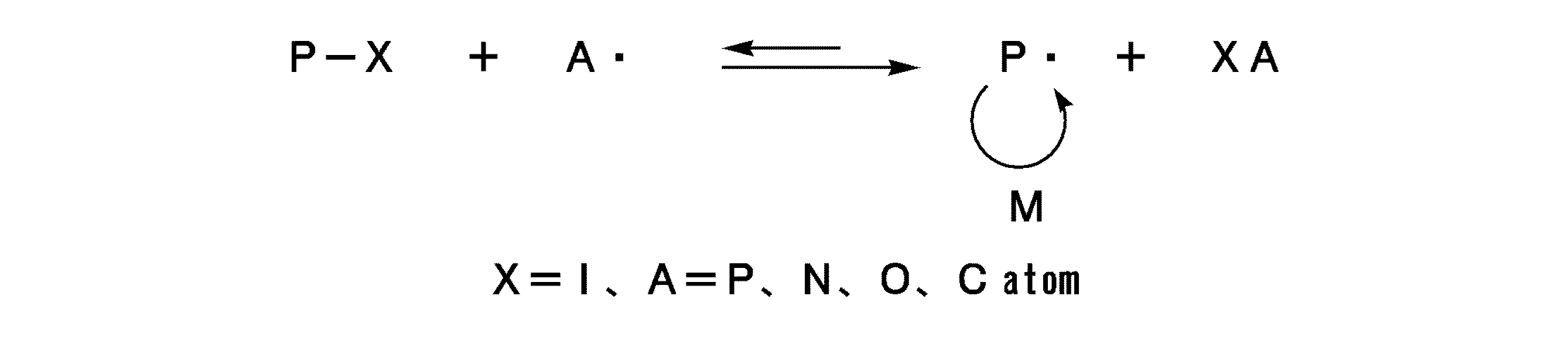WO2015152189A1 - 易分散性セルロース組成物の製造方法、易分散性セルロース組成物、セルロース分散樹脂組成物及びセルロース用の水系の分散処理剤の製造方法 - Google Patents
易分散性セルロース組成物の製造方法、易分散性セルロース組成物、セルロース分散樹脂組成物及びセルロース用の水系の分散処理剤の製造方法 Download PDFInfo
- Publication number
- WO2015152189A1 WO2015152189A1 PCT/JP2015/060029 JP2015060029W WO2015152189A1 WO 2015152189 A1 WO2015152189 A1 WO 2015152189A1 JP 2015060029 W JP2015060029 W JP 2015060029W WO 2015152189 A1 WO2015152189 A1 WO 2015152189A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cellulose
- resin
- polymer dispersant
- composition
- segment
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L1/00—Compositions of cellulose, modified cellulose or cellulose derivatives
- C08L1/02—Cellulose; Modified cellulose
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F293/00—Macromolecular compounds obtained by polymerisation on to a macromolecule having groups capable of inducing the formation of new polymer chains bound exclusively at one or both ends of the starting macromolecule
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J3/00—Processes of treating or compounding macromolecular substances
- C08J3/02—Making solutions, dispersions, lattices or gels by other methods than by solution, emulsion or suspension polymerisation techniques
- C08J3/03—Making solutions, dispersions, lattices or gels by other methods than by solution, emulsion or suspension polymerisation techniques in aqueous media
- C08J3/05—Making solutions, dispersions, lattices or gels by other methods than by solution, emulsion or suspension polymerisation techniques in aqueous media from solid polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/17—Amines; Quaternary ammonium compounds
- C08K5/175—Amines; Quaternary ammonium compounds containing COOH-groups; Esters or salts thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/36—Sulfur-, selenium-, or tellurium-containing compounds
- C08K5/41—Compounds containing sulfur bound to oxygen
- C08K5/42—Sulfonic acids; Derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L101/00—Compositions of unspecified macromolecular compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/04—Homopolymers or copolymers of ethene
- C08L23/06—Polyethylene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2301/00—Characterised by the use of cellulose, modified cellulose or cellulose derivatives
- C08J2301/02—Cellulose; Modified cellulose
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2323/00—Characterised by the use of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Derivatives of such polymers
- C08J2323/02—Characterised by the use of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Derivatives of such polymers not modified by chemical after treatment
- C08J2323/04—Homopolymers or copolymers of ethene
- C08J2323/06—Polyethene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2401/00—Characterised by the use of cellulose, modified cellulose or cellulose derivatives
- C08J2401/02—Cellulose; Modified cellulose
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2453/00—Characterised by the use of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives of such polymers
Definitions
- the present invention is attracting attention for its function as an excellent filler, it is a hydrophilic substance, so that it is difficult to disperse in a resin or the like, and at present, a wide range of fine cellulose fibers whose use is not promoted.
- a method for producing an easily dispersible cellulose composition in which cellulose is treated with a polymer dispersant to increase the resin dispersibility of cellulose The present invention relates to a method for producing a water-based dispersion treatment agent used in the above, an easily dispersible cellulose composition, and a technique for providing a cellulose-dispersed resin composition using the same.
- Cellulose fiber is a basic skeletal material of all plants, has accumulated over a trillion tons on the earth, and is a resource that can be regenerated by tree planting, so its effective use is desired.
- Cellulose fibers are fibers that are 5 times stronger than steel and have a low linear thermal expansion coefficient that is 1/50 that of glass, despite being 1/5 lighter than steel.
- Patent Document 1 a technique has been proposed in which cellulose fibers are contained as a filler in a matrix such as a resin to impart mechanical strength.
- Patent Document 1 In order to further improve the mechanical strength of the cellulose fiber, the cellulose fiber is defibrated so that the cellulose nanofiber (CNF, microfibrillated plant fiber) is dispersed in the additive.
- CNF cellulose nanofiber
- CNF cellulose nanocrystals
- CNF is a fiber obtained by subjecting cellulose fibers to a treatment such as mechanical defibration, and is a fiber having a fiber width of about 4 to 100 nm and a fiber length of about 5 ⁇ m or more.
- CNC is a crystal obtained by subjecting cellulose fibers to chemical treatment such as acid hydrolysis, and is a crystal having a crystal width of about 10 to 50 nm and a crystal length of about 500 nm.
- CNF and CNC are collectively referred to as nanocellulose.
- Nanocellulose has a high specific surface area (250 to 300 m 2 / g), is lighter and has higher strength than steel.
- Nanocellulose is less thermally deformed than glass.
- Nanocellulose which has high strength and low thermal expansion, is a material that is useful as a sustainable resource material.
- composite materials, airgel materials, and CNCs that combine nanocellulose and polymer materials such as resins to achieve high strength and low thermal expansion.
- Development and creation of highly functional materials by introducing functional functional groups into nanocellulose, an optically anisotropic material using chiral nematic liquid crystal phase by self-organization of the material.
- nanocellulose since nanocellulose has abundant hydroxyl groups, it has an aspect that is inferior in compatibility with a versatile resin that is hydrophilic and strongly polar, hydrophobic and nonpolar.
- Non-Patent Document 1 a surfactant is adsorbed on cellulose nanocrystals (cellulose nanowhiskers) to improve the organic solvent dispersibility of cellulose nanocrystals.
- Non-Patent Document 2 an isotactic polypropylene (iPP) composite material using cellulose nanocrystals adsorbing a surfactant as a reinforcing material is produced, and the tensile strength is improved by about 1.4 times compared to iPP alone. Yes.
- iPP isotactic polypropylene
- the cellulose fiber when cellulose is used as a reinforcing material for a thermoplastic resin, the cellulose fiber is hydrophilic and specified for the purpose of suppressing the generation of cellulose agglomerates and uniformly dispersing the cellulose in the resin.
- An additive having a HLB value (hydrophilic / lipophilic balance) (low molecular surfactant) is used, and the cellulose fibers are dispersed in the additive.
- JP 2008-266630 A International Publication No. 2012/111408
- polymer design with a wide variety of structures is possible by monomer design, so molecular design according to purpose and application becomes possible. That is, since an infinite number of structures can be designed as a polymer dispersant, synthesis of a higher performance dispersant adapted to the type of resin to be dispersed can be expected by monomer design. It is considered that various types of polymer dispersants such as olefin polymers, acrylic polymers, ester polymers, urethane polymers, and the like can be used. Among them, particularly acrylic polymers can be polymerized under mild conditions and can be obtained relatively easily. In addition, since there are a wide variety of acrylic monomers, an infinite number of compositions are selected. Since it is easy to design a molecule according to the purpose and application, it is expected to be more useful.
- polymer dispersants such as olefin polymers, acrylic polymers, ester polymers, urethane polymers, and the like can be used. Among them, particularly acrylic polymers can be polymerized under mild conditions
- the block copolymer has a structure in which two or more types of polymer segments with different constituent components are contained in one polymer chain, so that different functionality can be given to each polymer segment by devising the monomer composition.
- the polymer segment A has a component having high affinity with a general-purpose resin.
- the block copolymer can be designed so as to have a highly adsorbing component with cellulose as the polymer segment B (B chain), it can be used as a dispersant to Are considered to act effectively, and it can be expected to suppress aggregation and stabilize the dispersion of cellulose in the general-purpose resin. That is, when a block copolymer having a resin affinity segment A and a cellulose adsorptive segment B is used as a dispersant for a cellulose-dispersed resin composition, the dispersibility of cellulose is improved and the mechanical strength is sufficiently increased as a filler. I can expect that.
- the present inventors considered that a method of treating the polymer dispersant with cellulose is important in order to fully exhibit the function of the polymer dispersant.
- a method of treating the polymer dispersant with cellulose is important in order to fully exhibit the function of the polymer dispersant.
- it is a prerequisite for achieving good dispersion to suppress the generation of cellulose agglomerates. Therefore, as a pretreatment method for obtaining a cellulose-dispersed resin composition that has achieved a good dispersion state, it is important how to treat the polymer dispersant on cellulose without aggregating the cellulose.
- the present invention has been made in view of the above-mentioned background, and the purpose thereof is conventionally without performing surface modification of nanocellulose or introduction of a functional group into nanocellulose as in the prior art.
- Dispersion with a polymeric dispersant which has been developed to disperse fine hydrophobic substances such as pigments, can be applied to cellulose, which is a hydrophilic substance, by a simple method and use a large amount of organic solvent.
- an easily dispersible cellulose composition that can easily and efficiently treat a polymer dispersant into cellulose and easily disperse cellulose in a hydrophobic substance such as a resin in a system using water as a main medium. It is another object of the present invention to provide a useful cellulose-dispersed resin composition.
- the present inventor has made it easy to use cellulose in a hydrophobic substance such as a resin, which is necessary for obtaining a cellulose-dispersed resin composition using a polymer dispersant.
- a method for obtaining an easily dispersible cellulose composition made dispersible in a simple manner without using a large amount of an organic solvent has been found, whereby a polymer dispersant can be applied to a hydrophilic substance, cellulose.
- the present invention has been achieved by finding that a good cellulose-dispersed resin composition can be obtained while suppressing the generation of cellulose agglomerates.
- the present invention relates to a method for producing an easily dispersible cellulose composition having improved dispersibility of cellulose in a resin, and a block copolymer structure having a resin affinity segment A and a cellulose adsorbing segment B
- a water-based dispersion treatment agent containing the polymer dispersant is prepared by dissolving a polymer dispersant having a hydrophilic organic solvent solution, adding a surfactant thereto, and then adding water.
- a method for producing an easily dispersible cellulose composition characterized in that an easily dispersible cellulose composition is obtained by adding the obtained aqueous dispersion treating agent to water-containing or dried cellulose.
- the surfactant is selected from the group consisting of a carboxylic acid salt of an aliphatic amine, an inorganic acid salt, and a quaternary ammonium salt. It is at least one selected; the hydrophilic organic solvent is an alcohol solvent or a glycol solvent; the cellulose is selected from the group consisting of cellulose nanofiber, cellulose nanocrystal, pulp, lignocellulose, and wood flour. It is mentioned that it is a cellulose fiber of the at least 1 sort (s) of a water-containing state or dry state.
- the polymer dispersant is a block copolymer that further satisfies all the following requirements (1) to (5). It is done. (1) 90% by mass or more of the constituent components of the AB block copolymer are composed of methacrylate monomers; (2) In the cellulose adsorbing segment B, 50% by mass or more of the constituent components are composed of a methacrylate monomer having at least one hydroxyl group and / or a methacrylate monomer having a urea group, and Not compatible; (3) The number average molecular weight in terms of polystyrene in the gel permeation chromatography of the resin affinity segment A is 500 to 20000, and the ratio of the resin affinity segment A in the whole AB copolymer is 5 to 95% by mass; (4) The number average molecular weight in terms of polystyrene in the gel permeation chromatography of the cellulose adsorbing segment
- 70% by mass or more of the constituents of the cellulose adsorbent segment B of (2) described above is a hydroxyl group.
- a methacrylate monomer having at least one and / or a methacrylate monomer having a urea group, and further 3 to 15% by mass of the constituent components contain methacrylic acid and / or carboxy group neutralized with an alkali.
- having a methacrylate monomer having a quaternary ammonium base for example, having a methacrylate monomer having a quaternary ammonium base.
- the polymer dispersant uses an organic iodine compound as a starting compound, and a phosphorus compound, a nitrogen compound, an oxygen compound, or a carbon compound as a catalyst. It is synthesized by a reversible chain transfer catalytic polymerization (RTCP) method which is a living radical polymerization method.
- RTCP reversible chain transfer catalytic polymerization
- the present invention provides, as another embodiment, an easily dispersible cellulose composition characterized by being obtained by any one of the above-described methods for producing an easily dispersible cellulose composition.
- the present invention is a cellulose-dispersed resin composition obtained by melt-kneading a resin composition containing the above-described easily dispersible cellulose composition and a resin.
- the resin composition is obtained by melt-kneading while containing a liquid component derived from the easily dispersible cellulose composition; and the resin is a thermoplastic resin.
- the polymer dispersant having a block copolymer structure having a resin affinity segment A and a cellulose adsorptive segment B is dissolved in a hydrophilic organic solvent solution. Easily dispersible with improved dispersibility of cellulose in resin, characterized by producing a water-based dispersion treatment agent containing a polymer dispersant by adding a surfactant and then adding water A method for producing an aqueous dispersion treatment agent used for producing a cellulose composition is provided.
- an aqueous dispersion treatment agent for cellulose and a readily dispersible cellulose composition which makes it possible to treat a polymer dispersant into cellulose in a medium system. More specifically, an aqueous dispersion treatment containing a polymer dispersant useful for cellulose can be stably produced by a simple method using a surfactant.
- This aqueous dispersion treatment agent is a useful agent that can be used regardless of whether the cellulose is in a water-containing state or a dry state. That is, according to the present invention, the polymer dispersant that has been applied to hydrophobic pigments and the like in the past can be treated with water as a main component solvent in the step of treating the hydrophilic substance cellulose.
