WO2009107731A1 - 排ガス処理方法及び装置 - Google Patents
排ガス処理方法及び装置 Download PDFInfo
- Publication number
- WO2009107731A1 WO2009107731A1 PCT/JP2009/053575 JP2009053575W WO2009107731A1 WO 2009107731 A1 WO2009107731 A1 WO 2009107731A1 JP 2009053575 W JP2009053575 W JP 2009053575W WO 2009107731 A1 WO2009107731 A1 WO 2009107731A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- exhaust gas
- mercury
- combustion exhaust
- halogenating agent
- catalyst
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
- B01D53/86—Catalytic processes
- B01D53/8643—Removing mixtures of carbon monoxide or hydrocarbons and nitrogen oxides
- B01D53/8656—Successive elimination of the components
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/46—Removing components of defined structure
- B01D53/48—Sulfur compounds
- B01D53/50—Sulfur oxides
- B01D53/501—Sulfur oxides by treating the gases with a solution or a suspension of an alkali or earth-alkali or ammonium compound
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/46—Removing components of defined structure
- B01D53/64—Heavy metals or compounds thereof, e.g. mercury
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
- B01D53/86—Catalytic processes
- B01D53/8621—Removing nitrogen compounds
- B01D53/8625—Nitrogen oxides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
- B01D53/86—Catalytic processes
- B01D53/869—Multiple step processes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2251/00—Reactants
- B01D2251/20—Reductants
- B01D2251/206—Ammonium compounds
- B01D2251/2062—Ammonia
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2251/00—Reactants
- B01D2251/30—Alkali metal compounds
- B01D2251/304—Alkali metal compounds of sodium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2251/00—Reactants
- B01D2251/40—Alkaline earth metal or magnesium compounds
- B01D2251/404—Alkaline earth metal or magnesium compounds of calcium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/10—Noble metals or compounds thereof
- B01D2255/102—Platinum group metals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/207—Transition metals
- B01D2255/20707—Titanium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/207—Transition metals
- B01D2255/20715—Zirconium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/207—Transition metals
- B01D2255/20723—Vanadium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/207—Transition metals
- B01D2255/20769—Molybdenum
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/207—Transition metals
- B01D2255/20776—Tungsten
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/209—Other metals
- B01D2255/2092—Aluminium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/30—Silica
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/50—Zeolites
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/30—Sulfur compounds
- B01D2257/302—Sulfur oxides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/40—Nitrogen compounds
- B01D2257/404—Nitrogen oxides other than dinitrogen oxide
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/60—Heavy metals or heavy metal compounds
- B01D2257/602—Mercury or mercury compounds
Definitions
- the present invention relates to an exhaust gas treatment method and apparatus for removing nitrogen oxides, sulfur oxides, and mercury from exhaust gas discharged from a combustion apparatus such as a boiler.
- Mercury present in the flue gas contains metallic mercury that is insoluble in water and mercury chloride that is soluble in water, and is difficult to dissolve in water when mercury is in the form of metallic mercury.
- the mercury removal rate by the wet desulfurization apparatus is low.
- mercury can be removed because HgCl or HgCl 2 of the exhaust gas is dissolved in water by gas-liquid contact with a wet desulfurization apparatus. That is, if metal mercury can be converted to mercury chloride in the presence of a catalyst such as a denitration catalyst, mercury can be removed by a downstream desulfurization apparatus.
- FIG. 3 An example of a conventional exhaust gas treatment method using this will be described with reference to FIG.
- a supply point 21 for injecting a mercury chlorinating agent from 40 into the exhaust gas is installed.
- the exhaust gas from the boiler 10 is introduced into the reducing denitration device 60.
- the exhaust gas into which NH 3 and HCl have been injected undergoes a reaction between NH 3 and NO x in the reductive denitration device 60, and at the same time, metal Hg is oxidized to HgCl 2 in the presence of HCl.
- the wet desulfurization apparatus 100 removes HgCl 2 simultaneously with the removal of SO 2 in the exhaust gas.
- excess HCl is contained in the exhaust gas exiting the reducing denitration device 60, but it is absorbed by the alkaline aqueous solution such as lime milk by the desulfurization device 100, so that it is not discharged from the chimney.
- the above method proposes a system that sprays a chlorinating agent such as HCl in the upstream of the denitration catalyst, oxidizes (chlorinates) mercury on the catalyst, and removes mercury in the downstream wet-type desulfurization equipment.
- an object of the present invention is to provide an exhaust gas treatment method and apparatus capable of reducing the amount of mercury halogenating agent such as highly corrosive mercury chlorinating agent while maintaining high mercury removal efficiency in exhaust gas treatment. Is an offer.
- the present invention provides a method for treating boiler flue gas containing NO x , SO x and mercury, the step of adding a mercury halogenating agent and ammonia to the flue gas, and the mercury halogenation
- a process in which the combustion exhaust gas to which the agent and ammonia are added is brought into contact with the CO and HC oxidation catalyst, and the combustion exhaust gas in contact with the CO and HC oxidation catalyst is reductively denitrated in the presence of a solid catalyst and the mercury metal is mercury halide.
- a step of wet desulfurizing the combustion exhaust gas subjected to reductive denitration with an alkali absorbing solution and removing the mercury halide with the alkali absorbing solution are examples of wet desulfurizing the combustion exhaust gas subjected to reductive denitration with an alkali absorbing solution and removing the mercury halide with the alkali absorbing solution.
