WO2005122770A2 - Beizmittel zur bekämpfung von phytopathogenen pilzen - Google Patents
Beizmittel zur bekämpfung von phytopathogenen pilzen Download PDFInfo
- Publication number
- WO2005122770A2 WO2005122770A2 PCT/EP2005/006483 EP2005006483W WO2005122770A2 WO 2005122770 A2 WO2005122770 A2 WO 2005122770A2 EP 2005006483 W EP2005006483 W EP 2005006483W WO 2005122770 A2 WO2005122770 A2 WO 2005122770A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- chlorine
- methyl
- seed
- fluorine
- hydrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC(C)CC(C)c(cccc1)c1NC(c1c[n](C)cc1*)=O Chemical compound CC(C)CC(C)c(cccc1)c1NC(c1c[n](C)cc1*)=O 0.000 description 2
- YEGIVZPZMYYGHT-UHFFFAOYSA-N CC(C)CC(C)c(cccc1)c1NC(c1c(C(F)(F)F)cccc1)=O Chemical compound CC(C)CC(C)c(cccc1)c1NC(c1c(C(F)(F)F)cccc1)=O YEGIVZPZMYYGHT-UHFFFAOYSA-N 0.000 description 1
- GZNCWLFUAXRBFQ-UHFFFAOYSA-N CC(C)CC(C)c(cccc1)c1NC(c1c[n](C)cc1C(F)(F)F)=O Chemical compound CC(C)CC(C)c(cccc1)c1NC(c1c[n](C)cc1C(F)(F)F)=O GZNCWLFUAXRBFQ-UHFFFAOYSA-N 0.000 description 1
- LBWGCYNZIUGHGN-UHFFFAOYSA-O CC(CC(C)(C)C)c(cccc1)c1NC(c(c(C(F)(F)F)[nH+][n]1C)c1F)=O Chemical compound CC(CC(C)(C)C)c(cccc1)c1NC(c(c(C(F)(F)F)[nH+][n]1C)c1F)=O LBWGCYNZIUGHGN-UHFFFAOYSA-O 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/56—1,2-Diazoles; Hydrogenated 1,2-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/18—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof
- A01N37/22—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof the nitrogen atom being directly attached to an aromatic ring system, e.g. anilides
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/06—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings
- A01N43/10—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings with sulfur as the ring hetero atom
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/36—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom five-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/74—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,3
- A01N43/78—1,3-Thiazoles; Hydrogenated 1,3-thiazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/14—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D231/16—Halogen atoms or nitro radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- the present invention relates to the use of fungicidally active carboxamides for the treatment of seed, corresponding pickling agents containing these carboxamides, a method for controlling phytopathogenic fungi by treating the seed with fungicidally active carboxamides, and seed which has been treated with the fungicidally active carboxamides.
- M stands for one of the following rings Ml to M4
- R 1 represents hydrogen, halogen, C ⁇ -C3 alkyl or C ⁇ -C 3 haloalkyl having 1 to 7 fluorine, chlorine and / or bromine atoms
- A represents one of the following radicals AI to A8:
- R 2 is Ci-Cs-alkyl
- R 3 is hydrogen, halogen, C ⁇ -C 3 alkyl or C r C is 3 -haloalkyl having 1 to 7 fluorine, chlorine and / or bromine atoms, R 4 represents hydrogen, halogen or CC 3 alkyl;
- R 5 C ⁇ -C3 alkyl or C ⁇ -C stands for halogen, 3 haloalkyl having 1 to 7 fluorine, chlorine and / or bromine atoms
- R 6 is hydrogen, halogen, C ⁇ -C3 alkyl, amino, mono - or di (! C 3 -C -alltyl) amino
- R 7 is hydrogen, halogen, -C 3 alkyl or C ⁇ -C is 3 -haloalkyl having 1 to 7 fluorine, chlorine and / or bromine atoms
- R 8 is halogen, CC 3 alkyl or C ⁇ -C is 3 -haloalkyl having 1 to 7 fluorine, chlorine and / or bromine atoms
- R 9 is halogen, -Cs-alkyl or C ⁇ -C haloalkyl having 1 to 7 Fluorine, chlorine and / or bromine atoms
- R 10 is hydrogen, halogen, C r C 3 alkyl or C, -C 3 haloalkyl having 1 to 7 fluorine, chlorine and / or bromine atoms
- R 11 is is hydrogen, halogen, C ⁇ -C l 3 Al or C r C 3 -haloalkyl having 1 to 7 fluorine, chlorine and / or bromine atoms
- seed dressing very good for the treatment (seed dressing) of seeds against attack by phytopathogenic fungi.
- phytopathogenic fungi can be significantly better controlled / controlled by the use of the active compounds according to the invention as mordant than by spray treatment with foliar application.
- the carboxamides which can be used for the treatment of seeds are generally defined by the formula (I). Preference is given to using carboxamides of the formula (I) in which the radicals have the following meanings.
- M is preferably M-1.
- M is also preferred for M-2.
- R is preferably hydrogen.
- R is for the case that M stands for Ml, moreover preferably for fluorine, where fluorine is particularly preferably in the 4-, 5- or 6-position, very particularly preferably in the 4- or 6-position, in particular in the 4-position of the anilide radical stands.
- R is in the event that M is Ml, moreover preferably chlorine, chlorine being particularly preferably in the 4-, 5- or 6-position, very particularly preferably in the 4- or 6-position, in particular in the 4-position of the anilide residue.
- R stands for the case that M is M-1, furthermore preferably methyl, where methyl is particularly preferably in the 3-position of the anilide radical [cf. above formula (I)].
- R in the case where M is M-2, M-3 or M-4, furthermore preferably represents methyl, where methyl is particularly preferably in the 5-position (M-2, M-3) or in the 3-position ( M-4).
- R 1 preferably represents hydrogen, fluorine, chlorine, methyl, ethyl, n-, isopropyl, monofluoromethyl, difluoromethyl, trifluoromethyl, monochloromethyl, dichloromethyl or trichloromethyl.
- R 1 particularly preferably represents hydrogen, fluorine, chlorine, methyl, ethyl or trifluoromethyl.
- R 1 very particularly preferably represents hydrogen or methyl.
- R 1 is in particular hydrogen. In addition, R 1 is in particular methyl.
- A is preferably the radicals AI.
- A is also very particularly preferably the radical A2.
- A is also very particularly preferably the radical A3.
- A is also most preferably the radical A5.
- R 2 is preferably methyl, ethyl, n- or iso-propyl.
- R 2 particularly preferably represents methyl or isopropyl.
- R 2 is very particularly preferably methyl.
- R 3 is preferably iodine, methyl, difluoromethyl or trifluoromethyl.
- R 3 particularly preferably represents methyl, difluoromethyl or trifluoromethyl.
