WO2007024536A2 - Process for preparing maytansinoid antibody conjugates - Google Patents
Process for preparing maytansinoid antibody conjugates Download PDFInfo
- Publication number
- WO2007024536A2 WO2007024536A2 PCT/US2006/031653 US2006031653W WO2007024536A2 WO 2007024536 A2 WO2007024536 A2 WO 2007024536A2 US 2006031653 W US2006031653 W US 2006031653W WO 2007024536 A2 WO2007024536 A2 WO 2007024536A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cell
- drug
- antibody
- mixture
- chromatography
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C[C@@](C(OC(CC(N(C)c(cc(CC(C)=CC=CC(C(C1)(N2)O)OC)cc3OC)c3Cl)=O)C3(C)OC3C(C)C1OC2=O)=O)N(C)C(*)=O Chemical compound C[C@@](C(OC(CC(N(C)c(cc(CC(C)=CC=CC(C(C1)(N2)O)OC)cc3OC)c3Cl)=O)C3(C)OC3C(C)C1OC2=O)=O)N(C)C(*)=O 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/6807—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug or compound being a sugar, nucleoside, nucleotide, nucleic acid, e.g. RNA antisense
- A61K47/6809—Antibiotics, e.g. antitumor antibiotics anthracyclins, adriamycin, doxorubicin or daunomycin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/20—Interleukins [IL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/68033—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug being a maytansine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6867—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from a cell of a blood cancer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2896—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against molecules with a "CD"-designation, not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2839—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the integrin superfamily
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2884—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against CD44
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/40—Immunoglobulins specific features characterized by post-translational modification
Definitions
- This invention pertains to a process for preparing conjugates of substantially high purity and stability, wherein the conjugates comprise a cell-binding agent chemically coupled to a drug.
- Such compounds are typically referred to as toxin, radionuclide, and drag "conjugates," respectively. Often they also are referred to as immunoconjugates, radioimmunoconjugates, and immunotoxins.
- Tumor cell killing occurs upon binding of the drag conjugate to a tumor cell and release or/and activation of the cytotoxic activity of the drug.
- the selectivity afforded by drug conjugates minimizes toxicity to normal cells, thereby enhancing tolerability of the drag in the patient.
- Patents 5,208,020 and 5,416,064 further describe conjugation of a modified antibody with an excess of a sulfhydryl-containing cytotoxic agent at pH 7, followed by purification on SephadexTM G25 chromatography columns. Purification of antibody-drug conjugates by size exclusion chromatography (SEC) also has been described (see, e.g., Liu et al., Proc. Natl. Acad. Set (USA), 93: 8618-8623 (1996), and Chari et al., Cancer Research, 52: 127-131 (1992)).
- SEC size exclusion chromatography
- the invention provides a process for preparing a conjugate of substantially high purity and stability comprising a cell-binding agent chemically coupled to a drag.
- the process comprises (a) contacting a cell-binding agent with a bifunctional crosslinking reagent to covalently attach a linker to the cell-binding agent and thereby prepare a first mixture comprising cell-binding agents having linkers bound thereto, (b) subjecting the first mixture to tangential flow filtration, adsorptive chromatography, adsorptive filtration, selective precipitation, or combination thereof, and thereby prepare a purified first mixture of cell- binding agents having linkers bound thereto, (c) conjugating a drug to the cell-binding agents having linkers bound thereto in the purified first mixture by reacting the cell-binding agents having linkers bound thereto with a drug in a solution having a pH of about 4 to about 9 to prepare a second mixture comprising (i) cell-binding agent chemically coupled through the linker to the drug, (ii
- the invention provides a process for preparing cell-binding agent-drug conjugates of substantially high purity and stability.
- Such compositions can be used for treating diseases because of the high purity and stability of the conjugates.
- Compositions comprising a cell- binding agent, such as an antibody, chemically coupled to a drug, such as a maytansinoid, are described in, for example, U.S. Patent Application Publication No. 2004/0241174 Al.
- substantially high purity is considered to be: (a) greater than 90%, preferably greater than 95%, of conjugate species are monomeric, and/or (b) free drug level in the conjugate preparation is less than 2% (relative to total drug).
- the inventive process comprises (a) modifying the cell-binding agent with a bifunctional crosslinking reagent to covalently attach a linker to the cell-binding agent and thereby prepare a first mixture comprising cell-binding agents having linkers bound thereto, (b) subjecting the first mixture to tangential flow filtration, adsorptive chromatography, adsorptive filtration, selective precipitation, or combinations thereof, to purify the cell-binding agents having linkers bound thereto from other components of the first mixture and thereby prepare a purified first mixture of cell-binding agents having linkers bound thereto, (c) conjugating a drug to the cell-binding agents having linkers bound thereto in the purified first mixture by reacting the cell-binding agents having linkers bound thereto with the drug in a solution having a pH of about 4 to about 9 to prepare a second mixture comprising (i) cell-binding agent chemically coupled through the linker to the drug, (ii) free drug, and (iii)
- tangential flow filtration also known as cross flow filtration, ultrafiltration and diafiltration
- adsorptive chromatography resins are utilized in the purification steps.
- TFF tangential flow filtration
- step c tangential flow filtration
- step d adsorptive chromatography resin
- the adsorptive chromatography resin is a non-ion exchange resin.
- TFF is utilized in both purification steps, or adsorptive chromatography resins are utilized in both purification steps.
- an adsorptive chromatography resin is utilized in the first purification step, and TFF is utilized in the second purification step.
- TFF is utilized in the first and/or second purification step as well.
- Any suitable TFF systems may be utilized, including a Pellicon type system (Millipore, Billerica, MA), a Sartocon Cassette system (Sartorius AG, Edgewood, NY), and a Centrasette type system (Pall Corp., East Hills, NY).
- any suitable adsorptive chromatography resin may be utilized.
- Preferred adsorptive chromatography resins include resins for hydroxyapatite chromatography, hydrophobic charge induction chromatography (HCIC), hydrophobic interaction chromatography (HIC), ion exchange chromatography, mixed mode ion exchange chromatography, immobilized metal affinity chromatography (IMAC), dye ligand chromatography, affinity chromatography, reversed phase chromatography, and combinations thereof.
- Suitable hydroxyapatite resins include ceramic hydroxyapatite (CHT Type I and Type II, Bio-Rad Laboratories, Hercules, CA), HA Ultrogel hydroxyapatite (Pall Corp., East Hills, NY), and ceramic fluoroapatite (CFT Type I and Type II, Bio-Rad Laboratories, Hercules, CA).
- CHT Type I and Type II Ceramic hydroxyapatite
- HA Ultrogel hydroxyapatite Pall Corp., East Hills, NY
- CFT Type I and Type II Bio-Rad Laboratories, Hercules, CA
- An example of a suitable HCIC resin is MEP Hypercel resin (Pall Corp., East Hills, NY).
- HIC resins examples include Butyl-Sepharose, Hexyl-Sepaharose, Phenyl-Sepharose, and Octyl Sepharose resins (all from GE Healthcare, Piscataway, NJ), as well as Macro-prep Methyl and Macro-Prep t-Butyl resins (Biorad Laboratories, Hercules, CA).
- suitable ion exchange resins include SP- Sepharose, CM-Sepharose, and Q-Sepharose resins (all from GE Healthcare, Piscataway, NJ), and Unosphere S resin (Bio-Rad Laboratories, Hercules, CA).
- suitable mixed mode ion exchangers include Bakerbond ABx resin (JT Baker, Phillipsburg NJ).
- suitable IMAC resins include Chelating Sepharose resin (GE Healthcare, Piscataway, NJ) and Profinity IMAC resin (Bio-Rad Laboratories, Hercules, CA).
- suitable dye ligand resins include Blue Sepharose resin (GE Healthcare, Piscataway, NJ) and Affi-gel Blue resin (Bio-Rad Laboratories, Hercules, CA).
- suitable affinity resins include Protein A Sepharose resin (e.g., MabSelect, GE Healthcare, Piscataway, NJ), where the cell binding agent is an antibody, and lectin affinity resins, e.g.
- Lentil Lectin Sepharose resin (GE Healthcare, Piscataway, NJ), where the cell binding agent bears appropriate lectin binding sites.
- an antibody specific to the cell binding agent may be used.
- Such an antibody can be immobilized to, for instance, Sepharose 4 Fast Flow resin (GE Healthcare, Piscataway, NJ).
- suitable reversed phase resins include C4, C8, and Cl 8 resins (Grace Vydac, Hesperia, CA).
- a first mixture is produced comprising the cell-binding agent having linkers bound thereto, as well as reactants and other byproducts.
- Purification of the modified cell-binding agent from reactants and by-products is carried out by subjecting the first mixture to a purification process.
- the first mixture can be purified using tangential flow filtration (TFF), e.g., a membrane-based tangential flow filtration process, adsorptive chromatography, adsorptive filtration, or selective precipitation, or any other suitable purification process, as well as combinations thereof.
- TMF tangential flow filtration
- This first purification step provides a purified first mixture, i.e., an increased concentration of the cell-binding agents having linkers bound thereto and a decreased amount of unbound bifunctional crosslinking reagent, as compared to the first mixture prior to purification in accordance with the invention.
- a drug is conjugated to the cell-binding agents having linkers bound thereto in the first purified mixture by reacting the cell-binding agents having linkers bound thereto with a drug in a solution having a pH from about 4 to about 9, whereupon a second mixture comprising (i) the cell-binding agent chemically coupled through the linker to the drug, (ii) free drug, and (iii) reaction by-products is produced.
- the conjugation reaction is performed at a pH of about 4 to about pH 9
- the reaction is preferably performed at a pH of about 6 or below or at a pH of about 6.5 or greater, most preferably at a pH of about 4 to about 6 or at a pH of about 6.5 to about 9, and especially at a pH of 4 to less than 6 or at a pH of greater than 6.5 to 9.
- the conjugation step is performed at a pH of about 6.5 or greater, some sulhydryl-containing drugs may be prone to dimerize by disulfide-bond formation. Removal of trace metals and/or oxygen from the reaction mixture, as well as optional addition of antioxidants or the use of linkers with more reactive leaving groups, or addition of drug in more than one aliquot, may be required to allow for efficient reaction in such a situation.
- purification of the modified cell binding agent may be omitted.
- the drug may be added simultaneously with the crosslinking reagent or at some later point, e.g., 1, 2, 3, or more hours after addition of the crosslinking reagent to the cell binding agent.
- the inventive method may optionally include the addition of sucrose to the conjugation step used in the inventive method to increase solubility and recovery of the cell- binding agent-drug conjugates.
- sucrose is added at a concentration of about 0.1% (w/v) to about 20% (w/v) (e.g., about 0.1% (w/v), 1% (w/v), 5% (w/v), 10% (w/v), 15% (w/v), or 20% (w/v)).
- sucrose is added at a concentration of about 1% (w/v) to 10% (w/v) (e.g., about 2% (w/v), about 4% (w/v), about 6% (w/v), or about 8% (w/v)).
- the conjugation reaction also can comprise the addition of a buffering agent.
- a buffering agent Any suitable buffering agent known in the art can be used. Suitable buffering agents include, for example, a citrate buffer, an acetate buffer, a succinate buffer, and a phosphate buffer.
- the second mixture is subjected to a purification step.
- the second mixture can be purified using tangential flow filtration (TFF), e.g., a membrane-based tangential flow filtration process, adsorptive chromatography, absorptive filtration, selective precipitation, or any other suitable purification process, as well as combinations thereof, which are set-forth herein.
- TMF tangential flow filtration
- This second purification step provides a purified second mixture, i.e., an increased concentration of the cell-binding agents chemically coupled through the linkers to the drug and a decreased amount of one or more other components of the second mixture, as compared to the second mixture prior to purification in accordance with the invention.
- the cell-binding agent can be any suitable agent that binds to a cell, typically and preferably an animal cell (e.g., a human cell).
- the cell-binding agent preferably is a peptide or a polypeptide.
- Suitable cell-binding agents include, for example, antibodies (e.g., monoclonal antibodies and fragments thereof), lymphokines, hormones, growth factors, nutrient-transport molecules (e.g., transferrin), and any other agent or molecule that specifically binds a target molecule on the surface of a cell.
- antibody refers to any immunoglobulin, any immunoglobulin fragment, such as Fab, F(ab') 2 , dsFv, sFv, diabodies, and triabodies, or immunoglobulin chimera, which can bind to an antigen on the surface of a cell (e.g., which contains a complementarity determining region (CDR)).
- CDR complementarity determining region
- Any suitable antibody can be used as the cell-binding agent.
- One of ordinary skill in the art will appreciate that the selection of an appropriate antibody will depend upon the cell population to be targeted.
- cell surface molecules i.e., antigens
- a particular cell population typically and preferably a diseased cell population
- Cell surface expression profiles are known for a wide variety of cell types, including tumor cell types, or, if unknown, can be determined using routine molecular biology and histochemistry techniques.
- the antibody can be polyclonal or monoclonal, but is most preferably a monoclonal antibody.
- polyclonal antibodies refer to heterogeneous populations of antibody molecules, typically contained in the sera of immunized animals.
- Monoclonal antibodies refer to homogenous populations of antibody molecules that are specific to a particular antigen.
- Monoclonal antibodies are typically produced by a single clone of B lymphocytes ("B cells").
- B cells B cells
- Monoclonal antibodies may be obtained using a variety of techniques known to those skilled in the art, including standard hybridoma technology (see, e.g., K ⁇ hler and Milstein, Eur. J.
- the hybridoma method of producing monoclonal antibodies typically involves injecting any suitable animal, typically and preferably a mouse, with an antigen (i.e., an "immunogen"). The animal is subsequently sacrificed, and B cells isolated from its spleen are fused with human myeloma cells.
- an antigen i.e., an "immunogen”
- hybrid cell is produced (i.e., a "hybridoma"), which proliferates indefinitely and continuously secretes high titers of an antibody with the desired specificity in vitro.
- Any appropriate method known in the art can be used to identify hybridoma cells that produce an , antibody with the desired specificity. Such methods include, for example, enzyme-linked immunosorbent assay (ELISA), Western blot analysis, and radioimmunoassay.
- ELISA enzyme-linked immunosorbent assay
- the population of hybridomas is screened to isolate individual clones, each of which secretes a single antibody species to the antigen.
- each hybridoma is a clone derived from fusion with a single B cell, all the antibody molecules it produces are identical in structure, including their antigen binding site and isotype.
- Monoclonal antibodies also may be generated using other suitable techniques including EBV-hybridoma technology (see, e.g., Haskard and Archer, J Immunol.
- the monoclonal antibody can be isolated from or produced in any suitable animal, but is preferably produced in a mammal, more preferably a mouse or human, and most preferably a human. Methods for producing an antibody in mice are well known to those skilled in the art and are described herein. With respect to human antibodies, one of ordinary skill in the art will appreciate that polyclonal antibodies can be isolated from the sera of human subjects vaccinated or immunized with an appropriate antigen. Alternatively, human antibodies can be generated by adapting known techniques for producing human antibodies in non-human animals such as mice (see, e.g., U.S. Patents 5,545,806, 5,569,825, and 5,714,352, and U.S. Patent Application Publication No.
- human antibodies While being the ideal choice for therapeutic applications in humans, human antibodies, particularly human monoclonal antibodies, typically are more difficult to generate than mouse monoclonal antibodies. Mouse monoclonal antibodies, however, induce a rapid host antibody response when administered to humans, which can reduce the therapeutic or diagnostic potential of the antibody-drug conjugate. To circumvent these complications, a monoclonal antibody preferably is not recognized as "foreign" by the human immune system.
- phage display can be used to generate the antibody.
- phage libraries encoding antigen-binding variable (V) domains of antibodies can be generated using standard molecular biology and recombinant DNA techniques (see, e.g., Sambrook et al. (eds.), Molecular Cloning, A Laboratory Manual, 3 rd Edition, Cold Spring Harbor Laboratory Press, New York (2001)). Phage encoding a variable region with the desired specificity are selected for specific binding to the desired antigen, and a complete human antibody is reconstituted comprising the selected variable domain.
