WO2021199587A1 - 二次電池用負極活物質およびこれを用いた二次電池 - Google Patents
二次電池用負極活物質およびこれを用いた二次電池 Download PDFInfo
- Publication number
- WO2021199587A1 WO2021199587A1 PCT/JP2021/001564 JP2021001564W WO2021199587A1 WO 2021199587 A1 WO2021199587 A1 WO 2021199587A1 JP 2021001564 W JP2021001564 W JP 2021001564W WO 2021199587 A1 WO2021199587 A1 WO 2021199587A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- silicon
- negative electrode
- secondary battery
- active material
- phase
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/364—Composites as mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/366—Composites as layered products
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
- H01M4/386—Silicon or alloys based on silicon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/485—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of mixed oxides or hydroxides for inserting or intercalating light metals, e.g. LiTi2O4 or LiTi2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/46—Separators, membranes or diaphragms characterised by their combination with electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/027—Negative electrodes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
- This disclosure mainly relates to a negative electrode active material for a secondary battery.
- Patent Document 2 describes a lithium silicate phase represented by Li 2z SiO (2 + z) ⁇ 0 ⁇ z ⁇ 2 ⁇ , silicon particles dispersed in the lithium silicate phase, and dispersion in the lithium silicate phase.
- a negative electrode active material for a non-aqueous electrolyte secondary battery which comprises metal particles mainly composed of one or more metals or alloys selected from Fe, Pb, Zn, Sn, Cu, Ni and Cr. ing.
- Patent Document 2 in a non-aqueous electrolyte secondary battery using a silicon material as a negative electrode active material, the initial charge / discharge efficiency can be improved and the cycle life can be extended. However, the effect of extending the cycle life is not sufficient and there is room for improvement.
- One aspect of the present disclosure includes a silicon-containing material, which comprises a lithium ion conductive phase, silicon particles dispersed in the lithium ion conductive phase, and vanadium dispersed in the lithium ion conductive phase.
- Another aspect of the present disclosure relates to a secondary battery comprising a positive electrode, a negative electrode, an electrolyte and a separator interposed between the positive electrode and the negative electrode, wherein the negative electrode contains the negative electrode active material for the secondary battery.
- FIG. 1 is a cross-sectional view schematically showing a negative electrode active material (silicon-containing material) for a secondary battery according to an embodiment of the present disclosure.
- FIG. 2 is a schematic perspective view in which a part of the secondary battery according to the embodiment of the present disclosure is cut out.
- the negative electrode active material for a secondary battery includes a silicon-containing material. Further, the silicon-containing material includes a lithium ion conductive phase, silicon particles dispersed in the lithium ion conductive phase, and vanadium-containing particles dispersed in the lithium ion conductive phase.
- the silicon-containing material has a so-called sea-island structure.
- the silicon particles (islands) are dispersed in the matrix (sea) of the lithium ion conductive phase and are covered with the lithium ion conductive phase.
- the contact between the silicon particles and the electrolyte is restricted, so side reactions are suppressed.
- the stress generated by the expansion and contraction of the silicon particles is relaxed by the matrix of the lithium ion conductive phase.
- particles containing vanadium are dispersed in the lithium ion conductive phase. Vanadium improves the particle strength of silicon-containing materials. Vanadium is considered to form a high-hardness microregion within the lithium-ion conduction phase. By uniformly distributing high-hardness minute regions in the matrix of the lithium ion conductive phase, expansion and contraction of the silicon-containing material due to charge and discharge are suppressed, and cracks occur in the silicon-containing material and particles collapse. Isolation of a part of the silicon-containing material is suppressed. Therefore, the cycle life of the secondary battery is greatly improved.
- Vanadium suppresses the expansion and contraction of the silicon-containing material due to charging and discharging, and suppresses the generation of cracks and the collapse of the particles of the silicon-containing material by simply adding a smaller amount than iron to the lithium-ion conductive phase.
- the content of vanadium contained in the silicon-containing material may be, for example, 1000 ppm or less, 500 ppm or less, 300 ppm or less, or 100 ppm or less.
- the content of vanadium contained in the silicon-containing material is preferably 5 ppm or more, preferably 10 ppm or more, or 30 ppm or more, from the viewpoint of increasing the effect of increasing the hardness of the lithium ion conductive phase.
- the content of vanadium contained in the silicon-containing material can be measured by, for example, inductively coupled plasma emission spectroscopy (ICP-AES). Specifically, a sample of a silicon-containing material is completely dissolved in a heated acid solution (mixed acid of hydrofluoric acid, nitric acid and sulfuric acid), carbon in the solution residue is filtered off, and then the obtained filtrate is obtained. Is analyzed by ICP-AES, and the spectral intensity of vanadium is measured. Subsequently, a calibration curve is prepared using a standard solution of commercially available elements, and the vanadium content is calculated.
- ICP-AES inductively coupled plasma emission spectroscopy
- vanadium may be contained in at least one selected from the group consisting of vanadium alloys and vanadium carbide. That is, the vanadium-containing particles may be an alloy containing vanadium, vanadium carbide, or the like. Among them, vanadium carbide has a Vickers hardness of about 2800, and has an effect of remarkably increasing the hardness of the lithium ion conductive phase.
- the particles containing vanadium may further contain iron. That is, the vanadium-containing particles may be formed of an alloy containing vanadium and iron, or an alloy containing vanadium carbide and iron.
- the alloy containing vanadium and iron may contain elemental vanadium and / or vanadium carbide in a dispersed state. Since iron is excellent in malleability, cracks and collapses of the silicon-containing material that may occur when charging and discharging are repeated can be remarkably suppressed. Therefore, the cycle life is greatly improved. Further, such an effect peculiar to iron is sufficiently exhibited even if the amount of iron is reduced when vanadium and iron are used in combination.
- the iron content in the silicon-containing material may be, for example, 0.01% by mass or more and 10% by mass or less, or 0.1% by mass or more and 3% by mass or less.
- the iron content contained in the silicon-containing material can be measured by ICP-AES in the same manner as vanadium, for example. Specifically, a sample of a silicon-containing material is completely dissolved in a heated acid solution, carbon in the solution residue is filtered off, and then the obtained filtrate is analyzed by ICP-AES to obtain the spectral intensity of iron. To measure. Subsequently, a calibration curve is prepared using a standard solution of commercially available elements, and the iron content is calculated.
- the average particle size of the silicon particles contained in the silicon-containing material may be, for example, 1 nm or more and 1000 nm or less.
- the average particle size of the silicon particles is preferably 500 nm or less, more preferably 200 nm or less, and even more preferably 50 nm or less. That is, the silicon particles may be fine particles.
- the smaller the average particle size of the silicon particles the smaller the volume change during charging and discharging, and the better the structural stability. In addition, the expansion and contraction of silicon particles are made uniform, and particle cracking is suppressed.
- the average particle size of the silicon particles is measured by observing the cross section of the silicon-containing material using SEM or TEM. Specifically, it is obtained by averaging the maximum diameters of any 100 silicon particles.
