WO2014049552A2 - Process for the preparation of rivaroxaban - Google Patents
Process for the preparation of rivaroxaban Download PDFInfo
- Publication number
- WO2014049552A2 WO2014049552A2 PCT/IB2013/058897 IB2013058897W WO2014049552A2 WO 2014049552 A2 WO2014049552 A2 WO 2014049552A2 IB 2013058897 W IB2013058897 W IB 2013058897W WO 2014049552 A2 WO2014049552 A2 WO 2014049552A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- carbonate
- formula
- process according
- dialkyl
- base
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
Definitions
- the present invention provides a process for the preparation of rivaroxaban.
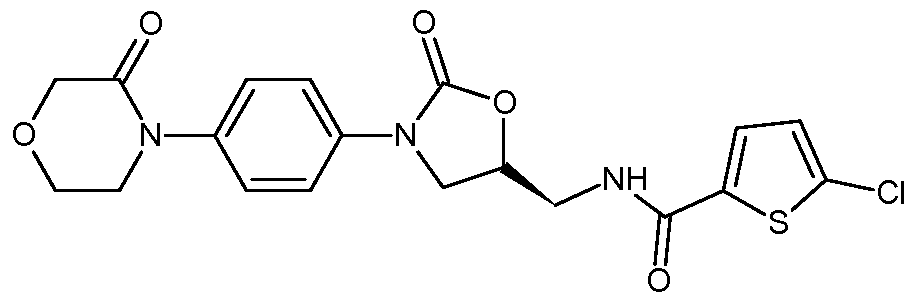
- Rivaroxaban chemically is 5-chloro-N-( ⁇ (5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl) phenyl]- 1 ,3-oxazolidin-5-yl ⁇ methyl)-2-thiophenecarboxamide of Formula I.
- Rivaroxaban is used as an anti-thrombotic agent.
- U.S. Patent No. 7,157,456 provides rivaroxaban and processes for its preparation.
- Example 44 of the '456 Patent provides a process of making rivaroxaban.
- Example 44 does not disclose a step of cyclizing a compound of Formula II with a dialkyl carbonate to produce the compound of Formula I.
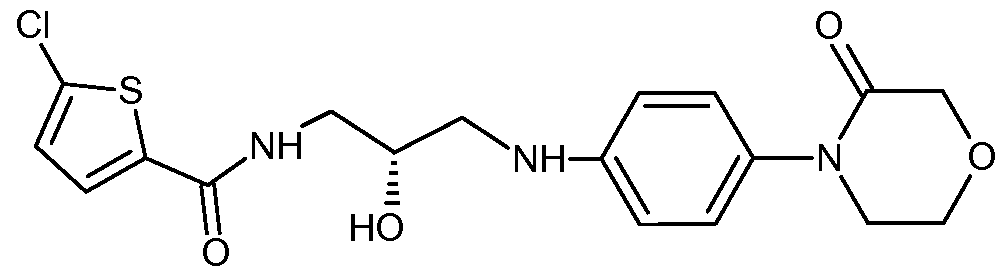
- U.S. Patent No. 8,106, 192 provides a process for the preparation of rivaroxaban, wherein N- ⁇ (R)-2-hydroxy-3-[4-(3-oxomorpholin-4-yl)phenylamino]propyl ⁇ -5- chlorothiophene-2-carboxamide is treated with ⁇ , ⁇ -carbonyldiimidazole in the presence of 1 -methyl-2-pyrrolidone and toluene.
- ⁇ , ⁇ -Carbonyldiimidazole is costly, toxic, moisture sensitive, and may produce toxic by-products during the reaction.
- the present invention provides processes for the preparation of rivaroxaban.
- Embodiments of the process may include one or more of the following features.
- the compound of Formula II may be cyclized with the dialkyl carbonate in the presence of a base.
- the dialkyl carbonate may be selected from one or more of dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate.
