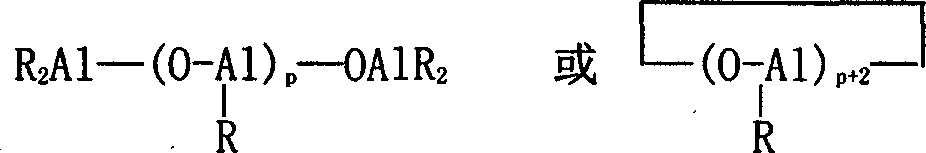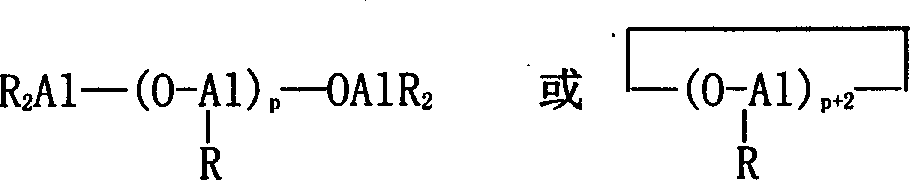CN1069100C - Composite solid catalyst and its preparation method and use - Google Patents
Composite solid catalyst and its preparation method and use Download PDFInfo
- Publication number
- CN1069100C CN1069100C CN95106710A CN95106710A CN1069100C CN 1069100 C CN1069100 C CN 1069100C CN 95106710 A CN95106710 A CN 95106710A CN 95106710 A CN95106710 A CN 95106710A CN 1069100 C CN1069100 C CN 1069100C
- Authority
- CN
- China
- Prior art keywords
- compound
- composition
- magnesium
- catalyzer
- titanium
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000011949 solid catalyst Substances 0.000 title claims abstract description 38
- 238000002360 preparation method Methods 0.000 title claims abstract description 12
- 239000002131 composite material Substances 0.000 title abstract description 5
- 239000003054 catalyst Substances 0.000 claims abstract description 40
- 238000006116 polymerization reaction Methods 0.000 claims abstract description 31
- 150000001336 alkenes Chemical class 0.000 claims abstract description 23
- 239000000654 additive Substances 0.000 claims abstract description 19
- 150000001875 compounds Chemical class 0.000 claims description 88
- -1 magnesium halide Chemical class 0.000 claims description 67
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 64
- 239000011777 magnesium Substances 0.000 claims description 52
- 229910052749 magnesium Inorganic materials 0.000 claims description 39
- 239000000203 mixture Substances 0.000 claims description 34
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical group CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 claims description 33
- 239000007787 solid Substances 0.000 claims description 33
- 238000000034 method Methods 0.000 claims description 29
- 238000013329 compounding Methods 0.000 claims description 23
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 20
- 229910052799 carbon Inorganic materials 0.000 claims description 19
- 230000000996 additive effect Effects 0.000 claims description 17
- 150000001721 carbon Chemical group 0.000 claims description 17
- 125000000217 alkyl group Chemical group 0.000 claims description 16
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 13
- 229910052782 aluminium Inorganic materials 0.000 claims description 13
- 229940058344 antitrematodals organophosphorous compound Drugs 0.000 claims description 13
- 150000002903 organophosphorus compounds Chemical class 0.000 claims description 13
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 claims description 12
- 125000000538 pentafluorophenyl group Chemical group FC1=C(F)C(F)=C(*)C(F)=C1F 0.000 claims description 12
- 239000003701 inert diluent Substances 0.000 claims description 11
- 238000001556 precipitation Methods 0.000 claims description 11
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical group C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 10
- 229910052751 metal Inorganic materials 0.000 claims description 10
- 235000011007 phosphoric acid Nutrition 0.000 claims description 10
- 239000010936 titanium Substances 0.000 claims description 10
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims description 9
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims description 9
- 238000005660 chlorination reaction Methods 0.000 claims description 9
- 239000001257 hydrogen Substances 0.000 claims description 9
- 229910052739 hydrogen Inorganic materials 0.000 claims description 9
- 230000008569 process Effects 0.000 claims description 9
- 239000004593 Epoxy Substances 0.000 claims description 8
- 229910052736 halogen Inorganic materials 0.000 claims description 8
- 150000002367 halogens Chemical class 0.000 claims description 8
- 239000002184 metal Substances 0.000 claims description 8
- CYIDZMCFTVVTJO-UHFFFAOYSA-N pyromellitic acid Chemical compound OC(=O)C1=CC(C(O)=O)=C(C(O)=O)C=C1C(O)=O CYIDZMCFTVVTJO-UHFFFAOYSA-N 0.000 claims description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 8
- 125000005234 alkyl aluminium group Chemical group 0.000 claims description 7
- 229910052796 boron Inorganic materials 0.000 claims description 7
- 125000005843 halogen group Chemical group 0.000 claims description 7
- 239000002904 solvent Substances 0.000 claims description 7
- XJDNKRIXUMDJCW-UHFFFAOYSA-J titanium tetrachloride Chemical compound Cl[Ti](Cl)(Cl)Cl XJDNKRIXUMDJCW-UHFFFAOYSA-J 0.000 claims description 7
- LRWZZZWJMFNZIK-UHFFFAOYSA-N 2-chloro-3-methyloxirane Chemical compound CC1OC1Cl LRWZZZWJMFNZIK-UHFFFAOYSA-N 0.000 claims description 6
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 claims description 6
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 claims description 6
- 125000003545 alkoxy group Chemical group 0.000 claims description 6
- 125000003118 aryl group Chemical group 0.000 claims description 6
- UJMDYLWCYJJYMO-UHFFFAOYSA-N benzene-1,2,3-tricarboxylic acid Chemical compound OC(=O)C1=CC=CC(C(O)=O)=C1C(O)=O UJMDYLWCYJJYMO-UHFFFAOYSA-N 0.000 claims description 6
- 235000011147 magnesium chloride Nutrition 0.000 claims description 6
- STCOOQWBFONSKY-UHFFFAOYSA-N tributyl phosphate Chemical compound CCCCOP(=O)(OCCCC)OCCCC STCOOQWBFONSKY-UHFFFAOYSA-N 0.000 claims description 6
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims description 5
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 5
- 229910052801 chlorine Inorganic materials 0.000 claims description 5
- 239000000460 chlorine Substances 0.000 claims description 5
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 5
- 229910052735 hafnium Inorganic materials 0.000 claims description 5
- 239000012456 homogeneous solution Substances 0.000 claims description 5
- 150000002431 hydrogen Chemical class 0.000 claims description 5
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 claims description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 5
- 229910052719 titanium Inorganic materials 0.000 claims description 5
- MCULRUJILOGHCJ-UHFFFAOYSA-N triisobutylaluminium Chemical compound CC(C)C[Al](CC(C)C)CC(C)C MCULRUJILOGHCJ-UHFFFAOYSA-N 0.000 claims description 5
- 229910052726 zirconium Inorganic materials 0.000 claims description 5
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 claims description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 claims description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 4
- LGRFSURHDFAFJT-UHFFFAOYSA-N Phthalic anhydride Natural products C1=CC=C2C(=O)OC(=O)C2=C1 LGRFSURHDFAFJT-UHFFFAOYSA-N 0.000 claims description 4
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 claims description 4
- 150000001350 alkyl halides Chemical class 0.000 claims description 4
- JHIWVOJDXOSYLW-UHFFFAOYSA-N butyl 2,2-difluorocyclopropane-1-carboxylate Chemical compound CCCCOC(=O)C1CC1(F)F JHIWVOJDXOSYLW-UHFFFAOYSA-N 0.000 claims description 4
- XSIFPSYPOVKYCO-UHFFFAOYSA-N butyl benzoate Chemical compound CCCCOC(=O)C1=CC=CC=C1 XSIFPSYPOVKYCO-UHFFFAOYSA-N 0.000 claims description 4
- NMJJFJNHVMGPGM-UHFFFAOYSA-N butyl formate Chemical compound CCCCOC=O NMJJFJNHVMGPGM-UHFFFAOYSA-N 0.000 claims description 4
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 claims description 4
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 4
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical class C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 claims description 4
- MGWAVDBGNNKXQV-UHFFFAOYSA-N diisobutyl phthalate Chemical compound CC(C)COC(=O)C1=CC=CC=C1C(=O)OCC(C)C MGWAVDBGNNKXQV-UHFFFAOYSA-N 0.000 claims description 4
- MTZQAGJQAFMTAQ-UHFFFAOYSA-N ethyl benzoate Chemical compound CCOC(=O)C1=CC=CC=C1 MTZQAGJQAFMTAQ-UHFFFAOYSA-N 0.000 claims description 4
- UAIZDWNSWGTKFZ-UHFFFAOYSA-L ethylaluminum(2+);dichloride Chemical compound CC[Al](Cl)Cl UAIZDWNSWGTKFZ-UHFFFAOYSA-L 0.000 claims description 4
- VBJZVLUMGGDVMO-UHFFFAOYSA-N hafnium atom Chemical compound [Hf] VBJZVLUMGGDVMO-UHFFFAOYSA-N 0.000 claims description 4
- 239000007788 liquid Substances 0.000 claims description 4
- KYTZHLUVELPASH-UHFFFAOYSA-N naphthalene-1,2-dicarboxylic acid Chemical class C1=CC=CC2=C(C(O)=O)C(C(=O)O)=CC=C21 KYTZHLUVELPASH-UHFFFAOYSA-N 0.000 claims description 4
- 150000007524 organic acids Chemical group 0.000 claims description 4
- 238000006884 silylation reaction Methods 0.000 claims description 4
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 4
- VOITXYVAKOUIBA-UHFFFAOYSA-N triethylaluminium Chemical compound CC[Al](CC)CC VOITXYVAKOUIBA-UHFFFAOYSA-N 0.000 claims description 4