- the lithium ion conductive phase may be at least one selected from the group consisting of, for example, a silicon oxide phase, a silicate phase and a carbon phase.
- the silicate phase is suitable for obtaining a silicon-containing material having a small irreversible capacity at the initial stage of charge and discharge and a high capacity.
- the silicon oxide phase contains silicon dioxide as its main component (for example, 95 to 100% by mass).
- the composition of the composite material containing the silicon oxide phase and the silicon particles dispersed therein can be represented as SiO x as a whole.
- SiO x has a structure in which fine particles of silicon are dispersed in amorphous SiO 2.
- the oxygen content ratio x to silicon is, for example, 0.5 ⁇ x ⁇ 2.0, and more preferably 0.8 ⁇ x ⁇ 1.5.
- the carbon phase may be composed of, for example, amorphous carbon having low crystallinity (that is, amorphous carbon).
- amorphous carbon may be, for example, hard carbon, soft carbon, or other carbon.
- the silicate phase may contain at least one selected from the group consisting of alkali metal elements and Group II elements.
- the inclusion of such elements further significantly reduces the irreversible capacitance of the silicate phase.
- a silicate phase containing lithium (hereinafter, also referred to as a lithium silicate phase) is preferable because the irreversible capacity is small and the initial charge / discharge efficiency is high.
- the alkali metal element and Group II element for example, Li, K, Na, Mg, Ca, Sr, Ba and the like can be used.
- the silicate phase may further contain an element M other than the alkali metal element and the Group II element.
- the element M is selected from the group consisting of, for example, B, Al, Zr, Nb, Ta, La, Y, Ti, P, Bi, Zn, Sn, Pb, Sb, Co, Er, F and W. Can be at least one species.
- the silicate phase preferably contains a lithium silicate represented by the formula: Li 2z SiO 2 + z (0 ⁇ z ⁇ 2).
- Lithium silicate is lightweight and has excellent lithium ion conductivity.
- the lithium silicate may contain any other element as long as it is an oxide phase containing Li, Si and O.
- the atomic ratio of O to Si in the lithium silicate phase: O / Si is, for example, greater than 2 and less than 4.
- O / Si is greater than 2 and less than 3.
- the content of silicon particles in the silicon-containing material is preferably, for example, 30% by mass or more and 80% by mass or less from the viewpoint of increasing the capacity and improving the cycle characteristics. ..
- the content of the silicon particles is preferably, for example, 30% by mass or more and 80% by mass or less from the viewpoint of increasing the capacity and improving the cycle characteristics. ..
- the content of silicon particles in the silicon-containing material is preferably 40% by mass or more, more preferably 50% by mass or more.
- the composition of the silicate phase can be analyzed by the following method. It is desirable to analyze the composition using a silicon-containing material or a negative electrode mixture layer in a discharged state. Further, from the viewpoint of eliminating the influence of the decomposition products of the electrolyte, it is desirable to analyze the sample of the silicon-containing material in the battery before the charge / discharge cycle or at the beginning of the cycle.
- the content of B, Na, K and Al contained in the silicate layer can be determined by quantitative analysis in accordance with, for example, JIS R3105 (1995) (analysis method for silicic acid glass).
- the Ca content is determined by quantitative analysis in accordance with JIS R3101 (1995) (analysis method for soda-lime glass).
- the content of each element contained in the silicon-containing material can be measured by ICP-AES in the same manner as vanadium, for example. Specifically, a sample of a silicon-containing material is completely dissolved in a heated acid solution, carbon in the solution residue is filtered off, and then the obtained filtrate is analyzed by ICP-AES to analyze each element. Measure the spectral intensity. Subsequently, a calibration curve is prepared using a commercially available standard solution of each element, and the content of each element is calculated.
- the silicon-containing material may be taken out of the battery by, for example, the following method. Specifically, the battery is disassembled, the negative electrode is taken out, and the negative electrode is washed with anhydrous ethyl methyl carbonate or dimethyl carbonate to remove the electrolyte. Next, the negative electrode mixture layer is peeled off from the negative electrode current collector and pulverized in a mortar to obtain sample powder. Next, the sample powder is dried in a dry atmosphere for 1 hour and immersed in weakly boiled 6M hydrochloric acid for 10 minutes to remove alkali metals such as Na and Li that may be contained in the binder or the like.
- sample powder is washed with ion-exchanged water, separated by filtration, and dried at 200 ° C. for 1 hour. Then, by heating in an oxygen atmosphere to remove the carbon component, only the particles of the silicon-containing material can be isolated.
- the silicon-containing material may contain a silicate phase, a silicon oxide phase, silicon particles, and the like.
- Si-NMR By using Si-NMR, these can be distinguished and quantified.
- the Si content obtained by ICP-AES as described above is the sum of the amount of Si constituting the silicon particles, the amount of Si in the lithium ion conductive phase, and the amount of Si in the silicon oxide phase.
- the amount of Si constituting the silicon particles and the amount of Si in the silicon oxide phase can be separately quantified by using Si-NMR. Therefore, the amount of Si in the silicate phase can be quantified by subtracting the amount of Si constituting the silicon particles and the amount of Si in the silicon oxide phase from the Si content obtained by ICP-AES.
- a mixture containing silicate having a known Si content and silicon particles in a predetermined ratio may be used.
- Measuring device Solid-state nuclear magnetic resonance spectrum measuring device (INOVA-400) manufactured by Varian. Probe: Varian 7mm CPMAS-2 MAS: 4.2kHz MAS speed: 4kHz Pulse: DD (45 ° pulse + signal capture time 1H decouple) Repeat time: 1200 sec to 3000 sec Observation width: 100 kHz Observation center: Around -100ppm Signal capture time: 0.05sec Accumulation number: 560 Sample amount: 207.6 mg
- the quantification of each element in the silicon-containing material is also possible by SEM-EDX analysis, Auger electron spectroscopy (AES), laser ablation ICP mass spectrometry (LA-ICP-MS), X-ray photoelectron spectroscopy (XPS), etc. Is.
- the quantification of each element in the silicon-containing material by SEM-EDX analysis can be performed by observing the cross section of the silicon-containing material in the cross section of the negative electrode mixture layer.
- the cross-sectional observation can be performed by, for example, the following method. First, the battery is disassembled, the negative electrode is taken out, and a cross section of the negative electrode mixture layer is obtained using a cross section polisher (CP). Observe the cross section of the negative electrode mixture layer using a scanning electron microscope (SEM). From the cross-sectional image of the reflected electron image of the negative electrode mixture layer, 10 silicon-containing materials having a maximum particle diameter of 5 ⁇ m or more are randomly selected, and element mapping analysis is performed for each of them by energy dispersive X-ray (EDX). .. Calculate the content area of the target element using image analysis software. The observation magnification is preferably 2000 to 20000 times. The measured values of the content areas of the 10 predetermined elements of the obtained particles are averaged and obtained.
- a film is formed on the surface of the silicon-containing material due to decomposition of the electrolyte during the charging / discharging process.
