WO2006088518A2 - Process for preparing chromium conversion coatings for iron and iron alloys - Google Patents
Process for preparing chromium conversion coatings for iron and iron alloys Download PDFInfo
- Publication number
- WO2006088518A2 WO2006088518A2 PCT/US2005/041413 US2005041413W WO2006088518A2 WO 2006088518 A2 WO2006088518 A2 WO 2006088518A2 US 2005041413 W US2005041413 W US 2005041413W WO 2006088518 A2 WO2006088518 A2 WO 2006088518A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- grams
- water soluble
- iron
- aqueous solution
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/05—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions
- C23C22/06—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6
- C23C22/34—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 containing fluorides or complex fluorides
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/05—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions
- C23C22/06—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6
- C23C22/48—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 not containing phosphates, hexavalent chromium compounds, fluorides or complex fluorides, molybdates, tungstates, vanadates or oxalates
- C23C22/50—Treatment of iron or alloys based thereon
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2222/00—Aspects relating to chemical surface treatment of metallic material by reaction of the surface with a reactive medium
- C23C2222/10—Use of solutions containing trivalent chromium but free of hexavalent chromium
Definitions
- This invention relates to a process for preparing zirconium-chromium conversion coatings on iron and iron alloys.
- the process comprises pretreating iron and its alloys with effective amounts of an acidic aqueous solution containing trivalent chromium compounds, at least one hexafluorozirconate and optionally, tetrafluoroborates and/or hexafluorosilicates, zinc compounds, surfactants, wetting agents and/or thickeners.
- this invention relates to a process for pretreating iron and iron alloys to improve the alloys adhesion bonding and corrosion resistant properties.
- the process comprises treating iron and its alloys with an acidic aqueous solution containing effective amounts of at least one water-soluble trivalent chromium compound, a water soluble alkali metal hexafluorozirconate, at least one water soluble alkali metal tetrafluoroborate and/or hexafluorosilicate, at least one water soluble divalent zinc compound, and effective amounts of water soluble thickeners and/or water soluble surfactants.
- a chemical pretreatment for iron alloys e.g. steel that is analogous to an aluminum pretreatment is not available but would be desirable. Such a coating would impart good paint adhesion to the steel substrate without needing a grit- blasted surface and thereby increase the corrosion resistance of the painted steel.
- a pretreatment of the alloy would also serve to prevent flash-rusting and allow for expanded handling times of the steel parts before they need to be painted. Such a pretreatment could be produced by merely immersing the components or steel parts in a tank of the pretreating solution, or by spraying the solution onto the alloy, or by wiping the solution on to the alloy.
- This invention relates to a process for preparing conversion coatings on iron and its alloys at ambient temperatures or higher e.g. ranging up to about 120 0 F. More specifically, this invention relates to a process of preparing conversion coatings on iron alloys such as steel to improve its corrosion resistance and adhesion bonding properties.
- the trivalent chromium process (TCP) of this invention comprises an acidic aqueous solution having a pH ranging from about 2.5 to 5.5 and preferably 3.7 to 4.0, and per liter of said acidic solution, from about 0.01 to 22 grams of a water soluble trivalent chromium compound, about 0.01 to 12 grams of an alkali metal hexafluorozirconate, from 0.0 to 12 grams of at least one fluorocompound selected from the group consisting of tetrafluoroborates, hexafluorosilicates and various combinations thereof in any ratio, from 0.0 to 10 grams of at least one water soluble divalent zinc compound, from 0.0 to 10 grams and preferable 0.5 to 1.5 grams of at least one water-soluble thickener, and/or from 0.0 to 10 and preferably 0.5 to 1.5 grams of at least one water soluble non-ionic, cationic or anionic surfactant or wetting agent.
- the pretreatment process of this invention also provides improved corrosion resistance of painted steel compared to steel that is painted by the direct-to-metal method.
- Fig. 1 is a photo of MIL-C-53022 primer on steel, with no grit blast.
- Fig. 2 is a photo of MEL-C-53022 primer on steel with TCP-P coating, with no grit blast.
- Fig. 3 is a photo of MIL-C-53022 primer on steel with Aqua Zen treatment, with no grit blast.
- Fig. 4 is a photo of MIL-C-53022 primer on steel with grit blast treatment.
- Fig. 5 is a photo of MEL-C-53022 primer on steel with TCP-P coating with grit blast treatment.
