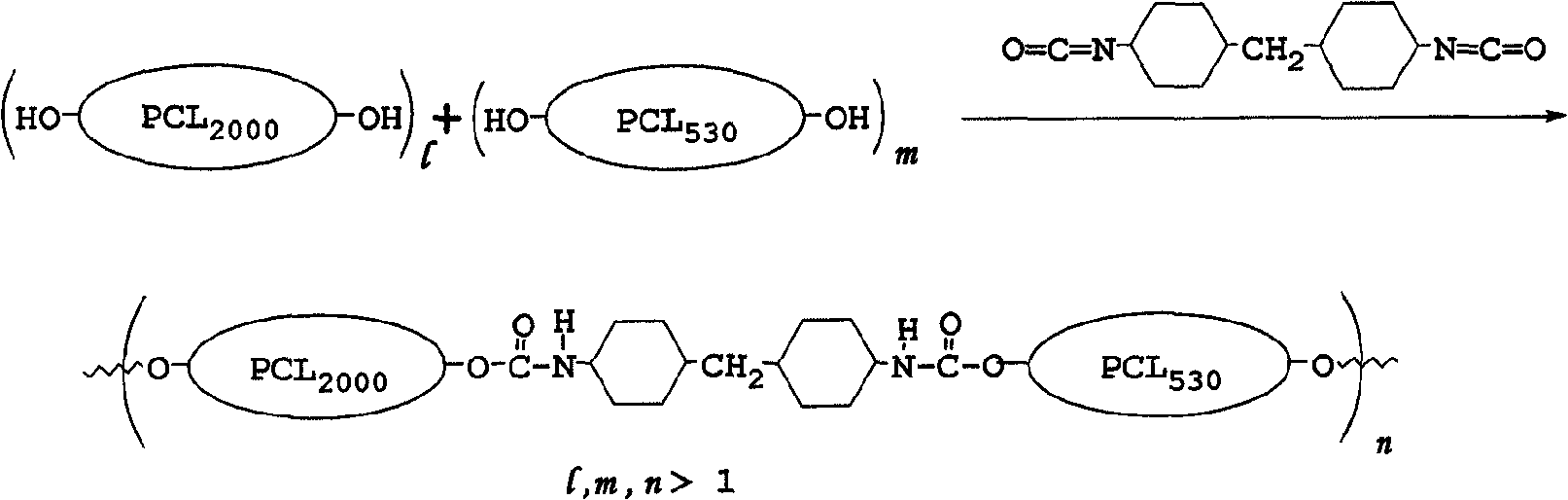
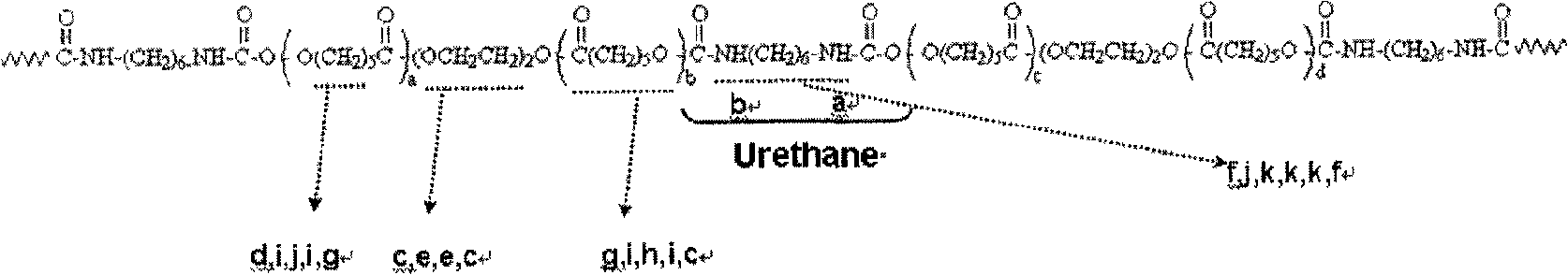
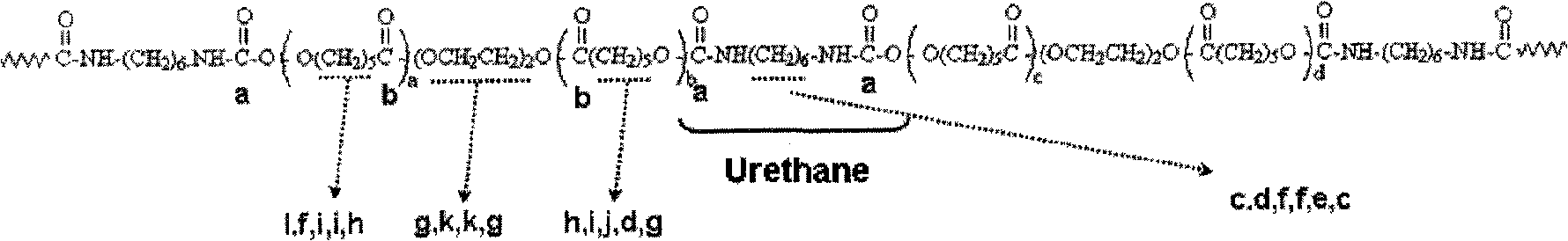
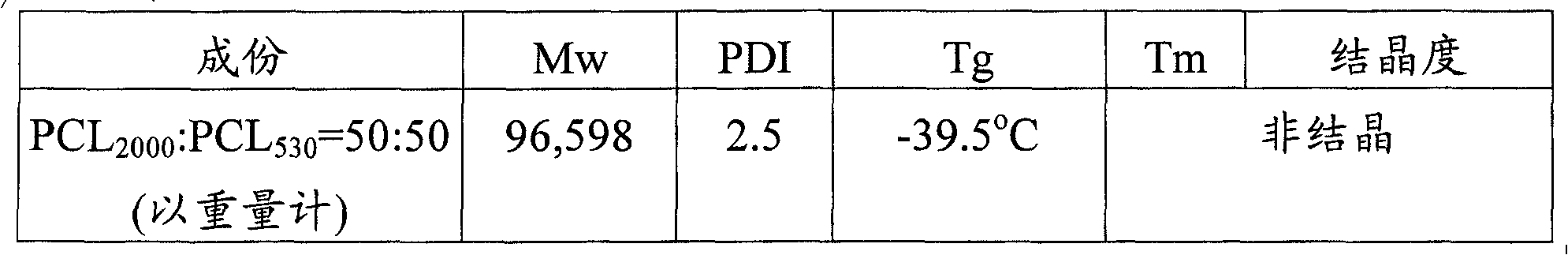
Embodiment
In the Bioabsorbable polyester material degradation process, fs is that water molecules enters noncrystalline domain (amorphous region) and is hydrolyzed at ester bond (ester bond), and next water molecules just can enter crystal region (crystalline region) and carries out hydrolysis more slowly.Most polyester materials have crystallographic property (for example: the PCL material has hypocrystalline (semi crystalline) character, and the degradation time of mentioning PCL in the past in the document is in general all greater than 2 years).The present invention utilizes the aliphatics coupler (for example: H
12MDI) polyester with the differing molecular chain length becomes multipolymer, wherein the amino-formate bond that is produced (Urethanelinkage) can reduce the crystallization degree of original material, and after polyester material degree of crystallinity reduces, the molecular chain that is easier to make large quantity of moisture enter material is hydrolyzed, therefore this material degradation time in animal body will shorten, and reaches the purpose of controlled material degradation time.
The invention provides novel Bioabsorbable aliphatic polyester copolymer, this Bioabsorbable aliphatic polyester copolymer copolymerization first polyester and second polyester in the presence of coupler form, and wherein but first polyester and second polyester have identical repeating unit have different weight average molecular weights.Bioabsorbable aliphatic polyester copolymer of the present invention is easy to prepare and be beneficial to mass production.In addition, aliphatic polyester copolymer of the present invention also has favorable mechanical characteristic and regulatable biological decomposition time, but wherein the biology resolving time of this aliphatic polyester copolymer, can utilize weight or molar ratio between first and second polyester to adjust.
First and second polyester is polymerized by the monomer with identical repeating unit, and its difference it should be noted that only in the molecular weight difference of first and second polyester the number of the hydroxy functional group on first and second polyester is not less than 2.Described repeating unit is derived from following monomer: caprolactone (caprolactone), butyrolactone (butyrolactone), D, L-lactic acid lactide (D, L-lactide), D-lactic acid lactide (D-lactide), L-lactic acid lactide (L-lactide), D, L-lactic acid (D, L-lactic acid), D-lactic acid (D-lactic acid), L-lactic acid (L-lactic acid), glycollide (glycolide), glycolic acid (glycolic acid), hydroxycaproic acid (hydroxyhexonoic acid), hydroxybutyric acid (hydroxy butyric acid), valerolactone (valerolactone), hydroxypentanoic acid (hydroxy valeric acid), oxysuccinic acid (malic acid) or its multipolymer.When the hydroxy number that has when employed first and second polyester was 2, the aliphatic polyester copolymer of gained was a linear copolymer.In addition, the hydroxy number that has when employed first and second polyester is greater than 2 the time, and the aliphatic polyester copolymer of gained is the network multipolymer.
The weight-average molecular weight of first and second polyester is between 150~50000 dalton (Dalton), preferably between 200~30000 dalton, more preferably between 200~20000 dalton.The part by weight of first and second polyester can be between 9.5: 0.5~0.5: 9.5, preferably between 8: 2~2: 8, more preferably between 7: 3~3: 7.In addition, the physical strength of described Bioabsorbable aliphatic polyester copolymer can and become big along with the polyester weight ratio of constituents example increase with higher molecular weight.
According to a preferred embodiment of the invention, but the biology resolving time of this aliphatic polyester copolymer also can utilize the molecular weight differences between first and second polyester to adjust, wherein the weight-average molecular weight gap of first polyester and second polyester can be more than 200 dalton, is preferably more than 500 dalton.In addition, but the biology resolving time of this aliphatic polyester copolymer can be according to the weight-average molecular weight gap of first polyester and second polyester and in 1~36 month scope, adjusts.For example, biology can be transferred to for 1~December the decomposition cycle meeting the demand that short-term is implanted, or should can transfer to 13~36 months the decomposition cycle to meet the demand of long-term implantation by biology.
Coupler of the present invention can comprise and has more than 2 or 2-aliphatic isocyanates of N=C=O functional group, its can with the hydroxyl reaction of first and second polyester, this coupler is epoxide, acid anhydrides or isocyanic ester, can comprise two-1,4-cyclohexyl diisocyanate (bis-1,4-cyclohexyldiisocyanate, H
12MDI), hexamethylene diisocyanate (diisocyanatohexane, HDI), 5-isocyanato-1-(isocyanato methyl)-1,3,3-trimethyl-cyclohexane (5-Isocyanato-1-(isocyanatomethyl)-1,3,3-trimethylcyclohexane, IPDI), tetramethyl-benzene dimethyl vulcabond (tetramethyl-m-xylylene diisocyanate, TMXDI) or its mixture.
In embodiment of the present invention, this aliphatic polyester copolymer with Bioabsorbable further by first polyester, second polyester, coupler, and Preparation of Catalyst form, wherein this catalyst pack contains organo-metallic catalyst, amine catalyst or its mixture, wherein this organo-metallic catalyst comprises dibutyl tin laurate (dibutyltin dilaurate), the metatitanic acid tert-butyl ester (tertbutyl titanate), dibutyl tin (dibutyltin), stannous octoate (stannous octoate), or its mixture.This amine catalyst comprises N, N '-dimethylcyclohexylamine (N, N-Dimethylcyclohexylamine), 1,1,3,3-tetramethyl guanidine (1,1,3,3-tertamethylguanidine), Tetramethyl Ethylene Diamine (tetramethylethylenediamine), Triethylene Diamine (triethylene diamine), tripropylene glycol (tripropylene glycol), N, N '-lupetazin (N, N '-dimethylpiperazine), N, N, N ', N '-tetramethyl--1,3-butanediamine (N, N, N ', N '-tetramethyl-1,3-butanediamine), tri methyl piperazine (trimethylpiperazine), 1, and two (2-the hydroxypropyl)-2-methylpiperazines of 4-(1,4-bis (2-hydroxylproply)-2-methylpiperazine), N-hydroxyethyl piperazine (N-hydroxyethylpiperazine), 1,3,5-three (dimethylaminopropyl) six hydrogen-s-triazine (1,3,5-tris (dimethylaminopropyl) hexahydrostriazine), dimethyl benzene methylamine (dimethylbenzylamine), 4-ethyl morpholine (4-ethylmorpholine), 2,2-dimorpholine benzyl ethyl ether (2,2-dimorpholinoethyl ether), triethylamine (triethylamine), 2,2 '-two (2-ethyls-2-azo bicyclic ethers) (2,2 '-bis (2-ethyl-2-azobicycloether)), two azo double-octanes (diazobicyclooctane), dimethylamino propylamine (dimethylaminopropylamine), diethylamino ethamine (diethylaminoethylamine) or its mixture.
The method for preparing this Bioabsorbable aliphatic polyester copolymer is hard-core, can be bulk polymerization, solution polymerization, letex polymerization, dispersion polymerization, suspension polymerization or reaction extrusion molding.
About solution polymerization, at first first and second polyester (but first polyester and second polyester have identical repeating unit have different weight average molecular weights) is dissolved in the organic solvent.Then, (temperature of reaction is between 30 ℃~200 ℃, preferably between 40 ℃~150 ℃ to carry out copolymerization this coupler (and catalyzer) to be added said mixture.Above-mentioned organic solvent can be N, dinethylformamide (N, N-dimethylformamide, DMF), N, N-N,N-DIMETHYLACETAMIDE (N, N-dimethylacetamide, DMAc), dimethyl sulfoxide (DMSO) (DMSO), tetrahydrofuran (THF) (THF), chloroform, methylene dichloride (DCM), two divinyl oxides, benzene or its mixture.About bulk polymerization, it mixes first and second polyester and coupler in its temperature more than fusing point, then coupler (and catalyzer) is added said mixture, to carry out copolymerization.This temperature of reaction can be between 40 ℃~250 ℃, preferably between 50 ℃~200 ℃.About reactive extrusion molding, it mixes (and catalyzer) with first and second polyester and coupler, and drops into twin screw extruder, and extruder temperature is set between 50 ℃~250 ℃, preferably between 60 ℃~220 ℃.
The present invention also provides medical implanted device, it comprises described Bioabsorbable aliphatic polyester copolymer, and wherein this implanted device is used for the reparation of soft or hard tissues such as nerve, endocranium, ligament, tendon, inguinal hernia, shoulder rotation sleeve, meniscus, muscle, joint, spinal cord, cranium face or jaw face.In addition, the shape of this medical implanted device can be hollow conduit, multiple channel duct, film, membranaceous coiling thing, tabular or spongy (porousness) structure of above-mentioned form.
About the nerve reparation, in general, the reason of nervous system injury can be generalized into following several: burn (3) geneogenous deformity (4) tumour or the cancer of (1) external force or mechanical force injury (2) burn and scald or chemical substance cause.Generally speaking, after the nerve structure of nerve tract was subjected to acute (for example mechanical injury) or chronic (for example growth of tumor) injury, nerve fiber begins that promptly a succession of change takes place and physiological function is slowly lost.Therefore, for the patient of peripheral injured nerve, the most frequently used clinically method that also extensively adopts is to use the mode of micrurgy with fascicular neurorrhaphy impaired or fracture to the patient.
The comparatively unessential one section nerve of needs of patients excision self (normally nervus peronaeus, sural nerve) is repaired injured position, therefore can cause the secondary nervosa injury of patient but in this case.Since patient self can capture the limited length of repairing the injury nerve, bottleneck when having caused treatment, therefore developing the artificial neuron prosthetic device seems extremely important, an artificial neuron prosthetic device need possess following characteristics are arranged: 1. support and quarantine measures, the neural axon regeneration space at fracture or two ends, illness position is provided, provide neural axon to extend simultaneously and attach interface, guiding is neural towards the certain orientation growth, avoids aixs cylinder to be entangled with; 2. support and the employed device of quarantine measures, it must be the mutual connectivity structure of porousness, and nutrition and metabolite mass-energy are freed in and out inside and outside with device in the neurotization reparation, is beneficial to the psychological need of nervous tissue.3. to be preferably bioabsorbable material with device made in the neurotization reparation, and degradation time the best between several months to 1 year, so, can reduce the foreign body reaction chance that bioabsorbable material implants.
The medical implant apparatus that is used for neural reparation of the present invention (for example: N is dissolved in organic solvent with aliphatic polyester copolymer of the present invention, dinethylformamide, N, N-N,N-DIMETHYLACETAMIDE, dimethyl sulfoxide (DMSO), tetrahydrofuran (THF), chloroform, methylene dichloride, two divinyl oxides, benzene or its mixture) in, to obtain Biodegradable polyester copolymer solution.Then, in surface or cylindric mould, utilize peptizer to carry out solidification forming then this Biodegradable polyester copolymer solution coat.After removing this cylindric mould, can obtain hollow and porous aliphatic polyester copolymer conduit, as the described medical implant apparatus that is used for neural reparation.
The thickness of described duct wall can be between 0.05~1.5mm.And peptizer can be water or wrap aqueous organic solvent, wherein the weight ratio of water and organic solvent is 1: 3~20: 1, and this organic solvent can be amine, ketone, alcohol or its mixture, be preferably ketone and alcohols, for example N, dinethylformamide, N,N-dimethylacetamide, acetone, methylethylketone, methyl alcohol, ethanol, propyl alcohol, Virahol and butanols.
In some embodiments of the invention, have low-molecular-weight oligomer and also can be prepared into this Biodegradable polyester copolymer solution to form medical implant apparatus with aliphatic polyester copolymer of the present invention.The low-molecular weight oligo thing that is fit to can be poly-own lactones trivalent alcohol (PCLTL), poly-own lactones dibasic alcohol (PCLDL), polycaprolactone polyol (PCL), poly(lactic acid) (PLA), polyoxyethylene glycol (PEG), polypropylene glycol (PPG), poly-butyl ether glycol (PTMG) or its mixture.
This hole that is used for the neural medical implant apparatus of repairing when bore hole size can utilize the kind that changes oligomer, molecular weight and recently adjust with the weight of polyester copolymer.The molecular weight of this oligomer is between 200~4000.And the weight ratio of this oligomer and this polyester copolymer is between 1: 20~1: 1, preferably between 1: 10~2: 3.In addition, the hole of hollow of the present invention and porous aliphatic polyester copolymer conduit can link to each other mutually and connect.The hole that should link to each other and connect can provide preferable microenvironment, and required nutrition of cell and metabolite mass-energy are freed in and out inside and outside with device in the neurotization reparation, was beneficial to the psychological need of nervous tissue.
The bootable spinous process axle of medical implant apparatus that is used for neural reparation of the present invention is prolonging a specific direction growth, and avoids nerve to tangle.Because this material that is used for the neural medical implant apparatus of repairing is an aliphatic polyester copolymer of the present invention, this biology that is used for the neural medical implant apparatus of repairing can decompose the cycle and can optionally be adjusted into for 3~December, to avoid neural extrusion and foreign body reaction.
That causes behind the resection operation when the damaged reason of endocranium tissue (duralike tissue) mostly is damaged (2) tumour that causes behind the injury of (1) external force or mechanical force or its debridement surgical or cancerous lesion greatly and takes place is damaged.Generally speaking, dura defect can cause that celiolymph leaks outside, epilepsy and tissue are stained with complication such as sticking, can impose endocranium substituent (dural substitutes) clinically, and endocranium is repaired, and produces to prevent above-mentioned complication.And an artificial meninx substituent prosthetic device need possess following characteristics are arranged: 1. isolated material/device, be preferably multilayered structure, and wherein, the face of pressing close to cerebral tissue or nervous tissue is preferably smooth-flat-surface, can prevent the sticking phenomenon of being stained with of cerebral tissue or nervous tissue; And the upper end is preferably uneven surface near the skull face, can help the endocranium tissue (for example: fibrous tissue (fibrous tissue)) attaching and hyperplasia.2. isolated material/device when repairing defect, can prevent celiolymph leak outside (Cerebrospinalfluid (CSF) leakage).3. isolated material/device is preferably the mutual connectivity structure of porousness, makes nutrition and metabolite mass-energy free in and out the psychological need that is beneficial to organize inside and outside device.And it is made that device is preferably bioabsorbable material, the injury that this can reduce foreign body in vivo reaction chance and be avoided second operation to cause.
According to one preferred embodiment of the present invention, the invention provides a kind of endocranium substituent and manufacture method thereof that endocranium is repaired that be used for by aliphatic polyester copolymer of the present invention constituted.
The manufacture method of aliphatic polyester copolymer endocranium substituent provided by the present invention comprises following steps.At first, aliphatic polyester copolymer and low-molecular weight oligo thing are dissolved in the organic solvent, form bioresorbable aliphatic polyester polymer solution.Then, with this bioresorbable aliphatic polyester polymer solution coat in die surface, this die surface has concavo-convex or rough surface structure, and the height of this concavo-convex or uneven surface is between 10 μ m~1000 μ m, most preferably between 20 μ m~500 μ m, next bioresorbable aliphatic polyester polymer solution is poured in the above-mentioned container with concavo-convex or uneven surface, make this bioresorbable macromolecular solution have solid shape, for example, the film of the about 0.01mm to 5mm of thickness most preferably is the film of 0.1mm to 3mm.Then, the container that will scribble the mould of bioresorbable aliphatic polyester polymer solution or interior Sheng bioresorbable aliphatic polyester polymer solution is again inserted in the solidification liquid (coagulant) and is contacted with solidification liquid, to solidify the porousness dura mater substituent that forms multilayered structure.The hole of this porousness dura mater substituent when bore hole size can utilize the kind that changes oligomer, molecular weight and recently adjust with the weight of polyester copolymer.The low-molecular weight oligo thing that is fit to can be poly-own lactones trivalent alcohol (PCLTL), poly-own lactones dibasic alcohol (PCLDL), polycaprolactone polyol (PCL), poly(lactic acid) (PLA), polyoxyethylene glycol (PEG), polypropylene glycol (PPG), poly-butyl ether glycol (PTMG) or its mixture.The molecular weight of this oligomer is between 200~4000.And the weight ratio of oligomer and polyester copolymer is between 1: 20~1: 1, preferably between 1: 10~2: 3.Employed organic solvent for example can be: N, dinethylformamide, N,N-dimethylacetamide, dimethyl sulfoxide (DMSO) (DMSO), tetrahydrofuran (THF) (THF), chloroform, methylene dichloride (DCM), two divinyl oxides, benzene or its mixture).
It should be noted that adjusting suitable solidification liquid can prepare upper surface and have less or than the porousness dura mater substituent of small holes, to form smooth upper surface.According to the present invention, this peptizer can be water or wrap aqueous organic solvent, wherein the weight ratio of water and organic solvent is 1: 3~20: 1, and this organic solvent can be amine, ketone, alcohol or its mixture, be preferably ketone and alcohols, N for example, dinethylformamide, N,N-dimethylacetamide, acetone, methylethylketone, methyl alcohol, ethanol, propyl alcohol, Virahol and butanols.
Particularly, the artificial meninx substituent of the porousness of multilayered structure of the present invention can be made for the bioresorbable macromolecular material, and it is made to be preferably aliphatic polyester copolymer of the present invention.
The artificial meninx substituent of the porousness of multilayered structure is designed among the present invention:
(a) multilayered structure: upper strata--the face of pressing close to cerebral tissue or nervous tissue is a smooth-flat-surface, can prevent from so that contact friction causes between cerebral tissue or nervous tissue and the material to be stained with sticking phenomenon; Middle--this position is preferably cellular structure, so, can allow host tissue (host tissue) replace the artificial meninx substituent of Bioabsorbable (for example: guiding endocranium tissue growth and reparation) of implantation gradually; The upper strata--the face near skull is preferably uneven surface, and (for example: (fibrous tissue fibrous tissue) attaches and hyperplasia can to help the endocranium tissue.
(b) has the material of preferable rubber elastomer characteristics: when the clinicist repairs defect, when sewing up artificial meninx substituent and original endocranium and organizing, when suture passes the made worker's meninx substituent of aliphatic polyester copolymer, because of aliphatic polyester copolymer material of the present invention owing to contain the carbamate bond, therefore has preferable rubber elastomer characteristics, when suture passes through the made artificial meninx substituent of aliphatic polyester copolymer material, can produce the deflation effect voluntarily, can prevent that celiolymph from passing through the slit that is produced via suture and causing and leak outside.
(c) Bioabsorbable and cellular structure: artificial meninx substituent/device that aliphatic polyester copolymer is made, last middle level (near skull and position, middle layer) is the mutual connectivity structure of porousness, so, nutrition and metabolite mass-energy are freed in and out inside and outside device, be beneficial to the demand of endocranium tissue growth.And it is made that this device is preferably bioabsorbable material, and degradation time the best is lower than 12 months, so, and the injury that can reduce foreign body in vivo reaction chance and avoid second operation to cause.
Except above-mentioned said application, aliphatic polyester copolymer of the present invention also can be used as environmentally-friendly plastic bag or other commodity, to replace the plastic material that biology can't decompose.
Following spy enumerates several embodiment, and in conjunction with the accompanying drawings, in order to explanation the present invention, can be more clear in the hope of making the present invention:
The preparation of aliphatic polyester copolymer
Embodiment 1
At first be averaged molecular weight and be 2000 polycaprolactone (PCL2000) 75 grams and put into round-bottomed bottle, and it is dissolved in the 58.5 gram N,N-dimethylacetamide fully.Then, add 22.18 gram H
12MDI as coupler and 60mg dibutyl tin laurate (DBTDL) as catalyzer, after 60 ℃ stirring finishes down, is that 530 polycaprolactone (PCL530) 25 grams (being dissolved in 58.5 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe the variation of resin viscosity, and mend an amount of H respectively
12MDI, DMAc and DBTDL, the reaction times is eight hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 31.93 grams, 234 grams and 0.4 gram, after reacting completely, adds the isocyanic ester of 0.3 gram dibutylamine with consume residual again, and stopped reaction.Above-mentioned reaction process is as shown below.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing, and spends 24 hours and utilizes ether to extract with soxhlet's extractor.The structure evaluation spectrum of this polyester copolymer (
1H-NMR,
13C-NMR, FT-IR) respectively as shown in Figure 1, 2, 3.C, d, e, g, h, i and j represent at each peak caprolactone-CH among Fig. 1
2-hydrogen signal, peaks such as a, b then represent carbamate bond (Urethanelinkage)-hydrogen signal of NH-, as follows:
Please refer to Fig. 2, peaks such as c, d, e, f are H
12The carbon signal of MDI (coupler), and a, the carbon signal of carbonyl is represented at peaks such as b, the carbon signal of own lactones is represented on peaks such as d, f, g, h, i, j, k and l, and the carbon signal as the dimethyl Asia (DMSO) of measuring solvent is represented on the DMSO peak.
Please refer to Fig. 3, peak 1,2,3,4,5 and 6 show the FT-IR signal of embodiment 1 described aliphatic polyester copolymer.
In addition, the weight-average molecular weight (Mw) of embodiment 1 described aliphatic polyester copolymer, polydispersity index (polydispersity index, PDI), glass transition temp (glass transition temperature, Tg), fusing point (Tm), and degree of crystallinity (crystallinity) is also measured, and its result is as shown in table 1.
Table 1
Degree of crystallinity converts out with following formula:
C=ΔH/(wPCL×ΔHref)
C: degree of crystallinity
WPCL: polycaprolactone (PCL) is in the weight fraction of multipolymer
Δ Href:136.1J/g, the enthalpy that crystalline polycaprolactone (PCL) fusion is required
Embodiment 2
At first be averaged molecular weight and be 2000 polycaprolactone (PCL2000) 50 grams and put into round-bottomed bottle, and make it be dissolved in 58.5 gram N,N-dimethylacetamide fully.Then, add 31.28 gram H
12MDI as coupler and 60mg dibutyl tin laurate (DBTDL) as catalyzer, after 60 ℃ stirring finishes down, is that 530 polycaprolactone (PCL530) 50 grams (being dissolved in 58.5 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe resin viscosity and change, and mend an amount of H respectively
12MDI, DMAc and DBTDL, the reaction times is eight hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 38.16 grams, 234 grams and 0.4 gram, after reacting completely, adds the isocyanic ester of 0.1 gram dibutylamine with consume residual again, and stopped reaction.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing,, and spend 24 hours and utilize ether to extract with soxhlet's extractor.The weight-average molecular weight (Mw) of embodiment 2 described aliphatic polyester copolymers, polydispersity index (PDI), glass transition temp (Tg), fusing point (Tm) and degree of crystallinity are also measured, and its result is as shown in table 2.
Table 2
Embodiment 3
At first be averaged molecular weight and be 2000 polycaprolactone (PCL2000) 25 grams and put into round-bottomed bottle, and make it be dissolved in 58.5 gram N,N-dimethylacetamide fully.Then, add 40.35 gram H
12MDI as coupler and 60 milligrams of dibutyl tin laurates (DBTDL) as catalyzer, after 60 ℃ stirring finishes down, is that 530 polycaprolactone (PCL530) 75 grams (being dissolved in 58.5 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe resin viscosity and change, and mend an amount of H respectively
12MDI, DMAc and DBTDL, the reaction times is eight hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 45.19 grams, 234 grams and 0.4 gram, after reacting completely, adds the isocyanic ester of 0.2 gram dibutylamine with consume residual again, and stopped reaction.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing,, and spend 24 hours and utilize ether to extract with soxhlet's extractor.The weight-average molecular weight (Mw) of embodiment 3 described aliphatic polyester copolymers, polydispersity index (PDI), glass transition temp (Tg), fusing point (Tm) and degree of crystallinity are also measured, and its result is as shown in table 3.
Table 3
Embodiment 4
At first be averaged molecular weight and be 10000 polycaprolactone (PCL10000) 75 grams and put into round-bottomed bottle, and make it be dissolved in 58.5 gram N,N-dimethylacetamide fully.Then, add 14.32 gram H
12MDI as coupler and 60 milligrams of dibutyl tin laurates (DBTDL) as catalyzer, after 60 ℃ stirring finishes down, is that 530 polycaprolactone (PCL530) 25 grams (being dissolved in 58.5 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe resin viscosity and change, and mend an amount of H respectively
12MDI, DMAc and DBTDL, the reaction times is eight hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 18.62 grams, 234 grams and 0.4 gram, after reacting completely, adds the isocyanic ester of 0.2 gram dibutylamine with consume residual again, and stopped reaction.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing,, and spend 24 hours and utilize ether to extract with soxhlet's extractor.The weight-average molecular weight (Mw) of embodiment 4 described aliphatic polyester copolymers, polydispersity index (PDI), glass transition temp (Tg), fusing point (Tm) degree of crystallinity is also measured, and its result is as shown in table 4.
Table 4
| Composition |
Mw |
PDI |
Tg |
Tm |
Degree of crystallinity |
| PCL
10000∶PCL
530=75: 25 (by weight)
|
111,523 |
2.6 |
-60.3℃ |
60.2℃ |
44.8% |
Embodiment 5
At first be averaged molecular weight and be 10000 polycaprolactone (PCL10000) 100 grams and put into round-bottomed bottle, and make it be dissolved in 117 gram N,N-dimethylacetamide fully.Then, add 62.62 gram H
12MDI as coupler and 60 milligrams of dibutyl tin laurates (DBTDL) as catalyzer, after 60 ℃ stirring finishes down, is that 530 polycaprolactone (PCL530) 100 grams (being dissolved in 117 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe resin viscosity and change, and mend an amount of H respectively
12MDI, DMAc, and DBTDL, the reaction times is eight hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 67.62 grams, 468 grams and 0.8 gram, after reacting completely, adds the isocyanic ester of 0.2 gram dibutylamine with consume residual again, and stopped reaction.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing,, and spend 24 hours and utilize ether to extract with soxhlet's extractor.The weight-average molecular weight (Mw) of embodiment 5 described aliphatic polyester copolymers, polydispersity index (PDI), glass transition temp (Tg), fusing point (Tm), and degree of crystallinity is also measured, its result is as shown in table 5.
Table 5
| Composition |
Mw |
PDI |
Tg |
Tm |
Degree of crystallinity |
| PCL
10000∶PCL
530=50: 50 (by weight)
|
84,320 |
2.4 |
-55.9℃ |
37.8℃ |
17.7% |
Embodiment 6
At first be averaged molecular weight and be 10000 polycaprolactone (PCL10000) 25 grams and put into round-bottomed bottle, and make it be dissolved in 58.5 gram N,N-dimethylacetamide fully.Then, add 37.73 gram H
12MDI as coupler and 60 milligrams of dibutyl tin laurates (DBTDL) as catalyzer, after 60 ℃ stirring finishes down, is that 530 polycaprolactone (PCL530) 75 grams (being dissolved in 58.5 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe resin viscosity and change, and mend an amount of H respectively
12MDI, DMAc and DBTDL, the reaction times is eight hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 49.8 grams, 234 grams and 0.4 gram, after reacting completely, adds the isocyanic ester of 0.3 gram dibutylamine with consume residual again, and stopped reaction.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing,, and spend 24 hours and utilize ether to extract with soxhlet's extractor.The weight-average molecular weight (Mw) of embodiment 6 described aliphatic polyester copolymers, polydispersity index (PDI), glass transition temp (Tg), fusing point (Tm) and degree of crystallinity are also measured, and its result is as shown in table 6.
Table 6
Embodiment 7
At first be averaged molecular weight and be 10000 polycaprolactone (PCL10000) 80 grams and put into round-bottomed bottle, and make it be dissolved in 58.5 gram N,N-dimethylacetamide fully.Then, add 4.72 gram H
12MDI as coupler and 60 milligrams of dibutyl tin laurates (DBTDL) as catalyzer, after 60 ℃ stirring finishes down, is that 2000 polycaprolactone (PCL2000) 20 grams (being dissolved in 58.5 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe resin viscosity and change, and mend an amount of H respectively
12MDI, DMAc and DBTDL, the reaction times is eight hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 7.31 grams, 234 grams and 0.4 gram, after reacting completely, adds the isocyanic ester of 0.2 gram dibutylamine with consume residual again, and stopped reaction.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing,, and spend 24 hours and utilize ether to extract with soxhlet's extractor.The weight-average molecular weight (Mw) of embodiment 7 described aliphatic polyester copolymers, polydispersity index (PDI), glass transition temp (Tg), fusing point (Tm) and degree of crystallinity are also measured, and its result is as shown in table 7.
Table 7
| Composition |
Mw |
PDI |
Tg |
Tm |
Degree of crystallinity |
| PCL
10000∶PCL
2000=80: 20 (by weight)
|
984,567 |
1.65 |
-49.9℃ |
58.6℃ |
31.3% |
Embodiment 8
At first be averaged molecular weight and be 2000 polycaprolactone (PCL2000) 25 grams and put into round-bottomed bottle, and make it be dissolved in 58.5 gram N,N-dimethylacetamide fully.Then, add 40.35 gram H
12MDI as coupler and 1 gram Triethylene Diamine as catalyzer, 85 ℃ stir down finish after, be that 530 polycaprolactone (PCL530) 75 grams (being dissolved in 58.5 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe resin viscosity and change, and mend an amount of H respectively
12MDI, DMAc and TEDA, the reaction times is 48 hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 45.19 grams, 234 grams and 6 grams, after reacting completely, adds the isocyanic ester of 0.3 gram dibutylamine with consume residual again, and stopped reaction.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing,, and spend 24 hours and utilize ether to extract with soxhlet's extractor.The weight-average molecular weight (Mw) of embodiment 8 described aliphatic polyester copolymers, polydispersity index (PDI) is also measured, and its result is as shown in table 8.
Table 8
| Composition |
Mw |
PDI |
| PCL
2000∶PCL
530=25: 75 (by weight)
|
103,471 |
2.1 |
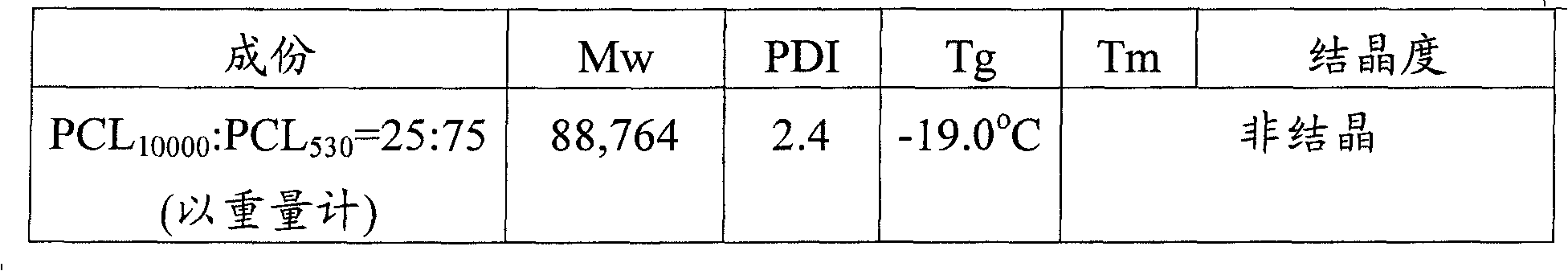
Embodiment 9
At first be averaged molecular weight and be 10000 polycaprolactone (PCL10000) 25 grams and put into round-bottomed bottle, and make it be dissolved in 58.5 gram N,N-dimethylacetamide fully.Then, add 37.73 gram H
12MDI as coupler and 1 gram Triethylene Diamine as catalyzer, 80 ℃ stir down finish after, be that 530 polycaprolactone (PCL530) 75 grams (being dissolved in 58.5 gram N,N-dimethylacetamide) splash in the round-bottomed bottle with the speed of 2ml/min with molecular-weight average.In polyreaction, observe resin viscosity and change, and mend an amount of H respectively
12MDI, DMAc, and TEDA, the reaction times is 48 hours.Last H
12The total amount that MDI, DMAc and DBTDL add is respectively 49.8 grams, 234 grams and 6 grams, after reacting completely, adds the isocyanic ester of 0.1 gram dibutylamine with consume residual again, and stopped reaction.
Then, the resin that negate should be finished (allocate solid-state part=20%) is coated glass substrate, to remove DMAc, obtains product with 80 ℃ of bakings 3 hours, with its pulverizing,, and spend 24 hours and utilize ether to extract with soxhlet's extractor.The weight-average molecular weight (Mw) of embodiment 9 described aliphatic polyester copolymers, polydispersity index (PDI) is also measured, and its result is as shown in table 9.
Table 9
| Composition |
Mw |
PDI |
| PCL
10000∶PCL
530=25: 75 (by weight)
|
195,500 |
1.9 |
Embodiment 10
Being averaged molecular weight and being 2000 polycaprolactone (PCL2000) 25 grams and molecular-weight average and be 530 polycaprolactone (PCL530) 75 grams puts into reaction flask and continues down to stir at 100 ℃.Then, add 31.93 gram H
12MDI and 0.1 gram DBTDL.Low whipping speed 30rpm stirred 15 minutes down, made the product of gained place baking oven to dry by the fire 8 hours down at 90 ℃.Measure the weight-average molecular weight (Mw) of the aliphatic polyester copolymer of gained at last, polydispersity index (PDI), as shown in table 10.
Table 10
| Composition |
Mw |
PDI |
| PCL
2000∶PCL
530=75: 25 (by weight)
|
184,233 |
1.85 |
Embodiment 11
Being averaged molecular weight and being 2000 polycaprolactone (PCL2000) 50 grams and molecular-weight average and be 530 polycaprolactone (PCL530) 50 grams puts into reaction flask and continues down to stir at 100 ℃.Then, add 38.16 gram H
12MDI and 0.1 gram DBTDL.Low whipping speed 300rpm stirred 5 minutes down, made the product of gained place baking oven to dry by the fire 6 hours down at 80 ℃.Measure the weight-average molecular weight (Mw) of the aliphatic polyester copolymer of gained at last, polydispersity index (PDI), as shown in table 11.
Table 11
| Composition |
Mw |
PDI |
| PCL
2000∶PCL
530=50: 50 (by weight)
|
191,735 |
1.81 |
Embodiment 12
Being averaged molecular weight and being 2000 polycaprolactone (PCL2000) 75 grams and molecular-weight average and be 530 polycaprolactone (PCL530) 75 grams puts into reaction flask and continues down to stir at 100 ℃.Then, add 43.58 gram H
12MDI and 0.1 gram DBTDL.Low whipping speed 30rpm stirred 15 minutes down, made the product of gained place baking oven to dry by the fire 8 hours down at 90 ℃.Measure its weight-average molecular weight of aliphatic polyester copolymer (Mw) of gained at last, polydispersity index (PDI), as shown in table 12.
Table 12
| Composition |
Mw |
PDI |
| PCL
2000∶PCL
530=25: 75 (by weight)
|
36,715 |
2.12 |
Embodiment 13
The preparation of the L-lactic acid lactide glycol (A) of short chain
Get 100g L-lactic acid lactide and place round-bottomed bottle, and be heated to 110 ℃ of dissolvings fully.Then, add 2.87 gram ethylene glycol (EG) and 1.5 gram (0.5mol%) stannous octoates (as catalyzer), wherein the mol ratio of this L-lactic acid lactide and ethylene glycol is 15: 1.Be heated to 140 ℃, continuing after 6 hours, the product of gained is being dissolved in methylene dichloride, and in ether, precipitates.After the oven dry, obtain L-lactic acid lactide glycol (A).At last, measure its weight-average molecular weight (Mw), polydispersity index (PDI), as shown in table 13.
The preparation of the L-lactic acid lactide glycol (B) of long-chain
The L-lactic acid lactide (L-lactide) of getting 100g places round-bottomed bottle, and is heated to 110 ℃ of dissolvings fully.Then, add 2.15 gram ethylene glycol (EG) and 48 gram (0.5mol%) stannous octoates (as catalyzer), wherein the mol ratio of this L-lactic acid lactide and ethylene glycol is 20: 1.Be heated to 140 ℃, continuing after 6 hours, the product of gained is being dissolved in methylene dichloride, and in ether, precipitates.After the oven dry, obtain L-lactic acid lactide glycol (B).At last, measure its weight-average molecular weight (Mw), polydispersity index (PDI), as shown in table 13.
Table 13
| Product |
Composition |
Mw |
PDI |
| The L-lactic acid lactide glycol (A) of short chain |
Lactic acid lactide: EG=15: 1 (mol ratio) |
3,781 |
1.15 |
| The L-lactic acid lactide glycol (B) of long-chain |
Lactic acid lactide: EG=20: 1 (mol ratio) |
4,569 |
1.17 |
With L-lactic acid lactide is the preparation that repeats unitary aliphatic polyester copolymer
With 50 gram molecular weights be 3,781 L-lactic acid lactide glycol (A) (PLA3781) and 50 gram molecular weights be that 4,569 L-lactic acid lactide glycol (B) (PLA4,569) places reaction flask and 190 ℃ of stirrings.Then, add 6.63 gram H
12MDI and 1 gram DBTDL are in reaction flask.Low whipping speed 50rpm stirs after 5 minutes down, makes the product of gained place baking oven to dry by the fire 6 hours down at 120 ℃.Measure the weight-average molecular weight (Mw) of the aliphatic polyester copolymer of gained at last, polydispersity index (PDI), as shown in table 14.
Table 14
| Composition |
Mw |
PDI |
| PLA
3781∶PLA
4569=50: 50 (by weight)
|
26,585 |
2.35 |
Embodiment 14
Being averaged molecular weight and being 2000 polycaprolactone (PCL2000) 200 grams and molecular-weight average and be 10000 polycaprolactone (PCL10000) 200 grams puts into reaction flask and continues down to stir at 100 ℃.Then, add 47.16 gram H
12MDI and 0.6 gram DBTDL.Said mixture is carried out reacting and extruding polymerization.Polymerization is selected parallel dual-screw extruding machine for use with extruder, and the L/D value is 48.The setting reaction conditions is: (a) screw speed is made as 30rpm, and (b) extruder temperature is set between 150 ℃~180 ℃, and die head temperature is located at 60 ℃.(c) charging and to cover material speed be 50ml/min.At last, measure the weight-average molecular weight (Mw) of gained aliphatic polyester copolymer, polydispersity index (PDI), as shown in Table 15.
Table 15
| Composition |
Mw |
PDI |
| PCL
2000∶PCL
10000=50: 50 (by weight)
|
68,697 |
2.34 |
Embodiment 15
Aliphatic polyester copolymer and commodity PCL raw material (molecular-weight average 80000, by Aldrich Co., Ltd makes sale) to embodiment of the invention 1-6 gained carry out external accelerated degradation test, and testing method is as follows.
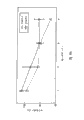
The aliphatic polyester copolymer and the commodity PCL raw material of embodiment of the invention 1-6 gained are made 20 * 20mm test piece that thickness is 0.2mm.Then, preparation phosphoric acid buffer (phosphate buffersaline), and adjust its pH=7.4.Then above-mentioned test piece is inserted respectively in the container of the phosphoric acid buffer with 15ml, it is inserted 50 ± 1 ℃ baking oven, and shake with the speed of 20rpm, and the molecular-weight average of sampling analysis material changes week about, its result please refer to Fig. 4.As seen from the figure, but the present invention can utilize the difference of first polyester and second molecular weight of polyesters and part by weight to regulate and control the resolving time.
Embodiment 16
The testing method of the vivo degradation test of aliphatic polyester copolymer of the present invention is as follows.Please refer to the rat pattern muscle implantation test shown in Fig. 5 a, the rat (8-9 week age) of kind for being provided by Sprague Dawley farm is provided.The aliphatic polyester copolymer of embodiment 2 and embodiment 5 gained is made 12 * 5mm test piece that thickness is 0.5mm.Rats with left 6 is the control group, is left intact; Rat right side 5 is an experimental group, the test piece of implanting embodiment 2 and embodiment 5 is in rat leg muscle layer (n=4), the animal sacrifice time is respectively 1,3,6 and 9 months, to implant material (test piece of embodiment 2 and embodiment 5) behind the sacrifice takes out, analyze the molecular weight of implanting material, its result is shown in Fig. 5 b.
Embodiment 17
Aliphatic polyester copolymer and commodity PCL raw material (molecular-weight average 80000, by Aldrich Co., Ltd makes sale) to embodiment of the invention 1-6 gained carry out the material mechanical property detection, and method is as follows.Take the aliphatic polyester copolymer and the commodity PCL raw material of embodiment 1-6 gained, in 25 ± 1 ℃, left standstill 48 hours under the 50% relative humidity environment before the test.Then, use universal testing machine (Universal Material Testing Machine), model: Instron 4467 tests.According to ASTM D638 standard specifications, sample is cut into dumb-bell shape, and use 500N Load Cell, with draw speed: 150mm/min tests, and the result is shown in Fig. 6 and 7.
Be used for the preparation of the neural medical implant apparatus of repairing
Embodiment 18
Getting aliphatic polyester copolymer 20 gram of embodiment 5 gained and 20 gram molecular weights and be 300 polyoxyethylene glycol (PEG300) is dissolved among the 60 gram DMAc, to form homogeneous polymer solution.Then, this polymers soln is poured in the cylindric spreader, this cylindric spreader has the center hole that diameter is 2.2m.Then, with external diameter be the pole of 1.5mm by this tubular spreader, make pole surface coated one layer thickness be about the polymers soln of 0.35mm.
Then, the pole of this surface coated polymers soln is placed solidification liquid (at this, this solidification liquid is a water), in this solidification liquid, polymers soln solidifies gradually and forms porousness aliphatic polyester copolymer tubes of material.Then, porousness aliphatic polyester copolymer tubes of material is detached pole, this hollow porousness aliphatic polyester copolymer tubes of material is being placed aqueous ethanolic solution clean (ethanol: water=1: 1), clean with clear water more at last, obtain hollow porousness aliphatic polyester copolymer tubes of material.Then, observe its structure, the mutual commissure of hole that it is inside and outside with SEM.And, therefore can be used as the medical implant apparatus that is used for neural reparation because the aliphatic polyester copolymer of gained of the present invention has Bioabsorbable.
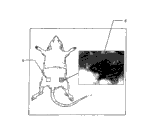
Please refer to Fig. 8 a and 8b, adult rat 10 (200~250 gram) is provided by Sprague Dawley farm, by the narcotic of abdominal injection 400mg/kg.Shave after the anesthesia except that its thigh part chaeta, and with excellent iodine sterilization skin, draw skin and the muscle layer of opening thigh middle part 8b with knife blade, and to start fixedly wound of device, expose sciatic nerve 12, block eight millimeters sciatic nerve 12 with shearing, block the back and cause 10 millimeters interval because of the nerve retraction, 10 millimeters above-mentioned hollow porousness aliphatic polyester copolymer tubes of material 20 is placed 16 of neural residul ends, and two neural residul ends 16 are sewed up two ends of this hollow porousness aliphatic polyester copolymer tubes of material with the 9-0 suture.After implant surgery is finished, remove this and start device, and careful suture muscles and skin, and return in rearging cage.
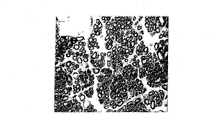
Perform the operation after back one month (n=3) and three months (n=4), experimental animal with 4% trioxymethylene perfusion, then, is taken out hollow porousness aliphatic polyester copolymer tubes of material and nerve in the lump, and placing 5% glutaraldehyde fixing, the fixed tissue is with resin-coated.Then, tissue slice is shown with Toluidine blue staining and with optics levy sem observation neurotization situation.
This regenerated nervous tissue is arranged in hollow aliphatic polyester copolymer conduit 20 places, can be by 20 tangent plane in this hollow aliphatic polyester copolymer conduit be observed the neurotization situation, please refer to Fig. 9 a and 9b, it shows respectively along A-A ' tangent line (performing the operation back month) and along the section tissue slice of B-B ' tangent line (performing the operation back three months).A-A ' tangent line is at the 3mm place of 20 1 ends in this hollow aliphatic polyester copolymer conduit, and as seen from the figure, the sciatic nerve of mouse has really to be repaired and regeneration.
Be used for the preparation of the medical implant apparatus of endocranium reparation
Embodiment 19
Get aliphatic polyester copolymer 20 gram of embodiment 5 gained, and 20 gram molecular weights are that 300 polyoxyethylene glycol (PEG300) adds in the 60 gram DMAc organic solvents, formation solution stirs.Then with solution coat in tabular die surface with convex-concave surface, the thickness of coating is about 2mm.Then the tabular mould (having convex-concave surface) of surface coverage polyester copolymer solution is inserted in 25 ℃ ± 2 ℃ the solidification liquid (solidification liquid consists of solution), form porousness aliphatic polyester copolymer thin-film material with multilayered structure to be frozen into.Then the porousness aliphatic polyester copolymer film that forms is inserted in the scavenging solution that contains 40wt% acetone soaking and washing 2 hours, and re-used clean water logging bubble at last and cleaned 6 hours, obtain porousness aliphatic polyester copolymer thin-film material after the drying.
This porousness aliphatic polyester copolymer thin-film material is made into is of a size of 15 * 12mm endocranium substituent.The New Zealand white rabbit usefulness that (2-3kg) tests this endocranium substituent is provided.At first, with rabbit impose vetatar (Ketamine hydrochloride) (50mg/Kg) and xylazine (Xylazine) (2%, 1ml/Kg) injection.Then, it is imposed top frontal lobe craniotomy, please refer to Figure 10 a, prescind endocranium (size 12 * 10mm of this rabbit 100
2).Please refer to Figure 10 b, it shows Figure 10 a rabbit forehead 10b enlarged view partly, and in experimental group, the endocranium tissue 104 (using the No.7-0 suture line) that this orthogonal endocranium substituent 102 is sutured in the breach periphery is located.In control group, the endocranium indentation, there is directly used the 7-0 suture, does not use to appoint and closes the endocranium substituent.At last, use N0.4-0 suture skull film and skin.The experimental animal of experimental group and control group was sacrificed in the time of 6,3,1 months respectively.
Porousness aliphatic polyester endocranium substituent shows via rabbit mode performance assessment result, this porousness aliphatic polyester endocranium substituent is implanted to there is no except a little foreign body reaction after 1 month and is observed other untoward reaction, therefore judges that it has good bio-compatibility.
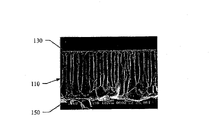
Please refer to Figure 11 a, it shows embodiment 19 employed porousness aliphatic polyester endocranium substituents, and please refer to Figure 11 b, and it shows the SEM spectrogram of porousness aliphatic polyester endocranium substituent 102.Shown in Figure 11 b~11d, endocranium substituent 102 has cellular structure 110, and has smooth-flat-surface 130 (shown in Figure 11 c), and a uneven surface 150 (shown in Figure 11 d).In addition, please refer to Figure 12 a and 12b, it shows the SEM spectrogram of smooth-flat-surface 130 and uneven surface 150 respectively, and Figure 13 is presented at endocranium substituent 102 tissue slice (performing the operation after month) on every side.Can be observed by above-mentioned diagram, after implanting 1 month, observe newborn tissue in the pore space structure of the upper strata (slightly making face) of discovery endocranium substituent 102 and middle layer (cellular structure layer) and produce, in addition between the smooth-flat-surface 130 of this endocranium substituent 102 and uneven surface 150 and host's flexible back meninges (Pia mater) 203, be coated with monolayer cell 201.And be not as assembling many immunocytes between implant and host's the contact surface.This represents the artificial meninx of mPCL after implanting 1 month, and the host has begun to carry out the action of tissue repair.
In addition, find main tissue 207 and capillary blood vessel 209 in endocranium substituent 102, on behalf of host's endocranium, this repairing and regeneration.Moreover the smooth-flat-surface 130 of endocranium substitution device 102 can avoid cerebral tissue to be stained with sticking and uneven surface 150 can promote cerebral tissue propagation.Please refer to Figure 13, be stained with sticking phenomenon without any tissue as seen from the figure and be observed.
It should be noted that, implant rabbit skull position after 1 month at endocranium substitution device 102, observing artificial meninx substituent surface morphology by SEM finds, be covered with the preceding slight crack of not finding of many implantation in the endocranium substitution device 102 integral surface structures, this is the phenomenon of material production degraded, analyze the difference of its degraded front and back molecular weight, aliphatic polyester copolymer is after rabbit pattern skull position is implanted 1 month, the molecular weight and molecular weight amplitude is 3.9%, implant after 3 months, the molecular weight and molecular weight amplitude is 8.2%, implants after 6 months, and the molecular weight and molecular weight amplitude is 23.3% (weight-average molecular weight, Mw), so show that this endocranium substitution device 102 can degrade in animal body really voluntarily, and increase the phenomenon that increase is arranged with the implantation time, this just had been found that 1 middle of the month.
One of major technique feature of the present invention is that aliphatic polyester copolymer of the present invention has identical repeating unit by at least two kinds but the aliphatic polyester institute copolymerization of different molecular weight forms.But therefore the biology resolving time of this aliphatic polyester copolymer can utilize molecular weight differences between different polyester, weight ratio, mol ratio, and the selection of coupler adjust.Moreover above-mentioned variable has also changed the crystallization property of this aliphatic polyester copolymer simultaneously, but has therefore increased its variation on the biology capacity of decomposition.
Though the present invention discloses as above with preferred embodiment, so it is not in order to limit the present invention.Any the technical staff in the technical field of the invention, without departing from the spirit and scope of the present invention, Ying Kezuo changes arbitrarily and retouching.Therefore, protection scope of the present invention should be as the criterion with appended claims institute restricted portion.