- R 3 is very particularly preferably methyl.
- R is preferably hydrogen, fluorine, chlorine or methyl.
- R 4 particularly preferably represents hydrogen or fluorine.
- R 4 is very particularly preferably fluorine.
- R 5 is preferably chlorine, bromine, iodine, methyl, difluoromethyl or trifluoromethyl.
- R 5 is particularly preferably iodine, difluoromethyl or trifluoromethyl.
- R 5 very particularly preferably represents iodine or trifluoromethyl.
- R 6 is preferably hydrogen, chlorine, methyl or amino.
- R 6 particularly preferably represents methyl or amino.
- R 6 is very particularly preferably methyl.
- R 7 is preferably hydrogen, chlorine, methyl, difluoromethyl or trifluoromethyl.
- R 7 particularly preferably represents chlorine, methyl, difluoromethyl or trifluoromethyl.
- R 7 is very particularly preferably methyl, difluoromethyl or trifluoromethyl.
- R 8 is preferably chlorine, bromine, iodine, methyl or trifluoromethyl.
- R 8 particularly preferably represents bromine, iodine, methyl or trifluoromethyl.
- R 8 most preferably represents bromine, iodine or methyl.
- R 9 is preferably chlorine, bromine, iodine, methyl, isopropyl or trifluoromethyl.
- R 9 particularly preferably represents methyl, iso-propyl or trifluoromethyl.
- R 9 very particularly preferably represents methyl or trifluoromethyl.
- R 10 is preferably fluorine, chlorine, methyl or trifluoromethyl.
- R 10 particularly preferably represents chlorine or trifluoromethyl.
- R 10 very particularly preferably represents chlorine.
- R 1 ' is preferably chlorine, iodine, methyl, difluoromethyl or trifluoromethyl.
- R 11 particularly preferably represents methyl. Difluoromethyl or trifluoromethyl.
- R 1! is very particularly preferably trifluoromethyl.
- Carboxamides of the formula (I-b) are furthermore particularly preferably usable for the treatment of seed
- R, R 1 and R 5 have the meanings given above and R 1 is in particular hydrogen or methyl.
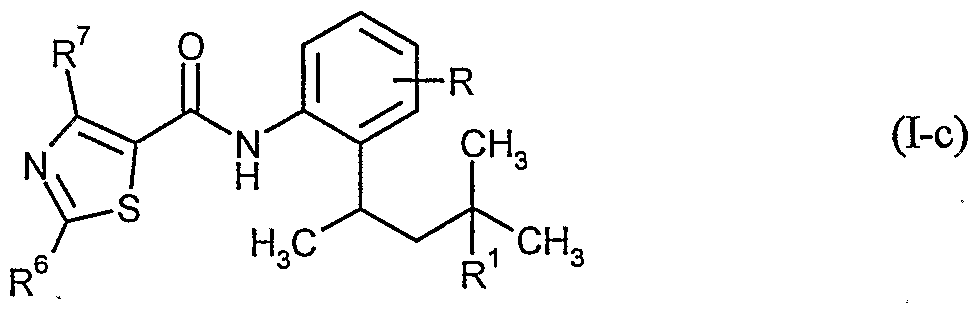
- Carboxamides of the formula (I-c) are furthermore particularly preferably usable for the treatment of seed
- R, R 1 , R and R 7 have the meanings given above and R 1 is in particular hydrogen or methyl.
- Carboxamides of the formula (I-d) can furthermore be used with particular preference for the treatment of seed.
- Carboxamides of the formula (Ie) are furthermore particularly preferably usable for the treatment of seed
- R, R and R 11 have the meanings given above and R 1 is in particular hydrogen or methyl.
- Carboxamides of the formula (I-f) are furthermore particularly preferably usable for the treatment of seed
- R, R 1 , R 2 , R 3 and R 4 have the meanings given above and R 1 is in particular hydrogen or methyl.
- Carboxamides of the formula (I-g) are furthermore particularly preferably usable for the treatment of seed
- R, R 1 and R 5 have the meanings given above and R 1 is in particular hydrogen or methyl.
- Carboxamides of the formula (Ij) are furthermore particularly preferably usable for the treatment of seed
- R, R 1 and R 8 have the abovementioned meanings and R 1 is in particular hydrogen or methyl.
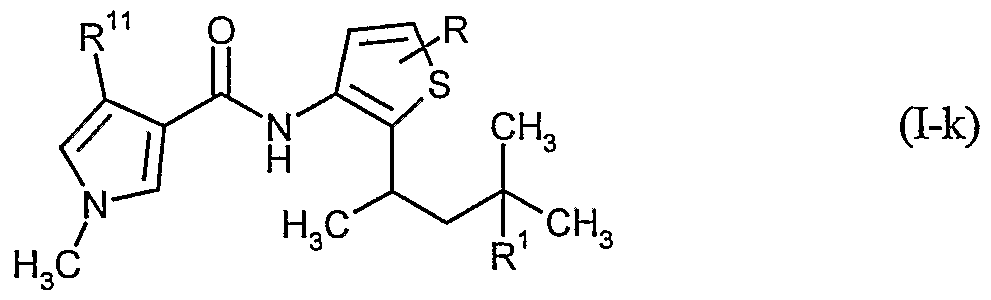
- Carboxamides of the formula (I-k) are furthermore particularly preferably usable for the treatment of seed
- R, R 1 and R u have the meanings given above and R 1 is in particular hydrogen or methyl.
- the formula (I) comprises in particular the following carboxamides which can preferably be used for seed treatment: (1-1) N- [2- (1,3-dimemylbutyl) phenyl] -1,3-dimethyl-1H-pyrazole-4-carboxamide of the formula
- the active compounds according to the invention have very good fungicidal properties and can be used in the treatment of seed, in particular for controlling phytopathogenic fungi, such as Ascomycetes and Basidiomycetes.
- the active compounds according to the invention are particularly suitable in the treatment of seed for controlling Pyrenophora, Rhizoctonia, Tilletia and Ustilago species.
- Pyrenophora species such as, for example, P. allosuri, P. alternarina, P. avenae, P. bartramiae, P. bondarzewii, P. bromi, P. bryophila, P. buddleiae, P. buplewi, P. ca ertn, P calvescens var. moravica, P. carthami, P. centranthi, P. cerastii, P. chengii, P. chrysanthemi, P. convolvuli, P. coppeyana, P. cytisi, P. dactylidis, P. dictyoides, P. echinopis, P.
- Rh. graminea P. teres, P. tetrarrhenae, P. tranzschelii, P. trifolii, P. tritici-repentis, P. typhicola, P. ushuwaiensis, P. villosa, Rliizoctonia species, such as R r. aerea, Rh. alba, Eh. alpina, Rh. anaticula, Rh. anom ⁇ la, Rlt. apocynacearum, Rh. arachnion, Rh. asclerotica, Rh. bataticola, Rh. borealis, PJi. callae, Rh. candida, Rh. carotae, Rh.