- Nucleic acid sequences encoding the reconstituted antibody are introduced into a suitable cell line, such as a myeloma cell used for hybridoma production, such that human antibodies having the characteristics of monoclonal antibodies are secreted by the cell (see, e.g., Janeway et al., supra, Huse et al., supra, and U.S. Patent 6,265,150).
- a suitable cell line such as a myeloma cell used for hybridoma production
- monoclonal antibodies can be generated from mice that are transgenic for specific human heavy and light chain immunoglobulin genes.
- Such methods are known in the art and described in, for example, U.S. Patents 5,545,806 and 5,569,825, and Janeway et al., supra.
- the antibody is a humanized antibody.
- a "humanized” antibody is one in which the complementarity-determining regions (CDR) of a mouse monoclonal antibody, which form the antigen binding loops of the antibody, are grafted onto the framework of a human antibody molecule. Owing to the similarity of the frameworks of mouse and human antibodies, it is generally accepted in the art that this approach produces a monoclonal antibody that is antigenically identical to a human antibody but binds the same antigen as the mouse monoclonal antibody from which the CDR sequences were derived. Methods for generating humanized antibodies are well known in the art and are described in detail in, for example, Janeway et al., supra, U.S.
- Humanized antibodies can also be generated using the antibody resurfacing technology described in U.S. Patent 5,639,641 and Pedersen et al., J MoI Biol, 235: 959- 973 (1994). While the antibody employed in the conjugate of the inventive composition most preferably is a humanized monoclonal antibody, a human monoclonal antibody and a mouse monoclonal antibody, as described above, are also within the scope of the invention.
- Antibody fragments that have at least one antigen binding site, and thus recognize and bind to at least one antigen or receptor present on the surface of a target cell also are within the scope of the invention.
- proteolytic cleavage of an intact antibody molecule can produce a variety of antibody fragments that retain the ability to recognize and bind antigens.
- limited digestion of an antibody molecule with the protease papain typically produces three fragments, two of which are identical and are referred to as the Fab fragments, as they retain the antigen binding activity of the parent antibody molecule.
- F(ab') 2 fragment Cleavage of an antibody molecule with the enzyme pepsin normally produces two antibody fragments, one of which retains both antigen-binding arms of the antibody molecule, and is thus referred to as the F(ab') 2 fragment.
- Reduction of a F(ab') 2 fragment with dithiothreitol or mercaptoethylamine produces a fragment referred to as a Fab' fragment.
- a single-chain variable region fragment (sFv) antibody fragment which consists of a truncated Fab fragment comprising the variable (V) domain of an antibody heavy chain linked to a V domain of a light antibody chain via a synthetic peptide, can be generated using routine recombinant DNA technology techniques (see, e.g., Janeway et al., supra).
- disulfide-stabilized variable region fragments (dsFv) can be prepared by recombinant DNA technology (see, e.g., Reiter et al., Protein Engineering, 7: 697-704 (1994)).
- Antibody fragments in the context of the invention are not limited to these exemplary types of antibody fragments.
- Antibody fragments that recognizes and binds to a desired cell surface receptor or antigen can be employed.
- Antibody fragments are further described in, for example, Parham, J Immunol, 131: 2895-2902 (1983), Spring et al, J Immunol, 113: 470-478 (1974), and Nisonoff et A., Arch. Biochem. Biophys., 89: 230-244 (1960).
- Antibody-antigen binding can be assayed using any suitable method known in the art, such as, for example, radioimmunoassay (RIA), ELISA, Western blot, immunoprecipitation, and competitive inhibition assays (see, e.g., Janeway et al., supra, and U.S. Patent Application Publication No. 2002/0197266 Al).
- the antibody can be a chimeric antibody or an antigen binding fragment thereof.
- chimeric it is meant that the antibody comprises at least two immunoglobulins, or fragments thereof, obtained or derived from at least two different species (e.g., two different immunoglobulins, such as a human immunoglobulin constant region combined with a murine immunoglobulin variable region).
- the antibody also can be a domain antibody (dAb) or an antigen binding fragment thereof, such as, for example, a camelid antibody (see, e.g., Desmyter et al., Nature Struct.
- the monoclonal antibody J5 is a murine IgG2a antibody that is specific for Common Acute Lymphoblastic Leukemia Antigen (CALLA) (Rite et al., Nature, 283: 583-585 (1980)), and can be used to target cells that express CALLA (e.g., acute lymphoblastic leukemia cells).
- CALLA Common Acute Lymphoblastic Leukemia Antigen
- the monoclonal antibody MY9 is a murine IgGl antibody that binds specifically to the CD33 antigen (Griffin et al., Leukemia Res., 8: 521 (1984)), and can be used to target cells that express CD33 (e.g., acute myelogenous leukemia (AML) cells).
- AML acute myelogenous leukemia
- the monoclonal antibody anti-B4 (also referred to as B4) is a murine IgGl antibody that binds to the CD19 antigen on B cells (Nadler et al., J. Immunol., 131: 244-250 (1983)), and can be used to target B cells or diseased cells that express CD19 (e.g., non-Hodgkin's lymphoma cells and chronic lymphoblastic leukemia cells).
- N901 is a murine monoclonal antibody that binds to the CD56 (neural cell adhesion molecule) antigen found on cells of neuroendocrine origin, including small cell lung tumor, which can be used in the conjugate to target drugs to cells of neuroendocrine origin.
- the J5, MY9, and B4 antibodies preferably are resurfaced or humanized prior to their use as part of the conjugate. Resurfacing or humanization of antibodies is described in, for example, Roguska et al., Proc. Natl. Acad. Sci. USA, 91: 969-73 (1994).
- the monoclonal antibody C242 binds to the CanAg antigen (see, e.g., U.S. Patent 5,552,293), and can be used to target the conjugate to CanAg expressing tumors, such as colorectal, pancreatic, non-small cell lung, and gastric cancers.
- HuC242 is a humanized form of the monoclonal antibody C242 (see, e.g., U.S. Patent 5,552,293).
- the hybridoma from which HuC242 is produced is deposited with ECACC identification Number 90012601.
- HuC242 can be prepared using CDR-grafting methodology (see, e.g., U.S.
- HuC242 can be used to target the conjugate to tumor cells expressing the CanAg antigen, such as, for example, colorectal, pancreatic, non-small cell lung, and gastric cancer cells.
- an anti-MUCl antibody can be used as the cell-binding agent in the conjugate.
- Anti-MUCl antibodies include, for example, anti-HMFG-2 (see, e.g., Taylor-Papadimitriou et al., Int. J. Cancer, 28: 17-21 (1981)), hCTMOl (see, e.g., van Hof et al., Cancer Res., 56: 5179-5185 (1996)), and DS6.
- Prostate cancer cells also can be targeted with the conjugate by using an anti-prostate-specific membrane antigen (PSMA) as the cell-binding agent, such as J591 (see, e.g., Liu et al., Cancer Res., 57: 3629-3634 (1997)).
- PSMA anti-prostate-specific membrane antigen
- cancer cells that express the Her2 antigen such as breast, prostate, and ovarian cancers, can be targeted using the antibody trastuzumab.
- Anti-IGF-IR antibodies that bind to insulin-like growth factor receptor also can be used in the conjugate.
- Particularly preferred antibodies are humanized monoclonal antibodies, examples of which include huN901, huMy9-6, huB4, huC242, trastuzumab, bivatuzumab, sibrotuzumab, and rituximab (see, e.g., U.S. Patents 5,639,641 and 5,665,357, U.S. Provisional Patent Application No. 60/424,332 (which is related to U.S. Patent Application Publication No.
- the antibody is the huN901 humanized monoclonal antibody or the huMy9-6 humanized monoclonal antibody.
- Other preferred antibodies include CNTO95, huDS6, huB4, and huC242.
- Other humanized monoclonal antibodies are known in the art and can be used in connection with the invention.
- the cell-binding agent preferably is an antibody
- the cell-binding agent also can be a non-antibody molecule.
- suitable non-antibody molecules include, for example, interferons (e.g., alpha, beta, or gamma interferon), lymphokines (e.g., interleukin 2 (IL-2), IL-3, IL-4, or IL-6), hormones (e.g., insulin), growth factors (e.g., EGF, TGF-alpha, FGF, and VEGF), colony-stimulating factors (e.g., G-CSF, M-CSF, and GM-CSF (see, e.g., Burgess, Immunology Today, 5: 155-158 (1984)), somatostatin, and transferrin (see, e.g., O'Keefe et al, J Biol.
- interferons e.g., alpha, beta, or gamma interferon
- GM-CSF 3 which binds to myeloid cells, can be used as a cell-binding agent to target acute myelogenous leukemia cells.
- IL-2 which binds to activated T-cells, can be used for prevention of transplant graft rejection, for therapy and prevention of graft- versus-host disease, and for treatment of acute T-cell leukemia.
- Epidermal growth factor (EGF) can be used to target squamous cancers such as lung cancer and head and neck cancer.
- Somatostatin can be used to target neuroblastoma cells and other tumor cell types.
- the conjugate can comprise any suitable drug, typically a cytotoxic agent.
- Suitable cytotoxic agents include, for example, maytansinoids and maytansinoid analogs, taxoids, CC- 1065 and CC-1065 analogs, and dolastatin and dolastatin analogs.
- the cytotoxic agent is a maytansinoid, including maytansinol and maytansinol analogs. Maytansinoids are compounds that inhibit microtubule formation and are highly toxic to mammalian cells.
- Suitable maytansinol analogues include those having a modified aromatic ring and those having modifications at other positions. Such maytansinoids are described in, for example, U.S. Patents 4,256,746, 4,294,757, 4,307,016, 4,313,946, 4,315,929, 4,322,348, 4,331,598, 4,361,650, 4,362,663, 4,364,866, 4,424,219, 4,371,533, 4,450,254, 5,475,092, 5,585,499, 5,846,545, and 6,333,410. [0034] Examples of maytansinol analogs having a modified aromatic ring include:
- Patent 4,424,219) (prepared by the reaction of maytansinol with H 2 S or P 2 S 5 ), (2) C-14-alkoxymethyl (demethoxy/CH 2 OR) (U.S. Patent 4,331,598), (3) C-14-hydroxymethyl or acyloxymethyl (CH 2 OH or CH 2 OAc) (U.S. Patent 4,450,254) (prepared from Nocardia), (4) C-15-hydroxy/acyloxy (U.S. Patent 4,364,866) (prepared by the conversion of maytansinol by Streptomyces), (5) C-15-methoxy (U.S.
- Patents 4,313,946 and 4,315,929) isolated from Trewia nudiflora
- (6) C-18-N-demethyl U.S. Patents 4,362,663 and 4,322,348
- 4,5-deoxy U.S. Patent 4,371,533
- the conjugate utilizes the thiol- containing maytansinoid DMl, also known as N 2 -deacetyl-N 2 -(3-mercapto-l-oxopropyl)- maytansine, as the cytotoxic agent.
- DMl also known as N 2 -deacetyl-N 2 -(3-mercapto-l-oxopropyl)- maytansine
- the conjugate utilizes the thiol- containing maytansinoid DM4, also known as N -deacetyl-N ⁇ (4-methyl-4-mercapto-l- oxopentyl)-maytansine, as the cytotoxic agent.
- DM4 is represented by formula (II):
- maytansines may be used in the context of the invention, including, for example, thiol and disulfide-containing maytansinoids bearing a mono or di-alkyl substitution on the carbon atom bearing the sulfur atom.
- Particularly preferred is a maytansinoid having at the C-3 position (a) C- 14 hydroxymethyl, C- 15 hydroxy, or C-20 desmethyl functionality, and (b) an acylated amino acid side chain with an acyl group bearing a hindered sulfhydryl group, wherein the carbon atom of the acyl group bearing the thiol functionality has one or two substituents, said substituents being CH 3 , C 2 H 5 , linear or branched alkyl or alkenyl having from 1 to 10 carbon atoms, cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl, or heterocyclic aromatic or heterocycloalkyl radical, and further wherein one of
- R 1 and R 2 are each independently CH 3 , C 2 H 5 , linear alkyl or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl or heterocyclic aromatic or heterocycloalkyl radical, and wherein
- R 2 also can be H, wherein A, B, D are cycloalkyl or cycloalkenyl having 3-10 carbon atoms, simple or substituted aryl, or heterocyclic aromatic, or heterocycloalkyl radical, wherein R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 11 , and R 12 are each independently H, CH 3 , C 2 H 5 , linear alkyl or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl or heterocyclic aromatic, or heterocycloalkyl radical, wherein 1, m, n, o, p, q, r, s, and t are each independently zero or an integer from 1 to 5, provided that at least two of 1, m, n, o, p, q, r, s and t are not zero at
- Preferred embodiments of formula (III) include compounds of formula (III) wherein (a) R 1 is H, R 2 is methyl and Z is H, (b) R 1 and R 2 are methyl and Z is H, (c) R 1 is H 5
- Such additional maytansines also include compounds represented by formula (IV- L) 5 (IV-D), or (IV-D 3 L):
- Y represents (CR 7 R 8 )i(CR 5 R 6 ) m (CR 3 R 4 ) n CR 1 R 2 SZ, wherein R 1 and R 2 are each independently CH 3 , C 2 H 5 , linear alkyl, or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl, or heterocyclic aromatic or heterocycloalkyl radical, and wherein
- R 2 also can be H 3 wherein R 3 , R 4 , R 5 , R 6 , R 7 , and R 8 are each independently H, CH 3 , C 2 H 5 , linear alkyl or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl, or heterocyclic aromatic or heterocycloalkyl radical, wherein 1, m, and n are each independently an integer of from 1 to 5, and in addition n can be zero, wherein Z is H, SR, or COR wherein R is linear or branched alkyl or alkenyl having from 1 to 10 carbon atoms, cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, or simple or substituted aryl or heterocyclic aromatic or heterocycloalkyl radical, and wherein May represents a maytansinoid which bears the side chain at C-3, C- 14
- Preferred embodiments of formulas (IV-L), (IV-D) and (IV-D 5 L) include compounds of formulas (IV-L), (IV-D) and (IV-D 5 L) wherein (a) R 1 is H 5 R 2 is methyl, R 5 ,
- R 6 , R 7 , and R 8 are each H, 1 and m are each 1 , n is O 5 and Z is H, (b) R 1 and R 2 are methyl,
- R 5 , R 6 , R 7 , Rg are each H, 1 and m are I 5 n is O 5 and Z is H 5 (c) R 1 is H, R 2 is methyl, R 5 , R 6 ,
- R 7 , and R 8 are each H, 1 and m are each 1, n is 0, and Z is -SCH 3 , or (d) Ri and R 2 are methyl, R 5 , R 6 , R 7 , R 8 are each H 5 1 and m are I 5 n is O 5 and Z is -SCH 3 .
- cytotoxic agent is represented by formula (IV-L).
- Additional preferred maytansines also include compounds represented by formula (V):
- Y represents (CR 7 R 8 )i(CR 5 R 6 ) m (CR 3 R 4 ) n CR 1 R 2 SZ, wherein R 1 and R 2 are each independently CH 3 , C 2 H 5 , linear alkyl, or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl or heterocyclic aromatic or heterocycloalkyl radical, and wherein
- R 2 also can be H, wherein R 3 , R 4 , R 5 , R 6 , R 7 , and R 8 are each independently H, CH 3 , C 2 H 5 , linear alkyl or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl, or heterocyclic aromatic or heterocycloalkyl radical, wherein 1, m, and n are each independently an integer of from 1 to 5, and in addition n can be zero, and wherein Z is H, SR or COR, wherein R is linear alkyl or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, or simple or substituted aryl or heterocyclic aromatic or heterocycloalkyl radical.
