WO2022090252A2 - Sulfated c19 steroid hormones to protect against aging-associated proteotoxicity - Google Patents
Sulfated c19 steroid hormones to protect against aging-associated proteotoxicity Download PDFInfo
- Publication number
- WO2022090252A2 WO2022090252A2 PCT/EP2021/079715 EP2021079715W WO2022090252A2 WO 2022090252 A2 WO2022090252 A2 WO 2022090252A2 EP 2021079715 W EP2021079715 W EP 2021079715W WO 2022090252 A2 WO2022090252 A2 WO 2022090252A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- sul
- age
- sulfate
- salts
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/365—Lactones
- A61K31/366—Lactones having six-membered rings, e.g. delta-lactones
- A61K31/37—Coumarins, e.g. psoralen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/565—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol
- A61K31/566—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol having an oxo group in position 17, e.g. estrone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/565—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol
- A61K31/568—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol substituted in positions 10 and 13 by a chain having at least one carbon atom, e.g. androstanes, e.g. testosterone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/565—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol
- A61K31/568—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol substituted in positions 10 and 13 by a chain having at least one carbon atom, e.g. androstanes, e.g. testosterone
- A61K31/5685—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol substituted in positions 10 and 13 by a chain having at least one carbon atom, e.g. androstanes, e.g. testosterone having an oxo group in position 17, e.g. androsterone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
Definitions
- the present invention is encompassed within the field of medicine and provides a compound for use in the prevention of aging-associated proteotoxicity caused by protein-aggregation diseases and/or to increase the lifespan of a eukaryotic organism.
- Animals can extend life span by activating different genetic pathways. This increase of longevity is a regulated process that relay in the coordination of different tissues and environmental signals. Hormones are key players in tissues and cell communication. Consequently, they are involved in different pathways that regulate longevity, among those insulin and insulin-like growth factor, TGFD or dafachronic acids which are described to affect life span at least in the model organism Caenorhabditis elegans. Gonad is an endocrine tissue that produces steroids hormones to regulate different physiological aspects of the organism, including longevity. In C. elegans, germline ablation extends life span by non-completely understood mechanisms.
- dafachronic acid by the somatic gonad as well as the transcription factor encoded by daf-16, homologue to the human FOXO, and the nuclear receptors encoded genes daf-12, nhr-80 and nhr-492.
- Steroid hormones The classical function of steroid hormones is considered to be the activation of hormones receptors to transcribe their target genes. Steroid hormones are not only produced in gonads but also in other tissues. Those produced in the nervous system are known as neurosteroids. Neurosteroids, in addition to bind to hormone receptors, modulate neurotransmission either through direct interaction with neurotransmitter receptors or by other mechanisms. Steroid hormones can be sulfated by a sulfotransferase enzyme, generating a profound change in the chemical features of the hormone that impairs its function as hormone receptor activator.
- sulfated hormones are considered to be an inactive reservoir of hormones that can be activated upon removal of the sulfate moiety by the activity of hormone sulfatases.
- Sulfated steroid hormones can also be active as neurosteroids, regulating neurotransmission.
- Some sulfated steroid hormones like dehidroepiandrosterone sulfated (DHEAS), have long been related to aging. The level of this hormone declines with age and in age-related diseases such as sarcopenia or Alzheimer’s disease, which has generated the speculation of a causative effect.
- DHEAS dehidroepiandrosterone sulfated
- inhibition of the steroid sulfatase increases the percentage of sulfated hormones and, associated with that, an increase in longevity and the improvement of the symptoms related to protein aggregation diseases. This increase in longevity is mainly dependent on the same factors described for longevity caused by germline ablation.
- Treatment with STX64 a specific inhibitor of the steroid sulfatase enzyme, mimics the beneficial effects in longevity and protein aggregation diseases observed in the mutant.
- treatment with STX64 also ameliorates the cognitive symptoms and plaque formation in a mammalian model of Alzheimer’s disease.
- the observed phenotypes are recapitulated by treatment with sulfated C19 androgens steroid hormones but not with the non-sulfated forms or the sulfated C21 pregnenolone hormone, indicating that the causative beneficial effect of sul-2 inhibition is due to the increase of sulfated C19 steroid hormones rather than reduction of the non-sulfated form.
- This invention thus demonstrates that STX64 or specific sulfated C19 steroid hormones are a possible treatment for aging and/or aging-related diseases, more particularly specific sulfated C19 steroid hormones extend lifespan and protect against aging-associated proteotoxicity.
- Figure 1 Reduction of activity of sul-2 increases longevity and affects the levels of sulphated steroid hormones.
- FIG. 2 Genetic interactions and cellular location of sul-2 expression. Genetic analysis shows that sul-2 mimics most of the genetic interaction described for animals without germline, but do not affect fertility, a sul-2 enhances longevity in daf-2(e1370) background, b daf-16 transcription factor is required for sul-2 longevity, c, d, e.
- the essential factors for germline-loss longevity kri-1 (ok1251), tcer-1 (tm1452), and partially nhr-80(tm1011) are requiered for sul-2 longevity, f nhr-49(nr2041) does not suppres sul-2 increased lifespan.
- g,h sul-2 deletion has not significant increase of longevity in germline-less mutants glp-1 (e2141) and mes-1(bn7).
- i Brood size of sul-2 mutants are not significant different to wild type. Mean ⁇ SEM; One-way ANOVA test; ns. j Reproductive period of sul-2 mutants are not affected (25°C).
- k Dietary restriction conditions (DR) show enhanced longevity in sul-2 deletion background. I, daf-12 transcription factor is required for sul-2 longevity, m
- the Rieske-like oxygenase daf-36 is necessary for the increase of longevity upon inhibition of steroid hormone sulfatase, STX64 (1 pg/ml).
- n sul-2 is transcriptional expressed in sensory neurons, mainly ADF and ASE in the head, and PHA and PHB in the tail. Scale bar 10 urn.
- Figure 3 Reduction of steroid hormone sulfatase activity ameliorates the symptoms of proteotoxicity models in C. elegans and murine model, a NL5901 strain expressing a-synuclein in muscle cells reduce mobility with age at a slower rate in a sul-2(gk187) background (n > 17 per day).
- Statistic test vs a-synuclein control, b or under treatment with STX64 (1 pg/ml)( (n > 9 per day). Additional biological replicates assays are shown in Fig. 12 a and c.
- DA dopaminergic
- mice per groups o Temporal course of p-amyloid deposition in APP-PS1 mice and the effect of 3-4 weeks STX64 oral treatment on p-amyloid area in the frontal cortex (upper panel) and the hippocampus (lower panel).
- the number of microphotographs used was more than 3 in all the mice used, p Effect of oral administration with STX64 in more than 15-month-old APP-PS1 mice, and comparation with APP-PS1 and wild type mice older than 15 months in the passive avoidance test.
- the number of mice in each group were >5.
- * represent significant differences between vehicle-and STX64 administrated APP-PS1 mice.
- * represent significant differences between the short-term and long-term memory sessions (STM and LTM, respectively) and the training session in the same experimental group; and, + represent significant differences between the STM and LTM sessions between each experimental group and p-amyloid group.
- Figure 5 Longevity assays of sul-2 and genetic interactions with daf-2.
- sul-2 mutants do not show visible phenotypes but are long-lived and enhance developmental phenotypes of daf-2 mutants, a sul-2(pv17) is long lived and fer-15(b26) does not affect their lifespan, b sul-2 mutants are long-lived at 20 °C. c and at 25 °C.
- d Pumping rate of sul-2 mutants are similar to wild type at adult day 1 , while pv17 allele keeps higher pumping rate than wild type at adult day 6 stage. Worms were growth at 16° C until L4, then shifted to 25° C considering this Day 0 of adulthood.
- sul-2(gk17) at day 6 seems to distribute into two different populations, one with almost no thrashing and other with values similar to wild type a sul- 2(pv17).
- a small percentage of daf-2(e979) animals arrest development in dauer larvae at 16 °C
- sul-2(pv17) mutant does not show any larval arrest but enhances dauer arrest of daf-2(e979).
- Most daf-2(e1370) animals arrest in dauer stage when develop at 25 °C, but small percentage arrest in L1 stage.
- FIG. 6 Identification of pv17 allele and curated sequence of SUL-2.
- the pv17 allele is a missense mutation that changes the glycine indicated with asterisk to an aspartic acid residue. The mutation is located close to an evolutionary constrained region (ECR) , indicated with the bar at the bottom and also near to the catalytic core of sulfatases (in green).
- ECR evolutionary constrained region
- This allele deletes the sequence that encodes to the catalytic core of sulfatases (in green) and generates a frame shift, conserving only the four first aminoacids of the original sequence; therefore we consider gk187 a null allele.
