WO2019131901A1 - ピリジルアミノ酢酸化合物を含有する医薬製剤 - Google Patents
ピリジルアミノ酢酸化合物を含有する医薬製剤 Download PDFInfo
- Publication number
- WO2019131901A1 WO2019131901A1 PCT/JP2018/048228 JP2018048228W WO2019131901A1 WO 2019131901 A1 WO2019131901 A1 WO 2019131901A1 JP 2018048228 W JP2018048228 W JP 2018048228W WO 2019131901 A1 WO2019131901 A1 WO 2019131901A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- glaucoma
- pharmaceutical preparation
- omidenepag
- ester
- ocular hypertension
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/444—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a six-membered ring with nitrogen as a ring heteroatom, e.g. amrinone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/557—Eicosanoids, e.g. leukotrienes or prostaglandins
- A61K31/5575—Eicosanoids, e.g. leukotrienes or prostaglandins having a cyclopentane, e.g. prostaglandin E2, prostaglandin F2-alpha
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/14—Esters of carboxylic acids, e.g. fatty acid monoglycerides, medium-chain triglycerides, parabens or PEG fatty acid esters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
- A61K9/0051—Ocular inserts or implants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/06—Antiglaucoma agents or miotics
Definitions
- the present invention is a pharmaceutical preparation for treating or preventing glaucoma or ocular hypertension containing Omidenepag or an ester thereof or a salt thereof as an active ingredient, wherein the other glaucoma or ocular hypertensive agents are insufficiently effective It relates to a pharmaceutical preparation to be administered to a patient.
- Glaucoma is an intractable eye disease that may cause blindness due to increased intraocular pressure due to various etiologies and damage to internal tissues of the eye (retina, optic nerve, etc.).
- intraocular pressure reduction therapy is generally used, and drug therapy, laser therapy, surgical therapy and the like are representative thereof.
- sympathomimetics nonselective stimulants such as dipivefrin, alpha 2 receptor agonists, such as brimonidine
- sympatholytic timolol, befunolol, carteolol, nipradilol, betaxolol, levobunolol, Mechipurano Beta receptor blockers such as roll (Metipranolol), alpha 1 receptor blockers such as bunazosin hydrochloride), parasympathetic agonists (such as pilocarpine), carbonic anhydrase inhibitors (such as acetazolamide), prostaglandins (such as isopropyl uno Drugs such as prostone, latanoprost, travoprost, bimatoprost, etc., Rho kinase inhibitors (lipasdil) are used.
- Omidenepag is a compound described in Patent Literature 1 and Patent Literature 2 as one of a vast number of pyridylaminoacetic acid compounds. Since these pyridylaminoacetic acid compounds have an EP2 agonist activity (Patent Document 2), it is described that they can be expected to be a glaucoma therapeutic agent because they are expected to have an intraocular pressure lowering effect (Patent Document 1). Furthermore, Patent Literatures 3 and 4 describe that the combination of Omidenepag with other glaucoma therapeutic agents such as Timolol enhances the intraocular pressure lowering action, and Patent Literature 5 discloses that Omidenepag has a specific content.
- Patent Document 6 describes that Omidenepag is useful as a therapeutic agent for diseases accompanied by elevated intraocular pressure.
- Patent Documents 7 to 9 describe specific formulations containing omidenepag as an active ingredient.
- Patent Documents 1 to 9 The entire contents of Patent Documents 1 to 9 and the documents described in the present specification are incorporated as the disclosure of the present specification.
- Omidenepag or an ester thereof or a salt thereof exhibits excellent intraocular pressure-lowering action on patients whose other glaucoma or ocular hypertensive agents are insufficiently effective.
- the present invention has been completed. Specifically, the present invention provides the following.
- a pharmaceutical preparation for treating or preventing glaucoma or ocular hypertension which comprises Omidenepag or an ester thereof or a salt thereof as an active ingredient, which is administered to a patient whose other glaucoma or ocular hypertensive agents are not effective Pharmaceutical preparations.
- the intraocular pressure is further lowered by the active ingredient to treat the glaucoma or ocular hypertension, and the glaucomatous or ocular hypertension is high.
- the pharmaceutical preparation according to the above [1] which is for treating or preventing ocular hypertension.
- a method of treating or preventing glaucoma or ocular hypertension comprising administering to a patient a pharmaceutical preparation containing Omidenepag or an ester thereof or a salt thereof as an active ingredient, said patient comprising other glaucoma or hyperoculoid The method, wherein the antihypertensive drug is an inadequately effective patient.
- the intraocular pressure is further lowered by the active ingredient to treat the glaucoma or ocular hypertension, and the glaucomatous or ocular hypertension is high.
- a method for treating or preventing glaucoma or ocular hypertension comprising administering to a patient a pharmaceutical preparation comprising Omidenepag or an ester or salt thereof as an active ingredient, wherein the glaucoma is Omidenepag or an ester thereof Or a glaucoma treatment resistant to glaucoma treatment with other active ingredients other than their salts, and the ocular hypertension is a hypertensive disease resistant to ocular hypertension treatment with the other active ingredients.
- a method for treating or preventing glaucoma or ocular hypertension comprising administering to a patient a pharmaceutical preparation containing Omidenepag or an ester thereof or a salt thereof as an active ingredient, which comprises the following steps (1) Omidenepag or an ester thereof Or a first treatment step of administering to the patient other glaucoma or ocular hypertension treatments other than their salts, (2) determining whether the first treatment step has insufficient treatment or insufficient preventive effect; (3) A second treatment wherein a pharmaceutical preparation containing Omidenepag or an ester thereof or a salt thereof as an active ingredient is further administered to a patient when the first treatment step has insufficient treatment or insufficient preventive effect Process, Method including. [20] Use of the pharmaceutical preparation according to any one of the above [1] to [9] in the manufacture of a medicament for treating or preventing glaucoma or ocular hypertension.
- each structure of said [1] to [20] can select 2 or more arbitrarily, and can combine them.
- Omidenepag or an ester thereof or a salt thereof exhibits excellent intraocular pressure-lowering action on patients whose other glaucoma or ocular hypertensive agents are ineffective.
- Omidenepag or an ester thereof or a salt thereof is useful as a pharmaceutical preparation for treating or preventing glaucoma or ocular hypertension, even for patients who are not effective for other glaucoma or ocular hypertensive agents.
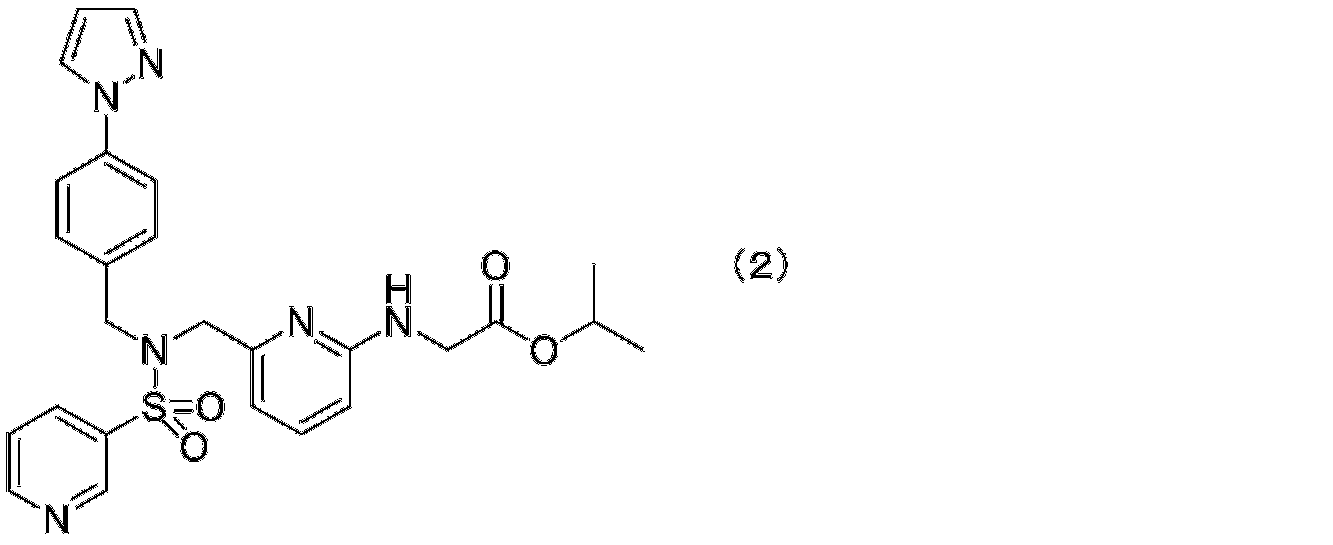
- Omidenepag contained in the pharmaceutical preparation of the present invention has the following formula (1): (CAS registration number; 1187451-41-7), (6- ⁇ [4- (pyrazol-1-yl) benzyl] (pyridin-3-ylsulfonyl) aminomethyl ⁇ pyridine-2- Also known as ylamino) acetic acid.
- the ester of Omidenepag contained in the pharmaceutical preparation of the present invention is preferably an ester formed by dehydration condensation of the carboxyl group of Omidenepag with a monohydric alcohol having 1 to 6 carbon atoms, and more preferably the carboxyl group of Omidenepag is An ester formed by dehydration condensation with a C 2 to C 5, more preferably C 3 to C 4 monohydric alcohol is preferred.
- esters include methyl ester, ethyl ester, n-propyl ester, isopropyl ester, n-butyl ester, isobutyl ester, sec-butyl ester, tert-butyl ester, n-pentyl ester or n-hexyl ester
- ethyl ester, n-propyl ester and isopropyl ester are mentioned, and more preferably isopropyl ester is mentioned.
- isopropyl ester of Omidenepag is represented by the following formula (2): A compound represented by the formula (CAS Registry Number; 1187451-19-9), and Omidenepag isopropyl or (6- ⁇ [4- (pyrazol-1-yl) benzyl] (pyridin-3-ylsulfonyl) aminomethyl ⁇ pyridine -2-ylamino) It is also called isopropyl acetate.
- the salt of omidenepag or the salt of ester of omidenepag contained in the pharmaceutical preparation of the present invention is not particularly limited as long as it is a pharmacologically acceptable salt.
- inorganic acid salts such as hydrochloride, hydrobromide, hydroiodide, nitrate, sulfate or phosphate; acetate, trifluoroacetate, benzoate, oxalic acid Salt, malonate, succinate, maleate, fumarate, tartrate, citrate, methanesulfonate, ethanesulfonate, trifluoromethanesulfonate, benzenesulfonate, p-toluenesulfone Acid salts, organic acid salts such as glutamate or aspartate; metal salts such as sodium salts, potassium salts, calcium salts or magnesium salts; inorganic salts such as ammonium salts; or organic amine salts such as triethy
- Omidenepag or an ester thereof or a salt thereof contained in the pharmaceutical preparation of the present invention is disclosed in U.S. Patent Application Publication No. 2012/0190852 (Patent Document 1) and U.S. Patent Application Publication No. 2011/0054172 (Patent Document) It can manufacture according to the method as described in 2), the normal method in the said technical field, etc.
- the content of omidenepag or an ester thereof or a salt thereof contained in the pharmaceutical preparation of the present invention, types and amounts of additives, preferable forms such as administration forms, etc. are as described in these documents Can be adopted.
- omidenepag or an ester or a salt thereof used in the present application includes (1) Omidenepag, (2) ester of Omidenepag, (3) salt of Omidenepag, and (4) salt of ester of Omidenepag It is an included meaning.
- the content of omidenepag or its ester or a salt thereof contained in the pharmaceutical preparation of the present invention is not particularly limited, and depending on the administration form, the lower limit thereof is, for example, 0.000001% (w / v) Preferably it is 0.00001% (w / v), more preferably 0.00003% (w / v), 0.0001% (w / v), 0.001 (w / v), 0.. 01% (w / v), 0.1% (w / v) or 1% (w / v).
- the upper limit of the content is, for example, 30% (w / v), 25% (w / v), 20% (w / v), 15% (w / v) or 12% (w / v) Also, 0.03% (w / v), 0.01% (w / v), 0.005% (w / v), 0.003% (w / v) or 0.0027% (w / v) w / v). More specifically, the content may be a range combining any of the above lower limit and the upper limit, but is, for example, 0.000001 to 30% (w / v), preferably 0.00001 to 25.
- % (w / v) means the mass (g) of the active ingredient (omidenepag or its ester or a salt thereof) and additives (surfactant etc.) contained in 100 mL of the pharmaceutical preparation.
- Omidenepag 0.01% (w / v) means that the content of Omidenepag contained in 100 mL of the pharmaceutical preparation is 0.01 g.
- the lower limit of the content of omidenepag or its ester or a salt thereof contained in the pharmaceutical preparation of the present invention is preferably 0.0003% (w / v), preferably 0.001% (W / v) is more preferable, 0.0013% (w / v) is more preferable, and 0.0015% (w / v) is particularly preferable.
- the upper limit of the content is preferably 0.03% (w / v), more preferably 0.01% (w / v), still more preferably 0.005% (w / v), and 0.003 % (W / v) is particularly preferred and 0.0027% (w / v) is particularly preferred.
- the content may be a range combining any of the above lower limit and the upper limit, 0.0003 to 0.03% (w / v) is preferable, and 0.001 to 0.01 is preferable.
- % (W / v) is more preferable, 0.001 to 0.005% (w / v) is more preferable, and 0.001 to 0.003% (w / v) is particularly preferable, and 0.0013 to 0. 003% (w / v) is particularly preferred and 0.0015 to 0.0027% (w / v) is particularly preferred.
- the lower limit of the content of omidenepag or its ester or a salt thereof contained in the pharmaceutical preparation of the present invention is preferably 0.000001% (w / v), and 0. 000003 (w / v) is more preferable, 0.000005% (w / v) is more preferable, 0.00001% (w / v) is particularly preferable, and 0.00003% (w / v) is particularly preferable.
- the upper limit of the content is preferably 30% (w / v), more preferably 10% (w / v), still more preferably 1% (w / v), and 0.1% (w / v) Is particularly preferred, 0.01% (w / v) being particularly preferred. More specifically, the content may be a range combining any of the above lower limit and the upper limit, but 0.000001 to 30% (w / v) is preferable, 0.000003 to 10% (w / v) v) is more preferable, 0.000005 to 1% (w / v) is more preferable, 0.00001 to 0.1% (w / v) is particularly preferable, 0.00003 to 0.01% (w / v) Is particularly preferred.
- the said content is a preferable example of the ophthalmic injection, it is not restricted to the ophthalmic injection.
- the pharmaceutical preparation of this invention contains the salt of Omidenepag or its ester, it means that content of Omidenepag or its ester at the time of salt liberation will become said range.
- Additives can be used in the pharmaceutical preparation of the present invention as required.
- a surfactant for example, a buffer, a tonicity agent, a stabilizer, an antiseptic, an antioxidant, a thickening agent, a base, a pH adjuster and the like can be added.
- a surfactant which can be used as an additive of a medicine can be appropriately blended.
- surfactants include polyoxyethylene castor oil, polyoxyethylene hydrogenated castor oil, polyoxyethylene sorbitan fatty acid ester, vitamin E TPGS, polyoxyethylene fatty acid ester, polyoxyethylene polyoxypropylene glycol, sucrose fatty acid ester Etc.
- polyoxyethylene castor oil various polyoxyethylene castor oils having different polymerization numbers of ethylene oxide can be used, and the polymerization number of ethylene oxide is preferably 5 to 100, and 20 to 50 More preferably, 30 to 40 are particularly preferred, and 35 is most preferred.
- polyoxyethylene castor oil examples include polyoxyl 5 castor oil, polyoxyl 9 castor oil, polyoxyl 15 castor oil, polyoxyl 35 castor oil, polyoxyl 40 castor oil and the like, with polyoxyl 35 castor oil being most preferable.
- polyoxyethylene hydrogenated castor oil various polyoxyethylene hydrogenated castor oils having different polymerization numbers of ethylene oxide can be used.
- the polymerization number of ethylene oxide is preferably 10 to 100, more preferably 20 to 80, and 40 -70 are particularly preferred, 60 is most preferred.
- Specific examples of polyoxyethylene hydrogenated castor oil include polyoxyethylene hydrogenated castor oil 10, polyoxyethylene hydrogenated castor oil 40, polyoxyethylene hydrogenated castor oil 50, polyoxyethylene hydrogenated castor oil 60, etc.
- Ethylene hydrogenated castor oil 60 is most preferred.
- polyoxyethylene sorbitan fatty acid ester examples include polysorbate 80, polysorbate 60, polysorbate 40, polyoxyethylene sorbitan monolaurate, polyoxyethylene sorbitan trioleate, polysorbate 65 and the like, and polysorbate 80 is most preferable.
- Vitamin E TPGS is also referred to as tocopherol polyethylene glycol 1000 succinate.
- polyoxyethylene fatty acid esters examples include polyoxyl 40 stearate and the like.
- polyoxyethylene polyoxypropylene glycol examples include polyoxyethylene (160) polyoxypropylene (30) glycol, polyoxyethylene (42) polyoxypropylene (67) glycol, polyoxyethylene (54) polyoxypropylene (39) Examples thereof include glycol, polyoxyethylene (196) polyoxypropylene (67) glycol, and polyoxyethylene (20) polyoxypropylene (20) glycol.
- sucrose fatty acid ester sucrose stearic acid ester etc. are mentioned.
- the lower limit is preferably 0.001% (w / v), more preferably 0.01% (w / v), still more preferably 0.1% (w / v), and still more preferably 0.5% (w) / V) is particularly preferred, 0.8% (w / v) being most preferred.
- the upper limit is preferably 10% (w / v), more preferably 5% (w / v), still more preferably 4% (w / v), particularly preferably 3% (w / v), 2% (w / v) / V) is most preferred.
- the content may be a range combining any of the above lower limit and the upper limit, but 0.001 to 10% (w / v) is preferable, and 0.01 to 5% (w / v) Is more preferable, 0.03 to 4% (w / v) is more preferable, 0.05 to 3% (w / v) is particularly preferable, and 0.1 to 2% (w / v) is most preferable.
- a buffer that can be used as a pharmaceutical additive can be appropriately blended.
- the buffer include phosphoric acid or a salt thereof, boric acid or a salt thereof, citric acid or a salt thereof, acetic acid or a salt thereof, carbonic acid or a salt thereof, tartaric acid or a salt thereof, ⁇ -aminocaproic acid, trometamol etc. .
- examples of phosphate include sodium phosphate, sodium dihydrogen phosphate, disodium hydrogen phosphate, potassium phosphate, potassium dihydrogen phosphate, dipotassium hydrogen phosphate and the like, and boric acid
- examples of salts include borax, sodium borate, potassium borate and the like
- examples of citric acid or a salt thereof include citric acid monohydrate, sodium citrate, disodium citrate, trisodium citrate and the like
- examples of the acetate include sodium acetate and potassium acetate
- examples of the carbonate include sodium carbonate and sodium hydrogencarbonate.
- examples of the tartrate include sodium tartrate and potassium tartrate. Among them, boric acid or a salt thereof, or citric acid or a salt thereof is preferable.
- a buffer When a buffer is added to the pharmaceutical preparation of the present invention, its content can be appropriately adjusted depending on the type of buffer etc., but it is preferably 0.001 to 10% (w / v), preferably 0.01 to 10%. 5% (w / v) is more preferable, 0.05 to 3% (w / v) is more preferable, and 0.1 to 2% (w / v) is most preferable.
- an isotonicity agent which can be used as an additive of a medicine can be appropriately blended.
- tonicity agents include ionic tonicity agents and nonionic tonicity agents.
- examples of the ionic tonicity agent include sodium chloride, potassium chloride, calcium chloride, magnesium chloride and the like, and examples of the nonionic tonicity agent include glycerin, propylene glycol, sorbitol, mannitol and the like.
- a tonicity agent When a tonicity agent is added to the pharmaceutical preparation of the present invention, its content can be appropriately adjusted depending on the type of tonicity agent etc., but preferably 0.01 to 10% (w / v), 0.02 to 7% (w / v) is more preferable, 0.1 to 5% (w / v) is more preferable, 0.5 to 4% (w / v) is particularly preferable, and 0.8 to 3 % (W / v) is most preferred.
- a stabilizer which can be used as an additive of a medicine can be appropriately blended.
- the stabilizer include edetic acid, monosodium edetate, disodium edetate, tetrasodium edetate, sodium citrate and the like, with disodium edetate being particularly preferred.
- Edetate sodium may be a hydrate.
- the content thereof can be appropriately adjusted depending on the kind of the stabilizer etc., but 0.001 to 1% (w / v) is preferable, 0. 005 to 0.5% (w / v) is more preferable, and 0.01 to 0.1% (w / v) is most preferable.
- an antiseptic that can be used as an additive of a medicine can be appropriately blended.
- preservatives include benzalkonium chloride, benzalkonium bromide, benzethonium chloride, sorbic acid, potassium sorbate, methyl parahydroxybenzoate, propyl parahydroxybenzoate, chlorobutanol and the like.
- the content thereof can be appropriately adjusted according to the type of preservative, etc., but 0.0001 to 1% (w / v) is preferable, and 0.0005 to 0.1% (w / v) is more preferable, 0.001 to 0.05% (w / v) is more preferable, and 0.005 to 0.010% (w / v) is most preferable. Moreover, also when not containing a preservative, it is preferable.
- an antioxidant which can be used as an additive of a medicine can be appropriately blended.
- the antioxidant include ascorbic acid, tocophenol, dibutylhydroxytoluene, butylhydroxyanisole, sodium erythorbate, propyl gallate, sodium sulfite and the like.
- the content thereof can be appropriately adjusted depending on the type of antioxidant etc., but 0.0001 to 1% (w / v) is preferable, 0. 0005 to 0.1% (w / v) is more preferable, and 0.001 to 0.05% (w / v) is most preferable.
- a thickening agent which can be used as an additive of a medicine can be appropriately blended.
- thickening agents include methylcellulose, ethylcellulose, hydroxymethylcellulose, hydroxyethylcellulose, hydroxypropylcellulose, hydroxyethylmethylcellulose, hydroxypropylmethylcellulose, carboxymethylcellulose, carboxymethylcellulose sodium, hydroxypropyl methylcellulose acetate succinate, hydroxypropyl methylcellulose phthalate, Carboxymethyl ethyl cellulose, cellulose acetate phthalate, polyvinyl pyrrolidone, polyvinyl alcohol, carboxyvinyl polymer, polyethylene glycol and the like can be mentioned.
- a thickener When a thickener is added to the pharmaceutical preparation of the present invention, its content can be appropriately adjusted depending on the type of thickener, etc., but preferably 0.001 to 5% (w / v), 0.01 to 1% (w / v) is more preferable, and 0.1 to 0.5% (w / v) is most preferable.
- a base that can be used as a pharmaceutical additive can be appropriately blended.
- the base include water, physiological saline, dimethyl sulfoxide, polyethylene glycol such as PEG 400, tributyl citrate, acetyl tributyl citrate, benzyl benzoate, white petrolatum, liquid paraffin, etc., water, physiological saline Water, dimethyl sulfoxide, PEG 400 is preferred.
- the pH of the pharmaceutical preparation of the present invention is preferably 4.0 to 8.0, more preferably 4.5 to 7.5, particularly preferably 5.0 to 7.0, and most preferably 5.5 to 6.1. preferable. Also, the pH may be 6.0 to 8.0.
- hydrochloric acid, phosphoric acid, citric acid, acetic acid, sodium hydroxide, potassium hydroxide or the like may be added as a pH adjuster for adjusting the pH.
- the pharmaceutical preparation according to the present invention exhibits excellent intraocular pressure-lowering action even on patients whose other glaucoma or ocular hypertensive agents are efficacious, and therefore a glaucoma therapeutic or prophylactic agent and / or hypertensive disorder It is useful as a therapeutic or prophylactic agent and / or an intraocular pressure lowering agent.
- Glaucoma in the present invention includes primary open angle glaucoma, secondary open angle glaucoma, normal tension glaucoma, aqueous humor hyperglaucoma, primary closed angle glaucoma, secondary closed angle glaucoma, plateau iris glaucoma, mixed glaucoma, Developmental glaucoma, steroid glaucoma, exfoliation glaucoma, amyloid glaucoma, neovascular glaucoma, malignant glaucoma, capsular glaucoma of the lens, plateau iris syndrome etc., preferably primary open angle glaucoma, normal pressure glaucoma, primary closed angle glaucoma Particularly, the pharmaceutical preparation of the present invention is effective for primary open angle glaucoma.
- the pharmaceutical formulations of the present invention are preferably administered to patients whose other glaucoma or ocular hypertensive agents are ineffective.
- the other glaucoma or ocular hypertensive agents refer to any of glaucoma or ocular hypertensive agents containing an active ingredient (other active ingredients) other than omidenepag or its ester or a salt thereof, omidenepag or Specifically, the active ingredients other than the ester or their salts include nonselective sympathetic agonists, alpha 2 receptor agonists, alpha 1 receptor blockers, beta receptor blockers, parasympathetic agonists , Carbonic anhydrase inhibitors, prostaglandins, Rho kinase inhibitors and the like.
- nonselective sympathetic agonists include dipivefrine
- specific examples of ⁇ 2 receptor agonists include brimonidine and apraclonidine
- specific examples of ⁇ 1 receptor blockers include bunazosin
- beta receptor blockers include timolol, befnolol, carteolol, niprazilol, betaxolol, levobnolol, and methipranol
- specific examples of parasympathomimetics include pilocarpine
- carbonate Specific examples of dehydration enzyme inhibitors include dorzolamide, brinzolamide, acetazolamide
- specific examples of prostaglandins include latanoprost, isopropyl unoprostone, bimatoprost, travoprost, and specific examples of the Rho kinase inhibitor
- Fasudil cited Among them, prostaglandins are preferable, prostaglandin F 2 ⁇ derivatives are more preferable,
- the drop rate of intraocular pressure after treatment to the pre-treatment intraocular pressure is 18% or less, preferably 17% or less, more preferably 16% or less, still more preferably 15% or less, more preferably 14% or less, particularly preferably 13% or less, particularly preferably Is preferably a patient who is 12% or less, most preferably 10% or less.
- pretreatment intraocular pressure (mmHg)-post-treatment intraocular pressure ( ) is 4.5 mmHg or less, preferably 4.2 mmHg or less, more preferably 4 mmHg or less, more preferably 3.7 mmHg or less, more preferably 3.5 mmHg or less, particularly preferably 3.2 mmHg or less, particularly preferably It is also preferred that the patient is 3 mmHg or less, most preferably 2.5 mmHg or less.
- the treatment with this other glaucoma or ocular hypertensive agent is usually administered 1 to 3 drops at a time, 1 to 3 times a day, for 1 week or more, preferably 1 week or more, preferably 2 weeks or more, more preferably 4 weeks or more More preferably, it is carried out for a period of 2 months or more, particularly preferably 6 months or more, and most preferably 1 year or more.
- patients whose other glaucoma or ocular hypertensive agents are ineffective include patients who can not use or can not treat other glaucoma or ocular hypertensive agents due to side effects or the like.
- the patients targeted by the pharmaceutical preparation of the present invention are domestic animals such as cows and pigs, mammals including rabbits, monkeys, dogs, cats, and humans, preferably humans.
- the intraocular pressure of patients treated with other glaucoma or ocular hypertensive agents is at least 5%, for example 6% or more, preferably 7% or more, more preferably 8% or more. More preferably, it is 9% or more, more preferably 10% or more, particularly preferably 11% or more, particularly preferably 12% or more, most preferably 13% or more.
- the upper limit value of the drop rate of the intraocular pressure by the pharmaceutical preparation of the present invention is, for example, 40% or less, preferably 35% or less, more preferably 30% or less, still more preferably 28% or less, more preferably 26% or less, particularly preferably Preferably 24% or less, particularly preferably 22% or less, and most preferably 20% or less can be mentioned, and a range in which the above lower limit value and the upper limit value are appropriately combined can be selected appropriately.
- the drop rate of the intraocular pressure by the pharmaceutical preparation of the present invention is, for example, 5 to 40%, preferably 7 to 35%, more preferably 9 to 30%.
- the drop width of the intraocular pressure (the amount of change in intraocular pressure) after the treatment to the intraocular pressure before treatment with the pharmaceutical preparation of the present invention is at least 1.0 mmHg or more, preferably 1.2 mmHg or more It is more preferably 1.4 mmHg or more, still more preferably 1.6 mmHg or more, more preferably 1.8 mmHg or more, particularly preferably 2.0 mmHg or more, particularly preferably 2.5 mmHg or more, most preferably 2.9 mmHg or more. Is preferred.
- the upper limit value of the amount of change in intraocular pressure by the pharmaceutical preparation of the present invention is, for example, 10.0 mmHg or less, preferably 8.0 mmHg or less, more preferably 6.0 mmHg or less, still more preferably 5.5 mmHg or less, more preferably 5 .0 mmHg or less, particularly preferably 4.5 mmHg or less, particularly preferably 4.0 mmHg or less, most preferably 3.2 mmHg or less, and a range in which the above lower limit value and the upper limit value are combined appropriately can be selected as appropriate.
- the amount of change in intraocular pressure by the pharmaceutical preparation of the present invention is, for example, 1.0 to 10.0 mmHg, preferably 1.4 to 8.0 mmHg, and more preferably 1.8 to 6.0 mmHg.
- the “drop rate of intraocular pressure after treatment with the pharmaceutical preparation of the present invention after treatment (pressure drop rate)” and “fall width (amount of change in intraocular pressure)” defined here will be described later.
- [Method of administration] means the fall rate and fall width of intraocular pressure in the second treatment step as shown in (3) of the above, and the above-mentioned first treatment step shown in (1) of [Method of administration] Does not include intraocular pressure drop rates and changes in intraocular pressure due to treatment with other glaucoma or ocular hypertension treatments. Therefore, the pharmaceutical preparation of the present invention containing Omidenepag or the like as an active ingredient can be added to the therapeutic effect of other glaucoma or ocular hypertension therapeutic agents containing latanoprost or the like as an active ingredient to lower intraocular pressure.
- the pharmaceutical preparation of the present invention is used in combination with one or more, preferably one to three, more preferably one or two additional glaucoma or ocular hypertensive agents to lower intraocular pressure more strongly (eg They may be combined as a kit) and may contain additional active ingredients.
- additional glaucoma or ocular hypertensive agent e.g., commercially available or under development glaucoma or ocular hypertensive agents and the like are preferable, and commercially available glaucoma or ocular hypertensive agents and the like are more preferable
- nonselective sympathetic agonists alpha 2 receptor agonists, alpha 1 receptor blockers, beta receptor blockers, parasympathetic agonists, carbonic anhydrase inhibitors, prostaglandins
- examples thereof include glaucoma or ocular hypertension therapeutic agents containing a Rho kinase inhibitor and the like as an active ingredient.
- the additional active ingredients are specifically nonselective sympathetic agonists, alpha 2 receptor agonists, alpha 1 receptor blockers, beta receptor blockers, parasympathetic agonists, carbonic anhydrase inhibitors, Prostaglandins, Rho kinase inhibitors and the like can be mentioned.
- the intraocular pressure before treatment with the pharmaceutical preparation of the present invention described above is the effect of the additional glaucoma or hyperotensive agent or the additional active ingredient. It means the excluded part.
- nonselective sympathetic agonists include dipivefrine
- specific examples of ⁇ 2 receptor agonists include brimonidine and apraclonidine
- specific examples of ⁇ 1 receptor blockers include bunazosin
- beta receptor blockers include timolol, befnolol, carteolol, niprazilol, betaxolol, levobnolol, and methipranol
- specific examples of parasympathomimetics include pilocarpine
- carbonate Specific examples of dehydration enzyme inhibitors include dorzolamide, brinzolamide, acetazolamide
- specific examples of prostaglandins include isopropyl unoprostone, bimatoprost, travoprost, and specific examples of Rho kinase inhibitors Are Fasudil.
- the pharmaceutical preparation of the present invention can be administered orally or parenterally, for example, eye drop administration, intravitreal administration, intraconjunctival administration, intracameral administration, subconjunctival administration, subtenon administration or punctum plug administration It may be
- the dosage form of the pharmaceutical preparation of the present invention includes eye drops, eye ointments, injections, punctum plugs, tablets, capsules, granules, powders and the like, and in particular, eye drops, ophthalmic injections, punctum plugs Is preferred.
- Ophthalmic injections include injections for intravitreal administration, administration in the anterior chamber, administration in the conjunctival pouch, administration in the anterior chamber, administration under the conjunctiva or subtenon administration.
- the dosage form of the pharmaceutical preparation of the present invention can be manufactured according to the usual method in the technical field of medicine.
- oral agents such as tablets, capsules, granules, powders, etc., as well as extenders such as lactose, crystalline cellulose, starch, vegetable oil, lubricants such as magnesium stearate, talc, hydroxypropyl cellulose,
- Use binders such as polyvinyl pyrrolidone, disintegrants such as calcium carboxymethyl cellulose and low substituted hydroxypropyl methyl cellulose, coating agents such as hydroxypropyl methyl cellulose, macrogol, silicone resin, and coating agents such as gelatin film as needed , Can be formulated.
- the pharmaceutical preparation of the present invention can be stored in containers made of various materials.
- a container made of polyethylene, polypropylene or the like can be used, and when used as an eye drop, a container made of polyethylene in terms of ease of eye drop (hardness of container), stability of the present compound, etc. It is preferable to put in and save.
- the dosage form of the pharmaceutical preparation of the present invention is not particularly limited as long as it is a dosage and dosage sufficient to exert the desired medicinal effects, and it depends on the symptom of the disease, the age and body weight of the patient, the dosage form of the pharmaceutical preparation, etc. It can be selected appropriately. Specifically, in the case of eye drops, 1 to 5 drops, preferably 1 to 3 drops, more preferably 1 to 2 drops, particularly preferably 1 drop, per dose 1 to 4 times a day, preferably 1 drop. One to three times a day, more preferably once or twice a day, particularly preferably once a day, can be administered by eye drop daily to weekly. It is preferred to administer one drop daily once daily.
- one drop is usually about 0.01 to about 0.1 mL, preferably about 0.015 to about 0.07 mL, more preferably about 0.02 to about 0.05 mL. And particularly preferably about 0.03 mL.
- the dose of the drug is preferably 0.0001 to 30000 ⁇ g / eye, more preferably 0.0005 to 10000 ⁇ g / eye, and most preferably 0.001 to 5000 ⁇ g / eye.
- the administration interval is not particularly limited as long as it is sufficient to produce the desired effect, but once a week to once every three years It is preferably administered at intervals, once a week, once every two weeks, once a month, once every two months, once every three months, once every four months, once every five months More preferably, once every six months, once a year, once every two years or once every three years, once every two months, once every three months, every four months Most preferably, it is administered once, once every 5 months or once every 6 months. Also, the administration interval can be changed as appropriate.
- 0.01 to 5000 mg, preferably 0.1 to 1000 mg can be divided and administered in one to several times (2 to 5 times, preferably 2 to 3 times) per day.
- the administration method of the pharmaceutical preparation of the present invention is (1) a first treatment step of treating or preventing glaucoma or ocular hypertension with the above other active ingredients other than omidenepag or its ester or a salt thereof, (2) an optional step of judging whether the first treatment step has insufficient treatment or insufficient preventive effect, (3) a second treatment step of treating or preventing glaucoma or ocular hypertension by administering a pharmaceutical preparation containing Omidenepag or an ester thereof or a salt thereof as an active ingredient, following the first treatment step; including.
- the pharmaceutical preparation of the invention By administering the pharmaceutical preparation of the present invention to a patient based on the administration method as described above, regardless of the effect of treatment or prevention with the other active ingredient, or when the effect is insufficient
- the pharmaceutical preparation of the invention additionally provides an intraocular pressure-lowering effect, etc., and can further treat or prevent glaucoma or ocular hypertension.
- the above-mentioned administration method is as follows. First, the above-mentioned other glaucoma or ocular hypertensive therapeutic agent (other active ingredient) other than Omidenepag of the present invention or an ester or a salt thereof of the present invention is administered Treat or prevent ocular hypertension. After that, when it is predicted that the other active ingredient has insufficient therapeutic effect or insufficient prophylactic effect, “glaucoma resistant to glaucoma treatment with the other active ingredient” or “high with the other active ingredient” It is determined that the treatment is "Ocular hypertension resistant to treatment for ocular hypertension", and subsequently, Omidenepag of the present invention or an ester thereof or a salt thereof is administered.
- insufficient therapeutic effect and “probable in preventing effect” means that the above-mentioned “treatment with other glaucoma or ocular hypertensive agents does not have sufficient effect.
- the pharmaceutical preparation of the present invention is used as a secondary administration means. Is appropriate.
- the pharmaceutical preparation of the present invention is sufficiently treated or treated with the other active ingredient, in addition to the case where the treatment of glaucoma or ocular hypertension with the above-mentioned other active ingredient such as latanoprost is insufficient. Even if it was thought that the prevention had been completed, it had an unexpected effect that the treatment or prevention could be completed further, and it would solve the problem different from the conventional one. is there.
- the pharmaceutical preparation of the present invention can be expected to have an effect on the requirement for lowering the intraocular pressure or the prevention of a re-elevation (rebound) of the intraocular pressure.
- the Omidenepag of the present invention or an ester or salt thereof can be used for the preparation of a pharmaceutical preparation as described above.
- the following are representative preparation examples of the pharmaceutical preparation of the present invention.
- the compounding quantity of each component is a content in 100 mL of formulation in the following formulation example.
- desired types of pharmaceutical preparations can be obtained by appropriately adjusting the types and / or amounts of the Omidenepag and / or additives.
- 0.001g, 0.0011g, 0.0012g, 0.0013g, 0.0014g, 0.0015g, 0.0016g, 0.0017g, 0.0018g of the combination amount of Omidenepag of the said Preparation Examples 1-5 0.0019 g, 0.0021 g, 0.0022 g, 0.0023 g, 0.0024 g, 0.0025 g, 0.0026 g, 0.0027 g, 0.0028 g, 0.0029 g, 0.003 g, which is desired.
- Omidenepag isopropyl, polyoxyl 35 castor oil, glycerin, citric acid, sodium citrate, sodium edetate, benzalkonium chloride are dissolved in purified water, and after adjusting the pH, purified water is added to adjust the total volume, 0.002% (w / v) Omidenepag isopropyl eye drop A was prepared.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Ophthalmology & Optometry (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
さらに、特許文献3及び4には、オミデネパグにチモロール等の他の緑内障治療薬を組み合わせることで眼圧下降作用が増強されることが記載され、特許文献5には、オミデネパグが特定の含有量のときに特に優れた眼圧下降作用を示すことが記載され、特許文献6には、オミデネパグが高度に上昇した眼圧を伴う疾患の治療剤として有用であることが記載されている。
また、特許文献7~9には、オミデネパグを有効成分として含有する特定の製剤が記載されている。
オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する緑内障若しくは高眼圧症を治療または予防するための医薬製剤であって、他の緑内障または高眼圧症治療薬が効果不十分な患者に投与される医薬製剤。
〔2〕
前記緑内障若しくは高眼圧症を治療または予防が、前記他の緑内障または高眼圧症治療薬によって緑内障若しくは高眼圧症の治療または予防を行った後、さらに前記有効成分によって眼圧を下げて緑内障若しくは高眼圧症を治療または予防するものである、前記〔1〕に記載の医薬製剤。
〔3〕
前記他の緑内障または高眼圧症治療薬の有効成分がプロスタグランジンF2α誘導体である、前記〔1〕又は〔2〕に記載の医薬製剤。
〔4〕
前記他の緑内障または高眼圧症治療薬有効成分がラタノプロストである、前記〔1〕~〔3〕のいずれか一項に記載の医薬製剤。
〔5〕
オミデネパグ若しくはそのエステル又はそれらの塩の含有量が、0.001~0.003%(w/v)である、前記〔1〕~〔4〕のいずれか一項に記載の医薬製剤。
〔6〕
オミデネパグ若しくはそのエステル又はそれらの塩の含有量が、0.002%(w/v)である、前記〔1〕~〔5〕のいずれか一項に記載の医薬製剤。
〔7〕
オミデネパグ若しくはそのエステル又はそれらの塩が、オミデネパグ イソプロピルである、前記〔1〕~〔6〕のいずれか一項に記載の医薬製剤。
〔8〕
点眼剤である、前記〔1〕~〔7〕のいずれか一項に記載の医薬製剤。
〔9〕
オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する緑内障若しくは高眼圧症を治療または予防するための医薬製剤であって、前記緑内障が、オミデネパグ若しくはそのエステル又はそれらの塩以外の他の有効成分による緑内障治療に抵抗性の緑内障であり、前記高眼圧症が、前記他の有効成分による高眼圧症治療に抵抗性の高眼圧症である、医薬製剤。
〔10〕
オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を患者に投与することを含む、緑内障若しくは高眼圧症を治療または予防する方法であって、前記患者が、他の緑内障または高眼圧症治療薬が効果不十分な患者である、方法。
〔11〕
前記緑内障若しくは高眼圧症を治療または予防が、前記他の緑内障または高眼圧症治療薬によって緑内障若しくは高眼圧症の治療または予防を行った後、さらに前記有効成分によって眼圧を下げて緑内障若しくは高眼圧症を治療または予防するものである、前記〔10〕に記載の方法。
〔12〕
前記他の緑内障または高眼圧症治療薬の有効成分がプロスタグランジンF2α誘導体である、前記〔10〕又は〔11〕に記載の方法。
〔13〕
前記他の緑内障または高眼圧症治療薬有効成分がラタノプロストである、前記〔10〕~〔12〕のいずれか一項に記載の方法。
〔14〕
前記医薬製剤中におけるオミデネパグ若しくはそのエステル又はそれらの塩の含有量が、0.001~0.003%(w/v)である、前記〔10〕~〔13〕のいずれか一項に記載の方法。
〔15〕
前記医薬製剤中におけるオミデネパグ若しくはそのエステル又はそれらの塩の含有量が、0.002%(w/v)である、前記〔10〕~〔14〕のいずれか一項に記載の方法。
〔16〕
前記オミデネパグ若しくはそのエステル又はそれらの塩が、オミデネパグ イソプロピルである、前記〔10〕~〔15〕のいずれか一項に記載の方法。
〔17〕
前記投与が点眼投与である、前記〔10〕~〔16〕のいずれか一項に記載の方法。
〔18〕
オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を患者に投与することを含む、緑内障若しくは高眼圧症を治療または予防するための方法であって、前記緑内障が、オミデネパグ若しくはそのエステル又はそれらの塩以外の他の有効成分による緑内障治療に抵抗性の緑内障であり、前記高眼圧症が、前記他の有効成分による高眼圧症治療に抵抗性の高眼圧症である、方法。
〔19〕
オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を患者に投与することを含む、緑内障若しくは高眼圧症を治療または予防する方法であって、以下の工程
(1)オミデネパグ若しくはそのエステル又はそれらの塩以外の他の緑内障または高眼圧症治療薬を患者に投与する第一の処置工程、
(2)上記第一の処置工程では治療が不十分若しくは予防効果が不十分であるか否か判断する工程、
(3)上記第一の処置工程では治療が不十分若しくは予防効果が不十分である場合、オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を患者にさらに投与する第二の処置工程、
を含む方法。
〔20〕
緑内障若しくは高眼圧症を治療または予防するための薬剤の製造における、前記〔1〕~〔9〕のいずれか一項に記載の医薬製剤の使用。
本発明の医薬製剤に含有されるオミデネパグは、下記式(1):
で表される化合物(CAS登録番号;1187451-41-7)であり、(6-{[4-(ピラゾール-1-イル)ベンジル](ピリジン-3-イルスルホニル)アミノメチル}ピリジン-2-イルアミノ)酢酸ともいう。
で表される化合物(CAS登録番号;1187451-19-9)であり、オミデネパグ イソプロピル又は(6-{[4-(ピラゾール-1-イル)ベンジル](ピリジン-3-イルスルホニル)アミノメチル}ピリジン-2-イルアミノ)酢酸イソプロピルともいう。
本発明の医薬製剤には、必要に応じて添加剤を用いることができる。添加剤としては、例えば、界面活性剤、緩衝剤、等張化剤、安定化剤、防腐剤、抗酸化剤、粘稠化剤、基剤、pH調整剤等を加えることができる。
界面活性剤の例としては、ポリオキシエチレンヒマシ油、ポリオキシエチレン硬化ヒマシ油、ポリオキシエチレンソルビタン脂肪酸エステル、ビタミンE TPGS、ポリオキシエチレン脂肪酸エステル、ポリオキシエチレンポリオキシプロピレングリコール、ショ糖脂肪酸エステル等が挙げられる。
より具体的には、ポリオキシエチレンヒマシ油としては、酸化エチレンの重合数の異なる種々のポリオキシエチレンヒマシ油を用いることができ、酸化エチレンの重合数は5~100が好ましく、20~50がより好ましく、30~40が特に好ましく、35が最も好ましい。ポリオキシエチレンヒマシ油の具体例としては、ポリオキシル5ヒマシ油、ポリオキシル9ヒマシ油、ポリオキシル15ヒマシ油、ポリオキシル35ヒマシ油、ポリオキシル40ヒマシ油等が挙げられ、ポリオキシル35ヒマシ油が最も好ましい。
ビタミンE TPGSは、トコフェロールポリエチレングリコール1000コハク酸エステルともいう。
ポリオキシエチレンポリオキシプロピレングリコールとしては、ポリオキシエチレン(160)ポリオキシプロピレン(30)グリコール、ポリオキシエチレン(42)ポリオキシプロピレン(67)グリコール、ポリオキシエチレン(54)ポリオキシプロピレン(39)グリコール、ポリオキシエチレン(196)ポリオキシプロピレン(67)グリコール、ポリオキシエチレン(20)ポリオキシプロピレン(20)グリコール等が挙げられる。
ショ糖脂肪酸エステルとしては、ショ糖ステアリン酸エステル等が挙げられる。
緩衝剤の例としては、リン酸又はその塩、ホウ酸又はその塩、クエン酸又はその塩、酢酸又はその塩、炭酸又はその塩、酒石酸又はその塩、ε-アミノカプロン酸、トロメタモール等が挙げられる。より具体的には、リン酸塩としては、リン酸ナトリウム、リン酸二水素ナトリウム、リン酸水素二ナトリウム、リン酸カリウム、リン酸二水素カリウム、リン酸水素二カリウム等が挙げられ、ホウ酸塩としては、ホウ砂、ホウ酸ナトリウム、ホウ酸カリウム等が挙げられ、クエン酸又はその塩としては、クエン酸一水和物、クエン酸ナトリウム、クエン酸二ナトリウム、クエン酸三ナトリウム等が挙げられ、酢酸塩としては、酢酸ナトリウム、酢酸カリウム等が挙げられ、炭酸塩としては、炭酸ナトリウム、炭酸水素ナトリウム等が挙げられ、酒石酸塩としては、酒石酸ナトリウム、酒石酸カリウム等が挙げられる。中でも、ホウ酸若しくはその塩、又は、クエン酸若しくはその塩が好ましい。
等張化剤の例としては、イオン性等張化剤や非イオン性等張化剤等が挙げられる。
イオン性等張化剤としては、塩化ナトリウム、塩化カリウム、塩化カルシウム、塩化マグネシウム等が挙げられ、非イオン性等張化剤としてはグリセリン、プロピレングリコール、ソルビトール、マンニトール等が挙げられる。本発明の医薬製剤に等張化剤を配合する場合、その含有量は、等張化剤の種類などにより適宜調整することができるが、0.01~10%(w/v)が好ましく、0.02~7%(w/v)がより好ましく、0.1~5%(w/v)がさらに好ましく、0.5~4%(w/v)が特に好ましく、0.8~3%(w/v)が最も好ましい。
安定化剤の例としては、エデト酸、エデト酸一ナトリウム、エデト酸二ナトリウム、エデト酸四ナトリウム、クエン酸ナトリウム等が挙げられ、エデト酸二ナトリウムが特に好ましい。エデト酸ナトリウムは水和物であってもよい。本発明の医薬製剤に安定化剤を配合する場合、その含有量は、安定化剤の種類などにより適宜調整することができるが、0.001~1%(w/v)が好ましく、0.005~0.5%(w/v)がより好ましく、0.01~0.1%(w/v)が最も好ましい。
防腐剤の例としては、ベンザルコニウム塩化物、ベンザルコニウム臭化物、ベンゼトニウム塩化物、ソルビン酸、ソルビン酸カリウム、パラオキシ安息香酸メチル、パラオキシ安息香酸プロピル、クロロブタノール等が挙げられる。本発明の医薬製剤に防腐剤を配合する場合、その含有量は、防腐剤の種類などにより適宜調整することができるが、0.0001~1%(w/v)が好ましく、0.0005~0.1%(w/v)がより好ましく、0.001~0.05%(w/v)がさらに好ましく、0.005~0.010%(w/v)が最も好ましい。また、防腐剤を含有しない場合も好ましい。
抗酸化剤の例としては、アスコルビン酸、トコフェノール、ジブチルヒドロキシトルエン、ブチルヒドロキシアニソール、エリソルビン酸ナトリウム、没食子酸プロピル、亜硫酸ナトリウム等が挙げられる。本発明の医薬製剤に抗酸化剤を配合する場合、その含有量は、抗酸化剤の種類などにより適宜調整することができるが、0.0001~1%(w/v)が好ましく、0.0005~0.1%(w/v)がより好ましく、0.001~0.05%(w/v)が最も好ましい。
粘稠化剤の例としては、メチルセルロース、エチルセルロース、ヒドロキシメチルセルロース、ヒドロキシエチルセルロース、ヒドロキシプロピルセルロース、ヒドロキシエチルメチルセルロース、ヒドロキシプロピルメチルセルロース、カルボキシメチルセルロース、カルボキシメチルセルロースナトリウム、ヒドロキシプロピルメチルセルロースアセテートサクシネート、ヒドロキシプロピルメチルセルロースフタレート、カルボキシメチルエチルセルロース、酢酸フタル酸セルロース、ポリビニルピロリドン、ポリビニルアルコール、カルボキシビニルポリマー、ポリエチレングリコール等が挙げられる。
本発明の医薬製剤に粘稠化剤を配合する場合、その含有量は、粘稠化剤の種類などにより適宜調整することができるが、0.001~5%(w/v)が好ましく、0.01~1%(w/v)がより好ましく、0.1~0.5%(w/v)が最も好ましい。
基剤の例としては、水、生理食塩水、ジメチルスルホキシド、PEG400等のポリエチレングルコール、クエン酸トリブチル、クエン酸アセチルトリブチル、安息香酸ベンジル、白色ワセリン、流動パラフィン等が挙げられ、水、生理食塩水、ジメチルスルホキシド、PEG400が好ましい。
本発明における医薬製剤は、他の緑内障または高眼圧症治療薬が効果不十分な患者に対しても優れた眼圧下降作用を示すことから、緑内障治療若しくは予防剤、及び/又は、高眼圧症治療若しくは予防剤、及び/又は眼圧下降剤として有用である。本発明における緑内障としては、原発開放隅角緑内障、続発開放隅角緑内障、正常眼圧緑内障、房水産生過多緑内障、原発閉塞隅角緑内障、続発閉塞隅角緑内障、プラトー虹彩緑内障、混合型緑内障、発達緑内障、ステロイド緑内障、落屑緑内障、アミロイド緑内障、血管新生緑内障、悪性緑内障、水晶体の嚢性緑内障、plateau iris syndrome等が、好ましくは原発開放隅角緑内障、正常眼圧緑内障、原発閉塞隅角緑内障が挙げられ、特に本発明の医薬製剤は原発開放隅角緑内障に有効である。
また、本発明の医薬製剤によって、本発明の医薬製剤による治療前の眼圧に対する治療後の眼圧の下降幅(眼圧変化量)は、少なくとも1.0mmHg以上、好ましくは1.2mmHg以上、より好ましくは1.4mmHg以上、さらに好ましくは1.6mmHg以上、もっと好ましくは1.8mmHg以上、殊更好ましくは2.0mmHg以上、特に好ましくは2.5mmHg以上、最も好ましくは2.9mmHg以上であることが好適である。本発明の医薬製剤による当該眼圧変化量の上限値としては、例えば10.0mmHg以下、好ましくは8.0mmHg以下、より好ましくは6.0mmHg以下、さらに好ましくは5.5mmHg以下、もっと好ましくは5.0mmHg以下、殊更好ましくは4.5mmHg以下、特に好ましくは4.0mmHg以下、最も好ましくは3.2mmHg以下を挙げることができ、上記下限値と上限値を適宜組みあわせた範囲を適宜選択できる。好ましい本発明の医薬製剤による当該眼圧変化量は、例えば、1.0~10.0mmHg、好ましくは1.4~8.0mmHg、より好ましくは1.8~6.0mmHgである。
なお、ここで定義した「本発明の医薬製剤による治療前の眼圧に対する治療後の眼圧の下降率(眼圧下降率)」及び「下降幅(眼圧変化量)」は、後述する[投与方法]の(3)に示すような第二の処置工程での眼圧の下降率及び下降幅を意味し、[投与方法]の(1)に示す第一の処置工程のような、前記他の緑内障または高眼圧症治療薬による治療による眼圧降下率及び眼圧変化量を含まない。従って、オミデネパグ等を有効成分とする本発明の医薬製剤は、ラタノプロスト等を有効成分とする他の緑内障または高眼圧症治療薬の治療効果に上乗せして眼圧を下降できる。
本発明の医薬製剤は、経口でも、非経口でも投与することができ、例えば、点眼投与、硝子体内投与、結膜嚢内投与、前房内投与、結膜下投与、テノン嚢下投与又は涙点プラグ投与であってもよい。本発明の医薬製剤の剤形としては、点眼剤、眼軟膏、注射剤、涙点プラグ、錠剤、カプセル剤、顆粒剤、散剤等が挙げられ、特に点眼剤、眼科用注射剤、涙点プラグが好ましい。眼科用注射剤としては、硝子体内投与、前房内投与、結膜嚢内投与、前房内投与、結膜下投与又はテノン嚢下投与用の注射剤が挙げられる。本発明の医薬製剤の剤形は、薬品の技術分野における通常の方法に従って製造することができる。錠剤、カプセル剤、顆粒剤、散剤等の経口剤は、上記の添加剤のほか、乳糖、結晶セルロース、デンプン、植物油等の増量剤、ステアリン酸マグネシウム、タルク等の滑沢剤、ヒドロキシプロピルセルロース、ポリビニルピロリドン等の結合剤、カルボキシメチルセルロース カルシウム、低置換ヒドロキシプロピルメチルセルロース等の崩壊剤、ヒドロキシプロピルメチルセルロース、マクロゴール、シリコン樹脂等のコーティング剤、ゼラチン皮膜等の皮膜剤等を必要に応じて使用して、製剤化することができる。
本発明の医薬製剤の用法・用量は、所望の薬効を奏するのに十分な用法・用量であれば特に制限はなく、疾患の症状、患者の年齢や体重、医薬製剤の剤形等に応じて適宜選択できる。
具体的には、点眼剤の場合、1回量1~5滴、好ましくは1~3滴、より好ましくは1~2滴、特に好ましくは1滴を、1日1~4回、好ましくは1日1~3回、より好ましくは1日1~2回、特に好ましくは1日1回を、毎日~1週間毎に点眼投与することができる。1日1回1滴を毎日、点眼投与することが好ましい。ここで、1滴は、通常、約0.01~約0.1mLであり、好ましくは、約0.015~約0.07mLであり、より好ましくは、約0.02~約0.05mLであり、特に好ましくは約0.03mLである。
本発明の医薬製剤は、上記他の緑内障または高眼圧症治療薬による治療又は予防で十分な効果が得られない又は得られないと見込まれる場合に、続いて投与される。すなわち、本発明の医薬製剤の投与方法は、
(1)オミデネパグ若しくはそのエステル又はそれらの塩以外の上記他の有効成分により緑内障若しくは高眼圧症を治療または予防する第一の処置工程、
(2)上記第一の処置工程では治療が不十分若しくは予防効果が不十分であるか否か判断する任意工程、
(3)前記第一の処置工程に続き、オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を投与して緑内障若しくは高眼圧症を治療または予防する第二の処置工程、
を含む。
上記のような投与方法に基づいて本発明の医薬製剤を患者に投与することにより、上記他の有効成分による治療または予防による効果にかかわらず、若しくは当該効果が不十分であった場合に、本発明の医薬製剤により追加的に眼圧降下作用等が得られることとなり、さらに緑内障若しくは高眼圧症を治療または予防することができる。
例えば、他の有効成分としてラタノプロストを挙げると、ラタノプロストによって緑内障若しくは高眼圧症の治療及び予防を行った後、さらに眼圧の下降が必要な場合、又は、ラタノプロストでは治療または予防後に眼圧の再上昇(リバウンド)が起こる又は見込まれる場合に、本発明の医薬製剤によって当該眼圧下降の要求又は眼圧の再上昇(リバウンド)防止に対する効果が期待できる。
本発明のオミデネパグ若しくはそのエステル又はそれらの塩は、上記のような医薬製剤の製造に使用することができる。以下に本発明の医薬製剤における代表的な製剤例を示す。なお、下記製剤例において各成分の配合量は製剤100mL中の含量である。
点眼剤1(100mL中)
オミデネパグ イソプロピル 0.002g
ホウ酸 0.2g
グリセリン 2.0g
ポリソルベート80 0.5g
エデト酸二ナトリウム 0.05g
ベンザルコニウム塩化物 0.005g
希塩酸 適量
水酸化ナトリウム 適量
精製水 適量
点眼剤2(100mL中)
オミデネパグ イソプロピル 0.002g
リン酸二水素ナトリウム 0.2g
グリセリン 2.0g
ビタミンE TPGS 0.8g
エデト酸二ナトリウム 0.05g
ベンザルコニウム塩化物 0.005g
希塩酸 適量
水酸化ナトリウム 適量
精製水 適量
点眼剤3(100mL中)
オミデネパグ イソプロピル 0.002g
クエン酸三ナトリウム 0.2g
グリセリン 2.0g
ポリオキシエチレン硬化ヒマシ油60 0.3g
エデト酸二ナトリウム 0.05g
ベンザルコニウム塩化物 0.005g
希塩酸 適量
水酸化ナトリウム 適量
精製水 適量
注射剤1(100mL中)
オミデネパグ 0.003g
PEG400 適量
注射剤2(100mL中)
オミデネパグ 0.0003g
PEG400 適量
他の緑内障または高眼圧症治療薬が効果不十分な患者に対するオミデネパグ イソプロピルの効果を検討した。
精製水にオミデネパグ イソプロピル、ポリオキシル35ヒマシ油、グリセリン、クエン酸、クエン酸ナトリウム、エデト酸ナトリウム、ベンザルコニウム塩化物を溶解させ、pH調整後、精製水を加えて全量調整し、0.002%(w/v)オミデネパグ イソプロピル点眼剤Aを製造した。
原発開放隅角緑内障又は高眼圧症患者を対象に1~4週間のウォッシュアウト(washout)期間の後、導入期(-8週~ベースライン)としてラタノプロスト点眼液0.005%を8週間にわたって1日1回1滴、両眼点眼した。導入期終了時に点眼開始時からの眼圧の下降率が15%以下であったラタノプロスト点眼液治療抵抗性の原発開放隅角緑内障(ヒト、21人)又は高眼圧症患者(ヒト、5人)が治療期(ベースライン~4週)に移行し、上記0.002%(w/v)オミデネパグ イソプロピル点眼剤Aを4週間にわたって、1日1回1滴(約0.03mL)、毎日両眼点眼した。
結果を図1に示す。
以上から、オミデネパグ若しくはそのエステル又はそれらの塩は、他の緑内障または高眼圧症治療薬が効果不十分な患者に対して優れた眼圧下降作用を示すことがわかった。
Claims (20)
- オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する緑内障若しくは高眼圧症を治療または予防するための医薬製剤であって、他の緑内障または高眼圧症治療薬が効果不十分な患者に投与される医薬製剤。
- 前記緑内障若しくは高眼圧症を治療または予防が、前記他の緑内障または高眼圧症治療薬によって緑内障若しくは高眼圧症の治療または予防を行った後、さらに前記有効成分によって眼圧を下げて緑内障若しくは高眼圧症を治療または予防するものである、請求項1に記載の医薬製剤。
- 前記他の緑内障または高眼圧症治療薬の有効成分がプロスタグランジンF2α誘導体である、請求項1又は2に記載の医薬製剤。
- 前記他の緑内障または高眼圧症治療薬有効成分がラタノプロストである、請求項1~3のいずれか一項に記載の医薬製剤。
- オミデネパグ若しくはそのエステル又はそれらの塩の含有量が、0.001~0.003%(w/v)である、請求項1~4のいずれか一項に記載の医薬製剤。
- オミデネパグ若しくはそのエステル又はそれらの塩の含有量が、0.002%(w/v)である、請求項1~5のいずれか一項に記載の医薬製剤。
- オミデネパグ若しくはそのエステル又はそれらの塩が、オミデネパグ イソプロピルである、請求項1~6のいずれか一項に記載の医薬製剤。
- 点眼剤である、請求項1~7のいずれか一項に記載の医薬製剤。
- オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する緑内障若しくは高眼圧症を治療または予防するための医薬製剤であって、前記緑内障が、オミデネパグ若しくはそのエステル又はそれらの塩以外の他の有効成分による緑内障治療に抵抗性の緑内障であり、前記高眼圧症が、前記他の有効成分による高眼圧症治療に抵抗性の高眼圧症である、医薬製剤。
- オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を患者に投与することを含む、緑内障若しくは高眼圧症を治療または予防する方法であって、前記患者が、他の緑内障または高眼圧症治療薬が効果不十分な患者である、方法。
- 前記緑内障若しくは高眼圧症を治療または予防が、前記他の緑内障または高眼圧症治療薬によって緑内障若しくは高眼圧症の治療または予防を行った後、さらに前記有効成分によって眼圧を下げて緑内障若しくは高眼圧症を治療または予防するものである、請求項10に記載の方法。
- 前記他の緑内障または高眼圧症治療薬の有効成分がプロスタグランジンF2α誘導体である、請求項10又は11に記載の方法。
- 前記他の緑内障または高眼圧症治療薬有効成分がラタノプロストである、請求項10~12のいずれか一項に記載の方法。
- 前記医薬製剤中におけるオミデネパグ若しくはそのエステル又はそれらの塩の含有量が、0.001~0.003%(w/v)である、請求項10~13のいずれか一項に記載の方法。
- 前記医薬製剤中におけるオミデネパグ若しくはそのエステル又はそれらの塩の含有量が、0.002%(w/v)である、請求項10~14のいずれか一項に記載の方法。
- 前記オミデネパグ若しくはそのエステル又はそれらの塩が、オミデネパグ イソプロピルである、請求項10~15のいずれか一項に記載の方法。
- 前記投与が点眼投与である、請求項10~16のいずれか一項に記載の方法。
- オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を患者に投与することを含む、緑内障若しくは高眼圧症を治療または予防するための方法であって、前記緑内障が、オミデネパグ若しくはそのエステル又はそれらの塩以外の他の有効成分による緑内障治療に抵抗性の緑内障であり、前記高眼圧症が、前記他の有効成分による高眼圧症治療に抵抗性の高眼圧症である、方法。
- オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を患者に投与することを含む、緑内障若しくは高眼圧症を治療または予防する方法であって、以下の工程
(1)オミデネパグ若しくはそのエステル又はそれらの塩以外の他の緑内障または高眼圧症治療薬を患者に投与する第一の処置工程、
(2)上記第一の処置工程では治療が不十分若しくは予防効果が不十分であるか否か判断する工程、
(3)上記第一の処置工程では治療が不十分若しくは予防効果が不十分である場合、オミデネパグ若しくはそのエステル又はそれらの塩を有効成分として含有する医薬製剤を患者にさらに投与する第二の処置工程、
を含む方法。 - 緑内障若しくは高眼圧症を治療または予防するための薬剤の製造における、請求項1~9のいずれか一項に記載の医薬製剤の使用。
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201880083527.1A CN111526876A (zh) | 2017-12-28 | 2018-12-27 | 含有吡啶基氨基乙酸化合物的药物制剂 |
| KR1020207019947A KR102901189B1 (ko) | 2017-12-28 | 2018-12-27 | 피리딜아미노아세트산 화합물을 함유하는 의약 제제 |
| EP18896002.5A EP3733179A4 (en) | 2017-12-28 | 2018-12-27 | PHARMACEUTICAL PREPARATION CONTAINING A PYRIDYLAMINOACETIC ACID COMPOUND |
| RU2020124742A RU2776374C2 (ru) | 2017-12-28 | 2018-12-27 | Фармацевтический препарат, содержащий пиридиламиноуксусную кислоту |
| US16/957,749 US11666563B2 (en) | 2017-12-28 | 2018-12-27 | Pharmaceutical preparation containing pyridyl aminoacetic acid compound |
| CA3087019A CA3087019A1 (en) | 2017-12-28 | 2018-12-27 | Pharmaceutical preparation containing pyridyl aminoacetic acid compound |
| JP2019562181A JP7346305B2 (ja) | 2017-12-28 | 2018-12-27 | ピリジルアミノ酢酸化合物を含有する医薬製剤 |
| US18/314,909 US12290511B2 (en) | 2017-12-28 | 2023-05-10 | Pharmaceutical preparation containing pyridyl aminoacetic acid compound |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762611017P | 2017-12-28 | 2017-12-28 | |
| US62/611,017 | 2017-12-28 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/957,749 A-371-Of-International US11666563B2 (en) | 2017-12-28 | 2018-12-27 | Pharmaceutical preparation containing pyridyl aminoacetic acid compound |
| US18/314,909 Continuation US12290511B2 (en) | 2017-12-28 | 2023-05-10 | Pharmaceutical preparation containing pyridyl aminoacetic acid compound |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019131901A1 true WO2019131901A1 (ja) | 2019-07-04 |
Family
ID=67067440
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2018/048228 Ceased WO2019131901A1 (ja) | 2017-12-28 | 2018-12-27 | ピリジルアミノ酢酸化合物を含有する医薬製剤 |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US11666563B2 (ja) |
| EP (1) | EP3733179A4 (ja) |
| JP (1) | JP7346305B2 (ja) |
| KR (1) | KR102901189B1 (ja) |
| CN (1) | CN111526876A (ja) |
| CA (1) | CA3087019A1 (ja) |
| TW (1) | TWI842692B (ja) |
| WO (1) | WO2019131901A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021145355A1 (ja) * | 2020-01-15 | 2021-07-22 | 参天製薬株式会社 | 近視の治療、近視の予防および/または近視の進行抑制のための医薬組成物 |
| JPWO2022034909A1 (ja) * | 2020-08-13 | 2022-02-17 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111526876A (zh) | 2017-12-28 | 2020-08-11 | 参天制药株式会社 | 含有吡啶基氨基乙酸化合物的药物制剂 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110054172A1 (en) | 2008-03-12 | 2011-03-03 | Ryo Iwamura | Pyridylaminoacetic acid compound |
| US20120190852A1 (en) | 2009-03-30 | 2012-07-26 | Ube Industries, Ltd. | Medical composition for treatment or prophylaxis of glaucoma |
| US20140018350A1 (en) | 2012-07-13 | 2014-01-16 | Asahi Glass Co., Ltd. | Combination of sulfonamide compound and tafluprost |
| US20140018396A1 (en) | 2012-07-13 | 2014-01-16 | Santen Pharmaceutical Co., Ltd | Combination of sulfonamide compound |
| US20150196541A1 (en) | 2014-01-10 | 2015-07-16 | Santen Pharmaceutical Co., Ltd. | Pharmaceutical formulations comprising a pyridylaminoacetic acid compound |
| US20160317512A1 (en) | 2014-01-10 | 2016-11-03 | Santen Pharmaceutical Co., Ltd. | Pyridylaminoacetic acid compound and polyoxyethylene castor oil-containing pharmaceutical composition |
| US20160317664A1 (en) | 2014-01-10 | 2016-11-03 | Santen Pharmaceutical Co., Ltd. | Pharmaceutical composition containing pyridylaminoacetic acid compound |
| WO2017002941A1 (ja) | 2015-07-01 | 2017-01-05 | 参天製薬株式会社 | クエン酸エステルを含有するデポ剤 |
| WO2017006985A1 (ja) | 2015-07-09 | 2017-01-12 | 参天製薬株式会社 | ピリジルアミノ酢酸化合物を含有する予防及び/又は治療剤 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101113280B1 (ko) | 2009-11-17 | 2012-03-14 | 천공전기(주) | 인장 클램프 |
| EP2702087B1 (en) | 2011-04-28 | 2015-03-18 | Basf Se | Semiconductor materials based on thienothiophene-2,5-dione oligomers and polymers |
| WO2012165639A1 (ja) | 2011-06-03 | 2012-12-06 | キヤノン株式会社 | トナー |
| JP6193655B2 (ja) * | 2012-07-13 | 2017-09-06 | 参天製薬株式会社 | スルホンアミド化合物の組み合わせ |
| JP2017002941A (ja) | 2015-06-05 | 2017-01-05 | 日本精工株式会社 | ボールねじ |
| US10029330B2 (en) | 2015-06-17 | 2018-07-24 | The Boeing Company | Hybrid laser machining of multi-material stack-ups |
| CN111526876A (zh) | 2017-12-28 | 2020-08-11 | 参天制药株式会社 | 含有吡啶基氨基乙酸化合物的药物制剂 |
-
2018
- 2018-12-27 CN CN201880083527.1A patent/CN111526876A/zh active Pending
- 2018-12-27 KR KR1020207019947A patent/KR102901189B1/ko active Active
- 2018-12-27 CA CA3087019A patent/CA3087019A1/en active Pending
- 2018-12-27 JP JP2019562181A patent/JP7346305B2/ja active Active
- 2018-12-27 US US16/957,749 patent/US11666563B2/en active Active
- 2018-12-27 TW TW107147394A patent/TWI842692B/zh active
- 2018-12-27 WO PCT/JP2018/048228 patent/WO2019131901A1/ja not_active Ceased
- 2018-12-27 EP EP18896002.5A patent/EP3733179A4/en not_active Withdrawn
-
2023
- 2023-05-10 US US18/314,909 patent/US12290511B2/en active Active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110054172A1 (en) | 2008-03-12 | 2011-03-03 | Ryo Iwamura | Pyridylaminoacetic acid compound |
| US20120190852A1 (en) | 2009-03-30 | 2012-07-26 | Ube Industries, Ltd. | Medical composition for treatment or prophylaxis of glaucoma |
| US20140018350A1 (en) | 2012-07-13 | 2014-01-16 | Asahi Glass Co., Ltd. | Combination of sulfonamide compound and tafluprost |
| US20140018396A1 (en) | 2012-07-13 | 2014-01-16 | Santen Pharmaceutical Co., Ltd | Combination of sulfonamide compound |
| US20150196541A1 (en) | 2014-01-10 | 2015-07-16 | Santen Pharmaceutical Co., Ltd. | Pharmaceutical formulations comprising a pyridylaminoacetic acid compound |
| WO2015105144A1 (ja) * | 2014-01-10 | 2015-07-16 | 参天製薬株式会社 | ピリジルアミノ酢酸化合物を含む医薬製剤 |
| US20160317512A1 (en) | 2014-01-10 | 2016-11-03 | Santen Pharmaceutical Co., Ltd. | Pyridylaminoacetic acid compound and polyoxyethylene castor oil-containing pharmaceutical composition |
| US20160317664A1 (en) | 2014-01-10 | 2016-11-03 | Santen Pharmaceutical Co., Ltd. | Pharmaceutical composition containing pyridylaminoacetic acid compound |
| WO2017002941A1 (ja) | 2015-07-01 | 2017-01-05 | 参天製薬株式会社 | クエン酸エステルを含有するデポ剤 |
| WO2017006985A1 (ja) | 2015-07-09 | 2017-01-12 | 参天製薬株式会社 | ピリジルアミノ酢酸化合物を含有する予防及び/又は治療剤 |
Non-Patent Citations (6)
| Title |
|---|
| AIHARA, MAKOTO ET AL.: "The Intraocular pressure lowering effect of tafluprost in latanoprost hyporesponsive monkeys", JOURNAL OF JAPANESE OPHTHALMOLOGICAL SOCIETY, vol. 113, no. 01-017, pages 195, ISSN: 0029-0203 * |
| CHEMICAL ABSTRACTS, Columbus, Ohio, US; abstract no. 1187451-19-9 |
| KALOUCHE, G. ET AL.: "Activation of Prostaglandin FP and EP 2 Receptors Differently Modulates Myofibroblast Transition in a Model of Adult Primary Human Trabecular Meshwork Cells", INVEST OPHTHALMOL VIS SCI, vol. 57, no. 4, 2016, pages 1816 - 1825, XP055624516, ISSN: 0146-0404 * |
| KOCULUK, Y. ET AL.: "Efficacy of Monotherapy with Either Bimatoprost or Travoprost in Patients with Primary Open-Angle Glaucoma Resistant to Latanoprost Therapy", TURK J OPHTHALMOL, vol. 41, no. 5, 2011, pages 295 - 298, XP055624519, ISSN: 1300-0659 * |
| ODANI, NORIKO: "Development of intraocular pressure lowering drugs of a new action mechanism - Selective EP 2 receptor agonists, Omidenepag Isopropyl ophthalmic solution", PROGRAMS AND LECTURE ABSTRACTS OF THE 37TH ANNUAL SCIENTIFIC MEETING OF THE JAPANESE SOCIETY FOR OCULAR PHARMACOLOGY, XP055289484 * |
| See also references of EP3733179A4 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021145355A1 (ja) * | 2020-01-15 | 2021-07-22 | 参天製薬株式会社 | 近視の治療、近視の予防および/または近視の進行抑制のための医薬組成物 |
| JPWO2022034909A1 (ja) * | 2020-08-13 | 2022-02-17 | ||
| WO2022034909A1 (ja) * | 2020-08-13 | 2022-02-17 | 参天製薬株式会社 | セペタプロストを含有する医薬製剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3733179A1 (en) | 2020-11-04 |
| US20230277515A1 (en) | 2023-09-07 |
| TWI842692B (zh) | 2024-05-21 |
| JP7346305B2 (ja) | 2023-09-19 |
| JPWO2019131901A1 (ja) | 2020-12-10 |
| EP3733179A4 (en) | 2021-09-15 |
| TW201929854A (zh) | 2019-08-01 |
| US11666563B2 (en) | 2023-06-06 |
| KR102901189B1 (ko) | 2025-12-16 |
| CA3087019A1 (en) | 2019-07-04 |
| RU2020124742A3 (ja) | 2022-01-28 |
| KR20200103719A (ko) | 2020-09-02 |
| RU2020124742A (ru) | 2022-01-28 |
| US12290511B2 (en) | 2025-05-06 |
| CN111526876A (zh) | 2020-08-11 |
| US20200345713A1 (en) | 2020-11-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7726542B2 (ja) | ピリジルアミノ酢酸化合物を含む医薬製剤 | |
| JP5174777B2 (ja) | Rhoキナーゼ阻害剤とプロスタグランジン類からなる緑内障治療剤 | |
| JP6383058B2 (ja) | スルホンアミド化合物の組み合わせ | |
| JP4934653B2 (ja) | Rhoキナーゼ阻害剤とβ遮断薬からなる緑内障治療剤 | |
| US12290511B2 (en) | Pharmaceutical preparation containing pyridyl aminoacetic acid compound | |
| US20140018350A1 (en) | Combination of sulfonamide compound and tafluprost | |
| JP4482726B2 (ja) | Rhoキナーゼ阻害剤とプロスタグランジン類からなる緑内障治療剤 | |
| TWI794140B (zh) | 含有吡啶基胺基乙酸化合物之預防及/或治療劑 | |
| RU2776374C2 (ru) | Фармацевтический препарат, содержащий пиридиламиноуксусную кислоту | |
| HK40031074A (en) | Pharmaceutical preparation containing pyridyl aminoacetic acid compound | |
| HK40029199A (en) | Pharmaceutical preparation containing pyridyl aminoacetic acid compound | |
| JP2023110029A (ja) | オミデネパグの組合せ | |
| HK40025370A (en) | Omidenepag combination | |
| JP2012250952A (ja) | アデノシン誘導体とRhoキナーゼ阻害剤の組合せ剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18896002 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 3087019 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2019562181 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20207019947 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2018896002 Country of ref document: EP Effective date: 20200728 |
|
| WWR | Wipo information: refused in national office |
Ref document number: 1020207019947 Country of ref document: KR |
|
| WWC | Wipo information: continuation of processing after refusal or withdrawal |
Ref document number: 1020207019947 Country of ref document: KR |