- Rh. Grisea Rh coniothecioides, Rh. dichotoma, Rh. dimorpha, Rh. endophytica. filigata, Rh. endophytica, Rh ferruginea, Rh. floccosa, Rh. fragariae, Rh. fraxini, Rh. fuliginea, Rh. fumigata, Rh. globularis, Rh. goodyerae-repentis, Rh. gossypii var. anatolica, Rh. gossypii, Rh. Gracilis, Rh. Grisea,
- Rh. Hiemalis Rh. Juniperi, Rh. Lamellifera, Rh. Leguminicola, Rlt. lilacina, Rh. lupini, Rh. macrosclerotia, Rh. melongenae, Rh. microsclerotia, Rh. monilioides, Rh. monteithiana, Rh. muneratii, Rh. nandorii, Rh. oryzae, Rh. oryzae-sativae, Rh. pallida, Rh. pini-insignis, Rh. praticola,
- T. ahmadiana T. airina, T. ajrekari, T. alopecuri, T. anthoxanthi, T. apludae, T. arundinellae, T. asperifolia, T. asperitolioides, T. atacamensis, T. baldrati, T. bambusae, T. Banarasae, T. bangalorensis, T. barclayana, T. biharica, T. bolayi, T. boliviensis, T. boutelouae, T. brachypodii, T. brachypodii ramosi, T. brevifaciens, T. broml, T. bromina, T.
- T. elymandrae T. elymi, T. elymicola, T. elytrophori, T. eragrostidis, T. euphorbiae, T. fahrendorf ⁇ , T. festuca octoflorana, T. foetida, T. fusca, T. fusca var. bromo-tectorum, T. fusca var. guyotiana, T. fusca var. patagonica, T. georfischeri, T. gigaspora, T. goloskokovü, T. haynaldiae, T heterospora, T. holci, T. hordei var. spontanei, T.
- TJ. vetiveriae tuberculata
- T. viennotii T. vittata var. burmannii
- Ustilago species such as TJ. abstrusa, TJ. aegilopsidis, TJ. aeluropodis, TJ. qff ⁇ nis var. hilariae, TJ. agrestis, TJ. agropyrina, TJ. agrostis-palustris, TJ. ahmadiana, TJ. airae-caespitosae, TJ. alismatis, TJ.
- alopecurivora U. asineae, TJ. altilis, TJ. amadelpha var. glabriuscula, TJ. amphilophidis, TJ. amplexa, U. andropogonis-tectorum, TJ. aneilemae, J. anhweiana, TJ. anomala [var.] microspora, TJ. anomala var. avicularis, J. anomala var. carnea, TJ. anomala var. cordai, TJ. anomala var. muricata,
- TJ. anomala var. tovarae TJ. anihoxanthi, TJ. apscheronica, TJ. Arabidia alpinae, TJ. arctagrostis, TJ. arctica, TJ. arenariae bryophyllae, TJ. argentina, TJ. aristidarius, TJ. arundinellae-hirtae, J. asparagi- pygmaei, TJ. asprellae, TJ. avenae fsp. perennans, TJ. avenae subsp. alba, TJ. avicularis, TJ. bahuichivoensis, TJ. Barberi, J.
- microspora U. bullata f. brachypodii distachyi, U. bullata var. bonariensis, U. bullata var. macrospora, U. bungeana, U. calamagrostidis var. scrobiculata, U. calamagrostidis var. typica, U. calamagrostidis, U. cardamines, U. cariciphila, U. caricis-wallichianae, U. carnea, U. catherinae, U. caulicola, U. centrodomis, U. ceparum, U. cephalariae, U. chacoensis, U. chloridum, U.
- TJ. elymicola TJ. elytrigiae
- TJ. enneapogonis TJ. epicampida, TJ. eragrostidis japonicana, TJ. eriocauli, TJ. eriochloae, TJ. euphorbiae, TJ. fagopyri, TJ. festucae, TJ. festucarum, J. parliamentaryola, TJ. Gebruthiae, TJ. flectens, TJ. flowersii, TJ. foliorum, TJ. formosana, TJ. fueguina, J. gageae, TJ. garcesi, TJ.
- TJ. gaussenii TJ. gigantispora
- TJ. glyceriae TJ. goyazana
- TJ. gregaria TJ. Grossheimu
- TJ. gunnerae J. haesendocki var. chloraphorae
- TJ. halophiloides var. vargasu TJ. halophiloides
- J. haynaldiae TJ. heleochloae
- TJ. helictotrichi TJ. herteri var. bicolor
- TJ. hierochloae-odoratae TJ. Hieronymi var. insularis
- TJ. hilariicola J. himalensis, TJ. hitchcockiana
- TJ. holci-avenacei TJ. holubii
- U. hordei TJ. hordei fp avenae
- TJ. hsuii TJ. hyalino-bipolaris
- TJ. hydropiperis TJ. hyparrheniae
- TJ. hypodytes f. congoensis TJ. hypodytesf. sporoboli
- TJ. hypodytes var. agrestis TJ. idonea, TJ. imperatae, TJ. induta, TJ.
- TJ. ixophori TJ. jacksonii var. ventanensis, TJ. jacksonii, TJ. jaczevskyana var. sibirica, TJ. jaczevskyana var. typica, TJ. jaczevskyana, TJ. jagdishwari, J. jalalainenii, TJ. jehudana, TJ. johnstonii, TJ. kairamoi, TJ. Koreaica, TJ. kenjiana, TJ. kweichowensis, TJ. kyllingae, TJ. lachrymae-jobi, TJ. lepyrodiclidis, TJ. lidii, TJ.
- TJ. merxmuellerana TJ. mesatlantica, TJ. michnoana, TJ. microspora var. paspalicola, TJ. microspora, TJ. microstegii, TJ. microthelis, J. milli, TJ. modesta, TJ. moehringiae, TJ. moenchiae manticae, TJ. monermae, TJ. commonlyi var. minor, TJ. morinae, J. morobiana, J. muehlenbergiae var. tucumanensis, TJ. muricata, TJ. muscari-botryoidis, TJ.
- TJ. nannfeldtii TJ. nelsoniana
- TJ. nepalensis TJ. neyraudiae
- TJ. nigra TJ. nivalis
- TJ. nuda TJ. nuda var. hordei
- TJ. nuda var. tritici TJ. nyassae, U. okudairae, TJ. olida, TJ. olivacea var. macrospora, TJ. onopordi, TJ. onumae, TJ. opiziicola, TJ. oplismeni, U. orientalis, U.
