WO2013166833A1 - 包含有人环氧酶及阿霉素或类阿霉素的药用组合物、其制备方法及在制备多种药物的应用 - Google Patents
包含有人环氧酶及阿霉素或类阿霉素的药用组合物、其制备方法及在制备多种药物的应用 Download PDFInfo
- Publication number
- WO2013166833A1 WO2013166833A1 PCT/CN2012/086605 CN2012086605W WO2013166833A1 WO 2013166833 A1 WO2013166833 A1 WO 2013166833A1 CN 2012086605 W CN2012086605 W CN 2012086605W WO 2013166833 A1 WO2013166833 A1 WO 2013166833A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- doxorubicin
- cell
- natural
- extract
- drug
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/43—Enzymes; Proenzymes; Derivatives thereof
- A61K38/44—Oxidoreductases (1)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7028—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages
- A61K31/7034—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin
- A61K31/704—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin attached to a condensed carbocyclic ring system, e.g. sennosides, thiocolchicosides, escin, daunorubicin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/53—Lamiaceae or Labiatae (Mint family), e.g. thyme, rosemary or lavender
- A61K36/537—Salvia (sage)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/88—Liliopsida (monocotyledons)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y114/00—Oxidoreductases acting on paired donors, with incorporation or reduction of molecular oxygen (1.14)
- C12Y114/99—Miscellaneous (1.14.99)
- C12Y114/99001—Prostaglandin-endoperoxide synthase (1.14.99.1), i.e. cyclooxygenase
Definitions
- composition comprising human cyclooxygenase and doxorubicin or doxorubicin, preparation method thereof and application thereof in preparing medicine
- the present invention relates to a pharmaceutical composition for extracting from a natural plant, in particular to a natural human cyclooxygenase
- composition of (COX-2), natural doxorubicin (atm) or natural doxorubicin material such a pharmaceutical combination containing natural human cyclooxygenase, natural doxorubicin or natural doxorubicin material
- Method for preparing the same and the use of the pharmaceutical composition containing the natural human epoxidase, the natural doxorubicin or the natural doxorubicin substance in the preparation of a medicament for treating nephritis, in the preparation of a treatment for cholecystitis and bile polyps
- Use in medicine in the preparation of a medicament for treating tumors, or in the preparation of an anti-inflammatory drug.
- RNA has a broad anti-tumor spectrum, and has a role in various tumors. It is a non-specific periodic drug and has a killing effect on various growth cycles. It is mainly suitable for acute leukemia and is effective for acute lymphocytic leukemia and leukemia. Malignant lymphoma can be used as the first drug to be used interchangeably.
- Breast cancer, _ ⁇ , lung cancer, bladder cancer and other various cancers have a certain effect, and are often used in combination with other 3 ⁇ 4 ⁇ .
- This product is a broad-spectrum anti-tumor drug that produces a wide range of biochemical effects on the body and has a strong cytotoxic effect.
- the mechanism of action is mainly that the product is embedded in DNA to inhibit the synthesis of nucleic acids.
- COX Cyclooxygenase
- PGHS rostagierinhypcroxidesynthase PGHS
- COX-1 Human cyclooxygenase
- COX-2 Human cyclooxygenase
- Natural human cyclooxygenase is a protein, also an inducible enzyme that acts as an enzyme to accelerate the production of certain chemical signals that are active in inflammation and pain. When the activity of COX-2 is inhibited, the pain is alleviated.
- COX-2 can produce arachidonic acid, produce a variety of prostaglandins, and participate in a variety of physiological and pathological processes in the body.
- the current consensus is that COX-2 can pass It promotes cell proliferation, inhibits apoptosis, promotes angiogenesis, and suppresses immune function, and participates in the occurrence and development of tumors.
- a pharmaceutical composition comprising natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin material, Method for preparing a pharmaceutical composition containing natural human epoxidase, natural doxorubicin or natural doxorubicin material, and the like containing natural human epoxidase, natural doxorubicin or natural doxorubicin.
- COX-2 natural human cyclooxygenase
- natural doxorubicin atm
- natural doxorubicin material Method for preparing a pharmaceutical composition containing natural human epoxidase, natural doxorubicin or natural doxorubicin material, and the like containing natural human epoxidase, natural doxorubicin or natural doxorubicin
- a pharmaceutical composition for the treatment of nephritis, for the preparation of a medicament for treating cholecystitis and bile polyp
- the invention provides a pharmaceutical composition prepared by medicinal natural plant rich in human cyclooxygenase (COX-2) and natural doxorubicin or doxorubicin, which can realize natural mold by using traditional Chinese medicine.
- COX-2 human cyclooxygenase
- COX-2 natural doxorubicin or doxorubicin
- a technical solution for accomplishing the above inventive task is a pharmaceutical composition
- a pharmaceutical composition comprising natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin material, characterized in that the composition
- the raw material is Qizhen Grass (or Guizhou Qizhen Grass).
- the "raw material of the composition” in the above technical solution means: a powder obtained by dehydrating, drying, or pulverizing the whole plant (color roots) of Qizheng grass; or fresh of the rare grass Or its extract.
- the extract of the exotic grass refers to one of the following forms of extract:
- the alcohol extract obtained by ethanol extraction is concentrated and dried to obtain a powder
- An extract obtained by extracting the whole plant of Qizhencao or Qizhencao decoction by an organic solvent obtained by extracting the whole plant of Qizhencao or Qizhencao decoction by an organic solvent
- the pharmaceutical composition containing natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin may also contain pharmaceuticals in addition to the raw material Qizhencao or its extract.
- Excipients allowed in the field.
- the exotic grass is a one-year or perennial herb, roots, roots erect or vines rising, stalks have rooting at nodes, solitary or clumped, stems blunt-square (square), grooved, densely short Fur.
- the seeds of the Qizhencao (tentative name) were deposited with the China Center for Type Culture Collection of Wuhan University on April 2, 2011, and the deposit number is CCTCC No: P201102 (see attached sheet 1 of the manual).
- the plant has applied to the United States for the right to new plant varieties; the acceptance number is: 12/215.016. US trademark acceptance number: 77633688.
- the plant variety has been submitted to the Ministry of Agriculture and the materials have been added.
- a technical solution for accomplishing the second invention task of the present application is a method for preparing a pharmaceutical composition containing the natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin material,
- COX-2 natural human cyclooxygenase
- atm natural doxorubicin
- natural doxorubicin material natural doxorubicin material
- Step (1) to obtain a powder obtained by dehydrating, drying and pulverizing the whole plant of Qizheng grass
- the step (2) or extracting the extract of the grass using one of the following steps:
- the solution for accomplishing the third invention task of the present application is: the above-mentioned pharmaceutical composition containing natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin material in the preparation of therapeutic nephritis and Application in medicine.
- COX-2 natural human cyclooxygenase
- atm natural doxorubicin
- doxorubicin material in the preparation of therapeutic nephritis and Application in medicine.
- the solution to accomplish the fourth inventive task of the present application is: The above pharmaceutical composition containing natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin material is prepared for the treatment of cholecystitis and The application of drugs for bile polyps.