- the polymer dispersant can be processed into cellulose while suppressing cellulose aggregation due to the organic solvent, and an easily dispersible cellulose composition can be provided simply and efficiently.
- the present invention will be described in more detail with reference to the best mode for carrying out the invention.
- the feature of the present invention is that an easily dispersible cellulose composition in which dispersibility of cellulose into a resin is improved by the polymer dispersant, which is necessary for obtaining a cellulose-dispersed resin composition using the polymer dispersant. It is in the point which discovered the simple pre-processing method which can provide this. Specifically, a surfactant is used when water is added to a hydrophilic organic solvent solution containing a polymer dispersant having a specific block copolymer structure and a surfactant to form an aqueous dispersion treatment agent.
- an aqueous dispersion treatment agent containing a polymer dispersant that functions effectively with respect to cellulose is obtained by a very simple method in which the addition order is specified.
- an easily dispersible cellulose composition is provided, and by using this easily dispersible cellulose composition and complexing with resin, it is extremely easy to produce cellulose.
- a dispersed resin composition can be obtained.
- the obtained cellulose-dispersed resin composition has a cellulose dispersed well in the resin, and further has improved mechanical strength when injection molded.
- an easily dispersible cellulose composition having excellent compatibility with a resin can be obtained by a very simple method without using a large amount of an organic solvent, a wide range of excellent cellulose as a functional material can be expected. .
- the aqueous dispersion treatment agent for cellulose of the present invention can be prepared by adding water to a hydrophilic organic solvent solution of a polymer dispersant containing a surfactant.
- a polymer dispersant having a specific block copolymer structure is dissolved in a hydrophilic organic solvent solution, a surfactant is added thereto, and then water is added. It is essential to prepare an aqueous dispersion treatment agent containing a molecular dispersant.
- each material to be used will be described.
- the hydrophilic organic solvent used in the present invention may be any solvent as long as it is compatible with water and becomes uniform, but it is particularly preferable to use an alcohol solvent or a glycol solvent.
- a hydrophilic organic solvent solution of the polymer dispersant is used, and the concentration of the polymer dispersant in the organic solvent at that time is 5 to 80 mass. % Is preferred.
- the concentration of the polymer dispersant in the organic solvent is preferably 5 to 80% by mass, more preferably 5 to 50% by mass, and still more preferably 10 to 30% by mass.
- the hydrophilic organic solvent solution of the polymer dispersant used in the present invention the polymerization solution obtained when preparing the polymer dispersant can be used as it is, and in this case, it becomes a simpler method. Moreover, what diluted the polymerization solution with the hydrophilic organic solvent and adjusted to the above-mentioned density
- concentration range can be used. Of course, it is also possible to use a solution obtained by re-dissolving a polymer dispersant once precipitated from a polymerization solution with a hydrophilic organic solvent.
- the present invention is characterized by adding water to a hydrophilic organic solvent solution containing a polymer dispersant having a specific block copolymer structure and a surfactant in a specific method defined in the present invention.
- a hydrophilic organic solvent solution containing a polymer dispersant having a specific block copolymer structure and a surfactant in a specific method defined in the present invention.
- an aqueous dispersion treatment agent for cellulose comprising a polymer dispersant is produced.
- the concentration of the surfactant in the hydrophilic organic solvent solution is preferably 0.01 to 1% by mass. When the concentration is higher than 1% by mass, the content of the surfactant in the cellulose-dispersed resin composition is excessively increased, which acts as a plasticizer and may affect physical properties.
- the concentration of the surfactant in the hydrophilic organic solvent solution is preferably 0.01 to 1% by mass, and more preferably 0.05 to 0.5% by mass.
- the aqueous dispersion treatment agent for cellulose used in the present invention is obtained by adding water to a hydrophilic organic solvent solution of a polymer dispersant containing a surfactant, and the water in the total amount is 50% by mass. It is preferable that the hydrophilic organic solvent is 30% by mass or less. Since the method of the present invention is characterized in that the polymer dispersant is processed into cellulose using water as the main medium to form an easily dispersible cellulose composition, the aqueous dispersion treatment agent for cellulose of the present invention is It is preferable that the content of water is large and the amount of the hydrophilic organic solvent is small.
- the water content in the aqueous dispersion treatment agent for cellulose of the present invention is 50% by mass or more and is hydrophilic.
- the organic solvent is preferably 30% by mass or less. However, these concentrations are not limited.
- the polymer dispersant used in the present invention has a block copolymer structure having a resin affinity segment A and a cellulose adsorptive segment B, and further satisfies the following requirements (1) to (5): A block copolymer satisfying all of them is preferable.
- the polymer dispersant used in the present invention has a block copolymer structure having a resin affinity segment A and a cellulose adsorptive segment B.
- block copolymers in two or more types of monomer components having different properties, polymer segments formed by the monomer components are covalently bonded to each other, and two or more types of polymer segments having different properties are included in one polymer chain.
- a structured structure Taking an AB type block copolymer composed of two types of segments as an example, it has a structure in which polymer segment A and polymer segment B having different properties (functionality) are covalently bonded.
- the block copolymer has a portion where two or more types of monomer components having different properties in the polymer chain are localized for each monomer component, a random copolymer in which each monomer component is randomly arranged and In comparison, it can be expected that the performance of each component will be exhibited more. That is, by making the structure of the dispersant an AB block copolymer having a resin affinity segment A and a cellulose adsorptive segment B, the resin affinity performance and the cellulose adsorption performance are sufficiently exhibited by each polymer chain. Can be expected.
- the structure of the polymer dispersant used is an AB block copolymer structure having a resin affinity segment A and a cellulose adsorbing segment B.
- the present inventors consider the reason why the remarkable effects of the present invention are obtained by the present invention as follows. That is, by using the polymer dispersant having the block copolymer structure described above, when the cellulose is treated with an aqueous dispersion treatment agent for cellulose containing the polymer dispersant obtained as described above, The cellulose adsorbent segment B of the polymer dispersant and the multipoint interaction on the surface of the cellulose effectively coat the dispersant on the cellulose surface.
- the resin affinity segment A of the dispersant having the above structure makes the surface of the cellulose hydrophobic, and can improve the affinity with the cellulose and the resin. It becomes a composition.
- the resin composition obtained by combining with the resin using the composition has excellent dispersibility of cellulose due to the effect of the polymer dispersant, and the polymer dispersant covers the cellulose. It is considered that the strength of the interface between cellulose and resin was increased, and as a result, a resin composition excellent in strength and elastic modulus could be obtained.
- the polymer dispersant that can be used in the present invention is preferably one in which 90% by mass or more of its constituent components are composed of a methacrylate monomer. This is due to the following reasons. A dispersant composed of 90% by mass or more of a methacrylate-based monomer is not only good in the structure of the dispersant, but will be described in detail later, but in order to obtain a polymer dispersant with superior functionality, It is preferable to use a reversible chain transfer catalytic polymerization (RTCP) method, which is a living radical polymerization method, for the synthesis of the agent.
- RTCP reversible chain transfer catalytic polymerization
- the RTCP method can exhibit excellent living polymerizability mainly in methacrylic monomers, has a good polymerization yield, and has a molecular weight. This is because the distribution is narrow and block formation is easy. More specifically, when an acrylic monomer, a styrene monomer, or a vinyl monomer is present, the molecular weight distribution is widened or the polymerization yield is deteriorated.
- the component is preferably 90% by mass or more of a methacrylic monomer.
- the polymer dispersant used in the present invention is composed of 90% by mass or more of a methacrylic monomer, so that the difference in each block structure becomes a clear polymer dispersant, which is more functional. It will be.
- the polymer dispersant used in the present invention is a methacrylate monomer in which 50% by mass or more of the constituent components of the cellulose adsorbing segment B have one or more hydroxyl groups and / or It is preferably composed of a methacrylate monomer having a urea group and not compatible with the resin. This is due to the following reason. One reason is that, as described in the above (1), 90% by mass or more of the constituent components must be methacrylate monomers.
- cellulose has a hydroxyl group in its skeleton, and hydrogen bonds between the hydroxyl groups work, so cellulose is a very strong polymer that is insoluble in water.
- the proportion of the hydroxyl group component and / or urea group in the constituent components of the cellulose adsorbing segment B is preferably 60% by mass or more, and more preferably 70% by mass or more.
- the proportion of the methacrylate monomer having one or more hydroxyl groups constituting the segment B and / or the methacrylate monomer having a urea group is less than 50% by mass, the effect as the cellulose adsorptive segment B is insufficient. This is not preferable because the effect as a dispersant may not be sufficiently exhibited.
- the polymer dispersant used in the present invention preferably has a polystyrene-equivalent number average molecular weight of 500 to 20000 in gel permeation chromatography of resin affinity segment A. Further, in order to show resin affinity with resin (compatibility with resin), it is more preferably about 1000 to 8000. This is a molecular weight region where the resin affinity segment A is considered to have the highest resin affinity efficiency. Further, the proportion of the resin affinity segment A in the entire dispersant is preferably 5 to 95% by mass, more preferably 30 to 70% by mass. If it is less than 5% by mass, the resin affinity component is relatively small, and the resin affinity performance may not be fully exhibited, which is not preferable. On the other hand, when it is larger than 95% by mass, the cellulose adsorbing component is relatively small, and the cellulose adsorbing performance may not be sufficiently exhibited, which is not preferable.
- the polymer dispersant used in the present invention preferably has a polystyrene-equivalent number average molecular weight of 500 to 20000 in the gel permeation chromatography of the resin affinity segment B. Further, in order to show high cellulose adsorptivity with cellulose, it is more preferably about 1000 to 8000, and it is a molecular weight region where the cellulose adsorptive segment B seems to have the highest cellulose adsorption efficiency. Further, the proportion of the cellulose adsorbing segment B in the entire dispersant is preferably 5 to 95% by mass, more preferably 30 to 70% by mass.
- the cellulose adsorbing component If it is less than 5% by mass, the cellulose adsorbing component is relatively small, and the cellulose adsorbing performance cannot be sufficiently exhibited. On the other hand, when it is larger than 95% by mass, the resin affinity component is relatively small and the resin affinity performance cannot be sufficiently exhibited.
- the polymer dispersant used in the present invention preferably has a polystyrene-equivalent number average molecular weight of 1,000 to 40,000 in gel permeation chromatography. Further, it is more preferably about 2000 to 16000. When the molecular weight is increased, an aqueous dispersion of the polymer dispersant may not be stably formed, and the polymer dispersant may not be effectively processed into cellulose, which is not preferable.
- the molecular weight distribution index (weight average molecular weight / number average molecular weight) is preferably 1.0 to 1.6. More preferably, it is 1.0 to 1.5.
- the molecular weight distribution index of the polymer dispersant represents the degree of molecular weight distribution, and a small value means that the molecular weight distribution of the polymer dispersant is narrow, that is, the molecular weight is highly uniform.
- the narrow molecular weight distribution means that there are few high molecular weights or small molecular weights, the properties of the polymer dispersant are uniform, and the effect of providing a highly finely dispersed state brought about by the polymer dispersant is further improved. It means that you can make it.
- the resin affinity segment A constituting the preferred polymer dispersant used in the present invention hydrophobizes the surface of cellulose through the cellulose adsorptive segment B.
- the basis of the resin affinity is preferably similar to the structure of the target resin or has a hydrophobic property close to the target resin, and the conditions for the polymer dispersant suitably used in the present invention described above. From (1), a methacrylate monomer is preferred.
- Examples of the monomer component of the resin affinity segment A used in the present invention include methyl methacrylate, ethyl methacrylate, n-propyl methacrylate, isopropyl methacrylate, t-butyl methacrylate, hexyl methacrylate, 2-ethylhexyl methacrylate, lauryl methacrylate, and tetradecyl methacrylate.
- Alkyl such as octadecyl methacrylate, cyclohexyl methacrylate, bornyl methacrylate, isobornyl methacrylate, dicyclopentanyl methacrylate, dicyclopentenyloxyethyl methacrylate, benzyl methacrylate, tetrahydrofurfuryl methacrylate, octafluorooctyl methacrylate, tetrafluoroethyl methacrylate, etc. , Alkenyl, cycloal Le, aromatic ring, and methacrylate containing a halogen element.
- the cellulose adsorptive segment B constituting the polymer dispersant used in the present invention interacts with a hydroxyl group present on the surface of cellulose by hydrogen bonding.
- the cellulose adsorbent segment B suitable for the present invention is a methacrylate monomer and / or urea group in which 50% by mass or more of the constituent components of the cellulose adsorbent segment B have one or more hydroxyl groups. It is comprised with the methacrylate-type monomer which has.
- the hydroxyl group which exists in the surface of a cellulose forms a hydrogen bond
- the cellulose adsorptive segment B is effectively adsorbed with cellulose, and as a result, the cellulose is hydrophobized by the effect of the resin affinity component A in the structure.
- having a hydroxyl group or the like makes it easy to dissolve in a hydrophilic organic solvent, and is advantageous in the production of an aqueous dispersion treatment agent for cellulose containing a polymer dispersant used in the present invention.
- this segment B must not have compatibility (affinity) with resin which is a dispersion medium.
- affinity means mixing with each other, and indicates compatibility. If the resin and the segment B have an affinity, the segment B adsorbed to the cellulose is detached from the cellulose due to the affinity for the resin and may not show a good dispersion state, which is not preferable.
- the component of the cellulose adsorptive segment B constituting the polymer dispersant used in the present invention is preferably a methacrylate monomer from the above condition (1). Therefore, examples of the methacrylate monomer having one or more hydroxyl groups used in the present invention include 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, 3-hydroxypropyl methacrylate, 2-hydroxybutyl methacrylate, and 3-hydroxybutyl methacrylate. , Hydroxyl group-containing methacrylates such as 4-hydroxybutyl methacrylate, polyethylene glycol monomethacrylate, polypropylene glycol monomethacrylate, glyceryl monomethacrylate and the like.
- Examples of the methacrylate monomer having a urea group that forms the cellulose adsorptive segment B include methacryloyloxyethyl urea, methacryloyloxyethyl ethylene urea, and the like.
- the other monomer constituting the cellulose adsorbing segment B the above-mentioned alkyl, alkenyl, cycloalkyl, aromatic ring, halogen-containing methacrylate can be used, and further, a methacrylate monomer having an alkoxy group and a glycidyl group. Can also be used within the above-mentioned range of use.
- n 2 or more.
- 2-hydroxyethyl methacrylate (HEMA), glyceryl monomethacrylate, and methacryloyloxyethyl ethylene urea are general-purpose monomers and have a small molecular weight relative to one hydroxyl group or urea group. Since the number of functional groups can be increased, the effect is enhanced, and the compatibility with the resin is poor, which is preferable.
- the cellulose adsorptive segment B constituting the preferred polymer dispersant used in the present invention will be described. More preferably, it may be preferable to emulsify with as little surfactant as possible, in which case 3 to 15% by mass of the monomer component to be formed is formed in forming the cellulose adsorbing segment B.