- HVLLSGMXQDNUAL-UHFFFAOYSA-N triphenyl phosphite Chemical compound C=1C=CC=CC=1OP(OC=1C=CC=CC=1)OC1=CC=CC=C1 HVLLSGMXQDNUAL-UHFFFAOYSA-N 0.000 claims description 4
- 238000005406 washing Methods 0.000 claims description 4
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 claims description 3
- 102000040350 B family Human genes 0.000 claims description 3
- 108091072128 B family Proteins 0.000 claims description 3
- WOBVZKNOKPJLJU-UHFFFAOYSA-N CNC.[B+3] Chemical compound CNC.[B+3] WOBVZKNOKPJLJU-UHFFFAOYSA-N 0.000 claims description 3
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims description 3
- PPTSBERGOGHCHC-UHFFFAOYSA-N boron lithium Chemical compound [Li].[B] PPTSBERGOGHCHC-UHFFFAOYSA-N 0.000 claims description 3
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 claims description 3
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 claims description 3
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 3
- 238000002156 mixing Methods 0.000 claims description 3
- DIOQZVSQGTUSAI-UHFFFAOYSA-N n-butylhexane Natural products CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 claims description 3
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 claims description 3
- 229910052706 scandium Inorganic materials 0.000 claims description 3
- SIXSYDAISGFNSX-UHFFFAOYSA-N scandium atom Chemical compound [Sc] SIXSYDAISGFNSX-UHFFFAOYSA-N 0.000 claims description 3
- 229910052723 transition metal Inorganic materials 0.000 claims description 3
- 150000003624 transition metals Chemical class 0.000 claims description 3
- 239000008096 xylene Substances 0.000 claims description 3
- AUEJBKCWXPYRGZ-UHFFFAOYSA-N 1,1'-biphenyl diethoxysilane Chemical compound C(C)O[SiH2]OCC.C1(=CC=CC=C1)C1=CC=CC=C1 AUEJBKCWXPYRGZ-UHFFFAOYSA-N 0.000 claims description 2
- DURPTKYDGMDSBL-UHFFFAOYSA-N 1-butoxybutane Chemical compound CCCCOCCCC DURPTKYDGMDSBL-UHFFFAOYSA-N 0.000 claims description 2
- AOPDRZXCEAKHHW-UHFFFAOYSA-N 1-pentoxypentane Chemical compound CCCCCOCCCCC AOPDRZXCEAKHHW-UHFFFAOYSA-N 0.000 claims description 2
- HFZLSTDPRQSZCQ-UHFFFAOYSA-N 1-pyrrolidin-3-ylpyrrolidine Chemical compound C1CCCN1C1CNCC1 HFZLSTDPRQSZCQ-UHFFFAOYSA-N 0.000 claims description 2
- LTMRRSWNXVJMBA-UHFFFAOYSA-L 2,2-diethylpropanedioate Chemical compound CCC(CC)(C([O-])=O)C([O-])=O LTMRRSWNXVJMBA-UHFFFAOYSA-L 0.000 claims description 2
- LKMJVFRMDSNFRT-UHFFFAOYSA-N 2-(methoxymethyl)oxirane Chemical compound COCC1CO1 LKMJVFRMDSNFRT-UHFFFAOYSA-N 0.000 claims description 2
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 claims description 2
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 claims description 2
- IHVVZSPWQJWRDV-UHFFFAOYSA-N Butyl ethyl malonate Chemical compound CCCCOC(=O)CC(=O)OCC IHVVZSPWQJWRDV-UHFFFAOYSA-N 0.000 claims description 2
- CADAOKXOWMHTKV-UHFFFAOYSA-N C(C)O[Ti](OCC)OCC.[Cl] Chemical compound C(C)O[Ti](OCC)OCC.[Cl] CADAOKXOWMHTKV-UHFFFAOYSA-N 0.000 claims description 2
- FXTVTRUDVQINIM-UHFFFAOYSA-N C=1C=CC=CC=1C[Ti](C1C=CC=C1)CC1=CC=CC=C1 Chemical compound C=1C=CC=CC=1C[Ti](C1C=CC=C1)CC1=CC=CC=C1 FXTVTRUDVQINIM-UHFFFAOYSA-N 0.000 claims description 2
- MVHZHMKEBJJTCH-UHFFFAOYSA-N CN(C)C.CO[SiH3] Chemical compound CN(C)C.CO[SiH3] MVHZHMKEBJJTCH-UHFFFAOYSA-N 0.000 claims description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 claims description 2
- KKDBZWZRJNRBGA-UHFFFAOYSA-L Cl[Ti]Cl.[CH]1C=CC=C1 Chemical compound Cl[Ti]Cl.[CH]1C=CC=C1 KKDBZWZRJNRBGA-UHFFFAOYSA-L 0.000 claims description 2
- IEPRKVQEAMIZSS-UHFFFAOYSA-N Di-Et ester-Fumaric acid Natural products CCOC(=O)C=CC(=O)OCC IEPRKVQEAMIZSS-UHFFFAOYSA-N 0.000 claims description 2
- ONKUXPIBXRRIDU-UHFFFAOYSA-N Diethyl decanedioate Chemical compound CCOC(=O)CCCCCCCCC(=O)OCC ONKUXPIBXRRIDU-UHFFFAOYSA-N 0.000 claims description 2
- IEPRKVQEAMIZSS-WAYWQWQTSA-N Diethyl maleate Chemical compound CCOC(=O)\C=C/C(=O)OCC IEPRKVQEAMIZSS-WAYWQWQTSA-N 0.000 claims description 2
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 claims description 2
- GORXRFCHPRVABO-UHFFFAOYSA-N O(C)[SiH](OC)OC.C=C Chemical compound O(C)[SiH](OC)OC.C=C GORXRFCHPRVABO-UHFFFAOYSA-N 0.000 claims description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 claims description 2
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 claims description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 2
- 229910021552 Vanadium(IV) chloride Inorganic materials 0.000 claims description 2
- ILTMWECZMURSQF-UHFFFAOYSA-I [V+5].[Br-].[Br-].[Br-].[Br-].[Br-] Chemical compound [V+5].[Br-].[Br-].[Br-].[Br-].[Br-] ILTMWECZMURSQF-UHFFFAOYSA-I 0.000 claims description 2
- 150000007933 aliphatic carboxylic acids Chemical class 0.000 claims description 2
- 239000004411 aluminium Substances 0.000 claims description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical class OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 claims description 2
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 claims description 2
- 239000012965 benzophenone Substances 0.000 claims description 2
- HQMRIBYCTLBDAK-UHFFFAOYSA-M bis(2-methylpropyl)alumanylium;chloride Chemical compound CC(C)C[Al](Cl)CC(C)C HQMRIBYCTLBDAK-UHFFFAOYSA-M 0.000 claims description 2
- SIPUZPBQZHNSDW-UHFFFAOYSA-N bis(2-methylpropyl)aluminum Chemical compound CC(C)C[Al]CC(C)C SIPUZPBQZHNSDW-UHFFFAOYSA-N 0.000 claims description 2
- YHWCPXVTRSHPNY-UHFFFAOYSA-N butan-1-olate;titanium(4+) Chemical compound [Ti+4].CCCC[O-].CCCC[O-].CCCC[O-].CCCC[O-] YHWCPXVTRSHPNY-UHFFFAOYSA-N 0.000 claims description 2
- 229940043232 butyl acetate Drugs 0.000 claims description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 2
- YCURFOQQPNHZAO-UHFFFAOYSA-N butyl naphthalene-1-carboxylate Chemical compound C1=CC=C2C(C(=O)OCCCC)=CC=CC2=C1 YCURFOQQPNHZAO-UHFFFAOYSA-N 0.000 claims description 2
- DOIRQSBPFJWKBE-UHFFFAOYSA-N dibutyl phthalate Chemical compound CCCCOC(=O)C1=CC=CC=C1C(=O)OCCCC DOIRQSBPFJWKBE-UHFFFAOYSA-N 0.000 claims description 2
- CQYBWJYIKCZXCN-UHFFFAOYSA-N diethylaluminum Chemical compound CC[Al]CC CQYBWJYIKCZXCN-UHFFFAOYSA-N 0.000 claims description 2
- AHUXYBVKTIBBJW-UHFFFAOYSA-N dimethoxy(diphenyl)silane Chemical compound C=1C=CC=CC=1[Si](OC)(OC)C1=CC=CC=C1 AHUXYBVKTIBBJW-UHFFFAOYSA-N 0.000 claims description 2
- YYLGKUPAFFKGRQ-UHFFFAOYSA-N dimethyldiethoxysilane Chemical compound CCO[Si](C)(C)OCC YYLGKUPAFFKGRQ-UHFFFAOYSA-N 0.000 claims description 2
- POLCUAVZOMRGSN-UHFFFAOYSA-N dipropyl ether Chemical compound CCCOCCC POLCUAVZOMRGSN-UHFFFAOYSA-N 0.000 claims description 2
- 150000002148 esters Chemical class 0.000 claims description 2
- UHSDHNXHBQDMMH-UHFFFAOYSA-L ethanolate;titanium(4+);dichloride Chemical compound CCO[Ti](Cl)(Cl)OCC UHSDHNXHBQDMMH-UHFFFAOYSA-L 0.000 claims description 2
- RSIHJDGMBDPTIM-UHFFFAOYSA-N ethoxy(trimethyl)silane Chemical compound CCO[Si](C)(C)C RSIHJDGMBDPTIM-UHFFFAOYSA-N 0.000 claims description 2
- ANSXAPJVJOKRDJ-UHFFFAOYSA-N furo[3,4-f][2]benzofuran-1,3,5,7-tetrone Chemical compound C1=C2C(=O)OC(=O)C2=CC2=C1C(=O)OC2=O ANSXAPJVJOKRDJ-UHFFFAOYSA-N 0.000 claims description 2
- 239000007789 gas Substances 0.000 claims description 2
- PDPJQWYGJJBYLF-UHFFFAOYSA-J hafnium tetrachloride Chemical compound Cl[Hf](Cl)(Cl)Cl PDPJQWYGJJBYLF-UHFFFAOYSA-J 0.000 claims description 2
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 claims description 2
- 125000001183 hydrocarbyl group Chemical group 0.000 claims description 2
- 238000005984 hydrogenation reaction Methods 0.000 claims description 2
- 150000002680 magnesium Chemical class 0.000 claims description 2
- BLQJIBCZHWBKSL-UHFFFAOYSA-L magnesium iodide Chemical class [Mg+2].[I-].[I-] BLQJIBCZHWBKSL-UHFFFAOYSA-L 0.000 claims description 2
- 239000011976 maleic acid Substances 0.000 claims description 2
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 claims description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Natural products C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 claims description 2
- RPESBQCJGHJMTK-UHFFFAOYSA-I pentachlorovanadium Chemical class [Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[V+5] RPESBQCJGHJMTK-UHFFFAOYSA-I 0.000 claims description 2
- ISIJQEHRDSCQIU-UHFFFAOYSA-N tert-butyl 2,7-diazaspiro[4.5]decane-7-carboxylate Chemical compound C1N(C(=O)OC(C)(C)C)CCCC11CNCC1 ISIJQEHRDSCQIU-UHFFFAOYSA-N 0.000 claims description 2
- JMXKSZRRTHPKDL-UHFFFAOYSA-N titanium ethoxide Chemical compound [Ti+4].CC[O-].CC[O-].CC[O-].CC[O-] JMXKSZRRTHPKDL-UHFFFAOYSA-N 0.000 claims description 2
- UBZYKBZMAMTNKW-UHFFFAOYSA-J titanium tetrabromide Chemical compound Br[Ti](Br)(Br)Br UBZYKBZMAMTNKW-UHFFFAOYSA-J 0.000 claims description 2
