WO2014148396A1 - 植物保護剤及び植物病害の防除方法 - Google Patents
植物保護剤及び植物病害の防除方法 Download PDFInfo
- Publication number
- WO2014148396A1 WO2014148396A1 PCT/JP2014/056963 JP2014056963W WO2014148396A1 WO 2014148396 A1 WO2014148396 A1 WO 2014148396A1 JP 2014056963 W JP2014056963 W JP 2014056963W WO 2014148396 A1 WO2014148396 A1 WO 2014148396A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- iron
- iron oxide
- plant
- control method
- biox
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N63/00—Biocides, pest repellants or attractants, or plant growth regulators containing microorganisms, viruses, microbial fungi, animals or substances produced by, or obtained from, microorganisms, viruses, microbial fungi or animals, e.g. enzymes or fermentates
- A01N63/20—Bacteria; Substances produced thereby or obtained therefrom
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N59/00—Biocides, pest repellants or attractants, or plant growth regulators containing elements or inorganic compounds
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N59/00—Biocides, pest repellants or attractants, or plant growth regulators containing elements or inorganic compounds
- A01N59/16—Heavy metals; Compounds thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N59/00—Biocides, pest repellants or attractants, or plant growth regulators containing elements or inorganic compounds
- A01N59/26—Phosphorus; Compounds thereof
Definitions
- the present invention relates to a novel plant protective agent and a method for controlling plant diseases using the plant protective agent.
- microbial pesticides using soil microorganisms and plant endogenous microorganisms are well known, and some of these are commercially available. So far, nearly 20 types of microbial pesticides have been released, but problems such as difficulty in storage and handling have been pointed out. In addition, microbial pesticides cannot afford the possibility that microorganisms cannot exert their full potential under various environments and stresses and produce harmful metabolites under stress.
- Patent Document 1 Non-Patent Document 1
- Patent Document 2 to 4 have the following reports on the use of such iron oxide derived from microorganisms.
- Patent Document 2 an organic / inorganic composite material chemically modified with an organic group is obtained by subjecting a microorganism-derived ceramic to chemical treatment, and an organic group introduced into the organic / inorganic composite material is used. It has been reported that catalysts can be immobilized.
- Patent Document 3 reports that magnetic ceramics having unique properties can be obtained by heat-treating microorganism-derived ceramics.
- Patent Document 4 it is possible to obtain amorphous silica by acid-treating iron content of iron oxide produced by microorganisms and dissolving and removing the Fe component, the amorphous silica has an acid point that acts as a solid acid catalyst, It has been reported that it has superior acid strength and catalytic activity compared to an artificially synthesized silica catalyst.
- An object of the present invention is to provide a plant protection agent that is easy to handle and can be stably stored, can be used stably and safely, and a method for controlling plant diseases using the plant protection agent.
- OUMS1-BIOX L-BIOX® produced by OUMS1 (hereinafter also referred to as OUMS1-BIOX) for crop (plant) protection. Then, OUMS1-BIOX is safe with almost no direct antibacterial activity and cytotoxicity, and has the activity of remarkably inhibiting the invasion of pathogenic bacteria, and also has the activity of inducing resistance to plants. I got the knowledge.
- the present invention has been completed through further studies based on these findings, and provides the following plant protective agents and methods for controlling plant diseases.
- Plant protectant (I-1) A plant protective agent comprising amorphous and / or microcrystalline silicon and phosphorus-containing iron oxide.
- the iron oxide contains iron and oxygen as main components, The element ratio of iron, silicon, and phosphorus is 66 to 87: 2 to 27: 1 to 32, respectively (the total of the number of atoms% of iron, silicon, and phosphorus is 100).
- the plant protective agent as described in (I-1).
- II-3) The plant protective agent according to (I-1) or (I-2), wherein the iron oxide further contains 0.1 to 5% by weight of carbon.
- (I-4) The plant protective agent according to any one of (I-1) to (I-3), wherein the microcrystalline iron oxide is ferrihydrite and / or lipidocrosite.
- (I-5) The plant protective agent according to any one of (I-1) to (I-4), wherein the iron oxide is iron oxide produced by an iron-oxidizing bacterium.
- (I-6) The plant protective agent according to (I-5), wherein the iron oxide is iron oxide separated from an aggregated precipitate generated by an iron bacterial water purification method.
- (I-7) The plant protective agent according to (I-5) or (I-6), wherein the iron-oxidizing bacterium is a bacterium belonging to the genus Leptothrix and / or Gallionella.
- (I-8) The plant protective agent according to (I-5) or (I-6), wherein the iron-oxidizing bacterium is Leptothrix cholodnii OUMS1 (NITE BP-860).
- (I-9) The plant protective agent according to any one of (I-5) to (I-8), comprising a supernatant separated from a suspension of iron oxide and water produced by iron-oxidizing bacteria .
- (I-10) The plant protective agent according to any one of (I-1) to (I-9), which is in the form of a wettable powder.
- (I-11) The plant protective agent according to any one of (I-1) to (I-9), which is in the form of a spray.
- (I-12) Use of amorphous and / or microcrystalline silicon and phosphorus-containing iron oxides as plant protection agents.
- Plant disease control method (II) A method for controlling plant diseases, comprising a step of applying the plant protective agent according to any one of (I-1) to (I-11).
- the plant protective agent of the present invention is a safe material that hardly recognizes direct antibacterial activity and cytotoxicity, and has an activity that can remarkably inhibit the invasion of pathogenic bacteria when given to plants.
- the plant protective agent of the present invention can induce resistance through a plurality of pathways to plants. This indicates that it is possible to target pathogens that attempt to invade plants, and is highly valuable from the viewpoint of safety. Moreover, this has shown that it is effective with respect to a wide range of pathogens, and since it has many action points, it is thought that appearance of a resistant microbe can also be avoided.
- the plant protective agent of the present invention is easy to handle and can be stored stably.
- the conventional microbial pesticides cannot fully eliminate the effects of microorganisms under various stresses and the possibility of producing harmful metabolites, but the plant protection agent of the present invention does not have such problems. It can be used stably and safely.
- the present invention paves the way for the effective use of BIOX, which has become a problem in the treatment of water purification facilities. is there.
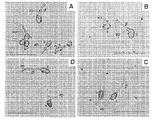
- Bar is 50 ⁇ m It is a graph which shows the germination rate of the gray mold fungus conidia spores and pea brown mold fungus spore which were processed with OUMS1-BIOX on the slide glass. The final concentration is 1/3 of the displayed concentration in the graph. It is a microscope picture which shows the morphogenesis of the gray mold fungus on the onion epidermis treated with ethanol. The bar is 30 ⁇ m. a, attachment device; gt, germ tube; It is a graph which shows the invasion rate of the gray mold fungus to an onion epidermis cell. Different alphabets indicate significant differences. It is a photograph showing the effect of OUMS1-BIOX on brown leaf blight infection in pea leaves.