- a methacrylate monomer having methacrylic acid and / or a carboxy group neutralized with an alkali or a methacrylate monomer having a quaternary ammonium base is used. These monomers are monomers that have been ionized.
- the reason for this is that, as a constituent of the cellulose adsorptive segment B, if there is a water-affinity monomer such as a neutralized carboxy group or a quaternary ammonium salt, in addition to the effect of increasing the adsorptivity to cellulose, By comprising in this way, it becomes what can be used conveniently as a water-system dispersion processing agent. That is, by introducing a functional group such as a carboxy group or a quaternary ammonium salt into the structure of the cellulose adsorbing segment B, the segment B is dissolved in water.
- a functional group such as a carboxy group or a quaternary ammonium salt
- the polymer dispersant having a polymer structure can be made to be self-emulsifying, and this structure has an effect of assisting the emulsifiability of the surfactant. As a result, an aqueous dispersion treatment agent can be easily obtained.
- methacrylate monomer having a methacrylic acid and / or carboxy group or the methacrylate monomer having a quaternary ammonium base used in this case, the following may be mentioned.
- methacrylates having carboxy groups such as methacrylic acid and methacrylate monomers having a hydroxyl group such as 2-hydroxyethyl methacrylate and a polybasic acid such as phthalic acid, dimethylaminoethyl methacrylate and diethylaminoethyl
- examples thereof include quaternary ammonium salt-containing methacrylate monomers quaternized with methyl chloride such as methacrylate, benzyl chloride, dimethyl sulfate and the like.
- quaternary ammonium salts of methacrylic acid, dimethylaminoethyl methacrylate, methyl chloride or benzyl chloride which have no possibility of being hydrolyzed, are preferable because they are highly versatile materials.
- this alkali is not specifically limited. Examples thereof include organic amines such as ammonia and dimethylaminoethanol, and hydroxides such as sodium hydroxide and potassium hydroxide.
- the amount of ionized methacrylate introduced into the cellulose adsorbing segment B is 3 to 15% by mass in the segment B.
- the amount is less than 3% by mass, the self-emulsifying property is insufficient, and a surfactant described later is required.
- the amount is more than 15% by mass, the water resistance may be deteriorated. More preferably, it is 5 to 13% by mass.
- this ionized methacrylate should not be introduced into the resin affinity segment. Since it is ionized, the compatibility is poor and the dispersibility of cellulose may be hindered.
- the polymer dispersant used in the present invention is preferably the AB type block copolymer having a block copolymer structure having a resin affinity segment A and a cellulose adsorptive segment B.
- the polymerization can proceed again by adding another monomer after polymerizing one monomer, and polymer segments having different configurations can be obtained. It is possible to synthesize a block copolymer having the same.
- Nitroxide polymerized polymerization using nitroxide hereinafter abbreviated as NMP method
- atom transfer radical polymerization method which is a method of extracting a protective group which is a halogen element with a metal complex.
- ATRP method reversible Addition Fragmentation
- organic compound such as RAFT method
- Method of using, method of using cobalt complex, iodine transfer polymerization, iodine Was a protecting group, phosphorus as a catalyst, nitrogen, oxygen, reversible chain transfer catalyzed polymerization process using a compound of the hydrocarbon Reversible Transfer Catalized Polymerization, hereinafter referred to as RTCP method
- RTCP method Reversible Transfer Catalized Polymerization
- the NMP method can be used for polymerization of acrylic monomers, styrene monomers, etc., but methacrylic monomers are not preferable because sufficient structural control cannot be performed. This is because a side reaction of the tertiary radical is caused by dissociation of the terminal. In general, a high temperature is required, and the nitroxide used is a special compound, which is expensive and does not guarantee safety in terms of the environment.
- the ATRP method uses an amine-based complex, the carboxyl group-containing monomer cannot be used as it is.
- the RAFT method is difficult to obtain a low molecular weight distribution when various monomers are used, and has problems such as sulfur odor and coloring.
- the method using organic tellurium is not preferable in this respect because the organic metal is expensive and the environmental safety is unknown, so that the safety test is costly.
- the living radical polymerization method used in the synthesis of the polymer dispersant suitable for the present invention is a polymerization method that can be easily performed only by using a polymerization initiating compound and a catalyst in combination with conventional radical polymerization.
- the RTCP method is suitable.
- the polymerization method proceeds by the reaction mechanism represented by the following general reaction formula 1, that is, the polymerization proceeds by a reversible active reaction of the dormant species Polymer-X (PX) to the growth radical. .
- the polymerization reaction mechanism may vary depending on the type of catalyst, but is considered to proceed as follows.
- P ⁇ generated from the radical initiator reacts with XA to produce catalyst A ⁇ in situ.
- A. acts as an activator of PX, and this catalysis activates PX at a high frequency.
- an organic iodine compound when used as a polymerization initiating compound, an organic radical generated by heat or light reacts with a monomer to generate a polymer terminal radical.
- an organic radical generated by heat or light reacts with a monomer to generate a polymer terminal radical.
- the iodine radicals generated sequentially are combined with the polymer terminal radicals and stabilized, it is possible to prevent the termination reaction from occurring. Since living radical polymerization proceeds by this repetition, the molecular weight and structure of the obtained AB block copolymer can be easily controlled to those desired.
- the organic iodine compound is not particularly limited as long as it can generate iodine radicals by the action of heat or light.
- Specific examples of the organic iodine compound include, for example, alkyl iodides such as 2-iodo-1-phenylethane and 1-iodo-1-phenylethane; 2-cyano-2-iodopropane, 2-cyano-2-iodobutane Cyano group-containing iodides such as 1-cyano-1-iodocyclohexane and 2-cyano-2-iodovaleronitrile.
- organic iodine compound may be used as it is, or an organic iodine compound synthesized by a conventionally known method may be used.
- the organic iodine compound can be obtained, for example, by reacting an azo compound such as azobisisobutyronitrile with iodine.
- organic halides having halogen atoms other than iodine such as bromine and chlorine, and iodide salts such as quaternary ammonium iodide and sodium iodide, a halogen exchange reaction is caused in the reaction system.
- Organic iodine compounds may be generated.
- a catalyst capable of generating iodine radicals by extracting iodine atoms from iodine compounds examples include phosphorus compounds such as phosphorus halides, phosphite compounds and phosphinate compounds; nitrogen compounds such as imide compounds; oxygen compounds such as phenol compounds; diphenylmethane compounds and cyclopentadiene. And hydrocarbon compounds containing an active carbon atom such as a series compound. In addition, you may use these catalysts individually by 1 type or in combination of 2 or more types.
- the hydrophilic organic solvent used in the present invention is not particularly limited as long as it is compatible with water and dissolves the polymer dispersant, but is preferably an alcohol solvent or a glycol solvent.
- the alcohol solvent for example, methanol, ethanol, propanol, butanol, pentanol, hexanol and the like can be used.
- glycol solvent for example, ethylene glycol, propylene glycol, diethylene glycol, ethylene glycol methyl ether, propylene glycol Methyl ether, propylene glycol propyl ether, diethylene glycol methyl ether, diethylene glycol butyl ether, diethylene glycol dimethyl ether and the like can be used.
- hydrophilic organic solvents such as methylamine, ethylamine, propylamine, butylamine, dimethylamine, diethylamine, trimethylamine, triethylamine, ethylenediamine, diethylenetriamine, dimethylformamide, dimethylacetamide, pyrrolidone, methylpyrrolidone, ethylpyrrolidone, etc.
- the amides can also be used. According to the study by the present inventors, it is more preferable to use a hydrophilic organic solvent having a boiling point of 170 ° C. or less in consideration of removal of the hydrophilic organic solvent by drying.
- surfactant used in the present invention.
- general surfactants such as a cationic surfactant, an anionic surfactant, and a nonionic surfactant can be used as the surfactant.
- an aqueous dispersion treatment agent for cellulose containing the above-described polymer dispersant can be more stably formed, and the dispersion By using a treating agent, it becomes possible to perform an effective treatment with cellulose.
- Examples of the cationic surfactant used in the present invention include carboxylates, inorganic acid salts and quaternary ammonium salts of aliphatic amines.
- Specific examples of the carboxylate or inorganic acid salt of aliphatic amines include laurylamine, methyllaurylamine, dimethyllaurylamine, myristylamine, palmitylamine, stearylamine, methylstearylamine, dimethylstearylamine, methyl Use acetate, propionate, lactate, citric acid, hydrochloride, sulfate, nitrate, phosphate, etc.
- the quaternary ammonium salts of aliphatic amines include trimethyl lauryl ammonium chloride, dimethyl dilauryl ammonium chloride, methyl tristearyl ammonium chloride, trimethyl stearyl ammonium chloride, dimethyl distearyl ammonium chloride, methyl tristearyl ammonium chloride, trimethyl stearyl.
- Ammonium bromide, trimethyloleyl ammonium chloride, dimethyl distearyl ammonium chloride, methyl trioleyl ammonium chloride and the like can be used.
- the anionic surfactant is not particularly limited, and a general surfactant can be used.
- alkylbenzene sulfonates such as sodium dodecylbenzene sulfonate
- alkyl naphthalene sulfonates such as sodium dodecyl naphthalene sulfonate
- polystyrene sulfonates such as sodium polystyrene sulfonate
- alkyl sulfates such as sodium dodecyl sulfonate.
- a salt or the like can be used.
- the nonionic surfactant is not particularly limited, and a general surfactant can be used.
- polyoxyethylene alkyl ethers such as polyoxyethylene lauryl ether, polyethylene glycol fatty acid esters such as polyethylene glycol monolaurate, glycerin fatty acid esters such as glycerol monostearate, and the like can be used.
- the cellulose used in the present invention may be at least one selected from the group consisting of cellulose nanofiber (hereinafter referred to as CNF), cellulose nanocrystal (hereinafter referred to as CNC), pulp, lignocellulose, and wood flour. preferable. In particular, it is more preferable to use CNF or CNC. In the present invention, CNF and CNC are referred to as “nanocellulose”. Hereinafter, each cellulose will be described in detail.
- Plant fibers used as raw materials for cellulose are natural celluloses obtained from natural plant materials such as wood, bamboo, hemp, jute, kenaf, cotton, beet, agricultural waste and cloth, pulp (paper) and Examples include regenerated cellulose fibers such as rayon and cellophane.
- wood include Sitka spruce, cedar, cypress, eucalyptus, and acacia.
- the paper include, but are not limited to, deinked waste paper, corrugated waste paper, magazines, and copy paper. .
- One kind of plant fiber may be used alone, or two or more kinds selected from these may be used.
- Lignocellulose is a main component of plant fibers, and is mainly composed of cellulose, hemicellulose, and lignin, and has a structure in which each is combined to form plant fibers. Pulp is obtained by removing hemicellulose and lignin from the plant fiber containing lignocellulose by mechanical treatment and / or chemical treatment to increase the pure content of cellulose. Bleaching is also performed as necessary, and the amount of delignification can be adjusted to adjust the amount of lignin in the pulp.
- pulp chemical pulp obtained by pulping plant fibers by mechanical treatment and / or chemical treatment [kraft pulp (KP), sulfite pulp (SP)], semi-chemical pulp (SCP), chemi-ground pulp (CGP) ), Chemimechanical pulp (CMP), groundwood pulp (GP), refiner mechanical pulp (RMP), thermomechanical pulp (TWP), chemithermomechanical pulp (CTMP), and deinked waste paper pulp based on these pulps Corrugated waste paper pulp and magazine waste paper pulp are preferable.
- KP kraft pulp
- SP sulfite pulp
- SCP semi-chemical pulp
- CGP Chemimechanical pulp
- CMP Chemimechanical pulp
- GP groundwood pulp
- RMP refiner mechanical pulp
- TWP thermomechanical pulp
- CTMP chemithermomechanical pulp
- deinked waste paper pulp based on these pulps Corrugated waste paper pulp and magazine waste paper pulp are preferable.
- various kraft pulps derived from conifers having strong fiber strength [unleaved kraft pulps of conifers (NUKP), unbleached kraft pulps exposed to oxygen of conifers (NOKP), and bleached kraft pulps of conifers (NBKP)] are particularly preferable.
- the lignin content in the pulp is not particularly limited, but is usually about 0 to 40% by mass, preferably about 0 to 10% by mass. The lignin content can be measured by the Klason method.
- Nanocellulose that can be suitably used in the present invention is cellulose obtained by unraveling (defibrating) a material containing cellulose fibers (such as wood pulp) to a nanosize level, and includes CNF and CNC.
- cellulose microfibrils single cellulose nanofibers
- Single cellulose nanofibers with a width of about 4 nm exist as a minimum unit and are the basic skeletal material of plants.
- Nanocellulose is composed of multiple cellulose microfibrils or cellulose microfibrils. It is a nano-sized cellulose formed as described above.
- CNF is a fiber obtained by subjecting cellulose fibers to a treatment such as mechanical defibration, and is a fiber having a fiber width of about 4 to 200 nm and a fiber length of about 5 ⁇ m or more.
- the specific surface area of the CNF preferably about 70 ⁇ 300m 2 / g, more preferably about 70 ⁇ 250m 2 / g, more preferably about 100 ⁇ 200m 2 / g.
- the average fiber diameter of CNF is usually about 4 to 200 nm, preferably about 4 to 150 nm, and particularly preferably about 4 to 100 nm.
- Examples of a method for defibrating plant fibers and preparing CNF include a method for defibrating cellulose fiber-containing materials such as pulp.
- a method for defibrating cellulose fiber-containing materials such as pulp.
- the defibrating method for example, an aqueous suspension or slurry of a cellulose fiber-containing material is mechanically ground by a refiner, a high-pressure homogenizer, a grinder, a uniaxial or multiaxial kneader (preferably a biaxial kneader), a bead mill or the like.
- a method of defibration by crushing or beating can be used. You may process combining the said defibrating method as needed.
- the defibrating methods described in JP2011-213754A and JP2011-195738A can be used.
- CNC is a crystal obtained by subjecting cellulose fibers to chemical treatment such as acid hydrolysis, and is a crystal having a crystal width of about 4 to 70 nm and a crystal length of about 25 to 3000 nm.
- the specific surface area of the CNC is preferably about 90 to 900 m 2 / g, more preferably about 100 to 500 m 2 / g, and still more preferably about 100 to 300 m 2 / g.
- the average crystal width of the CNC is usually about 10 to 50 nm, preferably about 10 to 30 nm, and particularly preferably about 10 to 20 nm.
- the average crystal length of the CNC is usually about 500 nm, preferably about 100 to 500 nm, and particularly preferably about 100 to 200 nm.
- a known method can be adopted as a method of preparing a CNC by defibrating plant fibers.
