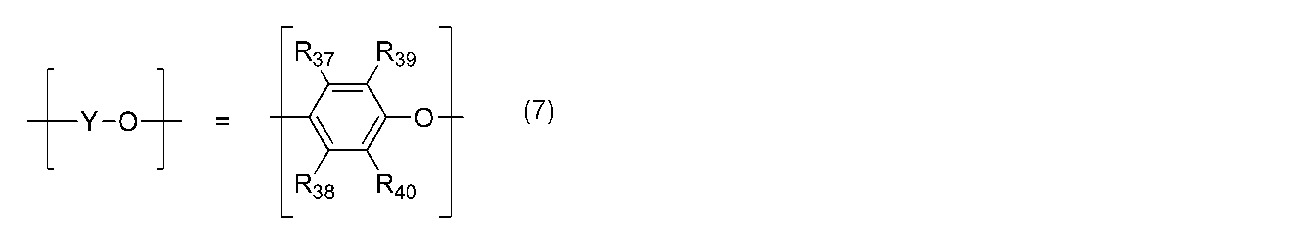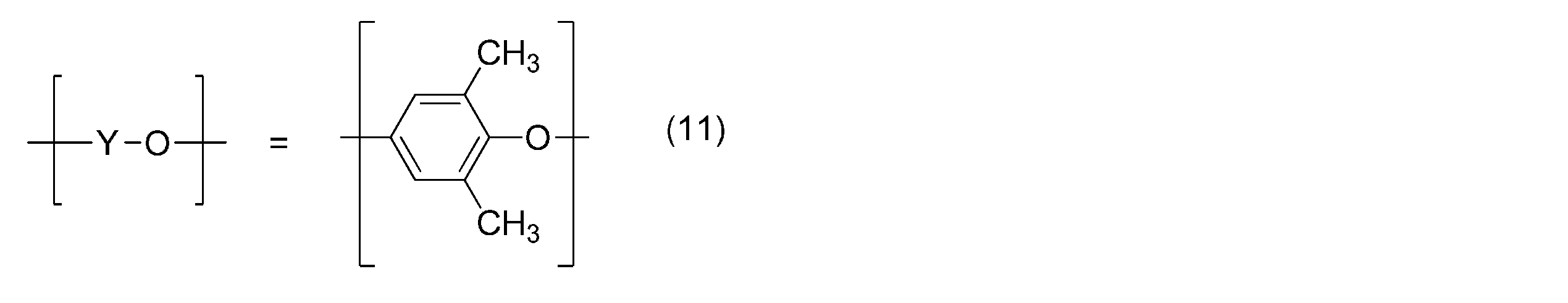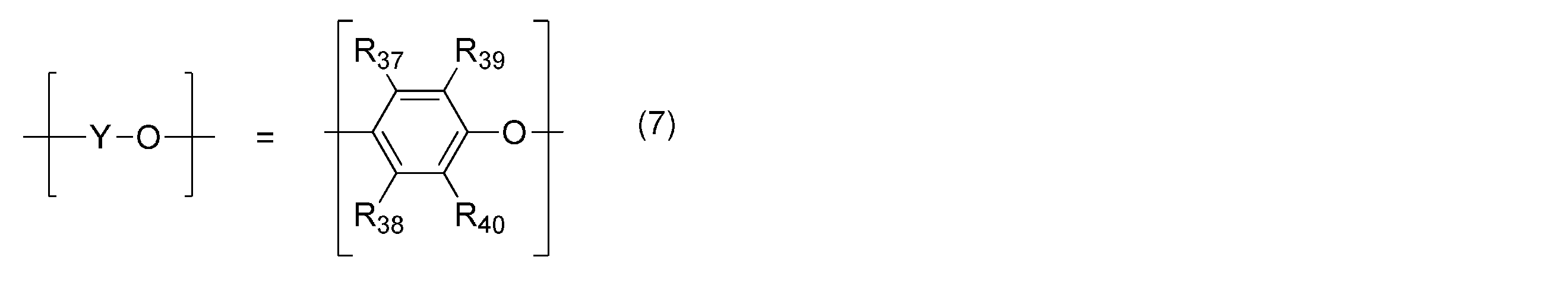WO2013141298A1 - プリプレグ及び積層板 - Google Patents
プリプレグ及び積層板 Download PDFInfo
- Publication number
- WO2013141298A1 WO2013141298A1 PCT/JP2013/058075 JP2013058075W WO2013141298A1 WO 2013141298 A1 WO2013141298 A1 WO 2013141298A1 JP 2013058075 W JP2013058075 W JP 2013058075W WO 2013141298 A1 WO2013141298 A1 WO 2013141298A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- prepreg
- mass
- resin composition
- group
- prepreg according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 Cc(c(*)c1*)c(*)c(*)c1OC Chemical compound Cc(c(*)c1*)c(*)c(*)c1OC 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K1/00—Printed circuits
- H05K1/02—Details
- H05K1/03—Use of materials for the substrate
- H05K1/0313—Organic insulating material
- H05K1/0353—Organic insulating material consisting of two or more materials, e.g. two or more polymers, polymer + filler, + reinforcement
- H05K1/0373—Organic insulating material consisting of two or more materials, e.g. two or more polymers, polymer + filler, + reinforcement containing additives, e.g. fillers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/14—Layered products comprising a layer of metal next to a fibrous or filamentary layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/18—Layered products comprising a layer of synthetic resin characterised by the use of special additives
- B32B27/20—Layered products comprising a layer of synthetic resin characterised by the use of special additives using fillers, pigments, thixotroping agents
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/28—Layered products comprising a layer of synthetic resin comprising synthetic resins not wholly covered by any one of the sub-groups B32B27/30 - B32B27/42
- B32B27/285—Layered products comprising a layer of synthetic resin comprising synthetic resins not wholly covered by any one of the sub-groups B32B27/30 - B32B27/42 comprising polyethers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
- B32B27/365—Layered products comprising a layer of synthetic resin comprising polyesters comprising polycarbonates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/38—Layered products comprising a layer of synthetic resin comprising epoxy resins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B5/00—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts
- B32B5/02—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by structural features of a fibrous or filamentary layer
- B32B5/024—Woven fabric
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/4007—Curing agents not provided for by the groups C08G59/42 - C08G59/66
- C08G59/4014—Nitrogen containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/34—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from hydroxy compounds or their metallic derivatives
- C08G65/38—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from hydroxy compounds or their metallic derivatives derived from phenols
- C08G65/40—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from hydroxy compounds or their metallic derivatives derived from phenols from phenols (I) and other compounds (II), e.g. OH-Ar-OH + X-Ar-X, where X is halogen atom, i.e. leaving group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/34—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from hydroxy compounds or their metallic derivatives
- C08G65/48—Polymers modified by chemical after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/241—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using inorganic fibres
- C08J5/244—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using inorganic fibres using glass fibres
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/249—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs characterised by the additives used in the prepolymer mixture
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/01—Use of inorganic substances as compounding ingredients characterized by their specific function
- C08K3/013—Fillers, pigments or reinforcing additives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/36—Silica
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
- C08L63/04—Epoxynovolacs
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L69/00—Compositions of polycarbonates; Compositions of derivatives of polycarbonates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L71/00—Compositions of polyethers obtained by reactions forming an ether link in the main chain; Compositions of derivatives of such polymers
- C08L71/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08L71/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
- C08L71/12—Polyphenylene oxides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L71/00—Compositions of polyethers obtained by reactions forming an ether link in the main chain; Compositions of derivatives of such polymers
- C08L71/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08L71/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
- C08L71/12—Polyphenylene oxides
- C08L71/123—Polyphenylene oxides not modified by chemical after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L71/00—Compositions of polyethers obtained by reactions forming an ether link in the main chain; Compositions of derivatives of such polymers
- C08L71/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08L71/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
- C08L71/12—Polyphenylene oxides
- C08L71/126—Polyphenylene oxides modified by chemical after-treatment
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B3/00—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties
- H01B3/18—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances
- H01B3/30—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes
- H01B3/42—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes polyesters; polyethers; polyacetals
- H01B3/427—Polyethers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2260/00—Layered product comprising an impregnated, embedded, or bonded layer wherein the layer comprises an impregnation, embedding, or binder material
- B32B2260/02—Composition of the impregnated, bonded or embedded layer
- B32B2260/021—Fibrous or filamentary layer
- B32B2260/023—Two or more layers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2260/00—Layered product comprising an impregnated, embedded, or bonded layer wherein the layer comprises an impregnation, embedding, or binder material
- B32B2260/04—Impregnation, embedding, or binder material
- B32B2260/046—Synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/02—Synthetic macromolecular fibres
- B32B2262/0276—Polyester fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2264/00—Composition or properties of particles which form a particulate layer or are present as additives
- B32B2264/10—Inorganic particles
- B32B2264/102—Oxide or hydroxide
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/20—Properties of the layers or laminate having particular electrical or magnetic properties, e.g. piezoelectric
- B32B2307/202—Conductive
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/20—Properties of the layers or laminate having particular electrical or magnetic properties, e.g. piezoelectric
- B32B2307/206—Insulating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/702—Amorphous
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2457/00—Electrical equipment
- B32B2457/08—PCBs, i.e. printed circuit boards
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2371/00—Characterised by the use of polyethers obtained by reactions forming an ether link in the main chain; Derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2371/00—Characterised by the use of polyethers obtained by reactions forming an ether link in the main chain; Derivatives of such polymers
- C08J2371/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08J2371/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
- C08J2371/12—Polyphenylene oxides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2463/00—Characterised by the use of epoxy resins; Derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2203/00—Applications
- C08L2203/20—Applications use in electrical or conductive gadgets
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K2201/00—Indexing scheme relating to printed circuits covered by H05K1/00
- H05K2201/01—Dielectrics
- H05K2201/0137—Materials
- H05K2201/0158—Polyalkene or polyolefin, e.g. polyethylene [PE], polypropylene [PP]
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K2201/00—Indexing scheme relating to printed circuits covered by H05K1/00
- H05K2201/02—Fillers; Particles; Fibers; Reinforcement materials
- H05K2201/0203—Fillers and particles
- H05K2201/0206—Materials
- H05K2201/0209—Inorganic, non-metallic particles
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31678—Of metal
Definitions
- the present invention relates to a prepreg and a laminate used for a printed wiring board for forming an electric circuit.
- a resin composition having a low dielectric constant and a low dielectric loss tangent such as a fluorine resin, a cyanate ester resin, a polyphenylene ether resin, or a vinyl compound mainly containing styrene is used.
- a resin composition having a low dielectric constant and a low dielectric loss tangent such as a fluorine resin, a cyanate ester resin, a polyphenylene ether resin, or a vinyl compound mainly containing styrene is used.
