WO2006018315A1 - Diagnostic method for disorders using copeptin - Google Patents
Diagnostic method for disorders using copeptin Download PDFInfo
- Publication number
- WO2006018315A1 WO2006018315A1 PCT/EP2005/009001 EP2005009001W WO2006018315A1 WO 2006018315 A1 WO2006018315 A1 WO 2006018315A1 EP 2005009001 W EP2005009001 W EP 2005009001W WO 2006018315 A1 WO2006018315 A1 WO 2006018315A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- copeptin
- vasopressin
- seq
- diseases
- amino acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6893—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids related to diseases not provided for elsewhere
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/74—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving hormones or other non-cytokine intercellular protein regulatory factors such as growth factors, including receptors to hormones and growth factors
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
- G01N2333/575—Hormones
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
- G01N2333/575—Hormones
- G01N2333/5757—Vasoactive intestinal peptide [VIP] or related peptides
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S930/00—Peptide or protein sequence
- Y10S930/01—Peptide or protein sequence
- Y10S930/15—Oxytocin or vasopressin; related peptides
Definitions
- the present invention relates to the use of copeptin and/or its precursors or fragments and/or its splice variants, fragments comprising copeptin and/or combinations and/or derivatives thereof in medical diagnosis as humoral bio- marker for disorders associated with or caused by non-phy ⁇ siological alterations of vasopressin release from the neurohypophysis, as there are, for example, cardiac disea ⁇ ses, renal disaeases, inflammatory diseases and sepsis and diseases/disorders of the central nervous system and neuro ⁇ degenerative diseases and others as mentioned below.
- Vasopressin has also been discussed as a marker for the prognosis of probability of survival of patients with car ⁇ diac arrest [11] and consequently it was used for the treat ⁇ ment of such patients [12] .
- the object of the invention was to overcome the effects of the disadvantageous half-life of vasopressin and the varia ⁇ ble results in measurements and to develop a method, use and a kit for the detection and determination of the molecules associated with the release of vasopressin, more specially copeptin, for the diagnosis of cardiovascular diseases and sepsis .