- the silicon-containing material may have a conductive layer on its surface. Therefore, the mapping analysis by EDX is performed on the region inside 1 ⁇ m or more from the peripheral edge of the cross section of the silicon-containing material so that the coating film and the conductive layer are not included in the measurement range.
- ⁇ SEM-EDX measurement conditions > Processing equipment: JEOL, SM-09010 (Cross Section policer) Processing conditions: Acceleration voltage 6kV Current value: 140 ⁇ A Vacuum degree: 1 x 10 -3 to 2 x 10 -3 Pa Measuring device: Electron microscope SU-70 manufactured by Hitachi Acceleration voltage during analysis: 10 kV Field: Free mode Probe current mode: Medium Probe current range: High Anode Ap .: 3 OBJ Ap .: 2 Analysis area: 1 ⁇ m square Analysis software: EDAX Genesis CPS: 20500 Lsec: 50 Time constant: 3.2
- the crystallite size of the silicon particles dispersed in the lithium ion conductive phase is preferably 30 nm or less.
- the crystallite size of the silicon particles is 30 nm or less, the amount of volume change due to expansion and contraction of the silicon particles due to charge and discharge can be reduced, and the cycle characteristics can be further improved.
- the crystallite size of the silicon particles is more preferably 2 nm or more and 30 nm or less, and further preferably 2 nm or more and 20 nm or less.
- the crystallite size of the silicon particles is 20 nm or less, the expansion and contraction of the silicon particles can be made uniform, the fine cracks of the particles due to the expansion and contraction of the charged and discharged silicon particles can be reduced, and the cycle characteristics can be improved.
- the crystallite size of the silicon particles is calculated by Scherrer's formula from the half width of the diffraction peak attributed to the Si (111) plane of the X-ray diffraction (XRD) pattern of the silicon particles.
- At least a part of the surface of the silicon-containing material may be coated with a conductive layer.
- a conductive layer By forming a conductive layer on the surface of the silicon-containing material, the conductivity of the silicon-containing material can be dramatically increased.
- a carbon material is preferable.
- the carbon material preferably contains at least one selected from the group consisting of carbon compounds and carbonaceous materials.
- the thickness of the conductive layer is so thin that it does not substantially affect the average particle size of the silicon-containing material.
- the thickness of the conductive layer is preferably 1 to 200 nm, more preferably 5 to 100 nm, in consideration of ensuring conductivity and diffusivity of lithium ions.
- the thickness of the conductive layer can be measured by observing the cross section of the silicon-containing material using SEM or TEM (transmission electron microscope).
- Examples of the carbon compound include a compound containing carbon and hydrogen, and a compound containing carbon, hydrogen and oxygen.
- amorphous carbon having low crystallinity, graphite having high crystallinity, and the like can be used.
- Examples of amorphous carbon include carbon black, coal, coke, charcoal, and activated carbon.
- Examples of graphite include natural graphite, artificial graphite, graphitized mesophase carbon particles and the like. Of these, amorphous carbon is preferable because it has a low hardness and a large buffering action against silicon particles whose volume changes with charge and discharge.
- the amorphous carbon may be easily graphitized carbon (soft carbon) or non-graphitized carbon (hard carbon).
- Examples of carbon black include acetylene black and Ketjen black.
- a raw material mixture containing a Si raw material and a Li raw material in a predetermined ratio is used as the raw material for lithium silicate.
- a vanadium raw material vanadium carbide, vanadium iron alloy, etc.
- the raw material mixture may contain the above-mentioned alkali metal element, Group II element, and / or element M.
- the raw material mixture is dissolved and the melt is passed through a metal roll to form flakes to prepare lithium silicate.
- the flaky silicate is crystallized by heat treatment at a temperature above the glass transition point and below the melting point in an atmospheric atmosphere. It is also possible to use the flaky silicate without crystallizing it. It is also possible to produce a silicate by a solid phase reaction by firing at a temperature below the melting point without dissolving the raw material mixture.
- Silicon oxide can be used as the Si raw material.
- Li raw material for example, lithium carbonate, lithium oxide, lithium hydroxide, lithium hydride and the like can be used. These may be used alone or in combination of two or more.
- raw materials for alkali metal elements, group II elements, and element M include oxides, hydroxides, carbonic acid compounds, hydrides, nitrates, and sulfates of each element.
- the raw material silicon is mixed with the lithium silicate to form a composite.
- a silicon-containing material which is a composite particle of lithium silicate and silicon particles (hereinafter, also referred to as silicate composite particle) is produced through the following steps (a) to (c).
- Step (a) The raw material silicon powder and the lithium silicate powder are mixed, for example, in a mass ratio of 20:80 to 95: 5.
- the raw material silicon coarse particles of silicon having an average particle size of several ⁇ m to several tens of ⁇ m may be used.
- Step (b) Next, using a pulverizer such as a ball mill, the mixture of the raw material silicon and the lithium silicate is pulverized and compounded while being pulverized. At this time, an organic solvent may be added to the mixture and wet pulverized. The organic solvent plays a role of preventing the object to be crushed from adhering to the inner wall of the crushing container.
- a pulverizer such as a ball mill
- organic solvent alcohol, ether, fatty acid, alkane, cycloalkane, silicic acid ester, metal alkoxide and the like can be used.
- the raw material silicon and lithium silicate may be separately atomized and then mixed. Further, silicon nanoparticles and amorphous lithium silicate nanoparticles may be prepared and mixed without using a pulverizer.
- a known method such as a gas phase method (for example, a plasma method) or a liquid phase method (for example, a liquid phase reduction method) may be used for producing nanoparticles.
- Step (c) the mixture is pressurized and sintered in a state of being heated to 600 ° C. to 1000 ° C. in an inert gas atmosphere (for example, an atmosphere of argon, nitrogen, etc.).
- an inert gas atmosphere for example, an atmosphere of argon, nitrogen, etc.
- a sintering device such as a hot press that can pressurize under an inert atmosphere can be used.
- the silicate softens and flows to fill the gaps between the silicon particles.
- a dense block-shaped sintered body having the silicate phase as the sea portion and the silicon particles as the island portion.
- At least a part of the surface of the composite particle may be coated with a conductive material to form a conductive layer.
- a CVD method using hydrocarbon gas such as acetylene or methane as a raw material, coal pitch, petroleum pitch, phenol resin, etc. are mixed with the composite particles to create an inert atmosphere. Examples thereof include a method of heating and carbonizing at 700 ° C. to 950 ° C. in an atmosphere of (for example, argon, nitrogen, etc.). Further, carbon black may be attached to the surface of the composite particles.
- the step of washing the composite particles (including the case where the composite particles have a conductive layer on the surface) with an acid may be performed.
- an acidic aqueous solution it is possible to dissolve and remove a trace amount of alkaline components that may be generated when the raw material silicon and lithium silicate are composited.
- an aqueous solution of an inorganic acid such as hydrochloric acid, hydrofluoric acid, sulfuric acid, nitric acid, phosphoric acid or carbonic acid, or an aqueous solution of an organic acid such as citric acid or acetic acid can be used.
