WO2011139252A2 - Efervescent formulations comprising cefdinir - Google Patents
Efervescent formulations comprising cefdinir Download PDFInfo
- Publication number
- WO2011139252A2 WO2011139252A2 PCT/TR2011/000127 TR2011000127W WO2011139252A2 WO 2011139252 A2 WO2011139252 A2 WO 2011139252A2 TR 2011000127 W TR2011000127 W TR 2011000127W WO 2011139252 A2 WO2011139252 A2 WO 2011139252A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pharmaceutical formulation
- cefdinir
- formulation according
- effervescent
- taste
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0002—Galenical forms characterised by the drug release technique; Application systems commanded by energy
- A61K9/0007—Effervescent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/54—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame
- A61K31/542—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame ortho- or peri-condensed with heterocyclic ring systems
- A61K31/545—Compounds containing 5-thia-1-azabicyclo [4.2.0] octane ring systems, i.e. compounds containing a ring system of the formula:, e.g. cephalosporins, cefaclor, or cephalexine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
Definitions
- the present invention relates to effervescent pharmaceutical dosage forms comprising cefdinir as the active agent, and their preparation.
- Cefdinir which physically appears as a white powder has very poor solubility in common organic solvents such as methanol, ethanol, and acetonitrile and in water.
- Cefdinir do not get wet or dissolve in water owing to its hydrophobic character. Moreover, cefdinir has a very bitter taste that it is difficult to mask it with flavoring agents or taste regulating agents.
- OMNICEF ® The product under the name OMNICEF ® is available in capsule and suspension forms on the market.
- Administration of solid oral dosage forms such as capsules, pills and tablets by oral route generally causes difficulty in swallowing for geriatric and pediatric patients.
- alternatively developed suspension dosage forms indicate higher bioavailability, the use of this dosage form brings along the possibility of excessive and/or uncontrolled dose intake particularly in pediatric and geriatric patients.
- water dispersible forms are preferred since they are more convenient to use for geriatric and pediatric patients and they solve the solubility problem of cefdinir.
- excipients such as taste regulating agent, aroma and/or flavoring agents are used to eliminate the bitter taste of cefdinir, a desirable and pleasant taste of it can not be achieved.
- the inventors have surprisingly found that the problems in the prior art can be solved by the pharmaceutical formulations formulated in effervescent form prepared according to the present invention.
- the subject of the present invention is effervescent formulations comprising cefdinir and the procedures related to their preparation.
- the effervescent formulations comprising cefdinir wherein two different taste regulating agents are used and the ratio of the first taste regulating agent having 31-90% (w/w) water solubility at 25 °C to the second taste regulating agent having 5-30% (w/w) water solubility at 25°C is in the range of 5:1 to 1:1, have achieved to increase the solubility of cefdinir and eliminate the bitter taste of cefdinir.
- the first aspect of the present invention is the effervescent formulations comprising cefdinir as the active agent in which two different taste regulating agents are used and the ratio of the first taste regulating agent having 31-90% (w/w) water solubility at 25°C to the second taste regulating agent having 5-30% (w/w) water solubility at 25°C is in the range of 5:1 to 1:1.
- effervescent formulations present in the text comprises effervescent tablets, effervescent granules and effervescent powders.
- taste regulating agent having 31-90 % (w/w) water solubility at 25 °C means that the amount of taste regulating agent dissolved in 100 g water is in the range of 31-90 g at 25°C.
- taste regulating agent having 5-30 % (w/w) water solubility at 25°C means that the amount of taste regulating agent dissolved in 100 g water is in the range of 5-30 g at 25°C.
- Cefdinir that can be used in the effervescent formulations, which are the subject of the present invention, can be in the form of its solvates, hydrates, enantiomers, racemates, organic salts, inorganic salts, polymorphs, crystal and amorphous forms or in free form and/or combination thereof.
- the first taste regulating agent that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising dextrose, fructose, glucose, lactitol, maltitol, maltose, sorbitol, saccharine sodium, sodium cyclamate, sodium chloride, potassium chloride, sucrose and xylitol or combinations thereof.
- sodium chloride is used as the first taste regulating agent in the formulations according to the present invention.
- the second taste regulating agent that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising acesulfame, aspartame, saccharine and sucralose, or combinations thereof.
- sucralose is used as the second taste regulating agent in the formulations according to the present invention.
- another aspect of the present invention is the effervescent formulations comprising cefdinir as the active agent in which sodium chloride and sucralose are used as taste regulating agents and the ratio of sodium chloride to sucralose is in the range of 5:1 to 1:1.
- the ratio of active agent to the taste regulating agent comprising two different agents is significant in masking the bitter taste of cefdinir. Therefore, it has been observed that unpleasant taste of cefdinir is successfully masked in the formulations in which the ratio of cefdinir to the combination of taste regulating agents is in the range of 10:1 to 1:1, preferably 8:1 to 1:1 and more preferably 5:1 to 2:1.
- another aspect of the present invention is effervescent formulations wherein the ratio of cefdinir to the combination of taste regulating agents is in the range of 10:1 to 1:1, preferably 8:1 to 1:1 and more preferably 5:1 to 2:1.
- the inventors have also found that the effervescent formulations comprising cefdinir, in which organic base is used in addition to cefdinir and the ratio of cefdinir to the organic base is in the range of 5:1 to 1:5, provide dissolution of cefdinir in water entirely. Therefore, the developed effervescent formulations pertaining to the present invention have both provided a high solubility of cefdinir in water and eliminated the unpleasant taste of cefdinir to a large extent.
- another aspect of the present invention is the effervescent formulations comprising cefdinir in which organic base is used in addition to cefdinir and the ratio of cefdinir to the organic base is in the range of 5:1 to 1 :5.
- primary amines, secondary amines, tertiary amines and/or heterocyclic compounds containing nitrogen can be used as the organic base in the present invention.
- the organic base that is to be used in the formulation can be selected from, but not limited to, a group comprising ethanolamine, isopropanolamine, 1-dioxy-l-methylamino-sorbitol, 1- dioxy- 1 -methylamino-D-glucitol, tris(hydroxymethyl)aminomethane, N-(Tri(hydroxymethyl) methyl)glycine, N,N-Bis(2-hydroxyethyl)glycine, 2-methyl aminophenol.
- 1- dioxy-l-methylamino-sorbitol and tris(hydroxymethyl)aminomethane are used.
- Another aspect of the present invention is the effervescent formulations comprising pharmaceutically acceptable excipients in addition to the active agent cefdinir.
- excipients selected from, but not limited to, a group comprising binders, lubricants, humectants, disintegrants, diluents, effervescent acid and effervescent base can be used in the effervescent formulation pertaining to the present invention apart from cefdinir, taste regulating agent and the organic base.
- the binder that can be used in the effervescent formulations pertaining to the present invention can be selected from, but not limited to, a group comprising ethyl cellulose, gelatine, hydroxyethyl cellulose, hydroxymethyl cellulose, hydroxypropyl cellulose, hypromellose, magnesium aluminum silicate, methyl cellulose, povidone.
- povidon is used in scope of the present invention.
- the inventors have found that the dissolution time of the pharmaceutical formulation when 1- 8% binder is used is shorter compared to the formulations comprising a higher amount of binder.
- another aspect of the present invention is effervescent formulations which comprise the binder in the ratio of 1-8%, preferably in the ratio of 1-7%, most preferably in the ratio of 2-5% with respect to the total weight of the composition and cefdinir as the active agent.
- cefdinir: binder ratio is in the range of 5:1 to 1:3, preferably 4:1 to 2:1 in the effervescent formulations comprising cefdinir, the water solubility of cefdinir has increased.
- another aspect of the present invention is the effervescent formulations comprising cefdinir wherein the ratio of cefdinir to the binder is in the range of 5:1 to 1:3, preferably in the range of 4: 1 to 2: 1.
- the lubricant that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising calcium stearate, magnesium stearate, polyethylene glycol, PEG 6000, polyvinyl alcohol, potassium benzoate, sodium benzoate.
- PEG 6000 is used as lubricant in the formulations pertaining to the present invention.
- the disintegrant that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising carboxymethyl cellulose calcium, carboxymethyl cellulose sodium, microcrystalline cellulose, silicon dioxide, croscarmellose sodium, crospovidone, hydroxypropyl cellulose, methyl cellulose, povidone, magnesium aluminum silicate and starch or combinations thereof.
- the diluent that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising calcium carbonate, calcium sulphate, dibasic calcium phosphate, tribasic calcium phosphate, calcium phosphate, calcium sulphate, microcrystalline cellulose, lactose, magnesium carbonate, magnesium oxide, maltodextrin, maltose, mannitol, sodium chloride, sorbitol, starch and xylitol or combinations thereof.
- the effervescent acid that is used in effervescent formulations pertaining to the present invention can be selected from organic acids such as citric acid, tartaric acid, malic acid, fumaric acid.
- citric acid is used as effervescent acid.
- the effervescent base that is used in effervescent formulations pertaining to the present invention can be selected from sodium hydrogen carbonate, sodium carbonate, potassium carbonate and potassium hydrogen carbonate.
- sodium hydrogen carbonate is used as effervescent base.
- cefdinir or its pharmaceutically acceptable salts, hydrates, solvates or combinations thereof can be used.
- the effervescent formulation of the present invention can comprise 5-30% cefdinir or pharmaceutically acceptable solvates, hydrates, enantiomers, racemates, organic salts, inorganic salts, polymorphs, crystal and amorphous forms of cefdinir and 1-20% organic base; 1-8% binder; 0.1-2% lubricant; 0.1-8% taste regulating agents, 0-25% diluent; 0-15% disintegrant; 0-10% humectant; 0.2-6% coloring agent and/or flavoring agent, 10-60% effervescent acid and 15-50% effervescent base with respect to the total amount of unit dose.
- the present invention relates to the processes which can be used for the preparation of effervescent formulations comprising pharmaceutically acceptable excipients in addition to cefdinir as the active agent.
- the process in scope of the present invention comprises the granulation of the active agent cefdinir through conventional wet and/or dry granulation methods present in the prior art or mixing cefdinir and the other excipients through dry blending method and then pulverizing them and optionally compressing them in tablet compressing machine to obtain tablet forms.
- the pharmaceutical formulation prepared according to the present invention is used in the manufacture of a medicament so as to be used in upper respiratory infections such as ear, nose, throat, otitis media, sinusitis, tonsillitis, pharyngitis; lower respiratory tract infections such as pyelonephritis, cystitis and urethritis; skin or soft tissue infections such as froncle, pyoderma, impetigo; in the treatment and prophylaxis of gonorrhea and lyme diseases.
- upper respiratory infections such as ear, nose, throat, otitis media, sinusitis, tonsillitis, pharyngitis
- lower respiratory tract infections such as pyelonephritis, cystitis and urethritis
- skin or soft tissue infections such as froncle, pyoderma, impetigo
- in the treatment and prophylaxis of gonorrhea and lyme diseases in the manufacture of a medicament so as to be used in upper respiratory infections such as
- Example 1 Formulation and process for the preparation of effervescent tablet
- the formulation to be used in scope of said invention comprises granulating sodium hydrogen carbonate and cefdinir with aqueous organic base solution; then adding citric acid and sodium chloride into the obtained granule and granulating the mixture obtained with binder solution; following this, adding sucralose, lubricant, coloring agent and flavoring agent to the granule mixture obtained and mixing it again; then compressing the obtained mixture in tablet compressing machine to obtain tablets.