- the plant protective agent of the present invention is characterized by containing amorphous and / or microcrystalline silicon and phosphorus-containing iron oxide.
- the iron oxide in the present invention contains iron and oxygen as main components, and the element ratio of iron, silicon and phosphorus is 66 to 87: 2 to 27: 1 to 32, preferably 70 to 75: 5, respectively, in terms of atomic%. It is desirable that it is ⁇ 15: 5 ⁇ 20% (the sum of the atomic percentages of iron, silicon and phosphorus is 100).
- the microcrystalline iron oxide is preferably ferrihydrite and / or lipidocrosite.
- Ferrihydrite means low crystalline iron oxide. It is called 2-line ferrihydrite or 6-line ferrihydrite depending on the number of peaks appearing in the X-ray diffraction pattern.
- the composition of 2-line ferrihydrite is Fe 4 (O, OH, H 2 O) and the composition of 6-line ferrihydrite is Fe 4.6 (O, OH, H 2 O) 12 (RA Eggleton and RW Fitzpatrick, “ New data and a revised structural model for ferrihydrite ”, Clays and Clay Minerals, Vol. 36, No. 2, pp111-124, 1988).
- Repidocrocite is crystalline iron oxide whose chemical formula is represented by ⁇ -FeOOH.
- iron oxide in the present invention both iron oxide prepared by a synthesis method and iron oxide generated by iron-oxidizing bacteria can be used.
- Examples of the method for synthesizing iron oxide in the present invention include reacting an iron compound, a silicon compound, and a phosphorus compound as follows.
- An iron compound, a silicon compound, and a phosphorus compound were dissolved in a solvent at a predetermined ratio, and an alkaline aqueous solution (for example, ammonia, sodium hydroxide, potassium hydroxide, calcium carbonate, etc.) was added dropwise with stirring to obtain a pH of about 10.
- an alkaline aqueous solution for example, ammonia, sodium hydroxide, potassium hydroxide, calcium carbonate, etc.
- the precipitate is washed with distilled water and collected by centrifugation.
- the obtained product is dried under reduced pressure and pulverized to prepare silicon- and phosphorus-containing iron oxide.
- iron compound examples include iron nitrate, iron sulfate, iron chloride, and iron carbonate. Among these, iron nitrate is preferable.
- silicon compound examples include sodium silicate and potassium silicate.
- phosphorus compound examples include phosphoric acid, sodium phosphate, and potassium phosphate.
- Examples of the medium for reacting the iron compound and the like include aqueous solutions and alcohols, but aqueous solutions are preferred.
- the temperature during the reaction is 10 to 50 ° C., preferably 20 to 30 ° C.
- the iron-oxidizing bacteria is not particularly limited as long as it forms amorphous and / or microcrystalline silicon- and phosphorus-containing iron oxides.
- Examples of iron-oxidizing bacteria include Toxothrix sp., Leptothrix sp., Crenothrix sp., And Clonothrix sp. , Gallionella sp., Siderocapsa sp., Siderococcus sp., Sideromonas sp., Planktomyces sp. Can do.
- Leptothrix ochracea which is a bacterium belonging to the genus Leptothrix, can produce biogenous iron oxide having a hollow fibrous sheath structure.
- Galionella ferruginea which is a bacterium belonging to the genus Galionella is capable of generating helical biogenous iron oxide.
- Chronosilix bacteria are branched tubes or filaments
- Toxosilicus bacteria are filamentous (harp-like, fan-shaped)
- Sideromonas bacteria are short stems
- Siderocapsa bacteria are capsules
- Siderococcus bacteria Is known to produce spherical biogenous iron oxides (for example, “Sadao Kojima, Ryuichi Sudo, Mitsuo Chihara:“ Environmental Microbiology Encyclopedia ”Kodansha (1995)”).
- the size of biogenous iron oxide varies depending on its type, but is usually about 0.1 to 3000 ⁇ m. More specifically, in the case of a sheath, a spiral, a branched tube, a thread, and a short stem, the diameter is usually about 0.1 to 5 ⁇ m and the length is about 5 to 3000 ⁇ m. In the case of a capsule, the length is usually about 1.2 to 24 ⁇ m. Further, if it is spherical, it is usually about 0.1 to 1 ⁇ m.
- Iron oxide produced by iron-oxidizing bacteria such as bacteria belonging to the genus Leptothrix is generally amorphous iron oxide.
- Leptothrix Korodini OUMS1 strain As an example of the genus Bacteria belonging to the genus Leptothrix, Leptothrix Korodini OUMS1 strain can be mentioned.
- the Leptothrix Korodini OUMS1 strain was issued on December 25, 2009 by the National Institute for Product Evaluation Technology Patent Microorganisms Deposit Center (2-5-8 Kazusa Kamashi, Kisarazu City, Chiba Prefecture, Japan) )), The deposit number is NITE P-860. This strain has now been transferred to an international deposit and its deposit number is NITE BP-860.
- the iron oxide produced by Leptolith Korodini OUMS1 strain has the structure of ferrihydrite and lepidochrosite, and is an aggregate of ferrihydrite nanoparticles or lepidochrosite nanoparticles.
- the primary particle size of the ferrihydrite nanoparticles is preferably about 3 to 5 nm, and the primary particle size of the lipidocrosite nanoparticles is preferably about 30 to 50 nm.
- biogenous iron oxide produced by Leptotrix Korodini examples include microtubes, nanotubes, hollow strings, capsules, strings and spherical aggregates, strings, rods, and the like.
- the size of these biogenous iron oxides is as follows: microtube shape: diameter 0.3-4 ⁇ m, length 5-200 ⁇ m, nanotube shape: diameter 300-450 nm, length 5-200 ⁇ m, hollow string shape: length 3-10 ⁇ m, Capsule shape: major axis 0.5 to 7 ⁇ m, minor axis 0.5 to 3 ⁇ m, string shape: length 0.5 to 5 ⁇ m, rod shape: length 5 to 30 ⁇ m.
- the method for obtaining biogenous iron oxide is not particularly limited, and various methods can be used.
- a method for obtaining from an aggregated precipitate produced by an iron-oxidizing bacterium present in a bio-water purification method (iron bacterial water purification method) or a water purification plant (see JP-A-2005-272251), and JP-A-10-338526 Can be used as a method for obtaining biogenous iron oxide, and the description thereof can be used as appropriate.
- the “iron bacterial water purification method” refers to the purification of microorganisms, in contrast to the rapid filtration water purification method that removes impurities in the raw water using only the coagulation effect of a coagulant such as polyaluminum chloride (PAC).
- a coagulant such as polyaluminum chloride (PAC).
- impurities are removed by action.
- examples of the method for removing impurities by the action of purifying microorganisms include a method for coagulating and precipitating impurities in raw water using the aggregating action of microorganisms such as iron-oxidizing bacteria.
- water purification is performed using microorganisms, and a so-called ⁇ slow filtration water purification method (natural filtration) that only forms a microorganism film on the surface of the sand layer described above and filters the raw water through the sand layer.
- a so-called “medium speed filtration water purification method” in which the filtration layer is washed to prevent the filtration layer from being blocked and maintain the filtration rate.
- the genus Leptothrix is a dominant bacterium in the filtration layer of the iron bacteria water purification method, and mainly produces biogenous iron oxide having a hollow fibrous sheath structure.
- the inventors of the present invention have an excellent characteristic that the biogenous iron oxide having a hollow fiber-like sheath structure produced by a Leptococcus bacterium has a hollow with an inner diameter of about 1.0 ⁇ m and an outer diameter of about 1.2 ⁇ m, and is a substantially uniform particle. I have confirmed.
- the “iron bacterial water purification method” means including the phenomenon itself of agglomerating and removing iron ions and the like in the raw water by the same action as described above, and water purification on a practical scale for the purpose of genuine water purification. Is meant to include small-scale implementation at the laboratory level.
- biogenous iron oxide that can be used in the present invention
- a product obtained by separating biogenous iron oxide contained therein from an aggregated precipitate generated by an iron bacterial water purification method can be suitably used.
- the method for separating biogenous iron oxide is not particularly limited as long as it is a method capable of separating biogenous iron oxide from the aggregated precipitate.
- the suspension of the aggregated precipitate is not allowed to pass through the biogenous iron oxide.
- the suspension may be passed through a sieve having a pore size (mesh size) through which only impurities pass (mesh size), a mesh, a filter, an insulator net used for papermaking, or the like.
- the above-mentioned aggregated precipitate is a precipitate obtained by agglomeration of iron ions or the like in the raw water by the aggregation action of iron-oxidizing bacteria in the above-described iron bacterial water purification method.
- the aggregated precipitate referred to in the present invention is sufficient if impurities in the raw water are aggregated due to the aggregation action of iron-oxidizing bacteria, and particularly includes aggregates that are not settled (precipitated). That is, the aggregated precipitate referred to in the present invention may be in a suspended state in water or the like, or may be in a suspension state in which the precipitate is resuspended by washing or the like. Furthermore, the dry state which evaporated the water
- the method for obtaining the aggregated precipitate is not particularly limited, and the sediment deposited on the filtration layer in the water purification facility may be scraped, or backwash water (washing) in the slow (medium speed) filtration water purification method. Water). Moreover, the filtration residue filtered separately with the filtration apparatus may be sufficient, and the precipitation acquired with the centrifuge may be sufficient. Furthermore, the aggregate obtained by sedimentation by natural sedimentation may be a sediment obtained by decantation.
- biogenous iron oxide from the aggregated precipitate generated by iron-oxidizing bacteria present in water purification plants and the like will be described below.
- precipitates formed by iron-oxidizing bacteria present in water purification plants using natural filtration methods such as Leptothrix ocracea (hereinafter referred to as ⁇ L. Collect.
- the constituent element ratio and structure of biogenous iron oxide from which L. ochracea forms a precipitate vary depending on the temperature and water quality of the environment where the iron-oxidizing bacteria survive, but L. ochracea produces a precipitate. If it is the conditions which can do, it will not specifically limit,
- the biogenous iron oxide which mainly has the structure of a hollow fiber-like sheath structure can be obtained.
- biogenous iron oxide can be obtained by removing impurities such as sand from the washed sludge using a sieve. If necessary, the obtained biogenous iron oxide may be sorted by specific gravity using a centrifuge.
- biogenous iron oxide produced by the above iron-oxidizing bacteria varies depending on the iron-oxidizing bacteria used for production and the conditions at the time of production, but includes a hollow fiber sheath structure, a spiral shape, a granular shape, and a filamentous shape. It is what has been.
- biogenous iron oxide having a hollow fiber sheath structure may be mainly included, and granular biogenous iron oxide may be mainly included.
- any iron oxide produced by the iron-oxidizing bacterium can be used for the plant protection agent of the present invention regardless of the shape or any of the above shapes.
- the composition contains iron and oxygen as main components and further contains silicon, phosphorus and the like.
- the biogenous iron oxide may further contain carbon in a proportion of 0.1 to 5% by weight, particularly 0.2 to 2% by weight.
- This composition changes appropriately depending on the environment in which iron-oxidizing bacteria are present. Therefore, the synthesized iron oxides such as 2-line ferrihydrite are different in that their compositions do not contain phosphorus and silicon. Furthermore, it is clear from the sample measurement results by SEM that each constituent element is uniformly distributed in biogenous iron oxide.
- the supernatant liquid separated from the suspension of iron oxide and water may be used as a component of the plant protection agent.
- the supernatant liquid separated from the suspension of iron oxide and water may be used as a component of the plant protection agent.
- the supernatant liquid separated from the suspension of iron oxide and water may be used as a component of the plant protection agent.
- the supernatant liquid separated from the suspension of iron oxide and water may be used as a component of the plant protection agent.
- the supernatant liquid separated from the suspension of iron oxide and water may be used as a component of the plant protection agent.
- iron oxide in the present invention is a safe material because it hardly recognizes direct antibacterial activity and cytotoxicity. It has an activity that can be inhibited.
- the iron oxide in the present invention can induce resistance to plants through a plurality of pathways. This indicates that it is possible to target pathogens that attempt to invade plants, which is also highly valuable in terms of safety. This also indicates that the iron oxide is effective against a wide range of pathogens, and since there are many action points, it is considered that the emergence of resistant bacteria can be avoided.
- the iron oxide in the present invention has the characteristics that it is easy to handle and can be stably stored, and can be used stably and safely.
- the plant protective agent of the present invention can also be referred to as a plant disease control composition in view of the effect of the iron oxide.
- the plant protective agent of the present invention includes a carrier, a surfactant, a wetting agent, a preservative, a sticking agent, a stabilizer, a colorant, an emulsifier, a dispersant, a penetrating agent, a thickener, as components other than the iron oxide.