- Fig. 6 is a photo of MIL-C-53022 primer on steel with Aqua Zen treatment with grit blast treatment.
- This invention relates to the process of using an acidic aqueous solution having a pH ranging from about 2.5 to 5.5, and preferably from about 2.5 to 4.5 or 3.7 to 4.0 for preparing a zirconium-chromium conversion coating on iron and its alloys such as steel to improve adhesion bonding and the corrosion-resistance properties of the alloy.
- the process comprises preparing the pretreatment coating by using an acidic aqueous solution at temperatures ranging up to about 12O 0 F or higher which comprises from about 0.01 to 22 grams and preferably from about 0.01 to 10 grams e.g. 5.0 to 7.0 grams of at least one water soluble trivalent chromium compound e.g.
- chromium sulfate about 0.01 to 12 grams and preferably about 1.0 to 8.0 grams e.g. 6.0 to 8.0 grams of at least one alkali metal hexafluorozirconate, about 0.0 to 12 grams and preferably from about 0.01 to 1.2 grams e.g. 0.12 to 0.24 grams of at least one fluorocompound selected from the group consisting of alkali metal tetrafluoroborates, alkali metal hexafluorosilicates and various mixtures or combinations thereof in any ratio, and from about 0.0 to 10 grams and preferably 0.001 to 10 or 0.1 to 5.0 grams of at least one divalent zinc compound such as zinc sulfate.
- the solution is applied at about room temperature to the substrate via immersion, spray or wipe-on techniques similar to the process used for aluminum pretreatments.
- Solution dwell time ranges from about 1.0 to 10 minutes. With this solution, the 1.0 to 10 minute dwell time yields an optimum film for color change, paint adhesion, and corrosion resistance. More importantly for this invention, the 1.0 to 10 minute dwell time yields appreciable color change to the as- deposited coating that ranges from royal blue to blue-gray depending primarily on the chemical composition of the aqueous solution.
- the remaining unreacted solution is subsequently rinsed from the substrate with tap or deionized water. No additional post- treatments of the alloy are necessary.
- the pretreatment coating is allowed to dry thoroughly before subsequent painting.
- an unique feature is the addition of a thickener to the solution that aids in optimum film formation during spray and wipe- on applications by slowing down solution evaporation. This also mitigates the formation of powdery deposits that degrade paint adhesion.
- the addition of thickeners also aids in proper film formation during large area applications and mitigates the diluent effect of rinse water that remains on the substrate during processing from previous steps. This feature of the process yields films that have no streaks and are an improvement in coloration and corrosion protection.
- Water-soluble thickeners such as the cellulose compounds are present in the acidic aqueous solution in amounts ranging from about 0.0 to 10 grams per liter and preferably 0.5 to 1.5 e.g., or about 1.0 gram per liter of the aqueous solution.
- an effective but small amount of at least one water-soluble surfactant or wetting agent can be added to the acidic solution in amounts ranging from about 0.0 to 10 grams and preferably from 0.5 to 1.5 grams e.g. 1.0 gram per liter of the acidic solution.
- water soluble surfactants known in the prior art and therefore for purpose of this invention the surfactants can be selected from the group consisting of non-ionic, cationic and anionic surfactants.
- the trivalent chromium is added to the solution as a water-soluble trivalent chromium compound, preferably as a trivalent chromium salt.
- the chromium salt can be added conveniently to the solution in its water soluble form wherein the valence of the chromium is plus 3.
- some of the preferred chromium compounds are incorporated in the solution in the form of Cr 2 (SO 4 ) 3 , (NH 4 )Cr(SO 4 ⁇ or KCr(SQO 2 and any mixtures of these compounds.
- a preferred trivalent chromium salt concentration is within the range of about 5.0 to 7.0 grams per liter of the aqueous solution. It has been found that particularly good results are obtained from these processes when the trivalent chromium compound is present in solution in the preferred ranges.
- the alkali metal tetrafluoroborates and/or hexafluorosilicates can be added to the acidic solutions in amounts as low as 0.01 grams per liter up to the solubility limits of the compounds.
- about 50% weight percent of the fluorosilicate is added based on the weight of the fluorozirconate.
- about 4.0 grams per liter of fluorosilicate is added to the solution.
- An alternative is to add about 0.01 to 100 weight percent of the fluoroborate salt based on the weight of the fluorozirconate salt.
- 1.0 to 10 weight percent of the fluoroborate salt can be added based on the weight of the fluorozirconate salt.