- Fig. 1 shows the standard curve for the sandwich-immuno ⁇ assay for copeptin immunoreactivity using the peptide PAY33 as a standard material.
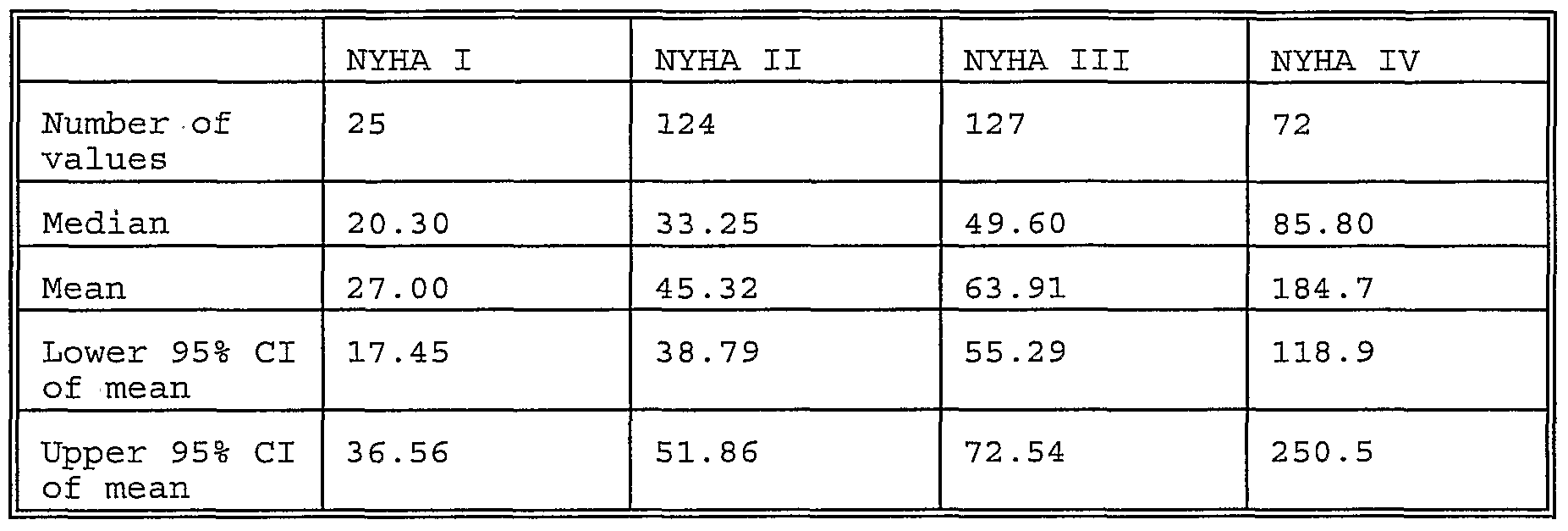
- Fig. 2 shows the concentration of copeptin immunoreacti ⁇ vity in samples of healthy individuals and diffe ⁇ rent groups of patients (sepsis, cardiac in ⁇ farction and increased arterial blood pressure) .
- amino acid sequences of the peptides are the following (numbers refer to corresponding positions in the human pro- vasopressin-neurophysin 2-copeptin-precursors (positions 132-147 and 149-164) :
- the polyclonal antibodies from sheep were purified using ligand specific affinity purification.
- the peptides PATV 17 and PLAY 17 were linked to SuIfoLink-Gel supplied by Pierce (Boston, USA) .
- the binding occurred according to the protocol of Pierce. 5 mg of peptide were added per 5 ml of gel .
- the sample was added and chromatographed at a flow rate of lml/minute in solvent A.
- the flow was continuously monitored in a UV-meter at wave length of 280 and 368 nm to determine the degree of tagging.
- the absorp ⁇ tion ratio 368/280 nm of labelled antibody was 0,1.
- the fractions containing monomeric antibodies were collected (retention time 8-10 minutes) and taken up in 3 ml 10OmM sodium phosphate, 150 mM NaCl, 5% bovine serum albumin, 0,1 % sodium azide, pH 7.4)
- pep ⁇ tide PAY 33 As a standard material chemically synthesized peptide (pep ⁇ tide PAY 33) was used which corresponds to positions 132-164 of vasopressin-neurophysin 2-copeptin precursor.
- the stan ⁇ dard was diluted in normal horse serum (Sigma) .
- the chemiluminescence bound to the tube was determined in a luminometer (Berthold, LB952T, basic reagents Brahms AG) . Using the software MultiCalc (Spline Fit) the concentrations of the samples were determined.
- copeptin a biomarker candidate for use in the diagnosis (positive or negative diagnosis) of CHF, the monitoring of the course and evolution of CHF and the monitoring and control of a CHF therapy. Further, in view of current attempts to evaluate the usefulness of vasoporessin receptor antagonists in the therapy of heart failure [27] , the determination of copeptin in serum or plasma samples of heart failure patients can allow the identification of such patients who would benefit more than others from a treatment with vasopressin receptor antagonists.