- NLLZTRMHNHVXJJ-UHFFFAOYSA-J titanium tetraiodide Chemical compound I[Ti](I)(I)I NLLZTRMHNHVXJJ-UHFFFAOYSA-J 0.000 claims description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 2
- RJIFVNWOLLIBJV-UHFFFAOYSA-N tributyl benzene-1,2,4-tricarboxylate Chemical compound CCCCOC(=O)C1=CC=C(C(=O)OCCCC)C(C(=O)OCCCC)=C1 RJIFVNWOLLIBJV-UHFFFAOYSA-N 0.000 claims description 2
- JCVQKRGIASEUKR-UHFFFAOYSA-N triethoxy(phenyl)silane Chemical compound CCO[Si](OCC)(OCC)C1=CC=CC=C1 JCVQKRGIASEUKR-UHFFFAOYSA-N 0.000 claims description 2
- BDZBKCUKTQZUTL-UHFFFAOYSA-N triethyl phosphite Chemical compound CCOP(OCC)OCC BDZBKCUKTQZUTL-UHFFFAOYSA-N 0.000 claims description 2
- 125000005591 trimellitate group Chemical group 0.000 claims description 2
- CYTQBVOFDCPGCX-UHFFFAOYSA-N trimethyl phosphite Chemical compound COP(OC)OC CYTQBVOFDCPGCX-UHFFFAOYSA-N 0.000 claims description 2
- OJAJJFGMKAZGRZ-UHFFFAOYSA-N trimethyl(phenoxy)silane Chemical compound C[Si](C)(C)OC1=CC=CC=C1 OJAJJFGMKAZGRZ-UHFFFAOYSA-N 0.000 claims description 2
- JLTRXTDYQLMHGR-UHFFFAOYSA-N trimethylaluminium Chemical compound C[Al](C)C JLTRXTDYQLMHGR-UHFFFAOYSA-N 0.000 claims description 2
- LFXVBWRMVZPLFK-UHFFFAOYSA-N trioctylalumane Chemical compound CCCCCCCC[Al](CCCCCCCC)CCCCCCCC LFXVBWRMVZPLFK-UHFFFAOYSA-N 0.000 claims description 2
- PTVDYMGQGCNETM-UHFFFAOYSA-N trityl 2-methylprop-2-enoate Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(OC(=O)C(=C)C)C1=CC=CC=C1 PTVDYMGQGCNETM-UHFFFAOYSA-N 0.000 claims description 2
- JBIQAPKSNFTACH-UHFFFAOYSA-K vanadium oxytrichloride Chemical compound Cl[V](Cl)(Cl)=O JBIQAPKSNFTACH-UHFFFAOYSA-K 0.000 claims description 2
- JTJFQBNJBPPZRI-UHFFFAOYSA-J vanadium tetrachloride Chemical compound Cl[V](Cl)(Cl)Cl JTJFQBNJBPPZRI-UHFFFAOYSA-J 0.000 claims description 2
- DUNKXUFBGCUVQW-UHFFFAOYSA-J zirconium tetrachloride Chemical compound Cl[Zr](Cl)(Cl)Cl DUNKXUFBGCUVQW-UHFFFAOYSA-J 0.000 claims description 2
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims 3
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 claims 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 claims 2
- 150000002118 epoxides Chemical class 0.000 claims 2
- JHPBZFOKBAGZBL-UHFFFAOYSA-N (3-hydroxy-2,2,4-trimethylpentyl) 2-methylprop-2-enoate Chemical compound CC(C)C(O)C(C)(C)COC(=O)C(C)=C JHPBZFOKBAGZBL-UHFFFAOYSA-N 0.000 claims 1
- UFERIGCCDYCZLN-UHFFFAOYSA-N 3a,4,7,7a-tetrahydro-1h-indene Chemical compound C1C=CCC2CC=CC21 UFERIGCCDYCZLN-UHFFFAOYSA-N 0.000 claims 1
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 claims 1
- XJONFIGVOQMBIP-UHFFFAOYSA-L Cl[Zr](Cl)C1C=CC=C1 Chemical compound Cl[Zr](Cl)C1C=CC=C1 XJONFIGVOQMBIP-UHFFFAOYSA-L 0.000 claims 1
- XRDCOVBFSVCOJU-UHFFFAOYSA-N [B].CC1=CC=CC(N)=C1C Chemical compound [B].CC1=CC=CC(N)=C1C XRDCOVBFSVCOJU-UHFFFAOYSA-N 0.000 claims 1
- 239000002253 acid Substances 0.000 claims 1
- 150000001335 aliphatic alkanes Chemical class 0.000 claims 1
- 235000010290 biphenyl Nutrition 0.000 claims 1
- 239000004305 biphenyl Substances 0.000 claims 1
- DIOQZVSQGTUSAI-NJFSPNSNSA-N decane Chemical compound CCCCCCCCC[14CH3] DIOQZVSQGTUSAI-NJFSPNSNSA-N 0.000 claims 1
- 125000003963 dichloro group Chemical group Cl* 0.000 claims 1
- 235000019439 ethyl acetate Nutrition 0.000 claims 1
- OBNCKNCVKJNDBV-UHFFFAOYSA-N ethyl butyrate Chemical compound CCCC(=O)OCC OBNCKNCVKJNDBV-UHFFFAOYSA-N 0.000 claims 1
- FKRCODPIKNYEAC-UHFFFAOYSA-N ethyl propionate Chemical compound CCOC(=O)CC FKRCODPIKNYEAC-UHFFFAOYSA-N 0.000 claims 1
- 125000001188 haloalkyl group Chemical group 0.000 claims 1
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims 1
- APPHYFNIXVIIJR-UHFFFAOYSA-K scandium bromide Chemical compound Br[Sc](Br)Br APPHYFNIXVIIJR-UHFFFAOYSA-K 0.000 claims 1
- 230000003197 catalytic effect Effects 0.000 abstract description 3
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 abstract 1
- 230000001376 precipitating effect Effects 0.000 description 16
- 238000009826 distribution Methods 0.000 description 13
- 239000000243 solution Substances 0.000 description 12
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 10
- 230000015572 biosynthetic process Effects 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 9
- 238000003756 stirring Methods 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- 238000003786 synthesis reaction Methods 0.000 description 7
- 239000004698 Polyethylene Substances 0.000 description 5
- 238000006555 catalytic reaction Methods 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- 229920000573 polyethylene Polymers 0.000 description 5
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 5
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 5
- QMBQEXOLIRBNPN-UHFFFAOYSA-L zirconocene dichloride Chemical compound [Cl-].[Cl-].[Zr+4].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 QMBQEXOLIRBNPN-UHFFFAOYSA-L 0.000 description 5
- 239000004743 Polypropylene Substances 0.000 description 4
- 238000001816 cooling Methods 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 229910001629 magnesium chloride Inorganic materials 0.000 description 4
- 239000011259 mixed solution Substances 0.000 description 4
- 239000002245 particle Substances 0.000 description 4
- 229920001155 polypropylene Polymers 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- 239000004711 α-olefin Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 3
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 3
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 3
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- 238000012674 dispersion polymerization Methods 0.000 description 3
- 239000000413 hydrolysate Substances 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 239000012495 reaction gas Substances 0.000 description 3
- 229910000077 silane Inorganic materials 0.000 description 3
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 2
- KWKAKUADMBZCLK-UHFFFAOYSA-N 1-octene Chemical compound CCCCCCC=C KWKAKUADMBZCLK-UHFFFAOYSA-N 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 125000001118 alkylidene group Chemical group 0.000 description 2
- 238000012661 block copolymerization Methods 0.000 description 2
- 150000001639 boron compounds Chemical class 0.000 description 2
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 238000007334 copolymerization reaction Methods 0.000 description 2
- 230000006837 decompression Effects 0.000 description 2
- 229960000935 dehydrated alcohol Drugs 0.000 description 2
- 150000001993 dienes Chemical class 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 239000012442 inert solvent Substances 0.000 description 2
- 238000001746 injection moulding Methods 0.000 description 2
- 150000002899 organoaluminium compounds Chemical class 0.000 description 2
- 229920000098 polyolefin Polymers 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 239000012429 reaction media Substances 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- 229910052720 vanadium Inorganic materials 0.000 description 2
- 238000010792 warming Methods 0.000 description 2
- KMOUUZVZFBCRAM-OLQVQODUSA-N (3as,7ar)-3a,4,7,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound C1C=CC[C@@H]2C(=O)OC(=O)[C@@H]21 KMOUUZVZFBCRAM-OLQVQODUSA-N 0.000 description 1
- CDULGHZNHURECF-UHFFFAOYSA-N 2,3-dimethylaniline 2,4-dimethylaniline 2,5-dimethylaniline 2,6-dimethylaniline 3,4-dimethylaniline 3,5-dimethylaniline Chemical group CC1=CC=C(N)C(C)=C1.CC1=CC=C(C)C(N)=C1.CC1=CC(C)=CC(N)=C1.CC1=CC=C(N)C=C1C.CC1=CC=CC(N)=C1C.CC1=CC=CC(C)=C1N CDULGHZNHURECF-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical compound OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 241000234282 Allium Species 0.000 description 1
- 235000002732 Allium cepa var. cepa Nutrition 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- GXBYFVGCMPJVJX-UHFFFAOYSA-N Epoxybutene Chemical compound C=CC1CO1 GXBYFVGCMPJVJX-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 1
- 101000905241 Mus musculus Heart- and neural crest derivatives-expressed protein 1 Proteins 0.000 description 1
- AFBPFSWMIHJQDM-UHFFFAOYSA-N N-methylaniline Chemical compound CNC1=CC=CC=C1 AFBPFSWMIHJQDM-UHFFFAOYSA-N 0.000 description 1
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 1
- 229910007926 ZrCl Inorganic materials 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 125000004067 aliphatic alkene group Chemical group 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 1
- 125000005037 alkyl phenyl group Chemical group 0.000 description 1