- the pv17 allele location is indicated with asterisk, the new DNA fragment indentified in yellow and the new exon in purple.
- FIG. 7 Treatment with STX64 phenocopies longevity and genetic interaction of sul-2 mutants, a STX64 in non-UV E. coli increases lifespan of wild type, b, c Dosis curves of STX64 in non-UV E.coli show a significat effect at 1 pg/ml and 5pg/ml.
- d Photographs of wild type and daf-2 (e1370) at 25 °C. DMSO (STX64 vehicle) does not affect development of wild type, neither STX64 treatment, daf-2(e1370) arrests mostly in dauer stage at 25°C, but mainly arrests in L1 when treated with STX64, similar interaction is also observed in the sul-2 mutants. Photographs were taken in a Leica scope. Arrow heads: dauers, arrows: L1s.
- Figure 8 Genetic interactions of sul-2(pv17) allele, sul-2 point mutation allele pv17 shows similar phenotypes in longevity interactions assayed as sul-2 deletion, except in glp-1 background, a, b sul-2(pv17) enhances longevity of two daf-2 alleles, e1370 and m577. c, d sul-2(pv17) longevity is suppresed by two alleles of daf-16, mu86 and m26. e sul-2(pv17) longevity is suppresed by tcer-1(tm1452).
- f sul- 2(pv17) longevity is partially suppreses by nhr-80(tm1011).
- g sul-2(pv17) longevity is not suppresed by nhr-49(nr2041).
- h sul-2(pv17) increases lifespan in glp-1 background, i sul- 2(pv17) longevity is suppresed by daf-12(m20).
- Figure 9 sul-2 mutants affect DAF-16 location in intestinal cells, but they do not affect reproduction or gonad morphology
- a Micrographs show representative images of Pdaf- 16: :gfp:: daf-16 location in wild type (left panel) and sul-2 mutants (central and right panels). Both sul-2 mutants increase the nuclear location of DAF-16 in intestinal cells, like in germ-line less animals. Scale 20 pm.
- One-way ANOVA test c sul-2 mutants have similar brood size to control at 20 °C.
- One-way ANOVA test ns. D. the reproductive period of sul-2 mutants are not affected at 20 °C.
- e sul-2 mutants show normal gonad morphology. Micrographs of one representative gonadal arm in late L4 for each strain are shown.
- sul-2 is expressed in amphid and phasmid sensory neurons.
- Upper panel shows colocalization in the head with the most anterior pair amphids, possibly ASK, ADF or ADL neurons, but not with the most posterior pair, ASG or ASE.
- the four neurons where sul-2 is expressed colocalize with FiTC staining in PHAs and PHBs phasmids neurons, d Identification of the two main pair of amphid where sul-2 is exppresed by colocalization with the tph-1 neuron-specific promoter for ADF, upper panel, and the posterior neurons expressed by flp-6 promotor, ASE amphids, bottom panel.
- sul-2 is not expressed in gonadal tissues and is not affected in neuronal functions, a Integrated mCHERRY reporter of the sul-2 transcriptional unit is only expressed in sensory neurons, there is not expression in other tissues. Image in fluorescence and merged with the bright field image, b Inset of integrated worms, i) In heads, sul-2 is expressed in few amphid neurons (white arrows).
- Figure 12 Neurodegeneration phenotypes are ameliorated during aging when steroid sulfatase function is reduced, and a-synuclein aggregation decreases, a sul-2 has a beneficial effect during adulthood in muscular Parkinson’s disease model.
- c The protective effect of STX64 in muscular Parkinson’s disease model is present throughout aging.
- FIG. 13 Treatment with sulfated C19 steroid hormones phenocopy sul-2 inactivation, a Paralysis phenotype of GMC101 strain, Alzheimer’s dissease model, is reduced with TS and ES, but do not with DHEAS (1 g/ml). Assays performed with normal NGM plates. Data display from three independent biological replicates, n>130. b Similar to daf-2(e 1370); sul-2, percentage of L1 arrest in daf-2 (e1370) increase with DHEAS.TS or ES. c, d Aditional biological replicates of longevity curve with sulfated C19 steroid hormones. ES (1 pg/ml) increases lifespan in wild type background but does not increase further in sul-2(pv17), while DHEAS and TS do not affect (1 pg/ml).
- Figure 14 Model of regulation of longevity by SUL-2.
- the sulfatase SUL-2 and the sulfotransferase (probably ssu-1) regulates the level of sulfated steroid hormones.
- High level of sulfated steroid hormone provokes an increase of longevity, which depend on the same factors to the longevity generated by germline reduction (daf-16, daf-12, kri-1 , tcer-1).
- germline reduction daf-16, daf-12, kri-1 , tcer-1).
- the fact that both sulfatase and sulfotransferase are expressed in sensory neurons suggests a coordination of the sulfated state of the hormones with environmental signals.
- Figure 15 The fact that both sulfatase and sulfotransferase are expressed in sensory neurons suggests a coordination of the sulfated state of the hormones with environmental signals.
- FIG. 16 3 months treatment with STX64 in 15 months old mice improves cognition. 15 months mice still keep cognition capacities, but they lose this capacity during the following months.
- FIG. 17 6 months treatment with STX64 in 15 months old mice improves cognition. 15 months mice keep cognition capacities, but they lose this capacity during the following months. Age of mice when assayed was 21 months.
- Figure 18 1 -month treatment with STX64 in 18 months old mice improves cognition. 18 months mice have impaired cognition capacities. Age of mice when assayed was 19 months. A: Locomotion index were similar in both groups (vehicle and STX groups). B: Treated animals showed significant improvement in short term and long-term memory in passive avoidance test. * p ⁇ 0.05. ** p ⁇ 0.005. *** p> 0.0005. 1-way ANOVA.
- FIG. 19 3-month treatment with STX64 in 18 months old mice improves cognition. 18 months mice have impaired cognition capacities. Age of mice when assayed was 21 months. A: Locomotion index were similar in both groups (vehicle and STX groups). B: Object recognition is improved after three months treatment. C: short term and long-term memory in passive avoidance test. * p ⁇ 0.05. ** p ⁇ 0.005. *** p> 0.0005. 1-way ANOVA. B: * p ⁇ 0.05. ** p ⁇ 0.005. *** p> 0.0005. 1-way ANOVA.
- FIG 20 This figure shows the results of an "Open Field” test and of a "Y maze” test.
- the "Open Field” is a test where the mouse must explore an unknown space for 15 minutes. In this test, both mobility and habituation (cognitive parameter that measures the individual's ability to recognize the environment) can be studied.
- the "Y maze” test is a test where the mouse explores the maze for 6 minutes. This test evaluates the distance traveled as a measure of mobility and the ability to visit triplets of arms without repeating any of them as a sign of immediate memory. The results show that treatment with STX-64, E-S or both tend to improve the motor ability of 16-month-old mice.
- habituation ability and immediate memory of aged animals are improved by treatment with STX, E-S and the combination of both
- FIG 21 In this figure the evaluation of learning and memory abilities of aged mice was determined in the object recognition test and the passive avoidance test.
- object recognition after training with identical objects, tests the ability to identify different objects exposed together with a familiar one. When this happens, the object discrimination index is positive and higher than 0.1.
- passive avoidance the aim is to teach the mouse that its preferred place does not suit it and for them it is associated with an aversive stimulus.
- the entry latency to the preferred site is established and the aversive stimulus is administered.
- STM and LTM short- and long-term memory tests if the mouse associated the preferred place to the aversive stimulus, the input latency increases with respect to training.
- Vehicle-treated aged mice showed impairments in learning ability and thus memory. In both tests, mice treated with STX, E-S and the combination of both showed significantly both learning (STM) and ability to store information (memory).
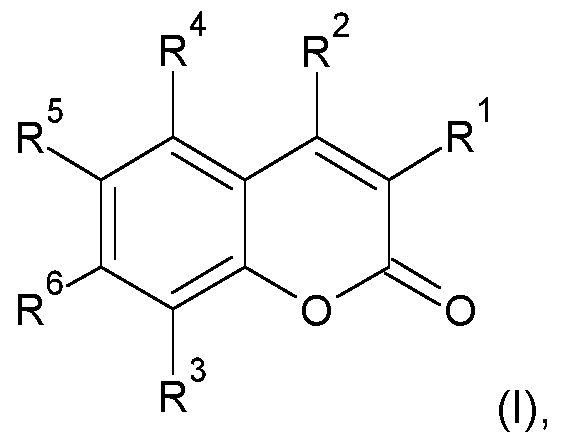
- an aspect of the present invention refers to a composition, preferably a pharmaceutical composition, nutraceutical composition or dietary supplement comprising epitestosterone sulfate (ES), or any salts or esters thereof, and/or compounds capable of inhibiting the steroid sulfatase of Formula (I): wherein:
- Ri-Re are independently selected from hydrogen, halogen (fluorine, chlorine, bromine, iodine or astatine), hydroxyl, sulfamate (OSO2NH2), alkyl and salts thereof;
- the composition is for use in Age-related cognitive impairment associated with Mild Cognitive Impairment (MCI), Age-Associated Memory Impairment (AAMI), or Age-Related Cognitive Decline (ARCD). More particularly, the composition is for use in Age- related cognitive impairment associated with Age-Associated Memory Impairment (AAMI).