- Hyde JF, North WG, Ben-Jonathan N The vasopressin- associated glycopeptide is not a prolactin-releasing factor: studies with lactating Brattleboro rats. Endo ⁇ crinology 124: 35-40, 1989;
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Immunology (AREA)
- Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- Urology & Nephrology (AREA)
- Hematology (AREA)
- Biotechnology (AREA)
- Analytical Chemistry (AREA)
- Cell Biology (AREA)
- Pathology (AREA)
- Food Science & Technology (AREA)
- Medicinal Chemistry (AREA)
- Physics & Mathematics (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Endocrinology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Peptides Or Proteins (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| HK08101167.7A HK1114171B (en) | 2004-08-19 | 2005-08-19 | Copeptin-based diagnostic kit for disorders |
| JP2007526400A JP4932714B2 (en) | 2004-08-19 | 2005-08-19 | Disease diagnosis method using copeptin |
| CN2005800214349A CN101010591B (en) | 2004-08-19 | 2005-08-19 | A kit for diagnosis of diseases based on copeptin |
| DE200560002836 DE602005002836T2 (en) | 2004-08-19 | 2005-08-19 | DIAGNOSTIC PROCEDURE FOR ILLNESSES USING COPEPTIN |
| MXPA06014775A MXPA06014775A (en) | 2004-08-19 | 2005-08-19 | Diagnostic method for disorders using copeptin. |
| EP20050782853 EP1738178B1 (en) | 2004-08-19 | 2005-08-19 | Diagnostic method for disorders using copeptin |
| US11/573,595 US7807397B2 (en) | 2004-08-19 | 2005-08-19 | Diagnostic method for disorders using copeptin |
| US12/860,311 US20110008911A1 (en) | 2004-08-19 | 2010-08-20 | Diagnostic method for disorders using copeptin |
| US15/443,717 US20170242037A1 (en) | 2004-08-19 | 2017-02-27 | Diagnostic method for disorders using copeptin |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP20040019732 EP1628136A1 (en) | 2004-08-19 | 2004-08-19 | Method of diagnosis of disease using copeptin |
| EP04019732.9 | 2004-08-19 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/573,595 A-371-Of-International US7807397B2 (en) | 2004-08-19 | 2005-08-19 | Diagnostic method for disorders using copeptin |
| US12/860,311 Division US20110008911A1 (en) | 2004-08-19 | 2010-08-20 | Diagnostic method for disorders using copeptin |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006018315A1 true WO2006018315A1 (en) | 2006-02-23 |
Family
ID=34926232
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2005/009001 Ceased WO2006018315A1 (en) | 2004-08-19 | 2005-08-19 | Diagnostic method for disorders using copeptin |
Country Status (10)
| Country | Link |
|---|---|
| US (3) | US7807397B2 (en) |
| EP (2) | EP1628136A1 (en) |
| JP (2) | JP4932714B2 (en) |
| CN (2) | CN101010591B (en) |
| AR (1) | AR050215A1 (en) |
| AT (1) | ATE375518T1 (en) |
| DE (1) | DE602005002836T2 (en) |
| ES (1) | ES2296224T3 (en) |
| MX (1) | MXPA06014775A (en) |
| WO (1) | WO2006018315A1 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008012019A2 (en) | 2006-07-24 | 2008-01-31 | B.R.A.H.M.S. Aktiengesellschaft | Method for controlling the therapy of patients suffering from cardiac insufficiency by means of in vitro determination of threshold values of vasoactive peptides |
| WO2008058517A3 (en) * | 2006-11-12 | 2008-10-23 | Brahms Ag | Diagnosis and risk stratification of infections and chronic diseases of the respiratory tract and lungs by means of provasopressin, particularly copeptin or neurophysin ii |
| JP2010507076A (en) * | 2006-10-18 | 2010-03-04 | アクセラ インコーポレーテッド | Measurement of multiple analytes over a wide concentration range by light diffraction. |
| JP2010507786A (en) * | 2006-10-26 | 2010-03-11 | ブラームズ アクチェンゲゼルシャフト | Risk stratification for acute coronary syndrome using provasopressin or fragments thereof and partial peptides, particularly copeptin or neurophysin II |
| JP2010511877A (en) * | 2006-12-08 | 2010-04-15 | ブラームズ アクチェンゲゼルシャフト | Diagnosis and risk stratification of cardiac dysfunction using neurophysin |
| US9018350B2 (en) | 2011-05-26 | 2015-04-28 | Phoenix Pharmaceuticals, Inc. | Phoenixin peptides |
| CN109679924A (en) * | 2018-12-25 | 2019-04-26 | 广州万孚生物技术股份有限公司 | The anti-human monoclonal antibody and the preparation method and application thereof with peptide element of high-affinity |
| US10954298B2 (en) | 2011-06-30 | 2021-03-23 | B.R.A.H.M.S. Gmbh | Method of obtaining a binder to prepro-vasopressin or fragments thereof |
Families Citing this family (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20140046407A1 (en) * | 2001-08-31 | 2014-02-13 | Bio Control Medical (B.C.M.) Ltd. | Nerve stimulation techniques |
| DE102006046996A1 (en) * | 2006-10-01 | 2008-04-03 | Brahms Aktiengesellschaft | Diagnosis process for respiratory infections involves using procalcitonin as marker for assessing patient risk level |
| DE102006050497A1 (en) * | 2006-10-26 | 2008-04-30 | Brahms Aktiengesellschaft | Risk stratification process for acute coronary syndrome involves finding levels of provasoppressin or its fragments and part-peptides by in-vitro diagnosis |
| EP2180322A1 (en) * | 2008-10-22 | 2010-04-28 | BRAHMS Aktiengesellschaft | Prognostic biomarkers for the progression of primary chronic kidney disease |
| ES2772930T3 (en) * | 2008-10-31 | 2020-07-08 | Brahms Gmbh | Prohormone arginine vasopressin as a predictive biomarker for diabetes |
| US20100312581A1 (en) * | 2009-06-08 | 2010-12-09 | Peter James Wachtell | Process and system for efficient allocation of medical resources |
| US20120129187A1 (en) | 2009-06-16 | 2012-05-24 | B.R.A.H.M.S. Gmbh | Diagnostical use of peroxiredoxin 4 |
| JP5827323B2 (en) * | 2010-06-18 | 2015-12-02 | ベー.エル.アー.ハー.エム.エス ゲゼルシャフト ミット ベシュレンクテル ハフツング | Biomarkers for predicting cancer incidence |
| EP2776064A4 (en) * | 2011-11-11 | 2015-08-05 | Woomera Therapeutics | ANTAGONISTS OF PRO-VASOPRESSIN AND USES THEREOF |
| GB201210686D0 (en) * | 2012-06-15 | 2012-08-01 | Holsboermaschmeyer Neurochemie Gmbh | V1B receptor antagonist for use in the treatment of patients having an elevated AVP level and/or an elevated copeptin level |
| GB201310782D0 (en) | 2013-06-17 | 2013-07-31 | Max Planck Innovation Gmbh | Method for predicting a treatment response to a CRHR1 antagonist and/or V1B antagonist in a patient with depressive and/or anxiety symptoms |
| US9687526B2 (en) | 2015-01-30 | 2017-06-27 | Par Pharmaceutical, Inc. | Vasopressin formulations for use in treatment of hypotension |
| US9937223B2 (en) | 2015-01-30 | 2018-04-10 | Par Pharmaceutical, Inc. | Vasopressin formulations for use in treatment of hypotension |
| US9744239B2 (en) | 2015-01-30 | 2017-08-29 | Par Pharmaceutical, Inc. | Vasopressin formulations for use in treatment of hypotension |
| US9744209B2 (en) | 2015-01-30 | 2017-08-29 | Par Pharmaceutical, Inc. | Vasopressin formulations for use in treatment of hypotension |
| US9750785B2 (en) | 2015-01-30 | 2017-09-05 | Par Pharmaceutical, Inc. | Vasopressin formulations for use in treatment of hypotension |
| US9925233B2 (en) | 2015-01-30 | 2018-03-27 | Par Pharmaceutical, Inc. | Vasopressin formulations for use in treatment of hypotension |
| WO2016128383A1 (en) | 2015-02-10 | 2016-08-18 | B.R.A.H.M.S Gmbh | Free histone proteins as biomarkers |
| EP3279667A1 (en) | 2016-08-02 | 2018-02-07 | Abbott Laboratories | Cardiac troponin i and copeptin as biomarkers for short-term mortality in acute exacerbation of chronic obstructive pulmonary disease (aecopd) |
| CN109061140A (en) * | 2018-08-31 | 2018-12-21 | 广州华澳生物科技有限公司 | A kind of and peptide element immunochromatographiassay assay reagent card and preparation method thereof |
| CN109596839A (en) * | 2018-12-25 | 2019-04-09 | 广州万孚生物技术股份有限公司 | People and peptide element fast quantitative measurement method for detecting and kit |
| JP7640168B2 (en) * | 2019-01-28 | 2025-03-05 | ベー.エル.アー.ハー.エム.エス ゲーエムベーハー | Method for predicting treatment response in patients with nocturnal enuresis - Patent Application 20070123633 |
| CN111007270A (en) * | 2019-11-27 | 2020-04-14 | 迪瑞医疗科技股份有限公司 | Peptide-neutralizing chemiluminescence immunoassay kit and preparation method thereof |
| CN113406339A (en) * | 2021-08-19 | 2021-09-17 | 山东康华生物医疗科技股份有限公司 | Dry-type immunofluorescence quantitative method human Copeptin (CPP) detection kit |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004006860A2 (en) * | 2002-07-16 | 2004-01-22 | Woomera Therapeutics, Inc. | Compositions and uses thereof for identifying and targeting provasopressin-expressing cancer cells |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6413783B1 (en) * | 1997-09-18 | 2002-07-02 | Meso Scale Technologies, Llc | Assay sonication apparatus and methodology |
| US6822082B2 (en) * | 2000-06-30 | 2004-11-23 | Zymogenetics, Inc. | Polynucleotides for mammalian secreted protein, Z1055G2P |
| US20040253637A1 (en) * | 2001-04-13 | 2004-12-16 | Biosite Incorporated | Markers for differential diagnosis and methods of use thereof |
| CN1166953C (en) * | 2001-05-30 | 2004-09-15 | 上海晶泰生物技术有限公司 | Fast gold mark test reagent kit for one-step multiple-index test of myocardial infarction |
| IL161446A0 (en) * | 2001-11-26 | 2004-09-27 | Daiichi Suntory Pharma Co Ltd | Medicinal compositions for nasal absorption |
| EP1588159A4 (en) * | 2002-12-24 | 2008-03-12 | Biosite Inc | Method and system for disease detection using marker combinations |
| PL2084543T3 (en) * | 2006-10-26 | 2018-04-30 | B.R.A.H.M.S Gmbh | Risk stratification for acute coronary syndrome by means of fragments/partial peptides of provasopressin, especially copeptin or neurophysin ii |
| DE102006053442A1 (en) * | 2006-11-12 | 2008-05-15 | Brahms Aktiengesellschaft | Diagnosis and risk stratification of infections and chronic diseases of the respiratory and lungs by means of proVasopressin, in particular copeptin or neurophysin II |
| DE102006060112A1 (en) * | 2006-12-20 | 2008-06-26 | Brahms Aktiengesellschaft | Diagnosis and risk stratification using the new marker CT-proADM |
| DE102007010834A1 (en) * | 2007-03-03 | 2008-09-04 | Brahms Aktiengesellschaft | Method for in-vitro diagnosis or risk classification or outcome prognosis of heart failure for New york heart association patient, involves utilizing determination of marker proatrial natriuretic peptide |
-
2004
- 2004-08-19 EP EP20040019732 patent/EP1628136A1/en not_active Withdrawn
-
2005
- 2005-08-19 MX MXPA06014775A patent/MXPA06014775A/en active IP Right Grant
- 2005-08-19 CN CN2005800214349A patent/CN101010591B/en not_active Expired - Lifetime
- 2005-08-19 US US11/573,595 patent/US7807397B2/en active Active
- 2005-08-19 CN CN201110359380.4A patent/CN102539789B/en not_active Expired - Lifetime
- 2005-08-19 DE DE200560002836 patent/DE602005002836T2/en not_active Expired - Lifetime
- 2005-08-19 JP JP2007526400A patent/JP4932714B2/en not_active Expired - Lifetime
- 2005-08-19 ES ES05782853T patent/ES2296224T3/en not_active Expired - Lifetime
- 2005-08-19 AR ARP050103483 patent/AR050215A1/en active IP Right Grant
- 2005-08-19 EP EP20050782853 patent/EP1738178B1/en not_active Expired - Lifetime
- 2005-08-19 AT AT05782853T patent/ATE375518T1/en active
- 2005-08-19 WO PCT/EP2005/009001 patent/WO2006018315A1/en not_active Ceased
-
2010
- 2010-08-20 US US12/860,311 patent/US20110008911A1/en not_active Abandoned
-
2011
- 2011-08-01 JP JP2011168242A patent/JP2011227095A/en active Pending
-
2017
- 2017-02-27 US US15/443,717 patent/US20170242037A1/en not_active Abandoned
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004006860A2 (en) * | 2002-07-16 | 2004-01-22 | Woomera Therapeutics, Inc. | Compositions and uses thereof for identifying and targeting provasopressin-expressing cancer cells |
Non-Patent Citations (6)
| Title |
|---|
| GABREELS B A T F ET AL: "The vasopressin precursor is not processed in the hypothalamus of Wolfram syndrome patients with diabetes insipidus: Evidence for the involvement of PC2 and 7B2", JOURNAL OF CLINICAL ENDOCRINOLOGY AND METABOLISM, vol. 83, no. 11, November 1998 (1998-11-01), pages 4026 - 4033, XP008055034, ISSN: 0021-972X * |
| NIJENHUIS MARGA ET AL: "Familial neurohypophysial diabetes insipidus in a large Dutch kindred: Effect of the onset of diabetes on growth in children and cell biological defects of the mutant vasopressin prohormone", JOURNAL OF CLINICAL ENDOCRINOLOGY AND METABOLISM, vol. 86, no. 7, July 2001 (2001-07-01), pages 3410 - 3420, XP008055032, ISSN: 0021-972X * |
| NIJENHUIS MARGA ET AL: "Mutations in the vasopressin prohormone involved in diabetes insipidus impair endoplasmic reticulum export but not sorting", JOURNAL OF BIOLOGICAL CHEMISTRY, vol. 274, no. 30, 23 July 1999 (1999-07-23), pages 21200 - 21208, XP008055030, ISSN: 0021-9258 * |
| NORTH W G ET AL: "IMMUNOHISTOCHEMICAL EVALUATION OF VASOPRESSIN EXPRESSION IN BREAST FIBROCYSTIC DISEASE AND DUCTAL CARCINOMA IN SITU (DCIS)", ENDOCRINE PATHOLOGY, HUMANA PRESS, TOTOWA, NJ, US, vol. 14, no. 3, 2003, pages 257 - 262, XP008043447, ISSN: 1046-3976 * |
| NORTH W G: "NEUROPEPTIDE PRODUCTION BY SMALL CELL CARCINOMA VASOPRESSIN AND OXYTOCIN AS PLASMA MARKERS OF DISEASE", JOURNAL OF CLINICAL ENDOCRINOLOGY AND METABOLISM, NEW YORK, NY, US, vol. 73, no. 6, 1991, pages 1316 - 1320, XP008043564, ISSN: 0021-972X * |
| YAMAJI T ET AL: "PROPRESSOPHYSIN IN HUMAN BLOOD A POSSIBLE MARKER OF ECTOPIC VASOPRESSIN PRODUCTION", JOURNAL OF CLINICAL ENDOCRINOLOGY AND METABOLISM, vol. 59, no. 3, 1984, pages 505 - 512, XP008055037, ISSN: 0021-972X * |
Cited By (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008012019A2 (en) | 2006-07-24 | 2008-01-31 | B.R.A.H.M.S. Aktiengesellschaft | Method for controlling the therapy of patients suffering from cardiac insufficiency by means of in vitro determination of threshold values of vasoactive peptides |
| US7915002B2 (en) | 2006-07-24 | 2011-03-29 | B.R.A.H.M.S Gmbh | Method for controlling the therapy of patients suffering from cardiac insufficiency by means of in vitro determination of threshold values of vasoactive peptides |
| JP2010507076A (en) * | 2006-10-18 | 2010-03-04 | アクセラ インコーポレーテッド | Measurement of multiple analytes over a wide concentration range by light diffraction. |
| JP2010507786A (en) * | 2006-10-26 | 2010-03-11 | ブラームズ アクチェンゲゼルシャフト | Risk stratification for acute coronary syndrome using provasopressin or fragments thereof and partial peptides, particularly copeptin or neurophysin II |
| US9261517B2 (en) | 2006-10-26 | 2016-02-16 | B.R.A.H.M.S Gmbh | Diagnosis of acute coronary syndrome, myocardial infarction, or angina pectoris by means of neurophysin II |
| US8501485B2 (en) | 2006-10-26 | 2013-08-06 | B.R.A.H.M.S. Gmbh | Risk stratification for acute coronary syndrome by determining copeptin |
| JP2013178249A (en) * | 2006-10-26 | 2013-09-09 | Brahms Gmbh | Risk stratification for acute coronary syndrome by means of provasopressin or fragments and partial peptides thereof, in particular copeptin or neurophysin ii |
| JP2010509576A (en) * | 2006-11-12 | 2010-03-25 | ブラームズ アクチェンゲゼルシャフト | Diagnosis and / or stratification of risk of respiratory and pulmonary infections and chronic diseases with provasopressin, especially copeptin or neurophysin II |
| WO2008058517A3 (en) * | 2006-11-12 | 2008-10-23 | Brahms Ag | Diagnosis and risk stratification of infections and chronic diseases of the respiratory tract and lungs by means of provasopressin, particularly copeptin or neurophysin ii |
| EP2378290A1 (en) | 2006-11-12 | 2011-10-19 | B.R.A.H.M.S GmbH | Diagnosis and risk stratification of infections and chronic illnesses of the airways and lungs by means of Pro-Vasopressin, in particular Copeptin or Neurophysin II |
| US8158368B2 (en) | 2006-11-12 | 2012-04-17 | B.R.A.H.M.S. Gmbh | Diagnosis and risk stratification of infections and chronic diseases of the respiratory tract and lungs by means of provasopressin, particularly copeptin or neurophysin II |
| JP2013083664A (en) * | 2006-11-12 | 2013-05-09 | Brahms Gmbh | Diagnosis and/or risk stratification of infections and chronic diseases of respiratory tract and lungs by means of provasopressin, particularly copeptin or neurophysin ii |
| US10718783B2 (en) | 2006-11-12 | 2020-07-21 | Brahms Gmbh | Diagnosis and risk stratification of infections and chronic diseases of the respiratory tract and lungs by means of provasopressin, particularly copeptin or neurophysin II |
| US20170059583A1 (en) * | 2006-11-12 | 2017-03-02 | Brahms Gmbh | Diagnosis and risk stratification of infections and chronic diseases of the respiratory tract and lungs by means of provasopressin, particularly copeptin or neurophysin ii |
| JP2010511877A (en) * | 2006-12-08 | 2010-04-15 | ブラームズ アクチェンゲゼルシャフト | Diagnosis and risk stratification of cardiac dysfunction using neurophysin |
| JP2013210385A (en) * | 2006-12-08 | 2013-10-10 | Brahms Gmbh | Diagnosis of heart malfunction and layering of risk using neurophysin |
| US9534045B2 (en) | 2011-05-26 | 2017-01-03 | Phoenix Pharmaceuticals, Inc. | Phoenixin peptides |
| US9018350B2 (en) | 2011-05-26 | 2015-04-28 | Phoenix Pharmaceuticals, Inc. | Phoenixin peptides |
| US10836812B2 (en) | 2011-05-26 | 2020-11-17 | Phoenix Pharmaceuticals, Inc. | Phoenixin peptides |
| US12103965B2 (en) | 2011-05-26 | 2024-10-01 | Phoenix Pharmaceuticals, Inc. | Diagnostic kit |
| US10954298B2 (en) | 2011-06-30 | 2021-03-23 | B.R.A.H.M.S. Gmbh | Method of obtaining a binder to prepro-vasopressin or fragments thereof |
| CN109679924A (en) * | 2018-12-25 | 2019-04-26 | 广州万孚生物技术股份有限公司 | The anti-human monoclonal antibody and the preparation method and application thereof with peptide element of high-affinity |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1738178B1 (en) | 2007-10-10 |
| HK1172690A1 (en) | 2013-04-26 |
| ATE375518T1 (en) | 2007-10-15 |
| HK1114171A1 (en) | 2008-10-24 |
| DE602005002836T2 (en) | 2008-07-31 |
| JP4932714B2 (en) | 2012-05-16 |
| DE602005002836D1 (en) | 2007-11-22 |
| CN101010591A (en) | 2007-08-01 |
| AR050215A1 (en) | 2006-10-04 |
| US20170242037A1 (en) | 2017-08-24 |
| US20090221009A1 (en) | 2009-09-03 |
| CN102539789A (en) | 2012-07-04 |
| JP2011227095A (en) | 2011-11-10 |
| EP1628136A1 (en) | 2006-02-22 |
| JP2008510151A (en) | 2008-04-03 |
| US7807397B2 (en) | 2010-10-05 |
| EP1738178A1 (en) | 2007-01-03 |
| ES2296224T3 (en) | 2008-04-16 |
| CN101010591B (en) | 2011-12-21 |
| US20110008911A1 (en) | 2011-01-13 |
| MXPA06014775A (en) | 2007-03-23 |
| CN102539789B (en) | 2015-04-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1738178B1 (en) | Diagnostic method for disorders using copeptin | |
| US8501415B2 (en) | Identification of TSH receptor autoantibodies using affinity-purified antibodies | |
| CN1759319B (en) | Usage of peptide of ADM Among the regional parts of Biological fluid in preparing immune diagnosticum | |
| US6248546B1 (en) | Assay of IGFBP complex | |
| Birken et al. | Metabolism of hCG and hLH to multiple urinary forms | |
| JP4656493B2 (en) | Sandwich immunoassay for identifying partial proANP peptides | |
| CN114113637A (en) | Thyroid stimulating hormone receptor antigen reagent and thyroid stimulating hormone receptor antibody quantitative determination kit | |
| US6537760B1 (en) | Receptor binding assay for detecting TSH-receptor auto-antibodies | |
| Takada et al. | Measurement of free insulin-like growth factor-I using immunoradiometric assay | |
| EP0351804B1 (en) | HCG peptides for use in antibody purification procedures | |
| US7015003B1 (en) | Receptor binding assays and reagent kit for detecting TSH receptor autoantibodies | |
| EP0721105A1 (en) | Sandwich immunoassay for N-peptide | |
| HK1114171B (en) | Copeptin-based diagnostic kit for disorders | |
| HK1172690B (en) | Copeptin-based kit | |
| Voellmy et al. | A novel two-site immunoradiometric assay for β-endorphin using nitrocellulose as solid phase |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU LV MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005782853 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007526400 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1499/MUMNP/2006 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2006/014775 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580021434.9 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005782853 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11573595 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2005782853 Country of ref document: EP |