- An antifoaming agent etc. can be suitably mix
- the plant protective agent of the present invention is appropriately formulated by known methods in the form of wettable powder, granular wettable powder, flowable powder, emulsion, spraying powder, powder, aerosol, paste, suspension, liquid, etc. It may be made.
- the plant protection agent of the present invention may be used as it is, or diluted to a predetermined concentration with a diluent.
- the content of the iron oxide in the plant protective agent of the present invention is not particularly limited as long as the effect of the present invention is obtained, but is preferably 0.003 to 99% by weight, more preferably 0.013 to 95% by weight, still more preferably 0.067 to 90% by weight.
- agricultural chemicals such as an insecticide, an acaricide, a herbicide, may be used together besides the said iron oxide.
- the application method of the plant protective agent of the present invention is not particularly limited as long as the effects of the present invention are obtained.
- the spraying treatment to the plant body the spraying treatment to the soil surface, the infusion treatment into the soil, the plant seeds Spray treatment, smear treatment on plant seeds, immersion treatment in plant seeds, and the like.
- the application amount of the plant protective agent of the present invention can be appropriately selected according to the target disease, target plant, dosage form, degree of occurrence of the disease, application method, and the like.
- Plants targeted by the plant protective agent of the present specification are not limited to these, but examples thereof include rice, wheat, barley, peas, onions, corn, grapes, apples, pears, peaches, oysters, and citrus. , Soybean, strawberry, green beans, potato, cabbage, lettuce, tomato, cucumber, sugar beet, spinach, eggplant, watermelon, pumpkin, sugar cane, bell pepper, sugar beet, sweet potato, taro, cotton, sunflower, tulip, chrysanthemum and the like.
- the plant disease targeted by the plant protective agent of the present invention is not particularly limited as long as the effect of the present invention is obtained, and examples thereof include the following plant diseases.
- Gray mold disease such as tomato, cucumber, beans, strawberry, potato, cabbage, eggplant, lettuce
- mycorrhizal disease Sclerotinia sclerotiorum such as tomato, cucumber, legume, strawberry, potato, rapeseed, cabbage, eggplant, lettuce
- Seedling blight of various vegetables such as tomato, cucumber, beans, radish, watermelon, eggplant, rape, pepper, spinach, sugar beet (Rhizoctoniaoctspp., Pythium spp., Fusarium spp., Phythophthora spp., Sclerotiniarotsclerotiorum, etc.)
- Cucumber downy mildew Pseudoperonospora) cubensis
- powdery mildew Sphaerotheca ⁇ fuliginea
- anthrax Coldletotrichum lagenarium
- vine blight Mycosphaerella melonis
- vine split Feusa
- Grape downy mildew (Plasmopora viticola), rust (Phakopsora ampelopsidis), powdery mildew (Uncinula necator), black mildew (Elsinoe ampelina), late rot (Glomerella cingulata), black rot (Guignardia bidwellii), vine Wart disease (Phomopsis viticola), soot spot disease (Zygophiala jamaicensis), gray mold disease (Botrytis cinerea), bud blight disease (Diaporthe medusaea), purple coat feather disease (Helicobasidium mompa), white coat disease (Rosellinia necatrix); apple Powdery mildew (Podosphaera leucotricha), black spot disease (Venturia inaequalis), spotted leaf disease (Alternaria alternata (Apple pathotype)), red star disease (Gymnosporangium yamadae), mon
- Sunflower sclerotia Sunflower sclerotia (Sclerotinia sclerotiorum); rose black spot (Diplocarpon rosae), powdery mildew (Sphaerotheca pannosa), downy mildew (Peronospora sparsa), plague (Phytophthora megasperma); indici), white rust (Puccinia horiana), plague (Phytophthora cactorum);
- Rice blast (Pyricularia oryze), blight (Thanatephorus cucumeris), sesame leaf blight (Cochliobolus miyabeanus), idiot seedling (Gibberella fujikuroi), seedling blight (Pythium spp., Fusarium spp., Trichoderma spp.
- Rice rot (Claviceps virens), Smut (Tilletia barelayana); wheat powdery mildew (Erysiphe graminis f.sp.hordei), rust (Puccinia striiformis, Puccinia gramini) Puccinia recondita, Puccinia hordei), leafy leaf disease (Pyrenophora graminea), reticular leaf disease (Pyrenophora teres), red mold disease (Fusarium graminearum, Fusarium culmorum, Fusarium avenaceum, Microdochium nivale), snow rot (incolia, arnata, Micronectriella nivalis), naked scab (Ustilago nuda, Ustilago tritici, Ustilago nigra, Ustilago avenae),
- the plant diseases targeted by the plant protective agent of the present invention are preferably gray mold disease (Botrytis cinerea) and pea brown spot disease (Mycosphaerella pinodes).
- OUMS1-BIOX was prepared by culturing OUMS1 using the following culture solution (SIGP medium (Sawayama et al., Curr Microbiol (2011) 63: 173-180)).
- Test example 1 antibacterial / cytotoxicity test of OUMS1-BIOX
- Botrytis cinerea is a polycytic important plant pathogen that occurs in the process of cultivation, distribution and storage of flower buds and sugar beets. It is easy to develop drug resistance and causes great damage all over the world. Pea brown rot fungus is the most important disease of international peas, and no completely resistant varieties have been found.
- Figures 1 and 2 show the images observed with an optical microscope (Olympus IX70-22FL / PH) 16 hours after inoculation.
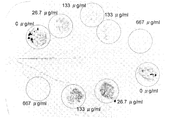
- the germination rate of gray mold and brown pea fungus (number of spores that formed germ tubes ⁇ total number of spores ⁇ The result of 100) is shown in FIG.
- Test example 2 effect on pathogen invasion behavior
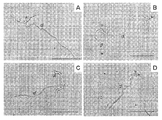
- the onion model epidermis Using the onion model epidermis, the effects of gray mold on germination, attachment formation, and invasion behavior (perforation) were investigated.
- the epidermis of 1 cm 2 onion was peeled off, and low molecular weight antibacterial substances were removed with 70% ethanol. After washing with water, float on 100 ⁇ l of distilled water placed on a slide glass, put 5 ⁇ l of 0, 0.08, 0.4, 2 mg / ml OUMS1-BIOX solution on this, and conidial spores of gray mold 10 ⁇ l (10 5 spores / ml) was inoculated (cultured at 23 ° C. for 1 month under a BL lamp on PDA medium). The sample was allowed to stand at 23 ° C. for 18 hours under wet chamber conditions, then stained with cotton blue, and observed under an optical microscope. The observation image of the light microscope is shown in FIG. 4, and the result of the invasion rate of the mold fungus fungus into the onion epidermis cells (the number of spores that have successfully penetrated ⁇ the number of spores that have formed an attachment ⁇ 100) is shown in FIG.
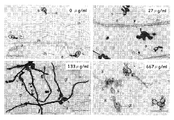
- Test example 3 (inoculation experiment) The lesion formation of brown spot fungus on OUMS1-BIOX-treated pea leaves was investigated.
- Test Example 4 Induction of defense-related gene expression by OUMS1-BIOX
- OUMS1-BIOX was found to have an activity of significantly suppressing the invasion behavior of pathogenic bacteria, but its effect on plants was unknown. Therefore, the expression of defense-related genes in Arabidopsis thaliana Col-0 3 hours after OUMS1-BIOX treatment was analyzed by RT-PCR. 40 ⁇ l of OUMS1-BIOX prepared at 2 mg / ml was added to the 6-week-old leaves after sowing, and fixed with liquid nitrogen after 3 hours, the Trizol method (TRIzol Total RNA was extracted with reagents (Life Technologies).
- Test Example 5 (Effect of natural BIOX on invasion behavior of pathogenic bacteria) BIOX produced by L. cholodnii OUMS1 was found to have an inhibitory effect on the invasion of spores from pathogenic fungi. It was decided to investigate whether or not there is a water purification tank installed in the Faculty of Agriculture of Okayama University and BIOX collected from a water purification plant in Joyo City, Kyoto Prefecture. Represented as Ou-Biox and Kyoto Biox, respectively. Each natural BIOX used was prepared according to the methods of Patent Documents 2 and 3.
- FIG. 8 shows the results of the rate of invasion of the brown spot fungus into the onion epidermis cells (the number of spores that successfully invaded / the number of spores that formed an attachment device ⁇ 100).
- the brown mold fungus Mycosphaerella pinodes spores in the water-treated control group formed an appendage immediately after germination and immediately invaded the epidermal cells.
- the invasion rate was significantly suppressed although an appendage was formed immediately after germination.
- Example 1 (dispersion in the field) Suspend 10 g of BIOX collected from OUMS1-BIOX or the water purification tank in the Faculty of Agriculture, Okayama University with 10 L of water and mix thoroughly. Put this in a sprayer and spray on the crop: Komatsuna (14 days after sowing) under the following conditions. As a result, an excellent inhibitory effect against gray mold and anthrax is obtained. Spraying water: 1L / 25 m 2 corresponds Example 2 (spraying in the field) Suspend 10 g of BIOX collected from OUMS1-BIOX or the water purification tank in the Faculty of Agriculture, Okayama University with 10 L of water and mix thoroughly.
- Spray water volume 1L / 25 m 2 equivalent
- Example 3 Suspend 10 g of BIOX collected from OUMS1-BIOX or the water purification tank in the Faculty of Agriculture, Okayama University with 10 L of water and mix thoroughly. Put this in a sprayer and spray on the crop: cucumber (28 days after sowing) under the following conditions. As a result, an excellent effect of suppressing anthrax is obtained.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Zoology (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- Plant Pathology (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Wood Science & Technology (AREA)
- Environmental Sciences (AREA)
- Inorganic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Virology (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
(I-1) 非晶質及び/又は微結晶性のケイ素及びリン含有酸化鉄を含む植物保護剤。
(I-2) 前記酸化鉄が、鉄及び酸素を主成分として含有し、
鉄、ケイ素及びリンの元素比率が原子数%で各々66~87:2~27:1~32(鉄、ケイ素及びリンの原子数%の合計は100)である、
(I-1)に記載の植物保護剤。
(I-3) 前記酸化鉄が、更に0.1~5重量%の炭素を含有する、(I-1)又は(I-2)に記載の植物保護剤。
(I-4) 前記微結晶性酸化鉄が、フェリハイドライト及び/又はレピドクロサイトである、(I-1)~(I-3)のいずれか一項に記載の植物保護剤。
(I-5) 前記酸化鉄が、鉄酸化細菌によって生成された酸化鉄である、(I-1)~(I-4)のいずれか一項に記載の植物保護剤。
(I-6) 前記酸化鉄が、鉄バクテリア浄水法によって生じた凝集沈殿物から分離された酸化鉄である、(I-5)に記載の植物保護剤。
(I-7) 前記鉄酸化細菌が、レプトスリックス属(Leptothrix)及び/又はガリオネラ属(Gallionella)に属する細菌である、(I-5)又は(I-6)に記載の植物保護剤。
(I-8) 前記鉄酸化細菌が、レプトスリックス・コロディニ(Leptothrix cholodnii) OUMS1(NITE BP-860)である、(I-5)又は(I-6)に記載の植物保護剤。
(I-9) 鉄酸化細菌によって生成された酸化鉄と水との懸濁液から分離した上澄み液を含有する、(I-5)~(I-8)のいずれか一項に植物保護剤。
(I-10) 水和剤の形態である、(I-1)~(I-9)のいずれか一項に記載の植物保護剤。
(I-11) 散布剤の形態である、(I-1)~(I-9)のいずれか一項に記載の植物保護剤。
(I-12) 非晶質及び/又は微結晶性のケイ素及びリン含有酸化鉄の植物保護剤としての使用。
(II-1) (I-1)~(I-11)のいずれか一項に記載の植物保護剤を施用する工程を備えた、植物病害の防除方法。
本発明における酸化鉄の合成法としては、鉄化合物、ケイ素化合物、リン化合物を次のように反応させることを例示できる。
鉄酸化細菌としては、非晶質及び/又は微結晶性のケイ素及びリン含有酸化鉄を形成するものであればよく、特に限定されるものではない。鉄酸化細菌としては、例えば、トキソシリックス属細菌(Toxothrix sp.)、レプトスリックス属細菌(Leptothrix sp.)、クレノシリックス属細菌(Crenothrix sp.)、クロノシリックス属細菌(Clonothrix sp.)、ガリオネラ属細菌(Gallionella sp.)、シデロカプサ属細菌(Siderocapsa sp.)、シデロコッカス属細菌(Siderococcus sp.)、シデロモナス属細菌(Sideromonas sp.)、プランクトミセス属細菌(Planktomyces sp.)などを挙げることができる。
1 OUMS1-BIOXの作成
OUMS1-BIOXは、OUMS1を以下の培養液(SIGP培地(Sawayama et al., Curr Microbiol (2011) 63: 173-180))を用いて培養することにより作成した。
Na2SiO3-9H2O 0.2 g
CaCl2-2H2O 0.044 g
MgSO4-7H2O 0.041 g
グルコース 1 g (終濃度0.1%)
ペプトン 1 g (終濃度0.1%)
Na2HPO2-12H2O 0.076 g
KH2PO4-2H2O 0.02 g
HEPES 2.383 g
FeSO4 0.05 mmol
上で得られたOUMS1-BIOXの乾燥標品2 mgを秤量し、1 mlの蒸留水を加えた後、20分間超音波処理してコロイド様溶液を調製した。これら一連の操作でも、一部沈殿が認められたので、以下の試験例では水に分散できた上澄みだけを用いた。
植物保護への応用の可能性を探る目的で、灰色かび病菌ボトリティス・シネレア(Botrytis cinerea)やエンドウ褐紋病菌ミコスファエレラ・ピノデス(Mycosphaerella pinodes)の形態形成(発芽や侵入行動)に及ぼす影響について調べた。ボトリティス・シネレアは、花卉及び蔬菜の栽培や流通及び保存の過程で発生する多犯性の重要植物病原菌で、薬剤耐性も発達し易く、世界中で大きい被害を及ぼしている。また、エンドウ褐紋病菌は、国際的なエンドウの最重要病害で、これに対する完全な抵抗性品種は見出されていない。
タマネギのモデル表皮を用いて、灰色かび病菌の発芽、付着器形成、侵入行動(穿孔)に対する影響を調べた。
OUMS1-BIOX処理エンドウ葉における褐紋病菌の病斑形成について調べた。
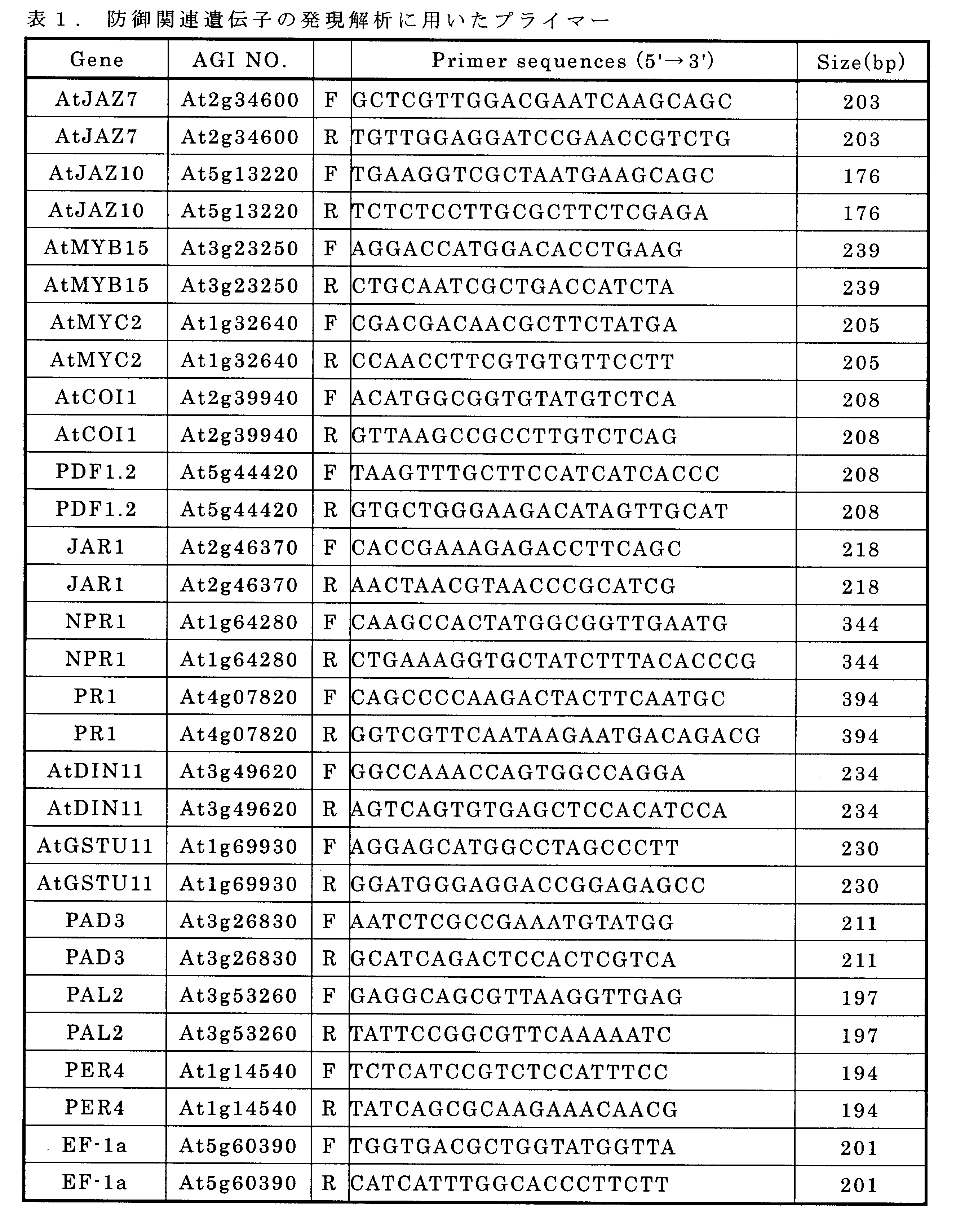
上記の様にOUMS1-BIOXは、病原菌の侵入行動を顕著に抑制する活性が認められたが、植物に対する効果は不明であった。そこで、OUMS1-BIOXを処理して3時間後のシロイヌナズナCol-0における防御関連遺伝子発現についてRT-PCR法によって解析した。播種後6週齢の葉に2 mg/mlに調製したOUMS1-BIOXを40μl添加し、3時間後に液体窒素で固定した後、トリゾール法(TRIzol 試薬;Life Technologies)で全RNAを抽出した。RNA (500 ng)にOligo-dT プライマー、Nuclease free waterを加えた後に70℃10分→直ぐに氷上で急冷して熱変性を行った後に逆転写酵素(タカラ)、逆転写用バッファー、dNTPを加えて逆転写反応(42℃、60分)を行いcDNAを得た。得られたcDNAを用い、表1に記載したプライマーを用いてRT-PCR(Promega、GoTaq Green Master Mix)を行い、各遺伝子の発現量を調べた。各遺伝子の発現量の結果を図7に示す。
レプトスリックス・コロディニ(L. cholodnii) OUMS1が生成するBIOXには病原菌胞子からの侵入を阻害する作用があることが判明したが、この効果が自然界に生息する酸化鉄生成菌由来の天然のBIOXにも見られるのかについて、岡山大学農学部内に設置された浄水タンク及び京都府城陽市内の浄水場から採取したBIOXを使って調べることとした。それぞれ、Ou-Biox及び京都Bioxと表記する。なお、使用したそれぞれの天然BIOXは特許文献2及び3の方法に従って調製した。
OUMS1-BIOX又は岡山大学農学部内浄水タンクから採取したBIOX 10 gを10Lの水で懸濁し十分に混合する。これを、噴霧器に入れ、作物:コマツナ(播種後、14日後)に対して以下の条件で散布を行う。結果として、優れた灰色かび病や炭疽病の抑制効果が得られる。
散布水量:1L/25 m2相当
実施例2(圃場での散布)
OUMS1-BIOX又は岡山大学農学部内浄水タンクから採取したBIOX 10 gを10Lの水で懸濁し十分に混合する。これを、噴霧器に入れ、作物:エンドウ(播種後、28日後)に対して以下の条件で散布を行う。結果として、優れた褐紋病の抑制効果が得られる。
散布水量:1L/25 m2相当
実施例3(圃場での散布)
OUMS1-BIOX又は岡山大学農学部内浄水タンクから採取したBIOX 10 gを10Lの水で懸濁し十分に混合する。これを、噴霧器に入れ、作物:キュウリ(播種後、28日後)に対して以下の条件で散布を行う。結果として、優れた炭疽病の抑制効果が得られる。
散布水量:1L/25 m2相当
Claims (12)
- 非晶質及び/又は微結晶性のケイ素及びリン含有酸化鉄を含む植物保護剤を施用する工程を備えた、植物病害の防除方法。
- 前記酸化鉄が、鉄及び酸素を主成分として含有し、
鉄、ケイ素及びリンの元素比率が原子数%で各々66~87:2~27:1~32(鉄、ケイ素及びリンの原子数%の合計は100)である、
請求項1に記載の防除方法。 - 前記酸化鉄が、更に0.1~5重量%の炭素を含有する、請求項1又は2に記載の防除方法。
- 前記微結晶性酸化鉄が、フェリハイドライト及び/又はレピドクロサイトである、請求項1~3のいずれか一項に記載の防除方法。
- 前記酸化鉄が、鉄酸化細菌によって生成された酸化鉄である、請求項1~4のいずれか一項に記載の防除方法。
- 前記酸化鉄が、鉄バクテリア浄水法によって生じた凝集沈殿物から分離された酸化鉄である、請求項5に記載の防除方法。
- 前記鉄酸化細菌が、レプトスリックス属(Leptothrix)及び/又はガリオネラ属(Gallionella)に属する細菌である、請求項5又は6に記載の防除方法。
- 前記鉄酸化細菌が、レプトスリックス・コロディニ(Leptothrix cholodnii) OUMS1(NITE BP-860)である、請求項5又は6に記載の防除方法。
- 前記植物保護剤が、鉄酸化細菌によって生成された酸化鉄と水との懸濁液から分離した上澄み液を含有する、請求項5~8のいずれか一項に防除方法。
- 前記植物保護剤が、水和剤の形態である、請求項1~9のいずれか一項に記載の防除方法。
- 前記植物保護剤が、散布剤の形態である、請求項1~9のいずれか一項に記載の防除方法。
- 非晶質及び/又は微結晶性のケイ素及びリン含有酸化鉄を含む植物保護剤。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015506747A JP6366073B2 (ja) | 2013-03-19 | 2014-03-14 | 植物保護剤及び植物病害の防除方法 |
| US14/778,140 US20160278383A1 (en) | 2013-03-19 | 2014-03-14 | Plant protection agent and method for controlling plant disease |
| EP14770794.7A EP2987409B1 (en) | 2013-03-19 | 2014-03-14 | Plant protection agent and method for controlling plant disease |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-056674 | 2013-03-19 | ||
| JP2013056674 | 2013-03-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014148396A1 true WO2014148396A1 (ja) | 2014-09-25 |
Family
ID=51580080
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/056963 Ceased WO2014148396A1 (ja) | 2013-03-19 | 2014-03-14 | 植物保護剤及び植物病害の防除方法 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20160278383A1 (ja) |
| EP (1) | EP2987409B1 (ja) |
| JP (1) | JP6366073B2 (ja) |
| WO (1) | WO2014148396A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019167338A (ja) * | 2018-03-23 | 2019-10-03 | 国立大学法人山口大学 | 植物病害の抑制剤及び植物病害の抑制方法 |
| JP2021169378A (ja) * | 2020-04-14 | 2021-10-28 | 国立大学法人 岡山大学 | チューブ状酸化鉄粒子、その製造方法、及び、抗菌性赤色顔料 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10338526A (ja) | 1997-06-04 | 1998-12-22 | Yoshida Seibutsu Kenkyusho:Kk | パイプ状微粒子酸化鉄の製造方法 |
| JP2000044414A (ja) * | 1998-07-24 | 2000-02-15 | Taki Chem Co Ltd | 植物保護剤 |
| JP2003137703A (ja) * | 2001-11-06 | 2003-05-14 | Dainichiseika Color & Chem Mfg Co Ltd | 植物の有害生物防除剤および有害生物防除方法 |
| JP2005272251A (ja) | 2004-03-25 | 2005-10-06 | Japan Science & Technology Agency | 鞘状酸化鉄粒子の生産方法、及びその利用 |
| WO2010110435A1 (ja) | 2009-03-27 | 2010-09-30 | 国立大学法人岡山大学 | 有機・無機複合材料及びその製造方法 |
| WO2011074587A1 (ja) | 2009-12-15 | 2011-06-23 | 国立大学法人岡山大学 | 磁性セラミックス及びその製造方法 |
| WO2011074586A1 (ja) | 2009-12-15 | 2011-06-23 | 国立大学法人岡山大学 | 酸化物の生成能を有する新規微生物 |
| WO2012124703A1 (ja) | 2011-03-15 | 2012-09-20 | 国立大学法人岡山大学 | 新規多孔質アモルファスシリカ及びその製造方法 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001503724A (ja) * | 1996-06-14 | 2001-03-21 | ドズネラドゼ、デビッド | リン酸塩組成物とその利用 |
-
2014
- 2014-03-14 US US14/778,140 patent/US20160278383A1/en not_active Abandoned
- 2014-03-14 WO PCT/JP2014/056963 patent/WO2014148396A1/ja not_active Ceased
- 2014-03-14 JP JP2015506747A patent/JP6366073B2/ja active Active
- 2014-03-14 EP EP14770794.7A patent/EP2987409B1/en not_active Not-in-force
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10338526A (ja) | 1997-06-04 | 1998-12-22 | Yoshida Seibutsu Kenkyusho:Kk | パイプ状微粒子酸化鉄の製造方法 |
| JP2000044414A (ja) * | 1998-07-24 | 2000-02-15 | Taki Chem Co Ltd | 植物保護剤 |
| JP2003137703A (ja) * | 2001-11-06 | 2003-05-14 | Dainichiseika Color & Chem Mfg Co Ltd | 植物の有害生物防除剤および有害生物防除方法 |
| JP2005272251A (ja) | 2004-03-25 | 2005-10-06 | Japan Science & Technology Agency | 鞘状酸化鉄粒子の生産方法、及びその利用 |
| WO2010110435A1 (ja) | 2009-03-27 | 2010-09-30 | 国立大学法人岡山大学 | 有機・無機複合材料及びその製造方法 |
| WO2011074587A1 (ja) | 2009-12-15 | 2011-06-23 | 国立大学法人岡山大学 | 磁性セラミックス及びその製造方法 |
| WO2011074586A1 (ja) | 2009-12-15 | 2011-06-23 | 国立大学法人岡山大学 | 酸化物の生成能を有する新規微生物 |
| WO2012124703A1 (ja) | 2011-03-15 | 2012-09-20 | 国立大学法人岡山大学 | 新規多孔質アモルファスシリカ及びその製造方法 |
Non-Patent Citations (7)
| Title |
|---|
| "Environmental Microorganism Pictorial Book", 1995, KODANSHA, LTD. |
| HORTICULTURAL RESEARCH ( JAPAN), vol. 11+2, 2012, pages 198, XP008181171 * |
| R.A. EGGLETON; R.W. FITZPATRICK: "New data and a revised structural model for ferrihydrite", CLAYS AND CLAY MINERALS, vol. 36, no. 2, 1988, pages 111 - 124 |
| SAWAYAMA ET AL., CURR MICROBIOL, vol. 63, 2011, pages 173 - 180 |
| SAWAYAMA ET AL., CURR. MICROBIOL., vol. 63, 2011, pages 173 - 180 |
| See also references of EP2987409A4 |
| THE JAPANESE SOCIETY FOR HORTICULTURAL SCIENCE CHU SHIKOKU SHIBU KENKYU HAPPYO YOSHI, no. 49, 2010, pages 21, XP008181913 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019167338A (ja) * | 2018-03-23 | 2019-10-03 | 国立大学法人山口大学 | 植物病害の抑制剤及び植物病害の抑制方法 |
| JP7233690B2 (ja) | 2018-03-23 | 2023-03-07 | 国立大学法人山口大学 | 植物病害の抑制剤及び植物病害の抑制方法 |
| JP2021169378A (ja) * | 2020-04-14 | 2021-10-28 | 国立大学法人 岡山大学 | チューブ状酸化鉄粒子、その製造方法、及び、抗菌性赤色顔料 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2987409A4 (en) | 2016-10-12 |
| EP2987409A1 (en) | 2016-02-24 |
| JP6366073B2 (ja) | 2018-08-01 |
| US20160278383A1 (en) | 2016-09-29 |
| JPWO2014148396A1 (ja) | 2017-02-16 |
| EP2987409B1 (en) | 2018-10-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Tseng et al. | An endophytic Trichoderma strain promotes growth of its hosts and defends against pathogen attack | |
| Shang et al. | Copper sulfide nanoparticles suppress Gibberella fujikuroi infection in rice (Oryza sativa L.) by multiple mechanisms: contact-mortality, nutritional modulation and phytohormone regulation | |
| Abd-Alla et al. | Assessment of silver nanoparticles contamination on faba bean-Rhizobium leguminosarum bv. viciae-Glomus aggregatum symbiosis: implications for induction of autophagy process in root nodule | |
| Hussain et al. | Nano-managing silver and zinc as bio-conservational approach against pathogens of the honey bee | |
| WO2021153759A1 (ja) | 植物病害防除組成物及び植物病害防除方法 | |
| Baka et al. | Antifungal activity of silver/silicon dioxide nanocomposite on the response of faba bean plants (Vicia faba L.) infected by Botrytis cinerea | |
| Hamed et al. | Green production of silver nanoparticles, evaluation of their nematicidal activity against Meloidogyne javanica and their impact on growth of faba bean | |
| Thamke et al. | Evaluation of risk assessment of new industrial pollutant, ionic liquids on environmental living systems | |
| BR112017014230B1 (pt) | Combinação sintética e métodos para preparar uma semente compreendendo uma população de endófitos, e para modular uma característica de planta | |
| JP2013502902A (ja) | フザリウムおよびフザリウムマイコトキシンに対する生物的制御剤 | |
| CA3204746A1 (en) | Antifungal methylobacterium compositions and methods of use | |
| Scholz et al. | Verticillium dahliae-Arabidopsis interaction causes changes in gene expression profiles and jasmonate levels on different time scales | |
| WO2012020772A1 (ja) | 植物病害防除組成物およびその用途 | |
| Andargie et al. | Arabidopsis thaliana: a model host plant to study plant–pathogen interaction using rice false smut isolates of Ustilaginoidea virens | |
| JP2018002646A (ja) | 植物用薬剤 | |
| CN108347945B (zh) | 抗植物致病组合物 | |
| Cabot et al. | Salinity is a prevailing factor for amelioration of wheat blast by biocontrol agents | |
| WO2021153758A1 (ja) | 植物病害防除組成物及び植物病害防除方法 | |
| Sima et al. | Efficiently reducing the plant growth inhibition of CuO NPs using rice husk-derived biochar: experimental demonstration and mechanism investigation | |
| JP6366073B2 (ja) | 植物保護剤及び植物病害の防除方法 | |
| Maffei et al. | Electrophysiology and plant responses to biotic stress | |
| Zhou et al. | Biological control of the native endophytic fungus Pochonia chlamydosporia from the root nodule of Dolichos lablab on Fusarium wilt of banana TR4 | |
| Aseel et al. | Biosynthesized silver nanoparticles mediated by Ammi visnaga extract enhanced systemic resistance and triggered multiple defense-related genes, including SbWRKY transcription factors, against tobacco mosaic virus infection | |
| JP2024501816A (ja) | フサリシジンa、フサリシジンb及び殺真菌剤を含む混合物及び組成物 | |
| JP2012188367A (ja) | 植物の菌類病害防除剤および菌類病害防除方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14770794 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2015506747 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14778140 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014770794 Country of ref document: EP |