- Preferred embodiments of formula (V) include compounds of formula (V) wherein (a) R 1 is H, R 2 is methyl, R 5 , R 6 , R 7 , and R 8 are each H; 1 and m are each 1; n is 0; and Z is H, (b) R 1 and R 2 are methyl; R 5 , R 6 , R 7 , R 8 are each H, 1 and m are 1; n is 0; and Z is H, (c) R 1 is H, R 2 is methyl, R 5 , R 6 , R 7 , and R 8 are each H 5 1 and m are each 1, n is 0, and Z is -SCH 3 , or (d) R 1 and R 2 are methyl, R 5 , R 6 , R 7 , R 8 are each H, 1 and m are 1, n is 0, and Z is -SCH 3 .
- Still further preferred maytansines include compounds represented by formula (VI-L), (VI-D), or (VI-D 3 L):
- VI-L VI-D
- VI-D VI-D, L
- Y 2 represents (CR 7 R 8 ) 1 (CR 5 R 6 ) m (CR 3 R 4 ) n CR 1 R 2 SZ 2 , wherein R 1 and R 2 are each independently CH 3 , C 2 H 5 , linear alkyl or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl or heterocyclic aromatic or heterocycloalkyl radical, and wherein
- R 2 also can be H, wherein R 3 , R 4 , R 5 , R 6 , R 7 , and R 8 are each independently H, CH 3 , C 2 H 5 , linear cyclic alkyl or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from
- n 3 to 10 carbon atoms, phenyl, substituted phenyl or heterocyclic aromatic or heterocycloalkyl radical, wherein 1, m, and n are each independently an integer of from 1 to 5, and in addition n can be zero, wherein Z 2 is SR or COR, wherein R is linear alkyl or alkenyl having from 1 to 10 carbon atoms, branched or cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, or simple or substituted aryl or heterocyclic aromatic or heterocycloalkyl radical, and wherein May is a maytansinoid.
- Additional preferred maytansines include compounds represented by formula
- R 1 and R 2 are each independently CH 3 , C 2 H 5 , linear branched or alkyl or alkenyl having from 1 to 10 carbon atoms, cyclic alkyl or alkenyl having from 3 to 10 carbon atoms, phenyl, substituted phenyl or heterocyclic aromatic or heterocycloalkyl radical, and in addition R 2 can be H, wherein A, B, and D each independently is cycloalkyl or cycloalkenyl having 3 to 10 carbon atoms, simple or substituted aryl, or heterocyclic aromatic or heterocycloalkyl radical, wherein R 3 , R 4 , R 5 , R 6 , R 7 , Rg, R
- Preferred embodiments of formula (VII) include compounds of formula (VII), wherein R 1 is H and R 2 is methyl.
- the cytotoxic agent used in the conjugate can be a taxane or derivative thereof.
- Taxanes are a family of compounds that includes paclitaxel (Taxol®), a cytotoxic natural product, and docetaxel (Taxotere®), a semi-synthetic derivative, which are both widely used in the treatment of cancer. Taxanes are mitotic spindle poisons that inhibit the depolymerization of tubulin, resulting in cell death.
- docetaxel and paclitaxel are useful agents in the treatment of cancer, their antitumor activity is limited because of their non-specific toxicity towards normal cells. Further, compounds like paclitaxel and docetaxel themselves are not sufficiently potent to be used in conjugates of cell-binding agents.
- a preferred taxane for use in the preparation of a cytotoxic conjugate is the taxane of formula (VIII):
- the cytotoxic also can be CC- 1065 or a derivative thereof.
- CC-1065 is a potent anti-tumor antibiotic isolated from the culture broth of Streptomyces zelensis.
- CC- 1065 is about 1000-fold more potent in vitro than commonly used anti-cancer drugs, such as doxorubicin, methotrexate, and vincristine (Bhuyan et al. 5 Cancer Res., 42: 3532-3537 (1982)).
- CC-1065 and its analogs are disclosed in U.S. Patents 5,585,499, 5,846,545, 6,340,701, and 6,372,738.
- the cytotoxic potency of CC-1065 has been correlated with its alkylating activity and its DNA-binding or DNA-intercalating activity. These two activities reside in separate parts of the molecule.
- the alkylating activity is contained in the cyclopropapyrroloindole (CPI) subunit and the DNA-binding activity resides in the two pyrroloindole subunits of CC-1065.
- CPI cyclopropapyrroloindole
- CC- 1065 analogs are known in the art and also can be used as the cytotoxic agent in the conjugate (see, e.g., Warpehoski et al., J Med. Chem., 31: 590-603 (1988)).
- a series of CC-1065 analogs has been developed in which the CPI moiety is replaced by a cyclopropabenzindole (CBI) moiety (Boger et al., J Org. Chem., 55: 5823- 5833 (1990), and Boger et al., Bioorg. Med. Chem. Lett, 1: 115-120 (1991)).
- CBI cyclopropabenzindole
- These CC- 1065 analogs maintain the high in vitro potency of the parental drug, without causing delayed toxicity in mice.
- these compounds are alkylating agents that covalently bind to the minor groove of DNA to cause cell death.
- CC-1065 analogs can be greatly improved by changing the in vivo distribution through targeted delivery to a tumor site, resulting in lower toxicity to non-targeted tissues, and thus, lower systemic toxicity.
- conjugates of analogs and derivatives of CC-1065 with cell-binding agents that specifically target tumor cells have been generated (see, e.g., U.S. Patents 5,475,092, 5,585,499, and 5,846,545). These conjugates typically display high target-specific cytotoxicity in vitro, and anti-tumor activity in human tumor xenograft models in mice (see, e.g., Chari et al., Cancer Res., 55: 4079-4084 (1995)).
- Drugs such as methotrexate, daunorubicin, doxorubicin, vincristine, vinblastine, melphalan, mitomycin C, chlorambucil, calicheamicin, tubulysin and tubulysin analogs, duocarmycin and duocarmycin analogs, dolastatin and dolastatin analogs also can be used in the context of the invention.
- Doxarubicin and daunorubicin compounds can also be used as the drug.
- the drug conjugates may be prepared by in vitro methods.
- a linking group is used.
- Suitable linking groups are well known in the art and include disulfide groups, acid labile groups, photolabile groups, peptidase labile groups, and esterase labile groups.
- Preferred linking groups are disulfide groups.
- conjugates can be constructed using a disulfide exchange reaction between the antibody and the drug or prodrug.
- the drug molecules also can be linked to a cell-binding agent through an intermediary carrier molecule such as serum albumin.
- the cell-binding agent is modified by reacting a bifunctional crosslinking reagent with the cell-binding agent, thereby resulting in the covalent attachment of a linker molecule to the cell-binding agent.
- a "bifunctional crosslinking reagent” is any chemical moiety that covalently links a cell-binding agent to a drug, such as the drugs described herein.
- a portion of the linking moiety is provided by the drug.
- the drug comprises a linking moiety that is part of a larger linker molecule that is used to join the cell-binding agent to the drug.
- the side chain at the C-3 hydroxyl group of maytansine is modified to have a free sulfhydryl group (SH).
- This thiolated form of maytansine can react with a modified cell-binding agent to form a conjugate. Therefore, the final linker is assembled from two components, one of which is provided by the crosslinking reagent, while the other is provided by the side chain from DMl.
- any suitable bifunctional crosslinking reagent can be used in connection with the invention, so long as the linker reagent provides for retention of the therapeutic, e.g., cytotoxicity, and targeting characteristics of the drug and the cell-binding agent, respectively.
- the linker molecule joins the drug to the cell-binding agent through chemical bonds (as described above), such that the drug and the cell-binding agent are chemically coupled (e.g., covalently bonded) to each other.
- the linking reagent is a cleavable linker. More preferably, the linker is cleaved under mild conditions, i.e., conditions within a cell under which the activity of the drug is not affected.
- cleavable linkers examples include disulfide linkers, acid labile linkers, photolabile linkers, peptidase labile linkers, and esterase labile linkers.
- Disulfide containing linkers are linkers cleavable through disulfide exchange, which can occur under physiological conditions.
- Acid labile linkers are linkers cleavable at acid pH. For example, certain intracellular compartments, such as endosomes and lysosomes, have an acidic pH (pH 4-5), and provide conditions suitable to cleave acid labile linkers.
- Photo labile linkers are useful at the body surface and in many body cavities that are accessible to light. Furthermore, infrared light can penetrate tissue.
- Peptidase labile linkers can be used to cleave certain peptides inside or outside cells (see e.g., Trouet et al, Proc. Natl. Acad. Sci. USA, 79: 626-629 (1982), and Umemoto et al., Int. J. Cancer, 43: 677-684 (1989)).
- the drug is linked to a cell-binding agent through a disulfide bond.
- the linker molecule comprises a reactive chemical group that can react with the cell-binding agent.
- Preferred reactive chemical groups for reaction with the cell-binding agent are N- succinimidyl esters and JV-sulfosuccinimidyl esters.
- linker molecule comprises a reactive chemical group, preferably a dithiopyridyl group, that can react with the drug to form a disulfide bond.
- linker molecules include, for example, N-succinimidyl 3-(2-pyridyldithio)propionate (SPDP) (see, e.g., Carlsson et al., Biochem. J, 173: 723-737 (1978)), N-succinimidyl 4-(2-pyridyldithio)butanoate (SPDB) (see, e.g., U.S.
- SPDP N-succinimidyl 3-(2-pyridyldithio)propionate
- SPDB N-succinimidyl 4-(2-pyridyldithio)butanoate
- Patent 4,563,304 N-succinimidyl 4-(2-pyridyldithio)pentanoate (SPP) (see, e.g., CAS Registry number 341498-08-6), and other reactive cross-linkers which are described in U.S. Patent 6,913,748, which is incorporated herein in its entirety by reference.
- SPP N-succinimidyl 4-(2-pyridyldithio)pentanoate
- other reactive cross-linkers which are described in U.S. Patent 6,913,748, which is incorporated herein in its entirety by reference.
- cleavable linkers preferably are used in the inventive method, a non- cleavable linker also can be used to generate the above-described conjugate.
- a non-cleavable linker is any chemical moiety that is capable of linking a drug, such as a maytansinoid, a taxane, or a CC- 1065 analog, to a cell-binding agent in a stable, covalent manner.
- non- cleavable linkers are substantially resistant to acid-induced cleavage, light-induced cleavage, peptidase-induced cleavage, esterase-induced cleavage, and disulfide bond cleavage, at conditions under which the drug or the cell-binding agent remains active.
- Suitable crosslinking reagents that form non-cleavable linkers between a drug and the cell-binding agent are well known in the art.
- non-cleavable linkers include linkers having an N-succinimidyl ester or N-sulfosuccinimidyl ester moiety for reaction with the cell-binding agent, as well as a maleimido- or haloacetyl-based moiety for reaction with the drug.
- Crosslinking reagents comprising a maleimido-based moiety include N- succinimidyl 4-(maleimidomethyl)cyclohexanecarboxylate (SMCC), N-succinimidyl-4-(N- maleimidomethyl)-cyclohexane-l-carboxy-(6-amidocaproate), which is a "long chain" analog of SMCC (LC-SMCC), ⁇ -maleimidoundecanoic acid N-succinimidyl ester (KMUA), ⁇ -maleimidobutyric acid N-succinimidyl ester (GMBS), ⁇ -maleimidocaproic acid N- hydroxysuccinimide ester (EMCS), m-maleimidobenzoyl-N-hydroxysuccinimide ester (MBS), N-( ⁇ -maleimidoacetoxy)-succinimide ester (AMAS), succinimidyl-6-( ⁇ - maleimid
- Cross-linking reagents comprising a haloacetyl-based moiety include N-succinimidyl-4-(iodoacetyl)-aminobenzoate (SIAB) 5 N-succinimidyl iodoacetate (SIA), N-succinimidyl bromoacetate (SBA), and N- succinimidyl 3-(bromoacetamido)propionate (SBAP).
- SIAB N-succinimidyl-4-(iodoacetyl)-aminobenzoate
- SIA N-succinimidyl iodoacetate
- SBA N-succinimidyl bromoacetate
- SBAP N- succinimidyl 3-(bromoacetamido)propionate
- linkers can be derived from dicarboxylic acid based moieties.
- Suitable dicarboxylic acid based moieties include, but are not limited to, ⁇ , ⁇ - dicarboxylic acids of the general formula (IX):
- X is a linear or branched alkyl, alkenyl, or alkynyl group having 2 to 20 carbon atoms
- Y is a cycloalkyl or cycloalkenyl group bearing 3 to 10 carbon atoms
- Z is a substituted or unsubstituted aromatic group bearing 6 to 10 carbon atoms, or a substituted or unsubstituted heterocyclic group wherein the hetero atom is selected from N, O or S, and wherein 1, m, and n are each 0 or 1, provided that 1, m, and n are all not zero at the same time.
- Patent Application No. 10/960,602 which corresponds to U.S. Patent Application Publication
- the drug can be first modified to introduce a reactive ester suitable to react with a cell-binding agent. Reaction of these maytansinoids containing an activated linker moiety with a cell-binding agent provides another method of producing a cleavable or non-cleavable cell-binding agent maytansinoid conjugate.
- This example demonstrates the purification of an antibody modified with a heterobifunctional modification reagent using TFF.
- the huN901 monoclonal antibody (final concentration of 8 mg/ml) was incubated with N-succinimidyl 4-(2-pyridyldithio)pentanoate (SPP, 5.6-fold molar excess) for approximately 180 minutes at 20° C in 50 mM potassium phosphate buffer (pH 7.5) containing 50 mM NaCl, 2 mM EDTA, and 5% ethanol.
- SPP N-succinimidyl 4-(2-pyridyldithio)pentanoate
- the reaction mixture was purified using a Pellicon XL TFF system (Millipore, Billerica, MA), and the antibody was diafiltered (5 volumes) into 50 mM potassium phosphate, 50 mM NaCl (pH 6.5), and 2 mM EDTA using a 10,000 molecular weight cutoff membrane (UltracelTM regenerated cellulose membrane, Millipore, Billerica, MA). Both samples were conjugated with DMl (1.7 fold molar excess over the unbound linker) for 18 hours at pH 6.5 in potassium phosphate buffer containing 50 mM NaCl and a final concentration of 3% DMA.
- TFF drug conjugate product of at least equivalent quality to the nonadsorptive chromatography (G25) process while being more convenient and scaleable.
- This example demonstrates the purification of an antibody modified with a heterobifunctional modification reagent using adsorptive chromatography.
- the huB4 antibody was modified with N-succinimidyl 4-(2- pyridyldithio)butanoate (SPDB, 5.4 fold molar excess) for 120 minutes at room temperature in 50 mM potassium phosphate buffer (pH 6.5) containing 50 mM NaCl, 2 mM EDTA, and 5% ethanol.
- SPDB N-succinimidyl 4-(2- pyridyldithio)butanoate
- the reaction mixture was purified using the SephadexTM G25F resin as described in Example 1.
- reaction mixture was loaded onto a column of ceramic hydroxyapatite (CHT, Bio-Rad Laboratories, Hercules, CA), which was equilibrated in 12.5 mM potassium phosphate buffer (pH 6.5) and eluted with 80 mM potassium phosphate buffer (pH 6.5).
- CHT ceramic hydroxyapatite
- the CNTO95 antibody (final concentration of 10 mg/ml) was modified with N- succinimidyl 4-(2-pyridyldithio)butanoate (SPDB, 4.5 fold molar excess) for 120 minutes at 20° C in 10 mM sodium phosphate buffer (pH 7.5) containing 2.7 % sucrose and 5% ethanol.
- SPDB N- succinimidyl 4-(2-pyridyldithio)butanoate
- the reaction mixture was purified using SephadexTM G25F resin in 12.5 mM potassium phosphate buffer (pH 6.6) containing 12.5 mM NaCl and 0.5 mM EDTA.