- Another aspect of the present invention is a method for treating boiler flue gas containing NO x , SO x and mercury, the step of contacting the flue gas with CO, HC oxidation catalyst, and the CO, HC oxidation catalyst Adding a mercury halogenating agent and ammonia to the combustion exhaust gas in contact with the catalyst, reducing and denitrating the combustion exhaust gas containing the mercury halogenating agent and ammonia in the presence of a solid catalyst, and oxidizing metallic mercury to mercury halide And a step of performing wet desulfurization of the reductive denitrated combustion exhaust gas with an alkali absorbing liquid and removing the mercury halide with the alkali absorbing liquid.
- the mercury halogenating agent is preferably ammonium chloride or HCl.
- the CO and HC oxidation catalyst may be Pt, Ru, Rh, Pd, Ir on the support using at least one selected from the group consisting of TiO 2 , SiO 2 , ZrO 2 , Al 2 O 3 and zeolite. , Au, Ag, V, W, Mo, Ni, Co, Fe, Cr, Cu, and Mn, a catalyst having at least one selected from the group consisting of Mn as an active component is preferable.
- the present invention is an apparatus for treating a combustion exhaust gas containing NO x , SO x and mercury, the mercury halogenating agent injection device for adding a mercury halogenating agent to the combustion exhaust gas, and the combustion
- An ammonia injection device for injecting ammonia into the exhaust gas, a CO and HC oxidation catalyst for oxidizing unburned components in the combustion exhaust gas, a reduction denitration device for reducing and denitrating the combustion exhaust gas in the presence of a solid catalyst, and an alkali absorbent
- a wet desulfurization apparatus that performs wet desulfurization in order.
- An exhaust gas treatment apparatus is an apparatus for treating combustion exhaust gas containing NO x , SO x and mercury, and CO, HC oxidation catalyst for oxidizing unburned components in the combustion exhaust gas,
- a mercury halogenating agent injection device for adding a mercury halogenating agent to the combustion exhaust gas
- an ammonia injection device for injecting ammonia into the combustion exhaust gas
- a reduction denitration device for reducing and denitrating the combustion exhaust gas in the presence of a solid catalyst
- the exhaust gas treatment method of the present invention after removing unburned components contained in the exhaust gas by the CO and HC oxidation catalyst, reductive denitration is performed in the presence of the solid catalyst, thereby the mercury halogenation reaction of CO and HC. Inhibition can be avoided, and a mercury halogenation reaction can be efficiently performed with a small amount of a mercury halogenating agent. Therefore, highly corrosive HCl used as a mercury halogenating agent can be reduced, and the concern about flue corrosion can be improved. Furthermore, the energy used for the heat source or steam required for vaporizing HCl can be suppressed.
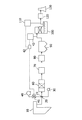
- FIG. 1 schematically shows an embodiment of an exhaust gas treatment apparatus.
- a boiler 10 in order of the flow path of the exhaust gas, a boiler 10, an NH 3 supply location 20 into which NH 3 is injected, and a mercury halogenating agent injection into which a mercury halogenating agent is injected.
- Location 21, CO, HC oxidation catalyst 50, denitration device 60, air heater 70, heat recovery device 80, dust collector 90, desulfurization device 100, reheater 120, and chimney 130 are installed.
- the NH 3 supply portion 20, NH 3 and tank 30 is connected to the mercury halogenating agent injection point 21, a mercury halogenating agent tank 40 is connected.
- a mercury halogenating agent flow rate control valve 41 for controlling the flow rate of the mercury halogenating agent is provided between the mercury halogenating agent injection site 21 and the mercury halogenating agent tank 40.
- a mercury halogenating agent monitor for detecting the concentration of the mercury halogenating agent in the gas is installed between the dust collector 90 and the desulfurization apparatus 100.
- An Hg monitor that detects the Hg concentration in the gas is installed between the desulfurization apparatus 100 and the reheater 120.
- a mercury halogenating agent supply amount control device that receives detection data of the mercury halide monitor and the Hg monitor and transmits the supply amount of the mercury halogenating agent to the flow control valve 41 is installed.
- the exhaust gas to be treated in the present invention is a boiler 10 exhaust gas from a thermal power plant or factory that burns fuel containing sulfur, mercury, etc. of coal, heavy oil, etc.
- Heating furnace exhaust gas from factories, oil refining factories, petrochemical factories, etc. may be used, and those having low NO x concentration, containing carbon dioxide, oxygen, SO x , dust, or moisture and having large exhaust gas emissions are preferred.
- the injection of ammonia from the NH 3 tank 30 into the exhaust gas is performed by a conventional method.
- the NH 3 injection means one constituted by an NH 3 injection pipe and a plurality of spray nozzles is used.
- an injection method there is a method in which NH 3 is vaporized and then diluted by adding air, an inert gas, water vapor or the like to the NH 3 . At this time, it is effective to arrange the nozzles so that the reducing agent flows uniformly in the downstream catalyst, and in some cases, a plurality of nozzles are arranged in a direction perpendicular to the gas flow.
- What forms a water-soluble metal compound is not limited to a mercury chlorinating agent, but can be halogen such as bromine other than chlorine-based compounds, bromine compounds such as HBr, Br 2 , and CaBr 2 . Therefore, in the present invention, bromine-based mercury halogenating agents such as bromine, HBr, Br 2 , and CaBr 2 can also be applied as the additive injected to oxidize mercury.
- the mercury halogenating agent in the mercury halogenating agent tank 40 is a chlorinating agent in which mercury in exhaust gas reacts with the mercury chlorinating agent in the presence of the catalyst to generate HgCl 2 and / or HgCl.
- HCl ammonium chloride
- chlorine hypochlorous acid
- ammonium hypochlorite chlorous acid
- ammonium chlorite chloric acid
- ammonium chlorate chloric acid
- ammonium chlorate perchloric acid
- ammonium perchlorate and other amines of the above acids
- salts and other salts include salts and other salts.
- the amount of the mercury halogenating agent added to the exhaust gas should be a stoichiometric amount or slightly more than the slightly water-soluble mercury such as metallic mercury.
- the concentration of the added mercury halogenating agent is 1000 ppm or less with respect to the exhaust gas, and is actually about 10 to 500 ppm.
- HCl When HCl is used as the mercury halogenating agent, hydrogen chloride or hydrochloric acid may be used as a chemical agent.
- concentration of hydrochloric acid is not particularly limited, and examples thereof include concentrated hydrochloric acid to dilute hydrochloric acid of about 5%.
- a conventional metering pump for chemicals may be used, or HCl may be sprayed or vaporized using a spray grid or a vaporizer and sprayed.
- an aqueous salt solution for adding a salt such as ammonium chloride.
- a solid the compound that evaporates (including sublimation) in the exhaust gas.
- the mercury halogenating agent may be added before or after the addition of ammonia to the exhaust gas.
- the CO and HC oxidation catalyst 50 it is possible to divert an NH 3 decomposition catalyst conventionally used (Japanese Patent Laid-Open No. 2004-237244). That is, the CO and HC oxidation catalyst 50 has at least one selected from the group consisting of TiO 2 , SiO 2 , ZrO 2 , Al 2 O 3 and zeolite as a support, and Pt, Ru, Rh, Pd on the support. , Ir, V, W, Mo, Ni, Co, Fe, Cr, Cu, and a catalyst having at least one selected from the group consisting of Mn as an active component.
- titanium oxide or silicon oxide as a carrier
- a composite oxide containing TiO 2 or SiO 2 as a carrier.
- the activity of the catalyst in which noble metals such as Pt, Ru, Pd, and Ir, and oxides such as vanadium, tungsten, and molybdenum are supported as active metal species is good.
- a composite oxide Ti oxide in order to increase the specific surface area and solid acid amount of the CO and HC oxidation catalyst, a composite oxide Ti oxide can be used as a support.
- the metal forming the composite oxide of Ti include silicon (Si), zirconium (Zr), aluminum (Al), and tungsten (W).
- composite oxides such as Ti and Si, Ti and Zr, Ti and Al, and Ti and W can be used. Since any of these composite oxides hardly forms a sulfate, a stable structure can be maintained, and the specific surface area and the amount of solid acid can be increased.
- a ternary composite oxide such as Ti and Si + Zr, Ti and Si + W, Ti and Si + Al, Ti and Zr + Al, Ti and Zr + W, or the like can be used.
- the carrier as described above is selected from the group consisting of Pt, Ru, Rh, Pd, Ir, V, W, Mo, Ni, Co, Fe, Cr, Cu and Mn. At least one kind can be supported and used as an active ingredient.
- the composition ratio of the catalyst used in the present invention is not particularly limited, but as an example, the active component is V, W, Mo, Ni with respect to 100 parts by weight of a support made of a kind of oxide or composite oxide.
- Non-metallic elements such as Co, Fe, Cr, Cu and Mn have oxides of 0.5 to 20 parts by weight as oxides, and noble metal systems of Pt, Ru, Rh, Pd, Ir, Au, and Ag.
- a composition of 0.01 to 2 parts by weight as the metal is preferable. More specifically, a composition in which the active ingredient is 0.02 part by weight of Pt with respect to 100 parts by weight of the TiO 2 carrier (JP 2004-237244 A).
- a dual function catalyst described in Japanese Patent Application No. 2007-215818 is also applicable.
- the CO and HC oxidation catalyst 50 can be arbitrarily selected according to the system configuration.
- any shape integrally formed such as a pellet, plate, cylinder, corrugate, or honeycomb can be used. It can be a shape.
- the solid catalyst used in the denitration apparatus 60 for example, a metal oxide such as V, W, Mo or the like supported on titania, silica, zirconia and their composite oxides and / or zeolite as a support is used. Can do.
- a honeycomb-shaped catalyst, a stack of these, a catalyst filled with a granular catalyst, or the like is used.
- the desulfurization apparatus 100 may be a wet desulfurization apparatus generally used in flue gas treatment or a desulfurization apparatus in which a cooling tower is installed in front of an absorption tower, and is not particularly limited, and a normal wet desulfurization apparatus can be used.
- the absorbent used for wet desulfurization include aqueous solutions (alkali absorbent) of absorbents such as calcium carbonate, calcium oxide, calcium hydroxide, sodium carbonate, and caustic soda.
- the heat recovery device 80 and the reheater 120 are configured by a gas heater that exchanges heat energy using a heat medium as a medium.
- the heat recovery device 80 and the reheater 120 are only required to cool and heat the temperature of the exhaust gas, and may be separate systems or gas gas heaters that directly exchange heat.
- NH 3 and a mercury halogenating agent such as HCl are supplied to the exhaust gas from the boiler 10 from the NH 3 tank 30 and the mercury halogenating agent tank 40.
- the exhaust gas is introduced into a device having a CO and HC oxidation catalyst, and unburned CO and HC in the exhaust gas are oxidized.
- An example of the oxidation reaction is shown below.
- HC exists in the exhaust gas in a state such as HCHO, C 2 H 4 , and C 6 H 6 .
- the exhaust gas in which the unburned matter is oxidized is introduced into the denitration device 60.
- the reaction between NH 3 and NO x is performed, and at the same time, metal Hg is oxidized to HgCl 2 in the presence of HCl. Examples of these reactions are shown below. 4NO + 4NH 3 + O 2 ⁇ 4N 2 + 6H 2 O Hg + 2HCl + 1 / 2O 2 ⁇ HgCl 2 + H 2 O
- the exhaust gas entering the denitration device contains an excessive mercury halogenating agent added by HCl, HBr spray device or NH 4 Cl supply device for oxidizing mercury. .
- HCl HCl
- HBr spray device NH 4 Cl supply device for oxidizing mercury.
- NH 4 Cl supply device for oxidizing mercury.
- the addition of a mercury halogenating agent used to oxidize mercury contained in exhaust gas can be suppressed to a very small amount, and as a result, smoke caused by highly corrosive HCl. Can improve road corrosion concerns.
- FIG. 2 shows another embodiment.
- the same components as those in FIG. 1 are denoted by the same reference numerals, and description thereof is omitted.
- the CO and HC oxidation catalyst 50 is installed upstream of the denitration device 60 and in front of the NH 3 and mercury halogenating agent supply locations 20 and 21.
- the unburned matter in the exhaust gas discharged from the boiler 10 is oxidized by the CO and HC oxidation catalyst 50 without being influenced by NH 3 and a mercury halogenating agent such as HCl supplied thereafter. It becomes possible to make it.
- the exhaust gas containing NH 3 and HCl supplied thereafter can efficiently perform the oxidation reaction of mercury contained in the exhaust gas in the denitration catalyst.
- dust is removed by the dust collector 90 through the air heater 70 and the heat recovery unit 80, and then, simultaneously with the removal of SO 2 in the exhaust gas by the wet desulfurization apparatus 100, the removal of HgCl 2 as in the embodiment of FIG. Is done.
- a conventionally used SO 3 reduction catalyst can be installed between the NH 3 and mercury halogenating agent supply locations 20 and 21 and the denitration catalyst 60. Accordingly, it is possible to advance the oxidation reaction of mercury contained in the exhaust gas stream on the denitration catalyst after performing the oxidation treatment of CO and HC without increasing SO 3 in the exhaust gas. Further, since the SO 3 reduction catalyst can also oxidize CO and HC, the CO and HC oxidation catalyst 50 in FIG. 1 can be a catalyst having an SO 3 reduction function. As an example, a catalyst described in JP-A-2006-136869 can be mentioned.
Landscapes
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Environmental & Geological Engineering (AREA)
- Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Analytical Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Exhaust Gas Treatment By Means Of Catalyst (AREA)
- Catalysts (AREA)
- Treating Waste Gases (AREA)
Abstract
Description
20 NH3供給箇所
21 水銀ハロゲン化剤供給箇所
30 NH3タンク
40 水銀ハロゲン化剤タンク
41 流量制御弁
43 水銀ハロゲン化剤供給量制御装置
44 水銀ハロゲン化剤モニター
50 CO、HC酸化触媒
60 脱硝触媒
70 エアヒータ80 熱回収器
90 集塵機
100 脱硫装置
110 Hgモニター
120 再加熱器
130 煙突
CO+1/2O2→CO2
HC+O2→CO2+H2O
4NO+4NH3+O2→4N2+6H2O
Hg+2HCl+1/2O2→HgCl2+H2O
HgCl2+CO+H2O→Hg+2HCl+CO2
HgCl2+HC+H2O+O2→Hg+2HCl+CO2
SO2+1/2O2→SO3
Claims (7)
- NOx、SOx及び水銀を含むボイラ燃焼排ガスを処理する方法であって、前記燃焼排ガスに水銀ハロゲン化剤及びアンモニアを加える工程と、この水銀ハロゲン化剤とアンモニアが添加された燃焼排ガスをCO、HC酸化触媒に接触させる工程と、前記CO、HC酸化触媒に接触させた燃焼排ガスを固体触媒の存在下に還元脱硝するとともに金属水銀をハロゲン化水銀に酸化する工程と、還元脱硝された燃焼排ガスをアルカリ吸収液により湿式脱硫するとともに、前記アルカリ吸収液により前記ハロゲン化水銀を除去する工程とを含む排ガス処理方法。
- NOx、SOx及び水銀を含むボイラ燃焼排ガスを処理する方法であって、前記燃焼排ガスをCO、HC酸化触媒に接触させる工程と、前記CO、HC酸化触媒に接触させた燃焼排ガスに水銀ハロゲン化剤及びアンモニアを加える工程と、この水銀ハロゲン化剤とアンモニアが添加された燃焼排ガスを固体触媒の存在下に還元脱硝するとともに金属水銀をハロゲン化水銀に酸化する工程と、還元脱硝された燃焼排ガスをアルカリ吸収液により湿式脱硫するとともに、前記アルカリ吸収液により前記ハロゲン化水銀を除去する工程とを含む排ガス処理方法。
- 水銀ハロゲン化物及びアンモニアの添加後、還元脱硝の前に排ガスをSO3還元機能を持つCO、HC酸化触媒に接触させる工程をさらに含む、請求項1又は2に記載の排ガス方法。
- 水銀ハロゲン化剤が塩化アンモニウム又はHCl、Cl2である請求項1~3のいずれか一項に記載の排ガス処理方法。
- CO、HC酸化触媒が、TiO2、SiO2、ZrO2、Al2O3およびゼオライトからなる群より選ばれる少なくとも1種類を担体として、前記担体上にPt、Ru、Rh、Pd、Ir、Au、Ag、V、W、Mo、Ni、Co、Fe、Cr、CuおよびMnからなる群より選ばれる少なくとも1種類を活性成分として担持した触媒である請求項1~4のいずれか一項に記載の排ガス処理方法。
- NOx、SOx及び水銀を含む燃焼排ガスを処理する装置であって、前記燃焼排ガスに水銀ハロゲン化剤を添加する水銀ハロゲン化剤注入装置および前記燃焼排ガスにアンモニアを注入するアンモニア注入装置と、前記燃焼排ガス中の未燃分を酸化するCO、HC酸化触媒と、前記燃焼排ガスを固体触媒の存在下に還元脱硝する還元脱硝装置と、アルカリ吸収液により湿式脱硫する湿式脱硫装置とを順に備えた排ガス処理装置。
- NOx、SOx及び水銀を含む燃焼排ガスを処理する装置であって、前記燃焼排ガス中の未燃分を酸化するCO、HC酸化触媒と、前記燃焼排ガスに水銀ハロゲン化剤を添加する水銀ハロゲン化剤注入装置および前記燃焼排ガスにアンモニアを注入するアンモニア注入装置と、前記燃焼排ガスを固体触媒の存在下に還元脱硝する還元脱硝装置と、アルカリ吸収液により湿式脱硫する湿式脱硫装置とを順に備えた排ガス処理装置。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0904317-9A BRPI0904317A2 (pt) | 2008-02-28 | 2009-02-26 | Método e equipamento para tratamento de gás de exaustão |
| ES09716136.8T ES2588371T3 (es) | 2008-02-28 | 2009-02-26 | Proceso y equipo para el tratamiento de gases de escape |
| KR1020107003685A KR101126840B1 (ko) | 2008-02-28 | 2009-02-26 | 배기가스 처리 방법 및 장치 |
| US12/670,871 US8420034B2 (en) | 2008-02-28 | 2009-02-26 | Method and apparatus for treating exhaust gas |
| CN2009801006619A CN101932376B (zh) | 2008-02-28 | 2009-02-26 | 废气的处理方法和装置 |
| EP09716136.8A EP2248574B1 (en) | 2008-02-28 | 2009-02-26 | Process and equipment for the treatment of exhaust gas |
| CA2694862A CA2694862C (en) | 2008-02-28 | 2009-02-26 | Method and apparatus for treating exhaust gas |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008-047460 | 2008-02-28 | ||
| JP2008047460A JP5319934B2 (ja) | 2008-02-28 | 2008-02-28 | 排ガス処理方法及び装置 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009107731A1 true WO2009107731A1 (ja) | 2009-09-03 |
Family
ID=41016118
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/053575 Ceased WO2009107731A1 (ja) | 2008-02-28 | 2009-02-26 | 排ガス処理方法及び装置 |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US8420034B2 (ja) |
| EP (1) | EP2248574B1 (ja) |
| JP (1) | JP5319934B2 (ja) |
| KR (1) | KR101126840B1 (ja) |
| CN (1) | CN101932376B (ja) |
| BR (1) | BRPI0904317A2 (ja) |
| CA (1) | CA2694862C (ja) |
| ES (1) | ES2588371T3 (ja) |
| PL (1) | PL2248574T3 (ja) |
| RU (1) | RU2429900C1 (ja) |
| WO (1) | WO2009107731A1 (ja) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011120981A (ja) * | 2009-12-09 | 2011-06-23 | Babcock Hitachi Kk | 酸素燃焼方式の排ガス処理装置と該排ガス処理装置の運用方法 |
| CN105312047A (zh) * | 2014-07-18 | 2016-02-10 | 中国钢铁股份有限公司 | 脱硝催化剂之制造方法及其应用 |
| US11065578B2 (en) * | 2012-04-11 | 2021-07-20 | ADA-ES, Inc. | Control of wet scrubber oxidation inhibitor and byproduct recovery |
| US11118127B2 (en) | 2011-05-13 | 2021-09-14 | ADA-ES, Inc. | Process to reduce emissions of nitrogen oxides and mercury from coal-fired boilers |
| US11213787B2 (en) | 2010-02-04 | 2022-01-04 | ADA-ES, Inc. | Method and system for controlling mercury emissions from coal-fired thermal processes |
| US11298657B2 (en) | 2010-10-25 | 2022-04-12 | ADA-ES, Inc. | Hot-side method and system |
| US11384304B2 (en) | 2012-08-10 | 2022-07-12 | ADA-ES, Inc. | Method and additive for controlling nitrogen oxide emissions |
Families Citing this family (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5517460B2 (ja) * | 2009-01-19 | 2014-06-11 | バブコック日立株式会社 | 脱硝装置 |
| WO2011058906A1 (ja) * | 2009-11-16 | 2011-05-19 | 株式会社Ihi | 水銀除去装置及び水銀除去方法 |
| JP5593100B2 (ja) * | 2010-03-17 | 2014-09-17 | バブコック日立株式会社 | ボイラプラント |
| CN102218267A (zh) * | 2010-04-15 | 2011-10-19 | 中国科学院城市环境研究所 | 一种用于催化降解燃煤烟气中多环芳烃(PAHs)的方法 |
| US8110164B2 (en) * | 2010-06-23 | 2012-02-07 | Baoquan Zhang | Flue-Gas purification and reclamation system and method thereof |
| JP5517778B2 (ja) * | 2010-06-25 | 2014-06-11 | 三菱重工業株式会社 | 噴霧装置及び水銀除去システム |
| JP5945093B2 (ja) * | 2010-09-06 | 2016-07-05 | 株式会社Ihi | 水銀除去装置 |
| JP5529701B2 (ja) | 2010-09-28 | 2014-06-25 | 三菱重工業株式会社 | ガス分析装置、水銀除去システム及び水銀除去方法 |
| EP2628527B1 (en) | 2010-10-15 | 2019-05-22 | Mitsubishi Hitachi Power Systems, Ltd. | System for processing mercury in exhaust gas |
| US9126143B2 (en) | 2011-10-12 | 2015-09-08 | Greenville Envirotech Co., Ltd. | Process and apparatus for removal of nitrogen oxides, sulfur oxides and mercury from off gas through oxidization |
| CN102527212B (zh) * | 2011-12-07 | 2013-08-28 | 南京钢铁股份有限公司 | 焦化厂氨性槽罐尾气综合治理方法及系统 |
| CN102716652B (zh) * | 2012-07-09 | 2014-06-25 | 常州大学 | 复杂废气综合处理系统 |
| JP6095923B2 (ja) * | 2012-09-14 | 2017-03-15 | 三菱日立パワーシステムズ株式会社 | 排ガス中の水銀処理システム |
| JP5583198B2 (ja) * | 2012-12-14 | 2014-09-03 | 三菱重工業株式会社 | 石炭乾留装置 |
| WO2014103074A1 (ja) * | 2012-12-28 | 2014-07-03 | 三菱重工業株式会社 | Coシフト触媒、coシフト反応装置及びガス化ガスの精製方法 |
| WO2014110882A1 (zh) * | 2013-01-18 | 2014-07-24 | 北京神雾环境能源科技集团股份有限公司 | 搭载蓄热式旋转换向加热器的粉状固体燃料锅炉 |
| CN103933838B (zh) * | 2013-01-21 | 2016-06-15 | 山东大学 | 煤中氯元素循环利用实现污染物联合脱除的装置及方法 |
| WO2015051363A1 (en) * | 2013-10-04 | 2015-04-09 | University Of Cincinnati | Method and system for removing gaseous mercury in flue gases |
| JP2015100758A (ja) | 2013-11-26 | 2015-06-04 | 三菱重工業株式会社 | 排ガス処理システム |
| CN103638793A (zh) * | 2013-12-17 | 2014-03-19 | 张继惟 | 一种用于高浓度SO2或NOx烟气净化的技术系统 |
| CN103939930A (zh) * | 2014-04-15 | 2014-07-23 | 山东大学 | 一种基于烟气调质实现单质汞转化的系统和方法 |
| CN104258700A (zh) * | 2014-09-22 | 2015-01-07 | 中国华能集团清洁能源技术研究院有限公司 | 一种循环流化床锅炉脱硫脱硝系统 |
| WO2017002168A1 (ja) * | 2015-06-29 | 2017-01-05 | 三菱日立パワーシステムズ株式会社 | 排気ガス処理設備、及び排気ガス処理方法 |
| CN105344234A (zh) * | 2015-10-30 | 2016-02-24 | 无锡华光新动力环保科技股份有限公司 | 一种烟气协同催化脱硝脱汞工艺 |
| KR101575699B1 (ko) * | 2015-11-02 | 2015-12-08 | 정재억 | 복합악취 제거설비 |
| CN105642339B (zh) * | 2015-12-08 | 2019-04-02 | 长春工业大学 | 一种无需还原气的同时脱硫脱硝催化剂及制备方法 |
| JP6587933B2 (ja) * | 2015-12-25 | 2019-10-09 | 三菱重工業株式会社 | 脱硝装置 |
| CN106140142B (zh) * | 2016-06-27 | 2017-10-13 | 北京化工大学 | 一种铬钨锆复合氧化物脱硝催化剂及其制备方法和应用 |
| CN107551757A (zh) * | 2016-06-30 | 2018-01-09 | 中冶长天国际工程有限责任公司 | 一种烟气脱硫脱硝方法和装置 |
| CN106362587A (zh) * | 2016-11-07 | 2017-02-01 | 上海东化环境工程有限公司 | 一种催化裂化烟气的脱硫脱硝工艺 |
| CN108295621B (zh) * | 2018-01-29 | 2019-07-12 | 中冶长天国际工程有限责任公司 | 一种多工序烟气净化系统及其控制方法 |
| CN108479344B (zh) * | 2018-04-08 | 2021-01-19 | 中冶长天国际工程有限责任公司 | 一种脱硫脱硝除氨系统 |
| CN108371872B (zh) * | 2018-04-08 | 2023-07-25 | 中冶长天国际工程有限责任公司 | 高效脱硝的脱硫脱硝装置 |
| CN108607322B (zh) * | 2018-05-10 | 2020-12-04 | 中冶长天国际工程有限责任公司 | 一种多工况烟气集中独立净化处理系统及其控制方法 |
| JP2020044461A (ja) * | 2018-09-14 | 2020-03-26 | 三菱日立パワーシステムズ株式会社 | 脱硝装置 |
| CN109603456B (zh) * | 2019-01-25 | 2021-08-03 | 米凯利科技(北京)有限公司 | 利用卤化物晶体的干法烟气脱汞系统 |
| CN112390285A (zh) * | 2020-12-03 | 2021-02-23 | 攀枝花钢企欣宇化工有限公司 | 一种钒钛系废催化剂的回收方法 |
| RU2755091C1 (ru) * | 2021-02-16 | 2021-09-13 | Открытое акционерное общество "Всероссийский дважды ордена Трудового Красного Знамени теплотехнический научно-исследовательский институт" (ОАО "ВТИ") | Аппарат для мокрой сероочистки дымовых газов |
| BE1030151B1 (nl) | 2021-12-30 | 2023-07-31 | Indaver Nv | Werkwijze en inrichting voor reiniging van rookgas afkomstig van de verbranding van chloorhoudend materiaal en verkregen gereinigd rookgas |
| BE1030154B1 (nl) | 2021-12-30 | 2023-07-31 | Indaver Nv | Werkwijze en inrichting voor onttrekken van zuur uit rookgas afkomstig van de verbranding van chloorhoudend materiaal en verkregen product |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10230137A (ja) | 1997-02-19 | 1998-09-02 | Mitsubishi Heavy Ind Ltd | 排ガス処理方法及び排ガス処理装置 |
| JP2003305338A (ja) * | 2002-04-15 | 2003-10-28 | Babcock Hitachi Kk | 排ガス浄化用触媒および浄化方法 |
| JP2004237244A (ja) | 2003-02-07 | 2004-08-26 | Mitsubishi Heavy Ind Ltd | 排ガス中の水銀除去方法およびそのシステム |
| JP2006136869A (ja) | 2004-10-14 | 2006-06-01 | Mitsubishi Heavy Ind Ltd | 排ガス処理触媒、排ガス処理方法および排ガス処理装置 |
| JP2006263700A (ja) * | 2005-02-28 | 2006-10-05 | Mitsubishi Heavy Ind Ltd | 排ガス中の水銀除去システムおよび除去方法 |
| JP2007215818A (ja) | 2006-02-17 | 2007-08-30 | Nippon Chem Ind Co Ltd | 消臭剤組成物 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001198434A (ja) * | 2000-01-18 | 2001-07-24 | Mitsubishi Heavy Ind Ltd | 排ガス中の水銀処理方法および排ガスの処理システム |
| US6960329B2 (en) * | 2002-03-12 | 2005-11-01 | Foster Wheeler Energy Corporation | Method and apparatus for removing mercury species from hot flue gas |
| JP2004337781A (ja) * | 2003-05-16 | 2004-12-02 | Mitsubishi Heavy Ind Ltd | 排ガス処理方法、排ガス処理システム及び触媒酸化装置 |
| TWI306410B (en) * | 2003-10-22 | 2009-02-21 | Nippon Catalytic Chem Ind | Method for treating exhaust gas |
| US7438876B2 (en) * | 2003-12-02 | 2008-10-21 | Cichanowicz J Edward | Multi-stage heat absorbing reactor and process for SCR of NOx and for oxidation of elemental mercury |
| JP4768636B2 (ja) * | 2004-02-04 | 2011-09-07 | バッテル メモリアル インスティチュート | 硫黄酸化物吸着剤および放出の制御 |
| US7597864B2 (en) * | 2004-07-16 | 2009-10-06 | Breen Energy Solutions | Control of mercury and other elemental metal emissions using reaction stabilization device |
| CN101098835A (zh) * | 2005-01-06 | 2008-01-02 | 太平洋水泥株式会社 | 水泥窑燃烧废气处理装置及处理方法 |
-
2008
- 2008-02-28 JP JP2008047460A patent/JP5319934B2/ja active Active
-
2009
- 2009-02-26 EP EP09716136.8A patent/EP2248574B1/en active Active
- 2009-02-26 BR BRPI0904317-9A patent/BRPI0904317A2/pt not_active IP Right Cessation
- 2009-02-26 KR KR1020107003685A patent/KR101126840B1/ko active Active
- 2009-02-26 CN CN2009801006619A patent/CN101932376B/zh active Active
- 2009-02-26 CA CA2694862A patent/CA2694862C/en active Active
- 2009-02-26 ES ES09716136.8T patent/ES2588371T3/es active Active
- 2009-02-26 WO PCT/JP2009/053575 patent/WO2009107731A1/ja not_active Ceased
- 2009-02-26 PL PL09716136.8T patent/PL2248574T3/pl unknown
- 2009-02-26 US US12/670,871 patent/US8420034B2/en active Active
- 2009-02-26 RU RU2010106054/05A patent/RU2429900C1/ru active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10230137A (ja) | 1997-02-19 | 1998-09-02 | Mitsubishi Heavy Ind Ltd | 排ガス処理方法及び排ガス処理装置 |
| JP2003305338A (ja) * | 2002-04-15 | 2003-10-28 | Babcock Hitachi Kk | 排ガス浄化用触媒および浄化方法 |
| JP2004237244A (ja) | 2003-02-07 | 2004-08-26 | Mitsubishi Heavy Ind Ltd | 排ガス中の水銀除去方法およびそのシステム |
| JP2006136869A (ja) | 2004-10-14 | 2006-06-01 | Mitsubishi Heavy Ind Ltd | 排ガス処理触媒、排ガス処理方法および排ガス処理装置 |
| JP2006263700A (ja) * | 2005-02-28 | 2006-10-05 | Mitsubishi Heavy Ind Ltd | 排ガス中の水銀除去システムおよび除去方法 |
| JP2007215818A (ja) | 2006-02-17 | 2007-08-30 | Nippon Chem Ind Co Ltd | 消臭剤組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2248574A4 * |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011120981A (ja) * | 2009-12-09 | 2011-06-23 | Babcock Hitachi Kk | 酸素燃焼方式の排ガス処理装置と該排ガス処理装置の運用方法 |
| US11213787B2 (en) | 2010-02-04 | 2022-01-04 | ADA-ES, Inc. | Method and system for controlling mercury emissions from coal-fired thermal processes |
| US11298657B2 (en) | 2010-10-25 | 2022-04-12 | ADA-ES, Inc. | Hot-side method and system |
| US11118127B2 (en) | 2011-05-13 | 2021-09-14 | ADA-ES, Inc. | Process to reduce emissions of nitrogen oxides and mercury from coal-fired boilers |
| US11065578B2 (en) * | 2012-04-11 | 2021-07-20 | ADA-ES, Inc. | Control of wet scrubber oxidation inhibitor and byproduct recovery |
| US11384304B2 (en) | 2012-08-10 | 2022-07-12 | ADA-ES, Inc. | Method and additive for controlling nitrogen oxide emissions |
| CN105312047A (zh) * | 2014-07-18 | 2016-02-10 | 中国钢铁股份有限公司 | 脱硝催化剂之制造方法及其应用 |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2588371T3 (es) | 2016-11-02 |
| KR20100033546A (ko) | 2010-03-30 |
| EP2248574A4 (en) | 2012-02-22 |
| CN101932376A (zh) | 2010-12-29 |
| KR101126840B1 (ko) | 2012-03-27 |
| PL2248574T3 (pl) | 2016-12-30 |
| JP2009202107A (ja) | 2009-09-10 |
| EP2248574B1 (en) | 2016-06-29 |
| CN101932376B (zh) | 2013-07-10 |
| US8420034B2 (en) | 2013-04-16 |
| US20110162345A1 (en) | 2011-07-07 |
| RU2429900C1 (ru) | 2011-09-27 |
| JP5319934B2 (ja) | 2013-10-16 |
| CA2694862C (en) | 2012-08-07 |
| EP2248574A1 (en) | 2010-11-10 |
| BRPI0904317A2 (pt) | 2015-06-30 |
| CA2694862A1 (en) | 2009-09-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5319934B2 (ja) | 排ガス処理方法及び装置 | |
| US6638485B1 (en) | Process for treating exhaust gas and exhaust gas treating equipment | |
| JP4838579B2 (ja) | 水銀除去システムおよび水銀除去方法 | |
| JP4388542B2 (ja) | 水銀除去方法及び水銀除去システム | |
| CN102119051B (zh) | 废气处理装置及废气处理系统 | |
| JP2004237244A (ja) | 排ガス中の水銀除去方法およびそのシステム | |
| JP2001198434A (ja) | 排ガス中の水銀処理方法および排ガスの処理システム | |
| JP4898751B2 (ja) | 排ガス処理装置及び排ガス処理システム | |
| JP4959650B2 (ja) | 排ガス処理装置及び排ガス処理システム | |
| US7501104B2 (en) | Apparatus and method for treating discharge gas | |
| JP5398193B2 (ja) | 排ガス処理装置、排ガス処理システム及び排ガス中の水銀酸化性能管理システム | |
| KR100550603B1 (ko) | 건식으로 질소산화물을 제거하는 장치 및 방법 | |
| JP5419400B2 (ja) | 排ガス処理装置及び排ガス処理システム |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980100661.9 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09716136 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2694862 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009716136 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 315/MUMNP/2010 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 20107003685 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010106054 Country of ref document: RU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12670871 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: PI0904317 Country of ref document: BR Kind code of ref document: A2 Effective date: 20100208 |