- the base may be selected from sodium carbonate, potassium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
- the base may be added to a solution of N- ⁇ (R)-2-hydroxy-3-[4-(3- oxomorpholin-4-yl)phenylamino]propyl ⁇ -5-chlorothiophene-2-carboxamide (Formula II) in the alkyl carbonate and the mixture refluxed.
- the process may further include the steps of (a) recovering the solvent under vacuum at 60°C to 65°C; (b) treating the solid material obtained with dichloromethane; and (c) filtering the solid material to remove any inorganic salt.
- the process may still further include (a) recovering the solvent under vacuum; and (b) crystallizing the material obtained from dichloromethane.
- the alkyl groups in the dialkyl carbonate may be the same or different.
- the invention relates to a process for the preparation of rivaroxaban of Formula I
- the process includes the steps of adding the base to a solution of N- ⁇ (R)-2-hydroxy-3-[4-(3-oxomorpholin-4-yl) phenylamino]propyl ⁇ -5-chlorothiophene-2-carboxamide (Formula II) in the alkyl carbonate and refluxing the resultant mixture;
- Embodiments of the process may include one or more of the following steps.
- the dialkyl carbonate may be selected from one or more of dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate.
- the base may be selected from sodium carbonate, potassium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
- the solvent may be recovered under vacuum at a temperature of 60°C to 65°C.
- the alkyl groups in the dialkyl carbonate may be the same or different.
- a first aspect of the present invention provides a process for the preparation of rivaroxaban of Formula I
- a second aspect of the present invention provides a process for the preparation of rivaroxaban of Formula I
- dimethyl carbonate diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate.
- a third aspect of the present invention provides a process for the preparation of rivaroxaban of Formula I
- the compound of Formula II may be prepared according to the process provided in the art, for example, the process described in U.S. Patent No. 8,106,192.
- the compound of Formula II is cyclized with a dialkyl carbonate, optionally in the presence of a base.
- the dialkyl carbonate may be, for example, dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate or combinations of dialkyl carbonates. It is expected that other dialkyl carbonates in which the alkyl groups are not the same will also function in the above reaction to cyclize the compound of Formula II.
- the dialkyl carbonate is a compound in which the alkyl groups in the dialkyl carbonate are the same.
- the alkyl groups in the dialkyl carbonate are not the same.
- the base may be, for example, sodium carbonate, potassium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
- the compound of Formula II may be heated with dialkyl carbonate for about 1 hour to about 8 hours above the boiling point of the alkanol produced during the reaction.
- the product may be isolated from the mixture by methods including concentration, distillation, decantation, filtration, evaporation, centrifugation, or a combination thereof, and may further be crystallized.
- dialkyl carbonate refers to a carbonate group flanked by two alkyl groups.
- alkyl refers to saturated, aliphatic hydrocarbon groups, either straight or branched-chain, containing from one to four carbon atoms, as exemplified by methyl, ethyl, propyl, isopropyl, butyl, or isobutyl.
- alkanol refers to an “alkyl” as defined above containing at least one hydroxyl group.
- the diethyl carbonate may be replaced by any of dimethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, diisobutyl carbonate or other dialkyl carbonate.
- the base used in the example, potassium carbonate may be replaced by sodium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Description
PROCESS FOR THE PREPARATION OF RIVAROXABAN
Field of the Invention
The present invention provides a process for the preparation of rivaroxaban.
Background of the Invention
Rivaroxaban chemically is 5-chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl) phenyl]- 1 ,3-oxazolidin-5-yl}methyl)-2-thiophenecarboxamide of Formula I.
Formula I
Rivaroxaban is used as an anti-thrombotic agent.
U.S. Patent No. 7,157,456 provides rivaroxaban and processes for its preparation. Example 44 of the '456 Patent provides a process of making rivaroxaban. However, Example 44 does not disclose a step of cyclizing a compound of Formula II with a dialkyl carbonate to produce the compound of Formula I.