- MCI Mild Cognitive Impairment
- AAMI Age-Associated Memory Impairment
- ARCD Age-Related Cognitive Decline
- the composition is for use in Age- related cognitive impairment associated with Age-Associated Memory Impairment (AAMI).
- the Age-related cognitive impairment preferably associated with Age- Associated Memory Impairment (AAMI)
- AAMI Age- Associated Memory Impairment
- the cognitive function might be reduced by about 5%, about 10%, about 30%, or more in the subject in need thereof, compared to cognitive function expected in an age-matched normal subject and the treatment reduces such difference, preferably in at least 0.1%, 1%, 2%, 3%, 4%, 5% or more.
- the composition is for use in a method for treating age-related cognitive impairment, preferably in a method to improve and/or treat age-related short- and/or long-term memory impairment, in a subject in need thereof.
- the composition is for use in a method for treating age-related cognitive impairment, preferably in a method to improve and/or treat age-related short- and/or long-term memory, in a subject in need thereof.
- composition for use comprises epitestosterone sulfate (ES) or any salts or esters thereof.
- ES epitestosterone sulfate
- composition for use comprises the sulfatase inhibitor STX64 of Formula (II):
- An additional aspect refers to a non-therapeutic method of improving age-related cognitive function, or age-related cognitive impairment in a subject in need thereof; in particular, for improving Age-related cognitive impairment associated with Mild Cognitive Impairment (MCI), Age-Associated Memory Impairment (AAMI), or Age-Related Cognitive Decline (ARCD); more particularly, for improving Age-related cognitive impairment associated with Age-Associated Memory Impairment (AAMI); the method comprising the steps of providing a composition, preferably a nutraceutical composition or dietary supplement, comprising epitestosterone sulfate (ES), or any salts or esters thereof, and/or a compound capable of inhibiting the steroid sulfatase of Formula (I): wherein:
- Ri-Re are independently selected from hydrogen, halogen (fluorine, chlorine, bromine, iodine or astatine), hydroxyl, sulfamate (OSO2NH2), alkyl and salts thereof;
- R1-R6 is a sulfamate group; and two or more of R1-R6 are linked together to form an additional cyclic structure; and administering an effective amount of the compound to a human subject in need thereof.
- a subject whose cognitive function might be reduced by about 5%, about 10%, about 30%, or more, compared to cognitive function expected in an age-matched normal subject.
- the method comprises the administration of epitestosterone sulfate (ES) or any salts or esters thereof. In an embodiment, the method comprises the administration of the sulfatase inhibitor STX64 of Formula (II):
- pharmaceutically acceptable carrier or “pharmaceutically acceptable diluent” means any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, compatible with pharmaceutical administration.
- pharmaceutically acceptable excipient refers to any substance formulated alongside the active ingredient of a medication, included for the purpose of long-term stabilization, bulking up solid formulations that contain potent active ingredients in small amounts, or to confer a therapeutic enhancement on the active ingredient in the final dosage form, such as facilitating drug absorption, reducing viscosity, or enhancing solubility.
- Excipients can also be useful in the manufacturing process, to aid in the handling of the active substance concerned such as by facilitating powder flowability or non-stick properties, in addition to aiding in vitro stability such as prevention of denaturation or aggregation over the expected shelf life.
- the use of such media and agents for pharmaceutically active substances is well known in the art.
- Acceptable carriers, excipients, or stabilizers are nontoxic to recipients at the dosages and concentrations employed and, without limiting the scope of the present invention, include: additional buffering agents; preservatives; co-solvents; antioxidants, including ascorbic acid and methionine; chelating agents such as EDTA; metal complexes (e.g., Zn-protein complexes); biodegradable polymers, such as polyesters; salt-forming counterions, such as sodium, polyhydric sugar alcohols; amino acids, such as alanine, glycine, glutamine, asparagine, histidine, arginine, lysine, ornithine, leucine, 2-phenylalanine, glutamic acid, and threonine; organic sugars or sugar alcohols, such as lactitol, stachyose, mannose, sorbose, xylose, ribose, ribitol, myoinisitose, myoinis
- “Improving” cognitive function includes promoting cognitive function and/or preserving cognitive function in a subject.
- “Cognitive function” or “cognitive status” refers to any higher order intellectual brain process or brain state, respectively, involved in learning and/or memory including, but not limited to, attention, information acquisition, information processing, working memory, short-term memory, long-term memory, anterograde memory, retrograde memory, memory retrieval, discrimination learning, decision-making, inhibitory response control, attentional set-shifting, delayed reinforcement learning, reversal learning, the temporal integration of voluntary behavior, and expressing an interest in one's surroundings and self-care.
- cognitive function may be measured, for example and without limitation, by the Alzheimer's Disease Assessment Scale-cognitive subscale (ADAS-cog), the clinical global impression of change scale (CGIC-plus scale), the Alzheimer's Disease Cooperative Study Activities of Daily Living Scale (ADCS-ADL), clinical global impression of change scale (CIBIC-plus scale); the Mini Mental State Exam (MMSE); the Neuropsychiatric Inventory (NPI); the Clinical Dementia Rating Scale (CDR); the Cambridge Neuropsychological Test Automated Battery (CANTAB) or the Sandoz Clinical Assessment-Geriatric (SCAG).
- ADAS-cog the Alzheimer's Disease Assessment Scale-cognitive subscale
- CGIC-plus scale the clinical global impression of change scale
- ADCS-ADL Alzheimer's Disease Cooperative Study Activities of Daily Living Scale
- CBIC-plus scale clinical global impression of change scale
- SCAG Sandoz Clinical Assessment-Geriatric
- cognitive function may be measured using imaging techniques such as Positron Emission Tomography (PET), functional magnetic resonance imaging (fMRI), Single Photon Emission Computed Tomography (SPECT), or any other imaging technique that allows one to measure brain activity.
- PET Positron Emission Tomography
- fMRI functional magnetic resonance imaging
- SPECT Single Photon Emission Computed Tomography
- cognitive function may be measured in various conventional ways known in the art, including using a Morris Water Maze (MWM), Barnes circular maze, elevated radial arm maze, T maze or any other mazes in which the animals use spatial information. Other tests known in the art may also be used to assess cognitive function, such as novel object recognition and odor recognition tasks. Cognitive function may also be measured using imaging techniques such as Positron Emission Tomography (PET), functional magnetic resonance imaging (fMRI), Single Photon Emission Computed Tomography (SPECT), or any other imaging technique that allows one to measure brain function. In animals, cognitive function may also be measured with electrophysiological techniques.
- PET Positron Emission Tomography
- fMRI functional magnetic resonance imaging
- SPECT Single Photon Emission Computed Tomography
- electrophysiological techniques any other imaging technique that allows one to measure brain function.
- Age-related cognitive impairment refers to cognitive function in aged subjects that is not as robust as that expected in an age-matched normal subject (i.e. subjects with mean scores for a given age in a cognitive test) or as that expected in young adult subjects. In some cases, cognitive function is reduced by about 5%, about 10%, about 30%, or more, compared to cognitive function expected in an age-matched normal subject. In some cases, cognitive function is as expected in an age-matched normal subject, but reduced by about 5%, about 10%, about 30%, about 50% or more, compared to cognitive function expected in a young adult subject. Age-related cognitive impairment may be associated with Mild Cognitive Impairment (MCI), Age-Associated Memory Impairment (AAMI), and Age-Related Cognitive Decline (ARCD).
- MCI Mild Cognitive Impairment
- AAMI Age-Associated Memory Impairment
- ARCD Age-Related Cognitive Decline
- MCI Mild Cognitive Impairment
- memory complaint as reported by patient, informant, or physician
- ADLs normal activities of daily living
- normal global cognitive function (4) abnormal memory for age (defined as scoring more than 1.5 standard deviations below the mean for a given age), and (5) absence of indicators of dementia (as defined by DSM-IV guidelines; see also Petersen et al., Srch. Neurol. 56: 303-308 (1999); Petersen, “Mild cognitive impairment: Aging to Alzheimer's Disease.” Oxford University Press, N.Y. (2003)).