- a laminate for a printed wiring board uses a prepreg obtained by uniformly dispersing components in a resin composition in an organic solvent, impregnating or applying the obtained varnish to a substrate such as glass cloth, and drying. Produced.
- a low-polarity resin such as polyphenylene ether is often used as the main component of the resin having low dielectric loss tangent, and the above-mentioned organic solvent is also low-polarity toluene according to the polarity of the main component (for example, Patent Document 3). reference).
- toluene tends to be used from the viewpoint of environmental pollution, and it is required to use another solvent.
- it is not generally used to use a polar solvent as a resin component for such a low dielectric loss tangent, and the effect of residual solvent in the prepreg when using a polar solvent on the prepared prepreg, etc. Not enough consideration has been made.
- This invention makes it a subject to provide the prepreg which implement
- the present inventors have determined that the dielectric loss tangent at 10 GHz of the laminate produced using the prepreg is set to 0. by setting the residual amount of the polar solvent in the prepreg to 3% by mass or less. As a result, the present invention has been reached.
- the present invention [1] A prepreg produced by impregnating or coating a base material with a resin composition containing polyphenylene ether as a main component, an inorganic filler and a polar solvent, and then performing a drying step,
- the content of the polar solvent in the prepreg is 3% by mass or less
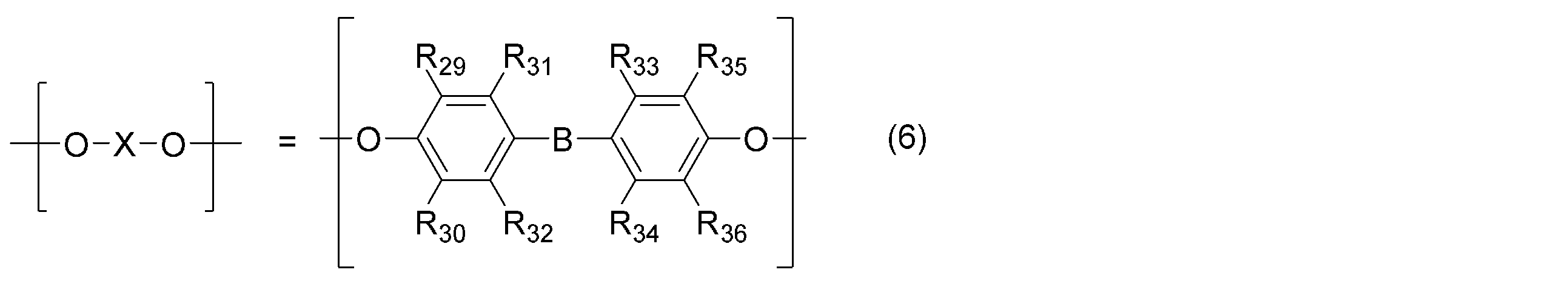
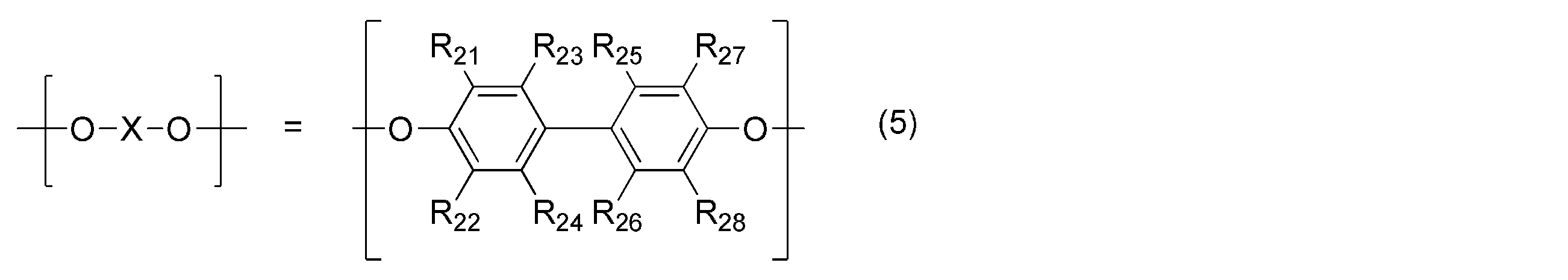
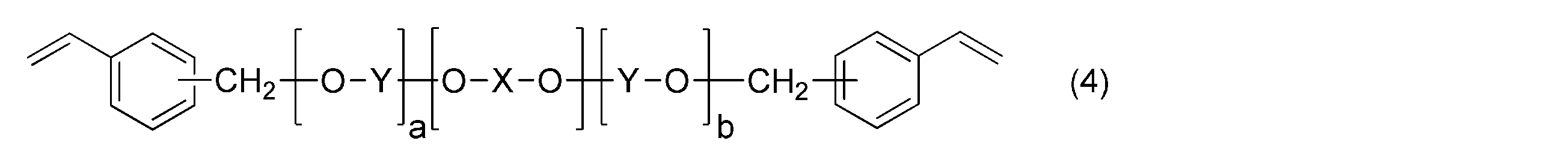
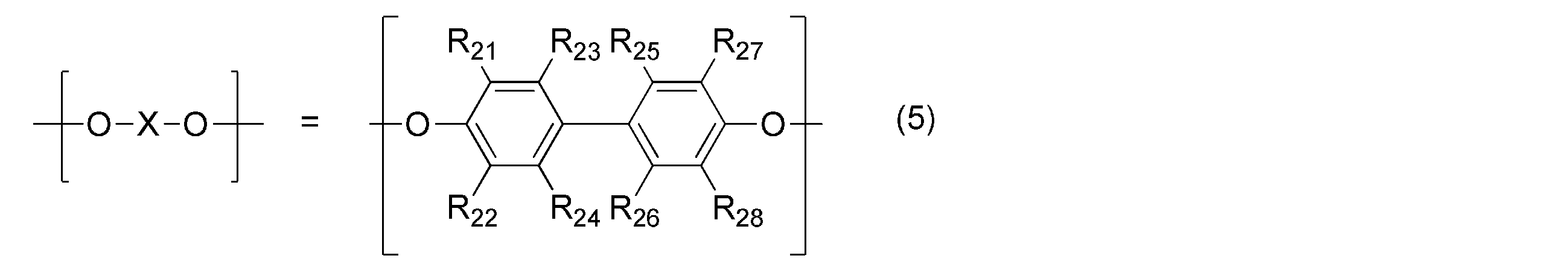
- the polyphenylene ether is represented by the general formula (4): (Wherein — (O—X—O) — represents the general formula (5): (R 21 , R 22 , R 23 , R 27 , R 28 may be the same or different and are an alkyl group or phenyl group having 6 or less carbon atoms. R 24 , R 25 , R 26 are the same or different.
- R 29 , R 30 , R 31 , R 32 , R 33 , R 34 , R 35 , R 36 may be the same or different and are a hydrogen atom, an alkyl group having 6 or less carbon atoms, or a phenyl group.
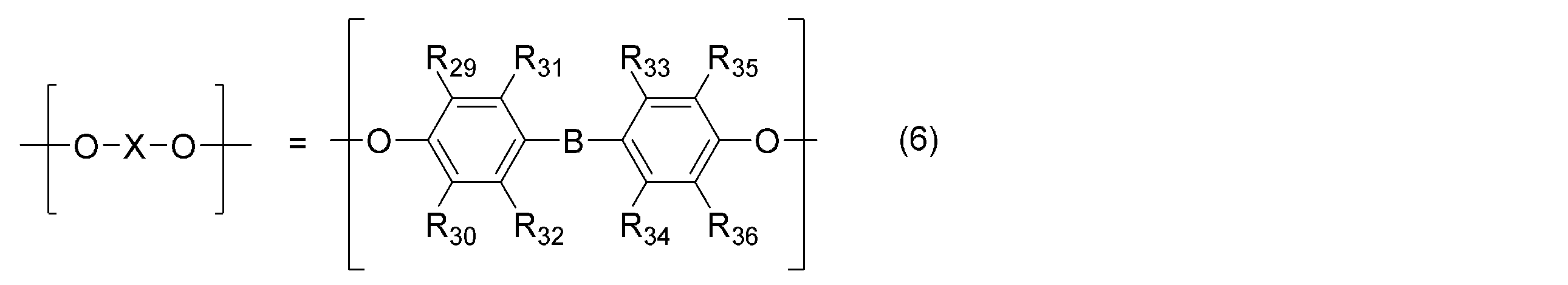
- B- is a linear, branched or cyclic divalent hydrocarbon group having 20 or less carbon atoms. It consists of the structure represented by.
- R 39 and R 40 may be the same or different, and are an alkyl group or phenyl group having 6 or less carbon atoms.
- R 37 and R 38 may be the same or different, and are a hydrogen atom or 6 or less carbon atoms. It is an alkyl group or a phenyl group.
- One type of structure or two or more types of structures are randomly arranged. a and b each represents an integer of 0 to 100, at least one of which is not 0.
- a printed wiring board including an insulating layer and a conductor layer formed on a surface of the insulating layer, wherein the insulating layer includes the prepreg according to any one of [1] to [13]. Including printed wiring board. Is provided.
- the prepreg of the present invention achieves a low dielectric loss tangent when a laminated board is produced, is suitable for a printed wiring board material corresponding to high density, and has extremely high industrial practicality.
- the prepreg of the present invention can be stably produced during the production of the prepreg or after the production, the phenomenon that the resin powder falls off from the prepreg and the powder fall off are suppressed.
- the present invention is a prepreg produced by impregnating or applying a varnish containing a resin composition containing polyphenylene ether as a main component, an inorganic filler and a polar solvent to a substrate, and then performing a drying step,
- the present invention relates to a prepreg in which the content of the polar solvent in the prepreg is 3% by mass or less, and a dielectric loss tangent at 10 GHz of a laminate produced using the prepreg is 0.001 to 0.007.
- polyphenylene ether As polyphenylene ether, general formula (1): (In the formula, R 1 , R 2 , R 3 , and R 4 may be the same or different and each represents an alkyl group, aryl group, halogen, or hydrogen having 6 or less carbon atoms.) It is preferable that the polymer comprises at least a repeating unit represented by:
- the polymer has the general formula (2): (R 5 , R 6 , R 7 , R 11 , R 12 may be the same or different, and are an alkyl group or phenyl group having 6 or less carbon atoms. R 8 , R 9 , R 10 are the same or different.
- R 13 , R 14 , R 15 , R 16 , R 17 , R 18 , R 19 , R 20 may be the same or different and are a hydrogen atom, an alkyl group having 6 or less carbon atoms, or a phenyl group.
- A- is a linear, branched or cyclic divalent hydrocarbon group having 20 or less carbon atoms.
- It may further contain a repeating unit represented by:
- a modified polyphenylene ether partially or wholly functionalized with an ethylenically unsaturated group such as a vinylbenzyl group, an epoxy group, an amino group, a hydroxyl group, a mercapto group, a carboxyl group, and a silyl group is used. You can also. These may be used alone or in combination of two or more.
- the method for producing the modified polyphenylene ether is not particularly limited as long as the effects of the present invention can be obtained.
- a product functionalized with a vinylbenzyl group is obtained by dissolving a bifunctional phenylene ether oligomer and vinylbenzyl chloride in a solvent, reacting by adding a base with heating and stirring, and then solidifying the resin.
- Those functionalized with a carboxyl group are produced, for example, by melting and kneading an unsaturated carboxylic acid or a functionalized derivative thereof with polyphenylene ether in the presence or absence of a radical initiator.
- it is produced by dissolving polyphenylene ether and an unsaturated carboxylic acid or a functional derivative thereof in an organic solvent in the presence or absence of a radical initiator and reacting in a solution.
- the polyphenylene ether preferably contains a modified polyphenylene ether having an ethylenically unsaturated group at both ends.
- the ethylenically unsaturated group include alkenyl groups such as ethenyl group, allyl group, acrylic group, methacryl group, propenyl group, butenyl group, hexenyl group, and octenyl group, cycloalkenyl groups such as cyclopentenyl group and cyclohexenyl group, Examples thereof include alkenylaryl groups such as vinylbenzyl group and vinylnaphthyl group, and vinylbenzyl group is preferred.
- the two ethylenically unsaturated groups at both ends may be the same functional group or different functional groups.
- the polyphenylene ether in the present invention has the general formula (4): (Wherein — (O—X—O) — represents the general formula (5): (R 21 , R 22 , R 23 , R 27 , R 28 may be the same or different and are an alkyl group or phenyl group having 6 or less carbon atoms. R 24 , R 25 , R 26 are the same or different.
- R 29 , R 30 , R 31 , R 32 , R 33 , R 34 , R 35 , R 36 may be the same or different and are a hydrogen atom, an alkyl group having 6 or less carbon atoms, or a phenyl group.
- B- is a linear, branched or cyclic divalent hydrocarbon group having 20 or less carbon atoms. It consists of the structure represented by.
- R 39 and R 40 may be the same or different, and are an alkyl group or phenyl group having 6 or less carbon atoms.
- R 37 and R 38 may be the same or different, and are a hydrogen atom or 6 or less carbon atoms. It is an alkyl group or a phenyl group.
- One type of structure or two or more types of structures are randomly arranged.
- a and b each represents an integer of 0 to 100, at least one of which is not 0.
- Examples of —B— in the general formula (6) include methylene, ethylidene, 1-methylethylidene, 1,1-propylidene, 1,4-phenylenebis (1-methylethylidene), 1,3-phenylenebis (1 -Methylethylidene), cyclohexylidene, phenylmethylene, naphthylmethylene, 1-phenylethylidene, and the like, but are not limited thereto.
- R 21 , R 22 , R 23 , R 27 , R 28 , R 39 , R 40 are alkyl groups having 3 or less carbon atoms, and R 24 , R 25 , R 26 , R 29 , R Polyphenylene ether in which 30 , R 31 , R 32 , R 33 , R 34 , R 35 , R 36 , R 37 , R 38 is a hydrogen atom or an alkyl group having 3 or less carbon atoms is preferred, and in particular, the general formula (5) or — (O—X—O) — represented by the general formula (6) is the formula (8), the general formula (9), or the general formula (10), and represented by the general formula (7) — A polyphenylene ether having a structure in which (YO) — is the formula (11) or the formula (12) or the formula (11) and the formula (12) are randomly arranged is more preferable.
- R 31 , R 32 , R 33 , and R 34 may be the same or different, and are a hydrogen atom or a methyl group.
- —B— is linear, branched or cyclic having 20 or less carbon atoms. Divalent hydrocarbon group
- the method for producing the modified polyphenylene ether having the structure represented by the formula (4) is not particularly limited.
- a bifunctional phenylene ether obtained by oxidative coupling of a bifunctional phenol compound and a monofunctional phenol compound. It can be produced by converting the terminal phenolic hydroxyl group of the oligomer to vinyl benzyl ether.
- Such modified polyphenylene ether can be obtained from, for example, Mitsubishi Gas Chemical Co., Ltd. (OPE-2St 1200, etc.).
- the number average molecular weight of the polyphenylene ether is preferably in the range of 500 to 3000, more preferably in the range of 1000 to 2500 in terms of polystyrene by the GPC method. If the number average molecular weight is 500 or more, it is difficult to be sticky when it is formed into a coating, and if it is 3000 or less, it is possible to prevent a decrease in solubility in a solvent, and during prepreg production or after production, a resin from the prepreg. The phenomenon of powder falling off and powder falling off are suppressed.
- the resin composition of the present invention is preferably composed mainly of polyphenylene ether in order to reduce the dielectric loss tangent, and the content of polyphenylene ether is 50% by mass or more based on the amount of the resin composition. Is particularly preferred.
- the quantity of a resin composition shall mean the total mass of each component except the inorganic filler and solvent in a varnish.
- the resin composition of the present invention includes epoxy resin, cyanate ester compound, polycarbonate, maleimide compound, styrene and / or substituted styrene oligomer, phosphazene compound, phenol resin, and silicone resin. Powders or the like, or a combination of two or more of these may be included.
- the epoxy resin is not particularly limited as long as it has two or more epoxy groups in one molecule.
- a compound obtained by epoxidizing a double bond, a compound obtained by reaction of a hydroxyl group-containing silicone resin and epichlorohydrin, and the like can be mentioned. These may be halogenated.
- bisphenol A type epoxy resin, phenol novolak type epoxy resin, cresol novolak type epoxy resin, and novolak type epoxy resin modified with naphthalene skeleton are preferable.
- the content of the epoxy resin in the resin composition is preferably in the range of 1 to 30% by mass and more preferably in the range of 5 to 15% by mass with respect to the amount of the resin composition from the viewpoint of electrical characteristics.
- the cyanate ester compound is not particularly limited as long as it is a compound having two or more cyanato groups in the molecule.
- bisphenol A type cyanate ester resin and its prepolymer naphthol aralkyl type cyanate ester resin, 1,3- or 1,4-dicyanatobenzene, 1,3,5-tricyanatobenzene, 1, 3-, 1,4-, 1,6-, 1,8-, 2,6- or 2,7-dicyanatonaphthalene, 1,3,6-tricyanatonaphthalene, 4,4-dicyanatobiphenyl, bis (4-dicyanatophenyl) methane, 2,2-bis (4-cyanatophenyl) propane, 2,2-bis (3,5-dibromo-4-cyanatophenyl) propane, bis (4-cyanatophenyl) Ether, bis (4-cyanatophenyl) thioether, bis (4-cyanatophenyl) sulfone, tris (4-cyanatophenyl
- bisphenol A type cyanate ester resins and their prepolymers and naphthol aralkyl type cyanate ester resins are particularly preferred in terms of heat resistance.
- These cyanate ester compounds may be used alone or in combination of two or more.
- the content of the cyanate ester compound is preferably in the range of 1 to 30% by mass and more preferably in the range of 5 to 20% by mass with respect to the amount of the resin composition from the viewpoint of electrical characteristics.
- the polycarbonate is preferably a brominated polycarbonate oligomer from the viewpoint of imparting flame retardancy.
- the molecular weight of the brominated polycarbonate oligomer is not particularly limited, but those having a weight average molecular weight of 500 to 3500 are preferred.
- the content of brominated polycarbonate oligomer is preferably in the range of 1 to 10% by mass, more preferably in the range of 2 to 8.5% by mass, and further in the range of 2 to 5% by mass with respect to the amount of the resin composition. preferable.
- the content of the brominated polycarbonate is in the above range, not only flame retardancy can be imparted but also powder falling can be suppressed.
- the resin composition in the present invention may contain polystyrene having a crosslinked structure or an oligomer of styrene and / or substituted styrene which is a copolymer of styrene and another aromatic vinyl compound.
- aromatic vinyl compound include, but are not limited to, ⁇ -methylstyrene, vinyltoluene, divinylbenzene, chlorostyrene, bromostyrene, and the like.
- Examples of the method for producing a styrene oligomer include, but are not limited to, copolymerization with a divinyl compound, combined use of a peroxide and radiation treatment.
- a styrene monomer and divinylbenzene are used under a polymerization catalyst.
- examples thereof include a method of producing by suspension polymerization or solution polymerization.
- the shape of the styrene oligomer powder used in the present invention is not particularly limited, and for example, any shape such as a spherical shape and an indefinite shape can be used.
- the varnish of the present invention contains the above resin composition, inorganic filler and polar solvent.
- any inorganic filler that is generally used in laminate applications can be used.
- silicas such as natural silica, fused silica, synthetic silica, amorphous silica, hollow silica, molybdenum compounds such as molybdenum oxide and zinc molybdate, zinc borate, zinc stannate, alumina, clay, kaolin, talc , Fired clay, fired kaolin, fired talc, mica, short glass fibers (glass fine powders such as E glass and D glass), hollow glass and the like.
- silicas, talc, and short glass fibers are preferable, and silicas are particularly preferable from the viewpoint of electrical characteristics.
- These inorganic fillers can be used alone or in combination of two or more.
- silica when silica is used as the inorganic filler, its average particle size (D50) is not particularly limited, but considering dispersibility, mesoporous silica having an average particle size (D50) of 0.1 to 3 ⁇ m, spherical Preferable examples include fused silica, spherical synthetic silica, and hollow spherical silica.
- D50 is the median diameter (median diameter), and is the diameter at which the masses on the large side and the small side are equal when the particle size distribution of the measured powder is divided into two. Generally, it is measured by a wet laser diffraction / scattering method.
- the content of the inorganic filler in the varnish is preferably in the range of 10 to 200 parts by mass, particularly preferably in the range of 40 to 100 parts by mass with respect to 100 parts by mass of the resin composition.
- the polar solvent contained in the varnish of the present invention is not particularly limited as long as it has polarity.
- Examples thereof include acetone, methyl ethyl ketone, propylene glycol monomethyl ether acetate, N, N-dimethylformamide, and dimethylacetamide.
- methyl ethyl ketone and propylene glycol monomethyl ether acetate are preferable from the viewpoint of solubility of the resin composition.
- the content of the polar solvent in the varnish is not particularly limited, but the total amount of the resin composition and the inorganic filler is 100 masses from the viewpoint of the remaining amount of the solvent, the uniform dispersion of the resin component, and the impregnation property into the glass cloth.
- the amount is preferably 10 to 180 parts by mass with respect to parts.
- the varnish of the present invention can contain other optional components such as a silane coupling agent, a wetting and dispersing agent, a curing accelerator, a crosslinking type curing agent, a polymerization inhibitor, and an additive.
- Additives include UV absorbers, antioxidants, photopolymerization initiators, optical brighteners, photosensitizers, dyes, pigments, thickeners, lubricants, antifoaming agents, dispersants, leveling agents, brighteners Etc. can be used in appropriate combinations as desired.
- the varnish of the present embodiment can be prepared according to a conventional method, and any varnish that uniformly contains the above-described resin composition, inorganic filler, polar solvent, and other optional components described above can be obtained.
- the preparation method is not particularly limited.
- the varnish of this embodiment can be easily prepared by sequentially blending various components in a polar solvent and sufficiently stirring.
- the prepreg according to the present invention is obtained by impregnating or coating the above varnish on a substrate and then drying it.
- the drying conditions during the production of the prepreg are not particularly limited as long as the amount of the residual polar solvent in the prepreg after drying is 3% by mass or less, and the following examples can be given.
- the size of the prepreg impregnated into the substrate is preferably 100 to 600 mm in width and 200 to 1000 mm in length, from the viewpoint of efficiently reducing the amount of residual solvent, and is 300 to 550 mm in width and 300 to 700 mm in length. More preferably.
- As the drying method it is preferable to dry at room temperature for 5 seconds to 10 minutes, then dry with a nitrogen stream, or use a steam type heat source.
- the drying temperature is preferably in the range of 120 to 220 ° C., more preferably in the range of 150 to 200 ° C., from the viewpoint of not accelerating the residual amount of solvent in the prepreg and the curing of the resin composition.
- the drying time is preferably 2 to 15 minutes and more preferably 3 to 10 minutes from the viewpoint of not promoting the drying efficiency of the solvent and the curing of the resin composition.
- the pressure during drying may be reduced pressure or normal pressure, but it is preferably performed at normal pressure from the viewpoint of cost.
- the total amount of the resin composition and the inorganic filler with respect to the total amount of the prepreg including the base material after the drying step is preferably in the range of 30 to 90% by mass.
- known base materials used for various printed wiring board materials can be used as the base material used when producing the prepreg.
- Examples thereof include woven fabrics such as E glass, D glass, S glass, T glass, NE glass, quartz, and liquid crystal polyester.
- the thickness of the woven fabric is not particularly limited, it is used for laminated board applications in the range of 0.01 to 0.2 mm.
- the woven fabric that has been subjected to ultra-opening treatment and plugging treatment has dimensional stability. From the aspect, it is preferable.
- a glass woven fabric surface-treated with a silane coupling agent such as epoxy silane treatment or amino silane treatment is preferred from the viewpoint of moisture absorption heat resistance.
- a liquid crystal polyester woven fabric is preferable from the viewpoint of electrical characteristics.
- the metal foil-clad laminate according to the present invention is formed by lamination using the above prepreg. Specifically, one or a plurality of the prepregs described above are stacked, and a metal foil is disposed on one or both sides thereof, for example, a temperature of 180 to 220 ° C., a heating time of 100 to 300 minutes, and a surface pressure of 20 to 40 kg / cm 2. It is manufactured by laminating and molding.
- the thickness of the metal foil to be used is not particularly limited as long as it is used for the printed wiring board material, but it is preferably 3 to 35 ⁇ m. In consideration of conductor loss in a high frequency region, an electrolytic copper foil having a small mat surface roughness is preferable.
- a 35 ⁇ m copper foil is disposed on both surfaces of one prepreg of the present invention, laminated under the above conditions, an inner layer circuit is formed, and this circuit is blackened. Processing is performed to obtain an inner layer circuit board. It is also possible to form a multilayer board by combining and molding this inner layer circuit board and the prepreg of the present invention.
- the metal foil-clad laminate of this embodiment can be suitably used as a printed wiring board.
- the printed wiring board can be manufactured according to a conventional method, and the manufacturing method is not particularly limited.
- an example of the manufacturing method of a printed wiring board is shown.
- a metal foil-clad laminate such as the copper clad laminate described above is prepared.
- an etching process is performed on the surface of the metal foil-clad laminate to form an inner layer circuit, thereby producing an inner layer substrate.
- the inner layer circuit surface of the inner layer substrate is subjected to a surface treatment to increase the adhesive strength as necessary, then the required number of the prepregs are stacked on the inner layer circuit surface, and a metal foil for the outer layer circuit is stacked on the outer surface.
- a multilayer laminate is produced in which an insulating layer made of a cured material of the base material and the thermosetting resin composition is formed between the inner layer circuit and the metal foil for the outer layer circuit.
- a plated metal film is formed on the wall surface of the hole to connect the inner layer circuit and the metal foil for the outer layer circuit.
- a printed wiring board is manufactured by performing an etching process on the metal foil for forming an outer layer circuit.
- the printed wiring board obtained in the above production example has an insulating layer and a conductor layer formed on the surface of the insulating layer, and the insulating layer includes the prepreg of the present embodiment described above.
- Example 1 In the general formula (4), — (O—X—O) — is represented by the formula (8), — (Y—O) — is represented by the formula (11), and a and b are 0 to 100 polyphenylene ether (OPE) -2St 1200, manufactured by Mitsubishi Gas Chemical Co., Ltd., number average molecular weight 1187, vinyl group equivalent: 590 g / eq., 69 parts by mass, brominated bisphenol A type epoxy resin 1 (E153, manufactured by DIC Corporation, epoxy equivalent: 400 g / eq., Weight average molecular weight 400-800) 11.5 parts by mass, cresol novolac epoxy resin (N680, manufactured by DIC Corporation, epoxy equivalent: 215 g / eq., Weight average molecular weight 2400) 1.0 part by mass Bisphenol A type cyanate ester (CA210, manufactured by Mitsubishi Gas Chemical Company, cyanate equivalent: 139 g / eq.) 13.5 parts by mass, brominated
- the obtained varnish was impregnated and coated on an E glass cloth having a thickness of 0.08 mm and dried by heating at 170 ° C. for 5 minutes using a dryer (explosion-proof steam dryer, manufactured by Takasugi Seisakusho Co., Ltd.)
- a prepreg of 55% by mass of the resin composition was obtained.
- 18 ⁇ m copper foil (3EC- ⁇ , manufactured by Mitsui Mining & Smelting Co., Ltd.) was placed on both sides of the 8 sheets of 55% by mass prepreg, and vacuum pressing was performed for 150 minutes at a pressure of 30 kg / cm 2 and a temperature of 210 ° C.
- An 18 ⁇ m copper-clad laminate with a thickness of 0.8 mm was obtained.
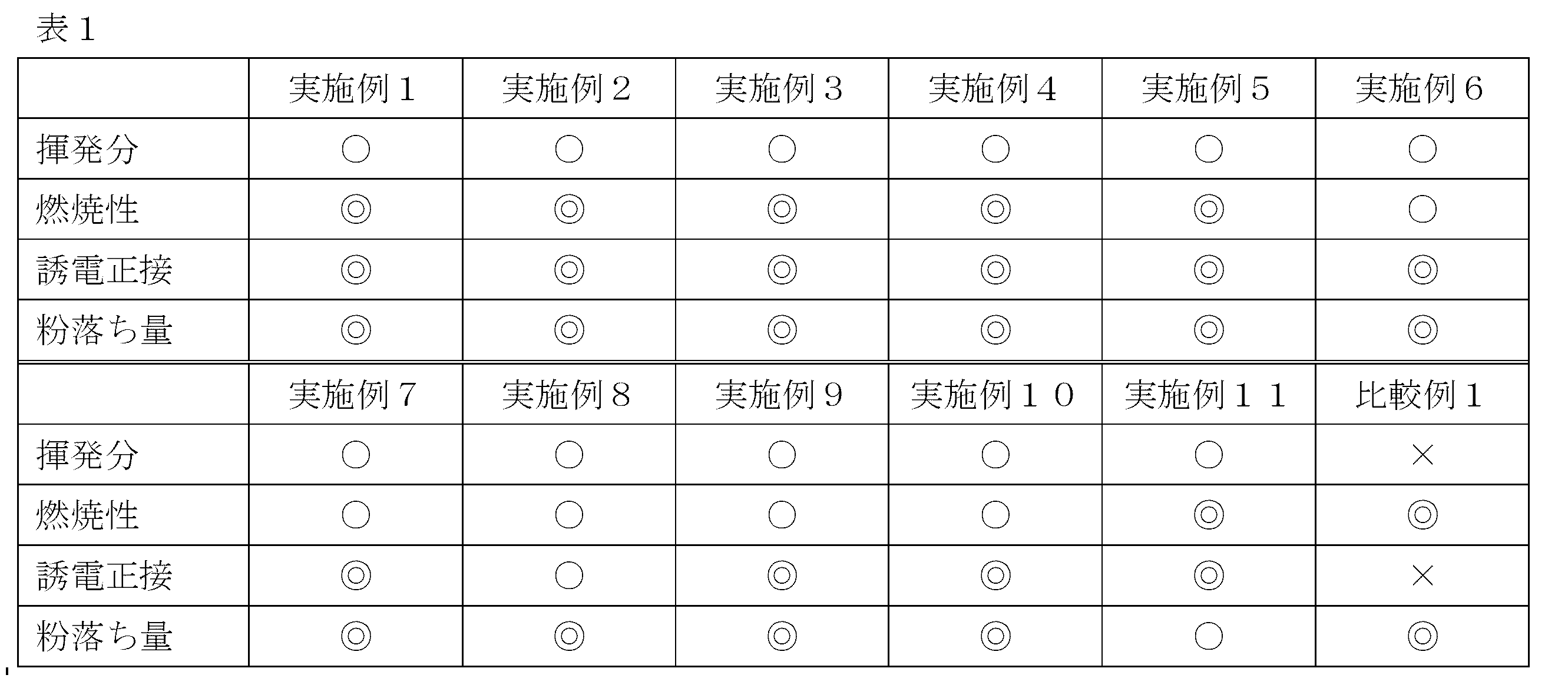
- the volatile matter, combustibility, dielectric loss tangent, and amount of dust fall were evaluated. The results are shown in Table 1.
- Dielectric loss tangent Using a test piece from which a copper foil of a 0.8 mm thick copper-clad laminate is removed, measure the dielectric loss tangent of 10 GHz by cavity resonator perturbation method (Agilent 8722ES, manufactured by Agilent Technologies). When the average value was lower than 0.0065, ( ⁇ ), when it was 0.0065 to 0.007, ( ⁇ ), and when it was larger than 0.007, ( ⁇ ).
- Amount of powder fall A prepreg (size: 40 mm x 345 mm) is placed on an aluminum sheet, a cylindrical SUS roll (weight: 1 kg, diameter: 25 mm) is passed over the prepreg, and the prepreg weight before and after that The reduction rate was calculated, and the case where the average value of the three measurement results was 5 wt% or less was evaluated as ( ⁇ ⁇ ⁇ ), and the case where it was larger than 5 wt% and smaller than 10 wt% was marked as ( ⁇ ).
- Example 2 The procedure was the same as Example 1 except that 13.5 parts by mass of the ⁇ -naphthol aralkyl cyanate obtained in Synthesis Example 1 was used in place of the bisphenol A type cyanate used in Example 1.
- Example 3 The procedure was the same as Example 1 except that 7.5 parts by mass of the bisphenol A type cyanate ester used in Example 1 and 6 parts by mass of the ⁇ -naphthol aralkyl type cyanate ester used in Example 2 were used.
- Example 4 67.5 parts by mass of polyphenylene ether used in Example 1, 3.0 parts by mass of cresol novolac type epoxy resin, 7.5 parts by mass of brominated bisphenol A type epoxy resin 1 and 8.5 parts by mass of brominated polycarbonate The procedure was the same as in Example 3 except that the part was changed to the part.
- Example 5 The polyphenylene ether used in Example 1 was 71.5 parts by mass, the brominated bisphenol A type epoxy resin 1 was 13.0 parts by mass, the brominated polycarbonate was 2.0 parts by mass, and no cresol novolac type epoxy resin was used. The procedure was the same as in Example 3 except that.
- Example 6 81.8 parts by mass of polyphenylene ether used in Example 1, 5.7 parts by mass of bisphenol A type cyanate ester, 4.5 parts by mass of ⁇ -naphthol aralkyl type cyanate ester used in Example 2, Brominated bisphenol A type epoxy resin 1, cresol novolak type epoxy resin and brominated polycarbonate were not used, but brominated bisphenol A type epoxy resin 2 (DER515, manufactured by Dow Chemical Company, epoxy equivalent: 550 g / eq., Weight average molecular weight) 1060-1120) 3.4 parts by mass, brominated novolac epoxy resin (BREN, Nippon Kayaku Co., Ltd., epoxy equivalent: 285 g / eq., Weight average molecular weight 1940) 3.4 parts by mass, bisphenol A Type epoxy resin 1 (828EL, manufactured by Mitsubishi Chemical Corporation, epoxy equivalent: 1 0 g / eq., Except for using 1.1 parts by weight average molecular weight from 368 to 388) were the same
- Example 7 The same procedure as in Example 6 was conducted except that 3.4 parts by mass of the cresol novolac type epoxy resin used in Example 1 was used without using the brominated novolak type epoxy resin used in Example 6.
- Example 8 52.0 parts by mass of polyphenylene ether used in Example 1, 9.0 parts by mass of cresol novolac type epoxy resin, 15.0 parts by mass of bisphenol A type cyanate ester, ⁇ -naphthol aralkyl used in Example 2 12.0 parts by mass of type cyanate ester, brominated bisphenol A type epoxy resin 1, brominated polycarbonate was not used, and 3.0 parts by mass of bisphenol A type epoxy resin 1 used in Example 6 was used.
- the procedure was the same as Example 3 except that 9.0 parts by mass of bisphenol A type epoxy resin 2 (E-1051, manufactured by DIC Corporation, epoxy equivalent: 475 g / eq., Weight average molecular weight 900) was used.
- Example 9 The polyphenylene ether used in Example 1 was 68.0 parts by mass, the cresol novolac type epoxy resin was 6.0 parts by mass, the bisphenol A type epoxy resin 1 used in Example 6 was 2.0 parts by mass, and in Example 8.
- Example 10 76.0 parts by mass of polyphenylene ether used in Example 1, 4.5 parts by mass of cresol novolac type epoxy resin, 1.5 parts by mass of bisphenol A type epoxy resin 1 used in Example 6,
- the bisphenol A type epoxy resin 2 used was 4.5 parts by mass
- the bisphenol A type cyanate ester was 7.5 parts by mass
- the ⁇ -naphthol aralkyl type cyanate ester used in Example 2 was 6.0 parts by mass. Except for this, the procedure was the same as in Example 8.
- Example 11 69.0 parts by mass of polyphenylene ether used in Example 1, 5.0 parts by mass of cresol novolac type epoxy resin, 2.0 parts by mass of bisphenol A type epoxy resin 2 used in Example 8, and bisphenol A type cyanide
- the brominated bisphenol A type epoxy resin 1 was prepared using 5.0 parts by mass of the acid ester, 4.0 parts by mass of the ⁇ -naphthol aralkyl cyanate ester used in Example 2, and 15.0 parts by mass of the brominated polycarbonate. The procedure was the same as Example 1 except that it was not used.
- Example 1 A prepreg and a copper clad laminate were obtained in the same manner as in Example 1 except that the temperature and drying time with a dryer were 100 ° C. for 5 minutes, and the volatile content was 3 wt% or more. The results are shown in Table 1.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- General Chemical & Material Sciences (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Textile Engineering (AREA)
- Inorganic Chemistry (AREA)
- Reinforced Plastic Materials (AREA)
- Laminated Bodies (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Polyethers (AREA)
Description
通常、プリント配線板用の積層板は、樹脂組成物における成分を有機溶媒に均一分散させ、得られたワニスをガラスクロス等の基材に含浸又は塗布し、乾燥させて得られたプリプレグを用いて作製される。
低誘電正接性を有する樹脂の主成分としてはポリフェニレンエーテル等の極性の低いものがよく用いられ、上述した有機溶媒も主成分の極性に合わせて極性の低いトルエン等が用いられる(例えば特許文献3参照)。
しかし、トルエンは環境汚染の観点から使用が自粛される傾向にあり、別の溶媒を使用することが求められている。
一方、このような低誘電正接用の樹脂成分として極性溶媒を使用することは一般に行われておらず、極性溶媒を使用した場合におけるプリプレグ中の溶媒残存量が作製されたプリプレグ等に与える影響について十分な検討がなされていない。
[1]ポリフェニレンエーテルを主成分として含有する樹脂組成物と、無機充填材及び極性溶媒とを含有するワニスを基材に含浸又は塗布し、その後乾燥工程を経て作製されるプリプレグであって、
該プリプレグ中の前記極性溶媒の含有量が3質量%以下であり、
該プリプレグを用いて作製した積層板の10GHzにおける誘電正接が、0.001~0.007となる、プリプレグ。
[2]前記極性溶媒が、アセトン、メチルエチルケトン、プロピレングリコールモノメチルエーテルアセテート、N,N―ジメチルホルムアミド及び、ジメチルアセトアミドからなる群より選択される少なくとも1種である、[1]に記載のプリプレグ。
[3]前記ワニス中における極性溶媒の含有量が、前記樹脂組成物及び前記無機充填材の合計100質量部に対し、10~180質量部である、[1]又は[2]に記載のプリプレグ。
[4]前記乾燥工程においては、120~200℃で2~30分の処理を行う、[1]~[3]のいずれか一項に記載のプリプレグ。
[5]前記樹脂組成物が、エポキシ樹脂、フェノール樹脂、シアン酸エステル化合物及びポリカーボネートからなる群より選択される少なくとも1種を更に含有する、[1]~[4]のいずれか一項に記載のプリプレグ。
[6]前記ポリフェニレンエーテルの数平均分子量が500~3000である、[1]~[5]のいずれか一項に記載のプリプレグ。
[7]前記樹脂組成物におけるポリフェニレンエーテルの含有量が、50質量%以上である、[1]~[6]のいずれか一項に記載のプリプレグ。
[8]前記ポリフェニレンエーテルが、一般式(4):
(式中、-(O-X-O)-は、一般式(5):
(R21,R22,R23,R27,R28は、同一又は異なってもよく、炭素数6以下のアルキル基又はフェニル基である。R24,R25,R26は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。)
又は一般式(6):
(R29,R30,R31,R32,R33,R34,R35,R36は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。-B-は、炭素数20以下の直鎖状、分岐状又は環状の2価の炭化水素基である。)
で表される構造からなる。-(Y-O)-は、一般式(7):
(R39,R40は、同一又は異なってもよく、炭素数6以下のアルキル基又はフェニル基である。R37,R38は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。)
で表され、1種類の構造又は2種類以上の構造がランダムに配列している。a,bは、少なくともいずれか一方が0でない、0~100の整数を示す。)
で表されるものである、[1]~[7]のいずれか一項に記載のプリプレグ。
[9]前記樹脂組成物が、エポキシ樹脂を含有し、かつ該樹脂組成物における該エポキシ樹脂の含有量が、1~30質量%である、[1]~[8]のいずれか一項に記載のプリプレグ。
[10]前記樹脂組成物が、シアン酸エステル化合物を含有し、かつ該樹脂組成物における該シアン酸エステル化合物の含有量が、1~30質量%である、[1]~[9]のいずれか一項に記載のプリプレグ。
[11]前記樹脂組成物が、臭素化ポリカーボネートオリゴマーを含有し、かつ該樹脂組成物における該ポリカーボネートオリゴマーの含有量が、2~10質量%である、[1]~[10]のいずれか一項に記載のプリプレグ。
[12]前記無機充填材が、天然シリカ、溶融シリカ、合成シリカ、アモルファスシリカ、中空シリカ、ガラス短繊維及びタルクからなる群より選択される少なくとも1種である、[1]~[11]のいずれか一項に記載のプリプレグ。
[13]粉落ち量が5質量%以下である、[1]~[12]のいずれか1項に記載のプリプレグ。
[14][1]~[13]のいずれか一項に記載のプリプレグを用いて作製された、積層板。
[15][1]~[13]のいずれか一項に記載のプリプレグ及び金属箔を用いて作製された、金属箔張積層板。
[16]絶縁層と、前記絶縁層の表面に形成された導体層とを含むプリント配線板であって、前記絶縁層が、[1]~[13]のいずれか一項に記載のプリプレグを含む、プリント配線板。
を、提供するものである。
また、本発明のプリプレグは、プリプレグの製造時、あるいは製造後に、プリプレグより樹脂粉の脱落する現象、粉落ちが抑制され、安定的に生産することが可能である。
(式中、R1、R2、R3、及びR4は、同一又は異なってもよく、炭素数6以下のアルキル基、アリール基、ハロゲン、又は水素を表す。)
で表される繰り返し単位を少なくとも含んでなる重合体であることが好ましい。該重合体は、一般式(2):
(R5,R6,R7,R11,R12は、同一又は異なってもよく、炭素数6以下のアルキル基又はフェニル基である。R8,R9,R10は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。)
で表される繰り返し単位、及び/又は、一般式(3):
(R13,R14,R15,R16,R17,R18,R19,R20は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。-A-は、炭素数20以下の直鎖状、分岐状又は環状の2価の炭化水素基である。)
で表される繰り返し単位をさらに含んでもよい。
(式中、-(O-X-O)-は、一般式(5):
(R21,R22,R23,R27,R28は、同一又は異なってもよく、炭素数6以下のアルキル基又はフェニル基である。R24,R25,R26は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。)
又は一般式(6):
(R29,R30,R31,R32,R33,R34,R35,R36は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。-B-は、炭素数20以下の直鎖状、分岐状又は環状の2価の炭化水素基である。)
で表される構造からなる。-(Y-O)-は、一般式(7):
(R39,R40は、同一又は異なってもよく、炭素数6以下のアルキル基又はフェニル基である。R37,R38は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。)
で表され、1種類の構造又は2種類以上の構造がランダムに配列している。a,bは、少なくともいずれか一方が0でない、0~100の整数を示す。)
で表される変性ポリフェニレンエーテルを含むことが特に好ましい。
また、このような変性ポリフェニレンエーテルは、例えば、三菱ガス化学(株)(OPE-2St 1200など)から入手することができる。
ここで、樹脂組成物の量とは、ワニスにおける無機充填材及び溶媒を除いた各成分の合計質量をいうものとする。
この中でもビスフェノールA型エポキシ樹脂、フェノールノボラック型エポキシ樹脂、クレゾールノボラック型エポキシ樹脂、ナフタレン骨格変性したノボラック型エポキシ樹脂が好ましい。
スチレンオリゴマーの製法としては、例えば、ジビニル化合物との共重合、過酸化物の併用や放射線処理等が挙げられるが、これらに限定されず、例えばスチレンモノマーとジビニルベンゼンを使用し、重合触媒下で懸濁重合、又は溶液重合などにより製造する方法が挙げられる。
本発明で使用されるスチレンオリゴマー粉末の形状は、特に限定されないが、例えば、球状、不定形状等、何れも使用可能である。
プリプレグの製造時における乾燥条件は、乾燥後のプリプレグ中の残存極性溶媒量が3質量%以下となるようにするものであれば特に限定されないが、以下の例を挙げることができる。
基材に含浸させるプリプレグの大きさは、幅100~600mm、長さ200~1000mmであることが、残存溶媒量を効率的に減らせる観点から好ましく、幅300~550mm、長さ300~700mmであることがより好ましい。
乾燥方法としては、5秒~10分の間室温下で乾燥させた後、窒素気流による乾燥、スチーム式等の熱源を用いた乾燥方法が好ましい。
乾燥温度はプリプレグにおける溶媒の残存量と、樹脂組成物の硬化を促進させない観点から、120~220℃の範囲で行うことが好ましく、150~200℃の範囲で行うことがより好ましい。
乾燥時間は、溶媒の乾燥効率と樹脂組成物の硬化を促進させない観点から、2~15分で行うことが好ましく、3~10分で行うことがより好ましい。
また、乾燥時の圧力としては減圧でも常圧であってもよいが、コスト的な観点から常圧で行うことが好ましい。
温度計、攪拌器、滴下漏斗及び還流冷却器を取りつけた反応器を予めブラインにより0~5℃に冷却しておき、そこへ塩化シアン7.47g(0.122mol)、35%塩酸9.75g(0.0935mol)、水76ml、及び塩化メチレン44mlを仕込んだ。
この反応器内の温度を-5~+5℃、pHを1以下を保ちながら、撹拌下、式(2)におけるRがすべて水素原子であるα-ナフトールアラルキル樹脂(SN485、OH基当量:214g/eq.軟化点:86℃、新日鐵化学(株)製)20g(0.0935mol)、及びトリエチルアミン14.16g(0.14mol)を塩化メチレン92mlに溶解した溶液を滴下漏斗により1時間かけて滴下し、滴下終了後、更にトリエチルアミン4.72g(0.047mol)を15分間かけて滴下した。
滴下終了後、同温度で15分間撹拌後、反応液を分液し、有機層を分取した。得られた有機層を水100mlで2回洗浄した後、エバポレーターにより減圧下で塩化メチレンを留去し、最終的に80℃で1時間濃縮乾固させて、α-ナフトールアラルキル樹脂のシアン酸エステル化物(α-ナフトールアラルキル型シアン酸エステル)が23.5g得られた。
一般式(4)における-(O-X-O)-が式(8)、-(Y-O)-が式(11)で表され、a及びbが0~100であるポリフェニレンエーテル(OPE-2St 1200、三菱ガス化学(株)製、数平均分子量1187、ビニル基当量:590g/eq.)69質量部、臭素化ビスフェノールA型エポキシ樹脂1(E153、DIC(株)製、エポキシ当量:400g/eq.、重量平均分子量400~800)11.5質量部、クレゾールノボラック型エポキシ樹脂(N680、DIC(株)製、エポキシ当量:215g/eq.、重量平均分子量2400)1.0質量部、ビスフェノールA型シアン酸エステル(CA210、三菱ガス化学(株)製、シアネート当量:139g/eq.)13.5質量部、臭素化ポリカーボネート(FG8500、帝人化成(株)製)5質量部、球状シリカ(SC2050、(株)アドマテックス製、平均粒子径0.5μm)50質量部、メチルエチルケトンとプロピレングリコールモノメチルエーテルアセテートをそれぞれ35質量部混合し、固形分65質量%に希釈したワニスを得た。この得られたワニスを厚さ0.08mmのEガラスクロスに含浸塗工し、乾燥機(耐圧防爆型スチーム乾燥機、(株)高杉製作所製)を用いて170℃、5分加熱乾燥し、樹脂組成物55質量%のプリプレグを得た。この55質量%のプリプレグ8枚を重ねた両面に18μm銅箔(3EC―■、三井金属鉱業(株)製)を配置し、圧力30kg/cm2、温度210℃で150分間真空プレスを行い、厚さ0.8mmの18μm銅張り積層板を得た。得られたプリプレグと銅張り積層板を用いて、揮発分、燃焼性、誘電正接、粉落ち量の評価を行った。結果を表1に示す。
1)揮発分:プリプレグ(サイズ:520mm×345mm)を、作製直後に乾固させ(160℃、15分)、その前後でのプリプレグ重量減少率を算出し、3質量%以下である場合を(○)、3質量%より大きい場合を(×)とした。
2)燃焼性:銅張り積層板の銅箔をエッチングにより除去したのちに、UL94垂直燃焼試験法に準拠して評価し、V-0であった場合を(◎)、V-1であった場合を(○)とした。
3)誘電正接:厚さ0.8mmの銅張り積層板の銅箔を除去した試験片を使用し、空洞共振器摂動法(Agilent 8722ES、アジレントテクノロジー製)にて10GHzの誘電正接の測定を2回実施し、その平均値が0.0065より低かった場合を(◎)、0.0065~0.007であった場合を(○)、0.007よりも大きい場合を(×)とした。
4)粉落ち量:プリプレグ(サイズ:40mm×345mm)をアルミ製シート上に配置し、プリプレグ上に円柱型SUS製ロール(重量:1kg、直径:25mm)を通過させ、その前後でのプリプレグ重量減少率を算出し、3回の測定結果の平均値が、5wt%以下である場合を(◎)、5wt%より大きく、10wt%より小さい場合を(○)とした。
実施例1で使用したビスフェノールA型シアン酸エステルに代えて合成例1で得られたα-ナフトールアラルキル型シアン酸エステルを13.5質量部用いた以外は実施例1と同様にした。
実施例1で使用したビスフェノールA型シアン酸エステルを7.5質量部、実施例2で使用したα-ナフトールアラルキル型シアン酸エステルを6質量部用いた以外は実施例1と同様にした。
実施例1で使用したポリフェニレンエーテルを67.5質量部、クレゾールノボラック型エポキシ樹脂を3.0質量部、臭素化ビスフェノールA型エポキシ樹脂1を7.5質量部、臭素化ポリカーボネートを8.5質量部とした以外は実施例3と同様にした。
実施例1で使用したポリフェニレンエーテルを71.5質量部、臭素化ビスフェノールA型エポキシ樹脂1を13.0質量部、臭素化ポリカーボネートを2.0質量部とし、クレゾールノボラック型エポキシ樹脂を使用しなかった以外は実施例3と同様にした。
実施例1で使用したポリフェニレンエーテルを81.8質量部、ビスフェノールA型シアン酸エステルを5.7質量部、実施例2で使用したα-ナフトールアラルキル型シアン酸エステルを4.5質量部とし、臭素化ビスフェノールA型エポキシ樹脂1、クレゾールノボラック型エポキシ樹脂及び臭素化ポリカーボネートを使用せず、臭素化ビスフェノールA型エポキシ樹脂2(DER515、ダウケミカル社製、エポキシ当量:550g/eq.、重量平均分子量1060~1120)を3.4質量部、臭素化ノボラック型エポキシ樹脂(BREN、日本化薬(株)製、エポキシ当量:285g/eq.、重量平均分子量1940)を3.4質量部、ビスフェノールA型エポキシ樹脂1(828EL、三菱化学(株)製、エポキシ当量:190g/eq.、重量平均分子量368~388)を1.1質量部使用した以外は実施例3と同様にした。
実施例6で使用した臭素化ノボラック型エポキシ樹脂を使用せず、実施例1で使用したクレゾールノボラック型エポキシ樹脂を3.4質量部使用した以外は実施例6と同様にした。
実施例1で使用したポリフェニレンエーテルを52.0質量部、クレゾールノボラック型エポキシ樹脂を9.0質量部、ビスフェノールA型シアン酸エステルを15.0質量部、実施例2で使用したα-ナフトールアラルキル型シアン酸エステルを12.0質量部とし、臭素化ビスフェノールA型エポキシ樹脂1、臭素化ポリカーボネートを使用せず、実施例6で使用したビスフェノールA型エポキシ樹脂1を3.0質量部使用し、ビスフェノールA型エポキシ樹脂2(E-1051、DIC(株)製、エポキシ当量:475g/eq.、重量平均分子量900)を9.0質量部使用した以外は実施例3と同様にした。
実施例1で使用したポリフェニレンエーテルを68.0質量部、クレゾールノボラック型エポキシ樹脂を6.0質量部、実施例6で使用したビスフェノールA型エポキシ樹脂1を2.0質量部、実施例8で使用したビスフェノールA型エポキシ樹脂2を6.0質量部、ビスフェノールA型シアン酸エステルを10.0質量部、実施例2で使用したα-ナフトールアラルキル型シアン酸エステルを8.0質量部とした以外は実施例8と同様にした。
実施例1で使用したポリフェニレンエーテルを76.0質量部、クレゾールノボラック型エポキシ樹脂を4.5質量部、実施例6で使用したビスフェノールA型エポキシ樹脂1を1.5質量部、実施例8で使用したビスフェノールA型エポキシ樹脂2を4.5質量部、ビスフェノールA型シアン酸エステルを7.5質量部、実施例2で使用したα-ナフトールアラルキル型シアン酸エステルを6.0質量部とした以外は実施例8と同様にした。
実施例1で使用したポリフェニレンエーテルを69.0質量部、クレゾールノボラック型エポキシ樹脂を5.0質量部、実施例8で使用したビスフェノールA型エポキシ樹脂2を2.0質量部、ビスフェノールA型シアン酸エステルを5.0質量部、実施例2で使用したα-ナフトールアラルキル型シアン酸エステルを4.0質量部、臭素化ポリカーボネートを15.0質量部用い、臭素化ビスフェノールA型エポキシ樹脂1を使用しなかった以外は実施例1と同様にした。
乾燥機による温度及び乾燥時間を100℃、5分とし、揮発分が3wt%以上になった以外は実施例1と同様に行い、プリプレグ、銅張積層板を得た。結果を表1に示す。
Claims (16)
- ポリフェニレンエーテルを主成分として含有する樹脂組成物と、無機充填材及び極性溶媒とを含有するワニスを基材に含浸又は塗布し、その後乾燥工程を経て作製されるプリプレグであって、
該プリプレグ中の前記極性溶媒の含有量が3質量%以下であり、
該プリプレグを用いて作製した積層板の10GHzにおける誘電正接が、0.001~0.007となる、プリプレグ。 - 前記極性溶媒が、アセトン、メチルエチルケトン、プロピレングリコールモノメチルエーテルアセテート、N,N―ジメチルホルムアミド及び、ジメチルアセトアミドからなる群より選択される少なくとも1種である、請求項1に記載のプリプレグ。
- 前記ワニス中における極性溶媒の含有量が、前記樹脂組成物及び前記無機充填材の合計100質量部に対し、10~180質量部である、請求項1又は2に記載のプリプレグ。
- 前記乾燥工程においては、120~200℃で2~30分の処理を行う、請求項1~3のいずれか一項に記載のプリプレグ。
- 前記樹脂組成物が、エポキシ樹脂、フェノール樹脂、シアン酸エステル化合物及びポリカーボネートからなる群より選択される少なくとも1種を更に含有する、請求項1~4のいずれか一項に記載のプリプレグ。
- 前記ポリフェニレンエーテルの数平均分子量が500~3000である、請求項1~5のいずれか一項に記載のプリプレグ。
- 前記樹脂組成物におけるポリフェニレンエーテルの含有量が、50質量%以上である、請求項1~6のいずれか一項に記載のプリプレグ。
- 前記ポリフェニレンエーテルが、一般式(4):
(式中、-(O-X-O)-は、一般式(5):
(R21,R22,R23,R27,R28は、同一又は異なってもよく、炭素数6以下のアルキル基又はフェニル基である。R24,R25,R26は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。)
又は一般式(6):
(R29,R30,R31,R32,R33,R34,R35,R36は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。-B-は、炭素数20以下の直鎖状、分岐状又は環状の2価の炭化水素基である。)
で表される構造からなる。-(Y-O)-は、一般式(7):
(R39,R40は、同一又は異なってもよく、炭素数6以下のアルキル基又はフェニル基である。R37,R38は、同一又は異なってもよく、水素原子、炭素数6以下のアルキル基又はフェニル基である。)
で表され、1種類の構造又は2種類以上の構造がランダムに配列している。a,bは、少なくともいずれか一方が0でない、0~100の整数を示す。)
で表されるものである、請求項1~7のいずれか一項に記載のプリプレグ。 - 前記樹脂組成物が、エポキシ樹脂を含有し、かつ該樹脂組成物における該エポキシ樹脂の含有量が、1~30質量%である、請求項1~8のいずれか一項に記載のプリプレグ。
- 前記樹脂組成物が、シアン酸エステル化合物を含有し、かつ該樹脂組成物における該シアン酸エステル化合物の含有量が、1~30質量%である、請求項1~9のいずれか一項に記載のプリプレグ。
- 前記樹脂組成物が、臭素化ポリカーボネートオリゴマーを含有し、かつ該樹脂組成物における該ポリカーボネートオリゴマーの含有量が、2~10質量%である、請求項1~10のいずれか一項に記載のプリプレグ。
- 前記無機充填材が、天然シリカ、溶融シリカ、合成シリカ、アモルファスシリカ、中空シリカ、ガラス短繊維及びタルクからなる群より選択される少なくとも1種である、請求項1~11のいずれか一項に記載のプリプレグ。
- 粉落ち量が5質量%以下である、請求項1~12のいずれか1項に記載のプリプレグ。
- 請求項1~13のいずれか一項に記載のプリプレグを用いて作製された、積層板。
- 請求項1~13のいずれか一項に記載のプリプレグ及び金属箔を用いて作製された、金属箔張積層板。
- 絶縁層と、前記絶縁層の表面に形成された導体層とを含むプリント配線板であって、
前記絶縁層が、請求項1~13のいずれか一項に記載のプリプレグを含む、プリント配線板。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014506268A JP6056849B2 (ja) | 2012-03-23 | 2013-03-21 | プリプレグ及び積層板 |
| CN201380012278.4A CN104169343B (zh) | 2012-03-23 | 2013-03-21 | 预浸料和层压板 |
| KR1020147024447A KR20140138677A (ko) | 2012-03-23 | 2013-03-21 | 프리프레그 및 적층판 |
| EP13764176.7A EP2829568B1 (en) | 2012-03-23 | 2013-03-21 | Prepreg and laminated board |
| US14/381,784 US20150044484A1 (en) | 2012-03-23 | 2013-03-21 | Prepreg and laminated board |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012066574 | 2012-03-23 | ||
| JP2012-066574 | 2012-03-23 | ||
| JP2012-191444 | 2012-08-31 | ||
| JP2012191444 | 2012-08-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013141298A1 true WO2013141298A1 (ja) | 2013-09-26 |
Family
ID=49222755
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/058075 Ceased WO2013141298A1 (ja) | 2012-03-23 | 2013-03-21 | プリプレグ及び積層板 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20150044484A1 (ja) |
| EP (1) | EP2829568B1 (ja) |
| JP (2) | JP6056849B2 (ja) |
| KR (1) | KR20140138677A (ja) |
| CN (1) | CN104169343B (ja) |
| MY (1) | MY167826A (ja) |
| TW (1) | TWI572649B (ja) |
| WO (1) | WO2013141298A1 (ja) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014240474A (ja) * | 2013-05-17 | 2014-12-25 | 三菱瓦斯化学株式会社 | 樹脂組成物、プリプレグ、樹脂シート、金属箔張積層板及びプリント配線板 |
| CN104629341A (zh) * | 2013-11-08 | 2015-05-20 | 中山台光电子材料有限公司 | 低介电树脂组合物,应用其的半固化胶片、覆铜箔基板、电路板 |
| US9051465B1 (en) | 2012-02-21 | 2015-06-09 | Park Electrochemical Corporation | Thermosetting resin composition containing a polyphenylene ether and a brominated fire retardant compound |
| US9243164B1 (en) | 2012-02-21 | 2016-01-26 | Park Electrochemical Corporation | Thermosetting resin composition containing a polyphenylene ether and a brominated fire retardant compound |
| WO2016038878A1 (ja) * | 2014-09-09 | 2016-03-17 | パナソニックIpマネジメント株式会社 | 硬化性組成物、プリプレグ、樹脂付き金属箔、金属張積層板、及びプリント配線板 |
| JP2016042169A (ja) * | 2014-08-19 | 2016-03-31 | 三菱瓦斯化学株式会社 | 電子写真感光体 |
| JP2016044208A (ja) * | 2014-08-20 | 2016-04-04 | 日立化成株式会社 | モールドアンダーフィル用樹脂組成物及び電子部品装置 |
| JP2018062568A (ja) * | 2016-10-12 | 2018-04-19 | 三菱瓦斯化学株式会社 | プリント配線板用樹脂組成物、プリプレグ、金属箔張積層板、積層樹脂シート、樹脂シート、及びプリント配線板 |
| WO2019167391A1 (ja) * | 2018-02-27 | 2019-09-06 | 京セラ株式会社 | プリプレグおよび回路基板用積層板 |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6788807B2 (ja) * | 2015-04-28 | 2020-11-25 | 三菱瓦斯化学株式会社 | 樹脂組成物、プリプレグ、金属箔張積層板、樹脂シート、及びプリント配線板 |
| JP2017031276A (ja) * | 2015-07-30 | 2017-02-09 | パナソニックIpマネジメント株式会社 | 熱硬化性樹脂組成物、並びに、それを用いた樹脂ワニス、樹脂付金属箔、樹脂フィルム、金属張積層板及びプリント配線板 |
| TWI580714B (zh) | 2016-03-10 | 2017-05-01 | 台燿科技股份有限公司 | 樹脂組合物及其應用 |
| WO2019208129A1 (ja) * | 2018-04-27 | 2019-10-31 | 三菱瓦斯化学株式会社 | 熱硬化性組成物、プリプレグ、金属箔張積層板、樹脂シート及びプリント配線板 |
| KR102672360B1 (ko) * | 2018-06-01 | 2024-06-04 | 미츠비시 가스 가가쿠 가부시키가이샤 | 수지 조성물, 프리프레그, 금속박 피복 적층판, 수지 시트 및 프린트 배선판 |
| KR20200038814A (ko) | 2018-10-04 | 2020-04-14 | 주식회사 엘지화학 | 회로 기판 제조용 연속 시트의 제조 방법 및 이로부터 제조된 회로 기판 제조용 연속 시트 |
| KR102400111B1 (ko) * | 2019-02-08 | 2022-05-19 | 주식회사 엘지화학 | 반도체 패키지용 열경화성 수지 조성물, 프리프레그 및 금속박 적층판 |
| US20250236693A1 (en) | 2022-01-31 | 2025-07-24 | Nippon Steel Chemical & Material Co., Ltd. | Resin composition, and molded product, cured product, film, composite material, cured composite material, laminate, metal foil with resin, and varnish for circuit board material obtainedtherefrom |
| US20250075033A1 (en) * | 2023-02-07 | 2025-03-06 | Shandong Xingshun New Material Co Ltd. | Low-molecular asymmetric thermosetting polyphenylene oxide resin polymer, and preparation method, purification method and use thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11124433A (ja) | 1997-10-22 | 1999-05-11 | Mitsubishi Gas Chem Co Inc | フェノールノボラック型シアン酸エステルプレポリマー |
| JP2005239767A (ja) | 2004-02-24 | 2005-09-08 | Matsushita Electric Works Ltd | 樹脂組成物並びにそれを用いたプリプレグおよび積層板 |
| JP2010138364A (ja) * | 2008-02-12 | 2010-06-24 | Mitsubishi Gas Chemical Co Inc | 樹脂組成物、プリプレグおよび金属箔張り積層板 |
| JP2010138366A (ja) * | 2008-04-01 | 2010-06-24 | Mitsubishi Gas Chemical Co Inc | 樹脂組成物、プリプレグおよび金属箔張り積層板 |
| JP2010174242A (ja) | 2009-12-28 | 2010-08-12 | Sumitomo Bakelite Co Ltd | ビフェニルアラルキル型シアン酸エステル樹脂、並びにビフェニルアラルキル型シアン酸エステル樹脂を含む樹脂組成物、及び、当該樹脂組成物を用いてなるプリプレグ、積層板、樹脂シート、多層プリント配線板、並びに半導体装置 |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4912172A (en) * | 1987-09-03 | 1990-03-27 | General Electric Company | Compositions comprising polyphenylene ethers, polyepoxides and aluminum or zinc diketone salt |
| CA1336845C (en) * | 1987-09-03 | 1995-08-29 | John Edward Hallgren | Curable polymer compositions comprising polyphenylene ethers and polyepoxides |
| WO2001042359A1 (en) * | 1999-12-13 | 2001-06-14 | Dow Global Technologies Inc. | Flame retardant phosphorus element-containing epoxy resin compositions |
| US7413791B2 (en) * | 2003-01-28 | 2008-08-19 | Matsushita Electric Works, Ltd. | Poly (phenylene ether) resin composition, prepreg, and laminated sheet |
| JPWO2007097209A1 (ja) * | 2006-02-22 | 2009-07-09 | 味の素株式会社 | エポキシ樹脂組成物 |
| US7919567B2 (en) * | 2006-06-07 | 2011-04-05 | Dow Global Technologies Llc | Oligomeric halogenated chain extenders for preparing epoxy resins |
| JP5104507B2 (ja) * | 2007-04-26 | 2012-12-19 | 日立化成工業株式会社 | セミipn型複合体の熱硬化性樹脂を含有する樹脂ワニスの製造方法、並びにこれを用いたプリント配線板用樹脂ワニス、プリプレグ及び金属張積層板 |
| JP2009027423A (ja) * | 2007-07-19 | 2009-02-05 | Sony Computer Entertainment Inc | 通信システム、通信装置、通信プログラム、通信プログラムを記憶したコンピュータ読み取り可能な記憶媒体 |
| JP5292783B2 (ja) * | 2007-11-27 | 2013-09-18 | 三菱瓦斯化学株式会社 | 硬化性樹脂組成物およびその硬化物 |
| JP2009179730A (ja) * | 2008-01-31 | 2009-08-13 | Mitsubishi Gas Chem Co Inc | 硬化性樹脂組成物およびプリプレグならびに銅張積層板 |
| JP5245496B2 (ja) * | 2008-03-31 | 2013-07-24 | Dic株式会社 | エポキシ樹脂組成物、及びその硬化物 |
| JP2011001411A (ja) * | 2009-06-17 | 2011-01-06 | Risho Kogyo Co Ltd | 高誘電率樹脂組成物を用いたプリプレグ、および銅張積層板 |
| JP5184480B2 (ja) * | 2009-09-29 | 2013-04-17 | パナソニック株式会社 | 樹脂組成物、樹脂組成物の製造方法、樹脂ワニス、プリプレグ、金属張積層板、及びプリント配線板 |
| TWI540170B (zh) * | 2009-12-14 | 2016-07-01 | Ajinomoto Kk | Resin composition |
| JP5408046B2 (ja) * | 2010-06-10 | 2014-02-05 | 味の素株式会社 | 樹脂組成物 |
-
2013
- 2013-03-21 EP EP13764176.7A patent/EP2829568B1/en active Active
- 2013-03-21 KR KR1020147024447A patent/KR20140138677A/ko not_active Ceased
- 2013-03-21 MY MYPI2014002538A patent/MY167826A/en unknown
- 2013-03-21 WO PCT/JP2013/058075 patent/WO2013141298A1/ja not_active Ceased
- 2013-03-21 US US14/381,784 patent/US20150044484A1/en not_active Abandoned
- 2013-03-21 CN CN201380012278.4A patent/CN104169343B/zh active Active
- 2013-03-21 JP JP2014506268A patent/JP6056849B2/ja active Active
- 2013-03-22 TW TW102110303A patent/TWI572649B/zh active
-
2016
- 2016-08-17 JP JP2016159839A patent/JP6299821B2/ja active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11124433A (ja) | 1997-10-22 | 1999-05-11 | Mitsubishi Gas Chem Co Inc | フェノールノボラック型シアン酸エステルプレポリマー |
| JP2005239767A (ja) | 2004-02-24 | 2005-09-08 | Matsushita Electric Works Ltd | 樹脂組成物並びにそれを用いたプリプレグおよび積層板 |
| JP2010138364A (ja) * | 2008-02-12 | 2010-06-24 | Mitsubishi Gas Chemical Co Inc | 樹脂組成物、プリプレグおよび金属箔張り積層板 |
| JP2010138366A (ja) * | 2008-04-01 | 2010-06-24 | Mitsubishi Gas Chemical Co Inc | 樹脂組成物、プリプレグおよび金属箔張り積層板 |
| JP2010174242A (ja) | 2009-12-28 | 2010-08-12 | Sumitomo Bakelite Co Ltd | ビフェニルアラルキル型シアン酸エステル樹脂、並びにビフェニルアラルキル型シアン酸エステル樹脂を含む樹脂組成物、及び、当該樹脂組成物を用いてなるプリプレグ、積層板、樹脂シート、多層プリント配線板、並びに半導体装置 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2829568A4 |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9051465B1 (en) | 2012-02-21 | 2015-06-09 | Park Electrochemical Corporation | Thermosetting resin composition containing a polyphenylene ether and a brominated fire retardant compound |
| US9243164B1 (en) | 2012-02-21 | 2016-01-26 | Park Electrochemical Corporation | Thermosetting resin composition containing a polyphenylene ether and a brominated fire retardant compound |
| JP2014240474A (ja) * | 2013-05-17 | 2014-12-25 | 三菱瓦斯化学株式会社 | 樹脂組成物、プリプレグ、樹脂シート、金属箔張積層板及びプリント配線板 |
| CN104629341A (zh) * | 2013-11-08 | 2015-05-20 | 中山台光电子材料有限公司 | 低介电树脂组合物,应用其的半固化胶片、覆铜箔基板、电路板 |
| JP2016042169A (ja) * | 2014-08-19 | 2016-03-31 | 三菱瓦斯化学株式会社 | 電子写真感光体 |
| JP2016044208A (ja) * | 2014-08-20 | 2016-04-04 | 日立化成株式会社 | モールドアンダーフィル用樹脂組成物及び電子部品装置 |
| WO2016038878A1 (ja) * | 2014-09-09 | 2016-03-17 | パナソニックIpマネジメント株式会社 | 硬化性組成物、プリプレグ、樹脂付き金属箔、金属張積層板、及びプリント配線板 |
| JP2016056367A (ja) * | 2014-09-09 | 2016-04-21 | パナソニックIpマネジメント株式会社 | 硬化性組成物、プリプレグ、樹脂付き金属箔、金属張積層板、及びプリント配線板 |
| CN106574111A (zh) * | 2014-09-09 | 2017-04-19 | 松下知识产权经营株式会社 | 固化性组合物、预浸料坯、带有树脂的金属箔、覆金属层叠板、以及印刷布线板 |
| US20170226302A1 (en) * | 2014-09-09 | 2017-08-10 | Panasonic Intellectual Property Management Co., Ltd. | Curable composition, prepreg, metal foil with resin, metal-clad laminate and printed wiring board |
| JP2018062568A (ja) * | 2016-10-12 | 2018-04-19 | 三菱瓦斯化学株式会社 | プリント配線板用樹脂組成物、プリプレグ、金属箔張積層板、積層樹脂シート、樹脂シート、及びプリント配線板 |
| WO2019167391A1 (ja) * | 2018-02-27 | 2019-09-06 | 京セラ株式会社 | プリプレグおよび回路基板用積層板 |
| US11457530B2 (en) | 2018-02-27 | 2022-09-27 | Kyocera Corporation | Prepreg and laminate for circuit board |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201348308A (zh) | 2013-12-01 |
| JP6056849B2 (ja) | 2017-01-11 |
| KR20140138677A (ko) | 2014-12-04 |
| JPWO2013141298A1 (ja) | 2015-08-03 |
| MY167826A (en) | 2018-09-26 |
| JP6299821B2 (ja) | 2018-03-28 |
| EP2829568A1 (en) | 2015-01-28 |
| CN104169343B (zh) | 2018-09-14 |
| TWI572649B (zh) | 2017-03-01 |
| JP2016191073A (ja) | 2016-11-10 |
| EP2829568A4 (en) | 2015-12-02 |
| US20150044484A1 (en) | 2015-02-12 |
| CN104169343A (zh) | 2014-11-26 |
| EP2829568B1 (en) | 2018-03-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6299821B2 (ja) | プリプレグ及び積層板 | |
| JP5846396B2 (ja) | 樹脂組成物、プリプレグ及び樹脂シート並びに金属箔張り積層板 | |
| JP6536565B2 (ja) | 樹脂組成物、プリプレグ、樹脂シート、金属箔張積層板及びプリント配線板 | |
| JP5369838B2 (ja) | 樹脂組成物、プリプレグおよび金属箔張り積層板 | |
| JP6168308B2 (ja) | 樹脂組成物、プリプレグ、金属箔張積層板及びプリント配線板 | |
| WO2019230945A1 (ja) | 樹脂組成物、プリプレグ、金属箔張積層板、樹脂シート及びプリント配線板 | |
| JP6010872B2 (ja) | 樹脂組成物、プリプレグ、樹脂シート及び金属箔張り積層板 | |
| JP2018131519A (ja) | 樹脂組成物、樹脂シート及びプリント配線板 | |
| JP6156075B2 (ja) | 樹脂組成物、プリプレグ、樹脂シート、金属箔張積層板及びプリント配線板 | |
| WO2013047041A1 (ja) | 樹脂組成物、プリプレグ及び金属箔張り積層板 | |
| WO2022124129A1 (ja) | 樹脂組成物、プリプレグ、樹脂シート、金属箔張積層板、及びプリント配線板 | |
| CN108148178B (zh) | 一种热固性树脂组合物 | |
| WO2022124130A1 (ja) | 銅箔張積層板及びプリント配線板 | |
| JP2017157783A (ja) | プリント配線基板 | |
| JP2021008634A (ja) | プリプレグ |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13764176 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2014506268 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14381784 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20147024447 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013764176 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |