WO2007134343A2 - Chloride heap leaching - Google Patents
Chloride heap leaching Download PDFInfo
- Publication number
- WO2007134343A2 WO2007134343A2 PCT/ZA2007/000025 ZA2007000025W WO2007134343A2 WO 2007134343 A2 WO2007134343 A2 WO 2007134343A2 ZA 2007000025 W ZA2007000025 W ZA 2007000025W WO 2007134343 A2 WO2007134343 A2 WO 2007134343A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- solution
- potential
- heap
- copper
- chloride
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B3/00—Extraction of metal compounds from ores or concentrates by wet processes
- C22B3/04—Extraction of metal compounds from ores or concentrates by wet processes by leaching
- C22B3/045—Leaching using electrochemical processes
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B15/00—Obtaining copper
- C22B15/0063—Hydrometallurgy
- C22B15/0065—Leaching or slurrying
- C22B15/0067—Leaching or slurrying with acids or salts thereof
- C22B15/0069—Leaching or slurrying with acids or salts thereof containing halogen
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B15/00—Obtaining copper
- C22B15/0063—Hydrometallurgy
- C22B15/0065—Leaching or slurrying
- C22B15/0067—Leaching or slurrying with acids or salts thereof
- C22B15/0071—Leaching or slurrying with acids or salts thereof containing sulfur
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B15/00—Obtaining copper
- C22B15/0095—Process control or regulation methods
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B3/00—Extraction of metal compounds from ores or concentrates by wet processes
- C22B3/04—Extraction of metal compounds from ores or concentrates by wet processes by leaching
- C22B3/06—Extraction of metal compounds from ores or concentrates by wet processes by leaching in inorganic acid solutions, e.g. with acids generated in situ; in inorganic salt solutions other than ammonium salt solutions
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P10/00—Technologies related to metal processing
- Y02P10/20—Recycling
Definitions
- This invention relates to a hydrometallurgical method for the recovery of copper from copper sulphide minerals such as bornite, chalcocite, chalcopyrite, covellite and enargite.
- Chalcopyrite is one of the most refractory copper sulphide minerals in relation to leaching in acidic ferric chloride and ferric sulphate systems at low temperature. This is exemplified by the mineral's slow leaching kinetics, which level off with time. This has been attributed to a process of "passivation", but uncertainty in regard to the mechanism still remains.
- the mineral is subjected to the process of "passivation", which is typified by the leveling-off of the leaching kinetics.
- passivation This defines the fundamental problem of oxidative dissolution of chalcopyrite in such systems.
- Many methods have been suggested to alleviate the problem of "passivation", one of which is thermophile bioleaching at elevated temperatures.
- bioleaching is carried out in a heap of low-grade, chalcopyrite-bearing ores. The process is operated in such a way that the heap temperature is raised sequentially from atmospheric to moderate thermophile or thermophile levels to achieve improved rates of chalcopyrite dissolution.
- the success of this strategy depends to a large extent on sufficient levels of available pyrite present in the ore and the successful oxidation thereof to achieve the required heat generation.
- thermophile bioleaching The recovery of copper from a low-grade, transitional and hypogene ore, which contains insufficient pyrite to generate a heap temperature for moderate thermophile, or thermophile bioleaching, remains problematic.
- the invention aims to address, at least partly, this situation.
- the use of the invention is however not confined to these circumstances and may be extended to the leaching of high-grade concentrates at elevated temperatures.
- the invention provides a method of recovering copper from a material, in a heap, that contains a copper sulphide mineral, which includes the steps of leaching the material in an acidic chloride or a mixed chloride / sulphate solution in the presence of dissolved oxygen, maintaining the mineral's surface potential below 600 mV (vs. SHE) to cause dissolution of the copper sulphide, and recovering copper from the solution.
- the potential is maintained within the range of 550 mV (vs. SHE) to 600 mV (vs. SHE) for optimum chalcopyrite leaching. The optimum potential value depends on the concentration of chloride.
- the method may be carried out at ambient or at an elevated temperature.
- the copper sulphide mineral may include bornite, chalcocite, chalcopyrite, covellite or enargite. These are non-limiting examples.
- the method of the invention may be applied to the leaching of copper sulphide in a low-grade, transitional and hypogene ore.
- the use of the invention is however not confined to these ore types and may include the leaching of low-grade supergene ores.
- the leaching may be carried out in an ore column, dump, heap or vat, collectively referred to herein, for the sake of convenience as "a heap”.
- the pH of the solution may be less than 3 and preferably is between pH 1 and pH 2.
- the pH may be controlled in any appropriate way, for example by the addition of H 2 SO 41 HCl Or HNO 3 .
- the dissolved oxygen level is preferably in excess of 1 ppm.