- a specific example comprises from about 6.0 to 8.0 grams per liter of potassium hexafiuorozirconate, about 5.0 to 7.0 grams per liter of chromium HI sulfate basic, about 0.1 to 5.0 grams per liter of divalent zinc sulfate and about 0.12 to 0.24 grams per liter of potassium tetrafluoroborate and/or hexafluorosilicate.
- the fiuoroborates and/or fluorosilicates is that the solution is stable while the pH is maintained between about 2.5 and 5.5.
- the solutions may require small adjustments to the pH by the addition of effective amounts of a dilute acid or base to maintain the pH hi the range of about 2.5 to 5.5 and preferably from 2.5 to 4.5 or 3.7 to 4.0.
- the solution may contain at least one divalent zinc compound to improve the color and corrosion protection of the alloy when compared to other treatment or compositions that do not contain zinc.
- the amount of the zinc compounds can be varied to adjust the color imparted to the coating, from as little as about 0.001 grams per liter up to 10 grams per liter e.g. 0.1 to 5.0 grams of Zinc 2 +cation.
- the divalent zinc can be supplied by any chemical compound e.g. salt that dissolves in water and is compatible with the other components in the acid solution.
- Divalent zinc compounds that are water soluble at the required concentrations preferably include, for example, zinc acetate, zinc telluride, zinc tetrafluoroborate, zinc molybdate, zinc hexafluorosilicate, zinc sulfate and the like or any combination thereof in any ratio.
- the pretreatment of the iron alloys can be carried out at various temperatures including the temperature of the solution which ranges from ambient e.g. from about room temperature up to about 120°F or higher up to about 200 0 F. Room temperature is preferred, however, in that this eliminates the necessity for heating equipment.
- the coating may be air dried by any of the methods known in the art including, for example, oven drying, forced-air drying, exposure to infra-red lamps, and the like.
- the term "iron alloys" includes any iron alloy such as steel containing small but effective amounts of various other metals and non-metals such as carbon.
- a stable acidic aqueous solution having a pH ranging from about 3.4 to 4.0 for pretreating steel to provide a corrosion-resistant and a color recognized coating thereon comprises, per liter of solution, about 3.0 grams of trivalent chromium sulfate basic, about 4.0 grams of potassium hexafluorozirconate and about 1.0 gram of zinc sulfate.
- a stable acidic aqueous solution for treating steel to form a corrosion-resistant coating thereon comprises, per liter of solution, about 3.0 grams of trivalent chromium sulfate basic, about 4.0 grams of potassium hexafluorozirconate, and from about 0.0 to 0.12 grams of potassium tetrafluoroborate.
- a stable acidic aqueous solution for treating steel to provide a corrosion-resistant and a color recognized coating thereon comprises, per liter of solution, about 3.0 grams of trivalent chromium sulfate basic, about 4.0 grams of potassium hexafluorozirconate, about 0.12 grams of potassium tetrafluoroborate and about 2.0 grams of divalent zinc sulfate.
- TCP-P potassium hexafluorozirconate
- Coupons were then allowed to dry in a rack at ambient conditions. The resulting coatings were a deep royal blue in color, easily visible from across the laboratory. This is a critical indication for quality control during processing so that the processors have a way of telling that the coating is complete.
- control sets of 4130 were prepared by cleaning (Turco HTC) in the above-identified solution. A subset of these steel panels or coupons were grit blasted with alumina grit media to produce an average profile of 1.0 to 1.5 mils, a standard for a direct-to-metal painting. These steel coupons were then painted with MIL-C-53022 primer to approximately 1.0 mil thick. One sub-set of coupons was painted with Aqua Zen "wash primer" before the primer.
- the Aqua Zen is an industry standard coating used to promote adhesion and improve corrosion performance of the coating system.