U.S. Patent No. 8,106, 192 provides a process for the preparation of rivaroxaban, wherein N- {(R)-2-hydroxy-3-[4-(3-oxomorpholin-4-yl)phenylamino]propyl}-5- chlorothiophene-2-carboxamide is treated with Ν,Ν-carbonyldiimidazole in the presence of 1 -methyl-2-pyrrolidone and toluene. Ν,Ν-Carbonyldiimidazole is costly, toxic, moisture sensitive, and may produce toxic by-products during the reaction.
The prior art processes for the preparation of rivaroxaban make use of chromatography or reagents that are costly, unstable, and have safety concerns.
Accordingly, these processes are not suitable at industrial scale.
The present inventors have developed a simple, safe, efficient, economical, industrially feasible process that provides rivaroxaban in good yield.
Summary of the Invention
The present invention provides processes for the preparation of rivaroxaban.
In one general aspect, a process for the preparation of rivaroxaban of Formula I
Formula I
Includes a step of cyclizing a compound of Formula II
Formula II
with a dialkyl carbonate.
Embodiments of the process may include one or more of the following features. For example, the compound of Formula II may be cyclized with the dialkyl carbonate in the presence of a base. The dialkyl carbonate may be selected from one or more of dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate. The base may be selected from sodium carbonate, potassium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
In the process, the base may be added to a solution of N- {(R)-2-hydroxy-3-[4-(3- oxomorpholin-4-yl)phenylamino]propyl} -5-chlorothiophene-2-carboxamide (Formula II) in the alkyl carbonate and the mixture refluxed.
The process may further include the steps of (a) recovering the solvent under vacuum at 60°C to 65°C; (b) treating the solid material obtained with dichloromethane; and (c) filtering the solid material to remove any inorganic salt. The process may still further include (a) recovering the solvent under vacuum; and (b) crystallizing the material obtained from dichloromethane.
In the process, the alkyl groups in the dialkyl carbonate may be the same or different.
In another general aspect, the invention relates to a process for the preparation of rivaroxaban of Formula I
Formula I
using a process that includes cyclizing a compound of Formula II
Formula II
with a dialkyl carbonate in the presence of a base. The process includes the steps of adding the base to a solution of N- {(R)-2-hydroxy-3-[4-(3-oxomorpholin-4-yl) phenylamino]propyl}-5-chlorothiophene-2-carboxamide (Formula II) in the alkyl carbonate and refluxing the resultant mixture;
recovering the solvent under vacuum;
treating the solid material obtained with dichloromethane;
filtering the solid material to remove any inorganic salt;
recovering the solvent under vacuum; and
crystallizing the material obtained from dichloromethane.
Embodiments of the process may include one or more of the following steps. For example, the dialkyl carbonate may be selected from one or more of dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate. The base may be selected from sodium carbonate, potassium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
In the process, the solvent may be recovered under vacuum at a temperature of 60°C to 65°C.
In the process, the alkyl groups in the dialkyl carbonate may be the same or different.
Detailed Description of the Invention
A first aspect of the present invention provides a process for the preparation of rivaroxaban of Formula I
Formula I
wherein the process comprises cyclizing a compound of Formula II
Formula II
with a dialkyl carbonate.
A second aspect of the present invention provides a process for the preparation of rivaroxaban of Formula I
Formula I
Formula II
with dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate.
A third aspect of the present invention provides a process for the preparation of rivaroxaban of Formula I
Formula I
wherein the process comprises cyclizing a compound of Formula II
Formula II
with a diethyl carbonate.
The compound of Formula II may be prepared according to the process provided in the art, for example, the process described in U.S. Patent No. 8,106,192. The compound of Formula II is cyclized with a dialkyl carbonate, optionally in the presence of a base. The dialkyl carbonate may be, for example, dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate or combinations of dialkyl carbonates. It is expected that other dialkyl carbonates in which the alkyl groups are not the same will also function in the above reaction to cyclize the compound of Formula II. Therefore in one general aspect, the dialkyl carbonate is a compound in which the alkyl groups in the dialkyl carbonate are the same. In another
general aspect, the alkyl groups in the dialkyl carbonate are not the same. The base may be, for example, sodium carbonate, potassium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof. The compound of Formula II may be heated with dialkyl carbonate for about 1 hour to about 8 hours above the boiling point of the alkanol produced during the reaction. The product may be isolated from the mixture by methods including concentration, distillation, decantation, filtration, evaporation, centrifugation, or a combination thereof, and may further be crystallized.
The term "dialkyl carbonate", as used herein, refers to a carbonate group flanked by two alkyl groups.
The term "alkyl", as used herein, refers to saturated, aliphatic hydrocarbon groups, either straight or branched-chain, containing from one to four carbon atoms, as exemplified by methyl, ethyl, propyl, isopropyl, butyl, or isobutyl.
The term "alkanol", as used herein, refers to an "alkyl" as defined above containing at least one hydroxyl group.
The term "about", as used herein, when used along with values assigned to certain measurements and parameters means a variation of up to 10% from such values, or in case of a range of values, means up to a 10% variation from both the lower and upper limits of such ranges.
While the present invention has been described in terms of its specific
embodiments, certain modifications and equivalents will be apparent to those skilled in the art, and are intended to be included within the scope of the present invention.
EXAMPLE
Preparation of 5-Chloro-N-i K5S V2-Oxo-3 - Γ4-Γ3 -Oxo-4-MorpholinvnPhenyll - 1.3- Oxazolidin-5-Yl}Methyl)-2-Thiophenecarboxamide (Formula I)
Potassium carbonate (0.64 g; 4.6 mmoles) was added to a solution of N- {(R)-2- hydroxy-3-[4-(3-oxomorpholin-4-yl)phenylamino]propyl}-5-chlorothiophene-2- carboxamide (Formula II; 1 g; 2.24 mmoles) in diethyl carbonate (4 mL). The mixture was refluxed for 4 hours. The solvent was recovered under vacuum at 60°C to 65°C. The solid material obtained was treated with dichloromethane (10 mL) and filtered to remove
the inorganic salt. The solvent was recovered under vacuum at 35°C and the material obtained was crystallized from dichloromethane (5 mL).
Yield = 0.9 g (85%).
It should be understood that variations in the above processes are contemplated. For example, the diethyl carbonate may be replaced by any of dimethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, diisobutyl carbonate or other dialkyl carbonate. Similarly, the base used in the example, potassium carbonate, may be replaced by sodium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
Claims
1. A process for the preparation of rivaroxaban of Formula I
Formula I
wherein the process comprises cyclizing a compound of Formula II
Formula II
with a dialkyl carbonate.
2. The process according to claim 1, wherein the compound of Formula II is cyclized with the dialkyl carbonate in the presence of a base.
3. The process according to claim 1 , wherein the dialkyl carbonate is selected from one or more of dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate.
4. The process according to claim 2, wherein the dialkyl carbonate is selected from one or more of dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate.
5. The process according to claim 2, wherein the base is selected from sodium carbonate, potassium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
6. The process according to claim 2, wherein the base is added to a solution of N- {(R)-2-hydroxy-3-[4-(3-oxomorpholin-4-yl)phenylamino]propyl} -5-chlorothiophene-2- carboxamide (Formula II) in the alkyl carbonate and the mixture refluxed.
7. The process according to claim 6, further comprising:
recovering the solvent under vacuum at 60°C to 65°C;
treating the solid material obtained with dichloromethane; and
filtering the solid material to remove any inorganic salt.
8. The process according to claim 7, further comprising:
recovering the solvent under vacuum; and
crystallizing the material obtained from dichloromethane.
9. The process according to claim 1, wherein the alkyl groups in the dialkyl carbonate are the same.
10. A process for the preparation of rivaroxaban of Formula I
Formula I
wherein the process comprises cyclizing a compound of Formula II
Formula II
with a dialkyl carbonate in the presence of a base, the method comprising:
adding the base to a solution of N- {(R)-2-hydroxy-3-[4-(3-oxomorpholin-4-yl) phenylamino]propyl} -5-chlorothiophene-2-carboxamide (Formula II) in the alkyl carbonate and refluxing the resultant mixture;
recovering the solvent under vacuum;
treating the solid material obtained with dichloromethane;
filtering the solid material to remove any inorganic salt;
recovering the solvent under vacuum; and
crystallizing the material obtained from dichloromethane.
1 1. The process according to claim 10, wherein the dialkyl carbonate is selected from one or more of dimethyl carbonate, diethyl carbonate, dipropyl carbonate, diisopropyl carbonate, dibutyl carbonate, or diisobutyl carbonate.
12. The process according to claim 10, wherein the base is selected from sodium carbonate, potassium carbonate, calcium carbonate, sodium hydroxide, potassium hydroxide, or a mixture thereof.
13. The process according to claim 10, wherein the solvent is recovered under vacuum at 60°C to 65°C.
14. The process according to claim 10, wherein the alkyl groups in the dialkyl carbonate are the same.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN2600DEN2015 IN2015DN02600A (en) | 2012-09-26 | 2013-09-26 | |
| EP13805537.1A EP2900663A2 (en) | 2012-09-26 | 2013-09-26 | Process for the preparation of rivaroxaban |
| US14/430,279 US20150218145A1 (en) | 2012-09-26 | 2013-09-26 | Process for the preparation of rivaroxaban |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN3005DE2012 | 2012-09-26 | ||
| IN3005/DEL/2012 | 2012-09-26 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2014049552A2 true WO2014049552A2 (en) | 2014-04-03 |
| WO2014049552A3 WO2014049552A3 (en) | 2014-05-15 |
Family
ID=49765588
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2013/058897 Ceased WO2014049552A2 (en) | 2012-09-26 | 2013-09-26 | Process for the preparation of rivaroxaban |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20150218145A1 (en) |
| EP (1) | EP2900663A2 (en) |
| IN (1) | IN2015DN02600A (en) |
| WO (1) | WO2014049552A2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016030669A1 (en) * | 2014-08-25 | 2016-03-03 | Cipla Limited | Process for the preparation of rivaroxaban |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7157456B2 (en) | 1999-12-24 | 2007-01-02 | Bayer Healthcare Ag | Substituted oxazolidinones and their use in the field of blood coagulation |
| US8106192B2 (en) | 2003-01-07 | 2012-01-31 | Bayer Pharma Aktiengesellschaft | Method for producing 5-chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-1,3-oxazolidin-5-yl}methyl)-2-thiophenecarboxamide |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2655369A1 (en) * | 1976-12-03 | 1978-06-08 | Schering Ag | 5- (SUBST. PHENYL) -OXAZOLIDINONE AND THEIR SULFUR ANALOGS AND PROCESS FOR THEIR PRODUCTION |
| EP2354128A1 (en) * | 2010-02-10 | 2011-08-10 | Sandoz Ag | Method for the preparation of rivaroxaban |
| US20130253187A1 (en) * | 2010-09-14 | 2013-09-26 | Medichem, S.A. | Process for Determining the Suitability for Distribution of a Batch of Thiophene-2-Carboxamide Derivative |
| WO2013046211A1 (en) * | 2011-09-27 | 2013-04-04 | Symed Labs Limited | Processes for the preparation of 5-chloro-n-({(5s)-2-oxo-3-[4-(3-oxo-4-morpholinyl) phenyl]-1,3-oxazolidin-5-yl}methyl)-2-thiophene-carboxamide and intermediates thereof |
| WO2013164833A1 (en) * | 2012-05-02 | 2013-11-07 | Symed Labs Limited | Improved process for preparing rivaroxaban using novel intermediates |
-
2013
- 2013-09-26 EP EP13805537.1A patent/EP2900663A2/en not_active Withdrawn
- 2013-09-26 US US14/430,279 patent/US20150218145A1/en not_active Abandoned
- 2013-09-26 IN IN2600DEN2015 patent/IN2015DN02600A/en unknown
- 2013-09-26 WO PCT/IB2013/058897 patent/WO2014049552A2/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7157456B2 (en) | 1999-12-24 | 2007-01-02 | Bayer Healthcare Ag | Substituted oxazolidinones and their use in the field of blood coagulation |
| US8106192B2 (en) | 2003-01-07 | 2012-01-31 | Bayer Pharma Aktiengesellschaft | Method for producing 5-chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-1,3-oxazolidin-5-yl}methyl)-2-thiophenecarboxamide |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016030669A1 (en) * | 2014-08-25 | 2016-03-03 | Cipla Limited | Process for the preparation of rivaroxaban |
Also Published As
| Publication number | Publication date |
|---|---|
| IN2015DN02600A (en) | 2015-09-18 |
| US20150218145A1 (en) | 2015-08-06 |
| EP2900663A2 (en) | 2015-08-05 |
| WO2014049552A3 (en) | 2014-05-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2012103960A1 (en) | Process for making trisubstituted silyloxyethyl triflates | |
| WO2017097275A1 (en) | Solid forms of (2r,4s)-5-(biphenyl-4-yl)-4-[(3-carboxypropionyl)amino]-2- -methylpentanoic acid ethyl ester, its salts and a preparation method | |
| AU2011300365A1 (en) | Processes for the preparation of 4-{4-[5(S)-(aminomethyl)-2-oxo-1,3-oxazolidin-3-yl]phenyl} morpholin-3-one | |
| KR101653064B1 (en) | A Method for Gadobutrol | |
| WO2013156936A1 (en) | Process for the preparation of rivaroxaban and intermediates thereof | |
| WO2013120464A1 (en) | A process for the preparation of rivaroxaban based on saving of 1,1'-carbonyl diimidazole. | |
| JP2018518504A (en) | Methods for the synthesis of rapamycin derivatives | |
| KR100917698B1 (en) | Improved process for the preparation of letrozole | |
| WO2012156983A1 (en) | Processes for the preparation of 5-chloro-n-({(5s)-2-oxo-3-[4-(3-oxo-4-morpholinyl) phenyl]-1,3-oxazolidin-5-yl}methyl)-2-thiophenecarboxamide | |
| CN106317114B (en) | A kind of preparation method of Tedizolid Phosphate | |
| KR100877849B1 (en) | Efficient preparation method of 3-hydroxytetrahydrofuran | |
| WO2012131017A1 (en) | Process for the production of deferasirox | |
| WO2014049552A2 (en) | Process for the preparation of rivaroxaban | |
| US20010014739A1 (en) | Purifying process of tetrakis (fluoroaryl) borate.magnesium halide, tetrakis (fluoroaryl) borate.ether complex and producing process of the same, and producing process of tetrakis (fluoroaryl) borate derivative | |
| EP2384324B1 (en) | Process for the preparation of efavirenz | |
| GB2551591B (en) | Liquid-liquid extraction of DMF | |
| WO2016021079A1 (en) | Anhydrosugar alcohol purification method, anhydrosugar alcohol, and resin | |
| WO2013175431A1 (en) | Process for the preparation of rivaroxaban | |
| WO2012041263A2 (en) | A method of manufacturing 2-({(5s)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]- l,3-oxazolidin-5-yl}methyl)-lh-isoindol-l,3(2h)-dione with a high optical purity | |
| CN101838235B (en) | 3-phenyl-3'-pyridylallylamine compound and synthesis method thereof | |
| KR20080077619A (en) | Variable Method of Making Rapamycin 42-Ester from Rapamycin 42-Ester Boronate | |
| CN104530060B (en) | A kind of preparation method of Bio key intermediate (3aS, 6aR)-lactone | |
| CN104098604A (en) | Fosaprepitant dimeglumine preparation method | |
| JPWO2004106314A1 (en) | Method for producing biphenyl compound containing oxetane ring | |
| JP7018840B2 (en) | Method for Producing Phenylpiperazine Pyridine Methyl Acetate |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13805537 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14430279 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2013805537 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013805537 Country of ref document: EP |