WO2015178241A1 - 電気化学素子電極用複合粒子および電気化学素子電極用複合粒子の製造方法 - Google Patents
電気化学素子電極用複合粒子および電気化学素子電極用複合粒子の製造方法 Download PDFInfo
- Publication number
- WO2015178241A1 WO2015178241A1 PCT/JP2015/063561 JP2015063561W WO2015178241A1 WO 2015178241 A1 WO2015178241 A1 WO 2015178241A1 JP 2015063561 W JP2015063561 W JP 2015063561W WO 2015178241 A1 WO2015178241 A1 WO 2015178241A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composite
- water
- active material
- electrode active
- composite particles
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G11/00—Hybrid capacitors, i.e. capacitors having different positive and negative electrodes; Electric double-layer [EDL] capacitors; Processes for the manufacture thereof or of parts thereof
- H01G11/22—Electrodes
- H01G11/30—Electrodes characterised by their material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G11/00—Hybrid capacitors, i.e. capacitors having different positive and negative electrodes; Electric double-layer [EDL] capacitors; Processes for the manufacture thereof or of parts thereof
- H01G11/22—Electrodes
- H01G11/30—Electrodes characterised by their material
- H01G11/48—Conductive polymers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G11/00—Hybrid capacitors, i.e. capacitors having different positive and negative electrodes; Electric double-layer [EDL] capacitors; Processes for the manufacture thereof or of parts thereof
- H01G11/22—Electrodes
- H01G11/30—Electrodes characterised by their material
- H01G11/50—Electrodes characterised by their material specially adapted for lithium-ion capacitors, e.g. for lithium-doping or for intercalation
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/134—Electrodes based on metals, Si or alloys
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/139—Processes of manufacture
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
- H01M4/621—Binders
- H01M4/622—Binders being polymers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
- H01M4/624—Electric conductive fillers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/139—Processes of manufacture
- H01M4/1395—Processes of manufacture of electrodes based on metals, Si or alloys
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
- H01M4/386—Silicon or alloys based on silicon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/13—Energy storage using capacitors
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02T—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO TRANSPORTATION
- Y02T10/00—Road transport of goods or passengers
- Y02T10/60—Other road transportation technologies with climate change mitigation effect
- Y02T10/70—Energy storage systems for electromobility, e.g. batteries
Definitions
- the present invention relates to a composite particle for an electrochemical element electrode and a method for producing a composite particle for an electrochemical element electrode.
- Lithium ion secondary batteries have a relatively high energy density and are used in mobile fields such as mobile phones and notebook personal computers.
- the electric double layer capacitor can be rapidly charged and discharged, the electric double layer capacitor is expected to be used as an auxiliary power source for an electric vehicle or the like in addition to being used as a memory backup small power source for a personal computer or the like.
- the lithium ion capacitor that takes advantage of the lithium ion secondary battery and the electric double layer capacitor has higher energy density and output density than the electric double layer capacitor.
- Application to applications that could not meet the specifications for capacitor performance is being considered.
- lithium ion secondary batteries have been studied for application not only to in-vehicle applications such as hybrid electric vehicles and electric vehicles, but also to power storage applications.
- Electrodes for electrochemical devices include coated electrodes manufactured by a method in which a slurry for coated electrodes containing an electrode active material, a binder resin, a conductive agent, etc. is coated on a current collector and the solvent is removed by heat or the like.
- a slurry for coated electrodes containing an electrode active material, a binder resin, a conductive agent, etc. is coated on a current collector and the solvent is removed by heat or the like.
- this method has a high cost and a poor working environment, and the manufacturing apparatus tends to be large.
- Patent Document 1 discloses composite particles obtained by spraying and drying a slurry for composite particles containing an electrode active material, a binder resin, and a dispersion medium. A method of forming an electrode active material layer using composite particles is disclosed.
- an electrode active material layer is formed by pressurizing composite particles.
- the pressure during pressurization is too strong, the electrode active material may be destroyed, and the resulting electrochemical device is stored at high temperature. There was a possibility that the characteristics deteriorated.
- An object of the present invention is to provide a composite particle for an electrochemical element electrode capable of producing an electrochemical element having excellent high-temperature storage characteristics.
- the present inventor has found that the above object can be achieved by using a composite of a water-soluble polymer and crystalline cellulose, and has completed the present invention.
- Electrode active material (A), particulate binder resin (B), water-soluble polymer (C) and composite of water-soluble polymer (d ′) and crystalline cellulose (d ′′) (D)
- a composite particle for an electrochemical element electrode comprising: (2) 0.1 to 10 parts by weight of the water-soluble polymer (C) and 0.1 to 2.0 parts by weight of the composite (D) with respect to 100 parts by weight of the electrode active material (A)
- Composite particles for chemical element electrodes (3)
- the composite (D) is a composite particle for an electrochemical element electrode according to (1) or (2), which has a property of being dispersed in water.
- the particulate binder resin (B) includes at least one of a conjugated diene polymer or an acrylate polymer, (1) to the composite particles for electrode for an electrochemical element according to any one of (3), (5) The composite particles for an electrochemical element electrode according to any one of (1) to (4), wherein the composite (D) has a primary average particle diameter of 10 ⁇ m or less.
- the step of obtaining the composite (D) by surface treatment, the electrode active material (A), the particulate binder resin (B), the water-soluble polymer (C), and the composite (D) Provided is a method for producing composite particles for electrochemical device electrodes, comprising: a step of mixing to obtain a composite particle slurry; and a step of obtaining composite particles for an electrochemical device electrode by granulating the composite particle slurry.
- an electrochemical element electrode composite particle capable of producing an electrochemical element having excellent high-temperature storage characteristics, and to provide a method for producing this electrochemical element electrode composite particle. it can.
- the composite particle for an electrochemical element electrode of the present invention (hereinafter sometimes referred to as “composite particle”) includes an electrode active material (A), a particulate binder resin (B), a water-soluble polymer (C), and a water-soluble polymer.
- positive electrode active material means an electrode active material for a positive electrode
- negative electrode active material means an electrode active material for a negative electrode
- the “positive electrode active material layer” means an electrode active material layer provided on the positive electrode
- the “negative electrode active material layer” means an electrode active material layer provided on the negative electrode.
- the positive electrode active material is an active material that can be doped and dedoped with lithium ions, and is broadly classified into an inorganic compound and an organic compound.
- Examples of the positive electrode active material made of an inorganic compound include transition metal oxides, transition metal sulfides, lithium-containing composite metal oxides of lithium and transition metals, and the like.
- Examples of the transition metal include Ti, V, Cr, Mn, Fe, Co, Ni, Cu, and Mo.
- Transition metal oxides include MnO, MnO 2 , V 2 O 5 , V 6 O 13 , TiO 2 , Cu 2 V 2 O 3 , amorphous V 2 O—P 2 O 5 , MoO 3 , V 2 O. 5 , V 6 O 13 and the like. Among them, MnO, V 2 O 5 , V 6 O 13 and TiO 2 are preferable from the viewpoint of cycle stability and capacity.
- the lithium-containing composite metal oxide include a lithium-containing composite metal oxide having a layered structure, a lithium-containing composite metal oxide having a spinel structure, and a lithium-containing composite metal oxide having an olivine structure.
- lithium-containing composite metal oxide having a layered structure examples include lithium-containing cobalt oxide (LiCoO 2 ) (hereinafter sometimes referred to as “LCO”), lithium-containing nickel oxide (LiNiO 2 ), and Co—Ni—Mn.
- LCO lithium-containing cobalt oxide
- LiNiO 2 lithium-containing nickel oxide
- Co—Ni—Mn examples thereof include lithium composite oxides, lithium composite oxides of Ni—Mn—Al, and lithium composite oxides of Ni—Co—Al.
- lithium-containing composite metal oxide having a spinel structure examples include lithium manganate (LiMn 2 O 4 ) and Li [Mn 3/2 M 1/2 ] O 4 in which a part of Mn is substituted with another transition metal (here, M may be Cr, Fe, Co, Ni, Cu or the like.
- Li x MPO 4 (wherein, M is Mn, Fe, Co, Ni, Cu, Mg, Zn, V, Ca, Sr, Ba, Ti, Al, and the like) is a lithium-containing composite metal oxide having an olivine structure.
- An olivine type lithium phosphate compound represented by at least one selected from Si, B, and Mo, 0 ⁇ X ⁇ 2) may be mentioned.
- a conductive polymer such as polyacetylene or poly-p-phenylene can be used.
- An iron-based oxide having poor electrical conductivity may be used as a positive electrode active material covered with a carbon material by allowing a carbon source material to be present during reduction firing. These compounds may be partially element-substituted.
- the positive electrode active material may be a mixture of the above inorganic compound and organic compound.
- the positive electrode active material may be any material that can reversibly carry lithium ions and anions such as tetrafluoroborate.
- carbon allotropes can be preferably used, and electrode active materials used in electric double layer capacitors can be widely used.
- Specific examples of the allotrope of carbon include activated carbon, polyacene (PAS), carbon whisker, carbon nanotube, and graphite.
- the volume average particle size of the positive electrode active material is appropriately selected in consideration of other components of the electrode for an electrochemical element, but preferably 1 from the viewpoint of improving the characteristics of the electrochemical element such as load characteristics and cycle characteristics. It is ⁇ 50 ⁇ m, more preferably 2 to 30 ⁇ m.
- examples of the negative electrode active material in the case where the electrochemical device is a lithium ion secondary battery include a material that can transfer electrons in the negative electrode of the electrochemical device.
- a material that can occlude and release lithium can be usually used as the negative electrode active material in the case where the electrochemical device is a lithium ion secondary battery.
- Examples of the negative electrode active material preferably used for the lithium ion secondary battery include a negative electrode active material formed of carbon.
- Examples of the negative electrode active material formed of carbon include natural graphite, artificial graphite, and carbon black. Among them, graphite such as artificial graphite and natural graphite is preferable, and natural graphite is particularly preferable.
- the negative electrode active material preferably used for the lithium ion secondary battery is a negative electrode active material containing a metal.
- a negative electrode active material containing at least one selected from the group consisting of tin, silicon, germanium and lead is preferable.
- the negative electrode active material containing these elements can reduce the irreversible capacity.
- negative electrode active materials containing silicon are preferable.
- a negative electrode active material containing silicon By using a negative electrode active material containing silicon, the electric capacity of the lithium ion secondary battery can be increased.
- a negative electrode active material containing silicon expands and contracts greatly (for example, about 5 times) with charge and discharge, but the composite particles of the present invention have a strength that can withstand the expansion and contraction of the negative electrode active material containing silicon. Have. Therefore, in the negative electrode manufactured using the composite particles of the present invention, it is possible to effectively suppress a decrease in battery performance due to expansion and contraction of the negative electrode active material containing silicon.
- Examples of the negative electrode active material containing silicon include silicon-containing compounds (hereinafter sometimes referred to as “silicon-containing compounds”) and metallic silicon.
- the silicon-containing compound is a compound of silicon and another element, and examples thereof include SiO, SiO 2 , SiO x (0.01 ⁇ x ⁇ 2), SiC, and SiOC.
- SiO x , SiOC, and SiC are preferable, SiO x and SiOC are more preferable from the viewpoint of battery life, and SiO x is particularly preferable from the viewpoint of suppressing swelling of the negative electrode.
- SiO x is a compound that can be formed from one or both of SiO and SiO 2 and metallic silicon. This SiO x can be produced, for example, by cooling and precipitating silicon monoxide gas generated by heating a mixture of SiO 2 and metal silicon.
- the amount of the silicon-containing compound in the negative electrode active material is preferably 1 to 50% by weight, more preferably 5 to 40% by weight, and particularly preferably 10 to 30% by weight. %. If the compounding amount of the silicon-containing compound is too small, the capacity when a lithium ion secondary battery is produced becomes small. Moreover, when there are too many compounding quantities of a silicon-containing compound, a negative electrode will swell. Moreover, as a negative electrode active material, 1 type may be used independently and 2 or more types may be used in combination by arbitrary ratios.
- the negative electrode active material is preferably sized in the form of particles.
- the volume average particle diameter of the negative electrode active material for a lithium ion secondary battery is preferably 0.1 to 100 ⁇ m, more preferably 0.5 to 50 ⁇ m, and still more preferably 0.8 to 20 ⁇ m.
- the tap density of the negative electrode active material for the lithium ion secondary battery is not particularly limited, but those having a density of 0.6 g / cm 3 or more are preferably used.
- the negative electrode active material formed with the said carbon is mentioned.
- the particulate binder resin (B) used in the present invention is not particularly limited as long as it is a substance capable of binding the above electrode active materials to each other.
- a dispersion type particulate binder resin having a property of being dispersed in a solvent is preferable.
- the dispersion-type particulate binder resin include high molecular compounds such as silicon polymers, fluorine-containing polymers, conjugated diene polymers, acrylate polymers, polyimides, polyamides, and polyurethanes.
- a fluorine-containing polymer, a conjugated diene polymer, and an acrylate polymer are preferred, and a conjugated diene polymer and an acrylate polymer are preferred. More preferred. These polymers can be used alone or in combination as a dispersion type particulate binder resin.
- the fluorine-containing polymer is a polymer containing a monomer unit containing a fluorine atom.
- Specific examples of the fluorine-containing polymer include polytetrafluoroethylene, polyvinylidene fluoride, tetrafluoroethylene / perfluoroalkyl vinyl ether copolymer, ethylene / tetrafluoroethylene copolymer, ethylene / chlorotrifluoroethylene copolymer, A perfluoroethylene propene copolymer may be mentioned.
- the conjugated diene polymer is a homopolymer of a conjugated diene monomer, a copolymer obtained by polymerizing a monomer mixture containing a conjugated diene monomer, or a hydrogenated product thereof.
- 1,3-butadiene is used in that the flexibility when used as an electrode can be improved and the resistance to cracking can be increased. It is more preferable.
- the monomer mixture may contain two or more of these conjugated diene monomers.