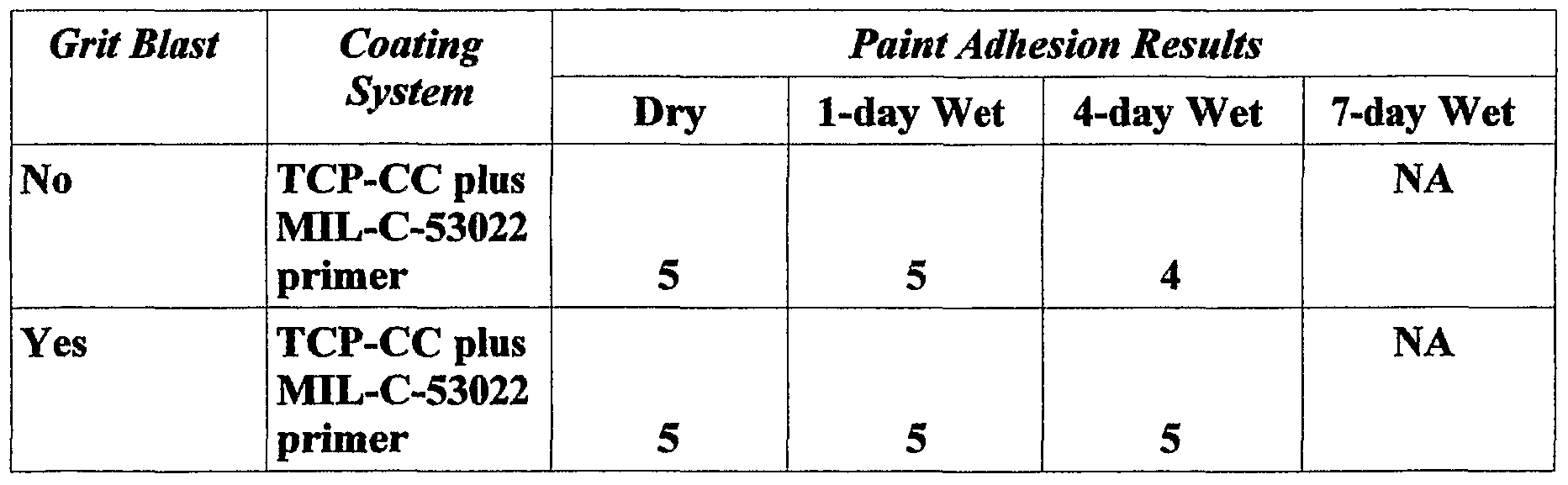
- the painted coupons sat for 14 days at ambient conditions to allow the paint to cure. After curing, the coupons were subjected to painted adhesion and painted corrosion tests. Table 1 describes the paint adhesion results. Ratings of 4 and 5 are considered passing, and 0 to 3 are failing.
- TCP-P coating provides excellent paint adhesion to the 4130 steel whether or not the steel was grit blasted.
- the TCP-P coating also shows better overall performance in comparison to the two controls panels which are commonly used on DoD equipment.
- the TCP-P coating is a better alternative to the Aqua Zen treatment and shows excellent performance with the MIL-C-53022 primer without the grit blast, where paint adhesion is very poor with only the primer.
- Fig's. 1 through 6 (photos) show examples of 7-day wet-tape adhesion for each coating system described in Table 1.
- Conversion coatings were applied to 4130 steel as follows: Test coupons were prepared by the same process as in Example A.
- the composition (TCP-CC) solution comprised 3.0 grams per liter of chromium sulfate basic, 6.0 grams per liter of potassium hexafluorozirconate, 0.18 grams per liter of potassium tetrafluoroborate, and 2.0 grams per liter of zinc sulfate.
- Table 2 sets-forth the paint adhesion results from these coated coupons that are comparable to the results in Table 1.
- TCP-CC color change composition
- the water soluble surfactants can be added to the trivalent chromium solutions in amounts ranging from about 0 to 10 grams per liter and preferably about 0.5 to 1.5 grams per liter.
- the surfactants are added to the aqueous solution to provide better wetting properties by lowering the surface tension thereby insuring complete coverage, and a more uniform film on the iron alloy substrates.
- the surfactants include at least one water soluble compound selected from the group consisting of non-ionic, anionic, and cationic surfactants.
- Some of the better known water soluble surfactants include the monocarboxyl imidoazoline, alkylsulfate sodium salts (DUPONOL®), tridecyloxy poly(alkyleneoxy ethanol), ethoxylated or propoxylated alkylphenol (IGEP AL®), alkylsulfonamides, alkaryl sulfonates, palmiticalkanol amides (CENTROL®), octylphenyl polyethoxy ethanol (TRITON®), sorbitan monopalmitate (SPAN®), dodecylphenyl polyethyleneglycol ether (e.g.
- alkyl pyrrolidone alkyl pyrrolidone
- polyalkoxylated fatty acid esters alkylbenzene sulfonates
- Other known water soluble surfactants include the alkylphenol alkoxylates, preferably the nonylphenol ethoxylates, and adducts of ethylene oxide with fatty amines; also see the publication: “Surfactants and Detersive Systems", published by John Wiley & Sops in Kirk-Othmer's Encyclopedia of Chemical Technology, 3 rd Ed.
- thickening agents can be added to retain the aqueous solution on the surface for sufficient contact time.
- the thickeners employed are known inorganic and preferably the organic water soluble thickeners added to the trivalent chromium solutions in effective amounts e.g. at sufficient concentrations ranging from about 0 to 10 grams per liter and preferably 0.5 to 1.5 grams per liter of the acidic solution.
- Specific examples of some preferred thickeners include the cellulose compounds, e.g. hydroxypropyl cellulose (e.g. Klucel), ethyl cellulose, hydroxyethyl cellulose, hydroxymethyl cellulose, or methyl cellulose and mixtures thereof.
- Other water soluble inorganic thickeners include colloidal silica, clays such as bentonite, starches, gum arabic, tragacanth, agar and various combinations.
- the solution can be applied via immersion, spray or wipe-on techniques.
- the TCP solutions can be used at elevated temperatures ranging up to 65 °C or higher and optimally applied via immersion to further improve the corrosion resistance of the coatings.
- Solution dwell time ranges from about 1 to 60 minutes, and preferably 5 to 15 minutes at about 8O 0 F. After dwelling, the remaining solution is then thoroughly rinsed from the substrate with tap or deionized water. No additional chemical manipulations of the deposited films are necessary for excellent performance. However, an application of a strong oxidizing solution can yield a film having additional corrosion resistance. The additional corrosion resistance is presumed to be due to the formation of hexavalent chromium in the film derived from the trivalent chromium.
- aqueous solutions may be sprayed from a spray tank apparatus designed to replace immersion tanks. This concept also reduces active chemical volume from about 1,000 gallons to about 30 to 50 gallons. Another feature of this invention is that this process provides coatings with corrosion resistance that is better or at least equivalent to other known coatings produced by sulfuric, chromic, or boric-sulfuric compositions.
Landscapes
- Chemical & Material Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Chemical Treatment Of Metals (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2005327545A AU2005327545A1 (en) | 2005-02-15 | 2005-11-14 | Process for preparing chromium conversion coatings for iron and iron alloys |
| MX2007009798A MX2007009798A (en) | 2005-02-15 | 2005-11-14 | Process for preparing chromium conversion coatings for iron and iron alloys. |
| JP2007555077A JP2008530360A (en) | 2005-02-15 | 2005-11-14 | Method for preparing chromium conversion coatings for iron and iron alloys |
| EP05851686A EP1848840A2 (en) | 2005-02-15 | 2005-11-14 | Process for preparing chromium conversion coatings for iron and iron alloys |
| CA002598397A CA2598397A1 (en) | 2005-02-15 | 2005-11-14 | Process for preparing chromium conversion coatings for iron and iron alloys |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/058,715 US20100032060A1 (en) | 2005-02-15 | 2005-02-15 | Process for preparing chromium conversion coatings for magnesium alloys |
| US11/076,106 | 2005-02-15 | ||
| US11/058,715 | 2005-02-15 | ||
| US11/076,106 US20060180247A1 (en) | 2005-02-15 | 2005-02-15 | Process for preparing chromium conversion coatings for iron and iron alloys |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2006088518A2 true WO2006088518A2 (en) | 2006-08-24 |
| WO2006088518A3 WO2006088518A3 (en) | 2007-11-01 |
Family
ID=36916890
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2005/041413 Ceased WO2006088518A2 (en) | 2005-02-15 | 2005-11-14 | Process for preparing chromium conversion coatings for iron and iron alloys |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP1848840A2 (en) |
| JP (1) | JP2008530360A (en) |
| KR (1) | KR20070118085A (en) |
| AU (1) | AU2005327545A1 (en) |
| CA (1) | CA2598397A1 (en) |
| MX (1) | MX2007009798A (en) |
| WO (1) | WO2006088518A2 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20120187350A1 (en) * | 2011-01-21 | 2012-07-26 | Craig Matzdorf | Aluminum alloy coated pigments and corrosion-resistant coatings |
| EP2880103A4 (en) * | 2012-08-01 | 2016-04-06 | Us Navy | OXIDE COATED METAL PIGMENTS AND FILMOGENIC COMPOSITIONS |
| US9797044B2 (en) | 2011-07-15 | 2017-10-24 | Yuken Industry Co., Ltd. | Aqueous acidic composition for forming chromium-containing chemical conversion coating on iron-based member, and iron-based member having chemical conversion coating formed using the composition |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6461449B1 (en) * | 1997-10-07 | 2002-10-08 | Henkel Corporation | Conversion coating zinciferous surfaces to resist blackening and white rust |
| US20040206266A1 (en) * | 2001-02-14 | 2004-10-21 | Metal Coatings International Inc. | Particulate metal alloy coating for providing corrosion protection |
| US7018486B2 (en) * | 2002-05-13 | 2006-03-28 | United Technologies Corporation | Corrosion resistant trivalent chromium phosphated chemical conversion coatings |
-
2005
- 2005-11-14 JP JP2007555077A patent/JP2008530360A/en active Pending
- 2005-11-14 EP EP05851686A patent/EP1848840A2/en active Pending
- 2005-11-14 WO PCT/US2005/041413 patent/WO2006088518A2/en not_active Ceased
- 2005-11-14 CA CA002598397A patent/CA2598397A1/en not_active Abandoned
- 2005-11-14 KR KR1020077021175A patent/KR20070118085A/en not_active Withdrawn
- 2005-11-14 MX MX2007009798A patent/MX2007009798A/en unknown
- 2005-11-14 AU AU2005327545A patent/AU2005327545A1/en not_active Abandoned
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20120187350A1 (en) * | 2011-01-21 | 2012-07-26 | Craig Matzdorf | Aluminum alloy coated pigments and corrosion-resistant coatings |
| US8277688B2 (en) * | 2011-01-21 | 2012-10-02 | The United States Of America As Represented By The Secretary Of The Navy | Aluminum alloy coated pigments and corrosion-resistant coatings |
| US9797044B2 (en) | 2011-07-15 | 2017-10-24 | Yuken Industry Co., Ltd. | Aqueous acidic composition for forming chromium-containing chemical conversion coating on iron-based member, and iron-based member having chemical conversion coating formed using the composition |
| EP2880103A4 (en) * | 2012-08-01 | 2016-04-06 | Us Navy | OXIDE COATED METAL PIGMENTS AND FILMOGENIC COMPOSITIONS |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2008530360A (en) | 2008-08-07 |
| CA2598397A1 (en) | 2006-08-24 |
| KR20070118085A (en) | 2007-12-13 |
| AU2005327545A1 (en) | 2006-08-24 |
| EP1848840A2 (en) | 2007-10-31 |
| WO2006088518A3 (en) | 2007-11-01 |
| MX2007009798A (en) | 2007-10-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6511532B2 (en) | Post-treatment for anodized aluminum | |
| EP1404894B1 (en) | Corrosion resistant coatings for aluminum and aluminum alloys | |
| US20060180247A1 (en) | Process for preparing chromium conversion coatings for iron and iron alloys | |
| US6527841B2 (en) | Post-treatment for metal coated substrates | |
| US6521029B1 (en) | Pretreatment for aluminum and aluminum alloys | |
| EP1853750B1 (en) | Process for sealing phosphoric acid anodized aluminums | |
| US7811391B2 (en) | Composition and process for preparing protective coatings on metal substrates | |
| EP1863952B1 (en) | Composition and process for preparing protective coatings on metal substrates | |
| US20070099022A1 (en) | Non-chromium post-treatment for aluminum coated steel | |
| US20100032060A1 (en) | Process for preparing chromium conversion coatings for magnesium alloys | |
| EP1848840A2 (en) | Process for preparing chromium conversion coatings for iron and iron alloys | |
| US20070095437A1 (en) | Non-chromium conversion coatings for ferrous alloys |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580048346.8 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2598397 Country of ref document: CA Ref document number: 2007555077 Country of ref document: JP Ref document number: MX/a/2007/009798 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005851686 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005327545 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 3687/CHENP/2007 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2005327545 Country of ref document: AU Date of ref document: 20051114 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1200701802 Country of ref document: VN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005327545 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020077021175 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005851686 Country of ref document: EP |
|
| ENPW | Started to enter national phase and was withdrawn or failed for other reasons |
Ref document number: PI0519911 Country of ref document: BR Kind code of ref document: A2 Free format text: PEDIDO CONSIDERADO RETIRADO EM RELACAO AO BRASIL E ARQUIVADO POR NAO ATENDER AS DETERMINACOES REFERENTES A ENTRADA DO PEDIDO NA FASE NACIONAL. |