- AAMI Alzheimer's disease
- a patient may be considered to have AAMI if he or she is at least 50 years old and meets all of the following criteria: a) The patient has noticed a decline in memory performance, b) The patient performs worse on a standard test of memory compared to young adults, c) All other obvious causes of memory decline, except normal aging, have been ruled out (in other words, the memory decline cannot be attributed to other causes such as a recent heart attack or head injury, depression, adverse reactions to medication, Alzheimer's disease, etc.).
- Age Related Cognitive Decline refers to declines in memory and cognitive abilities that are a normal consequence of aging in humans (e.g., Craik & Salthouse, 1992). This is also true in virtually all mammalian species. Age-Associated Memory Impairment refers to older persons with objective memory declines relative to their younger years, but cognitive functioning that is normal relative to their age peers (Crook et al., 1986). Age-Consistent Memory Decline is a less pejorative label which emphasizes that these are normal developmental changes (Crook, 1993; Larrabee, 1996), are not pathophysiological (Smith et al., 1991), and rarely progress to overt dementia (Youngjohn & Crook, 1993). The DSM-IV (1994) has codified the diagnostic classification of ARCD.
- treatment and “therapy”, as used in the present application, refer to a set of hygienic, pharmacological, surgical and/or physical means used with the intent to cure and/or alleviate a disease and/or symptom with the goal of remediating the health problem.
- treatment and “therapy” include preventive and curative methods, since both are directed to the maintenance and/or reestablishment of the health of an individual or animal. Regardless of the origin of the symptoms, disease and disability, the administration of a suitable medicament to alleviate and/or cure a health problem should be interpreted as a form of treatment or therapy within the context of this application.
- prevention refers to a set of hygienic, pharmacological, surgical and/or physical means used to prevent the onset and/or development of a disease and/or symptoms.
- prevention encompasses prophylactic methods, since these are used to maintain the health of an animal or individual.
- sulfatase inhibitor refers to any substance capable of reducing the activity of an enzyme of the esterase class that catalyzes the hydrolysis of sulfate esters.
- the substance may be a molecule that binds to any of the following elements: the gene that encodes the sulfatase enzyme, transcription factors of said gene, any of the expression products of said gene, for example, without being limited thereto, the messenger RNA or the sulfatase enzyme, and decreases or inhibits the expression and the activity of the molecule to which it binds, and/or its intracellular or extracellular signaling, thereby leading to total or partial inhibition of the activity of the sulfatase enzyme.
- the inhibitor may be selected from the list consisting of, without being limited thereto: antagonists against the sulfatase enzyme (preferably chemical), silencing RNA or specific antibody against the sulfatase enzyme (preferably, the antibody is monoclonal); in the present invention, this antibody may be defined as a neutralizing antibody against the effect of the sulfatase enzyme.
- Examples of chemical inhibitors of the activity of the sulfatase enzyme are, without being limited thereto, alternative substrates such as those in the series 2-(hydroxyphenyl) indol sulfate, synthetic or natural steroids which present inhibitory activity against STS, such as 5-androstene-3p, 17p-diol-3 sulfate, competitive inhibitors such as E1-MTP or EMATE, non-oestrogenic inhibitors such as Dll-14 (CAS NO: 186303-55-9), COUMATE (4-methylcoumarin-7-O-sulphamate) or STX64 (i.e., compound of Formula (II)), or others, such as KW-2581 or STX213, whose IC50 against the sulfatase enzyme has been determined in different studies (Purohit & Foster, 2012, J.
- steroid hormone sulfatase refers to any sulfatase enzyme involved in the metabolism of steroids. In particular, the enzymes catalyze the conversion of sulfated steroid precursors to the free steroid.
- An exemplary STS found in humans has been sequenced, characterized and the data have been deposited in the UniProtKB database under the accession number P08842.
- steroid hormone sulfatase inhibitor refers to any substance capable of reducing the activity of a steroid hormone sulfatase.
- the substance may be a molecule that binds to any of the following elements: the gene that encodes the STS enzyme, transcription factors of said gene, any of the expression products of said gene, for example, without being limited thereto, the messenger RNA or the STS enzyme, and decreases or inhibits the expression and the activity of the molecule to which it binds, and/or its intracellular signaling, thereby leading to total or partial inhibition of the activity of the STS enzyme.
- the inhibitor may be selected from the list consisting of, without being limited thereto: antagonists against the STS enzyme (preferably chemical), silencing RNA or specific antibody against the STS enzyme (preferably, the antibody is monoclonal); in the present invention, this antibody may be defined as a neutralising antibody against the effect of the STS enzyme.
- Examples of chemical inhibitors of the activity of the STS enzyme are, without being limited thereto, alternative substrates such as those in the series 2-(hydroxyphenyl) indol sulfate, synthetic or natural steroids which present inhibitory activity against STS, such as 5-androstene-3p, 17p- diol-3 sulfate, competitive inhibitors such as E1-MTP or EMATE, non-oestrogenic inhibitors such as Dll-14, COUMATE (4-methylcoumarin-7-O-sulphamate) or STX64 (i.e., compound of Formula (II)), or others, such as KW-2581 or STX213, whose IC50 against the sulfatase enzyme has been determined in different studies (Purohit & Foster, 2012, J. Endocrinol., 212(2):99-110).
- alternative substrates such as those in the series 2-(hydroxyphenyl) indol sulfate, synthetic or natural steroids which present inhibitory activity against STS, such as
- protein-aggregation disease refers to any disease in which certain proteins become structurally abnormal and thereby disrupt the function of cells, tissues and organs of the body. Often the proteins fail to fold into their normal configuration; in this misfolded state, the proteins can become toxic in some way or they can lose their normal function.
- protein-aggregation diseases include systemic AL amyloidosis, Alzheimer's Disease, Diabetes mellitus type 2, Parkinson's disease, Transmissible spongiform encephalopathy e.g.
- Bovine spongiform encephalopathy Fatal Familial Insomnia, Huntington's Disease, Medullary carcinoma of the thyroid, Cardiac arrhythmias, Atherosclerosis, Rheumatoid arthritis, Aortic medial amyloid, Prolactinomas, Familial amyloid polyneuropathy, Hereditary non-neuropathic systemic amyloidosis, Dialysis related amyloidosis, Finnish amyloidosis, Lattice corneal dystrophy, Cerebral amyloid angiopathy, Cerebral amyloid angiopathy (Icelandic type), Sporadic Inclusion Body Myositis, Amyotrophic lateral sclerosis (ALS), Prion-related or Spongiform encephalopathies, such as Creutzfeld-Jacob, Dementia with Lewy bodies, Frontotemporal dementia with Parkinsonism, Spinocerebellar ataxias, Spinocerebellar ataxia, Spin
- protein aggregate refers to any accumulation of abnormally folded proteins which cause and/or are associated with the negative progression of a protein-aggregation disease.
- amloid refers to a form of protein aggregates wherein the aggregates form unbranched fibers that bind Congo Red and then show green birefringence when viewed between crossed polarizers (for example, see Eisenberg & Jucker, 2012. Cell. 148(6):1188- 203 and Sipe et al., 2012. Amyloid. 19(4): 167-70).
- oligomer refers to any accumulation of abnormally folded proteins which cause and/or are associated with the negative progression of a protein-aggregation disease and does not satisfy the definition of an amyloid.
- polyglutamine oligomers cause and/or are associated with the negative progression of Huntington’s disease (see Hoffner & Dijan, 2014. Brain Sci. 4(1): 91-122).
- Gonad is a key tissue in the regulation of life span. Germline regulates longevity by inhibiting the production of dafachronic acid in the somatic gonads. Consistently, germline ablation or mutations that abolish the generation of germline, increase life span by activation of dafachronic acid synthesis. Gonads are also the classical tissue that produces sex steroids, although is not the only one. Our data indicates that inhibition of the sulfatase activity either by mutation or by STX64 raises the level of a very specific set of sulfated steroid hormones, which in fact generate an increase in longevity. This increase in longevity depends on common factors involved in life span extension produced by germline loss, suggesting that both processes are in fact linked.
- C. elegans In C. elegans, cell proliferation of the somatic cells only occurs during development and in larval stages but not in the adult stage; Therefore, increasing longevity is due to the maintenance of the postmitotic cells.
- One of the stresses observed in C. elegans adult cells is the aggregation of endogenous proteins, which generates cellular misfunction. This age-related formation of aggregates is also observed upon ectopic expression of aggregation-prone proteins, like - amyloid ora-synuclein. Long-lived mutants such as daf-2 or glp-1 delay the aggregation toxicity through a different mechanism including chaperon expression and degradation by proteasome or autophagy.