- Comparative Example 1 Formulation and process for the preparation of effervescent tablet
- the formulation comprises granulating sodium hydrogen carbonate and cefdinir with aqueous organic base solution; then adding citric acid and sodium chloride into the obtained granule and granulating the mixture obtained with binder solution; following this, adding sucralose, lubricant, coloring agent and flavoring agent to the granule mixture obtained and mixing it again; then compressing the obtained mixture in tablet compressing machine to obtain tablets.
- Comparative Example 2 Formulation and process for the preparation of effervescent tablet
- the formulation comprises granulating sodium hydrogen carbonate and cefdinir with aqueous organic base solution; then adding citric acid and sodium chloride into the obtained granule and granulating the mixture obtained with binder solution; following this, adding lubricant, coloring agent and flavoring agent to the granule mixture obtained and mixing it again; then compressing the obtained mixture in tablet compressing machine to obtain tablets.
- Comparative Example 3 Formulation and process for the preparation of effervescent tablet
- the formulation comprises granulating sodium hydrogen carbonate and cefdinir with aqueous organic base solution; then adding citric acid into the obtained granule and granulating the mixture obtained with binder solution; following this, adding sucralose, lubricant, coloring agent and flavoring agent to the granule mixture obtained and mixing it again; then compressing the obtained mixture in tablet compressing machine to obtain tablets.
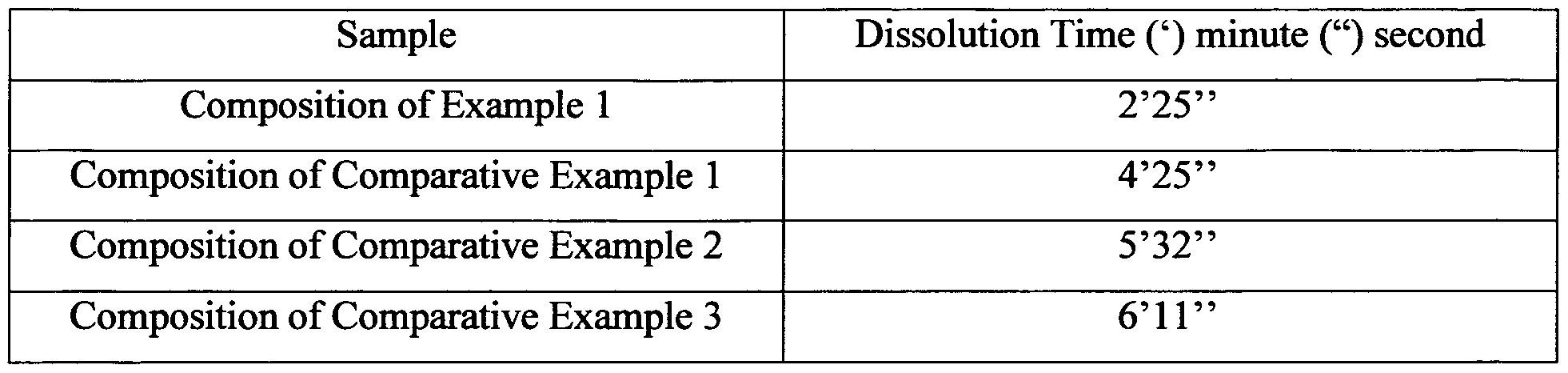
- Table 1 indicates the comparative data of dissolution time of the compositions for each example.
- Table 2 to Table 5 the results of the bitterness values of each cefdinir compositions which are calculated according to the European Pharmacopoeia 5.0 (2.8.15, pg. 221) are shown.
- the inventors have observed that the optimum dissolution time is less than 5 minutes for the effervescent formulations comprising cefdinir.
- Panelist* a member of a taste panel of six people
- k* the correction factor for each panelist
- Y* Dilution Factor (DF) of solution D which is a diluted solution having still a bitter test
- X* number of milliliters of solution D which, when diluted to 10 ml with water, still has a bitter taste
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention relates to effervescent dosage forms comprising cefdinir as the active agent, and the preparation of said formulations.
Description
EFERVESCENT FORMULATIONS COMPRISING CEFDINIR
The present invention relates to effervescent pharmaceutical dosage forms comprising cefdinir as the active agent, and their preparation.
Background of the Invention
The chemical structure of the molecule cefdinir, which has the chemical name (6R,7R)-7- [[(2Z)-(2-amino-4-tMazolil)(hydroxyir^
azabicyclo[4.2.0]oct-2-en-2-carboxylic acid and was first disclosed in the patent numbered BE897864, is shown in Formula I. This molecule, which is a third generation cephalosporin, is indicated for the treatment of many diseases caused by gram positive and gram negative bacteria.
Formula I
Cefdinir which physically appears as a white powder has very poor solubility in common organic solvents such as methanol, ethanol, and acetonitrile and in water.
Cefdinir do not get wet or dissolve in water owing to its hydrophobic character. Moreover, cefdinir has a very bitter taste that it is difficult to mask it with flavoring agents or taste regulating agents.
The product under the name OMNICEF® is available in capsule and suspension forms on the market. Administration of solid oral dosage forms such as capsules, pills and tablets by oral route generally causes difficulty in swallowing for geriatric and pediatric patients. Although alternatively developed suspension dosage forms indicate higher bioavailability, the use of this dosage form brings along the possibility of excessive and/or uncontrolled dose intake particularly in pediatric and geriatric patients.
In the solid oral and suspension dosage forms, some methods have been tried in order to eliminate the bitter taste of cefdinir. In the tablets containing coating, it has been tried to mask the bitter taste of cefdinir with the help of the coating material. However, cefdinir has such a bitter taste that coating materials are unable to mask it. On the other hand, the coatings in the
suspension dosage forms provide a solution for low solubility problem of cefdinir while they remain incapable of masking the bitter taste of it.
Alternatively, water dispersible forms are preferred since they are more convenient to use for geriatric and pediatric patients and they solve the solubility problem of cefdinir. Although some excipients such as taste regulating agent, aroma and/or flavoring agents are used to eliminate the bitter taste of cefdinir, a desirable and pleasant taste of it can not be achieved.
Accordingly, it is clearly seen that the problems such as difficulty in swallowing or low solubility of cefdinir can be eliminated to some extent. However, masking the bitter taste of cefdinir is a challenging task to overcome.
As it can be seen, new dosage forms and formulations need to be developed so as to provide reliable and user friendly dosage form alternatives for pediatric and geriatric patients, to overcome the solubility problems of cefdinir and especially, to eliminate the bitter taste of cefdinir.
The inventors have surprisingly found that the problems in the prior art can be solved by the pharmaceutical formulations formulated in effervescent form prepared according to the present invention.
Description of the Invention
The subject of the present invention is effervescent formulations comprising cefdinir and the procedures related to their preparation. Surprisingly, it has been found that the effervescent formulations comprising cefdinir, wherein two different taste regulating agents are used and the ratio of the first taste regulating agent having 31-90% (w/w) water solubility at 25 °C to the second taste regulating agent having 5-30% (w/w) water solubility at 25°C is in the range of 5:1 to 1:1, have achieved to increase the solubility of cefdinir and eliminate the bitter taste of cefdinir.
The inventors have found that these effervescent formulations developed pertaining to the present invention can be administered by pediatric and geriatric patients easily, can lead to the increase in the solubility of cefdinir and can mask the bitter and durable taste of cefdinir entirely.
The choice of the taste regulating agent with respect to their water solubility values enables fine tuning of the solubility problem of cefdinir and the ratio of these two agents provides effective taste masking of the formed solution.
Accordingly, the first aspect of the present invention is the effervescent formulations comprising cefdinir as the active agent in which two different taste regulating agents are used and the ratio of the first taste regulating agent having 31-90% (w/w) water solubility at 25°C to the second taste regulating agent having 5-30% (w/w) water solubility at 25°C is in the range of 5:1 to 1:1.
The term "effervescent formulations" present in the text comprises effervescent tablets, effervescent granules and effervescent powders.
The term "taste regulating agent having 31-90 % (w/w) water solubility at 25 °C" means that the amount of taste regulating agent dissolved in 100 g water is in the range of 31-90 g at 25°C.
The term "taste regulating agent having 5-30 % (w/w) water solubility at 25°C" means that the amount of taste regulating agent dissolved in 100 g water is in the range of 5-30 g at 25°C.
Cefdinir that can be used in the effervescent formulations, which are the subject of the present invention, can be in the form of its solvates, hydrates, enantiomers, racemates, organic salts, inorganic salts, polymorphs, crystal and amorphous forms or in free form and/or combination thereof.
The first taste regulating agent that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising dextrose, fructose, glucose, lactitol, maltitol, maltose, sorbitol, saccharine sodium, sodium cyclamate, sodium chloride, potassium chloride, sucrose and xylitol or combinations thereof. Preferably, sodium chloride is used as the first taste regulating agent in the formulations according to the present invention.
The second taste regulating agent that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising acesulfame, aspartame, saccharine and sucralose, or combinations thereof. Preferably, sucralose is used as the second taste regulating agent in the formulations according to the present invention.
According to this, another aspect of the present invention is the effervescent formulations comprising cefdinir as the active agent in which sodium chloride and sucralose are used as taste regulating agents and the ratio of sodium chloride to sucralose is in the range of 5:1 to 1:1.
The inventors have found that the ratio of active agent to the taste regulating agent comprising two different agents is significant in masking the bitter taste of cefdinir. Therefore, it has been observed that unpleasant taste of cefdinir is successfully masked in the formulations in which the ratio of cefdinir to the combination of taste regulating agents is in the range of 10:1 to 1:1, preferably 8:1 to 1:1 and more preferably 5:1 to 2:1.
According to this, another aspect of the present invention is effervescent formulations wherein the ratio of cefdinir to the combination of taste regulating agents is in the range of 10:1 to 1:1, preferably 8:1 to 1:1 and more preferably 5:1 to 2:1.
In the present invention, the inventors have also found that the effervescent formulations comprising cefdinir, in which organic base is used in addition to cefdinir and the ratio of cefdinir to the organic base is in the range of 5:1 to 1:5, provide dissolution of cefdinir in water entirely. Therefore, the developed effervescent formulations pertaining to the present invention have both provided a high solubility of cefdinir in water and eliminated the unpleasant taste of cefdinir to a large extent.
According to this, another aspect of the present invention is the effervescent formulations comprising cefdinir in which organic base is used in addition to cefdinir and the ratio of cefdinir to the organic base is in the range of 5:1 to 1 :5.
In the effervescent formulations pertaining to the present invention, primary amines, secondary amines, tertiary amines and/or heterocyclic compounds containing nitrogen can be used as the organic base in the present invention.
The organic base that is to be used in the formulation can be selected from, but not limited to, a group comprising ethanolamine, isopropanolamine, 1-dioxy-l-methylamino-sorbitol, 1- dioxy- 1 -methylamino-D-glucitol, tris(hydroxymethyl)aminomethane, N-(Tri(hydroxymethyl) methyl)glycine, N,N-Bis(2-hydroxyethyl)glycine, 2-methyl aminophenol. Preferably, 1- dioxy-l-methylamino-sorbitol and tris(hydroxymethyl)aminomethane are used.
Another aspect of the present invention is the effervescent formulations comprising pharmaceutically acceptable excipients in addition to the active agent cefdinir.
Various excipients selected from, but not limited to, a group comprising binders, lubricants, humectants, disintegrants, diluents, effervescent acid and effervescent base can be used in the effervescent formulation pertaining to the present invention apart from cefdinir, taste regulating agent and the organic base.
The binder that can be used in the effervescent formulations pertaining to the present invention can be selected from, but not limited to, a group comprising ethyl cellulose, gelatine, hydroxyethyl cellulose, hydroxymethyl cellulose, hydroxypropyl cellulose, hypromellose, magnesium aluminum silicate, methyl cellulose, povidone. Preferably, povidon is used in scope of the present invention.
The inventors have found that the dissolution time of the pharmaceutical formulation when 1- 8% binder is used is shorter compared to the formulations comprising a higher amount of binder.
According to this, another aspect of the present invention is effervescent formulations which comprise the binder in the ratio of 1-8%, preferably in the ratio of 1-7%, most preferably in the ratio of 2-5% with respect to the total weight of the composition and cefdinir as the active agent.
The inventors have also found that when cefdinir: binder ratio is in the range of 5:1 to 1:3, preferably 4:1 to 2:1 in the effervescent formulations comprising cefdinir, the water solubility of cefdinir has increased.
Accordingly, another aspect of the present invention is the effervescent formulations comprising cefdinir wherein the ratio of cefdinir to the binder is in the range of 5:1 to 1:3, preferably in the range of 4: 1 to 2: 1.
The lubricant that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising calcium stearate, magnesium stearate, polyethylene glycol, PEG 6000, polyvinyl alcohol, potassium benzoate, sodium benzoate. Preferably, PEG 6000 is used as lubricant in the formulations pertaining to the present invention.
The disintegrant that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising carboxymethyl cellulose calcium, carboxymethyl cellulose sodium, microcrystalline cellulose, silicon dioxide, croscarmellose sodium, crospovidone, hydroxypropyl cellulose, methyl cellulose, povidone, magnesium aluminum silicate and starch or combinations thereof.
The diluent that can be used in effervescent formulations of the present invention can be selected from, but not limited to, a group comprising calcium carbonate, calcium sulphate, dibasic calcium phosphate, tribasic calcium phosphate, calcium phosphate, calcium sulphate, microcrystalline cellulose, lactose, magnesium carbonate, magnesium oxide, maltodextrin, maltose, mannitol, sodium chloride, sorbitol, starch and xylitol or combinations thereof.
The effervescent acid that is used in effervescent formulations pertaining to the present invention can be selected from organic acids such as citric acid, tartaric acid, malic acid, fumaric acid. Preferably, citric acid is used as effervescent acid.
The effervescent base that is used in effervescent formulations pertaining to the present invention can be selected from sodium hydrogen carbonate, sodium carbonate, potassium carbonate and potassium hydrogen carbonate. Preferably, sodium hydrogen carbonate is used as effervescent base.
In effervescent formulation of the present invention, cefdinir or its pharmaceutically acceptable salts, hydrates, solvates or combinations thereof can be used.
The effervescent formulation of the present invention can comprise 5-30% cefdinir or pharmaceutically acceptable solvates, hydrates, enantiomers, racemates, organic salts, inorganic salts, polymorphs, crystal and amorphous forms of cefdinir and 1-20% organic base; 1-8% binder; 0.1-2% lubricant; 0.1-8% taste regulating agents, 0-25% diluent; 0-15% disintegrant; 0-10% humectant; 0.2-6% coloring agent and/or flavoring agent, 10-60% effervescent acid and 15-50% effervescent base with respect to the total amount of unit dose.
In another aspect, the present invention relates to the processes which can be used for the preparation of effervescent formulations comprising pharmaceutically acceptable excipients in addition to cefdinir as the active agent.
According to this, the process in scope of the present invention comprises the granulation of the active agent cefdinir through conventional wet and/or dry granulation methods present in
the prior art or mixing cefdinir and the other excipients through dry blending method and then pulverizing them and optionally compressing them in tablet compressing machine to obtain tablet forms.
According to another aspect of the present invention, the pharmaceutical formulation prepared according to the present invention is used in the manufacture of a medicament so as to be used in upper respiratory infections such as ear, nose, throat, otitis media, sinusitis, tonsillitis, pharyngitis; lower respiratory tract infections such as pyelonephritis, cystitis and urethritis; skin or soft tissue infections such as froncle, pyoderma, impetigo; in the treatment and prophylaxis of gonorrhea and lyme diseases.
Example 1: Formulation and process for the preparation of effervescent tablet
Sodium chloride: sucralose ratio is 3:1.
The formulation to be used in scope of said invention comprises granulating sodium hydrogen carbonate and cefdinir with aqueous organic base solution; then adding citric acid and sodium chloride into the obtained granule and granulating the mixture obtained with binder solution; following this, adding sucralose, lubricant, coloring agent and flavoring agent to the granule mixture obtained and mixing it again; then compressing the obtained mixture in tablet compressing machine to obtain tablets.
Comparative Example 1: Formulation and process for the preparation of effervescent tablet
* Sodium chloride: sucralose ratio is 8:1.
The formulation comprises granulating sodium hydrogen carbonate and cefdinir with aqueous organic base solution; then adding citric acid and sodium chloride into the obtained granule and granulating the mixture obtained with binder solution; following this, adding sucralose, lubricant, coloring agent and flavoring agent to the granule mixture obtained and mixing it again; then compressing the obtained mixture in tablet compressing machine to obtain tablets.
Comparative Example 2: Formulation and process for the preparation of effervescent tablet
*Only sodium chloride is used a s taste regulating agent
The formulation comprises granulating sodium hydrogen carbonate and cefdinir with aqueous organic base solution; then adding citric acid and sodium chloride into the obtained granule and granulating the mixture obtained with binder solution; following this, adding lubricant, coloring agent and flavoring agent to the granule mixture obtained and mixing it again; then compressing the obtained mixture in tablet compressing machine to obtain tablets.
Comparative Example 3: Formulation and process for the preparation of effervescent tablet
*Only sucralose is used as taste regulating agent
The formulation comprises granulating sodium hydrogen carbonate and cefdinir with aqueous organic base solution; then adding citric acid into the obtained granule and granulating the mixture obtained with binder solution; following this, adding sucralose, lubricant, coloring agent and flavoring agent to the granule mixture obtained and mixing it again; then compressing the obtained mixture in tablet compressing machine to obtain tablets.
Results:
The inventors have studied on the dissolution data and bitterness values of the compositions illustrated in each example given above. Based on the studies on the comparison of their dissolution data and the bitterness values, the results shown in Tables 1-6 are obtained. Table 1 indicates the comparative data of dissolution time of the compositions for each example. In Table 2 to Table 5, the results of the bitterness values of each cefdinir compositions which are calculated according to the European Pharmacopoeia 5.0 (2.8.15, pg. 221) are shown.
Table 1: Comparative Data of Dissolution of Effervescent Tablets Comprising Cefdinir
The inventors have observed that the optimum dissolution time is less than 5 minutes for the effervescent formulations comprising cefdinir.
The results of the dissolution data of compositions of Example 1, Comparative example 1, Comparative example 2, Comparative example 3 clearly indicate that the effervescent tablet of the present invention in which sodium chloride and sucralose are used as two different taste regulating agent and the ratio of sodium chloride to sucralose is in the range of 5:1 to 1:1, has a dissolution time that is less than 5 minutes. This shows that the effervescent tablets prepared according to the present invention is more preferable than the other tablets via their higher dissolution rate.
Table 2: Results of the Taste Trial for Cefdinir Composition of Example 1
Panelist*: a member of a taste panel of six people k*: the correction factor for each panelist
Y*: Dilution Factor (DF) of solution D which is a diluted solution having still a bitter test
X*: number of milliliters of solution D which, when diluted to 10 ml with water, still has a bitter taste
Yxk
Bitterness value: a value calculated by using the expression of
xxo.i
Table 3: Results of the Taste Trial for Cefdinir Composition of Comparative Example 1
Table 4: Results of the Taste Trial for Cefdinir Composition of Comparative Example 2
Taste trials were performed by a taste panel comprising 6 persons. The average bitterness value of each effervescent composition of Example 1, Comparative example 1, Comparative example 2, Comparative example 3 are calculated according to the European Pharmacopoeia 5.0. Six panelists tasted the solutions of compositions prepared according to the method described in European Pharmacopoeia 5.0. The results are represented in Tables 2-5 and they clearly indicate that average bitterness value of the composition of example 1 is the lowest one. Therefore, it can be deduced that effervescent tablets formulated according to the present invention in which sodium chloride and sucralose are used as two different taste regulating agent and the ratio of sodium chloride to sucralose is in the range of 5:1 to 1:1 shown in example 1 is much preferred via its more pleasant taste.
Claims
1. A pharmaceutical formulation formulated in effervescent form comprising cefdinir as the active agent characterized in that two different taste regulating agents are used and the ratio of the first taste regulating agent having 31-90% (w/w) water solubility at 25°C and the second taste regulating agent having 5-30% (w/w) water solubility at 25°C is in the range of 5:1 and 1:1.
2. The pharmaceutical formulation according to claim 1, wherein cefdinir is present in the form of its solvates, hydrates, enantiomers, racemates, organic salts, inorganic salts, polymorphs, in crystal and amorphous forms or in free form and/or combination thereof.
3. The pharmaceutical formulation according to claim 1, wherein the first taste regulating agent used in effervescent formulations of the present invention is selected from a group comprising dextrose, fructose, glucose, lactitol, maltitol, maltose, sorbitol, saccharine sodium, sodium cyclamate, sodium chloride, potassium chloride, sucrose and xylitol or combinations thereof.
4. The pharmaceutical formulation according to claim 3, wherein sodium chloride is used as the first taste regulating agent.
5. The pharmaceutical formulation according to claim 1, wherein the second taste regulating agent used in effervescent formulations of the present invention is selected from a group comprising acesulfame, aspartame, saccharine and sucralose, or combinations thereof.
6. The pharmaceutical formulation according to claim 5, wherein sucralose is used as the second taste regulating agent.
7. The pharmaceutical formulation according to claim 1, wherein the ratio of cefdinir to the combination of taste regulating agents is in the range of 10: 1 to 1 : 1.
8. The pharmaceutical formulation according to claim 1, wherein organic base is used in addition to cefdinir and the taste regulating agents and the ratio of cefdinir to the organic base is in the range of 5:1 to 1 :5.
9. The pharmaceutical formulation according to claim 8, wherein primary amines, secondary amines, tertiary amines and/or heterocyclic compounds containing nitrogen can be used as the organic base.
10. The pharmaceutical formulation according to claim 9, wherein organic base that is to be used in the formulation is selected from a group comprising ethanolamine, isopropanolamine, 1 -dioxy- 1 -methylamino-sorbitol, 1 -dioxy- 1 -methylamino-D-glucitol, tris(hy(koxymethyl)aminomethane, N-(Tri(hydroxymethyl)methyl)glycine, N,N-Bis(2- hydroxyethyl)glycine, 2-methyl aminophenol.
