WO2014180020A1 - 具有增效作用的杀菌组合物 - Google Patents
具有增效作用的杀菌组合物 Download PDFInfo
- Publication number
- WO2014180020A1 WO2014180020A1 PCT/CN2013/077180 CN2013077180W WO2014180020A1 WO 2014180020 A1 WO2014180020 A1 WO 2014180020A1 CN 2013077180 W CN2013077180 W CN 2013077180W WO 2014180020 A1 WO2014180020 A1 WO 2014180020A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polyhexamethylene
- melon
- wettable powder
- polyhexamethylene biguanide
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/40—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having a double or triple bond to nitrogen, e.g. cyanates, cyanamides
- A01N47/42—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having a double or triple bond to nitrogen, e.g. cyanates, cyanamides containing —N=CX2 groups, e.g. isothiourea
- A01N47/44—Guanidine; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/18—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/50—1,3-Diazoles; Hydrogenated 1,3-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/54—1,3-Diazines; Hydrogenated 1,3-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
- A01N43/653—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/84—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms six-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,4
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/28—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N<
- A01N47/38—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N< containing the group >N—CO—N< where at least one nitrogen atom is part of a heterocyclic ring; Thio analogues thereof
Definitions
- the invention belongs to the field of agricultural plant protection, in particular to a bactericidal composition with improved performance,
- bactericidal composition that is included.
- Polyhexamethylene 3 ⁇ 41 melon is a safe and efficient broad-spectrum antibacterial agent developed by Aveciago Company of the United Kingdom. It is colorless and odorless, low in dosage, widely used in toxic drugs, fast in action, and in a small amount. Low, and can form a layer of cation on the surface of the article for a long time to inhibit the bacteria, and will not produce antibacterial bacteria characteristics, its safety I" is recognized by the US FDA and EPA, its semi-lethal dose to mice LD50>5000mg/kg, which is an actual non-toxic grade.
- polyhexamethylene 3 ⁇ 41 melon is used as a cationic bactericide in agricultural applications, it can quickly adsorb to the surface of bacterial cells, rapidly attacking and destroying the plasma membrane, and the components of the medulla are leaking. In order to achieve the best bactericidal and sterilizing effect, the pathogenic bacteria and virus sedge are removed from the root. Common derivatives such as poly(hexamethylene biguanide hydrochloride).
- Prochloraz is a broad-spectrum fungicide that has significant control effects on a variety of crops caused by ascomycetes and deuteromycetes, as well as most fungicides, fungicides, insecticides, Herbicides are mixed and have good control effects. It has a therapeutic and eradicative effect on a variety of diseases on field crops, fruits and vegetables, turf and ornamental plants.
- Epoxiconazol is a systemic trifing fungicide. Its mechanism of action is to inhibit the synthesis of ergosterol, which hinders the formation of cell walls. It not only has good protection, treatment and eradication activity, but also has systemic and better residual active epoxiconazole, which can increase the activity of the fungi of the crop, lead to the contraction of the fungal suction device and inhibit the invasion of the bacteria. This is the fluorine ring. Frustrated in all three frustrations. Leaf spot, powdery mildew, rust on bananas, onions, celery, kidney beans, melons, asparagus, peanuts, beets, etc. And the diseases such as anthracnose and white rot on the grapes have good control effects.
- Benthiviricarb-isopropyl is a kind of phenothiamine which has strong prevention, treatment, osmotic activity and good persistence and rain repellency.
- fentanyl was effective at controlling the late blight of potatoes and tomatoes, downy mildew of grapes and other crops at lower doses.
- the combination of thiazide and other fungicides will also have a very good effect.
- Zoxamide is a new type of broad-spectrum fungicide mainly used for the prevention and treatment of various bacteria such as yellow jHH mildew, pear black scab, apple scab, citrus anthracnose, grape black pox, etc. , fungal diseases.
- the mechanism of bactericidal action mainly includes destroying the nuclear structure of the pathogen, causing it to lose the heart part, failing to die and interfering with the metabolism of the pathogenic cells, causing its physiological disorder and eventually leading to death.
- Azoxystrobin is a methoxy acrylate fungicide with high efficiency and broad spectrum. It is a disease of almost all fungi (Ascomycota, Basidiomycotina, Flagellate, and Deuteromyces). Such as powdery mildew, rust, sclerotium, net blotch, downy mildew, rice blast, etc. have good activity. It can be used for stem and leaf spray, seed treatment, and soil treatment. It is mainly used for cereals, 7J rice, peanuts, grapes, potatoes, fruit trees, vegetables, coffee, lawns, etc.
- Prothioconazole is a new broad-spectrum trithione bactericide developed by Bayer. It is mainly used to control many diseases such as cereals and wheat bean crops. Propionate is low in toxicity and non- teratogenic. , mutagenic, non-toxic to embryos, safe for humans and the environment. The mechanism of action is to inhibit demethylation at the 14 position of the precursor of sterol in the fungus, lanosterol or 2,4-methylenedihydrolane.
- Difenoconazole is a three-frozen fungicide with systemic activity. It is a sterol demethylation inhibitor. It can be internally absorbed into the leaves of plants and has a high redistribution ability. It can be ffiih. The infection and prevention of the spread of lesions have better protection and therapeutic effects. It has special effects on powdery mildew, rust, leaf spot, scab, scab, and anthrax in various crops.
- fenamidone The mechanism and characteristics of fenamidone are similar to those of famoxadone and methoxy acrylate fungicides, «1; at the level of the original threat of hydrogenated genomic Q-cytochrome C ⁇ Block electron transfer to suppress line bold breathing.
- fenamidone Suitable for wheat, cotton, grapes, tobacco, lawn, sunflower, rose Rose, potato, tomato and so on. All kinds of downy mildew, plague, Phytophthora, sputum, black spot, spot rot, etc.
- Polyoxin is a metabolite produced by Streptomyces aureus and belongs to the antibiotic antibiotics. It has a systemic conduction of 13 ⁇ 4. The mechanism of action is to interfere with the biosynthesis of the chitin of the cell wall of the pathogen, so that the cell wall of the cell cannot be biosynthesized and the pathogen is killed. After the germ tube and mycelium insecticide, the part is enlarged, ruptured, and overflows with the contents of the cell, but cannot develop normally, leading to death. Because 13 ⁇ 43 ⁇ 4 has the effect of inhibiting the spore formation of diseased bacteria and the enlargement of lesions.
- Iprodione is a high-efficiency, broad-spectrum, contact-killing fungicide with dimethyl-betaamine, which has a certain therapeutic and protective effect, and can also be absorbed by the roots for systemic action. It can effectively prevent fungi that are resistant to benzimidazole systemic fungicides. It is suitable for controlling early defoliation, gray mold, early blight and other diseases of various fruit trees, vegetables, melons and other crops.
- Acibenzolar is a highly efficient, broad-spectrum, new fungicide.
- strawberry powdery mildew, melon powdery mildew, cucumber powdery mildew, pear black spot disease ⁇ ! Harm has good control effect.
- Activated esters not only have a broad spectrum of bactericidal activity, but also have good protection and therapeutic effects. It has no cross-resistance with other commonly used fungicides and has a longer duration than conventional fungicides. It is highly selective, safe for crops, humans and animals, and beneficial to the environment, and has no pollution to the environment.
- Dithianon is a protective fungicide for many leaf diseases of many pome fruits and stone fruits, and has a multi-action mechanism.
- a series of fungal enzymes are inhibited by reacting with sulfur-containing groups and interfering with cellular respiration, which ultimately leads to disease death. It has a good protective activity and also has a certain therapeutic activity.
- Suitable crops include prunes and stone fruits such as apples, pears, peaches, apricots, cherries, citrus, coffee, grapes, strawberries, hops, etc.
- the control object can control almost all fruit tree diseases such as black spot disease, mildew disease, leaf spot disease, rust, charcoal surface, scab, downy mildew, brown rot and so on.
- Pyraclostrobin is a new broad-spectrum fungicide. Mechanism of action: It is a mitochondrial respiratory inhibitor. That is, by preventing electron transfer in cytochrome synthesis. It has protection, treatment and leaf penetration conduction. The results of field efficacy test of pyraclostrobin cream showed cucumber powdery mildew, downy mildew and banana black star Disease, leaf spot disease has a control effect.
- Trifloxystrobin is a new class of fluorochemical fungicides successfully developed from the natural product Strobilurins as a bactericide lead compound. It has the characteristics of high efficiency, broad spectrum, protection, treatment, eradication, infiltration, systemic activity, rain-resistant scouring and long-lasting effect. It is effective against strains which are resistant to 1, 4-demethylase inhibitors, benzamides, dicarboxyamines and benzidine, and have no cross-resistance with existing fungicides.
- Picoxystrobin is a systemic fungicide. It is widely used to control wheat leaf foliar diseases such as leaf blight, leaf rust, wing blight, brown spot, powdery mildew, etc. Compared with other methoxy acrylate fungicides, P-oxygenate has a stronger therapeutic effect on wheat leaf blast, net blotch and moiré.
- Fluazinam is a 2,6-dinitroaniline compound and is a protective fungicide.
- the disease caused by gray grape cells can be controlled by the dosage of 50 ⁇ 100g (ai) /100L.
- This product belongs to the genus Alternaria, Botrytis, Phytophthora, Trichoderma, Sclerotinia and Black genus
- the bacterium is very effective, and it has good effects against benzopyrimidine and dicarboximide-like fungicides. It is resistant to rain erosion, has a long-lasting effect, and has excellent control of the growth of food plants. It also has excellent control effect on cruciferous plant tuberculosis, and also has good control effect on rice blast disease caused by Rhizopus.
- Thifluzamide is a thiazolamide fungicide with strong systemic conductivity and long-lasting properties.
- Thioflavone is a succinate dehydrogenase inhibitor. Due to its high fluorine content, it is highly competitive in biochemical processes and is difficult to recover once it is combined with a substrate or enzyme.
- Thiofuramide is active against pathogenic fungi such as Rhizoctonia, Puccinia, Black powder, Sphaerotheca, Vola, and genus, especially for basidiomycetes. Diseases such as sheath blight and blight have special effects.
- Dimethomorph is a broad-spectrum bactericide of morpholine, which has a unique action on the fungi of the genus Aspergillus and the genus Phytophthora, mainly causing the decomposition of the sporangia wall, thus making the fungus# 3 ⁇ 4 dead.
- Dimethomorph is a fungicide of the genus Oomycetes. Its function is to destroy the formation of cell wall membrane, and it has effects on all stages of the life history of oocysts, especially in the stage of sporangia and oospore formation. At very low concentrations, «0.50 ⁇ g/ml) is inhibited, and there is no resistance to 3 ⁇ 4S with phenylindoles.
- the technical problem to be solved by the invention is: for the anti-f and soil residue problems of the bactericide in practical application, the bactericides of two different sterilization principles are screened to compound, so as to improve the control effect of the bactericide and delay the generation of resistance, Reduce the amount of application, control costs.
- the present invention provides a synergistic bactericidal composition
- the active ingredient A is a polyhexamethylene view or a 3 ⁇ 4 ⁇ 4 farmer 4 ⁇ 1: acceptable salt compound
- active component B is selected from the group consisting of cyprodinil, epoxiconazole, phenothimethamine, fenhexamide, azoxystrobin, prothioconazole, difenoconazole , imidacloprid, polymycin, iprodione, activated ester, dicyandiamide, pyraclostrobin, trifloxystrobin, P-oxypide, fluazinam, thiafuramide, dimethomorph A fungicide in the middle.
- the composition of the present invention mainly comprises A and B as active components, and may also be composed only of A and B.
- the weight ratio between the components of component A and component B is 1: 50 to 50: 1, preferably 1: 30 to 30: 1, further male 1: 10 to 10: 1 .
- the polyhexamethylene biguanide of the present invention may exist in the form of its salt, and the difference in the control effect of the compound of polyhexamethylene biguanide and its salt form is not obvious.
- Polyhexamethylene 3 ⁇ 41 melon salt suitable for the present invention is polyhexamethylene 3 ⁇ 41 melon hydrochloride, polyhexamethylene 3 ⁇ 41 melon nitrate, polyhexamethylene 3 ⁇ 41 melon carbonate, polyhexamethylene Base 3 ⁇ 41 guaranium, polyhexamethylene 3 ⁇ 4 3 ⁇ 43 ⁇ 4 acid salt, poly hexa One of methyl 3 ⁇ 41 melon stearate, polyhexamethylene 3 ⁇ 41 melon vinegar. Volt-selected polyhexamethylene 3 ⁇ 4) 3 ⁇ 43 ⁇ 4, as shown below.
- the bactericidal composition of the present invention may further comprise an active component and a pesticide adjuvant, wherein the amount of the active component may be selected according to specific conditions, such as an active component having a mass content of 6 to 92% and a pesticide of 94 to 8%. Auxiliary ingredients.
- the bactericidal composition is prepared from the active ingredient together with the pesticide adjuvant into a pesticide-acceptable dosage form such as a powder or a wettable powder.
- the present invention provides the use of a bactericidal composition
- a bactericidal composition comprising component A (polyhexamethylene guanidine or its agriculturally acceptable salt compound) and component B for controlling crop diseases in agricultural fields, especially in the prevention and treatment Use of yellow mold or wheat rust.
- the application method for controlling crop diseases can be selected by soaking seeds or spraying with water on the surface of the crop during the crop growth period according to different control diseases, or smearing on the surface of the control.
- compositions further comprise a carrier, an adjuvant and/or a surfactant.
- a carrier an adjuvant and/or a surfactant.
- Common auxiliaries can be mixed during the application.
- Suitable auxiliaries may be solid or liquid, they are usually used in the processing of dosage forms, such as natural or recycled minerals, mu dispersants, wetting agents, adhesives, thickeners, adhesives .
- the method of application of the compositions of the present invention comprises the use of the compositions of the present invention in aerial parts of plants, particularly leaves or foliage.
- the frequency of administration and the amount administered will depend on the biological and climatic conditions of the pathogen.
- the plant growth site such as rice fields, may be wetted with a liquid formulation of the composition, or the composition may be applied to the soil in solid form, such as in the form of granules (soil application), the composition may be passed from the soil to the plant through the roots of the plant. In vivo (systemic action), or through seed coating, soaking methods to eradicate and prevent the occurrence of diseases.
- compositions may be applied only with the active ingredient, or may be mixed with the additives.
- the compositions of the present invention can be prepared in a variety of dosage forms such as wettable powders, suspensions, oil suspensions, water-dispersible granules, aqueous emulsions, microemulsions.
- the compositions may be applied by spraying, misting, dusting, spreading or pouring, and the like.
- composition of the present invention can be prepared into various dosage forms by a known method, and the active ingredient and the auxiliary agent, such as a solid carrier, can be uniformly mixed with a surfactant and ground, if necessary, to prepare a preparation.
- auxiliary agent such as a solid carrier
- the above solvent may be selected from aromatic hydrocarbons, ⁇ «8-12 carbon atoms, such as xylene mixture or substituted benzene, phthalate esters, such as dibutyl phthalate or dicaprylic acid, , such as ring burned or paraffin, alcohol and ethylene glycol and their ethers and esters, such as ethanol, ethylene glycol, ethylene glycol monomethyl; ketones, such as cyclohexanone, strong polar solvents, such as N-A Base-2-pyrrolidone, dimethyl sulfoxide or dimethylformam furnace, and vegetable or vegetable oils such as soybean oil.
- aromatic hydrocarbons ⁇ «8-12 carbon atoms
- phthalate esters such as dibutyl phthalate or dicaprylic acid
- alcohol and ethylene glycol and their ethers and esters such as ethanol, ethylene glycol, ethylene glycol monomethyl
- ketones such as cyclohexanone
- strong polar solvents such as N-A Base
- the above solid carriers are usually natural mineral fillers such as talc, kaolin, montmorillonite or activated clay.
- a highly dispersible silicic acid or a highly dispersible adsorbent polymer carrier such as a particulate adsorbent carrier or a non-adsorbing carrier, and the particulate adsorbent carrier is porous, such as pumice, bentonite or strontium.
- a non-adsorbing carrier such as calcite or sand.
- a large amount of a pre-granulated material of inorganic or organic nature can be used as a carrier, in particular dolomite.
- the surfactant is lignosulfonic acid, naphthalenesulfonic acid, phenolsulfonic acid, alkaline earth metal salt or amine salt, mercaptoarylsulfonate, mercaptosulfur , mercapto mmit, fatty alkylate, fatty acid and sulfated fatty alcohol glycol ether, condensate of sulfonated naphthalene and naphthalene derivatives with formaldehyde, condensate of naphthalene or naphthalenesulfonic acid with phenol and formaldehyde, poly Oxyethylene octyl phenyl ether, ethoxylated isooctyl phenol, octyl phenol, nonyl phenol, alkyl aryl polyglycol ether, tributyl benzene polyglycol ether
- the two active ingredients in the compositions of the present invention exhibit synergistic effects, the activity of which is more pronounced than the expected sum of activity using a single compound, and the individual activity of a single compound.
- the synergistic effect is manifested by allowing a reduced application rate, a broader fungicidal control profile, quicker effect, longer lasting control effect, better control of plant harmful fungi by only one or a few applications, and broadening of possible application. Intervals. These properties are particularly desirable in the practice of plant fungi control.
- the other characteristics exhibited by the bactericidal composition of the present invention are mainly as follows: 1.
- the compounding of the composition of the present invention has obvious synergistic effect; 2. Since the chemical composition of the two single agents of the composition is greatly different, the effect The mechanism is completely different, there is no cross-resistance, and the problem of resistance caused by single use alone can be delayed; 3.
- the composition of the invention has good safety and control effect on crops. It has been verified by i that the fungicidal composition of the present invention has stable chemical properties, remarkable synergistic effect, and exhibits obvious synergistic effect and complementary effect on the control object. With ⁇ l 3 ⁇ 43 ⁇ 4
- the active ingredient polyhexamethylene 3 ⁇ 4
- ketone polymycin, iprodione, activated ester, dicyandiamide, pyraclostrobin, trifloxystrobin, p-oxypide, fluazinam, thiafuramide, dimethomorph One type, fully mixed with various auxiliaries and fillers, etc., and pulverized by an ultrafine pulverizer to obtain a wettable powder.
- Hungry Example 1 62% polyhexamethylene salt salt ship cyprodinil WP Polyhexamethylene 3 ⁇ 4) 3 ⁇ 43 ⁇ 4 ship 60%, cyprodinil 2%, burnt naphthalene sulfonate 4%, 12-alkyl sulfonium
- Hungry case 3 65% polyhexamethylene watch ship ⁇ cyprodinil WP
- Polyhexamethylene 3 ⁇ 4 3 ⁇ 4 ship 2%, cyprodinil 63%, lignin sulfonate 5%, methylnaphthalene sulfonate sodium formate condensate 7 Q / Q , sodium decyl sulfate 3 Q / Q , Silicon 3 ⁇ 4 ⁇ complement to 100 Q /o.
- Example 5 30% polyhexamethylene face vinegar. epoxiconazole wettable powder
- Example 6 75% polyhexamethylene carbonate, epoxiconazole wettable powder
- the soil is made up to 100%.
- Example 7 62% polyhexamethylene 3 ⁇ 4) 3 ⁇ 43 ⁇ 4 ⁇ phenothiamine wettable powder
- Example 8 40% polyhexamethylene 3 ⁇ 4
- Example 9 75% polyhexamethylene surface ⁇ phenothiamine wettable powder
- Example 10 85% polyhexamethylene 3 ⁇ 43 ⁇ 43 ⁇ 4 acid salt. Benzoyl WP wettable powder
- Example 11 60% polyhexamethylene surface ⁇ Benzamidamide wettable powder
- Example 12 75% polyhexamethylene surface sulfate ⁇ Benzoyl wettable powder
- Example 13 88% polyhexamethylene guanidate ⁇ azoxystrobin WP
- Example 14 50% polyhexamethylene surface ⁇ Azoxystrobin wettable powder
- Example 17 50% polyhexamethylene surface ⁇ Prothioconazole wettable powder
- Example 18 85% polyhexamethylene hard fat ship ⁇ prothioconazole wettable powder
- Example 20 50% polyhexamethylene 3 ⁇ 4M carbonate ⁇ Difenoconazole wettable powder
- Hungry Case 21 86% polyhexamethylene 3 ⁇ 4M3 ⁇ 4 acid salt. Difenoconazole wettable powder
- Polyhexamethylene 3 ⁇ 4) 3 ⁇ 43 ⁇ 4 ship 60%, Iprodione 2%, thiophene 3%, carboxymethyl starch sodium 1%, 12% sulfonate 4%, lignin sulfonene 4%, 1% xanthan gum, attapulgite to 100%.
- Example 29 40% polyhexamethylene hard fat ship ⁇ Isobacteria wettable powder
- Example 31 85% polyhexamethylene 3 ⁇ 4 3 ⁇ 4 acid salt ⁇ Activated ester wettable powder
- Example 32 60% polyhexamethylene surface ⁇ Activated ester wettable powder
- Example 33 78% polyhexamethylene surface sulfate ⁇ Activated ester wettable powder
- Example 34 88% polyhexamethylene guanidate ⁇ dicyandione wettable powder
- Example 37 85% polyhexamethylene 3 ⁇ 4
- Example 38 50% polyhexamethylene 3 ⁇ 4
- Example 40 88% polyhexamethylene 3 ⁇ 4
- Example 41 50% polyhexamethylene carbonate, chytrid WP
- Example 42 86% polyhexamethylene 3 ⁇ 4 3 ⁇ 4 acid salt ⁇ Fusarium ester wettable powder
- Hungry case 44 40% polyhexamethylene melon hard fat ship ⁇ picoxystrobin wettable powder
- Polyhexamethylene 3 ⁇ 41 melon 2%, picoxystrobin 73%, methyl naphthalene sulfonate formaldehyde condensate 2%, sodium lignosulfonate 5%, decyl thiophene 3%, diatomaceous earth make up to 100%.
- Example 46 85% polyhexamethylene 3 ⁇ 43 ⁇ 43 ⁇ 4 acid salt. Fluidine amine wettable powder
- Polyhexamethylene 3 ⁇ 4) 3 ⁇ 43 ⁇ 4 ship 83%, fluazinam 2%, polyoxyethylene octylphenyl ether 1%, sodium lignosulfonate 5%, white carbon black 3%, diatomaceous earth to 100% .
- Example 49 82% polyhexamethylene 3 ⁇ 4
- Example 50 50% polyhexamethylene surface ⁇ Thiofuramide wettable powder
- Example 51 82% polyhexamethylene hard fat ship ⁇ Thiofan earning wettable powder Polyhexamethylene 3 ⁇ 41 melon hard fat ship 2%, thiofuran 80%, lignin sulfonate 4%, alumina 5%, polyoxyethylene octyl phenyl ether 2%, attapulgite to 100 %.
- Example 52 85% polyhexamethylene 3 ⁇ 4M ⁇ dimethomorph wettable powder
- Example 54 75% polyhexamethylene salt salt ship ⁇ Dimethomorph wettable powder
- Polyhexamethylene 3 ⁇ 41 ship 2%, dimethomorph 73%, pull-open powder 1%, mercapto polyoxyethyl ether sulfonate
- Test object Yellow mold pathogen
- the incidence of cucumber leaves was investigated, and the disease index and control effect were calculated.
- the control effect is converted into the probability value (y)
- the liquid height (g/ml) is converted into a logarithmic value (X)
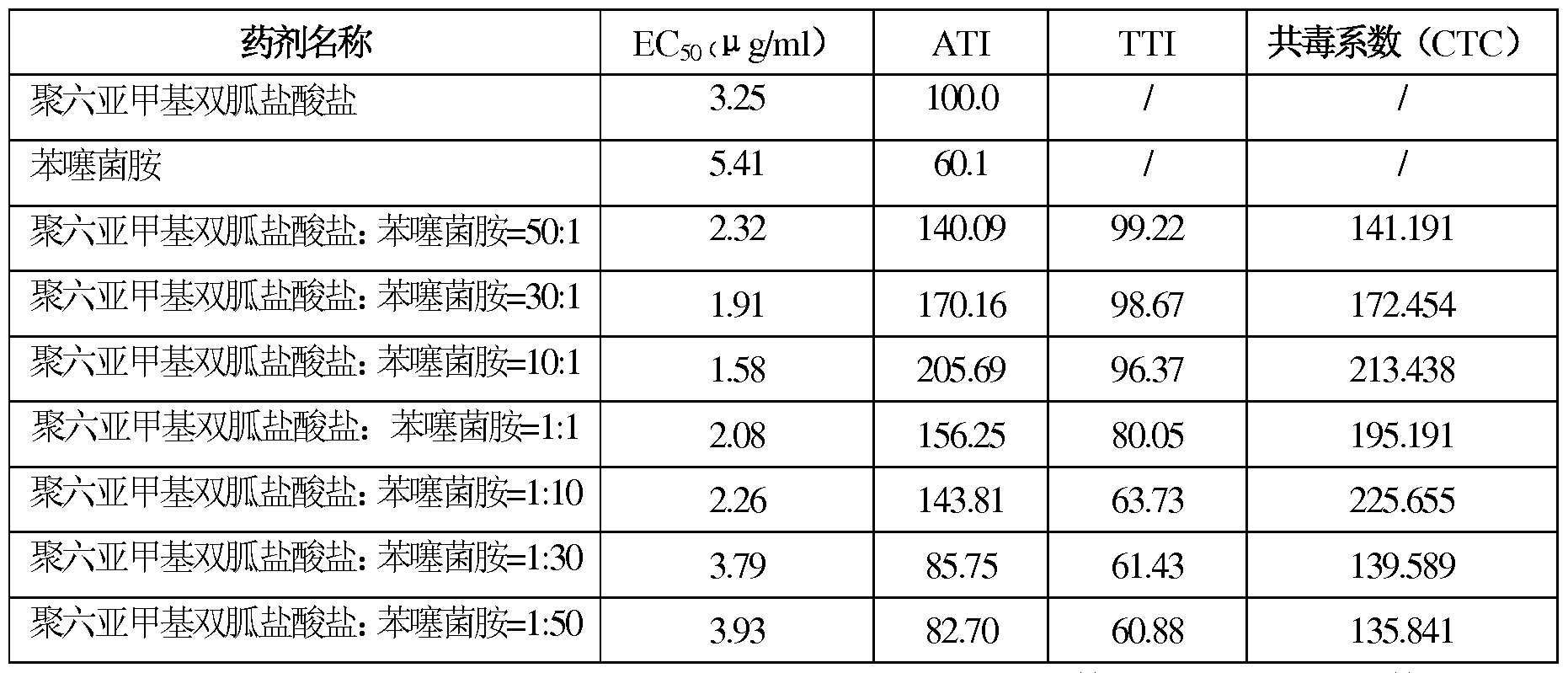
- the virulence equation is calculated by the least squares method and the neutral concentration EC50 is suppressed, and the virulence of the drug is calculated according to the method of Sun Yunpei. Exponential Co-toxicity Coefficient (CTC).
- Measured virulence index (ATI) (standard drug EC50 / test drug EC50) *100
- the ratio of 3 ⁇ 4M3 ⁇ 4 acid salt to flucyclazole is between 1:30 and 30:1, and the co-toxicity coefficient of polyhexamethylene 3 ⁇ 43 ⁇ 4M3 ⁇ 4 3 ⁇ 4 and epoxiconazole is above 120, and the synergistic effect is obvious.
- the effect of treatment is significantly improved, indicating that there is a significant synergistic effect between the two stalks of wheat rust, especially the ratio of polyhexamethylene 3 ⁇ 41 and tilfuramide in the ratio of 1:30-30:1.
- the co-toxicity coefficient of methyl 3 ⁇ 41 acid salt and tilfuramide is above 120, and the synergistic effect is obvious.
- Test method In the early stage of the disease, the first spray was immediately performed, and after 7 days, the second application was carried out, each treatment of 4 plots, 20 square meters per plot. The incidence of the disease was investigated before the drug and 10 days after the second drug. The plot was randomly sampled at 5 points, and 5 crops were investigated at each point. The percentage of the lesion area per leaf on the whole plant was counted and graded, and the disease index was calculated. And control effects.
- Anticipated effect (%) X+Y-XY/100 (where X, ⁇ is a single dose)
- Grade 1 Leaf lesions less than 5, length less than lcm; Grade 3: 6-10 leaf lesions, some lesions are longer than lcm; Grade 5: 11-25 leaf lesions, some lesions are connected into pieces The lesion area accounts for 10-25% of the leaf area; Grade 7: More than 26 leaf lesions, the lesions are connected into pieces, and the lesion area accounts for 26-50% of the leaf area; Grade 9: The lesions are connected into pieces. The area of the lesion is more than 50% of the leaf area or the whole leaf is dead.
- Polyhexamethylene biguanide or its salt is combined with cyprodinil (or cyprodinil), trifloxystrobin, picoxystrobin, fluazinam, thiazolamide, etc. to control rice blast Field efficacy test
- Example 33 20% polyhexamethylene biguanide 3.5 3.73 24.49 7.1 15% activated ester wettable powder 131.5 3.32 4.62 80.3 Expected control effect after mixing 81.7
- Example 41 20% polyhexamethylene biguanide 67.5 3.68 13.99 46.2 30% trifloxystrobin suspension 67.5 4.06 16.24 43.4 anti-effect 69.5 after mixing
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP13884051.7A EP2995200B1 (en) | 2013-05-07 | 2013-06-13 | Sterilization composition with synergism |
| EP19170011.1A EP3566579B1 (en) | 2013-05-07 | 2013-06-13 | Fungicidal composition having synergistic effect |
| EA201592133A EA028248B1 (ru) | 2013-05-07 | 2013-06-13 | Фунгицидный состав синергетического действия |
| ES13884051T ES2741438T3 (es) | 2013-05-07 | 2013-06-13 | Composición de esterilización con sinergismo |
| US14/889,924 US9867373B2 (en) | 2013-05-07 | 2013-06-13 | Fungicidal composition having synergistic effect |
| PL13884051T PL2995200T3 (pl) | 2013-05-07 | 2013-06-13 | Kompozycja do sterylizacji o synergicznym działaniu |
| AU2013388780A AU2013388780B2 (en) | 2013-05-07 | 2013-06-13 | Sterilization composition with synergism |
| CA2911669A CA2911669C (en) | 2013-05-07 | 2013-06-13 | Fungicidal composition having synergistic effect |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201310165069.5A CN103229779B (zh) | 2013-05-07 | 2013-05-07 | 具有增效作用的杀菌组合物 |
| CN201310165069.5 | 2013-05-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014180020A1 true WO2014180020A1 (zh) | 2014-11-13 |
Family
ID=48877913
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2013/077180 Ceased WO2014180020A1 (zh) | 2013-05-07 | 2013-06-13 | 具有增效作用的杀菌组合物 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US9867373B2 (zh) |
| EP (2) | EP2995200B1 (zh) |
| CN (6) | CN104365648B (zh) |
| AU (1) | AU2013388780B2 (zh) |
| CA (3) | CA3038363C (zh) |
| EA (1) | EA028248B1 (zh) |
| ES (1) | ES2741438T3 (zh) |
| PL (1) | PL2995200T3 (zh) |
| WO (1) | WO2014180020A1 (zh) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10743535B2 (en) | 2017-08-18 | 2020-08-18 | H&K Solutions Llc | Insecticide for flight-capable pests |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104642334A (zh) * | 2013-11-15 | 2015-05-27 | 南京华洲药业有限公司 | 一种含丙硫菌唑和嘧菌环胺的杀菌组合物及其应用 |
| CN104170833B (zh) * | 2014-09-02 | 2016-01-13 | 海南正业中农高科股份有限公司 | 含寡糖与聚六亚甲基双胍或其盐的组合物 |
| CN104886123B (zh) * | 2015-06-17 | 2017-05-17 | 广西田园生化股份有限公司 | 一种含毒氟磷和氟啶胺的杀菌组合物及其杀菌剂 |
| WO2018060101A1 (de) * | 2016-09-30 | 2018-04-05 | Flechsig Patent Company Llc | Verfahren zum schutz von pflanzen vor schädlingen unter verwendung einer pestizid-zusammensetzung mit einstellbarer persistenz und abbaubarkeit |
| CN107372568A (zh) * | 2017-07-18 | 2017-11-24 | 广东中迅农科股份有限公司 | 一种杀菌组合物 |
| CN107736370A (zh) * | 2017-10-17 | 2018-02-27 | 惠州市无龄康态健康科技有限公司 | 一种增效杀菌剂 |
| CN111466402A (zh) * | 2019-01-24 | 2020-07-31 | 陕西嘉益蓝德生物工程有限公司 | 一种含有聚六亚甲基双胍盐酸盐和春雷霉素的杀菌组合物及用途 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1335062A (zh) * | 1993-06-25 | 2002-02-13 | 拜尔公司 | 杀真菌组合物 |
| CN1409596A (zh) * | 1999-12-13 | 2003-04-09 | 拜尔公司 | 杀真菌活性化合物的组合 |
| WO2011030094A2 (en) * | 2009-09-09 | 2011-03-17 | Syngenta Limited | Fungicidal mixtures |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2003265309B2 (en) * | 2002-07-26 | 2009-10-29 | Microban Products Company | Durable antimicrobial leather |
| US7074459B2 (en) * | 2003-05-23 | 2006-07-11 | Stockel Richard F | Method for preserving wood |
| WO2006094127A1 (en) * | 2005-03-02 | 2006-09-08 | Osmose, Inc. | Wood preservative formulations comprising dichlorophen |
| US20060276468A1 (en) * | 2005-05-12 | 2006-12-07 | Blow Derek P | Wood preservative formulations comprising Imazalil |
| US20070048344A1 (en) * | 2005-08-31 | 2007-03-01 | Ali Yahiaoui | Antimicrobial composition |
| EP2071954A1 (de) * | 2007-12-19 | 2009-06-24 | Bayer CropScience AG | Verwendung von polymeren Guanidin-Derivaten zum Bekämpfen von unerwünschten Mikro-organismen im Pflanzenschutz |
| GB0903375D0 (en) * | 2009-02-27 | 2009-04-08 | Bio Technics Ltd | Disinfectant composition comprising a biguanide compound |
| WO2010134279A1 (ja) * | 2009-05-20 | 2010-11-25 | 日本曹達株式会社 | エマルションまたはマイクロエマルション製剤調製用組成物 |
| CN101962442B (zh) * | 2009-07-21 | 2013-03-27 | 铜陵高聚生物科技有限公司 | 一种盐酸聚六亚甲基双胍的制备方法 |
| CN102461653B (zh) * | 2010-11-14 | 2013-04-24 | 中国科学院华南植物园 | 聚六亚甲基双胍盐酸盐在防治柑桔酸腐病上的应用及其保鲜剂 |
| CN102067857A (zh) * | 2011-02-23 | 2011-05-25 | 陕西美邦农药有限公司 | 一种含有多抗霉素的杀菌组合物 |
| US10016354B2 (en) * | 2011-03-23 | 2018-07-10 | Basf Se | Compositions containing polymeric, ionic compounds comprising imidazolium groups |
| CN102283251B (zh) * | 2011-07-04 | 2013-03-13 | 朱晓鸣 | 一种农用杀菌液 |
-
2013
- 2013-05-07 CN CN201410084232.XA patent/CN104365648B/zh active Active
- 2013-05-07 CN CN201410083182.3A patent/CN103975944B/zh active Active
- 2013-05-07 CN CN201410082931.0A patent/CN103975942B/zh active Active
- 2013-05-07 CN CN201410084231.5A patent/CN104365647B/zh active Active
- 2013-05-07 CN CN201410083022.9A patent/CN103975943B/zh active Active
- 2013-05-07 CN CN201310165069.5A patent/CN103229779B/zh active Active
- 2013-06-13 EP EP13884051.7A patent/EP2995200B1/en not_active Not-in-force
- 2013-06-13 EP EP19170011.1A patent/EP3566579B1/en active Active
- 2013-06-13 CA CA3038363A patent/CA3038363C/en active Active
- 2013-06-13 CA CA2911669A patent/CA2911669C/en active Active
- 2013-06-13 ES ES13884051T patent/ES2741438T3/es active Active
- 2013-06-13 US US14/889,924 patent/US9867373B2/en not_active Expired - Fee Related
- 2013-06-13 PL PL13884051T patent/PL2995200T3/pl unknown
- 2013-06-13 CA CA2989158A patent/CA2989158C/en active Active
- 2013-06-13 EA EA201592133A patent/EA028248B1/ru unknown
- 2013-06-13 AU AU2013388780A patent/AU2013388780B2/en not_active Ceased
- 2013-06-13 WO PCT/CN2013/077180 patent/WO2014180020A1/zh not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1335062A (zh) * | 1993-06-25 | 2002-02-13 | 拜尔公司 | 杀真菌组合物 |
| CN1409596A (zh) * | 1999-12-13 | 2003-04-09 | 拜尔公司 | 杀真菌活性化合物的组合 |
| WO2011030094A2 (en) * | 2009-09-09 | 2011-03-17 | Syngenta Limited | Fungicidal mixtures |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2995200A4 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10743535B2 (en) | 2017-08-18 | 2020-08-18 | H&K Solutions Llc | Insecticide for flight-capable pests |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2013388780A1 (en) | 2015-11-26 |
| CN104365648A (zh) | 2015-02-25 |
| CN103975942A (zh) | 2014-08-13 |
| CA2989158C (en) | 2019-05-21 |
| EP3566579A2 (en) | 2019-11-13 |
| CN103975944A (zh) | 2014-08-13 |
| EP3566579A3 (en) | 2020-01-08 |
| US9867373B2 (en) | 2018-01-16 |
| CA2911669A1 (en) | 2014-11-13 |
| EP3566579B1 (en) | 2022-08-10 |
| CN104365648B (zh) | 2016-06-01 |
| EA201592133A1 (ru) | 2016-09-30 |
| US20160113280A1 (en) | 2016-04-28 |
| CN103229779B (zh) | 2014-09-17 |
| CN103229779A (zh) | 2013-08-07 |
| EP2995200B1 (en) | 2019-06-05 |
| AU2013388780B2 (en) | 2017-03-30 |
| EP2995200A4 (en) | 2017-02-08 |
| CN103975942B (zh) | 2015-08-26 |
| CN104365647A (zh) | 2015-02-25 |
| CN103975944B (zh) | 2015-08-26 |
| CA2911669C (en) | 2018-01-23 |
| PL2995200T3 (pl) | 2019-12-31 |
| EA028248B1 (ru) | 2017-10-31 |
| CA2989158A1 (en) | 2014-11-13 |
| ES2741438T3 (es) | 2020-02-11 |
| CA3038363C (en) | 2022-03-15 |
| CN103975943A (zh) | 2014-08-13 |
| CN103975943B (zh) | 2015-08-26 |
| EP2995200A1 (en) | 2016-03-16 |
| CN104365647B (zh) | 2016-06-01 |
| CA3038363A1 (en) | 2014-11-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN104094947B (zh) | 具有增效作用的杀菌组合物 | |
| WO2014180020A1 (zh) | 具有增效作用的杀菌组合物 | |
| WO2014026396A1 (zh) | 具有增效作用的杀菌组合物 | |
| US10694746B2 (en) | Fungicidal composition | |
| US11219213B2 (en) | Fungicidal composition | |
| WO2018006462A1 (zh) | 一种杀菌剂组合物 | |
| WO2018054396A1 (zh) | 一种杀菌剂组合物 | |
| CN104365649B (zh) | 具有增效作用的杀菌组合物 | |
| EP3679795A1 (en) | Bactericidal agent composition and method for controlling crop disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13884051 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2911669 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14889924 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013884051 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2013388780 Country of ref document: AU Date of ref document: 20130613 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 201592133 Country of ref document: EA |