- the chloride concentration may be controlled at a level of 5 to 100 g/L added via HCI or any suitable chloride salt including NaCI, MgCI 2 , saline water ("salares"), sea water or chloride containing process water.
- the mineral's surface potential can be controlled by manipulating variables within the leaching system. In one approach, the ratio of Cu(II) to Cu(I) is controlled. When the method of the invention is applied to a column, dump, heap or vat leaching situation, then the ratio of Cu(II) to Cu(I) within a leach lixiviant is manipulated.
- dissolution may possibly occur in accordance with the following scheme of reactions, which comprises a sequential non- oxidative / oxidative process:
- Equation 2 is slightly more thermodynamically favourable than Equation 1 in chloride or chloride / sulphate mixed solutions, both equations are used as a starting point in a proposed reaction mechanism and result in the same intermediate oxidation reaction, as reflected in Equation 6, and in the same overall reaction, as set out in Equation 9.
- Equations 7 and 8 can be written as:
- Equation 6 is the net result of each reaction route. It is believed that, under these conditions, the oxidation of hydrogen sulphide perturbs the equilibrium portrayed in Equation 1 or 2, and results in an overall dissolution process which is given by Equation 9.
- Eh determines the mixed potential at mineral's surface that controls the mechanism of chalcopyrite dissolution.
- Figure 1 is a schematic diagram, which conceptualizes experimental observations in regard to the route of elemental sulphur formation, type (morphology) of sulphur formed, as well as the deportment of the sulphur;
- Figure 2 is a graph which depicts the effect of chloride concentration on the formal potential of the Cu(II) / Cu(I) couple;
- Figure 3(a) shows graphs of copper dissolution versus time at low solution potential
- Figure 3(b) shows graphs of the corresponding solution potential versus time profiles of
- Figure 4 shows the results of a series of tests, in which the effect of dissolved oxygen on the rate of chalcopyrite dissolution is demonstrated at low solution potential (in the absence of ferric ions) and high solution potential (in the presence of ferric ions);
- Figure 5 is a graph of copper dissolution versus time, to illustrate the defined solution potential range for optimum chalcopyrite leaching, and to highlight the results achieved when leaching under conditions in accordance with the invention
- Figure 6 is a comparative graph, which depicts copper dissolution versus time, to illustrate the importance of the presence of dissolved oxygen when leaching within a defined optimum solution potential range
- Figure 7 (a) shows a graph of copper dissolution versus time, to illustrate the feasibility of a two-stage (reductive / oxidative) leach, in a variation of the invention