11. The pharmaceutical formulation according to claim 8, wherein said formulation comprises pharmaceutically acceptable excipients in addition to cefdinir, combination of taste regulating agents and organic base.
12. The pharmaceutical formulation according to claim 11, wherein pharmaceutically acceptable excipients selected from a group comprising binders, lubricants, humectants, disintegrants, diluents, and effervescent couple in addition to cefdinir, combination of taste regulating agents and organic base.
13. The pharmaceutical formulation accoding to claim 12, wherein the binder is selected from a group comprising ethyl cellulose, gelatine, hydroxyethyl cellulose, hydroxymethyl cellulose, hydroxypropyl cellulose, hypromellose, magnesium aluminum silicate, methyl cellulose, povidone.
14. The pharmaceutical formulation according to claim 13, wherein povidone is used as the binder.
15. The pharmaceutical formulation according to claim 13, wherein said formulation comprises 1-8% binder.
16. The pharmaceutical formulation according to claim 13, wherein cefdinir: binder ratio is in the range of 5:1 to 1:3.
17. The pharmaceutical formulation according to claim 12, wherein the lubricant is selected from a group comprising calcium stearate, magnesium stearate, polyethylene glycol, PEG 6000, polyvinyl alcohol, potassium benzoate, sodium benzoate.
18. The pharmaceutical formulation according to claim 17, wherein PEG 6000 is used as the lubricant.
19. The pharmaceutical formulation according to claim 12, wherein the disintegrant is selected from a group comprising carboxymethyl cellulose calcium, carboxymethylcellulose sodium, microcrystalline cellulose, silicon dioxide, croscarmellose sodium, crospovidone, hydroxypropyl cellulose, methyl cellulose, povidone, magnesium aluminum silicate, starch or combinations thereof.
20. The pharmaceutical formulation according to claim 12, wherein the diluent is selected from a group comprising calcium carbonate, calcium sulphate, dibasic calcium phosphate, tribasic calcium phosphate, calcium sulphate, microcrystalline cellulose, lactose, magnesium carbonate, magnesium oxide, maltodextrin, maltose, mannitol, sodium chloride, sorbitol, starch, xylitol or combinations thereof.
21. The pharmaceutical formulation according to claim 12, wherein the effervescent acid is selected from organic acids such as citric acid, tartaric acid, malic acid, fumaric acid.
22. The pharmaceutical formulation according to claim 12, wherein the effervescent base is selected from basic agents such as sodium hydrogen carbonate, sodium carbonate, potassium carbonate and potassium hydrogen carbonate.
23. The pharmaceutical formulation according to claim 12, wherein said formulation comprises 5-30% cefdinir or pharmaceutically acceptable solvates, hydrates, enantiomers, racemates, organic salts, inorganic salts, polymorphs, crystal and amorphous forms of cefdinir and 1-20% organic base; 1-8% binder; 0.1-2% lubricant; 0.1-8% taste regulating agents, 0-25% diluent; 0-15% disintegrant; 0-10% humectant; 0.2-6% coloring agent and/or flavoring agent, 10-60% effervescent acid and 15-50% effervescent base with respect to the total amount of unit dose with respect to the total amount of unit dose.
24. The process for preparation of the pharmaceutical formulation according to the proceeding claims, wherein said process comprises mixing the components through dry blending method and powderizing them or granulation of the active agent cefdinir and various excipients through conventional wet and/or dry granulation methods and optionally compressing these in tablet compressing machine to obtain tablet forms.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP11721664.8A EP2566448B1 (en) | 2010-05-04 | 2011-05-02 | Efervescent formulations comprising cefdinir |
| US13/668,922 US20130129791A1 (en) | 2010-05-04 | 2012-11-05 | Preparations of effervescent formulations comprising second and third generation cephalosporin and uses thereof |
| US14/309,273 US20150140111A1 (en) | 2010-05-04 | 2014-06-19 | Preparations of effervescent formulations comprising second and third generation cephalosporin and uses thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| TR201003547 | 2010-05-04 | ||
| TR2010/03547 | 2010-05-04 |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/TR2011/000129 Continuation-In-Part WO2011139253A2 (en) | 2010-05-04 | 2011-05-02 | Pharmaceutical compositions comprising ceftibuten |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/TR2011/000124 Continuation-In-Part WO2011142731A2 (en) | 2010-05-04 | 2011-05-13 | Formulations comprising a third generation cephalosporin and clavulanic acid |
| US13/668,922 Continuation-In-Part US20130129791A1 (en) | 2010-05-04 | 2012-11-05 | Preparations of effervescent formulations comprising second and third generation cephalosporin and uses thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2011139252A2 true WO2011139252A2 (en) | 2011-11-10 |
| WO2011139252A3 WO2011139252A3 (en) | 2012-03-01 |
Family
ID=44276339
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/TR2011/000121 Ceased WO2011139249A2 (en) | 2010-05-04 | 2011-05-02 | Pharmaceutical composition comprising cefdinir |
| PCT/TR2011/000127 Ceased WO2011139252A2 (en) | 2010-05-04 | 2011-05-02 | Efervescent formulations comprising cefdinir |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/TR2011/000121 Ceased WO2011139249A2 (en) | 2010-05-04 | 2011-05-02 | Pharmaceutical composition comprising cefdinir |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP2566448B1 (en) |
| WO (2) | WO2011139249A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015140306A (en) * | 2014-01-28 | 2015-08-03 | 花王株式会社 | Solid composition |
| JP2018090640A (en) * | 2018-03-16 | 2018-06-14 | 花王株式会社 | Solid composition |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TR201010212A2 (en) * | 2010-12-08 | 2012-06-21 | Bi̇lgi̇ç Mahmut | Solid oral dosage form containing cefdinir. |
| CN103110595B (en) * | 2012-12-31 | 2014-05-28 | 广东博洲药业有限公司 | Cefdinir dispersible tablet and preparation method thereof |
| CN115607553B (en) * | 2021-07-16 | 2024-02-23 | 广州白云山天心制药股份有限公司 | Medicine containing cefdinir |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BE897864A (en) | 1982-09-30 | 1984-03-29 | Fujisawa Pharmaceutical Co | PROCESS FOR PRODUCING 7-SUBSTITUTED 3-VINYL-3-CEPHEM COMPOUNDS AND NOVEL PRODUCTS THUS OBTAINED |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PT1166651E (en) * | 1996-02-29 | 2005-04-29 | Fujisawa Pharmaceutical Co | FAST DISINTEGRATION GRANULES OF A SYNTHETIC SWEETENER CONTAINING SILICON ACID AND / OR SILICON DIOXIDE |
| TWI344847B (en) * | 2003-04-28 | 2011-07-11 | Takeda Chemical Industries Ltd | Composition for injection |
| WO2004104010A1 (en) * | 2003-05-20 | 2004-12-02 | Ranbaxy Laboratories Limited | Crystalline form of cefdinir |
| US20050131079A1 (en) * | 2003-12-10 | 2005-06-16 | Pujara Chetan P. | Cefdinir oral suspension |
| WO2006106529A1 (en) * | 2005-04-05 | 2006-10-12 | Lupin Limited | A co-spray dried composition of cefepime with base and process for preparation thereof |
| CN1706389A (en) * | 2005-05-26 | 2005-12-14 | 济南平志医药科技有限公司 | Effervescent cefdinir prepn and its prepn process |
| US20070128268A1 (en) * | 2005-12-07 | 2007-06-07 | Herwig Jennewein | Pharmaceutical compositions comprising an antibiotic |
| US20080103124A1 (en) * | 2006-10-25 | 2008-05-01 | Astellas Pharma Inc. | Cefdinir-containing pharmaceutical composition |
| DE102007002924A1 (en) * | 2007-01-19 | 2008-07-24 | Bayer Healthcare Ag | ß-lactam-containing formulations with increased stability in aqueous solution |
-
2011
- 2011-05-02 WO PCT/TR2011/000121 patent/WO2011139249A2/en not_active Ceased
- 2011-05-02 WO PCT/TR2011/000127 patent/WO2011139252A2/en not_active Ceased
- 2011-05-02 EP EP11721664.8A patent/EP2566448B1/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BE897864A (en) | 1982-09-30 | 1984-03-29 | Fujisawa Pharmaceutical Co | PROCESS FOR PRODUCING 7-SUBSTITUTED 3-VINYL-3-CEPHEM COMPOUNDS AND NOVEL PRODUCTS THUS OBTAINED |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015140306A (en) * | 2014-01-28 | 2015-08-03 | 花王株式会社 | Solid composition |
| JP2018090640A (en) * | 2018-03-16 | 2018-06-14 | 花王株式会社 | Solid composition |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2566448B1 (en) | 2015-06-17 |
| WO2011139249A3 (en) | 2012-03-01 |
| WO2011139249A2 (en) | 2011-11-10 |
| WO2011139252A3 (en) | 2012-03-01 |
| EP2566448A2 (en) | 2013-03-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2008320458B2 (en) | Stabilized pediatric suspension of carisbamate | |
| US8614315B2 (en) | Cefdinir and cefixime formulations and uses thereof | |
| EP2566448B1 (en) | Efervescent formulations comprising cefdinir | |
| EP2563340A2 (en) | Water soluble pharmaceutical composition | |
| EP2566449B1 (en) | Pharmaceutical compositions comprising ceftibuten | |
| EP2515850B1 (en) | Pharmaceutical compositions comprising cefdinir as an active agent | |
| US20130129791A1 (en) | Preparations of effervescent formulations comprising second and third generation cephalosporin and uses thereof | |
| WO2011093831A2 (en) | Effervescent formulations comprising cefprozil as active agent | |
| WO2011129792A1 (en) | Water dispersible formulations comprising cefpodoxime proxetil | |
| EP2566451B1 (en) | Pharmaceutical compositions comprising cefditoren pivoxil | |
| WO2012060786A2 (en) | Cefpodoxime proxetil formulations comprising viscosity agent | |
| WO2013109230A1 (en) | Pharmaceutical compositions comprising tadalafil | |
| WO2011078830A1 (en) | Rapidly dispersing effervescent formulation | |
| EP2481395A1 (en) | Sachet, effervescent tablet and dry syrup of otilonium | |
| EP2566450A2 (en) | Pharmaceutical compositions comprising cefetamet | |
| WO2014007775A1 (en) | A novel formulation having fast dissolution | |
| WO2012060787A1 (en) | Oral dosage forms comprising cefdinir and carboxymethyl cellulose calcium | |
| WO2013109201A1 (en) | Pharmaceutical compositions comprising cefprozil and clavulanic acid | |
| US9456981B2 (en) | Stable orally disintegrating tablets of hyoscyamine | |
| EP2663289A2 (en) | Cefpodoxime proxetil formulations comprising taste regulating agent | |
| WO2013109224A1 (en) | Pharmaceutical compositions comprising diclofenac | |
| WO2012078121A2 (en) | Solid oral dosage form comprising cefdinir | |
| WO2012060789A2 (en) | Production method for cefdinir formulations |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11721664 Country of ref document: EP Kind code of ref document: A2 |
|
| NENP | Non-entry into the national phase in: |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011721664 Country of ref document: EP |