- This example demonstrates the beneficial effects of conjugating a modified antibody with a drug at a pH of above 6.5.
- Example 2 The modified antibody was then divided into two groups. In the first group, conjugation was performed in 12.5 mM potassium phosphate at pH 6.5 containing 12.5 mM
- the conjugation reaction was at pH 7.5.
- the conjugated antibody was purified over NAP-IO columns.
- huB4 humanized monoclonal antibody was modified with either (a) a 4.9-fold molar excess of SPDB relative to antibody, or (b) a 4.8-fold molar excess of SPDB relative to antibody.
- reaction was in 50 mM potassium phosphate, 50 mM potassium chloride, and 2 mM EDTA (pH 6.5) in 5% ethanol for a total of 120 minutes at room temperature.
- Sample (a) was purified over a column of SephadexTM G25F resin equilibrated in 50 mM potassium phosphate, 50 mM sodium chloride, and 2 mM EDTA at pH 6.5.
- Sample (b) was purified equivalently except that the chromatography buffer was adjusted to pH 7.5. Both samples were conjugated with DM4 (1.7 fold molar excess over bound linker) for 18 hours at room temperature in a final concentration of dimethylacetamide (DMA) of 3%.
- DMA dimethylacetamide
- sample (a) was conjugated at pH 6.5
- sample (b) was conjugated at pH 7.5.
- the samples were then purified over a column of SephadexTM G25F resin equilibrated in 9.6 mM potassium phosphate and 4.2 mM sodium chloride at pH 6.5. Both samples were incubated at 4° C for up to 7 months and subjected to analysis of released free drug at intervals. The resulting data are set forth in Table 3.
- release of free drug is substantially slower from sample (b) that had been conjugated at pH 7.5 relative to sample (a) that had been conjugated at pH 6.5. Accordingly, drug conjugate product prepared at pH 7.5 is shown to be more stable with respect to release of free drug over time as compared to the drug conjugate product prepared at pH 6.5. The conjugation at pH 7.5 also shows a better drug incorporation than at pH 6.5, thereby requiring less drug to be used.
- This example demonstrates the beneficial effects of conjugating a modified antibody with a drug at a pH of below 6.0.
- the huN901 monoclonal antibody (final concentration of 8 mg/ml) was incubated with N-succinimidyl 4-(2-pyridyldithio)pentanoate (SPP, 5.6-fold molar excess) for approximately 180 minutes at 20° C in 50 mM potassium phosphate buffer (pH 7.5) containing 50 mM NaCl, 2 mM EDTA, and 5% ethanol.
- SPP N-succinimidyl 4-(2-pyridyldithio)pentanoate
- the reaction mixture was purified using a column of SephadexTM G25F resin equilibrated and eluted in 50 mM sodium citrate buffer (pH 5.0) containing 50 mM NaCl and 2 mM EDTA.
- reaction mixture was purified using a column of SephadexTM G25F resin equilibrated and eluted in 50 mM potassium phosphate buffer (pH 6.5) containing 50 mM NaCl and 2 mM
- linker/antibody ratios were determined by treatment with dithiothreitol to release pyridine-2-thione, which has an extinction coefficient of 8,080
- the first group had a 4.3 linker/antibody ratio.
- the second group had a 4.2 linker/antibody ratio.
- conjugate that is made by conjugating the modified antibody with the drug at a pH of 5.0 reaches a higher and more stable level of bound drug during the course of the conjugation reaction than conjugate made at a conjugation pH of 6.5.
- the results indicate that a higher drug/antibody level is achieved upon conjugation at pH 5.0 than, when using the same amount of drug at a conjugation pH of 6.5, thereby indicating more efficient usage of drug at pH 5.0.
- the conjugate monomer amounts were determined over time. The resulting data are set forth in Table 5.
- conjugate that is made by conjugating the modified antibody with the drug at a pH of 5.0 has a higher level of conjugate monomer than conjugate made at a conjugation pH of 6.5.
- This example further demonstrates benefits of conjugating a drug to a modified antibody at a pH of less than 6.
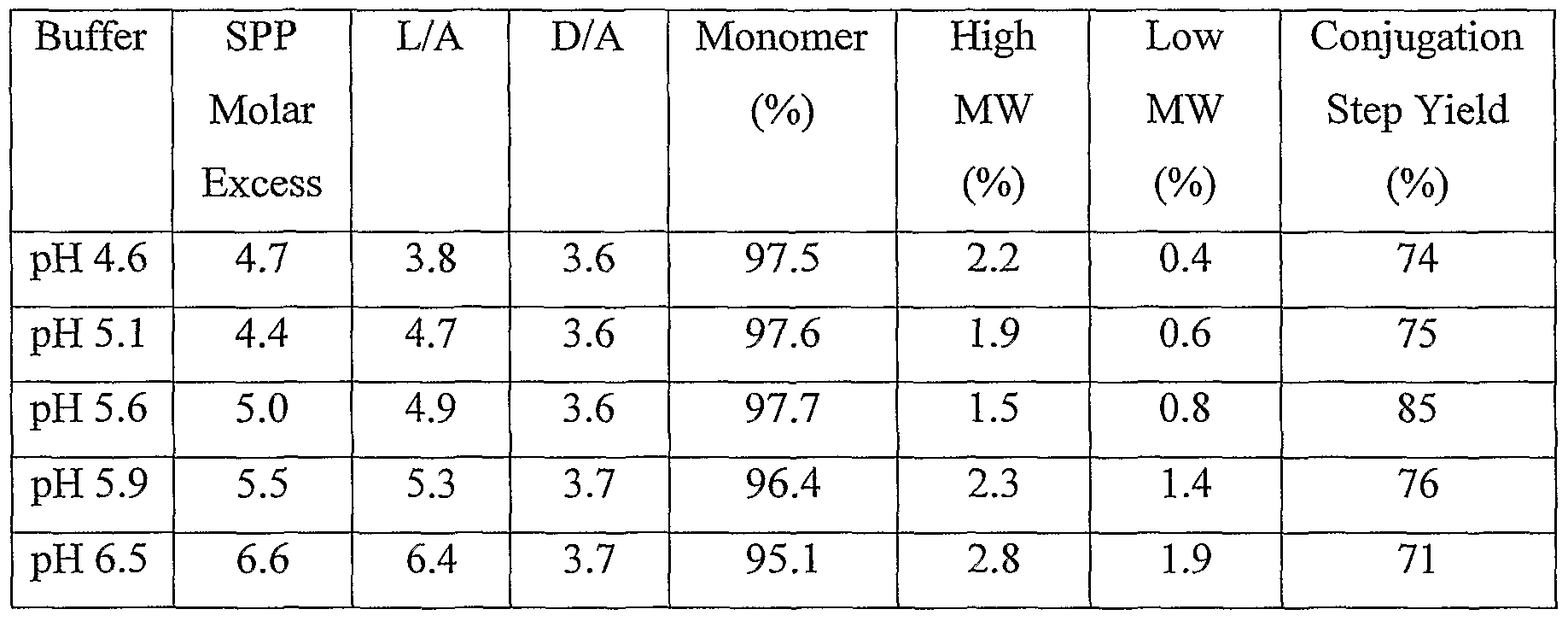
- BIWA 4 antibody was modified with SPP (molar excess of SPP as shown in Table 6) for 120-140 minutes at room temperature in 50 mM potassium phosphate buffer (pH 6.5), 50 mM NaCl, 2 mM EDTA, and 5% ethanol. Aliquots of modified antibody were purified on separate NAP 25 columns equilibrated in buffers having various pH values (pH 4.6 — 6.5). The pH 4.6 - 5.9 buffers were composed of 35 mM sodium citrate, 150 mM sodium chloride, and 2 mM EDTA. The pH 6.5 buffer was PBS with 2 mM EDTA.
- Modified antibody at each pH was conjugated with DMl (1.7 fold molar excess over linker) in dimethylacetamide (DMA, final concentration of 3%). After incubation for 17-18 hours at room temperature, the conjugated antibody samples were purified by chromatography on NAP 25 columns equilibrated in PBS (pH 6.5). Linker/antibody ratios (L/A in Table 6) were determined by treatment with dithiothreitol to release pyridine-2- thione, which has an extinction coefficient of 8,080 M -1 Cm "1 at 343 nM. Drug/antibody ratios were determined spectrophotometrically (wavelengths of 280 nm and 252 nm) for the conjugation step.
- DMA dimethylacetamide
- Conjugate monomer, high molecular weight species, and low molecular weight species were determined by SEC-HPLC using a TSKG3000SWXL column equilibrated and developed in 0.2 M potassium phosphate buffer (pH 7.0) containing 0.2 M potassium chloride and 20% isopropanol. [0098] The results of this analysis are set forth in Table 6.
- the humanized monoclonal antibody CNTO95 was modified at a concentration of 20 mg/mL with the bifunctional modifying reagent SPDB at a molar excess of SPDB over antibody of 4.6 for 120 min at 20° C.
- the modification buffer was 44 mM phosphate buffer (pH 7.5) containing 5.3% sucrose and 5% ethanol.
- One aliquot of the modified antibody was purified over SephadexTM G25F resin (standard four-step process), equilibrated and eluted in 12.5 mM potassium phosphate buffer (pH 7.5) containing 12.5 mM NaCl, and was subsequently conjugated with DM4 (1.7 fold molar excess of drug over bound linker) at a final modified antibody concentration of 10 mg/mL in 12.5 mM potassium phosphate buffer (pH 7.5) containing 12.5 mM NaCl and 10% DMA for 20 hours at room temperature.
- a second aliquot of the modified antibody was conjugated immediately at the end of the 120 minute modification reaction (three-step process), without being further purified.
- Linker/antibody ratios were determined by treatment with dithiothreitol to release pyridine-2-thione, which has an extinction coefficient of 8,080 M -1 Cm "1 at 343 nM.
- Drug/antibody (D/ A) ratios and yield were determined spectrophotometrically (wavelengths of 280 nm and 252 nm) for the conjugation step. Percentages of monomer were assayed by SEC-HPLC. Percentages of free drug were assayed by HPLC on a Hisep column. The results of these analyses are set forth in Table 7.
- This example demonstrates an improved means of purifying antibody that has been modified with a heterobifunctional modification reagent and then conjugated with a maytansinoid.
- a second conjugate sample was purified by a Pellicon XL TFF system (Millipore,
- a third conjugate sample was purified using a column of MEP Hypercell resin equilibrated in 50 mM Tris (pH 8.0), and eluted with 50 mM sodium acetate (pH 4.0).
- a fourth conjugate sample was purified using a column of UNOsphere S resin equilibrated in 50 mM sodium phosphate (pH 6.5) and eluted with 0.2 M NaCl and 50 mM sodium phosphate (pH 6.5).
- a fifth conjugate sample was purified using a column of CHT resin (Bio-Rad
- a sixth conjugate sample was purified using a column of SP Sepharose resin equilibrated in 35 mM sodium citrate, 10 mM sodium chloride (pH 5.0), and eluted with 0.25
- Conjugate monomer was determined by SEC-HPLC using a column of
- the conjugation step yield was determined by dividing the yield of conjugated antibody by the amount of modified antibody that was conjugated (determined spectrophotometrically at a wavelength of 280 nm).
- CHT ceramic hydroxyapatite
- CFT ceramic fluoroapatite
- both the CHT and CFT resins may be used in non-adsorptive mode so that the desired product (substantially monomeric conjugate) is not retained by the resins, whereas high molecular weight species are retained and thereby separated from the desired product.
- a standard buffer/solvent composition for conjugation comprises 3% DMA, 50 mM potassium phosphate, 50 mM NaCl, and 2 mM EDTA at pH 6.5 (as utilized in Example 1)
- other compositions are more compatible with some of the chromatographic steps described herein and provide other benefits relative to the standard process.
- conjugation may be performed in 3% DMA, 12.5 mM potassium phosphate, 12.5 mM NaCl, and 0.5 mM EDTA at pH 6.5.
- the amount of DM4 incorporated relative to the amount of linker incorporated in huB4 antibody was about 10% higher than for the standard conditions.
