WO2019177139A1 - 皮膚状態改善用組成物 - Google Patents
皮膚状態改善用組成物 Download PDFInfo
- Publication number
- WO2019177139A1 WO2019177139A1 PCT/JP2019/010773 JP2019010773W WO2019177139A1 WO 2019177139 A1 WO2019177139 A1 WO 2019177139A1 JP 2019010773 W JP2019010773 W JP 2019010773W WO 2019177139 A1 WO2019177139 A1 WO 2019177139A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- skin
- lactococcus
- composition
- present
- skin condition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/66—Microorganisms or materials therefrom
- A61K35/74—Bacteria
- A61K35/741—Probiotics
- A61K35/744—Lactic acid bacteria, e.g. enterococci, pediococci, lactococci, streptococci or leuconostocs
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/135—Bacteria or derivatives thereof, e.g. probiotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/02—Bacterial antigens
- A61K39/09—Lactobacillales, e.g. aerococcus, enterococcus, lactobacillus, lactococcus, streptococcus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/0056—Mouth soluble or dispersible forms; Suckable, eatable, chewable coherent forms; Forms rapidly disintegrating in the mouth; Lozenges; Lollipops; Bite capsules; Baked products; Baits or other oral forms for animals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/16—Emollients or protectives, e.g. against radiation
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/20—Bacteria; Culture media therefor
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K2035/11—Medicinal preparations comprising living procariotic cells
- A61K2035/115—Probiotics
Definitions
- the present invention relates to a composition for improving skin condition.
- the skin consists of the epidermis, dermis, and subcutaneous tissue from the outside and retains moisture in the body, protecting the body from physical and chemical stimuli and pathogenic microorganisms.
- the epidermis is divided into a basal layer, a spiny layer, a granule layer, and a stratum corneum from the deep layer to the surface layer.
- stratum corneum stratum corneum accumulates from several to several tens of layers, forming a strong stratum corneum barrier, retaining moisture and protecting the skin from dryness and external irritation.
- UV light is divided into UVA, UVB and UVC depending on the wavelength, UVB passes through the epidermis and reaches the upper dermis, and UVA reaches the deep dermis.
- Sunburn caused by UVB irradiation starts several hours after exposure and peaks after 24 hours. In mild cases, it is a mild burning sensation and erythema. In advanced cases, dark red with edema, blisters, and erosions It becomes spots and is accompanied by a strong burning sensation.
- skin tissue with erythema dilation of capillaries in the dermal epithelial layer and swelling of vascular endothelial cells are observed. It is desirable to improve the deterioration of the skin condition caused by drying and ultraviolet rays at an early stage from the viewpoint of restoration of normal skin function and cosmetics.
- Patent Document 1 a fermented milk product of lactic acid bacteria belonging to Lactococcus lactis having a skin function improving effect is known as a microorganism-derived food material (Patent Document 1).
- Patent Document 1 the technique of Patent Document 1 is intended to prevent and reduce stress-induced skin blood flow reduction, and is not intended to improve skin function due to UV damage.
- An object of the present invention is to provide a novel food material effective for improving the skin condition.
- Lactococcus lactis subspices lactis JCM5805 a kind of bacteria belonging to the genus Lactococcus, has an effect of improving the skin condition in mice and humans.
- the present invention is based on these findings.
- a composition for improving skin condition and an agent for improving skin condition comprising Lactococcus bacteria as an active ingredient.
- the composition and preparation according to [1] or [2] above, wherein the skin condition deteriorated by exposure to light is a decrease in skin water content and / or an increase in skin redness.
- [13] Use of a bacterium belonging to the genus Lactococcus or a composition containing the same for improving the skin condition, as a skin condition improving agent, or in the method described in [12] above.
- a method for treating, preventing and ameliorating photodermatoses comprising ingesting or administering an effective amount of a Lactococcus bacterium or a composition containing the same to a subject in need thereof.
- a therapeutic, preventive or ameliorating agent for photodermatoses or in the method according to [16] above, for the manufacture of a therapeutic, preventive or ameliorating agent for photodermatoses.
- a lactococcus bacterium or a composition containing the same for use in the treatment, prevention or amelioration of photodermatoses, or for use in the method of [16] above.
- composition of [1] and [15] above is referred to as “the composition of the present invention”, and the agent of [1] and [15] above is referred to as “the agent of the present invention”. Each has something to say.
- composition and the preparation of the present invention contain, as an active ingredient, lactic acid bacteria, which are food materials that have been eaten by humans for many years. Therefore, the composition and the preparation of the present invention can be used for improving the skin condition, and are advantageous in that they can be taken for a long time without causing any side effects.
- FIG. 1 is a diagram showing the relationship between Lactococcus lactis subsp. Lactis JCM5805 strain and strains equivalent to the strain (the strain derived from the strain and the strain from which the strain is derived).
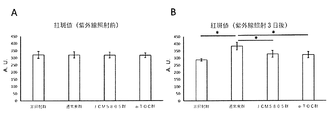
- FIG. 2 is a graph showing the effect of improving skin damage (erythema) by ingesting lactic acid bacteria (JCM5805) in Example 1.
- A A graph showing the erythema value before ultraviolet irradiation.
- B It is the graph showing the erythema value 3 days after ultraviolet irradiation. Expressed as mean ⁇ standard deviation. * Indicates p ⁇ 0.05 (Tukey-Kramer test).
- FIG. 1 is a diagram showing the relationship between Lactococcus lactis subsp. Lactis JCM5805 strain and strains equivalent to the strain (the strain derived from the strain and the strain from which the strain is derived).
- FIG. 2 is a graph showing the effect of improving skin damage (erythema) by
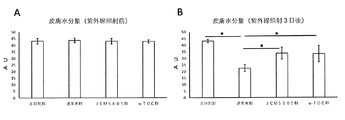
- FIG. 3 is a graph showing an improvement effect of skin damage (skin dryness) due to intake of lactic acid bacteria (JCM5805) in Example 1.
- A A graph showing the amount of skin moisture before ultraviolet irradiation.
- B It is the graph showing the amount of skin moisture 3 days after ultraviolet irradiation. Expressed as mean ⁇ standard deviation. * Indicates p ⁇ 0.05 (Tukey-Kramer test).
- FIG. 4 is a diagram showing the effect of improving skin damage (skin dryness) by ingesting lactic acid bacteria (JCM5805) in Example 1.
- B It is the graph showing the amount of skin moisture transpiration 3 days after ultraviolet irradiation. Expressed as mean ⁇ standard deviation. * Indicates p ⁇ 0.05 (Tukey-Kramer test).
- the Lactococcus bacterium used as an active ingredient in the present invention is a lactic acid cocci belonging to the genus Lactococcus .
- bacteria of the genus Lactococcus include, for example, Lactococcus lactis subspices lactis JCM5805, Lactococcus lactis subspices lactis JCM20101, Lactococcus lactis subspices lactis NBRC12007, Lactococcus lactis subspices lactis NRIC1150, Lactococcus galviae NBRC100934, Lactococcus lactis sub-species Cremolis JCM16167, Lactococcus lactis sub-species Cremolis NBRC100676, Lactococcus lactis sub-species Holdoniae JCM1180 and Lactococcus lactis subsidies J.
- Lactococcus lactis sub-species lactis JCM5805 and lac Tococcus lactis subspecies lactis JCM20101 is preferred, and Lactococcus lactis subspices lactis JCM5805 is particularly preferred.
- the JCM strain is from RIKEN, BioResource Center, Microbial Materials Development Office (http://jcm.brc.riken.jp/en/), and the NBRC strain is an independent administrative product evaluation technology. From the National Institute for Biological and Genetic Resources (http://www.nbrc.nite.go.jp), NRIC strains are available from the Tokyo University of Agriculture, strain storage room (http://nodaiweb.university.jp/nric/) Each can be obtained.
- Lactococcus lactis subspices lactis JCM5805 in addition to the above-mentioned specific strains, Lactococcus lactis subspices lactis JCM5805, Lactococcus lactis subspices lactis JCM20101, Lactococcus lactis subspices lactis NBRC12007, Lactococcus Lactis subspices lactis NRIC1150, Lactococcus galviaeae NBRC100934, Lactococcus lactis subspices cremoris JCM16167, Lactococcus lactis subspices cremoris NBRC100676, Lactococcus lactis subspices Holdnia cospice Lactococcus lactis -A strain equivalent to that of Holdoniae JCM11040 can be used.
- the equivalent strain refers to a strain derived from the above strain, a strain from which the above strain is derived, or a progeny strain of the strain. Equivalent strains may be stored in other strain storage institutions.
- FIG. 1 shows a strain derived from Lactococcus lactis subsp. Lactis JCM5805 and a strain derived from Lactococcus lactis subsp. Lactis JCM5805.
- the equivalent strain of Lactococcus lactis subspices lactis JCM5805 shown in FIG. 1 can also be used as an active ingredient of the present invention. Including.
- lactic acid strains that can be used as active ingredients of the present invention are RIKEN, BioResource Center, Microbial Materials Development Office (1-3 Takanodai, Tsukuba City, Ibaraki Prefecture), American type culture collection (USA), Independent Administration It can be obtained from the National Institute of Technology and Evaluation (2-8-8, Kazusa-Kamashita, Kisarazu City, Chiba Prefecture), Tokyo University of Agriculture, strain storage room (1-1 1-1 Sakuragaoka, Setagaya-ku, Tokyo), etc.
- the Lactococcus bacterium as an active ingredient of the present invention may be in the form of a culture.
- a culture is a living cell, dead cell, live cell or crushed product of dead cell, lyophilized product of live cell or dead cell, crushed product of the lyophilized product, culture solution, culture fluid extraction Such as a part of Lactococcus bacteria and processed products of Lactococcus bacteria.
- the treated product includes, for example, a product obtained by treating a Lactococcus bacterium by enzyme treatment, heat treatment or the like, or a product obtained by ethanol precipitation of the treated product.
- Lactococcus bacteria can be cultured by a known method using a known medium.
- a known medium an MRS medium, a GAM medium, or an LM17 medium can be used, and inorganic salts, vitamins, amino acids, antibiotics, serum, and the like may be added as appropriate.
- the culture may be performed at 25 to 40 ° C. for several hours to several days.
- Lactococcus bacteria are collected by centrifugation or filtration. When used as dead bacteria, it may be used after being sterilized and inactivated by an autoclave or the like.
- the composition and agent of the present invention can be provided as an active ingredient, Lactococcus bacterium alone, or mixed with an active ingredient, Lactococcus bacterium, and other ingredients (for example, pharmaceutical additives). Can also be provided.
- the blending amount of Lactococcus bacteria in the composition and the agent of the present invention can be arbitrarily determined according to the purpose, application, form, dosage form, symptom, weight, etc., but the present invention is not limited thereto.
- the content can be 0.0005 to 99% (w / w) of the total amount, more preferably 0.001 to 50% (w / w). can do.
- the preparation of the present invention can be composed of a bacterium belonging to the genus Lactococcus
- the composition of the present invention can be composed of a bacterium belonging to the genus Lactococcus and other components.
- skin condition is used to mean a normal skin condition and a deteriorated skin condition.
- the skin condition can be evaluated using either or both of skin redness and skin moisture content as an index.
- the redness of the skin can be measured according to a known method (for example, measurement of red spot value, measurement of color difference, and measurement of hemoglobin amount index).
- the skin water content can be measured according to a known method such as measurement of water content and measurement of water transpiration.
- the amount of moisture can be measured by a capacitance method.
- Moisture transpiration should be measured by a method that calculates the transpiration from the temperature difference and humidity difference of the moisture passing through each sensor, assuming that the moisture evaporating from the skin and the surface of the object diffuses according to Fick's law. Can do.
- “improvement of skin condition” means that the skin condition is improved, and includes not only improving the deteriorated skin condition but also preventing further deterioration of the deteriorated skin condition. Furthermore, it includes not only improving normal skin condition but also preventing deterioration of normal skin condition. Improvement of skin condition can be assessed using either or both of skin redness and skin moisture as an indicator, and if either or both reduction of skin redness and increase of skin moisture is observed It can be evaluated that the skin condition has improved.
- “aggravated skin condition” means a state in which the skin is damaged by a stimulus in daily life.
- the stimulus in daily life include light exposure, changes in drying and humidity, changes in temperature, natural products, Examples include external stimuli such as exposure to foreign substances such as chemical substances, and disturbances in lifestyle such as sleep and meals.
- Deteriorated skin condition can also be assessed using either or both skin redness and skin moisture as an indicator, and either or both increased skin redness and decreased skin moisture were observed In some cases, it can be evaluated as a worsened skin condition.
- an increase in skin redness and a decrease in skin water content can be determined on the basis of normal skin condition, and redness increased from normal skin and skin that has deteriorated due to less water content than normal skin. Indicates the state.
- examples of the deteriorated skin state include a state in which the redness of the skin has increased and a state in which the skin moisture content has decreased.
- the state in which the redness of the skin is increased is a state in which the skin is reddish, and so-called reddish face and hot flashes are also included, and the state in which the skin water content is reduced is a state in which the skin is dry.
- the normal skin state can be rephrased as a state in which the skin barrier function is functioning normally, and this state is a state in which there is little or no awareness of crusting, snuffing, or hot flashes.
- a skin condition deteriorated by light exposure can be mentioned.
- the light exposure is a normal light exposure in daily life or a light exposure exceeding this (for example, tanning), and the wavelength of the light is not limited and includes daily light, but preferably a wavelength of 315 to 280 nm called ultraviolet light, especially UVB.
- Light exposure can be specified by the amount of ultraviolet irradiation, and light exposure that exceeds light exposure in daily life can be light exposure that exceeds the amount of ultraviolet irradiation in daily life.
- aggravated skin condition can be rephrased as photodermatosis. Therefore, according to the present invention, there are provided a composition for treating, preventing and ameliorating photodermatoses, and a therapeutic agent, prophylactic agent and ameliorating agent for photodermatoses, comprising Lactococcus bacteria as an active ingredient. Is done.
- photodermatosis is used in the meaning including solar dermatitis, sunburn, photoaging, and photosensitivity.
- composition and the preparation of the present invention can be provided in the form of pharmaceuticals (for example, pharmaceutical compositions), quasi drugs, foods, feeds, etc., and can be carried out according to the following description.
- the Lactococcus bacterium as an active ingredient of the present invention has an effect of improving skin condition, it is ingested or administered to subjects whose skin is damaged by daily life, outdoor activities, etc., or subjects who are easily damaged. can do.
- the ingestion target and the administration target are not limited to humans, and may be mammals other than humans (domestic animals such as cows and horses and pets such as dogs and cats).
- the Lactococcus genus bacterium which is an active ingredient of the present invention When the Lactococcus genus bacterium which is an active ingredient of the present invention is provided as a food, it can be provided as it is as a food, or it can be provided by containing it in a food.
- the food thus provided is a food containing an effective amount of the active ingredient of the present invention.
- “containing an effective amount” of the active ingredient of the present invention means that the active ingredient of the present invention is ingested within the range described later when an amount normally consumed in each food is ingested.
- Food means health food, functional food, dietary supplement, health functional food (eg, food for specified health use, nutritional functional food, functional indication food), special-purpose food (eg, infant food, pregnant woman) Food, food for patients) and supplements.
- the food according to the present invention is used as a feed when a non-human animal ingests the Lactococcus bacterium, which is an active ingredient of the present invention. That is, in the present invention, “food” is used to mean “feed”.
- the Lactococcus bacterium which is an active ingredient of the present invention, has an effect of improving the skin condition, and therefore can be contained in foods taken daily or can be provided as a supplement. That is, the composition and preparation of the present invention can be provided in the form of food. In this case, the composition and the preparation of the present invention can be provided in a unit package form in which the amount taken per serving is predetermined. Examples of the unit packaging form per serving include a form in which a certain amount is specified by a pack, packaging, can, bottle, or the like. In order to make the various effects of the composition and agent of the present invention better, the intake per serving can be determined according to the intake per lactococcus bacterium described later.
- the food item of the present invention may be provided together with a document or the like on which the explanation items regarding the intake amount are displayed on the package or the explanation items are described.
- the effective intake per day is divided into two or more (preferably two or three times). It may be the intake amount. Therefore, in the unit packaging form of the composition and the agent of the present invention, a lactococcus bacterium can be blended with the amount of human daily intake described later, or 2 minutes of human daily daily intake described later. Lactococcus bacteria can be blended in an amount of 1/3 or 1/3.
- the composition and the preparation of the present invention are preferably provided in “unit package form per meal” in which the intake per meal is the effective intake per day for convenience of intake.
- the form of “food” is not particularly limited, and may be, for example, a beverage form, a semi-liquid or gel form, or a solid or powder form.
- Examples of the “supplement” include tablets manufactured by adding an excipient, a binder and the like to the active ingredient of the present invention and kneading them, and capsules enclosed in capsules.
- the food provided by the present invention is not particularly limited as long as it contains the active ingredient of the present invention.
- soft drinks, carbonated drinks, fruit juice drinks, vegetable juice drinks, fruit juices and vegetable juices Non-alcoholic beverages such as beverages, milk, soy milk, milk beverages, drink-type yogurt, drink-type jelly, coffee, cocoa, tea beverages, energy drinks, sports drinks, mineral water, near water; rice, noodles, Foods and beverages containing carbohydrates such as breads and pasta; Western confectionery such as cookies, cakes and chocolates, Japanese confectionery such as buns and sheepskin, candy, gums, yogurt, jelly and pudding, frozen confectionery, ice confectionery, snack confectionery, etc.
- the mineral water includes both foaming and non-foaming mineral water.
- Tea beverages include fermented tea, semi-fermented tea and non-fermented tea, for example, black tea, green tea, barley tea, brown rice tea, sencha tea, gyokuro tea, roasted tea, oolong tea, turmeric tea, puer tea, rooibos tea. , Rose tea, chrysanthemum tea, herbal tea (for example, mint tea, jasmine tea).
- fruits used in fruit juice-containing beverages and fruit and vegetable juice-containing beverages include apples, mandarin oranges, grapes, bananas, pears, peaches, mangoes, acais, blueberries, and ume.
- vegetables used in vegetable juice-containing beverages, fruit juices, and vegetable juice-containing beverages include tomato, carrot, celery, pumpkin, cucumber, and watermelon.
- the intake of the Lactococcus genus bacterium which is the active ingredient of the present invention, can be determined depending on the sex, age and weight, symptom, intake time, dosage form, administration route, combined drug and the like of the intake subject.
- the daily intake amount of human beings is, for example, 0.5 to 1000 mg, preferably 5 to 500 mg, more preferably 10 to 10 times as dry cells. It can be 300 mg, more preferably 10 to 100 mg, particularly preferably about 50 mg.
- the daily intake amount of humans is, for example, 1 ⁇ 10 8 to 1 ⁇ 10 14 , preferably 1 ⁇ 10 9 to The number may be 1 ⁇ 10 13 , more preferably 1 ⁇ 10 10 to 1 ⁇ 10 12 , and particularly preferably about 1 ⁇ 10 11 .
- the above-mentioned effective ingestion amount may be ingested once a day, or may be ingested in several times.
- the intake amount and intake timing of the above Lactococcus bacterium and the following intake period are applicable when the Lactococcus bacterium as an active ingredient of the present invention is used for any non-therapeutic purpose or therapeutic purpose, For therapeutic purposes, intake can be read as administration.
- compositions and the preparation of the present invention can exert their effects better by long-term ingestion, for example, can be continuously ingested for 3 days or more, preferably 6 days or more, more preferably 10 days or more. Can be ingested continuously.
- “continuously” means to continue daily intake.
- an effective intake for a certain period for example, one week
- compositions and the preparation of the present invention use lactic acid bacteria, which are food materials that human beings have been eating for many years, as active ingredients, so there is no concern about side effects even if they are used continuously, and they are highly safe. For this reason, when the composition and agent of the present invention are used in combination with an existing skin condition improving agent, the dose of the existing drug can be reduced, and side effects of the existing drug can be reduced or eliminated.
- the composition and the agent of the present invention or Lactococcus bacterium
- compositions, the preparation and the food of the present invention may be marked with an indication of having an effect of improving the skin condition.
- the composition, the preparation and the food of the present invention may be given the following part or all of the following indications.
- "improvement of a skin state" is used by the meaning containing the following displays in this invention. ⁇ For those who are concerned about sunburn ⁇ For those who are concerned about skin damage in daily life ⁇ For those who are concerned about dryness of their skin ⁇ For those who are concerned about hot flashes ⁇ I am concerned about erythema on my skin For those who are concerned about the redness of their skin
- a method for improving skin condition comprising ingesting or administering an effective amount of a Lactococcus bacterium or a composition comprising the same to a subject in need thereof.
- treatment or prevention of photodermatoses which comprises ingesting or administering an effective amount of a Lactococcus bacterium or a composition comprising the same to a subject in need thereof.
- the subject of ingestion or administration is a mammal including a human, preferably a human.
- the skin condition improving method of the present invention and the treatment, prevention, and improving method of the present invention can be carried out according to the description of the composition and agent of the present invention and the active ingredient of the present invention.
- a bacterium belonging to the genus Lactococcus or a composition containing the same for the manufacture of a composition for improving skin condition or for the manufacture of an agent for improving skin condition.
- the genus Lactococcus for the manufacture of a composition for the treatment, prevention or amelioration of photodermatoses, or for the manufacture of a therapeutic, preventive or amelioration agent for photodermatoses.
- a bacterium or a composition comprising it. The use of the present invention can be carried out according to the description of the composition and agent of the present invention and the active ingredient of the present invention.
- the present invention also provides the use of a Lactococcus bacterium or a composition comprising the same for improving the skin condition, as an agent for improving the skin condition, or in the method for improving the skin condition of the present invention.
- a Lactococcus bacterium or a composition comprising the same for improving the skin condition, as an agent for improving the skin condition, or in the method for improving the skin condition of the present invention.
- it is also possible to treat, prevent or ameliorate photodermatological disease, as a therapeutic agent, prophylactic or ameliorating agent for photodermatoses, or in the treatment, prevention or amelioration method of the present invention.
- Use of a Lactococcus bacterium or a composition comprising the same is provided.
- the use of the present invention can be carried out according to the description of the composition and agent of the present invention and the active ingredient of the present invention.
- the present invention further provides a Lactococcus bacterium or a composition containing the same for use in improving the skin condition or for use in the method for improving the skin condition of the present invention.
- the present invention also provides a bacterium belonging to the genus Lactococcus or a composition containing the same for use in the treatment, prevention or amelioration of photodermatoses, or for use in the treatment, prevention or amelioration method of the present invention. Is done.
- the Lactococcus bacterium of the present invention and the composition containing the same can be implemented according to the description of the composition and agent of the present invention and the active ingredient of the present invention.
- non-therapeutic means not including the act of operating, treating, or diagnosing a human (that is, a medical act on a human), and specifically, receiving a doctor or doctor's instructions It means that the person does not include a method of performing surgery, treatment or diagnosis on humans.
- Example 1 Effect of improving skin damage due to ingestion of lactic acid bacteria (JCM5805) (1) Test method A. Grouping Four-week-old female hairless mice (HOS: HR-1, Nippon SLC Co., Ltd.) solid feed AIN-93G (Oriental) Yeast Kogyo Co., Ltd.) was fed freely and acclimated for 7 days. Acclimatized mice are divided into 4 groups, the group that receives normal food without UV irradiation is referred to as “non-irradiated group” (6 animals), and the group that receives UV light and receives normal food is “normal food group” (6 animals).
- non-irradiated group 6 animals
- normal food group 6 animals
- Lactococcus lactis ssp lactis JCM5805 with ultraviolet irradiation Lactococcus lactis subsp. lactis JCM5805 ( hereinafter, the group gives a) of "JCM5805" in the diet as “JCM5805” group (6 rats), UV irradiation
- the group to which ⁇ -tocopherol was fed with diet was designated as “ ⁇ -TOC group” (6 animals), and the test was started.
- the average body weight of each group at the start of the test was 17.86 ⁇ 1.67 g in the non-irradiated group, 18.89 ⁇ 0.89 g in the normal diet group, 18.48 ⁇ 0.66 g in the JCM5805 group, and the ⁇ -TOC group was 18.26 ⁇ 1.05 g, and no significant difference was confirmed between the groups.
- Vitamin E has been shown to reduce UV-induced skin damage (J. Agric. Food Chem., 2010, 58, 7013-7020, J. Nutr. Sci. Vitaminol. 54, 117- 123, 2008), ⁇ -tocopherol, one of vitamin E, was used as a positive control. During the test period, mice were housed in individual cages.
- Test feed At the start of the test, AIN-93G was freely fed to the non-irradiated group and the normal diet group as the test feed, and 1 mg of JCM5805 per animal per day was dried to the JCM5805 group and ⁇ -TOC group, respectively. Bacteria or 2.5 mg of ⁇ -tocopherol (manufactured by Wako Pure Chemical Industries, Ltd.) was freely fed with AIN-93G. The feeding period of the test feed was 17 days.
- UVB ultraviolet rays
- an oven type ultraviolet irradiation device DF-312 Donafix manufactured by ATTO
- the erythema value was determined by measuring the density of hemoglobin, which is known as an index of redness and blood color of the skin, using a Megzometer MX18 (Courage & Khazaka).
- the meggermeter MX18 measures the reflected light from the skin when irradiated with light having wavelengths of 568 nm and 660 nm from the measurement probe applied to the skin, and the measured value is displayed as a relative value.
- the erythema value was measured seven times for each mouse, and the average value of five values excluding the maximum value and the minimum value was calculated, and this was used as the measurement value.
- the skin moisture content was measured using a Corneometer CM825 (Courage & Khazaka).
- the Corneometer CM825 measures the amount of water contained in 15 ⁇ m (mainly stratum corneum) from the skin surface as the capacitance due to the electric field generated in the skin through the probe tip by the capacitance method, and the measured value is a relative value. Is displayed.
- the skin moisture content was measured seven times for each mouse, and the average value of five values excluding the maximum and minimum values was calculated and used as the measured value.
- Skin moisture transpiration was measured using a Tevameter TM300 (Courage & Khazaka).
- the Tevameter TM300 measures the temperature difference and humidity difference of moisture passing through the temperature / humidity sensor arranged in the probe tip, and measures the skin moisture transpiration (g / h / m 2 ) from the measured value.
- the final value when the measurement using the tevameter was automatically completed was taken as the measurement value.
- JCM5805 dry dead cells have an improvement effect on skin damage typified by ultraviolet irradiation, and may have an improvement effect on skin damage that usually occurs in daily life. Indicated.
- Example 2 Effect of improving skin condition by ingestion of lactic acid bacteria (JCM5805) (1) Preparation of food containing lactic acid bacteria Hard capsules containing 50 mg (1.0 ⁇ 10 11 or more) of dried dead cells of JCM5805 and 150 mg of corn starch as food containing lactic acid bacteria was made. A hard capsule containing 200 mg of corn starch without JCM5805 was prepared as a placebo food.
- Test method The subjects were 70 Japanese men and women aged 30 to 60 who are prone to skin problems. Randomly divide subjects into two groups so as not to show bias in gender, age, BMI, doctor's skin symptom assessment (texture visual assessment, skin quality assessment, wound-related assessment), etc. A “test group” (9 males, 26 females, 35 in total) was used, and a group fed with placebo food was designated as a “control group” (10 males, 25 females, 35 in total).
- the test group contains the lactic acid bacteria-containing food prepared in (1) above, the control group contains the placebo food, and each capsule contains 8 capsules once daily. Ingestion continued for a week (56 days). During the study period, subjects continued their pre-test lifestyle.
- the skin state before and after the test food intake was evaluated using the change in skin redness as an index. Specifically, before the start of the test food intake period (8 weeks) (hereinafter sometimes referred to as “before test food intake”) and when the test food intake period ends (hereinafter referred to as “after test food intake”).
- the measurement probe is applied to the center of the subject's left face connecting the lower armpit and the lip, and the color difference and hemoglobin amount index (hereinafter also referred to as “Hb index”).
- the color difference and the Hb index were measured using a spectrocolorimeter (CM-2600d, manufactured by Konica Minolta) and analyzed with analysis software (CM-SA, manufactured by Konica Minolta).
- L * value is a parameter representing brightness. When the measured value is high, it is close to white (bright), when the measured value is low, it is close to black (dark), and the a * value is red to green.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Mycology (AREA)
- General Health & Medical Sciences (AREA)
- Microbiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Zoology (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Nutrition Science (AREA)
- Wood Science & Technology (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Physiology (AREA)
- Immunology (AREA)
- Biochemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Food Science & Technology (AREA)
- General Engineering & Computer Science (AREA)
- Virology (AREA)
- Toxicology (AREA)
- Dermatology (AREA)
- Biomedical Technology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Tropical Medicine & Parasitology (AREA)
- Polymers & Plastics (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
[1]ラクトコッカス属細菌を有効成分として含んでなる、皮膚状態の改善用組成物および皮膚状態の改善剤。
[2]皮膚状態が、光暴露により悪化した皮膚状態である、上記[1]に記載の組成物および用剤。
[3]光暴露により悪化した皮膚状態が、皮膚水分量の低下および/または皮膚の赤みの増加である、上記[1]または[2]に記載の組成物および用剤。
[4]ラクトコッカス属細菌を死菌の形態で含む、上記[1]~[3]のいずれかに記載の組成物および用剤。
[5]ラクトコッカス属細菌が、ラクトコッカス・ラクティスである、上記[1]~[4]のいずれかに記載の組成物および用剤。
[6]ラクトコッカス属細菌が、ラクトコッカス・ラクティス・サブスピーシズ・ラクティスJCM5805である、上記[1]~[5]のいずれかに記載の組成物および用剤。
[7]ヒト1日当たりの有効摂取量のラクトコッカス属細菌を含んでなる、上記[1]~[6]のいずれかに記載の組成物および用剤。
[8]ヒト1日当たりの有効摂取量が、乾燥菌体として0.5~1000mgである、上記[7]に記載の組成物および用剤。
[9]ヒト1日当たりの有効摂取量が、菌数として1×108~1×1014個である、上記[7]に記載の組成物および用剤。
[10]単位包装形態である、上記[1]~[9]のいずれかに記載の組成物および用剤。
[11]食品組成物である、上記[1]~[10]のいずれかに記載の組成物および用剤。
[12]有効量のラクトコッカス属細菌またはそれを含む組成物を、それを必要とする対象に摂取させるか、あるいは投与することを含んでなる、皮膚状態の改善方法。
[13]皮膚状態の改善剤の製造のための、皮膚状態の改善剤としての、あるいは、上記[12]に記載の方法における、ラクトコッカス属細菌またはそれを含む組成物の使用。
[14]皮膚状態の改善に用いるための、あるいは、上記[12]の方法に用いるための、ラクトコッカス属細菌またはそれを含む組成物。
[15]ラクトコッカス属細菌を有効成分として含んでなる、光線性皮膚疾患の治療、予防および改善用組成物並びに光線性皮膚疾患の治療剤、予防剤および改善剤。
[16]有効量のラクトコッカス属細菌またはそれを含む組成物を、それを必要とする対象に摂取させるか、あるいは投与することを含んでなる、光線性皮膚疾患の治療、予防および改善方法。
[17]光線性皮膚疾患の治療剤、予防剤または改善剤の製造のための、光線性皮膚疾患の治療剤、予防剤または改善剤としての、あるいは、上記[16]に記載の方法における、ラクトコッカス属細菌またはそれを含む組成物の使用。
[18]光線性皮膚疾患の治療、予防または改善に用いるための、あるいは、上記[16]の方法に用いるための、ラクトコッカス属細菌またはそれを含む組成物。
・日焼けの気になる方に
・日常生活の肌へのダメージが気になる方に
・肌の乾燥が気になる方に
・肌のほてりが気になる方に
・肌の紅斑が気になる方に
・肌の赤みが気になる方に
・赤ら顔の気になる方に
・手荒れが気になる方に
(1)試験方法
ア 群分け
4週齢の雌へアレスマウス(HOS:HR-1、日本エスエルシー社)に固形飼料AIN-93G(オリエンタル酵母工業社製)を自由摂食させて7日間馴化飼育した。馴化後のマウスを4群に分け、紫外線照射を行わず通常食を与える群を「非照射群」(6匹)とし、紫外線照射を行い通常食を与える群を「通常食群」(6匹)とし、紫外線照射を行いラクトコッカス・ラクティス・サブスピーシズ・ラクティスJCM5805(Lactococcus lactis subsp. lactis JCM5805)(以下、「JCM5805」という)を混餌で与える群を「JCM5805群」(6匹)とし、紫外線照射を行いα-トコフェロールを混餌で与える群を「α-TOC群」(6匹)とし、試験を開始した。試験開始時の各群の平均体重は、非照射群が17.86±1.67g、通常食群が18.89±0.89g、JCM5805群が18.48±0.66g、α-TOC群が18.26±1.05gであり、群間で有意な差は確認されなかった。ビタミンEは紫外線によって誘導される皮膚障害を軽減する効果が認められていることから(J. Agric. Food Chem., 2010, 58, 7013-7020、J. Nutr. Sci. Vitaminol. 54, 117-123,2008)、ビタミンEの一つであるα-トコフェロールを陽性対照とした。試験期間中、マウスは個別ケージで飼育した。
試験開始とともに、被験飼料として、非照射群および通常食群にはAIN-93Gを自由摂食させ、JCM5805群およびα-TOC群にはそれぞれ1日1匹あたり1mgのJCM5805の乾燥死菌体または2.5mgのα-トコフェロール(和光純薬社製)をAIN-93Gとともに自由に混餌摂食させた。被験飼料の摂食期間は17日間とした。
被験飼料の摂食開始日から起算して14日目にマウスに紫外線を照射した。具体的には、線量として90mJ/cm2(本試験の線量)に相当する量の波長312nmの紫外線(UVB)を単回で照射した。照射の光源としては、オーブン型紫外線照射装置DF-312ドナフィクス(ATTO社製)を用いた。札幌における冬季の日積算UV-B量(2kJ/m2:気象庁ウェブサイトhttp://www.data.jma.go.jp/gmd/env/uvhp/uvb_monthave_sap.html)に鑑みると、本試験の線量である90mJ/cm2(900J/m2)相当量は、日常生活で受ける紫外線量と同程度であるといえる。
紫外線照射前および紫外線照射から3日後(紫外線照射3日後)に、マウス尻尾の付根から2cmの背部正中線付近に測定プローブを当てて、マウス背部の紅斑値、皮膚水分量および皮膚水分蒸散量を測定した。具体的な測定方法は後記の通りである。なお、各プローブの接続にはマルチプローブアダプター(MPA5、Courage&Khazaka社製)を使用した。
測定した紅斑値、皮膚水分量、皮膚水分蒸散量について、一段階多重比較検定であるTukey-Kramer法を用いて、4群間で有意差検定を行い評価した。有意水準は5%未満とした。
結果は、図2~4に示される通りであった。図2に示される通り、通常食群では紫外線照射3日後において、非照射群と比較して紅斑値の有意な上昇がみられたが、JCM5805群およびα-TOC群では、通常食群と比較して紅斑値の上昇が有意に抑制された。図3に示される通り、通常食群では紫外線照射3日後において、非照射群と比較して皮膚水分量の有意な低下がみられたが、JCM5805群およびα-TOC群では、通常食群と比較して皮膚水分量の低下が有意に抑制された。図4に示される通り、通常食群では紫外線照射3日後において、非照射群と比較して皮膚水分蒸散量の有意な上昇がみられたが、JCM5805群およびα-TOC群では、通常食群と比較して皮膚水分蒸散量の上昇が有意に抑制された。
(1)乳酸菌含有食品の調製
乳酸菌含有食品として、JCM5805の乾燥死菌体50mg(1.0×1011個以上)とコーンスターチ150mgを含むハードカプセルを作製した。プラセボ食品として、JCM5805を含まず200mgのコーンスターチを含むハードカプセルを作製した。
皮膚のトラブルが起こりやすい30歳以上60歳未満の日本人男女70名を被験者とした。被験者を性別、年齢、BMI、医師による肌症状評価(キメ目視評価、肌質評価、挫創関連評価)等に偏りを示さないようにランダムに2群に分け、乳酸菌含有食品を摂取させた群を「試験群」(男性9名、女性26名、合計35名)とし、プラセボ食品を摂取させた群を「対照群」(男性10名、女性25名、合計35名)とした。
被験食品摂取前後の肌の状態として皮膚の赤みの変化を指標として評価した。具体的には、被験食品の摂取期間(8週間)の開始前(以下、「被験食品摂取前」ということがある。)および被験食品の摂取期間が終了した時(以下、「被験食品摂取後」ということがある。)に、被験者の左顔の耳朶下と唇端とを結んだ中心部に測定プローブを当てて、色差およびヘモグロビン量指数(以下、「Hbインデックス」ということがある。)を測定した。色差およびHbインデックスは、分光測色計(CM-2600d、コニカミノルタ社製)を用いて測定し、解析ソフトウェア(CM-SA、コニカミノルタ社製)で解析した。各被験者の同一箇所について5回実施し、L*値、a*値、b*値およびHbインデックスを得た。5回の測定のうちL*値が最大または最小となった測定回を除いた3回分の各値の平均値を算出し、これを測定値とした。被験食品摂取前後の測定値を対応のあるt検定により評価した。ここで、L*値は明るさを表すパラメータであり、測定値が高いと白(明るい)に近いことを、測定値が低いと黒(暗い)に近いことを、a*値は赤~緑を表すパラメータであり、測定値が高いと赤に近いことを、測定値が低いと緑に近いことを、b*値は黄~青を表すパラメータであり、測定値が高いと黄に近いことを、測定値が低いと青に近いことを示すことが知られている。また、Hbインデックスは、肌の赤みを示す指標であることが知られている。
試験期間中に2名の被験者が脱落したため、試験群33名および対照群35名の被験者を解析対象者とした。結果は、表1に示される通りであった。
Claims (14)
- ラクトコッカス属細菌を有効成分として含んでなる、皮膚状態の改善用組成物。
- 皮膚状態が、光暴露により悪化した皮膚状態である、請求項1に記載の組成物。
- 光暴露により悪化した皮膚状態が、皮膚水分量の低下および/または皮膚の赤みの増加である、請求項1または2に記載の組成物。
- ラクトコッカス属細菌を死菌の形態で含む、請求項1~3のいずれか一項に記載の組成物。
- ラクトコッカス属細菌が、ラクトコッカス・ラクティスである、請求項1~4のいずれか一項に記載の組成物。
- ラクトコッカス属細菌が、ラクトコッカス・ラクティス・サブスピーシズ・ラクティスJCM5805である、請求項1~5のいずれか一項に記載の組成物。
- ヒト1日当たりの有効摂取量のラクトコッカス属細菌を含んでなる、請求項1~6のいずれか一項に記載の組成物。
- ヒト1日当たりの有効摂取量が、乾燥菌体として0.5~1000mgである、請求項7に記載の組成物。
- ヒト1日当たりの有効摂取量が、菌数として1×108~1×1014個である、請求項8に記載の組成物。
- 単位包装形態である、請求項1~9のいずれか一項に記載の組成物。
- 食品組成物である、請求項1~10のいずれか一項に記載の組成物。
- 有効量のラクトコッカス属細菌またはそれを含む組成物を、それを必要とする対象に摂取させるか、あるいは投与することを含んでなる、皮膚状態の改善方法。
- 皮膚状態の改善剤の製造のための、皮膚状態の改善剤としての、あるいは、請求項12に記載の方法における、ラクトコッカス属細菌またはそれを含む組成物の使用。
- 皮膚状態の改善に用いるための、あるいは、請求項12の方法に用いるための、ラクトコッカス属細菌またはそれを含む組成物。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SG11202008959RA SG11202008959RA (en) | 2018-03-15 | 2019-03-15 | Composition for improving skin conditions |
| EP19767482.3A EP3766363B1 (en) | 2018-03-15 | 2019-03-15 | Composition for improving skin conditions |
| US16/980,714 US11497778B2 (en) | 2018-03-15 | 2019-03-15 | Composition for improving skin conditions |
| NZ768504A NZ768504B2 (en) | 2019-03-15 | Composition for improving skin conditions | |
| CN201980017850.3A CN111818812B (zh) | 2018-03-15 | 2019-03-15 | 皮肤状态改善用组合物 |
| AU2019233471A AU2019233471B2 (en) | 2018-03-15 | 2019-03-15 | Composition for improving skin conditions |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018-048311 | 2018-03-15 | ||
| JP2018048311A JP6846376B2 (ja) | 2018-03-15 | 2018-03-15 | 皮膚状態改善用組成物 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019177139A1 true WO2019177139A1 (ja) | 2019-09-19 |
Family
ID=67906705
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/010773 Ceased WO2019177139A1 (ja) | 2018-03-15 | 2019-03-15 | 皮膚状態改善用組成物 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US11497778B2 (ja) |
| EP (1) | EP3766363B1 (ja) |
| JP (1) | JP6846376B2 (ja) |
| CN (1) | CN111818812B (ja) |
| AU (1) | AU2019233471B2 (ja) |
| SG (1) | SG11202008959RA (ja) |
| WO (1) | WO2019177139A1 (ja) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018051895A1 (ja) * | 2016-09-15 | 2018-03-22 | キリン株式会社 | 乳酸菌を有効成分として含む、皮膚における特定細菌の異常増殖に起因する皮膚の状態の悪化を防止又は改善するための組成物 |
| KR102754494B1 (ko) * | 2020-10-30 | 2025-01-14 | 중앙대학교 산학협력단 | 락토코커스 중앙젠시스를 포함하는 피부 손상 예방 또는 치료용 조성물 |
| JP2022145446A (ja) * | 2021-03-19 | 2022-10-04 | キリンホールディングス株式会社 | 紙臭が抑制された容器詰茶飲料、及びその製造方法 |
| JP7248250B2 (ja) * | 2021-04-28 | 2023-03-29 | キリンホールディングス株式会社 | 乳酸球菌の増殖促進剤 |
| JP2024065261A (ja) * | 2022-10-31 | 2024-05-15 | 日清食品ホールディングス株式会社 | 皮膚状態改善剤 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006069940A (ja) | 2004-09-01 | 2006-03-16 | Fujicco Co Ltd | ストレス由来の皮膚血流低下改善用組成物、皮膚機能改善用組成物およびそれらを用いた飲食品 |
| JP2013526265A (ja) * | 2009-05-11 | 2013-06-24 | ネステク ソシエテ アノニム | プロバイオティクス微生物を含有する朝食用シリアル |
| JP2013233097A (ja) * | 2012-05-07 | 2013-11-21 | Koiwai Nyugyo Kk | 風味良好なドリンクタイプヨーグルト |
| JP2017081838A (ja) * | 2015-10-26 | 2017-05-18 | キリン株式会社 | 抗原に対する特異的な細胞傷害性t細胞(ctl)及び抗体産生を増強させる方法 |
| JP2017534589A (ja) * | 2014-09-23 | 2017-11-24 | コリア インスティテュート オブ オリエンタル メディシン | ティンダル化乳酸菌死菌体を有効成分として含む皮膚保湿又はシワ改善用組成物 |
| WO2018051895A1 (ja) * | 2016-09-15 | 2018-03-22 | キリン株式会社 | 乳酸菌を有効成分として含む、皮膚における特定細菌の異常増殖に起因する皮膚の状態の悪化を防止又は改善するための組成物 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5527690B2 (ja) * | 2007-11-19 | 2014-06-18 | 株式会社明治 | 免疫調節性機能誘導剤及び食品組成物 |
| EP2586448B1 (en) * | 2010-06-28 | 2018-02-07 | Kabushiki Kaisha Yakult Honsha | Skin properties improving agent for oral administration |
| JP5950827B2 (ja) * | 2010-12-28 | 2016-07-13 | キリンホールディングス株式会社 | 乳酸菌を含むインターフェロン産生誘導剤 |
-
2018
- 2018-03-15 JP JP2018048311A patent/JP6846376B2/ja active Active
-
2019
- 2019-03-15 CN CN201980017850.3A patent/CN111818812B/zh active Active
- 2019-03-15 EP EP19767482.3A patent/EP3766363B1/en active Active
- 2019-03-15 AU AU2019233471A patent/AU2019233471B2/en active Active
- 2019-03-15 SG SG11202008959RA patent/SG11202008959RA/en unknown
- 2019-03-15 US US16/980,714 patent/US11497778B2/en active Active
- 2019-03-15 WO PCT/JP2019/010773 patent/WO2019177139A1/ja not_active Ceased
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006069940A (ja) | 2004-09-01 | 2006-03-16 | Fujicco Co Ltd | ストレス由来の皮膚血流低下改善用組成物、皮膚機能改善用組成物およびそれらを用いた飲食品 |
| JP2013526265A (ja) * | 2009-05-11 | 2013-06-24 | ネステク ソシエテ アノニム | プロバイオティクス微生物を含有する朝食用シリアル |
| JP2013233097A (ja) * | 2012-05-07 | 2013-11-21 | Koiwai Nyugyo Kk | 風味良好なドリンクタイプヨーグルト |
| JP2017534589A (ja) * | 2014-09-23 | 2017-11-24 | コリア インスティテュート オブ オリエンタル メディシン | ティンダル化乳酸菌死菌体を有効成分として含む皮膚保湿又はシワ改善用組成物 |
| JP2017081838A (ja) * | 2015-10-26 | 2017-05-18 | キリン株式会社 | 抗原に対する特異的な細胞傷害性t細胞(ctl)及び抗体産生を増強させる方法 |
| WO2018051895A1 (ja) * | 2016-09-15 | 2018-03-22 | キリン株式会社 | 乳酸菌を有効成分として含む、皮膚における特定細菌の異常増殖に起因する皮膚の状態の悪化を防止又は改善するための組成物 |
Non-Patent Citations (3)
| Title |
|---|
| HELSINKI: "Ethical Guidelines for Medical and Health Research Involving Human Subjects", MINISTRY OF EDUCATION, CULTURE, SPORTS, SCIENCE AND TECHNOLOGY |
| J. AGRIC. FOOD CHEM., vol. 58, 2010, pages 7013 - 7020 |
| J. NUTR.SCI. VITAMINOL., vol. 54, 2008, pages 117 - 123 |
Also Published As
| Publication number | Publication date |
|---|---|
| SG11202008959RA (en) | 2020-10-29 |
| AU2019233471A1 (en) | 2020-10-22 |
| NZ768504A (en) | 2025-08-29 |
| US11497778B2 (en) | 2022-11-15 |
| EP3766363A4 (en) | 2022-01-05 |
| JP2019156793A (ja) | 2019-09-19 |
| US20210008127A1 (en) | 2021-01-14 |
| AU2019233471B2 (en) | 2024-03-28 |
| JP6846376B2 (ja) | 2021-03-24 |
| CN111818812A (zh) | 2020-10-23 |
| CN111818812B (zh) | 2023-09-26 |
| EP3766363B1 (en) | 2026-04-15 |
| EP3766363A1 (en) | 2021-01-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019177139A1 (ja) | 皮膚状態改善用組成物 | |
| US10441618B2 (en) | Moringa extract | |
| JP2017534589A (ja) | ティンダル化乳酸菌死菌体を有効成分として含む皮膚保湿又はシワ改善用組成物 | |
| JP7041508B2 (ja) | 概日リズム改善用組成物 | |
| US12016890B2 (en) | Composition for preventing or treating menopause, containing Lactobacillus intestinalis | |
| AU2017337936A1 (en) | Novel Lactobacillus Sakei And Composition Comprising The Same | |
| KR102122652B1 (ko) | 락토바실러스 루테리 제제를 이용한 피부개선용 조성물 | |
| KR20210063728A (ko) | 장기능 개선용 발효 키위 분말 및 이의 제조방법 | |
| Kenig et al. | Helichrysum italicum (Roth) G. Don and Helichrysum arenarium (L.) Moench infusions in reversing the traits of metabolic syndrome: a double-blind randomized comparative trial | |
| KR101669362B1 (ko) | 발효 태반 조성물을 유효성분으로 하는 면역 증강 또는 항피로 조성물과 그의 용도 | |
| JP6482930B2 (ja) | 皮膚化粧料および飲食品 | |
| dos Santos Baião et al. | A new functional beetroot formulation enhances adherence to nitrate supplementation and health outcomes in clinical practice | |
| JP2009107965A (ja) | セラミド合成促進剤、並びに皮膚外用剤及び飲食品 | |
| CN100534446C (zh) | 护肤用口服组合物 | |
| JP2022089939A (ja) | 肝機能改善剤 | |
| Sharma et al. | Honey as Potential Cosmeceutical Agent and Functional Food | |
| JP7154473B2 (ja) | 疲労回復用および/または疲労蓄積予防用組成物 | |
| JP7206623B2 (ja) | 糖代謝異常の予防および改善用組成物 | |
| KR20180075763A (ko) | 마치현 주정 추출물을 유효성분으로 함유하는 알콜성 간손상 예방, 개선 및 치료용 조성물 | |
| JP7112688B2 (ja) | 花粉症を予防及び/又は改善するための組成物 | |
| JP2018070569A (ja) | 肝星細胞の活性化抑制剤及び肝星細胞の活性化抑制用食品組成物 | |
| HK40042697A (en) | Composition for improving skin conditions | |
| Raza et al. | Development of anti-aging toffee by incorporating Beta vulgaris, Punica granatum and Saccharam officinarum and study of its acceptability and stability | |
| TW200417366A (en) | Remedy | |
| JP2026065722A (ja) | Trpv1活性化剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19767482 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2019767482 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2019767482 Country of ref document: EP Effective date: 20201015 |
|
| ENP | Entry into the national phase |
Ref document number: 2019233471 Country of ref document: AU Date of ref document: 20190315 Kind code of ref document: A |
|
| WWG | Wipo information: grant in national office |
Ref document number: 11202008959R Country of ref document: SG |
|
| WWP | Wipo information: published in national office |
Ref document number: 11202008959R Country of ref document: SG |
|
| WWG | Wipo information: grant in national office |
Ref document number: 768504 Country of ref document: NZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2019767482 Country of ref document: EP |