial disorders using a farnesyl transferase inhibitor |
| US9180127B2 (en) | 2009-12-29 | 2015-11-10 | Dana-Farber Cancer Institute, Inc. | Type II Raf kinase inhibitors |
| CA2856291C (en) | 2011-11-17 | 2020-08-11 | Dana-Farber Cancer Institute, Inc. | Inhibitors of c-jun-n-terminal kinase (jnk) |
| EP2909194A1 (en) | 2012-10-18 | 2015-08-26 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 (cdk7) |
| US9758522B2 (en) | 2012-10-19 | 2017-09-12 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged small molecules as inducers of protein degradation |
| US10000483B2 (en) | 2012-10-19 | 2018-06-19 | Dana-Farber Cancer Institute, Inc. | Bone marrow on X chromosome kinase (BMX) inhibitors and uses thereof |
| US10047070B2 (en) | 2013-10-18 | 2018-08-14 | Dana-Farber Cancer Institute, Inc. | Polycyclic inhibitors of cyclin-dependent kinase 7 (CDK7) |
| EP3057955B1 (en) | 2013-10-18 | 2018-04-11 | Syros Pharmaceuticals, Inc. | Heteroaromatic compounds useful for the treatment of prolferative diseases |
| US9862688B2 (en) | 2014-04-23 | 2018-01-09 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged janus kinase inhibitors and uses thereof |
| US10017477B2 (en) | 2014-04-23 | 2018-07-10 | Dana-Farber Cancer Institute, Inc. | Janus kinase inhibitors and uses thereof |
| KR102451223B1 (en) * | 2014-05-23 | 2022-10-05 | 밍사이트 파마슈티칼스, 인크. | Treatment of autoimmune disease |
| AU2015371251B2 (en) * | 2014-12-23 | 2020-06-11 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 (CDK7) |
| HK1246645A1 (en) | 2015-03-27 | 2018-09-14 | 达纳-法伯癌症研究所股份有限公司 | Inhibitors of cyclin-dependent kinases |
| US10702527B2 (en) | 2015-06-12 | 2020-07-07 | Dana-Farber Cancer Institute, Inc. | Combination therapy of transcription inhibitors and kinase inhibitors |
| KR101845931B1 (en) * | 2015-06-18 | 2018-04-05 | 한국화학연구원 | Heteroaryl derivatives or pharmaceutically acceptable salts thereof, preparation method thereof and pharmaceutical composition for use in preventing or treating PI3 kinase related diseases |
| EP3347018B1 (en) | 2015-09-09 | 2021-09-01 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| SG11201804210XA (en) * | 2015-11-20 | 2018-06-28 | Mingsight Pharmaceuticals Inc | Treatment of autoimmune disease |
| JP2019112306A (en) * | 2016-04-28 | 2019-07-11 | 宇部興産株式会社 | Pharmaceutical composition for treating and/or preventing chronic obstructive pulmonary disease |
| JP2019112305A (en) * | 2016-04-28 | 2019-07-11 | 宇部興産株式会社 | Pharmaceutical composition for treating and/or preventing atopic dermatitis |
| JP2019112307A (en) * | 2016-04-28 | 2019-07-11 | 宇部興産株式会社 | Pharmaceutical composition administered in combination with substituted dihydro pyrrolopyrazole compound and other psoriasis therapeutic agents |
| CN107337674B (en) * | 2016-04-29 | 2019-09-20 | 江苏吉贝尔药业股份有限公司 | For the oxinane amine derivative of DPP-IV inhibitor, its pharmaceutical composition and preparation and purposes |
| US12187701B2 (en) | 2018-06-25 | 2025-01-07 | Dana-Farber Cancer Institute, Inc. | Taire family kinase inhibitors and uses thereof |
| JP7660063B2 (en) | 2018-12-28 | 2025-04-10 | ダナ-ファーバー キャンサー インスティテュート, インコーポレイテッド | Inhibitors of cyclin-dependent kinase 7 and their uses |
| EP4003335A4 (en) * | 2019-07-23 | 2024-01-10 | Dana-Farber Cancer Institute, Inc. | CYCLINE-DEPENDENT KINASE 7 INHIBITORS AND THEIR USES |
| WO2022005175A1 (en) | 2020-06-30 | 2022-01-06 | 보령제약 주식회사 | Method for preparing compound as pi3k inhibitor and intermediate compound for preparing same |