- the solution to accomplish the fifth inventive task of the present application is: the above-mentioned pharmaceutical composition containing natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin substances in the preparation of a medicament for treating tumors Application in .
- the solution to accomplish the sixth inventive task of the present application is: the above pharmaceutical composition containing natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin material in the preparation of an anti-inflammatory drug application.
- COX-2 natural human cyclooxygenase
- atm natural doxorubicin
- doxorubicin material in the preparation of an anti-inflammatory drug application.
- the administration method is: oral tablets, capsules, oral liquid or syrup, oral steaming liquid, topical eye drops, topical eye ointment, external application of bruises Analgesic ointment, topical anti-inflammatory, hemostasis, stimulating, analgesic powder, do not rule out injection administration.
- the dosage to be administered is:
- the daily measurement of the adult is equivalent to the raw material of Guizhou Qizheng grass powder 5g-6g, and the recommended amount of this application is 5.4g. That is, three times a day, 1.8 g each time.
- the treatment period (treatment) is 7 days. It takes two courses to treat general inflammation; six courses are required to treat chronic inflammation. It usually takes 6 courses to treat nephritis, cholecystitis and tumors.
- the pharmaceutical composition of the present invention can rapidly treat inflammation, eliminate swelling, and relieve pain; it can also remove viruses, inhibit tumor cells, and activate the vital system of the body. Therefore, the pharmaceutical composition of the present invention is useful for treating various nephropathy such as chronic nephritis, acute cholecystitis (including bile reflux cholecystitis), human lymph, lung, liver, stomach, breast, fibroid, leukemia and the like.
- the pharmaceutical composition of the present invention can also be used for the treatment of trachoma and fire eyes.
- the pharmaceutical composition of the present invention can be used as a topical drug to rub, and can treat bruises, water and fire, and sore pain and pus.
- the fresh product of the raw material of the pharmaceutical composition of the invention, the external application of erosion can promote fracture healing.
- the pharmaceutical composition of the present invention also has a cosmetic effect and can be used for the development of a cosmetic product.
- a pharmaceutical composition containing natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin is used in the preparation of a medicament for treating nephritis
- the diagnosis of nephritis patients is as usual.
- Standards are diagnosed; standards such as "effective”, “effective”, and “healed” are also in accordance with traditional standards.
- the dose of the medicament of the present invention is: Adults three times a day, 1.8 g each time.
- the treatment is 7 days. When the treatment is more than 6 courses, the treatment results are compared as follows:
- Cure time 7-15 days £ When a pharmaceutical composition containing natural human cyclooxygenase (COX-2), natural doxorubicin (atm) or natural doxorubicin is used in the preparation of a medicament for treating cholecystitis, The diagnosis of patients with cholecystitis is diagnosed according to the usual criteria; the criteria for "effective", “effective", “healing”, etc. are also in accordance with traditional standards. The dose of the medicament of the present invention is: 1.8 g per adult three times a day. The treatment lasts for 7 days. When the treatment is more than 6 courses, the treatment results are compared as follows:
- the dose of the medicament of the present invention is: Adult three times a day, each time 1.8 go for 7 days. When the treatment is more than 8 courses, the treatment results are compared as follows:
- the extract of traditional Chinese medicine has obvious inhibitory effect on the proliferation of fine fetuses in A549 cells, and the inhibitory effect is drug concentration-dependent.
- the extract of traditional Chinese medicine has a significant inhibitory effect on cell proliferation in HT1080 cells, and the inhibitory effect is drug concentration.
- the extract of traditional Chinese medicine has a significant inhibitory effect on cell proliferation in Hela cells, and the inhibitory effect is drug concentration dependent.
- the extract of traditional Chinese medicine has a significant inhibitory effect on cell proliferation in MDA-MB-231 cells, and the inhibitory effect is drug-dependent.
- the extract of traditional Chinese medicine is made from 8 grams of dry medicine and 120 grams of medicinal juice.
- the concentration of 1 to 15 is 55.56ug/ml. It has obvious toxic effects on cells. Under the toxic effect, it migrates to the lower chamber due to the death of upper chamber cells or low activity. The number of cells is reduced. When the drug concentration is lower than 55.56 ug/ml, there is substantially no toxic effect on the cells. (Experimental test data attached).
- the invention provides a pharmaceutical composition prepared by medicinal natural plant rich in human cyclooxygenase (COX-2) and natural doxorubicin or doxorubicin, which can realize natural mold by using traditional Chinese medicine.
- COX-2 human cyclooxygenase
- COX-2 natural doxorubicin or doxorubicin
- Figure 1 is a curve showing the inhibition of proliferation and inhibition of A549 cell line in vitro by Chinese herbal extracts
- Figure 2 is a curve showing the inhibition of proliferation and inhibition of HT29 cell line in vitro by traditional Chinese medicine extract
- Figure 3 is a curve showing the inhibition of proliferation and inhibition of PC3 cell line in vitro by Chinese herbal extracts
- Figure 4 is a curve showing the inhibition of proliferation and inhibition of HT1080 cell line in vitro by traditional Chinese medicine extract
- Figure 5 is a curve showing the inhibition of proliferation and inhibition of Hela cell line in vitro by Chinese herbal extract
- Figure 6 is a curve showing the inhibition of proliferation and inhibition of MDA-MB-231 cell line in vitro by traditional Chinese medicine extract
- Figure 7 is a curve showing the inhibition of proliferation and inhibition of HepG2 cell line in vitro by traditional Chinese medicine extract
- Map Figure 9 is a graph showing the inhibition effect of the extract of traditional Chinese medicine on the migration ability of A549 cell line in vitro;