- TJ. raciborskiana TJ. Radians, TJ. Ravida, TJ. rechingeri, TJ. reticulata, TJ. reticulispora, TJ. rhei, TJ. rhynchelytri, TJ. Rwandensis, TJ. sabouriana, TJ. Salviae, TJ. sanctae-catharinae, TJ. Scaura, TJ. sc ⁇ llae, J. scitaminea var. sacchari-barberi, TJ. scitaminea var. sacchari-ofcinarum, J. scitaminea, J. scleranthi, TJ. scrobiculata, TJ.
- TJ. striifornis var. agrostidis TJ. striiformis var. dactylidis
- TJ. striiformis var. hold J. striiformis var. phlei, TJ. striiformis var. poae
- TJ. stygia TJ. sumnevicziana
- TJ. superba TJ. sydowiana,.
- TJ. symbiotics TJ. taenia, TJ. taiana, TJ. Tanakae, TJ. tenuispora, TJ. thaxteri, TJ.
- the good plant tolerance of the usable active ingredients in the necessary concentrations for controlling plant diseases allows treatment of the seed.
- the active compounds according to the invention can thus be used as mordants.
- the present invention therefore more particularly relates to a method of protecting seeds and germinating plants from the infestation of phytopathogenic fungi by treating the seed with an agent according to the invention.
- the invention also relates to the use of the seed treatment agents of the invention for protecting the seed and the germinating plant from phytopathogenic fungi.
- the invention relates to seed which has been treated with an agent according to the invention for protection against phytopathogenic fungi.
- One of the advantages of the present invention is that because of the particular systemic properties of the compositions of the invention, treatment of the seed with these agents not only protects the seed itself but also the resulting plants after emergence from phytopathogenic fungi. In this way, the immediate treatment of the culture at the time of sowing or shortly afterwards can be omitted.
- mixtures according to the invention can also be used in particular in the case of transgenic seed.
- compositions according to the invention are suitable for the protection of seeds of any plant variety used in agriculture, in the greenhouse, in forests or in horticulture.
- these are seeds of cereals (such as wheat, barley, rye, millet and oats), corn, cotton, soy, rice, potatoes, sunflower, bean, coffee, turnip (eg sugarbeet and fodder), peanut, vegetables ( like tomato, cucumber, onions and lettuce), lawn and ornamental plants.
- cereals such as wheat, barley, rye, millet and oats
- corn cotton, soy, rice, potatoes, sunflower, bean, coffee, turnip (eg sugarbeet and fodder), peanut, vegetables (like tomato, cucumber, onions and lettuce), lawn and ornamental plants.
- turnip eg sugarbeet and fodder
- peanut like tomato, cucumber, onions and lettuce
- lawn and ornamental plants e.g sugarbeet and fodder
- the agent according to the invention is applied to the seed alone or in a suitable
- the seed is treated in a condition that is so stable that no damage occurs during the treatment.
- the treatment of the seed can be done at any time between harvesting and sowing.
- seed is used which has been separated from the plant and freed from flasks, shells, stems, hull, wool or pulp.
- seed may be used which has been harvested, cleaned and dried to a moisture content below 15% by weight.
- seed can also be used, which after drying, for example, treated with water and then dried again.
- the agents according to the invention can be applied directly, ie without containing further components and without being diluted.
- suitable formulations and methods for seed treatment are known to those skilled in the art and are described e.g. in the following documents: US 4,272,417 A, US 4,245,432 A, US 4,808,430 A, US 5,876,739 A, US 2003/0176428 AI, WO 2002/080675 AI, WO 2002/028186 A2.
- the active compound combinations that can be used according to the invention can be converted into the customary seed dressing formulations, such as solutions, emulsions, suspensions, powders, foams, slurries or other seed coating compositions, as well as ULV formulations.
- formulations are prepared in a known manner by mixing the active ingredients or combinations of active ingredients with conventional additives, such as conventional extenders and solvents or diluents, dyes, wetting agents, dispersants, emulsifiers, defoamers, preservatives, secondary thickeners, adhesives, gibberellins and also Water.
- conventional additives such as conventional extenders and solvents or diluents, dyes, wetting agents, dispersants, emulsifiers, defoamers, preservatives, secondary thickeners, adhesives, gibberellins and also Water.
- Dyes which may be present in the seed dressing formulations which can be used according to the invention are all dyes customary for such purposes. There are both in
- Water sparingly soluble pigments as well as water-soluble dyes usable.
- water-soluble dyes usable.
- Suitable wetting agents which may be present in the seed dressing formulations which can be used according to the invention are all wetting-promoting substances customary for the formulation of agrochemical active compounds.
- Preferably used are alkylnaphthalene sulfonates, such as diisopropyl or diisobutyl naphthalene sulfonates.
- dispersants and / or emulsifiers which may be present in the seed dressing formulations which can be used according to the invention, all are used for the formulation of agrochemicals
- nonionic or anionic dispersants or mixtures of nonionic or anionic dispersants.
- Particularly suitable nonionic dispersants are, in particular, ethylene oxide-propylene oxide, block polymers, alkylphenol polyglycol ethers and tri-stryrylphenol polyglycol ethers and their phosphated or sulfated derivatives.
- Suitable anionic dispersants are, in particular, lignosulfonates, polyacrylic acid salts and arylsulfonate-formaldehyde condensates.
- Defoamers which may be present in the seed-dressing formulations which can be used according to the invention are all foam-inhibiting substances customary for the formulation of agrochemical active compounds.
- Preferably usable are silicone defoamer and magnesium stearate.
- Preservatives which may be present in the seed dressing formulations which can be used according to the invention are all substances which can be used for such purposes in agrochemical compositions. Examples include dichlorophen and Benzylalkohomemiformal.
- Suitable secondary thickeners which may be present in the seed dressing formulations which can be used according to the invention are all substances which can be used for such purposes in agrochemical compositions. Preference is given to cellulose derivatives, acrylic acid derivatives, xanthan, modified clays and finely divided silica.
- Suitable adhesives which may be present in the seed dressing formulations which can be used according to the invention are all customary binders which can be used in pickling agents.
- Preferably mentioned are polyvinylpyrrolidone, polyvinyl acetate, polyvinyl alcohol and Tylose.
- gibberellins which may be present in the seed dressing formulations which can be used according to the invention preference is given to substances of the formula
- gibberellins of the formula (II) may be mentioned:
- the gibberellins of the formula (IT) are known (see R. Wegler “Chemie der” convinced “Cackeaudlingsbekungsmittel”, Volume 2, Springer Verlag, Berlin-Heidelberg-New York, 1970, pages 401-412).
- the seed dressing formulations which can be used according to the invention can be used either directly or after prior dilution with water for the treatment of seed of various kinds.
- the concentrates or the preparations obtainable therefrom by dilution with water can be used for dressing the seeds of cereals such as wheat, barley, rye, oats and triticale, as well as the seeds of corn, rice, rape, peas, beans, cotton, sunflowers and beets or even vegetable seeds of various nature.
- the seed dressing formulations or their diluted preparations which can be used according to the invention can also be used for pickling seeds of transgenic plants. In this case, additional synergistic effects may occur in interaction with the substances formed by expression.
- the seed dressing formulations which can be used according to the invention or the preparations prepared therefrom by the addition of water
- all mixing devices customarily usable for the dressing can be considered.
- the seed is placed in a mixer which adds the desired amount of seed dressing formulations, either as such or after prior dilution with water, and mixes until evenly distributed the formulation on the seed .
- a drying process follows.
- the application rate of the seed dressing formulations which can be used according to the invention can be varied within a relatively wide range. It depends on the respective content of the active ingredients in the formulations and on the seed.
- the application rates of active ingredient combination are generally between 0.001 and 50 g per kilogram of seed, preferably between 0.01 and 15 g per kilogram of seed.
- the application of the active ingredients is carried out as a dry dressing. They are prepared by stripping the respective active substance with rock flour to a finely powdered mixture, which ensures a uniform distribution on the seed surface.
- Seeds are sown in seed boxes with 2 x 50 seeds in unit soil.
- Perlite infected with Rhizoctonia solani (5 ml / seed box) is sprinkled between the seeds, covered with Lecaton and then cultivated in the greenhouse at a temperature of about 22 ° C. and 15 hours a day for light.
- the application of the active ingredients is carried out as a commercial formulation.
- the preparation is placed in a pickling cup and placed on a pickling shaker.
- the infected seed is filled and shaken for about 2-3 minutes until homogenized.
- the evaluation takes place at a time when the Kraiikheitssymptome are completely and well recognized. In this case, 0% means an efficiency which corresponds to that of the control, while an efficiency of 100% means that no infestation is observed.
- Rice seed (variety: Koshihikari) is soaked for 10 days at 15 ° C in distilled water, transferred to fresh water and soaked with aeration for 1 day at 32 ° C. Thereafter, the seed is dried for a few hours at room temperature. Dissolve 7.6 mg of the test substance in 200 ⁇ l of acetone in a test tube, add an aliquot of the seed (equivalent to 3.8 g of dry seed), mix and blow off the acetone. Seeds are sown on soil in a 7.5 cm diameter plastic vessel and cultured in an incubator for 3 days at 32 ° C under high humidity in the dark. The seedlings are then cultivated in the greenhouse for 2 weeks at an average of 21 ° C.
- Each 5 seedlings are transferred into 5 plastic containers with a diameter of 12 cm and cultivated in the greenhouse for 5-6 weeks at an average of 25 ° C.
- Rhizoctonia solani mycelia grown on sterile barley kernels are spread at the foot of the rice plant (5 cm from the soil surface) and incubated in a high humidity glass chamber at an average of 27 ° C.
- Type of cereal spring barley Seed per plot: 40g Plot size: 2 m 2 Number of repetitions: 3 Development stage: Beginning to middle flowering
- the application of the active ingredients is carried out as a commercial formulation.
- the preparation is placed in a pickling cup and placed on a pickling shaker.
- the infected seed is filled and shaken for about 2-3 minutes until homogenized.
- the sowing in the field takes place after practice ground preparation at a time, which favors the disease infestation.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Dentistry (AREA)
- Wood Science & Technology (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Agronomy & Crop Science (AREA)
- General Health & Medical Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Pretreatment Of Seeds And Plants (AREA)
- Plural Heterocyclic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (18)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP05751644A EP1761128B1 (de) | 2004-06-21 | 2005-06-16 | Beizmittel zur bekämpfung von phytopathogenen pilzen |
| AT05751644T ATE499004T1 (de) | 2004-06-21 | 2005-06-16 | Beizmittel zur bekämpfung von phytopathogenen pilzen |
| EA200700019A EA012432B1 (ru) | 2004-06-21 | 2005-06-16 | Действующие на грибы rhizoctonia solani карбоксамиды и их применение в средствах обработки семенного материала, способ борьбы с поражением грибами rhizoctonia solani и семенной материал, обработанный этими карбоксамидами |
| MXPA06014646A MXPA06014646A (es) | 2004-06-21 | 2005-06-16 | Agentes desinfectantes para la lucha contra hongos fitopatogenos. |
| BRPI0512322-4A BRPI0512322A (pt) | 2004-06-21 | 2005-06-16 | desinfetante para combater fungos fitopatogênicos |
| CA002570978A CA2570978A1 (en) | 2004-06-21 | 2005-06-16 | Seed dressing for controlling phytopathogenic fungi |
| US11/629,982 US20080139389A1 (en) | 2004-06-21 | 2005-06-16 | Seed Dressing for Controlling Phytopathogenic Fungi |
| JP2007517149A JP5001147B2 (ja) | 2004-06-21 | 2005-06-16 | 植物病原性真菌類の抑制のための保護剤 |
| KR1020077001351A KR101234606B1 (ko) | 2004-06-21 | 2005-06-16 | 식물병원성 진균 방제용 보호제 |
| DK05751644.5T DK1761128T3 (da) | 2004-06-21 | 2005-06-16 | Bejdsningsmidler til bekæmpelse af fytopatogene svampe |
| CN2005800206342A CN101031204B (zh) | 2004-06-21 | 2005-06-16 | 用于防治植物致病真菌的保护剂 |
| DE502005011000T DE502005011000D1 (de) | 2004-06-21 | 2005-06-16 | Beizmittel zur bekämpfung von phytopathogenen pilzen |
| AU2005253725A AU2005253725A1 (en) | 2004-06-21 | 2005-06-16 | Protectant for controlling phytopathogenic fungi |
| PL05751644T PL1761128T3 (pl) | 2004-06-21 | 2005-06-16 | Zaprawy do zwalczania fitopatogenicznych grzybów |
| IL180178A IL180178A0 (en) | 2004-06-21 | 2006-12-19 | Protectant for controlling phytopathogenic fungi |
| TNP2006000422A TNSN06422A1 (en) | 2004-06-21 | 2006-12-20 | Protectant for controlling phytopathogenic fungi |
| NO20070341A NO20070341L (no) | 2004-06-21 | 2007-01-18 | Beskyttelsesmiddel for kontrollering av fytopatogene sopper |
| US15/131,759 US20160227778A1 (en) | 2004-06-21 | 2016-04-18 | Seed dressing for controlling phytopathogenic fungi |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102004029972.2 | 2004-06-21 | ||
| DE102004029972A DE102004029972A1 (de) | 2004-06-21 | 2004-06-21 | Beizmittel zur Bekämpfung von phytopathogenen Pilzen |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/629,982 A-371-Of-International US20080139389A1 (en) | 2004-06-21 | 2005-06-16 | Seed Dressing for Controlling Phytopathogenic Fungi |
| US15/131,759 Continuation US20160227778A1 (en) | 2004-06-21 | 2016-04-18 | Seed dressing for controlling phytopathogenic fungi |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005122770A2 true WO2005122770A2 (de) | 2005-12-29 |
| WO2005122770A3 WO2005122770A3 (de) | 2007-02-22 |
Family
ID=35385382
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2005/006483 Ceased WO2005122770A2 (de) | 2004-06-21 | 2005-06-16 | Beizmittel zur bekämpfung von phytopathogenen pilzen |
Country Status (26)
| Country | Link |
|---|---|
| US (2) | US20080139389A1 (de) |
| EP (1) | EP1761128B1 (de) |
| JP (1) | JP5001147B2 (de) |
| KR (1) | KR101234606B1 (de) |
| CN (1) | CN101031204B (de) |
| AR (1) | AR052972A1 (de) |
| AT (1) | ATE499004T1 (de) |
| AU (1) | AU2005253725A1 (de) |
| BR (1) | BRPI0512322A (de) |
| CA (1) | CA2570978A1 (de) |
| DE (2) | DE102004029972A1 (de) |
| DK (1) | DK1761128T3 (de) |
| EA (1) | EA012432B1 (de) |
| ES (1) | ES2360119T3 (de) |
| IL (1) | IL180178A0 (de) |
| MA (1) | MA28974B1 (de) |
| MX (1) | MXPA06014646A (de) |
| NO (1) | NO20070341L (de) |
| PE (1) | PE20060085A1 (de) |
| PL (1) | PL1761128T3 (de) |
| TN (1) | TNSN06422A1 (de) |
| TW (1) | TWI364257B (de) |
| UA (1) | UA86072C2 (de) |
| UY (1) | UY28964A1 (de) |
| WO (1) | WO2005122770A2 (de) |
| ZA (1) | ZA200610631B (de) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006036827A1 (en) * | 2004-09-27 | 2006-04-06 | E.I. Dupont De Nemours And Company | Fungicidal mixtures of thiophene derivative |
| WO2006105888A3 (de) * | 2005-04-06 | 2007-03-15 | Bayer Cropscience Ag | Synergistische fungizide wirkstoffkombinationen |
| WO2006131221A3 (de) * | 2005-06-07 | 2007-08-23 | Bayer Cropscience Ag | Carboxamide |
| WO2008006541A3 (en) * | 2006-07-12 | 2008-05-08 | Syngenta Participations Ag | Method of controlling or preventing pathogenic damage in a plant propagation material |
| EP2027773A1 (de) * | 2007-08-24 | 2009-02-25 | Bayer CropScience AG | Verwendung von N-[2-(1,3-Dimethylbutyl)phenyl]-5-fluor-1,3-dimethyl-1H-pyrazol-4-carboxamid |
| JP2009522358A (ja) * | 2006-01-05 | 2009-06-11 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | カルボキサミド節足動物駆除物質の液体調合物 |
| EA013265B1 (ru) * | 2005-04-08 | 2010-04-30 | Исис Инновейшн Лимитед | Гликозилирование белков |
| EP2192115A1 (de) * | 2003-10-23 | 2010-06-02 | Bayer CropScience AG | 1,3-dimethylbutylcarboxanilide zur bekämpfung von unerwünschten mikrorganismen |
| WO2009098223A3 (en) * | 2008-02-05 | 2010-08-12 | Basf Se | Plant health composition |
| WO2009098218A3 (en) * | 2008-02-05 | 2010-09-02 | Basf Se | Plant health composition |
| EP2272345A1 (de) | 2009-07-07 | 2011-01-12 | Bayer CropScience AG | Verfahren für erhöhtes Wachstum von Setzlingen und/oder frühes Aufgehen von Saat |
| WO2012065945A1 (en) * | 2010-11-15 | 2012-05-24 | Bayer Cropscience Ag | 5-halogenopyrazole(thio)carboxamides |
| CN103081612A (zh) * | 2013-02-25 | 2013-05-08 | 贵州省亚热带作物研究所 | 防治薏苡黑穗病的方法 |
| US8748342B2 (en) | 2009-12-08 | 2014-06-10 | Basf Se | Pesticidal mixtures |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19716257A1 (de) | 1997-04-18 | 1998-10-22 | Bayer Ag | Fungizide Wirkstoffkombination |
| DE102004005787A1 (de) | 2004-02-06 | 2005-08-25 | Bayer Cropscience Ag | Carboxamide |
| DE102004005786A1 (de) * | 2004-02-06 | 2005-08-25 | Bayer Cropscience Ag | Haloalkylcarboxamide |
| DE102004045242A1 (de) | 2004-09-17 | 2006-03-23 | Bayer Cropscience Ag | Synergistische fungizide Wirkstoffkombinationen |
| DE102005015850A1 (de) * | 2005-04-07 | 2006-10-12 | Bayer Cropscience Ag | Synergistische fungizide Wirkstoffkombinationen |
| DE102005022147A1 (de) * | 2005-04-28 | 2006-11-02 | Bayer Cropscience Ag | Wirkstoffkombinationen |
| DE102005023835A1 (de) * | 2005-05-24 | 2006-12-07 | Bayer Cropscience Ag | Fungizide Wirkstoffkombination |
| EA019439B1 (ru) * | 2008-10-21 | 2014-03-31 | Басф Се | Применение карбоксамидов на культивируемых растениях |
| MX2012000566A (es) * | 2009-07-16 | 2012-03-06 | Bayer Cropscience Ag | Combinaciones sinergicas de principios activos con feniltriazoles. |
| WO2017020180A1 (en) * | 2015-07-31 | 2017-02-09 | Bayer Cropscience (China) Co., Ltd. | Method and use of penflufen for controlling corn head smut |
Family Cites Families (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL134133C (de) * | 1965-11-05 | |||
| US4272417A (en) * | 1979-05-22 | 1981-06-09 | Cargill, Incorporated | Stable protective seed coating |
| US4245432A (en) * | 1979-07-25 | 1981-01-20 | Eastman Kodak Company | Seed coatings |
| US4808430A (en) * | 1987-02-27 | 1989-02-28 | Yazaki Corporation | Method of applying gel coating to plant seeds |
| GB9117486D0 (en) * | 1991-08-13 | 1991-09-25 | Schering Ag | Fungicides |
| ES2169773T3 (es) * | 1995-04-11 | 2002-07-16 | Mitsui Chemicals Inc | Derivados de tiofeno sustituido y fungicidas agricolas y horticolas que contienen dichos derivados como ingrediente activo. |
| JP3164762B2 (ja) * | 1995-04-11 | 2001-05-08 | 三井化学株式会社 | 置換チオフェン誘導体およびこれを有効成分とする農園芸用殺菌剤 |
| US5876739A (en) * | 1996-06-13 | 1999-03-02 | Novartis Ag | Insecticidal seed coating |
| US5914344A (en) * | 1996-08-15 | 1999-06-22 | Mitsui Chemicals, Inc. | Substituted carboxanilide derivative and plant disease control agent comprising same as active ingredient |
| US6503904B2 (en) * | 1998-11-16 | 2003-01-07 | Syngenta Crop Protection, Inc. | Pesticidal composition for seed treatment |
| US6838473B2 (en) * | 2000-10-06 | 2005-01-04 | Monsanto Technology Llc | Seed treatment with combinations of pyrethrins/pyrethroids and clothiandin |
| ES2271109T3 (es) * | 2000-11-08 | 2007-04-16 | Syngenta Participations Ag | Pirrocarboxamidas y pirrolcarbotioamidas y sus usos agroquimicos. |
| US20020134012A1 (en) * | 2001-03-21 | 2002-09-26 | Monsanto Technology, L.L.C. | Method of controlling the release of agricultural active ingredients from treated plant seeds |
| JP5034142B2 (ja) * | 2001-04-20 | 2012-09-26 | 住友化学株式会社 | 植物病害防除剤組成物 |
| DE10136065A1 (de) * | 2001-07-25 | 2003-02-13 | Bayer Cropscience Ag | Pyrazolylcarboxanilide |
| DE10229595A1 (de) * | 2002-07-02 | 2004-01-15 | Bayer Cropscience Ag | Phenylbenzamide |
| JP2004123587A (ja) * | 2002-10-01 | 2004-04-22 | Mitsui Chemicals Inc | 芝病害防除剤 |
| DE10303589A1 (de) * | 2003-01-29 | 2004-08-12 | Bayer Cropscience Ag | Pyrazolylcarboxanilide |
| DE10349501A1 (de) * | 2003-10-23 | 2005-05-25 | Bayer Cropscience Ag | Synergistische fungizide Wirkstoffkombinationen |
| IN2004DE01799A (de) * | 2003-10-23 | 2007-01-12 | Bayer Cropscience Ag | |
| DK1751109T3 (da) * | 2004-02-12 | 2009-08-03 | Bayer Cropscience Sa | Fungicid sammensætning omfattende et pyridylethylbenzamidderivat og en forbindelse, der er i stand til at hæmme elektrontransporten i phytopathogene svampeorganismers respirationskæde |
| US20080058352A1 (en) * | 2004-03-26 | 2008-03-06 | Tormo I Blasco Jordi | Fungicidal Mixtures |
| DE102005022147A1 (de) * | 2005-04-28 | 2006-11-02 | Bayer Cropscience Ag | Wirkstoffkombinationen |
| EP2272345A1 (de) * | 2009-07-07 | 2011-01-12 | Bayer CropScience AG | Verfahren für erhöhtes Wachstum von Setzlingen und/oder frühes Aufgehen von Saat |
-
2004
- 2004-06-21 DE DE102004029972A patent/DE102004029972A1/de not_active Withdrawn
-
2005
- 2005-06-16 CN CN2005800206342A patent/CN101031204B/zh not_active Expired - Lifetime
- 2005-06-16 KR KR1020077001351A patent/KR101234606B1/ko not_active Expired - Lifetime
- 2005-06-16 BR BRPI0512322-4A patent/BRPI0512322A/pt not_active IP Right Cessation
- 2005-06-16 MX MXPA06014646A patent/MXPA06014646A/es not_active Application Discontinuation
- 2005-06-16 DK DK05751644.5T patent/DK1761128T3/da active
- 2005-06-16 UA UAA200700554A patent/UA86072C2/ru unknown
- 2005-06-16 WO PCT/EP2005/006483 patent/WO2005122770A2/de not_active Ceased
- 2005-06-16 CA CA002570978A patent/CA2570978A1/en not_active Abandoned
- 2005-06-16 DE DE502005011000T patent/DE502005011000D1/de not_active Expired - Lifetime
- 2005-06-16 EP EP05751644A patent/EP1761128B1/de not_active Expired - Lifetime
- 2005-06-16 AT AT05751644T patent/ATE499004T1/de active
- 2005-06-16 JP JP2007517149A patent/JP5001147B2/ja not_active Expired - Lifetime
- 2005-06-16 EA EA200700019A patent/EA012432B1/ru not_active IP Right Cessation
- 2005-06-16 PL PL05751644T patent/PL1761128T3/pl unknown
- 2005-06-16 US US11/629,982 patent/US20080139389A1/en not_active Abandoned
- 2005-06-16 AU AU2005253725A patent/AU2005253725A1/en not_active Abandoned
- 2005-06-16 ES ES05751644T patent/ES2360119T3/es not_active Expired - Lifetime
- 2005-06-17 UY UY28964A patent/UY28964A1/es not_active Application Discontinuation
- 2005-06-17 AR ARP050102512A patent/AR052972A1/es unknown
- 2005-06-20 TW TW094120358A patent/TWI364257B/zh not_active IP Right Cessation
- 2005-06-20 PE PE2005000701A patent/PE20060085A1/es not_active Application Discontinuation
-
2006
- 2006-12-18 ZA ZA200610631A patent/ZA200610631B/en unknown
- 2006-12-19 IL IL180178A patent/IL180178A0/en unknown
- 2006-12-20 TN TNP2006000422A patent/TNSN06422A1/en unknown
- 2006-12-29 MA MA29598A patent/MA28974B1/fr unknown
-
2007
- 2007-01-18 NO NO20070341A patent/NO20070341L/no not_active Application Discontinuation
-
2016
- 2016-04-18 US US15/131,759 patent/US20160227778A1/en not_active Abandoned
Cited By (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2192115A1 (de) * | 2003-10-23 | 2010-06-02 | Bayer CropScience AG | 1,3-dimethylbutylcarboxanilide zur bekämpfung von unerwünschten mikrorganismen |
| AU2005289678B2 (en) * | 2004-09-27 | 2011-07-07 | Corteva Agriscience Llc | Fungicidal mixtures of thiophene derivative |
| WO2006036827A1 (en) * | 2004-09-27 | 2006-04-06 | E.I. Dupont De Nemours And Company | Fungicidal mixtures of thiophene derivative |
| EA014663B1 (ru) * | 2005-04-06 | 2010-12-30 | Байер Кропсайенс Аг | Фунгицидные комбинации биологически активных веществ, их применение, семенной материал, способ борьбы с нежелательными фитопатогенными грибами |
| WO2006105888A3 (de) * | 2005-04-06 | 2007-03-15 | Bayer Cropscience Ag | Synergistische fungizide wirkstoffkombinationen |
| JP2008537945A (ja) * | 2005-04-06 | 2008-10-02 | バイエル・クロツプサイエンス・アクチエンゲゼルシヤフト | 相乗作用を示す殺菌活性物質組合せ |
| EA013265B1 (ru) * | 2005-04-08 | 2010-04-30 | Исис Инновейшн Лимитед | Гликозилирование белков |
| WO2006131221A3 (de) * | 2005-06-07 | 2007-08-23 | Bayer Cropscience Ag | Carboxamide |
| JP2009522358A (ja) * | 2006-01-05 | 2009-06-11 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | カルボキサミド節足動物駆除物質の液体調合物 |
| WO2008006541A3 (en) * | 2006-07-12 | 2008-05-08 | Syngenta Participations Ag | Method of controlling or preventing pathogenic damage in a plant propagation material |
| EP2027773A1 (de) * | 2007-08-24 | 2009-02-25 | Bayer CropScience AG | Verwendung von N-[2-(1,3-Dimethylbutyl)phenyl]-5-fluor-1,3-dimethyl-1H-pyrazol-4-carboxamid |
| WO2009098218A3 (en) * | 2008-02-05 | 2010-09-02 | Basf Se | Plant health composition |
| WO2009098223A3 (en) * | 2008-02-05 | 2010-08-12 | Basf Se | Plant health composition |
| EA018967B1 (ru) * | 2008-02-05 | 2013-12-30 | Басф Се | Способ повышения выхода и/или повышения жизнеспособности и/или увеличения устойчивости или сопротивления к факторам абиотического стресса и фунгицидные смеси |
| EA018987B1 (ru) * | 2008-02-05 | 2013-12-30 | Басф Се | Композиция для улучшения жизнеспособности растения |
| EP2272345A1 (de) | 2009-07-07 | 2011-01-12 | Bayer CropScience AG | Verfahren für erhöhtes Wachstum von Setzlingen und/oder frühes Aufgehen von Saat |
| WO2011003533A2 (en) | 2009-07-07 | 2011-01-13 | Bayer Cropscience Ag | Process for improving seedling growth and/or early emergence of crops |
| US8748342B2 (en) | 2009-12-08 | 2014-06-10 | Basf Se | Pesticidal mixtures |
| US9049859B2 (en) | 2009-12-08 | 2015-06-09 | Basf Se | Pesticidal mixtures |
| US9585391B2 (en) | 2009-12-08 | 2017-03-07 | Basf Se | Pesticidal mixtures |
| WO2012065945A1 (en) * | 2010-11-15 | 2012-05-24 | Bayer Cropscience Ag | 5-halogenopyrazole(thio)carboxamides |
| CN103081612A (zh) * | 2013-02-25 | 2013-05-08 | 贵州省亚热带作物研究所 | 防治薏苡黑穗病的方法 |
| CN103081612B (zh) * | 2013-02-25 | 2014-12-10 | 贵州省亚热带作物研究所 | 防治薏苡黑穗病的方法 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1761128A2 (de) | Beizmittel zur bekämpfung von phytopathogenen pilzen | |
| EP1677598B1 (de) | Synergistische fungizide wirkstoffkombinationen | |
| Poswal et al. | Current trends in the use of traditional and organic methods for the control of crop pests and diseases in Nigeria | |
| EA012504B1 (ru) | Средство на основе замещённых циклических кетоенолов и защитных веществ, применение средства для борьбы с насекомыми и паукообразными и способ борьбы с насекомыми и/или паукообразными | |
| WO2006002824A9 (de) | Verwendung von 3-(2,4,6-trimethylphenyl)-4-neopentylcarbonyloxy-5,5-tetramethylen ν dihydrofuran-2-on zur bekämpfung von psylliden | |
| DE102005015850A1 (de) | Synergistische fungizide Wirkstoffkombinationen | |
| KR20120099396A (ko) | 식물병원성 박테리아를 구제하기 위한 사이클릭 케토에놀의 용도 | |
| CN108495556B (zh) | 有害生物防治组合物和有害生物防治方法 | |
| CN107950552A (zh) | 一种含申嗪霉素的复配种衣剂 | |
| CN101248804A (zh) | 溴氰菊酯和八角茴香油杀虫剂组合物 | |
| KR20080071998A (ko) | 살선충성 조성물 및 방법 | |
| values were calculated by Probit | Relative efficacy and residual toxicity of margosa (Azadirachta indica) and indian beech (Pongamia pinnata) oils in stemborer (Chilo partellus) of maize (Zea mays) | |
| JP4239181B2 (ja) | 植物種子 | |
| KR102146506B1 (ko) | 토마틴을 유효성분으로 포함하는 식물병 방제용 조성물 | |
| CN101553117B (zh) | 3-(2,4,6-三甲基苯基)-4-新戊基碳酰氧基-5,5-四亚甲基-△3-二氢呋喃-2-酮用于防治瘿蚊科双翅目的用途 | |
| CN114041468A (zh) | 一种含硫虫酰胺和四唑虫酰胺的杀虫组合物及其预防或控制动物害虫的用途 | |
| CN1040612C (zh) | 杀虫和杀螨组合物 | |
| CN112544626A (zh) | 含有2,4-二氯苯氧乙酸的除草剂及其制备方法 | |
| CN105394069B (zh) | 一种多杀霉素和戊吡虫胍复配杀虫组合物、制剂及其应用 | |
| CN106305732A (zh) | 一种含有氟螨嗪的杀虫杀螨组合物 | |
| CN103875699B (zh) | 一种含拟除虫菊酯类杀虫剂的组合物及其应用 | |
| CN116171994B (zh) | 一种含敌稗、硝磺草酮和氰氟草酯三元除草组合物 | |
| CN113875766B (zh) | 一种杀虫组合物 | |
| CN115956570B (zh) | 一种杀虫组合物 | |
| JP4993469B2 (ja) | シロアリ防除剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Country of ref document: MX Ref document number: PA/a/2006/014646 Ref document number: 2005751644 Country of ref document: EP Ref document number: 7574/DELNP/2006 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006/10631 Country of ref document: ZA Ref document number: 552165 Country of ref document: NZ Ref document number: 2570978 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 180178 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007517149 Country of ref document: JP Ref document number: 06127741 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005253725 Country of ref document: AU Ref document number: 200580020634.2 Country of ref document: CN Ref document number: 12006502615 Country of ref document: PH |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: DZP2007000037 Country of ref document: DZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020077001351 Country of ref document: KR Ref document number: 1200700127 Country of ref document: VN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200700019 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: 2005253725 Country of ref document: AU Date of ref document: 20050616 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005253725 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020077001351 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005751644 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11629982 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0512322 Country of ref document: BR |