- conjugated diene polymer is a copolymer of the above conjugated diene monomer and a monomer copolymerizable therewith
- examples of the copolymerizable monomer include ⁇ , Examples thereof include a ⁇ -unsaturated nitrile compound and a vinyl compound having an acid component.
- conjugated diene polymers include conjugated diene monomer homopolymers such as polybutadiene and polyisoprene; aromatic vinyl monomers such as carboxy-modified styrene-butadiene copolymer (SBR). Monomer / conjugated diene monomer copolymer; vinyl cyanide monomer / conjugated diene monomer copolymer such as acrylonitrile / butadiene copolymer (NBR); hydrogenated SBR, hydrogenated NBR, etc. Is mentioned.
- conjugated diene monomer homopolymers such as polybutadiene and polyisoprene
- aromatic vinyl monomers such as carboxy-modified styrene-butadiene copolymer (SBR).
- SBR carboxy-modified styrene-butadiene copolymer
- Monomer / conjugated diene monomer copolymer Monomer / conjugated diene monomer copolymer
- the ratio of the conjugated diene monomer unit in the conjugated diene polymer is preferably 20 to 60% by weight, more preferably 30 to 55% by weight.
- the ratio of the conjugated diene monomer unit is too large, the resistance to the electrolytic solution tends to be lowered when an electrode is produced using composite particles containing the particulate binder resin (B).
- the ratio of the conjugated diene monomer unit is too small, there is a tendency that sufficient adhesion between the composite particles and the current collector cannot be obtained.
- the acrylate polymer has the general formula (1): CH 2 ⁇ CR 1 —COOR 2 (wherein R 1 represents a hydrogen atom or a methyl group, R 2 represents an alkyl group or a cycloalkyl group. R 2 further represents A monomer unit derived from a compound represented by an ether group, a hydroxyl group, a phosphate group, an amino group, a carboxyl group, a fluorine atom, or an epoxy group. Copolymer obtained by polymerizing a polymer containing, specifically, a homopolymer of a compound represented by the general formula (1) or a monomer mixture containing the compound represented by the general formula (1) It is a coalescence.
- Specific examples of the compound represented by the general formula (1) include methyl (meth) acrylate, ethyl (meth) acrylate, propyl (meth) acrylate, isopropyl (meth) acrylate, and (meth) acrylate n.
- Acid esters carboxylic acid-containing (meth) acrylic acid esters such as 2- (meth) acryloyloxyethylphthalic acid and 2- (meth) acryloyloxyethylphthalic acid; fluorine such as perfluorooctylethyl (meth) acrylic acid Group-containing (meth) acrylic acid ester; Phosphoric acid group-containing (meth) acrylic acid esters such as ethyl phosphate; Epoxy group-containing (meth) acrylic acid esters such as glycidyl (meth) acrylate; Amino group content such as dimethylaminoethyl (meth) acrylate ( (Meth) acrylic acid ester; and the like.
- fluorine such as perfluorooctylethyl (meth) acrylic acid Group-containing (meth) acrylic acid ester
- Phosphoric acid group-containing (meth) acrylic acid esters such as ethyl phosphate
- (meth) acryl means “acryl” and “methacryl”.
- (Meth) acryloyl means “acryloyl” and “methacryloyl”.
- (meth) acrylic acid esters can be used alone or in combination of two or more.
- (meth) acrylic acid alkyl ester is preferable from the viewpoint of being able to reduce the swellability with respect to the electrolytic solution and improving cycle characteristics, and methyl (meth) acrylate and (meth) acrylic are preferred. More preferred are ethyl acrylate, n-butyl (meth) acrylate, and alkyl (meth) acrylate having an alkyl group with 6 to 12 carbon atoms.
- the acrylate polymer is a copolymer of the compound represented by the general formula (1) and a monomer copolymerizable therewith
- the copolymerizable monomer For example, carboxylic acid esters having two or more carbon-carbon double bonds, aromatic vinyl monomers, amide monomers, olefins, diene monomers, vinyl ketones, and heterocyclic rings
- examples include ⁇ , ⁇ -unsaturated nitrile compounds and vinyl compounds having an acid component.
- the above copolymerizable monomers it is difficult to be deformed when an electrode is produced, and it can be strong, and sufficient adhesion between the electrode active material layer and the current collector can be obtained. It is preferable to use an aromatic vinyl monomer.
- the aromatic vinyl monomer include styrene.
- the proportion of the (meth) acrylic acid ester unit in the acrylate polymer is preferably 50 to 95% by weight from the viewpoint of improving flexibility when used as an electrode and increasing resistance to cracking. More preferably, it is 60 to 90% by weight.
- Examples of the ⁇ , ⁇ -unsaturated nitrile compound used in the polymer constituting the particulate binder resin (B) include acrylonitrile, methacrylonitrile, ⁇ -chloroacrylonitrile, and ⁇ -bromoacrylonitrile. These may be used alone or in combination of two or more. Among these, acrylonitrile and methacrylonitrile are preferable, and acrylonitrile is more preferable.
- the proportion of ⁇ , ⁇ -unsaturated nitrile compound units in the particulate binder resin (B) is preferably 0.1 to 40% by weight, more preferably 0.5 to 30% by weight, and still more preferably 1 to 20%. % By weight.
- the proportion of ⁇ , ⁇ -unsaturated nitrile compound units in the particulate binder resin (B) is preferably 0.1 to 40% by weight, more preferably 0.5 to 30% by weight, and still more preferably 1 to 20%. % By weight.
- vinyl compound having an acid component examples include acrylic acid, methacrylic acid, itaconic acid, maleic acid, and fumaric acid. These may be used alone or in combination of two or more. Among these, acrylic acid, methacrylic acid, and itaconic acid are preferable, methacrylic acid and itaconic acid are more preferable, and itaconic acid is particularly preferable in terms of good adhesion.
- the proportion of the vinyl compound unit having an acid component in the particulate binder resin (B) is preferably 0.5 to 10% by weight, more preferably from the viewpoint of improving the stability of the composite particle slurry. It is 1 to 8% by weight, more preferably 2 to 7% by weight.
- the particulate binder resin (B) used in the present invention is particulate, it has good binding properties, and can suppress deterioration of the capacity of the produced electrode and repeated charge / discharge.
- the particulate binder resin (B) include those in which binder resin particles such as latex are dispersed in water, and powders obtained by drying such a dispersion.
- the average particle diameter of the particulate binder resin (B) is preferably from the viewpoint that the strength and flexibility of the obtained electrode are good while the stability when the slurry for composite particles is made good.
- the thickness is from 001 to 100 ⁇ m, more preferably from 10 to 1000 nm, still more preferably from 50 to 500 nm.
- the method for producing the particulate binder resin (B) used in the present invention is not particularly limited, and a known polymerization method such as an emulsion polymerization method, a suspension polymerization method, a dispersion polymerization method or a solution polymerization method may be employed. it can.
- the particulate binder resin (B) used in the present invention may be particles having a core-shell structure obtained by stepwise polymerization of a mixture of two or more monomers.
- the compounding amount of the particulate binder resin (B) in the composite particle for an electrochemical element electrode of the present invention is such that the electrode has good adhesion between the water-soluble polymer (C) and the composite (D).
- the amount is preferably 0.1 to 50 parts by weight, more preferably 0.5 to 20 parts by weight, still more preferably 1 to 15 parts by weight based on 100 parts by weight of the active material (A).
- the water-soluble polymer (C) used in the present invention refers to a polymer having an undissolved content of less than 10.0% by weight when 0.5 g of the polymer is dissolved in 100 g of pure water at 25 ° C.
- water-soluble polymer (C) examples include cellulosic polymers such as carboxymethylcellulose, methylcellulose, ethylcellulose and hydroxypropylcellulose, and ammonium salts or alkali metal salts thereof, alginates such as propylene glycol alginate, and alginic acid.
- Alginates or polymers such as sodium, sulfonic acid polymers, polyacrylic acid, and poly (meth) acrylates such as sodium poly (meth) acrylate, polyvinyl alcohol, modified polyvinyl alcohol, poly-N-vinylacetamide, poly Ethylene oxide, polyvinyl pyrrolidone, polycarboxylic acid, oxidized starch, phosphate starch, casein, various modified starches, chitin, chitosan derivatives Etc., and the like.
- “(modified) poly” means “unmodified poly” or “modified poly”.
- water-soluble polymers (C) can be used alone or in combination of two or more.
- cellulose-based polymers, polyacrylic acid, polyvinyl alcohol and poly-N-vinylacetamide are preferable from the viewpoint of improving the dispersibility of the composite (D) and high adhesiveness.
- carboxymethylcellulose or its ammonium salt or alkali metal salt is particularly preferable.
- the blending amount of the water-soluble polymer (C) in the composite particles is not particularly limited as long as the effect of the present invention is not impaired.
- the solid content is converted to 100 parts by weight of the electrode active material (A).
- the amount is preferably 0.1 to 10 parts by weight, more preferably 0.2 to 5 parts by weight, still more preferably 0.25 to 5 parts by weight, particularly preferably 0.3 to 2 parts by weight.
- the composite (D) used in the present invention is a composite of a water-soluble polymer (d ′) and crystalline cellulose (d ′′).
- the water-soluble polymer (d ′) used in the present invention is a polymer having an undissolved content of less than 10.0% by weight when 25 g of polymer is dissolved in 100 g of pure water at 25 ° C. .
- Specific examples of the water-soluble polymer (d ′) include cellulosic polymers such as carboxymethylcellulose, methylcellulose, ethylcellulose and hydroxypropylcellulose, and ammonium salts or alkali metal salts thereof, alginates such as propylene glycol alginate, and Alginates such as sodium alginate and polysaccharides such as polymers, dextrin, galarginan and xanthan gum, sulfonic acid polymers, polyacrylic acid, and poly (meth) acrylates such as sodium poly (meth) acrylate, polyvinyl alcohol , Modified polyvinyl alcohol, poly-N-vinylacetamide, polyethylene oxide, polyvinylpyrrolidone, polycarboxylic acid,
- water-soluble polymers (d ′) can be used alone or in combination of two or more.
- cellulose-based polymers and polysaccharide-based polymers are preferable in that the reinforcing effect by crystalline cellulose (d ′′) is high and the strength of the composite particles is improved.
- carboxymethylcellulose or its ammonium is preferred. Salts or alkali metal salts are particularly preferred, and among the polysaccharide-based polymers, dextrin, xanthan gum and garaginan are particularly preferred.
- the crystalline cellulose (d ′′) used in the present invention is obtained by subjecting cellulose materials such as plant pulp fibers and refined linters to depolymerization treatment such as acid hydrolysis, alkali hydrolysis, and enzymatic decomposition. It is done.
- the crystalline region of the cellulose material can be extracted by depolymerization treatment, and the crystallinity of crystalline cellulose (d ′′) is preferably 10% or more, more preferably 30% or more, and 50% or more. More preferably.
- the crystallinity was calculated by the Segal method from the diffraction intensity value of the X-ray diffraction pattern obtained with the X-ray diffraction apparatus defined in JIS K 0131-1996 (“General Rules for X-ray Diffraction Analysis”).
- the average degree of polymerization of crystalline cellulose (d ′′) is preferably 30 to 400.
- the crystalline cellulose (d ′′) used in the present invention is hardly soluble in water. Specifically, the undissolved content when 0.5 g of crystalline cellulose (d ′′) is dissolved in 100 g of pure water at 25 ° C. is 90% by weight or more, preferably 95% by weight or more, more preferably 99%. % By weight or more.
- the composite (D) of the water-soluble polymer (d ′) and the crystalline cellulose (d ′′) is prepared by, for example, mixing and polishing the water-soluble polymer (d ′) and the crystalline cellulose (d ′′) in a wet manner. It is obtained by crushing and dry crushing. Thereby, crystalline cellulose (d ′′) surface-treated with the water-soluble polymer (d ′) can be obtained as the composite (D). That is, the water-soluble polymer (d ′) can be used as a surface treatment agent for crystalline cellulose (d ′′).
- crystalline cellulose (d ′′) usually precipitates in water, but when the complex (D) surface-treated with the water-soluble polymer (d ′) is stirred in water, the water-soluble polymer (d ') Swells and dissolves, and the crystalline cellulose (d' ') is dispersed in water in a colloidal form.
- the composite (D) in the present invention is obtained by dispersing the composite (D) by adding the composite (D) to pure water and sufficiently stirring with a stirrer such as a homogenizer (for example, at 15000 rpm for 5 minutes).
- the (primary average particle diameter) is 20 ⁇ m or less, preferably 10 ⁇ m or less, and when the dispersion is left for 10 minutes or more, preferably 30 minutes or more, no obvious precipitation occurs.
- the average particle size is a volume average particle size (d50) calculated by measuring with a laser diffraction particle size distribution analyzer (for example, SALD-3100; manufactured by Shimadzu Corporation).
- the amount of the water-soluble polymer (d ′) in the composite (D) is not particularly limited as long as it does not impair the effects of the present invention, but with respect to 100 parts by weight of the composite (D),
- the amount in terms of solid content is preferably 0.5 to 50 parts by weight, more preferably 1 to 45 parts by weight, still more preferably 1.5 to 40 parts by weight.
- the blending amount of the crystalline cellulose (d ′′) in the composite (D) is preferably 50 to 99.5 parts by weight in terms of solid content with respect to 100 parts by weight of the composite (D), more preferably. Is 55 to 99 parts by weight, more preferably 60 to 98.5 parts by weight.
- the blending amount of the water-soluble polymer (d ′) in the composite (D) is too small, the dispersion stability of the electrode active material in the composite particle slurry used for producing composite particles tends to deteriorate. .
- strength of the composite particle obtained will become weak.