- 125000003710 aryl alkyl group Chemical group 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 150000001733 carboxylic acid esters Chemical class 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 150000001925 cycloalkenes Chemical class 0.000 description 1
- HGCIXCUEYOPUTN-UHFFFAOYSA-N cyclohexene Chemical compound C1CCC=CC1 HGCIXCUEYOPUTN-UHFFFAOYSA-N 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 150000001941 cyclopentenes Chemical class 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 125000000219 ethylidene group Chemical group [H]C(=[*])C([H])([H])[H] 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 238000012685 gas phase polymerization Methods 0.000 description 1
- 239000003502 gasoline Substances 0.000 description 1
- 150000008282 halocarbons Chemical class 0.000 description 1
- 150000002366 halogen compounds Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 239000003350 kerosene Substances 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000012968 metallocene catalyst Substances 0.000 description 1
- CPOFMOWDMVWCLF-UHFFFAOYSA-N methyl(oxo)alumane Chemical compound C[Al]=O CPOFMOWDMVWCLF-UHFFFAOYSA-N 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000012452 mother liquor Substances 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- HJWLCRVIBGQPNF-UHFFFAOYSA-N prop-2-enylbenzene Chemical compound C=CCC1=CC=CC=C1 HJWLCRVIBGQPNF-UHFFFAOYSA-N 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 150000003377 silicon compounds Chemical class 0.000 description 1
- 238000010557 suspension polymerization reaction Methods 0.000 description 1
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 description 1
- VIPCDVWYAADTGR-UHFFFAOYSA-N trimethyl(methylsilyl)silane Chemical compound C[SiH2][Si](C)(C)C VIPCDVWYAADTGR-UHFFFAOYSA-N 0.000 description 1
- 238000012725 vapour phase polymerization Methods 0.000 description 1
Landscapes
- Transition And Organic Metals Composition Catalysts For Addition Polymerization (AREA)
Abstract
The invention provides a composite solid catalyst and a preparation method thereof. The invention also provides a catalyst system for olefin polymerization, which comprises a composite solid catalyst and a cocatalyst, and additives can be added if necessary.
The composite solid catalyst and the catalyst system thereof provided by the invention have high and stable catalytic activity, and the prepared product has excellent processing performance.
Description
The present invention relates to a kind of compounding solid catalyst and method for making thereof, the invention still further relates to the catalyst system of described compounding solid catalyst preparation and the purposes of this catalyst system.More particularly, what the present invention relates to is compounding solid catalyst and its catalyst system that is used for olefinic polymerization, and this catalyst system is specially adapted to the heterogeneous polymerization or the suspension polymerization of alkene.
As everyone knows, be raw material with alkene, can in the presence of a kind of catalyst system, produce polyolefine, described catalyst system is to comprise metallocene catalyst and by organo-aluminium compound, particularly the promotor formed of aikyiaiurnirsoxan beta.Yet,, therefore be not suitable for being used for the form of suspension of aliphatic hydrocarbon or the method for the heterogeneous polymerization carried out with the gas phase form because this catalyzer will use with aromatic hydrocarbons solution form.
After the homogeneous phase metallocene catalysis agent carrierization of prior art, not high by the catalyst weight calculated activity, and the distribution of prepared polyolefin molecular weight is very narrow, and MWD only is 2-3, and the poor processability energy consumption is big.
For many years, people seek making great efforts, and attempt to find a kind of highly active solid catalyst that alkene heterogeneous (being total to) polymerization process comprises metallocene that is used for, and particularly seek the high solid catalyst of a kind of metallocene content.This catalyzer is applicable to alkene heterogeneous polymerization method.In described method, catalyst particle size size-grade distribution and form are easy to control, and the polyolefinic molecular-weight average of gained is adjustable and have wide molecular weight distribution, so that prepared polymkeric substance or multipolymer can be used injection moulding, molding or extrusion molding.And wish directly to use alkylaluminium cpd, lack usefulness or, be beneficial to suitability for industrialized production without aikyiaiurnirsoxan beta.
The inventor is through studying for a long period of time, prepared a kind ofly to have highly active compounding solid catalyst in alkene (being total to) polymerization, and it is metallocene and the Nonmetallocene compound that is stated from the magnesium halide.And prepared with described compounding solid catalyst and to be used for alkene (being total to) polymeric catalyst system.
The purpose of this invention is to provide a kind of compounding solid catalyst that contains the compound of metallocene compound and Nonmetallocene compound and magnesium.
Second purpose of the present invention provided the preparation method of described compounding solid catalyst.
The 3rd purpose of the present invention provided and has been used for alkene (being total to) polymeric catalyst system with the preparation of described compounding solid catalyst.
The 4th purpose of the present invention provides with described catalyst system carries out alkene (being total to) polymeric method.
The invention provides a kind of compounding solid catalyst.
Catalyst system provided by the invention comprises described compounding solid catalyst and organo-aluminium compound or aikyiaiurnirsoxan beta or both mixtures, and selectively is added with organic silicon compound and/or organoboron compound.
Described compounding solid catalyst and method for making thereof and described catalyst system and uses thereof will be described in detail belows.
This composite catalyst is to be dissolved in the solvent system of the solvent system of organic epoxy compounds and organo phosphorous compounds or organo phosphorous compounds and the homogeneous solution that forms by magnesium halide, can add inert diluent when dissolving.This homogeneous solution and metallocene compound and/or Nonmetallocene compound form solids.In case of necessity, in reaction system, can add precipitation additive.If desired, the solids of formation can be handled with monobasic or polybasic ester.After this, use metallocene compound and/or Nonmetallocene compound treatment again, solids after treatment washs with inert diluent, has just obtained containing metallocene, Nonmetallocene compound, compounding solid catalysts such as magnesium halide, organo phosphorous compounds.In case of necessity, in said process, can add organic boride.
1, the preparation of magnesium halide solution
Magnesium halide solution is meant that magnesium halide is dissolved in organo phosphorous compounds, or the homogeneous solution that obtains in organo phosphorous compounds and the organic epoxy compounds composition solvent system.Here the solvent system of indication comprises use or does not use inert diluent.
(1) magnesium halide
Magnesium halide is selected from a kind of in the group of being made up of magnesium dichloride, dibrominated magnesium, two magnesium iodides or their composition; Or the title complex of magnesium dihalide and water, alcohols; Or the derivative that replaced by-oxyl or halo-oxyl of the halogen atom in the magnesium dihalide.
(2) organo phosphorous compounds
The hydrocarbyl carbonate or the halo hydrocarbyl carbonate that comprise ortho-phosphoric acid or phosphorous acid.Example has: ortho-phosphoric acid trimethyl, ortho-phosphoric acid triethyl, ortho-phosphoric acid tri-n-butyl, ortho-phosphoric acid triphenylmethyl methacrylate, trimethyl phosphite, triethyl-phosphite, tributyl phosphate, triphenyl phosphite etc. or their composition.
(3) organic epoxy compounds
Comprise the compound such as oxide compound, glycidyl ether, inner ether of aliphatics alkene, diolefine or the halogenated aliphatic alkene or the diolefine of 2-8 carbon atom, example has: oxyethane, propylene oxide, butylene oxide ring, butadiene oxide, butadiene double oxide, epoxy chloropropane, methyl glycidyl ether, diglycidylether, tetrahydrofuran (THF) etc. or their composition.
(4) preparation of magnesium halide solution
The granularity of the magnesium halide that uses will under agitation be easy to dissolving, be dissolved under the heating and carry out, solvent temperature is 0-150 ℃, preferred 20-80 ℃, can add inert diluent during dissolving, as hexane, heptane, octane, decane, benzene,toluene,xylene, 1, as long as 2-ethylene dichloride, chlorobenzene etc. and other hydro carbons or halogenated hydrocarbon compound help the magnesium halide dissolved all can use.Above-mentioned inert solvent can use separately, use also capable of being combined, and its consumption is in every mole of magnesium halide, and organic epoxy compounds is the 0-10 mole, preferred 0.3-6 mole, organo phosphorous compounds is the 0.1-3 mole, preferred 0.3-1.5 mole.
2, the preparation of compounding solid catalyst
With above-mentioned magnesium halide solution that makes and metallocene compound, the inclusive NAND metallocene compound, or with metallocene compound and Nonmetallocene compound mixed together, or metallocene compound adds the Nonmetallocene compound earlier with after magnesium halide solution mixes again, or the Nonmetallocene compound with add metallocene compound again after magnesium halide solution mixes, separate out solids.In the solids precipitation process, can add precipitation additive in case of necessity, precipitation additive can add system with solid magnesium halide, also can generate the back at the magnesium halide homogeneous solution and add.Metallocene compound can be pure compound, also can be the solution or the suspension that form with inert diluent; Also can be the solution or the suspension that mix back formation with organoboron compound.The Nonmetallocene compound is like this too.Before or after solid separates out, also can add monobasic or multi-carboxylate, so that the solids of separating out is handled.In each above-mentioned process, can add organoboron compound, preferably contain the boron compound of fluorinated aromatic hydrocarbon base, as three (pentafluorophenyl group) boron (C
6F
5)
3B, four (pentafluorophenyl group) boron lithium (Li (C
6F
5)
4B), four (pentafluorophenyl group) boron (Dimethyl Ammonium), four (pentafluorophenyl group) boron (xylidine).
Solids to separate out operation be at-80 ℃-200 ℃, carry out under preferred-40 ℃-120 ℃.
Wherein the mass ratio of magnesium halide, metallocene compound, Nonmetallocene compound is:
M
1∶Mg=0.001-1.0
M
1∶M
2=0.01-100
Preferred quality than scope is
M
1∶Mg=0.005-0.5
M
1∶M
2=0.05-50
In the formula: M
1Total amount for metal in the metallocene compound
M
2Total amount for metal in the Nonmetallocene compound
Mg: be the content of magnesium in the magnesium halide;
(1) Nonmetallocene compound
The Nonmetallocene compound is meant halogen compound, hydroxyhalide, alkoxy halide, hydrohalogen and the alkyl halide of periodictable III B, IV B, V B and VI B group 4 transition metal.Halogen can be chlorine or bromine or iodine, alkyl in alkoxy halide and the alkyl halide is generally 1-12 carbon atom, it can be straight or branched, preferred IV B, V B group 4 transition metal halogenide, for example tri-chlorination scandium, the tetrabormated scandium, titanium tetrachloride, titanium tetrabromide, titanium tetra iodide, four titanium butoxide, purity titanium tetraethoxide, one chlorine triethoxy titanium, dichloro diethoxy titanium, trichlorine one ethanolato-titanium, zirconium tetrachloride, the tetrabormated zirconium, hafnium tetrachloride, hafnium, the oxychlorination hafnium, vanadium tetrachloride, five vanadium chlorides, the pentabromo-vanadium, vanadium oxytrichloride etc. or their composition.
(2) metallocene compound
Metallocene compound is by general formula R
1R
2R
3R
4Represented compound, M is an IV B family metallic element in the formula, preferred Ti, Zr, Hf; R
1The representative ring alkadienyl, or by the alkyl of the preferred 1-6 of at least one an alkyl carbon atom such as methyl, ethyl, or at least one alkoxyl group, alkoxyl group such as methoxyl group, oxyethyl group, propoxy-or the butoxy of preferred 1-6 carbon atom, or the loop chain dialkylene of silylation replacement, or R
1Expression condensed loop chain dialkylene, the described group of preferred 7-14 carbon atom such as indenyl or tetrahydroindenyl or green onion are basic or by fused rings alkadienyl that above-mentioned substituting group replaced; R
2, R
3Or R
4Can be identical also can be different, the representative ring alkadienyl preferably has conjugated C
5Cycloalkadienyl such as cyclopentadienyl; Contained the alkyl preferable methyl of 1-6 carbon atom by at least one, ethyl, or a loop chain dialkylene and a condensed loop chain dialkylene that contains the preferred methoxyl group of alkoxyl group, oxyethyl group or the silylation replacement of 1-6 carbon atom; The preferred phenyl of aryl that contains 6-19 carbon atom; The alkyl preferable methyl, the ethyl that contain 1-6 carbon atom; The preferred cyclohexyl of cycloalkyl that contains 5-7 carbon atom; The preferred methoxyl group of alkoxyl group, oxyethyl group, propoxy-, the butoxy that contain 1-12 carbon atom; The preferred chlorine of halogen, bromine; Hydrogen atom.Condition is: work as R
2The loop chain dialkylene of representative ring alkadienyl, replacement, or when condensed loop chain dialkylene or the fused rings alkadienyl that replaces, R
1And R
2Keyed jointing directly, or the preferred ethylidene of low-grade alkylidene by a low-grade alkylidene such as 1-4 carbon atom, or the preferred inferior dimethylsilyl of silicylene, inferior diethylsilyl, the mutual keyed jointing of tetramethyl-disilane base.The example of metallocene compound has: molybdenyl dichloride (cyclopentadienyl) titanium, two (cyclopentadienyl) dibenzyl titanium, dichloride ethylenebis (4,5,6,7-tetrahydrochysene 11 indenyls) titanium, two (indenyl) methylcyclopentadienyl zirconiums of molybdenyl dichloride (five dipentadiene bases) lithium chlorination, two (cyclopentadienyl) phenoxy group zirconiums of chlorination, two (indenyl) zirconiums of protochloride dimethylsilyl, chlorination ethylenebis (4,5,6,7-tetrachloro 11 indenyls) zirconium, two (cyclopentadienyl) hafniums of a chlorine one hydrogenation, two (encircling luxuriant dialkylene) the alkylphenyl group hafniums of chlorination etc. or their composition.
(3) precipitation additive
Precipitation additive comprises compounds such as organic acid anhydride, organic acid, ketone, ether, and example has: diacetyl oxide, Tetra hydro Phthalic anhydride, Succinic anhydried, MALEIC ANHYDRIDE, pyromellitic acid anhydride, acetic acid, propionic acid, butyric acid, vinylformic acid, methacrylic acid, acetone, butanone, benzophenone, methyl ether, ether, propyl ether, butyl ether, amyl ether etc. or their composition.
(4) monobasic or multi-carboxylate
Monobasic or multi-carboxylate comprise aliphatic carboxylic acid esters, and aromatic carboxylic acid esters, and example has: methyl-formiate, butyl formate, butylacetate, butyl propionate, ethyl benzoate, butyl benzoate, the naphthoic acid butyl ester, diethyl malonate, butyl ethyl malonate, Polycizer W 260, ethyl sebacate, n-butyl phthalate, diisobutyl phthalate, dimixo-octyl phthalate, diethyl maleate, maleic acid n-butyl, the naphthalene dicarboxylic acids diethyl ester, the naphthalene dicarboxylic acids dibutylester, triethyl trimellitate, tributyl trimellitate, the benzene-1,2,3-tricarboxylic acid triethyl, connection benzenetricarboxylic acid tri-n-butyl, the pyromellitic acid tetra-ethyl ester, pyromellitic acid four butyl esters etc. or their composition.
Above-mentioned solids is mingled with multiple complicated complex compound and impurity, needs further to handle.Solids can be with the mixture process of metallocene compound or itself and inert diluent; The mixture process of available Nonmetallocene compound or itself and inert diluent; Or both handle simultaneously.Solids after treatment washs with inert diluent subsequently.Solids after the washing is compounding solid catalyst of the present invention, can solids or the use down of suspension state.
The feature and the chemical constitution of compounding solid catalyst of the present invention are as follows;
The prepared compounding solid catalyst of the present invention is the pulverulent solids particulate, and median size is the 2-40 micron, and specific surface area is at least 30 meters
2More than/the gram, preferred 100 meters
2More than/the gram.Its chemical constitution is: magnesium 10-30% (wt), halogen 40-80% (wt), M
10.1-10% (wt), M
20.1-10% (wt), carboxylicesters 0-25% (wt), organo phosphorous compounds 0.1-2.5% (wt), organoboron compound 0-20% (wt), and inert diluent 0-15% (wt).(M
1Be the total amount of metal in the Nonmetallocene compound; M
2Be the total amount of metal in the metallocene compound).
3, be used for alkene (being total to) polymeric catalyst system
Of the present inventionly be used for that alkene (being total to) polymeric catalyst system comprises compounding solid catalyst of the present invention and aluminum alkyls or by the aluminoxane catalyst of aluminum alkyls preparation.According to the kind difference of alkene, can add or not add additive.
(1) alkylaluminium cpd
Alkylaluminium cpd is by general formula ALRnX
3-nExpression, R is H in the formula, carbonatoms is the alkyl of 1-20, preferred alkyl, aralkyl, aryl; X is a halogen, preferred chlorine and bromine, and n is the integer of 0<n<≤3, example has: trialkylaluminiums such as trimethyl aluminium, triethyl aluminum, triisobutyl aluminium, trioctylaluminum; Alkyl aluminium hydrides such as one hydrogen diethyl aluminum, a hydrogen diisobutyl aluminum; Aluminum alkyls muriates such as aluminium diethyl monochloride, a chloro-di-isobutyl aluminum, sesquialter ethyl aluminum chloride, ethyl aluminum dichloride.
(2) aikyiaiurnirsoxan beta
Aikyiaiurnirsoxan beta is the partial hydrolysate of alkylaluminium cpd, and it can be earlier be made by aluminum alkyls and water reaction, also can be replaced by aluminum alkyls in polymerization reaction system and an amount of water.
Expression, R is an alkyl in the formula, preferable methyl, ethyl, isobutyl-; Part R also can be replaced by halogen and/or hydroxyl among a plurality of R; P is for being 1 integer at least.
Above-mentioned aikyiaiurnirsoxan beta has also comprised the partial hydrolysate of several aluminum alkyls mixtures, also comprised partial hydrolysate further with the reacted product of water (aikyiaiurnirsoxan beta that promptly contains part of hydroxyl), also comprise the product (being partially halogenated aikyiaiurnirsoxan beta) that is further replaced by halogen.
(3) additive
Alkene (being total to) the polymeric catalyst system that is used for of the present invention is usually without additive.But to (being total to) polymerization of some alkene, but also doping.Useful additives is silicoorganic compound, by formula RnSi (OR ')
4-nExpression, 0<n in the formula≤3, R and R ' they are identical or different alkyl, cycloalkyl, aryl, halogenated alkyl etc., R also can be halogen atom or hydrogen atom.Example has: trimethylammonium methoxy silane, trimethylethoxysilane, trimethyl phenoxysilane, dimethyldimethoxysil,ne, dimethyldiethoxysilane, dimethoxydiphenylsilane, phenylbenzene diethoxy silane, phenyl triethoxysilane, phenyltrimethoxysila,e, ethene Trimethoxy silane and their composition.
Additive also can be organoboron compound, and being preferably with the fluoro aromatic ring is that the boron compound of dentate is as three (pentafluorophenyl group) boron (C
6F
5)
3B, four (pentafluorophenyl group) boron lithium (Li (C
6F
5)
4B), four (pentafluorophenyl group) boron (Dimethyl Ammonium), four (pentafluorophenyl group) boron (monomethylaniline).
Wherein said additive also can be the composition of silicoorganic compound and organoboron compound.
4, (being total to) polymerization of alpha-olefin
Being used for the polymeric alpha-olefin has ethene, propylene, 1-butylene, 4-methylpentene-1,1-hexene, 1-octene, vinylbenzene, vinyl toluene etc., and these alkene can not only carry out equal polymerization separately, and can carry out random copolymerization, block copolymerization.Also can select conjugated dienes or non-conjugated dienes as comonomer during copolymerization.Above-mentioned alpha-olefin has also comprised cycloolefin, as cyclopentenes, tetrahydrobenzene etc.
Can adopt liquid polymerization during polymerization, also can adopt vapour phase polymerization, when carrying out liquid polymerization, can use hexane, heptane, hexanaphthene, petroleum naphtha, raffinate oil, inert solvent such as aliphatic saturated hydrocarbon such as hydrogenated gasoline, kerosene, benzene,toluene,xylene or aromatic hydrocarbon makes reaction medium.
Also available alkene itself is made reaction medium, can carry out prepolymerization earlier before the polymerization, and polymerization methods can adopt intermittent type, semi continuous or continous way.
Polymerization temperature is 0-150 ℃, with 40-100 ℃ for well, in order to regulate molecular weight, adopt hydrogen to make molecular weight regulator.
Catalyst system of the present invention can be used for alkene gas-phase polymerization or liquid polymerization.
5, positively effect of the present invention
(1) catalyst activity is high especially, thus in the resulting polymers residual catalyst content very pettiness can still can obtain well behaved polymkeric substance without any aftertreatment.
(2) prolong with polymerization time, catalyst activity reduction is little, is not only applicable to homopolymerization and closes, and also can be applicable to random and block copolymerization.
(3) resulting polymers narrow diameter distribution, fine powder content seldom, apparent density is big, and is very favourable to the polymerization process and the course of processing.
(4) molecular weight distribution of resulting polymers can be regulated, and can obtain the very wide polymkeric substance of molecular weight distribution, can expand the range of application of polymkeric substance.
(5) the catalyst system suitability is wide, can be applicable to various polymerization techniques, also can be applicable to the polymerization of various alkene.
(6) the catalyzer synthesis condition is not harsh, and the raw material consumption amount is lower, the plant factor height.
(7) during industrial application, existing apparatus need not big change, is convenient to industrialization.
(8) use the present invention, can make and contain the extra high catalyzer of metallocene compound.
As above-mentioned, the invention provides a kind of compounding solid catalyst and be used for alkene (being total to) polymeric catalyst system; The present invention the preparation method of compounding solid catalyst also is provided, also provide with catalyst system of the present invention and carried out alpha-olefin (being total to) polymeric method.Those skilled in the art are fully aware of, and the present invention can also carry out various modifications or improvement, but only otherwise leave spirit of the present invention, all within the scope of the present invention.The present invention's scope required for protection will propose in claims.
Below, will be explained in more detail the present invention with example.
The synthesis of solid catalyzer
Example 1
Under nitrogen protection; the 4.1g magnesium chloride joined 64ml toluene is housed; 6.4ml epoxy chloropropane in the vial of the 500ml after washing, drying, nitrogen fill row of 7ml tributyl phosphate, and makes it dissolving; add the 1.1g phthalic anhydride then; after the stirring, add the 0.4973g bis cyclopentadienyl zirconium dichloride, and make system temperature drop to-30 ℃ simultaneously; in vial, slowly drip 48ml then and leach mother liquor then, at room temperature wash the precipitating thing twice with 88ml toluene.Mixed solution with 35ml titanium tetrachloride and 52ml toluene mixed 2 hours with the precipitating thing down at 100 ℃ again, so handle twice after, under 110 ℃, wash precipitating thing twice with 88ml toluene, wash twice with the 88ml hexane again, drain the 5.7g solid catalyst.
This catalyzer is a brown powder, and Zr/Mg (wt) is 0.024, and Zr/Ti (wt) is that 0.253 specific surface area is 190m
2/ g.
Embodiment 2
In the 500ml vial, the 3g magnesium chloride is dissolved in the mixed solution of 48ml toluene, 4.8ml epoxy chloropropane and 5.1ml tributyl phosphate composition, add the 0.8g phthalic anhydride then and mix the back cooling, between cooldown period, add the 0.2579g bis cyclopentadienyl zirconium dichloride, when system temperature slowly drips the 36ml titanium tetrachloride to-30 ℃ the time in system, slowly be warming up to 80 ℃ again, add 2.4mlDIBP then and stir, filter then, get the precipitating thing.At room temperature wash the precipitating thing twice with 88ml toluene, then, the precipitating thing is mixed with 48ml toluene, 35ml titanium tetrachloride, stirred 2 hours down at 100 ℃, after so handling twice, use 88ml toluene wash precipitating thing twice again, make precipitating thing and 15ml bis cyclopentadienyl zirconium dichloride toluene solution (0.2579gCpZrCl then
2) mixed 15 minutes, wash the precipitating thing with toluene then.Wash secondary with the 86ml hexane again and drain, get solid catalyst 4.5g, by analysis, this catalyzer is a brown powder, and Zr/Mg (wt) is 0.107, and Zr/Ti (wt) is 0.303, and specific surface area is 336.3m
2/ g.
Embodiment 3
Press embodiment 2, just the bis cyclopentadienyl zirconium dichloride in the intermediate temperature drop process does not add, and other are identical, gets solid catalyst 5.6g at last.
By analysis, Zr/Mg in this catalyzer (wt) is 0.0828, and Zr/Ti (wt) is 0.257, and specific surface area is 185.3m
2/ g.
Embodiment 4
Magnesium chloride 3g is dissolved in 40ml toluene, the 3.2ml epoxy chloropropane, in the mixed solution that 3.4ml tributyl phosphate and 1.5ml dehydrated alcohol are formed, other are with synthetic catalyst embodiment 3.
Get solid catalyst 3.8g at last.
By analysis, Zr/Mg (wt) is 0.198, and Zr/Ti (wt) is 0.841, and specific surface area is 198.4m
2/ g.
Embodiment 5
In the 500ml vial, the 3g magnesium chloride is dissolved in 40ml toluene, 3.2ml in the mixed solution that epoxy chloropropane and 3.4ml tributyl phosphate are formed, add phthalic anhydride 0.8g then, mixing the back cooling makes system temperature reach-30 ℃, slowly be warming up to 80 ℃ after slowly dripping titanium tetrachloride 36ml, and then stirred 15 minutes, filter, wash precipitating thing secondary with toluene 80ml, add toluene 48ml and titanium tetrachloride 35ml then in the precipitating thing and mixed 2 hours down at 100 ℃, so handle twice after, again with toluene 80ml washing precipitating thing, add toluene 80ml again, dehydrated alcohol 2ml after stirring 30 minutes under 110 ℃, filters in the precipitating thing, in the precipitating thing, add bis cyclopentadienyl zirconium dichloride toluene solution 15ml, (contain Cp
2ZrCl
20.2579g) add hexane 80ml to system after mixing half an hour, stir half an hour, filter, wash the precipitating thing 1 time with toluene 80ml, wash precipitating thing twice with hexane 80ml again, drain, must solid catalyst 4.0g.
By analysis, this catalyzer Zr/Mg (wt) is 0.12, and Zr/Ti (wt) is 0.740, and specific surface area is 107.5m
2/ g.
Olefinic polymerization
Embodiment 1
The stainless steel stirring tank; with nitrogen replacement qualified after; replace nitrogen with reaction gas again, and positive pressure protection is stand-by, under nitrogen protection; in still, add the 500ml refined hexane; 0.69ml triethyl aluminum and 0.07ml silane (DPMS) are used the 300ml hexane again, and the solid catalyst of 15mg synthesis of solid catalyzer embodiment 1 gained is poured in the still; and feeding appropriate amount of hydrogen; feed hot water then and heat up in the stirring tank chuck, logical propylene reaction keeps 70 ℃ of temperature of reaction; reaction pressure 0.68MPa; react and stop after 2 hours, cooling decompression is emitted the still interpolymer; get polymkeric substance 165g after the oven dry; its apparent density is 0.458g/ml, and distribution of particle sizes is the 20-60 order, accounts for 96.8%; molecular weight distribution value Mw/Mn:4.09, the MI value is 17.43.
Embodiment 2
In the 2L stainless steel cauldron, add the 400ml refined hexane, the water of the triethyl aluminum of 0.69ml and 15 microlitres reacted 15 minutes down at 50 ℃, be charged to 0.4MPa with reaction gas then, drain into normal pressure twice then, after this use the 400ml hexane that the catalyzer of 0.07ml silane (DPMS) and 12.4mg synthesis of solid catalyzer embodiment 1 gained is poured in the still, add appropriate amount of hydrogen, logical hot water heats up in the stirring tank chuck then, logical propylene reaction keeps 70 ℃ of temperature of reaction, reaction pressure 0.63MPa, reacted 2 hours, cooling decompression is emitted the still interpolymer, gets 105g after the oven dry, its apparent density is 0.46g/ml, granularity is the 20-60 order, accounts for 93.6%, and the MI value is 19.28.
Embodiment 3
Change 25.5 milligrams into by polymerization embodiment 1 solid catalysis dosage, reaction pressure changes 0.6MPa into, gets polypropylene 261g at last, and apparent density is 0.4588/ml, size-grade distribution: the 20-60 order accounts for 93.8%, and molecular weight distribution mw/mn is 4.90, and the MI value is 29.46.
Embodiment 4
By polymerization embodiment 1, catalyzer changes the solid catalyst 15.8mg by synthesis of solid catalyzer example 2 gained into, keep-ups pressure to be 0.6MPa, gets polypropylene 115g at last, and it shows density 0.427, and 20-80 order particle accounts for 94.8%.
Embodiment 5
By polymerization embodiment 1, the solid catalysis dosage that catalyzer changes into by synthesis of solid catalyzer embodiment 3 gained is 31.4mg, keeps propylene pressure 0.60MPa, gets polypropylene 260g at last, and its apparent density is 0.429, and 20-80 order particle accounts for 91.8%.
Embodiment 6
By polymerization embodiment 1, catalyzer is that the solid catalysis dosage of synthesis of solid catalyzer embodiment 3 gained is 27.8mg, and keeping propylene pressure is 0.63MPa, and aluminum alkyls changes triisobutyl aluminium 0.72ml into, gets polypropylene 190g at last, and apparent density is 0.40.
Embodiment 7
By polymerization example 1, catalyzer changes the solid catalyst by synthetic catalyst example 4 gained into, 10.0mg, and reaction gas changes ethene into, and silane does not add, and the maintenance ethylene pressure is 0.6MPa, gets polyethylene 105g at last, performance density 0.374.Molecular weight distribution mw/mn is 3.51.
Embodiment 8
By polymerization embodiment 7, just the reaction times changes 4 hours into, and catalytic amount is 12.5mg, gets polyethylene 270g at last, and apparent density is 0.391.
Embodiment 9
By polymerization embodiment 7, catalytic amount changes 12.2mg into, and aluminum alkyls changes triisobutyl aluminium into, and 0.70ml gets polyethylene 185g at last, and apparent density is 0.376, and molecular weight distribution mw/mn is 4.53.
Embodiment 10
By polymerization embodiment 7, solid catalysis dosage is 13.1mg, and aluminum alkyls changes methylaluminoxane (available from German Schering Corp 30% toluene solution) 1.2ml into, triisobutyl aluminium 0.17ml, get polyethylene 180g at last, apparent density is 0.351, and molecular weight distribution is that Mw/Mn is 9.81.
Embodiment 11
By polymerization embodiment 7, catalyzer changes the solid catalyst 9.3mg of synthetic catalyst example 5 gained into, and other get polyethylene 85g, apparent density 0.363 together at last.
Claims (32)
1, a kind of compounding solid catalyst comprises magnesium halide, metallocene compound, Nonmetallocene compound, organo phosphorous compounds or organic epoxy compounds or both, and wherein the mass ratio of magnesium halide, metallocene compound, Nonmetallocene compound is:
M
1∶Mg=0.001-1.0
M
1∶M
2=0.01-100
In the formula: M
1Total amount for metal in the metallocene compound
M
2Total amount for metal in the Nonmetallocene compound
Mg: be the content of magnesium in the magnesium halide;
Wherein the Nonmetallocene compound is halogenide, alkoxy halide, hydrohalogen, the alkyl halide of the transition metal of periodictable III B, IV B, VI B family;
Alkyl in described alkoxy halide and the alkyl halide is the alkyl of the straight or branched of 1-12 carbon atom.
2, press the catalyzer of claim 1, wherein
M
1∶Mg=0.005-0.5
M
1∶M
2=0.05-50
3, by claim 1 or 2 catalyzer, wherein magnesium halide is to be selected from a kind of in magnesium dichloride, dibrominated magnesium, two magnesium iodides or their composition.
4, by claim 1 or 2 catalyzer, wherein magnesium halide is a magnesium dihalide and water, pure title complex; Or the halogen atom in the magnesium dihalide is by the derivative of-oxyl or the replacement of halo-oxyl.
5, by the catalyzer of claim 1 or 2, wherein metallocene compound is by general formula R
1R
2R
3R
4The compound that M represents, M is an IV B family metallic element in the periodictable in the formula;
R
1The representative ring alkadienyl; By the loop chain dialkylene of the alkyl of at least one 1-6 carbon atom, alkoxyl group replacement; The loop chain dialkylene that is replaced by silylation; Thick and the loop chain dialkylene; The fused rings alkadienyl that replaces;
R
2, R
3, R
4Identical or inequality, be above-mentioned replacement or unsubstituted loop chain dialkylene, condensed loop chain dialkylene; Aryl; Alkyl; Silylation; The cycloalkyl of 5-7 carbon atom; Halogen atom; The alkoxyl group of 1-12 carbon atom; Hydrogen atom.
6, catalyzer by claim 1 or 2, wherein metallocene compound is selected from by molybdenyl dichloride (cyclopentadienyl) titanium, two (cyclopentadienyl) dibenzyl titanium, dichloride ethylenebis (4,5,6,7-tetrahydrochysene-1-indenyl) titanium, molybdenyl dichloride cyclopentadienyl zirconium, two (indenyl) methylcyclopentadienyl zirconiums of chlorination, two (encircling luxuriant dialkylene) the phenoxy group zirconiums of chlorination, two (indenyl) zirconiums of protochloride dimethyl, chlorination ethylenebis (4,5,6,7-tetrahydrochysene-1-indenyl) zirconium, two (cyclopentadienyl) zirconiums of one chlorine, one hydrogenation, the dichloro hafnocene, a kind of in two (tetrahydroindene) hafniums of protochloride dimethyl or their composition.
7, press the catalyzer of claim 1 or 2, wherein the Nonmetallocene compound is selected from a kind of in tri-chlorination scandium, tribromide scandium, titanium tetrachloride, titanium tetrabromide, titanium tetra iodide, four titanium butoxide, purity titanium tetraethoxide, a chlorine triethoxy titanium, dichloro diethoxy titanium, trichlorine one ethanolato-titanium, zirconium tetrachloride, tetrabormated zirconium, hafnium tetrachloride, hafnium, oxychlorination hafnium, vanadium tetrachloride, five vanadium chlorides, pentabromo-vanadium, the vanadium oxytrichloride or their composition.
8, by the catalyzer of claim 1 or 2, wherein organo phosphorous compounds is the hydrocarbyl carbonate or the halo hydrocarbyl carbonate of ortho-phosphoric acid or phosphorous acid.
9, by claim 1 or 2 catalyzer, wherein organo phosphorous compounds is selected from a kind of in ortho-phosphoric acid trimethyl, ortho-phosphoric acid triethyl, ortho-phosphoric acid tri-n-butyl, ortho-phosphoric acid triphenylmethyl methacrylate, trimethyl phosphite, triethyl-phosphite, tributyl phosphate, the triphenyl phosphite or their composition.
10, press the catalyzer of claim 1 or 2, the aliphatics alkane that wherein organic epoxy compounds is a 2-8 carbon atom, the epoxide of alkene, the epoxide of halogenated aliphatic alkane, alkene, glycidyl ether, inner ether.
11, press the catalyzer of claim 1 or 2, wherein organic epoxy compounds is selected from a kind of in oxyethane, butylene oxide ring, propylene oxide, epoxidized butadiene thing, divinyl di-epoxide, epoxy chloropropane, methyl glycidyl ether, diglycidylether, the tetrahydrofuran (THF) or their composition.
12, the preparation method of the described compounding solid catalyst of claim 1 comprises
(1) preparation magnesium halide solution; Magnesium halide is dissolved in the solvent system of the solvent system of organic epoxy compounds and organo phosphorous compounds or organo phosphorous compounds and forms homogeneous solution;
(2) separate out solids
Magnesium halide solution and metallocene compound or Nonmetallocene compound or the mixture of the two are mixed, separate out solids;
Wherein the mass ratio of magnesium halide, metallocene compound, Nonmetallocene compound is:
M
1∶Mg=0.001-1.0
M
1∶M
2=0.01-100
In the formula: M
1Total amount for metal in the metallocene compound
M
2Total amount for metal in the Nonmetallocene compound
Mg: be the content of magnesium in the magnesium halide;
(3) processing of solids
The described solids of mixture process with metallocene compound and/or Nonmetallocene compound or itself and inert diluent;
(4) the described solids crossed of carrying out washing treatment.
13, by the method for claim 12, wherein in the solids precipitation process, add precipitation additive.
14, by the method for claim 12, wherein in the solids precipitation process, add monobasic or multi-carboxylate.
15, by the method for claim 13, wherein precipitation additive is selected from organic acid anhydride; Organic acid acetic; Ketone compounds; A kind of in the ether compound or their composition.
16, by the method for claim 14, wherein monobasic or multi-carboxylate are selected from the aliphatic carboxylic acid esters; A kind of in the aromatic carboxylic acid esters or their composition.
17, by the method for claim 12, wherein said inert diluent is selected from hexane, heptane, octane, decane, benzene,toluene,xylene, 1, a kind of in 2-ethylene dichloride, the chlorobenzene or their composition.
18,, after wherein separating out solids and be metallocene compound or Nonmetallocene compound and magnesium halide solution mixing, add Nonmetallocene compound or metallocene compound again by the method for claim 12.
19, by the method for claim 12, wherein in the step of (1)-(4), add organoboron compound.
20, by the method for claim 19, wherein organoboron compound is selected from three (pentafluorophenyl group) boron (C
6F
5)
3B, four (pentafluorophenyl group) boron lithium (Li (C
6F
5)
4B), four (pentafluorophenyl group) boron (Dimethyl Ammonium), four (pentafluorophenyl group) boron (xylidine) (CH
3)
2NH
2(C
6F
5)
4A kind of among the B or their composition.
21, the catalyst system used of a kind of alkene (being total to) polymerization comprises the composition of the described a kind of compounding solid catalyst of aforesaid right requirement and a kind of aluminum alkyls or its aikyiaiurnirsoxan beta or aluminum alkyls and aikyiaiurnirsoxan beta.
22, by the catalyst system of claim 21, also add additive.
23, by the catalyst system of claim 21, alkylaluminium cpd wherein is by general formula AlR
nX
3-nThe compound of expression, R is the alkyl of hydrogen or 1-20 carbon atom in the formula; X is that halogen, n are the integer of 0<n<3.
24, by the catalyst system of claim 21, alkylaluminium cpd wherein is selected from a kind of in trimethyl aluminium, triethyl aluminum, triisobutyl aluminium, trioctylaluminum, a hydrogen diethyl aluminum, a hydrogen diisobutyl aluminum, aluminium diethyl monochloride, a chloro-di-isobutyl aluminum, sesquialter ethyl aluminum chloride, the ethyl aluminum dichloride or their composition.
26, by the catalyst system of claim 22, wherein additive is silicoorganic compound, and it is by general formula R nSi (OR ')
4-nShown compound, the integer of 0≤n in the formula≤3, R are hydrogen or halogen atom, R and R ' they are alkyl, cycloalkyl, aryl, haloalkyl of the same race or not of the same race.
27, by the catalyst system of claim 26, wherein silicoorganic compound are selected from by a kind of in trimethylammonium methoxy silane, trimethylethoxysilane, trimethyl phenoxysilane, dimethyldimethoxysil,ne, dimethyldiethoxysilane, dimethoxydiphenylsilane, phenylbenzene diethoxy silane, phenyl triethoxysilane, phenyltrimethoxysila,e, the ethene Trimethoxy silane or their composition.
28, by the catalyst system of claim 22, wherein additive is an organoboron compound.
29, by the catalyst system of claim 22, wherein additive is the composition of silicoorganic compound and organoboron compound.
30, with the catalyst system of claim 22-29, carry out alkene gas phase or liquid polymerization.
31, by the method for claim 13, wherein precipitation additive is selected from a kind of in diacetyl oxide, phthalic anhydride, Succinic anhydried, cis-butenedioic anhydride, pyromellitic acid anhydride, acetic ester, propionic ester, butyric ester, acrylate, methacrylic ester, acetone, butanone, benzophenone, methyl ether, ether, propyl ether, butyl ether, the amyl ether or their composition.
32, by the method for claim 14, wherein monobasic or multi-carboxylate are selected from methyl-formiate, butyl formate, butylacetate, butyl propionate, ethyl benzoate, butyl benzoate, the naphthoic acid butyl ester, diethyl malonate, butyl ethyl malonate, Polycizer W 260, ethyl sebacate, n-butyl phthalate, diisobutyl phthalate, dimixo-octyl phthalate, diethyl maleate, maleic acid n-butyl, the naphthalene dicarboxylic acids diethyl ester, the naphthalene dicarboxylic acids dibutylester, triethyl trimellitate, tributyl trimellitate, biphenyl three triethylenetetraminehexaacetic acid esters, connection benzenetricarboxylic acid tri-n-butyl, the pyromellitic acid tetra-ethyl ester, a kind of in pyromellitic acid four butyl esters or their composition.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN95106710A CN1069100C (en) | 1995-06-16 | 1995-06-16 | Composite solid catalyst and its preparation method and use |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN95106710A CN1069100C (en) | 1995-06-16 | 1995-06-16 | Composite solid catalyst and its preparation method and use |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1138589A CN1138589A (en) | 1996-12-25 |
| CN1069100C true CN1069100C (en) | 2001-08-01 |
Family
ID=5075979
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN95106710A Expired - Fee Related CN1069100C (en) | 1995-06-16 | 1995-06-16 | Composite solid catalyst and its preparation method and use |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN1069100C (en) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1096474C (en) * | 1998-12-30 | 2002-12-18 | 化学工业部北京化工研究院 | Composite catalyst for synthesizing wide distribution ethylene polymer and its preparation method and application |
| US6743873B2 (en) * | 2002-05-28 | 2004-06-01 | Rohm And Haas Company | Olefin polymerization catalyst composition and preparation thereof |
| US6630547B1 (en) * | 2002-06-11 | 2003-10-07 | Equistar Chemicals, Lp | Use of silanes to control molecular weight in olefin polymerizations |
| EP2607388B1 (en) | 2010-08-19 | 2015-08-12 | China Petroleum & Chemical Corporation | Catalytic composition for polymerization of olefin and preparation method thereof |
| CN102453131B (en) * | 2010-10-19 | 2013-08-14 | 中国石油化工股份有限公司 | Catalyst ingredient for olefin polymerization and preparation method thereof |
| DK2586801T3 (en) | 2011-10-29 | 2015-01-26 | China Petroleum & Chemical | The propylene copolymer, process for its preparation and to compositions and articles containing same |
| CN103087237B (en) * | 2011-10-29 | 2015-03-11 | 中国石油化工股份有限公司 | Preparation method of propylene random copolymer |
| CN105199024B (en) * | 2014-06-24 | 2018-01-23 | 中国石油化工股份有限公司 | A kind of catalytic component for vinyl polymerization, catalyst and preparation method thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN85100997A (en) * | 1985-04-01 | 1987-01-10 | 化工部北京化工研究院 | The catalyst system that is used for olefinic polymerization and copolymerization |
| CN1053433A (en) * | 1989-12-22 | 1991-07-31 | 英国石油化学品有限公司 | Be used to prepare the catalyzer and the prepolymer of polyolefins |
-
1995
- 1995-06-16 CN CN95106710A patent/CN1069100C/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN85100997A (en) * | 1985-04-01 | 1987-01-10 | 化工部北京化工研究院 | The catalyst system that is used for olefinic polymerization and copolymerization |
| CN1053433A (en) * | 1989-12-22 | 1991-07-31 | 英国石油化学品有限公司 | Be used to prepare the catalyzer and the prepolymer of polyolefins |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1138589A (en) | 1996-12-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1080268C (en) | Catalyst compositions and process for preparing polyolefins | |
| CN1034125C (en) | Metallocene catalysts with lewis acids and aluminum alkyls | |
| CN1134467C (en) | Shear thining ethylene/alpha-olefin interpolymers and their preparation | |
| CN85100997A (en) | The catalyst system that is used for olefinic polymerization and copolymerization | |
| CN1092206C (en) | Process and catalyst for preventing reactor fouling | |
| CN1202140C (en) | Olfin polymerization catalyst componnet, its prepn. and use | |
| CN1049897C (en) | Catalyst composition for use in the polymerization and copolymerization of ethylene | |
| CN1044372C (en) | Solid precursor of a catalytic system for the polymerization of olefins, process for its preparation, catalytic system comprising this solid precursor and process for the polymerization of olefins | |
| CN1086191C (en) | Catalyst for ethylene polymerization or copolymerization and preparation method thereof | |
| CN1098866C (en) | Gas-phase method total density polyvinyl high-efficiency catalyst | |
| CN1149586A (en) | Stereo rigid metallocene compound | |
| JPH075655B2 (en) | Catalyst component and polymerization catalyst system | |
| CN85108908A (en) | Polymerizing catalyst and its preparation and application | |
| CN1414980A (en) | Mixed metal alkoxide and cycloalkadienyl catalysts for preparing polyolefins | |
| CN1561349A (en) | Supported phosphinimine polymerization catalyst | |
| CN1069100C (en) | Composite solid catalyst and its preparation method and use | |
| CN1157291A (en) | Catalyst for production of olefin polymers | |
| GB1586267A (en) | Catalyst for polymerizing olefins and process for the polymerization of olefins | |
| CN1160059A (en) | Process for producing olefin polymers | |
| CN1233671C (en) | Catalyt and its component for polymerization of ethylene | |
| CN1227271C (en) | Solid catalytic composition for olefin polymerization | |
| CN1324055C (en) | Ethene polymerization catalyst, its production and polymerization kinetic behaviour control | |
| CN1274721C (en) | Catalyst for ethene polymerization or multipolymer, preparing method thereof | |
| CN1124293C (en) | Composite catalyst for preparing polypropylene with wide molecular weight distribution and its prepn | |
| CN1867594A (en) | Olefin Polymerization Process |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C17 | Cessation of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20010801 Termination date: 20100616 |