| CN115073471B (en) * | 2021-03-11 | 2023-03-21 | 沈阳药科大学 | Pyrazolotetrahydropyrrole derivatives, preparation method and application in medicine |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3526633A (en) * | 1968-03-06 | 1970-09-01 | American Cyanamid Co | Substituted 2,4,5,6-tetrahydropyrrolo(3,4-c)pyrazoles |
| GB9911053D0 (en) | 1999-05-12 | 1999-07-14 | Pharmacia & Upjohn Spa | 4,5,6,7-tetrahydroindazole derivatives process for their preparation and their use as antitumour agents |
| US6387900B1 (en) | 1999-08-12 | 2002-05-14 | Pharmacia & Upjohn S.P.A. | 3(5)-ureido-pyrazole derivatives process for their preparation and their use as antitumor agents |
| KR20020060160A (en) | 1999-08-12 | 2002-07-16 | 파마시아 이탈리아 에스.피.에이. | 3(5)-Amino-pyrazole derivatives, process for their preparation and their use as antitumor agents |
| ES2349762T3 (en) * | 2000-08-10 | 2011-01-11 | Pfizer Italia S.R.L. | BICYCLE-PYRAZOLES ACTIVE AS KINASE INHIBITORS, PROCEDURES FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS THAT INCLUDE THEM. |
| AU2002215053A1 (en) | 2000-11-27 | 2002-06-24 | Pharmacia Italia S.P.A. | Phenylacetamido- pyrazole derivatives and their use as antitumor agents |
| CA2434066A1 (en) * | 2001-01-26 | 2002-09-12 | Pharmacia Italia S.P.A. | Chromane derivatives, process for their preparation and their use as antitumor agents |
| WO2003028720A1 (en) | 2001-09-26 | 2003-04-10 | Pharmacia Italia S.P.A. | Aminoindazole derivatives active as kinase inhibitors, process for their preparation and pharmaceutical compositions containing them |
| UA81790C2 (en) * | 2002-12-19 | 2008-02-11 | Фармация Италия С.П.А. | Substituted derivatives of pyrolopyrazol as kinaze inhibitors |
-
2003
- 2003-04-12 UA UAA200507098A patent/UA81790C2/en unknown
- 2003-12-04 AP AP2005003343A patent/AP2005003343A0/en unknown
- 2003-12-04 GE GEAP20038905A patent/GEP20084435B/en unknown
- 2003-12-04 EP EP10179134.1A patent/EP2266987B1/en not_active Expired - Lifetime
- 2003-12-04 CN CN2003801064477A patent/CN1726217B/en not_active Expired - Lifetime
- 2003-12-04 US US10/539,145 patent/US7407971B2/en not_active Expired - Lifetime
- 2003-12-04 ES ES10179134.1T patent/ES2635191T3/en not_active Expired - Lifetime
- 2003-12-04 WO PCT/EP2003/050942 patent/WO2004056827A2/en not_active Ceased
- 2003-12-04 AU AU2003300255A patent/AU2003300255B9/en not_active Ceased
- 2003-12-04 ME MEP-524/08A patent/MEP52408A/en unknown
- 2003-12-04 KR KR1020127002938A patent/KR20120038465A/en not_active Ceased
- 2003-12-04 RS YUP-2005/0462A patent/RS20050462A/en unknown
- 2003-12-04 NZ NZ540644A patent/NZ540644A/en not_active IP Right Cessation
- 2003-12-04 HR HR20050562A patent/HRP20050562A2/en not_active Application Discontinuation
- 2003-12-04 EA EA200501009A patent/EA010596B1/en not_active IP Right Cessation
- 2003-12-04 PL PL377285A patent/PL377285A1/en unknown
- 2003-12-04 ES ES03799536.2T patent/ES2605848T3/en not_active Expired - Lifetime
- 2003-12-04 EP EP03799536.2A patent/EP1575954B1/en not_active Expired - Lifetime
- 2003-12-04 BR BR0317455-7A patent/BR0317455A/en not_active IP Right Cessation
- 2003-12-04 OA OA1200500186A patent/OA13015A/en unknown
- 2003-12-04 CA CA2508069A patent/CA2508069C/en not_active Expired - Fee Related
- 2003-12-04 MX MXPA05006791A patent/MXPA05006791A/en active IP Right Grant
- 2003-12-04 JP JP2004561484A patent/JP4739761B2/en not_active Expired - Fee Related
- 2003-12-04 KR KR1020057011433A patent/KR101223826B1/en not_active Expired - Fee Related
- 2003-12-05 MY MYPI20034670A patent/MY141196A/en unknown
- 2003-12-16 TW TW092135527A patent/TWI327470B/en not_active IP Right Cessation
- 2003-12-18 AR ARP030104702A patent/AR042525A1/en unknown
-
2005
- 2005-05-23 IS IS7857A patent/IS7857A/en unknown
- 2005-05-26 ZA ZA200504298A patent/ZA200504298B/en unknown
- 2005-06-06 IL IL169019A patent/IL169019A/en not_active IP Right Cessation
- 2005-06-09 NO NO20052805A patent/NO333178B1/en not_active IP Right Cessation
- 2005-06-14 CU CU20050117A patent/CU23550B7/en not_active IP Right Cessation
- 2005-06-17 MA MA28347A patent/MA27569A1/en unknown
- 2005-06-17 TN TNP2005000163A patent/TNSN05163A1/en unknown
- 2005-06-17 CR CR7880A patent/CR7880A/en unknown
- 2005-06-17 EC EC2005005864A patent/ECSP055864A/en unknown
-
2008
- 2008-07-29 US US12/181,772 patent/US8557857B2/en not_active Expired - Fee Related
- 2008-10-16 CR CR10370A patent/CR10370A/en not_active Application Discontinuation
-
2011
- 2011-02-14 JP JP2011028813A patent/JP2011144180A/en not_active Withdrawn
- 2011-10-06 IL IL215617A patent/IL215617A0/en unknown
-
2012
- 2012-09-13 US US13/614,499 patent/US8481585B2/en not_active Expired - Fee Related
- 2012-09-13 US US13/614,545 patent/US8557845B2/en not_active Expired - Fee Related
-
2013
- 2013-08-27 US US14/010,819 patent/US8785448B2/en not_active Expired - Lifetime
Cited By (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7468376B2 (en) | 2003-02-27 | 2008-12-23 | Palau Pharma, S.A. | Pyrazolopyridine derivates |
| US8536194B2 (en) | 2003-02-27 | 2013-09-17 | Palau Pharma, S.A. | Pyrazolopyridine derivates |
| US8642779B2 (en) | 2003-09-23 | 2014-02-04 | Vertex Pharmaceuticals Incorporated | Compositions useful as inhibitors of protein kinases |
| US7652135B2 (en) | 2003-09-23 | 2010-01-26 | Vertex Pharmaceuticals Incorporated | Compositions useful as inhibitors of protein kinases |
| US7572914B2 (en) | 2003-12-19 | 2009-08-11 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| US7550598B2 (en) | 2004-08-18 | 2009-06-23 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| US8288536B2 (en) | 2004-10-15 | 2012-10-16 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| US7713973B2 (en) | 2004-10-15 | 2010-05-11 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| US7612200B2 (en) | 2004-12-07 | 2009-11-03 | Locus Pharmaceuticals, Inc. | Inhibitors of protein kinases |
| US7741479B2 (en) | 2004-12-07 | 2010-06-22 | Locus Pharmaceuticals, Inc. | Urea inhibitors of MAP kinases |
| WO2006072831A1 (en) | 2005-01-10 | 2006-07-13 | Pfizer Inc. | Pyrrolopyrazoles, potent kinase inhibitors |
| EA011815B1 (en) * | 2005-01-10 | 2009-06-30 | Пфайзер Инк. | Pyrrolopyrazoles, potent kinase inhibitors |
| US8067591B2 (en) | 2005-01-10 | 2011-11-29 | Agouron Pharmaceuticals, Inc. | Pyrrolopyrazole, potent kinase inhibitors |
| KR100919343B1 (en) * | 2005-01-10 | 2009-09-25 | 화이자 인코포레이티드 | Pyrrolopyrazoles, potent kinase inhibitors |
| US8530652B2 (en) | 2005-01-10 | 2013-09-10 | Agouron Pharmaceuticals, Inc. | Pyrrolopyrazoles, potent kinase inhibitors |
| CN101115760B (en) * | 2005-01-10 | 2010-11-03 | 辉瑞大药厂 | Kinase inhibitors pyrrolopyrazoles |
| US8119655B2 (en) | 2005-10-07 | 2012-02-21 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| AP2369A (en) * | 2005-12-21 | 2012-02-29 | Pfizer Prod Inc | Carbonylamino pyrrolopyrazoles, potent kinase inhibitors. |
| AU2006327866B2 (en) * | 2005-12-21 | 2012-06-14 | Pfizer Products Inc. | Carbonylamino pyrrolopyrazoles, potent kinase inhibitors |
| EA015513B1 (en) * | 2005-12-21 | 2011-08-30 | Пфайзер Продактс Инк. | Carbonylaminopyrrolopyrazoles as effective kinase inhibitors |
| US7884117B2 (en) | 2005-12-21 | 2011-02-08 | Pfizer Inc. | Carbonylamino pyrrolopyrazoles, potent kinase inhibitors |
| CN101346382B (en) * | 2005-12-21 | 2011-07-06 | 辉瑞产品公司 | Carbonylamino pyrrolopyrazoles, potent kinase inhibitors |
| WO2007072153A3 (en) * | 2005-12-21 | 2007-10-25 | Pfizer Prod Inc | Carbonylamino pyrrolopyrazoles, potent kinase inhibitors |
| JP2009520805A (en) * | 2005-12-21 | 2009-05-28 | ファイザー・プロダクツ・インク | Carbonylaminopyrrolopyrazole, an effective kinase inhibitor |
| WO2007099171A2 (en) | 2006-03-03 | 2007-09-07 | Nerviano Medical Sciences S.R.L. | Bicyclo-pyrazoles active as kinase inhibitors |
| WO2007099171A3 (en) * | 2006-03-03 | 2007-11-15 | Nerviano Medical Sciences Srl | Bicyclo-pyrazoles active as kinase inhibitors |
| US9012487B2 (en) | 2006-03-03 | 2015-04-21 | Nerviano Medical Sciences S.R.L. | Bicyclo-pyrazoles active as kinase inhibitors |
| US8227472B2 (en) | 2006-10-11 | 2012-07-24 | Nerviano Medical Sciences S.R.L. | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| US8623876B2 (en) | 2006-10-11 | 2014-01-07 | Nerviano Medical Sciences S.R.L. | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| WO2008043745A1 (en) * | 2006-10-11 | 2008-04-17 | Nerviano Medical Sciences S.R.L. | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors |
| US11780853B2 (en) | 2007-02-07 | 2023-10-10 | Pfizer Inc. | Substituted N-pyrimidin-4-yl-3-aminopyrrolo[3,4-C]pyrazoles as protein kinase C inhibitors |
| EA017937B1 (en) * | 2007-02-07 | 2013-04-30 | Пфайзер Инк. | 3-AMINOPYRROLO[3,4-c]PYRAZOLE-5(1H,4H,6H)-CARBALDEHYDE DERIVATIVES AS PKC INHIBITORS |
| US11220518B2 (en) | 2007-02-07 | 2022-01-11 | Pfizer Inc. | Substituted n-pyrimidin-4-yl-3-amino-pyrrolo[3,4-c]pyrazoles as protein kinase C inhibitors |
| US9518060B2 (en) | 2007-02-07 | 2016-12-13 | Pfizer Inc. | Substituted pyrrolo[3,4-c]pyrazoles as PKC kinase inhibitors |
| US10316045B2 (en) | 2007-02-07 | 2019-06-11 | Pfizer Inc. | Substituted pyrrolo[3,4-c]pyrazoles as pkc kinase inhibitors |
| CN101646673B (en) * | 2007-02-07 | 2014-06-25 | 辉瑞大药厂 | 3-Amino-pyrrolo[3,4-C]pyrazole-5(1H,4H,6H)carbaldehyde derivatives as PKC inhibitors |
| US8877761B2 (en) | 2007-02-07 | 2014-11-04 | Pfizer Inc. | N-pyrimidin-4-yl-3-amino-pyrrolo[3,4-c]pyrazole derivatives as PKC kinase inhibitors |
| US8183255B2 (en) | 2007-02-07 | 2012-05-22 | Pfizer, Inc. | N-pyrimidin-4-yl-3-amino-pyrrolo[3,4-c]pyrazole derivatives as PKC kinase inhibitors |
| WO2008096260A1 (en) * | 2007-02-07 | 2008-08-14 | Pfizer Inc. | 3-amino-pyrrolo[3,4-c] pyrazole- 5 (1h, 4h, 6h) carbaldehyde derivatives as pkc inhibitors |
| US8999981B2 (en) | 2007-04-12 | 2015-04-07 | Pfizer Inc. | 3-amido-pyrrolo[3,4-C]pyrazole-5(1H, 4H,6H) carbaldehyde derivatives |
| AU2008237660B2 (en) * | 2007-04-12 | 2011-12-22 | Pfizer Inc. | 3-amido-pyrrolo [3, 4-C] pyrazole-5 (1H, 4H, 6H) carbaldehyde derivatives as inhibitors of protein kinase C |
| US8114871B2 (en) * | 2007-04-12 | 2012-02-14 | Pfizer Inc. | 3-amido-pyrrolo[3,4-C]pyrazole-5(1H,4H,6H) carbaldehyde derivatives |
| US8278450B2 (en) | 2007-04-18 | 2012-10-02 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| KR20170077168A (en) | 2014-10-31 | 2017-07-05 | 우베 고산 가부시키가이샤 | Substituted dihydropyrrolopyrazole compound |
| US10131679B2 (en) | 2014-10-31 | 2018-11-20 | Ube Industries, Ltd. | Substituted dihydropyrrolopyrazole compound |
| US10968238B2 (en) | 2014-10-31 | 2021-04-06 | Ube Industries, Ltd. | Substituted dihydropyrrolopyrazole compound |
| KR20180018661A (en) | 2015-06-15 | 2018-02-21 | 우베 고산 가부시키가이샤 | Substituted dihydropyrrolopyrazole derivatives |
| US10273252B2 (en) | 2015-06-15 | 2019-04-30 | Ube Industries, Ltd. | Substituted dihydropyrrolopyrazole derivative |
| US10894798B2 (en) | 2015-06-15 | 2021-01-19 | Ube Industries, Ltd. | Substituted dihydropyrrolopyrazole derivative |
| RU2765718C1 (en) * | 2015-06-15 | 2022-02-02 | Убе Индастриз, Лтд. | Substituted dihydropyrrolopyrazole derivative |
| WO2020100944A1 (en) | 2018-11-14 | 2020-05-22 | 宇部興産株式会社 | Dihydropyrrolopyrazole derivative |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1575954B1 (en) | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors | |
| EP1309590B1 (en) | Azaindole derivatives, process for their preparation, and their use as antitumor agents | |
| OA13170A (en) | Pyrazolo-quinazoline derivatives, process for their preparation and their use as kinase inhibitors. | |
| AU2001266079A1 (en) | Azaindole derivatives, process for their preparation, and their use as antitumor agents | |
| WO2008043745A1 (en) | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors | |
| WO2003014090A1 (en) | Amino-phthalazinone derivatives active as kinase inhibitors, process for their preparation and pharmaceutical compositions containing them | |
| HK1086256B (en) | Substituted pyrrolo-pyrazole derivatives as kinase inhibitors | |
| AU2002325383A1 (en) | Amino-phthalazinone derivatives active as kinase inhibitors process for their preparation and pharmaceutical compositions containing them |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: P-2005/0462 Country of ref document: YU |
|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005/04298 Country of ref document: ZA Ref document number: 200504298 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2508069 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2318/DELNP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 169019 Country of ref document: IL Ref document number: 2003300255 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 540644 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: P20050562A Country of ref document: HR Ref document number: AP/P/2005/003343 Country of ref document: AP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 377285 Country of ref document: PL Ref document number: 1020057011433 Country of ref document: KR Ref document number: 20038A64477 Country of ref document: CN Ref document number: 05059507 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: DZP5005000213 Country of ref document: DZ Ref document number: DZP2005000213 Country of ref document: DZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2005/006791 Country of ref document: MX Ref document number: 2004561484 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200501009 Country of ref document: EA |
|
| REEP | Request for entry into the european phase |
Ref document number: 2003799536 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 8905 Country of ref document: GE Ref document number: 2003799536 Country of ref document: EP Ref document number: 20050269 Country of ref document: UZ Ref document number: 1200501021 Country of ref document: VN |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020057011433 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003799536 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1-2005-501127 Country of ref document: PH |
|
| ENP | Entry into the national phase |
Ref document number: PI0317455 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007004705 Country of ref document: US Ref document number: 10539145 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 10539145 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: CR2008-010370 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 215617 Country of ref document: IL |