- sulfotransferases and sulfatases are expressed in different tissues, including the nervous system, similar to what we observe in C. elegans.
- C19 steroid hormones have also been involved in longevity. For instance, dehydroepiandrosterone sulfate (DHEAS) declines with age and has been used as a marker of aging, raising speculations of a causative effect on sarcopenia, poor cognitive function and other aging associated diseases6 including Alzheimer’s disease.
- DHEAS dehydroepiandrosterone sulfate
- Our data show that inhibition of the steroid sulfatase by mutation or by STX64 treatment extends lifespan in C. elegans and protects against aging-associated proteotoxicity in nematodes. Interestingly, similar effects were observed upon treatment with some specific sulfated C19 steroid hormones.
- the present invention provides a method for increasing the lifespan of eukaryotic organisms comprising the steps of: providing a sulfated C19 androgen compound, or a compound capable of inhibiting the steroid sulfatase selected from the list consisting of 2- (hydroxyphenyl) indol sulfate, 5-androstene-3p, Dll-14, 17p-diol-3 sulfate, E1-MTP, EMATE, COUMATE, STX64 (compound of formula II), KW-2581 , STX213, morpholine, silencing RNA and specific antibody against the Steroid sulfatase (STS), or steryl-sulfatase (EC 3.1.6.2), formerly known as arylsulfatase C; or the sulfatase inhibitor of Formula (I): wherein:
- Ri-Re are independently selected from hydrogen, halogen (fluorine, chlorine, bromine, iodine or astatine), hydroxyl, sulfamate (OSO2NH2), alkyl and salts thereof;
- R1-R6 is a sulfamate group; and two or more of R1-R6 are linked together to form an additional cyclic structure; and administering an effective amount of the compound to a eukaryotic organism, such that the lifespan of the eukaryotic organism is increased.
- the present invention further provides methods and compositions for improving age-related cognitive function and/or treating disorders involving age-related cognitive dysfunction, age-related cognitive impairment or the risk thereof in a subject in need thereof by administering a sulfated C19 androgen compound, or a compound capable of inhibiting the steroid sulfatase selected from the list consisting of 2- (hydroxyphenyl) indol sulfate, 5-androstene-3p, DU-14, 17p-diol-3 sulfate, E1-MTP, EMATE, COUMATE, STX64 (compound of formula II), KW-2581 , STX213, morpholine, silencing RNA and specific antibody against the Steroid sulfatase (STS), or steryl-sulfatase (EC 3.1.6.2), formerly known as arylsulfatase C; or the sulfatase inhibitor of Formula (I):
- Ri-Re are independently selected from hydrogen, halogen (fluorine, chlorine, bromine, iodine or astatine), hydroxyl, sulfamate (OSO2NH2), alkyl and salts thereof;
- R1-R6 is a sulfamate group; and two or more of R1-R6 are linked together to form an additional cyclic structure.
- the compound of any of the above methods is a sulfated C19 androgen selected from the list consisting of dehidroepiandrosterone sulfate (DHEAS), testosterone sulfate (TS), epitestosterone sulfate (ES) or Androsterone sulfate (AS).
- DHEAS dehidroepiandrosterone sulfate
- TS testosterone sulfate
- ES epitestosterone sulfate
- AS Androsterone sulfate
- the sulfated C19 androgen is selected from the group consisting of testosterone sulfate (TS), epitestosterone sulfate (ES) or Androsterone sulfate (AS).
- Testosterone sulfate (https://pubchem.ncbi.nlm.nih.gov/compound/Testosterone-sulfate) is an endogenous, naturally occurring steroid and minor urinary metabolite of testosterone, of chemical name [(8R,9S,10R,13S,14S,17S)-10,13-dimethyl-3-oxo-
- Testosterone 17p-sulfate Testosterone 17p-sulfuric acid
- 17p- (Sulfooxy)androst-4-en-3-one Epitestosterone structurally differs from testosterone only in the configuration at the hydroxybearing carbon, C17.
- Testosterone 17a-sulfate also known as Testosterone 17a-sulfate has the following chemical name N,N-diethylethanamine;[(8R,9S,10R,13S,14S,17R)-10,13- dimethyl-3-oxo-1 ,2,6,7,8,9,11 ,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-17-yl] hydrogen sulfate, and formula:
- AS Androsterone sulfate
- 3a-hydroxy-5a-androstan- 17-one 3a-sulfate is an endogenous, naturally occurring steroid and one of the major urinary metabolites of androgens.
- steroid sulfate which is formed from sulfation of androsterone by the steroid sulfotransferase SULT2A1 and can be desulfated back into androsterone by steroid sulfatase, of chemical name [(3R,5S,8R,9S,10S,13S,14S)-10,13-Dimethyl-17-oxo- 1 ,2, 3, 4, 5, 6, 7, 8, 9,11 ,12,14,15,16-tetradecahydrocyclopenta[a]phenanthren-3-yl] hydrogen sulfate, and formula
- any of the above mentioned sulfated C19 androgens includes the corresponding salts and esters thereof.
- any of the above mentioned sulfated C19 androgens includes the corresponding pharmaceutically acceptable salts, pharmaceutically acceptable solvates, isotopic variants (preferably comprising deuterium atoms and/or one or more carbon atoms with 13C), different crystalline forms such as polymorphs, pharmaceutically acceptable esters, stereoisomers, tautomers, analogs and derivatives thereof.
- Solvates such as (A) or (B), where R H, alkyl or aryl and are formed by addition of water or an alcohol to the parent compound:
- the compound capable of inhibiting the steroid sulfatase of any of the above methods is the sulfatase inhibitor of Formula (I):
- Ri-Re are independently selected from hydrogen, halogen (fluorine, chlorine, bromine, iodine or astatine), hydroxyl, sulfamate (OSO2NH2), alkyl and salts thereof;
- R1-R6 is a sulfamate group; and two or more of R1-R6 are linked together to form an additional cyclic structure.
- the compound capable of inhibiting the steroid sulfatase is the sulfatase inhibitor STX64 of Formula (II):
- STX64 of Formula (II) above includes the corresponding salts and esters thereof.
- STX64 of Formula (II) above includes the corresponding pharmaceutically acceptable salts, pharmaceutically acceptable solvates, isotopic variants (preferably comprising deuterium atoms and/or one or more carbon atoms with 13C), different crystalline forms such as polymorphs, pharmaceutically acceptable esters, stereoisomers, tautomers, analogs and derivatives thereof.
- the compound of any of the above methods is a combination of a sulfated C19 androgen, preferably testosterone sulfate (TS), epitestosterone sulfate (ES) or Androsterone sulfate (AS), and a sulfatase inhibitor selected from the list consisting of 2-(hydroxyphenyl) indol sulfate, 5-androstene-3p, Dll-14, 17p-diol-3 sulfate, E1-MTP, EMATE, COUMATE, STX64, KW-2581 , STX213, morpholine, silencing RNA and specific antibody against the STS enzyme; or the sulfatase inhibitor of Formula (I):
- a sulfated C19 androgen preferably testosterone sulfate (TS), epitestosterone sulfate (ES) or Androsterone sulfate (AS), and a sulfatase inhibitor selected from the list consist
- Ri-Re are independently selected from hydrogen, halogen (fluorine, chlorine, bromine, iodine or astatine), hydroxyl, sulfamate (OSO2NH2), alkyl and salts thereof;
- Ri-Re is a sulfamate group; and two or more of Ri-Re are linked together to form an additional cyclic structure.
- the sulfatase inhibitor is STX64 and the sulfated C19 androgen is epitestosterone sulfate (ES).
- lifespan increasing compounds when used in a method for increasing the lifespan of a eukaryotic organism, shall be referred to as “lifespan increasing compounds”.
- a lifespan increasing compound is a compound that is useful for the prevention of aging-associated proteotoxicity and/or to increase the lifespan of a eukaryotic organism.
- Lifespan as used herein means the length of time (e.g. days or years) a cell or organism survives before dying (chronological lifespan).
- a lifespan increasing compound is preferably a compound that prevents aging-associated proteotoxicity caused by protein-aggregation diseases and/or increases the lifespan of a eukaryotic organism.
- Protein aggregates such as amyloids and oligomers have been associated with a number of diseases. In some cases, these protein aggregates can become toxic and can cause significant damage to cells and tissue. This toxicity is thought to be one of the contributing factors causing and/or contributing to the pathology of protein-aggregation diseases.
- the lifespan increasing compounds of the present invention protect against and treat protein-aggregation diseases.
- the protein-aggregation disease is selected from a list consisting of systemic AL amyloidosis, Alzheimer's Disease, Diabetes mellitus type 2, Parkinson's disease, Transmissible spongiform encephalopathy e.g.