- these conditions are more compatible with loading onto resins such as cation exchange and CHT resins.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Immunology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Cell Biology (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Peptides Or Proteins (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Cephalosporin Compounds (AREA)
Abstract
Description
Claims
Priority Applications (27)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2006283726A AU2006283726C1 (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
| JP2008527970A JP5350792B2 (en) | 2005-08-24 | 2006-08-14 | Method for preparing maytansinoid antibody complex |
| MX2008002597A MX2008002597A (en) | 2005-08-24 | 2006-08-14 | Process for preparing purified drug conjugates. |
| CN2006800342426A CN101267841B (en) | 2005-08-24 | 2006-08-14 | Method for preparing maytansinoid antibody conjugates |
| IL305084A IL305084A (en) | 2005-08-24 | 2006-08-14 | Process for preparing purified drug conjugates |
| KR1020087004084A KR101301011B1 (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
| NZ595430A NZ595430A (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
| ES06801436T ES2390826T3 (en) | 2005-08-24 | 2006-08-14 | Procedure for preparing purified drug conjugates |
| PL06801436T PL1928503T3 (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
| BRPI0615049-7A BRPI0615049B1 (en) | 2005-08-24 | 2006-08-14 | PROCESS FOR THE PREPARATION OF AN ANTIBODY- MAITANSINOID CONJUGATE |
| HRP20120794TT HRP20120794T1 (en) | 2005-08-24 | 2006-08-14 | PROCEDURE FOR THE PREPARATION OF Purified Drug Conjugates |
| NZ565964A NZ565964A (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates by purifying with tangential flow filtration |
| EP06801436A EP1928503B1 (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
| EP19156303.0A EP3539572A1 (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
| SI200631435T SI1928503T1 (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
| EA200800657A EA013327B1 (en) | 2005-08-24 | 2006-08-14 | METHOD FOR PREPARING PURIFIED CONJUGATES OF MEDICINES |
| IL282138A IL282138B2 (en) | 2005-08-24 | 2006-08-14 | A process for preparing purified drug compounds |
| CA2620343A CA2620343C (en) | 2005-08-24 | 2006-08-14 | Process for preparing antibody maytansinoid conjugates |
| HK08112150.3A HK1120407B (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
| RS20120425A RS52470B (en) | 2005-08-24 | 2006-08-14 | PROCEDURE FOR PREPARATION OF PURE CONJUGATED MEDICINES |
| DK06801436.4T DK1928503T3 (en) | 2005-08-24 | 2006-08-14 | Process for Preparation of Purified Drug Conjugates |
| IL189461A IL189461A (en) | 2005-08-24 | 2008-02-12 | Process for preparing cell-binding agent-cytotoxic agent conjugates |
| ZA2008/01564A ZA200801564B (en) | 2005-08-24 | 2008-02-15 | Process for preparaing maytansinoid antibody conjugates |
| IL220816A IL220816A (en) | 2005-08-24 | 2012-07-08 | Process for preparing cell-binding agent-cytotoxic agent conjugates |
| IL226985A IL226985A (en) | 2005-08-24 | 2013-06-17 | Process for preparing purified drug conjugates |
| CR20150350A CR20150350A (en) | 2005-08-24 | 2015-07-03 | PROCESS TO PREPARE ANTIBODY AND MAYTANSINOID CONJUGATES |
| IL248076A IL248076B (en) | 2005-08-24 | 2016-09-27 | Process for preparing purified drug conjugates |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US71085805P | 2005-08-24 | 2005-08-24 | |
| US60/710,858 | 2005-08-24 | ||
| US79771306P | 2006-05-04 | 2006-05-04 | |
| US60/797,713 | 2006-05-04 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP11004940.0A Previously-Filed-Application EP2399609B1 (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2007024536A2 true WO2007024536A2 (en) | 2007-03-01 |
| WO2007024536A3 WO2007024536A3 (en) | 2007-07-05 |
Family
ID=37666423
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2006/031653 Ceased WO2007024536A2 (en) | 2005-08-24 | 2006-08-14 | Process for preparing maytansinoid antibody conjugates |
Country Status (24)
| Country | Link |
|---|---|
| US (7) | US7811572B2 (en) |
| EP (4) | EP3539572A1 (en) |
| JP (3) | JP5350792B2 (en) |
| KR (1) | KR101301011B1 (en) |
| CN (2) | CN101267841B (en) |
| AU (1) | AU2006283726C1 (en) |
| BR (1) | BRPI0615049B1 (en) |
| CA (6) | CA2893252C (en) |
| CR (2) | CR9742A (en) |
| CY (1) | CY1113206T1 (en) |
| DK (1) | DK1928503T3 (en) |
| EA (1) | EA013327B1 (en) |
| EC (1) | ECSP088212A (en) |
| ES (2) | ES2390826T3 (en) |
| HR (1) | HRP20120794T1 (en) |
| IL (6) | IL305084A (en) |
| MX (1) | MX2008002597A (en) |
| NZ (4) | NZ716641A (en) |
| PL (1) | PL1928503T3 (en) |
| PT (1) | PT1928503E (en) |
| RS (1) | RS52470B (en) |
| SI (1) | SI1928503T1 (en) |
| WO (1) | WO2007024536A2 (en) |
| ZA (1) | ZA200801564B (en) |
Cited By (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010126979A1 (en) | 2009-04-29 | 2010-11-04 | Bio-Rad Laboratories, Inc. | Purification of immunoconjugates |
| KR20120088549A (en) * | 2009-06-03 | 2012-08-08 | 이뮤노젠 아이엔씨 | Splicing method |
| WO2012143499A2 (en) | 2011-04-21 | 2012-10-26 | Bayer Intellectual Property Gmbh | Novel binder-drug conjugates (adcs) and their use |
| WO2012135517A3 (en) * | 2011-03-29 | 2013-02-21 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| WO2014055842A1 (en) * | 2012-10-04 | 2014-04-10 | Immunogen, Inc. | Process for preparing stable antibody maytansinoid conjugates |
| US8795673B2 (en) | 2011-03-29 | 2014-08-05 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| US8933205B2 (en) | 2005-08-24 | 2015-01-13 | Immunogen, Inc. | Process for preparing purified drug conjugates |
| WO2015009740A2 (en) | 2013-07-15 | 2015-01-22 | Cell Signaling Technology, Inc. | Anti-mucin 1 binding agents and uses thereof |
| CN104524592A (en) * | 2008-04-30 | 2015-04-22 | 伊缪诺金公司 | Cross-linkers and their uses |
| EP2903450A4 (en) * | 2012-10-04 | 2016-05-11 | Immunogen Inc | Use of an ion exchange membrane to remove impurities from cell-binding agent cytotoxic agent conjugates |
| WO2016207089A1 (en) | 2015-06-22 | 2016-12-29 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (adcs) and antibody prodrug conjugates (apdcs) with enzymatically cleavable groups |
| WO2017060322A2 (en) | 2015-10-10 | 2017-04-13 | Bayer Pharma Aktiengesellschaft | Ptefb-inhibitor-adc |
| WO2017162663A1 (en) | 2016-03-24 | 2017-09-28 | Bayer Pharma Aktiengesellschaft | Prodrugs of cytotoxic active agents having enzymatically cleavable groups |
| WO2018112334A1 (en) | 2016-12-16 | 2018-06-21 | Bluefin Biomedicine, Inc. | Anti-cub domain-containing protein 1 (cdcp1) antibodies, antibody drug conjugates, and methods of use thereof |
| WO2018114798A1 (en) | 2016-12-21 | 2018-06-28 | Bayer Aktiengesellschaft | Prodrugs of cytotoxic active agents having enzymatically cleavable groups |
| WO2018114578A1 (en) | 2016-12-21 | 2018-06-28 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (adcs) having enzymatically cleavable groups |
| US10022453B2 (en) | 2013-12-23 | 2018-07-17 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (ADCs) with kinesin spindel protein (KSP) |
| US10035817B2 (en) | 2012-10-04 | 2018-07-31 | Immunogen, Inc. | Method of purifying cell-binding agent-cytotoxic agent conjugates with a PVDF membrane |
| US20180311375A1 (en) * | 2017-04-27 | 2018-11-01 | Cadila Healthcare Limited | Process of preparing antibody-drug conjugate |
| WO2019105835A1 (en) | 2017-11-29 | 2019-06-06 | Bayer Consumer Care Ag | Combinations of copanlisib and anetumab ravtansine |
| WO2020022363A1 (en) | 2018-07-25 | 2020-01-30 | 第一三共株式会社 | Effective method for manufacturing antibody-drug conjugate |
| WO2020084115A1 (en) | 2018-10-25 | 2020-04-30 | Pharma Mar, S.A. | Antibody drug conjugates comprising ecteinascidin derivatives |
| US10640508B2 (en) | 2017-10-13 | 2020-05-05 | Massachusetts Institute Of Technology | Diazene directed modular synthesis of compounds with quaternary carbon centers |
| WO2020234114A1 (en) | 2019-05-21 | 2020-11-26 | Bayer Aktiengesellschaft | A novel stable high concentration formulation for anetumab ravtansine |
| US10918627B2 (en) | 2016-05-11 | 2021-02-16 | Massachusetts Institute Of Technology | Convergent and enantioselective total synthesis of Communesin analogs |
| US10918735B2 (en) | 2012-12-04 | 2021-02-16 | Massachusetts Institute Of Technology | Substituted pyrazino[1′,2′:1,5]pyrrolo[2,3-b]indole-1,4-diones for cancer treatment |
| WO2021043951A1 (en) | 2019-09-05 | 2021-03-11 | Pharma Mar, S.A. | Drug antibody conjugates |
| US11001636B2 (en) | 2016-06-15 | 2021-05-11 | Bayer Pharma Aktiengesellschaft | Specific antibody-drug-conjugates (ADCs) with KSP inhibitors and anti-CD123-antibodies |
| WO2021214126A1 (en) | 2020-04-21 | 2021-10-28 | Pharma Mar, S.A. | Drug antibody conjugates |
| US11160871B2 (en) | 2015-10-28 | 2021-11-02 | Tarveda Therapeutics, Inc. | SSTR-targeted conjugates and particles and formulations thereof |
| US11274121B2 (en) | 2018-01-12 | 2022-03-15 | Immunogen, Inc. | Methods for antibody drug conjugation, purification, and formulation |
| US11433140B2 (en) | 2016-12-21 | 2022-09-06 | Bayer Pharma Aktiengesellschaft | Specific antibody drug conjugates (ADCs) having KSP inhibitors |
| US11535634B2 (en) | 2019-06-05 | 2022-12-27 | Massachusetts Institute Of Technology | Compounds, conjugates, and compositions of epipolythiodiketopiperazines and polythiodiketopiperazines and uses thereof |
| US11634508B2 (en) | 2019-07-10 | 2023-04-25 | Cybrexa 2, Inc. | Peptide conjugates of cytotoxins as therapeutics |
| US11932650B2 (en) | 2017-05-11 | 2024-03-19 | Massachusetts Institute Of Technology | Potent agelastatin derivatives as modulators for cancer invasion and metastasis |
| US12030888B2 (en) | 2021-02-24 | 2024-07-09 | Massachusetts Institute Of Technology | Himastatin derivatives, and processes of preparation thereof, and uses thereof |
| WO2025005240A1 (en) | 2023-06-30 | 2025-01-02 | 第一三共株式会社 | Antibody-drug conjugate production method including purification step using activated carbon material |
| US12234212B2 (en) | 2019-07-10 | 2025-02-25 | Cybrexa 3, Inc. | Peptide conjugates of microtubule-targeting agents as therapeutics |
Families Citing this family (67)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1392359B2 (en) | 2001-05-11 | 2013-03-13 | Ludwig Institute for Cancer Research Ltd. | Specific binding proteins and uses thereof |
| US20100056762A1 (en) | 2001-05-11 | 2010-03-04 | Old Lloyd J | Specific binding proteins and uses thereof |
| JP5149620B2 (en) | 2004-07-23 | 2013-02-20 | エンドサイト,インコーポレイテッド | Bivalent linker and conjugate thereof |
| US20110166319A1 (en) * | 2005-02-11 | 2011-07-07 | Immunogen, Inc. | Process for preparing purified drug conjugates |
| US9090693B2 (en) * | 2007-01-25 | 2015-07-28 | Dana-Farber Cancer Institute | Use of anti-EGFR antibodies in treatment of EGFR mutant mediated disease |
| CN104127878A (en) | 2007-03-14 | 2014-11-05 | 恩多塞特公司 | Binding ligand linked drug delivery conjugates of tubulysins |
| ES2542152T3 (en) | 2007-03-15 | 2015-07-31 | Ludwig Institute For Cancer Research Ltd. | Treatment method using EGFR antibodies and Src inhibitors and related formulations |
| US9877965B2 (en) | 2007-06-25 | 2018-01-30 | Endocyte, Inc. | Vitamin receptor drug delivery conjugates for treating inflammation |
| BRPI0812970A2 (en) | 2007-06-25 | 2019-09-24 | Endocyte Inc | conjugates containing hydrophilic spacers |
| CN108424454B (en) | 2007-08-14 | 2022-05-31 | 路德维格癌症研究所有限公司 | Monoclonal antibody 175 targeting EGF receptor, and derivatives and uses thereof |
| US20100092495A1 (en) * | 2008-04-30 | 2010-04-15 | Immunogen Inc. | Potent cell-binding agent drug conjugates |
| US20110076232A1 (en) * | 2009-09-29 | 2011-03-31 | Ludwig Institute For Cancer Research | Specific binding proteins and uses thereof |
| AR078471A1 (en) * | 2009-10-02 | 2011-11-09 | Sanofi Aventis | MAITANSINOID COMPOUNDS AND THE USE OF THESE TO PREPARE CONJUGATES WITH AN ANTIBODY WHICH ARE USED AS ANTI-BANKING AGENTS AND THE PREPARATION PROCEDURE OF THESE CONJUGATES |
| TW201129384A (en) | 2010-02-10 | 2011-09-01 | Immunogen Inc | CD20 antibodies and uses thereof |
| AR084020A1 (en) | 2010-11-30 | 2013-04-17 | Genentech Inc | ANTIBODIES FOR THE LOW AFFINITY HEMATOENCEPHALIC BARRIER RECEIVER AND ITS USES |
| JP6049642B2 (en) | 2011-02-15 | 2016-12-21 | イミュノジェン・インコーポレーテッド | Method for preparing complex |
| MX2013011201A (en) * | 2011-03-29 | 2013-12-16 | Immunogen Inc | Process for manufacturing conjugates of improved homogeneity. |
| CN103648532A (en) | 2011-06-21 | 2014-03-19 | 伊缪诺金公司 | Novel maytansinoid derivatives with peptide linker and conjugates thereof |
| US20140350228A1 (en) * | 2011-12-13 | 2014-11-27 | Immunogen, Inc. | Use of n-hydroxysuccinimide to improve conjugate stability |
| US10080805B2 (en) | 2012-02-24 | 2018-09-25 | Purdue Research Foundation | Cholecystokinin B receptor targeting for imaging and therapy |
| US20140080175A1 (en) | 2012-03-29 | 2014-03-20 | Endocyte, Inc. | Processes for preparing tubulysin derivatives and conjugates thereof |
| WO2013151649A1 (en) * | 2012-04-04 | 2013-10-10 | Sialix Inc | Glycan-interacting compounds |
| EP2848686B1 (en) * | 2012-04-04 | 2024-12-25 | Perseus Proteomics Inc. | Conjugate of anti-cdh3 (p-cadherin) antibody and drug |
| HK1211208A1 (en) | 2012-08-22 | 2016-05-20 | Immunogen, Inc. | Cytotoxic benzodiazepine derivative |
| WO2014058947A1 (en) | 2012-10-12 | 2014-04-17 | Sanofi | Compositions and methods for treating cancer using pi3k inhibitor and anti-cd19 maytansinoid immunoconjugate |
| EP2908818A4 (en) | 2012-10-16 | 2016-07-13 | Endocyte Inc | Drug delivery conjugates containing unnatural amino acids and methods for using |
| JP6494533B2 (en) | 2013-02-28 | 2019-04-03 | イミュノジェン・インコーポレーテッド | Complexes comprising maytansinoids as cell binding agents and cytotoxic agents |
| WO2014134486A2 (en) | 2013-02-28 | 2014-09-04 | Immunogen, Inc. | Conjugates comprising cell-binding agents and cytotoxic agents |
| US9498532B2 (en) | 2013-03-13 | 2016-11-22 | Novartis Ag | Antibody drug conjugates |
| NZ710929A (en) | 2013-03-15 | 2018-02-23 | Novartis Ag | Antibody drug conjugates |
| MX379256B (en) | 2013-03-15 | 2025-03-10 | Regeneron Pharma | BIOLOGICALLY ACTIVE MOLECULES, CONJUGATES THEREOF, AND THERAPEUTIC USES. |
| WO2014182970A1 (en) * | 2013-05-08 | 2014-11-13 | Zymeworks Inc. | Bispecific her2 and her3 antigen binding constructs |
| WO2014194030A2 (en) | 2013-05-31 | 2014-12-04 | Immunogen, Inc. | Conjugates comprising cell-binding agents and cytotoxic agents |
| MX2016003256A (en) | 2013-09-12 | 2016-06-07 | Halozyme Inc | Modified anti-epidermal growth factor receptor antibodies and methods of use thereof. |
| TWI541022B (en) | 2013-12-18 | 2016-07-11 | 應克隆公司 | Compound and treatment method for fibroblast growth factor receptor-3 (FGFR3) |
| EP3160513B1 (en) | 2014-06-30 | 2020-02-12 | Glykos Finland Oy | Saccharide derivative of a toxic payload and antibody conjugates thereof |
| CA2953371C (en) | 2014-06-30 | 2021-08-24 | Tarveda Therapeutics, Inc. | Targeted conjugates and particles and formulations thereof |
| KR20170040249A (en) | 2014-08-12 | 2017-04-12 | 노파르티스 아게 | Anti-cdh6 antibody drug conjugates |
| CN106999604B (en) * | 2014-09-02 | 2021-08-03 | 伊缪诺金公司 | Methods for formulating antibody drug conjugate compositions |
| EP3189057A1 (en) | 2014-09-03 | 2017-07-12 | ImmunoGen, Inc. | Cytotoxic benzodiazepine derivatives |
| ES2815353T3 (en) | 2014-09-03 | 2021-03-29 | Immunogen Inc | Cytotoxic benzodiazepine derivatives |
| CN104208719B (en) * | 2014-09-24 | 2017-05-03 | 北京天广实生物技术股份有限公司 | ADC (antibody-drug conjugate) cation exchange chromatographic purification method |
| IL313511A (en) | 2014-11-12 | 2024-08-01 | Seagen Inc | Glycan-interacting compounds and methods of use |
| EP3217962A4 (en) * | 2014-11-13 | 2018-06-27 | The Curators of the University of Missouri | Multiple human antibody-nanoparticle conjugates and methods of formation |
| TW201711702A (en) | 2015-06-04 | 2017-04-01 | 應克隆公司 | Therapies utilizing compounds to fibroblast growth factor receptor-3 (FGFR3) |
| EP3310813A1 (en) | 2015-06-17 | 2018-04-25 | Novartis AG | Antibody drug conjugates |
| EP3373969A4 (en) | 2015-11-12 | 2019-08-14 | Siamab Therapeutics, Inc. | GLYCAN-INTERACTING COMPOUNDS AND METHODS OF USE |
| FR3045058B1 (en) * | 2015-12-11 | 2017-12-29 | Ifp Energies Now | NOVEL POLYAMINES, PROCESS FOR THEIR SYNTHESIS AND THEIR USE FOR THE SELECTIVE REMOVAL OF H2S FROM A GASEOUS EFFLUENT COMPRISING CO2 |
| WO2017161206A1 (en) | 2016-03-16 | 2017-09-21 | Halozyme, Inc. | Conjugates containing conditionally active antibodies or antigen-binding fragments thereof, and methods of use |
| EP3541847A4 (en) | 2016-11-17 | 2020-07-08 | Seattle Genetics, Inc. | GLYCAN INTERACTING COMPOUNDS AND METHOD FOR USE |
| JOP20190187A1 (en) | 2017-02-03 | 2019-08-01 | Novartis Ag | Anti-ccr7 antibody drug conjugates |
| KR102653141B1 (en) | 2017-03-03 | 2024-04-01 | 씨젠 인크. | Glycan-interacting compounds and methods of use |
| CN110177569A (en) * | 2017-03-30 | 2019-08-27 | 江苏恒瑞医药股份有限公司 | The preparation method of antibody drug conjugates |
| WO2018185618A1 (en) | 2017-04-03 | 2018-10-11 | Novartis Ag | Anti-cdh6 antibody drug conjugates and anti-gitr antibody combinations and methods of treatment |
| WO2018195243A1 (en) | 2017-04-20 | 2018-10-25 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives and conjugates thereof |
| TW202413360A (en) | 2017-12-28 | 2024-04-01 | 美商伊繆諾金公司 | Benzodiazepine derivatives |
| EP3552631A1 (en) | 2018-04-10 | 2019-10-16 | Inatherys | Antibody-drug conjugates and their uses for the treatment of cancer |
| EP3947395A1 (en) | 2019-03-29 | 2022-02-09 | ImmunoGen, Inc. | Cytotoxic bis-benzodiazepine derivatives and conjugates thereof with cell-binding agents for inhibiting abnormal cell growth or for treating proliferative diseases |
| JP7561141B2 (en) | 2019-04-26 | 2024-10-03 | イミュノジェン・インコーポレーテッド | Camptothecin derivatives |
| CN113125732B (en) * | 2019-12-31 | 2023-08-08 | 科美博阳诊断技术(上海)有限公司 | Homogeneous detection kit for interleukin 6 and application thereof |
| US20230181756A1 (en) | 2020-04-30 | 2023-06-15 | Novartis Ag | Ccr7 antibody drug conjugates for treating cancer |
| AU2022441090A1 (en) | 2022-02-17 | 2024-08-22 | Novelty Nobility Inc. | Antibody-drug conjugate. |
| WO2024246657A1 (en) * | 2023-05-29 | 2024-12-05 | Intas Pharmaceuticals Ltd. | Method for conjugation and purification of antibody drug conjugate |
| AU2024292473A1 (en) | 2023-07-19 | 2026-01-29 | Iovance Biotherapeutics, Inc. | Treatment of cancer patients with tumor infiltrating lymphocyte therapies in combination with trop-2 targeting adc |
| WO2025027529A1 (en) | 2023-07-31 | 2025-02-06 | Advesya | Anti-il-1rap antibody drug conjugates and methods of use thereof |
| TW202530255A (en) | 2023-12-15 | 2025-08-01 | 法商亞維西亞有限公司 | Anti il-1rap binding domains and antibody-drug conjugates thereof |
| WO2025192691A1 (en) * | 2024-03-14 | 2025-09-18 | 東レ株式会社 | Pharmaceutical composition for treating and/or preventing cancer |
Family Cites Families (163)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US585089A (en) * | 1897-06-22 | durey | ||
| US585499A (en) * | 1897-06-29 | Oil-can | ||
| US552293A (en) * | 1895-12-31 | ktjhn | ||
| SE430062B (en) | 1977-03-04 | 1983-10-17 | Pharmacia Fine Chemicals Ab | COUPLING OR TIOLATION REAGENTS |
| US4137230A (en) | 1977-11-14 | 1979-01-30 | Takeda Chemical Industries, Ltd. | Method for the production of maytansinoids |
| US4307016A (en) | 1978-03-24 | 1981-12-22 | Takeda Chemical Industries, Ltd. | Demethyl maytansinoids |
| US4256746A (en) | 1978-11-14 | 1981-03-17 | Takeda Chemical Industries | Dechloromaytansinoids, their pharmaceutical compositions and method of use |
| JPS55102583A (en) | 1979-01-31 | 1980-08-05 | Takeda Chem Ind Ltd | 20-acyloxy-20-demethylmaytansinoid compound |
| JPS55162791A (en) | 1979-06-05 | 1980-12-18 | Takeda Chem Ind Ltd | Antibiotic c-15003pnd and its preparation |
| JPS5645483A (en) | 1979-09-19 | 1981-04-25 | Takeda Chem Ind Ltd | C-15003phm and its preparation |
| EP0028683A1 (en) | 1979-09-21 | 1981-05-20 | Takeda Chemical Industries, Ltd. | Antibiotic C-15003 PHO and production thereof |
| JPS5645485A (en) | 1979-09-21 | 1981-04-25 | Takeda Chem Ind Ltd | Production of c-15003pnd |
| WO1982001188A1 (en) | 1980-10-08 | 1982-04-15 | Takeda Chemical Industries Ltd | 4,5-deoxymaytansinoide compounds and process for preparing same |
| US4450254A (en) | 1980-11-03 | 1984-05-22 | Standard Oil Company | Impact improvement of high nitrile resins |
| US4315929A (en) | 1981-01-27 | 1982-02-16 | The United States Of America As Represented By The Secretary Of Agriculture | Method of controlling the European corn borer with trewiasine |
| US4313946A (en) | 1981-01-27 | 1982-02-02 | The United States Of America As Represented By The Secretary Of Agriculture | Chemotherapeutically active maytansinoids from Trewia nudiflora |
| US4563304A (en) | 1981-02-27 | 1986-01-07 | Pharmacia Fine Chemicals Ab | Pyridine compounds modifying proteins, polypeptides or polysaccharides |
| JPS57192389A (en) | 1981-05-20 | 1982-11-26 | Takeda Chem Ind Ltd | Novel maytansinoid |
| US4664911A (en) | 1983-06-21 | 1987-05-12 | Board Of Regents, University Of Texas System | Immunotoxin conjugates employing toxin B chain moieties |
| EP0192009B1 (en) | 1985-01-22 | 1991-09-04 | Saint-Gobain Vitrage International | Process for the preparation of a powder based on indium formiate for producing a thin layer on a substrate, especially on glass. |
| GB8600582D0 (en) * | 1986-01-10 | 1986-02-19 | Ca Minister Nat Defence | Purifying biological materials |
| US5225539A (en) | 1986-03-27 | 1993-07-06 | Medical Research Council | Recombinant altered antibodies and methods of making altered antibodies |
| GB8607679D0 (en) | 1986-03-27 | 1986-04-30 | Winter G P | Recombinant dna product |
| US4859449A (en) | 1987-09-14 | 1989-08-22 | Center For Molecular Medicine And Immunology | Modified antibodies for enhanced hepatocyte clearance |
| US5241078A (en) | 1988-06-14 | 1993-08-31 | Cetus Oncology | Coupling agents and sterically hindered disulfide linked conjugates prepared therefrom |
| US5024834A (en) | 1988-07-12 | 1991-06-18 | Cetus Corporation | Thioether linked immunotoxin conjugates |
| CA2006408A1 (en) | 1988-12-27 | 1990-06-27 | Susumu Iwasa | Bispecific monoclonal antibody, its production and use |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US5714149A (en) | 1989-02-10 | 1998-02-03 | Celltech Therapeutics Limited | Crosslinked antibodies and processes for their preparation |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| CA2026147C (en) | 1989-10-25 | 2006-02-07 | Ravi J. Chari | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| US6316003B1 (en) | 1989-12-21 | 2001-11-13 | Whitehead Institute For Biomedical Research | Tat-derived transport polypeptides |
| US5137877B1 (en) | 1990-05-14 | 1996-01-30 | Bristol Myers Squibb Co | Bifunctional linking compounds conjugates and methods for their production |
| GB9015198D0 (en) | 1990-07-10 | 1990-08-29 | Brien Caroline J O | Binding substance |
| ATE300615T1 (en) | 1990-08-29 | 2005-08-15 | Genpharm Int | TRANSGENIC MICE CAPABLE OF PRODUCING HETEROLOGOUS ANTIBODIES |
| US5545806A (en) | 1990-08-29 | 1996-08-13 | Genpharm International, Inc. | Ransgenic non-human animals for producing heterologous antibodies |
| US5256294A (en) | 1990-09-17 | 1993-10-26 | Genentech, Inc. | Tangential flow filtration process and apparatus |
| CA2048078A1 (en) | 1990-11-15 | 1992-05-16 | Wolfgang A. Wrasidlo | Chemical modification of antibodies for creation of immunoconjugates |
| US5252714A (en) | 1990-11-28 | 1993-10-12 | The University Of Alabama In Huntsville | Preparation and use of polyethylene glycol propionaldehyde |
| SE9102074D0 (en) | 1991-07-03 | 1991-07-03 | Kabi Pharmacia Ab | TOMOUR ANTIGEN SPECIFIC ANTIBODY |
| SE470006B (en) | 1991-09-26 | 1993-10-25 | Corline Systems Ab | New conjugate, its preparation and use, and substrates prepared with the conjugate |
| ES2341666T3 (en) | 1991-12-02 | 2010-06-24 | Medimmune Limited | PRODUCTION OF AUTHORTIC BODIES OF REPERTORIES OF ANTIQUE RPOS SEGMENTS EXPRESSED ON THE FAGOS SURFACE. |
| ES2149768T3 (en) | 1992-03-25 | 2000-11-16 | Immunogen Inc | CONJUGATES OF BINDING AGENTS OF CELLS DERIVED FROM CC-1065. |
| US5639641A (en) | 1992-09-09 | 1997-06-17 | Immunogen Inc. | Resurfacing of rodent antibodies |
| US5556623A (en) | 1993-03-30 | 1996-09-17 | Eli Lilly And Company | Antibody-drug conjugates |
| GB9320575D0 (en) | 1993-10-06 | 1993-11-24 | Amp Gmbh | Coaxial connector having improved locking mechanism |
| IL111748A0 (en) | 1993-12-03 | 1995-01-24 | Zeneca Ltd | Proteins |
| DE19503164A1 (en) * | 1994-02-04 | 1995-08-10 | Siemens Comp Inc | Optically coupled data switching system and handset cradle switch |
| US5919758A (en) | 1994-03-22 | 1999-07-06 | Beth Israel Deaconess Medical Center | Modified polypeptides with altered biological activity |
| US5747446A (en) | 1994-03-22 | 1998-05-05 | Beth Israel Deaconess Medical Center | Modified polypeptides with increased biological activity |
| US5580853A (en) | 1994-03-22 | 1996-12-03 | New England Deaconess Hospital | Modified polypeptides with increased biological activity |
| US5612474A (en) | 1994-06-30 | 1997-03-18 | Eli Lilly And Company | Acid labile immunoconjugate intermediates |
| WO1996039183A1 (en) | 1995-05-31 | 1996-12-12 | Fred Hutchinson Cancer Research Center | Compositions and methods for targeted delivery of effector molecules |
| US6265150B1 (en) | 1995-06-07 | 2001-07-24 | Becton Dickinson & Company | Phage antibodies |
| US5714352A (en) | 1996-03-20 | 1998-02-03 | Xenotech Incorporated | Directed switch-mediated DNA recombination |
| GB9610992D0 (en) | 1996-05-24 | 1996-07-31 | Glaxo Group Ltd | Concentrated antibody preparation |
| WO1998026747A2 (en) | 1996-12-17 | 1998-06-25 | Terman David S | Superantigen based methods and compositions for treatment of diseases |
| US6462070B1 (en) | 1997-03-06 | 2002-10-08 | The General Hospital Corporation | Photosensitizer conjugates for pathogen targeting |
| US6371975B2 (en) | 1998-11-06 | 2002-04-16 | Neomend, Inc. | Compositions, systems, and methods for creating in situ, chemically cross-linked, mechanical barriers |
| AU736707B2 (en) | 1997-06-11 | 2001-08-02 | Anaphore, Inc. | Trimerising module |
| US6171586B1 (en) | 1997-06-13 | 2001-01-09 | Genentech, Inc. | Antibody formulation |
| US5958677A (en) | 1997-07-28 | 1999-09-28 | The New York Blood Center, Inc. | Method for purifying viral nucleic acids |
| CA2297070A1 (en) | 1997-08-01 | 1999-02-11 | Morphosys Ag | Novel method and phage for the identification of nucleic acid sequences encoding members of a multimeric (poly)peptide complex |
| US5965714A (en) * | 1997-10-02 | 1999-10-12 | Connaught Laboratories, Inc. | Method for the covalent attachment of polysaccharides to protein molecules |
| ES2245805T3 (en) | 1997-10-03 | 2006-01-16 | Galenica Pharmaceuticals, Inc. | POLISACARIDS FORMING IMINAS, PREPARATION OF THE SAME AND THE USE OF THE SAME AS ASSISTANTS OF IMMUNO STIMULANTS. |
| US6121236A (en) | 1998-03-24 | 2000-09-19 | The Children's Medical Center Corporation | Multivalent ligands which modulate angiogenesis |
| US5963761A (en) * | 1998-04-15 | 1999-10-05 | Xerox Corporation | Area coverage sensor calibration and algorithm for seam detection noise eliminator on a seamed photoreceptor |
| CA2329897A1 (en) | 1998-04-28 | 1999-11-04 | Galenica Pharmaceuticals, Inc. | Polysaccharide-antigen conjugates |
| US5981564A (en) | 1998-07-01 | 1999-11-09 | Universite Laval | Water-soluble derivatives of paclitaxel, method for producing same and uses thereof |
| US7067109B1 (en) | 1998-07-13 | 2006-06-27 | Board Of Regents, The University Of Texas System | Cancer treatment kits comprising therapeutic conjugates that bind to aminophospholipids |
| AUPQ014799A0 (en) | 1999-05-04 | 1999-05-27 | Access Pharmaceuticals Australia Pty Limited | Amplification of folate-mediated targeting to tumor cells using polymers |
| AU775373B2 (en) * | 1999-10-01 | 2004-07-29 | Immunogen, Inc. | Compositions and methods for treating cancer using immunoconjugates and chemotherapeutic agents |
| ES2276708T3 (en) | 1999-11-24 | 2007-07-01 | Immunogen, Inc. | CYTOTOXIC AGENTS UNDERSTANDING TAXANS AND THERAPEUTIC USE. |
| ATE344801T1 (en) | 1999-12-29 | 2006-11-15 | Immunogen Inc | CYTOTOXIC AGENTS CONTAINING DOXORUBICIN AND DAUNORUBICIN AND THEIR THERAPEUTIC USE |
| WO2001058479A1 (en) | 2000-02-08 | 2001-08-16 | The Penn State Research Foundation | Immunotherapy using interleukin 13 receptor subunit alpha 2 |
| US6632979B2 (en) | 2000-03-16 | 2003-10-14 | Genentech, Inc. | Rodent HER2 tumor model |
| US7097840B2 (en) | 2000-03-16 | 2006-08-29 | Genentech, Inc. | Methods of treatment using anti-ErbB antibody-maytansinoid conjugates |
| NZ539683A (en) | 2000-07-24 | 2006-11-30 | Microcell Corp | Microcell electrochemical devices and assemblies, and method of making and using the same |
| US6596503B1 (en) | 2000-08-18 | 2003-07-22 | East Carolina University | Monoclonal antibody DS6, tumor-associated antigen CA6, and methods of use thereof |
| US6333410B1 (en) | 2000-08-18 | 2001-12-25 | Immunogen, Inc. | Process for the preparation and purification of thiol-containing maytansinoids |
| US20060062786A1 (en) | 2000-11-08 | 2006-03-23 | Human Genome Sciences, Inc. | Antibodies that immunospecifically bind to TRAIL receptors |
| BR0206985A (en) | 2001-01-29 | 2005-04-19 | Idec Pharma Corp | Modified Antibodies and Methods of Use |
| WO2002098885A1 (en) | 2001-02-07 | 2002-12-12 | Beth Israel Deaconess Medical Center | Modified psma ligands and uses related thereto |
| US20030003048A1 (en) | 2001-04-26 | 2003-01-02 | Chun Li | Diagnostic imaging compositions, their methods of synthesis and use |
| EP1392361A4 (en) | 2001-05-11 | 2009-08-05 | Univ Texas | THERAPY BASED ON ANTI-CD26 MONOCLONAL ANTIBODIES AGAINST DISORDERS ASSOCIATED WITH CELLS EXPRESSING CD26 |
| EP1258255A1 (en) * | 2001-05-18 | 2002-11-20 | Boehringer Ingelheim International GmbH | Conjugates of an antibody to CD44 and a maytansinoid |
| US6441163B1 (en) * | 2001-05-31 | 2002-08-27 | Immunogen, Inc. | Methods for preparation of cytotoxic conjugates of maytansinoids and cell binding agents |
| EP2277542B1 (en) | 2001-06-01 | 2014-04-16 | Cornell Research Foundation Inc. | Modified antibodies to prostrate-specific membrane antigen and uses thereof |
| US6576802B2 (en) | 2001-06-04 | 2003-06-10 | Shell Oil Company | One-step production of 1, 3-propanediol from ethylene oxide and syngas with a catalyst with a phospholanoalkane ligand |
| JP2005532415A (en) | 2001-12-11 | 2005-10-27 | メルク エンド カムパニー インコーポレーテッド | Staphylococcus aureus exopolysaccharide and method |
| US6716821B2 (en) | 2001-12-21 | 2004-04-06 | Immunogen Inc. | Cytotoxic agents bearing a reactive polyethylene glycol moiety, cytotoxic conjugates comprising polyethylene glycol linking groups, and methods of making and using the same |
| AU2003201824A1 (en) | 2002-01-03 | 2003-07-24 | Smithkline Beecham Corporation | Methods for preparing immunoconjugates |
| US7591994B2 (en) | 2002-12-13 | 2009-09-22 | Immunomedics, Inc. | Camptothecin-binding moiety conjugates |
| US8877901B2 (en) | 2002-12-13 | 2014-11-04 | Immunomedics, Inc. | Camptothecin-binding moiety conjugates |
| US6756397B2 (en) | 2002-04-05 | 2004-06-29 | Immunogen, Inc. | Prodrugs of CC-1065 analogs |
| US6534660B1 (en) | 2002-04-05 | 2003-03-18 | Immunogen, Inc. | CC-1065 analog synthesis |
| CA2478925C (en) | 2002-04-26 | 2016-06-07 | Robert Lee Fahrner | Non-affinity purification of proteins |
| SI2371392T1 (en) | 2002-05-02 | 2015-10-30 | Wyeth Holdings Llc | Calicheamicin derivative-carrier conjugates |
| US20090068178A1 (en) | 2002-05-08 | 2009-03-12 | Genentech, Inc. | Compositions and Methods for the Treatment of Tumor of Hematopoietic Origin |
| US6596757B1 (en) | 2002-05-14 | 2003-07-22 | Immunogen Inc. | Cytotoxic agents comprising polyethylene glycol-containing taxanes and their therapeutic use |
| WO2004001007A2 (en) | 2002-06-21 | 2003-12-31 | Idec Pharmaceuticals Corporation | Buffered formulations for concentrating antibodies and methods of use thereof |
| US7390898B2 (en) | 2002-08-02 | 2008-06-24 | Immunogen Inc. | Cytotoxic agents containing novel potent taxanes and their therapeutic use |
| JP2006500364A (en) | 2002-08-16 | 2006-01-05 | イムノージェン インコーポレーテッド | Crosslinkers with high reactivity and solubility and their use in the preparation of conjugates for targeted delivery of small molecule drugs |
| ATE409048T1 (en) | 2002-10-08 | 2008-10-15 | Fresenius Kabi De Gmbh | PHARMACEUTICALLY ACTIVE OLIGOSACCHARIDE CONJUGATES |
| PT2348125T (en) | 2002-10-30 | 2017-08-29 | Nuevolution As | Method for the synthesis of a bifunctional complex |
| BR0316101A (en) | 2002-11-07 | 2005-09-27 | Immunogen Inc | Anticd33 antibodies and process for treating acute meloid leukemia using the same. |
| KR101424624B1 (en) * | 2003-05-14 | 2014-07-31 | 이뮤노젠 아이엔씨 | Drug Conjugate Composition |
| US8088387B2 (en) | 2003-10-10 | 2012-01-03 | Immunogen Inc. | Method of targeting specific cell populations using cell-binding agent maytansinoid conjugates linked via a non-cleavable linker, said conjugates, and methods of making said conjugates |
| US7276497B2 (en) | 2003-05-20 | 2007-10-02 | Immunogen Inc. | Cytotoxic agents comprising new maytansinoids |
| PL3524611T3 (en) * | 2003-05-20 | 2021-06-14 | Immunogen, Inc. | Improved cytotoxic agents comprising new maytansinoids |
| CN102558352A (en) | 2003-06-27 | 2012-07-11 | 艾默根佛蒙特有限公司 | Antibodies directed to the deletion mutants of epidermal growth factor receptor and uses thereof |
| US20050074425A1 (en) | 2003-07-02 | 2005-04-07 | Polycord, Inc. | Method for delivering polymerized therapeutic agent compositions and compositions thereof |
| WO2005044998A2 (en) | 2003-11-05 | 2005-05-19 | Palingen, Inc. | Enhanced b cell cytotoxicity of cdim binding antibody |
| US20050175619A1 (en) | 2004-02-05 | 2005-08-11 | Robert Duffy | Methods of producing antibody conjugates |
| US20110064754A1 (en) | 2005-03-03 | 2011-03-17 | Center For Molecular Medicine And Immunology | Immunoconjugates Comprising Poxvirus-Derived Peptides and Antibodies Against Antigen-Presenting Cells for Subunit-Based Poxvirus Vaccines |
| EP1610818A4 (en) * | 2004-03-03 | 2007-09-19 | Millennium Pharm Inc | Modified antibodies to prostate-specific membrane antigen and uses thereof |
| WO2005115477A2 (en) | 2004-04-13 | 2005-12-08 | Quintessence Biosciences, Inc. | Non-natural ribonuclease conjugates as cytotoxic agents |
| US20060073528A1 (en) | 2004-05-14 | 2006-04-06 | Jean-Michel Lecerf | Measurement methods |
| RU2402548C2 (en) | 2004-05-19 | 2010-10-27 | Медарекс, Инк. | Chemical linkers and conjugates thereof |
| JP4806680B2 (en) | 2004-05-19 | 2011-11-02 | メダレックス インコーポレイテッド | Self-sacrificing linker and drug conjugate |
| KR20120064120A (en) | 2004-06-01 | 2012-06-18 | 제넨테크, 인크. | Antibody drug conjugates and methods |
| US7313946B2 (en) * | 2004-07-15 | 2008-01-01 | Matsuo Electric Co., Ltd. | Moisture detector |
| GT200500283A (en) | 2004-10-08 | 2006-05-08 | IMMUNOTHERAPY OF AUTOIMMUNE DISORDERS | |
| EP1817341A2 (en) | 2004-11-29 | 2007-08-15 | Seattle Genetics, Inc. | Engineered antibodies and immunoconjugates |
| US7408030B2 (en) * | 2005-01-13 | 2008-08-05 | North Carolina State University | Purification of immunoglobulins using affinity chromatography and peptide ligands |
| US20110166319A1 (en) | 2005-02-11 | 2011-07-07 | Immunogen, Inc. | Process for preparing purified drug conjugates |
| CA2597407C (en) | 2005-02-11 | 2013-09-10 | Immunogen, Inc. | Process for preparing stable drug conjugates |
| EA200702239A1 (en) | 2005-04-15 | 2008-10-30 | Иммьюноджен, Инк. | DESTRUCTION OF HETEROGENOUS OR MIXED CELL POPULATION IN TUMORS |
| GB2427360A (en) | 2005-06-22 | 2006-12-27 | Complex Biosystems Gmbh | Aliphatic prodrug linker |
| JP5436856B2 (en) | 2005-07-15 | 2014-03-05 | アンジオケム インコーポレーティッド | Use of an aprotinin polypeptide as a carrier in a pharmaceutical complex |
| WO2007019232A2 (en) | 2005-08-03 | 2007-02-15 | Immunogen, Inc. | Immunoconjugate formulations |
| EP3539572A1 (en) | 2005-08-24 | 2019-09-18 | ImmunoGen, Inc. | Process for preparing maytansinoid antibody conjugates |
| CN101443044A (en) | 2005-09-22 | 2009-05-27 | 哈达斯特医学研究服务与开发有限公司 | Dextran and arabinogalactan conjugates of therapeutically active compounds |
| BRPI0617546A2 (en) | 2005-09-26 | 2011-07-26 | Medarex Inc | drug-antibody conjugate, pharmaceutical formulation, method for killing a tumor cell, method for retarding or arresting tumor growth in a mammalian subject and compound |
| EP2298308B1 (en) | 2005-11-14 | 2013-01-16 | University Of Southern California | Integrin-binding small molecules |
| US7964415B2 (en) | 2006-04-26 | 2011-06-21 | Cardiogenics Inc. | Stable water-soluble polyethylenimine conjugates and methods of use thereof |
| US7307016B1 (en) * | 2006-06-07 | 2007-12-11 | Grace Semiconductor Manufacturing Corporation | Method of processing metal surface in dual damascene manufacturing |
| ES2673822T3 (en) | 2006-07-18 | 2018-06-25 | Sanofi | Antagonist antibody against EphA2 for cancer treatment |
| US20080213349A1 (en) | 2006-09-11 | 2008-09-04 | Deepak Ramesh Thakker | Liposome Complexes Containing Pharmaceutical Agents and Methods |
| US8969532B2 (en) | 2006-10-03 | 2015-03-03 | Novo Nordisk A/S | Methods for the purification of polypeptide conjugates comprising polyalkylene oxide using hydrophobic interaction chromatography |
| PL2019104T3 (en) | 2007-07-19 | 2014-03-31 | Sanofi Sa | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| KR20200058590A (en) | 2008-04-30 | 2020-05-27 | 이뮤노젠 아이엔씨 | Cross-linkers and their uses |
| EP2276506A4 (en) | 2008-04-30 | 2014-05-07 | Immunogen Inc | Potent conjugates and hydrophilic linkers |
| GB0811743D0 (en) | 2008-06-26 | 2008-07-30 | Hemosol Biopharma Inc | Composition |
| CA2742846A1 (en) | 2008-11-17 | 2010-05-20 | Enzon Pharmaceuticals, Inc. | Releasable fusogenic lipids for nucleic acids delivery systems |
| CN102365021B (en) | 2009-02-05 | 2015-07-15 | 伊缪诺金公司 | Novel benzodiazepine derivatives |
| BRPI1010620B8 (en) | 2009-06-03 | 2021-05-25 | Immunogen Inc | conjugation methods |
| FR2947269B1 (en) | 2009-06-29 | 2013-01-18 | Sanofi Aventis | NEW ANTICANCER COMPOUNDS |
| TW201116297A (en) | 2009-10-02 | 2011-05-16 | Sanofi Aventis | Antibodies that specifically bind to the EphA2 receptor |
| CN103037900B (en) | 2010-02-24 | 2016-04-06 | 伊缪诺金公司 | Folate receptor 1 antibody and immunoconjugate and use thereof |
| KR20110103182A (en) | 2010-03-12 | 2011-09-20 | 삼성전자주식회사 | Stereoscopic image display device |
| US20120149732A1 (en) | 2010-12-14 | 2012-06-14 | Alexander Chucholowski | Multifunctional linkers and methods for the use thereof |
| JP6049642B2 (en) | 2011-02-15 | 2016-12-21 | イミュノジェン・インコーポレーテッド | Method for preparing complex |
| WO2012135517A2 (en) | 2011-03-29 | 2012-10-04 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| RS58367B1 (en) | 2011-03-29 | 2019-03-29 | Immunogen Inc | Preparation of maytansinoid antibody conjugates by a one-step process |
| MX2013011201A (en) | 2011-03-29 | 2013-12-16 | Immunogen Inc | Process for manufacturing conjugates of improved homogeneity. |
| IL281714B2 (en) | 2011-04-01 | 2023-11-01 | Immunogen Inc | Methods for increasing efficacy of folr1 cancer therapy |
| US20130071482A1 (en) | 2011-09-20 | 2013-03-21 | The University Of Kentucky Research Foundation | Block copolymer cross-linked nanoassemblies as modular delivery vehicles |
| US20140350228A1 (en) | 2011-12-13 | 2014-11-27 | Immunogen, Inc. | Use of n-hydroxysuccinimide to improve conjugate stability |
| MX2015004258A (en) | 2012-10-04 | 2015-09-25 | Immunogen Inc | Use of an ion exchange membrane to remove impurities from cell-binding agent cytotoxic agent conjugates. |
| WO2014055842A1 (en) | 2012-10-04 | 2014-04-10 | Immunogen, Inc. | Process for preparing stable antibody maytansinoid conjugates |
| WO2014055877A1 (en) | 2012-10-04 | 2014-04-10 | Immunogen, Inc. | Use of a pvdf membrane to purify cell-binding agent cytotoxic agent conjugates |
| TR201905612T4 (en) | 2012-12-05 | 2019-05-21 | Univ Heidelberg Ruprecht Karls | Conjugates of proteins and polyvalent cell-penetrating peptides and their uses. |
-
2006
- 2006-08-14 EP EP19156303.0A patent/EP3539572A1/en active Pending
- 2006-08-14 PL PL06801436T patent/PL1928503T3/en unknown
- 2006-08-14 CA CA2893252A patent/CA2893252C/en active Active
- 2006-08-14 AU AU2006283726A patent/AU2006283726C1/en active Active
- 2006-08-14 EA EA200800657A patent/EA013327B1/en active IP Right Revival
- 2006-08-14 CN CN2006800342426A patent/CN101267841B/en active Active
- 2006-08-14 KR KR1020087004084A patent/KR101301011B1/en active Active
- 2006-08-14 DK DK06801436.4T patent/DK1928503T3/en active
- 2006-08-14 NZ NZ716641A patent/NZ716641A/en unknown
- 2006-08-14 IL IL305084A patent/IL305084A/en unknown
- 2006-08-14 NZ NZ609752A patent/NZ609752A/en unknown
- 2006-08-14 US US11/503,781 patent/US7811572B2/en active Active
- 2006-08-14 CA CA2794554A patent/CA2794554C/en active Active
- 2006-08-14 ES ES06801436T patent/ES2390826T3/en active Active
- 2006-08-14 NZ NZ565964A patent/NZ565964A/en unknown
- 2006-08-14 PT PT06801436T patent/PT1928503E/en unknown
- 2006-08-14 CA CA2620343A patent/CA2620343C/en active Active
- 2006-08-14 ES ES11004940.0T patent/ES2533992T3/en active Active
- 2006-08-14 BR BRPI0615049-7A patent/BRPI0615049B1/en active IP Right Grant
- 2006-08-14 SI SI200631435T patent/SI1928503T1/en unknown
- 2006-08-14 IL IL282138A patent/IL282138B2/en unknown
- 2006-08-14 WO PCT/US2006/031653 patent/WO2007024536A2/en not_active Ceased
- 2006-08-14 CA CA3250822A patent/CA3250822A1/en active Pending
- 2006-08-14 JP JP2008527970A patent/JP5350792B2/en active Active
- 2006-08-14 NZ NZ595430A patent/NZ595430A/en unknown
- 2006-08-14 RS RS20120425A patent/RS52470B/en unknown
- 2006-08-14 CA CA3190867A patent/CA3190867C/en active Active
- 2006-08-14 HR HRP20120794TT patent/HRP20120794T1/en unknown
- 2006-08-14 CA CA3000520A patent/CA3000520C/en active Active
- 2006-08-14 MX MX2008002597A patent/MX2008002597A/en active IP Right Grant
- 2006-08-14 CN CN201210308200.4A patent/CN102989000B/en active Active
- 2006-08-14 EP EP06801436A patent/EP1928503B1/en active Active
- 2006-08-14 EP EP20130178039 patent/EP2662096A1/en not_active Withdrawn
- 2006-08-14 EP EP11004940.0A patent/EP2399609B1/en active Active
-
2008
- 2008-02-12 IL IL189461A patent/IL189461A/en active IP Right Grant
- 2008-02-15 ZA ZA2008/01564A patent/ZA200801564B/en unknown
- 2008-02-18 CR CR9742A patent/CR9742A/en unknown
- 2008-02-22 EC EC2008008212A patent/ECSP088212A/en unknown
-
2010
- 2010-10-08 US US12/901,039 patent/US8383122B2/en active Active
-
2012
- 2012-07-08 IL IL220816A patent/IL220816A/en active IP Right Grant
- 2012-09-11 JP JP2012199467A patent/JP5738248B2/en active Active
- 2012-10-17 CY CY20121100972T patent/CY1113206T1/en unknown
-
2013
- 2013-02-25 US US13/776,097 patent/US8933205B2/en active Active
- 2013-06-17 IL IL226985A patent/IL226985A/en active IP Right Grant
-
2014
- 2014-05-12 JP JP2014098796A patent/JP6028001B2/en active Active
-
2015
- 2015-01-05 US US14/589,541 patent/US9789204B2/en active Active
- 2015-07-03 CR CR20150350A patent/CR20150350A/en unknown
-
2016
- 2016-09-27 IL IL248076A patent/IL248076B/en active IP Right Grant
-
2018
- 2018-04-27 US US15/964,418 patent/US11471536B2/en active Active
-
2022
- 2022-09-08 US US17/940,297 patent/US20230158168A1/en not_active Abandoned
-
2025
- 2025-01-16 US US19/023,875 patent/US20250255979A1/en active Pending
Non-Patent Citations (5)
| Title |
|---|
| "Antibodies: A Laboratory Manual", 1988, CSH PRESS |
| "Immunobiology", 2001, GARLAND PUBLISHING |
| HASKARD; ARCHER, J. IMMUNOL. METHODS, vol. 74, no. 2, 1984, pages 361 - 67 |
| KOHLER; MILSTEIN, EUR. J. IMMUNOL., vol. 5, 1976, pages 511 - 519 |
| REITER ET AL., PROTEIN ENGINEERING, vol. 7, 1994, pages 697 - 704 |
Cited By (76)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8933205B2 (en) | 2005-08-24 | 2015-01-13 | Immunogen, Inc. | Process for preparing purified drug conjugates |
| US11471536B2 (en) | 2005-08-24 | 2022-10-18 | Immunogen, Inc. | Process for preparing purified drug conjugates |
| US9789204B2 (en) | 2005-08-24 | 2017-10-17 | Immunogen, Inc. | Process for preparing purified drug conjugates |
| US11046762B2 (en) | 2008-04-30 | 2021-06-29 | Immunogen, Inc. | Cross-linkers and their uses |
| US10494431B2 (en) | 2008-04-30 | 2019-12-03 | Immunogen, Inc. | Cross-linkers and their uses |
| CN104524592A (en) * | 2008-04-30 | 2015-04-22 | 伊缪诺金公司 | Cross-linkers and their uses |
| EP2424875A4 (en) * | 2009-04-29 | 2012-10-31 | Bio Rad Laboratories | PURIFICATION OF IMMUNOCONJUGATES |
| US8481694B2 (en) | 2009-04-29 | 2013-07-09 | Bio-Rad Laboratories, Inc. | Purification of immunoconjugates |
| WO2010126979A1 (en) | 2009-04-29 | 2010-11-04 | Bio-Rad Laboratories, Inc. | Purification of immunoconjugates |
| EP2424875B1 (en) | 2009-04-29 | 2016-01-06 | Bio-Rad Laboratories, Inc. | Purification of immunoconjugates |
| US9771432B2 (en) | 2009-06-03 | 2017-09-26 | Immunogen, Inc. | Conjugation methods |
| US11498979B2 (en) | 2009-06-03 | 2022-11-15 | Immunogen, Inc. | Methods for preparing a purified maytansinoid conjugate in a solution |
| EP2437790A4 (en) * | 2009-06-03 | 2015-06-24 | Immunogen Inc | METHODS OF CONJUGATION |
| US10815309B2 (en) | 2009-06-03 | 2020-10-27 | Immunogen, Inc. | Methods for preparing antibody-drug conjugates |
| US10233257B2 (en) | 2009-06-03 | 2019-03-19 | Immunogen, Inc. | Methods for preparing antibody-drug conjugates |
| KR20190015590A (en) * | 2009-06-03 | 2019-02-13 | 이뮤노젠 아이엔씨 | Conjugation methods |
| US9376500B2 (en) | 2009-06-03 | 2016-06-28 | Immunogen, Inc. | Conjugation methods |
| KR101947176B1 (en) * | 2009-06-03 | 2019-02-12 | 이뮤노젠 아이엔씨 | Conjugation methods |
| KR102139019B1 (en) * | 2009-06-03 | 2020-07-28 | 이뮤노젠 아이엔씨 | Conjugation methods |
| KR20120088549A (en) * | 2009-06-03 | 2012-08-08 | 이뮤노젠 아이엔씨 | Splicing method |
| US9914748B2 (en) | 2011-03-29 | 2018-03-13 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| US9428543B2 (en) | 2011-03-29 | 2016-08-30 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| EP3545977A1 (en) * | 2011-03-29 | 2019-10-02 | ImmunoGen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| US8795673B2 (en) | 2011-03-29 | 2014-08-05 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| AU2016202832B2 (en) * | 2011-03-29 | 2018-05-10 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| US11744900B2 (en) | 2011-03-29 | 2023-09-05 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| US11090390B2 (en) | 2011-03-29 | 2021-08-17 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| WO2012135517A3 (en) * | 2011-03-29 | 2013-02-21 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| EA033468B1 (en) * | 2011-03-29 | 2019-10-31 | Immunogen Inc | Processes for preparing an "antibody-maytansinoid" conjugate |
| AU2012236398B2 (en) * | 2011-03-29 | 2016-02-11 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| US10435432B2 (en) | 2011-03-29 | 2019-10-08 | Immunogen, Inc. | Preparation of maytansinoid antibody conjugates by a one-step process |
| WO2012143496A2 (en) | 2011-04-21 | 2012-10-26 | Bayer Intellectual Property Gmbh | Novel binder-drug conjugates (adcs) and their use |
| WO2012143499A2 (en) | 2011-04-21 | 2012-10-26 | Bayer Intellectual Property Gmbh | Novel binder-drug conjugates (adcs) and their use |
| WO2014055842A1 (en) * | 2012-10-04 | 2014-04-10 | Immunogen, Inc. | Process for preparing stable antibody maytansinoid conjugates |
| EP2903450A4 (en) * | 2012-10-04 | 2016-05-11 | Immunogen Inc | Use of an ion exchange membrane to remove impurities from cell-binding agent cytotoxic agent conjugates |
| US10035817B2 (en) | 2012-10-04 | 2018-07-31 | Immunogen, Inc. | Method of purifying cell-binding agent-cytotoxic agent conjugates with a PVDF membrane |
| US10918735B2 (en) | 2012-12-04 | 2021-02-16 | Massachusetts Institute Of Technology | Substituted pyrazino[1′,2′:1,5]pyrrolo[2,3-b]indole-1,4-diones for cancer treatment |
| WO2015009740A2 (en) | 2013-07-15 | 2015-01-22 | Cell Signaling Technology, Inc. | Anti-mucin 1 binding agents and uses thereof |
| EP3699200A1 (en) | 2013-07-15 | 2020-08-26 | Cell Signaling Technology, Inc. | Anti-mucin 1 binding agents and uses thereof |
| US10022453B2 (en) | 2013-12-23 | 2018-07-17 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (ADCs) with kinesin spindel protein (KSP) |
| WO2016207089A1 (en) | 2015-06-22 | 2016-12-29 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (adcs) and antibody prodrug conjugates (apdcs) with enzymatically cleavable groups |
| US11123439B2 (en) | 2015-06-22 | 2021-09-21 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (ADCS) and antibody prodrug conjugates (APDCS) with enzymatically cleavable groups |
| US12144865B2 (en) | 2015-06-22 | 2024-11-19 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates with enzymatically cleavable groups |
| EP4406606A2 (en) | 2015-06-22 | 2024-07-31 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (adcs) and antibody prodrug conjugates (apdcs) with enzymatically cleavable g |
| WO2017060322A2 (en) | 2015-10-10 | 2017-04-13 | Bayer Pharma Aktiengesellschaft | Ptefb-inhibitor-adc |
| US11160871B2 (en) | 2015-10-28 | 2021-11-02 | Tarveda Therapeutics, Inc. | SSTR-targeted conjugates and particles and formulations thereof |
| US11685714B2 (en) | 2016-03-24 | 2023-06-27 | Bayer Pharma Aktiengesellschaft | Prodrugs of cytotoxic active agents having enzymatically cleavable groups |
| WO2017162663A1 (en) | 2016-03-24 | 2017-09-28 | Bayer Pharma Aktiengesellschaft | Prodrugs of cytotoxic active agents having enzymatically cleavable groups |
| US10918627B2 (en) | 2016-05-11 | 2021-02-16 | Massachusetts Institute Of Technology | Convergent and enantioselective total synthesis of Communesin analogs |
| US11643469B2 (en) | 2016-06-15 | 2023-05-09 | Bayer Pharma Aktiengesellschaft | Specific antibody-drug-conjugates (ADCs) with KSP inhibitors and anti-CD123-antibodies |
| US11001636B2 (en) | 2016-06-15 | 2021-05-11 | Bayer Pharma Aktiengesellschaft | Specific antibody-drug-conjugates (ADCs) with KSP inhibitors and anti-CD123-antibodies |
| WO2018112334A1 (en) | 2016-12-16 | 2018-06-21 | Bluefin Biomedicine, Inc. | Anti-cub domain-containing protein 1 (cdcp1) antibodies, antibody drug conjugates, and methods of use thereof |
| US11433140B2 (en) | 2016-12-21 | 2022-09-06 | Bayer Pharma Aktiengesellschaft | Specific antibody drug conjugates (ADCs) having KSP inhibitors |
| US12059472B2 (en) | 2016-12-21 | 2024-08-13 | Bayer Aktiengesellschaft | Prodrugs of cytotoxic active agents having enzymatically cleavable groups |
| WO2018114578A1 (en) | 2016-12-21 | 2018-06-28 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (adcs) having enzymatically cleavable groups |
| WO2018114798A1 (en) | 2016-12-21 | 2018-06-28 | Bayer Aktiengesellschaft | Prodrugs of cytotoxic active agents having enzymatically cleavable groups |
| US11478554B2 (en) | 2016-12-21 | 2022-10-25 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates (ADCS) having enzymatically cleavable groups |
| US11660351B2 (en) | 2016-12-21 | 2023-05-30 | Bayer Aktiengesellschaft | Antibody drug conjugates (ADCs) having enzymatically cleavable groups |
| US20180311375A1 (en) * | 2017-04-27 | 2018-11-01 | Cadila Healthcare Limited | Process of preparing antibody-drug conjugate |
| US11932650B2 (en) | 2017-05-11 | 2024-03-19 | Massachusetts Institute Of Technology | Potent agelastatin derivatives as modulators for cancer invasion and metastasis |
| US10640508B2 (en) | 2017-10-13 | 2020-05-05 | Massachusetts Institute Of Technology | Diazene directed modular synthesis of compounds with quaternary carbon centers |
| WO2019105835A1 (en) | 2017-11-29 | 2019-06-06 | Bayer Consumer Care Ag | Combinations of copanlisib and anetumab ravtansine |
| US11274121B2 (en) | 2018-01-12 | 2022-03-15 | Immunogen, Inc. | Methods for antibody drug conjugation, purification, and formulation |
| KR20210038904A (en) | 2018-07-25 | 2021-04-08 | 다이이찌 산쿄 가부시키가이샤 | Effective method for preparing antibody-drug conjugates |
| WO2020022363A1 (en) | 2018-07-25 | 2020-01-30 | 第一三共株式会社 | Effective method for manufacturing antibody-drug conjugate |
| WO2020084115A1 (en) | 2018-10-25 | 2020-04-30 | Pharma Mar, S.A. | Antibody drug conjugates comprising ecteinascidin derivatives |
| WO2020234114A1 (en) | 2019-05-21 | 2020-11-26 | Bayer Aktiengesellschaft | A novel stable high concentration formulation for anetumab ravtansine |
| US11535634B2 (en) | 2019-06-05 | 2022-12-27 | Massachusetts Institute Of Technology | Compounds, conjugates, and compositions of epipolythiodiketopiperazines and polythiodiketopiperazines and uses thereof |
| US12180228B2 (en) | 2019-06-05 | 2024-12-31 | Massachusetts Institute Of Technology | Compounds, conjugates, and compositions of epipolythiodiketopiperazines and polythiodiketopiperazines and uses thereof |
| US11634508B2 (en) | 2019-07-10 | 2023-04-25 | Cybrexa 2, Inc. | Peptide conjugates of cytotoxins as therapeutics |
| US12234212B2 (en) | 2019-07-10 | 2025-02-25 | Cybrexa 3, Inc. | Peptide conjugates of microtubule-targeting agents as therapeutics |
| US12410262B2 (en) | 2019-07-10 | 2025-09-09 | Cybrexa 2, Inc. | Peptide conjugates of cytotoxins as therapeutics |
| WO2021043951A1 (en) | 2019-09-05 | 2021-03-11 | Pharma Mar, S.A. | Drug antibody conjugates |
| WO2021214126A1 (en) | 2020-04-21 | 2021-10-28 | Pharma Mar, S.A. | Drug antibody conjugates |
| US12030888B2 (en) | 2021-02-24 | 2024-07-09 | Massachusetts Institute Of Technology | Himastatin derivatives, and processes of preparation thereof, and uses thereof |
| WO2025005240A1 (en) | 2023-06-30 | 2025-01-02 | 第一三共株式会社 | Antibody-drug conjugate production method including purification step using activated carbon material |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11471536B2 (en) | Process for preparing purified drug conjugates | |
| AU2017218969B2 (en) | Process for preparing maytansinoid antibody conjugates | |
| HK40014343A (en) | Process for preparing maytansinoid antibody conjugates | |
| HK1192713A (en) | Process for preparing maytansinoid antibody conjugates | |
| HK1165329B (en) | Process for preparing maytansinoid antibody conjugates |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200680034242.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 189461 Country of ref document: IL Ref document number: 2006283726 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 565964 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: CR2008-009742 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020087004084 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 885/CHENP/2008 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008527970 Country of ref document: JP Ref document number: MX/a/2008/002597 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 2620343 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08019768 Country of ref document: CO Ref document number: 08019768B Country of ref document: CO |
|
| ENP | Entry into the national phase |
Ref document number: 2006283726 Country of ref document: AU Date of ref document: 20060814 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006801436 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200800657 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: PI0615049 Country of ref document: BR Kind code of ref document: A2 Effective date: 20080221 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 220816 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: P-2012/0425 Country of ref document: RS |
|
| WWE | Wipo information: entry into national phase |
Ref document number: CR2015-000350 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 248076 Country of ref document: IL |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 305084 Country of ref document: IL |