- Theolas RC-591 (manufactured by Asahi Kasei Chemicals), Theolas RC-N-30 (manufactured by Asahi Kasei Chemicals), Neocell NEO-C91 (manufactured by Meidai Kako), Avicel CL611 (FMC)
- Commercial products such as Avicel BV1518 (manufactured by FMC) and the like, and among them, Theolas RC-591 (manufactured by Asahi Kasei Chemicals) and Theolas RC-N-30 (manufactured by Asahi Kasei Chemicals) are preferably used.
- the compounding amount of the composite (D) in the composite particles is not particularly limited as long as the effect of the present invention is not impaired, but in terms of solid content with respect to 100 parts by weight of the electrode active material (A).
- the amount is preferably 0.1 to 2.0 parts by weight, more preferably 0.2 to 1.5 parts by weight, still more preferably 0.3 to 1.0 parts by weight. If the blending amount of the composite (D) in the composite particles is too small, the reinforcing effect is reduced, and the cycle characteristics of the obtained electrochemical device are deteriorated. Moreover, when there are too many compounding quantities of the composite body (D) in a composite particle, granulation of a composite particle will become difficult.
- the weight ratio (C) / (D) of the water-soluble polymer (C) and the composite (D) in the composite particles is calculated in terms of solid content from the viewpoint of improving the dispersibility of the composite (D).
- the amount is preferably 0.2 to 15, more preferably 0.25 to 10, still more preferably 0.3 to 9.0, and particularly preferably 0.35 to 8.0.
- weight ratio (C) / (D) is too large, the reinforcing effect is reduced, and the cycle characteristics and high-temperature storage characteristics of the obtained electrochemical device are deteriorated.
- weight ratio (C) / (D) is too small, the storage stability of the slurry for composite particles will fall.
- the composite particle for an electrochemical element electrode of the present invention may contain a conductive agent (E) as necessary in addition to the above components.
- a conductive agent (E) is not particularly limited as long as it is a conductive material, but is preferably a particulate material having conductivity, for example, carbon black such as furnace black, acetylene black, and ketjen black; natural And graphite such as graphite and artificial graphite; and carbon fibers such as polyacrylonitrile-based carbon fiber, pitch-based carbon fiber, and vapor grown carbon fiber.
- the average particle diameter when the conductive agent (E) is a particulate material is not particularly limited, but is preferably smaller than the average particle diameter of the electrode active material, and sufficient conductivity can be expressed with a smaller amount of use. From the viewpoint, it is preferably 0.001 to 10 ⁇ m, more preferably 0.05 to 5 ⁇ m, and still more preferably 0.1 to 1 ⁇ m.
- the compounding amount of the conductive agent (E) in the composite particle for an electrochemical element electrode of the present invention is selected from the viewpoint of sufficiently reducing the internal resistance while keeping the capacity of the obtained electrochemical element high. ) 100 to 100 parts by weight, preferably 0.1 to 50 parts by weight, more preferably 0.5 to 15 parts by weight, still more preferably 1 to 10 parts by weight.
- the composite particles include other components such as an electrode active material (A), a particulate binder resin (B), a water-soluble polymer (C), a composite (D), and a conductive agent (E) added as necessary. It is obtained by using and granulating.
- the composite particle comprises an electrode active material (A), a particulate binder resin (B), a water-soluble polymer (C), and a composite (D).
- Each of the resin (B), the water-soluble polymer (C), and the composite (D) does not exist as independent particles, but is an electrode active material (A) that is a constituent component, a particulate binder resin ( Among the B), the water-soluble polymer (C), and the composite (D), one particle is formed by at least two components. Specifically, a plurality of particles (preferably several to several tens of particles) are formed by combining a plurality of the above-mentioned two or more component individual particles while maintaining a substantial shape.
- the electrode active material (A) is preferably bound by the particulate binder resin (B) to form particles.
- the minor axis diameter L s and the major axis diameter L l are values measured from a scanning electron micrograph image.
- the average particle diameter of the composite particles is preferably from 0.1 to 200 ⁇ m, more preferably from 1 to 150 ⁇ m, and even more preferably from 10 to 80 ⁇ m, from the viewpoint that an electrode layer having a desired thickness can be easily obtained.
- the average particle size is a volume average particle size calculated by measuring with a laser diffraction particle size distribution analyzer (for example, SALD-3100; manufactured by Shimadzu Corporation).
- the production method of the composite particles is not particularly limited, but is spray drying granulation method, rolling bed granulation method, compression granulation method, stirring granulation method, extrusion granulation method, crushing granulation method, fluidized bed granulation method.
- Composite particles can be obtained by production methods such as a granulation method, a fluidized bed multifunctional granulation method, and a melt granulation method.
- the production method of the composite particles may be appropriately selected from the viewpoints of ease of particle size control, productivity, ease of control of particle size distribution, etc. according to the components of the composite particles, etc.
- the spray-drying granulation method described in 1 is preferable because the composite particles can be produced relatively easily.
- the spray drying granulation method will be described.
- a slurry for composite particles containing an electrode active material (A), a particulate binder resin (B), a water-soluble polymer (C) and a composite (D) (hereinafter sometimes referred to as “slurry”).
- the slurry for composite particles includes an electrode active material (A), a particulate binder resin (B), a water-soluble polymer (C) and a composite (D), and a conductive agent (E) added as necessary. It can be prepared by dispersing or dissolving in a solvent. In this case, when the particulate binder resin (B) is dispersed in water as a solvent, it can be added in a state dispersed in water.
- water is preferably used, but a mixed solvent of water and an organic solvent may be used, or only an organic solvent may be used alone or in combination of several kinds.
- organic solvent examples include alcohols such as methyl alcohol, ethyl alcohol, and propyl alcohol; alkyl ketones such as acetone and methyl ethyl ketone; ethers such as tetrahydrofuran, dioxane, and diglyme; diethylformamide, dimethyl Amides such as acetamide, N-methyl-2-pyrrolidone, dimethylimidazolidinone; and the like.
- alcohols are preferred.
- water and an organic solvent having a lower boiling point than water the drying rate can be increased during spray drying. Thereby, the viscosity and fluidity of the slurry for composite particles can be adjusted, and the production efficiency can be improved.
- the viscosity of the composite particle slurry is preferably 10 to 3,000 mPa ⁇ s, more preferably 30 to 1,500 mPa ⁇ s at room temperature from the viewpoint of improving the productivity of granulation of the composite particles by spray drying. More preferably, it is 50 to 1,000 mPa ⁇ s.
- a dispersant or a surfactant when preparing the composite particle slurry, a dispersant or a surfactant may be added as necessary.
- the surfactant include amphoteric surfactants such as anionic, cationic, nonionic, and nonionic anions, and anionic or nonionic surfactants are preferable.
- the compounding amount of the surfactant is preferably 50 parts by weight or less, more preferably 0.1 to 10 parts by weight, still more preferably 0.5 to 5 parts by weight with respect to 100 parts by weight of the electrode active material (A). Part.
- the amount of the solvent used in preparing the slurry is such that the solid content concentration of the slurry is preferably 1 to 70% by weight, more preferably 5 to 70% by weight, from the viewpoint of uniformly dispersing the binder resin in the slurry. More preferably, the amount is 10 to 65% by weight.
- the order is not particularly limited.
- an electrode active material (A), a particulate binder resin (B), a water-soluble polymer (C), a composite (D), and a conductive agent (E) are added to a solvent.
- the electrode active material (A), the conductive agent (E) and the composite (D) are added and mixed, and finally dispersed in the solvent.
- the particulate binder resin (B) (for example, latex) is added and mixed, the particulate binder resin (B) and the composite (D) dispersed in a solvent are mixed with the electrode active material (A) and the conductive material.
- the mixing device for example, a ball mill, a sand mill, a bead mill, a pigment disperser, a pulverizer, an ultrasonic disperser, a homogenizer, a homomixer, a planetary mixer, or the like can be used.
- the mixing is preferably performed at room temperature to 80 ° C. for 10 minutes to several hours.
- Spray drying is a method of spraying and drying a slurry in hot air.
- An atomizer is used as an apparatus used for spraying slurry.
- a rotating disk system slurry is introduced almost at the center of the disk that rotates at high speed, and the slurry is released outside the disk by the centrifugal force of the disk. In this case, the slurry is atomized.
- the rotational speed of the disk depends on the size of the disk, but is preferably 5,000 to 30,000 rpm, more preferably 15,000 to 30,000 rpm.
- a pin-type atomizer is a type of centrifugal spraying device that uses a spraying plate, and the spraying plate has a plurality of spraying rollers removably mounted on a concentric circle along its periphery between upper and lower mounting disks. It consists of The slurry for composite particles is introduced from the center of the spray disk, adheres to the spray roller by centrifugal force, moves outward on the roller surface, and finally sprays away from the roller surface.
- the nozzle method is a method in which the slurry for composite particles is pressurized to form a mist from the nozzle and dried, or the slurry ejected from the nozzle is formed into a mist by air pressure and dried.
- the temperature of the slurry for composite particles to be sprayed is preferably room temperature, but may be higher than room temperature by heating.
- the hot air temperature during spray drying is preferably 25 to 250 ° C, more preferably 50 to 200 ° C, and still more preferably 80 to 150 ° C.
- the method of blowing hot air is not particularly limited.
- the method in which the hot air and the spraying direction flow side by side the method in which the hot air is sprayed at the top of the drying tower and descends with the hot air, and the sprayed droplets and hot air flow countercurrently. Examples include a contact method, and a method in which sprayed droplets first flow in parallel with hot air, then drop by gravity and contact countercurrent.
- the electrochemical element electrode of the present invention is an electrode formed by laminating an electrode active material layer containing the above composite particles on a current collector.
- a material for the current collector for example, metal, carbon, conductive polymer, and the like can be used, and metal is preferably used.
- metal copper, aluminum, platinum, nickel, tantalum, titanium, stainless steel, other alloys and the like are usually used. Among these, it is preferable to use copper, aluminum, or an aluminum alloy in terms of conductivity and voltage resistance. In addition, when high voltage resistance is required, high-purity aluminum disclosed in JP 2001-176757 A can be suitably used.
- the current collector is in the form of a film or a sheet, and the thickness thereof is appropriately selected depending on the purpose of use, but is preferably 1 to 200 ⁇ m, more preferably 5 to 100 ⁇ m, and still more preferably 10 to 50 ⁇ m.
- the composite particles When laminating the electrode active material layer on the current collector, the composite particles may be formed into a sheet shape and then laminated on the current collector. However, the composite particles may be directly pressed on the current collector. Is preferred.
- a method for pressure molding for example, a roll type pressure molding apparatus provided with a pair of rolls is used, and a roll type pressure molding apparatus is used to feed composite particles with a feeder such as a screw feeder while feeding a current collector with the roll.
- Roll pressure forming method for forming an electrode active material layer on the current collector, or dispersing composite particles on the current collector, adjusting the thickness by smoothing the composite particles with a blade
- a method of forming with a pressurizing apparatus, a method of filling composite particles into a mold, and pressurizing the mold to form are included.
- the roll pressure molding method is preferable.
- the composite particles of the present invention have high fluidity, they can be molded by roll press molding due to the high fluidity, thereby improving productivity.
- the roll temperature at the time of roll press molding is preferably 25 to 200 ° C., more preferably 50 to 150 ° C., from the viewpoint of ensuring sufficient adhesion between the electrode active material layer and the current collector. More preferably, it is 80 to 120 ° C.
- the press linear pressure between the rolls during roll press molding is preferably 10 to 1000 kN / m, more preferably 200 to 900 kN / m, and still more preferably 300 from the viewpoint of preventing the electrode active material (A) from being broken. ⁇ 600 kN / m.
- the molding speed at the time of roll press molding is preferably 0.1 to 20 m / min, more preferably 4 to 10 m / min.
- post-pressurization may be further performed as necessary in order to eliminate variations in the thickness of the formed electrochemical element electrode and increase the density of the electrode active material layer to increase the capacity.
- the post-pressing method is preferably a pressing process using a roll.
- the roll pressing step two cylindrical rolls are arranged vertically in parallel with a narrow interval, each is rotated in the opposite direction, and pressure is applied by interposing an electrode therebetween.
- the temperature of the roll may be adjusted as necessary, such as heating or cooling.
- the density of the electrode active material layer is not particularly limited, but is usually 0.30 to 10 g / cm 3 , preferably 0.35 to 8.0 g / cm 3 , more preferably 0.40 to 6.0 g / cm 3. It is.
- the thickness of the electrode active material layer is not particularly limited, but is usually 5 to 1000 ⁇ m, preferably 20 to 500 ⁇ m, more preferably 30 to 300 ⁇ m.
- An electrochemical element can be obtained by using the electrochemical element electrode obtained as described above for at least one of a positive electrode and a negative electrode, and further including a separator and an electrolytic solution.
- Examples of the electrochemical element include a lithium ion secondary battery and a lithium ion capacitor.
- separator for example, a polyolefin resin such as polyethylene or polypropylene, or a microporous film or nonwoven fabric containing an aromatic polyamide resin; a porous resin coat containing an inorganic ceramic powder; Specific examples include microporous membranes made of polyolefin resins (polyethylene, polypropylene, polybutene, polyvinyl chloride), and resins such as mixtures or copolymers thereof; polyethylene terephthalate, polycycloolefin, polyether sulfone, polyamide, Examples thereof include a microporous film made of a resin such as polyimide, polyimide amide, polyaramid, nylon, and polytetrafluoroethylene; a polyolefin fiber woven or non-woven fabric thereof; and an aggregate of insulating substance particles.
- a polyolefin resin such as polyethylene or polypropylene, or a microporous film or nonwoven fabric containing an aromatic polyamide resin
- the thickness of the separator is preferably 0.5 to 40 ⁇ m from the viewpoint of reducing the internal resistance due to the separator in the lithium ion secondary battery and from the viewpoint of excellent workability when manufacturing the lithium ion secondary battery. More preferably, the thickness is 1 to 30 ⁇ m, still more preferably 1 to 25 ⁇ m.
- Electrode As an electrolytic solution for a lithium ion secondary battery, for example, a nonaqueous electrolytic solution in which a supporting electrolyte is dissolved in a nonaqueous solvent is used.