- Bovine spongiform encephalopathy Fatal Familial Insomnia, Huntington's Disease, Medullary carcinoma of the thyroid, Cardiac arrhythmias, Atherosclerosis, Rheumatoid arthritis, Aortic medial amyloid, Prolactinomas, Familial amyloid polyneuropathy, Hereditary non-neuropathic systemic amyloidosis, Dialysis related amyloidosis, Finnish amyloidosis, Lattice corneal dystrophy, Cerebral amyloid angiopathy, Cerebral amyloid angiopathy (Icelandic type), Sporadic Inclusion Body Myositis, Amyotrophic lateral sclerosis (ALS), Prion-related or Spongiform encephalopathies, such as Creutzfeld-Jacob, Dementia with Lewy bodies, Frontotemporal dementia with Parkinsonism, Spinocerebellar ataxias, Spinocerebellar ataxia, Spin
- the protein-aggregation disease is selected from a list consisting of Alzheimer’s disease, Parkinson’s disease and Huntington’s disease and the sulfated C19 androgens of the present invention are preferably selected from the list consisting of testosterone sulfate (TS), epitestosterone sulfate (ES) or Androsterone sulfate (AS). More preferably, said sulfated C19 androgens are in the form of a pharmaceutical or nutraceutical composition or in the form of Dietary Supplements.
- TS testosterone sulfate
- ES epitestosterone sulfate
- AS Androsterone sulfate
- the protein-aggregation disease is a central nervous system localized protein-aggregation disease.
- the protein-aggregation disease is also a neurodegenerative disease.
- the term “neurodegenerative disease” refers to any disorder characterized by the progressive loss of structure or function of neurons, including death of neurons.
- Alzheimer’s disease is an example or a protein-aggregation disease and an example of a neurodegenerative disease.
- an effective amount of a lifespan increasing compound increases the lifespan of a eukaryotic organism, preferably a human, and/or protects against aging-associated proteotoxicity caused by a protein-aggregation disease.
- an effective amount of a lifespan increasing compound increases the lifespan of an eukaryotic organism by a statistically significant amount compared to the lifespan of an untreated organism. The lifespan of an untreated organism may be determined in parallel or may be obtained from separately conducted studies (control).
- an effective amount of a lifespan increasing compound increases the lifespan of an eukaryotic organism by at least 5%.
- an effective amount of a LAC increases the lifespan of an eukaryotic organism by at least 10, 15, 20, 25, 35, 50%, or 100% over control.
- eukaryotic organisms include single- and multi-cellular organisms, including higher-order organisms (such as mammals, which includes humans).
- the present method for increasing the lifespan of the eukaryotic organism can be used in order to generally increase the lifespan of the cells of a eukaryotic organism and to protect its cells against stress and/or against apoptosis.
- the present method can be used for treating or preventing a disease or condition induced or exacerbated by cellular senescence in a subject; for extending the lifespan of a subject; for treating or preventing a disease or condition relating to lifespan; for treating or preventing a disease or condition relating to the proliferative capacity of cells; for treating or preventing a disease or condition resulting from cell damage or death.
- the present method may be used to prevent aging and aging-related consequences, or diseases caused by protein-aggregation diseases.
- the protein-aggregation disease is selected from a list consisting of systemic AL amyloidosis, Alzheimer's Disease, Diabetes mellitus type 2, Parkinson's disease, Transmissible spongiform encephalopathy e.g.
- Bovine spongiform encephalopathy Fatal Familial Insomnia, Huntington's Disease, Medullary carcinoma of the thyroid, Cardiac arrhythmias, Atherosclerosis, Rheumatoid arthritis, Aortic medial amyloid, Prolactinomas, Familial amyloid polyneuropathy, Hereditary non-neuropathic systemic amyloidosis, Dialysis related amyloidosis, Finnish amyloidosis, Lattice corneal dystrophy, Cerebral amyloid angiopathy, Cerebral amyloid angiopathy (Icelandic type), Sporadic Inclusion Body Myositis, Amyotrophic lateral sclerosis (ALS), Prion-related or Spongiform encephalopathies, such as Creutzfeld-Jacob, Dementia with Lewy bodies, Frontotemporal dementia with Parkinsonism, Spinocerebellar ataxias, Spinocerebellar ataxia, Spin
- Hereditary lattice corneal dystrophy Corneal amyloidosis associated with trichiasis, Cataract, Calcifying epithelial odontogenic tumors, Pulmonary alveolar proteinosis, Inclusion-body myositis, Cutaneous lichen amyloidosis, and Non-neuropathic systemic amyloidosis, such as AL amyloidosis, AA amyloidosis, Familial Mediterranean fever, Senile systemic amyloidosis, Familial amyloidotic polyneuropathy, Hemodialysis-related amyloidosis, ApoAl amyloidosis, ApoAII amyloidosis, ApoAl V amyloidosis, Finnish hereditary amyloidosis, Lysozyme amyloidosis, Fibrinogen amyloidosis, Icelandic hereditary cerebral amyloid angiopathy, familial amyloidosis, and systemic amy
- the protein-aggregation disease is selected from a list consisting of Alzheimer’s disease, Parkinson’s disease and Huntington’s disease and the sulfated C19 androgens of the present invention are preferably selected from the list consisting of testosterone sulfate (TS), epitestosterone sulfate (ES) or Androsterone sulfate (AS). More preferably, said sulfated C19 androgens are in the form of a pharmaceutical or nutraceutical composition or in the form of Dietary Supplements.
- TS testosterone sulfate
- ES epitestosterone sulfate
- AS Androsterone sulfate
- the method of the present invention may also be applied during developmental and growth phases in mammals, preferably in humans, in order to, e.g., alter, retard or accelerate the developmental and/or growth process.
- all of the compounds of the present invention can be in the form of pharmaceutical compositions, nutraceutical compositions or dietary supplements.
- the compounds are in the form of a pharmaceutical composition further comprising a pharmaceutically acceptable carrier and/or diluent.
- the pharmaceutical composition may further comprise a pharmaceutically acceptable excipient.
- a pharmaceutical composition as described herein may also contain other substances. These substances include, but are not limited to, cryoprotectants, lyoprotectants, surfactants, bulking agents, anti-oxidants, and stabilizing agents. In some embodiments, the pharmaceutical composition may be lyophilized.
- cryoprotectant includes agents which provide stability to the compositions against freezing-induced stresses. Cryoprotectants may also offer protection during primary and secondary drying and long-term product storage.
- cryoprotectants include sugars, such as sucrose, glucose, trehalose, mannitol, mannose, and lactose; polymers, such as dextran, hydroxyethyl starch and polyethylene glycol; surfactants, such as polysorbates (e.g., PS-20 or PS-80); and amino acids, such as glycine, arginine, leucine, and serine.
- a cryoprotectant exhibiting low toxicity in biological systems is generally used.
- a lyoprotectant is added to a pharmaceutical composition described herein.
- the term "lyoprotectant” as used herein includes agents that provide stability to the compositions during the freeze-drying or dehydration process (primary and secondary freeze- drying cycles. This helps to minimize product degradation during the lyophilization cycle, and improve the long-term product stability.
- Non-limiting examples of lyoprotectants include sugars, such as sucrose or trehalose; an amino acid, such as monosodium glutamate, non-crystalline glycine or histidine; a methylamine, such as betaine; a lyotropic salt, such as magnesium sulfate; a polyol, such as trihydric or higher sugar alcohols, e.g., glycerin, erythritol, glycerol, arabitol, xylitol, sorbitol, and mannitol; propylene glycol; polyethylene glycol; pluronics; and combinations thereof.
- the amount of lyoprotectant added to a pharmaceutical composition is generally an amount that does not lead to an unacceptable amount of degradation when the pharmaceutical composition is lyophilized.
- a bulking agent is included in the pharmaceutical composition.
- bulking agents may also impart useful qualities in regard to modifying the collapse temperature, providing freeze-thaw protection, and enhancing the stability over long-term storage.
- Non-limiting examples of bulking agents include mannitol, glycine, lactose, and sucrose.
- Bulking agents may be crystalline (such as glycine, mannitol, or sodium chloride) or amorphous (such as dextran, hydroxyethyl starch) and are generally used in formulations in an amount from 0.5% to 10%.
- nontoxic solid carriers may be used which include, for example, pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharin, talcum, cellulose, glucose, sucrose, magnesium carbonate, and the like.
- the pharmaceutical composition may further comprise cryoprotectants, lyoprotectants, surfactants, bulking agents, anti-oxidants, stabilizing agents and pharmaceutically acceptable carriers.
- the pharmaceutical compositions are generally supplied in finely divided form along with a surfactant and propellant. The surfactant must, of course, be nontoxic, and is generally soluble in the propellant.
- esters or partial esters of fatty acids containing from 6 to 22 carbon atoms such as caproic, octanoic, lauric, palmitic, stearic, linoleic, linolenic, olesteric and oleic acids with an aliphatic polyhydric alcohol or its cyclic anhydride.
- Mixed esters, such as mixed or natural glycerides may be employed.
- a carrier can also be included, as desired, as with, e.g., lecithin for intranasal delivery.
- traditional binders and carriers may include, for example, polyalkalene glycols or triglycerides.
- the compounds are prepared for oral, sublingual, buccal, intranasal, intravenous, intramuscular, intraperitoneal and/or inhalation-mediated administration.
- the compounds of the invention are in the form of a nutraceutical composition or a dietary supplement or product.
- the compounds of the invention can be delivered to an organism using any available method and route suitable for compound delivery, including oral, parenteral, subcutaneous, intraperitoneal, intrapulmonary, and intranasal routes. It will be recognized by those of skill in the art that the form and character of the particular dosing regimen employed in the method of the invention will be dictated by the route of administration and other well- known variables, such as the size and age of the eukaryotic organism. Determination of such dosing regimens is within the purview of one skilled in the art. Administration of the compound could be performed in conjunction with any conventional therapies that are intended to treat a disease or disorder associated with aging including topical, oral, or injectable.
- the compounds of the invention may be administered using any route known to the skilled person.
- the compounds of the invention are administered transdermally, sublingually, intravenously, intranasally, intracerebroventricularly, intraarterially, intracerebrally, intramuscularly, intraperitoneally, orally or via inhalation.
- the compounds of the invention are administered transdermally, sublingually, intravenously, intraperitoneally, orally or via inhalation.
- the composition may be aerosolized and administered via, for example, an anesthesia mask.
- the compounds of the invention are administered transdermally, sublingually, intravenously, subcutaneously, orally or via inhalation.
- the composition is administered orally or sublingually.
- the compounds of the invention may be administered once or more than once.
- a skilled person will be able to ascertain the most effective dosage regimen for the patient.
- the most effective dosage regimen may be one where the patient is administered the composition twice daily, once a day, once every three days, once a week, once a month, once every three months, once every six months or once every year.
- sul-2 also enhanced the developmental phenotypes of mutants in the insulin/insulin like growth factor (IGF) receptor daf-2 (Fig. 5g-i), which was used for gene mapping and identification.
- IGF insulin/insulin like growth factor
- Fig. 5g-i insulin/insulin like growth factor receptor daf-2
- the pv17 allele introduces a single amino acid substitution (G46D) resulting in a reduction of function phenotype.
- the curated seguence slightly differs from the one published (Fig. 6).
- sul-2 encodes a sulfatase of steroid hormones
- Sulfatases are a large protein family involved in different biological processes and with a wide range of substrates.
- the placement of curated sul-2 in the sulfatases phylogenetic tree is uncertain, but when compared to mammalian sulfatases, sul-2 clusters closer to the Arylsulfatases type H, F, E, D and the steroid sulfatase type C (Fig. 1c) which probably originated from a common ancestor gene, sul-1 and sul-3 belong to a different family of sulfatases (Fig. 1c).
- sul-2 may exert its activity by modifying sulfated steroid hormones.
- STX6410 Steroid hormone sulfatases are conserved proteins that participate, among other processes, stimulating proliferation in hormone-depending cancers.
- Specific inhibitors for this type of enzymes have been generated, such as STX6410, which has been used to treat patients with hormone-depending cancers.
- STX64 treatment also phenocopies other sul-2 mutant phenotypes (Fig. 7d).
- STX64 does not further increase the longevity of sul- 2 deletion mutants, suggesting STX64 increases longevity is by inhibiting the sulfatase activity of SUL-2 (Fig. 1d).
- sul-2 mutations further extend the lifespan of daf-2 reduction of function mutants (Fig. 2a Fig. 8a-b), suggesting that sul-2 acts in a different pathway to regulate longevity.
- the increased longevity of sul-2 mutants is mainly suppressed by DAF-16 loss-of-function (Fig. 2b and Fig. 8c-d).
- Longevity conferred by lack of germline also requires DAF-1612, 13, which translocates to the nuclei mainly in intestinal cells.
- DAF-16 localises to the nucleus of most cells.
- DAF-16 localises predominantly to intestinal nuclei (Fig. 9a-b), suggesting a role for sul-2 in germline mediated longevity.
- DAF-36 converts cholesterol to 7-dehydrocholesterol in the first step of the biosynthetic pathway of A7-DA21 , 22. Therefore, DAF-36 is also needed for the increased longevity of germline-less animals23. Similarly, DAF-36 is required for the longevity conferred by the steroid sulfatase inhibitor STX64 (Fig. 2m) placing the signal generated by sulfated steroid hormones upstream of or parallel to the biosynthesis of DAs. sul-2 is expressed in sensory neurons
- sul-2 is expressed only in a few sensory neurons, mainly in the amphids ADF and ASE, and phasmids PHA and PHB. There is no detectable expression in the germline in any transgenic strains (Fig. 2n and Fig. 10, 11a-b). ASE neurons are responsible for the attractive response of Na+ and CI-, among others. Defects in odour sensing affect longevity.Therefore, we tested the ability of sul-2 mutants to respond to Cl- or Na+ and found no differences compared to wild type animals (Fig. 11c-d).
- sul-2 mutation increases the longevity of daf-10(m79), a long-lived mutant defective in sensory cilia formation25 (Fig. 11e). These results show that the longevity phenotype observed in sul- 2 mutants is not due to the impaired functionality of sensory neurons.
- Reduction of sul-2 improves aging associated diseases in C. elegans Aging is considered the main risk factor for the onset of neurodegenerative disorders like Parkinson, Huntington, or Alzheimer’s diseases. These disorders are caused by the progressive decline of proteostasis, which results in protein aggregation that compromises cellular functions and finally causes cell death.
- Germlinedeficient C. elegans delay the symptoms derived from the proteotoxicity of ectopically expressed human p-amyloid (PA).
- sul-2 mutations or STX64 treatment improves the symptoms of C. elegans models for neurodegenerative diseases.
- a C. elegans Parkinson’s disease model in which human a-synuclein expression in muscle cells causes age-dependent paralysis, sul-2 mutation or treatment with STX64 significantly improved mobility (Fig. 3a-b, Fig. 12a-c).
- sulfated hormones In mammals, sulfated hormones have been long considered inactive forms that function mainly as reservoirs and are activated by steroid sulfatases6, although a direct action of sulfated hormones in the reproductive and the nervous system has been observed. In this last tissue, those hormones are named neurosteroids and their main function is the modulation of neurotransmition. In order to sort out whether the beneficial effect of sul-2 inhibition is due to the reduction of non-sulfated hormones or the increase of sulfated hormones, we tested the commercially available sulfated steroid hormones that are highly presented in the mutant (Table 1).
- strains were synchronized by hypochlorite treatment of gravid adults, grown up during two generations at 16 °C.
- ES treatment started at L4 stage, control and treated plates were prepared fresh and animals transferred each three days until death. Animals lost or dead by non-physiological causes were censured. Survival curves were generated using the product-limit method of Kaplan and Meier. The log-rank (Mantel-Cox) test was used to evaluate differences in survival and p-values lower than 0.05 were considered significant. Controls of all lifespan assays were fer-15(b26) mutant background, to avoid progeny production. fer-15(b26) mutation does not have effect on life span.
- Figure 20 shows the results of an "Open Field” test and of a "Y maze” test.
- the "Open Field” is a test where the mouse must explore an unknown space for 15 minutes. In this test, both mobility and habituation (cognitive parameter that measures the individual's ability to recognize the environment) can be studied.
- the "Y maze” test is a test where the mouse explores the maze for 6 minutes. This test evaluates the distance traveled as a measure of mobility and the ability to visit triplets of arms without repeating any of them as a sign of immediate memory. The results show that treatment with STX-64, E-S or both tend to improve the motor ability of 16-month-old mice.
- habituation ability and immediate memory of aged animals are improved by treatment with STX, E-S and the combination of both (the compounds improved Age-Associated Memory Impairment).
- Figure 21 shows the evaluation of learning and memory abilities of aged mice was determined in the object recognition test and the passive avoidance test.
- object recognition after training with identical objects, tests the ability to identify different objects exposed together with a familiar one. When this happens, the object discrimination index is positive and higher than 0.1.
- passive avoidance the aim is to teach the mouse that its preferred place does not suit it and for them it is associated with an aversive stimulus.
- the entry latency to the preferred site is established and the aversive stimulus is administered.
- STM and LTM short- and long-term memory tests if the mouse associated the preferred place to the aversive stimulus, the input latency increases with respect to training.
