WO2013133270A1 - ホットスタンプ用鋼板及びその製造方法並びにホットスタンプ鋼材 - Google Patents
ホットスタンプ用鋼板及びその製造方法並びにホットスタンプ鋼材 Download PDFInfo
- Publication number
- WO2013133270A1 WO2013133270A1 PCT/JP2013/055992 JP2013055992W WO2013133270A1 WO 2013133270 A1 WO2013133270 A1 WO 2013133270A1 JP 2013055992 W JP2013055992 W JP 2013055992W WO 2013133270 A1 WO2013133270 A1 WO 2013133270A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- hot
- less
- steel sheet
- steel
- hot stamping
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/38—Ferrous alloys, e.g. steel alloys containing chromium with more than 1.5% by weight of manganese
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/58—Ferrous alloys, e.g. steel alloys containing chromium with nickel with more than 1.5% by weight of manganese
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21B—ROLLING OF METAL
- B21B1/00—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations
- B21B1/22—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations for rolling plates, strips, bands or sheets of indefinite length
- B21B1/24—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations for rolling plates, strips, bands or sheets of indefinite length in a continuous or semi-continuous process
- B21B1/26—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations for rolling plates, strips, bands or sheets of indefinite length in a continuous or semi-continuous process by hot-rolling, e.g. Steckel hot mill
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/04—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips to produce plates or strips for drawing, e.g. for deep-drawing
- C21D8/0421—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips to produce plates or strips for drawing, e.g. for deep-drawing characterised by the working steps
- C21D8/0436—Cold rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/04—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips to produce plates or strips for drawing, e.g. for deep-drawing
- C21D8/0447—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips to produce plates or strips for drawing, e.g. for deep-drawing characterised by the heat treatment
- C21D8/0463—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips to produce plates or strips for drawing, e.g. for deep-drawing characterised by the heat treatment following hot rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/46—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for sheet metals
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/46—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for sheet metals
- C21D9/48—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for sheet metals deep-drawing sheets
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/001—Ferrous alloys, e.g. steel alloys containing N
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/002—Ferrous alloys, e.g. steel alloys containing In, Mg, or other elements not provided for in one single group C22C38/001 - C22C38/60
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/06—Ferrous alloys, e.g. steel alloys containing aluminium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/08—Ferrous alloys, e.g. steel alloys containing nickel
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/12—Ferrous alloys, e.g. steel alloys containing tungsten, tantalum, molybdenum, vanadium, or niobium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/14—Ferrous alloys, e.g. steel alloys containing titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/16—Ferrous alloys, e.g. steel alloys containing copper
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/22—Ferrous alloys, e.g. steel alloys containing chromium with molybdenum or tungsten
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/24—Ferrous alloys, e.g. steel alloys containing chromium with vanadium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/28—Ferrous alloys, e.g. steel alloys containing chromium with titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/32—Ferrous alloys, e.g. steel alloys containing chromium with boron
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/54—Ferrous alloys, e.g. steel alloys containing chromium with nickel with boron
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/02—Pretreatment of the material to be coated, e.g. for coating on selected surface areas
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/02—Pretreatment of the material to be coated, e.g. for coating on selected surface areas
- C23C2/022—Pretreatment of the material to be coated, e.g. for coating on selected surface areas by heating
- C23C2/0224—Two or more thermal pretreatments
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/02—Pretreatment of the material to be coated, e.g. for coating on selected surface areas
- C23C2/024—Pretreatment of the material to be coated, e.g. for coating on selected surface areas by cleaning or etching
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/04—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor characterised by the coating material
- C23C2/06—Zinc or cadmium or alloys based thereon
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/04—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor characterised by the coating material
- C23C2/12—Aluminium or alloys based thereon
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/26—After-treatment
- C23C2/28—Thermal after-treatment, e.g. treatment in oil bath
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/26—After-treatment
- C23C2/28—Thermal after-treatment, e.g. treatment in oil bath
- C23C2/29—Cooling or quenching
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2/00—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor
- C23C2/34—Hot-dipping or immersion processes for applying the coating material in the molten state without affecting the shape; Apparatus therefor characterised by the shape of the material to be treated
- C23C2/36—Elongated material
- C23C2/40—Plates; Strips
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/004—Dispersions; Precipitations
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/12—All metal or with adjacent metals
- Y10T428/12493—Composite; i.e., plural, adjacent, spatially distinct metal components [e.g., layers, joint, etc.]
- Y10T428/12736—Al-base component
- Y10T428/1275—Next to Group VIII or IB metal-base component
- Y10T428/12757—Fe
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/12—All metal or with adjacent metals
- Y10T428/12493—Composite; i.e., plural, adjacent, spatially distinct metal components [e.g., layers, joint, etc.]
- Y10T428/12771—Transition metal-base component
- Y10T428/12785—Group IIB metal-base component
- Y10T428/12792—Zn-base component
- Y10T428/12799—Next to Fe-base component [e.g., galvanized]
Definitions
- the present invention relates to a steel sheet for hot stamping, a manufacturing method thereof, and a hot stamping steel material.
- a hot forming method called a hot stamp method has attracted attention.
- the steel sheet (work material) is heated to a predetermined temperature (generally, the temperature at which it becomes an austenite phase), and the strength of the work material is lowered in order to facilitate molding,
- a predetermined temperature generally, the temperature at which it becomes an austenite phase
- the strength of the work material is lowered in order to facilitate molding.
- a mold having a temperature lower than that of the workpiece for example, room temperature
- rapid cooling utilizing the temperature difference between the workpiece and the mold Heat treatment (quenching) is performed to increase the strength of the molded product.
- Patent Document 1 discloses that one or more of Mg oxides, sulfides, composite crystallized substances, and composite precipitates having an average particle size within a predetermined range are specified.
- Patent Document 2 discloses that punching (perforation) is performed after heating for hot stamping and in a high temperature state (hot) before pressing, thereby improving punchability, thereby providing delayed fracture resistance. Techniques for improving are disclosed.
- Patent Document 1 Although the technique disclosed in Patent Document 1 is an excellent technique, Mg that is generally not easy to be contained is present in the steel, and a product containing the Mg is highly controlled. Therefore, a technique that can be more easily implemented is desired.
- Patent Document 2 is based on the premise of hot drilling in which punching (drilling) is performed after heating for hot stamping and in a high temperature state (hot) before pressing. Technology. For this reason, high dimensional accuracy cannot be ensured in the steel material after hot stamping. Moreover, the shape which can be shape
- the present invention ensures good hydrogen embrittlement resistance and can be easily implemented even when the steel material after hot stamping is subjected to processing such as drilling in which stress remains. It aims at providing a steel plate, its manufacturing method, and hot stamped steel.
- the present inventors have conducted intensive studies as follows.
- the inventors focused on inclusions containing Mn and Mn oxides, which are relatively easy to produce in steel, and by functioning these as trapping sites for diffusible hydrogen and non-diffusible hydrogen.
- the new idea was to secure good hydrogen embrittlement resistance.
- steel sheets for hot stamping were prepared according to various manufacturing conditions and subjected to the hot stamping method.
- the steel materials obtained were investigated for hydrogen embrittlement resistance and toughness. Went.
- the concentration of inclusions containing Mn is excessively increased, a new problem has been found that a decrease in toughness becomes apparent in the steel material after hot stamping. That is, after the hot stamping, the concentration of inclusions containing Mn is within a predetermined range and the number density of Mn oxides in inclusions containing Mn of a predetermined size is set to a predetermined value or more. It was newly discovered that it is possible to ensure good hydrogen embrittlement resistance and good toughness even when the steel material is subjected to processing such as drilling where residual stress remains. .
- the concentration of inclusions containing Mn is within a predetermined range by raising the coiling temperature in the hot rolling process higher than before and performing cold rolling.
- the number ratio of Mn oxides in inclusions containing Mn of a predetermined size can be made a predetermined value or more.
- the present invention has been made on the basis of the above new findings, and the gist thereof is as follows.
- a steel sheet for hot stamping characterized by being not less than 0%.
- the chemical composition is mass%, Cr: 0.01 to 2.0%, Mo: 0.01 to 1.0% V: 0.01 to 0.5% W: 0.01 to 0.5% Ni: 0.01 to 5.0% and B: 0.0005 to 0.01%
- the chemical composition is mass%, Ti: 0.001 to 0.5%, Nb: 0.001 to 0.5% and Cu: 0.01 to 1.0%
- a method of manufacturing a steel sheet for hot stamping comprising: a cold rolling step of performing cold rolling at a rolling reduction of ⁇ 90% to obtain a cold rolled steel sheet.
- the chemical composition is mass%, Cr: 0.01 to 2.0%, Mo: 0.01 to 1.0% V: 0.01 to 0.5% W: 0.01 to 0.5% Ni: 0.01 to 5.0% and B: 0.0005 to 0.01%
- the chemical composition is mass%, Ti: 0.001 to 0.5%, Nb: 0.001 to 0.5% and Cu: 0.01 to 1.0%
- the manufacturing method of the steel sheet for hot stamping as described in said (7) or (8) characterized by containing 1 type, or 2 or more types selected from the group which consists of.
- a steel sheet for hot stamping obtained by the manufacturing method according to any one of (7) to (9) above is immersed in a hot dip galvanizing bath, and then heated to a temperature range of 600 ° C. or lower.
- the chemical composition is mass%, Cr: 0.01 to 2.0%, Mo: 0.01 to 1.0% V: 0.01 to 0.5% W: 0.01 to 0.5% Ni: 0.01 to 5.0% and B: 0.0005 to 0.01%
- the chemical composition is mass%, Ti: 0.001 to 0.5%, Nb: 0.001 to 0.5% and Cu: 0.01 to 1.0%
- a hot stamping method can be obtained because it can ensure good hydrogen embrittlement resistance and is easy to implement even when a process such as drilling where stress remains after hot stamping is performed.
- the application range (parts) can be expanded.
- C is the most important element for increasing the strength of a steel sheet by the hot stamp method. If the C content is less than 0.18%, it is difficult to ensure a strength of 1500 MPa or more after hot stamping. Therefore, the C content is 0.18% or more. On the other hand, if the C content exceeds 0.26%, the ductility after hot stamping becomes poor, and it becomes difficult to ensure a total elongation of 10% or more. Therefore, the C content is 0.26% or less.
- Si is an important element for controlling the concentration of Mn-containing inclusions and the ratio of the number of Mn oxides in the inclusions having a maximum length of 1.0 to 4.0 ⁇ m.
- Si content is more than 0.02%.
- Si content exceeds 0.05%, the generation of Mn oxide is excessively suppressed, and the number of Mn oxides in the inclusion containing Mn having a maximum length of 1.0 to 4.0 ⁇ m.
- the ratio is less than 10.0%, and it is difficult to stably obtain good hydrogen embrittlement resistance. Therefore, the Si content is 0.05% or less.
- Mn is the most important element in the present invention.
- Mn has the effect of increasing hydrogen embrittlement resistance by forming inclusions containing Mn in the steel. Further, the remaining Mn that has not formed inclusions has an effect of enhancing the hardenability.
- the Mn content is less than 1.0%, it is difficult to make the concentration of inclusions containing Mn 0.010% by mass or more. Therefore, the Mn content is 1.0% or more.
- the Mn content exceeds 1.5%, the effect of the above action is saturated, which is economically disadvantageous, and in addition, the mechanical properties may be deteriorated due to segregation of Mn. Therefore, the Mn content is 1.5% or less.
- P is an element generally contained as an impurity.
- the P content exceeds 0.03%, the hot workability is significantly reduced. Therefore, the P content is 0.03% or less.
- the lower limit of the P content does not need to be specified, but excessive reduction places a great load on the steel making process, so 0.001% or more is preferable.
- S is an element generally contained as an impurity.
- S content exceeds 0.02%, the hot workability is significantly lowered. Therefore, the S content is 0.02% or less.
- the lower limit of the S content does not need to be specified, but excessive reduction places a great load on the steel making process, so it is preferably 0.0005% or more.
- Al is an element having an effect of making steel healthy by deoxidation.
- the Al content is less than 0.001%, it is difficult to perform sufficient deoxidation. Therefore, the Al content is 0.001% or more.
- the Al content exceeds 0.5%, the generation of Mn oxide is excessively suppressed, making it difficult to secure the ratio of Mn oxide described later, and ensuring good hydrogen embrittlement resistance. It becomes difficult. Therefore, the Al content is 0.5% or less.
- N is an element generally contained as an impurity.
- Ti and B which are optional elements described later, are easily combined and consumed, and the effects of these elements are reduced. Therefore, the N content is 0.1% or less, preferably 0.01% or less.
- the lower limit of the N content does not need to be specified, but excessive reduction places a great load on the steel making process, so it is preferably made 0.001% or more.
- O forms a Mn oxide in the steel, functions as a trap site for diffusible hydrogen and non-diffusible hydrogen, and has an action of enhancing hydrogen embrittlement resistance.
- the O content is less than 0.0010%, the generation of Mn oxide is not sufficiently promoted, and the number ratio of Mn oxide in the inclusions containing Mn is less than 10.0%, and good hydrogen embrittlement resistance. Stabilization characteristics cannot be obtained stably. Therefore, the O content is 0.0010% or more.
- the O content exceeds 0.020%, coarse oxides are formed in the steel, and the mechanical properties of the steel are deteriorated. Therefore, the O content is 0.020% or less.
- the steel sheet of the present invention and the steel material of the present invention have the above-described component composition as an essential component composition, and, if necessary, one or two of Cr, Mo, V, W, Ni, B, Ti, Nb, and Cu. More than seeds can be included.
- the upper limit values for the contents of B, Cr, Mo, W, V and Ni are as described above.
- the B content is set to 0.0005% or more, or the content of any element of Cr, Mo, W, V and Ni is set to 0.01% or more. It is preferable that Moreover, since Ni has the effect
- Ti, Nb, and Cu all have the effect of increasing the strength. Therefore, you may contain 1 type, or 2 or more types of these elements.
- the Ti content exceeds 0.5%, the generation of Mn oxide is excessively suppressed, and it becomes difficult to secure the ratio of Mn oxide described later, and the favorable hydrogen embrittlement resistance is ensured. It becomes difficult. Therefore, the Ti content is 0.5%.
- Nb content exceeds 0.5%, the controllability of hot rolling may be impaired. Therefore, the Nb content is 0.5% or less.
- Cu content exceeds 1.0%, the surface property of a hot-rolled steel plate may be impaired.
- the Cu content is 1.0% or less.
- Ti preferentially bonds with N in steel to form nitrides, thereby suppressing B from being wasted by the formation of nitrides and making it possible to further enhance the effects of B. Therefore, when B mentioned above is contained, it is preferable to also contain Ti.
- the balance is Fe and impurities.
- ⁇ Concentration of inclusion containing Mn 0.010 mass% or more and less than 0.25 mass%> Inclusions containing Mn play an important role in suppressing hydrogen embrittlement as well as the ratio of the number of Mn oxides to the number of inclusions containing Mn having a maximum length of 1.0 to 4.0 ⁇ m, which will be described later. .
- concentration of the inclusion containing Mn shall be 0.010% or more.
- the concentration of inclusions containing Mn is 0.25% or more, the toughness may be reduced. Therefore, the concentration of inclusions containing Mn is less than 0.25%.
- concentration of the inclusion containing Mn is calculated
- EDS energy dispersive X-ray spectroscopy
- the number ratio of Mn oxide in the number of inclusions containing Mn having a maximum length of 1.0 to 4.0 ⁇ m plays an important role in suppressing hydrogen embrittlement together with the inclusions containing Mn described above. .
- the ratio of the number of Mn oxides in the number of inclusions containing Mn having a maximum length of 1.0 to 4.0 ⁇ m is 10.0% or more.
- the number ratio of Mn oxide in the number of inclusions containing Mn having a maximum length of 1.0 to 4.0 ⁇ m is obtained by the following procedure.
- the cross section of the steel sheet is observed using an SEM, and the maximum length (for example, the length of the long side if the inclusion is rectangular, the length of the long diameter if the inclusion is elliptical) is 1.0 to 4.0 ⁇ m.
- Select inclusions for investigation. These inclusions were subjected to EDS analysis, and those in which characteristic X-rays from Mn and characteristic X-rays from O (oxygen) were simultaneously detected were determined to be Mn oxides. Then, observation / analysis is performed in a plurality of fields until the total number of investigations exceeds 500, and the number ratio of Mn oxides in the total number of investigations is defined as the number ratio of Mn oxides.
- the maximum length of inclusions to be investigated is set to 1.0 ⁇ m or more because inclusions smaller than that have insufficient accuracy in analyzing constituent elements by EDS.
- the maximum length of inclusions to be investigated is 4.0 ⁇ m or less. Inclusions larger than this are, for example, a combination of a plurality of different inclusions. This is because it is not uniquely determined.
- the steel sheet of the present invention and the steel material of the present invention may be formed as a surface-treated steel sheet or surface-treated steel material by forming a plating layer on the surface for the purpose of improving corrosion resistance or the like.
- the plating layer may be a hot-dip plating layer or an electroplating layer.
- the hot dip plating layer include hot dip galvanizing, alloyed hot dip galvanizing, hot dip aluminum plating, hot dip Zn-Al alloy plating, hot dip Zn-Al-Mg alloy plating, hot dip Zn-Al-Mg-Si alloy plating, etc.
- the Examples of the electroplating layer include electrogalvanizing and electro-Zn—Ni alloy plating.
- the thickness of the plating layer is not particularly limited. However, for the steel sheet of the present invention, it is preferable to limit the upper limit of the thickness of the plating layer from the viewpoint of press formability.
- the thickness of the plating layer is preferably 50 ⁇ m or less from the viewpoint of galling resistance, and in the case of hot dip galvanization, from the viewpoint of suppressing the adhesion of Zn to the mold.
- the thickness of the plating layer is preferably 30 ⁇ m or less.
- the thickness of the plating layer is preferably 45 ⁇ m or less from the viewpoint of suppressing the occurrence of cracks in the alloy layer.
- the thickness of the plating layer is preferably 5 ⁇ m or more, and more preferably 10 ⁇ m or more.
- the thickness of the plating layer is preferably 10 ⁇ m or more, and more preferably 15 ⁇ m or more.
- the manufacturing method of the steel plate of the present invention includes a hot rolling process in which a steel piece having the above chemical composition is hot-rolled and then wound in a temperature range of 690 ° C. or higher to form a hot-rolled steel sheet, It can be manufactured by a manufacturing method including a cold rolling process in which cold rolling at a reduction rate of 90% is performed to obtain a cold rolled steel sheet.
- the steelmaking conditions and casting conditions when manufacturing the steel slab, and the cold rolling conditions applied to the hot-rolled steel sheet may be in accordance with ordinary methods. Further, pickling performed before the hot-rolled steel sheet is subjected to cold rolling may be performed by a conventional method.
- the steel slab having the above chemical composition is hot-rolled and then rolled into a hot-rolled steel sheet wound in a temperature range of 690 ° C. or more, and cold rolling with a reduction rate of 10 to 90% is performed. It is obtained by applying. Therefore, from the viewpoint of hydrogen embrittlement resistance after hot stamping and toughness, recrystallization annealing after cold rolling is unnecessary. However, from the viewpoint of workability such as blanking or pre-forming before being subjected to hot stamping, it is preferable to perform recrystallization annealing after cold rolling to achieve softening. Further, a plating layer may be provided for the purpose of improving the corrosion resistance after recrystallization annealing. In the case of performing hot dip plating, it is preferable to perform hot dip plating subsequent to recrystallization annealing using continuous hot dip plating equipment.
- a hot stamping steel sheet capable of obtaining a hot stamping steel material having good hydrogen embrittlement resistance and toughness can be obtained by the above-described manufacturing method. It is considered that the formation of cementite in the rolled steel sheet and the microstructure are related. That is, cementite is crushed together with other inclusions in the cold rolling process, which is a subsequent process of the hot rolling process, but depending on its size, the size and dispersion status after crushing, and The generation situation of the voids of the different. Moreover, the difference in hardness from the inclusions differs depending on the strength (hardness) of the microstructure, and this also affects the state of the inclusions and the voids. Furthermore, both cementite and the microstructure influence the situation of inclusions that are deformed without being crushed.
- the present inventors have carried out hot rolling on a steel slab having the above chemical composition, and then wound it in a temperature range of 690 ° C. or higher, and 10% to 90% of the hot rolled steel sheet thus obtained.
- the upper limit of the coiling temperature is not particularly limited.
- the winding temperature is preferably 850 ° C. or lower.
- the reduction rate in a cold rolling process according to the capability of an installation, and the plate
- the temperature of the steel slab used for hot rolling can be selected from 1200 to 1250 ° C., the rolling reduction from 30 to 90%, and the finishing temperature around 900 ° C.
- the annealing temperature is preferably 700 to 850 ° C. from the viewpoint of appropriate softening, but may be less than 700 ° C. for the purpose of imparting other mechanical properties, It may be higher than 850 ° C.
- recrystallization annealing it may be cooled to room temperature as it is, or may be immersed in a hot dipping bath in the cooling process to room temperature to form a hot dipped layer on the surface of the steel sheet.
- Si contained in the hot-dip aluminum plating layer affects the reaction between Al and Fe that occurs during heating before hot stamping.
- the Si content in the bath is preferably 1% or more, and more preferably 3% or more.
- the Si content in the bath is preferably 15% or less, and more preferably 12% or less. preferable.
- the hot dip galvanizing When the hot dip galvanizing is hot dip galvanizing, it is cooled to room temperature after being immersed in a hot dip galvanizing bath. An alloying treatment is performed by heating to a temperature range of °C or lower, and then cooled to room temperature.
- the hot dip galvanizing bath may contain 0.01 to 3% Al.
- Al affects the reaction between Zn and Fe.
- mutual diffusion of Zn and Fe can be suppressed by the reaction layer of Fe and Al.
- hot dip plating when hot dip galvanization, it can utilize in order to control to a suitable plating composition from viewpoints, such as workability and plating adhesiveness.
- These functions and effects of Al are manifested by setting the Al concentration in the hot dip galvanizing bath to 0.01 to 3%. Therefore, the Al concentration in the hot dip galvanizing bath may be selected according to the capacity and purpose of the equipment to be manufactured.
- the steel of the present invention can be obtained by subjecting the steel sheet of the present invention to hot stamping by a conventional method.
- the contents of the hydrogen embrittlement promotion test for evaluating the hydrogen embrittlement resistance, the content of the critical diffusible hydrogen content, and the content of the Charpy impact test for evaluating the toughness are first described. explain.
- the introduction of diffusible hydrogen into the test piece (steel plate) was performed by the cathodic charging method in an electrolytic solution. That is, the test piece is a cathode, and a platinum electrode arranged around the test piece is used as an anode. A current is passed between the two at a predetermined current density to generate hydrogen on the surface of the test piece, thereby diffusing into the test piece. Urged.
- the electrolytic solution was an aqueous solution in which NH 4 SCN and NaCl were dissolved in pure water by 0.3% and 3%, respectively.
- the tension corresponding to the residual stress which is another factor that causes hydrogen embrittlement, is called a “lever type” constant load tester using a weight (hereinafter referred to as “constant load test”). ").
- the constant load test piece was notched. The time until the test piece broke was recorded, and recovered immediately after the breakage. The electrolytic solution was removed, and immediately, the amount of diffusible hydrogen was measured by a temperature rising hydrogen analysis method using a gas chromatograph. The cumulative release amount from room temperature to 250 ° C. was defined as the diffusible hydrogen amount.
- the toughness was evaluated by a Charpy impact test in accordance with JIS Z 2242 regardless of the presence or absence of plating.
- the shape of the No. 4 test piece of JIS Z 2202 was applied to the test piece, and the thickness of the test piece was determined according to the steel sheet to be evaluated. Tests were conducted in the range of ⁇ 120 ° C. to 20 ° C. to determine the ductile brittle transition temperature.
- Example 1 Steel pieces having the chemical composition shown in Table 1 were cast. These steel slabs were heated to 1250 ° C. and subjected to hot rolling to obtain hot rolled steel sheets having a finishing temperature of 870 to 920 ° C. and a thickness of 2.8 mm. The coiling temperature was 700 ° C. After pickling, cold rolling was performed at a reduction rate of 50% to obtain a cold-rolled steel sheet having a thickness of 1.4 mm. These cold-rolled steel sheets were kept in a temperature range of 700 to 800 ° C. for 1 minute and subjected to recrystallization annealing under the condition of air-cooling to room temperature to obtain test materials (steel sheets for hot stamping).
- a test piece of 50 ⁇ 50 mm was taken from each test material, and constant current electrolysis was performed in an electrolytic solution in which acetylacetone and tetramethylammonium were dissolved in methanol.
- the current value was 500 mA, and the electrolysis time was 4 hours.
- the mass of the residue collected using a filter having a pore diameter of 0.2 ⁇ m was divided by the amount of electrolysis and expressed as a percentage. Thus, the density
- the cross-section of the specimen was observed with an SEM, and the inclusions were analyzed, that is, the constituent elements were investigated by counting, dimension measurement, and EDS. In this way, the number ratio of Mn oxide in the inclusions having a maximum length of 1.0 to 4.0 ⁇ m was determined.
- each test material was held in the atmosphere at 900 ° C. for 3 minutes, and then hot stamped by a method of sandwiching it with an experimental flat press die shown in FIG. That is, as shown in FIG. 2, the steel plate 22 was processed with the upper mold 21a and the lower mold 21b.
- the average cooling rate to 200 ° C. measured with a thermocouple was about 70 ° C./s. From these steel materials after hot stamping, JIS No. 5 tensile test pieces, constant load test pieces shown in FIG. 3, and Charpy impact test pieces were collected.
- the constant load test was performed by applying a tension corresponding to 90% of the tensile strength obtained in the tensile test.
- the current density was 0.01 to 1 mA / cm 2 .
- the measurement of diffusible hydrogen was performed at a heating rate of 100 ° C./hour.
- the Charpy impact test is conducted at test temperatures of 20 ° C, 0 ° C, -20 ° C, -40 ° C, -60 ° C, -80 ° C, -100 ° C, and -120 ° C. Asked.
- the sample specimens were collected in a tensile test piece and a constant load test piece with the tensile direction perpendicular to the rolling direction of the steel sheet, and with a Charpy test piece, the longitudinal direction was parallel to the rolling direction.
- the thickness of the tensile test piece was 1.4 mm, and the thickness of the other test pieces was 1.2 mm by grinding both sides. Table 2 shows the results.
- the steel sheet after hot stamping exhibited a tensile strength of 1500 MPa or more.
- concentration of inclusions containing Mn and the number ratio of Mn oxide in the inclusions having a maximum length of 1.0 to 4.0 ⁇ m are within the scope of the present invention.
- 2, 3, 6 to 10, and 14 to 16 have a critical diffusible hydrogen content Hc of 0.84 ppm or more, a ductile brittle transition temperature of ⁇ 60 ° C. or less, and good hydrogen embrittlement resistance and toughness. Had.
- the concentration of inclusions containing Mn is out of the scope of the present invention.
- the ductile brittle transition temperature was significantly higher than that of the inventive examples having comparable tensile strength, and the toughness was inferior.
- the number ratio of the Mn oxide in the inclusions having the maximum length of 1.0 to 4.0 ⁇ m is No. out of the scope of the present invention.
- Hc was remarkably smaller than that of the examples of the present invention, and the hydrogen embrittlement resistance was inferior.
- No. The concentration of the inclusion containing 13 Mn is within the range of the present invention, but the ductile brittle transition temperature is significantly higher than that of the present invention example having the same tensile strength. It is presumed that this is because the Al content is high (outside the scope of the present invention) and the Al-based oxide is contained at a high concentration.
- Example 2 Steel pieces having the chemical composition shown in Table 3 were cast. These steel slabs were heated to 1250 ° C. and subjected to hot rolling to obtain hot rolled steel sheets having a finishing temperature of 880 to 920 ° C. and a thickness of 3.0 mm. The coiling temperature was 700 ° C. After pickling, the steel sheet was cold-rolled at a reduction rate of 50% to obtain a cold-rolled steel sheet having a thickness of 1.5 mm. These cold-rolled steel sheets were kept in a temperature range of 700 to 800 ° C. for 1 minute and subjected to recrystallization annealing under the condition of air-cooling to room temperature to obtain test materials (steel sheets for hot stamping).
- Example 2 In the same manner as in Example 1, the concentration of Mn-containing inclusions and the ratio of the number of Mn oxides in the inclusions having a maximum length of 1.0 to 4.0 ⁇ m were determined. Further, the test material was held in the atmosphere at 900 ° C. for 5 minutes, and then formed into a hat shape shown in FIG. 4 by a hot stamp method. The average cooling rate to 200 ° C. measured with a thermocouple was about 35 ° C./s. A JIS No. 5 tensile test piece, a constant load test piece, and a Charpy impact test piece were collected from a test piece collection position 41 (hat head) shown in FIG. The relationship between the specimen sampling direction and the steel sheet rolling direction was the same as in Example 1.
- the plate thickness of the tensile test piece was 1.5 mm, and the plate thickness of the other test pieces was 1.3 mm by grinding both sides.
- the constant load test was performed by applying a tension corresponding to 90% of the tensile strength obtained in the tensile test.
- the current density was 0.01 to 1 mA / cm 2 .
- the diffusible hydrogen was measured at a heating rate of 100 ° C./hour.
- the Charpy impact test is conducted at test temperatures of 20 ° C, 0 ° C, -20 ° C, -40 ° C, -60 ° C, -80 ° C, -100 ° C, and -120 ° C. Asked. Table 4 shows the results.
- the steel sheet after hot stamping exhibited a tensile strength of 1580 MPa or more.
- both the concentration of inclusions containing Mn and the number ratio of Mn oxide in the inclusions having a maximum length of 1.0 to 4.0 ⁇ m are within the scope of the present invention.
- No. 18 to 24, 27, 28, and 31 had Hc of 0.91 ppm or more and a ductile brittle transition temperature of ⁇ 65 ° C. or less, and had good hydrogen embrittlement resistance and toughness.
- the concentration of inclusions containing Mn is higher than the range of the present invention. In Nos. 17 and 25, the ductile brittle transition temperature was significantly higher than that of the inventive examples, and the toughness was inferior.
- the number ratio of the Mn oxide in the inclusions having the maximum length of 1.0 to 4.0 ⁇ m is No. out of the scope of the present invention.
- Hc is smaller than the example of the present invention and inferior in hydrogen embrittlement resistance.
- No. The ratio of the number of 25 Mn oxides is within the scope of the present invention, but Hc is small. This is because the Mn content and O content are high (outside the scope of the present invention), and the size distribution of the Mn oxide is biased toward the larger side compared to the present invention example. It is presumed that there are few voids.
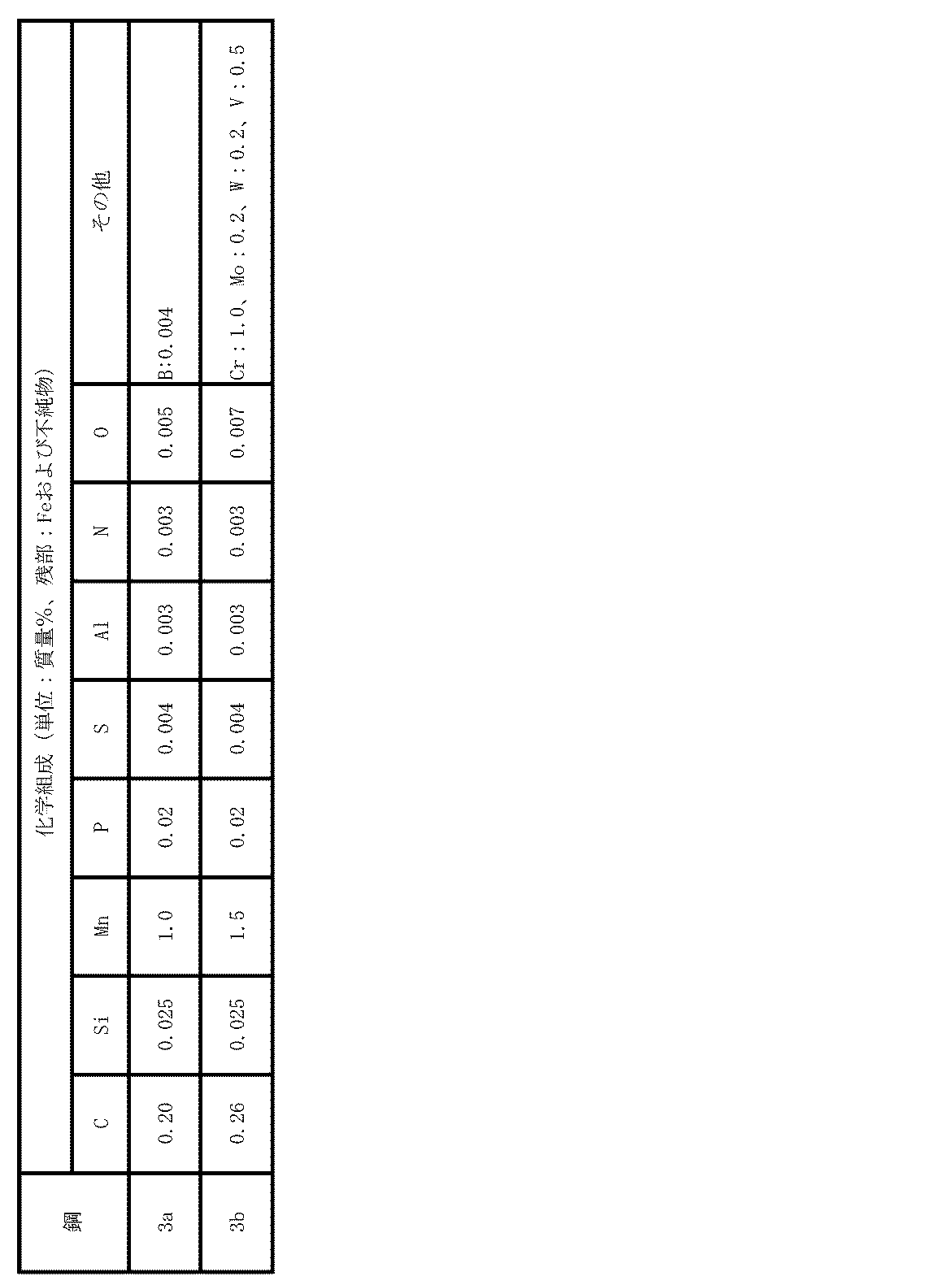
- Example 3 Steel pieces having the chemical composition shown in Table 5 were cast. These steel slabs were heated to 1200 ° C. and subjected to hot rolling to obtain hot rolled steel sheets having a finishing temperature of 880 to 920 ° C. and a thickness of 2.0 to 4.0 mm.
- the cooling conditions in the cooling bed (ROT) were controlled, and winding was performed at a plurality of winding temperatures. After pickling, cold rolling was performed at a reduction rate of 50% to obtain a cold rolled steel sheet.
- These cold-rolled steel sheets were held at 700 to 800 ° C. for 1 minute and subjected to recrystallization annealing under the condition of air-cooling to room temperature to obtain test materials (steel sheets for hot stamping).
- Example 2 In the same manner as in Example 1, the concentration of Mn-containing inclusions and the number ratio of Mn oxide in the Mn-containing inclusions having a maximum length of 1.0 to 4.0 ⁇ m were determined.
- Hot stamping was performed using the same flat plate mold as in Example 1.
- Tensile test pieces, constant load test pieces, and Charpy impact test pieces were collected from the steel sheet after hot stamping in the same manner as in Example 1.
- the plate thickness of the test piece was the same as that of the cold-rolled steel plate for the tensile test piece, and the other test pieces were formed by grinding 0.1 mm on both sides from the plate thickness of the cold-rolled steel plate.
- the constant load test, the measurement of diffusible hydrogen, and the Charpy impact test were also performed in the same manner as in Example 1.
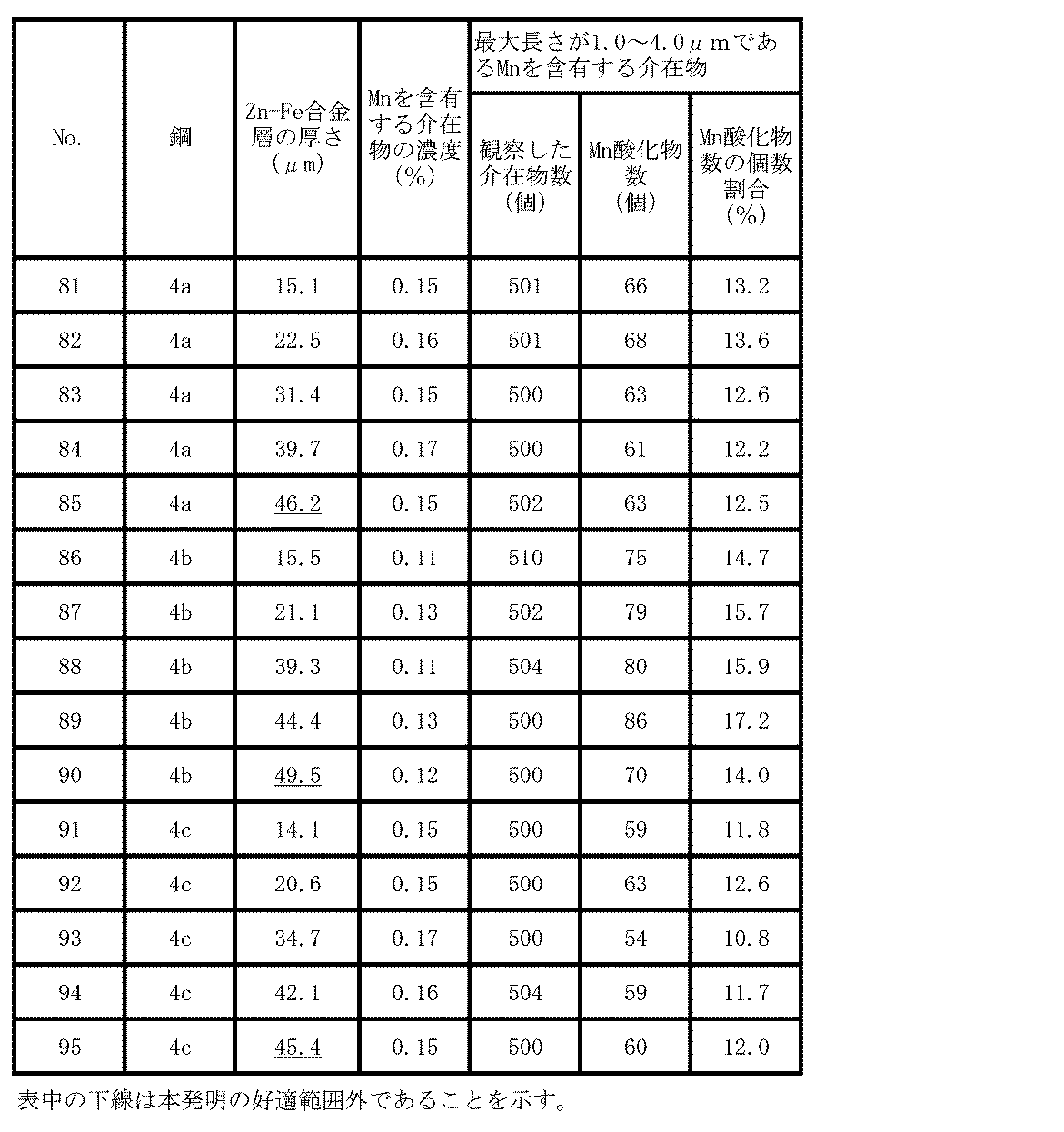
- Table 6 shows the finished sheet thickness of the hot-rolled sheet, the coiling temperature, the investigation results of inclusions, the hydrogen embrittlement resistance (Hc), and the toughness.
- the tensile strength of the steel sheet after hot stamping showed a tensile strength of 1500 to 1520 MPa for steel 3a and 1587 to 1622 MPa for steel 3b, regardless of the finished thickness.
- the lower the winding temperature the higher the tensile strength, and it is presumed that the strength of the test material is affected by the winding temperature.
- the concentration of inclusions containing Mn was within the scope of the present invention in any example, but the winding temperature deviated from the scope of the present invention.
- the number ratio of the Mn oxide in the inclusions containing Mn having a maximum length of 1.0 to 4.0 ⁇ m is out of the scope of the present invention ( Reflecting this, Hc is significantly smaller than the two examples of the present invention having the same finished plate thickness of the same steel, inferior in hydrogen brittleness resistance, and the ductile brittle transition temperature is also low. It was high and inferior in toughness as compared with two examples of the present invention having the same finished plate thickness of the same steel.
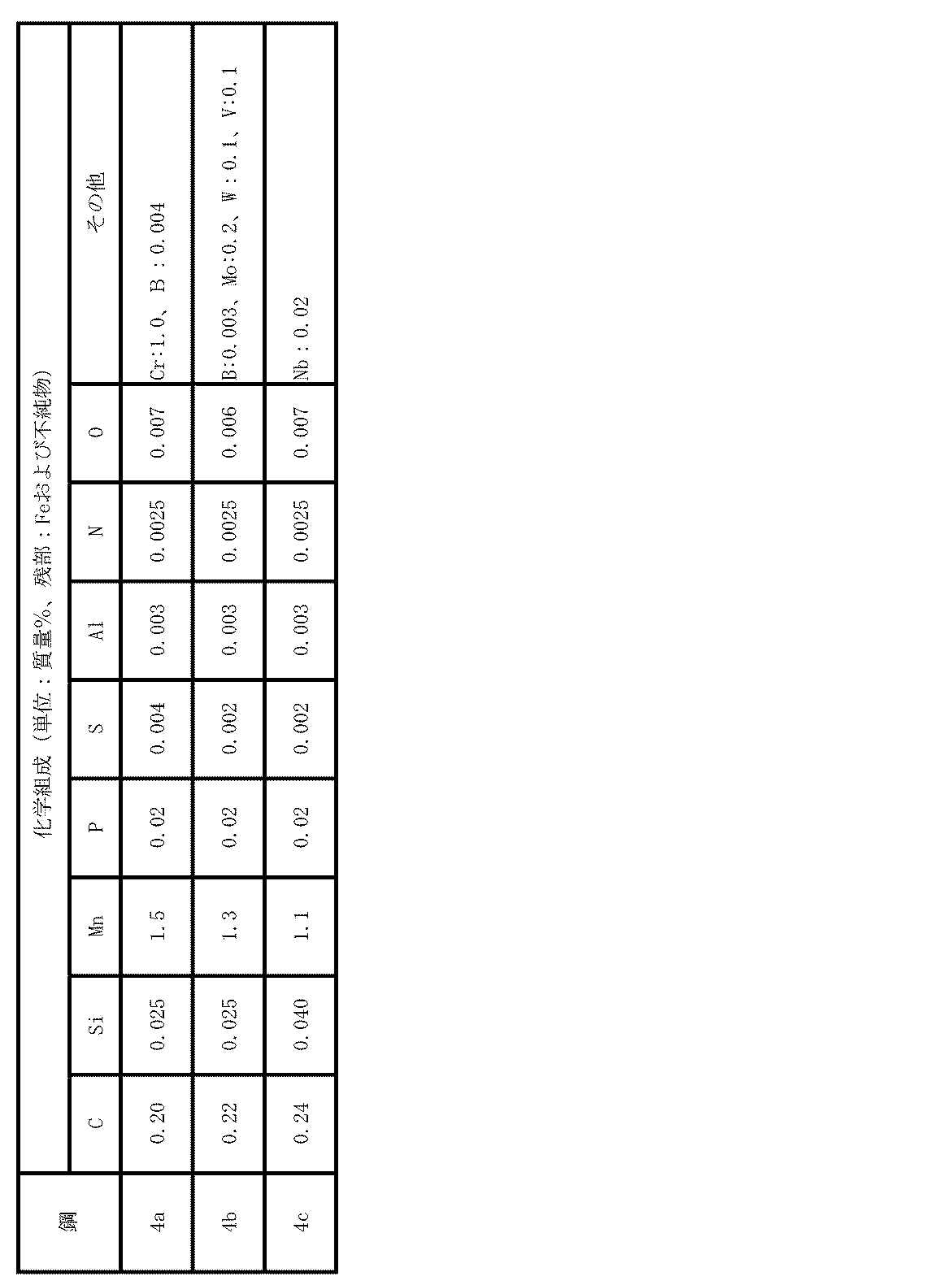
- Example 4 Steel pieces having the chemical composition shown in Table 7 were produced. These steel slabs were made into hot-rolled steel sheets having a thickness of 2.8 mm under the same conditions as in Example 1. After pickling, the steel pieces were cold-rolled (rolling ratio: 50%) to steel sheets having a thickness of 1.4 mm. These cold-rolled steel sheets were heated to 655 ° C. at an average heating rate of 19 ° C./s, followed by heating to 730-780 ° C. at an average heating rate of 2.5 ° C./s, immediately with an average cooling rate of 6.5. After cooling at °C / s and dipping in a 670 ° C.
- Example 2 a hat was hot stamped, and a JIS No. 5 tensile test piece, a punch test piece, and a Charpy impact test piece were collected from the head of the hat.
- the hot stamp was heated at 900 ° C. for 1 minute, the atmosphere was nitrogen containing 3% hydrogen, and the dew point was 0 ° C.
- Table 8 shows the analysis results regarding the inclusions
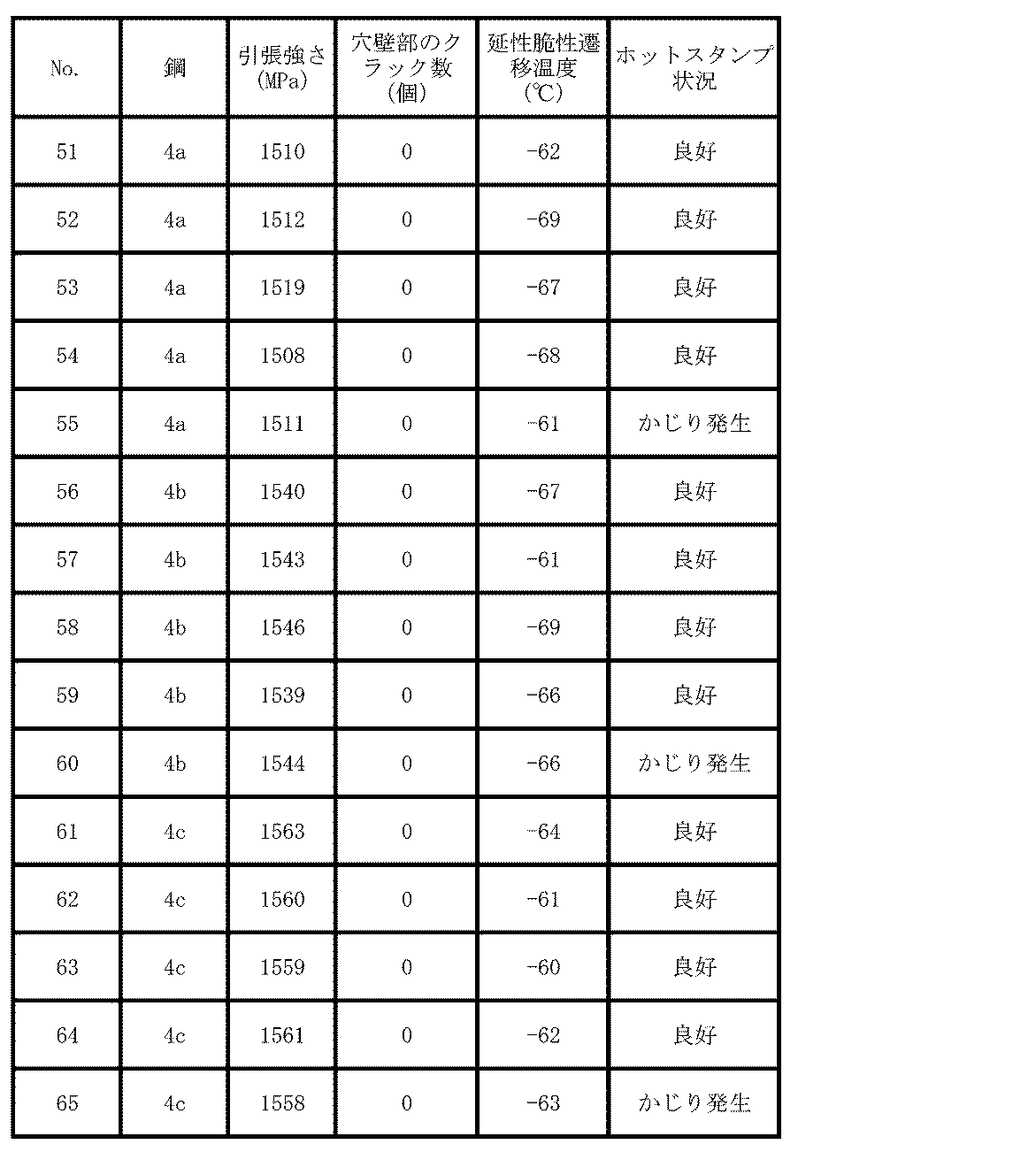
- Table 9 summarizes the test results regarding the hot stamp material.
- the concentration of Mn-containing inclusions and the number ratio of Mn oxide in the inclusions containing Mn having a maximum length of 1.0 to 4.0 ⁇ m are within the scope of the present invention. Therefore, the occurrence of cracks in the hole wall in the drilling test was not observed, and the ductile brittle transition temperature was ⁇ 60 ° C. or less, and a steel plate (member) having both hydrogen embrittlement resistance and toughness was obtained. No. with the thickness of the Al plating layer exceeding 50 ⁇ m. In 55, 60 and 65, galling occurred frequently in the hat-shaped vertical wall. On the other hand, no. In 51 to 54, 56 to 59, and 61 to 64, no galling occurred in the hat-shaped vertical wall portion.
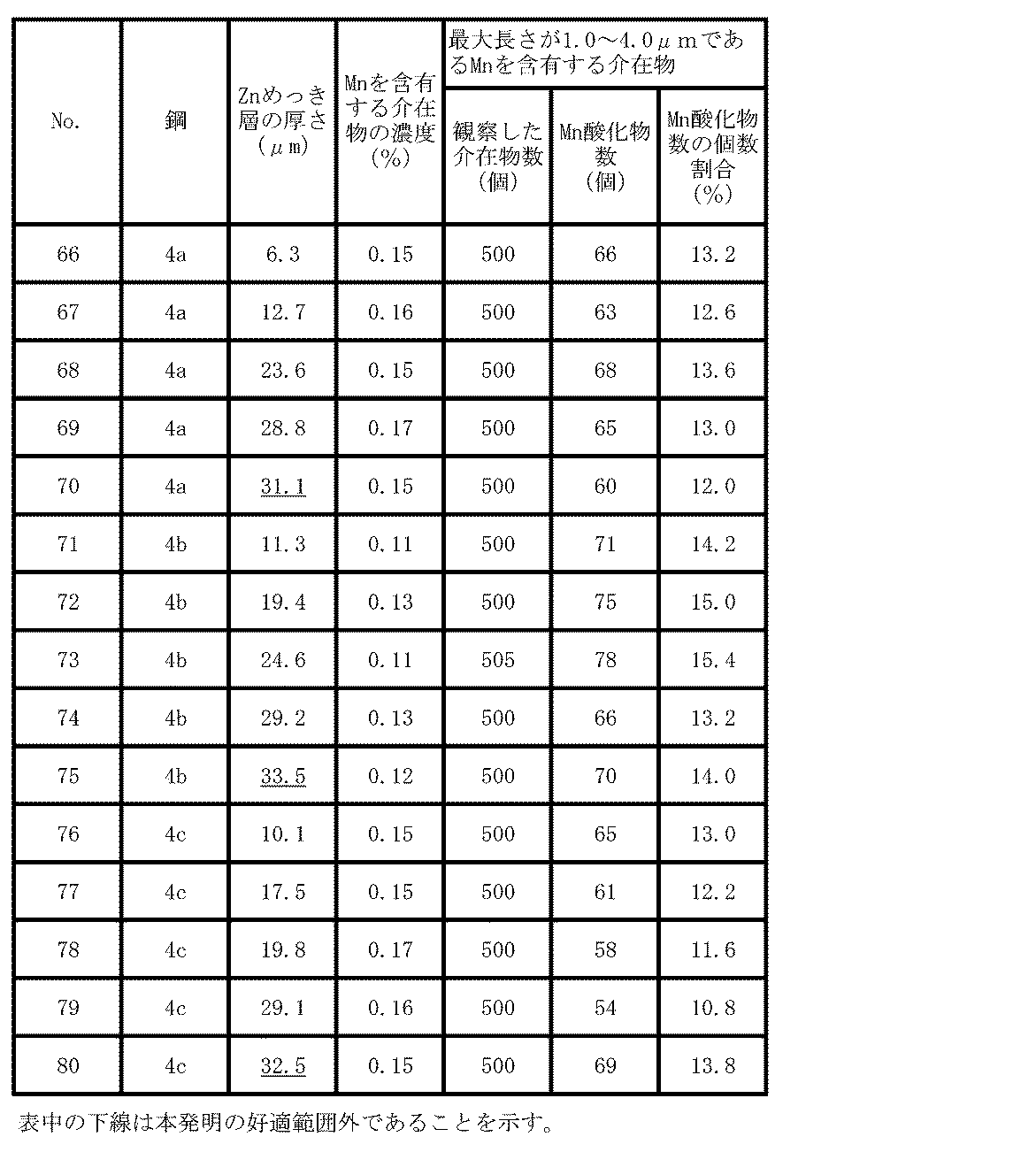
- Example 5 A steel piece having the chemical composition shown in Table 7 was formed into a hot-rolled steel sheet having a thickness of 2.8 mm under the same conditions as in Example 1. After pickling, the steel pieces were cold-rolled to a steel sheet having a thickness of 1.2 mm. These cold-rolled steel sheets were heated to 655 ° C. at an average heating rate of 19 ° C./s, followed by heating to 730-780 ° C. at an average heating rate of 2.5 ° C./s, immediately with an average cooling rate of 6.5.
- Example 2 Cooled at °C / s, immersed in a hot-dip galvanizing bath (containing 0.15% Al and impurities) at 460 ° C, taken out after 3 seconds, adjusted the amount of adhesion with a gas wiper, and then air-cooled to room temperature .
- Analysis of inclusions in the obtained steel plate was performed in the same manner as in Example 1. Further, in the same manner as in Example 2, a hat was hot stamped, and a JIS No. 5 tensile test piece, a perforated test piece, and a Charpy impact test piece were collected from the head of the hat. The hot stamp was heated at 900 ° C. for 1 minute, the atmosphere was nitrogen containing 3% hydrogen, and the dew point was 0 ° C. Table 10 shows the analysis results for inclusions, and Table 11 summarizes the test results for the hot stamp material.
- the concentration of Mn-containing inclusions and the ratio of the number of Mn oxides to inclusions containing Mn having a maximum length of 1.0 to 4.0 ⁇ m are within the scope of the present invention.
- No cracks were observed on the hole wall in the drilling test, and the ductile brittle transition temperature was ⁇ 60 ° C. or lower, and a steel plate (member) having both hydrogen embrittlement resistance and toughness was obtained.
- the thickness of the Zn plating layer was 30 ⁇ m or less. In 66 to 69, 71 to 74, and 76 to 79, no Zn adhesion to the mold occurred.
- Example 6 A steel slab having the chemical composition shown in Table 7 is a hot-rolled steel sheet having a thickness of 2.8 mm under the same conditions as in Example 1. After pickling, the steel slab is cold-rolled into a steel sheet having a thickness of 1.4 mm (reduction rate: 50%). These cold-rolled steel sheets were heated to 655 ° C. at an average heating rate of 19 ° C./s, followed by heating to 730-780 ° C. at an average heating rate of 2.5 ° C./s, immediately with an average cooling rate of 6.5.
- Example 2 a hat was hot stamped, and a JIS No. 5 tensile test piece, a perforated test piece, and a Charpy impact test piece were collected from the head of the hat.
- the hot stamp was heated at 900 ° C. for 1 minute, the atmosphere was nitrogen containing 3% hydrogen, and the dew point was 0 ° C.
- Table 12 summarizes the analysis results for inclusions
- Table 13 summarizes the test results for the hot stamp material.
- the concentration of Mn-containing inclusions and the ratio of the number of Mn oxides to inclusions containing Mn having a maximum length of 1.0 to 4.0 ⁇ m are within the scope of the present invention.
- a steel plate (member) having both hydrogen embrittlement resistance and toughness was obtained.
- the thickness of the galvannealed layer is 45 ⁇ m or less.
- 81 to 84, 86 to 89, and 91 to 94 no fine cracks occurred in the alloy layer after pressing.
- the hot stamping method can be obtained because it can ensure good hydrogen embrittlement resistance and is easy to implement even when processing such as drilling is performed after hot stamping.
- the application range (parts) can be expanded. Therefore, the present invention has high applicability in the steel plate processing industry.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Thermal Sciences (AREA)
- Physics & Mathematics (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Crystallography & Structural Chemistry (AREA)
- Heat Treatment Of Sheet Steel (AREA)
- Heat Treatment Of Articles (AREA)
- Heat Treatment Of Steel (AREA)
Abstract
Description
本発明者らは、鋼中に生成させることが比較的容易な、Mnを含有する介在物およびMn酸化物に着目し、これらを拡散性水素および非拡散性水素のトラップサイトとして機能させることにより、良好な耐水素脆化特性を確保することを新たに着想した。
(1)質量%で、
C:0.18~0.26%、
Si:0.02%超0.05%以下、
Mn:1.0~1.5%、
P:0.03%以下、
S:0.02%以下、
Al:0.001~0.5%、
N:0.1%以下、
O:0.0010~0.020%、
Cr:0~2.0%、
Mo:0~1.0%
V:0~0.5%、
W:0~0.5%、
Ni:0~5.0%
B:0~0.01%
Ti:0~0.5%、
Nb:0~0.5%、
Cu:0~1.0%、
残部:Feおよび不純物である化学組成を有し、
Mnを含有する介在物の濃度が0.010質量%以上0.25質量%未満、かつ、最大長さが1.0~4.0μmである前記介在物に占めるMn酸化物の個数割合が10.0%以上であることを特徴とするホットスタンプ用鋼板。
Cr:0.01~2.0%、
Mo:0.01~1.0%
V:0.01~0.5%、
W:0.01~0.5%、
Ni:0.01~5.0%および
B:0.0005~0.01%
からなる群から選択された1種または2種以上を含有することを特徴とする上記(1)に記載のホットスタンプ用鋼板。
Ti:0.001~0.5%、
Nb:0.001~0.5%および
Cu:0.01~1.0%
からなる群から選択された1種または2種以上を含有することを特徴とする上記(1)または(2)に記載のホットスタンプ用鋼板。
C:0.18~0.26%、
Si:0.02%超0.05%以下、
Mn:1.0~1.5%、
P:0.03%以下、
S:0.02%以下、
Al:0.001~0.5%、
N:0.1%以下、
O:0.0010~0.020%、
Cr:0~2.0%、
Mo:0~1.0%
V:0~0.5%、
W:0~0.5%、
Ni:0~5.0%
B:0~0.01%
Ti:0~0.5%、
Nb:0~0.5%、
Cu:0~1.0%、
残部がFeおよび不純物である化学組成を有する鋼片に熱間圧延を施した後に690℃以上の温度域で巻き取って熱間圧延鋼板とする熱間圧延工程と、前記熱間圧延鋼板に10~90%の圧下率の冷間圧延を施して冷間圧延鋼板とする冷間圧延工程とを含むことを特徴とするホットスタンプ用鋼板の製造方法。
Cr:0.01~2.0%、
Mo:0.01~1.0%
V:0.01~0.5%、
W:0.01~0.5%、
Ni:0.01~5.0%および
B:0.0005~0.01%
からなる群から選択された1種または2種以上を含有することを特徴とする上記(7)に記載のホットスタンプ用鋼板の製造方法。
Ti:0.001~0.5%、
Nb:0.001~0.5%および
Cu:0.01~1.0%
からなる群から選択された1種または2種以上を含有することを特徴とする上記(7)または(8)に記載のホットスタンプ用鋼板の製造方法。
C:0.18~0.26%、
Si:0.02%超0.05%以下、
Mn:1.0~1.5%、
P:0.03%以下、
S:0.02%以下、
Al:0.001~0.5%、
N:0.1%以下、
O:0.0010~0.020%、
Cr:0~2.0%、
Mo:0~1.0%
V:0~0.5%、
W:0~0.5%、
Ni:0~5.0%
B:0~0.01%
Ti:0~0.5%、
Nb:0~0.5%、
Cu:0~1.0%、
残部:Feおよび不純物である化学組成を有し、
Mnを含有する介在物の濃度が0.010質量%以上0.25質量%未満、かつ、最大長さが1.0~4.0μmである前記介在物に占めるMn酸化物の個数割合が10.0%以上であることを特徴とするホットスタンプ鋼材。
Cr:0.01~2.0%、
Mo:0.01~1.0%
V:0.01~0.5%、
W:0.01~0.5%、
Ni:0.01~5.0%および
B:0.0005~0.01%
からなる群から選択された1種または2種以上を含有することを特徴とする上記(13)に記載のホットスタンプ鋼材。
Ti:0.001~0.5%、
Nb:0.001~0.5%および
Cu:0.01~1.0%
からなる群から選択された1種または2種以上を含有することを特徴とする上記(13)または(14)に記載のホットスタンプ鋼材。
本発明に係るホットスタンプ用鋼板(以下、「本発明鋼板」ともいう。)およびホットスタンプ鋼材(以下、「本発明鋼材」ともいう。)の化学組成の限定理由について説明する。なお、以下の説明における「%」は「質量%」を意味する。
Cは、ホットスタンプ法によって鋼板を高強度化する上で最も重要な元素である。C含有量が0.18%未満では、ホットスタンプ後において1500MPa以上の強度を確保することが困難となる。したがって、C含有量は0.18%以上とする。
一方、C含有量が0.26%超では、ホットスタンプ後における延性が乏しくなり、10%以上の全伸びを確保することが困難となる。したがって、C含有量は0.26%以下とする。
Siは、Mnを含有する介在物の濃度および最大長さが1.0~4.0μmである前記介在物に占めるMn酸化物の個数割合を制御する上で重要な元素である。Si含有量が0.02%以下では、Mn酸化物の生成が過度に促進され、Mnを含有する介在物の濃度が0.25%以上となり、靭性の低下が著しくなる場合がある。したがって、Si含有量は0.02%超とする。一方、Si含有量が0.05%超では、Mn酸化物の生成が過度に抑制され、最大長さが1.0~4.0μmであるMnを含有する介在物に占めるMn酸化物の個数割合が10.0%未満となり、良好な耐水素脆化特性を安定して得ることが難しい。したがって、Si含有量は0.05%以下とする。
Mnは、本発明において最も重要な元素である。Mnは、鋼中にMnを含有する介在物を形成することにより、耐水素脆性を高める作用を有する。また、介在物を形成しなかった残りのMnは、焼入れ性を高める作用を有する。Mn含有量が1.0%未満では、Mnを含有する介在物の濃度を0.010質量%以上とすることが困難となる。したがって、Mn含有量は1.0%以上とする。一方、Mn含有量が1.5%を超えると、上記作用による効果は飽和してしまい、経済的に不利となり、加えて、Mnの偏析に起因する機械特性の低下を招く場合がある。したがって、Mn含有量は1.5%以下とする。
Pは、一般に不純物として含有される元素である。P含有量が0.03%超では熱間加工性の低下が著しくなる。したがって、P含有量は0.03%以下とする。P含有量の下限は特に規定する必要はないが、過度の低減は製鋼工程に多大な負荷をかけるので、0.001%以上とすることが好ましい。
Sは、一般に不純物として含有される元素である。S含有量が0.02%超では、熱間加工性の低下が著しくなる。したがって、S含有量は0.02%以下とする。S含有量の下限は特に規定する必要はないが、過度の低減は製鋼工程に多大な負荷をかけるので、0.0005%以上とすることが好ましい。
Alは、脱酸により鋼を健全化する作用を有する元素である。Al含有量が0.001%未満では、十分な脱酸を行うことが困難である。したがって、Al含有量は0.001%以上とする。一方、Al含有量が0.5%超では、Mn酸化物の生成が過度に抑制され、後述するMn酸化物の比率を確保することが困難となり、良好な耐水素脆化特性を確保することが困難となる。したがって、Al含有量は0.5%以下とする。
Nは、一般に不純物として含有される元素である。N含有量が0.1%超では、後述する任意元素であるTiやBと容易に結合して消費してしまい、それらの元素の作用効果を減じてしまう。したがって、N含有量は0.1%以下とし、好ましくは0.01%以下とする。N含有量の下限は特に規定する必要はないが、過度の低減は製鋼工程に多大な負荷をかけるので、0.001%以上とすることが好ましい。
Oは、鋼中にMn酸化物を形成し、拡散性水素および非拡散性水素のトラップサイトとして機能し、耐水素脆化特性を高める作用を有する。O含有量が0.0010%未満では、Mn酸化物の生成が十分に促進されず、Mnを含有する介在物に占めるMn酸化物の個数割合が10.0%未満となり、良好な耐水素脆化特性を安定して得られない。したがって、O含有量は0.0010%以上とする。一方、O含有量が0.020%超では、鋼中に粗大な酸化物を形成して、鋼材の機械的特性を劣化させる。したがって、O含有量は0.020%以下とする。
これらの元素は、いずれも焼入れ性を高める作用を有する。したがって、これらの元素の1種または2種以上を含有させてもよい。しかしながら、Bについては、上記上限値を超えて含有させると熱間加工性の劣化と延性の低下をもたらす。また、Cr、Mo、W、VおよびNiについては、上記上限値を超えて含有させても上記作用による効果は飽和してしまい、コスト的に不利になる。したがって、B、Cr、Mo、W、VおよびNiの含有量の上限値はそれぞれ上記のとおりとする。なお、上記作用による効果をより確実に得るには、B含有量を0.0005%以上とするか、Cr、Mo、W、VおよびNiのいずれかの元素の含有量を0.01%以上とすることが好ましい。また、Niは、Cuによる熱間圧延鋼板の表面性状の劣化を抑制する作用を有するので、後述するCuを含有させる場合には、Niも含有させることが好ましい。
Ti、NbおよびCuは、いずれも強度を高める作用を有する。したがって、これらの元素の1種または2種以上を含有させてもよい。しかしながら、Ti含有量が0.5%を超えると、Mn酸化物の生成が過度に抑制され、後述するMn酸化物の比率を確保することが困難となり、良好な耐水素脆化特性を確保することが困難となる。したがって、Ti含有量は0.5%とする。また、Nb含有量が0.5%を超えると、熱間圧延の制御性を損ねる場合がある。したがって、Nb含有量は0.5%以下とする。また、Cu含有量が1.0%を超えると、熱間圧延鋼板の表面性状を損ねる場合がある。したがって、Cu含有量は1.0%以下とする。上記作用による効果をより確実に得るには、Ti:0.001%以上、Nb:0.001%以上およびCu:0.01%以上のいずれかを含有させることが好ましい。また、Tiは、鋼中のNと優先的に結合して窒化物を形成することにより、Bが窒化物の形成によって浪費されるのを抑制し、Bによる作用効果をより高めることを可能にするので、上述したBを含有させる場合にはTiも含有させることが好ましい。
次に、本発明鋼板および本発明鋼材における、Mnを含有する介在物の濃度および最大長さが1.0~4.0μmであるMnを含有する介在物の数に占めるMn酸化物の個数割合に関する限定理由について説明する。
Mnを含有する介在物は、後述する最大長さが1.0~4.0μmであるMnを含有する介在物の数に占めるMn酸化物の個数割合とともに、水素脆化の抑制に重要な役割を果たす。Mnを含有する介在物の濃度が0.010%未満では、良好な耐水素脆化特性を得ることが困難である。したがって、Mnを含有する介在物の濃度は0.010%以上とする。一方、Mnを含有する介在物の濃度が0.25%以上では、靭性の低下をもたらす場合がある。したがって、Mnを含有する介在物の濃度は0.25%未満とする。
最大長さが1.0~4.0μmであるMnを含有する介在物の数に占めるMn酸化物の個数割合は、上述したMnを含有する介在物とともに、水素脆化の抑制に重要な役割を果たす。最大長さが1.0~4.0μmであるMnを含有する介在物の数に占めるMn酸化物の個数割合が10.0%未満では、良好な耐水素脆化特性を得ることが困難である。したがって、最大長さが1.0~4.0μmであるMnを含有する介在物の数に占めるMn酸化物の個数割合は10.0%以上とする。
本発明鋼板および本発明鋼材は、表面に、耐食性の向上等を目的としてめっき層を形成して表面処理鋼板または表面処理鋼材としてもよい。めっき層は溶融めっき層であってもよく電気めっき層であってもよい。溶融めっき層としては、溶融亜鉛めっき、合金化溶融亜鉛めっき、溶融アルミニウムめっき、溶融Zn-Al合金めっき、溶融Zn-Al-Mg合金めっき、溶融Zn-Al-Mg-Si合金めっき等が例示される。電気めっき層としては、電気亜鉛めっき、電気Zn-Ni合金めっき等が例示される。
本発明鋼板の製造方法について説明する。
本発明鋼板は、上記化学組成を有する鋼片に熱間圧延を施した後に690℃以上の温度域で巻き取って熱間圧延鋼板とする熱間圧延工程と、前記熱間圧延鋼板に10~90%の圧下率の冷間圧延を施して冷間圧延鋼板とする冷間圧延工程とを含む製造方法により製造することができる。ここで、鋼片を製造する際の製鋼条件および鋳造条件ならびに熱延鋼板に施す冷間圧延の条件は常法によればよい。また、熱延鋼板を冷間圧延に供する前に施す酸洗も常法によればよい。
本発明鋼材は、本発明鋼板に常法によりホットスタンプを施すことにより得ることができる。
拡散性水素の試験片(鋼板)中への導入は、電解液中での陰極チャージ法で行った。すなわち、試験片を陰極、試験片の周囲に配した白金電極を陽極とし、両者の間に所定の電流密度で通電して、試験片の表面に水素を発生させ、試験片内部への拡散を促した。電解液は、純水に、NH4SCN及びNaClを、それぞれ0.3%及び3%溶解させた水溶液とした。
表1に示す化学組成を有する鋼片を鋳造した。これらの鋼片を1250℃に加熱して熱間圧延に供し、仕上温度870~920℃で、厚さ2.8mmの熱延鋼板とした。巻取温度は700℃とした。酸洗後、50%の圧下率で冷間圧延を施して、板厚1.4mmの冷延鋼板を得た。それらの冷延鋼板を、700~800℃の温度域に1分間保持し、室温まで空冷する条件で再結晶焼鈍を施し、供試材(ホットスタンプ用鋼板)とした。
表3に示す化学組成を有する鋼片を鋳造した。これら鋼片を1250℃に加熱して熱間圧延に供し、仕上温度880~920℃で、厚さ3.0mmの熱延鋼板とした。巻取温度は700℃とした。酸洗後、50%の圧下率で冷間圧延して、板厚1.5mmの冷延鋼板を得た。それらの冷延鋼板を、700~800℃の温度域に1分間保持し、室温まで空冷する条件で再結晶焼鈍を施し、供試材(ホットスタンプ用鋼板)とした。実施例1と同じ方法で、Mnを含有する介在物の濃度および最大長さが1.0~4.0μmである前記介在物に占めるMn酸化物の個数割合を求めた。さらに、供試材を、大気中で900℃に5分間保持した後、図4に示すハット型に、ホットスタンプ法で成形した。熱電対を付けて測定した200℃までの平均冷却速度は、約35℃/sであった。図4に示す試験片採取位置41(ハット頭部)から、JIS5号引張試験片、定荷重試験片、及び、シャルピー衝撃試験片を採取した。試験片の採取方向と、鋼板の圧延方向の関係は、実施例1と同じとした。引張試験片の板厚は1.5mmとし、その他の試験片の板厚は、両面を研削し、1.3mmとした。定荷重試験は、引張試験で求めた引張強さの90%に相当する張力を付加して行った。電流密度は0.01~1mA/cm2とした。拡散性水素の測定は、加熱速度100℃/時間で行った。シャルピー衝撃試験は、試験温度20℃、0℃、-20℃、-40℃、-60℃、-80℃、-100℃、及び、-120℃で行い、吸収エネルギーの変化から延性脆性遷移温度を求めた。表4に結果を示す。
一方、Mnを含有する介在物の濃度が本発明の範囲よりも高いNo.17および25では、延性脆性遷移温度が、本発明例に比べて大幅に高く、靭性に劣っていた。また、最大長さが1.0~4.0μmである前記介在物に占めるMn酸化物の個数割合が本発明の範囲を外れるNo.26、29、30および32では、Hcが本発明例に比べて小さく、耐水素脆化特性に劣ることが解る。なお、No.25のMn酸化物の数の割合は本発明の範囲内であるが、Hcが小さい。これはMn含有量およびO含有量が高い(本発明の範囲外)ことで、Mn酸化物の大きさの分布が、本発明例に比べて大きい側に偏っているため、鋼との間の空隙が少ないからではないかと推定される。
表5に示す化学組成を有する鋼片を鋳造した。これらの鋼片を1200℃に加熱して熱間圧延に供し、仕上温度880~920℃で、厚さ2.0~4.0mmの熱延鋼板とした。冷却床(ROT)での冷却条件を制御して、複数の巻取温度で巻き取った。酸洗後、50%の圧下率で冷間圧延を施して冷延鋼板を得た。それらの冷延鋼板を、700~800℃に1分間保持し、室温まで空冷する条件で再結晶焼鈍を施し、供試材(ホットスタンプ用鋼板)とした。実施例1と同じ方法で、Mnを含有する介在物の濃度および最大長さが1.0~4.0μmであるMnを含有する介在物に占めるMn酸化物の個数割合を求めた。ホットスタンプは、実施例1と同じ平板金型で行った。ホットスタンプ後の鋼板から、引張試験片、定荷重試験片、及び、シャルピー衝撃試験片を、実施例1と同じ要領で採取した。試験片の板厚は、引張試験片は冷延鋼板と同じとし、他の試験片は、冷延鋼板の板厚から両面を0.1mm研削した厚さとした。定荷重試験、拡散性水素の測定およびシャルピー衝撃試験も実施例1と同じ要領で実施した。熱延板の仕上板厚、巻取温度、介在物の調査結果、耐水素脆化特性(Hc)および靭性を、まとめて表6に示す。
表7に示す化学組成を有する鋼片を製造した。これらの鋼片を、実施例1と同じ条件で、厚さ2.8mmの熱延鋼板とし、酸洗後、板厚1.4mmの鋼板に冷間圧延(圧下率:50%)した。これらの冷延鋼板を、平均加熱速度19℃/sで655℃まで加熱し、続いて、平均加熱速度2.5℃/sで730~780℃まで加熱し、直ちに、平均冷却速度6.5℃/sで冷却して、670℃の溶融アルミニウムめっき浴(10%のSiと不純物を含有)に浸漬し、5秒後に取り出し、ガスワイパーで付着量を調整した後、室温まで空冷した。得られた鋼板の介在物の分析を、実施例1と同じ要領で行った。また、実施例2と同じ要領で、ハット型にホットスタンプし、ハット頭部から、JIS5号引張試験片、穿孔試験用試験片、及び、シャルピー衝撃試験片を採取した。なお、ホットスタンプの加熱条件は、900℃に1分保持とし、雰囲気は、水素を3%含有した窒素とし、露点は0℃とした。表8に、介在物に関する分析結果を示し、表9に、ホットスタンプ材に関する試験結果をまとめて示す。
表7に示す化学組成を有する鋼片を、実施例1と同じ条件で、厚さ2.8mmの熱延鋼板とし、酸洗後、板厚1.2mmの鋼板に冷間圧延した。これらの冷延鋼板を、平均加熱速度19℃/sで655℃まで加熱し、続いて、平均加熱速度2.5℃/sで730~780℃まで加熱し、直ちに、平均冷却速度6.5℃/sで冷却して、460℃の溶融亜鉛めっき浴(0.15%のAlと不純物を含有)に浸漬し、3秒後に取り出し、ガスワイパーで付着量を調整した後、室温まで空冷した。得られた鋼板の介在物の分析を、実施例1と同じ要領で行った。また、実施例2と同じ要領で、ハット型にホットスタンプし、ハット頭部から、JIS5号引張試験片、穿孔試験片、及び、シャルピー衝撃試験片を採取した。なお、ホットスタンプの加熱条件は、900℃に1分保持とし、雰囲気は、水素を3%含有した窒素とし、露点は0℃とした。表10に、介在物に関する分析結果を示し、表11に、ホットスタンプ材に関する試験結果をまとめて示す。
表7に示す化学組成を有する鋼片を、実施例1と同じ条件で、厚さ2.8mmの熱延鋼板とし、酸洗後、板厚1.4mmの鋼板に冷間圧延(圧下率:50%)した。これらの冷延鋼板を、平均加熱速度19℃/sで655℃まで加熱し、続いて、平均加熱速度2.5℃/sで730~780℃まで加熱し、直ちに、平均冷却速度6.5℃/sで冷却して、460℃の溶融亜鉛めっき浴(0.13%のAl、0.03%のFeおよび不純物を含有)に浸漬し、3秒後に取り出し、ガスワイパーで付着量を調整した後、480℃に加熱して合金化溶融亜鉛めっき層を形成し、その後、室温まで空冷した。得られた鋼板の介在物の分析を、実施例1と同じ要領で行った。また、実施例2と同じ要領で、ハット型にホットスタンプし、ハット頭部から、JIS5号引張試験片、穿孔試験片、及び、シャルピー衝撃試験片を採取した。なお、ホットスタンプの加熱条件は、900℃に1分保持とし、雰囲気は、水素を3%含有した窒素とし、露点は0℃とした。表12に、介在物に関する分析結果を、表13に、ホットスタンプ材に関する試験結果をまとめて示す。
21b 下金型
22 鋼板
41 試験片採取位置
Claims (15)
- 質量%で、
C:0.18~0.26%、
Si:0.02%超0.05%以下、
Mn:1.0~1.5%、
P:0.03%以下、
S:0.02%以下、
Al:0.001~0.5%、
N:0.1%以下、
O:0.0010~0.020%、
Cr:0~2.0%、
Mo:0~1.0%
V:0~0.5%、
W:0~0.5%、
Ni:0~5.0%
B:0~0.01%
Ti:0~0.5%、
Nb:0~0.5%、
Cu:0~1.0%、
残部:Feおよび不純物である化学組成を有し、
Mnを含有する介在物の濃度が0.010質量%以上0.25質量%未満、かつ、最大長さが1.0~4.0μmである前記介在物に占めるMn酸化物の個数割合が10.0%以上であることを特徴とするホットスタンプ用鋼板。 - 前記化学組成が、質量%で、
Cr:0.01~2.0%、
Mo:0.01~1.0%
V:0.01~0.5%、
W:0.01~0.5%、
Ni:0.01~5.0%および
B:0.0005~0.01%
からなる群から選択された1種または2種以上を含有することを特徴とする請求項1に記載のホットスタンプ用鋼板。 - 前記化学組成が、質量%で、
Ti:0.001~0.5%、
Nb:0.001~0.5%および
Cu:0.01~1.0%
からなる群から選択された1種または2種以上を含有することを特徴とする請求項1または2に記載のホットスタンプ用鋼板。 - 表面に厚さ50μm以下の溶融アルミニウムめっき層を有することを特徴とする請求項1~3のいずれか1項に記載のホットスタンプ用鋼板。
- 表面に厚さ30μm以下の溶融亜鉛めっき層を有することを特徴とする請求項1~3のいずれか1項に記載のホットスタンプ用鋼板。
- 表面に厚さ45μm以下の合金化溶融亜鉛めっき層を有することを特徴とする請求項1~3のいずれか1項に記載のホットスタンプ用鋼板。
- 質量%で、
C:0.18~0.26%、
Si:0.02%超0.05%以下、
Mn:1.0~1.5%、
P:0.03%以下、
S:0.02%以下、
Al:0.001~0.5%、
N:0.1%以下、
O:0.0010~0.020%、
Cr:0~2.0%、
Mo:0~1.0%
V:0~0.5%、
W:0~0.5%、
Ni:0~5.0%
B:0~0.01%
Ti:0~0.5%、
Nb:0~0.5%、
Cu:0~1.0%、
残部:Feおよび不純物である化学組成を有する鋼片に熱間圧延を施した後に690℃以上の温度域で巻き取って熱間圧延鋼板とする熱間圧延工程と、前記熱間圧延鋼板に10~90%の圧下率の冷間圧延を施して冷間圧延鋼板とする冷間圧延工程とを含むことを特徴とするホットスタンプ用鋼板の製造方法。 - 前記化学組成が、質量%で、
Cr:0.01~2.0%、
Mo:0.01~1.0%
V:0.01~0.5%、
W:0.01~0.5%、
Ni:0.01~5.0%および
B:0.0005~0.01%
からなる群から選択された1種または2種以上を含有することを特徴とする請求項7に記載のホットスタンプ用鋼板の製造方法。 - 前記化学組成が、質量%で、
Ti:0.001~0.5%、
Nb:0.001~0.5%および
Cu:0.01~1.0%
からなる群から選択された1種または2種以上を含有することを特徴とする請求項7または8に記載のホットスタンプ用鋼板の製造方法。 - 請求項7~9のいずれか1項に記載の製造方法により得られたホットスタンプ用鋼板を溶融アルミニウムめっき浴に浸漬して鋼板表面に溶融アルミニウムめっき層を形成することを特徴とするホットスタンプ用鋼板の製造方法。
- 請求項7~9のいずれか1項に記載の製造方法により得られたホットスタンプ用鋼板を溶融亜鉛めっき浴に浸漬して鋼板表面に溶融亜鉛めっき層を形成することを特徴とするホットスタンプ用鋼板の製造方法。
- 請求項7~9のいずれか1項に記載の製造方法により得られたホットスタンプ用鋼板を溶融亜鉛めっき浴に浸漬したのちに600℃以下の温度域に加熱して鋼板表面に合金化溶融亜鉛めっき層を形成することを特徴とするホットスタンプ用鋼板の製造方法。
- 質量%で、
C:0.18~0.26%、
Si:0.02%超0.05%以下、
Mn:1.0~1.5%、
P:0.03%以下、
S:0.02%以下、
Al:0.001~0.5%、
N:0.1%以下、
O:0.0010~0.020%、
Cr:0~2.0%、
Mo:0~1.0%
V:0~0.5%、
W:0~0.5%、
Ni:0~5.0%
B:0~0.01%
Ti:0~0.5%、
Nb:0~0.5%、
Cu:0~1.0%、
残部:Feおよび不純物である化学組成を有し、
Mnを含有する介在物の濃度が0.010質量%以上0.25質量%未満、かつ、最大長さが1.0~4.0μmである前記介在物に占めるMn酸化物の個数割合が10.0%以上であることを特徴とするホットスタンプ鋼材。 - 前記化学組成が、質量%で、
Cr:0.01~2.0%、
Mo:0.01~1.0%
V:0.01~0.5%、
W:0.01~0.5%、
Ni:0.01~5.0%および
B:0.0005~0.01%
からなる群から選択された1種または2種以上を含有することを特徴とする請求項13に記載のホットスタンプ鋼材。 - 前記化学組成が、質量%で、
Ti:0.001~0.5%、
Nb:0.001~0.5%および
Cu:0.01~1.0%
からなる群から選択された1種または2種以上を含有することを特徴とする請求項13または14に記載のホットスタンプ鋼材。
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013542282A JP5541421B2 (ja) | 2012-03-07 | 2013-03-05 | ホットスタンプ用鋼板及びその製造方法並びにホットスタンプ鋼材 |
| CN201380012499.1A CN104160050B (zh) | 2012-03-07 | 2013-03-05 | 热冲压用钢板及其制造方法和热冲压钢材 |
| IN8225DEN2014 IN2014DN08225A (ja) | 2012-03-07 | 2013-03-05 | |
| EP13757523.9A EP2824207A4 (en) | 2012-03-07 | 2013-03-05 | STEEL PLATE FOR HOT MOLDING, MANUFACTURING METHOD AND HOT-STAINLESS STEEL MATERIAL |
| CA2865910A CA2865910C (en) | 2012-03-07 | 2013-03-05 | Steel sheet for hot stamping, method for production thereof, and hot stamping steel material |
| RU2014140161/02A RU2587106C2 (ru) | 2012-03-07 | 2013-03-05 | Стальной лист для горячей штамповки, способ его производства и горячештампованный стальной материал |
| BR112014021801-3A BR112014021801B1 (pt) | 2012-03-07 | 2013-03-05 | chapa de aço para estampagem a quente, método para sua produção |
| US14/382,704 US10161023B2 (en) | 2012-03-07 | 2013-03-05 | Steel sheet for hot stamping, method for production thereof, and hot stamping steel material |
| MX2014010602A MX366958B (es) | 2012-03-07 | 2013-03-05 | Plancha de acero para estampado en caliente, metodo para producirla y material de acero para estampado en caliente. |
| KR1020147027737A KR101629594B1 (ko) | 2012-03-07 | 2013-03-05 | 핫 스탬프용 강판 및 그 제조 방법, 및 핫 스탬프 강재 |
| ZA2014/06644A ZA201406644B (en) | 2012-03-07 | 2014-09-10 | Steel sheet for hot stamping, method for production thereof, and hot stamping steel material |
| US16/110,069 US20180363109A1 (en) | 2012-03-07 | 2018-08-23 | Steel sheet for hot stamping, method for production thereof, and hot stamping steel material |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012050935 | 2012-03-07 | ||
| JP2012-050935 | 2012-03-07 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/382,704 A-371-Of-International US10161023B2 (en) | 2012-03-07 | 2013-03-05 | Steel sheet for hot stamping, method for production thereof, and hot stamping steel material |
| US16/110,069 Division US20180363109A1 (en) | 2012-03-07 | 2018-08-23 | Steel sheet for hot stamping, method for production thereof, and hot stamping steel material |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013133270A1 true WO2013133270A1 (ja) | 2013-09-12 |
Family
ID=49116745
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/055992 Ceased WO2013133270A1 (ja) | 2012-03-07 | 2013-03-05 | ホットスタンプ用鋼板及びその製造方法並びにホットスタンプ鋼材 |
Country Status (12)
| Country | Link |
|---|---|
| US (2) | US10161023B2 (ja) |
| EP (1) | EP2824207A4 (ja) |
| JP (1) | JP5541421B2 (ja) |
| KR (1) | KR101629594B1 (ja) |
| CN (1) | CN104160050B (ja) |
| BR (1) | BR112014021801B1 (ja) |
| CA (1) | CA2865910C (ja) |
| IN (1) | IN2014DN08225A (ja) |
| MX (1) | MX366958B (ja) |
| RU (1) | RU2587106C2 (ja) |
| WO (1) | WO2013133270A1 (ja) |
| ZA (1) | ZA201406644B (ja) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160024610A1 (en) * | 2013-03-14 | 2016-01-28 | Nippon Steel & Sumitomo Metal Corporation | High strength steel sheet excellent in delayed fracture resistance and low temperature toughness, and high strength member manufactured using the same |
| WO2018220873A1 (ja) * | 2017-06-01 | 2018-12-06 | 日新製鋼株式会社 | 高強度Zn-Al-Mg系表面被覆鋼板およびその製造方法 |
| CN111902558A (zh) * | 2018-03-27 | 2020-11-06 | 株式会社神户制钢所 | 热冲压用钢板 |
| CN114807739A (zh) * | 2021-01-28 | 2022-07-29 | 宝山钢铁股份有限公司 | 一种镀铝钢板、热成形部件及制造方法 |
| US11530474B2 (en) | 2018-02-15 | 2022-12-20 | Nippon Steel Corporation | Fe—Al-based plated hot-stamped member and manufacturing method of Fe—Al-based plated hot-stamped member |
| US12312652B2 (en) | 2020-09-01 | 2025-05-27 | Hyundai Steel Company | Material for hot stamping and method of manufacturing the same |
Families Citing this family (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160145731A1 (en) * | 2014-11-26 | 2016-05-26 | GM Global Technology Operations LLC | Controlling Liquid Metal Embrittlement In Galvanized Press-Hardened Components |
| CN104846274B (zh) * | 2015-02-16 | 2017-07-28 | 重庆哈工易成形钢铁科技有限公司 | 热冲压成形用钢板、热冲压成形工艺及热冲压成形构件 |
| EP3278895B1 (en) * | 2015-03-31 | 2020-03-11 | Nippon Steel Corporation | Steel sheet for hot stamping, method for manufacturing same, and hot stamp molded article |
| CN105420598A (zh) * | 2015-12-14 | 2016-03-23 | 浙江海洋学院 | 一种船锚用低碳合金钢及其热处理工艺 |
| CN105546228A (zh) * | 2016-01-20 | 2016-05-04 | 浙江海洋学院 | 一种船舶用无缝钢管及其制备方法 |
| US10619223B2 (en) | 2016-04-28 | 2020-04-14 | GM Global Technology Operations LLC | Zinc-coated hot formed steel component with tailored property |
| US10385415B2 (en) | 2016-04-28 | 2019-08-20 | GM Global Technology Operations LLC | Zinc-coated hot formed high strength steel part with through-thickness gradient microstructure |
| MX2019001760A (es) * | 2016-08-16 | 2019-06-17 | Nippon Steel & Sumitomo Metal Corp | Pieza conformada por prensado en caliente. |
| CN106086683B (zh) * | 2016-08-24 | 2018-05-01 | 武汉钢铁有限公司 | 用薄板坯直接轧制的抗拉强度≥1700MPa薄热成形钢及生产方法 |
| CN106086632B (zh) * | 2016-08-24 | 2018-01-12 | 武汉钢铁有限公司 | 用薄板坯直接轧制的抗拉强度≥1100MPa薄热成形钢及生产方法 |
| CN106119692B (zh) * | 2016-08-24 | 2018-03-20 | 武汉钢铁有限公司 | 用中薄板坯直接轧制的抗拉强度≥1500MPa热成形钢及生产方法 |
| CN106119695B (zh) * | 2016-08-24 | 2018-03-20 | 武汉钢铁有限公司 | 用中薄板坯直接轧制的抗拉强度≥1100MPa热成形钢及生产方法 |
| CN106086685B (zh) * | 2016-08-24 | 2018-01-12 | 武汉钢铁有限公司 | 用薄板坯直接轧制的抗拉强度≥1500MPa薄热成形钢及生产方法 |
| CN106222555B (zh) * | 2016-08-24 | 2018-03-20 | 武汉钢铁有限公司 | 用薄板坯直接轧制的抗拉强度≥1300MPa薄热成形钢及生产方法 |
| CN106222556B (zh) * | 2016-08-24 | 2018-03-16 | 武汉钢铁有限公司 | 用中薄板坯直接轧制的抗拉强度≥1300MPa热成形钢及生产方法 |
| JP6572864B2 (ja) * | 2016-10-18 | 2019-09-11 | Jfeスチール株式会社 | 電磁鋼板製造用の熱延鋼板およびその製造方法 |
| CN106636890B (zh) * | 2016-11-11 | 2018-09-14 | 武汉钢铁有限公司 | 直接热成形用薄规格热轧钢板及其制造方法 |
| WO2018096387A1 (en) * | 2016-11-24 | 2018-05-31 | Arcelormittal | Hot-rolled and coated steel sheet for hot-stamping, hot-stamped coated steel part and methods for manufacturing the same |
| WO2018220412A1 (fr) * | 2017-06-01 | 2018-12-06 | Arcelormittal | Procede de fabrication de pieces d'acier a haute resistance mecanique et ductilite amelioree, et pieces obtenues par ce procede |
| DE102017218704A1 (de) * | 2017-10-19 | 2019-04-25 | Thyssenkrupp Ag | Verfahren zur Herstellung eines mit einem metallischen, vor Korrosion schützenden Überzug versehenen Stahlbauteils |
| MX2020009562A (es) * | 2018-03-20 | 2020-10-05 | Nippon Steel Corp | Cuerpo estampado en caliente. |
| US11338550B2 (en) * | 2018-03-20 | 2022-05-24 | Nippon Steel Corporation | Hot-stamped body |
| WO2019188622A1 (ja) * | 2018-03-27 | 2019-10-03 | 株式会社神戸製鋼所 | ホットスタンプ用鋼板 |
| CN112513310A (zh) | 2018-05-24 | 2021-03-16 | 通用汽车环球科技运作有限责任公司 | 改善压制硬化钢的强度和延性的方法 |
| CN112534078A (zh) | 2018-06-19 | 2021-03-19 | 通用汽车环球科技运作有限责任公司 | 具有增强的机械性质的低密度压制硬化钢 |
| US11530469B2 (en) | 2019-07-02 | 2022-12-20 | GM Global Technology Operations LLC | Press hardened steel with surface layered homogenous oxide after hot forming |
| CN112326551B (zh) * | 2020-11-13 | 2023-07-18 | 江苏省沙钢钢铁研究院有限公司 | 一种复合钢板性能的测试方法 |
| KR102904753B1 (ko) * | 2022-12-30 | 2025-12-24 | 현대제철 주식회사 | 핫 스탬핑 부품 |
| CN119824353A (zh) * | 2025-01-03 | 2025-04-15 | 鞍钢股份有限公司 | 铝合金预镀层钢带的制造方法 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000017371A (ja) * | 1998-07-02 | 2000-01-18 | Nippon Yakin Kogyo Co Ltd | 打抜き加工性に優れるFe−Ni系合金の製造方法 |
| JP2002080933A (ja) * | 2000-06-30 | 2002-03-22 | Nippon Steel Corp | 形状凍結性に優れた鋼板およびその製造方法 |
| JP2003166035A (ja) * | 2001-11-28 | 2003-06-13 | Nippon Steel Corp | 成形加工後の耐遅れ破壊性に優れた高強度薄鋼板及びその製造方法並びに高強度薄鋼板により作成された自動車用強度部品 |
| JP2006009116A (ja) | 2004-06-29 | 2006-01-12 | Nippon Steel Corp | 熱間プレス用鋼板 |
| JP2006029977A (ja) | 2004-07-15 | 2006-02-02 | Jfe Steel Kk | 自動車構造部材用鋼材の耐遅れ破壊特性評価方法および耐遅れ破壊特性に優れた自動車構造部材用鋼材 |
| WO2008066194A1 (en) * | 2006-11-28 | 2008-06-05 | Nippon Steel Corporation | Free-cutting steel excellent in manufacturability |
| JP2008144239A (ja) * | 2006-12-12 | 2008-06-26 | Nippon Steel Corp | 合金化溶融亜鉛めっき用鋼板及び合金化溶融亜鉛めっき鋼板 |
| JP2010174291A (ja) | 2009-01-28 | 2010-08-12 | Jfe Steel Corp | 熱間打抜き性に優れたダイクエンチ用鋼板 |
| JP2011184758A (ja) * | 2010-03-09 | 2011-09-22 | Jfe Steel Corp | 高強度プレス部材およびその製造方法 |
Family Cites Families (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3292671B2 (ja) * | 1997-02-10 | 2002-06-17 | 川崎製鉄株式会社 | 深絞り性と耐時効性の良好な冷延鋼板用の熱延鋼帯 |
| CA2231760A1 (en) * | 1998-03-11 | 1999-09-11 | Nisshin Steel Co., Ltd. | Cold-rolled steel strip and hot-dip coated cold-rolled steel strip for use as building material and manufacturing method thereof |
| CA2422753C (en) | 2000-09-21 | 2007-11-27 | Nippon Steel Corporation | Steel plate excellent in shape freezing property and method for production thereof |
| RU2197542C1 (ru) * | 2001-06-28 | 2003-01-27 | Открытое акционерное общество "Северсталь" | Способ производства листовой стали |
| CN1277634C (zh) | 2001-09-14 | 2006-10-04 | 纽科尔公司 | 铸造钢带的方法及钢带 |
| JP2004068128A (ja) * | 2002-08-09 | 2004-03-04 | Daido Steel Co Ltd | 切屑破砕性にすぐれた機械構造用鋼 |
| EP1630244B2 (en) * | 2003-04-23 | 2016-08-17 | Nippon Steel & Sumitomo Metal Corporation | Hot press formed product and method for production thereof |
| JP4443910B2 (ja) * | 2003-12-12 | 2010-03-31 | Jfeスチール株式会社 | 自動車構造部材用鋼材およびその製造方法 |
| JP4500124B2 (ja) * | 2004-07-23 | 2010-07-14 | 新日本製鐵株式会社 | ホットプレス用めっき鋼板の製造方法 |
| CA2701559C (en) * | 2004-09-15 | 2013-08-06 | Nippon Steel Corporation | High strength part and method of production of the same |
| CN100507053C (zh) | 2004-11-29 | 2009-07-01 | 宝山钢铁股份有限公司 | 一种800MPa冷轧热镀锌双相钢及其制造方法 |
| RU2277594C1 (ru) * | 2005-03-30 | 2006-06-10 | Открытое акционерное общество "Северсталь" | Способ производства холоднокатаных листов для глубокой вытяжки |
| SK288275B6 (sk) * | 2005-12-01 | 2015-06-02 | Posco | Oceľová doska na tvarovanie lisovaním za horúca s tepelnou úpravou a rázovými vlastnosťami, za horúca lisovaný diel z nej vyrobený a spôsob ich výroby |
| KR100878614B1 (ko) * | 2005-12-01 | 2009-01-15 | 주식회사 포스코 | 열처리 경화형 초고강도 강판, 이를 이용한 열처리 경화형부재와 그 제조방법 |
| EP1832667A1 (fr) * | 2006-03-07 | 2007-09-12 | ARCELOR France | Procédé de fabrication de tôles d'acier à très hautes caractéristiques de résistance, de ductilité et de tenacité, et tôles ainsi produites |
| JP5079795B2 (ja) * | 2007-04-11 | 2012-11-21 | 新日本製鐵株式会社 | 低温靭性に優れたプレス加工用溶融めっき高強度鋼板およびその製造方法 |
| CA2729942C (en) * | 2008-07-11 | 2013-08-06 | Nippon Steel Corporation | Aluminum plated steel sheet for rapid heating hot-stamping, production method of the same and rapid heating hot-stamping method by using this steel sheet |
| JP5126844B2 (ja) * | 2008-08-19 | 2013-01-23 | 新日鐵住金株式会社 | 熱間プレス用鋼板およびその製造方法ならびに熱間プレス鋼板部材の製造方法 |
| KR101141015B1 (ko) | 2009-06-26 | 2012-05-02 | 현대제철 주식회사 | 수소지연파괴 저항성이 우수한 강판 및 그 제조방법 |
| JP5457840B2 (ja) * | 2010-01-07 | 2014-04-02 | 株式会社神戸製鋼所 | 伸びおよび伸びフランジ性に優れた高強度冷延鋼板 |
| JP5064525B2 (ja) * | 2010-02-18 | 2012-10-31 | 新日本製鐵株式会社 | 異方性が小さく焼入性に優れた高炭素鋼板及びその製造方法 |
| JP5466576B2 (ja) * | 2010-05-24 | 2014-04-09 | 株式会社神戸製鋼所 | 曲げ加工性に優れた高強度冷延鋼板 |
| MX360249B (es) * | 2011-03-09 | 2018-10-26 | Nippon Steel & Sumitomo Metal Corp | Laminas de acero para estampado en caliente, metodo para fabricar las mismas y metodo para fabricar partes con alta resistencia. |
-
2013
- 2013-03-05 IN IN8225DEN2014 patent/IN2014DN08225A/en unknown
- 2013-03-05 US US14/382,704 patent/US10161023B2/en active Active
- 2013-03-05 KR KR1020147027737A patent/KR101629594B1/ko active Active
- 2013-03-05 BR BR112014021801-3A patent/BR112014021801B1/pt not_active IP Right Cessation
- 2013-03-05 MX MX2014010602A patent/MX366958B/es active IP Right Grant
- 2013-03-05 WO PCT/JP2013/055992 patent/WO2013133270A1/ja not_active Ceased
- 2013-03-05 RU RU2014140161/02A patent/RU2587106C2/ru not_active IP Right Cessation
- 2013-03-05 JP JP2013542282A patent/JP5541421B2/ja active Active
- 2013-03-05 CN CN201380012499.1A patent/CN104160050B/zh active Active
- 2013-03-05 CA CA2865910A patent/CA2865910C/en not_active Expired - Fee Related
- 2013-03-05 EP EP13757523.9A patent/EP2824207A4/en not_active Withdrawn
-
2014
- 2014-09-10 ZA ZA2014/06644A patent/ZA201406644B/en unknown
-
2018
- 2018-08-23 US US16/110,069 patent/US20180363109A1/en not_active Abandoned
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000017371A (ja) * | 1998-07-02 | 2000-01-18 | Nippon Yakin Kogyo Co Ltd | 打抜き加工性に優れるFe−Ni系合金の製造方法 |
| JP2002080933A (ja) * | 2000-06-30 | 2002-03-22 | Nippon Steel Corp | 形状凍結性に優れた鋼板およびその製造方法 |
| JP2003166035A (ja) * | 2001-11-28 | 2003-06-13 | Nippon Steel Corp | 成形加工後の耐遅れ破壊性に優れた高強度薄鋼板及びその製造方法並びに高強度薄鋼板により作成された自動車用強度部品 |
| JP2006009116A (ja) | 2004-06-29 | 2006-01-12 | Nippon Steel Corp | 熱間プレス用鋼板 |
| JP2006029977A (ja) | 2004-07-15 | 2006-02-02 | Jfe Steel Kk | 自動車構造部材用鋼材の耐遅れ破壊特性評価方法および耐遅れ破壊特性に優れた自動車構造部材用鋼材 |
| WO2008066194A1 (en) * | 2006-11-28 | 2008-06-05 | Nippon Steel Corporation | Free-cutting steel excellent in manufacturability |
| JP2008144239A (ja) * | 2006-12-12 | 2008-06-26 | Nippon Steel Corp | 合金化溶融亜鉛めっき用鋼板及び合金化溶融亜鉛めっき鋼板 |
| JP2010174291A (ja) | 2009-01-28 | 2010-08-12 | Jfe Steel Corp | 熱間打抜き性に優れたダイクエンチ用鋼板 |
| JP2011184758A (ja) * | 2010-03-09 | 2011-09-22 | Jfe Steel Corp | 高強度プレス部材およびその製造方法 |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160024610A1 (en) * | 2013-03-14 | 2016-01-28 | Nippon Steel & Sumitomo Metal Corporation | High strength steel sheet excellent in delayed fracture resistance and low temperature toughness, and high strength member manufactured using the same |
| US9353424B2 (en) * | 2013-03-14 | 2016-05-31 | Nippon Steel & Sumitomo Metal Corporation | High strength steel sheet excellent in delayed fracture resistance and low temperature toughness, and high strength member manufactured using the same |
| WO2018220873A1 (ja) * | 2017-06-01 | 2018-12-06 | 日新製鋼株式会社 | 高強度Zn-Al-Mg系表面被覆鋼板およびその製造方法 |
| JP2018204065A (ja) * | 2017-06-01 | 2018-12-27 | 日新製鋼株式会社 | 高強度Zn−Al−Mg系表面被覆鋼板およびその製造方法 |
| CN110678571A (zh) * | 2017-06-01 | 2020-01-10 | 日铁日新制钢株式会社 | 高强度Zn-Al-Mg系表面被覆钢板及其制造方法 |
| CN110678571B (zh) * | 2017-06-01 | 2022-02-18 | 日铁日新制钢株式会社 | 高强度Zn-Al-Mg系表面被覆钢板及其制造方法 |
| US12227851B2 (en) | 2017-06-01 | 2025-02-18 | Nippon Steel Nisshin Co., Ltd. | High-strength Zn—Al—Mg-based surface-coated steel sheet and method for producing same |
| US11530474B2 (en) | 2018-02-15 | 2022-12-20 | Nippon Steel Corporation | Fe—Al-based plated hot-stamped member and manufacturing method of Fe—Al-based plated hot-stamped member |
| CN111902558A (zh) * | 2018-03-27 | 2020-11-06 | 株式会社神户制钢所 | 热冲压用钢板 |
| US12312652B2 (en) | 2020-09-01 | 2025-05-27 | Hyundai Steel Company | Material for hot stamping and method of manufacturing the same |
| CN114807739A (zh) * | 2021-01-28 | 2022-07-29 | 宝山钢铁股份有限公司 | 一种镀铝钢板、热成形部件及制造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| MX2014010602A (es) | 2014-09-18 |
| US20150024237A1 (en) | 2015-01-22 |
| US20180363109A1 (en) | 2018-12-20 |
| CA2865910C (en) | 2017-10-17 |
| KR20140138829A (ko) | 2014-12-04 |
| RU2587106C2 (ru) | 2016-06-10 |
| EP2824207A1 (en) | 2015-01-14 |
| JP5541421B2 (ja) | 2014-07-09 |
| KR101629594B1 (ko) | 2016-06-13 |
| JPWO2013133270A1 (ja) | 2015-07-30 |
| CA2865910A1 (en) | 2013-09-12 |
| US10161023B2 (en) | 2018-12-25 |
| IN2014DN08225A (ja) | 2015-05-15 |
| EP2824207A4 (en) | 2016-03-09 |
| BR112014021801B1 (pt) | 2019-10-29 |
| CN104160050A (zh) | 2014-11-19 |
| RU2014140161A (ru) | 2016-04-27 |
| MX366958B (es) | 2019-08-01 |
| CN104160050B (zh) | 2016-05-18 |
| ZA201406644B (en) | 2016-09-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5541421B2 (ja) | ホットスタンプ用鋼板及びその製造方法並びにホットスタンプ鋼材 | |
| RU2566131C1 (ru) | Гальванизированный горячим способом стальной лист и способ его изготовления | |
| KR101721352B1 (ko) | 내 지연 파괴 특성과 저온 인성이 우수한 고강도 강판 및 그것을 사용하여 제조한 고강도 부재 | |
| EP3309273B1 (en) | Galvannealed steel sheet and method for manufacturing same | |
| JP5376090B2 (ja) | 亜鉛めっき鋼板及びその製造方法 | |
| CN102639739B (zh) | 耐氢脆化特性优异的最大拉伸强度为900MPa以上的高强度钢板及其制造方法 | |
| JP2011111671A (ja) | 延性及び耐遅れ破壊特性の良好な引張最大強度900MPa以上を有する高強度鋼板および高強度冷延鋼板の製造方法、高強度亜鉛めっき鋼板の製造方法 | |
| EP3006586B1 (en) | Heat-treated steel material and method for producing same | |
| CN107148486A (zh) | 高强度钢板、高强度热镀锌钢板、高强度热镀铝钢板和高强度电镀锌钢板、以及它们的制造方法 | |
| CN110475892B (zh) | 高强度冷轧钢板及其制造方法 | |
| JP4317491B2 (ja) | 熱間プレス用鋼板 | |
| KR20200087202A (ko) | 열간 프레스 강판 부재 및 그 제조 방법 | |
| KR20220033519A (ko) | 고강도 박강판 및 그의 제조 방법 | |
| WO2017009936A1 (ja) | 鋼板、溶融亜鉛めっき鋼板、及び合金化溶融亜鉛めっき鋼板、並びにそれらの製造方法 | |
| CN108884534A (zh) | 薄钢板和镀覆钢板、以及热轧钢板的制造方法、冷轧全硬钢板的制造方法、薄钢板的制造方法和镀覆钢板的制造方法 | |
| KR20240148911A (ko) | 아연 도금 강판, 부재 및 그들의 제조 방법 | |
| KR102561381B1 (ko) | 고강도 합금화 전기 아연 도금 강판 및 그 제조 방법 | |
| EP4600397A1 (en) | Steel plate, resistance spot welding method, resistance spot welding member, and method for manufacturing steel plate | |
| KR20250150125A (ko) | 강판, 부재 및 그들의 제조 방법 | |
| KR20250150126A (ko) | 강판, 부재 및 그들의 제조 방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2013542282 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13757523 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2865910 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14382704 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2014/010602 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 20147027737 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: IDP00201405964 Country of ref document: ID |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013757523 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2014140161 Country of ref document: RU Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112014021801 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112014021801 Country of ref document: BR Kind code of ref document: A2 Effective date: 20140903 |