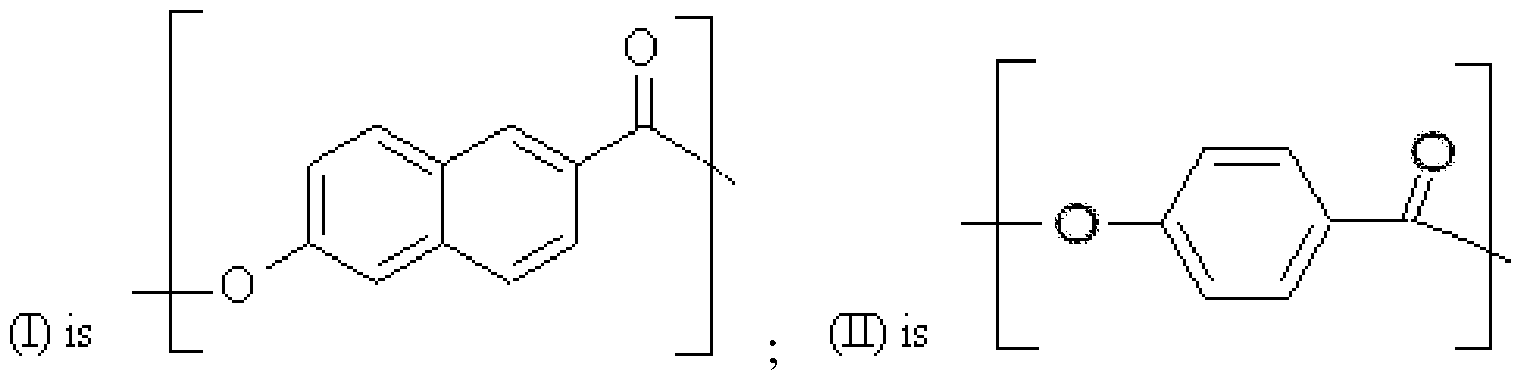WO2012012614A2 - Electrically conductive metal-coated fibers, continuous process for preparation thereof, and use thereof - Google Patents
Electrically conductive metal-coated fibers, continuous process for preparation thereof, and use thereof Download PDFInfo
- Publication number
- WO2012012614A2 WO2012012614A2 PCT/US2011/044817 US2011044817W WO2012012614A2 WO 2012012614 A2 WO2012012614 A2 WO 2012012614A2 US 2011044817 W US2011044817 W US 2011044817W WO 2012012614 A2 WO2012012614 A2 WO 2012012614A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fiber
- metal
- plated
- nickel
- coating
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/38—Polymers
- C09K19/3804—Polymers with mesogenic groups in the main chain
- C09K19/3809—Polyesters; Polyester derivatives, e.g. polyamides
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C18/00—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating
- C23C18/16—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating by reduction or substitution, e.g. electroless plating
- C23C18/1601—Process or apparatus
- C23C18/1633—Process of electroless plating
- C23C18/1635—Composition of the substrate
- C23C18/1639—Substrates other than metallic, e.g. inorganic or organic or non-conductive
- C23C18/1641—Organic substrates, e.g. resin, plastic
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C18/00—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating
- C23C18/16—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating by reduction or substitution, e.g. electroless plating
- C23C18/1601—Process or apparatus
- C23C18/1633—Process of electroless plating
- C23C18/1635—Composition of the substrate
- C23C18/1639—Substrates other than metallic, e.g. inorganic or organic or non-conductive
- C23C18/1642—Substrates other than metallic, e.g. inorganic or organic or non-conductive semiconductor
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C18/00—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating
- C23C18/16—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating by reduction or substitution, e.g. electroless plating
- C23C18/1601—Process or apparatus
- C23C18/1633—Process of electroless plating
- C23C18/1646—Characteristics of the product obtained
- C23C18/165—Multilayered product
- C23C18/1653—Two or more layers with at least one layer obtained by electroless plating and one layer obtained by electroplating
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C18/00—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating
- C23C18/16—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating by reduction or substitution, e.g. electroless plating
- C23C18/18—Pretreatment of the material to be coated
- C23C18/20—Pretreatment of the material to be coated of organic surfaces, e.g. resins
- C23C18/2006—Pretreatment of the material to be coated of organic surfaces, e.g. resins by other methods than those of C23C18/22 - C23C18/30
- C23C18/2046—Pretreatment of the material to be coated of organic surfaces, e.g. resins by other methods than those of C23C18/22 - C23C18/30 by chemical pretreatment
- C23C18/2073—Multistep pretreatment
- C23C18/2086—Multistep pretreatment with use of organic or inorganic compounds other than metals, first
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C18/00—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating
- C23C18/16—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating by reduction or substitution, e.g. electroless plating
- C23C18/18—Pretreatment of the material to be coated
- C23C18/20—Pretreatment of the material to be coated of organic surfaces, e.g. resins
- C23C18/22—Roughening, e.g. by etching
- C23C18/26—Roughening, e.g. by etching using organic liquids
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C18/00—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating
- C23C18/16—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating by reduction or substitution, e.g. electroless plating
- C23C18/18—Pretreatment of the material to be coated
- C23C18/20—Pretreatment of the material to be coated of organic surfaces, e.g. resins
- C23C18/28—Sensitising or activating
- C23C18/30—Activating or accelerating or sensitising with palladium or other noble metal
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C18/00—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating
- C23C18/16—Chemical coating by decomposition of either liquid compounds or solutions of the coating forming compounds, without leaving reaction products of surface material in the coating; Contact plating by reduction or substitution, e.g. electroless plating
- C23C18/31—Coating with metals
- C23C18/32—Coating with nickel, cobalt or mixtures thereof with phosphorus or boron
- C23C18/34—Coating with nickel, cobalt or mixtures thereof with phosphorus or boron using reducing agents
- C23C18/36—Coating with nickel, cobalt or mixtures thereof with phosphorus or boron using reducing agents using hypophosphites
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D5/00—Electroplating characterised by the process; Pretreatment or after-treatment of workpieces
- C25D5/54—Electroplating of non-metallic surfaces
- C25D5/56—Electroplating of non-metallic surfaces of plastics
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M11/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with inorganic substances or complexes thereof; Such treatment combined with mechanical treatment, e.g. mercerising
- D06M11/83—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with inorganic substances or complexes thereof; Such treatment combined with mechanical treatment, e.g. mercerising with metals; with metal-generating compounds, e.g. metal carbonyls; Reduction of metal compounds on textiles
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B1/00—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors
- H01B1/02—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors mainly consisting of metals or alloys
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K9/00—Screening of apparatus or components against electric or magnetic fields
- H05K9/0073—Shielding materials
- H05K9/0081—Electromagnetic shielding materials, e.g. EMI, RFI shielding
- H05K9/009—Electromagnetic shielding materials, e.g. EMI, RFI shielding comprising electro-conductive fibres, e.g. metal fibres, carbon fibres, metallised textile fibres, electro-conductive mesh, woven, non-woven mat, fleece, cross-linked
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2219/00—Aspects relating to the form of the liquid crystal [LC] material, or by the technical area in which LC material are used
- C09K2219/01—Aspects relating to the form of the liquid crystal [LC] material, or by the technical area in which LC material are used in the form of fibres, e.g. fibres after polymerisation of LC precursor
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/29—Coated or structually defined flake, particle, cell, strand, strand portion, rod, filament, macroscopic fiber or mass thereof
- Y10T428/2913—Rod, strand, filament or fiber
- Y10T428/2933—Coated or with bond, impregnation or core
- Y10T428/2964—Artificial fiber or filament
- Y10T428/2967—Synthetic resin or polymer
- Y10T428/2969—Polyamide, polyimide or polyester
Definitions
- This application relates to electrically conductive metal-plated fibers and continuous processes of preparing metal-plated fibers, as well as uses of the provided fibers or other fibers prepared by the provided processes.
- Electrical wires are typically made of highly conductive metals, such as copper. These metals afford the highest electrical conductivity for signal and power transfer, and can also be used for electromagnetic interference (EMI) shielding applications.
- EMI electromagnetic interference
- the weight of metal wires e.g., copper has a density of 8.96 g cm "
- Such applications include, but are not limited to, aerospace applications.
- Some known efforts to reduce the weight of electrical wiring systems have involved replacing standard gauge copper wire (eg., 22 gauge) with a smaller gauge wire (e.g., 26 or 28 gauge). However, because thinner wires do not have the necessary mechanical strength and durability required for many applications, replacing the gauge of wiring is typically not a feasible solution to the problem.
- Metal-coated polymer fibers have been proposed as a solution to the need in the art for improved conductive materials.
- Metal-coated fibers are typically made by metallizing poly(p-phenylene benzobisoxazole) (Zylon®) or poly(p- phenylene terephthalamide) (Kevlar®) fiber with highly conductive metals. Because the interior fiber has a high tensile strength and Young's modulus, low density, and small diameter, such metal-coated polymer fibers offer benefits over traditional conductive wires (such as copper wires) in flexibility, weight savings, and durability.
- EMI shielding Braided EMI shields are traditionally made from standard copper wire, but utilizing metal-clad fibers instead provides weight savings and can provide better shielding at high frequencies due to increased braid coverage and less windowing.
- metal-plated liquid crystalline polymer fibers comprising (a) a melt processable, thermotropic wholly aromatic liquid crystalline polymer fiber; (b) at least one coating of electroless-plated metal on said fiber; and (c) optionally, at least one coating of electroplated metal on said fiber.
- the wholly aromatic liquid crystalline polymer fiber is a polyester consisting essentially of repeating units of (I) and (II):
- the wholly aromatic liquid crystalline polymer fiber is selected from Vectran® fiber, Ekonol® fiber, and Xydar® fiber, and may be monofilament fiber or multi-filament fiber.
- the provided process comprises (a) etching the surface of a melt processable wholly aromatic liquid crystalline polymer fiber by contacting it with alkaline solution in the presence of ultrasonic agitation, wherein the alkaline solution does not comprise surfactant or solubilizer; (b) seeding the etched surface with catalyst by contacting the etched fiber with one or more electroless plating catalysts; (c) reducing the catalyst by contacting said fiber with a reducing solution; (d) electrolessly plating at least one coating of metal on said fiber; and (e) optionally, electroplating at least one coating of metal on said fiber.
- Metal-plated fibers prepared by the provided process show one or more of thermal stability, thermo-oxidative stability, mechanical flexibility, durability, strength, electrical conductivity, small diameter, and light weight.
- a polymeric article having electromagnetic interference shielding effectiveness comprising a provided metal- plated fiber or other fiber prepared by the provided process.
- the provided article comprises: (a) a melt processable, thermotropic wholly aromatic liquid crystalline polymer fiber; (b) at least one coating of electroless-plated metal on said fiber; and (c) optionally, at least one coating of electroplated metal on said fiber; wherein the fiber of (b) or (c) is adapted to be woven or braided to provide a polymeric article having electromagnetic interference shielding effectiveness.
- Figure 1 illustrates the chemical structure of one example (Vectran® fiber) of a wholly aromatic polyester liquid crystalline fiber (wherein x and y are variable) that may be used in the provided process to prepare a provided metal-plated fiber, a provided polymeric article having EMI shielding effectiveness, or combinations thereof;
- Figure 2 depicts a schematic drawing of a cross-section of one example of a metal-coated monofilament that may be prepared according to the provided process;
- Figure 3 depicts a schematic of one embodiment of a continuous process of producing metal-coated fibers, wherein in some embodiments, one or more optional rollers, ultrasonic agitation, tension control (for example, below 50 g), and combinations thereof are employed in at least the surface modification step.
- the fiber may be continuously transferred from bath to bath utilizing one or more rollers, wherein tension control is achieved by adjusting fiber transfer speed between each bath.
- de-ionized water rinsing between chemical baths may be used to remove any cross-contamination
- Figure 4 depicts a schematic of metallization of a bundle of monofilaments by the provided process.
- electrically conductive metal-plated high-temperature aromatic polymer fibers comprising (a) at least one coating of electroless- plated metal on the fiber and (b) optionally, at least one coating of electroplated metal.
- Said fibers are, in some embodiments, prepared by the provided continuous fabrication process comprising depositing one or more uniform layers of metals onto fibers through one or more of electroless and electroplating methods.
- Deposited electroless-plated metals may, in some embodiments, be selected from nickel, copper, silver, and alloys thereof. In some embodiments, the provided fibers comprise at least one coating of electroless-plated nickel/phosphorous alloy. Deposited electroplated metals may, in some embodiments, be selected from tin, nickel, copper, silver, gold, and alloys thereof. Whether deposited by electroless plating or electroplating methods, metals may be deposited in one, two, three, four, or more layers, each layer being of a metal that is the same as or different from the previous layer. In some embodiments, the deposited metal layers may have a cumulative thickness of from about ⁇ to about 10 ⁇ .
- the cumulative thickness of the deposited metal layers may be 1 ⁇ , 2 ⁇ , 3 ⁇ , 4 ⁇ , 5 ⁇ , 6 ⁇ , 7 ⁇ , 8 ⁇ , 9 ⁇ , 10 ⁇ , or combinations thereof.
- the fibers coated by the metal(s) are liquid crystalline polymer fibers.
- the fibers are melt processable, thermotropic wholly aromatic liquid crystalline polymer fibers. Examples include, but are not limited to, Vectran® fiber (Kuraray), Ekonol® fiber (Saint-Gobain), and Xydar® fiber (Solvay). Good results have been achieved with Vectran fiber.
- the process may be used on other types of high temperature aromatic fibers, such as Zylon® (PBO) fiber and Kevlar® (aramid) fiber, PEEK (polyether ether ketone) fiber, Ultem® (polyetherimide) fiber, and PPS
- Zylon® PBO
- Kevlar® Kevlar®
- PEEK polyether ether ketone
- Ultem® polyetherimide
- wholly aromatic polyester liquid crystalline fibers may be used in the provided process to form metal-plated fibers.
- Wholly aromatic polyester liquid crystalline polymers are known in the art, and many are commercially available. Examples include, but are not limited to, those comprising moieties derived from one or more of 6-hydroxy-2-naphthoic acid; 4,4'-biphenol; hydroquinone; p-hydroxybenzoic acid;
- suitable wholly aromatic liquid crystalline polymer fibers are melt processable, thermotropic polyesters of 2,6-dicarboxynaphthalene and p-oxybenzoyl moieties, or ring- substituted derivatives thereof. Accordingly, suitable fibers for use in the provided process to form metallized fibers consist essentially of repeating units of (I) and (II):
- At least one hydrogen of an aromatic ring of (I), (II), or both may optionally be substituted with an alkyl group, an alkoxy group, a halogen, or combinations thereof.
- Vectran fiber and uses thereof to form metallized fibers will be described.
- the scope of this present application is not intended to be limited by such illustration. Rather, the scope is intended to encompass other high temperature aromatic polymers, including without limitation, other wholly aromatic polyester liquid crystalline fibers.
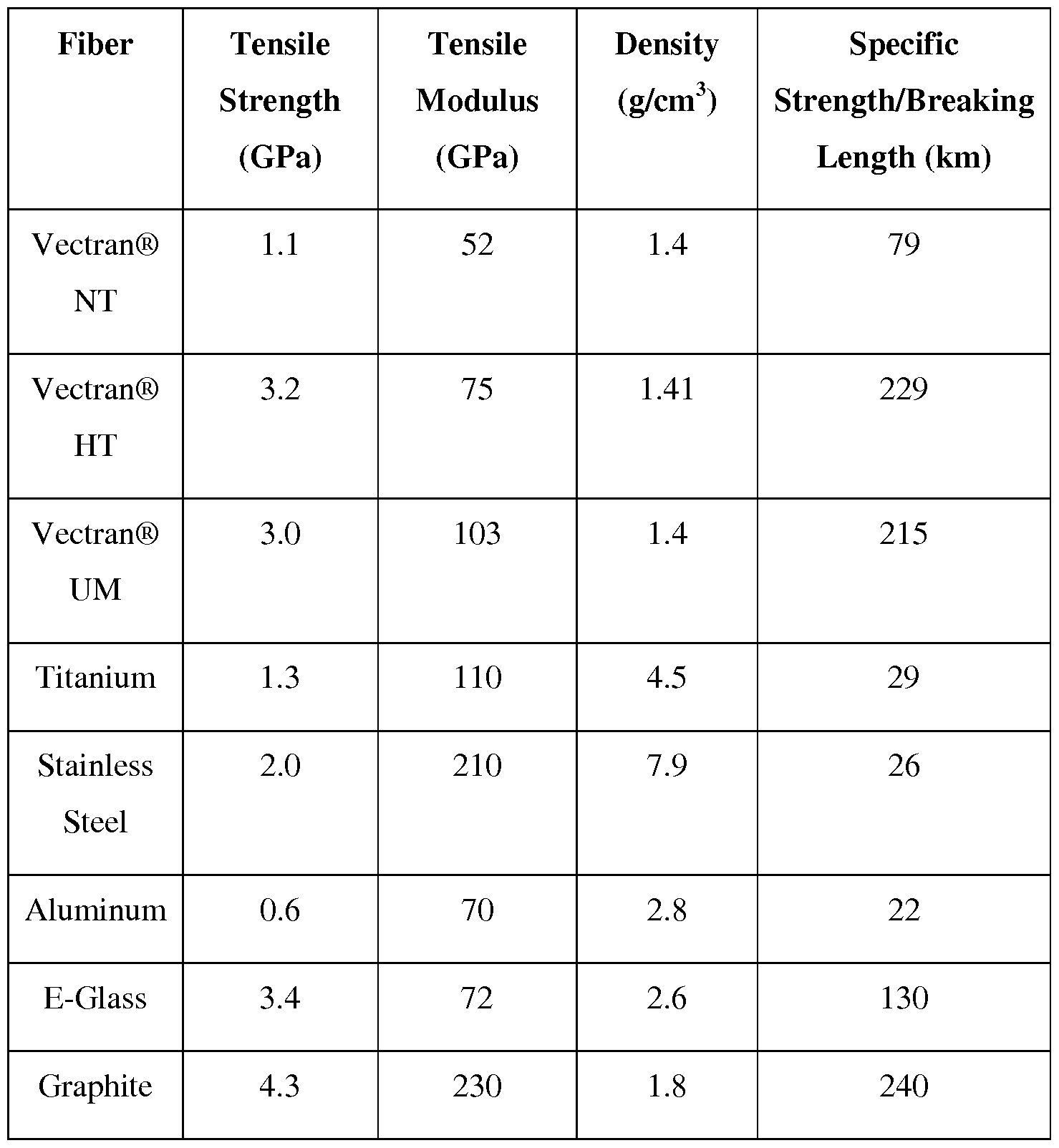
- Vectran fiber is a highly oriented multi-filament polyester-polyarylate liquid crystalline polymer fiber exhibiting a very high tensile strength and high melting temperature. Vectran fiber is three to five times stronger than other polyesters and is stronger than aramid fibers (Kevlar). In addition to having high strength, Vectran fiber has excellent rigidity, tenacity retention, abrasion resistance, moisture resistance, and property retention over a broad range of temperatures and chemical environments. Some properties of Vectran fiber, as compared to other high strength fibers, are illustrated in Tables 1-3. Table 1
- Vectran fiber is different from other high strength fibers, such as aramid fiber, poly(p-phenylene-2,6-benzobisoxazole) (PBO) fiber, and ultra-high molecular weight polyethylene (HMPE) fiber.
- Aramid fiber Kevlar®, DuPont
- PBO fiber Zylon®, Toyobo
- HMPE fiber Spectra®, Honeywell
- Vectran fiber is a thermotropic liquid crystalline polymer formed by melt-spinning through fine diameter capillaries, a process causing molecular chains to orient parallel to the fiber axis without chain folding.
- Vectran fiber is hydrophobic, resistant to hydrolytic degradation, and shows good tenacity retention in aggressive chemical exposure. Because the moisture absorbed by a fiber during the original manufacturing or metallization processes will remain with the fiber after metallization, hydrolytic stability of fibers is important for long-term stability, especially when the metallized fiber will be used at elevated temperatures. Vectran fiber has higher hydrolytic stability than other fibers, including Kevlar® and Zylon® fibers. Additionally, it has been reported that the tenacity retention of Vectran fiber is far superior to standard Aramid fiber, like Kevlar, after 300 hours thermal exposure at 250°C.
- the highly conductive metal-coated polymer fibers prepared by the provided process have advantages over copper wires in terms of flexibility, light weight, strength, durability, and tailored electrical/mechanical properties.
- the provided metal-plated fibers also have advantages over other metal-coated fibers.
- provided metal-coated polymers may have higher long-term hydrolytic stability, higher temperature capability, higher conductivity, or combinations thereof, with respect to metallized Kevlar, metallized Zylon, and other metallized fibers.
- metallized fibers such as Vectran may be used in applications such as EMI shielding.
- metal-plated Vectran may be woven or braided into a polymeric article having EMI shielding
- Vectran may be attractive substrates for metallization, there are challenges to metallizing such fibers.
- Vectran fiber is unique with respect to its formation and its properties, and such uniqueness presents challenges to its use in applications.
- the fiber is hydrophobic, exhibits high bundle stiffness, is sensitive to static, has thermoplastic properties, and it has a multi- layered fiber structure, all of which create unique challenges to processes of metallization.
- known processes for metallization of polymer fibers are not suitable for metallization of fibers such as Vectran® fibers
- fibers contemplated to be suitable for use in the provided process include, but are not limited to, PEEK (polyether ether ketone) fiber; Ultem® (polyetherimide) fiber; PPS (polyphenylene sulfide) fiber; and melt processable, thermotropic wholly aromatic liquid crystalline polymer fibers.
- the continuous process of metallizing aromatic polymer fibers comprises (a) surface modification (b) catalyzing, (c) reduction, (d) electroless plating of metal, and (e) optionally, electroplating metal.
- the metal- plated fiber comprises one or more coatings of electrolessly-plated metal, each coating being of the same or different metal as the prior coating.
- the metal-plated fiber further comprises one or more coatings of electroplated metal, each coating being of the same or different metal as the prior coating.
- the electrical conductivity of the resulting metal-coated fiber can be tuned over a very wide range depending on the plating thickness and composition of the metal coating.
- resistance of a metal-coated fiber may range from about 0.5 to about 300 Ohm per foot. Accordingly, resistance can be from 0.5-1, 1-5, 5-10, 10-20, 20-30, 30-40, 40-50, 50-60, 60-70, 70-80, 80-90, 90-100, 100-110, 110-120, 120-130, 130-140, 140-150, 150-160, 160-170, 170-180, 180-190, 190-200, 200- 210, 210-220, 220-230, 230-240, 240-250, 250-260, 260-270, 270-280, 280-290, 290-300 Ohms per foot, and combinations thereof.
- a yarn of polymeric fiber (whether a monofilament or multifilament tow) can be made highly conductive in a single continuous reel-to-reel method.
- the provided process allows highly conductive metals to be incorporated onto a polymer fiber, giving rise to electrical conductivity.
- the goal is to produce a light weight, mechanically robust material that contains a desired volume fraction of metal but has a metallic conductivity comparable to current state-of-the-art high strength copper alloy, such as CS-95 alloys.
- one or more highly conductive metals are deposited onto polymer fibers by an autocatalytic deposition process, commonly referred to as
- electroless plating The autocatalytic deposition process allows for uniform deposition of metal onto catalyzed surfaces of objects that are immersed in a solution.
- the electroless plating process occurs without application of an electrical current. Instead, deposition occurs through a controlled electrochemical reduction process.
- Various conductive metals can be deposited. In some embodiments, one or more of copper, nickel, silver, gold, and alloys thereof, may be deposited by the provided process. In some embodiments, one or more layers of metal may be deposited (via electroplating techniques) onto the electrolessly-plated metal coating(s).
- the provided process may be applied to wholly aromatic polyester liquid crystalline fibers (including, but not limited to Vectran® fibers) in order to produce metal-plated liquid crystalline polymer fibers.
- a continuous process for preparing metal-plated liquid crystalline polymer fibers comprising (a) etching the surface of a melt processable, thermotropic wholly aromatic liquid crystalline polymer fiber by contacting it with alkaline solution in the presence of ultrasonic agitation, wherein the alkaline solution does not comprise surfactant or solubilizer; (b) contacting the fiber of (a) with one or more electroless plating catalysts selected from salts of silver, nickel, gold, platinum, osmium, palladium, and rhodium; (c) contacting the fiber of (b) with a reducing solution; (d) electrolessly plating at least one coating of metal on the fiber of (c), the electroless-plated metal selected from nickel, copper, silver, and alloys thereof
- suitable fibers for use in the provided process consist essentially of repeating units of (I) and (II):
- At least one hydrogen of an aromatic ring of (I), (II), or both may optionally be substituted with an alkyl group, an alkoxy group, a halogen, or combinations thereof.
- One commercial example of such fiber is Vectran® fiber.
- the purpose of surface modification is to provide some interlocking mechanism on the Vectran® fiber for chemical and/or physical bonding with the
- Vectran® fiber is a thermotropic liquid crystalline polymer fiber which provides excellent resistance to a wide range of organic and inorganic chemicals.
- Conventional processes (such as those described in U.S. 5,302,415; 5,422,142; 5,453,299; 5,935,706; and 6,045,680) to uniformly metallize multiple-filament polymeric fibers of polyaramid, polyamide, or polyester involve strong acid surface preconditioning (often in combination with surfactant to help the acid to penetrate fiber bundles) followed by electroless nickel coating.
- US Patents 6,403,211 and 6,923,919 disclose a process of how to effectively etch a liquid crystalline polymer film with a heated potassium hydroxide (KOH) bath with ethanolamine solubilizer.
- KOH potassium hydroxide
- a LCP film is preconditioned insufficiently by KOH solution alone. It was unexpectedly observed, however, that the methods described with respect LCP films are not applicable to LCP fibers.
- the provided process allows for successful modification of Vectran® fiber surfaces with a heated alkaline solution alone (i.e., without any solubilizer or surfactant).
- the provided process comprises contacting the fiber with alkaline solution.
- the alkaline solution may be one or more of a strong base, including but not limited to, bases such as lithium hydroxide (LiOH), sodium hydroxide (NaOH), potassium hydroxide (KOH), rubidium hydroxide (RbOH), cesium hydroxide (CsOH), calcium hydroxide (Ca(OH)2), strontium hydroxide (Sr(OH)2), barium chloride (Ba(OH)2).
- bases such as lithium hydroxide (LiOH), sodium hydroxide (NaOH), potassium hydroxide (KOH), rubidium hydroxide (RbOH), cesium hydroxide (CsOH), calcium hydroxide (Ca(OH)2), strontium hydroxide (Sr(OH)2), barium chloride (Ba(OH)2).
- bases such as lithium hydroxide (LiOH), sodium hydroxide (NaOH), potassium hydroxide (KOH), rubidium hydroxide (RbOH), cesium hydroxide (CsOH), calcium hydroxide
- concentration, and the solution processing temperature may be selected to provide the desired characteristics.
- temperature may be from about 40°C-45°C, 45°C-50°C, 50°C-55°C, 55°C-60°C, 60°C-65°C, 65°C-70°C, 70°C- 75°C, 75°C-80°C, 80°C-85°C, 85°C-90°C, 90°C-95°C, 95°C-100°C, and combinations thereof.
- the temperature may be from about 45°C to 65°C; alternatively, from about 55°C to 65°C; alternatively, from about 50°C to 80°C; alternatively, from about 80°C to 100°C.
- the KOH solution has a concentration of from about 20 wt% to about 75 wt%, wherein the concentration is selected to avoid extensive fiber damage.
- concentration may be from 20-25 wt%, 25-30 wt%, 30-35 wt%, 35-40 wt%, 40-45 wt%, 45-50 wt%, 50-55 wt%, 55-60 wt%, 60-65 wt%, 65-70 wt%, 70-75 wt%, and combinations thereof.
- the concentration may be from about 30 wt% to about 45 wt%.
- the concentration may be from about 45 wt% to about 60 wt%.
- Vectran® monofilaments due to the small diameter of Vectran® monofilaments, surface modification as little as one micron deep will result in 16% loss of the whole fiber strength. Therefore, it is important that etching conditions be selected such that the KOH solution can etch each filament effectively and uniformly in as short a period of time as possible. In some embodiments, KOH etching should occur simultaneously with ultrasonic agitation.
- Vectran fiber is available in 5, 20, 40 ,80 and higher monofilament tows, and the provided processes may be used on the same to provide metal-clad Vectran fibers having a variable number of monofilaments. Good results have been obtained by etching a 40 monofilament tow of Vectran® fiber, while
- agitation may be from about 25-45 KHz; alternatively, from about 45-65 KHz; alternatively, from about 65-85 KHz; alternatively, from about 85-105 KHz; alternatively, from about 105-120 KHz.
- the period of time may be from about 50 to 100 seconds; alternatively, from about 100 to 200 seconds; alternatively, from about 10 to 50 seconds.
- a favorable KOH solution etching environment may be achieved with the combination of mechanical agitation arising due to continuous movement of yarn monofilaments during operation of the continuous process with additional agitation created by ultrasound.
- mechanical agitation arising due to continuous movement of yarn monofilaments during operation of the continuous process with additional agitation created by ultrasound.
- one or more optional rollers may be used to aid in the surface modification of the Vectran® fiber.
- the rollers may be selected from cylindrical and non-cylindrical rollers.
- a non-cylindrical roller may have a transverse cross-section having a triangular, hexagonal, octagonal, or other suitable shape adapted to, when in operation, provide alternating levels of tension on yarn.
- one or more rollers such as those described in US2008/0280045 Al may be used in some embodiments.
- the one or more rollers may be used to continuously transfer the Vectran® fiber from one chemical bath to another chemical bath, from a chemical bath to a rinse bath, from a rinse bath to chemical bath, and combinations thereof, which provides mechanical agitation to open up the fiber tow for better solution penetration.
- tension control of the continuously moving Vectran® fiber may be achieved by adjusting transfer speed between each bath.
- the catalysis process comprises seeding a catalyst onto the Vectran® fiber surface to initiate the electroless plating process.
- a catalyst For purposes of illustration, palladium (Pd) catalyst will be discussed. However, one of skill in the art will recognize that other catalysts may alternatively be used. For example, it is contemplated that suitable catalysts may be selected from salts of silver, nickel, gold, platinum, osmium, palladium, and rhodium.
- the fiber substrate is immersed in a mixed acidic colloidal solution of stannous chloride (SnCl 2 ) sensitizer and palladium chloride (PdCl 2 ) catalyst.
- the Sn(II) will be oxidized to Sn(IV) while the Pd(II) will be reduced back to Pd, and the Pd nucleus will be readily absorbed onto the fiber surface as the working catalyst.
- the initial nucleation sites generated by a separate Sn-Pd process may be as much as an order of magnitude more numerous than those produced by the mixed Sn-Pd approach.
- the higher the number of nucleation sites the better the metal-to- substrate adhesive properties.
- the etched fiber is immersed in a dilute catalyst solution for a sufficient period of time to allow the catalyst to migrate and penetrate into the etched fiber structure.
- the catalyst solution is a palladium chloride (PdCl 2 )/hydrochloric acid (HC1) solution and the Pd ions migrate and penetrate into the etched fiber structure.
- a suitable period for immersion may be from about 1-360 minutes. Accordingly, immersion may be from about 1- 30 seconds, 30-60 seconds, 60-90 seconds, 90-120 seconds, 120-150 seconds, 150-180 seconds, 180-210 seconds, 210-240 seconds, 240-270 seconds, 270-300 seconds, 300-330 seconds, 330-360 seconds, and combinations thereof. In some embodiments, immersion may be from 2-3 minutes, 3-4 minutes, 4-5 minutes, and combinations thereof.
- the acid/catalyst solution may comprise from about 0.01 to 0.5 g/L of catalyst.
- the catalyst concentration may be from about 0.01-0.05 g/L, 0.05-0.10 g/L, 0.10-0.15 g/L, 0.15-0.20 g/L, 0.20-0.25 g/L, 0.25-0.30 g/L, 0.30-0.35 g/L, 0.35-0.40 g/L, 0.40-0.45 g/L, 0.45-0.50 g/L, and combinations thereof.
- Good results have been obtained with a catalyst concentration of from about 0.1 to 0.3 g/L.
- the acid/catalyst solution may also comprise one or more surfactants (e.g., sodium lauryl sulfate or ammonia lauryl sulfate) to facilitate catalyst absorption onto the fiber surface.
- surfactants e.g., sodium lauryl sulfate or ammonia lauryl sulfate
- concentrations of catalyst in the acid/catalyst solution and period of immersion may be varied to accommodate different properties and characteristics of the specific catalyst chosen.
- the reducing solution comprises from about 0.01 wt to about 0.10 wt of reducing agent.
- the reducing agent concentration may be from about 0.01-0.05 wt , 0.05-0.10 wt , and combinations thereof. Good results have been obtained using a reducing agent concentration of from about 0.02 to 0.03 wt .
- immersion may be less than 60 seconds.
- immersion may be from about 15-60 seconds. Good results have been obtained when immersion is less than 30 seconds.
- reducing agents other than sodium borohydride and dimethylamine borane may be utilized and that concentrations of reducing agent in the reducing solution and period of immersion may be varied to accommodate different properties and characteristics of the specific reducing agent chosen.
- Electroless plating is an autocatalytic deposition process that places metal onto objects that are immersed in a plating solution, wherein a uniform metallic coating is deposited conformably onto catalytic surfaces under a controlled electrochemical reduction process without applying an electrical current. Electroless plating is, in a general manner, well known. However, challenges nevertheless remain, such as obtaining good adhesion of the plated metal to the fiber surface.
- the provided process achieves good adhesion of metal, in part, through the choice of plating alloy.
- a nickel sulfate based-electroless nickel solution (8 to 10 wt Phosphorus content) may be used for nickel metallization.
- Such a plating solution is capable of depositing a 20 micron nickel coating onto a catalyzed Vectran® fiber at 88°C in one hour.
- the suitability of nickel-phosphorus alloy coatings was surprising given the prior art teachings regarding electroless plating of fibers.
- US 5,935,706 and US 6,045,680 teach against use of nickel-phosphorus alloys to coat fibers.
- nickel-phosphorus alloys may be deposited.
- metals and metal alloys other than nickel may also be deposited by electroless plating. Examples include copper, silver and alloys thereof. In some embodiments, more than one layer of metal may be deposited by electroless plating.
- electroless plating techniques are used to provide a uniform metal coating over the fiber surface.
- a uniform metal coating may be greater than 85% of the fiber surface area.
- the coating may be 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100%, or combinations thereof, of the fiber surface area.
- the deposited metal coats the fiber. It does not, however, form a matrix in which the fiber is embedded or encased within metal and functions to reinforce the metal matrix.
- the provided process comprises preparing metal-plated polymer fibers with electroplated metal. In some embodiments, the provided process comprises preparing metal-plated polymer fibers without electroplated metal.
- a fiber having a uniform coating of electroplated metals may be achieved by, among other things, controlling voltage during the electroplating process.
- the resulting metal-plated fiber may be further processes by known methods.
- melt-processable, thermotropic wholly aromatic liquid crystalline polymer fiber 301 is sequentially transported through an etching station 302 in which the fiber is contacted with alkaline solution and ultrasonic agitation (not labeled); through a water rinse station 303; through a catalyst seeding station 304 in which the fiber is contacted with one or more electroless plating catalysts; through a reducing station 305 in which the fiber is contacted with a reducing solution; through a water rinse station 306; through an electroless plating station 307 wherein one or more coatings of electroless metal are deposited onto the fiber; through a water rinse station 308; through an electroplating station 309 wherein one or more coatings of electroplated metal are deposited onto the one or more coatings of electroless metal, the sum of which produces a provided metal-plated fiber 310.
- melt-processable, thermotropic wholly aromatic liquid crystalline polymer fiber 301 is sequentially transported through an etching station 302 in which the fiber is contacted with alkaline solution and ultras
- metal-plated melt processable wholly aromatic polyester liquid crystalline polymer fibers as well as a continuous process for preparation of electrically conductive metal-coated fibers.
- metal- plated polymer fibers consisting essentially of repeating units of (I) and (II):
- the metal-plated fiber prepared by a continuous process comprising (a) etching the surface of the fiber by contacting it with alkaline solution in the presence of ultrasonic agitation, wherein the alkaline solution does not comprise surfactant or solubilizer; (b) seeding the etched surface of (a) with catalyst by contacting the fiber with one or more electroless plating catalysts selected from salts of silver, nickel, gold, platinum, osmium, palladium, and rhodium; (c) reducing the catalyst by contacting the fiber of (b) with a reducing solution; (d) electrolessly plating at least one coating of metal on the fiber of (c), the electroless-plated metal selected from nickel, copper, silver, and alloys thereof; and (e) optionally,
- at least one hydrogen of an aromatic ring of (I), (II), or both, may be substituted with an alkyl group, an alkoxy group, a halogen, or combinations thereof.
- At least one hydrogen of an aromatic ring of (I), (II), or both may optionally be substituted with an alkyl group, an alkoxy group, a halogen, or combinations thereof.
- a provided polymeric article comprises a liquid crystalline polymer fiber with at least one coating of electroless-plated metal and at least one coating of electroplated metal.
- a provided polymeric article is adapted to provide a shielding effectiveness of from about 35 to about 80 decibels (db) across a frequency range of from about 0.1 to about 3000 MHz. Accordingly, shielding effectiveness may be from about 35-40 db, 40-45 db, 45-50 db, 50-55 db, 55-60 db, 60-65 db, 65-70 db, 70-75 db, 75-80 db, and combinations thereof.
- Vectran® fiber An illustrative procedure for preparing metallized Vectran® fiber using a continuous wet chemical process may be summarized as:
- Vectran® fiber is etched in a strong alkaline bath, such as potassium hydroxide or sodium hydroxide with a 30 to 60 wt concentration, with a soaking duration of 10 to 300 seconds.
- the alkaline etching solution is preheated to 45°C to 75°C under ultrasonic agitation at 25 to 120 KHz.
- the etched fiber is then thoroughly cleaned with plenty of deionized rinsed water for 30 to 240 seconds.
- the wet fiber is then soaked in an acidic catalyst aqueous solution, such as palladium, silver or nickel ion solution with weight concentration of 0.01 to 0.5 g/1 for 60 to 120 seconds.
- an acidic catalyst aqueous solution such as palladium, silver or nickel ion solution with weight concentration of 0.01 to 0.5 g/1 for 60 to 120 seconds.
- Surfactant such as sodium lauryl sulfate may also be added into the solution to facilitate the catalyst absorption to the fiber surface.
- the catalyzed fiber is then neutralized in a dilute acid bath, such as hydrochloric or sulfuric acid, and subsequently rinsed thoroughly with deionized water for 30 to 240 seconds before being immersed in the electroless plating solution.
- a dilute acid bath such as hydrochloric or sulfuric acid
- Electroless nickel, silver or copper can all be used for building up the conductive layer on the Vectran® fiber.
- the resulting conductive fiber can then be electroplated with copper, nickel, silver and gold to enhance its electrical
- the Vectran yarn was first etched in a 45 wt potassium hydroxide bath at 62°C under 40 KHz ultrasonic agitation for 80 seconds. The yarn was then thoroughly rinsed using deionized water. Subsequently, the wet yarn was passed through a series of process baths, including 240 seconds each in palladium catalyst bath, sodium borohydride reduction bath, hydrochloric neutralizing bath and deionized water rinse bath. Nickel sulfate/sodium hypophosphite base electroless nickel was used to for the nickel undercoating coating on the treated yarn.
- the solution was made up by using 6 vol of nickel sulfate, 15 vol of sodium hypophosphite, and 79 Vol% of deionized water.
- the bath was operated in 190°F at a PH value of 4.85 and constantly filtered through a 1 um filter. Generally, a soaking duration of two to three minutes will coat the yarn uniformly with a layer of phosphorus based electroless nickel of 0.5 to 0.75 micrometer.
- This electroless nickel-coated Vectran yarn exhibited an electrical resistance of -250 ohm/ft and was found conductive enough to facilitate the subsequent electroplating. Additional two minutes acid copper sulfate electroplating operated at a current of 3.75 amp resulted in a very highly conductive and uniform yarn with resistance of 2.06 ohm/ft.
- the Vectran yarn was first etched in a 45 wt potassium hydroxide bath at 62°C under 40 KHz ultrasonic agitation for 80 seconds. The yarn was then thoroughly rinsed using deionized water. Subsequently, the wet yarn was passed through a series of process baths, including 240 seconds each in palladium catalyst bath, sodium borohydride reduction bath, hydrochloric neutralizing bath and deionized water rinse bath. Nickel sulfate/sodium hypophosphite base electroless nickel was used to for the nickel undercoating coating on the treated yarn.
- the solution was made up by using 6 vol of nickel sulfate, 15 vol of sodium hypophosphite, and 79 Vol% of deionized water.
- the bath was operated in 190°F at a PH value of 4.85 and constantly filtered through a 1 um filter. Generally, a soaking duration of two to three minutes will coat the yarn uniformly with a layer of phosphorus based electroless nickel of 0.5 to 0.75 micrometer.
- This electroless nickel-coated Vectran yarn exhibited an electrical resistance of -250 ohm/ft and was found conductive enough to facilitate the subsequent electroplating. Additional two minutes acid copper sulfate electroplating operated at a current of 5.83 amp resulted in a very highly conductive and uniform yarn with electrical resistance of 1.23 ohm/ft.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Mechanical Engineering (AREA)
- General Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Textile Engineering (AREA)
- Crystallography & Structural Chemistry (AREA)
- Electromagnetism (AREA)
- Physics & Mathematics (AREA)
- Electrochemistry (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Chemical Or Physical Treatment Of Fibers (AREA)
- Chemically Coating (AREA)
- Shielding Devices Or Components To Electric Or Magnetic Fields (AREA)
- Electroplating Methods And Accessories (AREA)
- Polyesters Or Polycarbonates (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2011800459464A CN103221577A (en) | 2010-07-23 | 2011-07-21 | Conductive metal-coated fibers and continuous methods of making and using the same |
| EP11741504.2A EP2596149A2 (en) | 2010-07-23 | 2011-07-21 | Electrically conductive metal-coated fibers, continuous process for preparation thereof, and use thereof |
| CA2806480A CA2806480A1 (en) | 2010-07-23 | 2011-07-21 | Electrically conductive metal-coated fibers, continuous process for preparation thereof, and use thereof |
| BR112013001706A BR112013001706A2 (en) | 2010-07-23 | 2011-07-21 | continuous process for preparation and use of electrically conductive metal coated fibers |
| JP2013520863A JP2013533387A (en) | 2010-07-23 | 2011-07-21 | Conductive metal coated fiber, continuous method for its preparation, and its use |
| KR1020137004320A KR20130132754A (en) | 2010-07-23 | 2011-07-21 | Electrically conductive metal-coated fibers, continuous process for preparation thereof, and use thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US36723510P | 2010-07-23 | 2010-07-23 | |
| US61/367,235 | 2010-07-23 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2012012614A2 true WO2012012614A2 (en) | 2012-01-26 |
| WO2012012614A3 WO2012012614A3 (en) | 2012-03-08 |
Family
ID=44511504
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2011/044817 Ceased WO2012012614A2 (en) | 2010-07-23 | 2011-07-21 | Electrically conductive metal-coated fibers, continuous process for preparation thereof, and use thereof |
Country Status (8)
| Country | Link |
|---|---|
| US (3) | US20120021218A1 (en) |
| EP (1) | EP2596149A2 (en) |
| JP (1) | JP2013533387A (en) |
| KR (1) | KR20130132754A (en) |
| CN (1) | CN103221577A (en) |
| BR (1) | BR112013001706A2 (en) |
| CA (1) | CA2806480A1 (en) |
| WO (1) | WO2012012614A2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12410555B2 (en) | 2020-01-15 | 2025-09-09 | Kuraray Co., Ltd. | Metal-covered liquid crystal polyester multifilament |
Families Citing this family (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9416328B2 (en) | 2010-01-06 | 2016-08-16 | General Electric Company | System and method for treatment of fine particulates separated from syngas produced by gasifier |
| US9324472B2 (en) * | 2010-12-29 | 2016-04-26 | Syscom Advanced Materials, Inc. | Metal and metallized fiber hybrid wire |
| JP5970825B2 (en) * | 2012-01-17 | 2016-08-17 | 三菱自動車工業株式会社 | undercover |
| US9783890B2 (en) * | 2012-10-26 | 2017-10-10 | Rohm And Haas Electronic Materials Llc | Process for electroless plating and a solution used for the same |
| KR101469683B1 (en) * | 2013-05-31 | 2014-12-05 | 주식회사 불스원신소재 | Methods for Preparing of Electromagnetic Shielding Composite Using Copper-Nickel Plated Carbon Fiber Prepared by Electroless-Electronic Continuous Process and Electromagnetic Shielding Material Prepared by the Methods |
| CN104562114A (en) * | 2013-10-28 | 2015-04-29 | 黄文昌 | Preparation method and finished product of colored high-strength fiber material |
| US9716374B2 (en) * | 2014-01-03 | 2017-07-25 | Rohr, Inc. | Systems and methods for electrical harness construction |
| CN103757617B (en) * | 2014-01-09 | 2016-07-20 | 成都理工大学 | A kind of Ni-Cu-La-B quaternary alloy plating solution and the method for the plating of glass fibre chemistry |
| US20150204008A1 (en) * | 2014-01-22 | 2015-07-23 | Wen-Chang Huang | Manufacturing method of a colored high-strength fiber and a colored high-strength fiber |
| DE102014010777A1 (en) * | 2014-01-30 | 2015-07-30 | Dürr Systems GmbH | High voltage cables |
| KR101662759B1 (en) | 2015-01-09 | 2016-10-10 | 건국대학교 글로컬산학협력단 | Production method of metal plated fiber by adopting consecutive electroless plating and electroplating process, metal plated fiber produced by said method and a filter comprising siad metal plated fiber |
| US11234878B2 (en) | 2015-12-16 | 2022-02-01 | Precision Fabrics Group, Inc. | High performance, skin friendly, fabric for patient transfer and care and lifting devices made therefrom |
| JP6746842B2 (en) * | 2016-06-24 | 2020-08-26 | 学校法人関東学院 | Method for producing cycloolefin yarn with metal coating |
| JP6758578B2 (en) * | 2016-06-24 | 2020-09-23 | 学校法人関東学院 | Method for manufacturing cycloolefin yarn with metal film |
| JP6746843B2 (en) * | 2016-06-24 | 2020-08-26 | 学校法人関東学院 | Method for producing cycloolefin yarn with metal coating |
| US11528833B2 (en) | 2016-12-13 | 2022-12-13 | Amogreentech Co., Ltd. | Flexible electromagnetic wave shielding material, electromagnetic wave shielding-type circuit module comprising same and electronic device furnished with same |
| CN108267870B (en) * | 2016-12-30 | 2021-03-30 | 财团法人工业技术研究院 | Copper foil composite |
| US10556776B2 (en) | 2017-05-23 | 2020-02-11 | Otis Elevator Company | Lightweight elevator traveling cable |
| US10833318B2 (en) * | 2017-10-03 | 2020-11-10 | California Institute Of Technology | Three-dimensional architected pyrolyzed electrodes for use in secondary batteries and methods of making three-dimensional architected electrodes |
| US11691338B2 (en) * | 2019-05-02 | 2023-07-04 | Hamilton Sunstrand Corporation | Article containing conductive fibers |
| CN110983764B (en) * | 2019-12-20 | 2022-04-05 | 上海大学 | A kind of conductive aramid fiber with composite metal coating structure |
| JP7754807B2 (en) * | 2020-04-23 | 2025-10-15 | セーレン株式会社 | Conductive thread and article having wiring made of conductive thread |
| CN112267116A (en) * | 2020-10-28 | 2021-01-26 | 深圳亚太航空技术有限公司 | Light aramid fiber wave-proof sleeve |
| JP7643876B2 (en) * | 2021-01-19 | 2025-03-11 | ヤマハ株式会社 | Conductive yarn and fabric with elastic wiring |
| US11246248B1 (en) * | 2021-04-09 | 2022-02-08 | Micrometal Technologies, Inc. | Electrical shielding material composed of metallized stainless steel or low carbon steel monofilament yarns |
| CN113215630A (en) * | 2021-04-21 | 2021-08-06 | 飞荣达科技(江苏)有限公司 | High-performance carbon fiber and electroplating method thereof |
| US20220380674A1 (en) * | 2021-05-18 | 2022-12-01 | Ticona Llc | Photoplethysmographic Sensor Containing A Polymer Composition |
| US12428543B2 (en) | 2021-05-18 | 2025-09-30 | Ticona Llc | Connected medical device containing a liquid crystalline polymer composition having a low dielectric constant |
| CN114045566A (en) * | 2021-11-03 | 2022-02-15 | 常隆塑胶科技(苏州)有限公司 | Composite conductive fiber and composite process method thereof |
| CN114990880B (en) * | 2022-05-26 | 2024-05-07 | 惠州学院 | Conductive liquid crystal polyarylester fiber and preparation method thereof |
| EP4421680A1 (en) | 2023-02-24 | 2024-08-28 | Kordsa Teknik Tekstil Anonim Sirketi | Rfid tag for a tire having a flexible antenna |
| KR20240133312A (en) * | 2023-02-28 | 2024-09-04 | 재단법인대구경북과학기술원 | Highly-durable conductive fiber with retained electrical properties after washing and its manufacturing method |
| CN118516850B (en) * | 2024-06-03 | 2025-10-24 | 武汉纺织大学 | A method for preparing high-conductivity flexible liquid crystal polyarylate nanofiber membrane by in-situ self-assembly |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5302415A (en) | 1992-12-08 | 1994-04-12 | E. I. Du Pont De Nemours And Company | Electroless plated aramid surfaces and a process for making such surfaces |
| US5453299A (en) | 1994-06-16 | 1995-09-26 | E. I. Du Pont De Nemours And Company | Process for making electroless plated aramid surfaces |
| US5935706A (en) | 1996-05-30 | 1999-08-10 | E. I. Dupont De Nemours & Comp | Thermally stable metal coated polymeric monofilament or yarn |
| US6045680A (en) | 1996-05-30 | 2000-04-04 | E. I. Du Pont De Nemours And Company | Process for making thermally stable metal coated polymeric monofilament or yarn |
| US6403211B1 (en) | 2000-07-18 | 2002-06-11 | 3M Innovative Properties Company | Liquid crystal polymer for flexible circuits |
| US6923919B2 (en) | 2000-07-18 | 2005-08-02 | 3M Innovative Properties Company | Liquid crystal polymers for flexible circuits |
| US7166354B2 (en) | 2000-12-26 | 2007-01-23 | Mitsubishi Materials Corporation | Metal coated fiber and electroconductive composition comprising the same and method for production thereof and use thereof |
| US20080280045A1 (en) | 2003-12-08 | 2008-11-13 | Jar-Wha Lee | Method and apparatus for the treatment of individual filaments of a multifilament yarn |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4161470A (en) * | 1977-10-20 | 1979-07-17 | Celanese Corporation | Polyester of 6-hydroxy-2-naphthoic acid and para-hydroxy benzoic acid capable of readily undergoing melt processing |
| DE3338805A1 (en) * | 1983-10-26 | 1985-05-15 | Basf Ag, 6700 Ludwigshafen | FULLY FLAVORED POLYESTER |
| JP2581543B2 (en) * | 1986-07-04 | 1997-02-12 | エビナ電化工業 株式会社 | Method for producing plated wholly aromatic polyester liquid crystal polymer molded article |
| WO1990000634A2 (en) * | 1988-07-07 | 1990-01-25 | E.I. Du Pont De Nemours And Company | Metallized polymers and method |
| WO1997048832A2 (en) * | 1996-05-30 | 1997-12-24 | E.I. Du Pont De Nemours And Company | Process for making thermally stable metal coated polymeric monofilament or yarn |
| US6129878A (en) * | 1998-09-10 | 2000-10-10 | Celanese Acetate Llc | Process for direct on-bobbin heat treating of high denier filaments of thermotropic liquid crystalline polymers |
| JP3929925B2 (en) * | 2003-04-10 | 2007-06-13 | 東レエンジニアリング株式会社 | Method for producing metal-clad liquid crystal polyester substrate |
| JP5058973B2 (en) * | 2006-03-23 | 2012-10-24 | 株式会社きもと | Electroless plating forming material and electroless plating forming method using the same |
| EP2066826A1 (en) * | 2006-09-26 | 2009-06-10 | DSMIP Assets B.V. | Method for metallizing a component |
| US8025814B2 (en) * | 2008-06-23 | 2011-09-27 | Sumitomo Chemical Company, Limited | Resin composition and molded article using the same |
| JP2010100934A (en) * | 2008-09-24 | 2010-05-06 | Du Pont Toray Co Ltd | Conductive high tenacity fiber yarn and method for manufacturing the same |
| US20110287272A1 (en) * | 2008-12-23 | 2011-11-24 | E.I.Du Pont De Nemours And Company | Polymer compositions for metal coating, articles made therefrom and process for same |
| CN101766959B (en) * | 2009-12-22 | 2012-01-11 | 武汉智迅创源科技发展股份有限公司 | Small-bore nuclear pore filter membrane and production method thereof |
-
2011
- 2011-07-21 CA CA2806480A patent/CA2806480A1/en not_active Abandoned
- 2011-07-21 EP EP11741504.2A patent/EP2596149A2/en not_active Withdrawn
- 2011-07-21 US US13/187,927 patent/US20120021218A1/en not_active Abandoned
- 2011-07-21 BR BR112013001706A patent/BR112013001706A2/en not_active IP Right Cessation
- 2011-07-21 JP JP2013520863A patent/JP2013533387A/en active Pending
- 2011-07-21 CN CN2011800459464A patent/CN103221577A/en active Pending
- 2011-07-21 WO PCT/US2011/044817 patent/WO2012012614A2/en not_active Ceased
- 2011-07-21 KR KR1020137004320A patent/KR20130132754A/en not_active Ceased
-
2017
- 2017-08-23 US US15/684,360 patent/US20180187077A1/en not_active Abandoned
-
2025
- 2025-07-30 US US19/285,913 patent/US20260028532A1/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5302415A (en) | 1992-12-08 | 1994-04-12 | E. I. Du Pont De Nemours And Company | Electroless plated aramid surfaces and a process for making such surfaces |
| US5422142A (en) | 1992-12-08 | 1995-06-06 | E. I. Du Pont De Nemours And Company | Process for making electroless plated aramid fibrids |
| US5453299A (en) | 1994-06-16 | 1995-09-26 | E. I. Du Pont De Nemours And Company | Process for making electroless plated aramid surfaces |
| US5935706A (en) | 1996-05-30 | 1999-08-10 | E. I. Dupont De Nemours & Comp | Thermally stable metal coated polymeric monofilament or yarn |
| US6045680A (en) | 1996-05-30 | 2000-04-04 | E. I. Du Pont De Nemours And Company | Process for making thermally stable metal coated polymeric monofilament or yarn |
| US6403211B1 (en) | 2000-07-18 | 2002-06-11 | 3M Innovative Properties Company | Liquid crystal polymer for flexible circuits |
| US6923919B2 (en) | 2000-07-18 | 2005-08-02 | 3M Innovative Properties Company | Liquid crystal polymers for flexible circuits |
| US7166354B2 (en) | 2000-12-26 | 2007-01-23 | Mitsubishi Materials Corporation | Metal coated fiber and electroconductive composition comprising the same and method for production thereof and use thereof |
| US20080280045A1 (en) | 2003-12-08 | 2008-11-13 | Jar-Wha Lee | Method and apparatus for the treatment of individual filaments of a multifilament yarn |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12410555B2 (en) | 2020-01-15 | 2025-09-09 | Kuraray Co., Ltd. | Metal-covered liquid crystal polyester multifilament |
Also Published As
| Publication number | Publication date |
|---|---|
| US20260028532A1 (en) | 2026-01-29 |
| CA2806480A1 (en) | 2012-01-26 |
| EP2596149A2 (en) | 2013-05-29 |
| JP2013533387A (en) | 2013-08-22 |
| KR20130132754A (en) | 2013-12-05 |
| BR112013001706A2 (en) | 2016-05-31 |
| US20120021218A1 (en) | 2012-01-26 |
| WO2012012614A3 (en) | 2012-03-08 |
| US20180187077A1 (en) | 2018-07-05 |
| CN103221577A (en) | 2013-07-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20260028532A1 (en) | Electrically conductive metal-coated fibers, continuous process for preparation thereof, and use thereof | |
| US9324472B2 (en) | Metal and metallized fiber hybrid wire | |
| CN1040785C (en) | Electroless plated aramid surfaces and a process for making such surfaces | |
| KR101423169B1 (en) | A Method for Manufacturing of Shield Sheet for Preventing Electromagnetic Wave | |
| CN104894846A (en) | Preparation method of nickel-plated aromatic polyamide fiber | |
| JP6114521B2 (en) | Metal coated fiber | |
| US5453299A (en) | Process for making electroless plated aramid surfaces | |
| EP0807192B1 (en) | Process for batch-plating aramid fibers | |
| JP4060363B2 (en) | Production of polymer monofilaments or yarns coated with heat stable metals | |
| JP2005105386A (en) | Electroless silver plating solution for fiber | |
| KR101664857B1 (en) | Conductive yarn and method for preparing the same | |
| JP5941317B2 (en) | Metal coated fiber | |
| CN105133301B (en) | A kind of preparation method of nickel plating aromatic polyamide fibre | |
| Park et al. | Copper metallization of poly (ethylene terephthalate) fabrics via intermediate polyaniline layers | |
| JP2987556B2 (en) | Method for forming metal conductive layer on fluororesin body surface | |
| KR20210029886A (en) | Metal plated carbon fibers by non-electroplating process and manufacturing method thereof | |
| KR100466993B1 (en) | Process For Making Thermally Stable Metal Coated Polymeric Monofilament or Yarn | |
| JP5160057B2 (en) | Fiber material with silver plating | |
| JP5117656B2 (en) | Electroless plating pretreatment method and conductive material using the same | |
| JP6473307B2 (en) | Manufacturing method of conductor for electric wire | |
| US20200165729A1 (en) | Thermal decomposition metallization process | |
| JPH0274672A (en) | Production of metal-coated fibrous structure | |
| JP2019183179A (en) | Copper plating method of fiber |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11741504 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 2013520863 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2806480 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20137004320 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2011741504 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011741504 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013001706 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112013001706 Country of ref document: BR Kind code of ref document: A2 Effective date: 20130123 |