- Vehicle-treated aged mice showed impairments in learning ability and thus memory. In both tests, mice treated with STX, ES and the combination of both showed significantly both learning (STM) and ability to store information (memory) (the compounds improved Age-Associated
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Biomedical Technology (AREA)
- Neurosurgery (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Neurology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MX2023004825A MX2023004825A (en) | 2020-10-26 | 2021-10-26 | Epitestosterone sulphate and/or a steroid sulfatase inhibitor for use in treating or improving age related cognitive impairment. |
| CA3199813A CA3199813A1 (en) | 2020-10-26 | 2021-10-26 | Sulfated c19 steroid hormones to protect against aging-associated proteotoxicity |
| US18/250,692 US20240108637A1 (en) | 2020-10-26 | 2021-10-26 | Epitestosterone sulphate and/or a steroid sulfatase inhibitor for use in treating or improving age related cognitive impairment |
| JP2023525496A JP2023546515A (en) | 2020-10-26 | 2021-10-26 | Sulfated C19 steroid hormone to protect against age-related protein toxicity |
| AU2021372716A AU2021372716A1 (en) | 2020-10-26 | 2021-10-26 | Epitestosterone sulphate and/or a steroid sulfatase inhibitor for use in treating or improving age related cognitive impairment |
| CN202180087664.4A CN116801877A (en) | 2020-10-26 | 2021-10-26 | Epitestosterone sulfate and/or steroid sulfatase inhibitors for the treatment or improvement of age-related cognitive impairment |
| EP21814685.0A EP4232022A2 (en) | 2020-10-26 | 2021-10-26 | Epitestosterone sulphate and/or a steroid sulfatase inhibitor for use in treating or improving age related cognitive impairment |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP20382930.4A EP3988095A1 (en) | 2020-10-26 | 2020-10-26 | Sulfated c19 steroid hormones to extend lifespan and protect against aging-associated proteotoxicity |
| EP20382930.4 | 2020-10-26 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2022090252A2 true WO2022090252A2 (en) | 2022-05-05 |
| WO2022090252A3 WO2022090252A3 (en) | 2022-06-02 |
| WO2022090252A9 WO2022090252A9 (en) | 2022-07-28 |
Family
ID=73030060
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2021/079715 Ceased WO2022090252A2 (en) | 2020-10-26 | 2021-10-26 | Sulfated c19 steroid hormones to protect against aging-associated proteotoxicity |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20240108637A1 (en) |
| EP (2) | EP3988095A1 (en) |
| JP (1) | JP2023546515A (en) |
| CN (1) | CN116801877A (en) |
| AU (1) | AU2021372716A1 (en) |
| CA (1) | CA3199813A1 (en) |
| MX (1) | MX2023004825A (en) |
| WO (1) | WO2022090252A2 (en) |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2002257430A1 (en) * | 2001-05-01 | 2002-11-11 | Mcgill University | Androgen-mediated neuroprotection and uses thereof |
| EP1310258A1 (en) * | 2001-11-08 | 2003-05-14 | Etienne-Emile Baulieu | Enantiomers of steroids for the enhancement of memory and cognitive function |
| GB0511190D0 (en) * | 2005-06-01 | 2005-07-06 | Sterix Ltd | Use |
| ES2514140B1 (en) * | 2013-03-26 | 2015-08-03 | Universidad Pablo De Olavide | Use of the STX64 steroid sulfatase inhibitor for the treatment of aging |
| WO2016138135A1 (en) * | 2015-02-25 | 2016-09-01 | Teva Pharmaceuticals International Gmbh | Sigma-1 receptor modulators for treating huntington's disease |
| EP3988096A1 (en) * | 2020-10-26 | 2022-04-27 | Universidad Pablo de Olavide OTRI | Sulfated c19 steroid hormones to treat and/or prevent proteotoxicity in protein-aggregation diseases |
-
2020
- 2020-10-26 EP EP20382930.4A patent/EP3988095A1/en not_active Withdrawn
-
2021
- 2021-10-26 US US18/250,692 patent/US20240108637A1/en active Pending
- 2021-10-26 WO PCT/EP2021/079715 patent/WO2022090252A2/en not_active Ceased
- 2021-10-26 MX MX2023004825A patent/MX2023004825A/en unknown
- 2021-10-26 EP EP21814685.0A patent/EP4232022A2/en active Pending
- 2021-10-26 CN CN202180087664.4A patent/CN116801877A/en active Pending
- 2021-10-26 CA CA3199813A patent/CA3199813A1/en active Pending
- 2021-10-26 JP JP2023525496A patent/JP2023546515A/en active Pending
- 2021-10-26 AU AU2021372716A patent/AU2021372716A1/en active Pending
Non-Patent Citations (13)
| Title |
|---|
| "Remington's Pharmaceutical Sciences", 1980 |
| "UniProtKB", Database accession no. P08842 |
| CAS , no. 186303-55-9 |
| EISENBERGJUCKER, CELL, vol. 148, no. 6, 2012, pages 1188 - 203 |
| FOLSTEIN ET AL., J PSYCHIATRIC RES, vol. 12, 1975, pages 189 - 98 |
| HOFFNERDIJAN, BRAIN SCI., vol. 4, no. 1, 2014, pages 91 - 122 |
| KLUGER ET AL., J GERIATR PSYCHIATRY NEUROL, vol. 12, 1999, pages 168 - 79 |
| PETERSEN ET AL., SRCH. NEUROL., vol. 56, 1999, pages 303 - 308 |
| PETERSEN: "Mild cognitive impairment: Aging to Alzheimer's Disease", 2003, OXFORD UNIVERSITY PRESS |
| PUROHITFOSTER, J. ENDOCRINOL., vol. 212, no. 2, 2012, pages 99 - 110 |
| REY, L'EXAMEN CLINIQUE EN PSYCHOLOGIE, 1964 |
| ROBBINS ET AL., DEMENTIA, vol. 5, 1994, pages 266 - 81 |
| SIPE ET AL., AMYLOID, vol. 19, no. 4, 2012, pages 167 - 70 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2023546515A (en) | 2023-11-02 |
| CN116801877A (en) | 2023-09-22 |
| AU2021372716A9 (en) | 2024-07-25 |
| WO2022090252A3 (en) | 2022-06-02 |
| MX2023004825A (en) | 2023-07-21 |
| WO2022090252A9 (en) | 2022-07-28 |
| US20240108637A1 (en) | 2024-04-04 |
| AU2021372716A1 (en) | 2023-06-08 |
| EP3988095A1 (en) | 2022-04-27 |
| EP4232022A2 (en) | 2023-08-30 |
| CA3199813A1 (en) | 2022-05-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Maeda et al. | Retinopathy in mice induced by disrupted all-trans-retinal clearance | |
| Browne et al. | Treatment with a catalytic antioxidant corrects the neurobehavioral defect in ataxia–telangiectasia mice | |
| JP6876047B2 (en) | Compounds for the treatment of diseases associated with mitochondrial reactive oxygen species (ROS) production | |
| Kong et al. | Augmenter of liver regeneration-mediated mitophagy protects against hepatic ischemia/reperfusion injury | |
| US20260083700A1 (en) | Methods for treating and/or preventing protein-aggregation diseases | |
| JP2017520553A (en) | Ketobutyric acid compounds and compositions for treating age-related symptoms and diseases | |
| US20230405020A1 (en) | Sulfated c19 steroid hormones to treat and/or prevent proteotoxicity in protein-aggregation diseases | |
| US20240108637A1 (en) | Epitestosterone sulphate and/or a steroid sulfatase inhibitor for use in treating or improving age related cognitive impairment | |
| US8673977B2 (en) | Choline salts of succinic acid for the treatment of depression, anxiety, schizophrenia, sleep disorder, and epilepsy | |
| Tai et al. | Electrolytic lesions of the nucleus accumbens in rats which abolish the PREE enhance the locomotor response to amphetamine | |
| JP2019501972A (en) | Tacrolimus for the treatment of TDP-43 proteinosis | |
| HK40085227A (en) | Compositions for treating and/or preventing protein-aggregation diseases | |
| Stahlmann | Effects on connective tissue structures | |
| WO2025091016A1 (en) | Compositions comprising p75ntr inhibitors and recombinant nerve growth factor and methods for the treatment of corneal diseases and disorders using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21814685 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 3199813 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18250692 Country of ref document: US Ref document number: 2023525496 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202317034218 Country of ref document: IN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2021814685 Country of ref document: EP Effective date: 20230526 |
|
| ENP | Entry into the national phase |
Ref document number: 2021372716 Country of ref document: AU Date of ref document: 20211026 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202180087664.4 Country of ref document: CN |