- a lithium salt is preferably used.
- the lithium salt include LiPF 6 , LiAsF 6 , LiBF 4 , LiSbF 6 , LiAlCl 4 , LiClO 4 , CF 3 SO 3 Li, C 4 F 9 SO 3 Li, CF 3 COOLi, (CF 3 CO) 2 NLi , (CF 3 SO 2 ) 2 NLi, (C 2 F 5 SO 2 ) NLi, and the like.
- LiPF 6 , LiClO 4 , and CF 3 SO 3 Li that are easily soluble in a solvent and exhibit a high degree of dissociation are preferable.
- One of these may be used alone, or two or more of these may be used in combination at any ratio. Since the lithium ion conductivity increases as the supporting electrolyte having a higher degree of dissociation is used, the lithium ion conductivity can be adjusted depending on the type of the supporting electrolyte.
- the concentration of the supporting electrolyte in the electrolytic solution is preferably used at a concentration of 0.5 to 2.5 mol / L depending on the type of the supporting electrolyte. If the concentration of the supporting electrolyte is too low or too high, the ionic conductivity may decrease.
- the non-aqueous solvent is not particularly limited as long as it can dissolve the supporting electrolyte.
- non-aqueous solvents include carbonates such as dimethyl carbonate (DMC), ethylene carbonate (EC), diethyl carbonate (DEC), propylene carbonate (PC), butylene carbonate (BC), methyl ethyl carbonate (MEC);
- DMC dimethyl carbonate
- EC ethylene carbonate
- DEC diethyl carbonate
- PC propylene carbonate
- BC butylene carbonate
- MEC methyl ethyl carbonate
- esters such as ⁇ -butyrolactone and methyl formate
- ethers such as 1,2-dimethoxyethane and tetrahydrofuran
- sulfur-containing compounds such as sulfolane and dimethyl sulfoxide
- ionic liquids used also as supporting electrolytes used also as supporting electrolytes.
- a non-aqueous solvent may be used individually by 1 type, and may be used combining two or more types by arbitrary ratios. In general, the lower the viscosity of the non-aqueous solvent, the higher the lithium ion conductivity, and the higher the dielectric constant, the higher the solubility of the supporting electrolyte, but since both are in a trade-off relationship, the lithium ion conductivity depends on the type of solvent and the mixing ratio. It is recommended to adjust the conductivity.
- the nonaqueous solvent may be used in combination or in whole or in a form in which all or part of hydrogen is replaced with fluorine.
- the additive include carbonates such as vinylene carbonate (VC); sulfur-containing compounds such as ethylene sulfite (ES); and fluorine-containing compounds such as fluoroethylene carbonate (FEC).
- An additive may be used individually by 1 type and may be used combining two or more types by arbitrary ratios.
- an electrolyte solution for lithium ion capacitors the same electrolyte solution that can be used for the above-described lithium ion secondary battery can be used.
- Method for producing electrochemical element As a specific method for producing an electrochemical element such as a lithium ion secondary battery or a lithium ion capacitor, for example, a positive electrode and a negative electrode are overlapped via a separator, and this is wound or folded according to the shape of the battery. Examples of the method include putting the battery in a battery container, injecting an electrolyte into the battery container, and sealing the battery. Further, if necessary, an expanded metal; an overcurrent prevention element such as a fuse or a PTC element; a lead plate or the like may be inserted to prevent an increase in pressure inside the battery or overcharge / discharge.
- an electrochemical element such as a lithium ion secondary battery or a lithium ion capacitor
- a positive electrode and a negative electrode are overlapped via a separator, and this is wound or folded according to the shape of the battery. Examples of the method include putting the battery in a battery container, injecting an electrolyte into the battery container, and sealing the battery. Further, if necessary, an
- the shape of the lithium ion secondary battery may be any of a coin type, a button type, a sheet type, a cylindrical type, a square type, a flat type, and the like.
- the material of the battery container is not particularly limited as long as it inhibits the penetration of moisture into the battery, and is not particularly limited, such as a metal or a laminate such as aluminum.
- an electrochemical element having excellent high-temperature storage characteristics can be produced.
- the electrode active material layer is formed using the composite particle for an electrochemical element electrode of the present invention, the electrode active material layer can be formed with a weak pressure, and the destruction of the electrode active material can be prevented. .
- ⁇ Particle strength of composite particles> The composite particles obtained in Examples and Comparative Examples were subjected to a compression test using a micro compression tester (“MCT-W500” manufactured by Shimadzu Corporation).
- MCT-W500 micro compression tester
- a compressive strength (MPa) is measured when a particle is deformed until the diameter of the composite particle is displaced by 40% by applying a load at a loading speed of 4.46 mN / sec in the center direction of the composite particle at room temperature. did.
- a composite particle having a diameter of 40 to 60 ⁇ m was selected and a compression test was performed.
- Compressive strength is 1.00 MPa or more
- B Compressive strength is 0.90 MPa or more and less than 1.00 MPa
- C Compressive strength is 0.80 MPa or more and less than 0.90 MPa
- D Compressive strength is 0.70 MPa or more, 0.80 MPa Less than E: Compressive strength is less than 0.70 MPa
- Capacity maintenance ratio is 90% or more
- B: Capacity maintenance ratio is 80% or more and less than 90%
- C: Capacity maintenance ratio is 75% or more and less than 80%
- D: Capacity maintenance ratio is 70% or more and less than 75%
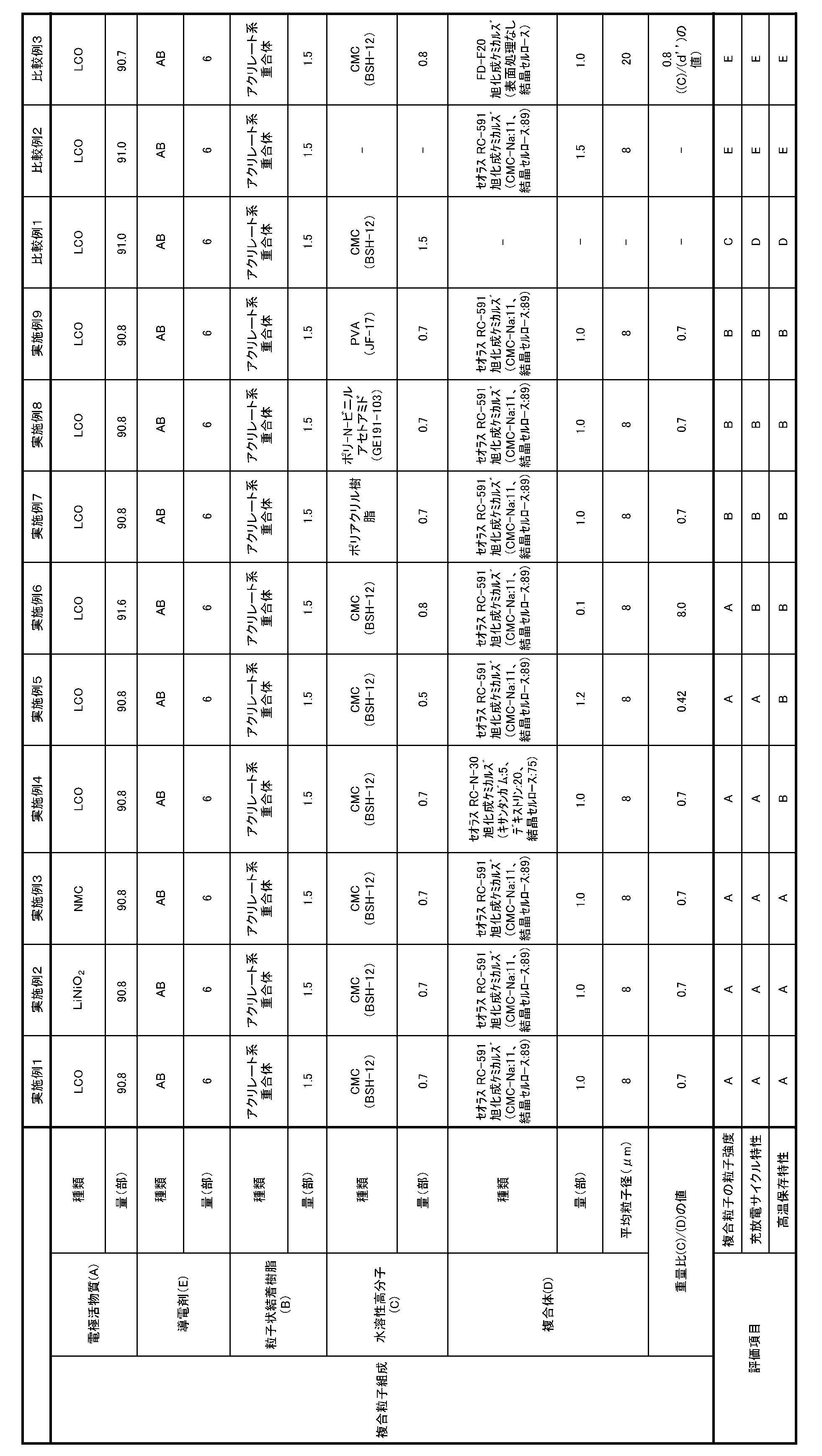
- Example 1 Manufacture of particulate binder resin (B)
- a 1 L SUS separable flask equipped with a stirrer, a reflux condenser and a thermometer add 130 parts of ion exchange water, and further add 0.8 parts of ammonium persulfate and 10 parts of ion exchange water as a polymerization initiator. , Heated to 80 ° C.
- the emulsion obtained above was continuously added to the separable flask over 3 hours. After further reaction for 2 hours, the reaction was stopped by cooling. 10% aqueous ammonia was added thereto to adjust the pH to 7.5, and an aqueous dispersion of an acrylate polymer as a particulate binder resin (B) was obtained. The polymerization conversion rate was 98%.
- LiCoO 2 (hereinafter sometimes referred to as “LCO”) as the positive electrode active material as the electrode active material (A) and acetylene black (hereinafter referred to as “AB”) as the conductive agent (E).
- LCO LiCoO 2
- AB acetylene black
- CMC carboxymethyl cellulose
- the slurry for composite particles in a spray dryer (manufactured by Okawara Kako Co., Ltd.) was used with a rotary disk type atomizer (diameter 65 mm), rotation speed 25,000 rpm, hot air temperature 150 ° C., and particle recovery outlet temperature 90 ° C. Then, spray drying granulation was performed to obtain composite particles.
- the composite particles had an average volume particle diameter of 40 ⁇ m.
- the composite particles obtained above are used for a press roll (roll temperature) of a roll press machine (“Hurano Giken Kogyo Co., Ltd.“ Rough Surface Heated Roll ”) using a quantitative feeder (“ Nikka Spray KV ”manufactured by Nikka). 100 ° C., press linear pressure 500 kN / m). An aluminum foil having a thickness of 20 ⁇ m is inserted between the rolls for pressing, and the composite particles for the secondary battery electrode supplied from the quantitative feeder are adhered onto the aluminum foil, followed by pressure molding at a molding speed of 1.5 m / min. Thus, a positive electrode having a positive electrode active material was obtained.
- the negative electrode slurry composition obtained above was applied onto a copper foil having a thickness of 20 ⁇ m using a comma coater so that the film thickness after drying was about 150 ⁇ m and dried. This drying was performed by conveying the copper foil in an oven at 60 ° C. at a speed of 0.5 m / min for 2 minutes. Thereafter, heat treatment was performed at 120 ° C. for 2 minutes to obtain a negative electrode raw material. This negative electrode raw material was rolled with a roll press to obtain a negative electrode having a negative electrode active material layer.
- a single-layer polypropylene separator (width 65 mm, length 500 mm, thickness 25 ⁇ m, manufactured by dry method, porosity 55%) was cut into a square of 5 ⁇ 5 cm 2 .
- the positive electrode for a lithium ion secondary battery obtained above was cut into a 4 ⁇ 4 cm 2 square and placed so that the current collector-side surface was in contact with the aluminum packaging exterior.
- the square separator obtained above was disposed on the surface of the positive electrode active material layer of the positive electrode for a lithium ion secondary battery.
- the negative electrode for a lithium ion secondary battery obtained above was cut into a square of 4.2 ⁇ 4.2 cm 2 and arranged on the separator so that the surface on the negative electrode active material layer side faced the separator. Further, containing the vinylene carbonate 2.0%, was charged with LiPF 6 solution having a concentration of 1.0 M.
- Example 2 Production of composite particle slurry, production of composite particles, production of positive electrode, lithium, except that electrode active material (A) used for production of composite particle slurry was 90.8 parts of LiNiO 2 An ion secondary battery was manufactured.
- Example 3 The slurry for composite particles in the same manner as in Example 1 except that the electrode active material (A) used for the production of the composite particle slurry was 90.8 parts of lithium manganese manganese cobaltate (NMC (111)) having a layered structure. , Composite particles, positive electrode, and lithium ion secondary battery.
- the electrode active material (A) used for the production of the composite particle slurry was 90.8 parts of lithium manganese manganese cobaltate (NMC (111)) having a layered structure.
- NMC lithium manganese manganese cobaltate
- Example 5 Example 1 except that the amount of the water-soluble polymer (C) used in the production of the composite particle slurry is 0.5 parts in terms of solid content and the amount of the composite (D) is 1.2 parts.
- a slurry for composite particles, a composite particle, a positive electrode, and a lithium ion secondary battery were manufactured.
- the weight ratio (C) / (D) of the water-soluble polymer (C) and the composite (D) was 0.42.
- Example 6 The amount of the electrode active material (A) used for the production of the composite particle slurry is 91.6 parts, the amount of the water-soluble polymer (C) is 0.8 parts in terms of solid content, and the amount of the composite (D) is Except that the amount was 0.1 parts, the production of the composite particle slurry, the production of the composite particles, the production of the positive electrode, and the production of the lithium ion secondary battery were performed in the same manner as in Example 1. The weight ratio (C) / (D) between the water-soluble polymer (C) and the composite (D) was 8.0.