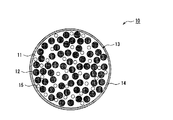
- FIG. 1 schematically shows a cross section of a silicate composite particle 10 coated with a conductive layer as an example of a silicon-containing material.
- the silicate composite particle (mother particle) 13 includes a lithium silicate phase 11 and silicon particles 12 dispersed in the lithium silicate phase 11.
- the silicate composite particle (mother particle) 13 has a sea-island structure in which fine silicon particles 12 are dispersed in the matrix of the lithium silicate phase 11.
- Particles 15 containing vanadium are also dispersed in the matrix of the lithium silicate phase 11.
- the vanadium-containing particles 15 usually have a smaller average particle size than the silicon particles 12.
- the surface of the mother particle 13 is covered with the conductive layer 14.
- the average particle size of the vanadium-containing particles 15 is measured by observing the cross section of the silicon-containing material using SEM or TEM. Specifically, it is obtained by averaging the maximum diameters of particles containing any 100 vanadium.
- a silicon oxide phase may be dispersed in the lithium silicate phase 11.
- the SiO 2 content in the silicate composite particles (mother particles) 13 measured by Si-NMR is preferably, for example, 30% by mass or less, and more preferably less than 7% by mass.
- the silicate composite particle (mother particle) 13 may contain other components in addition to the above.
- a carbon material, an oxide such as ZrO 2 , and a reinforcing material such as a carbide may be contained in an amount of less than 10% by mass with respect to the mother particle 13.
- the secondary battery according to the embodiment of the present disclosure includes a positive electrode, a negative electrode, an electrolyte, and a separator interposed between the positive electrode and the negative electrode.
- the negative electrode includes a negative electrode mixture layer containing a negative electrode active material containing the above silicon-containing material.
- the negative electrode includes, for example, a negative electrode current collector and a negative electrode mixture layer formed on the surface of the negative electrode current collector and containing a negative electrode active material.
- the negative electrode mixture layer can be formed by applying a negative electrode slurry in which the negative electrode mixture is dispersed in a dispersion medium to the surface of the negative electrode current collector and drying it. The dried coating film may be rolled if necessary.
- the negative electrode mixture contains the negative electrode active material containing the above silicon-containing material as an essential component, and may contain a binder, a conductive agent, a thickener, and the like as optional components. Since the silicon particles of the silicon-containing material can occlude a large amount of lithium ions, a high-capacity negative electrode can be obtained.
- the negative electrode active material may further contain other active material materials that electrochemically occlude and release lithium ions.
- a carbon-based active material is preferable. Since the volume of the silicon-containing material expands and contracts with charge and discharge, if the ratio of the silicon-containing material to the negative electrode active material increases, poor contact between the negative electrode active material and the negative electrode current collector tends to occur with charge and discharge.
- the ratio of the silicon-containing material to the total of the silicon-containing material and the carbon-based active material is preferably, for example, 0.5 to 15% by mass, and more preferably 1 to 5% by mass. This makes it easier to achieve both high capacity and improved cycle characteristics.
- Examples of the carbon-based active material include graphite, easily graphitized carbon (soft carbon), and non-graphitized carbon (hard carbon). Among them, graphite having excellent charge / discharge stability and a small irreversible capacity is preferable.
- Graphite means a material having a graphite-type crystal structure, and includes, for example, natural graphite, artificial graphite, graphitized mesophase carbon particles, and the like. As the carbon-based active material, one type may be used alone, or two or more types may be used in combination.
- the negative electrode current collector As the negative electrode current collector, a non-perforated conductive substrate (metal foil, etc.) and a porous conductive substrate (mesh body, net body, punching sheet, etc.) are used. Examples of the material of the negative electrode current collector include stainless steel, nickel, nickel alloy, copper, and copper alloy.
- binder examples include fluororesin, polyolefin resin, polyamide resin, polyimide resin, vinyl resin, styrene-butadiene copolymer rubber (SBR), polyacrylic acid and derivatives thereof.
- fluororesin polyolefin resin
- polyamide resin polyamide resin
- polyimide resin polyimide resin
- vinyl resin vinyl resin
- SBR styrene-butadiene copolymer rubber
- polyacrylic acid and derivatives thereof examples include fluororesin, polyolefin resin, polyamide resin, polyimide resin, vinyl resin, styrene-butadiene copolymer rubber (SBR), polyacrylic acid and derivatives thereof.
- SBR styrene-butadiene copolymer rubber
- Examples of the conductive agent include carbon black, conductive fibers, carbon fluoride, and organic conductive materials. One of these may be used alone, or two or more thereof may be used in combination.
- thickener examples include carboxymethyl cellulose (CMC) and polyvinyl alcohol. One of these may be used alone, or two or more thereof may be used in combination.
- CMC carboxymethyl cellulose
- polyvinyl alcohol examples include polyvinyl alcohol. One of these may be used alone, or two or more thereof may be used in combination.
- dispersion medium examples include water, alcohol, ether, N-methyl-2-pyrrolidone (NMP), and a mixed solvent thereof.
- the positive electrode includes, for example, a positive electrode current collector and a positive electrode mixture layer formed on the surface of the positive electrode current collector.
- the positive electrode mixture layer can be formed by applying a positive electrode slurry in which the positive electrode mixture is dispersed in a dispersion medium to the surface of the positive electrode current collector and drying it. The dried coating film may be rolled if necessary.
- the positive electrode mixture contains a positive electrode active material as an essential component, and can contain a binder, a conductive agent, etc. as an optional component.
- a lithium composite metal oxide can be used as the positive electrode active material.
- the lithium composite metal oxide include Li a CoO 2 , Li a NiO 2 , Li a MnO 2 , Li a Co b Ni 1-b O 2 , Li a Co b M 1-b O c , and Li a Ni. 1-b M b O c, Li a Mn 2 O 4, Li a Mn 2-b M b O 4, LiMePO 4, Li 2 MePO 4 F can be mentioned.
- M is at least one selected from the group consisting of Na, Mg, Sc, Y, Mn, Fe, Co, Ni, Cu, Zn, Al, Cr, Pb, Sb, and B.
- Me contains at least a transition element (eg, includes at least one selected from the group consisting of Mn, Fe, Co, Ni).
- a transition element eg, includes at least one selected from the group consisting of Mn, Fe, Co, Ni.
- the value a indicating the molar ratio of lithium is a value immediately after the production of the active material, and increases or decreases depending on charging and discharging.
- the binder and the conductive agent the same ones as those exemplified for the negative electrode can be used.
- the conductive agent graphite such as natural graphite or artificial graphite may be used.
- the positive electrode current collector a conductive substrate similar to the negative electrode current collector can be used.
- the material of the positive electrode current collector include stainless steel, aluminum, aluminum alloy, and titanium.
- the electrolyte contains a solvent and a lithium salt dissolved in the solvent.
- concentration of the lithium salt in the electrolyte is, for example, 0.5 to 2 mol / L.
- the electrolyte may contain known additives.
- an aqueous solvent or a non-aqueous solvent is used.
- a non-aqueous solvent for example, a cyclic carbonate ester, a chain carbonate ester, a cyclic carboxylic acid ester, or the like is used.
- the cyclic carbonate include propylene carbonate (PC) and ethylene carbonate (EC).
- the chain carbonic acid ester include diethyl carbonate (DEC), ethyl methyl carbonate (EMC), and dimethyl carbonate (DMC).
- DEC diethyl carbonate
- EMC ethyl methyl carbonate
- DMC dimethyl carbonate
- examples of the cyclic carboxylic acid ester include ⁇ -butyrolactone (GBL) and ⁇ -valerolactone (GVL).
- GBL ⁇ -butyrolactone
- GVL ⁇ -valerolactone
- lithium salt examples include a lithium salt of a chlorine-containing acid (LiClO 4 , LiAlCl 4 , LiB 10 Cl 10, etc.) and a lithium salt of a fluorine-containing acid (LiPF 6 , LiBF 4 , LiSbF 6 , LiAsF 6 , LiCF 3 SO 3). , LiCF 3 CO 2 etc.), Lithium salt of fluorine-containing acid imide (LiN (CF 3 SO 2 ) 2 , LiN (CF 3 SO 2 ) (C 4 F 9 SO 2 ), LiN (C 2 F 5 SO 2 ) 2 etc.), lithium halide (LiCl, LiBr, LiI, etc.) and the like can be used.
- One type of lithium salt may be used alone, or two or more types may be used in combination.
- the separator has high ion permeability and has appropriate mechanical strength and insulation.
- a microporous thin film, a woven fabric, a non-woven fabric, or the like can be used.
- the material of the separator for example, polyolefins such as polypropylene and polyethylene can be used.
- An example of the structure of a secondary battery is a structure in which an electrode group in which a positive electrode and a negative electrode are wound via a separator and an electrolyte are housed in an exterior body.
- another form of electrode group such as a laminated type electrode group in which a positive electrode and a negative electrode are laminated via a separator may be applied.
- the secondary battery may be in any form such as a cylindrical type, a square type, a coin type, a button type, and a laminated type.
- FIG. 2 is a schematic perspective view in which a part of the square secondary battery according to the embodiment of the present disclosure is cut out.
- the battery includes a bottomed square battery case 4, an electrode group 1 and an electrolyte housed in the battery case 4, and a sealing plate 5 for sealing the opening of the battery case 4.
- the electrode group 1 has a long strip-shaped negative electrode, a long strip-shaped positive electrode, and a separator interposed between them.
- the sealing plate 5 has a liquid injection port closed by the sealing 8 and a negative electrode terminal 6 insulated from the sealing plate 5 by a gasket 7.
- One end of the negative electrode lead 3 is attached to the negative electrode current collector by welding or the like.
- One end of the positive electrode lead 2 is attached to the positive electrode current collector by welding or the like.
- the other end of the negative electrode lead 3 is electrically connected to the negative electrode terminal 6.
- the other end of the positive electrode lead 2 is electrically connected to the sealing plate 5.
- Si powder 3N, average particle size 10 ⁇ m
- Li 2 Si 2 O 5 powder average particle size 10 ⁇ m
- simple substance vanadium and carbonized material are mixed.
- a predetermined amount of vanadium-iron alloy particles containing vanadium in a dispersed state was added and filled in a pot (SUS, volume: 500 mL) of a planetary ball mill (Fritsch, P-5). Twenty-four SUS balls (diameter 20 mm) were placed in a pot, the lid was closed, and the pot was pulverized at 200 rpm for 50 hours. Then, the powder was taken out in the inert atmosphere and heat-treated at 800 ° C. for 4 hours in the inert atmosphere to obtain silicate composite particles containing 30 ppm vanadium and 1.3% by mass of iron as the silicon-containing material A1.
- the silicon-containing material A1 is crushed, passed through a mesh of 40 ⁇ m, mixed with coal pitch (MCP250 manufactured by JFE Chemical Co., Ltd.), and heat-treated at 800 ° C. in an inert atmosphere to obtain the silicon-containing material A1.
- the surface was coated with carbon to form a conductive layer.
- the amount of carbon coated is about 5% by mass with respect to the total of the silicon-containing material A1 and the conductive layer.
- a silicon-containing material A1 having a conductive layer and having an average particle size of 5 ⁇ m was obtained.
- the vanadium and iron contents were measured by ICP-AES.
- the mixture was mixed at a mass ratio of: 1.0: 1.5, water was added, and then the mixture was stirred using a mixer (manufactured by Primix, TK hibismix) to prepare a negative electrode slurry.
- a negative electrode slurry was applied to both sides of the copper foil, the coating film was dried, and then rolled to obtain a negative electrode A2 having a negative electrode mixture layer having a density of 1.6 g / cm 3 formed on both sides of the copper foil. Obtained.
- Lithium cobalt oxide, acetylene black, and polyvinylidene fluoride are mixed at a mass ratio of 95: 2.5: 2.5, N-methyl-2-pyrrolidone (NMP) is added, and then a mixer (Primix) is added.
- NMP N-methyl-2-pyrrolidone
- Primemix a mixer
- a positive positive slurry was prepared by stirring using TK Hibismix (manufactured by TK Hibismix). Next, the positive electrode slurry was applied to both sides of the aluminum foil, the coating film was dried, and then rolled to obtain a positive electrode having a positive electrode mixture layer having a density of 3.6 g / cm 3 formed on both sides of the aluminum foil. rice field.
- a non-aqueous electrolyte solution was prepared by dissolving LiPF 6 at a concentration of 1.0 mol / L in a mixed solvent containing ethylene carbonate (EC) and ethyl methyl carbonate (EMC) in a volume ratio of 3: 7.
- EC ethylene carbonate
- EMC ethyl methyl carbonate
- Electrode group was inserted into the outer body made of an aluminum laminated film, vacuum dried at 105 ° C. for 2 hours, then a non-aqueous electrolytic solution was injected, and the opening of the outer body was sealed to obtain a secondary battery A1. ..
- the pause period between charging and discharging was set to 10 minutes.
- the number of cycles until reaching 80% of the discharge capacity of the first cycle was measured and used as the cycle life. The results are shown in Table 1.
- the secondary battery negative electrode according to the present disclosure is useful in a secondary battery that serves as a main power source for mobile communication devices, portable electronic devices, and the like.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Composite Materials (AREA)
- Engineering & Computer Science (AREA)
- Inorganic Chemistry (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Battery Electrode And Active Subsutance (AREA)
Abstract
Description
測定装置:バリアン社製、固体核磁気共鳴スペクトル測定装置(INOVA‐400)
プローブ:Varian 7mm CPMAS-2
MAS:4.2kHz
MAS速度:4kHz
パルス:DD(45°パルス+シグナル取込時間1Hデカップル)
繰り返し時間:1200sec~3000sec
観測幅:100kHz
観測中心:-100ppm付近
シグナル取込時間:0.05sec
積算回数:560
試料量:207.6mg
また、ケイ素含有材料中の各元素の定量は、SEM-EDX分析、オージェ電子分光分析(AES)、レーザアブレーションICP質量分析(LA-ICP-MS)、X線光電子分光分析(XPS)などでも可能である。
加工装置:JEOL製、SM-09010(Cross Section Polisher)
加工条件:加速電圧6kV
電流値:140μA
真空度:1×10-3~2×10-3Pa
測定装置:電子顕微鏡HITACHI製SU-70
分析時加速電圧:10kV
フィールド:フリーモード
プローブ電流モード:Medium
プローブ電流範囲:High
アノード Ap.:3
OBJ Ap.:2
分析エリア:1μm四方
分析ソフト:EDAX Genesis
CPS:20500
Lsec:50
時定数:3.2
リチウムイオン伝導相内に分散しているシリコン粒子の結晶子サイズは、30nm以下であることが好ましい。シリコン粒子の結晶子サイズが30nm以下である場合、充放電に伴うシリコン粒子の膨張収縮による体積変化量を小さくでき、サイクル特性が更に高められる。シリコン粒子の結晶子サイズは、より好ましくは2nm以上、30nm以下であり、更に好ましくは2nm以上、20nm以下である。シリコン粒子の結晶子サイズが20nm以下である場合、シリコン粒子の膨張収縮を均一化でき、充放電のシリコン粒子の膨張収縮による粒子の微細クラックが低減され、サイクル特性を向上させることができる。
リチウムシリケートの原料には、Si原料と、Li原料とを所定の割合で含む原料混合物を用いる。原料混合物には、バナジウム原料(炭化バナジウム、バナジウム鉄合金など)を添加する。また、原料混合物に、上述のアルカリ金属元素、第II族元素、および/または、元素Mを含ませてもよい。原料混合物を溶解し、融液を金属ロールに通してフレーク化してリチウムシリケートを作製する。その後フレーク化したシリケートを大気雰囲気で、ガラス転移点以上、融点以下の温度で熱処理により結晶化させる。なお、フレーク化したシリケートは結晶化させずに使用することも可能である。原料混合物を溶解せずに、融点以下の温度で焼成して固相反応によりシリケートを製造することも可能である。
次に、リチウムシリケートに原料シリコンを配合して複合化を行う。例えば、以下の工程(a)~(c)を経てリチウムシリケートとシリコン粒子との複合粒子(以下、シリケート複合粒子とも称する。)であるケイ素含有材料が作製される。
原料シリコンの粉末とリチウムシリケートの粉末とを、例えば、20:80~95:5の質量比で混合する。原料シリコンには平均粒径が数μm~数十μm程度のシリコンの粗粒子を用いればよい。
次に、ボールミルのような粉砕装置を用いて、原料シリコンとリチウムシリケートの混合物を微粒子化しながら粉砕および複合化する。このとき、混合物に有機溶媒を添加して、湿式粉砕してもよい。有機溶媒は、粉砕対象物の粉砕容器の内壁への付着を防ぐ役割を果たす。
次に、混合物を、例えば不活性ガス雰囲気(例えばアルゴン、窒素などの雰囲気)中で600℃~1000℃に加熱された状態で加圧し、焼結させる。焼結には、ホットプレスなど、不活性雰囲気下で加圧できる焼結装置を用い得る。焼結時、シリケートが軟化し、シリコン粒子間の隙間を埋めるように流動する。その結果、シリケート相を海部とし、シリコン粒子を島部とする緻密なブロック状の焼結体を得ることができる。得られた焼結体を粉砕すれば、シリケート複合粒子が得られる。
引き続き、複合粒子の表面の少なくとも一部を導電性材料で被覆して導電層を形成してもよい。導電性炭素材料で複合粒子の表面を被覆する方法としては、アセチレン、メタンなどの炭化水素ガスを原料に用いるCVD法、石炭ピッチ、石油ピッチ、フェノール樹脂などを複合粒子と混合し、不活性雰囲気(例えば、アルゴン、窒素などの雰囲気)中で、700℃~950℃で加熱して炭化させる方法などが例示できる。また、カーボンブラックを複合粒子の表面に付着させてもよい。
複合粒子(表面に導電層を有する場合を含む。)を酸で洗浄する工程を行ってもよい。例えば、酸性水溶液で複合粒子を洗浄することで、原料シリコンとリチウムシリケートとを複合化させる際に生じ得る微量のアルカリ成分を溶解させ、除去することができる。酸性水溶液としては、塩酸、フッ化水素酸、硫酸、硝酸、リン酸、炭酸などの無機酸の水溶液や、クエン酸、酢酸などの有機酸の水溶液を用いることができる。
負極は、例えば、負極集電体と、負極集電体の表面に形成され、かつ負極活物質を含む負極合材層とを具備する。負極合材層は、負極合材を分散媒に分散させた負極スラリーを、負極集電体の表面に塗布し、乾燥させることにより形成できる。乾燥後の塗膜を、必要により圧延してもよい。
正極は、例えば、正極集電体と、正極集電体の表面に形成された正極合材層とを具備する。正極合材層は、正極合材を分散媒に分散させた正極スラリーを、正極集電体の表面に塗布し、乾燥させることにより形成できる。乾燥後の塗膜を、必要により圧延してもよい。
電解質は、溶媒と、溶媒に溶解したリチウム塩を含む。電解質におけるリチウム塩の濃度は、例えば、0.5~2mol/Lである。電解質は、公知の添加剤を含有してもよい。
正極と負極との間には、セパレータを介在させることが望ましい。セパレータは、イオン透過度が高く、適度な機械的強度および絶縁性を備えている。セパレータとしては、微多孔薄膜、織布、不織布などを用いることができる。セパレータの材質としては、例えば、ポリプロピレン、ポリエチレンなどのポリオレフィンが用いられ得る。
[リチウムシリケートの合成]
炭酸リチウムと二酸化ケイ素とを、Li2CO3:SiO2=34:66のモル比となるように混合し、混合物を不活性ガス雰囲気中で1500℃、5時間溶解し、融液を金属ローラに通してフレーク状とし、その後、750℃で、5時間の熱処理を行い、リチウムシリケート(Li2Si2O5)を得た。リチウムシリケートは平均粒径10μmに粉砕した。
不活性雰囲気中で、Si粉末(3N、平均粒径10μm)とLi2Si2O5粉末(平均粒径10μm)とを、58:42の質量比で混合し、更に、単体のバナジウムと炭化バナジウムとを分散状態で含むバナジウムと鉄との合金粒子を所定量添加し、遊星ボールミル(フリッチュ製、P-5)のポット(SUS製、容積:500mL)に充填した。ポットにSUS製ボール(直径20mm)を24個入れて蓋を閉め、200rpmで50時間粉砕処理した。その後、不活性雰囲気中で粉末を取り出し、不活性雰囲気中、800℃で4時間熱処理し、バナジウム30ppmおよび鉄1.3質量%を含むシリケート複合粒子をケイ素含有材料A1として得た。
ケイ素含有材料A1の断面をTEMで観察した結果、Si粒子の平均粒径は50nm未満であった。ケイ素含有材料A1の粒子断面をSEMで観察した結果、シリケート相内にSi粒子および炭化バナジウムを含む粉末が略均一に分散していることが確認された。
導電層を有するケイ素含有材料A1と、黒鉛とを、5:95の質量比で含む混合物と、カルボキシメチルセルロースのナトリウム塩(CMC-Na)と、スチレン-ブタジエンゴム(SBR)とを、97.5:1.0:1.5の質量比で混合し、水を添加した後、混合機(プライミクス製、T.K.ハイビスミックス)を用いて攪拌し、負極スラリーを調製した。次に、銅箔の両面に負極スラリーを塗布し、塗膜を乾燥させた後、圧延して、銅箔の両面に密度1.6g/cm3の負極合材層が形成された負極A2を得た。
コバルト酸リチウムと、アセチレンブラックと、ポリフッ化ビニリデンとを、95:2.5:2.5の質量比で混合し、N-メチル-2-ピロリドン(NMP)を添加した後、混合機(プライミクス社製、T.K.ハイビスミックス)を用いて攪拌し、正極スラリーを調製した。次に、アルミニウム箔の両面に正極スラリーを塗布し、塗膜を乾燥させた後、圧延して、アルミニウム箔の両面に密度3.6g/cm3の正極合材層が形成された正極を得た。
エチレンカーボネート(EC)とエチルメチルカーボネート(EMC)とを3:7の体積比で含む混合溶媒にLiPF6を濃度1.0mol/Lで溶解して非水電解液を調製した。
各電極にタブをそれぞれ取り付け、タブが最外周部に位置するように、セパレータを介して正極および負極を渦巻き状に巻回することにより電極群を作製した。電極群をアルミニウムラミネートフィルム製の外装体内に挿入し、105℃で2時間真空乾燥した後、非水電解液を注入し、外装体の開口部を封止して、二次電池A1を得た。
Si粉末とLi2Si2O5粉末との混合物に炭化バナジウムを添加せず、鉄を添加して、鉄1.7質量%を含むシリケート複合粒子(ケイ素含有材料R1)を合成したこと以外は、実施例1と同様の方法で、負極R1および二次電池R1を得た。
(サイクル寿命)
実施例1および比較例1の各電池について、以下の条件で充放電を繰り返し、サイクル寿命を評価した。
25℃で、1It(800mA)の電流で電圧が4.2Vになるまで定電流充電を行い、その後、4.2Vの電圧で電流が1/20It(40mA)になるまで定電圧充電した。
25℃で、1It(800mA)の電流で電圧が2.75Vになるまで定電流放電を行った。
2 正極リード
3 負極リード
4 電池ケース
5 封口板
6 負極端子
7 ガスケット
8 封栓
10 導電層を有するシリケート複合粒子
11 リチウムシリケート相
12 シリコン粒子
13 シリケート複合粒子(母粒子)
14 導電層
15 バナジウムを含む粒子
Claims (11)
- ケイ素含有材料を含み、
前記ケイ素含有材料は、リチウムイオン伝導相と、前記リチウムイオン伝導相内に分散したシリコン粒子と、前記リチウムイオン伝導相内に分散したバナジウムを含む粒子と、を含む、二次電池用負極活物質。 - 前記ケイ素含有材料に含まれる前記バナジウムの含有量が、1000ppm以下である、請求項1に記載の二次電池用負極活物質。
- 前記バナジウムを含む粒子が、バナジウム合金および炭化バナジウムからなる群より選択される少なくとも1種を含む、請求項1または2に記載の二次電池用負極活物質。
- 前記バナジウムを含む粒子が、鉄を含む、請求項1~3のいずれか1項に記載の二次電池用負極活物質。
- 前記ケイ素含有材料に含まれる前記鉄の含有量が、0.01質量%以上、10質量%以下である、請求項4に記載の二次電池用負極活物質。
- 前記シリコン粒子の平均粒径は、1nm以上、1000nm以下である、請求項1~5のいずれか1項に記載の二次電池用負極活物質。
- 前記リチウムイオン伝導相は、ケイ素酸化物相、シリケート相および炭素相からなる群より選択される少なくとも1種である、請求項1~6のいずれか1項に記載の二次電池用負極活物質。
- 前記リチウムイオン伝導相は、前記シリケート相を含み、
前記シリケート相は、アルカリ金属元素および第II族元素からなる群より選択される少なくとも1種を含む、請求項7に記載の二次電池用負極活物質。 - 前記シリケート相は、さらに元素Mを含み、
前記元素Mは、B、Al、Zr、Nb、Ta、La、Y、Ti、P、Bi、Zn、Sn、Pb、Sb、Co、Er、FおよびWからなる群より選択される少なくとも1種である、請求項8に記載の二次電池用負極活物質。 - 前記シリケート相は、式:Li2zSiO2+z(0<z<2)で表されるリチウムシリケートを含む、請求項7~9のいずれか1項に記載の二次電池用負極活物質。
- 正極、負極、電解質および前記正極と前記負極との間に介在するセパレータを備え、
前記負極が、請求項1~10のいずれか1項に記載の二次電池用負極活物質を含む、二次電池。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2022511558A JP7664538B2 (ja) | 2020-03-30 | 2021-01-19 | 二次電池用負極活物質およびこれを用いた二次電池 |
| EP21782413.5A EP4131491A4 (en) | 2020-03-30 | 2021-01-19 | NEGATIVE ELECTRODE ACTIVE MATERIAL FOR SECONDARY BATTERIES, AND SECONDARY BATTERY USING SAME |
| US17/912,130 US12506141B2 (en) | 2020-03-30 | 2021-01-19 | Negative electrode active material for secondary batteries, and secondary battery using same |
| CN202180024438.1A CN115398674B (zh) | 2020-03-30 | 2021-01-19 | 二次电池用负极活性物质和使用其的二次电池 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020061364 | 2020-03-30 | ||
| JP2020-061364 | 2022-03-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021199587A1 true WO2021199587A1 (ja) | 2021-10-07 |
Family
ID=77929814
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2021/001564 Ceased WO2021199587A1 (ja) | 2020-03-30 | 2021-01-19 | 二次電池用負極活物質およびこれを用いた二次電池 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US12506141B2 (ja) |
| EP (1) | EP4131491A4 (ja) |
| JP (1) | JP7664538B2 (ja) |
| CN (1) | CN115398674B (ja) |
| WO (1) | WO2021199587A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024090206A1 (ja) * | 2022-10-25 | 2024-05-02 | パナソニックIpマネジメント株式会社 | 二次電池用負極及び二次電池 |
| WO2024090148A1 (ja) * | 2022-10-24 | 2024-05-02 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN119340378B (zh) * | 2024-12-16 | 2025-07-29 | 贝特瑞新材料集团股份有限公司 | 负极材料和二次电池 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013086979A (ja) * | 2011-10-13 | 2013-05-13 | Taiheiyo Cement Corp | リチウムイオン電池用正極活物質及びその製造法 |
| JP2014056722A (ja) * | 2012-09-13 | 2014-03-27 | Asahi Glass Co Ltd | リン酸化合物、二次電池用正極材料、および二次電池の製造方法 |
| JP2014120324A (ja) * | 2012-12-17 | 2014-06-30 | Taiheiyo Cement Corp | 二次電池正極活物質及びその製造方法 |
| WO2016035290A1 (ja) | 2014-09-03 | 2016-03-10 | 三洋電機株式会社 | 非水電解質二次電池用負極活物質及び非水電解質二次電池 |
| WO2016121320A1 (ja) | 2015-01-28 | 2016-08-04 | 三洋電機株式会社 | 非水電解質二次電池用負極活物質及び非水電解質二次電池 |
| CN107863506A (zh) * | 2017-10-20 | 2018-03-30 | 成都新柯力化工科技有限公司 | 一种用于锂电池的氮掺杂锡/铜多孔负极材料及制备方法 |
| WO2019131724A1 (ja) * | 2017-12-27 | 2019-07-04 | パナソニックIpマネジメント株式会社 | 二次電池用負極活物質及び二次電池 |
| WO2019151016A1 (ja) * | 2018-01-30 | 2019-08-08 | パナソニックIpマネジメント株式会社 | 二次電池用負極活物質及び二次電池 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4173687A (en) | 1978-06-20 | 1979-11-06 | Occidental Research Corporation | Current generating cell with alloy anode |
| WO2015064633A1 (ja) | 2013-10-30 | 2015-05-07 | 古河電気工業株式会社 | 負極活物質及びその製造方法並びにそれを用いた負極及び非水電解質二次電池 |
| WO2016121321A1 (ja) | 2015-01-28 | 2016-08-04 | 三洋電機株式会社 | 非水電解質二次電池用負極活物質及び非水電解質二次電池 |
| JPWO2016152505A1 (ja) | 2015-03-25 | 2017-12-07 | 株式会社村田製作所 | リチウムイオン二次電池 |
| CN107863503B (zh) * | 2017-10-12 | 2020-09-11 | 合肥国轩高科动力能源有限公司 | 一种改性硅酸铁锂正极材料的制备方法 |
-
2021
- 2021-01-19 JP JP2022511558A patent/JP7664538B2/ja active Active
- 2021-01-19 US US17/912,130 patent/US12506141B2/en active Active
- 2021-01-19 CN CN202180024438.1A patent/CN115398674B/zh active Active
- 2021-01-19 EP EP21782413.5A patent/EP4131491A4/en active Pending
- 2021-01-19 WO PCT/JP2021/001564 patent/WO2021199587A1/ja not_active Ceased
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013086979A (ja) * | 2011-10-13 | 2013-05-13 | Taiheiyo Cement Corp | リチウムイオン電池用正極活物質及びその製造法 |
| JP2014056722A (ja) * | 2012-09-13 | 2014-03-27 | Asahi Glass Co Ltd | リン酸化合物、二次電池用正極材料、および二次電池の製造方法 |
| JP2014120324A (ja) * | 2012-12-17 | 2014-06-30 | Taiheiyo Cement Corp | 二次電池正極活物質及びその製造方法 |
| WO2016035290A1 (ja) | 2014-09-03 | 2016-03-10 | 三洋電機株式会社 | 非水電解質二次電池用負極活物質及び非水電解質二次電池 |
| WO2016121320A1 (ja) | 2015-01-28 | 2016-08-04 | 三洋電機株式会社 | 非水電解質二次電池用負極活物質及び非水電解質二次電池 |
| CN107863506A (zh) * | 2017-10-20 | 2018-03-30 | 成都新柯力化工科技有限公司 | 一种用于锂电池的氮掺杂锡/铜多孔负极材料及制备方法 |
| WO2019131724A1 (ja) * | 2017-12-27 | 2019-07-04 | パナソニックIpマネジメント株式会社 | 二次電池用負極活物質及び二次電池 |
| WO2019151016A1 (ja) * | 2018-01-30 | 2019-08-08 | パナソニックIpマネジメント株式会社 | 二次電池用負極活物質及び二次電池 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP4131491A4 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024090148A1 (ja) * | 2022-10-24 | 2024-05-02 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池 |
| WO2024090206A1 (ja) * | 2022-10-25 | 2024-05-02 | パナソニックIpマネジメント株式会社 | 二次電池用負極及び二次電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4131491A4 (en) | 2024-11-13 |
| CN115398674B (zh) | 2025-06-20 |
| JP7664538B2 (ja) | 2025-04-18 |
| US12506141B2 (en) | 2025-12-23 |
| JPWO2021199587A1 (ja) | 2021-10-07 |
| CN115398674A (zh) | 2022-11-25 |
| US20230111143A1 (en) | 2023-04-13 |
| EP4131491A1 (en) | 2023-02-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113195406B (zh) | 二次电池用负极活性物质及二次电池 | |
| JP7710186B2 (ja) | 二次電池 | |
| US20240021806A1 (en) | Negative electrode material for non-aqueous electrolyte secondary battery, and non-aqueous electrolyte secondary battery | |
| WO2021241618A1 (ja) | 二次電池用負極活物質および二次電池 | |
| WO2023053947A1 (ja) | 二次電池 | |
| WO2023053946A1 (ja) | 二次電池 | |
| WO2021153077A1 (ja) | 二次電池用負極活物質およびその製造方法、ならびに二次電池 | |
| CN112119520B (zh) | 二次电池用负极活性物质和二次电池 | |
| CN115398674B (zh) | 二次电池用负极活性物质和使用其的二次电池 | |
| CN114902447B (zh) | 二次电池用负极活性物质及二次电池 | |
| US20250201831A1 (en) | Negative electrode active material for secondary batteries, and secondary battery | |
| JP7336690B2 (ja) | 二次電池用負極活物質および二次電池 | |
| WO2023053888A1 (ja) | 二次電池用負極活物質およびその製造方法、ならびに二次電池 | |
| WO2023042864A1 (ja) | 二次電池用負極活物質および二次電池 | |
| US20230078415A1 (en) | Negative electrode active material for secondary batteries, and secondary battery | |
| US20230411618A1 (en) | Negative electrode active material for nonaqueous electrolyte secondary batteries, and nonaqueous electrolyte secondary battery | |
| JP7417902B2 (ja) | 二次電池用負極活物質および二次電池 | |
| WO2021241388A1 (ja) | 二次電池用負極活物質および二次電池 | |
| US20240234722A9 (en) | Negative electrode active material for secondary battery, and secondary battery using same | |
| WO2023008098A1 (ja) | 二次電池用負極活物質および二次電池 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21782413 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2022511558 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2021782413 Country of ref document: EP Effective date: 20221031 |
|
| WWG | Wipo information: grant in national office |
Ref document number: 202180024438.1 Country of ref document: CN |