- a chemical method such as acid hydrolysis with sulfuric acid, hydrochloric acid, hydrobromic acid or the like can be used for the aqueous suspension or slurry of the cellulose fiber-containing material. You may process combining the said defibrating method as needed.
- the average value of the fiber diameter of nanocellulose in the present invention is an average value when measuring at least 50 nanocelluloses in the field of view of an electron microscope. It is.
- Nanocellulose has a high specific surface area (preferably about 200 to 300 m 2 / g), is lighter and has higher strength than steel. Nanocellulose also has low thermal deformation (low thermal expansion) compared to glass.
- Nanocellulose preferably has cellulose I-type crystals and a crystallinity as high as 50% or more.
- the cellulose I type crystallinity of the nanocellulose is more preferably 55% or more, and still more preferably 60% or more.
- the upper limit of the crystallinity of cellulose I type of nanocellulose is generally about 95% or about 90%.
- the cellulose type I crystal structure is, for example, as described in “The Dictionary of Cellulose”, the first edition of the first edition, pages 81 to 86, or pages 93 to 99, published by Asakura Shoten. Most natural celluloses are cellulose I Type crystal structure. In contrast, for example, cellulose fibers having a cellulose II, III, and IV structure, not a cellulose I crystal structure, are derived from cellulose having a cellulose I crystal structure. Above all, the I-type crystal structure has a higher crystal elastic modulus than other structures.
- nanocellulose having a cellulose I-type crystal structure is preferable.
- a composite material having a low linear expansion coefficient and a high elastic modulus can be obtained when a composite material of nanocellulose and a matrix resin is used.
- ethanol is added to a nanocellulose slurry to prepare a nanocellulose concentration of 0.5% by mass.
- vacuum filtration (5C filter paper manufactured by Advantech Toyo Co., Ltd.) is quickly started.
- the obtained wet web is heated and compressed at 110 ° C. and a pressure of 0.1 t for 10 minutes to obtain a CNF sheet of 50 g / m 2 .
- the polymerization degree of cellulose is about 500 to 10,000 for natural cellulose and about 200 to 800 for regenerated cellulose.
- Cellulose is a bundle of several celluloses that are linearly stretched by ⁇ -1,4 bonds, which are fixed by intramolecular or intermolecular hydrogen bonds to form crystals that are elongated chains. .
- the fact that many crystal forms exist in the crystal of cellulose has been clarified by analysis by X-ray diffraction or solid state NMR, but the crystal form of natural cellulose is only type I. From the X-ray diffraction and the like, it is estimated that the ratio of crystal regions in cellulose is about 50 to 60% for wood pulp and about 70% for bacterial cellulose.
- cellulose Due to the fact that cellulose is an extended chain crystal, cellulose not only has a high elastic modulus, but also exhibits a strength five times that of steel and a linear thermal expansion coefficient of 1/50 or less that of glass. Conversely, breaking the crystal structure of cellulose leads to the loss of excellent characteristics such as high elastic modulus and high strength of cellulose.
- an aqueous dispersion treatment agent for cellulose comprising the above-described polymer dispersant that characterizes the present invention, and processing this into cellulose, the polymer dispersant is obtained without breaking the cellulose crystals described above.
- an easily dispersible cellulose composition that is well dispersed can be obtained.
- the easily dispersible cellulose composition and combining with the resin it is possible to obtain a cellulose-dispersed resin composition in which the cellulose is well dispersed in the resin without breaking the cellulose crystal. .
- the cellulose crystals are present in an unbroken state, and because the cellulose has excellent dispersibility, it exhibits excellent mechanical properties such as high elastic modulus and high strength of cellulose in the resin.
- a high-strength resin composition can be obtained.
- an aqueous dispersion treatment agent for cellulose containing the above-described polymer dispersant is added to cellulose to produce an easily dispersible cellulose composition, This is combined with resin.
- the cellulose used in this case is a water-containing state. That is, by using water-containing cellulose, it is economical because water-based treatment can be carried out in cellulose treatment using an aqueous dispersion treatment agent for cellulose containing a polymer dispersant.
- the water-containing cellulose may contain a hydrophilic solvent such as alcohols, glycols, amines and amides in addition to water.
- Alcohols include methanol, ethanol, propanol, butanol, etc.
- glycols include ethylene glycol, propylene glycol, diethylene glycol, ethylene glycol methyl ether, propylene glycol methyl ether, propylene glycol propyl ether, diethylene glycol methyl ether, diethylene glycol butyl ether, diethylene glycol
- amines such as dimethyl ether include methylamine, ethylamine, propylamine, butylamine, dimethylamine, diethylamine, trimethylamine, triethylamine, ethylenediamine, and diethylenetriamine
- amides include dimethylformamide, dimethylacetamide, pyrrolidone, methylpyrrolidone, It may also contain such pyrrolidone.
- At least one water-containing or dry cellulose fiber selected from the group consisting of CNF, CNC, pulp, lignocellulose, and wood flour described above can be used.
- nanocellulose the cellulose-dispersed resin composition obtained by compounding with the resin is excellent in mechanical strength.
- the aqueous dispersion treatment agent for cellulose containing a polymer dispersant used in the present invention is prepared by adding water to a hydrophilic organic solvent solution containing a polymer dispersant and a cationic surfactant.
- a hydrophilic organic solvent solution containing a polymer dispersant and a cationic surfactant are prepared by adding water to a hydrophilic organic solvent solution containing a polymer dispersant and a cationic surfactant.
- an aqueous dispersion containing a polymer dispersant is first prepared by dissolving a polymer dispersant in a hydrophilic organic solvent solution, adding a surfactant to the solution, and then adding water. It is to prepare a treatment agent. According to the study of the present inventors, it is necessary to satisfy the order in order to allow the polymer dispersant to be effectively processed into cellulose.
- the hydrophilic organic solvent solution of the polymer dispersant used in the above may be the polymer solution of the polymer dispersant as it is, or the polymer solution diluted with a hydrophilic organic solvent. Furthermore, the polymerization solvent may be removed from the polymerization solution by precipitation or drying, and the polymer dispersant alone may be re-diluted with a hydrophilic organic solvent.
- the method of adding water to the hydrophilic organic solvent solution containing the polymer dispersant and the surfactant is to drop water into the solution while stirring the hydrophilic organic solvent solution containing the polymer dispersant and the surfactant. However, the method is not particularly limited.
- the polymer dispersant one having a block copolymer structure having the resin affinity segment A and the cellulose adsorbing segment B described above is used.
- Introducing methacrylate into the cellulose adsorbing segment to make it self-emulsifying and using an aqueous dispersion treatment agent is also an effective means for reducing the amount of surfactant and making an aqueous dispersion treatment agent more easily.
- an aqueous dispersion treatment agent containing a polymer dispersant is used as a method for treating cellulose with an aqueous dispersion treatment agent for cellulose containing the polymer dispersant prepared as described above used in the present invention. Is preferably added to cellulose having a water content of about 25% solids. In order to treat uniformly, it is preferable to add an aqueous dispersion treatment agent containing a polymer dispersant while stirring the cellulose and mix the cellulose and the polymer dispersant. In addition, conditions such as temperature and pressure at the time of addition and mixing in this case are not particularly limited. Also good.
- the stirring speed is not particularly limited.
- Cellulose can be used in a water-containing state or in a dry state, but it is preferable to use a water-containing state as described above from the viewpoint of ease of treatment during addition.
- the water-containing cellulose may contain an organic solvent other than water as long as the cellulose is not in an aggregated state.
- thermoplastic resin examples include olefin resins, nylon resins, polyamide resins, polycarbonate resins, polysulfone resins, polyester resins, cellulose resins such as triacetylated cellulose, and diacetylated cellulose.
- Polyamide resins include polyamide 6 (PA6, ring-opening polymer of ⁇ -caprolactam), polyamide 66 (PA66, polyhexamethylene adipamide), polyamide 11 (PA11, polyamide obtained by ring-opening polycondensation of undecane lactam), polyamide 12 (PA12, polyamide obtained by ring-opening polycondensation of lauryl lactam) and the like.
- PA6 ring-opening polymer of ⁇ -caprolactam
- PA66 polyhexamethylene adipamide
- PA11 polyamide obtained by ring-opening polycondensation of undecane lactam
- polyamide 12 PA12, polyamide obtained by ring-opening polycondensation of lauryl lactam
- olefin-based resins are preferred because of the advantage that a sufficient reinforcing effect can be obtained when a resin composition is used, and the advantage that they are inexpensive.
- the olefin resin examples include general-purpose resins such as polyethylene resin, polypropylene resin, vinyl chloride resin, styrene resin, (meth) acrylic resin, and vinyl ether resin. These thermoplastic resins may be used alone or as a mixed resin of two or more.
- thermoplastic resins may be used alone or as a mixed resin of two or more.
- olefin-based resins from the advantage that a sufficient reinforcing effect can be obtained when a resin composition is used and the advantage of being inexpensive, high density polyethylene (HDPE), low density polyethylene (LDPE), biopolyethylene, etc.
- Polyethylene resin (PE), polypropylene resin (PP), vinyl chloride resin, styrene resin, (meth) acrylic resin, vinyl ether resin and the like are preferable.
- the reaction system was kept at 40 ° C. and polymerized for 7 hours to obtain a polymer block A.
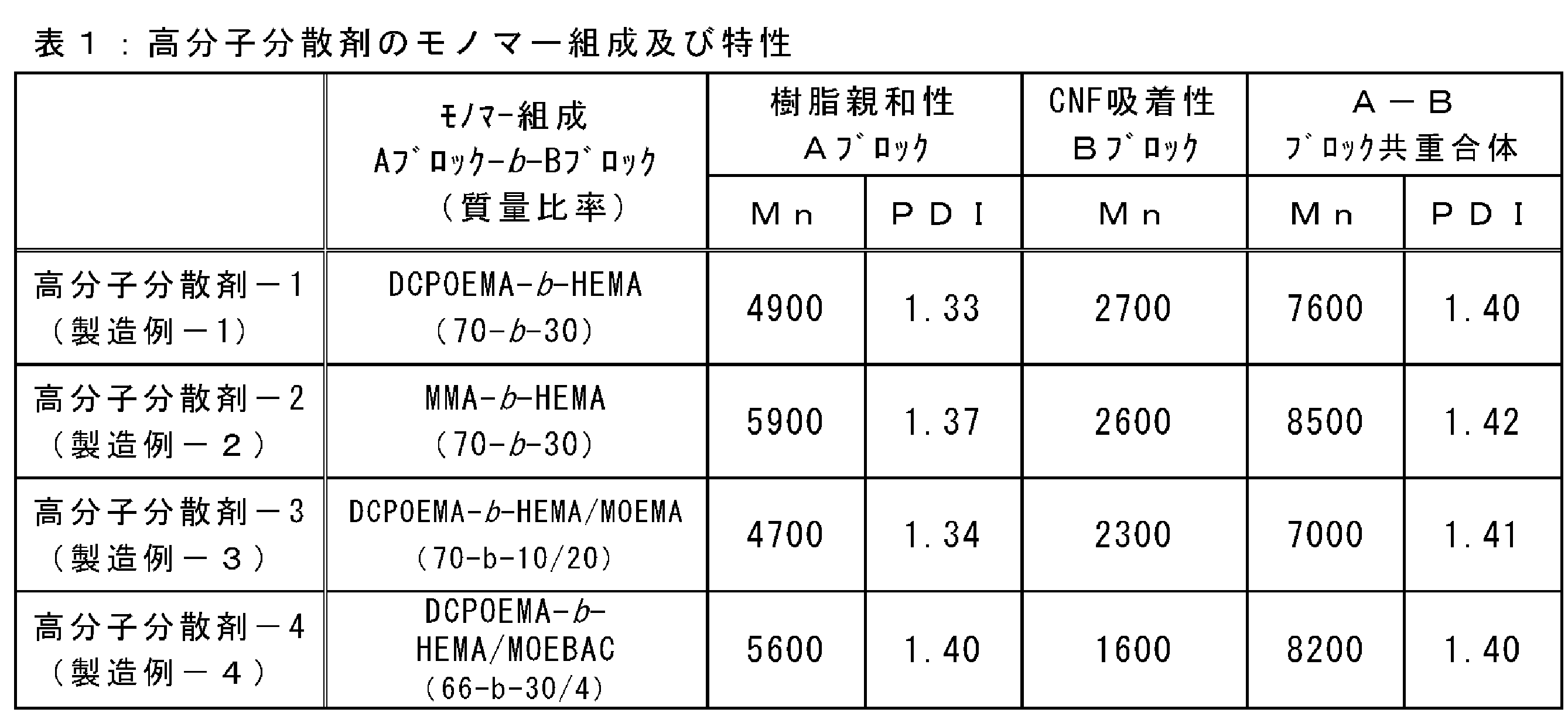
- the molecular weight was calculated by gel permeation chromatography (GPC) measurement using a THF solvent, and the number average molecular weight (hereinafter abbreviated as Mn) was 4900, and the weight average molecular weight (hereinafter abbreviated as Mw) was 6500. Its molecular weight distribution (hereinafter abbreviated as PDI value) was 1.33.
- Mn number average molecular weight
- Mw weight average molecular weight
- PDI value Its molecular weight distribution
- HEMA 2-hydroxyethyl methacrylate
- the polymerization rate is 97.0%
- Mn of the entire block copolymer is 7000
- Mw is 9900
- PDI value is 1.41. there were.
- 93 parts of Polymer Dispersant-3 composed of AB block copolymer was obtained by precipitation with water / methanol mixture, washing and drying.
- the polymerization rate was almost 100%, and the number average molecular weight was 8200 and PDI was 1.40.
- the calculated number average molecular weight of the polymer block B is 1600, the content of the methacrylate monomer having a hydroxyl group is 88.2% by mass, and the content of the monomer having an ionic group is 11.8%.
- Table 1 summarizes the monomer compositions and characteristics of the polymer dispersants 1 to 4 obtained as described above.
- the Mn of the B block which is a CNF affinity segment was calculated as a value obtained by subtracting the Mn of the A block from the Mn value of the AB block. The results are shown in Table 1.
- Example 1 (Preparation of a solution a of 10% by mass of a polymeric dispersant-1 aqueous dispersion treatment agent) 10 parts of the polymer dispersant-1 prepared above was dissolved in 25 parts of DMDG, 0.2 parts of oleylamine acetate as a surfactant was added thereto, and while stirring uniformly, water 64. 8 parts were added dropwise to obtain 100 parts of a 10% by weight polymer dispersion-1 aqueous dispersion treatment solution a. In the obtained solution a, Polymer Dispersant-1 was dispersed in a pale yellow turbid state, and no sedimentation was confirmed even after standing for 24 hours.
- Example 2 (Preparation of solution b of 10% by mass of polymer dispersant-2 aqueous dispersion treatment agent) A 10% by mass polymer dispersant was obtained in the same manner as in Example 1 except that the previously prepared polymer dispersant-2 was used instead of the polymer dispersant-1 used in Example 1. 100 parts of an aqueous dispersion treating agent solution b of -2. In the obtained solution b, the polymer dispersant-2 was dispersed in a white turbid state, and no settling was confirmed even when this was allowed to stand for 24 hours.
- Example 3 (Preparation of aqueous dispersion of polymer dispersant-4)
- the polymer dispersant-4 prepared earlier was used, and 5.2 parts of 10% aqueous solution of oleylamine acetate and 800 parts of water were dispersed. While stirring at high speed, 200 parts of the resin solution was gradually added. A bluish white aqueous dispersion was obtained. Even if it was stored for a long period of time, no sedimentation was observed and it was stable.
- Example 4 (Preparation of a solution d of 10% by weight of a polymeric dispersant-1 in an aqueous dispersant / use of an anionic surfactant) 10% by mass of polymer dispersant-1 as in Example 1 except that 10 parts of the previously prepared polymer dispersant-1 was used and 0.2 part of dodecylbenzenesulfonic acid was used as the surfactant. 100 parts of solution d of 1 aqueous dispersant was obtained. In the obtained solution d, Polymer Dispersant-1 was dispersed in a pale yellow turbid state, and even when this was allowed to stand for several days, sedimentation was hardly confirmed.
- Example 5 (Preparation of a solution e of a 10% by mass polymer dispersant-3 aqueous dispersant) The same procedure as in Example 1 was carried out except that the previously prepared polymer dispersant-3 was used instead of the polymer dispersant-1, and 10% by mass of the polymer dispersant-3 was obtained. 100 parts of an aqueous dispersant solution e was obtained. In the obtained solution e, the polymer dispersant-3 was dispersed in a white turbid state, but when left for several days, it slightly settled.
- Example 6 Cellulose treatment with aqueous dispersion treatment solution a and preparation of resin composition a
- 80 parts of fine particle polyethylene manufactured by Sumitomo Seika, flow beads HE3040 (trade name), hereinafter abbreviated as “fine particle PE”
- fine particle PE fine particle polyethylene
- Example 7 Cellulose treatment with aqueous dispersion treatment solution b and preparation of resin composition b
- the aqueous dispersion treating agent solution b of 10% by weight of the polymeric dispersing agent-2 obtained in Example 2 was used. Except for the above, the same procedure as in Example 6 was carried out to obtain 98 parts of a cellulose-dispersed resin composition b, which is a mixed composition of CNF-1 treated with the polymer dispersant-2 and particulate PE. It was.
- a resin mixture composition of a comparative example which is a mixed composition of CNF-1 treated with a DMDG solution of polymer dispersant-1 and fine-particle PE, was prepared. I got a part.
- Example 8 Cellulose treatment with polymer dispersant-1 aqueous dispersant solution d and preparation of resin composition d
- the 10% by weight of the polymer dispersion-1 aqueous dispersion treatment solution d prepared in Example 3 was used. Except for the above, the same procedure as in Example 6 was carried out to obtain 98 parts of a cellulose-dispersed resin composition d, which is a mixed composition of CNF-1 treated with the polymer dispersant-1 and particulate PE. It was.
- Example 9 Cellulose treatment with aqueous dispersant solution e of polymer dispersant-3 and preparation of resin composition e
- the aqueous dispersion treating agent solution a of the polymeric dispersant-1 used in Example 6 the aqueous dispersion dispersing agent solution e of 10% by mass of the polymeric dispersing agent-3 prepared in Example 5 was used. Except for the above, the same procedure as in Example 6 was performed to obtain 99 parts of a cellulose-dispersed resin composition e, which is a mixed composition of CNF-1 treated with the polymer dispersant-3 and fine-particle PE. It was.
- each of the obtained PE resin pellets in which CNF-1 was dispersed was used for injection molding to produce a dumbbell piece (dumbbell thickness: 2 mm), which was used as an evaluation sample.
- dumbbell thickness 2 mm
- Each of the dumbbell pieces of the obtained sample for evaluation was subjected to a tensile test at a tensile speed of 10 mm / min using a tensile tester (manufactured by Instron Co., Ltd .: universal testing machine 5900 series), and the tensile modulus and tensile strength. Were measured and evaluated. The results are summarized in Table 2.
- Example 10 Cellulose treatment with aqueous dispersion treatment solution a and preparation / non-drying of resin composition a
- 80 parts of fine particle PE and 50 parts of water were added to the obtained easily dispersible cellulose composition and mixed.
- a cellulose-dispersed resin which is a mixed composition of CNF-1 treated with the polymer dispersant-1 and finely divided PE, containing a liquid component without lightly filtering and drying the obtained mixture 150 parts of composition a ′ were obtained.
- the surfactants of Examples 6 to 10 of the present invention were used, and the aqueous dispersion treatment agents of the examples in which the order of addition was defined in the present invention were used.
- the sample for evaluation was produced and confirmed about the cellulose dispersion PE resin mixed composition, the tensile elasticity modulus and the tensile strength showed the high value.
- a cationic surfactant is used, a high value is exhibited.
- a water-based dispersion treatment agent of a polymer dispersant is prepared, the use of a cationic surfactant is particularly effective.
- biaxial extrusion kneading was performed while containing the liquid component, and further improvement in mechanical strength was confirmed.
- the aqueous dispersion is not used. It is considered that the polymer dispersant in the treatment agent was in a precipitated state and was not effectively adsorbed with cellulose, and the mechanical properties were low as in the above case. From the above, the method of applying the polymer dispersant as a dispersion treatment using the surfactant specified in the present invention is based on the cellulose fine powder that is hydrophilic while using water as the main medium. It was confirmed that it is effective as a treatment method that can sufficiently exhibit the effect of the polymer dispersant.
- a cellulose-dispersed PE resin is prepared by adding an aqueous dispersion treatment agent comprising a polymer dispersant using a surfactant in a specific order of addition to cellulose to obtain an easily dispersible cellulose composition.
- the cellulose-dispersed resin composition obtained by carrying out as a pretreatment step in the process has no problems caused by using a large amount of an organic solvent, and the molded product is excellent in tensile modulus and tensile strength. It will have.
- the cellulose-dispersed resin composition provided in the present invention is a housing for electrical appliances such as automobile members, televisions, telephones, watches, etc., a casing for mobile communication devices such as mobile phones, printing equipment, copying machines, sports equipment. It is useful as a structural material such as a casing.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Dispersion Chemistry (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Processes Of Treating Macromolecular Substances (AREA)
- Emulsifying, Dispersing, Foam-Producing Or Wetting Agents (AREA)
- Graft Or Block Polymers (AREA)
Abstract
Description
(1)前記A-Bブロック共重合体の構成成分の90質量%以上がメタクリレート系モノマーで構成されていること;
(2)前記セルロース吸着性セグメントBは、構成成分の50質量%以上が、水酸基を1個以上有するメタクリレート系モノマー及び/又は尿素基を有するメタクリレート系モノマーで構成されており、且つ、樹脂との相溶性がないこと;
(3)前記樹脂親和性セグメントAのゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が500~20000であり、且つ、前記A-B共重合体全体に占める該樹脂親和性セグメントAの割合が5~95質量%であること;
(4)前記セルロース吸着性セグメントBのゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が500~20000であり、且つ、前記A-B共重合体全体に占める該セルロース吸着性セグメントBの割合が5~95質量%であること;
(5)前記A-Bブロック共重合体のゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が3500~40000であり、且つ、分子量分布指数(重量平均分子量/数平均分子量)が1.0~1.6であること。
本発明の特徴は、高分子分散剤を用いたセルロース分散樹脂組成物を得るために必要となる、高分子分散剤によってセルロースの樹脂への分散性を良好なものとした易分散性セルロース組成物の提供を可能できる、簡便な前処理方法を見出した点にある。具体的には、特定のブロック共重合体構造を有する高分子分散剤及び界面活性剤を含む親水性有機溶剤溶液に水を添加し、水系の分散処理剤とする場合に、界面活性剤を用い、その添加順を特定のものとするという極めて簡便な方法で、セルロースに対して有効に機能する、高分子分散剤を含む水系の分散処理剤を得た点に大きな特徴を有する。この水系の分散処理剤をセルロースに添加混合することで、易分散性セルロース組成物が提供され、この易分散性セルロース組成物を用いて樹脂との複合化をすることで、極めて容易に、セルロース分散樹脂組成物を得ることができる。本発明者らの検討によれば、得られるセルロース分散樹脂組成物は、セルロースが樹脂中に良好に分散したものとなり、更に、射出成型した場合に、その機械的強度が向上したものとなる。このように、多量の有機溶剤を使用しない、極めて簡便な方法で、樹脂への相溶性に優れる易分散性セルロース組成物が得られるので、機能性材料として優れたセルロースの広範な利用が期待できる。
本発明のセルロース用の水系の分散処理剤は、界面活性剤を含む高分子分散剤の親水性有機溶剤溶液に水を添加することで作製することができる。特に、本発明では、まず、特定のブロック共重合体構造を有する高分子分散剤を親水性有機溶剤溶液に溶解し、これに界面活性剤を添加し、その後に水を添加することで、高分子分散剤を含有した水系の分散処理剤を作製することを必須とする。以下、使用するそれぞれの材料について説明する。
次に、本発明において好適に用いることのできる高分子分散剤について詳細に説明する。
本発明で用いる高分子分散剤は、樹脂親和性セグメントAと、セルロース吸着性セグメントBとを有するブロック共重合体構造を有するものを用いるが、更に、下記(1)~(5)の要件をすべて満たすブロック共重合体であることが好ましい。
(1)前記A-Bブロック共重合体の構成成分の90質量%以上がメタクリレート系モノマーで構成されていること;
(2)前記セルロース吸着性セグメントBは、構成成分の50質量%以上が、水酸基を1個以上有するメタクリレート系モノマー及び/又は尿素基を有するメタクリレート系モノマーで構成されており、且つ、樹脂との相溶性がないこと;
(3)前記樹脂親和性セグメントAのゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が500~20000であり、且つ、前記A-B共重合体全体に占める該樹脂親和性セグメントAの割合が5~95質量%であること;
(4)前記セルロース吸着性セグメントBのゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が500~20000であり、且つ、前記A-B共重合体全体に占める該セルロース吸着性セグメントBの割合が5~95質量%であること;
(5)前記A-Bブロック共重合体のゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が3500~40000であり、且つ、分子量分布指数(重量平均分子量/数平均分子量)が1.0~1.6であること;である。
本発明で使用される好適な高分子分散剤を構成する樹脂親和性セグメントAは、セルロース吸着性セグメントBを介して、セルロースの表面を疎水化する。ここで、樹脂親和性の基本は、対象となる樹脂の構造に類似又は対象となる樹脂に近い疎水性を有することが好ましく、また、前記した本発明に好適に用いられる高分子分散剤の条件(1)より、メタクリレート系モノマーであることが好ましい。本発明で用いる樹脂親和性セグメントAのモノマー成分としては、例えば、メチルメタクリレート、エチルメタクリレート、n-プロピルメタクリレート、イソプロピルメタクリレート、t-ブチルメタクリレート、ヘキシルメタクリレート、2-エチルヘキシルメタクリレート、ラウリルメタクリレート、テトラデシルメタクリレート、オクタデシルメタクリレート、シクロヘキシルメタクリレート、ボルニルメタクリレート、イソボロニルメタクリレート、ジシクロペンタニルメタクリレート、ジシクロペンテニルオキシエチルメタクリレート、ベンジルメタクリレート、テトラヒドロフルフリルメタクリレート、オクタフルオロオクチルメタクリレート、テトラフルオロエチルメタクリレート等の、アルキル、アルケニル、シクロアルキル、芳香環、ハロゲン元素含有のメタクリレート等が挙げられる。
本発明者らの検討によれば、本発明で使用される高分子分散剤を構成するセルロース吸着性セグメントBは、セルロースの表面に存在する水酸基に対して、水素結合により相互作用を示す。前記(2)の要件の通り、本発明に好適なセルロース吸着性セグメントBは、セルロース吸着性セグメントBの構成成分の50質量%以上が、水酸基を1個以上有するメタクリレート系モノマー及び/又は尿素基を有するメタクリレート系モノマーで構成される。このように構成することで、セルロースの表面に存在する水酸基と水素結合を形成し、及び、高分子鎖において多点相互作用を示すことで、セルロース吸着性セグメントBは、セルロースと効果的に吸着するものとなる。すなわち、このセルロース吸着性セグメントBが、セルロースと効果的に吸着し、この結果、その構造中の上記した樹脂親和成分Aの効果で、セルロースが疎水化される。また、水酸基等を有することで親水性有機溶剤に溶解しやすくなり、本発明で使用する、高分子分散剤を含有するセルロース用の水系の分散処理剤の作製において有利となり、また高分子分散剤は析出せずに安定な水系分散液を得ることができる。なお、このセグメントBは、分散媒体である樹脂に相溶性(親和性)を有してはいけない。本願において、親和性とは、お互いに混ざることを示し、相溶性を示すものである。樹脂と、セグメントBに親和性があると、セルロースと吸着したセグメントBが、樹脂にも親和することによってセルロースから脱離してしまい、良好な分散状態を示さない場合があるので好ましくない。
次に、上記に挙げた要件(1)~(5)の全てを具備する本発明に好適な高分子分散剤の合成方法について説明する。本発明で用いる高分子分散剤は、樹脂親和性セグメントAとセルロース吸着性セグメントBとを有するブロック共重合体構造である前記A-B型ブロック共重合体であることが好ましいが、その合成には、リビングラジカル重合法を用いることが好ましい。リビングラジカル重合とは、ラジカル重合において、分子構造の明確な高分子を得ることができる方法である。重合時、生長鎖末端のラジカルが安定化させるため、あるモノマーを重合した後に、続けて別のモノマーを添加することで、再び重合を進行させることができ、構成がそれぞれに異なる重合体セグメントを有するブロック共重合体を合成することができる。
(一般反応式1)
上記重合反応機構は、触媒の種類によって変わる可能性があるが、次のように進むと考えられる。上記一般反応式1では、ラジカル開始剤から発生したP・がXAと反応して、in situで触媒A・が生成する。A・は、P-Xの活性化剤として作用して、この触媒作用によってP-Xは高い頻度で活性化する。
次に、本発明で使用する、上記に挙げたような高分子分散剤を溶解する際に用いる親水性有機溶剤について説明する。本発明において使用する親水性有機溶剤としては、水と均一に相溶し、高分子分散剤を溶解させるものであれば特に限定されないが、アルコール系溶剤又はグリコール系溶剤であることが好ましい。アルコール系溶剤としては、例えば、メタノール、エタノール、プロパノール、ブタノール、ペンタノール、ヘキサノール等を用いることができ、グリコール系溶剤としては、例えば、エチレングリコール、プロピレングリコール、ジエチレングリコール、エチレングリコールメチルエーテル、プロピレングリコールメチルエーテル、プロピレングリコールプロピルエーテル、ジエチレングリコールメチルエーテル、ジエチレングリコールブチルエーテル、ジエチレングリコールジメチルエーテル等を用いることができる。また、親水性有機溶剤として、メチルアミン、エチルアミン、プロピルアミン、ブチルアミン、ジメチルアミン、ジエチルアミン、トリメチルアミン、トリエチルアミン、エチレンジアミン、ジエチレントリアミン等のアミン類や、ジメチルホルムアミド、ジメチルアセトアミド、ピロリドン、メチルピロリドン、エチルピロリドン等のアミド類等も用いることができる。本発明者らの検討によれば、親水性有機溶剤の乾燥による除去を考慮し、親水性有機溶剤としては、沸点が170℃以下のものを用いることがより好ましい。
本発明で使用する界面活性剤について説明する。本発明では、界面活性剤として、カチオン性界面活性剤、アニオン性界面活性剤、ノニオン性界面活性剤等の一般的な界面活性剤を用いることができる。本発明者らの検討によれば、中でも特にカチオン性界面活性剤を用いると、前記した高分子分散剤を含有するセルロース用の水系の分散処理剤をより安定に形成することができ、該分散処理剤を用いることで、セルロースにより効果的な処理をすることができるようになる。
本発明で使用するセルロースは、セルロースナノファイバー(以下、CNFと記載)、セルロースナノクリスタル(以下、CNCと記載)、パルプ、リグノセルロース、木粉からなる群から選ばれる少なくとも1種であることが好ましい。特に、CNF又はCNCを用いることがより好ましい。本発明では、CNF及びCNCを「ナノセルロース」と称す。以下、各セルロースについて、詳細に説明する。
本発明で使用する高分子分散剤を含有するセルロース用の水系の分散処理剤は、高分子分散剤及びカチオン性界面活性剤を含む親水性有機溶剤溶液に、水を添加することで作製する。特に重要なことは、まず、高分子分散剤を親水性有機溶剤溶液に溶解し、これに界面活性剤を添加し、その後に、水を添加することで高分子分散剤を含有した水系の分散処理剤を作製することである。本発明者らの検討によれば、高分子分散剤を、セルロースに効果的に処理することができるようにするためには、この順序を満たして作製する必要がある。すなわち、高分子分散剤と界面活性剤と親水性有機溶剤と水とを含有するものであっても、本発明で規定する順で作製したものでなければ、本発明の顕著な効果は得られない。換言すれば、高分子分散剤と界面活性剤と親水性有機溶剤と水とを含有するものを用いてセルロースの分散を実現しているとすれば、その実施系は、本発明で規定する方法を実施していることになる。
本発明で使用する、上記したようにして調製した高分子分散剤を含有するセルロース用の水系の分散処理剤を、セルロースに処理する方法としては、高分子分散剤を含有する水系の分散処理剤を、固形分25%程度の含水率を示すセルロースに添加するとよい。均一に処理するため、セルロースを撹拌しながら、高分子分散剤を含有する水系の分散処理剤を添加し、セルロース及び高分子分散剤を混合することが好ましい。また、この際における、添加、混合時における温度、圧力等の条件は特に制限するものでなく、常温常圧での条件の他、昇温、冷却、加圧、減圧等の条件で実施してもよい。また、撹拌速度も特に制限するものではない。セルロースは含水状態又は乾燥状態のものを用いることができるが、添加時の処理のし易さから、上記したような含水状態のものを用いることが好ましい。また、含水状態のセルロースには、セルロースが凝集状態とならない範囲で水以外の有機溶剤を含んでいてもよい。
本発明では、本発明により得られる高分子分散剤を含む易分散性セルロース組成物と、樹脂とを溶融混練することで、良好なセルロース分散樹脂組成物を得ることができる。この際に使用する樹脂としては、特に限定されないが、溶融混練してセルロース分散樹脂組成物を得ることが好ましいことから、熱可塑性樹脂を使用することが好ましい。熱可塑性樹脂としては、オレフィン系樹脂、ナイロン樹脂、ポリアミド系樹脂、ポリカーボネート系樹脂、ポリスルホン系樹脂、ポリエステル系樹脂、トリアセチル化セルロース、ジアセチル化セルロース等のセルロース系樹脂等がある。ポリアミド系樹脂としてはポリアミド6(PA6、ε-カプロラクタムの開環重合体)、ポリアミド66(PA66、ポリヘキサメチレンアジポアミド)、ポリアミド11(PA11、ウンデカンラクタムを開環重縮合したポリアミド)、ポリアミド12(PA12、ラウリルラクタムを開環重縮合したポリアミド)等が例示される。この中で、樹脂組成物とした場合の補強効果を十分に得ることができるという利点、安価であるという利点から、オレフィン系樹脂が好ましい。オレフィン系樹脂としては、ポリエチレン系樹脂、ポリプロピレン系樹脂、塩化ビニル樹脂、スチレン樹脂、(メタ)アクリル樹脂、ビニルエーテル樹脂等の汎用樹脂が挙げられる。これらの熱可塑性樹脂は、単独で使用してもよく、2種以上の混合樹脂として用いてもよい。オレフィン系樹脂の中でも、樹脂組成物とした場合の補強効果を十分に得ることができるという利点、安価であるという利点から、高密度ポリエチレン(HDPE)、低密度ポリエチレン(LDPE)、バイオポリエチレン等のポリエチレン系樹脂(PE)、ポリプロピレン系樹脂(PP)、塩化ビニル樹脂、スチレン樹脂、(メタ)アクリル樹脂、ビニルエーテル樹脂等が好ましい。
撹拌機、還流コンデンサー、温度計及び窒素導入管を取り付けた反応装置に、ジメチルジグリコール(以下、DMDGと略す)106部、ジシクロペンテニルオキシエチルメタクリレート(以下、DCPOEMAと略す)70部、ヨウ素1.0部、ジフェニルメタン(以下、DPMと略す)0.2部、重合開始剤として2,2’-アゾビス(4-メトキシ-2,4-ジメチルバレロニトリル)(商品名:V-70、和光純薬社製)5.0部を添加した。そして、窒素ガスを導入しながら撹拌し、マントルヒーターにて40℃に昇温した。反応系をそのまま40℃に保ち、7時間重合してポリマーブロックAを得た。重合の進行を、反応系中の固形物濃度より算出したところ、重合率は85%であった。また、THF溶媒によるゲルパーミエーションクロマトグラフィー(GPC)測定により分子量を算出し、数平均分子量(以下、Mnと略す)が4900、重量平均分子量(以下、Mwと略す)で6500であった。その分子量分布(以下、PDI値と略す)は1.33であった。上記で得られたポリマーブロックAは、後述するように、樹脂親和性セグメントとして機能するものとなる。
先の高分子分散剤-1の合成において、DCPOEMA70部を使用する代わりに、メチルメタクリレート(以下、MMAと略す)70部を使用し、また、DMDG106部を使用する代わりにプロピレングリコールプロピルエーテル(以下、PFGと略す)106部を使用した以外は製造例1と同様にして重合を実施した。その結果、樹脂親和性セグメントAとして機能させるためのMMAの重合で得られたセグメントは、重合率86%であり、Mnは5900、Mwは8100、PDI値は1.37であった。また、セルロース吸着性セグメントBとして機能させるためのHEMAの重合では、増粘したために、追加でPFG56部を添加して希釈した。その重合率は99%、ブロック共重合体全体のMnは8500、Mwは12000、PDI値は1.41であった。製造例1と同様に、水/メタノール混合液による析出、洗浄及び乾燥によりA-Bブロックコポリマーからなる高分子分散剤-2を90部得た。
先の高分子分散剤-1の合成において、HEMA30部を使用する代わりに、HEMA10部及びメトキシエチルメタクリレート(以下、MOEMAと略す)20部を使用したこと以外は製造例1と同様にして重合を実施した。その結果、樹脂親和性セグメントAとして機能させるためのDCPOEMAの重合では、重合率83%であり、Mnは4700、Mwは6300、PDI値は1.34であった。また、セルロース吸着性セグメントBとして機能させるためのHEMA/MOEMAの重合では、重合率は97.0%であり、ブロック共重合体全体のMnは7000、Mwは9900、PDI値は1.41であった。製造例1と同様に、水/メタノール混合液による析出、洗浄及び乾燥によりA-Bブロックコポリマーからなる高分子分散剤-3を93部得た。
実施例1と同様にして重合時間を9時間に替えて、ポリマーブロックAを得た。その重合率は100%で、数平均分子量が5600、PDIが1.40であった。次いで、HEMA30部、メタクリロイロキシエチルベンジルトリメチルアンモニウムクロライド(MOEBAC)の30質量%のプロピレングリコールモノプロピルエーテル13.3部を添加し、重合した。得られた重合物の分子量は、THF溶媒のGPCから、10ml/Lのリチウムブロマイドのジメチルホルムアミド溶液を展開溶媒とするGPCに換えて測定したところ、重合率はほぼ100%であり、数平均分子量が8200、PDIは1.40であった。算出されたポリマーブロックBの数平均分子量は1600であり、水酸基を有するメタクリレート系モノマーの含有量は88.2質量%であり、イオン性基を有するモノマーの含有量は11.8%である。
まず、針葉樹漂白クラフトパルプ(NBKP)(リファイナー処理済み、固形分:25%)600部に、水を19400部添加し、パルプスラリー濃度0.75質量%の水懸濁液(スラリー)を調製した。次に、得られたスラリーに対し、ビーズミルを用いて機械的解繊処理を行った。更に、解繊処理を行った後、フィルタープレスで脱水し、含水状態のCNF-1(固形分:25%)を570部得た。
先に調製した高分子分散剤-1を10部、DMDG25部に溶解させ、これに、界面活性剤としてオレイルアミン酢酸塩を0.2部添加し、均一に撹拌しながら、次に、水64.8部を滴下して、10質量%の高分子分散剤-1の水系分散処理剤の溶液aを100部得た。得られた溶液aは、高分子分散剤-1が淡い黄濁状に分散しており、これを24時間静置しても沈降は確認されなかった。
実施例1で用いた高分子分散剤-1に替えて、先に調製した高分子分散剤-2を使用したこと以外は実施例1と同様に実施して、10質量%の高分子分散剤-2の水系分散処理剤の溶液bを100部得た。得られた溶液bは、高分子分散剤-2が白濁状に分散しており、これを24時間静置しても沈降は確認されなかった。
実施例1で用いた高分子分散剤-1に替えて、先に調製した高分子分散剤-4を使用して、オレイルアミンの酢酸塩10%水溶液を5.2部、水800部をディスパーで高速撹拌しながら、樹脂溶液200部を徐々に添加した。青味がかった白色の水分散体が得られた。これを長期に保管しても全く沈降は見られず安定であった。
DMDGの89.8部に、先に調製した高分子分散剤-1を10部と、界面活性剤としてオレイルアミン酢酸塩の0.2部とを一緒に入れて溶解して、10質量%の高分子分散剤-1のDMDG溶液を100部得た。得られたDMDG溶液は、淡い黄色の透明液体であった。
オレイルアミン酢酸塩の0.2部を添加せず、界面活性剤を使用しなかったこと以外は実施例1と同様に実施して、高分子分散剤-1を10質量%含有した、高分子分散剤の水系分散剤溶液cを100部得た。得られた水系分散剤溶液cは、白濁状であるが、静置後1時間で高分子分散剤-1が分離沈降した状態となった。
先に調製した高分子分散剤-1を10部用い、界面活性剤としてドデシルベンゼンスルホン酸の0.2部を用いた以外は実施例1と同様にして、10質量%の高分子分散剤-1の水系分散剤の溶液dを100部得た。得られた溶液dは、高分子分散剤-1が淡い黄濁状に分散しており、これを数日静置しても沈降はほとんど確認されなかった。
高分子分散剤-1の代わりに、先に調製した高分子分散剤-3を使用したこと以外は実施例1と同様に実施し、高分子分散剤-3の10質量%の高分子分散剤の水系分散剤の溶液eを100部得た。得られた溶液eは、高分子分散剤-3が白濁状に分散していたが、数日静置すると、若干沈降気味であった。
先に調製した含水状態のCNF-1(固形分:25%)の40部に、実施例1で得た10質量%の高分子分散剤-1の水系分散処理剤の溶液aを100部添加し、十分に混合して、高分子分散剤-1で処理された易分散性セルロース組成物を得た。次に、得られた易分散性セルロース組成物に、微粒子状ポリエチレン(住友精化製、フロービーズHE3040(商品名)、以下「微粒子状のPE」と略す)80部を水50部で湿潤させた状態で添加し、混合した。更に、得られた混合物を、ろ過及び乾燥することで、高分子分散剤-1で処理されたCNF-1と、微粒子状のPEとの混合組成物であるセルロース分散樹脂組成物aを98部得た。
実施例6で使用した高分子分散剤-1の水系分散処理剤の溶液aの替りに、実施例2で得た10質量%の高分子分散剤-2の水系分散処理剤の溶液bを使用したこと以外は実施例6と同様に実施して、高分子分散剤-2で処理されたCNF-1と、微粒子状のPEとの混合組成物であるセルロース分散樹脂組成物bを98部得た。
先に調製した含水状態のCNF-1(固形分:25%)の40部に、比較例1で得た10質量%の高分子分散剤-1のDMDG溶液100部を添加し、十分に混合した。次に、この混合物にエタノール800部を添加して混合し、ろ過を実施した。更に、実施例6で使用したと同様の微粒子状のPE80部を、エタノール800部で湿潤させた状態で添加して混合し、ろ過を実施して、混合物から残存DMDGを除去したそして、乾燥することで、混合物中のエタノール及び水を除去し、高分子分散剤-1のDMDG溶液で処理したCNF-1と、微粒子状のPEとの混合組成物である比較例の樹脂混合組成物を99部得た。
実施例6で使用した高分子分散剤-1の水系分散処理剤の溶液aの替りに、比較例2の10質量%の高分子分散剤-1の水系分散剤の溶液cを使用したこと以外は実施例6と同様に実施して、高分子分散剤-1で処理したCNF-1と、微粒子状のPEとの混合組成物であるセルロース分散樹脂組成物cを98部得た。
実施例6で使用した高分子分散剤-1の水系分散処理剤の溶液aの替りに、実施例3で作製した10質量%の高分子分散剤-1の水系分散処理剤の溶液dを使用したこと以外は実施例6と同様に実施して、高分子分散剤-1で処理されたCNF-1と、微粒子状のPEとの混合組成物であるセルロース分散樹脂組成物dを98部得た。
実施例6で使用した高分子分散剤-1の水系分散処理剤の溶液aの替りに、実施例5で作製した10質量%の高分子分散剤-3の水系分散処理剤の溶液eを使用したこと以外は実施例6と同様に実施して、高分子分散剤-3で処理されたCNF-1と、微粒子状のPEとの混合組成物であるセルロース分散樹脂組成物eを99部得た。
上記した実施例5~8及び比較例3、4でそれぞれに得たCNF-1と微粒子状のPEとの混合組成物について、それぞれ、下記に述べる方法で、二軸押出混練、射出成型、引張試験による引張弾性率、引張強度の評価を実施した。具体的には、二軸押出混練を、混練温度140℃で実施し、ストランド状に吐出して冷却、ペレタイザーでカッティングして、CNF-1が分散したPE樹脂ペレットをそれぞれ調製した。また、得られたCNF-1が分散したPE樹脂ペレットのそれぞれに用い、射出成型を実施し、ダンベル片(ダンベル厚:2mm)を作製して評価用サンプルとした。得られた評価用サンプルのダンベル片のそれぞれについて、引張試験機(インストロン社製:万能試験機5900シリーズ使用)で、10mm/minの引張速度で引張試験を実施し、引張弾性率及び引張強度を測定し、評価した。その結果を表2にまとめて示した。
先に調製した含水状態のCNF-1(固形分:25%)の40部に、実施例1で得た高分子分散剤-1の水系分散処理剤の溶液aを100部添加し、十分に混合して、高分子分散剤-1で処理された易分散性セルロース組成物を得た。次に、得られた易分散性セルロース組成物に、微粒子状のPEを80部、水50部で湿潤させた状態で添加し、混合した。得られた混合物を軽くろ過し、乾燥処理することなく、液分を含んだ、高分子分散剤-1で処理されたCNF-1と、微粒子状のPEとの混合組成物であるセルロース分散樹脂組成物a’を、150部得た。
実施例9で得たセルロース分散樹脂組成物a’を用い、基本的には先の評価-1で行ったと同様にして、CNF-1が分散したPE樹脂ペレットを調製し、評価用サンプルを作製した。具体的には、二軸押出混練を、混練温度140℃で実施した際に、セルロース分散樹脂組成物a’中の液分除去用にベント孔を開放し、ストランド状に吐出して冷却、ペレタイザーでカッティングしてCNF-1が分散したPE樹脂ペレットを調製した。それ以外は、先の評価-1と同様に、得られた評価用サンプルについて引張弾性率及び引張強度を測定し、評価した。表2に、機械特性評価結果をまとめて示した。
Claims (12)
- セルロースの樹脂中への分散性を高めた易分散性セルロース組成物の製造方法であって、樹脂親和性セグメントAと、セルロース吸着性セグメントBとを有するブロック共重合体構造を有する高分子分散剤を親水性有機溶剤溶液に溶解し、これに界面活性剤を添加し、その後に水を添加することで前記高分子分散剤を含有した水系の分散処理剤を作製し、得られた水系の分散処理剤を、含水状態又は乾燥状態のセルロースに添加して易分散性セルロース組成物を得ることを特徴とする易分散性セルロース組成物の製造方法。
- 前記界面活性剤が、脂肪族アミン類のカルボン酸塩、無機酸塩及び4級アンモニウム塩からなる群から選ばれる少なくともいずれかである請求項1に記載の易分散性セルロース組成物の製造方法。
- 前記親水性有機溶剤が、アルコール系溶剤又はグリコール系溶剤である請求項1又は2に記載の易分散性セルロース組成物の製造方法。
- 前記セルロースが、セルロースナノファイバー、セルロースナノクリスタル、パルプ、リグノセルロース及び木粉からなる群から選ばれる少なくとも1種の、含水状態又は乾燥状態のセルロース繊維である請求項1~3のいずれか1項に記載の易分散性セルロース組成物の製造方法。
- 前記高分子分散剤が、更に、下記(1)~(5)の要件をすべて満たすブロック共重合体である請求項1~4のいずれか1項に記載の易分散性セルロース組成物の製造方法。
(1)前記A-Bブロック共重合体の構成成分の90質量%以上がメタクリレート系モノマーで構成されていること;
(2)前記セルロース吸着性セグメントBは、構成成分の50質量%以上が、水酸基を1個以上有するメタクリレート系モノマー及び/又は尿素基を有するメタクリレート系モノマーで構成されており、且つ、樹脂との相溶性がないこと;
(3)前記樹脂親和性セグメントAのゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が500~20000であり、且つ、前記A-B共重合体全体に占める該樹脂親和性セグメントAの割合が5~95質量%であること;
(4)前記セルロース吸着性セグメントBのゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が500~20000であり、且つ、前記A-B共重合体全体に占める該セルロース吸着性セグメントBの割合が5~95質量%であること;
(5)前記A-Bブロック共重合体のゲルパーミエーションクロマトグラフィーにおけるポリスチレン換算の数平均分子量が3500~40000であり、且つ、分子量分布指数(重量平均分子量/数平均分子量)が1.0~1.6であること。 - 前記(2)のセルロース吸着性セグメントBの構成成分の70質量%以上が、水酸基を1個以上有するメタクリレート系モノマー及び/又は尿素基を有するメタクリレート系モノマーで構成されており、更に、構成成分の3~15質量%が、アルカリで中和されたメタクリル酸及び/又はカルボキシ基を有するメタクリレート系モノマー又は第4級アンモニウム塩基を有するメタクリレート系モノマーで構成されている請求項5に記載の易分散性セルロース組成物の製造方法。
- 前記高分子分散剤が、有機ヨウ素化合物を開始化合物とし、リン化合物、窒素化合物、酸素化合物又は炭素化合物を触媒とするリビングラジカル重合法である可逆連鎖移動触媒重合(RTCP)法で合成されたものである請求項5又は6に記載の易分散性セルロース組成物の製造方法。
- 請求項1~7のいずれか1項に記載の易分散性セルロース組成物の製造方法により得られたことを特徴とする易分散性セルロース組成物。
- 請求項8に記載の易分散性セルロース組成物と、樹脂とを含む樹脂組成物を溶融混練して得られたものであることを特徴とするセルロース分散樹脂組成物。
- 前記樹脂組成物が、前記易分散性セルロース組成物に起因する液分を含んだまま溶融混練して得られたものである請求項9に記載のセルロース分散樹脂組成物。
- 前記樹脂が、熱可塑性樹脂である請求項9又は10に記載のセルロース分散樹脂組成物。
- 樹脂親和性セグメントAと、セルロース吸着性セグメントBとを有するブロック共重合体構造を有する高分子分散剤を親水性有機溶剤溶液に溶解し、これに界面活性剤を添加し、その後に水を添加することで高分子分散剤を含有した水系の分散処理剤を製造することを特徴とする、セルロースの樹脂中への分散性を高めた易分散性セルロース組成物の製造に用いる水系の分散処理剤の製造方法。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201580017739.6A CN106164155B (zh) | 2014-03-31 | 2015-03-30 | 易分散性纤维素组合物及制造方法、纤维素分散树脂组合物及水系纤维素分散剂的制造方法 |
| EP15773988.9A EP3127955B1 (en) | 2014-03-31 | 2015-03-30 | Production method for readily dispersible cellulose composition, readily dispersible cellulose composition, cellulose dispersion resin composition, and production method for water-based dispersant for cellulose |
| US15/125,868 US10077357B2 (en) | 2014-03-31 | 2015-03-30 | Production method for readily dispersible cellulose composition, readily dispersible cellulose composition, cellulose dispersion resin composition, and production method for water-based dispersant for cellulose |
| JP2015525664A JP5825653B1 (ja) | 2014-03-31 | 2015-03-30 | 易分散性セルロース組成物の製造方法、及びセルロース用の水系の分散処理剤の製造方法 |
| CA2944415A CA2944415C (en) | 2014-03-31 | 2015-03-30 | Production method for readily dispersible cellulose composition, readily dispersible cellulose composition, cellulose dispersion resin composition, and production method for water-based dispersant for cellulose |
| PH12016501851A PH12016501851B1 (en) | 2014-03-31 | 2016-09-21 | Production method for readily dispersible cellulose composition, readily dispersible cellulose composition, cellulose dispersion resin composition, and production method for water-based dispersant for cellulose |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014072483 | 2014-03-31 | ||
| JP2014-072483 | 2014-03-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015152189A1 true WO2015152189A1 (ja) | 2015-10-08 |
Family
ID=54240510
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/060029 Ceased WO2015152189A1 (ja) | 2014-03-31 | 2015-03-30 | 易分散性セルロース組成物の製造方法、易分散性セルロース組成物、セルロース分散樹脂組成物及びセルロース用の水系の分散処理剤の製造方法 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US10077357B2 (ja) |
| EP (1) | EP3127955B1 (ja) |
| JP (1) | JP5825653B1 (ja) |
| CN (1) | CN106164155B (ja) |
| CA (1) | CA2944415C (ja) |
| MY (1) | MY171305A (ja) |
| PH (1) | PH12016501851B1 (ja) |
| WO (1) | WO2015152189A1 (ja) |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016017096A (ja) * | 2014-07-04 | 2016-02-01 | 王子ホールディングス株式会社 | 繊維含有樹脂組成物の製造方法 |
| JP2016104865A (ja) * | 2014-03-31 | 2016-06-09 | 大日精化工業株式会社 | セルロース分散用分散剤含有の樹脂組成物及びセルロース用高分子分散剤 |
| JP2017128630A (ja) * | 2016-01-18 | 2017-07-27 | 旭化成株式会社 | セルロース製剤 |
| JP2017141323A (ja) * | 2016-02-08 | 2017-08-17 | 大日精化工業株式会社 | 易分散性セルロース組成物、易分散性セルロース組成物の製造方法、セルロース分散樹脂組成物及びセルロース分散樹脂組成物の製造方法 |
| US10077357B2 (en) | 2014-03-31 | 2018-09-18 | Dainichiseika Color & Chemicals Mfg. Co., Ltd. | Production method for readily dispersible cellulose composition, readily dispersible cellulose composition, cellulose dispersion resin composition, and production method for water-based dispersant for cellulose |
| WO2018181224A1 (ja) * | 2017-03-29 | 2018-10-04 | 日本製紙株式会社 | ゴム組成物およびその製造方法 |
| JP2019172891A (ja) * | 2018-03-29 | 2019-10-10 | 大阪瓦斯株式会社 | バイオマス樹脂組成物 |
| EP3438207A4 (en) * | 2016-03-31 | 2019-11-06 | Furukawa Electric Co., Ltd. | THERMOPLASTIC RESIN COMPOSITION, PRODUCTION METHOD FOR THERMOPLASTIC RESIN COMPOSITION, CELLULOSE REINFORCED RESIN PRODUCT AND CELLULOSE REINFORCED RESIN PRODUCT MANUFACTURING METHOD |
| WO2020003934A1 (ja) * | 2018-06-29 | 2020-01-02 | 新中村化学工業株式会社 | セルロース繊維分散用複合体及びセルロース繊維組成物 |
| JP2020007525A (ja) * | 2018-06-29 | 2020-01-16 | 新中村化学工業株式会社 | ナノセルロース組成物 |
| JP2020083983A (ja) * | 2018-11-21 | 2020-06-04 | 大日精化工業株式会社 | セルロース分散樹脂組成物、その製造方法、及びセルロース樹脂複合材 |
| JP2020128506A (ja) * | 2019-02-08 | 2020-08-27 | 王子ホールディングス株式会社 | 繊維状セルロース含有組成物及び塗料 |
| JP2021001292A (ja) * | 2019-06-24 | 2021-01-07 | 住友ゴム工業株式会社 | ナノセルロース・界面活性剤複合体 |
| US11466140B2 (en) | 2016-03-31 | 2022-10-11 | Furukawa Electric Co., Ltd. | Thermoplastic resin composition, method of producing thermoplastic resin composition, molded article of cellulose-reinforced resin, and method of producing molded article of cellulose-reinforced resin |
| US11566118B2 (en) | 2016-02-18 | 2023-01-31 | Starlite Co., Ltd. | Nanofiber dispersion, method of producing nanofiber dispersion, powdery nanofibers obtainable from the dispersion, resin composition containing the powdery nanofibers ad molding material for 3D printer using the resin composition |
| US11578192B2 (en) | 2017-09-29 | 2023-02-14 | Furukawa Electric Co., Ltd. | Molded article |
| US11597818B2 (en) | 2017-09-29 | 2023-03-07 | Furukawa Electric Co., Ltd. | Molded article |
| US11629244B2 (en) | 2016-03-31 | 2023-04-18 | Furukawa Electric Co., Ltd. | Thermoplastic resin composition, cellulose-reinforced thermoplastic resin composition, method of producing cellulose-reinforced thermoplastic resin composition, molded article of cellulose-reinforced resin, and method of producing molded article of cellulose-reinforced resin |
| US11746215B2 (en) | 2017-09-29 | 2023-09-05 | Furukawa Electric Co., Ltd. | Molded article |
| US11891498B2 (en) | 2017-10-31 | 2024-02-06 | Furukawa Electric Co., Ltd. | Molded article provided with a resin part |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102537413B1 (ko) * | 2017-07-11 | 2023-05-26 | 스미또모 가가꾸 가부시키가이샤 | 수성 수지 조성물 및 성형체 |
| JP6796111B2 (ja) * | 2018-07-13 | 2020-12-02 | 大王製紙株式会社 | 繊維状セルロース含有物及びその製造方法、繊維状セルロース乾燥体及びその製造方法、並びに繊維状セルロース複合樹脂及びその製造方法 |
| KR20210040430A (ko) * | 2018-08-30 | 2021-04-13 | 오지 홀딩스 가부시키가이샤 | 섬유상 셀룰로오스 함유 조성물, 액상 조성물 및 성형체 |
| CN109251259B (zh) * | 2018-09-12 | 2020-12-25 | 福州大学 | 一种基于催化剂和Finkelstein反应实现可控自由基聚合的方法 |
| CN110154186B (zh) * | 2019-04-09 | 2021-06-29 | 中国林业科学研究院林业新技术研究所 | 一种水性木材着色剂及着色方法 |
| WO2020240935A1 (ja) * | 2019-05-27 | 2020-12-03 | 中越パルプ工業株式会社 | 溶融混合物、溶融混合物の製造方法、組成物、組成物の製造方法並びに成形品 |
| CN112300419B (zh) * | 2020-09-28 | 2023-02-03 | 浙江理工大学 | 一种具有环境响应性的纤维素气凝胶的制备方法 |
| CN112979877A (zh) * | 2021-02-22 | 2021-06-18 | 文水县是大高分子材料有限公司 | 一种纤维素基分散剂及其制备方法和应用、水煤浆 |
| US12043808B2 (en) | 2021-12-28 | 2024-07-23 | Afton Chemical Corporation | Quaternary ammonium salt combinations for injector cleanliness |
| JP2024060659A (ja) * | 2022-10-20 | 2024-05-07 | 大日精化工業株式会社 | 高分子分散剤、セルロースファイバー樹脂組成物、及びその検査方法 |
| CN120437084B (zh) * | 2025-05-14 | 2025-11-21 | 山东润君药业有限公司 | 一种缓释载药颗粒及其制备方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002146116A (ja) * | 2000-11-13 | 2002-05-22 | Calp Corp | ポリオレフィン系樹脂組成物 |
| JP2009138024A (ja) * | 2007-12-03 | 2009-06-25 | Chuo Rika Kogyo Corp | 繊維含有脂肪族ポリエステル系樹脂組成物、及びその製造方法 |
| WO2014133019A1 (ja) * | 2013-02-26 | 2014-09-04 | 王子ホールディングス株式会社 | セルロース及び分散剤を含む組成物 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5322470B2 (ja) | 2007-03-26 | 2013-10-23 | 国立大学法人京都大学 | 表面改質ミクロフィブリル化セルロース及びこれを含有してなる複合化樹脂 |
| US9512321B2 (en) * | 2010-03-02 | 2016-12-06 | Dainichiseika Color & Chemicals Mfg. Co., Ltd. | Alkoxysilyl group-containing block copolymer, method for producing the same, resin-treated pigment, and pigment dispersion |
| WO2012111408A1 (ja) * | 2011-02-15 | 2012-08-23 | 日産化学工業株式会社 | 繊維状樹脂補強剤とその製造方法、およびそれを用いた樹脂組成物 |
| EP3127955B1 (en) | 2014-03-31 | 2024-11-27 | Dainichiseika Color & Chemicals Mfg. Co., Ltd. | Production method for readily dispersible cellulose composition, readily dispersible cellulose composition, cellulose dispersion resin composition, and production method for water-based dispersant for cellulose |
| MY174331A (en) | 2014-03-31 | 2020-04-08 | Dainichiseika Color Chem | Polymer dispersant for cellulose, aqueous dispersion treatment agent containing same, readily dispersible cellulose composition, cellulose dispersion resin composition, and dispersant-containing resin composition for cellulose dispersion |
-
2015
- 2015-03-30 EP EP15773988.9A patent/EP3127955B1/en active Active
- 2015-03-30 CN CN201580017739.6A patent/CN106164155B/zh active Active
- 2015-03-30 CA CA2944415A patent/CA2944415C/en active Active
- 2015-03-30 MY MYPI2016703453A patent/MY171305A/en unknown
- 2015-03-30 US US15/125,868 patent/US10077357B2/en active Active
- 2015-03-30 WO PCT/JP2015/060029 patent/WO2015152189A1/ja not_active Ceased
- 2015-03-30 JP JP2015525664A patent/JP5825653B1/ja active Active
-
2016
- 2016-09-21 PH PH12016501851A patent/PH12016501851B1/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002146116A (ja) * | 2000-11-13 | 2002-05-22 | Calp Corp | ポリオレフィン系樹脂組成物 |
| JP2009138024A (ja) * | 2007-12-03 | 2009-06-25 | Chuo Rika Kogyo Corp | 繊維含有脂肪族ポリエステル系樹脂組成物、及びその製造方法 |
| WO2014133019A1 (ja) * | 2013-02-26 | 2014-09-04 | 王子ホールディングス株式会社 | セルロース及び分散剤を含む組成物 |
Non-Patent Citations (8)
| Title |
|---|
| "New Energy and Industrial Technology Development Organization (NEDO", HEISEI 22 NENDO - HEISEI 24 NENDO SEIKA HOKOKUSHO, 2013, pages 39 - 42, XP008184964 * |
| KEITA SAKAKIBARA ET AL.: "Cellulose Nanofiber ni Tekishita Kobunshi Bunsanzai no Kaihatsu to Jushi Fukugo Zairyo eno Oyo", THE SOCIETY OF FIBER SCIENCE AND TECHNOLOGY, JAPAN YOKOSHU, vol. 68, no. 1, 2013, pages 2H15, XP008185728 * |
| KEITA SAKAKIBARA ET AL.: "Diblock Kyojugotai Tenka ni yoru Cellulose Nanofiber/Jushi Fukugo Zairyo no Sakusei", THE CELLULOSE SOCIETY OF JAPAN NENJI TAIKAI KOEN YOSHISHU, vol. 20, 2013, pages 67, XP008184963 * |
| KEITA SAKAKIBARA ET AL.: "Kobunshi Bunsanzai o Mochiita Cellulose Nanofiber Kyoka Fukugo Zairyo no Koseinoka to Kozo Hyoka", THE SOCIETY OF FIBER SCIENCE AND TECHNOLOGY, JAPAN YOKOSHU, vol. 68, no. 2, 2013, pages 72, XP008185297 * |
| KEITA SAKAKIBARA ET AL.: "Kobunshi Bunsanzai o Mochiita Cellulose Nanofiber Kyoka Jushi Zairyo no Kaihatsu", SEIKEI KAKO, vol. 24, 2013, pages 119 - 120, XP008184823 * |
| KEITA SAKAKIBARA ET AL.: "Polymer Dispersants for Cellulose Nanofiber Reinforced Polyolefin Composite Materials", FUNCTION & MATERIALS, vol. 34, no. 11, 2014, pages 40 - 45, XP008184746 * |
| KEITA SAKAKIBARA: "Polymer Brush no Tokusei o Ikashita Cellulose Nanofiber Kyoka Jushi Zairyo no Kaihatsu", SEN'I TO KOGYO, vol. 70, no. 8, 2014, pages 276 - 280, XP008184967 * |
| YOSHINOBU TSUJII: "Kobunshi Bunsanzai ni yoru Mokuzai Yurai NC no Kaimen Kino Seigyo to Jushi Fukugo Zairyo eno Oyo", DAI 280 KAI SEIZONKEN SYMPOSIUM, 2015, pages 59 - 62, XP008184966 * |
Cited By (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016104865A (ja) * | 2014-03-31 | 2016-06-09 | 大日精化工業株式会社 | セルロース分散用分散剤含有の樹脂組成物及びセルロース用高分子分散剤 |
| US10077357B2 (en) | 2014-03-31 | 2018-09-18 | Dainichiseika Color & Chemicals Mfg. Co., Ltd. | Production method for readily dispersible cellulose composition, readily dispersible cellulose composition, cellulose dispersion resin composition, and production method for water-based dispersant for cellulose |
| US10081001B2 (en) | 2014-03-31 | 2018-09-25 | Dainichiseika Color & Chemicals Mfg. Co., Ltd. | Polymer dispersant for cellulose, aqueous dispersion treatment agent containing same, readily dispersible cellulose composition, cellulose dispersion resin composition, and dispersant-containing resin composition for cellulose dispersion |
| JP2016017096A (ja) * | 2014-07-04 | 2016-02-01 | 王子ホールディングス株式会社 | 繊維含有樹脂組成物の製造方法 |
| JP2017128630A (ja) * | 2016-01-18 | 2017-07-27 | 旭化成株式会社 | セルロース製剤 |
| JP2017141323A (ja) * | 2016-02-08 | 2017-08-17 | 大日精化工業株式会社 | 易分散性セルロース組成物、易分散性セルロース組成物の製造方法、セルロース分散樹脂組成物及びセルロース分散樹脂組成物の製造方法 |
| US11566118B2 (en) | 2016-02-18 | 2023-01-31 | Starlite Co., Ltd. | Nanofiber dispersion, method of producing nanofiber dispersion, powdery nanofibers obtainable from the dispersion, resin composition containing the powdery nanofibers ad molding material for 3D printer using the resin composition |
| EP3438207A4 (en) * | 2016-03-31 | 2019-11-06 | Furukawa Electric Co., Ltd. | THERMOPLASTIC RESIN COMPOSITION, PRODUCTION METHOD FOR THERMOPLASTIC RESIN COMPOSITION, CELLULOSE REINFORCED RESIN PRODUCT AND CELLULOSE REINFORCED RESIN PRODUCT MANUFACTURING METHOD |
| US11629244B2 (en) | 2016-03-31 | 2023-04-18 | Furukawa Electric Co., Ltd. | Thermoplastic resin composition, cellulose-reinforced thermoplastic resin composition, method of producing cellulose-reinforced thermoplastic resin composition, molded article of cellulose-reinforced resin, and method of producing molded article of cellulose-reinforced resin |
| US11597817B2 (en) | 2016-03-31 | 2023-03-07 | Furukawa Electric Co., Ltd | Thermoplastic resin composition, method of producing thermoplastic resin composition, molded article of cellulose-reinforced resin, and method of producing molded article of cellulose-reinforced resin |
| US11466140B2 (en) | 2016-03-31 | 2022-10-11 | Furukawa Electric Co., Ltd. | Thermoplastic resin composition, method of producing thermoplastic resin composition, molded article of cellulose-reinforced resin, and method of producing molded article of cellulose-reinforced resin |
| WO2018181224A1 (ja) * | 2017-03-29 | 2018-10-04 | 日本製紙株式会社 | ゴム組成物およびその製造方法 |
| JPWO2018181224A1 (ja) * | 2017-03-29 | 2020-02-06 | 日本製紙株式会社 | ゴム組成物およびその製造方法 |
| JP7061998B2 (ja) | 2017-03-29 | 2022-05-02 | 日本製紙株式会社 | ゴム組成物およびその製造方法 |
| US11746215B2 (en) | 2017-09-29 | 2023-09-05 | Furukawa Electric Co., Ltd. | Molded article |
| US11597818B2 (en) | 2017-09-29 | 2023-03-07 | Furukawa Electric Co., Ltd. | Molded article |
| US11578192B2 (en) | 2017-09-29 | 2023-02-14 | Furukawa Electric Co., Ltd. | Molded article |
| US11891498B2 (en) | 2017-10-31 | 2024-02-06 | Furukawa Electric Co., Ltd. | Molded article provided with a resin part |
| JP2019172891A (ja) * | 2018-03-29 | 2019-10-10 | 大阪瓦斯株式会社 | バイオマス樹脂組成物 |
| JP7163046B2 (ja) | 2018-03-29 | 2022-10-31 | 大阪瓦斯株式会社 | バイオマス樹脂組成物 |
| US11149132B2 (en) | 2018-06-29 | 2021-10-19 | Shin-Nakamura Chemical Co., Ltd. | Composite for cellulose fiber dispersion and cellulose fiber composition |
| US11667769B2 (en) | 2018-06-29 | 2023-06-06 | Shin-Nakamura Chemical Co., Ltd. | Composite for cellulose fiber dispersion and cellulose fiber composition |
| JP2020007525A (ja) * | 2018-06-29 | 2020-01-16 | 新中村化学工業株式会社 | ナノセルロース組成物 |
| WO2020003934A1 (ja) * | 2018-06-29 | 2020-01-02 | 新中村化学工業株式会社 | セルロース繊維分散用複合体及びセルロース繊維組成物 |
| JP2020083983A (ja) * | 2018-11-21 | 2020-06-04 | 大日精化工業株式会社 | セルロース分散樹脂組成物、その製造方法、及びセルロース樹脂複合材 |
| JP2020128506A (ja) * | 2019-02-08 | 2020-08-27 | 王子ホールディングス株式会社 | 繊維状セルロース含有組成物及び塗料 |
| JP2021001292A (ja) * | 2019-06-24 | 2021-01-07 | 住友ゴム工業株式会社 | ナノセルロース・界面活性剤複合体 |
| JP7322543B2 (ja) | 2019-06-24 | 2023-08-08 | 住友ゴム工業株式会社 | ナノセルロース・界面活性剤複合体 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN106164155A (zh) | 2016-11-23 |
| CA2944415A1 (en) | 2015-10-08 |
| PH12016501851B1 (en) | 2021-03-10 |
| EP3127955A1 (en) | 2017-02-08 |
| EP3127955A4 (en) | 2018-01-03 |
| JPWO2015152189A1 (ja) | 2017-04-13 |
| EP3127955B1 (en) | 2024-11-27 |
| PH12016501851A1 (en) | 2016-12-19 |
| JP5825653B1 (ja) | 2015-12-02 |
| CA2944415C (en) | 2018-05-15 |
| US10077357B2 (en) | 2018-09-18 |
| MY171305A (en) | 2019-10-08 |
| US20170002182A1 (en) | 2017-01-05 |
| CN106164155B (zh) | 2018-09-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5825653B1 (ja) | 易分散性セルロース組成物の製造方法、及びセルロース用の水系の分散処理剤の製造方法 | |
| JP5904520B2 (ja) | セルロース用高分子分散剤の製造方法、セルロース用高分子分散剤、高分子分散剤含有の水系分散処理剤、易分散性セルロース組成物、セルロース分散樹脂組成物、セルロース分散用分散剤含有の樹脂組成物、及び、水系分散処理剤含有の樹脂組成物 | |
| JP6234037B2 (ja) | セルロース及び分散剤を含む組成物 | |
| JP6591304B2 (ja) | 易分散性セルロース組成物、易分散性セルロース組成物の製造方法、セルロース分散樹脂組成物及びセルロース分散樹脂組成物の製造方法 | |
| Kalia et al. | Nanofibrillated cellulose: surface modification and potential applications | |
| JP6245779B2 (ja) | 誘導体化cnfの製造方法及び高分子化合物樹脂組成物の製造方法 | |
| JP2014193959A (ja) | 植物繊維含有樹脂組成物及びその製造方法 | |
| Cheng et al. | Preparation of acetylated nanofibrillated cellulose from corn stalk microcrystalline cellulose and its reinforcing effect on starch films | |
| JP6737864B2 (ja) | 化学修飾セルロース繊維およびその製造方法 | |
| Builes et al. | Unsaturated polyester nanocomposites modified with fibrillated cellulose and PEO-b-PPO-b-PEO block copolymer | |
| JP2017052888A (ja) | セルロース微小繊維分散液、セルロース微小繊維分散液の製造方法及び繊維複合樹脂 | |
| Moreau et al. | Bifunctionalization of cellulose fibers by one-step Williamson’s etherification to obtain modified microfibrillated cellulose | |
| JP6871079B2 (ja) | 解繊セルロース繊維の製造方法、及び樹脂組成物の製造方法 | |
| JP6545758B2 (ja) | セルロース及び分散剤を含む組成物 | |
| JP6954884B2 (ja) | セルロース分散樹脂組成物、その製造方法、及びセルロース樹脂複合材 | |
| JP2024060659A (ja) | 高分子分散剤、セルロースファイバー樹脂組成物、及びその検査方法 | |
| JP7340195B2 (ja) | 微細セルロース繊維、その製造方法、スラリー及び複合体 | |
| Zhang | Hydrogen Bond Reconfiguration in Wood Polysaccharides Towards Functional Materials | |
| JP2025506191A (ja) | 多官能基化セルロースを調製する方法および多官能基化ナノフィブリル化セルロースを調製するためのその使用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2015525664 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15773988 Country of ref document: EP Kind code of ref document: A1 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015773988 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015773988 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15125868 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12016501851 Country of ref document: PH |
|
| ENP | Entry into the national phase |
Ref document number: 2944415 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |