WO2019059868A2 - Pharmaceutical combinations of tenofovir, emtricitabine and efavirenz - Google Patents
Pharmaceutical combinations of tenofovir, emtricitabine and efavirenz Download PDFInfo
- Publication number
- WO2019059868A2 WO2019059868A2 PCT/TR2018/050508 TR2018050508W WO2019059868A2 WO 2019059868 A2 WO2019059868 A2 WO 2019059868A2 TR 2018050508 W TR2018050508 W TR 2018050508W WO 2019059868 A2 WO2019059868 A2 WO 2019059868A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- component
- pharmaceutical composition
- oral pharmaceutical
- composition according
- weight
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC[C@@](CN=C(COC)O)*(C=C(C)C(*C)=*1)C1=O Chemical compound CC[C@@](CN=C(COC)O)*(C=C(C)C(*C)=*1)C1=O 0.000 description 1
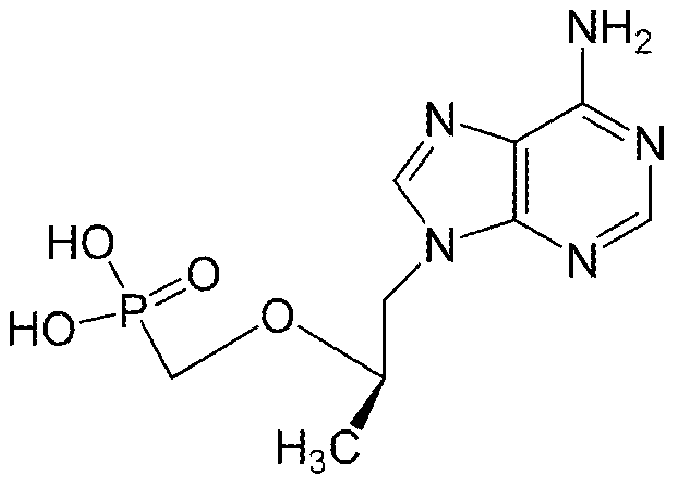
- SGOIRFVFHAKUTI-ZCFIWIBFSA-N C[C@H](C[n]1c(ncnc2N)c2nc1)OCP(O)(O)=O Chemical compound C[C@H](C[n]1c(ncnc2N)c2nc1)OCP(O)(O)=O SGOIRFVFHAKUTI-ZCFIWIBFSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2072—Pills, tablets, discs, rods characterised by shape, structure or size; Tablets with holes, special break lines or identification marks; Partially coated tablets; Disintegrating flat shaped forms
- A61K9/2086—Layered tablets, e.g. bilayer tablets; Tablets of the type inert core-active coat
- A61K9/209—Layered tablets, e.g. bilayer tablets; Tablets of the type inert core-active coat containing drug in at least two layers or in the core and in at least one outer layer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/536—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines ortho- or peri-condensed with carbocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/66—Phosphorus compounds
- A61K31/683—Diesters of a phosphorus acid with two hydroxy compounds, e.g. phosphatidylinositols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
- A61K31/7064—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines
- A61K31/7068—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines having oxo groups directly attached to the pyrimidine ring, e.g. cytidine, cytidylic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
- A61K9/1605—Excipients; Inactive ingredients
- A61K9/1629—Organic macromolecular compounds
- A61K9/1652—Polysaccharides, e.g. alginate, cellulose derivatives; Cyclodextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2095—Tabletting processes
Definitions
- the present invention relates to pharmaceutical combinations comprising tenofovir or a pharmaceutically acceptable salt thereof, emtricitabine or a pharmaceutically acceptable salt thereof and efavirenz or a pharmaceutically acceptable salt thereof.
- Tenofovir is a nucleotide analogue reverse transcriptase inhibitor (NtRTI).
- the chemical name of tenofovir is ( ⁇ [(2R)-1 -(6-amino-9H-purin-9-yl)propan-2-yl]oxy ⁇ methyl)phosphonic acid and has the structure shown in the following formula I.
- Tenofovir disoproxil fumarate is a prodrug of tenofovir and is marketed under the brand name VIREAD ® .
- Tenofovir disoproxil fumarate blocks the reverse transcriptase enzyme which is crucial for the human immunodeficiency virus 1 (HIV-1 ) and the hepatitis B virus. Therefore, it is used in the treatment of HIV-1 and hepatitis B.
- Tenofovir disoproxil fumarate has the following structural formula.
- Emtricitabine is a synthetic fluoro derivative of thiacytidine with potent antiviral activity.
- the chemical name of emtricitabine is 4-amino-5-fluoro-1 -[(2R,5S)-2-(hydroxymethyl)- 1 ,3-oxathiolan-5-yl]-1 ,2-dihydropyrimidin-2-one and has the structure shown in the following formula III.
- Emtricitabine with trade name EMTRIVA ® , is a nucleoside reverse transcriptase inhibitor (NRTI) for the treatment of HIV infection in adults and children.
- NRTI nucleoside reverse transcriptase inhibitor
- Emtricitabine is phosphorylated to form emtricitabine 5'-triphosphate within the cell.
- This metabolite inhibits the activity of human immunodeficiency virus (HIV) reverse transcriptase both by competing with the natural substrate deoxycytidine 5'-triphosphate and by incorporation into viral DNA causing a termination of DNA chain elongation.
- HIV human immunodeficiency virus
- Efavirenz is a synthetic non-nucleoside reverse transcriptase inhibitor (NNRTI) with antiviral activity. Efavirenz binds directly to the human immunodeficiency virus type 1 (HIV-1 ) RT, an RNA-dependent DNA polymerase, blocking its function in viral DNA replication.
- NRTI non-nucleoside reverse transcriptase inhibitor
- Efavirenz which is used as part of highly active antiretroviral therapy (HAART) for the treatment of a human immunodeficiency virus (HIV) type 1 , is marketed under the brand name SUSTIVA ®
- combinations of compounds can yield an equivalent antiviral effect with reduced toxicity, or an increase in drug efficacy.
- the decrease in dosages thanks to the administration of combinations can reduce the frequency of occurrence of drug-resistant variants of HIV.
- the prior art has many formulation examples of binary combinations of tenofovir disoproxil fumarate and emtricitabine for the treatment of HIV infection. It is also suggested to add a third antiviral active agent to this combination.
- This active agent can be a protease inhibitor (PI), a nucleoside reverse transcriptase inhibitor (NRTI), a non- nucleoside reverse transcriptase inhibitor (NNRTI) or an integrase inhibitor are some of them.
- all the compounds may be administered in a binary or ternary combinations at once.
- these compounds are delivered serially, sequentially, parallelly or simultaneously in separate pharmaceutical formulations or combinations. This administration, which is named as alternation therapy, is a little risky due to the possible delay, omission or misadministration of a second or a third compound.
- Patent document numbered EP1583542B1 basically emphasizes binary combinations of tenofovir disoproxil fumarate (tenofovir DF) and emtricitabine and aims to develop chemically stable tablet formulations of them.
- the main object of the present invention is to obtain pharmaceutical combinations comprising tenofovir disoproxil fumarate (tenofovir DF), emtricitabine and efavirenz eliminating all aforesaid problems and bringing additional advantages to the relevant prior art.
- tenofovir DF tenofovir disoproxil fumarate
- emtricitabine emtricitabine
- Another object of the present invention is to obtain combinations with high stability and patient compliance.
- a further object of the present invention is to develop combinations having improved dissolution rate.
- a further object of the present invention is to develop combinations having decreased friability and brittleness.
- a further object of the present invention is to develop combinations having increased crushing strength.
- Another object of the present invention is to provide a bilayer tablet dosage form which has surfactant in both layers.
- Another object of the present invention is to provide a bilayer tablet dosage form which has decreased amount of binder in both layers.
- Another object of the present invention is to provide a bilayer tablet dosage form which has decreased amount of lubricant in both layers.
- a further object of the present invention is to improve a process for preparing a bilayer tablet dosage form in which wet granulation is carried out for both layers.
- Figure 1 shows the top view of an embodiment of the oral dosage form subjected to the invention which has one score.
- Figure 2 shows the lateral view of the embodiment of the oral dosage form subjected to the invention which has one score.
- Figure 3 shows the lateral cross sectional area of the embodiment of the oral dosage form subjected to the invention which has one score.
- Figure 4 shows the lateral view of another embodiment of the oral dosage form subjected to the invention which has two scores.
- Figure 5 shows the lateral cross sectional area of the embodiment of the oral dosage form subjected to the invention which has two score.
- the present invention relates to an oral pharmaceutical composition
- an oral pharmaceutical composition comprising tenofovir or a pharmaceutically acceptable salt thereof, emtricitabine or a pharmaceutically acceptable salt thereof and efavirenz or a pharmaceutically acceptable salt thereof in combination.
- the composition comprises; -a first component (1 ) comprising tenofovir or a pharmaceutically acceptable salt thereof, emtricitabine or a pharmaceutically acceptable salt thereof and
- -a second component (2) comprising efavirenz or a pharmaceutically acceptable salt thereof.
- these two components are present discretely in a single dosage form and more preferably these components are layers.
- the first component (1 ) and the second component (2) are in contact with one another.
- these components can be separated by an inert layer.
- the composition is a solid dosage form which is in the form of coated tablet, trilayer tablet, bilayer tablet, multilayer tablet, orally disintegrating tablet, mini tablet, pellet, sugar pellet, buccal tablet, sublingual tablet, effervescent tablet, immediate release tablet, modified release tablet, gastric disintegrating tablet, pill, capsule, oral granule, powder, coated bead system, microsphere, tablet in tablet, inlay tablet, dragee, sachet or orally administrable film.
- composition is in the form of a bilayer tablet.
- the dosage form has at least one score (3) to enable splitting, thus serial, sequent and simultaneous administration of at least two smaller pieces than the whole dosage form.
- the dosage form is scored on one side as seen in Figure 1 , 2 and 3.
- the dosage form is scored bilaterally as seen in Figure 4 and 5.
- the dosage form further comprises a film coating.
- the composition comprises tenofovir disoproxil fumarate (tenofovir DF) with emtricitabine in the first component (1 ) and efavirenz in the second component (2).
- the composition comprises tenofovir DF in the amount of 1 -60% by weight of the total composition. Preferably this amount is between 5-50% by weight of the total composition. More preferably tenofovir DF is present in the amount of 10-30% by weight in the total composition.
- the composition comprises emtricitabine in the amount of 1 -60% by weight of the total composition. Preferably this amount is between 5-30% by weight of the total composition. More preferably emtricitabine is present in the amount of 5-20% by weight in the total composition.
- the composition comprises efavirenz in the amount of 5-70% by weight of the total composition. Preferably this amount is between 10-60% by weight of the total composition. More preferably efavirenz is present in the amount of 30-50% by weight in the total composition.
- the weight of the total composition is between 500 and 2500 mg, preferably lower than 1595 mg.
- the weight of the first layer (1 ) is between 150-1400 mg, preferably 300-1250 mg, more preferably 600-950 mg. In the most preferred embodiment, it is 750 mg.
- the weight of the second layer (2) is between 150-1400 mg, preferably 300-1250 mg, more preferably 600-950 mg. In the most preferred embodiment, it is 800 mg.
- Tenofovir DF is present in an amount of 100 to 1000 mg, preferably 200 to 500 mg and more preferably 300 mg in the composition.
- Emtricitabine is present in an amount of 100 to 1000 mg, preferably 100 to 500 mg and more preferably 200 mg in the composition.
- Efavirenz is present in an amount of 100 to 1000 mg, preferably 300 to 800 mg and more preferably 600 mg in the composition.
- the amount of tenofovir DF is between 10-80%, preferably 20-70%, more preferably 30- 50% by weight of the first component (1 ).
- the amount of emtricitabine is between 5-70%, preferably 10-50%, more preferably 15- 35% by weight of the first component (1 ).
- the first component (1 ) comprises at least one excipient selected from surfactants, solubilizers, wetting agents or mixtures thereof.
- the first component (1 ) comprises at least one surfactant which acts also as a wetting agent and a solubilizer and which is selected from the group comprising carboxylates, sulfates, sulfonates, phosphate esters.
- the said surfactant is selected from alkyl sulfates. More preferably, the selected surfactant is sodium lauryl sulfate (SLS).
- SLS sodium lauryl sulfate
- SLS is known to reduce interfacial tension between the solid particles and the granulation liquid and to improve wetting and deaggregation of active agent particles. Thus, it enables a homogeneous mixture and accordingly enhances physical properties of the final dosage form.
- the amount of sodium lauryl sulfate is lower than 1 % by weight of the first component (1 ). More preferably, this amount is lower than 0.5%.
- the dosage form comprising the composition is preferably in the form of a scored tablet which is supposed to be split in half and this kind of tablets should have the strength not to crumble to more than two pieces. Improvement in powder wettability can often improve the bioavailability of the formulation.
- the first component (1 ) further comprises at least one excipient selected from binders, disintegrants, lubricants, glidants or mixtures thereof.
- the first component (1 ) of the composition comprises at least one binder which is selected from the group comprising polyvinylpyrrolidone (PVP, povidone), microcrystalline cellulose, copovidone, copolyvidone, polyvinylpyrrolidone K30 (povidone K30), carnauba wax, pullulan, polymethacrylate, glyceryl behenate, hydroxypropyl cellulose (HPC), carboxymethyl cellulose (CMC), methyl cellulose (MC), hydroxyethyl cellulose, sodium carboxymethyl cellulose (Na CMC), ethyl cellulose, polymetacrylates, polyethylene oxide, polyvinyl alcohol, polycarbophil, polyvinyl acetate and its copolymers, gelatin, xanthan gum, guar gum
- the amount of binder in the first component (1 ) is lower than 10% by weight of the first component (1 ). More preferably, this amount is lower than 5%. This value range, which is much lower than the prior art, is important to enhance the final tablet dissolution rate since it is known that high amounts of binder have a negative influence on dissolution rate.
- the first component (1 ) comprises hydroxypropyl cellulose as binder. More preferably, Klucel LF, which has a low viscosity (75-150 cps), is used as hydroxypropyl cellulose. The amount of hydroxypropyl cellulose is between 0.1 -10%, preferably 1 -5% by weight of the first component (1 ).
- the first component (1 ) of the composition comprises at least one diluent which is selected from the group comprising lactose monohydrate, lactose, microcrystalline cellulose, mannitol, spray-dried mannitol, starch, dextrose, sucrose, fructose, maltose, sorbitol, xylitol, inositol, kaolin, inorganic salts, polysaccharides, sodium chloride, dextrates, lactitol, maltodextrin, sucrose-maltodextrin mixture, trehalose, sodium carbonate, sodium bicarbonate, calcium carbonate or mixtures thereof.
- diluent which is selected from the group comprising lactose monohydrate, lactose, microcrystalline cellulose, mannitol, spray-dried mannitol, starch, dextrose, sucrose, fructose, maltose, sorbitol, xylitol, in
- the first component (1 ) comprises lactose monohydrate as diluent.
- the amount of lactose monohydrate is between 1 -50%, preferably 5-40%, more preferably 5-20% by weight of the first component (1 ).
- the first component (1 ) of the composition comprises at least one disintegrant which is selected from the group comprising croscarmellose sodium, sodium starch glycolate, microcrystalline cellulose, sodium carbonate, hydroxylpropyl cellulose (HPC), cross-linked polyvinylpyrrolidone (crospovidone), copovidon, polycarbophil, low-substitue poloxamer, alginic acid and alginates, ion-exchange resins, magnesium aluminum silica, sodium carboxy methyl cellulose, carboxy methyl cellulose calcium, docusate sodium, guar gum, polyacrylin potasium, sodium alginate, sodium glycine carbonate or mixtures thereof.
- disintegrant which is selected from the group comprising croscarmellose sodium, sodium starch glycolate, microcrystalline cellulose, sodium carbonate, hydroxylpropyl cellulose (HPC), cross-linked polyvinylpyrrolidone (crospovidone), copovidon, polycarbophil, low-substi
- the first component (1 ) comprises two disintegrants which are microcrystalline cellulose and croscarmellose sodium.
- the amount of microcrystalline cellulose is between 0.1 -50%, preferably 1 -20%, more preferably 3-10% by weight of the first component (1 ).
- the amount of croscarmellose sodium is between 1 -50%, preferably 5-30%, more preferably 8-20% by weight of the first component (1 ).
- the first component (1 ) of the composition comprises at least one lubricant and at least one glidant which are selected from the group comprising sodium lauryl sulfate, sodium stearyl fumarate, magnesium stearate, colloidal silicon dioxide, zinc stearate, calcium stearate, mineral oil, talc, polyethylene glycol, glyceryl monostearate, glyceryl palmitostearate, magnesium lauryl sulfate, fumaric acid, zinc stearate, stearic acid, hydrogenated natural oils, silica, paraffin or mixtures thereof.
- the first component (1 ) comprises magnesium stearate as lubricant and colloidal silicon dioxide as glidant.
- the amount of magnesium stearate is lower than 2% by weight of the first component (1 ). More preferably, this amount is kept lower than 1 .5% to reduce the final tablet friability and brittleness which is essential for scored tablets.
- the amount of colloidal silicon dioxide is lower than 2% by weight of the first component
- this amount is lower than 1 %.
- This amount of colloidal silicon dioxide is required to prevent sedimentation during the compression of scored tablets.
- the amount of the first component (1 ) is between 10-90%, preferably 20-80%, more preferably 40-60% by weight of the total composition.
- the amount of efavirenz is between 10-95%, preferably 30-90%, more preferably 60-80% by weight of the second component (2).
- the second component (2) further comprises at least one excipient selected from binders, disintegrants, lubricants, glidants, surfactants, solubilizers, wetting agents or mixtures thereof.
- the second component (2) of the composition comprises at least one binder which is selected from the group comprising polyvinylpyrrolidone (PVP, povidone), microcrystalline cellulose, copovidone, copolyvidone, polyvinylpyrrolidone K30 (povidone K30), carnauba wax, pullulan, polymethacrylate, glyceryl behenate, hydroxypropyl cellulose (HPC), carboxymethyl cellulose (CMC), methyl cellulose (MC), hydroxyethyl cellulose, sodium carboxymethyl cellulose (Na CMC), ethyl cellulose, polymetacrylates, polyethylene oxide, polyvinyl alcohol, polycarbophil, polyvinyl acetate and its copolymers, gelatin, xanthan gum, guar gum, alginate, carrageen, collagen, agar, pectin, hyaluronic acid, carbomer, cellulose acetate phthalate, hydroxyethy
- PVP polyviny
- the second component (2) is lower than 15% by weight of the second component (2). More preferably, this amount is lower than 5%. This value range, which is much lower than the prior art, is important to enhance the final tablet dissolution rate since it is known that high amounts of binder have a negative influence on dissolution rate.
- the second component (2) comprises hydroxypropyl cellulose as binder. More preferably, Klucel LF, which has a low viscosity (75-150 cps), is used as hydroxypropyl cellulose. The amount of hydroxypropyl cellulose is between 0.1 -10%, preferably 1 -5% by weight of the second component (2).
- the second component (2) of the composition comprises at least one diluent which is selected from the group comprising lactose monohydrate, lactose, microcrystalline cellulose, mannitol, spray-dried mannitol, starch, dextrose, sucrose, fructose, maltose, sorbitol, xylitol, inositol, kaolin, inorganic salts, polysaccharides, sodium chloride, dextrates, lactitol, maltodextrin, sucrose- maltodextrin mixture, trehalose, sodium carbonate, sodium bicarbonate, calcium carbonate or mixtures thereof.
- diluent which is selected from the group comprising lactose monohydrate, lactose, microcrystalline cellulose, mannitol, spray-dried mannitol, starch, dextrose, sucrose, fructose, maltose, sorbitol, xylitol, inos
- the second component (2) comprises lactose monohydrate as diluent.
- the amount of lactose monohydrate is between 1 -50%, preferably 3-40%, more preferably 5-20% by weight of the second component (2).
- the second component (2) of the composition comprises at least one disintegrant which is selected from the group comprising croscarmellose sodium, sodium starch glycolate, microcrystalline cellulose, sodium carbonate, hydroxylpropyl cellulose (HPC), cross-linked polyvinylpyrrolidone (crospovidone), copovidon, polycarbophil, low-substitue poloxamer, alginic acid and alginates, ion-exchange resins, magnesium aluminum silica, sodium carboxy methyl cellulose, carboxy methyl cellulose calcium, docusate sodium, guar gum, polyacrylin potasium, sodium alginate, sodium glycine carbonate or mixtures thereof.
- disintegrant is selected from the group comprising croscarmellose sodium, sodium starch glycolate, microcrystalline cellulose, sodium carbonate, hydroxylpropyl cellulose (HPC), cross-linked polyvinylpyrrolidone (crospovidone), copovidon, polycarbophil, low-substitue
- the second component (2) comprises two disintegrants which are microcrystalline cellulose and croscarmellose sodium.
- the amount of microcrystalline cellulose is between 0.1 -50%, preferably 1 -20%, more preferably 2-10% by weight of the second component (2).
- the amount of croscarmellose sodium is between 0.5-50%, preferably 1 -30%, more preferably 2-10% by weight of the second component (2).
- the second component (2) of the composition comprises at least one lubricant and at least one glidant which are selected from the group comprising sodium lauryl sulfate, sodium stearyl fumarate, magnesium stearate, colloidal silicon dioxide, zinc stearate, calcium stearate, mineral oil, talc, polyethylene glycol, glyceryl monostearate, glyceryl palmitostearate, magnesium lauryl sulfate, fumaric acid, zinc stearate, stearic acid, hydrogenated natural oils, silica, paraffin or mixtures thereof.
- the second component (2) comprises magnesium stearate as lubricant and colloidal silicon dioxide as glidant.
- the amount of magnesium stearate is lower than 2% by weight of the second component (2). More preferably, this amount is kept lower than 1 .5% to reduce the final tablet friability and brittleness which is essential for scored tablets.
- the amount of colloidal silicon dioxide is lower than 2% by weight of the second component (2). More preferably, this amount is lower than 1 %. This amount of colloidal silicon dioxide is required to prevent sedimentation during the compression of scored tablets.
- the second component (2) of the composition comprises at least one surfactant which acts also as a wetting agent and a solubilizer and which is selected from the group comprising carboxylates, sulfates, sulfonates, phosphate esters.
- SLS is known to reduce interfacial tension between the solid particles and the granulation liquid and to improve wetting and deaggregation of active agent particles. Thus, it enables a homogeneous mixture and accordingly enhances physical properties of the final dosage form.
- the amount of sodium lauryl sulfate is higher than 2% by weight of the second component (2). In the preferred embodiment, this amount is between 2-10%, more preferably 2-5%. It has been seen that the presence of this amount of SLS in the second component (2) surprisingly increases the final tablet hardness, strength and dissolution rate.
- the composition is preferably in the form of a scored tablet which is supposed to be split in half and this kind of tablets should have the strength not to crumble to more than two pieces. Improvement in powder wettability can often improve the bioavailability of the formulation.
- the amount of the second component (2) is between 10-90%, preferably 20-80%, more preferably 40-60% by weight of the total composition.
- the coating is Opadry II.
- the amount of the film coating is 1 -10%, preferably %2-5 by weight of the total composition.
- the composition comprises;
- the below given formulations can be used in the solid oral pharmaceutical composition subjected to the invention. These examples are not limiting the scope of the present invention and should be considered under the light of the foregoing detailed disclosure.
- Example 1 Ratios by weight of the components
- Example 2 Ratios by weight of the total composition (coated bilayer tablet)
- compositions mentioned above are prepared by following these steps:
- the mixture composing the first component (1 ) is prepared by following these steps:
- the mixture composing the second component (2) is prepared by following these steps:
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Epidemiology (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
The present invention relates to a scored bilayer tablet comprising tenofovir or a pharmaceutically acceptable salt thereof, emtricitabine or a pharmaceutically acceptable salt thereof and efavirenz or a pharmaceutically acceptable salt thereof.
Description
PHARMACEUTICAL COMBINATIONS OF TENOFOVIR, EMTRICITABINE AND
EFAVIRENZ
Field of Invention
The present invention relates to pharmaceutical combinations comprising tenofovir or a pharmaceutically acceptable salt thereof, emtricitabine or a pharmaceutically acceptable salt thereof and efavirenz or a pharmaceutically acceptable salt thereof.
Background of Invention
Tenofovir is a nucleotide analogue reverse transcriptase inhibitor (NtRTI). The chemical name of tenofovir is ({[(2R)-1 -(6-amino-9H-purin-9-yl)propan-2-yl]oxy}methyl)phosphonic acid and has the structure shown in the following formula I.
Formula I: Tenofovir
Tenofovir disoproxil fumarate is a prodrug of tenofovir and is marketed under the brand name VIREAD®. Tenofovir disoproxil fumarate blocks the reverse transcriptase enzyme which is crucial for the human immunodeficiency virus 1 (HIV-1 ) and the hepatitis B virus. Therefore, it is used in the treatment of HIV-1 and hepatitis B. Tenofovir disoproxil fumarate has the following structural formula.
Formula II: Tenofovir disoproxil fumarate
Emtricitabine is a synthetic fluoro derivative of thiacytidine with potent antiviral activity. The chemical name of emtricitabine is 4-amino-5-fluoro-1 -[(2R,5S)-2-(hydroxymethyl)- 1 ,3-oxathiolan-5-yl]-1 ,2-dihydropyrimidin-2-one and has the structure shown in the following formula III.
Formula III: Emtricitabine
Emtricitabine, with trade name EMTRIVA®, is a nucleoside reverse transcriptase inhibitor (NRTI) for the treatment of HIV infection in adults and children.
Emtricitabine is phosphorylated to form emtricitabine 5'-triphosphate within the cell. This metabolite inhibits the activity of human immunodeficiency virus (HIV) reverse transcriptase both by competing with the natural substrate deoxycytidine 5'-triphosphate and by incorporation into viral DNA causing a termination of DNA chain elongation.
Efavirenz is a synthetic non-nucleoside reverse transcriptase inhibitor (NNRTI) with antiviral activity. Efavirenz binds directly to the human immunodeficiency virus type 1 (HIV-1 ) RT, an RNA-dependent DNA polymerase, blocking its function in viral DNA replication.
Efavirenz, which is used as part of highly active antiretroviral therapy (HAART) for the treatment of a human immunodeficiency virus (HIV) type 1 , is marketed under the brand name SUSTIVA®
Its chemical name is (4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluoromethyl)-1 H-3,1 - benzoxazin-2-one and it has the structure shown in the following formula IV.
Formula IV: Efavirenz
The use of combinations of compounds can yield an equivalent antiviral effect with reduced toxicity, or an increase in drug efficacy. The decrease in dosages thanks to the administration of combinations can reduce the frequency of occurrence of drug-resistant variants of HIV.
The prior art has many formulation examples of binary combinations of tenofovir disoproxil fumarate and emtricitabine for the treatment of HIV infection. It is also suggested to add a third antiviral active agent to this combination. This active agent can be a protease inhibitor (PI), a nucleoside reverse transcriptase inhibitor (NRTI), a non- nucleoside reverse transcriptase inhibitor (NNRTI) or an integrase inhibitor are some of them.
According to an approach in the art, all the compounds may be administered in a binary or ternary combinations at once. In another administration example, these compounds are delivered serially, sequentially, parallelly or simultaneously in separate pharmaceutical formulations or combinations. This administration, which is named as alternation therapy, is a little risky due to the possible delay, omission or misadministration of a second or a third compound.
Accordingly, it is more preferable to formulate a single dosage form comprising all the compounds. Therefore, overcoming the incompatibility problem of these compounds and providing the chemical stability of their combinations play an important role in the formulation success. Another important point to take into consideration is to maintain patient compliance, since the size of the dosage form enlarges due to the increase in the number of required layer to ensure the stability. Hence, one of the significant problems with multilayer tablets is the swallowing difficulty.
Patent document numbered EP1583542B1 basically emphasizes binary combinations of tenofovir disoproxil fumarate (tenofovir DF) and emtricitabine and aims to develop chemically stable tablet formulations of them. The document makes mention of a treatment in which tenofovir disoproxil fumarate, emtricitabine, and efavirenz are administered by two separate tablets. It is also mentioned the possibility to make a unitary and chemically stable combination of these three compounds. However, this statement is left unsupported, since there is no suggestions or examples for such a ternary combination formulation.
The main problem with combining these three active agents into a unitary and homogeneous composition is the incompatibilities among them. The prior art gives the teaching that tenofovir and its pharmaceutically acceptable salts are incompatible with efavirenz or its formulations. In some documents in the art, this incompatibility is assumed as a problem related not to efavirenz but to the excipients, such as surfactants, which are present in the efavirenz formulations. Consequently; surfactants, particularly SLS, are excluded from tenofovir DF and emtricitabine combinations.
The technical problem handled by the prior art is the stability issues of tenofovir DF and emtricitabine binary combinations. Accordingly, dry granulation is suggested to enhance the stability and improvements related to the preparing process have been carried out.
Although ternary combinations with efavirenz is mentioned in the prior art, these statements don't touch on the increased tablet size caused by the bilayer form and its disadvantages such as swallowing difficulty. Therefore, the prior art does not give any teaching, suggestion or motivation about how to enhance patient compliance and ensure high stability and high dissolution rate at the same time.
Besides, in the prior art, the amount of binder and lubricant are always kept high in the formulations to maintain the stability. But it is a well-known fact that the increase in the amount of binder causes the decrease in the dissolution rate. Besides, the tablet size shall get bigger in such case. On the other hand, high amounts of lubricant, especially magnesium stearate, decreases dissolution rate and crushing strength and may also increase tablet friability {Handbook of Pharmaceutical Excipients, Sixth edition, page 405).
Obviously, there is still a need for a bilayer ternary combination of these active agents which has high stability and at the same time high strength and low friability to enable the administration of a scored tablet form which will enhance the patient compliance.
Objects and Brief Description of the Invention
The main object of the present invention is to obtain pharmaceutical combinations comprising tenofovir disoproxil fumarate (tenofovir DF), emtricitabine and efavirenz eliminating all aforesaid problems and bringing additional advantages to the relevant prior art.
Another object of the present invention is to obtain combinations with high stability and patient compliance.
A further object of the present invention is to develop combinations having improved dissolution rate.
A further object of the present invention is to develop combinations having decreased friability and brittleness.
A further object of the present invention is to develop combinations having increased crushing strength. Another object of the present invention is to provide a bilayer tablet dosage form which has surfactant in both layers.
Another object of the present invention is to provide a bilayer tablet dosage form which has decreased amount of binder in both layers.
Another object of the present invention is to provide a bilayer tablet dosage form which has decreased amount of lubricant in both layers.
Another object of the present invention is to provide a bilayer tablet dosage form with a score which is enabling serial, sequent and simultaneous administration of two smaller pieces of this dosage form.
Yet, another object of the present invention is to provide a bilayer tablet dosage form comprising at least one film coating to protect the pharmaceutical composition against the moisture to maintain the stability.
A further object of the present invention is to improve a process for preparing a bilayer tablet dosage form in which wet granulation is carried out for both layers.
Detailed Description of Invention
In accordance with the objects outlined above, detailed features of the present invention are given herein.
List of figures:
Figure 1 shows the top view of an embodiment of the oral dosage form subjected to the invention which has one score.
Figure 2 shows the lateral view of the embodiment of the oral dosage form subjected to the invention which has one score.
Figure 3 shows the lateral cross sectional area of the embodiment of the oral dosage form subjected to the invention which has one score. Figure 4 shows the lateral view of another embodiment of the oral dosage form subjected to the invention which has two scores.
Figure 5 shows the lateral cross sectional area of the embodiment of the oral dosage form subjected to the invention which has two score.
The present invention relates to an oral pharmaceutical composition comprising tenofovir or a pharmaceutically acceptable salt thereof, emtricitabine or a pharmaceutically acceptable salt thereof and efavirenz or a pharmaceutically acceptable salt thereof in combination.
According to the preferred embodiment, the composition comprises;
-a first component (1 ) comprising tenofovir or a pharmaceutically acceptable salt thereof, emtricitabine or a pharmaceutically acceptable salt thereof and
-a second component (2) comprising efavirenz or a pharmaceutically acceptable salt thereof.
According to this preferred embodiment, these two components are present discretely in a single dosage form and more preferably these components are layers. According to the preferred embodiment, the first component (1 ) and the second component (2) are in contact with one another. In another embodiment, these components can be separated by an inert layer.
According to this preferred embodiment, the composition is a solid dosage form which is in the form of coated tablet, trilayer tablet, bilayer tablet, multilayer tablet, orally disintegrating tablet, mini tablet, pellet, sugar pellet, buccal tablet, sublingual tablet, effervescent tablet, immediate release tablet, modified release tablet, gastric disintegrating tablet, pill, capsule, oral granule, powder, coated bead system, microsphere, tablet in tablet, inlay tablet, dragee, sachet or orally administrable film.
According to the preferred embodiment the composition is in the form of a bilayer tablet.
According to this preferred embodiment, the dosage form has at least one score (3) to enable splitting, thus serial, sequent and simultaneous administration of at least two smaller pieces than the whole dosage form. According to an embodiment, the dosage form is scored on one side as seen in Figure 1 , 2 and 3.
According to another embodiment, the dosage form is scored bilaterally as seen in Figure 4 and 5.
According to the preferred embodiment, the dosage form further comprises a film coating.
According to the most preferred embodiment of the invention; the composition comprises tenofovir disoproxil fumarate (tenofovir DF) with emtricitabine in the first component (1 ) and efavirenz in the second component (2).
The composition comprises tenofovir DF in the amount of 1 -60% by weight of the total composition. Preferably this amount is between 5-50% by weight of the total composition. More preferably tenofovir DF is present in the amount of 10-30% by weight in the total composition.
The composition comprises emtricitabine in the amount of 1 -60% by weight of the total composition. Preferably this amount is between 5-30% by weight of the total composition. More preferably emtricitabine is present in the amount of 5-20% by weight in the total composition.
The composition comprises efavirenz in the amount of 5-70% by weight of the total composition. Preferably this amount is between 10-60% by weight of the total composition. More preferably efavirenz is present in the amount of 30-50% by weight in the total composition.
In one embodiment of the invention, the weight of the total composition is between 500 and 2500 mg, preferably lower than 1595 mg.
The weight of the first layer (1 ) is between 150-1400 mg, preferably 300-1250 mg, more preferably 600-950 mg. In the most preferred embodiment, it is 750 mg.
The weight of the second layer (2) is between 150-1400 mg, preferably 300-1250 mg, more preferably 600-950 mg. In the most preferred embodiment, it is 800 mg. Tenofovir DF is present in an amount of 100 to 1000 mg, preferably 200 to 500 mg and more preferably 300 mg in the composition.
Emtricitabine is present in an amount of 100 to 1000 mg, preferably 100 to 500 mg and more preferably 200 mg in the composition.
Efavirenz is present in an amount of 100 to 1000 mg, preferably 300 to 800 mg and more preferably 600 mg in the composition.
The amount of tenofovir DF is between 10-80%, preferably 20-70%, more preferably 30- 50% by weight of the first component (1 ).
The amount of emtricitabine is between 5-70%, preferably 10-50%, more preferably 15- 35% by weight of the first component (1 ).
According to the preferred embodiments, the first component (1 ) comprises at least one excipient selected from surfactants, solubilizers, wetting agents or mixtures thereof.
According to the most preferred embodiment, the first component (1 ) comprises at least one surfactant which acts also as a wetting agent and a solubilizer and which is selected from the group comprising carboxylates, sulfates, sulfonates, phosphate esters.
In the preferred embodiment of the invention, the said surfactant is selected from alkyl sulfates. More preferably, the selected surfactant is sodium lauryl sulfate (SLS).
SLS is known to reduce interfacial tension between the solid particles and the granulation liquid and to improve wetting and deaggregation of active agent particles. Thus, it enables a homogeneous mixture and accordingly enhances physical properties of the final dosage form.
In the preferred embodiment, the amount of sodium lauryl sulfate is lower than 1 % by weight of the first component (1 ). More preferably, this amount is lower than 0.5%.
It has been seen that the presence of this amount of SLS in the first component (1 ) surprisingly increases the final tablet hardness, strength and dissolution rate. Besides, it has been found that the timing of the addition of SLS in the first component (1 ) mixture is also important. The said advantages of SLS is more potent when it is added to the granules just before the final blending step of the mixture related to first component (1 ).
Improved hardness and strength of the tablet is significantly essential for this invention since the dosage form comprising the composition is preferably in the form of a scored tablet which is supposed to be split in half and this kind of tablets should have the strength not to crumble to more than two pieces. Improvement in powder wettability can often improve the bioavailability of the formulation.
According to this preferred embodiment, the first component (1 ) further comprises at least one excipient selected from binders, disintegrants, lubricants, glidants or mixtures thereof.
According to one embodiment of the invention, the first component (1 ) of the composition comprises at least one binder which is selected from the group comprising polyvinylpyrrolidone (PVP, povidone), microcrystalline cellulose, copovidone, copolyvidone, polyvinylpyrrolidone K30 (povidone K30), carnauba wax, pullulan, polymethacrylate, glyceryl behenate, hydroxypropyl cellulose (HPC), carboxymethyl cellulose (CMC), methyl cellulose (MC), hydroxyethyl cellulose, sodium carboxymethyl cellulose (Na CMC), ethyl cellulose, polymetacrylates, polyethylene oxide, polyvinyl alcohol, polycarbophil, polyvinyl acetate and its copolymers, gelatin, xanthan gum, guar gum, alginate, carrageen, kollagen, agar, pectin, hyaluronic acid, carbomer, cellulose acetate phthalate, hydroxyethyl methyl cellulose, polaxomer, polyethylene glycol (PEG), sugars, glycose syrups, natural gums, tragacanth gum, polyacrylamide, aluminum hydroxide, benthonite, laponite, setostearyl alcohol, polyoxyethylene-alkyl ethers, acacia mucilage, polydextrose or mixtures thereof.
According to the preferred embodiment, the amount of binder in the first component (1 ) is lower than 10% by weight of the first component (1 ). More preferably, this amount is lower than 5%. This value range, which is much lower than the prior art, is important to enhance the final tablet dissolution rate since it is known that high amounts of binder have a negative influence on dissolution rate.
According to preferred embodiments above, the first component (1 ) comprises hydroxypropyl cellulose as binder. More preferably, Klucel LF, which has a low viscosity (75-150 cps), is used as hydroxypropyl cellulose. The amount of hydroxypropyl cellulose is between 0.1 -10%, preferably 1 -5% by weight of the first component (1 ).
It has been seen that this selected binder and its amount in the formulation surprisingly enhances the dissolution profile. According to one embodiment of the invention, the first component (1 ) of the composition comprises at least one diluent which is selected from the group comprising lactose monohydrate, lactose, microcrystalline cellulose, mannitol, spray-dried mannitol, starch, dextrose, sucrose, fructose, maltose, sorbitol, xylitol, inositol, kaolin, inorganic salts, polysaccharides, sodium chloride, dextrates, lactitol, maltodextrin, sucrose-maltodextrin mixture, trehalose, sodium carbonate, sodium bicarbonate, calcium carbonate or mixtures thereof.
According to the preferred embodiment of the invention, the first component (1 ) comprises lactose monohydrate as diluent. The amount of lactose monohydrate is between 1 -50%, preferably 5-40%, more preferably 5-20% by weight of the first component (1 ).
According to one embodiment of the invention, the first component (1 ) of the composition comprises at least one disintegrant which is selected from the group comprising croscarmellose sodium, sodium starch glycolate, microcrystalline cellulose, sodium carbonate, hydroxylpropyl cellulose (HPC), cross-linked polyvinylpyrrolidone (crospovidone), copovidon, polycarbophil, low-substitue poloxamer, alginic acid and alginates, ion-exchange resins, magnesium aluminum silica, sodium carboxy methyl cellulose, carboxy methyl cellulose calcium, docusate sodium, guar gum, polyacrylin potasium, sodium alginate, sodium glycine carbonate or mixtures thereof.
In the preferred embodiment, the first component (1 ) comprises two disintegrants which are microcrystalline cellulose and croscarmellose sodium. The amount of microcrystalline cellulose is between 0.1 -50%, preferably 1 -20%, more preferably 3-10% by weight of the first component (1 ). The amount of croscarmellose sodium is between 1 -50%, preferably 5-30%, more preferably 8-20% by weight of the first component (1 ).
According to one embodiment of the invention, the first component (1 ) of the composition comprises at least one lubricant and at least one glidant which are selected from the group comprising sodium lauryl sulfate, sodium stearyl fumarate, magnesium stearate, colloidal silicon dioxide, zinc stearate, calcium stearate, mineral oil, talc, polyethylene glycol, glyceryl monostearate, glyceryl palmitostearate, magnesium lauryl sulfate, fumaric acid, zinc stearate, stearic acid, hydrogenated natural oils, silica, paraffin or mixtures thereof. In the preferred embodiment of the invention, the first component (1 ) comprises magnesium stearate as lubricant and colloidal silicon dioxide as glidant.
According to the most preferred embodiment, the amount of magnesium stearate is lower than 2% by weight of the first component (1 ). More preferably, this amount is kept lower than 1 .5% to reduce the final tablet friability and brittleness which is essential for scored tablets.
The amount of colloidal silicon dioxide is lower than 2% by weight of the first component
(1 ) . More preferably, this amount is lower than 1 %. This amount of colloidal silicon dioxide is required to prevent sedimentation during the compression of scored tablets.
The amount of the first component (1 ) is between 10-90%, preferably 20-80%, more preferably 40-60% by weight of the total composition.
The amount of efavirenz is between 10-95%, preferably 30-90%, more preferably 60-80% by weight of the second component (2).
According to any of these embodiments, the second component (2) further comprises at least one excipient selected from binders, disintegrants, lubricants, glidants, surfactants, solubilizers, wetting agents or mixtures thereof.
According to one embodiment of the invention, the second component (2) of the composition comprises at least one binder which is selected from the group comprising polyvinylpyrrolidone (PVP, povidone), microcrystalline cellulose, copovidone, copolyvidone, polyvinylpyrrolidone K30 (povidone K30), carnauba wax, pullulan, polymethacrylate, glyceryl behenate, hydroxypropyl cellulose (HPC), carboxymethyl cellulose (CMC), methyl cellulose (MC), hydroxyethyl cellulose, sodium carboxymethyl cellulose (Na CMC), ethyl cellulose, polymetacrylates, polyethylene oxide, polyvinyl alcohol, polycarbophil, polyvinyl acetate and its copolymers, gelatin, xanthan gum, guar gum, alginate, carrageen, collagen, agar, pectin, hyaluronic acid, carbomer, cellulose acetate phthalate, hydroxyethyl methyl cellulose, polaxomer, polyethylene glycol (PEG), sugars, glycose syrups, natural gums, tragacanth gum, polyacrylamide, aluminum hydroxide, benthonite, laponite, setostearyl alcohol, polyoxyethylene-alkyl ethers, acacia mucilage, polydextrose or mixtures thereof. According to the preferred embodiment, the amount of binder in the second component
(2) is lower than 15% by weight of the second component (2). More preferably, this amount is lower than 5%. This value range, which is much lower than the prior art, is important to enhance the final tablet dissolution rate since it is known that high amounts of binder have a negative influence on dissolution rate.
According to preferred embodiments above, the second component (2) comprises hydroxypropyl cellulose as binder. More preferably, Klucel LF, which has a low viscosity (75-150 cps), is used as hydroxypropyl cellulose. The amount of hydroxypropyl cellulose is between 0.1 -10%, preferably 1 -5% by weight of the second component (2).
It has been seen that this selected binder and its amount in the formulation surprisingly enhances the dissolution profile.
According to one embodiment of the invention, the second component (2) of the composition comprises at least one diluent which is selected from the group comprising lactose monohydrate, lactose, microcrystalline cellulose, mannitol, spray-dried mannitol, starch, dextrose, sucrose, fructose, maltose, sorbitol, xylitol, inositol, kaolin, inorganic salts, polysaccharides, sodium chloride, dextrates, lactitol, maltodextrin, sucrose- maltodextrin mixture, trehalose, sodium carbonate, sodium bicarbonate, calcium carbonate or mixtures thereof.
According to the preferred embodiment of the invention, the second component (2) comprises lactose monohydrate as diluent. The amount of lactose monohydrate is between 1 -50%, preferably 3-40%, more preferably 5-20% by weight of the second component (2).
According to one embodiment of the invention, the second component (2) of the composition comprises at least one disintegrant which is selected from the group comprising croscarmellose sodium, sodium starch glycolate, microcrystalline cellulose, sodium carbonate, hydroxylpropyl cellulose (HPC), cross-linked polyvinylpyrrolidone (crospovidone), copovidon, polycarbophil, low-substitue poloxamer, alginic acid and alginates, ion-exchange resins, magnesium aluminum silica, sodium carboxy methyl cellulose, carboxy methyl cellulose calcium, docusate sodium, guar gum, polyacrylin potasium, sodium alginate, sodium glycine carbonate or mixtures thereof.
In the preferred embodiment, the second component (2) comprises two disintegrants which are microcrystalline cellulose and croscarmellose sodium. The amount of microcrystalline cellulose is between 0.1 -50%, preferably 1 -20%, more preferably 2-10% by weight of the second component (2). The amount of croscarmellose sodium is between 0.5-50%, preferably 1 -30%, more preferably 2-10% by weight of the second component (2).
According to one embodiment of the invention, the second component (2) of the composition comprises at least one lubricant and at least one glidant which are selected from the group comprising sodium lauryl sulfate, sodium stearyl fumarate, magnesium stearate, colloidal silicon dioxide, zinc stearate, calcium stearate, mineral oil, talc, polyethylene glycol, glyceryl monostearate, glyceryl palmitostearate, magnesium lauryl sulfate, fumaric acid, zinc stearate, stearic acid, hydrogenated natural oils, silica, paraffin or mixtures thereof. In the preferred embodiment of the invention, the second component (2) comprises magnesium stearate as lubricant and colloidal silicon dioxide as glidant.
According to the most preferred embodiment, the amount of magnesium stearate is lower than 2% by weight of the second component (2). More preferably, this amount is kept lower than 1 .5% to reduce the final tablet friability and brittleness which is essential for scored tablets.
The amount of colloidal silicon dioxide is lower than 2% by weight of the second component (2). More preferably, this amount is lower than 1 %. This amount of colloidal silicon dioxide is required to prevent sedimentation during the compression of scored tablets.
According to one embodiment of the invention, the second component (2) of the composition comprises at least one surfactant which acts also as a wetting agent and a solubilizer and which is selected from the group comprising carboxylates, sulfates, sulfonates, phosphate esters.
In the preferred embodiment of the invention, the second component (2) comprises a surfactant which is selected from alkyl sulfates. More preferably, the selected surfactant is sodium lauryl sulfate (SLS).
SLS is known to reduce interfacial tension between the solid particles and the granulation liquid and to improve wetting and deaggregation of active agent particles. Thus, it enables a homogeneous mixture and accordingly enhances physical properties of the final dosage form.
The amount of sodium lauryl sulfate is higher than 2% by weight of the second component (2). In the preferred embodiment, this amount is between 2-10%, more preferably 2-5%. It has been seen that the presence of this amount of SLS in the second component (2) surprisingly increases the final tablet hardness, strength and dissolution rate.
Improved hardness and strength of the tablet is significantly essential for this invention since the composition is preferably in the form of a scored tablet which is supposed to be split in half and this kind of tablets should have the strength not to crumble to more than two pieces. Improvement in powder wettability can often improve the bioavailability of the formulation.
The amount of the second component (2) is between 10-90%, preferably 20-80%, more preferably 40-60% by weight of the total composition.
According to the preferred embodiment of the invention, the dosage form of the composition further comprises at least one film coating to protect the composition against the moisture to maintain the stability. Suitable coating ingredients are selected from the group comprising hypromellose, lactose monohydrate, hydroxypropyl cellulose, polyvinyl alcohol (PVA), ethyl cellulose, polyethylene glycol (PEG), talc, triethyl citrate, polyvinyl alcohol-polyethylene glycol copolymers (Kollicoat® IR), ethyl cellulose dispersions (Surelease®), polyvinylprolidone, polyvinylprolidone-vinyl acetate copolymer (PVP-VA), all kinds of Opadry®, pigments, dyes, titanium dioxide, iron oxide or mixtures thereof. According to one embodiment, the film coating comprises polyvinyl alcohol, polyethylene glycol, titanium dioxide, talc, pigment or mixtures thereof.
Preferably, the coating is Opadry II. The amount of the film coating is 1 -10%, preferably %2-5 by weight of the total composition.
According to these embodiments, the composition comprises;
— 10-80% of tenofovir disoproxil fumarate,
— 5-70% of emtricitabine,
— 0.1 -10% of hydroxypropyl cellulose,
— 1 -50% of lactose monohydrate,
— 0.1 -50% of microcrystalline cellulose,
— 1 -50% of croscarmellose sodium,
— lower than 2% of magnesium stearate,
— lower than 2% of colloidal silicon dioxide,
— lower than 1 % of sodium lauryl sulfate
by weight of the first component (1 );
— 10-95% of efavirenz,
— 0.1 -10% of hydroxypropyl cellulose,
— 1 -50% of lactose monohydrate
— 0.1 -50% of microcrystalline cellulose,
— 0.5-50% of croscarmellose sodium,
— lower than 2% of magnesium stearate,
— lower than 2% of colloidal silicon dioxide,
— 2-10% of sodium lauryl sulfate
by weight of the second component (2) and
— 1 -10% film coating by weight of the total composition.
These selected ratios ensure the required effective doses for the treatment, enhance stability and dissolution profile and reduce friability and brittleness of the film coated bilayer tablet subjected to the invention.
According to all these embodiments, the below given formulations can be used in the solid oral pharmaceutical composition subjected to the invention. These examples are not limiting the scope of the present invention and should be considered under the light of the foregoing detailed disclosure.
Example 1 : Ratios by weight of the components
The compositions mentioned above are prepared by following these steps:
- preparation of the mixture composing the first component by (1 ) by wet granulation - preparation of the mixture composing the second component (2) by wet granulation
- compressing these two mixtures together into scored bilayer tablets
- coating these scored bilayer tablets with a film coating solution
The mixture composing the first component (1 ) is prepared by following these steps:
- mixing tenofovir disoproxil fumarate, emtricitabine, microcrystalline cellulose, lactose monohydrate, hydroxypropyl cellulose and one-third of croscarmellose sodium
- granulating this mixture with water
- sieving the granules through a 5mm mesh and drying them
- sieving the dried granules again through a 1 mm mesh
- adding two-thirds of croscarmellose sodium, sodium lauryl sulfate and colloidal silicon dioxide (sieved through the same 1 mm mesh) to the sieved granules and mixing them for 15 minutes
- adding magnesium stearate (sieved through the same 1 mm mesh) and mixing them for 5 minutes
The mixture composing the second component (2) is prepared by following these steps:
- mixing efavirenz, microcrystalline cellulose, lactose monohydrate, hydroxypropyl cellulose and croscarmellose sodium
- granulating this mixture with water and sodium lauryl sulfate
- sieving the granules through a 5mm mesh and drying them
- sieving the dried granules again through a 1 mm mesh
- adding colloidal silicon dioxide (sieved through the same 1 mm mesh) to the sieved granules and mixing them for 15 minutes
- adding magnesium stearate (sieved through the same 1 mm mesh) and mixing them for 5 minutes
Claims
1 . An oral pharmaceutical composition comprising
-a first component (1 ) comprising tenofovir or a pharmaceutically acceptable salt thereof and emtricitabine or a pharmaceutically acceptable salt thereof and -a second component (2) comprising efavirenz or a pharmaceutically acceptable salt thereof;
wherein the composition is a solid dosage form which has at least one score (3).
2. The oral pharmaceutical composition according to claim 1 , wherein the first component (1 ) and the second component (2) are discrete layers.
3. The oral pharmaceutical composition according to claim 2, wherein the composition is in the form of a bilayer tablet.
4. The oral pharmaceutical composition according to claim 3, wherein the composition is in the form of a film-coated bilayer tablet.
5. The oral pharmaceutical composition according to any one of the preceding claims, wherein the said first component (1 ) comprises tenofovir disoproxil fumarate and emtricitabine and the said second component (2) comprises efavirenz.
6. The oral pharmaceutical composition according to any one of the preceding claims, wherein the first component (1 ) comprises a surfactant.
7. The oral pharmaceutical composition according to claim 6, wherein the surfactant is selected from the group comprising carboxylates, sulfates, sulfonates and phosphate esters.
8. The oral pharmaceutical composition according to claim 7, wherein the said surfactant is selected from sulfates which is sodium lauryl sulfate.
9. The oral pharmaceutical composition according to claim 8, wherein the amount of sodium lauryl sulfate lower than 1 % by weight of the first component (1 ).
10. The oral pharmaceutical composition according to any one of the preceding claims, wherein the first component (1 ) further comprises at least one excipient selected from binders, disintegrants, lubricants, glidants or mixtures thereof.
1 1 . The oral pharmaceutical composition according to claim 10, wherein the amount of binder is lower than 10% by weight of the first component (1 ).
1 2. The oral pharmaceutical composition according to claim 10, wherein the composition comprises magnesium stearate as lubricant in the amount of lower than 2% by weight of the first component (1 ).
1 3. The oral pharmaceutical composition according to claim 10, wherein the composition comprises colloidal silicon dioxide as glidant in the amount of lower than 2% by weight of the first component (1 ).
14. The oral pharmaceutical composition according to any one of the preceding claims, wherein the second component (2) comprises at least one excipient selected from binders, disintegrants, lubricants, glidants, surfactants, solubilizers, wetting agents or mixtures thereof.
1 5. The oral pharmaceutical composition according to claim 14, wherein the amount of binder is lower than 15% by weight of the second component (2).
1 6. The oral pharmaceutical composition according to claim 14, wherein the composition comprises magnesium stearate as lubricant in the amount of lower than 2% by weight of the second component (2).
17. The oral pharmaceutical composition according to claim 14, wherein the composition comprises colloidal silicon dioxide as glidant in the amount of lower than 2% by weight of the second component (2).
1 8. The oral pharmaceutical composition according to claim 14, wherein the composition comprises sodium lauryl sulfate as surfactant in the amount of higher than 2% by weight of the second component (2).
1 9. The oral pharmaceutical composition according to any one of the preceding claims, wherein the composition comprises;
— 10-80% of tenofovir disoproxil fumarate,
— 5-70% of emtricitabine,
— 0.1 -10% of hydroxypropyl cellulose,
— 1 -50% of lactose monohydrate,
— 0.1 -50% of microcrystalline cellulose,
— 1 -50% of croscarmellose sodium,
— lower than 2% of magnesium stearate,
— lower than 2% of colloidal silicon dioxide,
— lower than 1 % of sodium lauryl sulfate
by weight of the first component (1 );
— 10-95% of efavirenz,
— 0.1 -10% of hydroxypropyl cellulose,
— 1 -50% of lactose monohydrate
— 0.1 -50% of microcrystalline cellulose,
— 0.5-50% of croscarmellose sodium,
— lower than 2% of magnesium stearate,
— lower than 2% of colloidal silicon dioxide,
— 2-10% of sodium lauryl sulfate
by weight of the second component (2) and
— 1 -10% film coating by weight of the total composition.
20. A process for preparing the oral pharmaceutical composition according to claim 19, comprising the following steps:
- preparation of the mixture composing the first component by (1 ) by wet granulation
- preparation of the mixture composing the second component (2) by wet granulation
- compressing these two total mixtures together into scored bilayer tablets
- coating these scored bilayer tablets with a film coating solution
21 . A process according to claim 20, wherein the said mixture composing the first component (1 ) is prepared by following these steps:
- mixing tenofovir disoproxil fumarate, emtricitabine, microcrystalline cellulose, lactose monohydrate, hydroxypropyl cellulose and one-third of croscarmellose sodium
- granulating this mixture with water
- sieving the granules and drying them
- sieving the dried granules again
- adding two-thirds of croscarmellose sodium, sodium lauryl sulfate and colloidal silicon dioxide to the sieved granules and mixing them
- adding magnesium stearate and mixing them
22. A process according to claim 20, wherein the said mixture composing the second component (2) is prepared by following these steps:
- mixing efavirenz, microcrystalline cellulose, lactose monohydrate, hydroxypropyl cellulose and croscarmellose sodium
- granulating this mixture with water and sodium lauryl sulfate
- sieving the granules and drying them
- sieving the dried granules again
- adding colloidal silicon dioxide to the sieved granules and mixing them
- adding magnesium stearate and mixing them
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP18840094.9A EP3684341A2 (en) | 2017-09-20 | 2018-09-19 | Pharmaceutical combinations of tenofovir, emtricitabine and efavirenz |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| TR2017/13954A TR201713954A2 (en) | 2017-09-20 | 2017-09-20 | PHARMACEUTICAL COMBINATIONS OF TENOFOVIR, EMTRISITABIN AND EAVIRENZINE |
| TR2017/13954 | 2017-09-20 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2019059868A2 true WO2019059868A2 (en) | 2019-03-28 |
| WO2019059868A3 WO2019059868A3 (en) | 2019-04-25 |
Family
ID=65237119
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/TR2018/050508 Ceased WO2019059868A2 (en) | 2017-09-20 | 2018-09-19 | Pharmaceutical combinations of tenofovir, emtricitabine and efavirenz |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP3684341A2 (en) |
| TR (1) | TR201713954A2 (en) |
| WO (1) | WO2019059868A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114948970A (en) * | 2022-06-07 | 2022-08-30 | 安徽贝克生物制药有限公司 | Efavirenz-containing pharmaceutical composition and preparation method thereof |
| CN119700780A (en) * | 2024-11-28 | 2025-03-28 | 安徽贝克生物制药有限公司 | A method for preparing a composite tablet |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1583542B1 (en) | 2003-01-14 | 2008-06-18 | Gilead Sciences, Inc. | Compositions and methods for combination antiviral therapy |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI471145B (en) * | 2005-06-13 | 2015-02-01 | Bristol Myers Squibb & Gilead Sciences Llc | Unitary pharmaceutical dosage form |
| TWI375560B (en) * | 2005-06-13 | 2012-11-01 | Gilead Sciences Inc | Composition comprising dry granulated emtricitabine and tenofovir df and method for making the same |
| WO2008096369A2 (en) * | 2007-02-05 | 2008-08-14 | Matrix Laboratories Limited | Pharmaceutical formulation for use in hiv therapy |
-
2017
- 2017-09-20 TR TR2017/13954A patent/TR201713954A2/en unknown
-
2018
- 2018-09-19 WO PCT/TR2018/050508 patent/WO2019059868A2/en not_active Ceased
- 2018-09-19 EP EP18840094.9A patent/EP3684341A2/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1583542B1 (en) | 2003-01-14 | 2008-06-18 | Gilead Sciences, Inc. | Compositions and methods for combination antiviral therapy |
Non-Patent Citations (1)
| Title |
|---|
| "Handbook of Pharmaceutical Excipients", pages: 405 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114948970A (en) * | 2022-06-07 | 2022-08-30 | 安徽贝克生物制药有限公司 | Efavirenz-containing pharmaceutical composition and preparation method thereof |
| CN119700780A (en) * | 2024-11-28 | 2025-03-28 | 安徽贝克生物制药有限公司 | A method for preparing a composite tablet |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3684341A2 (en) | 2020-07-29 |
| WO2019059868A3 (en) | 2019-04-25 |
| TR201713954A2 (en) | 2019-04-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10420727B2 (en) | Pharmaceutical antiretroviral composition | |
| WO2014184553A1 (en) | Pharmaceutical antiretroviral compositions | |
| JP2006516570A5 (en) | ||
| CA2866133A1 (en) | Pharmaceutical antiretroviral combinations comprising lamivudine, festinavir and nevirapine | |
| WO2018122384A1 (en) | Oral pharmaceutical compositions comprising tadalafil and dapoxetine | |
| RU2019132494A (en) | PHARMACEUTICAL TABLETS WITH ELTROMBOP OLAMIN | |
| EP3326619B1 (en) | Solid oral pharmaceutical compositions comprising tenofovir and emtricitabine | |
| EP3551171A1 (en) | Solid oral pharmaceutical compositions of ticagrelor | |
| WO2009106954A1 (en) | Stable dosage forms of lamivudine and tenofovir | |
| JP2021075530A (en) | Pharmaceutical composition | |
| WO2019059868A2 (en) | Pharmaceutical combinations of tenofovir, emtricitabine and efavirenz | |
| CA2835272A1 (en) | Pharmaceutical antiretroviral composition | |
| WO2009106960A2 (en) | Stable compositions of lamivudine, tenofovir and efavirenz | |
| JP2021075529A (en) | Pharmaceutical composition | |
| WO2009037449A1 (en) | Solid pharmaceutical compositions comprising one or more herpes virus inhibitors and one or more reverse transcriptase inhibitors | |
| WO2021091498A1 (en) | Pharmaceutical compositions comprising tenofovir and emtricitabine | |
| US20170128482A1 (en) | Modified release pharmaceutical compositions of sofosbuvir and ribavirin | |
| WO2015014737A1 (en) | Multilayer tablet formulations comprising tenofovir and entecavir | |
| EP3338767A1 (en) | Capsule compositions comprising donepezil and memantine | |
| WO2022153330A1 (en) | Pharmaceutical compositions comprising acalabrutinib | |
| WO2017029226A1 (en) | Solid pharmaceutical composition of abacavir, lamivudine, and efavirenz | |
| WO2024084496A1 (en) | Pharmaceutical compositions comprising acalabrutinib maleate | |
| EA043966B1 (en) | SOLID PHARMACEUTICAL COMPOSITION BASED ON TICAGRELOR FOR ORAL USE | |
| JP2022167880A (en) | Solid preparation | |
| JP2021075528A (en) | Pharmaceutical composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2018840094 Country of ref document: EP Effective date: 20200420 |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18840094 Country of ref document: EP Kind code of ref document: A2 |