- Example 7 Except that the water-soluble polymer (C) used for the production of the composite particle slurry is a polyacrylic resin, the production of the composite particle slurry, the production of the composite particles, the production of the positive electrode, the lithium ion secondary, as in Example 1.
- the battery was manufactured.
- the polyacrylic resin was produced as follows.
- the obtained emulsion aqueous solution was continuously dropped into the separable flask over 4 hours.
- the reaction temperature was set to 80 ° C., and the reaction was further carried out for 2 hours.
- the reaction was stopped by cooling to obtain an aqueous dispersion containing a polyacrylic resin.
- the polymerization conversion rate was 99%. Moreover, it was 25000 when the weight average molecular weight of the obtained polyacrylic resin was measured by GPC. Moreover, the viscosity when the obtained polyacrylic resin was made into 1 weight% aqueous solution was 3000 (mPa * s).
- Example 8 A slurry for composite particles in the same manner as in Example 1 except that the water-soluble polymer (C) used for the production of the composite particle slurry was a poly-N-vinylacetamide (PNVA, GE191-103; Showa Denko) resin. , Composite particles, positive electrode, and lithium ion secondary battery.
- PNVA poly-N-vinylacetamide
- Example 9 Manufacture of slurry for composite particles, manufacture of composite particles, manufacture of positive electrode in the same manner as in Example 1 except that the water-soluble polymer (C) used for the slurry for composite particles was polyvinyl alcohol resin (PVA, JF-17). A lithium ion secondary battery was manufactured.
- the water-soluble polymer (C) used for the slurry for composite particles was polyvinyl alcohol resin (PVA, JF-17).
- PVA, JF-17 polyvinyl alcohol resin
- the amount of the electrode active material (A) used in the production of the composite particle slurry is 90.7 parts, the amount of the water-soluble polymer (C) is 0.8 parts in terms of solid content, and the composite (D) is substituted.
- the weight ratio (C) / (d ′′) between the water-soluble polymer (C) and the crystalline cellulose (d ′′) not subjected to surface treatment was 0.8.
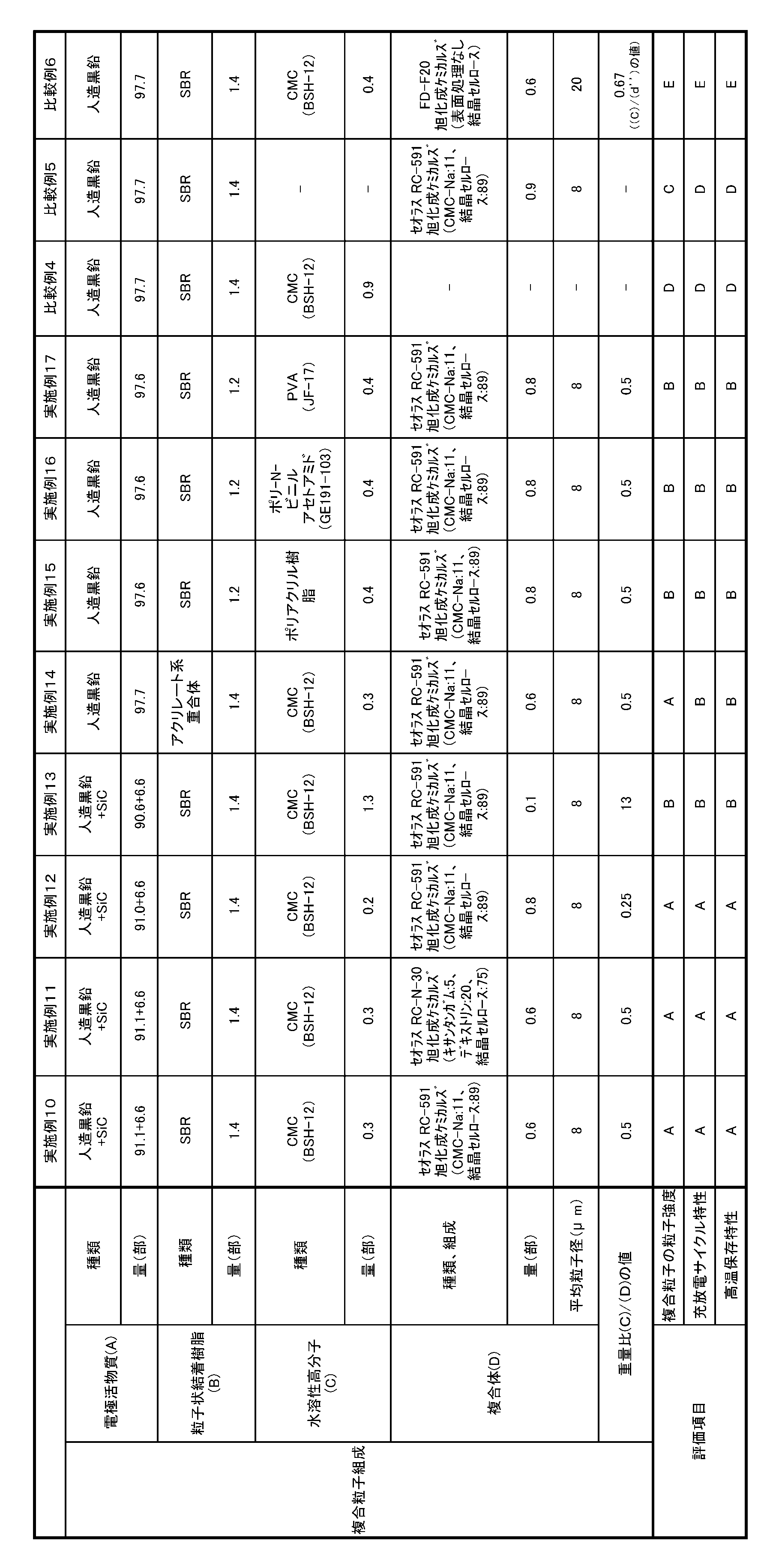
- Example 10 (Production of particulate binder resin (B)) In a 5 MPa pressure vessel equipped with a stirrer, 47 parts of styrene, 50 parts of 1,3-butadiene, 3 parts of methacrylic acid, 4 parts of sodium dodecylbenzenesulfonate, 150 parts of ion-exchanged water, 0.4 part of t-dodecyl mercaptan as a chain transfer agent Then, 0.5 part of potassium persulfate was added as a polymerization initiator, and after sufficiently stirring, the mixture was heated to 50 ° C. to initiate polymerization. When the polymerization conversion rate reached 96%, the reaction was stopped by cooling to obtain a styrene-butadiene copolymer (hereinafter sometimes referred to as “SBR”) as the particulate binder resin (B). .
- SBR styrene-butadiene copolymer
- the slurry for composite particles in a spray dryer (manufactured by Okawara Kako Co., Ltd.) was used with a rotary disk type atomizer (diameter 65 mm), rotation speed 25,000 rpm, hot air temperature 150 ° C., and particle recovery outlet temperature 90 ° C. Then, spray drying granulation was performed to obtain composite particles.
- the composite particles had an average volume particle diameter of 40 ⁇ m.
- LiCoO 2 having a layered structure as a positive electrode active material (hereinafter sometimes abbreviated as “LCO”) and polyvinylidene fluoride (PVDF; “KF-1100” manufactured by Kureha Chemical Co., Ltd.) as a positive electrode binder resin
- LCO positive electrode active material
- PVDF polyvinylidene fluoride
- H-100 acetylene black
- N-methylpyrrolidone N-methylpyrrolidone
- a single-layer polypropylene separator (width 65 mm, length 500 mm, thickness 25 ⁇ m, manufactured by dry method, porosity 55%) was cut into a square of 5 ⁇ 5 cm 2 .
- the positive electrode for a lithium ion secondary battery obtained above was cut into a 4 ⁇ 4 cm 2 square and placed so that the current collector-side surface was in contact with the aluminum packaging exterior.
- the square separator obtained above was disposed on the surface of the positive electrode active material layer of the positive electrode for a lithium ion secondary battery.
- the negative electrode for a lithium ion secondary battery obtained above was cut into a square of 4.2 ⁇ 4.2 cm 2 and arranged on the separator so that the surface on the negative electrode active material layer side faced the separator. Further, containing the vinylene carbonate 2.0%, was charged with LiPF 6 solution having a concentration of 1.0 M.
- Example 12 The amount of the electrode active material (A) used for the production of the composite particle slurry is 91.0 parts of artificial graphite and 6.6 parts of SiC, and the amount of the water-soluble polymer (C) is 0.2 parts in terms of solid content. Except that the amount of the composite (D) was 0.8 part, the production of the slurry for the composite particles, the production of the composite particles, the production of the negative electrode for the lithium ion secondary battery, the lithium ion secondary as in Example 10. The battery was manufactured. The weight ratio (C) / (D) of the water-soluble polymer (C) and the composite (D) was 0.25.
- the amount of the electrode active material (A) used in the production of the composite particle slurry is 90.6 parts of artificial graphite and 6.6 parts of SiC, and a 1.0% aqueous solution of CMC (BSH-6) is used as the water-soluble polymer (C). ; Manufactured by Daiichi Kogyo Seiyaku Co., Ltd.) in the same manner as in Example 10 except that 1.3 parts in terms of solid content is used and the amount of the complex (D) is 0.1 parts in terms of solid content. Production of slurry for particles, production of composite particles, production of negative electrode for lithium ion secondary battery, and production of lithium ion secondary battery were performed. The weight ratio (C) / (D) between the water-soluble polymer (C) and the composite (D) was 13.0.
- Example 14 Composite as in Example 10, except that the electrode active material (A) used for the production of the composite particle slurry was 97.7 parts of artificial graphite and the type of the particulate binder resin (B) was an acrylate polymer. Production of slurry for particles, production of composite particles, production of negative electrode for lithium ion secondary battery, and production of lithium ion secondary battery were performed. The same acrylate polymer as in Example 1 was used.
- the electrode active material (A) used for producing the composite particle slurry is 97.6 parts of artificial graphite, the amount of the particulate binder resin (B) is 1.2 parts, and the water-soluble polymer (C) is polyacrylic.
- Manufacture of lithium ion secondary batteries was performed.
- the weight ratio (C) / (D) between the water-soluble polymer (C) and the composite (D) was 0.5.
- the polyacrylic resin used was the same as in Example 7.
- Example 16 Composite particles as in Example 15 except that poly-N-vinylacetamide resin (PNVA, GE191-103; Showa Denko) was used as the water-soluble polymer (C) used in the production of the composite particle slurry. Slurry production, composite particle production, lithium ion secondary battery anode production, and lithium ion secondary battery production were carried out.
- PNVA poly-N-vinylacetamide resin
- C water-soluble polymer
- Example 17 Production of composite particle slurry and production of composite particles in the same manner as in Example 15 except that polyvinyl alcohol resin (PVA, JF-17) was used as the water-soluble polymer (C) used for production of the composite particle slurry.
- PVA, JF-17 polyvinyl alcohol resin
- C water-soluble polymer
- the electrode active material (A) used for the production of the composite particle slurry is 97.7 parts of artificial graphite, the amount of the water-soluble polymer (C) is 0.4 parts in terms of solid content, and is replaced with the composite (D).
- the slurry for composite particles was the same as in Example 10 except that 0.6 parts of FD-F20 (average particle diameter 20 ⁇ m, manufactured by Asahi Kasei Chemicals) was used as the crystalline cellulose (d ′′) that had not been surface-treated. Production of composite particles, production of negative electrodes for lithium ion secondary batteries, and production of lithium ion secondary batteries.
- the weight ratio (C) / (d ′′) between the water-soluble polymer (C) and the crystalline cellulose (d ′′) not subjected to surface treatment was 0.67.