- Figure 10 is a graph showing the inhibition effect of the extract of traditional Chinese medicine on the migration ability of PC3 cell line in vitro;
- Figure 11 is a graph showing the inhibition effect of Chinese herbal extracts on the migration ability of HT1080 cell line in vitro;
- Figure 12 is a graph showing the inhibitory effect of Chinese herbal extracts on the migration ability of HeLa cell lines in vitro;
- Figure 13 is a graph showing the inhibitory effect of Chinese herbal extracts on the migration ability of MDA-MB-231 cells in vitro
- Figure 14 is a graph showing the inhibitory effect of Chinese herbal extracts on the migration ability of HepG2 cells in vitro;
- Figure 15 is a curve showing the migration ability of HT29 cell line in vitro
- Figure 16 is a real-time cell analyzer
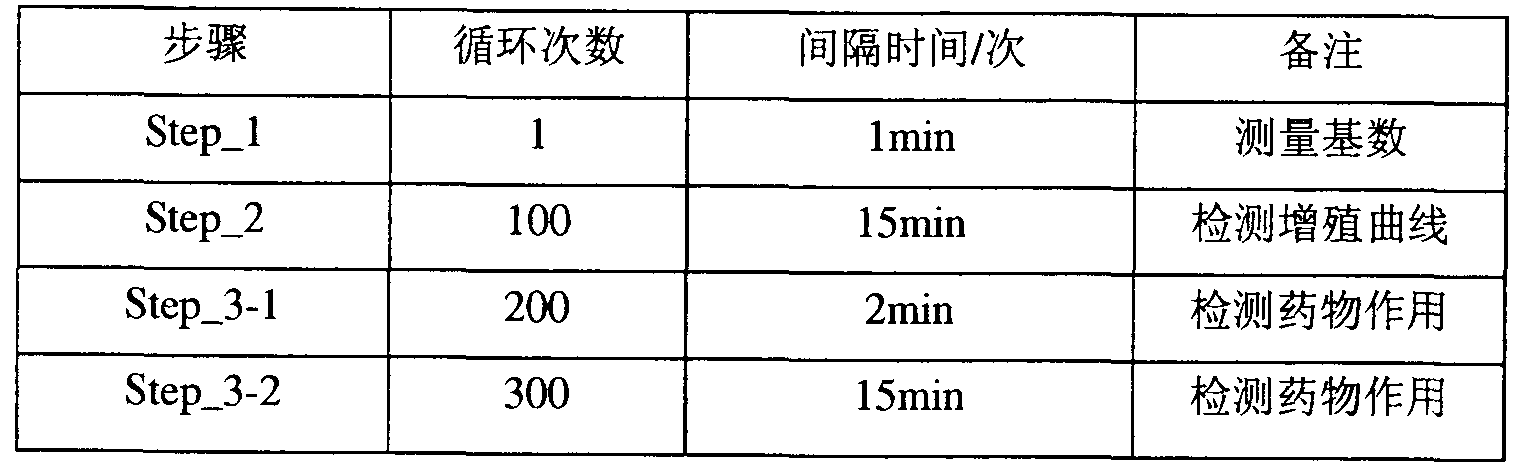
- Figure 17 and Figure 18 are measurement curves reflecting the beating characteristics of myocardial cells and their cell status
- Figure 19 shows the parameters and data analysis page in the cardiomyocyte model
- Figure 20 is a graph showing the effect of Chinese herbal extract on the activity of primary cardiomyocytes in neonatal rats;
- Figure 21 shows the changes in the beat curve of primary cardiomyocytes with time after dosing
- Figure 22 is a graph showing the effect of Chinese herbal extract on the beating rate
- Figure 23 is a graph showing the effect of Chinese herbal extract on the amplitude of the beat (Amplitude);
- Figure 24 shows the IC50 curve of Chinese herbal extracts affecting the frequency and amplitude of primary cardiomyocytes in neonatal rats.
- Example 1 A mixed pharmaceutical composition containing a natural human cyclooxygenase (COX-2), a natural doxorubicin (atm) or a natural doxorubicin substance, a preparation method and a clinical use of a tablet.
- COX-2 a natural human cyclooxygenase
- atm a natural doxorubicin
- doxorubicin substance a preparation method and a clinical use of a tablet.
- the extract of the preparation will be cleaned and sterilized, and the ratio of 10 kg of water of 1 kg of medicinal material is extracted by steam distillation to obtain volatile oil and distillate, and the volatile oil is reserved.
- the evaporating liquid is filtered for a period of more than 24 hours, and the supernatant is filtered.
- the ethanol is recovered to a non-alcoholic mash, concentrated into a thick paste, dried, pulverized, and the extract powder is taken, and the volatile oil and the precipitate are uniformly sprayed on the extract powder, and the micropowder is added with calcium citrate, lactose, de-lighted starch, and stearic acid.
- Pharmaceutical excipients such as magnesium sulfate are prepared in the corresponding dosage forms.
- the method of preparing the extracting powder tablet the dry extract is first pulverized into a fine powder, passed through a 5-6 mesh sieve, wetted by ethanol, and tableted.
- Spray rotary granulation method adding calcium citrate, lactose, luminescent starch, and placing the dry extract fine powder in a coating pan, and also spraying the mist into the mist while rotating, gradually granulating the wet powder.
- Powder powder preparation method medicinal materials are washed, sterilized, dried, and dried, processed micron Chinese medicine powder, particle size 10um (l-20um) over 300 mesh sieve, can be made into tablets;
- Capsule preparation method The ratio of 1 kg of medicine to 10 kg of water 3, plastic film preparation method: medicinal material processing: net, processing, purification, sterilization, crushing particle size 75um4.1 over 200um mesh sieve, filling capsules;
- Solvent extraction method Qizhen herbal materials, appropriate crushing, using 1 kg of medicinal materials 10 kg of water ratio, using impregnation method, percolation method, decoction method; optional ester esterity: petroleum ether. benzene. >ether>acetic acid B Ester>Acetone>Ethanol>Methanol>Water (hydrophilic), etc., organic solvent during extraction.
- the plant was screened and tested in Shanghai Testing and Screening Center of Shanghai New Drug Screening Center in 2006. The results showed that the plant contained natural human cyclooxygenase-2 ( COX -2) activity and immune biological activity, anti-inflammatory treatment. Has a very good effect. At the same time, it has a good inhibitory effect on lymphatic 1 ⁇ B cells and has a good application prospect. Attachment - 1 (Saving proof 1 page)
- the viability of the culture was monitored by the collection on March 26, 2011 and the result was survival.
- Cyclooxygenase produces prostaglandins and oxygen free radicals (ROS) by metabolizing arachidonic acid (AA). Cyclooxygenase
- C0X2 Cyclooxygenase 1
- C0X1 mainly regulates physiological functions.
- the xCELLigence real-time cell analysis system was used to detect the inhibition of cell proliferation by Chinese herbal extracts, and to detect the 1C50 of drug inhibition of cell proliferation.
- the cells used were A549, HT1 080, Hel. a, MDA-MB-231, HT29, HepG2 and PC3 cells.
- This experiment uses the xCELLigence real-time cell analysis system jointly developed by Essen and Roche.
- DMEM medium Item No. SH30022. 01B, Hyc] one (China).
- MEM medium Article number SH30024. 01B, Hyclone (China).
- F12 medium Item No. SH30526. 01 , Hyclone (China).
- MyCOA's 5A medium Item No. GNM 16600, Gino (China).
- PBS Item No. SH30256. 01B, Hyclone (China).
- Centrifuge tube article number 430791 , Corningo
- A549 non-small cell lung cancer cell line
- PC3 human prostate cancer cell line
- HT1080 human fibrosarcoma cell line
- Hela human cervical cancer cell line
- MDA MB-231 human breast cancer cell line
- HepG2 human liver cancer cell line
- the extract of Chinese medicine is 20 grams of medicine to get 125ml of medicine, the concentration is 160tng I ml.
- Figure 1 shows the effect of Chinese herbal extract on the proliferation and inhibition of A549 cell proliferation in vitro.
- A is a real-time dynamic map of cell growth and drug action.
- the abscissa indicates cell proliferation and drug action time;
- the ordinate indicates standardized cell index (NCI).
- NCI standardized cell index
- the cell index indicates the state of cell growth. The higher the value, the more living cells are reflected.
- the arrow indicates the dosing time, and the dosing concentration is shown in the figure.
- the figure shows the drug concentration-dependent dynamic response curve of the cells under the action of the Chinese herbal extract, and controll is a negative control, showing a normal cell growth dynamic curve.
- B is the IC50 value and IC50 fit curve of the drug at 12, 24, and 48 hours after the addition of the Chinese herbal extract to the A549 cells automatically calculated by the real-time cell analysis system. It can be seen from the figure that the extract of traditional Chinese medicine has a significant inhibitory effect on cell proliferation in A549 cells, and the inhibitory effect is drug-dependent.
- Figure 2 shows that the Chinese herbal extract inhibits the proliferation and inhibition of HT29 cell line in vitro.
- A is a real-time dynamic map of cell growth and drug action.
- B is the IC50 value and IC50 fit curve of the drug at 48 and 72 hours after the addition of the traditional Chinese medicine extract to the HT29 cells calculated by the real-time cell analysis system. It can be seen from the figure that the extract of traditional Chinese medicine has obvious inhibitory effect on cell proliferation of HT29 cells, and the inhibitory effect is drug concentration-dependent.
- FIG. 3 Chinese herbal extracts inhibit the proliferation of PC3 cell lines in vitro and observe the inhibitory effect.
- A is a real-time dynamic map of cell growth and drug action.
- B is the IC50 value and IC50 fit curve of the drug at 24, 48 and 72 hours after the addition of the traditional Chinese medicine extract to PC3 cells calculated by the real-time cell analysis system. It can be seen from the figure that the extract of traditional Chinese medicine has obvious inhibitory effect on cell proliferation in PC3 cells, and the inhibitory effect is drug concentration-dependent.
- Figure 4 shows that the Chinese herbal extract inhibits the proliferation and inhibition of HT1080 cell line in vitro.
- A is a real-time dynamic map of cell growth and drug action.
- B is the IC50 value and IC50 fit curve of the drug at 12, 24, and 48 hours after the addition of the Chinese herbal extract to the HT1080 cells, which was automatically detected by the real-time cell analysis system. It can be seen from the figure that the Chinese herbal extract has a significant inhibitory effect on cell proliferation in HT1080 cells, and the inhibitory effect is drug-dependent.
- Figure 5 shows the effect of Chinese herbal extract on the inhibition of proliferation and inhibition of Hda cell line in vitro.
- A is a real-time dynamic map of cell growth and drug action.
- B is the IC50 value and IC50 fit curve of the drug at 12, 24, and 48 hours after the addition of the Chinese herbal extract to the Hda cells automatically calculated by the real-time cell analysis system. It can be seen from the figure that the extract of traditional Chinese medicine has obvious inhibitory effect on cell proliferation of Hela cells, and the inhibitory effect is drug concentration dependent.
- Figure 6 shows that the Chinese herbal extract inhibits the proliferation and inhibition of MDA-MB-231 cell line in vitro.
- A is a real-time dynamic map of cell growth and drug action.
- B is the IC50 value and IC50 fit curve of the drug at 24, 48 and 72 hours after the addition of the Chinese herbal extract to the MDA-MB-231 cells, which was automatically detected by the real-time cell analysis system. It can be seen from the figure that the extract of traditional Chinese medicine has obvious inhibitory effect on cell proliferation of MDA-MB-231 cells, and the inhibitory effect is drug-dependent.
- FIG. 7 Chinese herbal extracts inhibited the proliferation and inhibition of HepG2 cell line in vitro.
- A is a real-time dynamic map of cell growth and drug action.
- B is the IC50 value and IC50 fit curve of the drug at 48 and 72 hours after the addition of the Chinese herbal extract to HepG2 cells, which was automatically detected by the real-time cell analysis system. It can be seen from the figure that the Chinese herbal extract has a significant inhibitory effect on cell proliferation in HepG2 cells, and the inhibitory effect is drug-dependent.
- the extract of traditional Chinese medicine has inhibitory effect on the proliferation of the selected 7 cell lines, showing a good concentration gradient, which has stronger effect on HT1080, HeLa, MDA-MB-231 and A549 cells, and on HepG2 and HT29.
- the role of PC3 cells is weak.
- Figure 8 Compound specificity map.
- the real-time impedance data obtained from the xCELLigence system yields compound-specific profiles that depend on the biological mechanism of action of the compound.
- specific compounds can be generated by adding compounds with different biological mechanisms of action and target specificity. For compounds that do not have a specific effect, these maps can be used to set some verifiable hypotheses in a predictive manner.
- the xCELLigence real-time cell analysis system was used to detect the inhibition of cell migration by a Chinese herbal extract provided by Guizhou traditional Chinese medicine.
- the cells used were A549, PC3, HT1080, Hela, MDA-MB-23 HepG2.
- This experiment uses the xCELLigence DP real-time cell analysis system jointly developed by Essen and Roche.
- DMEM medium Item No. SH30022.08B, Hyclone (China).
- MEM medium Item No. SH30024.01B, Hyclone (China).
- F12 medium Catalog No. SH30526.01, Hyclone (China). Serum: Cat. No. 16000-044, Gibco (USA).
- Centrifuge tube Item No. 430791, Corning.
- A549 non-small cell lung cancer cell line
- PC3 human prostate cell line
- HT1080 human fibrosarcoma cell line
- Hela human cervical cancer cell line
- MDA-MB-231 human breast cancer cell line
- HepG2 human liver cancer cell line
- the Chinese herbal extract is 20 g of the drug to obtain 125 ml of the drug juice, and the concentration is 160 mg/ml.
- a drug containing 10% serum medium was added according to Layout, and a drug prepared in serum-free medium was added to the upper chamber, and the baseline was detected on the DP system.
- Cells that were resuspended in serum-free medium and had been incubated with each compound for 1 h were then added to the upper chamber and detected on DP for 24 h to 48 h.
- a proliferation control of the cells was set up in parallel on the 96x E-plate.
- Baseline was tested on SP by adding a drug containing 10% serum medium according to Layout. Cells that had been resuspended in 10% serum medium and had been incubated with each compound for 1 h were then added and assayed on SP for 24h-48h.
- the acquisition signal settings are as follows:
- Fig. 9 shows the inhibitory effect of Chinese herbal extracts on the migration ability of A549 cells in vitro.
- the left panel shows the drug-inhibiting cell migration curve
- the right panel shows the real-time dynamic map of the effects of drugs on cell proliferation in parallel.
- the abscissa indicates cell culture and drug action time
- the ordinate indicates standardized cell index (NCI).
- NCI standardized cell index
- the cell index indicates the state of cell growth. In cell migration experiments, the higher the value, the greater the number of cells that migrate to the lower chamber; in cell proliferation experiments, the higher the value, the more living cells. Dosing concentrations are shown in the figure.
- the left panel shows the response curve of the drug concentration-dependent cell migration ability of the cells under the action of the traditional Chinese medicine extract
- Control is a negative control, showing the normal cell migration dynamic curve.
- the picture on the right is the concentration-dependent cell proliferation inhibition of cells under the action of traditional Chinese medicine extract.
- Control is a negative control, showing the normal fine Hangzhou Qualcomm New Drug Screening Center Essen Bio
- the concentration of the added drug is higher than 55.56 ug/ml, it has obvious toxic effects on the cells. Under the toxic effect, the number of cells migrating to the lower chamber is reduced due to the death or activity of the upper chamber cells.
- the drug concentration is lower than 55.56 ug/ml, there is almost no toxic effect on the cells, and the migration of the cells is also inhibited, and when the inhibition is drug-dependent, the drug has a certain inhibition on the migration of A549 cells. effect.
- Figure 10 shows the inhibitory effect of Chinese herbal extracts on the migration ability of PC3 cell lines in vitro.
- the left panel shows the drug-inhibiting cell migration curve, and the right panel shows the real-time dynamic map of the effect of the drug on cell proliferation in parallel. Dosing concentrations are shown in the figure.
- the left panel shows the response curve of the drug concentration-dependent cell migration ability of the cells under the action of the Chinese herbal extract, and Control is a negative control, showing a normal cell migration dynamic curve.
- Control is a negative control, showing a normal cell proliferation dynamic curve.
- Fig. 11 shows the inhibitory effect of Chinese herbal extract on the migration ability of HT1080 cell line in vitro.
- the left panel shows the drug-inhibiting cell migration curve
- the right panel shows the real-time dynamic map of the effects of drugs on cell proliferation in parallel. Dosing concentrations are shown in the figure.
- the left panel shows the response curve of the drug concentration-dependent cell migration ability of the cells under the action of the Chinese herbal extract, and Control is a negative control, showing a normal cell migration dynamic curve.
- Control is a negative control, showing a normal cell proliferation dynamic curve.
- the concentration of the added drug is higher than 18.52 ug/ml, it has obvious toxic effects on the cells. Under the toxic effect, the number of cells migrating to the lower chamber is reduced due to the death or activity of the upper chamber cells.
- the drug concentration is lower than or equal to 18.52 ug/ml, there is almost no toxic effect on the cells, and the migration of the cells is also inhibited, and when the inhibition is drug concentration dependent, the drug has a certain migration to A549 cells. Inhibition.
- Figure 12 shows the inhibitory effect of Chinese herbal extracts on the migration ability of HeLa cell lines in vitro.
- the left panel shows the drug-inhibiting cell migration curve, and the right panel shows the real-time dynamic map of the effects of drugs on cell proliferation in parallel. Dosing concentrations are shown in the figure.
- the left panel shows the response curve of the drug concentration-dependent cell migration ability of the cells under the action of the Chinese herbal extract, and Control is a negative control, showing a normal cell migration dynamic curve.
- Control is a negative control, showing a normal cell proliferation dynamic curve.
- the added drug has a significant toxic effect on the cells. Under this toxic effect, the number of cells migrating to the lower chamber is reduced due to the death or activity of the upper chamber cells. The drug has no significant inhibitory effect on cell migration.
- Figure 13 shows the inhibitory effect of Chinese herbal extracts on the migration ability of MDA-MB-231 cells in vitro.
- the left panel shows the drug-inhibiting cell migration curve
- the right panel shows the real-time dynamic map of the effects of drugs on cell proliferation in parallel. Dosing concentrations are shown in the figure.
- the left panel shows the response curve of the drug concentration-dependent cell migration ability of the cells under the action of the Chinese herbal extract, and Control is a negative control, showing a normal cell migration dynamic curve.
- Control is a negative control, showing a normal cell proliferation dynamic curve.
- the addition of drugs had a certain stimulating effect on the migration of MDA-MB-231 cells.
- the drug concentration is lower than 0.15 mg/ml, there is no obvious toxic effect on the cells.
- the higher the amount of the drug added the stronger the stimulation effect on cell migration.
- the concentration of the added drug is higher than 0.4 mg/ml, the cells have a relatively obvious toxic effect. Under the toxic effect, the number of cells migrating to the lower chamber is reduced due to the death or activity of the upper chamber cells.
- Fig. 14 shows the inhibitory effect of the Chinese herbal extract on the migration ability of HepG2 cell line in vitro.
- the left picture shows the drug inhibition cell migration curve
- the right picture shows A real-time dynamic map of the effects of drugs on cell proliferation in parallel. Dosing concentrations are shown in the figure.
- the left panel shows the response curve of the drug concentration-dependent cell migration ability of the cells under the action of the traditional Chinese medicine extract.
- Control is a negative control, showing the normal cell migration dynamic curve.
- Control is a negative control, showing a normal cell proliferation dynamic curve.
- the added drug has a significant toxic effect on the cells. Under this toxic effect, the number of cells migrating to the lower chamber is reduced due to the death or activity of the upper chamber cells. The drug has no significant inhibitory effect on cell migration.
- Figure 15 Detection of migration ability of HT29 cell line in vitro. As shown in the figure, when the cell concentrations were 80k/well, 40k/well, 20k/well, and l Ok/well, HT29 had no obvious cell migration ability, so it was impossible to detect the inhibition of cell migration ability.
- the Chinese herbal extract has obvious toxic effects on HeLa and HepG2 cells. Under the toxic effect, the number of cells migrating to the lower chamber is reduced due to the death or activity of the upper chamber cells. At low concentrations, the drug did not significantly inhibit cell migration. The drug has no inhibitory effect on cell migration.
- a real-time cardiomyocyte analysis system was used to detect and evaluate the drug heart safety of traditional Chinese medicine extracts provided by Guizhou traditional Chinese medicine on primary neonatal rat cardiomyocytes.
- the parameters used in this experiment were time-dependent in vitro myocardial cell beating frequency, myocardial cell beat amplitude, and myocardial cell beat rhythm and wave pattern.
- Essen Bio is a high-tech biotechnology company dedicated to the development and manufacture of our own proprietary impedance-based real-time cell detectors.
- Essen's innovative micro-electrode sensor mounted on the bottom of the microplate can be used for real-time, label-free cell detection with high throughput, high accuracy, high sensitivity and quantitative detection.
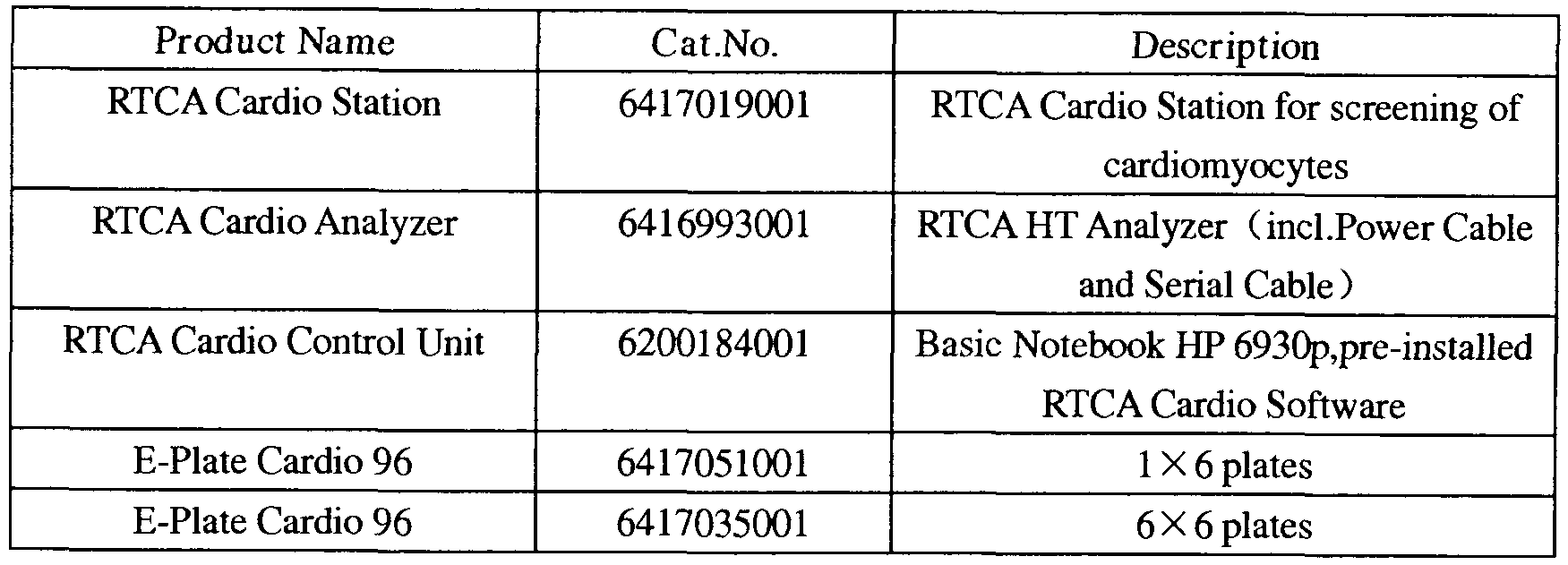
- the real-time cardiomyocyte system (XCELLigence RTCA Cardio System) developed by Essen Bio and Roche can dynamically monitor the excitatory contraction coupling reaction of cardiomyocytes, and detect and record the electrical signals of cardiomyocytes in real-time short-term/long-term time.
- Real-time monitoring can be performed by a real-time cardiomyocyte analysis system when the compound acts on cardiomyocytes to affect cell morphology and activity, or to cause changes in the degree of openness of the relevant channels, or to cause changes in contractile excitatory conductance.
- the system can be used to screen for drugs that affect changes in contractile excitatory conductance in cardiomyocytes, including ion channel and non-ion channel target drugs.
- Essen uses mouse embryonic stem cell-induced cardiomyocytes (CorAt, Axiogenesis) and human iPS-induced cardiomyocytes to validate more than 50 known compounds, and later supplemented with primary rat cardiomyocytes. .
- the results show that the real-time cardiomyocyte analysis system reliably and quantitatively detects the effects of arrhythmia compounds and non-ERG channels and voltage-gated calcium channel compounds on cardiomyocytes. Therefore, the system can be used as a tool for cardiotoxicity screening of compounds in early drug development, for high-throughput screening and drug toxicity studies.
- Work content :
- the goal of this project is to use an impedance detection system to perform drug heart safety testing and evaluation, to evaluate and identify the compounds caused by the original The ability to change the beating rate of rat cardiomyocytes.
- the real-time cardiomyocyte analysis system (xCELLigence RTCA Cardio System) continues Eisen's existing proprietary cell-based microelectronic impedance detection system (including RTCA SP, MP, DP system) for functional detection of myocardium Cellular beating and testing for drug cardiomyocyte cytotoxicity.
- RTCA SP RTCA SP
- MP MP
- DP DP system
- RTCA cardiomyocyte analysis system can also detect myocardial cells for excitement. The cells formed by contraction coupling are beating.
- the RTCA cardiomyocyte analysis system consists of the following components (see figure below):
- the cells were seeded into a 96-well cardiomyocyte assay plate to attach the cells to the surface of the electronic sensor.
- the RTCA test bench was placed in a carbon dioxide cell incubator (5% CO 2 , 37 ° C), and the cardiomyocyte test plate was placed on the RTCA test bench and connected to the analyzer and real-time cell analysis system console.
- the electrical impedance signal of the sensor surface can be detected in real time through the test bench and the analyzer.
- the measured electrical impedance signal is analyzed by RTCA real-time cell analysis system console and Cardio software to provide real-time quantitative information on the biological state of the cell, including cells. Information on quantity, vigor, morphology, and changes in cytoskeletal dynamics.
- Primary rat cardiomyocyte primary rat cardiomyocyte model can be used for many myocardial cell morphology, biochemistry, electrophysiology and pharmacology research. This model is a mature way to study drug transport, toxicity, and electrophysiological properties. Ten rats in the newborn 24 hours, 75% of the ethanol was disinfected and cut along the sternum, taking care to avoid cutting the digestive tract to prevent contamination.
- tissue block of the size is placed in a small glass bottle, and 1.5 ml of 0.07% trypsin and 0.07% collagenase I are added for digestion, and the digested supernatant is collected, and the appropriate amount (equal volume with the supernatant) is included.
- the DMEM medium of 10% fetal bovine serum terminates the action of the digestive enzyme, and the above digestion step is repeated until the tissue block disappears.
- the undigested tissue was filtered through a 200-mesh aperture mesh sieve.
- the supernatant was centrifuged for 5 min (800 r/min), the supernatant was discarded, and the precipitated cells were blown off with DMEM medium containing 10% fetal bovine serum, and inoculated into the culture flask.
- Carbon dioxide cell incubator (5% CO2, Hangzhou Qualcomm New Drug Screening Center Essen Bio
- the cells were statically cultured for 60 min (differential adherence), and the unattached cells were collected and inoculated into a gelatin-treated 96-well cardiomyocyte assay plate at a density of 15,000 cells per well. Detection is performed on the system.
- This experiment uses a real-time cardiomyocyte analysis system jointly developed by Essen and Roche.
- DMEM medium Item No. SH30022.08B, Hyclone (China).
- Collagenase Cat. No. 17101-015, Gibco (USA).
- HBSS solution 14175, Gibco (USA).
- the Chinese herbal extract is 20 g of the drug to obtain 125 ml of the drug juice, and the concentration is 160 mg/ml. During the experiment, it was diluted with the medium according to the actual concentration required, and then added to the cells.
- the primary neonatal rat cardiomyocytes were cultured, and 17000 cells were inoculated per well. After inoculation, they were placed in the real-time cardiomyocyte analysis system according to the following steps and parameters (see the acquisition signal setting table), and 48 hours. After changing the solution, the solution was changed for 2 hours, and after the cell signal was stabilized, it was added to the cell well according to the corresponding concentration of the compound. After the drug is added, the cell resetting detection system is used to collect the myocardial cell beating signal in real time. Two or more duplicate wells are used per compound.
- Each cell beat corresponds to the excitation and contraction of the muscle cells of the cardiomyocytes.
- the jitter includes positively occurring positive peaks (+P) and negative peaks (-P).
- the values of positive and negative peaks per unit time reflect the beating characteristics of cardiomyocytes and Cell state.
- the number of positive peaks in the Sweep Duration is m (+P1, +P2, +P3, +PmX
- the number of negative peaks is n (-P1, -P2, -P3, ..., - ⁇ , ⁇
- the time between a positive and negative peak is defined as the Beatins Period.
- the rising and falling processes of the jitter are described by parameters such as Rising Tine, Falling Time ⁇ IBD 50 , Rising Slope, and Falling Slope.
- the RTCA cardiomyocyte analysis software provides 12 parameters for analyzing the beating properties of cardiomyocytes. In this report, select the applicable parameters Hangzhou Qualcomm New Drug Screening Center Aisen Bio
- Ed NP-Negative Peak Standardized jitter amplitude is the beat amplitude of the selected time point Amplitude The ratio of the degree to the beat amplitude of the normalized time point.
- the Chinese herbal extract had an effect on cell viability only at a concentration of 16 mg/ml, and other concentrations had no effect on cell viability.
- the effect of the extract on myocardial cell beating is: There is no significant effect on the frequency, amplitude and rhythm of rat primary ventricular myocytes in a short period of time. After a long period of action, it has a certain cardiotoxic effect on ventricular myocytes. Especially at high concentration (16mg/ml), it can cause myocardial cell parameters after 2 hours of action.
- the low concentration of the drug (5.33 and lJ8mg/ml) caused a slight decrease in the frequency of myocardial cell beating and increased in the interval of 2-12 hours after dosing, and the beating returned to normal at 12 hours.
- the drug concentration below 0.59 mg/ml had no significant effect on myocardial cell beating.
- Chinese herbal extract (H20 ctrl, blank et, 7.32ug/ml, 21.95ug/ml, 65.84ug/ml, 0.20mg/ml,
- the above figure shows the real-time dynamic map of cell growth and drug action.
- the abscissa indicates cell culture and drug action time;
- the ordinate indicates standardized cell index (NCI).
- NCI standardized cell index
- the cell index indicates the state of cell growth. The higher the value, the more living cells are reflected.
- the arrow refers to the dosing period, and the dosing concentration is shown in the figure.
- the figure shows the drug concentration-dependent dynamic response curve of the cells under the action of the drug, and Ctrl is a negative control, showing a normal cell growth dynamic curve. (Note: H20 Ctrl control well added 10% (the same amount as the highest concentration of drug stock) H20)
- the figure shows the dose-effect relationship between drug concentration and time-dependent change of traditional Chinese medicine extracts of myocardial cell beat frequency.
- the parameter used is the Normalized Beating Rate (NBR).
- the parameter calculation method is the ratio of the beating frequency at the selected time point to the beating frequency of the normalized time point (Omin).
- Figure A shows the change of cell beating frequency within 1 h of dosing
- Figure B shows the change of cell beating frequency between lOmin before dosing and 24 h after dosing.
- the cell beating frequency is consistent within lOmin before dosing and the homogeneity is good.
- the drug had no obvious effect on cell beating.
- the high-concentration drug caused the cell beating frequency to decrease, and the jitter completely stopped after 6 hr. 5.
- 33ing/rnl drug caused a slight decrease in the beating frequency in a period of time, drug concentration Less than 1.78 mg/ml had no significant effect on cell beating. It indicates that the high concentration of the drug has a significant effect on the heartbeat, and the low concentration has no serious effect on the heartbeat.
- This figure shows the drug concentration and time-dependent changes in the amplitude of myocardial cell pulsation.
- the parameter used is the normalized runout amplitude (Normal ized
- the parameter calculation method is to select the ratio of the jitter amplitude at the time point to the jitter frequency of the normalized time point (Omin).
- Figure A shows the change of cell beating amplitude within 1 h of dosing
- Figure B shows the change of cell beating amplitude between lOmin before dosing and 24 h after dosing.
- the cell beating amplitude is stable within 10 min before dosing, and the drug has no significant effect on the cell beating amplitude in the short-term after dosing.
- the high concentration of 16 mg/ml drug causes the cell beating frequency to decrease, and the beating completely stops after 6 hr. 5.
- 33mg/ml drug caused a slight decrease in the frequency of beating, and the drug concentration was less than 1.78mg/ml had no significant effect on cell beating. It indicates that the high concentration of the drug has a significant effect on the heartbeat, and the low concentration has no serious effect on the heartbeat.
- the three time point IC50 values (beating rate/Amplitude) were calculated according to the cell beat frequency and amplitude. 5.
- the drug rapidly affects cell viability at high concentrations (16 mg/ml), leading to cell death.
- the Chinese medicine affects the frequency and amplitude of myocardial cell beating in primary neonatal rats in a concentration-dependent manner. High concentration (16mg/ml) will cause the cells to stop beating after about 6hr ; while medium concentration (5.33mg Ail ; 1.78mg/ml) can cause the cells to jump slightly slower after 6hr, slightly increasing the amplitude. . It indicates that the high concentration of the drug has a significant effect on the heartbeat, and the low concentration has no serious effect on the heartbeat.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Mycology (AREA)
- Botany (AREA)
- Microbiology (AREA)
- Gastroenterology & Hepatology (AREA)
- Medical Informatics (AREA)
- Alternative & Traditional Medicine (AREA)
- Biotechnology (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Immunology (AREA)
- Rheumatology (AREA)
- Pain & Pain Management (AREA)
- Urology & Nephrology (AREA)
- Medicines Containing Plant Substances (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2012379443A AU2012379443A1 (en) | 2012-05-10 | 2012-12-13 | Pharmaceutical composition containing human cyclooxygenase and doxorubicin or doxorubicin analogue, preparation method therefor and use thereof in preparing drugs |
| KR20147034747A KR20150020192A (ko) | 2012-05-10 | 2012-12-13 | 인간 사이클로옥시게나아제 및 독소루비신 또는 독소루비신 유사체를 함유한 약제 조성물, 이의 제조방법, 및 약물을 제조하는데 이의 용도 |
| US14/397,860 US20150118214A1 (en) | 2012-05-10 | 2012-12-13 | Pharmaceutical composition containing human cyclooxygenase and doxorubicin or doxorubicin analogue, preparation method therefor and use thereof in preparing drugs |
| JP2015510608A JP2015515993A (ja) | 2012-05-10 | 2012-12-13 | ヒトシクロオキシゲナーゼ及びドキソルビシン又はドキソルビシン類を含有する医薬組成物、その調製方法及び多種薬物の製造における使用 |
| EP12876488.3A EP2851079A4 (en) | 2012-05-10 | 2012-12-13 | PHARMACEUTICAL COMPOSITION WITH HUMAN CYCLOOXYGENASE AND DOXORUBICIN OR DOXORUBICIN ANALOGON, METHOD OF MANUFACTURING THEREOF, AND USE IN THE PRODUCTION OF MEDICAMENTS |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201210142501.4A CN102961440B (zh) | 2012-05-10 | 2012-05-10 | 含有人环氧酶及天然阿霉素或类阿霉素的药用组合物、其制备方法及在制备多种药物的应用 |
| CN201210142501.4 | 2012-05-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013166833A1 true WO2013166833A1 (zh) | 2013-11-14 |
Family
ID=47792059
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2012/086605 Ceased WO2013166833A1 (zh) | 2012-05-10 | 2012-12-13 | 包含有人环氧酶及阿霉素或类阿霉素的药用组合物、其制备方法及在制备多种药物的应用 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20150118214A1 (zh) |
| EP (1) | EP2851079A4 (zh) |
| JP (1) | JP2015515993A (zh) |
| KR (1) | KR20150020192A (zh) |
| CN (1) | CN102961440B (zh) |
| AU (1) | AU2012379443A1 (zh) |
| WO (1) | WO2013166833A1 (zh) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108159308A (zh) * | 2017-11-24 | 2018-06-15 | 花泽燕 | 一种胆囊息肉专用中药及其制备方法 |
| CN108157727A (zh) * | 2017-11-30 | 2018-06-15 | 慈中华 | 用于保持平和体质的固体饮料及其加工方法 |
| CN117721171A (zh) * | 2024-01-04 | 2024-03-19 | 中国人民解放军总医院第一医学中心 | 在细胞增殖过程中的氯化钴的加入方法 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1174065A (zh) * | 1997-07-16 | 1998-02-25 | 迟经惠 | 障碍贫血针剂 |
| CN101181349B (zh) * | 2007-11-13 | 2012-02-22 | 中国人民解放军第二军医大学 | 甘西鼠尾草提取物在制备治疗肾小球肾炎药物中的应用 |
-
2012
- 2012-05-10 CN CN201210142501.4A patent/CN102961440B/zh not_active Expired - Fee Related
- 2012-12-13 US US14/397,860 patent/US20150118214A1/en not_active Abandoned
- 2012-12-13 WO PCT/CN2012/086605 patent/WO2013166833A1/zh not_active Ceased
- 2012-12-13 EP EP12876488.3A patent/EP2851079A4/en not_active Withdrawn
- 2012-12-13 JP JP2015510608A patent/JP2015515993A/ja active Pending
- 2012-12-13 KR KR20147034747A patent/KR20150020192A/ko not_active Withdrawn
- 2012-12-13 AU AU2012379443A patent/AU2012379443A1/en not_active Abandoned
Non-Patent Citations (7)
| Title |
|---|
| LIU, XIAOLONG ET AL.: "Development and Utilization of Medicinal Plant Resources of Salvia in Anhui", CHINA JOURNAL OF CHINESE MATERIA MEDICA, vol. 21, no. 2, February 1996 (1996-02-01), pages 73, XP008175500 * |
| See also references of EP2851079A4 * |
| SHI BAINIAN; LIU JIANG; HE JIANGFANG; DAI LICHENG; ZHOU JIANFANG; ZHAO WEIFENG; GAN JIANHE: "Expression of epoxidation enzyme 2 in liver injury of hepatic failure model", ZHONGYUAN MEDICAL JOURNAL, vol. 34, 2007 |
| WANG, XIAOMING ET AL.: "Studies on the Chemical Constituents from Salvia Plectranthoides Griff", JOURNAL OF HUAZHONG UNIVERSITY OF SCIENCE AND TECHNOLOGY, vol. 31, no. 3, June 2002 (2002-06-01), pages 254, XP008175501 * |
| XUE PEINI; CHANG XIAOHONG; YANG LIRONG: "Progress in research of correlation between COX-2 and non-small cell carcinoma of the lung", JOURNAL OFYANAN UNIVERSITY, vol. 09, 2011 |
| ZHANG HUIFENG; GAO XIAOQIN; LI RONGSHAN: "P_(38)MAPK/COX 2 Experimental research in protection effect of signal transduction path in second window of pre-treatment of rat kidney ischemia", JOURNAL OF SHANXI MEDICAL UNIVERSITY, vol. 40, 2009 |
| ZOU YINGYING; YAO NAN; WANG FANG; GAO QIAN; SONG JINGLIN: "Characteristics of expression ofCDX2 and COX2 in human large intestine cancer and their relation with metastasis", JOURNAL OFKUNMING MEDICAL COLLEGE, vol. 31, 2010 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102961440B (zh) | 2015-01-07 |
| CN102961440A (zh) | 2013-03-13 |
| JP2015515993A (ja) | 2015-06-04 |
| EP2851079A1 (en) | 2015-03-25 |
| AU2012379443A1 (en) | 2014-11-27 |
| US20150118214A1 (en) | 2015-04-30 |
| KR20150020192A (ko) | 2015-02-25 |
| EP2851079A4 (en) | 2015-11-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Lu et al. | Ganoderma lucidum extracts inhibit growth and induce actin polymerization in bladder cancer cells in vitro | |
| Cui et al. | Triterpenoid saponins from the genus Camellia: structures, biological activities, and molecular simulation for structure–activity relationship | |
| CN109453212B (zh) | 一种具有抗癌作用的毛诃子提取物及其有效部位制备方法 | |
| Tao et al. | 1-O-tigloyl-1-O-deacetyl-nimbolinin B inhibits LPS-stimulated inflammatory responses by suppressing NF-κB and JNK activation in microglia cells | |
| Kim et al. | Effects of the roots of Liriope Platyphylla Wang Et tang on gastrointestinal motility function | |
| CN101570557B (zh) | 一种青钱柳中的化合物及其在医药领域的应用 | |
| WO2013166833A1 (zh) | 包含有人环氧酶及阿霉素或类阿霉素的药用组合物、其制备方法及在制备多种药物的应用 | |
| CN110946948A (zh) | 化风丹在制备抗乳腺癌药物中的应用 | |
| Qian-Yu et al. | Differences in anti-inflammatory effects between two specifications of Scutellariae Radix in LPS-induced macrophages in vitro | |
| CN101152173A (zh) | 甘草素在制备治疗神经退行性疾病药物中的用途 | |
| CN109602859B (zh) | 苗药廖氏化风丹在制备防治黑色素瘤药物中的应用 | |
| CN105687217B (zh) | 一种苯丙素类化合物及其药学上可接受的盐在制备治疗炎症性疾病的药物中的应用 | |
| CN109350614A (zh) | 一种荜茇酰胺生物碱的降糖用途 | |
| Zhang et al. | In vitro inhibition of proliferation of vascular smooth muscle cells by serum of rats treated with Dahuang Zhechong pill | |
| CN101167816B (zh) | 毛茛总苷的制备方法和应用 | |
| CN110772596B (zh) | 一种复方中药桃花精及其制备工艺与作为治疗糖尿病心肌病药物的应用 | |
| CN105663150B (zh) | 一种苯丙素类化合物及其药学上可接受的盐在制备治疗炎症性疾病的药物中的应用 | |
| CN101966254A (zh) | 生地黄水提物在制备治疗由于体内雌性激素分泌不足而引起的妇女更年期综合症药物中的应用 | |
| CN109575043A (zh) | 二氢血根碱的分离制备及在制备抗炎药物中的应用 | |
| CN105884841B (zh) | 一种苯丙素类化合物的制备方法 | |
| CN105708845B (zh) | 一种苯丙素类化合物及其药学上可接受的盐在制备治疗炎症性疾病的药物中的应用 | |
| CN106046071B (zh) | 一种苯丙素类化合物的制备方法 | |
| CN105732736B (zh) | 一种苯丙素类化合物的制备方法 | |
| CN106046072B (zh) | 苯丙素类化合物及其药学上可接受的盐和药物组合物 | |
| CN106176764B (zh) | 拟缺香茶菜中2a,3β,19a-三羟基-12烯-28-乌苏酸在制备抗肿瘤药物中的应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12876488 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14397860 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2015510608 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2012379443 Country of ref document: AU Date of ref document: 20121213 Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012876488 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012876488 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20147034747 Country of ref document: KR Kind code of ref document: A |