- Figure 7(b) shows graphs of the corresponding solution potential versus time profile of the two-stage (reductive / oxidative) leach
- Figure 8(a) is a schematic diagram which illustrates heap leaching in which a raffinate
- Figure 8(b) is a schematic diagram which illustrates an alternative heap leaching process wherein the raffinate Eh reduction is achieved by first leaching secondary copper sulphides and thereafter using the leaching solution to leach primary copper sulphides.
- CSR chalcopyrite copper source ratio
- Test I's solution contained only acid (98% H 2 SO 4 ), distilled water and sodium chloride (NaCI) to render a low potential, whereas Test I I's solution also included ferric sulphate (Fe 2 (SO 4 ) 3 ) to render a relatively high potential.
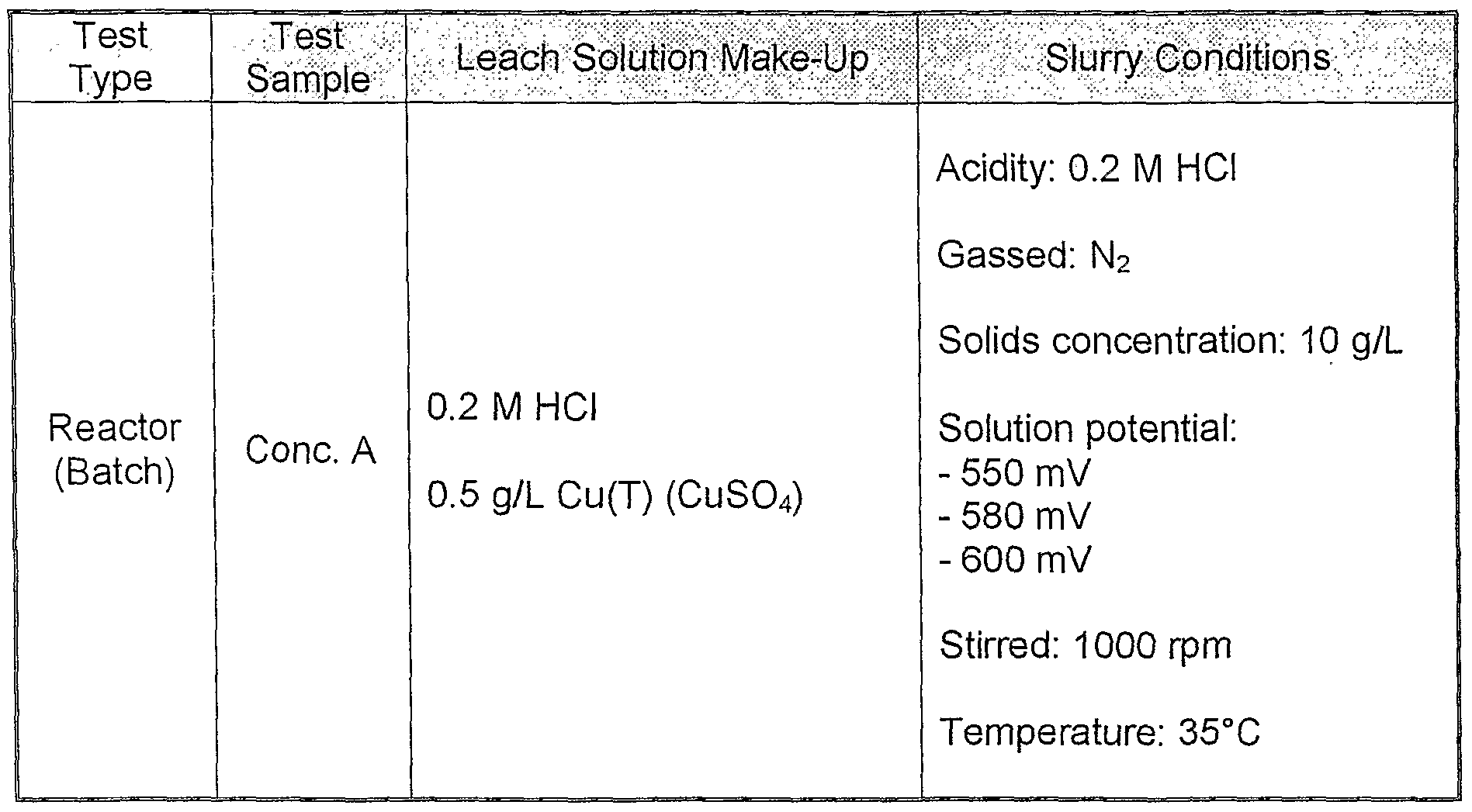
- Table 2 Test Conditions (Example 1)
- Figure 3(a) shows the copper dissolution transients of Tests I and II
- the low solution potential (0.2 M HCI) results show a marked difference in overall copper dissolution achieved in the presence of dissolved oxygen (air) against dissolution in the absence of dissolved oxygen (N 2 ). For example, 22.2% (air) vs. 11.0% (N 2 ) at 35°C, and 43.5% (air) vs. 15.4% (N 2 ) at 50 0 C, were achieved after 24 h.
- the redox sensor measured the solution potential of the slurry, which was controlled to a set point value by means of a control loop from a LabviewTM data acquisition system.
- the control loop caused the opening or closing of a solenoid valve, to allow for appropriate gas sparging.
- Each reactor was also enclosed in a temperature- controlled surround.
- the batch leaching experiments were all conducted at 35°C, and controlled at the following respective solution potentials: 540 mV, 550 mV, 580 mV, 600 mV and 620 mV.
- the tests at 550 mV, 580 mV and 600 mV were all controlled by air injection.
- Table 4 The details of the test conditions are summarized in Table 4.
- Figure 5 shows the copper dissolution transients of the five leaching tests, with 30.3% (540 mV), 73.1% (550 mV), 79.2% (580 mV), 76.2% (600 mV) and 22.5% (620 mV) copper dissolution achieved after 1000 h.
- the 550 mV test achieved 88.0% after 124O h.
- Two boundary conditions can be established, viz a lower boundary at 550 mV and an upper boundary at 600 mV, which define the solution potential range to achieve optimum chalcopyrite dissolution rates within the systems under investigation. This is depicted in Equation 13:
- Eh op timu m is the solution potential for optimum rate of chalcopyrite dissolution, in mV.
- E m i xed is the mixed potential (at the chalcopyrite mineral's surface), in mV.
- Equation 14 can be corroborated with potential measurements made with massive chalcopyrite electrodes during these and other tests.
- the following observations are made when the system is operated within the optimum solution potential range and in the presence of sufficient dissolved oxygen (more than 1 ppm), under the above conditions:
- Example 2 The importance of the presence of dissolved oxygen on the rate of chalcopyrite dissolution under conditions of low solution potential (in the absence of ferric ions) has already been illustrated in Example 2. In order to confirm this under conditions of controlled potential, more specifically within the optimum potential range of 550 mV to 600 mV, some batch leaching experiments were performed in the absence of dissolved oxygen. [0054] The tests' were executed on a fine-milled, chalcopyrite-bearing concentrate (Cone. A) in the same 1 L glass reactors as described in Example 3. The tests were all conducted at 35°C under gaseous nitrogen (saturated with water vapour), and at 550 mV, 580 mV and 600 mV. The solution potential was controlled at the desired set point by controlling the Cu(II) / Cu(I) ratio by means of electrical current. The test condition details are summarized in Table 5.
- Figure 6 shows the copper dissolution transients of these tests in comparison with those achieved at corresponding solution potentials, in the presence of dissolved oxygen (in Example 3).
- the overall copper dissolutions after a period of 1000 h are as follows:
- Example 5 The Feasibility of a Two-Stage (Reductive / Oxidative) Leach
- a batch leaching experiment was conducted to test whether chalcopyrite could also be leached successfully by the use of a variation of the aforementioned techniques of the invention.
- This constituted a two-stage leach, which included a period of initial leaching under reducing conditions of low solution potential (Stage 1), followed by leaching under oxidative conditions (Stage 2).
- Figures 7(a) and 7(b) show the copper dissolution and solution potential profiles for this test. Initially, very little copper dissolved during the period of nitrogen sparging, with only 11.1 % dissolution achieved after 139 h. The potential was as low as 500 mV over this period (Stage 1). The rate of copper dissolution increased significantly on introduction of oxygen, with an overall dissolution of 95.5% achieved after 787 h. The potential ranged from 570 mV to 591 mV over this period (Stage 2).
- FIGS. 8(a) and 8(b) are block diagram representations of the use of the method of the invention in two heap leaching processes.
- FIG. 8(a) ore 10 (ore C) is crushed (12) and agglomerated (14) with an aqueous acidic solution with a pH of below 2.0.
- This solution may be process raffinate or a solution in which the pH is controlled in any appropriate manner, for example by the addition of H 2 SO 4 , HCI or HNO 3 .
- the agglomeration (14) has two advantages viz it controls the amount of fine ore material which affects the percolation and aeration of the heap, and it solubilises acid-soluble secondary copper sulphides. This solubilization can be increased by allowing the agglomerated ore to cure before irrigating the heap.
- this solution may contain chloride at a level of 5 to 100 g/L added via suitable any chloride salt including NaCI, MgCI 2 , saline water ("salares”), sea water or chloride containing process water.
- chloride salt including NaCI, MgCI 2 , saline water ("salares"), sea water or chloride containing process water.
- the heap is leached (16) by irrigating the ore with a leach solution 18 with a pH of 2.0 or below, preferably 1.0.
- a leach solution 18 with a pH of 2.0 or below, preferably 1.0.
- the solution may contain the following: chloride at a level of 5 to 100 g/L added via HCI or any suitable chloride salt including NaCI, MgCI 2 or sea water; copper at a level of 0.05 to 10 g/L added via the corresponding chloride or sulphate salts; and iron at a level of 0 to 20 g/L added via the corresponding chloride or sulphate salts.
- the solution potential of the leach solution should preferably be maintained below 620 mV (vs. SHE), e.g. by manipulating the composition of the leach solution or via active potential reduction.
- Active potential reduction (20) may be achieved by passing the leach solution through a column containing material such as copper metal, certain types of activated carbon, by the addition reducing agents such ascorbic acid, by bubbling SO 2 or CO through the solution, or by promoting jarosite precipitation.
- Air 22 is introduced at the base of the heap to raise the dissolved oxygen level in the ore.
- the pregnant liquor solution 24 from the heap is subjected to solvent extraction 26 and copper 28 was then recovered by electrowinning 30.
- Solid residue 32 from the leached heap is disposed of in any suitable way.
- Figure 8(b) shows a modified process wherein the potential of the leach solution is reduced to the required level for primary copper sulphide leaching by first irrigating a heap consisting of secondary copper sulphide minerals 16A. The pregnant leach solution 34 of this heap is then used to irrigate a heap consisting of primary copper sulphide minerals 16B.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Metallurgy (AREA)
- Mechanical Engineering (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Life Sciences & Earth Sciences (AREA)
- Geology (AREA)
- Geochemistry & Mineralogy (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Environmental & Geological Engineering (AREA)
- General Chemical & Material Sciences (AREA)
- Electrochemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Automation & Control Theory (AREA)
- Inorganic Chemistry (AREA)
- Manufacture And Refinement Of Metals (AREA)
- Extraction Or Liquid Replacement (AREA)
Abstract
Description
Claims
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PL07784568T PL2024523T3 (en) | 2006-05-12 | 2007-05-09 | Chloride heap leaching |
| AU2007249176A AU2007249176B2 (en) | 2006-05-12 | 2007-05-09 | Chloride heap leaching |
| AT07784568T ATE538221T1 (en) | 2006-05-12 | 2007-05-09 | CHLORIDE HEAD LEACHING |
| CA2651516A CA2651516C (en) | 2006-05-12 | 2007-05-09 | Chloride heap leaching |
| AP2008004670A AP2268A (en) | 2006-05-12 | 2007-05-09 | Chloride heap leaching. |
| CN2007800262996A CN101506395B (en) | 2006-05-12 | 2007-05-09 | Chloride stack leaching |
| ES07784568T ES2379202T3 (en) | 2006-05-12 | 2007-05-09 | Leaching of chloride stacks |
| EA200802281A EA014569B1 (en) | 2006-05-12 | 2007-05-09 | Chloride heap leaching |
| EP07784568A EP2024523B1 (en) | 2006-05-12 | 2007-05-09 | Chloride heap leaching |
| US12/266,028 US8070851B2 (en) | 2006-05-12 | 2008-11-06 | Chloride heap leaching |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ZA2006/03788 | 2006-05-12 | ||
| ZA200603788 | 2006-05-12 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/266,028 Continuation US8070851B2 (en) | 2006-05-12 | 2008-11-06 | Chloride heap leaching |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2007134343A2 true WO2007134343A2 (en) | 2007-11-22 |
| WO2007134343A3 WO2007134343A3 (en) | 2008-04-03 |
Family
ID=38454574
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/ZA2007/000026 Ceased WO2007134344A1 (en) | 2006-05-12 | 2007-05-09 | Chloride tank leaching |
| PCT/ZA2007/000025 Ceased WO2007134343A2 (en) | 2006-05-12 | 2007-05-09 | Chloride heap leaching |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/ZA2007/000026 Ceased WO2007134344A1 (en) | 2006-05-12 | 2007-05-09 | Chloride tank leaching |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US8070851B2 (en) |
| EP (1) | EP2024523B1 (en) |
| CN (1) | CN101506395B (en) |
| AP (1) | AP2268A (en) |
| AR (2) | AR060898A1 (en) |
| AT (1) | ATE538221T1 (en) |
| AU (2) | AU2007249176B2 (en) |
| CA (1) | CA2651516C (en) |
| EA (1) | EA014569B1 (en) |
| ES (1) | ES2379202T3 (en) |
| PE (2) | PE20080200A1 (en) |
| PL (1) | PL2024523T3 (en) |
| WO (2) | WO2007134344A1 (en) |
| ZA (2) | ZA200809407B (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015059551A1 (en) | 2013-10-23 | 2015-04-30 | Bhp Chile Inc. | Heap leaching of copper |
| US9290827B2 (en) | 2011-03-04 | 2016-03-22 | Jx Nippon Mining & Metals Corporation | Method of leaching copper and gold from sulfide ores |
| WO2019193403A1 (en) | 2018-04-06 | 2019-10-10 | Nova Mineralis S.A. | Method for dissolving metallogenically primary copper metals obtained from ores and/or chalcopyrite concentrates that contain same |
| WO2020099966A1 (en) | 2018-11-14 | 2020-05-22 | Nova Mineralis S.A. | Solid-liquid-solid hydrometallurgical method for the solubilization of metals from sulfide copper minerals and/or concentrates |
| WO2022236437A1 (en) * | 2021-05-12 | 2022-11-17 | Corporacion Nacional Del Cobre De Chile | Sequential treatment process for the heap leaching of primary and secondary copper sulphides |
| US12421570B2 (en) | 2020-05-22 | 2025-09-23 | Anglo American Technical & Sustainability Service Ltd | Heap leaching |
| WO2025012848A3 (en) * | 2022-07-15 | 2026-01-08 | Corporacion Nacional Del Cobre De Chile | Heap leaching process with semi -autogenous heating |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20120039534A (en) * | 2009-05-26 | 2012-04-25 | 메타리치 리미티드 | Method of oxidative leaching of sulfide ores and/or concentrates |
| US9624560B2 (en) * | 2010-06-15 | 2017-04-18 | Teck Resources Limited | Recovery of residual copper from heap leach residues |
| AR082048A1 (en) * | 2010-06-30 | 2012-11-07 | Bhp Chile Inc | A METHOD FOR RECOVERING COPPER FROM A MENA CONTAINING COPPER SULFIDE MINERAL |
| US9816155B2 (en) * | 2012-08-22 | 2017-11-14 | Bhp Chile Inc. | Heap leaching method |
| FI125388B (en) * | 2013-06-07 | 2015-09-30 | Outotec Finland Oy | Process for the recovery of copper and precious metals |
| CL2014002238A1 (en) * | 2014-08-22 | 2014-10-17 | Compañia Minera Zaldivar Limitada | Leaching of copper sulphides in chloride-ferrous medium with bacteria |
| MX2017007686A (en) * | 2014-12-19 | 2017-11-10 | Smidth As F L | Methods for rapidly leaching chalcopyrite. |
| CL2015001298A1 (en) * | 2015-05-13 | 2016-05-20 | Univ Técnica Federico Santa María | Agglomeration drum and mineral agglomeration procedure for mineral pretreatment. |
| CL2016001188A1 (en) * | 2016-05-18 | 2017-02-24 | Antofagasta Minerales S A | Procedure for the improvement of copper leaching processes using calcium chloride |
| EP3529387A4 (en) | 2016-10-19 | 2020-06-10 | The University of British Columbia | METHOD FOR LEANING METAL SULFIDES WITH REAGENTS WITH THIOCARBONYL FUNCTIONAL GROUPS |
| CL2017001777A1 (en) * | 2017-07-05 | 2018-06-29 | Tecnologias Exponenciales En Minerales Spa | Method of extraction of base and precious metals by means of a pretreatment leading to the solubilization of its refractory or hypexgoldest matrices. |
| JP7206467B2 (en) * | 2017-12-21 | 2023-01-18 | ビーエイチピー チリ インコーポレイテッド | Water balance in chloride heap leaching |
| EP3728661B1 (en) * | 2017-12-21 | 2022-01-26 | Bhp Chile Inc. | Acid balance in a chloride heap leach |
| CN111826520B (en) * | 2020-06-28 | 2021-04-20 | 北京科技大学 | Using seawater-based solution to strengthen copper sulfide leaching experimental device and method |
| AU2021345381A1 (en) | 2020-09-18 | 2023-05-04 | Jetti Resources, Llc | Extracting base metals using a wetting agent and a thiocarbonyl functional group reagent |
| CN114574711B (en) * | 2022-03-10 | 2024-04-23 | 中国有色桂林矿产地质研究院有限公司 | A method for oxidative leaching of copper sulfide ore |
| WO2024178306A1 (en) | 2023-02-24 | 2024-08-29 | Cemvita Factory, Inc. | Process |
| CN116623001B (en) * | 2023-05-23 | 2025-10-31 | 中南大学 | Method for efficiently leaching chalcopyrite by reduction-oxidation two-stage method |
Family Cites Families (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3879272A (en) * | 1971-10-07 | 1975-04-22 | Duval Corp | Hydrometallurgical process for the production of copper |
| US3957602A (en) * | 1974-07-24 | 1976-05-18 | Cyprus Metallurgical Processes Corporation | Recovery of copper from chalcopyrite utilizing copper sulfate leach |
| US4276084A (en) * | 1979-10-01 | 1981-06-30 | Hazen Research, Inc. | Hydrometallurgical process for the recovery of lead |
| ZA835167B (en) * | 1982-08-10 | 1984-05-30 | Broken Hill Ass Smelters | Hydrometallurgical recovery of metal values from copper containing sulphide materials |
| CA1214043A (en) * | 1983-01-26 | 1986-11-18 | British Columbia Research Council | Biological-acid leach process |
| EP0214324B1 (en) * | 1985-09-05 | 1990-03-07 | Cheminvest A/S | Method for separation and leaching of the valuable metals in metal sulphide materials |
| US5316567A (en) * | 1992-06-19 | 1994-05-31 | Cominco Engineering Services Ltd. | Hydrometallurgical copper extraction process |
| CN1045476C (en) * | 1994-12-20 | 1999-10-06 | 康明柯工程服务有限公司 | Chloride-Assisted Hydrometallurgical Copper Extraction |
| US6277341B1 (en) * | 1997-03-03 | 2001-08-21 | Mintek | Process for the rapid leaching of chalcopyrite in the absence of catalysts |
| US6569391B1 (en) * | 1998-02-17 | 2003-05-27 | Ingesol Ltda. | Heap leaching copper ore using sodium nitrate |
| AU749257B2 (en) * | 1998-07-24 | 2002-06-20 | Intec Hellyer Metals Pty Ltd | Processing copper sulphide ores |
| US6802888B2 (en) * | 1998-12-14 | 2004-10-12 | Geobiotics, Llc | High temperature heap bioleaching process |
| US7189527B2 (en) * | 1999-09-03 | 2007-03-13 | Bioheap Limited | Bacterial oxidation of sulphide ores and concentrates |
| AUPR855201A0 (en) * | 2001-10-29 | 2001-11-29 | Technological Resources Pty Limited | Recovery of copper from chalcopyrite |
| FI113667B (en) | 2002-04-23 | 2004-05-31 | Outokumpu Oy | Method for recovering gold |
| JP2003328051A (en) * | 2002-05-17 | 2003-11-19 | Sumitomo Metal Mining Co Ltd | Leaching treatment method of copper sulfide ore containing chalcopyrite |
| JP4352823B2 (en) * | 2002-11-18 | 2009-10-28 | 住友金属鉱山株式会社 | Method for refining copper raw materials containing copper sulfide minerals |
| CA2432743C (en) * | 2002-11-26 | 2010-12-14 | Minera Michilla S.A. | Non-biochemical method to heap leach copper concentrates |
| CA2478516C (en) * | 2003-09-30 | 2007-12-11 | Jaguar Nickel Inc. | A process for the recovery of value metals from base metal sulfide ores |
| RU2373298C2 (en) | 2004-06-03 | 2009-11-20 | Дзе Юниверсити Оф Бритиш Коламбиа | Copper concentrate leaching method |
| FI117389B (en) | 2004-12-28 | 2006-09-29 | Outokumpu Oy | A process for hydrometallurgical treatment of a sulphide concentrate containing several precious metals |
| FI117941B (en) * | 2005-10-13 | 2007-04-30 | Outokumpu Technology Oyj | A process for dissolving metal sulfide minerals |
-
2007
- 2007-05-09 AP AP2008004670A patent/AP2268A/en active
- 2007-05-09 WO PCT/ZA2007/000026 patent/WO2007134344A1/en not_active Ceased
- 2007-05-09 WO PCT/ZA2007/000025 patent/WO2007134343A2/en not_active Ceased
- 2007-05-09 AU AU2007249176A patent/AU2007249176B2/en active Active
- 2007-05-09 AU AU2007249177A patent/AU2007249177B2/en not_active Ceased
- 2007-05-09 CN CN2007800262996A patent/CN101506395B/en active Active
- 2007-05-09 ES ES07784568T patent/ES2379202T3/en active Active
- 2007-05-09 CA CA2651516A patent/CA2651516C/en active Active
- 2007-05-09 AT AT07784568T patent/ATE538221T1/en active
- 2007-05-09 EA EA200802281A patent/EA014569B1/en not_active IP Right Cessation
- 2007-05-09 EP EP07784568A patent/EP2024523B1/en active Active
- 2007-05-09 PL PL07784568T patent/PL2024523T3/en unknown
- 2007-05-10 PE PE2007000567A patent/PE20080200A1/en active IP Right Grant
- 2007-05-11 PE PE2007000572A patent/PE20080202A1/en not_active Application Discontinuation
- 2007-05-11 AR ARP070102049A patent/AR060898A1/en active IP Right Grant
- 2007-05-11 AR ARP070102050A patent/AR060899A1/en active IP Right Grant
-
2008
- 2008-11-04 ZA ZA200809407A patent/ZA200809407B/en unknown
- 2008-11-04 ZA ZA200809406A patent/ZA200809406B/en unknown
- 2008-11-06 US US12/266,028 patent/US8070851B2/en active Active
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9290827B2 (en) | 2011-03-04 | 2016-03-22 | Jx Nippon Mining & Metals Corporation | Method of leaching copper and gold from sulfide ores |
| WO2015059551A1 (en) | 2013-10-23 | 2015-04-30 | Bhp Chile Inc. | Heap leaching of copper |
| US10041143B2 (en) | 2013-10-23 | 2018-08-07 | Bhp Chile Inc. | Heap leaching of copper |
| WO2019193403A1 (en) | 2018-04-06 | 2019-10-10 | Nova Mineralis S.A. | Method for dissolving metallogenically primary copper metals obtained from ores and/or chalcopyrite concentrates that contain same |
| WO2020099966A1 (en) | 2018-11-14 | 2020-05-22 | Nova Mineralis S.A. | Solid-liquid-solid hydrometallurgical method for the solubilization of metals from sulfide copper minerals and/or concentrates |
| WO2020099912A1 (en) | 2018-11-14 | 2020-05-22 | Nova Mineralis S.A. | Solid-liquid-solid method for the solubilisation of copper minerals and concentrates, independent of the redox potential and with low consumption of water and acid |
| US12421570B2 (en) | 2020-05-22 | 2025-09-23 | Anglo American Technical & Sustainability Service Ltd | Heap leaching |
| WO2022236437A1 (en) * | 2021-05-12 | 2022-11-17 | Corporacion Nacional Del Cobre De Chile | Sequential treatment process for the heap leaching of primary and secondary copper sulphides |
| ES2957847R1 (en) * | 2021-05-12 | 2025-05-20 | Corporacion Nac Del Cobre De Chile | SEQUENTIAL TREATMENT PROCESS FOR HEAP LEACHING OF PRIMARY AND SECONDARY COPPER SULFIDES |
| WO2025012848A3 (en) * | 2022-07-15 | 2026-01-08 | Corporacion Nacional Del Cobre De Chile | Heap leaching process with semi -autogenous heating |
Also Published As
| Publication number | Publication date |
|---|---|
| EA200802281A1 (en) | 2009-06-30 |
| PL2024523T3 (en) | 2012-05-31 |
| WO2007134344A1 (en) | 2007-11-22 |
| US20090173188A1 (en) | 2009-07-09 |
| CN101506395A (en) | 2009-08-12 |
| EP2024523A2 (en) | 2009-02-18 |
| ZA200809407B (en) | 2009-10-28 |
| AR060898A1 (en) | 2008-07-23 |
| AR060899A1 (en) | 2008-07-23 |
| EA014569B1 (en) | 2010-12-30 |
| CA2651516C (en) | 2012-09-18 |
| CN101506395B (en) | 2012-05-30 |
| EP2024523B1 (en) | 2011-12-21 |
| ES2379202T3 (en) | 2012-04-23 |
| AU2007249177A1 (en) | 2007-11-22 |
| CA2651516A1 (en) | 2007-11-22 |
| WO2007134343A3 (en) | 2008-04-03 |
| ZA200809406B (en) | 2010-03-31 |
| AU2007249176A1 (en) | 2007-11-22 |
| AU2007249176B2 (en) | 2011-09-08 |
| US8070851B2 (en) | 2011-12-06 |
| PE20080200A1 (en) | 2008-04-10 |
| AP2268A (en) | 2011-08-03 |
| ATE538221T1 (en) | 2012-01-15 |
| AU2007249177B2 (en) | 2011-01-20 |
| PE20080202A1 (en) | 2008-04-10 |
| AP2008004670A0 (en) | 2008-12-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8070851B2 (en) | Chloride heap leaching | |
| Lu et al. | The effect of chloride ions on the dissolution of chalcopyrite in acidic solutions | |
| Molleman et al. | The treatment of copper–gold ores by ammonium thiosulfate leaching | |
| Nazari et al. | Enhancing the kinetics of chalcopyrite leaching in the Galvanox™ process | |
| Mwase et al. | A conceptual flowsheet for heap leaching of platinum group metals (PGMs) from a low-grade ore concentrate | |
| Akbari et al. | Recovery of copper from a mixture of printed circuit boards (PCBs) and sulphidic tailings using bioleaching and solvent extraction processes | |
| US20110129891A1 (en) | Process for Controlled Homogeneous Acid Leaching | |
| US12503746B2 (en) | Oxidative heap leaching of base metals | |
| AU2007203580A1 (en) | Recovery of molybdenum from molybdenum bearing sulfide materials by bioleaching in the presence of iron | |
| Rademan et al. | The leaching characteristics of Ni–Cu matte in the acid–oxygen pressure leach process at Impala Platinum | |
| Nabizadeh et al. | Dissolution study of chalcopyrite concentrate in oxidative ammonia/ammonium carbonate solutions at moderate temperature and ambient pressure | |
| Sadeghieh et al. | Effect of water salinity on the bioleaching of copper, nickel and cobalt from the sulphidic tailing of Golgohar Iron Mine, Iran | |
| CA3218592A1 (en) | Sequential treatment process for primary and secondary copper sulphide heap leaching | |
| Esmailbagi et al. | Microbial assisted galvanic leaching of chalcopyrite concentrate in continuously stirred bioreactors | |
| Muñoz et al. | Silver-catalyzed bioleaching of low-grade copper ores. Part II: Stirred tank tests | |
| Sampson et al. | Testing the ability of a low grade sphalerite concentrate to achieve autothermality during biooxidation heap leaching | |
| van Hille et al. | Investigating heap bioleaching: Effect of feed iron concentration on bioleaching performance | |
| WO2000068445A1 (en) | Base metal recovery | |
| US12492449B2 (en) | Oxidative bioleaching of base metals | |
| EP3882365A1 (en) | Solid-liquid-solid method for the solubilisation of copper minerals and concentrates, independent of the redox potential and with low consumption of water and acid | |
| WO2025259816A1 (en) | Method of metal recovery | |
| JP3790152B2 (en) | Methods for leaching copper from copper sulfide ores | |
| Taghi Nazari | Enhancing the kinetics of pyrite catalyzed leaching of chalcopyrite | |
| Neale | Bioleaching technology in minerals processing | |
| Ghazali Jahromi | Atmospheric Leaching of Enargite in the Presence of Carbon Based Catalyst |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780026299.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07784568 Country of ref document: EP Kind code of ref document: A2 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2651516 Country of ref document: CA Ref document number: 2007249176 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007784568 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2007249176 Country of ref document: AU Date of ref document: 20070509 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200802281 Country of ref document: EA |