- the electrode active material (A), the particulate binder resin (B), the water-soluble polymer (C), the water-soluble polymer (d ′) and the crystalline cellulose (d ′′) The particle strength of the composite particle for an electrochemical device electrode containing the composite (D) with the lithium ion secondary battery obtained by using this composite particle was good in charge / discharge cycle characteristics and high-temperature storage characteristics. It was.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Power Engineering (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Composite Materials (AREA)
- Manufacturing & Machinery (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Electric Double-Layer Capacitors Or The Like (AREA)
Abstract
Description
(1) 電極活物質(A)、粒子状結着樹脂(B)、水溶性高分子(C)及び水溶性高分子(d')と結晶セルロース(d'')との複合体(D)を含む電気化学素子電極用複合粒子、
(2) 前記電極活物質(A)100重量部に対し、前記水溶性高分子(C)を0.1~10重量部、前記複合体(D)を0.1~2.0重量部それぞれ含み、前記水溶性高分子(C)と前記複合体(D)との重量比(C)/(D)が(C)/(D)=0.2~15である(1)記載の電気化学素子電極用複合粒子、
(3) 前記複合体(D)は、水中に分散する性質を有する(1)または(2)記載の電気化学素子電極用複合粒子、
(4) 前記粒子状結着樹脂(B)は、共役ジエン系重合体またはアクリレート系重合体の少なくとも一方を含む(1)~(3)の何れかに記載の電気化学素子電極用複合粒子、
(5) 前記複合体(D)の一次平均粒子径は、10μm以下である(1)~(4)の何れかに記載の電気化学素子電極用複合粒子、
(6) 前記複合体(D)は、水溶性高分子(d’)で表面処理された結晶セルロース(d’’)である(1)に記載の電気化学素子電極用複合粒子、
(7) さらに導電剤(E)を含む(1)~(6)の何れかに記載の電気化学素子電極用複合粒子、
(8) (1)~(7)のいずれかに記載の電気化学素子電極用複合粒子を製造する方法であって、前記水溶性高分子(d’)で前記結晶セルロース(d’’)を表面処理することにより前記複合体(D)を得る工程と、前記電極活物質(A)、前記粒子状結着樹脂(B)、前記水溶性高分子(C)及び前記複合体(D)を混合して複合粒子用スラリーを得る工程と、前記複合粒子用スラリーを造粒することにより、電気化学素子電極用複合粒子を得る工程とを有する電気化学素子電極用複合粒子の製造方法
が提供される。
電気化学素子がリチウムイオン二次電池である場合の正極活物質としては、リチウムイオンをドープ及び脱ドープ可能な活物質が用いられ、無機化合物からなるものと有機化合物からなるものとに大別される。
また、負極活物質としては、1種類を単独で用いてもよく、2種類以上を任意の比率で組み合わせて用いてもよい。
また、電気化学素子がリチウムイオンキャパシタである場合に好ましく用いられる負極活物質としては、上記炭素で形成された負極活物質が挙げられる。
本発明に用いる粒子状結着樹脂(B)としては、上述の電極活物質を相互に結着させることができる物質であれば特に限定はない。粒子状結着樹脂(B)としては、溶媒に分散する性質のある分散型の粒子状結着樹脂が好ましい。分散型の粒子状結着樹脂として、例えば、シリコン系重合体、フッ素含有重合体、共役ジエン系重合体、アクリレート系重合体、ポリイミド、ポリアミド、ポリウレタン等の高分子化合物が挙げられ、水溶性高分子(C)と複合体(D)との接着性が良好となる観点から、フッ素含有重合体、共役ジエン系重合体およびアクリレート系重合体が好ましく、共役ジエン系重合体およびアクリレート系重合体がより好ましい。これらの重合体は、それぞれ単独で、または2種以上混合して、分散型の粒子状結着樹脂として用いることができる。
本発明に用いる水溶性高分子(C)とは、25℃において、高分子0.5gを100gの純水に溶解させた場合の未溶解分が10.0重量%未満の高分子をいう。
本発明に用いる複合体(D)は、水溶性高分子(d’)と結晶セルロース(d’’)との複合体である。
結晶化度(%)={(Ic-Ia)/Ic}×100
(ここで、Ic:X線回析図の回折角2θ=22.5度での回折強度、Ia:同じく回析角2θ=18.5度付近のベースライン強度(極小値強度)である。)
また、結晶セルロース(d’’)の平均重合度は、30~400であることが好ましい。
本発明の電気化学素子電極用複合粒子は、上記各成分に加えて、必要に応じて導電剤(E)を含有していてもよい。また、電極活物質(A)が正極活物質である場合には、導電材(E)を含有することが好ましい。導電剤(E)としては、導電性を有する材料であれば特に限定されないが、導電性を有する粒子状の材料が好ましく、たとえば、ファーネスブラック、アセチレンブラック、及びケッチェンブラック等のカーボンブラック;天然黒鉛、人造黒鉛等の黒鉛;ポリアクリロニトリル系炭素繊維、ピッチ系炭素繊維、気相法炭素繊維等の炭素繊維;が挙げられる。導電剤(E)が粒子状の材料である場合の平均粒子径は、特に限定されないが、電極活物質の平均粒子径よりも小さいものが好ましく、より少ない使用量で十分な導電性を発現させる観点から、好ましくは0.001~10μm、より好ましくは0.05~5μm、さらに好ましくは0.1~1μmである。
複合粒子は、電極活物質(A)、粒子状結着樹脂(B)、水溶性高分子(C)、複合体(D)および必要に応じ添加される導電剤(E)等他の成分を用いて造粒することにより得られる。複合粒子は、電極活物質(A)、粒子状結着樹脂(B)、水溶性高分子(C)、複合体(D)を含んでなるが、電極活物質(A)、粒子状結着樹脂(B)、水溶性高分子(C)および複合体(D)のそれぞれが個別に独立した粒子として存在するのではなく、構成成分である電極活物質(A)、粒子状結着樹脂(B)、水溶性高分子(C)、複合体(D)のうち、少なくとも2成分以上によって一粒子を形成するものである。具体的には、前記2成分以上の個々の粒子が実質的に形状を維持した状態で複数個が結合して二次粒子を形成しており、たとえば、複数個(好ましくは数個~数十個)の電極活物質(A)が、粒子状結着樹脂(B)によって結着されて粒子を形成しているものが好ましい。
以下、噴霧乾燥造粒法について説明する。
本発明の電気化学素子電極は、上述の複合粒子を含む電極活物質層を集電体上に積層してなる電極である。集電体の材料としては、たとえば、金属、炭素、導電性高分子などを用いることができ、好適には金属が用いられる。金属としては、通常、銅、アルミニウム、白金、ニッケル、タンタル、チタン、ステンレス鋼、その他の合金等が使用される。これらの中で導電性、耐電圧性の面から、銅、アルミニウム又はアルミニウム合金を使用するのが好ましい。また、高い耐電圧性が要求される場合には特開2001-176757号公報等で開示される高純度のアルミニウムを好適に用いることができる。集電体は、フィルム又はシート状であり、その厚みは、使用目的に応じて適宜選択されるが、好ましくは1~200μm、より好ましくは5~100μm、さらに好ましくは10~50μmである。
上述のようにして得られる電気化学素子電極を正極又は負極のうちの少なくとも一方に用い、さらにセパレーターおよび電解液を備えることにより、電気化学素子を得ることができる。電気化学素子としては、例えば、リチウムイオン二次電池、リチウムイオンキャパシタ等が挙げられる。
セパレーターとしては、例えば、ポリエチレン、ポリプロピレンなどのポリオレフィン樹脂や、芳香族ポリアミド樹脂を含んでなる微孔膜または不織布;無機セラミック粉末を含む多孔質の樹脂コート;などを用いることができる。具体例を挙げると、ポリオレフィン系(ポリエチレン、ポリプロピレン、ポリブテン、ポリ塩化ビニル)、及びこれらの混合物あるいは共重合体等の樹脂からなる微多孔膜;ポリエチレンテレフタレート、ポリシクロオレフィン、ポリエーテルスルフォン、ポリアミド、ポリイミド、ポリイミドアミド、ポリアラミド、ナイロン、ポリテトラフルオロエチレン等の樹脂からなる微多孔膜;ポリオレフィン系の繊維を織ったもの又はその不織布;絶縁性物質粒子の集合体等が挙げられる。これらの中でも、セパレーター全体の膜厚を薄くすることができ、リチウムイオン二次電池内の活物質比率を上げて体積あたりの容量を上げることができるため、ポリオレフィン系の樹脂からなる微多孔膜が好ましい。
リチウムイオン二次電池用の電解液としては、例えば、非水溶媒に支持電解質を溶解した非水電解液が用いられる。支持電解質としては、リチウム塩が好ましく用いられる。リチウム塩としては、例えば、LiPF6、LiAsF6、LiBF4、LiSbF6、LiAlCl4、LiClO4、CF3SO3Li、C4F9SO3Li、CF3COOLi、(CF3CO)2NLi、(CF3SO2)2NLi、(C2F5SO2)NLiなどが挙げられる。中でも、溶媒に溶けやすく高い解離度を示すLiPF6、LiClO4、CF3SO3Liが好ましい。これらは1種類を単独で用いてもよく、2種類以上を任意の比率で組み合わせて用いてもよい。解離度の高い支持電解質を用いるほど、リチウムイオン伝導度が高くなるので、支持電解質の種類によりリチウムイオン伝導度を調節することができる。
なお、リチウムイオンキャパシタ用の電解液としては、上述のリチウムイオン二次電池に用いることができる電解液と同様のものを用いることができる。
リチウムイオン二次電池やリチウムイオンキャパシタ等の電気化学素子の具体的な製造方法としては、例えば、正極と負極とをセパレーターを介して重ね合わせ、これを電池形状に応じて巻く、折るなどして電池容器に入れ、電池容器に電解液を注入して封口する方法が挙げられる。さらに、必要に応じてエキスパンドメタル;ヒューズ、PTC素子などの過電流防止素子;リード板などを入れ、電池内部の圧力上昇、過充放電を防止してもよい。リチウムイオン二次電池の形状は、コイン型、ボタン型、シート型、円筒型、角形、扁平型など、何れであってもよい。電池容器の材質は、電池内部への水分の侵入を阻害するものであればよく、金属製、アルミニウムなどのラミネート製など特に限定されない。
実施例及び比較例で得られた複合粒子について、微小圧縮試験機(島津製作所製「MCT-W500」)を用いた圧縮試験を行った。圧縮試験においては、室温で複合粒子の中心方向へ荷重負荷速度4.46mN/secで荷重を加え、複合粒子の直径が40%変位するまで粒子を変形させたときの圧縮強度(MPa)を測定した。なお、この測定では直径が40~60μmの複合粒子を選び圧縮試験を行った。
A:圧縮強度が1.00MPa以上
B:圧縮強度が0.90MPa以上、1.00MPa未満
C:圧縮強度が0.80MPa以上、0.90MPa未満
D:圧縮強度が0.70MPa以上、0.80MPa未満
E:圧縮強度が0.70MPa未満
実施例及び比較例で得られたラミネート型のリチウムイオン二次電池について、60℃で0.5Cの定電流定電圧充電法にて、4.2Vになるまで定電流で充電し、その後、定電圧で充電し、次いで、0.5Cの定電流で3.0Vまで放電する充放電サイクル試験を行った。充放電サイクル試験は100サイクルまで行い、初期放電容量に対する100サイクル目の放電容量の比を容量維持率とした。容量維持率を下記基準にて評価し、結果を表1及び表2に示した。容量維持率が大きいほど繰り返し充放電による容量の減少が少ないことを示す。
A:容量維持率が90%以上
B:容量維持率が80%以上、90%未満
C:容量維持率が75%以上、80%未満
D:容量維持率が70%以上、75%未満
E:容量維持率が70%未満または評価不能
実施例および比較例で製造したリチウムイオン二次電池電極を用いて、コイン型セルのリチウムイオン二次電池を作製し、24時間静置した後に4.2V、0.1Cの充放電レートにて充放電の操作を行い、初期容量C0を測定した。さらに、4.2Vに充電し、60℃、14日間保存した後、4.2V、0.1Cの充放電レートにて充放電の操作を行い、高温保存後の容量C1を測定した。高温保存特性は、ΔC=C1/C0×100(%)で示す容量変化率にて評価し結果を表1及び表2に示した。この値が高いほど高温保存特性に優れることを示す。
A:85%以上
B:70%以上85%未満
C:60%以上70%未満
D:50%以上60%未満
E:50%未満
(粒子状結着樹脂(B)の製造)
撹拌機、還流冷却管及び温度計を備えた容量1LのSUS製セパラブルフラスコに、イオン交換水を130部加え、更に重合開始剤として過硫酸アンモニウムを0.8部、イオン交換水を10部加え、80℃に加温した。
電極活物質(A)として正極活物質のLiCoO2(以下、「LCO」ということがある。)90.8部と、導電剤(E)としてアセチレンブラック(以下、「AB」ということがある。)を6部、粒子状結着樹脂(B)として上記アクリレート系重合体を固形分換算量で1.5部、水溶性高分子(C)としてカルボキシメチルセルロース(以下、「CMC」ということがある。)(BSH-12;第一工業製薬社製)を0.7部、及び複合体(D)としてセオラスRC-591(水溶性高分子(d’)の種類:カルボキシメチルセルロースのナトリウム塩(以下、「CMC-Na」ということがある。)、水溶性高分子(d’)と結晶セルロース(d’’)との重量比=11:89、平均粒子径:8μm、旭化成ケミカルズ社製)を固形分換算量で1.0部混合し、さらにイオン交換水を固形分濃度が60%となるように加え、プラネタリーミキサーで混合して複合粒子用スラリーを得た。水溶性高分子(C)と複合体(D)との重量比(C)/(D)は0.7であった。
上記複合粒子用スラリーをスプレー乾燥機(大川原化工機社製)において、回転円盤方式のアトマイザ(直径65mm)を用い、回転数25,000rpm、熱風温度150℃、粒子回収出口の温度を90℃として、噴霧乾燥造粒を行い、複合粒子を得た。この複合粒子の平均体積粒子径は40μmであった。
上記で得られた複合粒子を、定量フィーダ(ニッカ社製「ニッカスプレーK-V))を用いてロールプレス機(ヒラノ技研工業社製「押し切り粗面熱ロール」)のプレス用ロール(ロール温度100℃、プレス線圧500kN/m)に供給した。プレス用ロール間に、厚さ20μmのアルミニウム箔を挿入し、定量フィーダから供給された上記二次電池電極用複合粒子をアルミニウム箔上に付着させ、成形速度1.5m/分で加圧成形し、正極活物質を有する正極を得た。
攪拌機付き5MPa耐圧容器に、スチレン47部、1,3-ブタジエン50部、メタクリル酸3部、ドデシルベンゼンスルホン酸ナトリウム4部、イオン交換水150部、連鎖移動剤としてt-ドデシルメルカプタン0.4部および重合開始剤として過硫酸カリウム0.5部を入れ、十分に攪拌した後、50℃に加温して重合を開始した。重合転化率が96%になった時点で冷却し反応を停止して、粒子状の負極用結着樹脂(スチレン・ブタジエン共重合体;以下、「SBR」と略記することがある。)を得た。
ディスパー付きプラネタリーミキサーに、負極活物質として比表面積4m2/gの人造黒鉛(平均粒子径:24.5μm)を98.3部、分散剤としてカルボキシメチルセルロースの1%水溶液(第一工業製薬社製「BSH-12」)を固形分相当で0.7部、負極用結着樹脂を固形分相当量で1.0部加え、イオン交換水で全固形分濃度が50%となるように調整して混合した。これを減圧下で脱泡処理し、負極用のスラリー組成物を得た。
上記で得られた負極用のスラリー組成物を、コンマコーターを用いて厚さ20μmの銅箔の上に乾燥後の膜厚が150μm程度になるように塗布し乾燥させた。この乾燥は、銅箔を0.5m/分の速度で60℃のオーブン内を2分間かけて搬送することにより行った。その後、120℃にて2分間加熱処理し、負極原反を得た。この負極原反をロールプレスで圧延し、負極活物質層を有する負極を得た。
単層のポリプロピレン製セパレーター(幅65mm、長さ500mm、厚さ25μm、乾式法により製造、気孔率55%)を、5×5cm2の正方形に切り抜いた。
電池の外装として、アルミ包材外装を用意した。上記で得られたリチウムイオン二次電池用正極を、4×4cm2の正方形に切り出し、集電体側の表面がアルミ包材外装に接するように配置した。リチウムイオン二次電池用正極の正極活物質層の面上に、上記で得られた正方形のセパレーターを配置した。さらに、上記で得られたリチウムイオン二次電池用負極を、4.2×4.2cm2の正方形に切り出し、負極活物質層側の表面がセパレーターに向かい合うように、セパレーター上に配置した。更に、ビニレンカーボネートを2.0%含有する、濃度1.0MのLiPF6溶液を充填した。このLiPF6溶液の溶媒はエチレンカーボネート(EC)とエチルメチルカーボネート(EMC)との混合溶媒(EC/EMC=3/7(体積比))である。さらに、アルミニウム包材の開口を密封するために、150℃でヒートシールをしてアルミニウム外装を閉口し、ラミネート型のリチウムイオン二次電池(ラミネート型セル)を製造した。
複合粒子用スラリーの製造に用いる電極活物質(A)を、LiNiO2 90.8部とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造に用いる電極活物質(A)を、層状構造を有するニッケルマンガンコバルト酸リチウム(NMC(111))90.8部とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造に用いる複合体(D)としてRC-N-30(水溶性高分子(d’)の種類:キサンタンガム及びデキストリン、キサンタンガムとデキストリンと結晶セルロース(d’’)との重量比=5:20:75、平均粒子径:8μm、旭化成ケミカルズ社製)を使用した以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造に用いる水溶性高分子(C)の量を固形分換算量で0.5部、複合体(D)の量を1.2部とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。水溶性高分子(C)と複合体(D)との重量比(C)/(D)は0.42であった。
複合粒子用スラリーの製造に用いる電極活物質(A)の量を91.6部、水溶性高分子(C)の量を固形分換算量で0.8部、複合体(D)の量を0.1部とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。水溶性高分子(C)と複合体(D)との重量比(C)/(D)は8.0であった。
複合粒子用スラリーの製造に用いる水溶性高分子(C)をポリアクリル樹脂とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。なお、ポリアクリル樹脂の製造は下記のようにして行った。
攪拌機、還流冷却管および温度計を備えた容量1LのSUS製セパラブルフラスコに、脱塩水を予め仕込み十分攪拌した後、70℃とし、過硫酸カリウム水溶液0.2部を添加した。
複合粒子用スラリーの製造に用いる水溶性高分子(C)をポリ-N-ビニルアセトアミド(PNVA、GE191-103;昭和電工社製)樹脂とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーに用いる水溶性高分子(C)をポリビニルアルコール樹脂(PVA、JF-17)とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造において複合体(D)を添加せず、電極活物質(A)の量を91.0部、水溶性高分子(C)の量を1.5部とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造において水溶性高分子(C)を添加せず、電極活物質(A)の量を91.0部、複合体(D)の量を1.5部とした以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造において用いる電極活物質(A)の量を90.7部、水溶性高分子(C)の量を固形分換算量で0.8部とし、複合体(D)に代えて表面処理が施されていない結晶セルロース(d’’)としてFD-F20(平均粒子径20μm、旭化成ケミカルズ社製)を1.0部用いた以外は、実施例1と同様に複合粒子用スラリーの製造、複合粒子の製造、正極の製造、リチウムイオン二次電池の製造を行った。水溶性高分子(C)と表面処理が施されていない結晶セルロース(d’’)との重量比(C)/(d'')は0.8であった。
(粒子状結着樹脂(B)の製造)
攪拌機付き5MPa耐圧容器に、スチレン47部、1,3-ブタジエン50部、メタクリル酸3部、ドデシルベンゼンスルホン酸ナトリウム4部、イオン交換水150部、連鎖移動剤としてt-ドデシルメルカプタン0.4部および重合開始剤として過硫酸カリウム0.5部を入れ、十分に攪拌した後、50℃に加温して重合を開始した。重合転化率が96%になった時点で冷却し反応を停止して、粒子状結着樹脂(B)としてのスチレン・ブタジエン共重合体(以下、「SBR」ということがある。)を得た。
電極活物質(A)として負極活物質の人造黒鉛(平均粒子径:24.5μm、黒鉛層間距離(X線回折法による(002)面の面間隔(d値)):0.354nm)91.1部及びSiC 6.6部と、粒子状結着樹脂(B)として上記SBRを固形分換算量で1.4部、水溶性高分子(C)としてカルボキシメチルセルロース(以下、「CMC」と略記することがある。)の1.0%水溶液(BSH-12;第一工業製薬社製)を固形分換算量で0.3部、複合体(D)としてセオラスRC-591(水溶性高分子(d')の種類:カルボキシメチルセルロースのナトリウム塩(以下、「CMC-Na」ということがある。)、水溶性高分子(d')と結晶セルロース(d'')との重量比=11:89、平均粒子径:8μm、旭化成ケミカルズ社製)を固形分換算量で0.6部混合し、さらにイオン交換水を固形分濃度が40%となるように加え、混合分散して複合粒子用スラリーを得た。水溶性高分子(C)と複合体(D)との重量比(C)/(D)は0.5であった。
上記複合粒子用スラリーをスプレー乾燥機(大川原化工機社製)において、回転円盤方式のアトマイザ(直径65mm)を用い、回転数25,000rpm、熱風温度150℃、粒子回収出口の温度を90℃として、噴霧乾燥造粒を行い、複合粒子を得た。この複合粒子の平均体積粒子径は40μmであった。
次に、得られた粒子をロールプレス機(押し切り粗面熱ロール、ヒラノ技研工業社製)のロール(ロール温度100℃、プレス線圧400kN/m)に供給し、成形速度20m/分でシート状に成形し、厚さ80μmのリチウムイオン二次電池用負極を得た。
正極活物質として層状構造を有するLiCoO2(以下、「LCO」と略記することがある。)92部に、正極用結着樹脂としてポリフッ化ビニリデン(PVDF;クレハ化学社製「KF-1100」)を固形分量が2部となるように加え、さらに、アセチレンブラック(電気化学工業社製「HS-100」)を6部、N-メチルピロリドン20部を加えて、プラネタリーミキサーで混合して正極用のスラリー組成物を得た。この正極用のスラリー組成物を厚さ18μmのアルミニウム箔に塗布し、120℃で30分乾燥した後、ロールプレスして厚さ60μmのリチウムイオン二次電池用正極を得た。
単層のポリプロピレン製セパレーター(幅65mm、長さ500mm、厚さ25μm、乾式法により製造、気孔率55%)を、5×5cm2の正方形に切り抜いた。
電池の外装として、アルミ包材外装を用意した。上記で得られたリチウムイオン二次電池用正極を、4×4cm2の正方形に切り出し、集電体側の表面がアルミ包材外装に接するように配置した。リチウムイオン二次電池用正極の正極活物質層の面上に、上記で得られた正方形のセパレーターを配置した。さらに、上記で得られたリチウムイオン二次電池用負極を、4.2×4.2cm2の正方形に切り出し、負極活物質層側の表面がセパレーターに向かい合うように、セパレーター上に配置した。更に、ビニレンカーボネートを2.0%含有する、濃度1.0MのLiPF6溶液を充填した。このLiPF6溶液の溶媒はエチレンカーボネート(EC)とエチルメチルカーボネート(EMC)との混合溶媒(EC/EMC=3/7(体積比))である。さらに、アルミニウム包材の開口を密封するために、150℃でヒートシールをしてアルミニウム外装を閉口し、ラミネート型のリチウムイオン二次電池(ラミネート型セル)を製造した。
複合体(D)としてRC-N-30(水溶性高分子(d')の種類:キサンタンガム及びデキストリン、キサンタンガムとデキストリンと結晶セルロース(d'')との重量比=5:20:75、平均粒子径:8μm、旭化成ケミカルズ社製)を使用した以外は、実施例10と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造に用いる電極活物質(A)の量を人造黒鉛91.0部及びSiC 6.6部とし、水溶性高分子(C)の量を固形分換算量で0.2部、複合体(D)の量を0.8部とした以外は、実施例10と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。水溶性高分子(C)と複合体(D)との重量比(C)/(D)は0.25であった。
複合粒子用スラリーの製造に用いる電極活物質(A)の量を人造黒鉛90.6部及びSiC 6.6部とし、水溶性高分子(C)としてCMCの1.0%水溶液(BSH-6;第一工業製薬社製)を固形分換算量で1.3部を用い、複合体(D)の量を固形分換算量で0.1部とした以外は、実施例10と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。水溶性高分子(C)と複合体(D)との重量比(C)/(D)は13.0であった。
複合粒子用スラリーの製造に用いる電極活物質(A)を人造黒鉛97.7部とし、粒子状結着樹脂(B)の種類をアクリレート系重合体とした以外は、実施例10と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。なお、アクリレート系重合体は、実施例1と同様のものを使用した。
複合粒子用スラリーの製造に用いる電極活物質(A)を人造黒鉛97.6部とし、粒子状結着樹脂(B)の量を1.2部とし、水溶性高分子(C)をポリアクリル樹脂0.4部とし、複合体(D)の量を0.8部とした以外は、実施例10と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。水溶性高分子(C)と複合体(D)との重量比(C)/(D)は0.5であった。なお、ポリアクリル樹脂は、実施例7と同様のものを使用した。
複合粒子用スラリーの製造に用いる水溶性高分子(C)として、ポリ-N-ビニルアセトアミド樹脂(PNVA、GE191-103;昭和電工社製)を用いた以外は、実施例15と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造に用いる水溶性高分子(C)として、ポリビニルアルコール樹脂(PVA、JF-17)を用いた以外は、実施例15と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造において複合体(D)を添加せず、電極活物質(A)を人造黒鉛97.7部とし、水溶性高分子(C)の量を固形分換算量で0.9部とした以外は、実施例10と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造において水溶性高分子(C)を添加せず、電極活物質(A)の量を人造黒鉛97.7部とし、複合体(D)の量を固形分換算量で0.9部とした以外は、実施例10と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。
複合粒子用スラリーの製造に用いる電極活物質(A)を人造黒鉛97.7部、水溶性高分子(C)の量を固形分換算量で0.4部とし、複合体(D)に代えて表面処理が施されていない結晶セルロース(d'')としてFD-F20(平均粒子径20μm、旭化成ケミカルズ社製)を0.6部用いた以外は、実施例10と同様に複合粒子用スラリーの製造、複合粒子の製造、リチウムイオン二次電池用負極の製造、リチウムイオン二次電池の製造を行った。水溶性高分子(C)と表面処理が施されていない結晶セルロース(d'')との重量比(C)/(d'')は0.67であった。
Claims (8)
- 電極活物質(A)、粒子状結着樹脂(B)、水溶性高分子(C)及び水溶性高分子(d')と結晶セルロース(d'')との複合体(D)を含む電気化学素子電極用複合粒子。
- 前記電極活物質(A)100重量部に対し、前記水溶性高分子(C)を0.1~10重量部、前記複合体(D)を0.1~2.0重量部それぞれ含み、前記水溶性高分子(C)と前記複合体(D)との重量比(C)/(D)が(C)/(D)=0.2~15である請求項1記載の電気化学素子電極用複合粒子。
- 前記複合体(D)は、水中に分散する性質を有する請求項1または2記載の電気化学素子電極用複合粒子。
- 前記粒子状結着樹脂(B)は、共役ジエン系重合体またはアクリレート系重合体の少なくとも一方を含む請求項1~3の何れか一項に記載の電気化学素子電極用複合粒子。
- 前記複合体(D)の一次平均粒子径は、10μm以下である請求項1~4の何れか一項に記載の電気化学素子電極用複合粒子。
- 前記複合体(D)は、水溶性高分子(d’)で表面処理された結晶セルロース(d’’)である請求項1に記載の電気化学素子電極用複合粒子。
- さらに導電剤(E)を含む請求項1~6の何れか一項に記載の電気化学素子電極用複合粒子。
- 請求項1~7のいずれかに記載の電気化学素子電極用複合粒子を製造する方法であって、
前記水溶性高分子(d’)で前記結晶セルロース(d’’)を表面処理することにより前記複合体(D)を得る工程と、
前記電極活物質(A)、前記粒子状結着樹脂(B)、前記水溶性高分子(C)及び前記複合体(D)を混合して複合粒子用スラリーを得る工程と、
前記複合粒子用スラリーを造粒することにより、電気化学素子電極用複合粒子を得る工程と
を有する電気化学素子電極用複合粒子の製造方法。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15796229.1A EP3147972B1 (en) | 2014-05-20 | 2015-05-12 | Composite particles for electrochemical device electrode and method for manufacturing composite particles for electrochemical device electrode |
| JP2016521043A JP6540692B2 (ja) | 2014-05-20 | 2015-05-12 | 電気化学素子電極用複合粒子および電気化学素子電極用複合粒子の製造方法 |
| US15/305,583 US11081699B2 (en) | 2014-05-20 | 2015-05-12 | Method for manufacturing electrochemical device electrode |
| CN201580021144.8A CN106233510B (zh) | 2014-05-20 | 2015-05-12 | 电化学元件电极用复合粒子和电化学元件电极用复合粒子的制造方法 |
| PL15796229T PL3147972T3 (pl) | 2014-05-20 | 2015-05-12 | Cząstki kompozytowe dla elektrody urządzenia elektrochemicznego i sposób wytwarzania cząstek kompozytowych dla elektrody urządzenia elektrochemicznego |
| KR1020167030295A KR102319301B1 (ko) | 2014-05-20 | 2015-05-12 | 전기 화학 소자 전극용 복합 입자 및 전기 화학 소자 전극용 복합 입자의 제조 방법 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014104041 | 2014-05-20 | ||
| JP2014-104041 | 2014-05-20 | ||
| JP2014-104042 | 2014-05-20 | ||
| JP2014104042 | 2014-05-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015178241A1 true WO2015178241A1 (ja) | 2015-11-26 |
Family
ID=54553914
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/063561 Ceased WO2015178241A1 (ja) | 2014-05-20 | 2015-05-12 | 電気化学素子電極用複合粒子および電気化学素子電極用複合粒子の製造方法 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US11081699B2 (ja) |
| EP (1) | EP3147972B1 (ja) |
| JP (1) | JP6540692B2 (ja) |
| KR (1) | KR102319301B1 (ja) |
| CN (1) | CN106233510B (ja) |
| HU (1) | HUE040706T2 (ja) |
| PL (1) | PL3147972T3 (ja) |
| WO (1) | WO2015178241A1 (ja) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017090553A1 (ja) * | 2015-11-27 | 2017-06-01 | 積水化学工業株式会社 | キャパシタ用電極材及びキャパシタ |
| JP6868751B1 (ja) * | 2019-12-24 | 2021-05-12 | 昭和電工株式会社 | 非水系二次電池電極、電極スラリー、及び非水系二次電池 |
| JP2021093292A (ja) * | 2019-12-10 | 2021-06-17 | 株式会社豊田中央研究所 | 電極、電極の製造方法、蓄電デバイス及び蓄電デバイスの製造方法 |
| WO2021131278A1 (ja) * | 2019-12-24 | 2021-07-01 | 昭和電工株式会社 | 非水系二次電池電極、電極スラリー、及び非水系二次電池 |
| JP2022058783A (ja) * | 2016-05-20 | 2022-04-12 | キョーセラ・エイブイエックス・コンポーネンツ・コーポレーション | マルチセル・ウルトラキャパシタ |
| WO2024142986A1 (ja) * | 2022-12-28 | 2024-07-04 | 日本ゼオン株式会社 | 複合粒子、二次電池用電極、および二次電池 |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20200287207A1 (en) * | 2019-03-06 | 2020-09-10 | Nanotek Instruments, Inc. | Process for producing porous particulates of graphene shell-protected alkali metal, electrodes, and alkali metal battery |
| CN114207882A (zh) * | 2019-08-13 | 2022-03-18 | Jsr株式会社 | 蓄电装置用组合物、蓄电装置电极用浆料、蓄电装置电极和蓄电装置 |
| US12573716B2 (en) | 2020-03-31 | 2026-03-10 | Zeon Corporation | Composition for electrochemical device functional layer, laminate for electrochemical device, and electrochemical device |
| WO2022140974A1 (zh) * | 2020-12-28 | 2022-07-07 | 宁德新能源科技有限公司 | SiOC复合材料及其制备方法和应用 |
| WO2022204974A1 (zh) * | 2021-03-30 | 2022-10-06 | 宁德新能源科技有限公司 | 电化学装置及电子装置 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005116855A (ja) * | 2003-10-09 | 2005-04-28 | Nippon Zeon Co Ltd | 電気二重層キャパシタ用電極の製造方法 |
| JP2005340072A (ja) * | 2004-05-28 | 2005-12-08 | Matsushita Electric Ind Co Ltd | 非水系二次電池用正極板の製造方法 |
| JP2005340071A (ja) * | 2004-05-28 | 2005-12-08 | Matsushita Electric Ind Co Ltd | 非水系二次電池用正極板の製造方法 |
| JP2005340073A (ja) * | 2004-05-28 | 2005-12-08 | Matsushita Electric Ind Co Ltd | 非水系二次電池用正極板の製造方法 |
| JP2005340074A (ja) * | 2004-05-28 | 2005-12-08 | Matsushita Electric Ind Co Ltd | 非水系二次電池用正極板の製造方法 |
| JP2006303395A (ja) * | 2005-04-25 | 2006-11-02 | Nippon Zeon Co Ltd | 電気化学素子電極用複合粒子の製造方法 |
| JP2013041821A (ja) * | 2011-07-20 | 2013-02-28 | Nippon Zeon Co Ltd | 電気化学素子正極電極用複合粒子、電気化学素子正極電極材料、及び電気化学素子正極電極 |
| WO2015098632A1 (ja) * | 2013-12-26 | 2015-07-02 | 日本ゼオン株式会社 | 電気化学素子電極用複合粒子 |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5032001A (en) | 1990-03-09 | 1991-07-16 | At&T Bell Laboratories | Optical fiber having enhanced bend resistance |
| JP4336927B2 (ja) * | 1999-01-28 | 2009-09-30 | 日本ゼオン株式会社 | リチウムイオン二次電池電極用バインダー組成物およびその利用 |
| JP2001176757A (ja) | 1999-12-14 | 2001-06-29 | Asahi Glass Co Ltd | 電気二重層キャパシタ |
| US6773838B2 (en) * | 2000-09-04 | 2004-08-10 | Matsushita Electric Industrial Co., Ltd. | Non-aqueous electrolyte secondary battery and negative electrode for the same |
| JP3615472B2 (ja) * | 2000-09-04 | 2005-02-02 | 松下電器産業株式会社 | 非水電解質電池 |
| JP4219705B2 (ja) | 2003-02-17 | 2009-02-04 | パナソニック株式会社 | 二次電池用電極の製造法 |
| JP4077432B2 (ja) * | 2003-07-07 | 2008-04-16 | Tdk株式会社 | 電気化学素子 |
| JP4449447B2 (ja) * | 2003-12-22 | 2010-04-14 | 日産自動車株式会社 | 固体電解質電池の製造方法 |
| JP5088807B2 (ja) * | 2004-04-19 | 2012-12-05 | パナソニック株式会社 | リチウムイオン二次電池およびその製造法 |
| KR100803470B1 (ko) * | 2004-04-19 | 2008-02-14 | 마쯔시다덴기산교 가부시키가이샤 | 리튬이온 2차전지 및 그 제조법 |
| WO2009069086A2 (en) * | 2007-11-27 | 2009-06-04 | Stroemme Maria | Composite materials including an intrinsically conducting polymer, and methods and devices |
| CN102906165B (zh) * | 2010-05-12 | 2016-05-25 | 多伦多大学董事局 | 产生导电聚合物和纤维素纳米复合物的方法 |
| JP5594548B2 (ja) * | 2010-08-25 | 2014-09-24 | トヨタ自動車株式会社 | 電池用電極の製造方法 |
| US9685658B2 (en) | 2011-07-15 | 2017-06-20 | Zeon Corporation | Composite particles for electrochemical device electrode, material for electrochemical device electrode, and electrochemical device electrode |
| US20140225300A1 (en) * | 2011-08-31 | 2014-08-14 | Zeon Corporation | Powder molding device and production method for powder molded product |
| CN103907226A (zh) * | 2011-09-14 | 2014-07-02 | 日本瑞翁株式会社 | 电化学元件用电极 |
| US20130189589A1 (en) * | 2012-01-23 | 2013-07-25 | Masdar Institute Of Science And Technology | Fabrication of cellulose polymer composites and their application as solid electrolytes |
| CN103396500B (zh) * | 2013-08-07 | 2016-08-17 | 中国科学院广州能源研究所 | 天然高分子衍生物-导电聚合物水性复合粘结剂及其应用 |
-
2015
- 2015-05-12 US US15/305,583 patent/US11081699B2/en not_active Expired - Fee Related
- 2015-05-12 CN CN201580021144.8A patent/CN106233510B/zh active Active
- 2015-05-12 HU HUE15796229A patent/HUE040706T2/hu unknown
- 2015-05-12 JP JP2016521043A patent/JP6540692B2/ja active Active
- 2015-05-12 KR KR1020167030295A patent/KR102319301B1/ko active Active
- 2015-05-12 WO PCT/JP2015/063561 patent/WO2015178241A1/ja not_active Ceased
- 2015-05-12 PL PL15796229T patent/PL3147972T3/pl unknown
- 2015-05-12 EP EP15796229.1A patent/EP3147972B1/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005116855A (ja) * | 2003-10-09 | 2005-04-28 | Nippon Zeon Co Ltd | 電気二重層キャパシタ用電極の製造方法 |
| JP2005340072A (ja) * | 2004-05-28 | 2005-12-08 | Matsushita Electric Ind Co Ltd | 非水系二次電池用正極板の製造方法 |
| JP2005340071A (ja) * | 2004-05-28 | 2005-12-08 | Matsushita Electric Ind Co Ltd | 非水系二次電池用正極板の製造方法 |
| JP2005340073A (ja) * | 2004-05-28 | 2005-12-08 | Matsushita Electric Ind Co Ltd | 非水系二次電池用正極板の製造方法 |
| JP2005340074A (ja) * | 2004-05-28 | 2005-12-08 | Matsushita Electric Ind Co Ltd | 非水系二次電池用正極板の製造方法 |
| JP2006303395A (ja) * | 2005-04-25 | 2006-11-02 | Nippon Zeon Co Ltd | 電気化学素子電極用複合粒子の製造方法 |
| JP2013041821A (ja) * | 2011-07-20 | 2013-02-28 | Nippon Zeon Co Ltd | 電気化学素子正極電極用複合粒子、電気化学素子正極電極材料、及び電気化学素子正極電極 |
| WO2015098632A1 (ja) * | 2013-12-26 | 2015-07-02 | 日本ゼオン株式会社 | 電気化学素子電極用複合粒子 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3147972A4 * |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017090553A1 (ja) * | 2015-11-27 | 2017-06-01 | 積水化学工業株式会社 | キャパシタ用電極材及びキャパシタ |
| CN107615428A (zh) * | 2015-11-27 | 2018-01-19 | 积水化学工业株式会社 | 电容器用电极材料及电容器 |
| JPWO2017090553A1 (ja) * | 2015-11-27 | 2018-09-13 | 積水化学工業株式会社 | キャパシタ用電極材及びキャパシタ |
| JP2022058783A (ja) * | 2016-05-20 | 2022-04-12 | キョーセラ・エイブイエックス・コンポーネンツ・コーポレーション | マルチセル・ウルトラキャパシタ |
| JP7441249B2 (ja) | 2016-05-20 | 2024-02-29 | キョーセラ・エイブイエックス・コンポーネンツ・コーポレーション | マルチセル・ウルトラキャパシタ |
| JP2021093292A (ja) * | 2019-12-10 | 2021-06-17 | 株式会社豊田中央研究所 | 電極、電極の製造方法、蓄電デバイス及び蓄電デバイスの製造方法 |
| JP7176504B2 (ja) | 2019-12-10 | 2022-11-22 | 株式会社豊田中央研究所 | 電極、電極の製造方法、蓄電デバイス及び蓄電デバイスの製造方法 |
| JP6868751B1 (ja) * | 2019-12-24 | 2021-05-12 | 昭和電工株式会社 | 非水系二次電池電極、電極スラリー、及び非水系二次電池 |
| WO2021131278A1 (ja) * | 2019-12-24 | 2021-07-01 | 昭和電工株式会社 | 非水系二次電池電極、電極スラリー、及び非水系二次電池 |
| WO2024142986A1 (ja) * | 2022-12-28 | 2024-07-04 | 日本ゼオン株式会社 | 複合粒子、二次電池用電極、および二次電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20170004973A (ko) | 2017-01-11 |
| EP3147972A1 (en) | 2017-03-29 |
| CN106233510A (zh) | 2016-12-14 |
| JP6540692B2 (ja) | 2019-07-10 |
| JPWO2015178241A1 (ja) | 2017-04-20 |
| EP3147972A4 (en) | 2017-10-11 |
| US11081699B2 (en) | 2021-08-03 |
| HUE040706T2 (hu) | 2019-03-28 |
| CN106233510B (zh) | 2018-09-28 |
| EP3147972B1 (en) | 2018-09-05 |
| US20170040613A1 (en) | 2017-02-09 |
| PL3147972T3 (pl) | 2019-03-29 |
| KR102319301B1 (ko) | 2021-10-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6954424B2 (ja) | 電気化学素子電極用複合粒子、電気化学素子電極及び電気化学素子 | |
| JP6217741B2 (ja) | 電気化学素子電極用複合粒子、電気化学素子電極用複合粒子の製造方法、電気化学素子電極および電気化学素子 | |
| JP6540692B2 (ja) | 電気化学素子電極用複合粒子および電気化学素子電極用複合粒子の製造方法 | |
| JP6344384B2 (ja) | 電気化学素子電極用複合粒子、電気化学素子電極用複合粒子の製造方法、電気化学素子電極および電気化学素子 | |
| JP6327249B2 (ja) | 電気化学素子電極用バインダー、電気化学素子電極用粒子複合体、電気化学素子電極、電気化学素子及び電気化学素子電極の製造方法 | |
| JP6380526B2 (ja) | 電気化学素子電極用複合粒子 | |
| JPWO2014192652A6 (ja) | 電気化学素子電極用バインダー、電気化学素子電極用粒子複合体、電気化学素子電極、電気化学素子及び電気化学素子電極の製造方法 | |
| JP6485359B2 (ja) | 電気化学素子電極用複合粒子 | |
| JP6451934B2 (ja) | 電気化学素子電極用複合粒子 | |
| JP6344143B2 (ja) | 電気化学素子電極用複合粒子の製造方法、電気化学素子電極の製造方法及び電気化学素子の製造方法 | |
| JP6398461B2 (ja) | 電気化学素子電極用複合粒子の製造方法 | |
| JP6365160B2 (ja) | 電気化学素子電極用複合粒子の製造方法、電気化学素子電極の製造方法及び電気化学素子の製造方法 | |
| JP6372273B2 (ja) | 電気化学素子電極用複合粒子、電気化学素子電極、電気化学素子、電気化学素子電極用複合粒子の製造方法及び電気化学素子電極の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15796229 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016521043 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15305583 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015796229 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015796229 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20167030295 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |