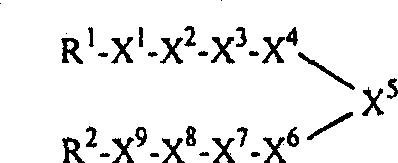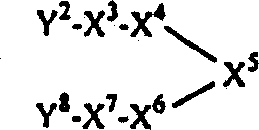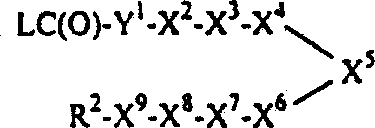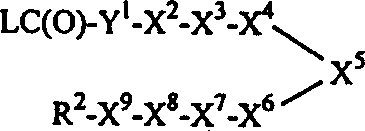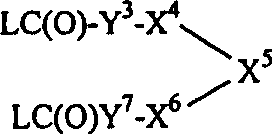CN1073103C - Compounds and compositions for treating diseases associated with tryptase activity - Google Patents
Compounds and compositions for treating diseases associated with tryptase activity Download PDFInfo
- Publication number
- CN1073103C CN1073103C CN97196877A CN97196877A CN1073103C CN 1073103 C CN1073103 C CN 1073103C CN 97196877 A CN97196877 A CN 97196877A CN 97196877 A CN97196877 A CN 97196877A CN 1073103 C CN1073103 C CN 1073103C
- Authority
- CN
- China
- Prior art keywords
- base
- compound
- alkylidene group
- alkyl
- inferior
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/16—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms
- C07D295/20—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms by radicals derived from carbonic acid, or sulfur or nitrogen analogues thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/16—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms
- C07D295/20—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms by radicals derived from carbonic acid, or sulfur or nitrogen analogues thereof
- C07D295/215—Radicals derived from nitrogen analogues of carbonic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/08—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms
- C07D211/18—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D211/26—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with substituted hydrocarbon radicals attached to ring carbon atoms with hydrocarbon radicals, substituted by nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/04—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D233/06—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to ring carbon atoms
- C07D233/08—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to ring carbon atoms with alkyl radicals, containing more than four carbon atoms, directly attached to ring carbon atoms
- C07D233/12—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to ring carbon atoms with alkyl radicals, containing more than four carbon atoms, directly attached to ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms
- C07D233/14—Radicals substituted by oxygen atoms
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pulmonology (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Hydrogenated Pyridines (AREA)
- Pyridine Compounds (AREA)
Abstract
本发明涉及用作类胰蛋白酶抑制剂的新化合物、其药学上可接受的盐和N-氧化物、其作为治疗药物的用途以及它们的制备方法。The present invention relates to novel compounds useful as tryptase inhibitors, their pharmaceutically acceptable salts and N-oxides, their use as therapeutic drugs and processes for their preparation.
Description
本申请要求1996年7月30登记的美国在先申请号60/023,139的优先权。This application claims priority to prior US Application Serial No. 60/023,139, filed July 30,1996.
发明领域:Field of invention:
本发明涉及通过施用新的类胰蛋白酶抑制剂而治疗与类胰蛋白酶活性有关的疾病的新方法和组合物。The present invention relates to new methods and compositions for treating diseases associated with tryptase activity by administering novel tryptase inhibitors.
本领域的描述:Description of the field:
从人类肥大细胞中分泌出来的主要的蛋白酶-类胰蛋白酶被认为与神经肽的加工和组织发炎有关。过敏几个小时后,血液中的类胰蛋白酶浓度升高(Schwartz等,(1987)N.Eng.J.Med.316:1622-1626);特异性抗原免疫激发后,特定主体的鼻腔和肺的灌洗液中类胰蛋白酶浓度升高(Castells等人,(1988)J.Allerg.Clin.Immunol.141:563-568);在支气管内的过敏原免疫激发后,特定的哮喘病人的肺灌洗液中类胰蛋白酶浓度升高。吸烟者的支气管肺泡灌洗液中类胰蛋白酶浓度常常显著升高,这个发现支持了这样的假定:从活化的肥大细胞中释放蛋白酶与吸烟者肺气肿的肺破坏有关。(Celenteron等人,(1988)Chest 94:119-123)。另外,类胰蛋白酶表现为成纤维细胞的强有力的分裂素,这暗示类胰蛋白酶可能参与了肺的纤维变性并与间隙性肺病有关(Rose等人,(1991)J.Clin.Invest.88:493-499)。Tryptase, the major protease secreted from human mast cells, is thought to be involved in neuropeptide processing and tissue inflammation. After a few hours of allergy, the concentration of tryptase in the blood increases (Schwartz et al., (1987) N.Eng.J.Med.316:1622-1626); Increased concentration of tryptase in the lavage fluid (Castells et al., (1988) J.Allerg.Clin.Immunol.141:563-568); After intrabronchial allergen immune challenge, the lungs of specific asthmatic patients Increased tryptase concentration in the lavage fluid. The finding that tryptase concentrations are often markedly elevated in the bronchoalveolar lavage fluid of smokers supports the hypothesis that release of the protease from activated mast cells is associated with lung destruction in smokers with emphysema. (Celenteron et al. (1988) Chest 94:119-123). In addition, tryptase appears to be a potent mitogen for fibroblasts, suggesting that tryptase may be involved in pulmonary fibrosis and is associated with interstitial lung disease (Rose et al., (1991) J.Clin.Invest.88 :493-499).
哮喘被认为是一种炎症(Hood等人,于1984在Benjamin-Cummings编辑的Immunology第二版中所说的)并经常以气管和支气管对免疫特异性过敏原和一般的化学或物理刺激的过敏反应的进行性发展为特征。这种疾病在其急性和慢性阶段涉及多种生化媒介物。哮喘的支气管内组织的过敏性反应被认为是由慢性炎症反应所引起的,这种炎症反应刺激和损害位于气管壁上的上皮并促进上皮以下组织的病理性增厚。对患有轻度哮喘的病人所进行的支气管活检发现气管壁中有炎症的特征。Asthma is considered an inflammatory condition (Hood et al., 1984 in Immunology, edited by Benjamin-Cummings, 2nd ed.) and is often characterized by hypersensitivity of the trachea and bronchi to immune-specific allergens and general chemical or physical stimuli. The progressive development of the reaction is characteristic. The disease involves multiple biochemical mediators in its acute and chronic phases. Hypersensitivity reactions in the endobronchial tissues of asthma are thought to result from chronic inflammatory responses that irritate and damage the epithelium lining the airway walls and promote pathological thickening of the subepithelial tissue. Bronchial biopsies of patients with mild asthma show signs of inflammation in the airway walls.
对吸入的过敏原的过敏性反应可以引发炎症过程,例如通过与位于细胞表面的IgE结合,过敏原可以活化位于上皮中和平滑肌组织下的肥大细胞和嗜碱性粒细胞。活化的肥大细胞释放一定量的进行炎症反应的执行媒介物或初级化学媒介物(例如组胺),并在原位产生大量的炎症的其它次级媒介物。(例如过氧化物,从媒介物得到的液体等)另外,通过肥大细胞的失粒还释放出几种大分子(例如蛋白多糖、类胰蛋白酶、食糜酶等)。Allergic reactions to inhaled allergens can trigger inflammatory processes, for example by binding to IgE located on the cell surface, allergens can activate mast cells and basophils located in the epithelium and beneath the smooth muscle tissue. Activated mast cells release quantities of executive or primary chemical mediators (such as histamine) that carry out the inflammatory response and generate large quantities of other secondary mediators of inflammation in situ. (eg peroxides, fluid from vehicle, etc.) In addition, several macromolecules are released by mast cell loss (eg proteoglycans, tryptase, chymase, etc.).
这些来自肥大细胞的执行媒介物可能就是造成在对空气中的过敏原的哮喘反应中支气管的早期收缩的原因。哮喘反应的初期在暴露于过敏原后大约15分钟达到峰值,并且一般随之于一两个小时的恢复。25-35%的病人感受到了呼吸功能的进一步衰退,这种衰退在暴露于过敏原后6-12小时后达到最大值。这种后期反应伴随着渗过细支气管组织的炎症细胞(例如嗜余弦细胞、嗜中性细胞、淋巴细胞等)数量的增多。这些渗出的细胞因源于肥大细胞的化学吸引素的释放而被吸引到位,然后在反应的后期被活化。后期的哮喘反应被认为是部分由粒细胞的分泌活性所介导的次级炎症反应。These executive mediators from mast cells may be responsible for the early constriction of the bronchi in the asthmatic response to airborne allergens. The initial phase of the asthmatic response peaks about 15 minutes after exposure to the allergen and is generally followed by an hour or two of recovery. A further decline in respiratory function is experienced by 25-35% of patients, with this decline reaching a maximum 6-12 hours after exposure to the allergen. This late response is accompanied by an increase in the number of inflammatory cells (eg, cosinophils, neutrophils, lymphocytes, etc.) that infiltrate the bronchiolar tissue. These extravasated cells are attracted into place by the release of mast cell-derived chemoattractants and then activated later in the response. The late asthmatic response is thought to be a secondary inflammatory response mediated in part by the secretory activity of granulocytes.
类胰蛋白酶与舒张血管和松弛支气管的神经肽的退化有关(Caughey等人,(1988)J.Pharmacol.Exp.Ther.244:133-137;Franconi等人,(1988)J.Pharmacol.Exp.Ther.248:947-951;和Tam等人(1990)Am.J.Respir.Cell Mol.Biol.3:27-32)及支气管对组胺的反应调控有关(Sekizawa等人,(1989)J.Clin.Invest.83:175-179)。这些发现提示,通过破坏舒张支气管的多肽,类胰蛋白酶可能会增大哮喘中支气管的收缩。类胰蛋白酶切断纤维蛋白原的α-链和高分子量的激肽原,这提示类胰蛋白酶作为一种局部的抗凝血药,发挥着与肝素同样的作用。类胰蛋白酶通过MMP-3活化prostromelysin(pro-MMP-3)和原蚀原酶(pro-MMP-1),这提示类胰蛋白酶与组织发炎和重建及类风湿性关节炎的关节毁坏有关。而且,服用类胰蛋白酶抑制剂避免了过敏原引起的绵羊的后期气管过敏反应的发展(Clark等人,(1995)Am.J.Respir.Crit.CareMed.152:2076-2083),并抑制皮内注射了过敏原的过敏绵羊中皮肤的直接反应(Molinari等人,(1995)Amer.Physiol.Soc.79(6):1966-1970)。所有上述的发现清楚地表明,类胰蛋白酶抑制剂可用作治疗哮喘及与呼吸道炎症有关的其它疾病的治疗药物。Tryptase has been implicated in the degradation of neuropeptides that relax blood vessels and bronchi (Caughey et al., (1988) J. Pharmacol. Exp. Ther. 244:133-137; Franconi et al., (1988) J. Pharmacol. Exp. Ther.248:947-951; related to Tam et al. (1990) Am.J.Respir.Cell Mol.Biol.3:27-32) and regulation of bronchial response to histamine (Sekizawa et al., (1989) J . Clin. Invest. 83:175-179). These findings suggest that tryptase may augment bronchoconstriction in asthma by disrupting bronchodilation polypeptides. Tryptase cleaves the α-chain of fibrinogen and high molecular weight kininogen, suggesting that tryptase acts as a local anticoagulant like heparin. Tryptase activates prostromelysin (pro-MMP-3) and protogenase (pro-MMP-1) through MMP-3, suggesting that tryptase is involved in tissue inflammation and remodeling and joint destruction in rheumatoid arthritis. Moreover, administration of tryptase inhibitors prevented the development of late airway hypersensitivity reactions in sheep induced by allergens (Clark et al., (1995) Am. J. Respir. Crit. Care Med. 152:2076-2083) and inhibited skin Direct skin reactions in allergic sheep intra-injected with allergens (Molinari et al. (1995) Amer. Physiol. Soc. 79(6):1966-1970). All of the above findings clearly indicate that tryptase inhibitors can be used as therapeutic agents in the treatment of asthma and other diseases associated with airway inflammation.
这里通过参考文献引入这些公开文件及本申请中所涉及的其它文献。These publications, as well as other documents referred to in this application, are hereby incorporated by reference.
发明概述Summary of the invention
本申请涉及式Ⅰ的化合物及其药学上可接受的盐、N-氧化物、其药物前体衍生物及其受保护的衍生物: This application relates to compounds of formula I and their pharmaceutically acceptable salts, N-oxides, their prodrug derivatives and their protected derivatives:
ⅠI
其中:in:
X5是C3-14亚环烷基、C3-14亚杂环烷基、C6-14亚芳基或C5-14亚芳杂环基;X 5 is C 3-14 cycloalkylene, C 3-14 heterocycloalkylene, C 6-14 arylene or C 5-14 arylene heterocyclyl;
X4和X6相互独立地为C0-2亚烷基;X 4 and X 6 are independently C 0-2 alkylene;
X1和X9相互独立地为共价键、-C(O)-、-C(O)O-、-OC(O)-、-C(O)N(R3)-、-N(R3)C(O)-、-S(O)2N(R3)-、-N(R3)S(O)2-、-OC(O)N(R3)-、-N(R3)C(O)O-、-N(R3)C(O)N(R3)-或-OC(O)O,其中每个R3独立地为氢、C1-3烷基或C3-8环烷基,条件是:X1和X9不全是共价键;X 1 and X 9 are independently covalent bonds, -C(O)-, -C(O)O-, -OC(O)-, -C(O)N(R 3 )-, -N( R 3 )C(O)-, -S(O) 2 N(R 3 )-, -N(R 3 )S(O) 2 -, -OC(O)N(R 3 )-, -N( R 3 )C(O)O-, -N(R 3 )C(O)N(R 3 )- or -OC(O)O, wherein each R 3 is independently hydrogen, C 1-3 alkyl Or C 3-8 cycloalkyl, provided that: X 1 and X 9 are not all covalent bonds;
X3和X7独立地为-C(O)-、-C(O)O-、-OC(O)-、-C(O)N(R3)-、-N(R3)C(O)-、-S(O)2N(R3)-、-N(R3)S(O)2-、-OC(O)N(R3)-、-N(R3)C(O)O-、-N(R3)C(O)N(R3)-或-OC(O)O-,其中R3如上所定义;X 3 and X 7 are independently -C(O)-, -C(O)O-, -OC(O)-, -C(O)N(R 3 )-, -N(R 3 )C( O)-, -S(O) 2 N(R 3 )-, -N(R 3 )S(O) 2 -, -OC(O)N(R 3 )-, -N(R 3 )C( O)O-, -N(R 3 )C(O)N(R 3 )- or -OC(O)O-, wherein R 3 is as defined above;
X2和X8独立地为C1-8亚烷基、C1-8亚杂烷基、-X10-X11-或-X11-X10-,其中X10是C0-4亚烷基或C3-4亚杂烷基,而X11是C3-8亚环烷基或C3-8亚杂环烷基;X 2 and X 8 are independently C 1-8 alkylene, C 1-8 heteroalkylene, -X 10 -X 11 - or -X 11 -X 10 -, wherein X 10 is C 0-4 alkylene Alkyl or C 3-4 heteroalkylene, and X 11 is C 3-8 cycloalkylene or C 3-8 heterocycloalkylene;
R1是R4-X12-或R3-X13-,其中R 1 is R 4 -X 12 - or R 3 -X 13 -, wherein
R4为氨基、脒基、胍基、1-亚氨基乙基或甲基氨基,R 4 is amino, amidino, guanidino, 1-iminoethyl or methylamino,
X12是C4-6亚烷基、C4-6亚杂烷基、氧代C4-6亚杂烷基、氧代C4-6亚烷基或-X14-X15-X16-,其中X15为C3-6亚环烷基、C5-6亚芳杂环基、C3-6亚杂环烷基或亚苯基,X14为Cn14亚烷基,而X16为Cn16亚烷基,其中n14和n16=0、1、2、3或4,X 12 is C 4-6 alkylene, C 4-6 heteroalkylene, oxo C 4-6 heteroalkylene, oxo C 4-6 alkylene or -X 14 -X 15 -X 16 -, wherein X 15 is C 3-6 cycloalkylene, C 5-6 arylene heterocyclyl, C 3-6 heterocycloalkylene or phenylene, X 14 is C n14 alkylene, and X 16 is C n16 alkylene, wherein n14 and n16=0, 1, 2, 3 or 4,
R5选自氮杂环丁-3-基、苯并咪唑-4-基、苯并咪唑-5-基、咪唑-1-基、咪唑-2-基、咪唑-4-基、2-咪唑啉-2-基、2-咪唑啉-3-基、2-甲基咪唑-1-基、4-甲基咪唑-1-基、5-甲基咪唑-1-基、1-甲基哌啶-3-基、1-甲基哌啶4-基、哌啶-3-基、哌啶-4-基、哌嗪-1-基、哌嗪-2-基、吡啶-3-基、吡啶-4-基、嘧啶-4-基、嘧啶-5-基、吡咯烷-3-基、1,4,5,6-四氢嘧啶-2-基、1,4,5,6-四氢嘧啶-4-基和1,4,5,6-四氢嘧啶-5-基和碳环酮或其硫代酮衍生物,该基团可以是未取代的或者是被一个或多个选自卤素、羟基、巯基、C1-8烷基、C3-14环烷基、C6-14芳基、C6-14芳基C1-4烷基、C1-8烷酰基、C1-8烷基氧基、C6-14芳基氧基、C3-14环烷基氧基、C1-4烷基氧基、C1-8烷基硫基、C3-14环烷基硫基、C6-14芳基硫基和-NR6R7的基团取代,其中R6和R7独立地选自氢、C1-8烷基、C1-8烷酰基、C3-14环烷基或C6-14芳基,而R is selected from the group consisting of azetidin-3-yl, benzimidazol-4-yl, benzimidazol-5-yl , imidazol-1-yl, imidazol-2-yl, imidazol-4-yl, 2-imidazole Lin-2-yl, 2-imidazolin-3-yl, 2-methylimidazol-1-yl, 4-methylimidazol-1-yl, 5-methylimidazol-1-yl, 1-methylpiper Pyridin-3-yl, 1-methylpiperidin-4-yl, piperidin-3-yl, piperidin-4-yl, piperazin-1-yl, piperazin-2-yl, pyridin-3-yl, Pyridin-4-yl, pyrimidin-4-yl, pyrimidin-5-yl, pyrrolidin-3-yl, 1,4,5,6-tetrahydropyrimidin-2-yl, 1,4,5,6-tetrahydropyrimidin-2-yl, 1,4,5,6-tetra Hydropyrimidin-4-yl and 1,4,5,6-tetrahydropyrimidin-5-yl and carbocyclones or their thioketone derivatives, which may be unsubstituted or replaced by one or more From halogen, hydroxyl, mercapto, C 1-8 alkyl, C 3-14 cycloalkyl, C 6-14 aryl, C 6-14 aryl C 1-4 alkyl, C 1-8 alkanoyl, C 1-8 alkyloxy, C 6-14 aryloxy, C 3-14 cycloalkyloxy, C 1-4 alkyloxy, C 1-8 alkylthio, C 3-14 ring Alkylthio, C 6-14 arylthio and -NR 6 R 7 are substituted by groups, wherein R 6 and R 7 are independently selected from hydrogen, C 1-8 alkyl, C 1-8 alkanoyl, C 3-14 cycloalkyl or C 6-14 aryl, while
X13是C0-6亚烷基、C2-6亚杂烷基、氧代C3-6亚杂烷基、氧代C2-6亚烷基或-X17-X18-X19-,其中X18与上述X15的定义相同,X17为Cn17亚烷基,而X19为Cn19亚烷基,其中n17和n19=0、1或2;而X 13 is C 0-6 alkylene, C 2-6 heteroalkylene, oxo C 3-6 heteroalkylene, oxo C 2-6 alkylene or -X 17 -X 18 -X 19 -, wherein X 18 is the same as defined above for X 15 , X 17 is C n17 alkylene, and X 19 is C n19 alkylene, wherein n17 and n19=0, 1 or 2; and
R2为R8-X20-或R9-X21-,其中:R 2 is R 8 -X 20 - or R 9 -X 21 -, wherein:
R8为氨基、1-亚氨基乙基或甲基氨基,R 8 is amino, 1-iminoethyl or methylamino,
X20为C4-6亚烷基、C4-6亚杂烷基、氧代C4-6亚杂烷基、氧代C4-6亚烷基或-X22-X23-X24-,其中X23与上述X15的定义相同,X22为Cn22亚烷基,而X24为Cn24亚烷基,其中n22和n24=0、1、2、3或4,条件是:当R8是氨基时,X20不是C4-6亚烷基或氧代C4-6亚烷基,而n22不是1、2、3或4,X 20 is C 4-6 alkylene, C 4-6 heteroalkylene, oxo C 4-6 heteroalkylene, oxo C 4-6 alkylene or -X 22 -X 23 -X 24 -, wherein X23 is as defined above for X15 , X22 is Cn22 alkylene, and X24 is Cn24 alkylene, wherein n22 and n24=0, 1, 2, 3 or 4, provided that: When R 8 is amino, X 20 is not C 4-6 alkylene or oxo C 4-6 alkylene, and n 22 is not 1, 2, 3 or 4,
R9与上述R5的定义相同,而 R9 is as defined above for R5 , and
X21是C0-6亚烷基、C2-6亚杂烷基、氧代C3-6亚杂烷基、氧代C2-6亚烷基或-X25-X26-X27-,其中X26与上述X15的定义相同,X25为Cn25亚烷基,而X27为Cn27亚烷基,其中n25和n27=0、1或2;X 21 is C 0-6 alkylene, C 2-6 heteroalkylene, oxo C 3-6 heteroalkylene, oxo C 2-6 alkylene or -X 25 -X 26 -X 27 -, wherein X 26 is the same as defined above for X 15 , X 25 is C n25 alkylene, and X 27 is C n27 alkylene, wherein n25 and n27=0, 1 or 2;
其中每个上述定义的亚烷基、亚环烷基、亚杂烷基、亚杂环烷基、亚苯基、亚芳基和亚杂芳基可以是未取代的或者被一个或多个选自卤素、羟基、巯基、C1-8烷基、C3-14环烷基、C6-14芳基、C6-14芳基C1-4烷基、C1-8烷酰基、C1-8烷基氧基、C6-14芳基氧基、C3-14环烷基氧基、C1-4烷基氧基、C1-8烷基硫基、C3-14环烷基硫基、C6-14芳基硫基和-NR6R7的基团所取代,其中R6和R7如上所定义,条件是:在R1、X2、X4、X6、X8和R2中所含的杂原子与X3、X5、X7和X9中所含的杂原子之间没有共价键。wherein each of the above-defined alkylene, cycloalkylene, heteroalkylene, heterocycloalkylene, phenylene, arylene and heteroarylene groups may be unsubstituted or replaced by one or more selected From halogen, hydroxyl, mercapto, C 1-8 alkyl, C 3-14 cycloalkyl, C 6-14 aryl, C 6-14 aryl C 1-4 alkyl, C 1-8 alkanoyl, C 1-8 alkyloxy, C 6-14 aryloxy, C 3-14 cycloalkyloxy, C 1-4 alkyloxy, C 1-8 alkylthio, C 3-14 ring Substituted by alkylthio, C 6-14 arylthio and -NR 6 R 7 , wherein R 6 and R 7 are as defined above, provided that: at R 1 , X 2 , X 4 , X 6 There is no covalent bond between the heteroatoms contained in , X 8 and R 2 and the heteroatoms contained in X 3 , X 5 , X 7 and X 9 .
本发明的第二个方面涉及式Ⅰ的化合物及其药学上可接受的盐、N-氧化物、其药物前体衍生物及其受保护的衍生物:ⅠA second aspect of the present invention relates to compounds of formula I and their pharmaceutically acceptable salts, N-oxides, prodrug derivatives and protected derivatives thereof: I
其中:in:
X4-X5-X6一起为C2-12亚烷基或C3-12亚杂烷基;X 4 -X 5 -X 6 together are C 2-12 alkylene or C 3-12 heteroalkylene;
X1和X9相互独立地为共价键、-C(O)-、-C(O)O-、-OC(O)-、-C(O)N(R3)-、-N(R3)C(O)-、-S(O)2N(R3)-、-N(R3)S(O)2-、-OC(O)N(R3)-、-N(R3)C(O)O-、-N(R3)C(O)N(R3)-或-OC(O)O-,其中每个R3独立地为氢、C1-3烷基或C3-8环烷基,条件是:X1和X9不全是共价键;X 1 and X 9 are independently covalent bonds, -C(O)-, -C(O)O-, -OC(O)-, -C(O)N(R 3 )-, -N( R 3 )C(O)-, -S(O) 2 N(R 3 )-, -N(R 3 )S(O) 2 -, -OC(O)N(R 3 )-, -N( R 3 )C(O)O-, -N(R 3 )C(O)N(R 3 )- or -OC(O)O-, wherein each R 3 is independently hydrogen, C 1-3 alkane Group or C 3-8 cycloalkyl, provided that: X 1 and X 9 are not all covalent bonds;
X3和X7独立地为-C(O)-、-C(O)O、-OC(O)-、-C(O)N(R3)-、-N(R3)C(O)-、-S(O)2N(R3)-、-N(R3)S(O)2-、-OC(O)N(R3)-、-N(R3)C(O)O-、-N(R3)C(O)N(R3)-或-OC(O)O-,其中R3如上所定义;X 3 and X 7 are independently -C(O)-, -C(O)O, -OC(O)-, -C(O)N(R 3 )-, -N(R 3 )C(O )-, -S(O) 2 N(R 3 )-, -N(R 3 )S(O) 2 -, -OC(O)N(R 3 )-, -N(R 3 )C(O )O-, -N(R 3 )C(O)N(R 3 )-or -OC(O)O-, wherein R 3 is as defined above;
X2和X8独立地为C1-8亚烷基、C1-8亚杂烷基、-X10-X11-或-X11-X10-,其中X10是C0-4亚烷基或C3-4亚杂烷基,而X11是C3-8亚环烷基或C3-8亚杂环烷基;X 2 and X 8 are independently C 1-8 alkylene, C 1-8 heteroalkylene, -X 10 -X 11 - or -X 11 -X 10 -, wherein X 10 is C 0-4 alkylene Alkyl or C 3-4 heteroalkylene, and X 11 is C 3-8 cycloalkylene or C 3-8 heterocycloalkylene;
R1是R4-X12-或R5-X13-,其中R 1 is R 4 -X 12 - or R 5 -X 13 -, wherein
R4为氨基、脒基、胍基、1-亚氨基乙基或甲基氨基,R 4 is amino, amidino, guanidino, 1-iminoethyl or methylamino,
X12是C4-6亚烷基、C4-6亚杂烷基、氧代C4-6亚杂烷基、氧代C4-6亚烷基或-X14-X15-X16-,其中X15为C3-6亚环烷基、C5-6亚芳杂环基、C3-6亚杂环烷基或亚苯基,X14为Cn14亚烷基,而X16为Cn16亚烷基,其中n14和n16=0、1、2、3或4,X 12 is C 4-6 alkylene, C 4-6 heteroalkylene, oxo C 4-6 heteroalkylene, oxo C 4-6 alkylene or -X 14 -X 15 -X 16 -, wherein X 15 is C 3-6 cycloalkylene, C 5-6 arylene heterocyclyl, C 3-6 heterocycloalkylene or phenylene, X 14 is C n14 alkylene, and X 16 is C n16 alkylene, wherein n14 and n16=0, 1, 2, 3 or 4,
R5选自氮杂环丁-3-基、苯并咪唑-4-基、苯并咪唑-5-基、咪唑-1-基、咪唑-2-基、咪唑-4-基、2-咪唑啉-2-基、2-咪唑啉-3-基、2-甲基咪唑-1-基、4-甲基咪唑-1-基、5-甲基咪唑-1-基、1-甲基哌啶-3-基、1-甲基哌啶-4-基、哌啶-3-基、哌啶-4-基、哌嗪-1-基、哌嗪-2-基、吡啶-3-基、吡啶-4-基、嘧啶-4-基、嘧啶-5-基、吡咯烷-3-基、1,4,5,6-四氢嘧啶-2-基、1,4,5,6-四氢嘧啶-4-基和1,4,5,6-四氢嘧啶-5-基和碳环酮或其硫代酮衍生物,该基团可以是未取代的或者是被一个或多个选自卤素、羟基、巯基、C1-8烷基、C3-14环烷基、C6-14芳基、C6-14芳基C1-4烷基、C1-8烷酰基、C1-8烷基氧基、C6-14芳基氧基、C3-14环烷基氧基、C1-4烷基氧基、C1-8烷基硫基、C3-14环烷基硫基、C6-14芳基硫基和-NR6R7的基团取代,其中R6和R7独立地选自氢、C1-8烷基、C1-8烷酰基、C3-14环烷基或C6-14芳基,而R is selected from the group consisting of azetidin-3-yl, benzimidazol-4-yl, benzimidazol-5-yl , imidazol-1-yl, imidazol-2-yl, imidazol-4-yl, 2-imidazole Lin-2-yl, 2-imidazolin-3-yl, 2-methylimidazol-1-yl, 4-methylimidazol-1-yl, 5-methylimidazol-1-yl, 1-methylpiper Pyridin-3-yl, 1-methylpiperidin-4-yl, piperidin-3-yl, piperidin-4-yl, piperazin-1-yl, piperazin-2-yl, pyridin-3-yl , pyridin-4-yl, pyrimidin-4-yl, pyrimidin-5-yl, pyrrolidin-3-yl, 1,4,5,6-tetrahydropyrimidin-2-yl, 1,4,5,6- Tetrahydropyrimidin-4-yl and 1,4,5,6-tetrahydropyrimidin-5-yl and carbocyclones or their thioketone derivatives, which may be unsubstituted or replaced by one or more Selected from halogen, hydroxyl, mercapto, C 1-8 alkyl, C 3-14 cycloalkyl, C 6-14 aryl, C 6-14 aryl C 1-4 alkyl, C 1-8 alkanoyl, C 1-8 alkyloxy, C 6-14 aryloxy, C 3-14 cycloalkyloxy, C 1-4 alkyloxy, C 1-8 alkylthio, C 3-14 Cycloalkylthio, C 6-14 arylthio and -NR 6 R 7 are substituted by groups, wherein R 6 and R 7 are independently selected from hydrogen, C 1-8 alkyl, C 1-8 alkanoyl , C 3-14 cycloalkyl or C 6-14 aryl, and
X13是C0-6亚烷基、C2-6亚杂烷基、氧代C3-6亚杂烷基、氧代C2-6亚烷基或-X17-X18-X19-,其中X18与上述X15的定义相同,X17为Cn17亚烷基,而X19为Cn19亚烷基,其中n17和n19=0、1或2;而X 13 is C 0-6 alkylene, C 2-6 heteroalkylene, oxo C 3-6 heteroalkylene, oxo C 2-6 alkylene or -X 17 -X 18 -X 19 -, wherein X 18 is the same as defined above for X 15 , X 17 is C n17 alkylene, and X 19 is C n19 alkylene, wherein n17 and n19=0, 1 or 2; and
R2为R8-X20-或R9-X21-,其中:R 2 is R 8 -X 20 - or R 9 -X 21 -, wherein:
R8与上述R4的定义相同,R 8 is as defined above for R 4 ,
X20为C4-6亚烷基、C4-6亚杂烷基、氧代C4-6亚杂烷基、氧代C4-6亚烷基或-X22-X23-X24-,其中X23与上述X15的定义相同,X22为Cn22亚烷基,而X24为Cn24亚烷基,其中n22和n24=0、1、2、-3或4,X 20 is C 4-6 alkylene, C 4-6 heteroalkylene, oxo C 4-6 heteroalkylene, oxo C 4-6 alkylene or -X 22 -X 23 -X 24 -, wherein X 23 is the same as defined above for X 15 , X 22 is C n22 alkylene, and X 24 is C n24 alkylene, wherein n22 and n24=0, 1, 2, -3 or 4,
R9与上述R5的定义相同,而 R9 is as defined above for R5 , and
X21是C0-6亚烷基、C2-6亚杂烷基、氧代C3-6亚杂烷基、氧代C2-6亚烷基或-X25-X26-X27-,其中X26与上述X15的定义相同,X25为Cn25亚烷基,而X27为Cn27亚烷基,其中n25和n27=0、1或2;X 21 is C 0-6 alkylene, C 2-6 heteroalkylene, oxo C 3-6 heteroalkylene, oxo C 2-6 alkylene or -X 25 -X 26 -X 27 -, wherein X 26 is the same as defined above for X 15 , X 25 is C n25 alkylene, and X 27 is C n27 alkylene, wherein n25 and n27=0, 1 or 2;
其中每个上述定义的亚烷基、亚环烷基、亚杂烷基、亚杂环烷基、亚苯基、亚芳基和亚杂芳基可以是未取代的或者被一个或多个选自卤素、羟基、巯基、C1-8烷基、C3-14环烷基、C6-14芳基、C6-14芳基C1-4烷基、C1-8烷酰基、C1-8烷基氧基、C6-14芳基氧基、C3-14环烷基氧基、C1-4烷基氧基、C1-8烷基硫基、C3-14环烷基硫基、C6-14芳基硫基和-NR6R7的基团所取代,其中R6和R7如上所定义,条件是:在R1、X2、X4、X6、X8和R2中所含的杂原子与X3、X5、X7和X9中所含的杂原子之间没有共价键。wherein each of the above-defined alkylene, cycloalkylene, heteroalkylene, heterocycloalkylene, phenylene, arylene and heteroarylene groups may be unsubstituted or replaced by one or more selected From halogen, hydroxyl, mercapto, C 1-8 alkyl, C 3-14 cycloalkyl, C 6-14 aryl, C 6-14 aryl C 1-4 alkyl, C 1-8 alkanoyl, C 1-8 alkyloxy, C 6-14 aryloxy, C 3-14 cycloalkyloxy, C 1-4 alkyloxy, C 1-8 alkylthio, C 3-14 ring Substituted by alkylthio, C 6-14 arylthio and -NR 6 R 7 , wherein R 6 and R 7 are as defined above, provided that: at R 1 , X 2 , X 4 , X 6 There is no covalent bond between the heteroatoms contained in , X 8 and R 2 and the heteroatoms contained in X 3 , X 5 , X 7 and X 9 .
本发明的第三个方面是一种含有式1的化合物或其药学上可接受的盐、N-氧化物、其药物前体衍生物与一种或多种合适的赋形剂的混合物的药物组合物。A third aspect of the present invention is a medicament containing a compound of formula 1 or a pharmaceutically acceptable salt thereof, an N-oxide, a prodrug derivative thereof and one or more suitable excipients combination.
本发明的第四个方面是一种治疗其发病机理和/或症状与类胰蛋白酶活性有关的动物疾病的方法,该方法包括以治疗有效量的式Ⅰ的化合物或其药学上可接受的盐、N-氧化物、其药物前体衍生物向该动物给药。The fourth aspect of the present invention is a method of treating an animal disease whose pathogenesis and/or symptoms are related to tryptase activity, which method comprises a therapeutically effective amount of a compound of formula I or a pharmaceutically acceptable salt thereof , N-oxide, prodrug derivative thereof is administered to the animal.
本发明的第五个方面是制备在“本发明的详细描述”中所提出的式Ⅰ的化合物及其药学上可接受的盐、N-氧化物、其药物前体衍生物及其受保护的衍生物的方法。The fifth aspect of the present invention is to prepare the compound of formula I and its pharmaceutically acceptable salt, N-oxide, its prodrug derivative and its protected Derivative method.
本发明的详细描述Detailed description of the invention
定义:definition:
除非特别声明,说明书和权利要求书中所使用的下列术语具有下列意义:Unless otherwise stated, the following terms used in the specification and claims have the following meanings:
“烷酰基”是指-C(O)R基,其中R为下列所定义的总共含有指定数目的碳原子的烷基(例如C1-8烷酰基包括甲酰基、乙酰基、丙酰基、丁酰基、异丁酰基、巴豆酰基、异巴豆酰基等)。"Alkanoyl" means a -C(O)R group, wherein R is an alkyl group as defined below containing a total of the indicated number of carbon atoms (e.g., C 1-8 alkanoyl includes formyl, acetyl, propionyl, butyl acyl, isobutyryl, crotonyl, isocrotonyl, etc.).
“烷基”(例如在烷基、芳基烷基、烷基氧基、烷基硫基中的烷基)是指含有指定数目的碳原子的直链或支链的、饱和或不饱和烃基(例如C1-8烷基包括甲基、乙基、丙基、异丙基、丁基、仲丁基、异丁基、叔丁基、乙烯基、烯丙基、1-丙烯基、异丙烯基、1-丁烯基、2-丁烯基、3-丁烯基、2-甲基烯丙基、乙炔基、1-丙炔基、2-丙炔基等)。"Alkyl" (eg, alkyl as in alkyl, arylalkyl, alkyloxy, alkylthio) means a straight or branched, saturated or unsaturated hydrocarbon group containing the indicated number of carbon atoms (For example, C 1-8 alkyl includes methyl, ethyl, propyl, isopropyl, butyl, sec-butyl, isobutyl, tert-butyl, vinyl, allyl, 1-propenyl, iso propenyl, 1-butenyl, 2-butenyl, 3-butenyl, 2-methallyl, ethynyl, 1-propynyl, 2-propynyl, etc.).
“亚烷基”是指含有指定数目的碳原子的直链的饱和或不饱和二价烃基(例如C0-6亚烷基包括亚甲基(-CH2-)、亚乙基(-(-CH2-)2-)、亚乙烯基(-CH=CH-)、亚乙炔基(-C≡C-)、2-亚丙烯基(-CH=CH-CH2-)、1-亚丙烯基(-CH2-CH=CH-)、四亚甲基(-(-CH2-)4-)、五亚甲基(-(-CH2-)5-)和六亚甲基(-(-CH2-)6-)等)。术语C0亚烷基是指代表一个共价键。"Alkylene" refers to a straight-chain saturated or unsaturated divalent hydrocarbon group containing the specified number of carbon atoms (for example, C 0-6 alkylene includes methylene (-CH 2 -), ethylene (-( -CH 2 -) 2 -), ethenylene (-CH=CH-), ethynylene (-C≡C-), 2-propenylene (-CH=CH-CH 2 -), 1- propenyl (-CH 2 -CH=CH-), tetramethylene (-(-CH 2 -) 4 -), pentamethylene (-(-CH 2 -) 5 -) and hexamethylene ( -(-CH 2 -) 6 -), etc.). The term C 0 alkylene is meant to represent a covalent bond.
“烷氧基”是指-OR基,其中R为如上所定义的含有指定数目的碳原子的烷基(例如C1-8烷氧基包括甲氧基、乙氧基、丙氧基、异丙氧基、丁氧基、异丁氧基等)。"Alkoxy" means an -OR group, wherein R is an alkyl group as defined above containing the indicated number of carbon atoms (e.g., C 1-8 alkoxy includes methoxy, ethoxy, propoxy, iso propoxy, butoxy, isobutoxy, etc.).
“烷基硫基”是指-SR基,其中R为如上所定义的含有指定数目的碳原子的烷基(例如C1-8烷基硫基包括甲基硫基、乙基硫基、丙基硫基、异丙基硫基、丁基硫基、异丁基硫基等)。"Alkylthio" means a -SR radical, where R is an alkyl group as defined above containing the indicated number of carbon atoms (e.g., C 1-8 alkylthio includes methylthio, ethylthio, propylthio, thiol, isopropylthio, butylthio, isobutylthio, etc.).
“动物”包括人类、非人类哺乳动物(例如狗、猫、兔子、牛、马、绵羊、山羊、猪、鹿等)和非哺乳动物(例如鸟类等)。"Animal" includes humans, non-human mammals (eg, dogs, cats, rabbits, cows, horses, sheep, goats, pigs, deer, etc.) and non-mammals (eg, birds, etc.).
“芳基”(例如在芳基、芳基烷基、芳基氧基和芳基硫基中的芳基)是指含有指定数目的碳原子的单环或多环芳香烃基,其中含有自由价的碳原子是芳香环及任何碳环酮或其硫代酮衍生物的一员(例如C6-14芳基包括苯基、萘基、蒽基、菲基、1,2,3,4-四氢萘-5-基、1-氧代-1,2-二氢萘-6-基、1-硫代-1,2-二氢萘-7-基等)。"Aryl" (as in aryl, arylalkyl, aryloxy, and arylthio) means a monocyclic or polycyclic aromatic hydrocarbon radical containing the indicated number of carbon atoms in which the free valence The carbon atom is a member of the aromatic ring and any carbocyclic ketone or its thioketone derivatives (for example, C 6-14 aryl includes phenyl, naphthyl, anthracenyl, phenanthrenyl, 1,2,3,4- tetrahydronaphthalen-5-yl, 1-oxo-1,2-dihydronaphthalen-6-yl, 1-thio-1,2-dihydronaphthalen-7-yl, etc.).
“亚芳基”是指含有指定数目的碳原子的二价单环或多环芳香烃基,其中含有自由价的碳原子是芳香环及任何碳环酮或其硫代酮衍生物的成员(例如C6-14亚芳基包括1,4-亚苯基、1,3-亚苯基、1,4-亚萘基、2,6-亚萘基、1,4-亚蒽基、2,6-亚蒽基、1,6-亚菲基、1,2,3,4-四氢-5,8-亚萘基、1-氧代-1,2-二氢-5,7-亚萘基、1-硫代-1,2-二氢-5,8-亚萘基等)。"Arylene" means a divalent monocyclic or polycyclic aromatic hydrocarbon radical containing the specified number of carbon atoms, wherein the carbon atoms containing free valences are members of the aromatic ring and any carbocyclic ketone or its thioketone derivatives (e.g. C 6-14 arylene includes 1,4-phenylene, 1,3-phenylene, 1,4-naphthylene, 2,6-naphthylene, 1,4-anthracene, 2, 6-Anthracene, 1,6-phenanthrene, 1,2,3,4-tetrahydro-5,8-naphthylene, 1-oxo-1,2-dihydro-5,7- naphthyl, 1-thio-1,2-dihydro-5,8-naphthylene, etc.).
“芳基氧基”是指-OR基,其中R为如上所定义的含有指定数目的碳原子的芳基(例如C6-14芳基氧基包括苯氧基、萘基氧基、蒽基氧基等)。"Aryloxy" means an -OR radical, wherein R is an aryl group as defined above containing the indicated number of carbon atoms (eg C 6-14 aryloxy includes phenoxy, naphthyloxy, anthracenyl oxygen, etc.).
“芳基硫基”是指-SR基,其中R为如上所定义的含有指定数目的碳原子的芳基(例如C6-14芳基硫基包括苯基硫基、萘基硫基、蒽基硫基等)。"Arylthio" means a -SR radical, where R is an aryl group as defined above containing the indicated number of carbon atoms (eg C 6-14 arylthio includes phenylthio, naphthylthio, anthracene thiol, etc.).
“环烷基”(例如在环烷基和环烷基氧基中的环烷基)是指含有指定数目的碳原子的饱和或不饱和单环或多环烃基,其中含有自由价的碳原子是非芳香环及任何碳环酮或其硫代酮衍生物的一员(例如C3-14环烷基包括环丙基、环丁基、环戊基、环己基、环己烯基、2,5-环己二烯基、双环[2.2.2]辛基、1,2,3,4-四氢萘-1-基、氧代环己基、二氧代环己基、硫代环己基等)。"Cycloalkyl" (as in cycloalkyl and cycloalkyloxy) means a saturated or unsaturated monocyclic or polycyclic hydrocarbon radical containing the indicated number of carbon atoms, wherein the free valence of carbon atoms Is a non-aromatic ring and a member of any carbocyclic ketone or its thioketone derivatives (for example, C 3-14 cycloalkyl includes cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclohexenyl, 2, 5-cyclohexadienyl, bicyclo[2.2.2]octyl, 1,2,3,4-tetrahydronaphthalene-1-yl, oxocyclohexyl, dioxocyclohexyl, thiocyclohexyl, etc.) .
“亚环烷基”是指含有指定数目的碳原子的二价饱和或不饱和单环或多环基团,其中含有自由价的碳原子是非芳香环及任何碳环酮或其硫代酮衍生物的成员(例如C3-6亚环烷基包括1,2-亚环丙基、1,2-亚环丁基、1,3-亚环丁基、1,2-亚环戊基、1,3-亚环戊基、1,4-亚环戊基、1,4-亚环己基、3-环己烯-1,2-亚基、2,5-环己二烯-1,4-亚基、1,4-双环[2.2.2]亚环辛基、1,2,3,4-四氢-1,4-亚萘基、5-氧代-1,3-亚环己基、2,5-二氧代-1,4-亚环己基、5-硫代-1,4-亚环己基等)。"Cycloalkylene" means a divalent saturated or unsaturated monocyclic or polycyclic group containing the specified number of carbon atoms, wherein the carbon atoms containing free valences are non-aromatic rings and any carbocyclic ketone or its thioketone derivative (for example, C 3-6 cycloalkylene includes 1,2-cyclopropylene, 1,2-cyclobutylene, 1,3-cyclobutylene, 1,2-cyclopentylene, 1,3-cyclopentylene, 1,4-cyclopentylene, 1,4-cyclohexylene, 3-cyclohexene-1,2-ylidene, 2,5-cyclohexadiene-1, 4-Ylidene, 1,4-bicyclo[2.2.2]cyclooctylene, 1,2,3,4-tetrahydro-1,4-naphthylidene, 5-oxo-1,3-cycloethylene Hexyl, 2,5-dioxo-1,4-cyclohexylene, 5-thio-1,4-cyclohexylene, etc.).
“环烷基氧基”是指-OR基,其中R为如上所定义的含有指定数目的碳原子的环烷基(例如C3-14环烷基氧基包括环丙基氧基、环丁基氧基、环戊基氧基、环己基氧基等)。"Cycloalkyloxy" means an -OR group, wherein R is a cycloalkyl group as defined above containing the indicated number of carbon atoms (eg C 3-14 cycloalkyloxy includes cyclopropyloxy, cyclobutyl oxy, cyclopentyloxy, cyclohexyloxy, etc.).
“环烷基硫基”是指-SR基,其中R为如上所定义的含有指定数目的碳原子的环烷基(例如C3-14环烷基硫基包括环丙基硫基、环丁基硫基、环戊基硫基、环己基硫基等)。"Cycloalkylthio" means a -SR group, wherein R is a cycloalkyl group as defined above containing the indicated number of carbon atoms (for example C 3-14 cycloalkylthio includes cyclopropylthio, cyclobutyl thiol, cyclopentylthio, cyclohexylthio, etc.).
“去保护”是指选择性反应完成后除去保护基团。"Deprotection" refers to the removal of a protecting group after the selective reaction is complete.
“疾病”具体地包括动物或其肢体的不健康的身体状况,包括可以是因对动物进行医学治疗或兽医治疗而引起或偶然发生的不健康的身体状况即这种治疗的副作用。"Disease" specifically includes an unhealthy physical condition of an animal or its limbs, including any unhealthy physical condition that may result from or occur incidentally to medical or veterinary treatment of an animal, a side effect of such treatment.
“卤素”是指氟、氯、溴或碘。"Halogen" means fluorine, chlorine, bromine or iodine.
“杂代亚烷基”是指在如上所定义的亚烷基中,1-5个指定的碳原子被选自N、O或S的杂原子所取代(例如分别为氮杂亚烷基、氧杂亚烷基和硫杂亚烷基),条件是:其中所含的O、N和S原子不与其它杂原子形成键。例如C3-12亚杂烷基包括氮杂C3-亚烷基,其中包括3-氮杂三亚甲基(-NHCH2CH2-)、2-氮杂三亚甲基(-CH2NHCH2-)等;ω-氮杂-C2-5-亚烷基,其中包括2-氮杂亚乙基(-NHCH2-)、3-氮杂三亚甲基、4-氮杂四亚甲基(-NHCH2CH2CH2-)和5-氮杂五亚甲基(-NHCH2CH2CH2CH2-);氧杂C3-亚烷基,其中包括3-氧杂三亚甲基(-OCH2CH2-)、2-氧杂三亚甲基(-CH2OCH2-)等;氧杂-C5-亚烷基如3-氧杂五亚甲基(-CH2CH2OCH2CH2-)等;硫杂C3-亚烷基,其中包括3-硫杂三亚甲基(-SCH2CH2-)、2-硫杂三亚甲基(-CH2SCH2-)等;ω-硫杂-C2-4-亚烷基,其中包括2-硫杂亚乙基(-SCH2-)、3-硫杂三亚甲基和4-硫杂四亚甲基(-SCH2CH2CH2-);二氮杂C6亚烷基,其中包括2,5-二氮杂六亚甲基(-CH2NHCH2CH2NHCH2-);氮杂氧杂C6亚烷基,其中包括2-氧杂-5-氮杂六亚甲基(-CH2OCH2CH2NHCH2-)等。"Heteroalkylene" means that in an alkylene group as defined above, 1-5 specified carbon atoms are replaced by heteroatoms selected from N, O or S (such as azaalkylene, azaalkylene, respectively oxaalkylene and thiaalkylene) with the proviso that the O, N and S atoms contained therein do not form bonds with other heteroatoms. For example, C 3-12 heteroalkylene includes aza C 3 -alkylene, including 3-azatrimethylene (-NHCH 2 CH 2 -), 2-azatrimethylene (-CH 2 NHCH 2 -), etc.; ω-aza-C 2-5 -alkylene, including 2-azaethylene (-NHCH 2 -), 3-azatrimethylene, 4-azatetramethylene (-NHCH 2 CH 2 CH 2 -) and 5-azapentamethylene (-NHCH 2 CH 2 CH 2 CH 2 -); oxaC 3 -alkylene, including 3-oxatrimethylene (-OCH 2 CH 2 -), 2-oxa trimethylene (-CH 2 OCH 2 -), etc.; oxa-C 5 -alkylene such as 3-oxa pentamethylene (-CH 2 CH 2 OCH 2 CH 2 -), etc.; thia C 3 -alkylene, including 3-thiatrimethylene (-SCH 2 CH 2 -), 2-thiatrimethylene (-CH 2 SCH 2 -) etc.; ω-thia-C 2-4 -alkylene, including 2-thiaethylene (-SCH 2 -), 3-thiatrimethylene and 4-thiatetramethylene (- SCH 2 CH 2 CH 2 -); Diaza C 6 alkylene, including 2,5-diazahexamethylene (-CH 2 NHCH 2 CH 2 NHCH 2 -); Azaoxa C 6 Alkylene groups include 2-oxa-5-azahexamethylene (-CH 2 OCH 2 CH 2 NHCH 2 -) and the like.
“亚杂芳基”是指在如上所定义的亚芳基中,1-5个指定的碳原子被选自N、O或S的杂原子所取代(例如C5-6亚杂芳基,包括亚呋喃基、亚噻吩基、亚吡咯基、亚咪唑基、亚吡啶基等)。"Heteroarylene" means that in an arylene group as defined above, 1-5 designated carbon atoms are replaced by heteroatoms selected from N, O or S (for example, C 5-6 heteroarylene, Including furylylene, thienylene, pyrrolylene, imidazolylidene, pyridinylene, etc.).
“亚杂环烷基”是指在如上所定义的亚环烷基中,1-5个指定的碳原子被选自N、O或S的杂原子所取代(例如C3-14亚杂环烷基包括2,4-亚吡咯烷基、2,4-亚吡咯啉基、2,4-亚咪唑啉基、3,5-亚吡唑啉基、1,4-亚哌啶基、1,4-亚哌嗪基、2,5-亚喹宁环基、2,5-亚吗啉基、1,3-亚异二氢吲哚基等)。"Heterocycloalkylene" means that in a cycloalkylene group as defined above, 1-5 specified carbon atoms are replaced by heteroatoms selected from N, O or S (for example, C 3-14 heterocycloalkylene Alkyl includes 2,4-pyrrolidinylidene, 2,4-pyrrolinylidene, 2,4-imidazolinylidene, 3,5-pyrazolinylidene, 1,4-piperidinylidene, 1 , 4-piperazinylene, 2,5-quininidine, 2,5-morpholinylene, 1,3-isoindolinylene, etc.).
“氧代亚杂烷基”是指在如上所定义的亚烷基中,1个指定的碳原子被选自N、O或S的杂原子所取代,而且与杂原子相邻的一个碳原子被一个羰基(C=O)所取代,例如分别为氧代氮杂亚烷基、氧代氧杂亚烷基和氧代硫杂亚烷基,条件是:其中所含的O、N和S原子不与其它杂原子形成键。例如,氧代C4-6亚杂代烷基包括氧代氮杂C3亚烷基,其中包括2-氮杂-3-氧代三亚甲基(-C(O)NHCH2-)、3-氮杂-2-氧代三亚甲基(-NHC(O)CH2-)等;氧代氧杂C3亚烷基,其中包括2-氧杂-3-氧代三亚甲基(-C(O)OCH2-)、3-氧杂-2-氧代三亚甲基(-OC(O)CH2-)等;和硫杂氧代C3亚烷基,其中包括2-硫杂-3-氧代三亚甲基(-C(O)SCH2-)、3-硫杂-2-氧代三亚甲基(-SC(O)CH2-)等。"Oxoheteroalkylene" means that in an alkylene group as defined above, one of the specified carbon atoms is replaced by a heteroatom selected from N, O or S, and a carbon atom adjacent to the heteroatom Substituted by a carbonyl group (C=O), such as oxoazaalkylene, oxooxaalkylene and oxothiaalkylene, respectively, provided that O, N and S contained therein Atoms do not form bonds with other heteroatoms. For example, oxoC 4-6 heteroalkylene includes oxoazepine C 3 alkylene, including 2-aza-3-oxotrimethylene (-C(O)NHCH 2 -), 3 -Aza-2-oxo-trimethylene (-NHC(O)CH 2 -), etc.; oxo-oxa C 3 alkylene, including 2-oxa-3-oxo-trimethylene (-C (O)OCH 2 -), 3-oxa-2-oxotrimethylene (-OC(O)CH 2 -), etc.; and thiaxo C 3 alkylene, including 2-thia- 3-oxotrimethylene (-C(O)SCH 2 -), 3-thia-2-oxotrimethylene (-SC(O)CH 2 -), and the like.
“离去基团”具有通常的合成有机化学中与其相关的意义,即在烷基化条件下可以被取代的原子或基团,包括卤素、羟基、烷基磺酰氧基(例如甲磺酰氧基、乙磺酰氧基等)、芳基磺酰氧基(例如苯磺酰氧基和对甲苯磺酰氧基、噻吩基氧基)、二卤代膦酰氧基、四卤代磷酰氧基等。"Leaving group" has the usual meaning associated with it in synthetic organic chemistry, that is, an atom or group that can be substituted under alkylation conditions, including halogen, hydroxyl, alkylsulfonyloxy (such as methylsulfonyl oxy, ethanesulfonyloxy, etc.), arylsulfonyloxy (such as benzenesulfonyloxy and p-toluenesulfonyloxy, thienyloxy), dihalophosphonyloxy, tetrahalophosphoryl Acyloxy, etc.
“选择性的”或“选择性地”是指在其后面所描述的事件或条件可以发生或不发生,而且该描述包括其中事件或条件发生的情况和其中事件或条件不发生的情况。例如术语“被一个或多个基团选择性地取代的”意思是所指定的基团可能被取代或可能不被取代,以使它落在本发明的范围内。"Optional" or "optionally" means that the event or condition described thereafter can or cannot occur, and that the description includes instances where the event or condition occurs and instances where it does not. For example, the term "optionally substituted with one or more groups" means that the specified group may or may not be substituted such that it falls within the scope of the invention.
“药学上可接受的N-氧化物”是指下列定义的药学上可接受的并具有所要的药理活性的、其中的氮原子处于氧化状态的化合物。式Ⅰ化合物的N-氧化物可以被本领域的普通技术人员用已知的方法制备。"Pharmaceutically acceptable N-oxide" means a compound defined below which is pharmaceutically acceptable and possesses the desired pharmacological activity, wherein the nitrogen atom is in an oxidized state. N-oxides of compounds of formula I can be prepared by methods known to those skilled in the art.
“氧代亚烷基”是指在如上所定义的亚烷基中,1个指定的碳原子被一个羰基(C=O)所取代,例如氧代C3亚烷基,包括3-氧代三亚甲基(-C(O)CH2CH2-)等。"Oxyalkylene" means that in an alkylene group as defined above, one of the specified carbon atoms is replaced by a carbonyl (C=O), such as oxo C 3 alkylene, including 3-oxo Trimethylene (-C(O)CH 2 CH 2 -) etc.
疾病的“病理学”是指疾病的基本性质、病因和发展以及发病过程中产生的结构和功能变化。The "pathology" of a disease refers to the basic nature, etiology and development of the disease, as well as the structural and functional changes that occur during the pathogenesis.
“药学上可接受的”是指可用于制备药物组合物的、一般是安全的、无毒的、没有生物学上的或其它的副作用,包括兽医用及人类药用时可以接受的。"Pharmaceutically acceptable" refers to those that can be used to prepare pharmaceutical compositions, are generally safe, non-toxic, and have no biological or other side effects, including acceptable for veterinary and human medicine.
“药学上可接受的盐”是指具有所要的药理活性的、如上所定义的药学上可以接受的盐。这种盐包括与无机酸(例如氢溴酸、盐酸、硝酸、磷酸、硫酸等)或与有机酸(例如乙酸、苯磺酸、苯甲酸、樟脑磺酸、对氯苯磺酸、肉桂酸、柠檬酸、环戊烷丙酸、1,2-乙烷二磺酸、乙磺酸、富马酸、葡庚糖酸、葡萄糖酸、谷氨酸、乙醇酸、己酸、庚酸、邻(4-羟基苯甲酰基)苯甲酸、2-羟基乙磺酸、羟基萘甲酸、乳酸、月桂基磺酸、马来酸、苹果酸、丙二酸、苯乙醇酸、甲磺酸、4-甲基双环[2.2.2]辛-2-烯-1-羧酸、4,4′-亚甲基双(3-羟基-2-烯-1-羧酸)、己二烯二酸、2-萘磺酸、草酸、3-苯基丙酸、丙酸、丙酮酸、水杨酸、硬脂酸、丁烯酸、酒石酸、叔丁基乙酸、对甲苯磺酸、三甲基乙酸等)形成的酸加合盐。"Pharmaceutically acceptable salt" refers to a pharmaceutically acceptable salt as defined above which possesses the desired pharmacological activity. Such salts include combinations with inorganic acids (such as hydrobromic acid, hydrochloric acid, nitric acid, phosphoric acid, sulfuric acid, etc.) or organic acids (such as acetic acid, benzenesulfonic acid, benzoic acid, camphorsulfonic acid, p-chlorobenzenesulfonic acid, cinnamic acid, Citric acid, cyclopentanepropionic acid, 1,2-ethanedisulfonic acid, ethanesulfonic acid, fumaric acid, glucoheptonic acid, gluconic acid, glutamic acid, glycolic acid, caproic acid, heptanoic acid, ortho ( 4-Hydroxybenzoyl)benzoic acid, 2-hydroxyethanesulfonic acid, hydroxynaphthoic acid, lactic acid, laurylsulfonic acid, maleic acid, malic acid, malonic acid, mandelic acid, methanesulfonic acid, 4-methanoic acid Bicyclo[2.2.2]oct-2-ene-1-carboxylic acid, 4,4′-methylene bis(3-hydroxy-2-ene-1-carboxylic acid), hexadienedioic acid, 2- Naphthalenesulfonic acid, oxalic acid, 3-phenylpropionic acid, propionic acid, pyruvic acid, salicylic acid, stearic acid, crotonic acid, tartaric acid, tert-butylacetic acid, p-toluenesulfonic acid, trimethylacetic acid, etc.) acid addition salts.
药学上可接受的盐还包括与碱的加合盐,当存在的酸性质子能与无机碱或有机碱反应时,可以形成这种碱加合盐。可以接受的无机碱包括氢氧化铝、氢氧化钙、氢氧化钾、碳酸钠和氢氧化钠。可以接受的有机碱包括二乙醇胺、乙醇胺、N-甲基葡萄糖胺、三乙醇胺、三甲胺等。Pharmaceutically acceptable salts also include addition salts with bases which may be formed when acidic protons present are capable of reacting with inorganic or organic bases. Acceptable inorganic bases include aluminum hydroxide, calcium hydroxide, potassium hydroxide, sodium carbonate and sodium hydroxide. Acceptable organic bases include diethanolamine, ethanolamine, N-methylglucamine, triethanolamine, trimethylamine, and the like.
“亚苯基”是指二价的芳基-C6H4-,包括1,4-亚苯基、1,3-亚苯基等。"Phenylene" refers to a divalent aryl group -C 6 H 4 -, including 1,4-phenylene, 1,3-phenylene and the like.
“药学上可接受的药物前体衍生物”是指如上所定义的药学上可接受的式Ⅰ的化合物的衍生物,该衍生物在活体内被转变为相应的式Ⅰ的化合物的非衍生物形式。这种药物前体包括具有N-酰化的吡啶基(即N(P)C5H9-)、N-酰化的氮杂亚烷基(例如-N(P)CH2CH2-)、N-酰化的氨基(即-NH2(P))、N-酰化的脒基(即-C(NP)NHP、-C(NH)NHP或-C(NP)NH2)、N-酰化的胍基(即-NHC(NP)NHP、-NHC(NH)NHP或-NHC(NP)NH2),其中P选自-C(O)R10(其中R10可以是C1-10烷氧基或顺式-2-C1-10烷酰氧基苯基乙烯基)、3-C1-10烷酰氧基丁酰基、R11-X28-(其中R11是羧基,而X28是C1-10亚烷基)或-C(O)OCH(R12)OC(O)R13(其中R12为氢、C1-10烷基或C3-10环烷基,而R13为C1-10烷基)。"Pharmaceutically acceptable prodrug derivative" means a pharmaceutically acceptable derivative of the compound of formula I as defined above which is converted in vivo into the corresponding non-derivative of the compound of formula I form. Such prodrugs include pyridyl groups with N - acylation (ie, N(P) C5H9- ), N-acylated azaalkylene groups (ie, -N(P) CH2CH2- ), , N-acylated amino group (ie -NH 2 (P)), N-acylated amidino group (ie -C(NP)NHP, -C(NH)NHP or -C(NP)NH 2 ), N - acylated guanidine (i.e. -NHC(NP)NHP, -NHC(NH)NHP or -NHC(NP)NH 2 ), wherein P is selected from -C(O)R 10 (wherein R 10 may be C 1 -10 alkoxy or cis-2-C 1-10 alkanoyloxyphenyl vinyl), 3-C 1-10 alkanoyloxybutyryl, R 11 -X 28 - (wherein R 11 is carboxyl , and X 28 is C 1-10 alkylene) or -C(O)OCH(R 12 )OC(O)R 13 (wherein R 12 is hydrogen, C 1-10 alkyl or C 3-10 cycloalkane group, and R 13 is C 1-10 alkyl).
“保护基团”具有通常的合成有机化学中与其相关的意义,即一种有选择地占据多官能团化合物中的活性位点、使得化学反应可以有选择地在另一个未保护的活性位点上进行、而且在选择性反应完成后可以很容易地被去掉的基团。"Protecting group" has the usual meaning associated with it in synthetic organic chemistry, that is, a selective occupation of an active site in a multifunctional compound so that chemical reactions can be selectively performed on another unprotected active site. A group that is carried out and can be easily removed after the selective reaction is complete.
“保护试剂”是指一种能与多官能团化合物反应而在活性位点产生一个保护基团的试剂。"Protecting reagent" refers to a reagent capable of reacting with a multifunctional compound to generate a protecting group at the active site.
化合物或基团的“受保护的衍生物”是指其中活性位点被保护基团占据的化合物或基团的衍生物。式Ⅰ化合物的受保护的衍生物其本身是类胰蛋白酶的活性抑制剂并且可用于制备式Ⅰ的其它化合物。活性氮原子的合适的保护基团包括叔丁氧基羰基、苄氧基羰基和任何其它合适的氨基保护基(例如参见T.W.Greene,Protective Groups in OrganicSynthesis,John Wiley & Sons,Inc.1981)。A "protected derivative" of a compound or group refers to a derivative of a compound or group in which the active site is occupied by a protecting group. Protected derivatives of the compounds of formula I are themselves inhibitors of tryptase activity and are useful in the preparation of other compounds of formula I. Suitable protecting groups for reactive nitrogen atoms include t-butoxycarbonyl, benzyloxycarbonyl and any other suitable amino protecting groups (see for example T.W. Greene, Protective Groups in Organic Synthesis, John Wiley & Sons, Inc. 1981).
疾病的“症状学”是指病人所感受到的任何病态的现象或偏离正常的结构、功能或感觉和疾病的指示、产生及其所提供的迹象。The "symptomology" of a disease refers to any morbid phenomenon or deviation from normal structure, function, or sensation experienced by a patient, and the indication, production, and signs provided by the disease.
“治疗有效量”是指治疗一种疾病时,足以有效地治疗这种疾病而需要向动物给药的数量。"Therapeutically effective amount" refers to the amount needed to be administered to animals when treating a disease, which is sufficient to effectively treat the disease.
疾病的“治疗”包括可以预先对还没有疾病症状的动物采取措施而预防动物发病、抑制疾病(即控制疾病的发展)或缓解疾病(即使疾病衰退)。"Treatment" of a disease includes taking measures in advance for animals that have no symptoms of the disease to prevent the disease, inhibit the disease (that is, control the development of the disease) or alleviate the disease (that is, the decline of the disease).
术语“q.s.”是指能充分达到所说的功能(例如使溶液达到所需要的体积即100%)所需要加入的数量。The term "q.s." refers to the quantity needed to add sufficient to achieve the stated function (eg, to bring the solution to the desired volume, ie 100%).
式Ⅰ的化合物以及用于制备它们的中间体和反应原料按照IUPAC命名规则命名,其中用作基本基团的特征基团按照下列递降的优先次序引用:酸、酯、酰胺和脒。而且,在本申请中以书面描述二价基时,前缀的序数代表基团的连接位置。同样地,就结构式所代表的二价基来说,结构式的表示方式代表连接位置。例如,其中R1为4-脒基苄基,X1和X9各为-NHC(O)-,X2为1,4-亚哌嗪基,X7为-C(O)O-,X8为4,1-亚哌啶基,而R2为R9-X21(其中R9为哌啶-4-基,而X21为3-氮杂三亚甲基)的式Ⅰ化合物用下列结构式表示: The compounds of formula I and the intermediates and reaction materials used to prepare them are named according to the IUPAC nomenclature, wherein the characteristic groups used as basic groups are cited in the following descending order of priority: acids, esters, amides and amidines. Moreover, when a divalent group is described in writing in this application, the ordinal number of the prefix represents the attachment position of the group. Likewise, as far as the divalent group represented by the structural formula is concerned, the representation of the structural formula represents the linking position. For example, wherein R 1 is 4-amidinobenzyl, X 1 and X 9 are each -NHC(O)-, X 2 is 1,4-piperazinylene, X 7 is -C(O)O-, X 8 is 4,1-piperidinylene, and R 2 is R 9 -X 21 (wherein R 9 is piperidin-4-yl, and X 21 is 3-azatrimethylene) for the compound of formula I The following structural formula represents:
当X3和X7各为-C(O)O-,X4和X6各为一个共价键,X5为顺式-1,5-亚环辛基,而P为氢时,该化合物被命名为:顺-1,5-亚环辛基-4-(4-脒基苄基氨基甲酰基)-1-哌嗪甲酸酯-4-(2-哌啶-4-基氨基乙基氨基甲酰基)-1-哌啶甲酸酯;When X 3 and X 7 are each -C(O)O-, X 4 and X 6 are each a covalent bond, X 5 is cis-1,5-cyclooctylene, and P is hydrogen, the The compound is named: cis-1,5-cyclooctylene-4-(4-amidinobenzylcarbamoyl)-1-piperazinecarboxylate-4-(2-piperidin-4-ylamino Ethylcarbamoyl)-1-piperidinecarboxylate;
当X3和X7各为-C(O)O-,X4和X6各为一个共价键,X5为顺式-1,5-亚环辛基,而P为3-羧基丙酰基时,该化合物被命名为:3-{4-[2-(1-{顺-5-[4-(4-脒基苄基氨基甲酰基)哌嗪-1-基羰基氧基]环辛氧基羰基}哌啶-4-基羰基氨基)乙基氨基]哌啶-1-基羰基}丙酸;When X 3 and X 7 are each -C(O)O-, X 4 and X 6 are each a covalent bond, X 5 is cis-1,5-cyclooctylene, and P is 3-carboxypropane When the acyl group is present, the compound is named: 3-{4-[2-(1-{cis-5-[4-(4-amidinobenzylcarbamoyl)piperazin-1-ylcarbonyloxy]cyclo Octyloxycarbonyl}piperidin-4-ylcarbonylamino)ethylamino]piperidin-1-ylcarbonyl}propionic acid;
当X3为-C(O)-,X7为-C(O)O-,X4为一个共价键,X6为亚甲基,X5为亚苯基,而P为氢时,该化合物被命名为:4-[4-(4-脒基苄基氨基甲酰基)哌嗪-1-基羰基]苄基-4-(2-哌啶-4-基氨基乙基氨基甲酰基)-1-哌啶甲酸酯;When X 3 is -C(O)-, X 7 is -C(O)O-, X 4 is a covalent bond, X 6 is methylene, X 5 is phenylene, and P is hydrogen, The compound is named: 4-[4-(4-amidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]benzyl-4-(2-piperidin-4-ylaminoethylcarbamoyl )-1-piperidinecarboxylate;
当X3和X7各为-C(O)O-,而X4-X5-X6为1,4-四亚甲基(即-CH2CH2CH2CH2-)时,该化合物被命名为:1,4-四亚甲基-4-脒基苄基氨基甲酰基-1-哌嗪甲酸酯;而When each of X 3 and X 7 is -C(O)O-, and X 4 -X 5 -X 6 is 1,4-tetramethylene (ie -CH 2 CH 2 CH 2 CH 2 -), the The compounds were named: 1,4-tetramethylene-4-amidinobenzylcarbamoyl-1-piperazinecarboxylate; and
当X3和X7各为-C(O)-,而X4-X5-X6为1,4-四亚甲基(即-CH2CH2CH2CH2-)时,该化合物被命名为:N-4-脒基苄基-4-{5-[4-(2-哌啶-4-基氨基乙基氨基甲酰基)哌啶-1-基羰基]戊酰基}-1-哌嗪甲酰胺。When each of X 3 and X 7 is -C(O)-, and X 4 -X 5 -X 6 is 1,4-tetramethylene (ie -CH 2 CH 2 CH 2 CH 2 -), the compound Named as: N-4-amidinobenzyl-4-{5-[4-(2-piperidin-4-ylaminoethylcarbamoyl)piperidin-1-ylcarbonyl]pentanoyl}-1 - piperazine carboxamide.
本发明的优选实施方式:Preferred embodiments of the present invention:
虽然在本发明的概述中提出了本发明的最宽的定义,然而优选某些式Ⅰ的化合物。例如,优选的式Ⅰ的化合物是其中X5为顺式-1,5-亚环辛基,X4和X6各为一个共价键,X4-X5-X6一起为C4-8亚烷基或者X5为亚苯基,X4和X6为C0-1亚烷基;X1和X9各自独立地为一共价键、-C(O)-、-NHC(O)-、-C(O)NH、-N(CH3)C(O)-或-S(O)2NH-(条件是:X1和X9不全是共价键);X3和X7独立地为-C(O)-或-C(O)O-;X2和X8独立地为-X10-X11-(其中X10为共价键或亚甲基,而X11为4,1-亚哌啶基或1,4-亚哌嗪基);R1为R4-X12-或R5-X13,其中R4为脒基、胍基或甲基氨基,X12为-X14-X15-X16-,其中X15为1,4-亚苯基或1,4-亚哌啶基,X14为Cn14亚烷基,而X16为Cn16亚烷基,其中n14和n16=0、1或2,R5为哌啶-4-基,而X13为C2-3亚烷基;和其中R2为R8-X20-或R9-X21-,R8为氨基、脒基、胍基、甲基氨基或1-亚氨基乙基,X20为-X22-X23-X24,其中X23为反式-1,4-亚环己基、1,4-亚苯基、4,1-亚吡啶基、1,4-亚哌啶基,X22为Cn22亚烷基,而X24为Cn24亚烷基,其中n22和n24=1或2,R9为苯并咪唑-5-基、咪唑-1-基、咪唑-4-基、2-咪唑啉-2-基、4-甲基咪唑-1-基、5-甲基咪唑-1-基、1-甲基哌啶-4-基、哌啶-4-基、哌嗪-1-基、吡啶-3-基、吡啶-4-基、1,4,5,6-四氢嘧啶-5-基或1,4,5,6-四氢-2-二氧代嘧啶-5-基,而X21为C1-6亚烷基、ω-氮杂C2-5亚烷基、3-氧代三亚烷基、ω-硫杂C2-4亚烷基、3-氧代-2-氮杂三亚烷基、3-氮杂-2-氧代三亚烷基或-X25-X26-X27-,其中X26为1,4-亚苯基,X25为Cn25亚烷基,而X27为Cn27亚烷基,其中n25和n27=0或1的式Ⅰ的化合物及其药学上可接受的盐、N-氧化物、药物前体衍生物及其受保护的衍生物。Although the broadest definition of the invention is set forth in the Summary of the Invention, certain compounds of formula I are preferred. For example, a preferred compound of formula I is wherein X 5 is cis-1,5-cyclooctylene, X 4 and X 6 are each a covalent bond, and X 4 -X 5 -X 6 together are C 4- 8 alkylene or X 5 is phenylene, X 4 and X 6 are C 0-1 alkylene; X 1 and X 9 are each independently a covalent bond, -C(O)-, -NHC(O )-, -C(O)NH, -N(CH 3 )C(O)- or -S(O) 2 NH- (provided that X 1 and X 9 are not all covalent bonds); X 3 and X 7 is independently -C(O)- or -C(O)O-; X 2 and X 8 are independently -X 10 -X 11 - (wherein X 10 is a covalent bond or methylene, and X 11 is 4,1-piperidinyl or 1,4-piperazinyl); R 1 is R 4 -X 12 - or R 5 -X 13 , wherein R 4 is amidino, guanidino or methylamino, X 12 is -X 14 -X 15 -X 16 -, wherein X 15 is 1,4-phenylene or 1,4-piperidinylene, X 14 is C n14 alkylene, and X 16 is C n16 Alkylene, wherein n14 and n16=0, 1 or 2, R 5 is piperidin-4-yl, and X 13 is C 2-3 alkylene; and wherein R 2 is R 8 -X 20 - or R 9 -X 21 -, R 8 is amino, amidino, guanidino, methylamino or 1-iminoethyl, X 20 is -X 22 -X 23 -X 24 , wherein X 23 is trans-1, 4-cyclohexylene, 1,4-phenylene, 4,1-pyridinyl, 1,4-piperidinyl, X 22 is C n22 alkylene, and X 24 is C n24 alkylene, Wherein n22 and n24=1 or 2, R 9 is benzimidazol-5-yl, imidazol-1-yl, imidazol-4-yl, 2-imidazolin-2-yl, 4-methylimidazol-1-yl , 5-methylimidazol-1-yl, 1-methylpiperidin-4-yl, piperidin-4-yl, piperazin-1-yl, pyridin-3-yl, pyridin-4-yl, 1, 4,5,6-tetrahydropyrimidin-5-yl or 1,4,5,6-tetrahydro-2-dioxopyrimidin-5-yl, and X 21 is C 1-6 alkylene, ω- Aza C2-5 alkylene, 3-oxotrialkylene, ω-thia C2-4 alkylene, 3-oxo-2-azatrialkylene, 3-aza-2-oxo Trialkylene or -X 25 -X 26 -X 27 -, where X 26 is 1,4-phenylene, X 25 is C n25 alkylene, and X 27 is C n27 alkylene, where n25 and Compounds of formula I with n27=0 or 1 and pharmaceutically acceptable salts, N-oxides, prodrug derivatives and protected derivatives thereof.
更优选的式Ⅰ的化合物是其中X5为顺式-1,5-亚环辛基,X4和X6各为一个共价键或X4-X5-X6一起为C4-8亚烷基;其中X1和X9各自独立地为一共价键、-C(O)-、-NHC(O)-、-C(O)NH或-S(O)2NH-(条件是:X1和X9不全是共价键);X3和X7独立地为-C(O)-或-C(O)O-;X2和X8独立地为-X10-X11-(其中X10为共价键或亚甲基,而X11为4,1-亚哌啶基或1,4-亚哌嗪基);R1为R4-X12-,其中R4为脒基或胍基,X12为-X14-X15-X16-,其中X15为1,4-亚苯基或1,4-亚哌啶基,X14为Cn14亚烷基,而X16为Cn16亚烷基,其中n14和n16=0、1或2;和R2为R8-X20-或R9-X21-,其中R8为氨基或甲基氨基,X20为-X22-X23-X24,其中X23为反式-1-4-亚环己基或1,4-亚苯基,X22为Cn22亚烷基,而X24为Cn24亚烷基,其中n22和n24=1或2,R9为咪唑-1-基、咪唑-4-基、4-甲基咪唑-1-基、5-甲基咪唑-1-基、哌啶-4-基或吡啶-4-基,而X21为C1-5亚烷基或3-氮杂三亚烷基的式Ⅰ的化合物及其药学上可接受的盐、N-氧化物、药物前体衍生物及其受保护的衍生物。A more preferred compound of formula I is wherein X 5 is cis-1,5-cyclooctylene, X 4 and X 6 are each a covalent bond or X 4 -X 5 -X 6 together are C 4-8 Alkylene; wherein X 1 and X 9 are each independently a covalent bond, -C(O)-, -NHC(O)-, -C(O)NH or -S(O) 2 NH- (provided : X 1 and X 9 are not all covalent bonds); X 3 and X 7 are independently -C(O)- or -C(O)O-; X 2 and X 8 are independently -X 10 -X 11 -(where X 10 is a covalent bond or methylene, and X 11 is 4,1-piperidinylene or 1,4-piperazinylene); R 1 is R 4 -X 12 -, wherein R 4 is amidino or guanidino, X 12 is -X 14 -X 15 -X 16 -, wherein X 15 is 1,4-phenylene or 1,4-piperidinyl, X 14 is C n14 alkylene , and X 16 is C n16 alkylene, wherein n14 and n16=0, 1 or 2; and R 2 is R 8 -X 20 - or R 9 -X 21 -, wherein R 8 is amino or methylamino, X 20 is -X 22 -X 23 -X 24 , wherein X 23 is trans-1-4-cyclohexylene or 1,4-phenylene, X 22 is C n22 alkylene, and X 24 is C N24 alkylene, wherein n22 and n24=1 or 2, R 9 is imidazol-1-yl, imidazol-4-yl, 4-methylimidazol-1-yl, 5-methylimidazol-1-yl, piper Pyridin-4-yl or pyridin-4-yl, and X 21 is a compound of formula I of C 1-5 alkylene or 3-azatrialkylene and pharmaceutically acceptable salts, N-oxides, Prodrug derivatives and protected derivatives thereof.
特别优选的式Ⅰ的化合物是其中X5为顺式-1,5-亚环辛基,而X4和X6各为一个共价键;其中X1和X9各自独立地为-C(O)-或-NHC(O)-;其中X3和X7各为-C(O)O-;X2和X8独立地为-X10-X11-(其中X10为共价键或亚甲基,而X11为1,4-亚哌嗪基);R1为R4-X12-,其中R4为脒基或胍基,X12为-X14-X15-X16-,其中X15为1,4-亚苯基,X14为一共价键,而X16为亚甲基;和R2为R8-X20-或R9-X21、其中R8为氨基,X20为-X22-X23-X24,其中X23为反式-1,4-亚环己基,X22为一共价键,而X24为亚甲基,R9为哌啶-4-基,而X21为亚乙基或三亚甲基的式Ⅰ的化合物及其药学上可接受的盐、N-氧化物、药物前体衍生物及其受保护的衍生物。A particularly preferred compound of formula I is wherein X 5 is cis-1,5-cyclooctylene, and X 4 and X 6 are each a covalent bond; wherein X 1 and X 9 are each independently -C( O)- or -NHC(O)-; wherein X 3 and X 7 are each -C(O)O-; X 2 and X 8 are independently -X 10 -X 11 - (wherein X 10 is a covalent bond or methylene, and X 11 is 1,4-piperazinyl); R 1 is R 4 -X 12 -, wherein R 4 is amidino or guanidino, and X 12 is -X 14 -X 15 -X 16 -, wherein X 15 is 1,4-phenylene, X 14 is a covalent bond, and X 16 is methylene; and R 2 is R 8 -X 20 - or R 9 -X 21 , wherein R 8 is amino, X 20 is -X 22 -X 23 -X 24 , wherein X 23 is trans-1,4-cyclohexylene, X 22 is a covalent bond, and X 24 is methylene, R 9 is piper Pyridin-4-yl, and X 21 is ethylene or trimethylene compound of formula I and pharmaceutically acceptable salts, N-oxides, prodrug derivatives and protected derivatives thereof.
特别优选的式Ⅰ的化合物是其中X4-X5-X6一起为C4-8亚烷基;X1和X9各自独立地为-C(O)-或-NHC(O)-;X3和X7独立地为-C(O)-或-C(O)O-;X2和X8独立地为-X10-X11-(其中X10为一共价键,而X11为1,4-亚哌嗪基);R1为R4-X12-,其中R4为脒基或胍基,而X12为-X14-X15-X16-,其中X15为1,4-亚苯基,X14为一共价键,而X16为亚甲基;和R2为R8-X20-,其中R8为脒基或胍基,而X20为-X22-X23-X24,其中X23为1,4-亚苯基,X22为一共价键,而X24为亚甲基的式Ⅰ的化合物及其药学上可接受的盐、N-氧化物、药物前体衍生物及其受保护的衍生物。最优选的式Ⅰ的化合物是下列化合物:A particularly preferred compound of formula I is wherein X 4 -X 5 -X 6 together represent a C 4-8 alkylene group; X 1 and X 9 are each independently -C(O)- or -NHC(O)-; X 3 and X 7 are independently -C(O)- or -C(O)O-; X 2 and X 8 are independently -X 10 -X 11 - (wherein X 10 is a covalent bond, and X 11 is 1,4-piperazinyl); R 1 is R 4 -X 12 -, wherein R 4 is amidino or guanidino, and X 12 is -X 14 -X 15 -X16-, wherein X 15 is 1 , 4-phenylene, X 14 is a covalent bond, and X 16 is methylene; and R 2 is R 8 -X 20 -, wherein R 8 is amidino or guanidino, and X 20 is -X 22 -X 23 -X 24 , wherein X 23 is 1,4-phenylene, X 22 is a covalent bond, and X 24 is a methylene compound of formula I and its pharmaceutically acceptable salt, N-oxidation Drugs, prodrug derivatives and protected derivatives thereof. The most preferred compounds of formula I are the following compounds:
4-胍基苄基-4-{7-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]-庚酰基}-1-哌嗪甲酰胺、4-guanidinobenzyl-4-{7-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]-heptanoyl}-1-piperazinecarboxamide,
4-胍基苄基-4-{8-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]-辛酰基}-1-哌嗪甲酰胺、4-guanidinobenzyl-4-{8-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]-octanoyl}-1-piperazinecarboxamide,
4-胍基苄基-4-{9-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]-壬酰基}-1-哌嗪甲酰胺、4-guanidinobenzyl-4-{9-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]-nonanoyl}-1-piperazinecarboxamide,
4-脒基苄基-4-{7-[4-(4-脒基苄基氨基甲酰基)哌嗪-1-基羰基]-庚酰基}-1-哌嗪甲酰胺、4-amidinobenzyl-4-{7-[4-(4-amidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]-heptanoyl}-1-piperazinecarboxamide,
顺式-1,5-亚环辛基-4-(4-脒基苄基氨基甲酰基)-1-哌嗪甲酸酯-4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯、cis-1,5-cyclooctylene-4-(4-amidinobenzylcarbamoyl)-1-piperazinecarboxylate-4-(2-piperidin-4-ylethylcarbamoyl )-1-piperazine carboxylate,
1,5-五亚甲基-二[4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯]、1,5-pentamethylene-bis[4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate],
顺式-1,5-亚环辛基-4-(4-脒基苄基氨基甲酰基)1-哌嗪甲酸酯-4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯、cis-1,5-Cyclooctylene-4-(4-amidinobenzylcarbamoyl)1-piperazinecarboxylate-4-(2-piperidin-4-ylethylcarbamoyl) -1-piperazine carboxylate,
顺式-1,5-亚环辛基-反式-4-(4-氨基环己基甲基氨基甲酰基)-1-哌嗪甲酸酯-4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯、cis-1,5-cyclooctylene-trans-4-(4-aminocyclohexylmethylcarbamoyl)-1-piperazinecarbamate-4-(4-guanidinobenzylcarbamoyl )-1-piperazine carboxylate,
顺式-1,5-亚环辛基-4-(4-脒基苯乙酰基)-1-哌嗪甲酸酯-4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯、cis-1,5-Cyclooctylene-4-(4-amidinophenylacetyl)-1-piperazinecarboxylate-4-(4-piperidin-4-ylbutyryl)-1-piper Zinc carboxylate,
1,4-四亚甲基-二[4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯]、1,4-Tetramethylene-bis[4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate],
顺式-1,5-亚环辛基-4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯-4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯、cis-1,5-cyclooctylene-4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate-4-(2-piperidin-4-ylethylcarbamoyl )-1-piperazine carboxylate,
4-胍基苄基-4-{6-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]-己酰基}-1-哌嗪甲酰胺、4-guanidinobenzyl-4-{6-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]-hexanoyl}-1-piperazinecarboxamide,
顺式-1,5-亚环辛基-4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯-4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯、cis-1,5-Cyclooctylene-4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate-4-(4-piperidin-4-ylbutyryl)-1 -piperazine carboxylate,
顺式-1,5-亚环辛基-4-(4-胍基苯乙酰基)-1-哌嗪甲酸酯-4-(2-哌啶-4-基乙基氨基甲酰基丁酰基)-1-哌嗪甲酸酯、cis-1,5-cyclooctylene-4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate-4-(2-piperidin-4-ylethylcarbamoylbutyryl )-1-piperazine carboxylate,
顺式-1,5-亚环辛基-4-(4-胍基苯乙酰基)-1-哌嗪甲酸酯-4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯、cis-1,5-cyclooctylene-4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate-4-(4-piperidin-4-ylbutyryl)-1-piper Zinc carboxylate,
4-胍基苄基-4-{5-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]-戊酰基}-1-哌嗪甲酰胺、4-guanidinobenzyl-4-{5-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]-pentanoyl}-1-piperazinecarboxamide,
3-氧杂-1,5-五亚甲基-二[4-(4-脒基苯乙酰基)哌嗪-1-基羰基]和3-oxa-1,5-pentamethylene-bis[4-(4-amidinophenylacetyl)piperazin-1-ylcarbonyl] and
顺式-1,5-亚环辛基-4-(4-脒基苯乙酰基)-1-哌嗪甲酸酯-4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯cis-1,5-cyclooctylene-4-(4-amidinophenylacetyl)-1-piperazinecarboxylate-4-(2-piperidin-4-ylethylcarbamoyl)- 1-piperazine carboxylate
及其药学上可接受的盐、N-氧化物、药物前体衍生物及其受保护的衍生物。药理学和用途:And pharmaceutically acceptable salts, N-oxides, drug prodrug derivatives and protected derivatives thereof. Pharmacology and Uses:
本发明的化合物是类胰蛋白酶抑制剂。这样的式Ⅰ的化合物可用于治疗疾病、特别是免疫介导的炎症,其中类胰蛋白酶的活性与疾病的病理学和/或症状学有关。例如,其中类胰蛋白酶的活性与疾病的病理学和/或症状学有关的免疫介导的炎症包括哮喘、过敏性鼻炎、风湿性脊椎炎、骨关节炎、通风性关节炎、风湿性关节炎、一般的关节炎、荨麻疹、血管性水肿、湿疹性皮炎、过敏症、过度增生性皮肤病、胃溃疡、炎症性肠病、眼结膜炎和青春期结膜炎、炎症性皮肤病等。The compounds of the present invention are tryptase inhibitors. Such compounds of formula I are useful in the treatment of diseases, especially immune-mediated inflammation, in which tryptase activity is associated with the pathology and/or symptomology of the disease. For example, immune-mediated inflammation in which tryptase activity is associated with the pathology and/or symptomology of the disease includes asthma, allergic rhinitis, rheumatoid spondylitis, osteoarthritis, rheumatoid arthritis, rheumatoid arthritis , General arthritis, urticaria, angioedema, eczematous dermatitis, allergies, hyperproliferative skin diseases, gastric ulcers, inflammatory bowel disease, ocular conjunctivitis and adolescent conjunctivitis, inflammatory skin diseases, etc.
适用于测定类胰蛋白酶活性及化合物对它的抑制作用的体外实验是已知的(例如参见Sturzebecher等人,(1992)Biol.Chem.Hoppe-Seyler373:1025-1030)。典型地,实验将测定由多肽碱底物水解诱导的类胰蛋白酶。关于测定类胰蛋白酶活性的体外实验的细节请阅以下的实施例33。Suitable in vitro assays for measuring tryptase activity and compounds' inhibition thereof are known (eg, see Sturzebecher et al. (1992) Biol. Chem. Hoppe-Seyler 373:1025-1030). Typically, experiments will measure tryptase induced by hydrolysis of a polypeptide base substrate. See Example 33 below for details on the in vitro assay for assaying tryptase activity.
炎症的合适的活体模型对于本领域普通技术人员是已知的,例如哮喘的活体模型是已知的(例如参见Larsen(1991)Experimental Models ofReversible Airway Obstruction.在West等人编辑的由ScientificFoundations Raven Press,New York出版的The Lung一书中)。关于哮喘的体外模型的细节请参阅下面的实施例2。另外,炎症性皮肤病(Walsh等人,(1995)Br.J.Pharmacol.114:1343-1350)和Armstrong等人,(1995)Prostaglandins 49:205-224)、关节炎(Peacock等人,(1995)Cell Immunol.160:178-184和Hourl等人,(1995)Curr.Opin.Rheumatol.7:201-205)和胃肠病(Anthony等人,(1995)Int.J.Exp.Pathol.76:215-224和Carter等人,(1995)Dig.Dis.Sci.40:192-197)的活体模型也是已知的。关于测定哮喘反应的活体实验的细节请参阅下面的实施例34。本发明的药物组合物:Suitable in vivo models of inflammation are known to those of ordinary skill in the art, for example in vivo models of asthma are known (see for example Larsen (1991) Experimental Models of Reversible Airway Obstruction. In West et al. edited by Scientific Foundations Raven Press, The Lung, New York Publishing). See Example 2 below for details on the in vitro model of asthma. In addition, inflammatory skin diseases (Walsh et al., (1995) Br.J.Pharmacol.114:1343-1350) and Armstrong et al., (1995) Prostaglandins 49:205-224), arthritis (Peacock et al., ( 1995) Cell Immunol.160:178-184 and Hourl et al., (1995) Curr.Opin.Rheumatol.7:201-205) and Gastroenterology (Anthony et al., (1995) Int.J.Exp.Pathol. 76:215-224 and Carter et al. (1995) Dig. Dis. Sci. 40:192-197) in vivo models are also known. See Example 34 below for details on the in vivo assay to measure asthmatic response. Pharmaceutical composition of the present invention:
一般来说,式Ⅰ的化合物可以以治疗有效量、按照通常的本领域可接受的方式、单独或与另一种治疗试剂一起给药。治疗有效量可以根据疾病的严重程度、病人的年龄和相对的健康状况、所使用的化合物的药力及其它因素而变化。例如,用于治疗哮喘的式Ⅰ化合物的治疗有效量可以在0.1μg/kg体重/天至1mg/kg体重/天的范围、典型地在1μg/kg体重/天至0.1mg/kg体重/天的范围内变化。因此,一个80kg重的哮喘病人的治疗有效量可以是10μg/天-10mg/天,典型地为0.1mg/天-10mg/天。In general, a compound of formula I may be administered in a therapeutically effective amount, either alone or in combination with another therapeutic agent, in a manner generally accepted in the art. A therapeutically effective amount can vary depending on the severity of the disease, the age and relative health of the patient, the potency of the compound being used, and other factors. For example, a therapeutically effective amount of a compound of formula I for the treatment of asthma may be in the range of 0.1 μg/kg body weight/day to 1 mg/kg body weight/day, typically 1 μg/kg body weight/day to 0.1 mg/kg body weight/day changes within the range. Thus, a therapeutically effective dose for an 80 kg asthmatic patient may be 10 μg/day to 10 mg/day, typically 0.1 mg/day to 10 mg/day.
在治疗哮喘时,可用于与式Ⅰ的化合物一起给药的其它治疗试剂包括β-肾上腺素激动剂(例如沙丁胺醇、叔丁喘宁、福莫特罗、非诺特罗、prenaline等)、甲基黄嘌呤(例如咖啡因、茶碱、氨茶碱、可可碱等)、色甘酸盐(例如色甘酸、奈多罗米等)和皮质类固醇(例如倍氯米松、去炎松、flurisolide、地塞米松等)。总之,本领域普通技术人员依靠个人的知识和本申请的公开,能够确定用于治疗一种给定的炎症所需要的式Ⅰ化合物的治疗有效量。In the treatment of asthma, other therapeutic agents that can be administered with the compound of formula I include beta-adrenergic agonists (such as albuterol, terbuterol, formoterol, fenoterol, prenaline, etc.), formazan Xanthines (such as caffeine, theophylline, aminophylline, theobromine, etc.), cromolyn (such as cromolyn, nedocromil, etc.) and corticosteroids (such as beclomethasone, triamcinolone, flurisolide, Semethasone, etc.). In general, one of ordinary skill in the art, relying on personal knowledge and the disclosure of this application, will be able to determine the therapeutically effective amount of a compound of formula I required to treat a given inflammation.
式Ⅰ化合物可以以药物组合物形式按照下列方式给药:口服、全身性给药(例如透皮给药、鼻腔内给药或栓剂给药)或非肠胃给药(例如肌肉注射、静脉注射或皮下注射)。组合物可以是片剂、药丸、胶囊、半固体、粉末、可持续性释放剂型、溶液、悬浮液、酏剂、气溶胶或其它合适形式的组合物,并且一般包括一种式Ⅰ的化合物和至少一种药学上可接受的赋形剂。可接受的赋形剂是无毒的帮助给药的试剂,并且对活性成分不产生负面影响。这样的赋形剂可以是固体、液体、半固体或者在气溶胶组合物的情况下,赋形剂可以是本领域技术人员一般可以得到的气态赋形剂。The compound of formula I can be administered in the form of a pharmaceutical composition in the following ways: oral, systemic administration (such as transdermal administration, intranasal administration or suppository administration) or parenteral administration (such as intramuscular injection, intravenous injection or subcutaneous injection). Compositions may be in the form of tablets, pills, capsules, semi-solids, powders, sustained release dosage forms, solutions, suspensions, elixirs, aerosols or other suitable forms of compositions and generally comprise a compound of formula I and at least one pharmaceutically acceptable excipient. Acceptable excipients are non-toxic agents that facilitate administration and do not adversely affect the active ingredient. Such excipients may be solid, liquid, semi-solid or, in the case of aerosol compositions, excipients may be gaseous excipients generally available to those skilled in the art.
固体的药用赋形剂包括淀粉、纤维素、滑石、葡萄糖、乳糖、蔗糖、明胶、麦芽糖、大米、面粉、白垩、硅胶、硬脂酸镁、硬脂酸钠、单硬脂酸甘油酯、氯化钠、干燥的脱脂奶等。液体和半固体的赋形剂可以选自水、乙醇、甘油、丙二醇和各种油类,包括石油、动物油、植物油和合成油(例如花生油、大豆油、矿物油、蓖麻油等)。优选的液态载体、特别是用于注射液的液体载体包括水、盐水、葡萄糖水溶液和甘醇。Solid pharmaceutical excipients include starch, cellulose, talc, glucose, lactose, sucrose, gelatin, maltose, rice, flour, chalk, silica gel, magnesium stearate, sodium stearate, glyceryl monostearate, Sodium chloride, dry skim milk, etc. Liquid and semisolid excipients can be selected from water, ethanol, glycerol, propylene glycol and various oils, including petroleum, animal, vegetable and synthetic oils (eg peanut oil, soybean oil, mineral oil, castor oil, etc.). Preferred liquid carriers, especially for injections, include water, saline, aqueous dextrose and glycol.
压缩气体可用于分散气溶胶形式的活性成分。适用于该目的的惰性气体是氮气、二氧化碳、氧化氮等。在Easton,Pa.:Mack PublishingCompany出版的A.R.Alfonso,Remington’s Pharmaceutical Sciences1985,17th ed.中记载了其它合适的药用载体及其制剂。Compressed gases may be used to disperse active ingredients in aerosol form. Inert gases suitable for this purpose are nitrogen, carbon dioxide, nitrogen oxide, and the like. Other suitable pharmaceutical carriers and their formulations are described in A.R. Alfonso, Remington's Pharmaceutical Sciences 1985, 17th ed., Easton, Pa.: Mack Publishing Company.
组合物中式Ⅰ化合物的用量可以随制剂的类型、单位剂量的大小、赋形剂的种类及药学领域的技术人员已知的其它因素而在很宽的范围内变化。一般地,用于治疗哮喘的式Ⅰ化合物的组合物将包含0.01%-10%重量、优选0.3%-1%重量的活性成分,其它为赋形剂。优选地,药物组合物以单剂量形式给药以进行连续治疗或者在特别需要减轻症状时随机地以单剂量形式给药。在实施例34中描述了具有代表性的含有式Ⅰ化合物的药物制剂。化学:The amount of the compound of formula I used in the composition can vary widely depending on the type of formulation, size of the unit dose, type of excipients and other factors known to those skilled in the pharmaceutical art. In general, compositions of compounds of formula I for use in the treatment of asthma will contain 0.01% to 10% by weight, preferably 0.3% to 1% by weight of active ingredient, the rest being excipients. Preferably, the pharmaceutical composition is administered in a single dose for continuous treatment or randomly when relief of symptoms is particularly desired. Representative pharmaceutical formulations containing compounds of formula I are described in Example 34. Chemical:
本发明的化合物由五个不同的小单元(即R1-、-X2-、-X4-X5-X6-、-X8-和R2-)组成,其中之间通过羰基、甲酰氧基、酰胺、磺酰胺、氨基甲酸酯或脲连接基团(即-C(O)-、-C(O)O-、-OC(O)-、-C(O)N(R3)-、-N(R3)C(O)-、-S(O)2N(R3)-、-N(R3)S(O)2-、-OC(O)N(R3)-、-N(R3)C(O)O-、-N(R3)C(O)N(R3)-或-OC(O)O-)连接。形成这种连接基团的方法是已知的,合适的试剂是容易得到的(例如参见March等人,Advanced OrganicChemistry,4th Ed.(Wiley 1992);Larock,Comprehensive OrganicTransformations(VCH 1989);和Furniss等人,Vogel’s Textbook ofPractical Organic Chemistry,5th Ed..(Longman 1989)。The compound of the present invention is composed of five different small units (namely R 1 -, -X 2 -, -X 4 -X 5 -X 6 -, -X 8 - and R 2 -), among which carbonyl, Formyloxy, amide, sulfonamide, carbamate, or urea linking groups (i.e. -C(O)-, -C(O)O-, -OC(O)-, -C(O)N( R 3 )-, -N(R 3 )C(O)-, -S(O) 2 N(R 3 )-, -N(R 3 )S(O) 2 -, -OC(O)N( R 3 )-, -N(R 3 )C(O)O-, -N(R 3 )C(O)N(R 3 )- or -OC(O)O-) linkage. Methods for forming such linking groups are known and suitable reagents are readily available (see for example March et al., Advanced Organic Chemistry, 4th Ed. (Wiley 1992); Larock, Comprehensive Organic Transformations (VCH 1989); and Furniss et al. people, Vogel's Textbook of Practical Organic Chemistry, 5th Ed.. (Longman 1989).
可以把包含式Ⅰ化合物的小单元单个组装或者组装成更大的小单元的组合体。下列反应式是制备式Ⅰ化合物的代表性的方法。应当明白,式Ⅰ化合物可以按照其它类似的方法制备。Small units comprising compounds of formula I can be assembled individually or into assemblies of larger small units. The following schemes are representative of methods for preparing compounds of formula I. It should be understood that compounds of formula I may be prepared by other similar methods.
其中X8为1,4-亚哌嗪基或1,4-亚哌啶基,而X9为-C(O)-、-OC(O)-或-N(R3)C(O)-的式Ⅰ化合物或者其中X8为C1-8亚烷基,而X9为-C(O)N(R3)-、-OC(O)N(R3)-或-N(R3)C(O)N(R3)-的式Ⅰ化合物可以通过使下式Ⅰ的化合物或其受保护的衍生物1与一种式R2-Y9-C(O)L的化合物或其受保护的衍生物反应(其中L为一种离去基团,Y9为一键、-O-或-N(R3)-,Y8为哌嗪-1-基、哌啶-4-基或HN(R3)-C1-8烷基,每个R1、R2、R3、X1、X2、X3、X4、X5、X6和X7如发明概述中所定义)、然后(必要时)脱保护来制备。另外,其中X8为1,4-亚哌嗪基或1,4-亚哌啶基,而X9为-NHC(O)-的式Ⅰ化合物或者其中X8为C1-8亚烷基,而X9为-NHC(O)N(R3)-的式Ⅰ化合物可以通过使一种合适的式Ⅰ的化合物或其受保护的衍生物与式R2-NC(O)的异氰酸酯或其受保护的衍生物反应,然后(必要时)脱保护来制备(细节请参阅下面的实施例8)。Where X 8 is 1,4-piperazinylene or 1,4-piperidinylene, and X 9 is -C(O)-, -OC(O)- or -N(R 3 )C(O) - or a compound of formula I wherein X 8 is C 1-8 alkylene, and X 9 is -C(O)N(R 3 )-, -OC(O)N(R 3 )- or -N(R 3 ) The formula I compound of C(O)N(R 3 )- can be obtained by making the following formula I compound or its protected derivative 1 reacting with a compound of formula R 2 -Y 9 -C(O)L or a protected derivative thereof (wherein L is a leaving group, Y 9 is a bond, -O- or -N( R 3 )-, Y 8 is piperazin-1-yl, piperidin-4-yl or HN(R 3 )-C 1-8 alkyl, each of R 1 , R 2 , R 3 , X 1 , X 2 , X3 , X4 , X5 , X6 and X7 are as defined in the Summary of the Invention), followed by (if necessary) deprotection. In addition, wherein X 8 is 1,4-piperazinylene or 1,4-piperidinylene, and X 9 is a compound of formula I of -NHC(O)- or wherein X 8 is C 1-8 alkylene , and X 9 is -NHC(O)N(R 3 )-The compound of formula I can be obtained by combining a suitable compound of formula I or its protected derivative with an isocyanate of formula R 2 -NC(O) or Its protected derivatives were reacted and then (if necessary) deprotected to prepare (see Example 8 below for details).
相似地,其中X2为1,4-亚哌嗪基或1,4-亚哌啶基,而X1为-C(O)-、-OC(O)-或-N(R3)C(O)-的式Ⅰ化合物或者其中X2为C1-8亚烷基,而X1为-C(O)N(R3)-、-OC(O)N(R3)-或-N(R3)C(O)N(R3)-的式Ⅰ化合物可以通过使下式2的化合物或其受保护的衍生物2与一种式R1-Y1-C(O)L的化合物或其受保护的衍生物反应(其中L为一种离去基团,Y1为一键、-O-或-N(R3)-,Y2为哌嗪-1-基、哌啶-4-基或HN(R3)-C1-8烷基,每个R1、R2、R3、X3、X4、X5、X6、X7、X8和X9如发明概述中所定义)、然后(必要时)脱保护来制备。另外,其中X2为1,4-亚哌嗪基或1,4-亚哌啶基,而X1为-NHC(O)-的式Ⅰ化合物或者其中X2为C1-8亚烷基,而X1为-NHC(O)N(R3)-的式Ⅰ化合物可以通过使一种式2的化合物或其受保护的衍生物与一种式R1-NC(O)的异氰酸酯或其受保护的衍生物反应,然后(必要时)脱保护来制备(细节请参阅下面的实施例14(b))。Similarly, wherein X 2 is 1,4-piperazinylene or 1,4-piperidinylene, and X 1 is -C(O)-, -OC(O)- or -N(R 3 )C (O)- compound of formula I or wherein X 2 is C 1-8 alkylene, and X 1 is -C(O)N(R 3 )-, -OC(O)N(R 3 )- or - The formula I compound of N(R 3 )C(O)N(R 3 )- can be obtained by making the compound of the following formula 2 or its protected derivatives 2 reacting with a compound of formula R 1 -Y 1 -C(O)L or a protected derivative thereof (wherein L is a leaving group, Y 1 is a bond, -O- or -N( R 3 )-, Y 2 is piperazin-1-yl, piperidin-4-yl or HN(R 3 )-C 1-8 alkyl, each of R 1 , R 2 , R 3 , X 3 , X 4 , X 5 , X 6 , X 7 , X 8 and X 9 are as defined in the Summary of the Invention), followed by (if necessary) deprotection. In addition, wherein X 2 is 1,4-piperazinylene or 1,4-piperidinylene, and X 1 is a compound of formula I of -NHC(O)- or wherein X 2 is C 1-8 alkylene , and X 1 is -NHC(O)N(R 3 )-The compound of formula I can be obtained by combining a compound of formula 2 or a protected derivative thereof with an isocyanate of formula R 1 -NC(O) or Its protected derivatives were reacted and then (if necessary) deprotected to prepare (see Example 14(b) below for details).
其中R1=R2,X2和/或X8为1,4-亚哌嗪基或1,4-亚哌啶基,X1为-C(O)-、-OC(O)-或-N(R3)C(O)-,而X9为-C(O)-、-OC(O)-或-N(R3)C(O)-的式Ⅰ化合物和/或其中X2和/或X8为C1-8亚烷基,X1为-C(O)N(R3)-、-OC(O)N(R3)-或-N(R3)C(O)N(R3)-,而X9为-C(O)N(R3)-、-OC(O)N(R3)-或-N(R3)C(O)N(R3)-的式Ⅰ化合物可以通过使下式3的化合物或其受保护的衍生物:3与2当量或更多当量的一种式R1-Y1-C(O)L的化合物或其受保护的衍生物反应(其中L为一种离去基团,Y1为一键、-O-或-N(R3)-,Y2和Y8独立地为哌嗪-1-基、哌啶-4-基或HN(R3)-C1-8烷基,每个R1、R2、R3、X3、X4、X5、X6和X7如发明概述中所定义)、然后(必要时)脱保护来制备。另外,其中R1=R2,X2和/或X8为1,4-亚哌嗪基或1,4-亚哌啶基,X1为-NHC(O)-和/或X9为-NHC(O)-的式Ⅰ化合物和/或其中X2和/或X8为C1-8亚烷基,而X1为-NHC(O)N(R3)-和/或X9为-NHC(O)N(R3)-的式Ⅰ化合物可以通过使下式3的化合物或其受保护的衍生物与2当量或更多当量的一种式R1-NC(O)的异氰酸酯或其受保护的衍生物反应,然后(必要时)脱保护来制备(细节请参阅下面的实施例10)。Wherein R 1 =R 2 , X 2 and/or X 8 are 1,4-piperazinylene or 1,4-piperidinylene, X 1 is -C(O)-, -OC(O)- or -N(R 3 )C(O)-, and X 9 is a compound of formula I of -C(O)-, -OC(O)- or -N(R 3 )C(O)- and/or wherein X 2 and/or X 8 is C 1-8 alkylene, X 1 is -C(O)N(R 3 )-, -OC(O)N(R 3 )- or -N(R 3 )C( O)N(R 3 )-, and X 9 is -C(O)N(R 3 )-, -OC(O)N(R 3 )- or -N(R 3 )C(O)N(R 3 )-The compound of formula I can be obtained by making the compound of the following formula 3 or its protected derivatives: 3 react with 2 or more equivalents of a compound of formula R 1 -Y 1 -C(O)L or a protected derivative thereof (wherein L is a leaving group, Y 1 is a bond, -O- or -N(R 3 )-, Y 2 and Y 8 are independently piperazin-1-yl, piperidin-4-yl or HN(R 3 )-C 1-8 alkyl, each R 1 , R 2 , R 3 , X 3 , X 4 , X 5 , X 6 and X 7 are as defined in the Summary of the Invention), followed by (if necessary) deprotection. In addition, wherein R 1 =R 2 , X 2 and/or X 8 are 1,4-piperazinylene or 1,4-piperidinylene, X 1 is -NHC(O)- and/or X 9 is -NHC(O)-compound of formula I and/or wherein X 2 and/or X 8 are C 1-8 alkylene, and X 1 is -NHC(O)N(R 3 )- and/or X 9 The formula I compound of -NHC(O)N(R 3 )- can be obtained by making the compound of the following formula 3 or its protected derivatives with 2 equivalents or more equivalents of a kind of formula R 1- NC(O) Isocyanates or their protected derivatives are reacted and then (if necessary) deprotected to prepare (see Example 10 below for details).
其中X1为-N(R3)C(O)-、-N(R3)C(O)O-或-N(R3)C(O)N(R3)-的式Ⅰ化合物可以通过使一种式R1-N(R3)H的胺或其受保护的衍生物与一种下式4的化合物或其受保护的衍生物反应来制备:4其中L为一种离去基团,Y1为一键、-O-或-N(R3)-,而每个R1、R2、R3、X2、X3、X4、X5、X6、X7、X8和X9如发明概述中所定义(细节请参阅下面的实施例20)。The compound of formula I wherein X 1 is -N(R 3 )C(O)-, -N(R 3 )C(O)O- or -N(R 3 )C(O)N(R 3 )- can be Prepared by reacting an amine of formula R 1 -N(R 3 )H or a protected derivative thereof with a compound of formula 4 or a protected derivative thereof: 4 wherein L is a leaving group, Y 1 is a bond, -O- or -N(R 3 )-, and each of R 1 , R 2 , R 3 , X 2 , X 3 , X 4 , X5 , X6 , X7 , X8 and X9 are as defined in the Summary of the Invention (see Example 20 below for details).
其中X2为1,4-亚哌嗪基或4,1-亚哌啶基,而X3为-C(O)-、-OC(O)-或-C(O)N(R3)-的式Ⅰ化合物或者其中X2为C1-8亚烷基,而X3为-N(R3)C(O)-、-N(R3)C(O)O-或-N(R3)C(O)N(R3)-的式Ⅰ化合物可以通过使一种式R1-X1-Y2的化合物或其受保护的衍生物与一种下式5的化合物或其受保护的衍生物反应来制备:5其中L为一种离去基团,Y3为-键、-O-或-N(R3)-,Y2为哌嗪-1-基、哌啶-4-基或HN(R3)-C1-8烷基,而每个R1、R2、R3、X1、X2、X3、X4、X5、X6、X7、X8和X9如发明概述中所定义(细节请参阅下面的Wherein X 2 is 1,4-piperazinylene or 4,1-piperidinylene, and X 3 is -C(O)-, -OC(O)- or -C(O)N(R 3 ) - the compound of formula I or wherein X 2 is C 1-8 alkylene, and X 3 is -N(R 3 )C(O)-, -N(R 3 )C(O)O- or -N( The formula I compound of R 3 )C(O)N(R 3 )- can be obtained by combining a compound of formula R 1 -X 1 -Y 2 or a protected derivative thereof with a compound of formula 5 or its The protected derivative is reacted to prepare: 5 where L is a leaving group, Y 3 is - bond, -O- or -N(R 3 )-, Y 2 is piperazin-1-yl, piperidin-4-yl or HN(R3) -C 1-8 alkyl, and each R 1 , R 2 , R 3 , X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , X 8 and X 9 are as in the summary of the invention defined (see below for details
实施例31)。Example 31).
其中X2和X8各自为1,4-亚哌嗪基或4,1-亚哌啶基,而X3和X7独立地为-C(O)-、-OC(O)-或-C(O)N(R3)-的式Ⅰ化合物或者其中X2和X8各自为C1-8亚烷基或C1-8亚杂烷基,而X3和X7独立地为-N(R3)C(O)-、-N(R3)C(O)O-或-N(R3)C(O)N(R3)-的式Ⅰ化合物可以通过使2当量或更多当量的一种式R1-X1-Y2的化合物或其受保护的衍生物与一种下式6的化合物或其受保护的衍生物反应来制备:6其中L为一种离去基团,Y3和Y7独立地为一键、-O-或-N(R3)、Y2为哌嗪-1-基、哌啶-4-基、HN(R3)-C1-8烷基或HN(R3)-C1-8杂烷基,而每个R1、X1、X4、X5和X6如发明概述中所定义(细节请参阅下面的实施例32)。Wherein X 2 and X 8 are each 1,4-piperazinylene or 4,1-piperidinylene, and X 3 and X 7 are independently -C(O)-, -OC(O)- or - C(O)N(R 3 )- compound of formula I or wherein X 2 and X 8 are each C 1-8 alkylene or C 1-8 heteroalkylene, and X 3 and X 7 are independently - The formula I compound of N(R 3 )C(O)-, -N(R 3 )C(O)O- or -N(R 3 )C(O)N(R 3 )- can be obtained by making 2 equivalents or Prepared by reacting more equivalents of a compound of formula R 1 -X 1 -Y 2 or a protected derivative thereof with a compound of formula 6 or a protected derivative thereof: 6 wherein L is a leaving group, Y 3 and Y 7 are independently a bond, -O- or -N(R 3 ), Y 2 is piperazin-1-yl, piperidin-4-yl, HN(R 3 )-C 1-8 alkyl or HN(R 3 )-C 1-8 heteroalkyl, and each of R 1 , X 1 , X 4 , X 5 and X 6 is as defined in the Summary of the Invention (See Example 32 below for details).
上述的酰化反应可以通过使一种活化的酯(例如一种酰氯衍生物)和一种合适的亲核试剂在一种合适的有机碱(例如N,N-二异丙基乙胺(DIEA)、N-甲基吗啉等,优选DIEA)和合适的溶剂(例如N,N-二甲基甲酰胺(DMF)、四氢呋喃(THF)、二氯甲烷等)存在下,在20-30℃、典型地大约23℃下一起反应几分钟至24小时而完成。另外,酰化反应可以通过使一种合适的羧酸和亲核试剂在一种合适的偶合剂(例如1-(3-二甲基氨基丙基)-3-乙基碳化二亚胺等)和一种合适的溶剂(例如DMF等)存在下,在20-30℃、典型地大约23℃下一起反应几小时至几天而完成。上述的用于制备式Ⅰ化合物的反应及进行反应的条件是描述性的,本领域的普通技术人员将会认识到,制备本发明的化合物时可以使用其它的反应条件和不同的反应原料。The above-mentioned acylation reaction can be carried out by making an activated ester (such as a kind of acid chloride derivative) and a kind of suitable nucleophilic reagent in a kind of suitable organic base (such as N,N-diisopropylethylamine (DIEA ), N-methylmorpholine, etc., preferably DIEA) and a suitable solvent (such as N,N-dimethylformamide (DMF), tetrahydrofuran (THF), dichloromethane, etc.), at 20-30 ° C , typically at about 23°C for a few minutes to 24 hours to complete. Alternatively, the acylation reaction can be carried out by reacting a suitable carboxylic acid and a nucleophile in a suitable coupling agent (for example, 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide, etc.) and In the presence of a suitable solvent (such as DMF, etc.), the reaction is carried out at 20-30°C, typically about 23°C, for several hours to several days. The reactions and conditions under which the above-described reactions are used to prepare the compounds of formula I are descriptive, and one of ordinary skill in the art will recognize that other reaction conditions and different reaction starting materials can be used to prepare the compounds of the present invention.
脱保护可以用任何能除去保护基并以合理的产率得到所要产物的手段来进行。在T.W.greene,Protective Groups in Organic Synthesis,John Wiley & Sons,Inc.1981中可以找到用于产生保护基和去掉保护基的技术的详细描述。Deprotection can be carried out by any means which removes the protecting group and affords the desired product in reasonable yield. A detailed description of techniques for creating and removing protecting groups can be found in T.W. greene, Protective Groups in Organic Synthesis, John Wiley & Sons, Inc. 1981.
一般地,用于制备式Ⅰ化合物的反应原料和中间体可以从商业上得到或者可以很容易地被本领域的普通技术人员制备。例如用于制备式Ⅰ化合物的中间体可以用上述的酰化反应方便地制备。当反应原料中存在多个反应位点时,有必要使用合适的保护化学来把反应指向所要的反应位点。In general, the starting materials and intermediates used in the preparation of compounds of formula I are commercially available or can be easily prepared by those of ordinary skill in the art. For example, intermediates useful in the preparation of compounds of formula I may be conveniently prepared by the acylation reactions described above. When multiple reaction sites are present in a reaction starting material, it is necessary to use appropriate protection chemistry to direct the reaction to the desired reaction site.
一种制备其中X4和X6各自为-C(O)O-的式Ⅰ化合物的方便的反应原料是下式7的化合物:7其中每个X4、X5和X6如发明概述中所定义。例如其中L为氯的式6的化合物可以通过使相应的二醇(例如顺式-1,5-环辛二醇、反式-1,4-亚环己基二甲醇、1,4-亚苯基二甲醇等)与三光气反应来制备(细节请参阅下面的实施例5)。A convenient starting material for the preparation of compounds of formula I wherein X and X are each -C(O)O- is a compound of formula 7 below: 7 wherein each of X 4 , X 5 and X 6 is as defined in the Summary of the Invention. For example, the compound of formula 6 wherein L is chlorine can be obtained by making the corresponding diol (such as cis-1,5-cyclooctanediol, trans-1,4-cyclohexylenedimethanol, 1,4-phenylene dimethanol, etc.) with triphosgene (for details, please refer to Example 5 below).
可用于制备式Ⅰ的化合物而且其中含有脒基的中间体可以通过用溶于乙醇的氯化氢处理相应的腈,然后和氨反应来制备。制备式Ⅰ化合物的其它方法:Intermediates which are useful in the preparation of compounds of formula I and which contain an amidine group can be prepared by treating the corresponding nitrile with hydrogen chloride in ethanol followed by reaction with ammonia. Other methods for preparing compounds of formula I:
其中R4为胍基的式Ⅰ化合物可以通过使一种相应的其中R4为氨基的式Ⅰ的化合物与氨基氰反应来制备。反应可以通过使胺与氯化氢进行反应,然后使提纯的产物与过量的氨基氰在大约65℃下反应大约2小时而完成(细节请参阅下面的实施例15)。Compounds of formula I wherein R4 is guanidino may be prepared by reacting a corresponding compound of formula I wherein R4 is amino with cyanamide. The reaction can be accomplished by reacting the amine with hydrogen chloride and then reacting the purified product with excess cyanamide at about 65°C for about 2 hours (see Example 15 below for details).
通过使游离碱形式的式Ⅰ化合物与一种药学上可接受的无机酸或有机酸反应来制备药学上可接受的酸加合盐形式的式Ⅰ的化合物。另外,通过使游离酸形式的式Ⅰ化合物与一种药学上可接受的无机碱或有机碱反应来制备式Ⅰ化合物的药学上可接受的碱加合盐。在本申请的定义部分提出了适用于制备式Ⅰ化合物的药学上可接受的盐的无机和有机酸碱。另外,使用反应原料或中间体的盐可以制备盐形式的式Ⅰ化合物。A compound of formula I in the form of a pharmaceutically acceptable acid addition salt is prepared by reacting a compound of formula I in free base form with a pharmaceutically acceptable inorganic or organic acid. Alternatively, pharmaceutically acceptable base addition salts of compounds of formula I are prepared by reacting the free acid form of a compound of formula I with a pharmaceutically acceptable inorganic or organic base. Inorganic and organic acids and bases suitable for the preparation of pharmaceutically acceptable salts of compounds of formula I are set forth in the definitions section of this application. Alternatively, the compounds of formula I may be prepared in the form of salts using salts of the starting materials or intermediates.
游离酸或游离碱形式的式Ⅰ化合物可以从相应的碱加合盐或酸加合盐形式制备。例如,用一种合适的碱(例如氢氧化铵溶液、氢氧化钠溶液等)处理酸加合盐形式的式Ⅰ化合物可以使它转变为相应的游离碱,用一种合适的酸(例如盐酸等)处理碱加合盐形式的式Ⅰ化合物可以使它转变为相应的游离酸。Compounds of formula I in free acid or free base form may be prepared from the corresponding base or acid addition salt forms. For example, a compound of formula I in the form of an acid addition salt can be converted to the corresponding free base by treating it with a suitable base (such as ammonium hydroxide solution, sodium hydroxide solution, etc.) etc.) treatment of a compound of formula I in base addition salt form converts it to the corresponding free acid.
式Ⅰ化合物的N-氧化物可以用本领域普通技术人员已知的方法制备。例如,通过用一种氧化剂(例如三氟过乙酸、过马来酸、过苯甲酸、过乙酸、间-氯过氧苯甲酸等)在一种合适的惰性有机溶剂(例如卤代烃如二氯甲烷)中在大约0℃下处理未氧化形式的式Ⅰ化合物可以制备N-氧化物。另外,可以从一种合适的反应原料的N-氧化物可以制备式Ⅰ化合物的N-氧化物。N-oxides of compounds of formula I can be prepared by methods known to those of ordinary skill in the art. For example, by using an oxidizing agent (such as trifluoroperacetic acid, permaleic acid, perbenzoic acid, peracetic acid, m-chloroperoxybenzoic acid, etc.) in a suitable inert organic solvent (such as a halogenated hydrocarbon such as di The N-oxides can be prepared by treating the unoxidized form of a compound of formula I in (chloromethane) at about 0°C. Alternatively, an N-oxide of a compound of formula I can be prepared from an N-oxide of a suitable reaction starting material.
通过用一种还原剂(例如硫、二氧化硫、三苯基膦、硼氢化锂、硼氢化钠、三氯化磷、三溴化磷等)在一种合适的惰性有机溶剂(例如乙腈、乙醇、二氧六环水溶液等)中在0-80℃下处理,可以从式Ⅰ化合物的N-氧化物制备未氧化形式的式Ⅰ化合物。By using a reducing agent (such as sulfur, sulfur dioxide, triphenylphosphine, lithium borohydride, sodium borohydride, phosphorus trichloride, phosphorus tribromide, etc.) in a suitable inert organic solvent (such as acetonitrile, ethanol, The compound of formula I in unoxidized form can be prepared from the N-oxide of the compound of formula I by treating in aqueous solution of dioxane, etc.) at 0-80°C.
式Ⅰ化合物的药物前体衍生物可以用本领域普通技术人员已知的方法制备(例如细节请参阅Saulnier等人,(1994)Bioorganic and MedicinalChemistry Letters.4:1985)。例如合适的药物前体可以通过使一种未衍生的式Ⅰ化合物与一种合适的氨基甲酰化剂(例如1,1-酰氧基烷基碳酰氯、碳酸对硝基苯基酯)反应来制备。Prodrug derivatives of compounds of formula I may be prepared by methods known to those of ordinary skill in the art (see, eg, Saulnier et al., (1994) Bioorganic and Medicinal Chemistry Letters. 4:1985 for details). For example, suitable prodrugs can be obtained by reacting an underivatized compound of formula I with a suitable carbamoylating agent (for example, 1,1-acyloxyalkyl carbonyl chloride, p-nitrophenyl carbonate) to prepare.
式Ⅰ化合物的受保护的衍生物可以用本领域普通技术人员已知的方法制备。在T.W.greene,Protective Groups in Organic Synthesis,JohnWiley & Sons,Inc.1981中可以找到用于产生保护基和去掉保护基的技术的详细描述。Protected derivatives of compounds of formula I can be prepared by methods known to those of ordinary skill in the art. A detailed description of techniques for creating and removing protecting groups can be found in T.W. greene, Protective Groups in Organic Synthesis, John Wiley & Sons, Inc. 1981.
总之,本发明的一个方面是一种制备式Ⅰ化合物的方法,该方法包括:In summary, one aspect of the present invention is a process for the preparation of compounds of formula I, the process comprising:
(a)使一种式1的化合物或其受保护的衍生物:1与一种式R2-Y9-C(O)L的化合物或其受保护的衍生物反应(其中L为一种离去基团,Y9为一键、-O-或-N(R3)-,Y8为哌嗪-1-基、哌啶-4-基或HN(R3)-C1-8烷基,每个R1、R2、R3、X1、X2、X3、X4、X5、X6和X7如发明概述中所定义)、然后(必要时)脱保护,制得其中X8为1,4-亚哌嗪基或1,4-亚哌啶基,而X9为-C(O)-、-OC(O)-或-N(R3)C(O)-的式Ⅰ化合物或者其中X8为C1-8亚烷基,而X9为-C(O)N(R3)-、-OC(O)N(R3)-或-NHC(O)N(R3)-的式Ⅰ化合物;(a) a compound of formula 1 or a protected derivative thereof: 1 reacting with a compound of formula R 2 -Y 9 -C(O)L or a protected derivative thereof (wherein L is a leaving group, Y 9 is a bond, -O- or -N( R 3 )-, Y 8 is piperazin-1-yl, piperidin-4-yl or HN(R 3 )-C 1-8 alkyl, each of R 1 , R 2 , R 3 , X 1 , X 2 , X 3 , X 4 , X 5 , X 6 and X 7 are as defined in the Summary of the Invention), followed by (if necessary) deprotection to obtain wherein X 8 is 1,4-piperazinylene or 1,4 -Piperidinylene, and X 9 is a compound of formula I of -C(O)-, -OC(O)- or -N(R 3 )C(O)- or wherein X 8 is C 1-8 alkylene group, and X 9 is a compound of formula I of -C(O)N(R 3 )-, -OC(O)N(R 3 )- or -NHC(O)N(R 3 )-;
(b)使一种式1的化合物或其受保护的衍生物与与式R2-NC(O)的异氰酸酯或其受保护的衍生物反应,然后(必要时)脱保护,制得其中X8为1,4-亚哌嗪基或1,4-亚哌啶基,而X9为-NHC(O)-的式Ⅰ化合物或者其中X8为C1-8亚烷基,而X9为-NHC(O)N(R3)-的式Ⅰ化合物;(b) reacting a compound of formula 1 or a protected derivative thereof with an isocyanate of formula R 2 -NC(O) or a protected derivative thereof, followed by (if necessary) deprotection, to obtain wherein X 8 is 1,4-piperazinyl or 1,4-piperidinyl, and X 9 is a compound of formula I of -NHC (O)- or wherein X 8 is C 1-8 alkylene, and X 9 A compound of formula I which is -NHC(O)N(R 3 )-;
(c)使一种下式2的化合物或其受保护的衍生物2与一种式R1-Y1-C(O)L的化合物或其受保护的衍生物反应(其中L为一种离去基团,Y1为一键、-O-或-N(R3)-,Y2为哌嗪-1-基、哌啶-4-基或HN(R3)-C1-8烷基,而每个R1、R2、R3、X3、X4、X5、X6、X7、X8和X9如发明概述中所定义)、然后(必要时)脱保护,制得其中X2为1,4-亚哌嗪基或1,4-亚哌啶基,而X1为-C(O)-、-OC(O)-或-N(R3)C(O)-的式Ⅰ化合物或者其中X2为C1-8亚烷基,而X1为-C(O)N(R3)-、-OC(O)N(R3)-或-NHC(O)N(R3)-的式Ⅰ化合物;(c) make a compound of the following formula 2 or its protected derivatives 2 reacting with a compound of formula R 1 -Y 1 -C(O)L or a protected derivative thereof (wherein L is a leaving group, Y 1 is a bond, -O- or -N( R 3 )-, Y 2 is piperazin-1-yl, piperidin-4-yl or HN(R 3 )-C 1-8 alkyl, and each R 1 , R 2 , R 3 , X 3 , X 4 , X 5 , X 6 , X 7 , X 8 and X 9 are as defined in the Summary of the Invention), followed by (if necessary) deprotection to obtain wherein X 2 is 1,4-piperazinylene or 1, 4-piperidinylene, and X 1 is a compound of formula I of -C(O)-, -OC(O)- or -N(R 3 )C(O)- or wherein X 2 is C 1-8 Alkyl, and X 1 is a compound of formula I of -C(O)N(R 3 )-, -OC(O)N(R 3 )- or -NHC(O)N(R 3 )-;
(d)使下式3的化合物或其受保护的衍生物:3与2当量或更多当量的一种式R1-Y1-C(O)L的化合物或其受保护的衍生物反应(其中L为一种离去基团,Y1为一键、-O-或-N(R3)-,Y2和Y8独立地为哌嗪-1-基、哌啶-4-基或HN(R3)-C1-8烷基,而每个R1、R2、R3、X3、X4、X5、X6和X7、如发明概述中所定义)反应,然后(必要时)脱保护,制得其中R1=R2,X2和/或X8为1,4-亚哌嗪基或1,4-亚哌啶基,X1为-C(O)-、-OC(O)-、-N(R3)C(O)-和/或X9为-C(O)-、-OC(O)-、-N(R3)C(O)-的式Ⅰ化合物和/或其中X2和/或X8为C1-8亚烷基,X1为-C(O)N(R3)-、-OC(O)N(R3)-或-N(R3)C(O)N(R3)-,而X9为-C(O)N(R3)-、-OC(O)N(R3)-或-N(R3)C(O)N(R3)-的式Ⅰ化合物;(d) a compound of the following formula 3 or a protected derivative thereof: 3 react with 2 or more equivalents of a compound of formula R 1 -Y 1 -C(O)L or a protected derivative thereof (wherein L is a leaving group, Y 1 is a bond, -O- or -N(R 3 )-, Y 2 and Y 8 are independently piperazin-1-yl, piperidin-4-yl or HN(R 3 )-C 1-8 alkyl, and each R 1 , R 2 , R 3 , X 3 , X 4 , X 5 , X 6 and X 7 , as defined in the Summary of the Invention) react, followed (if necessary) by deprotection, to produce wherein R 1 =R 2 , X 2 and/or X 8 are 1,4-piperazinylene or 1,4-piperidinylene, X 1 is -C(O)-, -OC(O)-, -N(R 3 )C (O)- and/or X 9 are -C(O)-, -OC(O)-, -N(R 3 )C(O)- compounds of formula I and/or wherein X 2 and/or X 8 is C 1-8 alkylene, X 1 is -C(O)N(R 3 )-, -OC(O)N(R 3 )- or -N(R 3 )C(O)N(R 3 )-, and X 9 is the formula I of -C(O)N(R 3 )-, -OC(O)N(R 3 )- or -N(R 3 )C(O)N(R 3 )- compound;
(f)使式3的化合物或其受保护的衍生物与2当量或更多当量的一种式R1-NC(O)的异氰酸酯或其受保护的衍生物反应,然后(必要时)脱保护,制得其中R1=R2,X2和/或X8为1,4-亚哌嗪基或1,4-亚哌啶基,X1为-NHC(O)-和/或X9为-NHC(O)-的式Ⅰ化合物和/或其中X2和/或X8为C1-8亚烷基,而X1为-NHC(O)N(R3)-和/或X9为-NHC(O)N(R3)-的式Ⅰ化合物;(f) reacting a compound of formula 3 or a protected derivative thereof with 2 equivalents or more of an isocyanate of formula R 1 -NC(O) or a protected derivative thereof, and then (if necessary) removing Protection, wherein R 1 = R 2 , X 2 and/or X 8 are 1,4-piperazinyl or 1,4-piperidinyl, X 1 is -NHC(O)- and/or X 9 is a compound of formula I of -NHC(O)- and/or wherein X 2 and/or X 8 are C 1-8 alkylene, and X 1 is -NHC(O)N(R 3 )- and/or X 9 is a compound of formula I of -NHC(O)N(R 3 )-;
(g)使一种式R1-N(R3)H的胺或其受保护的衍生物与一种下式4的化合物或其受保护的衍生物:4(其中L为一种离去基团,Y1为一键、-O-或-N(R3)-,而每个R1、R2、R3、X2、X3、X4、X5、X6、X7、X8和X9如发明概述中所定义)反应,然后(必要时)脱保护,制得其中X1为-N(R3)C(O)-、-N(R3)C(O)O-或-N(R3)C(O)N(R3)-的式Ⅰ化合物;(g) combining an amine of formula R 1 -N(R 3 )H or a protected derivative thereof with a compound of formula 4 or a protected derivative thereof: 4 (where L is a leaving group, Y 1 is a bond, -O- or -N(R 3 )-, and each R 1 , R 2 , R 3 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , X 8 and X 9 as defined in the Summary of the Invention) reaction, followed by (if necessary) deprotection, to obtain wherein X 1 is -N(R 3 )C(O)-, -N(R 3 )C(O)O- or -N(R 3 )C(O)N(R 3 )- compounds of formula I;
(h)使一种式R1-X1-Y2的化合物或其受保护的衍生物与一种下式5的化合物或其受保护的衍生物:5(其中L为一种离去基团,Y3为一键、-O-或-N(R3)-,Y2为哌嗪-1-基、哌啶-4-基或HN(R3)-C1-8烷基,而每个R1、R2、R3、X1、X2、X3、X4、X5、X6、X7、X8和X5如发明概述中所定义)反应,然后(必要时)脱保护,制得其中X2为1,4-亚哌嗪基或4,1-亚哌啶基,而X3为-C(O)-、-C(O)O-或C(O)N(R3)的式Ⅰ化合物,或其中X2是C1-8亚烷基,X3是-N(R3)C(O)-、-N(R3)C(O)O-或-N(R3)C(O)N(R3)-的式Ⅰ化合物;(h) a compound of formula R 1 -X 1 -Y 2 or a protected derivative thereof and a compound of the following formula 5 or a protected derivative thereof: 5 (where L is a leaving group, Y 3 is a bond, -O- or -N(R 3 )-, Y 2 is piperazin-1-yl, piperidin-4-yl or HN(R 3 )-C 1-8 alkyl, and each R 1 , R 2 , R 3 , X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , X 8 and X 5 are as invented as defined in the overview), followed by (if necessary) deprotection to obtain wherein X 2 is 1,4-piperazinylene or 4,1-piperidinylene and X 3 is -C(O)-, -C(O)O- or C(O)N(R 3 ) formula I compound, or wherein X 2 is C 1-8 alkylene, X 3 is -N(R 3 )C(O)-, -N(R 3 )C(O)O- or -N(R 3 )C(O)N(R 3 )- compounds of formula I;
(i)使2当量或更多当量的一种式R1-X1-Y2的化合物或其受保护的衍生物与一种下式6的化合物或其受保护的衍生物:6(其中L为一种离去基团,Y3和Y7独立地为一键、-O-或-N(R3)-,Y2为哌嗪-1-基、哌啶-4-基、HN(R3)-C1-8烷基或HN(R3)-C1-8杂烷基,而每个R1、X1、X4、X5和X6如发明概述中所定义)反应,然后(必要时)脱保护,制得其中X2和X8各自为1,4-亚哌嗪基或4,1-亚哌啶基,而X3和X7独立地为-C(O)-、-C(O)O-或-C(O)N(R3)-的式Ⅰ化合物或者其中X2和X8各自为C1-8亚烷基或C1-8亚杂烷基,而X3和X7各自独立地为-N(R3)C(O)-、-N(R3)C(O)O-或-N(R3)C(O)N(R3)-的式Ⅰ化合物;(i) 2 equivalents or more of a compound of formula R 1 -X 1 -Y 2 or a protected derivative thereof and a compound of the following formula 6 or a protected derivative thereof: 6 (where L is a leaving group, Y 3 and Y 7 are independently a bond, -O- or -N(R 3 )-, Y 2 is piperazin-1-yl, piperidine-4- group, HN(R 3 )-C 1-8 alkyl or HN(R 3 )-C 1-8 heteroalkyl, and each of R 1 , X 1 , X 4 , X 5 and X 6 is as described in the Summary of the Invention defined) reaction, followed by (if necessary) deprotection to obtain wherein X 2 and X 8 are each 1,4-piperazinylene or 4,1-piperidinylene, and X 3 and X 7 are independently -C(O)-, -C(O)O- or -C(O)N(R 3 )- compound of formula I or wherein X 2 and X 8 are each C 1-8 alkylene or C 1- 8 Heteroalkylene, and X 3 and X 7 are each independently -N(R 3 )C(O)-, -N(R 3 )C(O)O- or -N(R 3 )C(O) )N(R 3 )- compound of formula I;
(j)选择性地使一种其中R4为氨基的式Ⅰ化合物与氨基氰反应,制备其中R4为胍基的式Ⅰ化合物;(j) selectively reacting a compound of formula I wherein R is an amino group with cyanamide to prepare a compound of formula I wherein R is a guanidine group;
(k)选择性地把一种式Ⅰ化合物转变为其药学上可接受的盐;(k) selectively converting a compound of formula I into a pharmaceutically acceptable salt thereof;
(l)选择性地把一种盐形式的式Ⅰ化合物转变为非盐形式;(1) selectively converting a compound of formula I in a salt form into a non-salt form;
(m)选择性地把一种未氧化形式的式Ⅰ化合物转变为其药学上可接受N-氧化物;(m) selectively converting an unoxidized form of a compound of formula I to its pharmaceutically acceptable N-oxide;
(n)选择性地把一种N-氧化物形式的式Ⅰ化合物转变为其未氧化形式;(n) selectively converting a compound of formula I in the form of an N-oxide into its unoxidized form;
(o)选择性地把一种式Ⅰ化合物的非衍生物形式转变为其药物前体衍生物;(o) selectively converting a non-derivative form of a compound of formula I into a prodrug derivative thereof;
(p)选择性地把一种式Ⅰ化合物的药物前体衍生物转变为其非衍生物形式。(p) selectively converting a prodrug derivative of a compound of formula I into its non-derivative form.
在上述的方法中,所说的式Ⅰ是指其中每个R1、R2、R3、X1、X2、X3、X4、X5、X6、X7、X8和X9具有发明概述中所提出的最宽定义的式Ⅰ,这些方法特别适用于本发明的优选实施例。In the above method, said formula I means that each of R 1 , R 2 , R 3 , X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , X 8 and X 9 has Formula I with the broadest definition set forth in the Summary of the Invention, and these methods are particularly applicable to preferred embodiments of the invention.
实施例14-氨基苄基氨基甲酸叔丁基酯盐酸盐Embodiment 14-tert-butyl aminobenzylcarbamate hydrochloride
把溶于二氯甲烷(200ml)中的4-氨基苄基胺(50.34g,0.412mol)加入到1升的装有机械搅拌的3颈园底烧瓶中,将溶液冷却到0℃,用30分钟往上述溶液中滴加溶于二氯甲烷(200ml)中的碳酸二叔丁基酯(89.9g,0.412mol),在0℃下搅拌所得的悬浮液2小时,得到几乎均匀的溶液。随后用氢氧化钠水溶液(1.0M,500ml)和水(500ml)洗涤该二氯甲烷溶液,干燥有机层(MgSO4),过滤并真空浓缩,得到黄色油状物。将该油状物溶于乙醚∶甲醇(2∶1,225ml)中,并将溶液冷却到0℃,用HCl的二噁烷溶液(4.0M,115ml,0.412mol)酸化,与乙醚(200ml)混合,得到浓的浅黄色沉淀,过滤收集沉淀,再用乙醚(500ml)洗涤,真空干燥,得到4-氨基苄基氨基甲酸叔丁基酯(100.23g,0.387mol,产率94%),为浅黄色固体。1H-NMR(300MHz,DMSO-d6):10.40-10.20(br,s,3H),7.40(tr,1H),7.30(s,4H),4.10(d,2H),1.40(s,9H)。Add 4-aminobenzylamine (50.34 g, 0.412 mol) dissolved in dichloromethane (200 ml) into a 1-liter 3-necked round-bottomed flask equipped with mechanical stirring, and cool the solution to 0 ° C with 30 Di-tert-butyl carbonate (89.9 g, 0.412 mol) dissolved in dichloromethane (200 ml) was added dropwise to the above solution within minutes, and the resulting suspension was stirred at 0° C. for 2 hours to obtain an almost uniform solution. The dichloromethane solution was then washed with aqueous sodium hydroxide (1.0M, 500ml) and water (500ml), and the organic layer was dried ( MgSO4 ), filtered and concentrated in vacuo to give a yellow oil. The oil was dissolved in ether:methanol (2:1, 225ml), and the solution was cooled to 0°C, acidified with HCl in dioxane (4.0M, 115ml, 0.412mol), mixed with ether (200ml), A thick pale yellow precipitate was obtained, which was collected by filtration, washed with diethyl ether (500ml), and dried in vacuo to obtain tert-butyl 4-aminobenzylcarbamate (100.23g, 0.387mol, yield 94%) as light yellow solid. 1 H-NMR (300MHz,DMSO-d 6 ):10.40-10.20(br,s,3H),7.40(tr,1H),7.30(s,4H),4.10(d,2H),1.40(s,9H ).
实施例24-胍基苄基氨基甲酸叔丁基酯盐酸盐Example 24-guanidinobenzylcarbamate tert-butyl ester hydrochloride
把氨基腈(100g,2.4mol)加入到500mL园底烧瓶中,加热到60-65℃,直到完全熔融,将实施例1得到的4-氨基苄基氨基甲酸叔丁基酯盐酸盐(25.3g,97.8mmol.)直接加入到液体的氨基腈中,得到黄色溶液。于60-65℃下将溶液搅拌2小时,加入水(100ml),把水混合物冷却到室温,用乙醚(1L)洗涤,有机相用水反萃取(2X100ml),合并的水层用乙醚(500ml)洗涤,在冰水浴中冷却,然后用氢氧化钠水溶液(10M,100ml)碱化,得到慢慢结晶的不溶油状物,过滤收集结晶,用水洗涤,真空干燥,得到4-胍基苄基氨基甲酸叔丁基酯(18.3g,69.24mmol,产率70.8%),为无色结晶固体。1H-NMR(300MHz,DMSO-d6):9.70(s,1H),7.42(tr,1H),7.40(s,4H),7.25(d,2H),7.15(d,2H),4.10(d,2H),1.40(s,9H)Aminonitrile (100g, 2.4mol) was added to a 500mL round-bottomed flask, heated to 60-65°C until completely molten, and the tert-butyl 4-aminobenzylcarbamate hydrochloride (25.3 g, 97.8mmol.) was directly added to the liquid aminonitrile to obtain a yellow solution. The solution was stirred at 60-65°C for 2 hours, water (100ml) was added, the water mixture was cooled to room temperature, washed with diethyl ether (1L), the organic phase was back extracted with water (2X100ml), and the combined aqueous layer was washed with diethyl ether (500ml) Wash, cool in an ice-water bath, and then basify with aqueous sodium hydroxide solution (10M, 100ml) to obtain an insoluble oil that slowly crystallizes, collect the crystals by filtration, wash with water, and dry in vacuo to obtain 4-guanidinobenzylcarbamate Tert-butyl ester (18.3 g, 69.24 mmol, 70.8% yield), a colorless crystalline solid. 1 H-NMR (300MHz, DMSO-d 6 ): 9.70(s,1H), 7.42(tr,1H), 7.40(s,4H), 7.25(d,2H), 7.15(d,2H), 4.10( d,2H),1.40(s,9H)
实施例34-氯羰基-1-哌嗪甲酸叔丁基酯Example 34-Chlorocarbonyl-1-piperazinecarboxylic acid tert-butyl ester
把三光气(25g,84.2mmol)溶解于二氯甲烷(200ml)中,所得溶液冷却到0℃,将叔丁基1-哌嗪甲酸酯(40g,214.8mmol)和吡啶(35ml,432.7mmol)溶于二氯甲烷(100ml)中的混合物滴加到三光气的溶液中,用30分钟使混合物温热到室温,混合物用盐酸水溶液(0.1N,200ml)淬灭,水相用二氯甲烷(50ml)洗涤,干燥合并的有机相(MgSO4)并过滤,真空浓缩,得到4-氯羰基-1-哌嗪甲酸叔丁基酯(45.6g,71.6mmol,产率85%),为黄色固体。1H-NMR(300MHz,CDCl3):3.70(m,2H),3.60(m,2H),3.50(m,4H),1.50(s,9H)。Dissolve triphosgene (25g, 84.2mmol) in dichloromethane (200ml), the resulting solution was cooled to 0°C, and tert-butyl 1-piperazinecarboxylate (40g, 214.8mmol) and pyridine (35ml, 432.7mmol ) dissolved in dichloromethane (100ml) was added dropwise to the solution of triphosgene, the mixture was warmed to room temperature in 30 minutes, the mixture was quenched with aqueous hydrochloric acid (0.1N, 200ml), and the aqueous (50ml), dried the combined organic phases ( MgSO4 ) and filtered, concentrated in vacuo to give tert-butyl 4-chlorocarbonyl-1-piperazinecarboxylate (45.6g, 71.6mmol, 85% yield) as yellow solid. 1 H-NMR (300 MHz, CDCl 3 ): 3.70 (m, 2H), 3.60 (m, 2H), 3.50 (m, 4H), 1.50 (s, 9H).
实施例44-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸叔丁基酯三氟乙酸盐Example 44-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylic acid tert-butyl ester trifluoroacetate
在室温下用三氟乙酸(TFA,100ml)处理实施例2制备的叔丁基4-胍基苄基氨基甲酸酯(41.77g,0.158mol)30分钟,所得几乎无色的液体于45℃下真空浓缩,残留物用乙醚(3X400ml)研制、真空干燥得到无色的泡沫状物,将残留物溶解于甲醇中(200ml),往溶液中加入DIEA(55ml,0.32mol,根据估计的过量的TFA而确定数量),将混合物冷却到0℃,然后加入实施例3制备的溶于二氯甲烷(120ml)中的4-氯羰基-1-哌嗪甲酸叔丁基酯(39.3g,0.158mol),再加入一定量的DIEA(30ml),使反应混合物温热至室温,搅拌12小时,真空浓缩得到橙色油状物,将油状物和水(200ml)混合,得到浓的沉淀。该沉淀从乙腈和乙醚中重结晶,得到4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸叔丁基酯三氟乙酸盐(62.0g.0.126mol,产率80%),为浅黄色固体。1H-NMR(300MHz,DMSO-d6):10.15(s,1H),9.10(brs,2H),7.65(s,4H),7.40(tr,1H),7.25(dd,AB,4H),4.25(d,2H),3.55(m,4H),3.10(s,4H);The tert-butyl 4-guanidinobenzylcarbamate (41.77g, 0.158mol) prepared in Example 2 was treated with trifluoroacetic acid (TFA, 100ml) at room temperature for 30 minutes, and the resulting almost colorless liquid was Concentrate under vacuum, triturate the residue with diethyl ether (3×400ml) and dry in vacuo to obtain a colorless foam, dissolve the residue in methanol (200ml), add DIEA (55ml, 0.32mol, according to the estimated excess TFA to determine the quantity), the mixture was cooled to 0°C, and then tert-butyl 4-chlorocarbonyl-1-piperazinecarboxylate (39.3g, 0.158mol) dissolved in dichloromethane (120ml) prepared in Example 3 was added. ), and a certain amount of DIEA (30ml) was added, the reaction mixture was warmed to room temperature, stirred for 12 hours, and concentrated in vacuo to obtain an orange oil, which was mixed with water (200ml) to obtain a thick precipitate. The precipitate was recrystallized from acetonitrile and ether to obtain tert-butyl 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate trifluoroacetate (62.0g.0.126mol, yield 80% ), as a pale yellow solid. 1 H-NMR(300MHz,DMSO-d 6 ):10.15(s,1H),9.10(brs,2H),7.65(s,4H),7.40(tr,1H),7.25(dd,AB,4H), 4.25(d,2H),3.55(m,4H),3.10(s,4H);
电子喷雾法LRMS:计算值(C13H20N6O):MH+:277.4;MH2 +2/2;139.2;Electron spray method LRMS: calculated value (C 13 H 20 N 6 O): MH + : 277.4; MH 2 +2 /2 ; 139.2;
测定值:MH+:277.4;MH2 +2/2 139.3;Measured values: MH + : 277.4; MH 2 +2 /2 139.3;
实施例5顺-1,5-亚环辛基二(氯代甲酸酯)Example 5 cis-1,5-cyclooctylene bis(chloroformate)
把顺-1,5-环辛烷二醇(20.2g,0.14mol)溶于乙腈(250ml)中,往混合物中加入碳酸钾(41.4g,0.3mol),得到悬浮液,在氮气氛下将悬浮液冷却到0℃,在1小时内滴加光气(1.9M,甲苯中,220ml,0.42mol),将悬浮液温热到室温,搅拌12小时,然后加入1L乙醚,过滤出悬浮液中的不溶盐,浓缩。残留物用己烷重结晶,得到顺-1,5-亚环辛基二(氯代甲酸酯),为无色结晶固体。进一步用硅胶快速色谱法提纯,用己烷∶乙醚作为洗脱剂(10∶1)。1H-NMR(300MHz,CDCl3):5.00-4.85(m,2H),2.20-1.60(m,12H)。Dissolve cis-1,5-cyclooctanediol (20.2g, 0.14mol) in acetonitrile (250ml), add potassium carbonate (41.4g, 0.3mol) to the mixture to obtain a suspension, and dissolve under a nitrogen atmosphere The suspension was cooled to 0°C, phosgene (1.9M, in toluene, 220ml, 0.42mol) was added dropwise within 1 hour, the suspension was warmed to room temperature, stirred for 12 hours, then 1L ether was added, and the suspension was filtered insoluble salt, concentrated. The residue was recrystallized from hexane to give cis-1,5-cyclooctylene bis(chloroformate) as a colorless crystalline solid. It was further purified by flash chromatography on silica gel using hexane:ether as eluent (10:1). 1 H-NMR (300MHz, CDCl 3 ): 5.00-4.85 (m, 2H), 2.20-1.60 (m, 12H).
实施例6顺-1,5-亚环辛基氯代甲酸酯4-叔丁氧羰基-1-哌嗪甲酸酯Example 6 cis-1,5-cyclooctylene chloroformate 4-tert-butoxycarbonyl-1-piperazinecarboxylate
把溶于二氯甲烷(25ml)中的实施例5制备的顺-1,5-亚环辛基二(氯代甲酸酯)(1.91g,7.1mmol)滴加到溶于二氯甲烷(25ml)中的1-哌嗪甲酸叔丁基酯(1.3g,7.1mmol)和DIEA(1.3g,7.1mmol)的混合物中,室温搅拌混合物15分钟,用0.1M盐酸水溶液处理,干燥二氯甲烷层(MgSO4),过滤并浓缩,残留物用硅胶快速色谱法提纯,用乙醚和己烷作为洗脱剂,得到顺-1,5-亚环辛基氯代甲酸酯4-叔丁氧羰基-1-哌嗪甲酸酯(660mg,1.6mmol.产率22%)。1H-NMR(300MHz,CDCl3):5.00-4.90(m,1H),4.80-4.70(m,1H),3.40(s,8H),2.05-1.40(m,12H),1.40(s,9H)。The cis-1,5-cyclooctylene bis(chloroformate) (1.91g, 7.1mmol) prepared in Example 5 dissolved in dichloromethane (25ml) was added dropwise to dichloromethane ( In a mixture of tert-butyl-1-piperazinecarboxylate (1.3g, 7.1mmol) and DIEA (1.3g, 7.1mmol) in 25ml), the mixture was stirred at room temperature for 15 minutes, treated with 0.1M aqueous hydrochloric acid, and dried in dichloromethane layer (MgSO 4 ), filtered and concentrated, and the residue was purified by flash chromatography on silica gel using diethyl ether and hexane as eluents to give cis-1,5-cyclooctylene chloroformate 4-tert-butoxycarbonyl - 1-Piperazinecarboxylate (660 mg, 1.6 mmol. Yield 22%). 1 H-NMR(300MHz, CDCl 3 ):5.00-4.90(m,1H),4.80-4.70(m,1H),3.40(s,8H),2.05-1.40(m,12H),1.40(s,9H ).
实施例7顺-1,5-亚环辛基-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-叔丁氧羰基-1-哌嗪甲酸酯Example 7 cis-1,5-cyclooctylene-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-tert-butoxycarbonyl-1-piperazinecarboxylate
在室温下用纯三氟乙酸(1ml)处理实施例4制备的4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸叔丁基酯三氟乙酸盐(383.7g,1.06mmol)10分钟,真空浓缩混合物,得到无色油状物,将油状物溶于水(15ml),滴加5M氢氧化钠水溶液,将pH调节得到7-8,把溶于THF(10ml)中的实施例6制备的顺-1,5-亚环辛基氯代甲酸酯4-叔丁氧羰基-1-哌嗪甲酸酯(444.7mg,1.06mmol)加入到上述水溶液中,滴加1M氢氧化钠水溶液继续调节pH,直到pH没有变化,真空浓缩混合物,除去大部分的THF,然后加入乙醚(5ml)和5M氢氧化钠水溶液(足以把pH调节到14),得到稠的白色悬浮液。室温静置悬浮液15-30分钟,然后过滤收集沉淀,用水(2X15ml)洗涤。真空干燥得到顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-叔丁氧羰基-1-哌嗪甲酸酯(527mg,0.82mmol,产率77%),为无色固体。Treat tert-butyl 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate trifluoroacetate (383.7 g, 1.06 g) prepared in Example 4 with pure trifluoroacetic acid (1 ml) at room temperature mmol) for 10 minutes, the mixture was concentrated in vacuo to obtain a colorless oil, the oil was dissolved in water (15ml), and 5M aqueous sodium hydroxide solution was added dropwise to adjust the pH to 7-8. cis-1,5-cyclooctylene chloroformate 4-tert-butoxycarbonyl-1-piperazinecarboxylate (444.7mg, 1.06mmol) prepared in Example 6 was added to the above aqueous solution, and 1M Aqueous sodium hydroxide solution continued to adjust the pH until there was no change in pH and the mixture was concentrated in vacuo to remove most of the THF, then diethyl ether (5 mL) and 5M aqueous sodium hydroxide (enough to adjust the pH to 14) were added to give a thick white suspension . The suspension was allowed to stand at room temperature for 15-30 minutes, then the precipitate was collected by filtration and washed with water (2X15ml). Vacuum drying obtains cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-tert-butoxycarbonyl-1-piperazinecarboxylate (527mg , 0.82 mmol, yield 77%), as a colorless solid.
1H-NMR(300MHz,DMSO-d6):7.05(d,2H),7.00(tr,1H),6.70(d,2H),5.10(br,3H),4.65(m,2H),4.15(d,2H),3.30(s,16H),1.90-1.40(m,12H),1.40(s,9H)。 1 H-NMR (300MHz, DMSO-d 6 ): 7.05(d,2H),7.00(tr,1H),6.70(d,2H),5.10(br,3H),4.65(m,2H),4.15( d,2H), 3.30(s,16H), 1.90-1.40(m,12H), 1.40(s,9H).
实施例8顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯三氟乙酸盐(化合物1)Example 8 Cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarbamate 4(2-piperidin-4-ylethylcarbamoyl) -1-piperazinecarboxylate trifluoroacetate (compound 1)
以下制备式(Ⅰ)化合物,其中R1是4-胍基苄基,R2是2-哌啶-4-基乙基,X1和X9每个是-NHC(O)-,X2和X8每个是1,4-亚哌嗪基,X3和X7每个是-C(O)O-,X4和X6每个是共价键,X5是顺-1,5-亚环辛基。The compound of formula (I) is prepared as follows, wherein R 1 is 4-guanidinobenzyl, R 2 is 2-piperidin-4-ylethyl, X 1 and X 9 are each -NHC(O)-, X 2 and X 8 are each 1,4-piperazinylene, X 3 and X 7 are each -C(O)O-, X 4 and X 6 are each a covalent bond, X 5 is cis-1, 5-Cyclooctylene.
实施例7制备的顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-叔丁氧羰基-1-哌嗪甲酸酯(818mg,1.24mmol)用纯TFA(2ml)处理10分钟,混合物真空浓缩,得到无色油状物,残留物用乙醚(2X10ml)研制,真空干燥得到无色泡沫状物。然后将残留物溶于DMF(2ml),加入DIEA(700ml,4.0mmol)和叔丁基4-(2-异氰基乙基)-1-哌啶甲酸酯(3.2ml,0.39M,DMF中,1.25mmol),将混合物搅拌12小时,真空浓缩,残留物用水研制(2X,5ml),真空干燥得到黄色固体,该固体用TFA(2ml)处理,真空浓缩混合物,,残留物溶于水,通过制备性的反相HPLC从含水混合物中提纯,得到顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯,为无定型无色固体。等离子体吸收LRMS:计算值(C35H55N10O6):MH+:712.9;测定值MH+:713.2。Cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-tert-butoxycarbonyl-1-piperazinecarboxylate prepared in Example 7 (818mg, 1.24mmol) was treated with pure TFA (2ml) for 10 minutes, the mixture was concentrated in vacuo to give a colorless oil, the residue was triturated with ether (2X10ml) and dried in vacuo to give a colorless foam. The residue was then dissolved in DMF (2ml), DIEA (700ml, 4.0mmol) and tert-butyl 4-(2-isocyanoethyl)-1-piperidinecarboxylate (3.2ml, 0.39M, DMF medium, 1.25mmol), the mixture was stirred for 12 hours, concentrated in vacuo, the residue was triturated with water (2X, 5ml), dried in vacuo to give a yellow solid, the solid was treated with TFA (2ml), the mixture was concentrated in vacuo, the residue was dissolved in water , purified from the aqueous mixture by preparative reverse-phase HPLC to give cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(2 -piperidin-4-ylethylcarbamoyl)-1-piperazinecarboxylate as an amorphous colorless solid. Plasma Absorption LRMS: Calculated for (C 35 H 55 N 10 O 6 ): MH + : 712.9; Found MH + : 713.2.
按照实施例8的方法,使用不同的原料,制备以下式(Ⅰ)化合物:According to the method for embodiment 8, use different raw materials, prepare following formula (I) compound:
顺-1,5-亚环辛基4-(4-氨基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物2);计算值(C35H50N10O6):MH+:707.9,测定值MH+:707.7;cis-1,5-cyclooctylene 4-(4-aminobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate Ester (Compound 2); Calculated for (C 35 H 50 N 10 O 6 ): MH + : 707.9, Found MH + : 707.7;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-哌啶基甲基氨基甲酰基)-1-哌嗪甲酸酯(化合物3);计算值(C34H53N10O6):MH+:698.9,测定值MH+:699.7;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-piperidinylmethylcarbamoyl)-1-piperazine Formate (Compound 3); Calculated for (C 34 H 53 N 10 O 6 ): MH + : 698.9, Found MH + : 699.7;
顺-1,5-亚环辛基4-(反-4-氨基环己基甲基氨基甲酰基)-1-哌嗪甲酸酯4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物4);计算值(C35H55N10O6):MH+:712.9,测定值MH+:713.6;cis-1,5-cyclooctylene 4-(trans-4-aminocyclohexylmethylcarbamoyl)-1-piperazinecarboxylate 4-(4-guanidinobenzylcarbamoyl)-1- Piperazine carboxylate (Compound 4); Calculated (C 35 H 55 N 10 O 6 ): MH + : 712.9, Found MH + : 713.6;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯3-哌啶-4-基丙基氨基甲酰基-1-哌嗪甲酸酯(化合物5);计算值(C36H58N10O6):MH+:727.9,测定值MH+:727.9;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 3-piperidin-4-ylpropylcarbamoyl-1-piperazinecarbamate Ester (Compound 5); Calculated (C 36 H 58 N 10 O 6 ): MH + : 727.9, Found MH + : 727.9;
4-[4-(2-哌啶-4-基乙基氨基甲酰基)哌嗪-1-基羰基]苄基4(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物6);计算值(C34H48N10O5):MH+:677.8,测定值MH+:677.6;4-[4-(2-Piperidin-4-ylethylcarbamoyl)piperazin-1-ylcarbonyl]benzyl 4(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate (Compound 6); Calculated for (C 34 H 48 N 10 O 5 ): MH + : 677.8, Found MH + : 677.6;
4-[4-(3-哌啶-4-基丙基氨基甲酰基)哌嗪-1-基羰基]苄基4(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物7);计算值(C35H50N10O5):MH+:691.9,测定值MH+:691.5;4-[4-(3-Piperidin-4-ylpropylcarbamoyl)piperazin-1-ylcarbonyl]benzyl 4(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate (Compound 7); Calculated for (C 35 H 50 N 10 O 5 ): MH + : 691.9, Found MH + : 691.5;
4-[4-(4-哌啶-4-基丁基氨基甲酰基)哌嗪-1-基羰基]苄基4(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物8);计算值(C36H52N10O5):MH+:705.9,测定值MH+:705.9;4-[4-(4-Piperidin-4-ylbutylcarbamoyl)piperazin-1-ylcarbonyl]benzyl 4(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate (Compound 8); Calculated for (C 36 H 52 N 10 O 5 ): MH + : 705.9, Found MH + : 705.9;
4-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]苄基4(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯(化合物9);计算值(C34H48N10O5):MH+:677.8,测定值MH+.677.7;4-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]benzyl 4(2-piperidin-4-ylethylcarbamoyl)-1-piperazinecarboxylate (Compound 9); Calculated for (C 34 H 48 N 10 O 5 ): MH + : 677.8, Found MH + .677.7;
4-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]苄基4(3-哌啶-4-基丙基氨基甲酰基)-1-哌嗪甲酸酯(化合物10);计算值(C35H50N10O5):MH+:691.9,测定值MH+:691.3;4-[4-(4-Guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]benzyl 4(3-piperidin-4-ylpropylcarbamoyl)-1-piperazinecarboxylate (Compound 10); Calculated for (C 35 H 50 N 10 O 5 ): MH + : 691.9, Found MH + : 691.3;
4-[4-(2-哌啶-4-基乙基氨基甲酰基)哌嗪-1-基羰基甲基]苄基4(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物11);计算值(C35H50N10O5):MH+:691.9,测定值MH+:692.1;4-[4-(2-piperidin-4-ylethylcarbamoyl)piperazin-1-ylcarbonylmethyl]benzyl 4(4-guanidinobenzylcarbamoyl)-1-piperazine Ester (Compound 11); Calculated (C 35 H 50 N 10 O 5 ): MH + : 691.9, Found MH + : 692.1;
4-[4-(3-哌啶-4-基丙基氨基甲酰基)哌嗪-1-基羰基甲基]苄基4(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物12);计算值(C36H52N10O5):MH+:705.9,测定值MH+:705.6;4-[4-(3-piperidin-4-ylpropylcarbamoyl)piperazin-1-ylcarbonylmethyl]benzyl 4(4-guanidinobenzylcarbamoyl)-1-piperazine Ester (Compound 12); Calculated for (C 36 H 52 N 10 O 5 ): MH + : 705.9, Found MH + : 705.6;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-甲基氨基甲基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物13);计算值(C37H54N10O6):MH+:735.9,测定值MH+:735.7;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-methylaminomethylbenzylcarbamoyl)-1- Piperazine carboxylate (Compound 13); Calculated for (C 37 H 54 N 10 O 6 ): MH + : 735.9, Found MH + : 735.7;
顺-1,5-亚环辛基4-(4-胍基苯乙酰基)-1-哌嗪甲酸酯4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯(化合物14);计算值(C35H55N9O6):MH+:698.9,测定值MH+:698.2;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-(2-piperidin-4-ylethylcarbamoyl)-1-piper Mezincarboxylate (Compound 14); Calculated (C 35 H 55 N 9 O 6 ): MH + : 698.9, Found MH + : 698.2;
顺-1,5-亚环辛基4-(4-胍基苯乙酰基)-1-哌嗪甲酸酯4-(3-哌啶-4-基丙基氨基甲酰基)-1-哌嗪甲酸酯(化合物15);计算值(C36H57N2O6):MH+:712.9,测定值MH+:712.3;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-(3-piperidin-4-ylpropylcarbamoyl)-1-piper Oxyzinecarboxylate (Compound 15); Calculated (C 36 H 57 N 2 O 6 ): MH + : 712.9, Found MH + : 712.3;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-咪唑-1-基丁基氨基甲酰基)-1-哌嗪甲酸酯(化合物16);计算值(C35H53N11O6):MH+:724.9,测定值MH+:724.5;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-imidazol-1-ylbutylcarbamoyl)-1- Piperazine carboxylate (Compound 16); Calculated for (C 35 H 53 N 11 O 6 ): MH + : 724.9, Found MH + : 724.5;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-咪唑啉-2-基氨基丁基氨基甲酰基)-1-哌嗪甲酸酯(化合物17);计算值(C35H56N12O6):MH+:741.9,测定值MH+:741.7;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarbamate 4-(4-imidazolin-2-ylaminobutylcarbamoyl)- 1-Piperazinecarboxylate (Compound 17); Calculated (C 35 H 56 N 12 O 6 ): MH + : 741.9, Found MH + : 741.7;
顺-1,5-亚环辛基4-(反-4-氨基环己基甲基氨基甲酰基)-1-哌嗪甲酸酯4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物18);计算值(C35H56N10O6):MH+:713.9,测定值MH+:714.1;cis-1,5-cyclooctylene 4-(trans-4-aminocyclohexylmethylcarbamoyl)-1-piperazinecarboxylate 4-(4-guanidinobenzylcarbamoyl)-1- Piperazine carboxylate (Compound 18); Calculated (C 35 H 56 N 10 O 6 ): MH + : 713.9, Found MH + : 714.1;
顺-1,5-亚环辛基2-(1-叔丁酰氧基甲氧羰基哌啶-4-基)乙基氨基甲酰基-1-哌嗪甲酸酯4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物19);计算值(C42H66N10O10):MH+:872.1,测定值MH+:871.8;cis-1,5-cyclooctylene 2-(1-tert-butyryloxymethoxycarbonylpiperidin-4-yl)ethylcarbamoyl-1-piperazinecarboxylate 4-(4-guanidino Benzylcarbamoyl)-1-piperazinecarboxylate (Compound 19); Calculated for (C 42 H 66 N 10 O 10 ): MH + : 872.1, Found MH + : 871.8;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-[2-(1-甲基哌啶-4-基)乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物20);计算值(C36H58N10O6):MH2 +:364.0,测定值MH2 +/2:364.3;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-[2-(1-methylpiperidin-4-yl)ethyl Carbamoyl]-1-piperazinecarboxylate (Compound 20); Calculated for (C 36 H 58 N 10 O 6 ): MH 2 + : 364.0, Found MH 2 + /2: 364.3;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-咪唑-2-基氨基丙基氨基甲酰基)-1-哌嗪甲酸酯(化合物21);计算值(C34H54N12O6):MH+:727.9,测定值MH+:728.0;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-imidazol-2-ylaminopropylcarbamoyl)-1 - piperazine carboxylate (compound 21); calcd (C 34 H 54 N 12 O 6 ): MH + : 727.9, found MH + : 728.0;
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4-[1-(1-亚胺基乙基)哌啶-4-基甲基氨基甲酰基]-1-哌嗪甲酸酯(化合物22);计算值(C36H57N11O6):MH+:740.9,测定值MH+:740.5;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-[1-(1-iminoethyl)piperidin-4-yl Methylcarbamoyl]-1-piperazinecarboxylate (Compound 22); Calculated for (C 36 H 57 N 11 O 6 ): MH + : 740.9, Found MH + : 740.5;
顺-1,5-亚环辛基4-(4-胍基苄酰基氨基甲基)-1-哌啶甲酸酯4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯(化合物23);计算值(C36H57N9O6):MH+:712.9,测定值MH+:711.6;cis-1,5-cyclooctylene 4-(4-guanidinobenzoylaminomethyl)-1-piperidinecarboxylate 4-(2-piperidin-4-ylethylcarbamoyl)-1 - piperazine carboxylate (compound 23); calcd (C 36 H 57 N 9 O 6 ): MH + : 712.9, found MH + : 711.6;
顺-1,5-亚环辛基4-(4-脒基苄基氨基甲酰基)-1-哌嗪甲酸酯2-(1-甲氧羰基哌啶-4-基)乙基氨基甲酰基-1-哌嗪甲酸酯(化合物24);计算值(C37H57N9O8):MH+:756.9,测定值MH+:756.7;cis-1,5-cyclooctylene 4-(4-amidinobenzylcarbamoyl)-1-piperazinecarboxylate 2-(1-methoxycarbonylpiperidin-4-yl)ethylaminomethyl Acyl-1-piperazinecarboxylate (Compound 24); Calculated for (C 37 H 57 N 9 O 8 ): MH + : 756.9, Found MH + : 756.7;
3{4-[2-(4-{顺-5-[4-(4-脒基苄基氨基甲基)哌嗪-1-基羰氧基]环辛基氧羰基}哌嗪-1-基羰基氨基)乙基]哌啶-1-基羰基}丙酸(化合物25);计算值(C39H60N9O9):MH+:799.0,测定值MH+:798.6;3{4-[2-(4-{cis-5-[4-(4-amidinobenzylaminomethyl)piperazin-1-ylcarbonyloxy]cyclooctyloxycarbonyl}piperazine-1- (C 39 H 60 N 9 O 9 ): MH + : 799.0, Found MH + : 798.6;
按照实施例8,用活性酯代替异氰酸酯,制备以下式Ⅰ化合物:According to Example 8, with active ester instead of isocyanate, the following compound of formula I was prepared:
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-咪唑-4-基乙酰基-1-哌嗪甲酸酯(化合物26);计算值(C32H46N10O6):MH+:667.8,测定值MH+:667.7;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-imidazol-4-ylacetyl-1-piperazinecarboxylate (compound 26); Calculated (C 32 H 46 N 10 O 6 ): MH + : 667.8, Found MH + : 667.7;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(E-3-咪唑-4-基丙烯酰基)-1-哌嗪甲酸酯(化合物27);计算值(C33H46N10O6):MH+:679.9,测定值MH+:679.8;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(E-3-imidazol-4-ylacryloyl)-1-piper Oxyzinecarboxylate (Compound 27); Calculated for (C 33 H 46 N 10 O 6 ): MH + : 679.9, Found MH + : 679.8;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-咪唑-4-基丙酰基)-1-哌嗪甲酸酯(化合物28);计算值(C33H48N10O6):MH+:681.8,测定值MH+:681.7;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazine carboxylate 4-(3-imidazol-4-ylpropionyl)-1-piperazine Ester (Compound 28); Calculated (C 33 H 48 N 10 O 6 ): MH + : 681.8, Found MH + : 681.7;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(5-咪唑-4-基戊酰基)-1-哌嗪甲酸酯(化合物29);计算值(C35H52N10O6):MH+:709.9,测定值MH+:709.5;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazine carboxylate 4-(5-imidazol-4-ylpentanoyl)-1-piperazine Ester (Compound 29); Calculated (C 35 H 52 N 10 O 6 ): MH + : 709.9, Found MH + : 709.5;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(6-咪唑-4-基己酰基)-1-哌嗪甲酸酯(化合物30);计算值(C36H54N10O6):MH+:723.9,测定值MH+:723.4;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazine carboxylate 4-(6-imidazol-4-ylhexanoyl)-1-piperazine Ester (Compound 30); Calcd. (C 36 H 54 N 10 O 6 ): MH + : 723.9, Found MH + : 723.4;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-咪唑-1-基甲基苯基乙酰基)-1-哌嗪甲酸酯(化合物31);计算值(C39H52N10O6):MH+:757.9,测定值MH+:757.2;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-imidazol-1-ylmethylphenylacetyl)-1 - piperazine carboxylate (compound 31); calcd (C 39 H 52 N 10 O 6 ): MH + : 757.9, found MH + : 757.2;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-咪唑-1-基甲基苯甲酰基)-1-哌嗪甲酸酯(化合物32);计算值(C38H50N10O6):MH+:743.9,测定值MH+:743.7;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-imidazol-1-ylmethylbenzoyl)-1- Piperazine carboxylate (Compound 32); Calcd. (C 38 H 50 N 10 O 6 ): MH + : 743.9, Found MH + : 743.7;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-咪唑-4-基甲基苯甲酰基)-1-哌嗪甲酸酯(化合物33);计算值(C38H50N10O6):MH+:743.9,测定值MH+:743.6;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-imidazol-4-ylmethylbenzoyl)-1- Piperazine carboxylate (Compound 33); Calcd. (C 38 H 50 N 10 O 6 ): MH + : 743.9, Found MH + : 743.6;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲基)-1-哌嗪甲酸酯4-(7-咪唑-1-基庚酰基)-1-哌嗪甲酸酯(化合物34);计算值(C37H56N10O6):MH+:737.9,测定值MH+:737.6;cis-1,5-cyclooctylene 4-(4-guanidinobenzylaminomethyl)-1-piperazinecarboxylate 4-(7-imidazol-1-ylheptanoyl)-1-piperazine Ester (Compound 34); Calculated for (C 37 H 56 N 10 O 6 ): MH + : 737.9, Found MH + : 737.6;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-[6-(甲基咪唑-1-基)己酰基]-1-哌嗪甲酸酯(化合物35);计算值(C37H56N10O6):MH+:737.9,测定值MH+:737.3;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-[6-(methylimidazol-1-yl)hexanoyl]-1 - piperazine carboxylate (compound 35); calcd (C 37 H 56 N 10 O 6 ): MH + : 737.9, found MH + : 737.3;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-咪唑-1-基苯氧基乙酰基)-1-哌嗪甲酸酯(化合物36);计算值(C38H50N10O7):MH+:759.9,测定值MH+:759.3;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-imidazol-1-ylphenoxyacetyl)-1- Piperazine carboxylate (Compound 36); Calculated for (C 38 H 50 N 10 O 7 ): MH + : 759.9, Found MH + : 759.3;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-[6-(4-甲基咪唑-1-基)己酰基]-1-哌嗪甲酸酯和顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-[6-(4-methylimidazol-1-yl)hexanoyl] -1-piperazinecarboxylate and cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate;
4-[6-(5-甲基咪唑-1-基)己酰基]-1-哌嗪甲酸酯,混合物(化合物37);计算值(C37H56N10O6):MH+:737.9,测定值MH+:738.2;4-[6-(5-Methylimidazol-1-yl)hexanoyl]-1-piperazinecarboxylate, mixture (compound 37); calculated for (C 37 H 56 N 10 O 6 ): MH + : 737.9, found MH + : 738.2;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯(化合物38);计算值(C36H57N9O6):MH+:712.9,测定值MH+:712.4;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-piperidin-4-ylbutyryl)-1-piperazine Formate (Compound 38); Calculated for (C 36 H 57 N 9 O 6 ): MH + : 712.9, Found MH + : 712.4;
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯(化合物39);计算值(C36H56N8O6):MH+:697.9,测定值MH+:697.5;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-(4-piperidin-4-ylbutyryl)-1-piperazine Ester (Compound 39); Calculated (C 36 H 56 N 8 O 6 ): MH + : 697.9, Found MH + : 697.5;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(2-哌啶-4-基乙基)(甲基)氨基甲酰基-1-哌嗪甲酸酯(化合物40);计算值(C36H58N10O6):MH+:727.9,测定值MH+:727.6;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(2-piperidin-4-ylethyl)(methyl)amino Formyl-1-piperazinecarboxylate (Compound 40); Calculated for (C 36 H 58 N 10 O 6 ): MH + : 727.9, Found MH + : 727.6;
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4-(2-哌啶-4-基乙基)(甲基)氨基甲酰基-1-哌嗪甲酸酯(化合物41);计算值(C36H57N9O6):MH+:712.9,测定值MH+:712.7;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-(2-piperidin-4-ylethyl)(methyl)aminomethyl Acyl-1-piperazinecarboxylate (Compound 41); Calculated for (C 36 H 57 N 9 O 6 ): MH + : 712.9, Found MH + : 712.7;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(2-哌啶-4-基乙氧羰基)-1-哌嗪甲酸酯(化合物42);计算值(C35H55N9O7):MH+:714.9,测定值MH+:714.5;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(2-piperidin-4-ylethoxycarbonyl)-1-piper Mezincarboxylate (Compound 42); Calcd. (C 35 H 55 N 9 O 7 ): MH + : 714.9, Found MH + : 714.5;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲基)-1-哌嗪甲酸酯4-(4-咪唑-1-基苯基乙酰基)-1-哌嗪甲酸酯(化合物43);计算值(C38H50N10O6):MH+:743.9,测定值MH+:743.6;cis-1,5-cyclooctylene 4-(4-guanidinobenzylaminomethyl)-1-piperazinecarboxylate 4-(4-imidazol-1-ylphenylacetyl)-1-piperazine Mezincarboxylate (Compound 43); Calculated (C 38 H 50 N 10 O 6 ): MH + : 743.9, Found MH + : 743.6;
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4-(6-咪唑-1-基己酰基)-1-哌嗪甲酸酯(化合物44);计算值(C36H53N9O6):MH+:708.9,测定值MH+:708.8;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-(6-imidazol-1-ylhexanoyl)-1-piperazinecarboxylate Ester (Compound 44); Calcd. (C 36 H 53 N 9 O 6 ): MH + : 708.9, Found MH + : 708.8;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲基)-1-哌嗪甲酸酯4-(3-吡啶-4-基硫代丙酰基)-1-哌嗪甲酸酯(化合物45);计算值(C35H49N9O6):MH+:724.9,测定值MH+:724.4;cis-1,5-cyclooctylene 4-(4-guanidinobenzylaminomethyl)-1-piperazinecarboxylate 4-(3-pyridin-4-ylthiopropionyl)-1-piper Oxyzinecarboxylate (Compound 45); Calculated (C 35 H 49 N 9 O 6 ): MH + : 724.9, Found MH + : 724.4;
顺-1,5-亚环辛基4-(4-胍基苄基羰基)-1-哌嗪甲酸酯4-吡啶-4-基硫代乙酰基-1-哌嗪甲酸酯(化合物46);计算值(C34H47N9O6):MH+:710.9,测定值MH+:710.8;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbonyl)-1-piperazinecarboxylate 4-pyridin-4-ylthioacetyl-1-piperazinecarboxylate (compound 46); Calculated (C 34 H 47 N 9 O 6 ): MH + : 710.9, Found MH + : 710.8;
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4-(3-吡啶-4-基硫代丙酰基)-1-哌嗪甲酸酯(化合物47);计算值(C35H48N8O6):MH+:709.9,测定值MH+:709.3;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-(3-pyridin-4-ylthiopropionyl)-1-piperazine Formate (Compound 47); Calculated for (C 35 H 48 N 8 O 6 ): MH + : 709.9, Found MH + : 709.3;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(6-咪唑-4-基己酰基)-1-哌嗪甲酸酯(化合物48);计算值(C36H54N10O6):MH+:723.9,测定值MH+:723.5;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazine carboxylate 4-(6-imidazol-4-ylhexanoyl)-1-piperazine Ester (Compound 48); Calculated for (C 36 H 54 N 10 O 6 ): MH + : 723.9, Found MH + : 723.5;
顺-1,5-亚环辛基4-(4-苯并咪唑-6-基羰基)-1-哌嗪甲酸酯4-(胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物49);计算值(C35H47N10O6):MH+:703.8,测定值MH+:703.4;cis-1,5-cyclooctylene 4-(4-benzimidazol-6-ylcarbonyl)-1-piperazinecarboxylate 4-(guanidinobenzylcarbamoyl)-1-piperazinecarboxylate Ester (Compound 49); Calcd. (C 35 H 47 N 10 O 6 ): MH + : 703.8, Found MH + : 703.4;
顺-1,5-亚环辛基4-(4-脒基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(6-咪唑-1-基己酰基)-1-哌嗪甲酸酯(化合物50);计算值(C36H53N9O6):MH+:708.9,测定值MH+:708.6;cis-1,5-cyclooctylene 4-(4-amidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(6-imidazol-1-ylhexanoyl)-1-piperazine Ester (Compound 50); Calculated (C 36 H 53 N 9 O 6 ): MH + : 708.9, Found MH + : 708.6;
顺-1,5-亚环辛基4-(4-脒基苯甲酰基氨基甲基)-1-哌嗪甲酸酯4-(6-咪唑-4-基己酰基)-1-哌啶甲酸酯(化合物51);计算值(C37H54N8O6):MH+:707.9,测定值MH+:707.5;cis-1,5-cyclooctylene 4-(4-amidinobenzoylaminomethyl)-1-piperazinecarboxylate 4-(6-imidazol-4-ylhexanoyl)-1-piperidine Formate (Compound 51); Calculated for (C 37 H 54 N 8 O 6 ): MH + : 707.9, Found MH + : 707.5;
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4-(6-咪唑-4-基己酰基)-1-哌嗪甲酸酯(化合物52);计算值(C35H52N10O6):MH+:708.9,测定值MH+:708.4;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-(6-imidazol-4-ylhexanoyl)-1-piperazinecarboxylate Ester (Compound 52); Calcd. (C 35 H 52 N 10 O 6 ): MH + : 708.9, Found MH + : 708.4;
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4-吡啶-4-基氨基甲酰基乙酰基-1-哌嗪甲酸酯(化合物53);计算值(C35H47N9O7):MH+:706.8,测定值MH+:706.3;cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4-pyridin-4-ylcarbamoylacetyl-1-piperazinecarboxylate (Compound 53); Calculated for (C 35 H 47 N 9 O 7 ): MH + : 706.8, Found MH + : 706.3;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-吡啶-4-基氨基丙酰基)-1-哌嗪甲酸酯(化合物54);计算值(C35H50N10O6):MH+:707.9,测定值MH+:707.3;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-pyridin-4-ylaminopropionyl)-1-piperazine Formate (Compound 54); Calculated for (C 35 H 50 N 10 O 6 ): MH + : 707.9, Found MH + : 707.3;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯3-[吡啶-4-基(叔丁氧羰基)氨基]丙酰基-1-哌嗪甲酸酯(化合物55);计算值(C40H58N10O8):MH2 2+/2:404.5,测定值MH2 2+/2:404.2;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 3-[pyridin-4-yl(tert-butoxycarbonyl)amino]propionyl- 1-Piperazinecarboxylate (Compound 55); Calculated for (C 40 H 58 N 10 O 8 ): MH 2 2+ /2: 404.5, Found MH 2 2+ /2 : 404.2;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-哌嗪-1-基羰基丙酰基)-1-哌嗪甲酸酯(化合物56);计算值(C35H54N10O7):MH+:727.9,测定值MH+:727.5;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-piperazin-1-ylcarbonylpropionyl)-1-piper Oxyzinecarboxylate (Compound 56); Calculated (C 35 H 54 N 10 O 7 ): MH + : 727.9, Found MH + : 727.5;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-哌啶-1-基羰基氨基乙酰基-1-哌嗪甲酸酯(化合物57);计算值(C35H54N10O7):MH+:727.9,测定值MH+:727.5;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-piperidin-1-ylcarbonylaminoacetyl-1-piperazinecarboxylate Ester (Compound 57); Calculated for (C 35 H 54 N 10 O 7 ): MH + : 727.9, Found MH + : 727.5;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(咪唑-4-基戊酰基)-1-哌嗪甲酸酯(化合物58);计算值(C35H52N10O6):MH+:708.9,测定值MH+:709.4;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(imidazol-4-ylpentanoyl)-1-piperazinecarboxylate (Compound 58); Calculated for (C 35 H 52 N 10 O 6 ): MH + : 708.9, Found MH + : 709.4;
顺-1,5-亚环辛基4-(4-脒基苯甲酰基氨基甲基)-1-哌嗪甲酸酯4-(3-哌嗪-1-基羰基丙酰基)-1-哌嗪甲酸酯(化合物59);计算值(C36H54N8O7):MH+:711.9,测定值MH+:711.4;cis-1,5-cyclooctylene 4-(4-amidinobenzoylaminomethyl)-1-piperazinecarboxylate 4-(3-piperazin-1-ylcarbonylpropionyl)-1- Piperazine carboxylate (Compound 59); Calculated (C 36 H 54 N 8 O 7 ): MH + : 711.9, Found MH + : 711.4;
顺-1,5-亚环辛基4-(4-脒基苯甲酰基氨基甲基)-1-哌嗪甲酸酯4-哌啶-4-基羰基氨基乙酰基-1-哌嗪甲酸酯(化合物60);计算值(C36H54N8O7):MH+:711.9,测定值MH+:711.4;cis-1,5-cyclooctylene 4-(4-amidinobenzoylaminomethyl)-1-piperazinecarboxylate 4-piperidin-4-ylcarbonylaminoacetyl-1-piperazine Ester (Compound 60); Calculated (C 36 H 54 N 8 O 7 ): MH + : 711.9, Found MH + : 711.4;
顺-1,5-亚环辛基4-[3-(2-氨基嘧啶-5-基)丙酰基]-1-哌嗪甲酸酯4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物61);计算值(C34H49N11O6):MH+:708.8,测定值MH+:708.4;cis-1,5-cyclooctylene 4-[3-(2-aminopyrimidin-5-yl)propionyl]-1-piperazinecarboxylate 4-(4-guanidinobenzylcarbamoyl)- 1-Piperazinecarboxylate (Compound 61); Calculated for (C 34 H 49 N 11 O 6 ): MH + : 708.8, Found MH + : 708.4;
顺-1,5-亚环辛基4-[3-(6-氨基吡啶-3-基)丙酰基]-1-哌嗪甲酸酯4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物62);计算值(C35H50N10O6):MH+:707.8,测定值MH+:707.4;cis-1,5-cyclooctylene 4-[3-(6-aminopyridin-3-yl)propionyl]-1-piperazinecarboxylate 4-(4-guanidinobenzylcarbamoyl)- 1-Piperazinecarboxylate (Compound 62); Calculated for (C 35 H 50 N 10 O 6 ): MH + : 707.8, Found MH + : 707.4;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-[4-(4-吡啶-4-硫)丁酰基]-1-哌嗪甲酸酯(化合物63);计算值(C36H51N9O6):MH+:738.9,测定值MH+:738.4;cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-[4-(4-pyridine-4-thio)butyryl]-1 - piperazine carboxylate (compound 63); calcd (C 36 H 51 N 9 O 6 ): MH + : 738.9, found MH + : 738.4;
顺-1,5-亚环辛基4-[3-(2-氨基-2,4-二氧-1,2,3,4--四氢嘧啶-5-基)丙酰基]-1-哌嗪甲酸酯4-(4-胍基苄基氨基甲酰基-1-哌嗪甲酸酯(化合物64);计算值(C34H48N10O8):MH+:725.8,测定值MH+:725.2;cis-1,5-cyclooctylene 4-[3-(2-amino-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-5-yl)propionyl]-1- Piperazinecarboxylate 4-(4-guanidinobenzylcarbamoyl-1-piperazinecarboxylate (Compound 64); Calculated for (C 34 H 48 N 10 O 8 ): MH + : 725.8, found MH + :725.2;
顺-1,5-亚环辛基4-(4-脒基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯(化合物65);计算值(C36H56N8O6):MH+:697.9,测定值MH+:697.4;cis-1,5-cyclooctylene 4-(4-amidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(4-piperidin-4-ylbutyryl)-1-piperazine Formate (Compound 65); Calculated for (C 36 H 56 N 8 O 6 ): MH + : 697.9, Found MH + : 697.4;
顺-1,5-亚环辛基4-(4-脒基苄基氨基甲基)-1-哌啶甲酸酯4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯(化合物66);计算值(C37H57N7O6):MH+:696.9,测定值MH+:696.4;cis-1,5-cyclooctylene 4-(4-amidinobenzylaminomethyl)-1-piperidinecarboxylate 4-(4-piperidin-4-ylbutyryl)-1-piperazine Formate (Compound 66); Calculated for (C 37 H 57 N 7 O 6 ): MH + : 696.9, Found MH + : 696.4;
顺-1,5-亚环辛基4-(1-脒基哌啶-4-基乙酰基)-1-哌啶甲酸酯4-(6-咪唑-1-基己酰基)-1-哌嗪甲酸酯(化合物67);计算值(C35H57N9O6):MH+:700.9,测定值MH+:700.5;cis-1,5-cyclooctylene 4-(1-amidinopiperidin-4-ylacetyl)-1-piperidinecarboxylate 4-(6-imidazol-1-ylhexanoyl)-1- Piperazine carboxylate (Compound 67); Calculated (C 35 H 57 N 9 O 6 ): MH + : 700.9, Found MH + : 700.5;
顺-1,5-亚环辛基4-(1-脒基-4-哌啶基乙酰基)-1-哌嗪甲酸酯4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯(化合物68);计算值(C35H60N9O6):MH+:689.9,测定值MH+:689.4;cis-1,5-Cyclooctylene 4-(1-amidino-4-piperidinylacetyl)-1-piperazinecarboxylate 4-(4-piperidin-4-ylbutyryl)-1 - piperazine carboxylate (compound 68); calcd (C 35 H 60 N 9 O 6 ): MH + : 689.9, found MH + : 689.4;
顺-1,5-亚环辛基4-(1-脒基-4-哌啶基乙酰基)-1-哌嗪甲酸酯4-(6-咪唑-1-基己酰基)-1-哌嗪甲酸酯(化合物69);计算值(C35H57N9O6):MH+:700.9,测定值MH+:700.4;cis-1,5-Cyclooctylene 4-(1-amidino-4-piperidinylacetyl)-1-piperazinecarboxylate 4-(6-imidazol-1-ylhexanoyl)-1- Piperazine carboxylate (Compound 69); Calculated (C 35 H 57 N 9 O 6 ): MH + : 700.9, Found MH + : 700.4;
顺-1,5-亚环辛基4-(4-脒基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(6-咪唑-4-基己酰基)-1-哌嗪甲酸酯(化合物70);计算值(C36H53N9O6):MH+708.9,测定值MH+:708.4;cis-1,5-cyclooctylene 4-(4-amidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(6-imidazol-4-ylhexanoyl)-1-piperazine Ester (Compound 70); Calculated for (C 36 H 53 N 9 O 6 ): MH + 708.9, Found MH + : 708.4;
顺-1,5-亚环辛基4-(4-脒基苯甲酰基)-1-哌啶甲酸酯4-(6-咪唑-4-基己酰基)-1-哌嗪甲酸酯(化合物71);计算值(C37H54N8O6):MH+:709.9,测定值MH+:707.4;cis-1,5-cyclooctylene 4-(4-amidinobenzoyl)-1-piperidinecarboxylate 4-(6-imidazol-4-ylhexanoyl)-1-piperazinecarboxylate (Compound 71); Calculated for (C 37 H 54 N 8 O 6 ): MH + : 709.9, Found MH + : 707.4;
顺-1,5-亚环辛基4-(4-脒基苯基乙酰基)-1-哌嗪甲酸酯4-(6-咪唑-1-基己酰基)-1-哌嗪甲酸酯(化合物72);计算值(C36H52N8O6):MH+693.9,测定值MH+:693.4;cis-1,5-cyclooctylene 4-(4-amidinophenylacetyl)-1-piperazinecarboxylate 4-(6-imidazol-1-ylhexanoyl)-1-piperazinecarboxylate Ester (Compound 72); Calculated for (C 36 H 52 N 8 O 6 ): MH + 693.9, Found MH + : 693.4;
顺-1,5-亚环辛基4-(4-脒基苯基乙酰基)-1-哌嗪甲酸酯4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯(化合物73);计算值(C36H55N7O6):MH+:682.9,测定值MH+:682.4;cis-1,5-cyclooctylene 4-(4-amidinophenylacetyl)-1-piperazinecarboxylate 4-(4-piperidin-4-ylbutyryl)-1-piperazine Ester (Compound 73); Calculated for (C 36 H 55 N 7 O 6 ): MH + : 682.9, Found MH + : 682.4;
顺-1,5-亚环辛基4-(4-脒基苯基乙酰基)-1-哌嗪甲酸酯4-(6-咪唑-4-基己酰基)-1-哌嗪甲酸酯(化合物74);计算值(C36H52N8O6):MH+:693.9,测定值MH+:693.4;cis-1,5-cyclooctylene 4-(4-amidinophenylacetyl)-1-piperazinecarboxylate 4-(6-imidazol-4-ylhexanoyl)-1-piperazinecarboxylate Ester (Compound 74); Calculated for (C 36 H 52 N 8 O 6 ): MH + : 693.9, Found MH + : 693.4;
顺-1,5-亚环辛基4-(4-脒基苄基氨基甲酰基)-1-哌嗪甲酸酯4-[4-(2-(1-叔丁基羰基氧基甲氧基羰基)哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯(化合物75);计算值(C43H65N9O10):MH+:857.0,测定值MH+:856.6;cis-1,5-cyclooctylene 4-(4-amidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-[4-(2-(1-tert-butylcarbonyloxymethoxy (C 43 H 65 N 9 O 10 ): MH + : 857.0, found MH + :856.6;
实施例9顺-1,5-亚环辛基二(4-叔丁氧羰基-1-哌嗪甲酸酯)Example 9 cis-1,5-cyclooctylene bis(4-tert-butoxycarbonyl-1-piperazinecarboxylate)
把实施例5制备的顺-1,5-亚环辛基二(氯代甲酸酯)(3.69g,13.7mmol)和DIEA(7.2ml,41mmol)溶于DMF(25ml)中,加入-1-哌嗪甲酸叔丁基酯(5.1g,27.4mmol),在室温下搅拌混合物12小时,然后真空浓缩得到半固体残留物。将残留物在二氯甲烷(50ml)和水(50ml)之间分配,二氯甲烷层用0.1N盐酸水溶液洗涤(2X,25ml),干燥(MgSO4)并过滤,真空浓缩得到顺-1,5-亚环辛基二(4-叔丁氧羰基-1-哌嗪甲酸酯),为无定形固体。1H-NMR(300MHz,CDCl3):4.80(m,2H),3.40(brs,16H),2.00-1.40(m,12H),1.40(s,18H)。The cis-1,5-cyclooctylene bis(chloroformate) (3.69g, 13.7mmol) and DIEA (7.2ml, 41mmol) prepared in Example 5 were dissolved in DMF (25ml), and -1 - tert-butyl piperazinecarboxylate (5.1 g, 27.4 mmol), the mixture was stirred at room temperature for 12 hours, then concentrated in vacuo to give a semi-solid residue. The residue was partitioned between dichloromethane (50ml) and water (50ml), the dichloromethane layer was washed with 0.1N aqueous hydrochloric acid (2X, 25ml), dried ( MgSO4 ) and filtered, concentrated in vacuo to give cis-1, 5-Cyclooctylene bis(4-tert-butoxycarbonyl-1-piperazinecarboxylate), an amorphous solid. 1 H-NMR (300MHz, CDCl 3 ): 4.80 (m, 2H), 3.40 (brs, 16H), 2.00-1.40 (m, 12H), 1.40 (s, 18H).
实施例10顺-1,5-亚环辛基二[4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯](化合物76)Example 10 cis-1,5-cyclooctylenebis[4-(2-piperidin-4-ylethylcarbamoyl)-1-piperazinecarboxylate] (compound 76)
以下制备式Ⅰ化合物,其中R1和R2每个是2-哌啶-4-基乙基,X1和X9每个是-NHC(O)-,X2和X8每个是1,4-亚哌嗪基,X3和X7每个是-C(O)O,X4和X6每个是共价键,X5是顺-1,5-亚环辛基。Compounds of formula I are prepared as follows, wherein R and R are each 2-piperidin-4-ylethyl, X and X are each -NHC(O)-, X and X are each 1 , 4-piperazinylene, each of X 3 and X 7 is -C(O)O, each of X 4 and X 6 is a covalent bond, and X 5 is cis-1,5-cyclooctylene.
用纯TFA(1ml)把实施例9制备的顺-1,5-亚环辛基二(4-叔丁氧羰基-1-哌嗪甲酸酯)(47.9mg,0.088mmol)处理10分钟,得到无色油状物,真空浓缩混合物,残留物用乙醚(2X,5ml)研制,于真空中重复干燥,得到无定型固体。将固体残留物溶于DMF(5ml)和DIEA(100ml,0.5mmol)中,往溶液中加入4-(2-异氰酸酯基乙基)-1-哌啶甲酸叔丁基酯(460ml,0.39M,于DMF中,0.18mmol),搅拌混合物12小时,真空浓缩,残留物用水(2X,5ml)研制,真空干燥得到黄色油状物。固体用TFA(2ml)处理,真空浓缩混合物,残留物溶于水中,用制备性的反相HPLC从含水混合物中提纯,然后冷冻干燥得到顺-1,5-亚环辛基二[4-(2-哌啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯],为无色无定型固体。电子喷雾法LRMS:计算值(C34H60N8O6):MH+:677.9,测定值MH+:677.6;cis-1,5-cyclooctylene bis(4-tert-butoxycarbonyl-1-piperazinecarboxylate) (47.9 mg, 0.088 mmol) prepared in Example 9 was treated with pure TFA (1 ml) for 10 minutes, A colorless oil was obtained, the mixture was concentrated in vacuo, the residue was triturated with ether (2X, 5ml) and dried repeatedly in vacuo to give an amorphous solid. The solid residue was dissolved in DMF (5ml) and DIEA (100ml, 0.5mmol), and tert-butyl 4-(2-isocyanatoethyl)-1-piperidinecarboxylate (460ml, 0.39M, In DMF, 0.18 mmol), the mixture was stirred for 12 hours, concentrated in vacuo, the residue was triturated with water (2X, 5 mL), and dried in vacuo to give a yellow oil. The solid was treated with TFA (2 ml), the mixture was concentrated in vacuo, the residue was dissolved in water, purified from the aqueous mixture by preparative reverse phase HPLC, and lyophilized to give cis-1,5-cyclooctylenebis[4-( 2-piperidin-4-ylethylcarbamoyl)-1-piperazinecarboxylate], a colorless amorphous solid. Electron spray method LRMS: calculated value (C 34 H 60 N 8 O 6 ): MH + : 677.9, measured value MH + : 677.6;
按照实施例10的方法,使用不同的原料,得到顺-1,5-亚环辛基二[4-(4-甲基氨基甲基苄基氨基甲酰基)-1-哌嗪甲酸酯](化合物77)。计算值(C38H56N8O6):MH+:721.9,测定值MH+:721.7;According to the method of Example 10, different raw materials were used to obtain cis-1,5-cyclooctylene bis[4-(4-methylaminomethylbenzylcarbamoyl)-1-piperazinecarboxylate] (Compound 77). Calculated (C 38 H 56 N 8 O 6 ): MH + : 721.9, Found MH + : 721.7;
按照实施例10,使用活性酯代替异氰酸酯得到以下式Ⅰ化合物:According to Example 10, the following compound of formula I is obtained by using active ester instead of isocyanate:
顺-1,5-亚环辛基二[4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯](化合物78)。计算值(C36H62N6O6):MH+:675.9,测定值MH+:675.6;cis-1,5-cyclooctylenebis[4-(4-piperidin-4-ylbutyryl)-1-piperazinecarboxylate] (Compound 78). Calculated (C 36 H 62 N 6 O 6 ): MH + : 675.9, Found MH + : 675.6;
对-亚甲基亚苯基二[4-(4-哌啶-4-基丁酰基)-1-哌嗪甲酸酯]。(化合物79)计算值(C36H56N6O6):MH+:669.9,测定值MH+:669.4;p-Methylenephenylene bis[4-(4-piperidin-4-ylbutyryl)-1-piperazinecarboxylate]. (Compound 79) Calculated (C 36 H 56 N 6 O 6 ): MH + : 669.9, Found MH + : 669.4;
实施例114-(3-咪唑-1-基丙基氨基甲酰基)-1-哌嗪甲酸叔丁基酯Example 11 tert-butyl 4-(3-imidazol-1-ylpropylcarbamoyl)-1-piperazinecarboxylate
把实施例3制备的4-氯羰基-1-哌嗪甲酸叔丁基酯(188mg,0.76mmol)溶于二氯甲烷(10ml)中,加入DIEA(150ml,0.86mmol),用注射器加入1-(3-氨基丙基)咪唑(100ml,0.84mmol),搅拌混合物12小时,往混合物中加入二氯甲烷(10ml),用水洗涤有机层(1X,10ml),干燥(MgSO4)并过滤,浓缩得到4-(3-咪唑-1-基丙基氨基甲酰基)-1-哌嗪甲酸叔丁基酯(230mg,0.68mmol,90%产率),为无色油状物。1H-NMR(300MHz,DMSO-d6):7.60(s,1H),7.20(s,1H),6.85(s,1H),6.60(tr,1H),3.95(tr,2H),3.30(s,8H),3.00(q,2H),1.80(m,2H),1.40(s,9H)。实施例124-氨基甲基-1-苯氨基甲酸叔丁基酯盐酸盐Dissolve tert-butyl 4-chlorocarbonyl-1-piperazinecarboxylate (188mg, 0.76mmol) prepared in Example 3 in dichloromethane (10ml), add DIEA (150ml, 0.86mmol), and add 1- (3-Aminopropyl)imidazole (100ml, 0.84mmol), the mixture was stirred for 12 hours, dichloromethane (10ml) was added to the mixture, the organic layer was washed with water (1X, 10ml), dried (MgSO 4 ) and filtered, concentrated tert-Butyl 4-(3-imidazol-1-ylpropylcarbamoyl)-1-piperazinecarboxylate (230 mg, 0.68 mmol, 90% yield) was obtained as a colorless oil. 1 H-NMR (300MHz, DMSO-d 6 ): 7.60(s,1H), 7.20(s,1H), 6.85(s,1H), 6.60(tr,1H), 3.95(tr,2H), 3.30( s,8H), 3.00(q,2H), 1.80(m,2H), 1.40(s,9H). Example 12 tert-butyl 4-aminomethyl-1-phenylcarbamate hydrochloride
把4-氨基苄基胺(5.56g,45.6mmol)加入到水中(45ml),往溶液中加入柠檬酸(9.63g,50mmol),往溶液中滴加碳酸二叔丁基酯(9.94g,45.5mmol),室温搅拌混合物48小时,得到黄色悬浮液。过滤悬浮液,水溶液用过量固体碳酸钠碱化,用乙酸乙酯(3X,35ml)萃取,合并的萃取液用饱和氯化钠水溶液洗涤,干燥(MgSO4),过滤,真空浓缩得到白色固体,将固体溶于甲醇(30ml),溶液用溶于二噁烷中的HCl酸化(4M,8.4ml,33.6mmol),加入乙醚(100ml),得到悬浮液,过滤分离出颗粒物,真空干燥得到4-氨基甲基-1-苯氨基甲酸叔丁基酯盐酸盐(7.2g,27.8mmol,产率61%),为无色固体。1H-NMR(300MHz,DMSO-d6):9.43(s,1H),8.20(brs,3H),7.40(dd,AB,4H),3.92(m,2H),1.50(s,9H)。4-aminobenzylamine (5.56g, 45.6mmol) was added to water (45ml), citric acid (9.63g, 50mmol) was added to the solution, and di-tert-butyl carbonate (9.94g, 45.5 mmol), the mixture was stirred at room temperature for 48 hours to obtain a yellow suspension. The suspension was filtered, the aqueous solution was basified with excess solid sodium carbonate, extracted with ethyl acetate (3X, 35ml), the combined extracts were washed with saturated aqueous sodium chloride solution, dried ( MgSO4 ), filtered and concentrated in vacuo to give a white solid, The solid was dissolved in methanol (30ml) and the solution was acidified with HCl in dioxane (4M, 8.4ml, 33.6mmol) and diethyl ether (100ml) was added to give a suspension which was isolated by filtration and dried in vacuo to give 4- Aminomethyl-1-phenylcarbamate tert-butyl hydrochloride (7.2 g, 27.8 mmol, 61% yield) as a colorless solid. 1 H-NMR (300 MHz, DMSO-d 6 ): 9.43 (s, 1H), 8.20 (brs, 3H), 7.40 (dd, AB, 4H), 3.92 (m, 2H), 1.50 (s, 9H).
实施例134-异氰酸酯基甲基-1-苯氨基甲酸叔丁基酯Example 13 tert-butyl 4-isocyanatomethyl-1-phenylcarbamate
于0℃下将实施例12制备的4-氨基甲基-1-苯氨基甲酸叔丁基酯盐酸盐(3.39g,13.1mmol)溶于二氯甲烷(120ml)中,加入吡啶(4.3ml,53mmol)和三光气(1.3g,4.4mmol),用30分钟把混合物温热到室温,加入盐酸水溶液(0.5N,100ml)。干燥有机相(MgSO4),过滤,浓缩得到4-异氰酸酯基甲基-1-苯氨基甲酸叔丁基酯(2.7g,11mmol,产率,84%),为黄色固体。1H-NMR(300MHz,CDCl3):7.29(dd AB,4H),6.55(brs,1H),4.40(s,2H),1.55(s,9H)。Dissolve the tert-butyl 4-aminomethyl-1-phenylcarbamate hydrochloride (3.39g, 13.1mmol) prepared in Example 12 in dichloromethane (120ml) at 0°C, add pyridine (4.3ml , 53mmol) and triphosgene (1.3g, 4.4mmol), the mixture was warmed to room temperature in 30 minutes, and aqueous hydrochloric acid (0.5N, 100ml) was added. The organic phase was dried (MgSO 4 ), filtered, and concentrated to give tert-butyl 4-isocyanatomethyl-1-phenylcarbamate (2.7 g, 11 mmol, 84% yield) as a yellow solid. 1 H-NMR (300 MHz, CDCl 3 ): 7.29 (dd AB, 4H), 6.55 (brs, 1H), 4.40 (s, 2H), 1.55 (s, 9H).
实施例14顺-1,5-亚环辛基4-(4-氨基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-咪唑-1-基丙基氨基甲酰基)-1-哌嗪甲酸酯(化合物80)Example 14 cis-1,5-cyclooctylene 4-(4-aminobenzylcarbamoyl)-1-piperazinecarbamate 4-(3-imidazol-1-ylpropylcarbamoyl)- 1-piperazine carboxylate (compound 80)
以下制备式Ⅰ化合物,其中R1是4-氨基苄基,R2是3-咪唑-1-基丙基,X1和X9每个是-NHC(O)-,X2和X8每个是1,4-亚哌嗪基,X3和X7每个是-C(O)O,X4和X6每个是共价键,X5是顺-1,5-亚环辛基。The compound of formula I is prepared as follows, wherein R 1 is 4-aminobenzyl, R 2 is 3-imidazol-1-ylpropyl, X 1 and X 9 are each -NHC(O)-, X 2 and X 8 are each One is 1,4-piperazinyl, each of X 3 and X 7 is -C(O)O, each of X 4 and X 6 is a covalent bond, and X 5 is cis-1,5-cyclooctene base.
(a)用纯TFA(1ml)把实施例11制备的叔丁基4-(3-咪唑-1-基丙基氨基甲酰基)-1-哌嗪甲酸酯(225mg,0.67mmol)处理10分钟,真空浓缩混合物,残留物溶于二氯甲烷(10ml),将过量的DIEA(1.0ml)加入到溶液中,往溶液中加入实施例6制备的溶于二氯甲烷(5ml)中的顺-1,5-亚环辛基氯代甲酸酯4-叔丁氧羰基-1-哌嗪甲酸酯(279mg,0.67mmol),搅拌混合物1小时,再加入二氯甲烷(10ml),有机层用饱和碳酸氢钠水溶液(1X,10ml)洗涤,干燥(MgSO4)并过滤,浓缩得到粗的顺-1,5-亚环辛基4-(3-咪唑-1-基丙基氨基甲酰基)-1--哌嗪甲酸酯叔丁氧羰基-1-哌嗪甲酸酯加合物,为无色泡沫10 Minutes, the mixture was concentrated in vacuo, the residue was dissolved in dichloromethane (10ml), excess DIEA (1.0ml) was added to the solution, and the solution prepared in Example 6 was dissolved in dichloromethane (5ml). -1,5-Cyclooctylene chloroformate 4-tert-butoxycarbonyl-1-piperazinecarboxylate (279mg, 0.67mmol), the mixture was stirred for 1 hour, then dichloromethane (10ml) was added, organic The layer was washed with saturated aqueous sodium bicarbonate (1X, 10ml), dried ( MgSO4 ) and filtered, concentrated to give crude cis-1,5-cyclooctylene 4-(3-imidazol-1-ylpropylaminomethyl Acyl)-1-piperazinecarboxylate tert-butoxycarbonyl-1-piperazinecarboxylate adduct, colorless foam
(b)把(a)部分制备的产品用纯TFA(1ml)处理10分钟,真空浓缩混合物,残留物溶于DMF(10ml)中,加入过量的DIEA(1.5ml)和实施例13制备的4-异氰酸酯基甲基-1-苯氨基甲酸叔丁基酯(165mg,0.67mmol),搅拌混合物12小时,真空浓缩,残留物用纯TFA处理,真空浓缩混合物,残留物溶于水中(15ml),水溶液用乙醚萃取(1X,15ml),水相用1.0M的氢氧化钠水溶液碱化,然后用二氯甲烷萃取,干燥二氯甲烷(MgSO4),过滤,真空浓缩得到顺-1,5-亚环辛基4-(4-氨基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-咪唑-1-基丙基氨基甲酰基)-1-哌嗪甲酸酯,为无色泡沫。1H-NMR(300MHz,CDCl3):7.45(,4H),7.10(d,2H),7.05(s,1H),6.90(s,1H),6.60(d,2H),4.85-4.70(m,4H),4.30(d,2H),4.00(tr,2H),3.50-3.30(m,18H),2.00(m,2H),1.90-1.50(m,12H)。(b) Treat the product prepared in part (a) with pure TFA (1ml) for 10 minutes, concentrate the mixture in vacuo, dissolve the residue in DMF (10ml), add excess DIEA (1.5ml) and 4 - tert-butyl isocyanatomethyl-1-phenylcarbamate (165mg, 0.67mmol), the mixture was stirred for 12 hours, concentrated in vacuo, the residue was treated with pure TFA, the mixture was concentrated in vacuo, the residue was dissolved in water (15ml), The aqueous solution was extracted with diethyl ether (1X, 15ml), and the aqueous phase was basified with 1.0M aqueous sodium hydroxide solution, then extracted with dichloromethane, dried with dichloromethane (MgSO 4 ), filtered, and concentrated in vacuo to give cis-1,5- Cyclooctylene 4-(4-aminobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-imidazol-1-ylpropylcarbamoyl)-1-piperazinecarboxylate, for Colorless foam. 1 H-NMR (300MHz, CDCl 3 ): 7.45(,4H),7.10(d,2H),7.05(s,1H),6.90(s,1H),6.60(d,2H),4.85-4.70(m ,4H), 4.30(d,2H), 4.00(tr,2H), 3.50-3.30(m,18H), 2.00(m,2H), 1.90-1.50(m,12H).
按照实施例14的方法,使用不同的原料,得到以下式Ⅰ化合物:According to the method of Example 14, using different raw materials, the following compound of formula I was obtained:
顺-1,5-亚环辛基4-(4-氨基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(2-吡啶-4-基乙基氨基甲酰基)-1-哌嗪甲酸酯(化合物81);cis-1,5-cyclooctylene 4-(4-aminobenzylcarbamoyl)-1-piperazinecarboxylate 4-(2-pyridin-4-ylethylcarbamoyl)-1-piper Zinc carboxylate (compound 81);
顺-1,5-亚环辛基4-(4-氨基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-哌啶-4-基丙基)-1-哌啶甲酸酯(化合物82);和cis-1,5-cyclooctylene 4-(4-aminobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-piperidin-4-ylpropyl)-1-piperidinylmethyl ester (compound 82); and
顺-1,5-亚环辛基4-(4-氨基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(4-哌啶-4-基丁基)-1-哌嗪甲酸酯(化合物83);实施例15顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-咪唑-1-基丙基氨基甲酰基)-1-哌嗪甲酸酯(化合物84)cis-1,5-cyclooctylene 4-(4-aminobenzylcarbamoyl)-1-piperazine carboxylate 4-(4-piperidin-4-ylbutyl)-1-piperazine Ester (compound 83); Example 15 cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-imidazole-1- propylcarbamoyl)-1-piperazinecarboxylate (compound 84)
以下制备式Ⅰ化合物,其中R1是4-胍基苄基,R2是3-咪唑-1-基丙基,X1和X9每个是-NHC(O)-,X2和X8每个是1,4-亚哌嗪基,X3和X7每个是-C(O)O-,X4和X6每个是共价键,X5是顺-1,5-亚环辛基。The compound of formula I is prepared as follows, wherein R 1 is 4-guanidinobenzyl, R 2 is 3-imidazol-1-ylpropyl, X 1 and X 9 are each -NHC(O)-, X 2 and X 8 Each is 1,4-piperazinyl, each of X 3 and X 7 is -C(O)O-, each of X 4 and X 6 is a covalent bond, and X 5 is cis-1,5-ylidene Cyclooctyl.
把实施例14制备的顺-1,5-亚环辛基4-(4-氨基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-咪唑-1-基丙基氨基甲酰基)-1-哌嗪甲酸酯溶于甲醇中,加入乙醚和过量的HCl(4M,二噁烷中),浓缩化合物并真空干燥,加入过量的氨基氰(1.0g),将混合物在65℃加热2小时,得到黄色溶液。将混合物冷却到室温,用乙醚(3X,10ml)研制,不溶残留物溶于水中用制备性的反相HPLC从含水混合物中提纯,得到顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(3-咪唑-1-基丙基氨基甲酰基)-1-哌嗪甲酸酯,为无色无定型固体。电子喷雾法,LRMS:计算值(C34H51N11O6)MH+:710.9:测定值MH+:710.6。The cis-1,5-cyclooctylene 4-(4-aminobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-imidazol-1-ylpropylaminomethyl) prepared in Example 14 Acyl)-1-piperazinecarboxylate was dissolved in methanol, diethyl ether and excess HCl (4M in dioxane) were added, the compound was concentrated and dried in vacuo, an excess of cyanamide (1.0 g) was added, and the mixture was heated at 65 °C for 2 hours to obtain a yellow solution. The mixture was cooled to room temperature, triturated with diethyl ether (3X, 10 mL), and the insoluble residue was dissolved in water and purified from the aqueous mixture by preparative reverse-phase HPLC to give cis-1,5-cyclooctylene 4-(4- Guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(3-imidazol-1-ylpropylcarbamoyl)-1-piperazinecarboxylate, a colorless amorphous solid. Electrospray method, LRMS: Calculated for (C 34 H 51 N 11 O 6 ) MH + : 710.9: Found MH + : 710.6.
按照实施例15的方法,使用不同的原料,得到以下式Ⅰ化合物:According to the method of Example 15, using different raw materials, the following compound of formula I was obtained:
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-(2-吡啶-1-基乙基氨基甲酰基)-1-哌嗪甲酸酯(化合物85);计算值(C35H50N10O6)MH+:707.9;测定值MH+:707.6。cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-(2-pyridin-1-ylethylcarbamoyl)-1- Piperazine carboxylate (Compound 85); Calcd. (C 35 H 50 N 10 O 6 ) MH + : 707.9; Found MH + : 707.6.
顺-1,5-亚环辛基3-哌啶-4-基丙基-1-哌嗪甲酸酯4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯(化合物86);和Cis-1,5-cyclooctylene 3-piperidin-4-ylpropyl-1-piperazinecarboxylate 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate ( Compound 86); and
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯4-哌啶-4-基丁基-1-哌啶甲酸酯(化合物87);计算值(C37H60N8O5)MH+:697.9;测定值MH+:697.7。实施例16顺-1,5-亚环辛基氯代甲酸酯4-苄氧羰基-1-哌嗪甲酸酯Cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 4-piperidin-4-ylbutyl-1-piperidinecarboxylate ( Compound 87); Calcd. for (C 37 H 60 N 8 O 5 ) MH + : 697.9; Found MH + : 697.7. Example 16 cis-1,5-cyclooctylene chloroformate 4-benzyloxycarbonyl-1-piperazinecarboxylate
在0℃下,把溶于二氯甲烷(25ml)中的1-哌嗪甲酸苄基酯(1.0g,4.53mmol,1.0当量)和DIEA(0.88ml,4.98mmol,1.1当量)滴加到实施例5制备的溶于二氯甲烷(25ml)中的顺-1,5-亚环辛基二(氯代甲酸酯)(1.2g,4.53mmol,1.0当量)中,反应混合物搅拌22小时,同时温热到室温,混合物在二氯甲烷,0.05N盐酸水溶液和饱和氯化钠水溶液之间分配,用硫酸钠干燥有机层并浓缩,残留物用快速柱色谱法提纯残留物,用20%和30%的乙酸乙酯的己烷溶液洗脱,得到顺-1,5-亚环辛基氯代甲酸酯4-苄氧羰基-1-哌嗪甲酸酯(0.81g,1.81mmool,40%),为黄色油状物。IR:2939(s),2863(m),1770(s),1732(s),1696(s);1H-NMR(300MHz,CDCl3):7.35(s,5H),5.15(s,2H),4.95(m,1H),4.75(m,1H),3.45(s,8H),1.50-2.05(m,12H)。At 0°C, 1-piperazinecarboxylic acid benzyl ester (1.0g, 4.53mmol, 1.0eq) and DIEA (0.88ml, 4.98mmol, 1.1eq) dissolved in dichloromethane (25ml) were added dropwise to the In the cis-1,5-cyclooctylene bis(chloroformate) (1.2 g, 4.53 mmol, 1.0 equivalent) dissolved in dichloromethane (25 ml) prepared in Example 5, the reaction mixture was stirred for 22 hours, While warming to room temperature, the mixture was partitioned between dichloromethane, 0.05N aqueous hydrochloric acid and saturated aqueous sodium chloride, the organic layer was dried over sodium sulfate and concentrated, and the residue was purified by flash column chromatography with 20% and 30% ethyl acetate in hexane was eluted to give cis-1,5-cyclooctyl chloroformate 4-benzyloxycarbonyl-1-piperazinecarboxylate (0.81g, 1.81mmool, 40 %), as a yellow oil. IR: 2939(s), 2863(m), 1770(s), 1732(s), 1696(s); 1 H-NMR (300MHz, CDCl 3 ): 7.35(s, 5H), 5.15(s, 2H ), 4.95(m,1H), 4.75(m,1H), 3.45(s,8H), 1.50-2.05(m,12H).
实施例171-[顺-5-(4-苄氧羰基哌嗪-1-基羰基氧)环辛基氧羰基]-4-哌啶羧酸Example 171-[cis-5-(4-benzyloxycarbonylpiperazin-1-ylcarbonyloxy)cyclooctyloxycarbonyl]-4-piperidinecarboxylic acid
在0℃下将异3-哌啶甲酸(75mg.0.58mmol,1.1当量)和DIEA(0.23ml,1.33mmol,2.5当量)加入到实施例16制备的溶于二氯甲烷(10ml)中的顺-1,5-亚环辛基氯代甲酸酯4-苄氧羰基-1-哌嗪甲酸酯(0.24g,0.53mmol,1.0当量)中,得到白色悬浮液,搅拌悬浮液18小时,同时温热到室温,反应混合物在二氯甲烷和0.05N盐酸水溶液之间分配,浓缩有机相,得到1-[顺-5-(4-苄氧羰基哌嗪-1-基羰基氧)环辛基氧羰基]-4-哌啶羧酸(0.36g),为无色油状物。1HNMR(300MHz,CDCl3):7.35(s,5H),5.15(s,2H),4.75(m,2H),3.90(m,1H),3.45(s,8H),2.75(m,1H),2.50(m,3H),1.50-1.90(m,16H)。Add iso-3-piperidinecarboxylic acid (75 mg, 0.58 mmol, 1.1 eq) and DIEA (0.23 ml, 1.33 mmol, 2.5 eq) to the cis -1,5-Cyclooctylene chloroformate 4-benzyloxycarbonyl-1-piperazinecarboxylate (0.24g, 0.53mmol, 1.0eq), a white suspension was obtained, and the suspension was stirred for 18 hours, While warming to room temperature, the reaction mixture was partitioned between dichloromethane and 0.05N aqueous hydrochloric acid, and the organic phase was concentrated to give 1-[cis-5-(4-benzyloxycarbonylpiperazin-1-ylcarbonyloxy)cyclooctyl oxycarbonyl]-4-piperidinecarboxylic acid (0.36 g) as a colorless oil. 1 HNMR(300MHz, CDCl 3 ):7.35(s,5H),5.15(s,2H),4.75(m,2H),3.90(m,1H),3.45(s,8H),2.75(m,1H) ,2.50(m,3H),1.50-1.90(m,16H).
实施例18顺-1,5-亚环辛基4-苄氧羰基-1-哌嗪甲酸酯4[2-(1-叔丁氧羰基哌啶-4-基)乙基氨基甲酰基]-1-哌啶甲酸酯Example 18 cis-1,5-cyclooctylene 4-benzyloxycarbonyl-1-piperazinecarboxylate 4[2-(1-tert-butoxycarbonylpiperidin-4-yl)ethylcarbamoyl] -1-piperidinecarboxylate
将1-羟基苯并三唑(80mg.58.3mmol,1.1当量),叔-(2-氨基乙基)-1-哌啶甲酸叔丁基酯盐酸盐(0.14g,0.53mmol,1.0当量)和4-甲基吗啉(0.15ml,1.33mmol,2.5当量)加入到实施例17制备的溶于DMF(5ml)中的粗的1-[顺-5-(4-苄氧羰基哌嗪-1-基羰基氧)环辛基氧羰基]-4-哌啶羧酸(0.36g,0.53mmol,1.0当量)中,于0℃下将1-(3-二甲基氨基丙基)-3-乙基碳化二亚胺盐酸盐(0.13g,0.66mmol,1.25当量)加入到反应混合物中,于0℃搅拌溶液1.5小时,于23℃搅拌3天,反应混合物在二氯甲烷,0.05N盐酸水溶液,饱和碳酸氢钠水溶液,水(2份)和饱和氯化钠水溶液之间分配,干燥有机相(MgSO4),浓缩,用快速柱色谱法从残留物中提纯,用3%的甲醇的二氯甲烷溶液洗脱,得到顺-1,5-亚环辛基4-苄氧羰基-1-哌嗪甲酸酯4[2-(1-叔丁氧羰基哌啶-4-基)乙基氨基甲酰基]-1-哌啶甲酸酯(0.15g0.2mmol,37%,分两步),为黄包油状物。1HNMR(300MHz,CDCl3):7.35(s,5H),5.15(s,2H),4.80(m,2H),4.10(m,4H),3.45(m,10H),3.30(m,2H),2.70(m,2H),1.50-1.90(m,23H),1.45(s,9H);电子喷雾:LRMS:计算值(C40H62N5O9)(MH+):756.97;测定值757.0。1-Hydroxybenzotriazole (80mg.58.3mmol, 1.1 equivalent), tert-(2-aminoethyl)-1-piperidinecarboxylic acid tert-butyl ester hydrochloride (0.14g, 0.53mmol, 1.0 equivalent) and 4-methylmorpholine (0.15ml, 1.33mmol, 2.5eq) were added to the crude 1-[cis-5-(4-benzyloxycarbonylpiperazine- In 1-ylcarbonyloxy)cyclooctyloxycarbonyl]-4-piperidinecarboxylic acid (0.36g, 0.53mmol, 1.0eq), 1-(3-dimethylaminopropyl)-3 -Ethylcarbodiimide hydrochloride (0.13g, 0.66mmol, 1.25 equivalents) was added to the reaction mixture, the solution was stirred at 0°C for 1.5 hours, and at 23°C for 3 days, the reaction mixture was dichloromethane, 0.05N Partition between aqueous hydrochloric acid, saturated aqueous sodium bicarbonate, water (2 parts) and saturated aqueous sodium chloride, dry the organic phase (MgSO 4 ), concentrate, and purify from the residue by flash column chromatography with 3% methanol Eluted with dichloromethane solution to give cis-1,5-cyclooctylene 4-benzyloxycarbonyl-1-piperazinecarboxylate 4[2-(1-tert-butoxycarbonylpiperidin-4-yl) Ethylcarbamoyl]-1-piperidinecarboxylate (0.15g0.2mmol, 37%, in two steps), as yellow bag oil. 1 HNMR(300MHz, CDCl 3 ):7.35(s,5H),5.15(s,2H),4.80(m,2H),4.10(m,4H),3.45(m,10H),3.30(m,2H) , 2.70(m,2H), 1.50-1.90(m,23H), 1.45(s,9H); Electron spray: LRMS: Calculated (C 40 H 62 N 5 O 9 )(MH + ):756.97; Measured value 757.0.
实施例19顺-1,5-亚环辛基1-哌嗪甲酸酯4[2-(1-叔丁氧羰基哌啶-4-基)乙基氨基甲酰基1-1-哌啶甲酸酯Example 19 cis-1,5-cyclooctylene 1-piperazinecarboxylate 4[2-(1-tert-butoxycarbonylpiperidin-4-yl)ethylcarbamoyl 1-1-piperidinylmethyl Ester
在氮气氛下将乙醇(3ml)加入到实施例18制备的顺-1,5-亚环辛基4-苄氧羰基-1-哌嗪甲酸酯4[2-(1-叔丁氧羰基哌啶-4-基)乙基氨基甲酰基]-1-哌嗪甲酸酯(0.15g,0.20mmol,1.0当量)中,加入5%钯/炭(75mg,0.50wt当量)中,混合物于氢气气氛(1atm)和23℃下搅拌17小时,将反应混合物放于氮气氛下,过滤.浓缩滤液得到顺-1,5-亚环辛基1-哌嗪甲酸酯4[2-(1-叔丁氧羰基哌啶-4-基)乙基氨基甲酰基]-1-哌啶甲酸酯(110mg,0.18mmol,90%,),为无色油状物。1HNMR(300MHz,CDCl3):5.5(m,1H),4.9(m,2H),4.75(m,1H),4.1(m,4H),3.45(m,4H),3.25(m,2H),2.60-2.85(m,8H),2.10(m,1H),1.50-1.95(m,23H),1.45(s,9H);电子喷雾:LRMS:计算值(C32H56N5O7)(MH+):622.83;测定值622.7。实施例20顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基-l-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌啶甲酸酯(化合物88)Ethanol (3ml) was added to the cis-1,5-cyclooctylene 4-benzyloxycarbonyl-1-piperazinecarboxylate 4[2-(1-tert-butoxycarbonyl) prepared in Example 18 under a nitrogen atmosphere Piperidin-4-yl) ethylcarbamoyl]-1-piperazinecarboxylate (0.15g, 0.20mmol, 1.0 equivalent), was added to 5% palladium/carbon (75mg, 0.50wt equivalent), and the mixture was Stir under hydrogen atmosphere (1 atm) and 23 ℃ for 17 hours, put the reaction mixture under nitrogen atmosphere, and filter. -tert-butoxycarbonylpiperidin-4-yl)ethylcarbamoyl]-1-piperidinecarboxylate (110mg, 0.18mmol, 90%,), a colorless oil. 1 HNMR (300MHz, CDCl 3 ): 5.5(m, 1H), 4.9(m, 2H), 4.75(m, 1H), 4.1(m, 4H), 3.45(m, 4H), 3.25(m, 2H) ,2.60-2.85(m,8H),2.10(m,1H),1.50-1.95(m,23H),1.45(s,9H); Electron Spray: LRMS: Calculated (C 32 H 56 N 5 O 7 ) (MH + ): 622.83; found 622.7. Example 20 cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl-l-piperazinecarbamate 4(2-piperidin-4-ylethylcarbamoyl]- 1-piperidinecarboxylate (compound 88)
以下制备式Ⅰ化合物,其中R1是4-胍基苄基,R2是2-哌啶-4-基乙基,X1和X9每个是-NHC(O)-,X2是1,4-亚哌嗪基,X8是4,1-亚哌啶基,X3和X7每个是-C(O)O-,X4和X6每个是共价键,X5是顺-1,5-亚环辛基。The compound of formula I is prepared as follows, wherein R 1 is 4-guanidinobenzyl, R 2 is 2-piperidin-4-ylethyl, X 1 and X 9 are each -NHC(O)-, X 2 is 1 , 4-piperazinylene, X 8 is 4,1-piperidinylene, X 3 and X 7 are each -C(O)O-, X 4 and X 6 are each a covalent bond, X 5 is cis-1,5-cyclooctylene.
于0℃下将三光气(30mg,0.10mmol,0.58当量)和吡啶(30ml,0.39mmol,2.1当量)加入到实施例19制备的溶于二氯甲烷(2ml)中的顺-1,5-亚环辛基1-哌嗪甲酸酯4[2-(1-叔丁氧羰基哌啶-4-基)乙基氨基甲酰基]-1-哌嗪甲酸酯(0.11g,0.18mmol,1.0当量)中,于0℃下搅拌反应混合物3小时,混合物在二氯甲烷,0.05N盐酸水溶液和饱和氯化钠水溶液之间分配,干燥有机相(MgSO4),浓缩得到棕色油状残留物。往残留物中加入溶于DMF(2ml)中的4-胍基苄基胺盐酸盐(43mg,0.20mmol,1.1当量)和DIEA(0.16ml,0.90mmol,5.0当量),得到悬浮液,于23℃下搅拌悬浮液18.5小时,残留物溶于50%TFA的二氯甲烷(4ml)溶液中,于23℃下搅拌混合物45分钟,浓缩反应混合物,用乙醚研制残留物,真空干燥,用制备性的反相HPLC从残留物中提纯并冷冻干燥,得到顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基-1-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌啶甲酸酯,为无色无定型固体。电子喷雾,LRMS:计算值(C36H58N9O6)(MH+):712.91,测定值:712.8。Triphosgene (30mg, 0.10mmol, 0.58eq) and pyridine (30ml, 0.39mmol, 2.1eq) were added to the cis-1,5- Cyclooctylene 1-piperazinecarboxylate 4[2-(1-tert-butoxycarbonylpiperidin-4-yl)ethylcarbamoyl]-1-piperazinecarboxylate (0.11g, 0.18mmol, 1.0 equiv), the reaction mixture was stirred at 0°C for 3 hours, the mixture was partitioned between dichloromethane, 0.05N aqueous hydrochloric acid and saturated aqueous sodium chloride, the organic phase was dried (MgSO 4 ), and concentrated to give a brown oily residue. To the residue was added 4-guanidinobenzylamine hydrochloride (43 mg, 0.20 mmol, 1.1 equiv) and DIEA (0.16 ml, 0.90 mmol, 5.0 equiv) dissolved in DMF (2 ml) to obtain a suspension, which was The suspension was stirred at 23°C for 18.5 hours, the residue was dissolved in 50% TFA in dichloromethane (4 ml), the mixture was stirred at 23°C for 45 minutes, the reaction mixture was concentrated, the residue was triturated with ether, dried in vacuo, and prepared with Purified from the residue by precise reverse-phase HPLC and lyophilized to give cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl-1-piperazinecarboxylate 4(2-piperazine Pyridin-4-ylethylcarbamoyl]-1-piperidinecarboxylate as a colorless amorphous solid. Electron spray, LRMS: Calculated for (C 36 H 58 N 9 O 6 )(MH + ): 712.91 , measured value: 712.8.
按照实施例20,使用不同的原料,制备以下式Ⅰ化合物:According to Example 20, using different starting materials, the following compound of formula I was prepared:
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4(3-咪唑-4-基丙基氨基甲酰基]-1-哌嗪甲酸酯(化合物89),计算值(C34H50N10O6):MH+:695.9,测定值:MH+:695.4。cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4(3-imidazol-4-ylpropylcarbamoyl]-1-piperazine Formate (compound 89), calculated for (C 34 H 50 N 10 O 6 ): MH + : 695.9, found: MH + : 695.4.
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4(2-咪唑-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物90),计算值(C33H48N10O6):MH+:681.3,测定值:MH+:680.9。cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4(2-imidazol-4-ylethylcarbamoyl]-1-piperazine Formate (compound 90), calculated for (C 33 H 48 N 10 O 6 ): MH + : 681.3, found: MH + : 680.9.
顺-1.5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4(3-咪唑-1-基丙基氨基甲酰基]-1-哌嗪甲酸酯(化合物91),计算值(C34H50N10O6):MH+:695.9,测定值:MH+:694.9。cis-1.5-Cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4(3-imidazol-1-ylpropylcarbamoyl]-1-piperazinecarboxylate Ester (compound 91), calculated for (C 34 H 50 N 10 O 6 ): MH + : 695.9, found: MH + : 694.9.
顺-1,5-亚环辛基4-(4-胍基苯基乙酰基)-1-哌嗪甲酸酯4(4-咪唑-1-基丁基氨基甲酰基]-1-哌嗪甲酸酯(化合物92),计算值(C35H53N10O6):MH+:709.9,测定值:MH+:709.4。cis-1,5-cyclooctylene 4-(4-guanidinophenylacetyl)-1-piperazinecarboxylate 4(4-imidazol-1-ylbutylcarbamoyl]-1-piperazine Formate (compound 92), calculated for (C 35 H 53 N 10 O 6 ): MH + : 709.9, found: MH + : 709.4.
顺-1,5-亚环辛基4-(4-胍基苄基乙酰基)-1-哌嗪甲酸酯4(3-咪唑-1-基丙基氨基甲酰基]-1-哌嗪甲酸酯(化合物93),计算值(C35H51N10O6):MH+:709.9,测定值:MH+:710.4。cis-1,5-cyclooctylene 4-(4-guanidinobenzylacetyl)-1-piperazinecarboxylate 4(3-imidazol-1-ylpropylcarbamoyl]-1-piperazine Formate (compound 93), calculated for (C 35 H 51 N 10 O 6 ): MH + : 709.9, found: MH + : 710.4.
实施例213-哌啶-4-基丙酸盐酸盐Example 21 3-piperidin-4-yl propionate hydrochloride
把4-吡啶丙烯酸盐酸盐(12.0g,64.4mmol)和13.7g氧化铂悬浮于乙酸(80ml)中,在50-60psi下氢化12小时,混合物用水稀释,用硅藻土垫过滤,固体用水(200ml)洗涤,,真空浓缩合并的滤液和洗涤水,得到白色固体,将固体悬浮于少量甲醇中,混合物用乙醚(200ml)稀释,过滤分离颗粒物,用乙醚和己烷洗涤,空气干燥得到3-哌啶4-基丙酸盐酸盐(11.3g,58.1mmol,90%),为无色固体H1NMR(300MHz,MDSO-d6):8.75(br,s,2H),3.15(d,2H),2.75(t,1H),2.2(t,2H),1.75(d,2H),1.45(t,2H),1.25(br,q,2H)。4-Pyridine acrylate hydrochloride (12.0g, 64.4mmol) and 13.7g platinum oxide were suspended in acetic acid (80ml), hydrogenated at 50-60psi for 12 hours, the mixture was diluted with water, filtered through a Celite pad, and the solid was water (200ml), the combined filtrate and wash water were concentrated in vacuo to give a white solid, the solid was suspended in a small amount of methanol, the mixture was diluted with ether (200ml), the particulate was isolated by filtration, washed with ether and hexane, and air dried to give 3- Piperidin 4-ylpropion hydrochloride (11.3g, 58.1mmol, 90%), as a colorless solid H 1 NMR (300MHz, MDSO-d 6 ): 8.75(br,s,2H), 3.15(d, 2H), 2.75(t,1H), 2.2(t,2H), 1.75(d,2H), 1.45(t,2H), 1.25(br,q,2H).
实施例223-(1-叔丁氧羰基哌啶-4-基)丙酸Example 223-(1-tert-butoxycarbonylpiperidin-4-yl)propionic acid
把实施例21制备的3哌啶-4-丙酸(5.07g,26.2mmol)溶于2NNaOH(40ml,80mmo1),加入THF(40ml)和碳酸二(叔丁基)酯(6.21g,28.4mmol),得到悬浮液,将悬浮液搅拌22小时,用水稀释,真空浓缩,残留物用乙醚(2X,100ml)洗涤,水相用1.0N KHSO4水溶液酸化到pH2-3,用乙酸乙酯(3X,200ml)萃取,合并的有机相用食盐水洗涤,干燥(MgSO4),真空浓缩,得到3-[4-(1-叔丁氧羰基)-4-哌啶]丙酸(6.21g,24.1mmol,92%),为无色油状物,静置时结晶。H1NMR(300MHz,CDCl3):4.10(br,d,2H),2.65(br,t,2H),2.35(t,2H),1.70-.50(m,3H),1.45(s,9H),1.20-0.95(m,2H)。Dissolve 3-piperidine-4-propionic acid (5.07g, 26.2mmol) prepared in Example 21 in 2NNaOH (40ml, 80mmol), add THF (40ml) and di(tert-butyl)carbonate (6.21g, 28.4mmol) ), the suspension was obtained, the suspension was stirred for 22 hours, diluted with water, concentrated in vacuo, the residue was washed with diethyl ether (2X, 100ml), the aqueous phase was acidified to pH2-3 with 1.0N KHSO 4 aqueous solution, and washed with ethyl acetate (3X , 200ml), the combined organic phases were washed with brine, dried (MgSO 4 ), and concentrated in vacuo to obtain 3-[4-(1-tert-butoxycarbonyl)-4-piperidine]propionic acid (6.21g, 24.1 mmol, 92%), a colorless oil that crystallized on standing. H 1 NMR (300MHz, CDCl 3 ): 4.10(br,d,2H), 2.65(br,t,2H), 2.35(t,2H), 1.70-.50(m,3H), 1.45(s,9H ),1.20-0.95(m,2H).
实施例234-苄氧羰基-1-哌嗪甲酸叔丁基酯Example 234-Benzyloxycarbonyl-1-piperazinecarboxylic acid tert-butyl ester
用氯代甲酸苄基酯(2.0ml,2.39g,14.0mmol)处理溶于50ml冰冷却的二氯甲烷中的1-哌嗪甲酸叔丁基酯(2.01g,10.8mmo1)和DIEA(2.0ml,1.48g,11.5mmol),搅拌混合物42小时,在乙酸乙酯和0.5N的KHSO4之间分配,水相用乙酸乙酯萃取,合并的有机相用水和食盐水洗涤,干燥(MgSO4)并浓缩,用硅胶色谱法从残留物中提纯(乙酸乙酯∶己烷,1∶3),得到4-苄氧羰基-1-哌嗪甲酸叔丁基酯(3.33g,10.4mmol,96%),为无色固体。H1NMR(300MHz,CDCl3):7.35(b,s,5H),5.13(s,2H),3.55-3.25(m,8H),1.45(s,9H)。tert-Butyl-1-piperazinecarboxylate (2.01g, 10.8mmol) and DIEA (2.0ml , 1.48g, 11.5mmol), the mixture was stirred for 42 hours, partitioned between ethyl acetate and 0.5N KHSO 4 , the aqueous phase was extracted with ethyl acetate, the combined organic phases were washed with water and brine, dried (MgSO 4 ) and Concentrate and purify from the residue by silica gel chromatography (ethyl acetate:hexane, 1:3) to give tert-butyl 4-benzyloxycarbonyl-1-piperazinecarboxylate (3.33 g, 10.4 mmol, 96%) , is a colorless solid. H 1 NMR (300 MHz, CDCl 3 ): 7.35 (b, s, 5H), 5.13 (s, 2H), 3.55-3.25 (m, 8H), 1.45 (s, 9H).
实施例241-哌嗪甲酸苄基酯盐酸盐Example 241-Benzyl piperazinecarboxylate hydrochloride
把实施例23制备的4-苄氧羰基-1-哌嗪甲酸叔丁基酯(1.01g,3.16mmol)悬浮于4ml乙酸乙酯中,悬浮液在冰水浴中冷却,加入4NHCl(12ml,溶于1,4-二噁烷中),得到一溶液,溶液在冰浴上搅拌30分钟,再在室温下搅拌30分钟,反应混合物用乙醚(76ml)稀释,得到沉淀,过滤分离沉淀,用乙醚洗涤,真空干燥得到1-哌嗪甲酸苄基酯盐酸盐(740mg,2.78mmol,88%),为无色固体。H1NMR(300MHz,DMSO-d6):9.25(brs,2H),7.33(s,5H),5.06(s,2H),3.58(brs,4H),3.04(t,4H)。实施例254-[2-(4-苄氧羰基哌嗪-1-基羰基氨基)乙基]-1-吡啶甲酸叔丁基酯Suspend tert-butyl 4-benzyloxycarbonyl-1-piperazinecarboxylate (1.01 g, 3.16 mmol) prepared in Example 23 in 4 ml of ethyl acetate, cool the suspension in an ice-water bath, add 4N HCl (12 ml, dissolve In 1,4-dioxane), a solution was obtained. The solution was stirred on an ice bath for 30 minutes, and then stirred at room temperature for 30 minutes. The reaction mixture was diluted with diethyl ether (76ml) to obtain a precipitate, which was separated by filtration and washed with diethyl ether Washing and drying in vacuo gave benzyl 1-piperazinecarboxylate hydrochloride (740 mg, 2.78 mmol, 88%) as a colorless solid. H 1 NMR (300 MHz, DMSO-d 6 ): 9.25 (brs, 2H), 7.33 (s, 5H), 5.06 (s, 2H), 3.58 (brs, 4H), 3.04 (t, 4H). Example 254-[2-(4-Benzyloxycarbonylpiperazin-1-ylcarbonylamino)ethyl]-1-pyridinecarboxylic acid tert-butyl ester
把实施例22制备的3-[4-(1-叔丁氧羰基)-4-哌啶基]丙酸(2.16g,8.4mmol)溶于无水苯中(28ml)用三乙胺(1.35ml,951mg,9.40mmol)和二苯基磷酰叠氮(2.05ml,2.62g,9.53mmol)处理,反应混合物逐渐加热到回流,保持回流3.5小时,混合物冷却到室温,然后滴加到实施例24制备的于无水二氯甲烷(10ml)中的1-哌嗪甲酸苄基酯盐酸盐(2.44g,9.19mmol)和三乙胺(1.40ml)的悬浮液中,搅拌反应混合物43小时,用乙酸乙酯和0.5N KHSO4稀释,有机相用水,碳酸氢钠水溶液和食盐水洗涤,干燥(Na2SO4)并浓缩,用硅胶色谱法从残留物中提纯(先用4∶1的乙酸乙酯-己烷,然后用纯乙醚),得到4-[2-(4-苄氧羰基哌嗪-1-基羰基氨基)乙基]-1-哌啶甲酸叔丁基酯(3.84g,8.1mmol,96%),为白色固体。H1NMR(300MHz,CDCl3):7.35(s,5H),5.15(s,2H),4.50(brt,,1H),4.05(brs,2H)3.55-3.45(m,4H),3.40-3.30(m,4H),3.25(q,2H),2.65(t,2H),1.70(s,2H),1.45(s,11H),1.20-1.00(m,2H)。3-[4-(1-tert-butoxycarbonyl)-4-piperidinyl]propionic acid (2.16g, 8.4mmol) prepared in Example 22 was dissolved in anhydrous benzene (28ml) and triethylamine (1.35 ml, 951mg, 9.40mmol) and diphenylphosphoryl azide (2.05ml, 2.62g, 9.53mmol), the reaction mixture was gradually heated to reflux and maintained at reflux for 3.5 hours, the mixture was cooled to room temperature, and then added dropwise to Example 24 A suspension of 1-piperazinecarboxylate benzyl hydrochloride (2.44 g, 9.19 mmol) and triethylamine (1.40 ml) in anhydrous dichloromethane (10 ml) was prepared and the reaction mixture was stirred for 43 hours , diluted with ethyl acetate and 0.5N KHSO 4 , the organic phase was washed with water, aqueous sodium bicarbonate and brine, dried (Na 2 SO 4 ) and concentrated, and the residue was purified by silica gel chromatography (first with 4:1 ethyl acetate-hexane, then pure diethyl ether) to give tert-butyl 4-[2-(4-benzyloxycarbonylpiperazin-1-ylcarbonylamino)ethyl]-1-piperidinecarboxylate (3.84 g , 8.1 mmol, 96%) as a white solid. H 1 NMR(300MHz,CDCl 3 ):7.35(s,5H),5.15(s,2H),4.50(brt,,1H),4.05(brs,2H)3.55-3.45(m,4H),3.40-3.30 (m,4H), 3.25(q,2H), 2.65(t,2H), 1.70(s,2H), 1.45(s,11H), 1.20-1.00(m,2H).
实施例26顺-1,5-亚环辛基氯代甲酸酯4-[2-(4-叔丁氧羰基哌啶-4-基)乙基氨基甲酰基]-1-哌嗪甲酸酯Example 26 cis-1,5-cyclooctylene chloroformate 4-[2-(4-tert-butoxycarbonylpiperidin-4-yl)ethylcarbamoyl]-1-piperazinecarboxylic acid ester
把实施例25制备的叔丁基4-[2-(4-苄氧羰基哌嗪-1-基羰基氨基)乙基]-1-哌嗪甲酸酯(2.03g,4.28mmol)和10%的钯/炭(570mg)溶于乙醇(19ml)中的悬浮液在大气压下氢化过夜,过滤反应混合物,催化剂用乙醇洗涤,真空浓缩滤液和洗涤液,残留物溶于二氯甲烷(30ml)中,,用DIEA(500ml)处理,溶液滴加到实施例5制备的溶于冰冷却的二氯甲烷(75ml)中的顺-1,5-亚环辛基二(氯代甲酸酯)(4.15g,15.4mmol)中,搅拌反应混合物过夜,用0.5N KHSO4和二氯甲烷稀释,水相用二氯甲烷萃取,合并的有机相用食盐水洗涤,干燥(MgSO4)并浓缩,用硅胶色谱法从残留物中提纯(先用3∶1的乙酸乙酯-己烷,然后用纯乙酸乙酯),得到顺-1,5-亚环辛基氯代甲酸酯4-[2-(4-叔丁氧羰基哌啶-4-基)乙基氨基甲酰基]-1-哌嗪甲酸酯(1.02g,1.8mmol,42%),为浅黄色油状物。1H-NMR(300MHz,CDCl3):5.00-4.90(m,1H),4.85-4.75(m,1H),4.35(brt,1H),4.05(brd,2H),3.50-3.40(m,4H),3.35-3.30(m,4H),3.25(q,2H),2.65(t,2H),2.05-1.55(m,17H),1.40(s,9H),1.25-1.00(m,2H)。tert-butyl 4-[2-(4-benzyloxycarbonylpiperazin-1-ylcarbonylamino)ethyl]-1-piperazinecarboxylate (2.03g, 4.28mmol) prepared in Example 25 and 10% A suspension of palladium/charcoal (570 mg) dissolved in ethanol (19 ml) was hydrogenated overnight at atmospheric pressure, the reaction mixture was filtered, the catalyst was washed with ethanol, the filtrate and washings were concentrated in vacuo, and the residue was dissolved in dichloromethane (30 ml) ,, was treated with DIEA (500ml), and the solution was added dropwise to the cis-1,5-cyclooctylene bis(chloroformate) dissolved in ice-cooled methylene chloride (75ml) prepared in Example 5 ( 4.15 g, 15.4 mmol), the reaction mixture was stirred overnight, diluted with 0.5N KHSO 4 and dichloromethane, the aqueous phase was extracted with dichloromethane, the combined organic phases were washed with brine, dried (MgSO 4 ) and concentrated, and washed with Purification from the residue by silica gel chromatography (first with 3:1 ethyl acetate-hexane, then with pure ethyl acetate) gave cis-1,5-cyclooctylene chloroformate 4-[2 -(4-tert-butoxycarbonylpiperidin-4-yl)ethylcarbamoyl]-1-piperazinecarboxylate (1.02g, 1.8mmol, 42%), as light yellow oil. 1 H-NMR(300MHz, CDCl 3 ):5.00-4.90(m,1H),4.85-4.75(m,1H),4.35(brt,1H),4.05(brd,2H),3.50-3.40(m,4H ), 3.35-3.30(m,4H), 3.25(q,2H), 2.65(t,2H), 2.05-1.55(m,17H), 1.40(s,9H), 1.25-1.00(m,2H).
实施例274-氰基苯基乙酸Example 274-cyanophenylacetic acid
把2-(4-氰基苯基)乙醇(5.00g,34.0mmol)溶于丙酮(140ml)中并冷却到10-15℃,滴加溶于含水硫酸中的CrO3,保持内温在30℃以下,直到橙色不消失,得到悬浮液。搅拌悬浮液45分钟并过滤,固体用丙酮(150ml)洗涤,合并的滤液和洗涤液与2-丙醇(20ml)一起搅拌30分钟,过滤混合物,真空浓缩滤液,把残留物溶于乙酸乙酯中,溶液用0.5NKHSO4,水和食盐水洗涤,浓缩,残留物溶于二氯甲烷中,溶液用溶于水(100ml)中的氢氧化钠(1.56g)处理,水相用二氯甲烷处理,用浓盐酸酸化到pH1-2,得到沉淀,沉淀用水洗涤,空气干燥,真空干燥后得到4-氰基苯基乙酸(3.36g,20.7mmol,61%),为白色粉末。1H-NMR(300MHz,CDCl3):7.63(d,2H),7.40(d,2H),3.72(s,2H)。Dissolve 2-(4-cyanophenyl)ethanol (5.00g, 34.0mmol) in acetone (140ml) and cool to 10-15°C, add CrO 3 dissolved in aqueous sulfuric acid dropwise, and keep the internal temperature at 30 Below ℃, until the orange color does not disappear, a suspension is obtained. The suspension was stirred for 45 minutes and filtered, the solid was washed with acetone (150ml), the combined filtrate and washings were stirred with 2-propanol (20ml) for 30 minutes, the mixture was filtered, the filtrate was concentrated in vacuo, and the residue was dissolved in ethyl acetate , the solution was washed with 0.5NKHSO 4 , water and brine, concentrated, the residue was dissolved in dichloromethane, the solution was treated with sodium hydroxide (1.56 g) dissolved in water (100 ml), and the aqueous phase was treated with dichloromethane , acidified to pH 1-2 with concentrated hydrochloric acid to obtain a precipitate, which was washed with water, air-dried, and vacuum-dried to give 4-cyanophenylacetic acid (3.36g, 20.7mmol, 61%) as a white powder. 1 H-NMR (300 MHz, CDCl 3 ): 7.63 (d, 2H), 7.40 (d, 2H), 3.72 (s, 2H).
实施例284-(4-氰基苯基乙酰基)-1-哌嗪甲酸叔丁基酯Example 28 tert-butyl 4-(4-cyanophenylacetyl)-1-piperazinecarboxylate
把实施例27制备的4-氰基苯基乙酸(890mg,5.52mmol),二氯乙烷(1.16g,6.07mmol)和1-羟基苯并三唑水合物(820mg,6.07mmol)的混合物悬浮于THF(18ml)中,往得到的均相溶液中加入1-哌嗪甲酸叔丁基酯(1.04g,5.60mmol)和DIEA,真空浓缩溶液,残留物用0.2N KHSO4处理,过滤混合物,收集固体,用水洗涤,真空干燥,得到4-(4-氰基苯基乙酰基)-1-哌嗪甲酸叔丁基酯(1.68g,5.1mmol,92%),为固体。1H-NMR(300MHz,CDCl3):7.70(d,2H),7.35(d,2H),3.80(s,2H),3.70-3.60(m,2H),3.45-3.30(m,6H),1.40(s,9H)。实施例294-[4-(N-羟基脒基)苯基乙酰基]-1-哌嗪甲酸叔丁基酯Suspend the mixture of 4-cyanophenylacetic acid (890mg, 5.52mmol), dichloroethane (1.16g, 6.07mmol) and 1-hydroxybenzotriazole hydrate (820mg, 6.07mmol) prepared in Example 27 To the resulting homogeneous solution in THF (18ml) was added tert-butyl-piperazinecarboxylate (1.04g, 5.60mmol) and DIEA, the solution was concentrated in vacuo, the residue was treated with 0.2N KHSO4 , the mixture was filtered, The solid was collected, washed with water, and dried in vacuo to afford tert-butyl 4-(4-cyanophenylacetyl)-1-piperazinecarboxylate (1.68 g, 5.1 mmol, 92%) as a solid. 1 H-NMR (300MHz, CDCl 3 ): 7.70(d,2H),7.35(d,2H),3.80(s,2H),3.70-3.60(m,2H),3.45-3.30(m,6H), 1.40(s,9H). Example 294-[4-(N-hydroxyamidino)phenylacetyl]-1-piperazinecarboxylic acid tert-butyl ester
用羟胺盐酸盐(461mg,6.63mmol)和三乙胺(924ml,671mg,6.63mmol)处理溶于无水乙醇(10ml)中的实施例28制备的4-(4-氰基苯基乙酰基)-1-哌嗪甲酸叔丁基酯(1.68g,5.1mmol),混合物加热回流3.5小时,冷却到室温,真空浓缩,,残留物溶于乙醇,过滤,滤液冷却过夜,得到结晶产品,过滤分离结晶,用冷乙醇洗涤,空气干燥,得到4·[4-(N-羟基脒基)苯基乙酰基]·1-哌嗪甲酸叔丁基酯(1.62g,4.5mmol,88%)。With hydroxylamine hydrochloride (461mg, 6.63mmol) and triethylamine (924ml, 671mg, 6.63mmol) process the 4-(4-cyanophenylacetyl group prepared in Example 28) dissolved in absolute ethanol (10ml) )-1-tert-butyl piperazinecarboxylate (1.68g, 5.1mmol), the mixture was heated to reflux for 3.5 hours, cooled to room temperature, concentrated in vacuo, the residue was dissolved in ethanol, filtered, and the filtrate was cooled overnight to obtain a crystalline product, which was filtered The crystals were separated, washed with cold ethanol, and air-dried to obtain tert-butyl 4·[4-(N-hydroxyamidino)phenylacetyl]·1-piperazinecarboxylate (1.62g, 4.5mmol, 88%).
1H-NMR(300MHz,DMSO-d6):9.60(s,1H),7.60(d,2H),7.20(d,2H),5.75(s,2H),3.70(s,2H),3.40(brs,4H),3.25(brs,4H),1.40(s,9H)。 1 H-NMR (300MHz, DMSO-d 6 ): 9.60(s, 1H), 7.60(d, 2H), 7.20(d, 2H), 5.75(s, 2H), 3.70(s, 2H), 3.40( brs,4H), 3.25(brs,4H), 1.40(s,9H).
实施例304-哌嗪-1-基羰基甲基苄脒双(三氟乙酸酯)Example 304-piperazin-1-ylcarbonylmethylbenzamidine bis(trifluoroacetate)
把实施例29制备的4-[4-(N-羟基脒基)苯基乙酰基]-1-哌嗪甲酸叔丁基酯(653mg,1.81mmol)和10%钯/炭(200mg)悬浮于乙酸(12ml)中,往悬浮液中通入氢气过夜,过滤反应混合物,用乙酸洗涤催化剂,真空浓缩合并的滤液和洗涤液,残留物溶于TFA中,静置溶液1小时,真空浓缩,残留物从二氯甲烷和甲醇的混合物中共蒸发,然后悬浮于乙醚中,过滤收集颗粒物,干燥后得到4-哌嗪-1-基羰基甲基苄脒双(三氟乙酸酯)(1.04g,1.81mmol,100%),为白色固体。1H-NMR(300MHz,DMSO-d6):9.30(d,4H),9.15(brs,2H),7.70(d,2H),7.40(d,2H),3.85(s,2H),3.65(brd,4H),4.20-3.90(m,4H)。tert-butyl 4-[4-(N-hydroxyamidino)phenylacetyl]-1-piperazinecarboxylate (653 mg, 1.81 mmol) prepared in Example 29 and 10% palladium/carbon (200 mg) were suspended in In acetic acid (12ml), hydrogen gas was passed into the suspension overnight, the reaction mixture was filtered, the catalyst was washed with acetic acid, the combined filtrate and washings were concentrated in vacuo, the residue was dissolved in TFA, the solution was allowed to stand for 1 hour, and the residue was concentrated in vacuo. The product was co-evaporated from a mixture of dichloromethane and methanol, then suspended in ether, and the particles were collected by filtration and dried to give 4-piperazin-1-ylcarbonylmethylbenzamidine bis(trifluoroacetate) (1.04g, 1.81mmol, 100%), as a white solid. 1 H-NMR (300MHz, DMSO-d 6 ): 9.30(d,4H), 9.15(brs,2H), 7.70(d,2H), 7.40(d,2H), 3.85(s,2H), 3.65( brd, 4H), 4.20-3.90 (m, 4H).
实施例31顺-1,5-亚环辛基4-(4-脒基苯基乙酰基)-1-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物94)Example 31 cis-1,5-cyclooctylene 4-(4-amidinophenylacetyl)-1-piperazinecarboxylate 4(2-piperidin-4-ylethylcarbamoyl]- 1-piperazine carboxylate (compound 94)
以下制备式Ⅰ化合物,其中R1是4-脒基苄基,R2是2-哌啶-4-基乙基,X1和X9每个是-NHC(O)-,X2和X8每个是1,4-亚哌嗪基,X3和X7每个是-C(O)O-,X4和X6每个是共价键,X5是顺-1,5-亚环辛基。The compound of formula I is prepared as follows, wherein R 1 is 4-amidinobenzyl, R 2 is 2-piperidin-4-ylethyl, X 1 and X 9 are each -NHC(O)-, X 2 and X 8 each is 1,4-piperazinylene, each of X 3 and X 7 is -C(O)O-, each of X 4 and X 6 is a covalent bond, and X 5 is cis-1,5- Cyclooctylene.
把实施例30制备的4-哌嗪-1-基羰基甲基苄脒双(三氟乙酸酯)(80mg,0.17mmo1)溶于DMF(1.0ml)中,溶液用DIEA(150ml)处理,加入实施例26制备的溶于DMF(1.0ml)中的顺-1,5-亚环辛基氯代甲酸酯4-[2-(4-叔丁氧羰基哌嗪-1-基)乙基氨基甲酰基]-1-哌嗪甲酸酯(100mg),将混合物搅拌过夜,真空浓缩,残留物溶于二氯甲烷和TFA(1∶1)中,真空浓缩混合物,残留物用乙醚研制,得到泡沫状残留物。用制备性的反相HPLC从残留物中提纯,将纯化的馏分冷冻干燥,得到顺-1,5-亚环辛基4-(4-脒基苯基乙酰基)-1-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯,为无色固体。电子喷雾:LRMS:计算值(C35H54N8O8)MH+:683.9;MH2 +2/2:342.5;测定值:MH+:683.8;MH2 +2/2:342.3。The 4-piperazin-1-ylcarbonylmethylbenzamidine bis(trifluoroacetate) (80 mg, 0.17 mmol) prepared in Example 30 was dissolved in DMF (1.0 ml), and the solution was treated with DIEA (150 ml), Add cis-1,5-cyclooctylene chloroformate 4-[2-(4-tert-butoxycarbonylpiperazin-1-yl)ethyl prepared in Example 26 dissolved in DMF (1.0ml) Carbamoyl]-1-piperazinecarboxylate (100 mg), the mixture was stirred overnight, concentrated in vacuo, the residue was dissolved in dichloromethane and TFA (1:1), the mixture was concentrated in vacuo, the residue was triturated with ether , giving a foamy residue. Purification from the residue by preparative reverse-phase HPLC and lyophilization of the purified fractions afforded cis-1,5-cyclooctylidene 4-(4-amidinophenylacetyl)-1-piperazinecarboxylic acid Ester 4(2-piperidin-4-ylethylcarbamoyl ] -1-piperazinecarboxylate as a colorless solid. Electron spray: LRMS: Calcd . for ( C35H54N8O8 )MH + : 683.9; MH 2 +2 /2: 342.5; Found: MH + : 683.8; MH 2 +2 /2 : 342.3.
按照实施例31的方法,使用不同的原料,制备以下式Ⅰ化合物:According to the method of Example 31, using different starting materials, the following compound of formula I was prepared:
顺-1,5-亚环辛基4-(1-脒基哌啶-4-基乙酰基)-1-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物95);计算值(C34H59N9O6)MH+:690.9;测定值:MH+:690.6;cis-1,5-cyclooctylene 4-(1-amidinopiperidin-4-ylacetyl)-1-piperazinecarboxylate 4(2-piperidin-4-ylethylcarbamoyl] -1-piperazinecarboxylate (Compound 95); Calculated (C 34 H 59 N 9 O 6 ) MH + : 690.9; Found: MH + : 690.6;
顺-1,5-亚环辛基4-(4-脒基苯甲酰基氨基乙基)-1-哌啶甲酸酯4(2-哌啶-4-基乙基)氨基甲酰基]-1-哌嗪甲酸酯(化合物96);计算值(C36H56N8O6)MH+:697.9;测定值:MH+:697.7;cis-1,5-cyclooctylene 4-(4-amidinobenzoylaminoethyl)-1-piperidinecarboxylate 4(2-piperidin-4-ylethyl)carbamoyl]- 1-Piperazinecarboxylate (Compound 96); Calcd. (C 36 H 56 N 8 O 6 ) MH + : 697.9; Found: MH + : 697.7;
顺-1,5-亚环辛基4-(4-脒基苄基氨基甲酰基)-1-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物97);计算值(C35H55N9O6)MH+:698.9;测定值:MH+:698.7;cis-1,5-cyclooctylene 4-(4-amidinobenzylcarbamoyl)-1-piperazinecarboxylate 4(2-piperidin-4-ylethylcarbamoyl]-1- Piperazine carboxylate (Compound 97); Calcd. (C 35 H 55 N 9 O 6 ) MH + : 698.9; Found: MH + : 698.7;
顺-1,5-亚环辛基4-(4-脒基苯磺酰基氨基甲基)-1-哌啶甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物98);计算值(C35H56N8O7)MH+:733.9;测定值:MH+:733.4;cis-1,5-cyclooctylene 4-(4-amidinobenzenesulfonylaminomethyl)-1-piperidinecarboxylate 4(2-piperidin-4-ylethylcarbamoyl]-1 - piperazine carboxylate (Compound 98); Calcd. (C 35 H 56 N 8 O 7 ) MH + : 733.9; Found: MH + : 733.4;
顺-1,5-亚环辛基4-[2-(1-脒基哌啶4-基)乙基氨基甲酰基]-1-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物99);计算值(C35H62N10O6)MH+:719.9;测定值:MH+:719.5;cis-1,5-cyclooctylene 4-[2-(1-amidinopiperidin4-yl)ethylcarbamoyl]-1-piperazinecarboxylate 4(2-piperidin-4-yl Ethylcarbamoyl]-1-piperazinecarboxylate (Compound 99); Calculated for (C 35 H 62 N 10 O 6 ) MH + : 719.9; Found: MH + : 719.5;
顺-1,5-亚环辛基4-(4-脒基苯基氨基甲酰基甲基)-1-哌啶甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物100);计算值(C36H56N8O6)MH+:697.9;测定值:MH+:695.6;cis-1,5-cyclooctylene 4-(4-amidinophenylcarbamoylmethyl)-1-piperidinecarboxylate 4(2-piperidin-4-ylethylcarbamoyl]- 1-Piperazinecarboxylate (Compound 100); Calcd. (C 36 H 56 N 8 O 6 ) MH + : 697.9; Found: MH + : 695.6;
顺-1,5-亚环辛基4-(4-N-甲氧羰基脒基苄基氨基甲酰基)-1-哌嗪甲酸酯4(2-哌啶-4-基乙基氨基甲酰基]-1-哌嗪甲酸酯(化合物101);计算值(C37H57N9O8)MH+:756.9;测定值:MH+:756.4;cis-1,5-cyclooctylene 4-(4-N-methoxycarbonylamidinobenzylcarbamoyl)-1-piperazinecarboxylate 4(2-piperidin-4-ylethylaminomethyl Acyl]-1-piperazinecarboxylate (Compound 101); Calculated for (C 37 H 57 N 9 O 8 ) MH + : 756.9; Found: MH + : 756.4;
顺-1,5-亚环辛基4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯3-哌啶-4-基丙基-1-哌啶甲酸酯(化合物102);计算值(C36H58N8O5)MH+:683.9;测定值:MH+:683.3。Cis-1,5-cyclooctylene 4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate 3-piperidin-4-ylpropyl-1-piperidinecarboxylate ( Compound 102); Calcd. for (C 36 H 58 N 8 O 5 ) MH + : 683.9; Found: MH + : 683.3.
实施例321,5-五亚甲基二[4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯](化合物103)Example 321,5-pentamethylenebis[4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate] (compound 103)
以下制备式Ⅰ化合物,其中R1和R2每个是4-胍基苄基,X1和X9每个是-NHC(O)-,X2和X8每个是1,4-亚哌嗪基,X3和X7每个是-C(O)O-,X4-X6-X5一起是1,5-五亚甲基。Compounds of formula I are prepared as follows, wherein R and R are each 4-guanidinobenzyl, X and X are each -NHC(O)-, X and X are each 1,4- Piperazinyl, X 3 and X 7 are each -C(O)O-, X 4 -X 6 -X 5 together are 1,5-pentamethylene.
在室温下用纯三氟乙酸(1ml)把4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸叔丁基酯三氟乙酸盐(306mg,0.62mmol)处理10分钟,得到无色匀相溶液。浓缩液体,油状残留物用乙醚(3X 10ml)研制,真空干燥,得到无色泡沫状物。把脱保护的哌嗪盐溶于DMF(2.5ml),随后加入二异丙基乙基胺(0.5ml,3.1mmol)和1,5-亚正戊基二(氯代甲酸酯)(70mg,0.31mmol),室温搅拌混合物1小时,浓缩反应混合物,残留物用乙醚(3X10ml)研制,真空干燥,粗产品溶于水(5ml),用制备反相HPLC提纯,冷冻干燥,得到1,5-五亚甲基二[4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯],为无色无定型固体。电子喷雾:LRMS:计算值(C33H48N12O6)MH+:709.8;测定值:709.3。4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate tert-butyl trifluoroacetate (306 mg, 0.62 mmol) was treated with neat trifluoroacetic acid (1 ml) for 10 minutes at room temperature, A colorless homogeneous solution was obtained. The liquid was concentrated and the oily residue was triturated with ether (3X 10ml) and dried in vacuo to give a colorless foam. The deprotected piperazine salt was dissolved in DMF (2.5ml), followed by the addition of diisopropylethylamine (0.5ml, 3.1mmol) and 1,5-n-pentylene bis(chloroformate) (70mg , 0.31mmol), the mixture was stirred at room temperature for 1 hour, the reaction mixture was concentrated, the residue was triturated with diethyl ether (3X10ml), dried in vacuo, the crude product was dissolved in water (5ml), purified by preparative reverse-phase HPLC, and freeze-dried to give 1,5 - Pentamethylenebis[4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate], a colorless amorphous solid. Electrospray: LRMS: Calculated for ( C33H48N12O6 )MH + : 709.8 ; Found : 709.3.
按照实施例32的方法,使用不同的原料,制备以下式Ⅰ化合物。Following the procedure of Example 32, using different starting materials, the following compound of formula I was prepared.
1,5-四亚甲基二[4-(4-胍基苄基氨基甲酰基)-1-哌嗪甲酸酯](化合物104),计算值(C32H46N12O6)MH+:695.8;测定值:MH+:695.8;1,5-Tetramethylenebis[4-(4-guanidinobenzylcarbamoyl)-1-piperazinecarboxylate] (compound 104), calculated for (C 32 H 46 N 12 O 6 )MH + : 695.8; measured value: MH + : 695.8;
4-胍基苄基4-guanidinobenzyl
4-{5-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]戊酰基}-1-哌嗪甲酰胺,(化合物105),计算值(C32H46N12O6)MH+:663.8;测定值:MH+:663.4;4-{5-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]pentanoyl}-1-piperazinecarboxamide, (compound 105), calculated (C 32 H 46 N 12 O 6 ) MH + : 663.8; Measured value: MH + : 663.4;
4-胍基苄基4-guanidinobenzyl
4-{6-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]己酰基}-1-哌嗪甲酰胺,(化合物106),计算值(C33H48N12O4)MH+:677.8;测定值:MH+:677.4;4-{6-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]hexanoyl}-1-piperazinecarboxamide, (compound 106), calculated (C 33 H 48 N 12 O 4 ) MH + : 677.8; Measured value: MH + : 677.4;
4-胍基苄基4-guanidinobenzyl
4-{7-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]庚酰基}-1-哌嗪甲酰胺,(化合物107),计算值(C34H50N12O4)MH+:691.9;测定值:MH+:691.5;4-{7-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]heptanoyl}-1-piperazinecarboxamide, (compound 107), calculated (C 34 H 50 N 12 O 4 ) MH + : 691.9; Measured value: MH + : 691.5;
4-胍基苄基4-guanidinobenzyl
4-{8-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]辛酰基}-1-哌嗪甲酰胺,(化合物108),计算值(C37H57N9O8)MH+:756.9;测定值:MH+:756.4;4-{8-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]octanoyl}-1-piperazinecarboxamide, (compound 108), calculated (C 37 H 57 N 9 O 8 ) MH + : 756.9; Measured value: MH + : 756.4;
4-胍基苄基4-guanidinobenzyl
4-{9-[4-(4-胍基苄基氨基甲酰基)哌嗪-1-基羰基]壬酰基}-1-哌嗪甲酰胺,(化合物109),计算值(C36H54N12O4)MH+:719.9;测定值:MH+:719.5;4-{9-[4-(4-guanidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]nonanoyl}-1-piperazinecarboxamide, (compound 109), calculated (C 36 H 54 N 12 O 4 ) MH + : 719.9; Measured value: MH + : 719.5;
4-脒基苄基4-Amidinobenzyl
4-{7-[4-(4-脒基苄基氨基甲酰基)哌嗪-1-基羰基]庚酰基}-1-哌嗪甲酰胺,(化合物110),计算值(C34H48N10O4)MH+:661.8;测定值:MH+:661.3:4-{7-[4-(4-Amidinobenzylcarbamoyl)piperazin-1-ylcarbonyl]heptanoyl}-1-piperazinecarboxamide, (compound 110), calculated (C 34 H 48 N 10 O 4 ) MH + : 661.8; Measured value: MH + : 661.3:
1,5-五亚甲基二[4-(4-胍基苯基乙酰基)哌嗪-1-基羰基],(化合物111),计算值(C33H46N10O4),MH+:647.8;测定值:MH+:647.3;1,5-Pentamethylenebis[4-(4-guanidinophenylacetyl)piperazin-1-ylcarbonyl], (compound 111), calculated for (C 33 H 46 N 10 O 4 ), MH + : 647.8; measured value: MH + : 647.3;
1,6-六亚甲基二[4-(4-胍基苯基乙酰基)哌嗪-1-基羰基],(化合物112),计算值(C34H48N10O4),MH+:661.8;测定值:MH+:661;1,6-Hexamethylenebis[4-(4-guanidinophenylacetyl)piperazin-1-ylcarbonyl], (compound 112), calculated for (C 34 H 48 N 10 O 4 ), MH + : 661.8; measured value: MH + : 661;
1,7-七亚甲基二[4-(4-胍基苯基乙酰基)哌嗪-1-基羰基],(化合物113),计算值(C35H50N10O4),MH+:675.9;测定值:MH+:675.4;1,7-Heptamethylenebis[4-(4-guanidinophenylacetyl)piperazin-1-ylcarbonyl], (compound 113), calculated for (C 35 H 50 N 10 O 4 ), MH + : 675.9; measured value: MH + : 675.4;
3-氧杂-1,5-五亚甲基二[4-(4-胍基苯基乙酰基)哌嗪-1-基羰基],(化合物114),计算值(C32H44N10O7),MH+:681.8;测定值:MH+:681.4;3-Oxa-1,5-pentamethylenebis[4-(4-guanidinophenylacetyl)piperazin-1-ylcarbonyl], (compound 114), calculated (C 32 H 44 N 10 O 7 ), MH + : 681.8; measured value: MH + : 681.4;
实施例33类胰蛋白酶体外抑制试验Example 33 In vitro inhibition test of tryptase
把溶于Tris缓冲液(组成为:NaCl,100mM;Tris,50mM;2-[N-吗啉]乙烷磺酸,2.5mM,CaCl2,0.5mM;DMSO,10%;甘油,5%;聚氧乙烯山梨醇单月桂酸酯(吐温-20),0.05%;肝素,25ng/ml;pH8.2)中的人体类胰蛋白酶(15μg/ml)和试验化合物(各种浓度)在室温下温育1小时,加入Tosyl-Gly-Pro-Lys-对硝基苯胺,使试验混合物的最终浓度为0.5mM,将底物水解,进行光谱测定5分钟(405nm),使用标准数学模型从酶进展曲线计算表观抑制常数(Ki)。Dissolved in Tris buffer (composed of: NaCl, 100mM; Tris, 50mM; 2-[N-morpholine]ethanesulfonic acid, 2.5mM, CaCl 2 , 0.5mM; DMSO, 10%; glycerol, 5%; Human tryptase (15 μg/ml) and test compounds (various concentrations) in polyoxyethylene sorbitan monolaurate (Tween-20), 0.05%; heparin, 25 ng/ml; pH 8.2) at room temperature After incubation for 1 hour at 90°C, Tosyl-Gly-Pro-Lys-p-nitroaniline was added to give a final concentration of the assay mixture of 0.5 mM, the substrate was hydrolyzed, and spectroscopic measurements were performed for 5 minutes (405 nm), using standard mathematical models from the enzyme Apparent inhibition constants (Ki) were calculated from progression curves.
人体类胰蛋白酶可以从人肺和皮肤组织样品(例如见Smith etal.(1984)J.Biol.Chem.59;11046-11051;Braganza et al.(1991)Biochem30:4997-5007)及人体柱状细胞株提纯,也可以买到(例如,ICNBiomedicals,Irvine California;Athens Research and Technology,AthensGeorgia)。猪肠粘膜肝素和Tosyl-Gly-Pro-Lys-对硝基苯胺可以从Sigma化学公司得到。Human tryptase can be obtained from human lung and skin tissue samples (see, for example, Smith et al. (1984) J. Biol. Chem. 59; 11046-11051; Braganza et al. (1991) Biochem 30: 4997-5007) and human columnar cells Purified strains are also commercially available (eg, ICNBiomedicals, Irvine California; Athens Research and Technology, Athens Georgia). Porcine intestinal mucosal heparin and Tosyl-Gly-Pro-Lys-p-nitroaniline are available from Sigma Chemical Company.
按照本申请所述方法或本领域普通技术人员公知的方法,制备,并且试验了下式Ⅰ化合物对类胰蛋白酶的抑制活性。According to the method described in the present application or the method known to those skilled in the art, the compound of the following formula I was prepared and tested for its inhibitory activity against tryptase.
化合物1,Ki=0.003μM;化合物2,Ki=0.8μM;化合物3,Ki=0.07μM;化合物4,Ki=0.001μM;化合物5,Ki=0.2μM;化合物6,Ki=1μM;化合物7,Ki=0.3μM;化合物8,Ki=4μM;化合物9,Ki=0.4μM;化合物10,Ki=1μM;化合物11,Ki=0.09μM;化合物12,Ki=0.2μM;化合物13,Ki=0.02μM;化合物14,Ki=0.004μM;化合物15,Ki=0.5μM;化合物16,Ki=0.9μM;化合物17,Ki=1μM;化合物18,Ki=0.08μM;化合物19,Ki=1.2μM;化合物20,Ki=3.4μM;化合物21,Ki=0.5μM;化合物22,Ki=0.2μM;化合物23,Ki=4μM;化合物24,Ki=0.3μM;化合物25,Ki=0.002μM;化合物26,Ki=19μM;化合物27,Ki=2μM;化合物28,Ki=4μM;化合物29,Ki=1μM;化合物30,Ki=0.031μM;化合物31,Ki=1μM;化合物32,Ki=2μM;化合物33,Ki=1μM;化合物34,Ki=3μM;化合物35,Ki=0.8μM;化合物36,Ki=0.6μM;化合物37,Ki=0.07μM;化合物38,Ki=0.004μM;化合物39,Ki=0.004μM;化合物40,Ki=4μM;化合物41,Ki=0.7μM;化合物42,Ki=0.02μM;化合物43,Ki=0.4μM;化合物44,Ki=0.02μM;化合物45,Ki=0.08μM;化合物46,Ki=1μM;化合物47,Ki=0.3μM;化合物48,Ki=0.09μM;化合物49,Ki=2μM;化合物50,Ki=0.08μM;化合物51,Ki=1μM;化合物52,Ki=0.04μM;化合物53,Ki=6μM;化合物54,Ki=0.1μM;化合物55,Ki=2μM;化合物56,Ki=10μM;化合物57,Ki=2μM;化合物58,Ki=0.1μM;化合物59,Ki=0.5μM;化合物60,Ki=5μM;化合物61,Ki=41μM;化合物62,Ki=0.2μM;化合物63,Ki=2μM;化合物64,Ki=1μM;化合物65,Ki=0.001μM;化合物66,Ki=0.02μM;化合物67,Ki=3μM;化合物68,Ki=0.04μM;化合物69,Ki=0.5μM;化合物70,Ki=0.05μM;化合物71,Ki=0.8μM;化合物72,Ki=0.1μM;化合物73,Ki=0.002μM;化合物74,Ki=0.04μM;化合物75,Ki=0.01μM;化合物76,Ki=0.1μM;化合物77,Ki=6μM;化合物78,Ki=0.1μM;化合物79,Ki=1μM;化合物84,Ki=0.06μM;化合物85,Ki=0.9μM;化合物86,Ki=0.08μM;化合物87,Ki=0.05μM;化合物88,Ki=0.1μM;化合物89,Ki=0.1μM;化合物90,Ki=1μM;化合物91,Ki=0.1μM;化合物92,Ki=0.1μM;化合物93,Ki=0.02μM;化合物94,Ki=0.007μM;化合物95,Ki=0.02μM;化合物96,Ki=0.02μM;化合物97,Ki=0.0009μM;化合物98,Ki=0.03μM;化合物99,Ki=0.05μM;化合物100,Ki=0.009μM;化合物101,Ki=0.04μM;化合物102,Ki=0.08μM;化合物103,Ki=0.001μM;化合物104,Ki=0.003μM;化合物105,Ki=0.04μM;化合物106,Ki=0.004μM;化合物107,Ki=0.0001μM;化合物108,Ki=0.0005μM;化合物109,Ki=0.0007μM;化合物110,Ki=0.0008μM;化合物111,Ki=0.3μM;化合物112,Ki=0.09μM;化合物113,Ki=0.005μM;化合物114,Ki=0.058μM。Compound 1, K i =0.003 μM; Compound 2, K i =0.8 μM; Compound 3, K i =0.07 μM; Compound 4, K i =0.001 μM; Compound 5, K i =0.2 μM; Compound 6, K i Compound 7, K i =0.3 μM; Compound 8, K i =4 μM; Compound 9, K i =0.4 μM; Compound 10, K i =1 μM; Compound 11, K i =0.09 μM; Compound 12, K i = 0.2 μM; Compound 13, K i =0.02 μM; Compound 14, K i =0.004 μM; Compound 15, K i =0.5 μM; Compound 16, K i =0.9 μM; Compound 17, K i =1 μM ; Compound 18, K i =0.08 μM; Compound 19, K i =1.2 μM; Compound 20, K i =3.4 μM; Compound 21, K i =0.5 μM; Compound 22, K i =0.2 μM; Compound 23, K i = Compound 24, K i =0.3 μM; Compound 25, K i =0.002 μM; Compound 26, K i =19 μM; Compound 27, K i =2 μM; Compound 28, K i =4 μM; Compound 29, K i = 1 μM; Compound 30, K i =0.031 μM; Compound 31, K i =1 μM; Compound 32, K i =2 μM; Compound 33, K i =1 μM; Compound 34, K i =3 μM; Compound 35, K i =0.8 μM; Compound 36, K i =0.6 μM; Compound 37, K i =0.07 μM; Compound 38, K i =0.004 μM; Compound 39, K i =0.004 μM; Compound 40, K i =4 μM; Compound 41, K Compound 42, K i =0.02 μM; Compound 43, K i =0.4 μM; Compound 44, K i =0.02 μM; Compound 45, K i =0.08 μM; Compound 46, K i = 1 μM; Compound 47, K i =0.3 μM; Compound 48, K i =0.09 μM; Compound 49, K i =2 μM; Compound 50, K i =0.08 μM; Compound 51, K i =1 μM; Compound 52, K i =0.04 μM Compound 53, K i =6 μM; Compound 54, K i =0.1 μM; Compound 55, K i =2 μM; Compound 56, K i =10 μM; Compound 57, K i =2 μM; Compound 58, K i =0.1 μM Compound 59, K i =0.5 μM; Compound 60, K i =5 μM; Compound 61, K i =41 μM; Compound 62, K i =0.2 μM; Compound 63, K i =2 μM; Compound 64, K i =1 μM Compound 65, K i =0.001 μM; Compound 66, K i =0.02 μM; Compound 67, K i =3 μM; Compound 68, K i =0.04 μM; Compound 69, K i =0.5 μM; Compound 70, K i Compound 71, K i =0.8 μM; Compound 72, K i =0.1 μM; Compound 73, K i =0.002 μM; Compound 74, K i =0.04 μM; Compound 75, K i =0.01 μM; Compound 76, K i =0.1 μM; Compound 77, K i =6 μM; Compound 78, K i =0.1 μM; Compound 79, K i =1 μM; Compound 84, K i =0.06 μM; Compound 85, K i =0.9 μM Compound 86, K i =0.08 μM; Compound 87, K i =0.05 μM; Compound 88, K i =0.1 μM; Compound 89, K i =0.1 μM; Compound 90, K i =1 μM; Compound 91, K i Compound 92, K i =0.1 μM; Compound 93, K i =0.02 μM; Compound 94, K i =0.007 μM; Compound 95, K i =0.02 μM; Compound 96, K i =0.02 μM; Compound 97, K i =0.0009 μM; Compound 98, K i =0.03 μM; Compound 99, K i =0.05 μM; Compound 100, K i =0.009 μM; Compound 101, K i =0.04 μM; Compound 102, K i = Compound 103, K i =0.001 μM; Compound 104, K i =0.003 μM; Compound 105, K i =0.04 μM; Compound 106, K i =0.004 μM; Compound 107, K i =0.0001 μM; Compound 108 , K i =0.0005 μM; Compound 109, K i =0.0007 μM; Compound 110, K i =0.0008 μM; Compound 111, K i =0.3 μM; Compound 112, K i =0.09 μM; Compound 113, K i =0.005 μM; compound 114, K i =0.058 μM.
实施例33哮喘病体外试验Example 33 In vitro test of asthma
把具有双重反应者特征的变性绵羊(即表现为早期和晚期支气管收缩)用抗原(例如Ascaris suum)激发,在用抗原激发前0.5小时和用抗原激发后4和24小时用试验化合物和赋形剂通气雾剂过吸入方式向上述绵羊给药。在用第一个试验化合物或赋形剂治疗前和其后每隔0.5-1小时通过食管气囊导管监测特定肺抗性(SRL)。Transgenic sheep with double responder characteristics (i.e. exhibiting early and late bronchoconstriction) were challenged with antigen (e.g. Ascaris suum) with test compound and vehicle 0.5 h before antigen challenge and 4 and 24 h after antigen challenge. The above-mentioned sheep were administered by inhalation of aerosol spray. Specific lung resistance ( SRL ) was monitored by esophageal balloon catheter prior to treatment with the first test compound or vehicle and every 0.5-1 hour thereafter.
另外,在抗原激发前1-2天和在抗原激发后24小时用试验化合物和赋形剂给药监测气管的反应性。对于本申请,气管反应性被定义为使SRL增大400%(PC400)所需的氯化氨甲酰胆碱的累计剂量。通过气雾剂吸入方式给药0-30呼吸单位的1%氯化氨甲酰胆碱(1mL PBS中10mg),直到SRL提高400%时得到PC400值。Additionally, tracheal reactivity is monitored with test compound and vehicle administration 1-2 days prior to antigen challenge and 24 hours after antigen challenge. For the present application, airway reactivity was defined as the cumulative dose of carbachol required to increase SRL by 400% (PC 400 ). 0-30 respiratory units of 1% carbachol (10 mg in 1 mL PBS) were administered by aerosol inhalation until a PC 400 value was obtained at a 400% increase in SRL.
用赋形剂给药的绵羊在抗原激发后0-4小时表现出早期支气管收缩,在抗原激发后4到多于8小时后表现出晚期支气管收缩。另外赋形剂治疗的羊表现出对于氯化氨甲酰胆碱的过度反应性(即PC400值时观察到60%降低)。Sheep dosed with vehicle exhibited early bronchoconstriction 0-4 hours after antigen challenge and late bronchoconstriction 4 to more than 8 hours after antigen challenge. Additionally vehicle treated sheep showed hyperresponsiveness to carbachol (ie a 60% decrease in PC400 values was observed).
用类胰蛋白酶抑制剂治疗的绵羊没有表现晚期支气管缩小(即抗原激发后4-8小时SRL还保留在基础水平),另外用类胰蛋白酶抑制剂治疗的绵羊没有表现出对于氯化氨甲酰胆碱的过度反应性。Sheep treated with tryptase inhibitors did not show late bronchoconstriction (ie, SR L remained at basal levels 4-8 hours after antigen challenge), and sheep treated with tryptase inhibitors did not show response to ammonium chloride Acylcholine hyperresponsiveness.
实施例34含式Ⅰ化合物的典型药剂Example 34 Typical Medicaments Containing Compounds of Formula I
口服制剂Oral preparations
式Ⅰ化合物 10-100mgFormula I compound 10-100mg
柠檬酸单水合物 105mgCitric Acid Monohydrate 105mg
氢氧化钠 18mgSodium Hydroxide 18mg
调味剂flavoring agent
水足量加到 100mLAdd enough water to 100mL
静脉注射剂
式Ⅰ化合物 0.1-10mgFormula I compound 0.1-10mg
葡萄糖单水合物足量使达到等渗Sufficient glucose monohydrate to achieve isotonicity
柠檬酸单水合物 1.05mgCitric acid monohydrate 1.05mg
氢氧化钠 0.18mgSodium Hydroxide 0.18mg
注射用水足量加到 1.0mLAdd enough water for injection to 1.0mL
片剂Tablets
式Ⅰ化合物 1%Compound of formula Ⅰ 1%
微晶纤维素 73%Microcrystalline Cellulose 73%
硬脂酸 25%Stearic acid 25%
胶体二氧化硅 1%Colloidal silicon dioxide 1%
Claims (44)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US2313996P | 1996-07-30 | 1996-07-30 | |
| US60/023,139 | 1996-07-30 | ||
| US89577297A | 1997-07-17 | 1997-07-17 | |
| US08/895,772 | 1997-07-17 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1226892A CN1226892A (en) | 1999-08-25 |
| CN1073103C true CN1073103C (en) | 2001-10-17 |
Family
ID=26696778
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN97196877A Expired - Fee Related CN1073103C (en) | 1996-07-30 | 1997-07-30 | Compounds and compositions for treating diseases associated with tryptase activity |
Country Status (17)
| Country | Link |
|---|---|
| EP (1) | EP0934293A1 (en) |
| JP (1) | JP2001509787A (en) |
| KR (1) | KR20000029679A (en) |
| CN (1) | CN1073103C (en) |
| AU (1) | AU733621B2 (en) |
| CA (1) | CA2262542A1 (en) |
| CZ (1) | CZ29799A3 (en) |
| EE (1) | EE9900036A (en) |
| FI (1) | FI990171A7 (en) |
| HU (1) | HUP0003267A3 (en) |
| LV (1) | LV12291B (en) |
| NO (1) | NO990433L (en) |
| NZ (1) | NZ333713A (en) |
| PL (1) | PL331465A1 (en) |
| SI (1) | SI9720047A (en) |
| SK (1) | SK8599A3 (en) |
| WO (1) | WO1998004537A1 (en) |
Families Citing this family (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6221914B1 (en) | 1997-11-10 | 2001-04-24 | Array Biopharma Inc. | Sulfonamide bridging compounds that inhibit tryptase activity |
| ES2233025T3 (en) * | 1998-02-06 | 2005-06-01 | Max-Planck-Gesellschaft Zur Forderung Der Wissenschaften E.V. | AGENT INHIBITORS OF TRIPTASE. |
| US6613769B1 (en) * | 1998-02-06 | 2003-09-02 | Max-Planck-Gesellschaft zur Föderung der Wissenschaften. e.V. | Tryptase inhibitors |
| JP2003524598A (en) * | 1998-09-04 | 2003-08-19 | アルタナ ファルマ アクチエンゲゼルシャフト | New pyranocene |
| EP1208089B1 (en) | 1999-08-10 | 2004-10-13 | ALTANA Pharma AG | Diazocin-dione derivatives and their use as tryptase inhibitors |
| US6960588B1 (en) | 1999-09-14 | 2005-11-01 | Altana Pharma Ag | Tryptase inhibitors |
| AU1413301A (en) * | 1999-11-17 | 2001-05-30 | Sumitomo Pharmaceuticals Company, Limited | Diabetic remedy containing dipiperazine derivative |
| DE19955476A1 (en) * | 1999-11-18 | 2001-05-23 | Boehringer Ingelheim Pharma | Bis-basic compounds as tryptase inhibitors, process for their preparation and their use as medicaments |
| ES2222259T3 (en) * | 1999-12-20 | 2005-02-01 | Altana Pharma Ag | TRIPTASE INHIBITORS. |
| CA2392127A1 (en) | 1999-12-20 | 2001-06-28 | Byk Gulden Lomberg Chemische Fabrik Gmbh | Tryptase inhibitors |
| EP1358182B1 (en) * | 2001-01-31 | 2004-08-11 | ALTANA Pharma AG | Diazocine derivatives and their use as tryptase inhibitors |
| WO2002066430A1 (en) | 2001-02-21 | 2002-08-29 | Altana Pharma Ag | Tryptase inhibitors |
| JP2004518735A (en) | 2001-02-21 | 2004-06-24 | アルタナ ファルマ アクチエンゲゼルシャフト | Tryptase inhibitor |
| EP1370518A2 (en) * | 2001-03-15 | 2003-12-17 | ALTANA Pharma AG | Tryptase-inhibitors |
| WO2002074732A2 (en) * | 2001-03-15 | 2002-09-26 | Altana Pharma Ag | Tryptase-inhibitors |
| US6818787B2 (en) | 2001-06-11 | 2004-11-16 | Xenoport, Inc. | Prodrugs of GABA analogs, compositions and uses thereof |
| US7186855B2 (en) | 2001-06-11 | 2007-03-06 | Xenoport, Inc. | Prodrugs of GABA analogs, compositions and uses thereof |
| US8048917B2 (en) | 2005-04-06 | 2011-11-01 | Xenoport, Inc. | Prodrugs of GABA analogs, compositions and uses thereof |
| WO2002100347A2 (en) | 2001-06-11 | 2002-12-19 | Xenoport, Inc. | Prodrugs of gaba analogs, compositions and uses thereof |
| US7232924B2 (en) | 2001-06-11 | 2007-06-19 | Xenoport, Inc. | Methods for synthesis of acyloxyalkyl derivatives of GABA analogs |
| US6962941B2 (en) | 2001-06-19 | 2005-11-08 | Altana Pharma Ag | Tryptase inhibitors |
| EP1811986B1 (en) | 2004-11-04 | 2014-03-26 | XenoPort, Inc. | Gabapentin prodrug sustained release oral dosage forms |
| MX2011006353A (en) | 2008-12-19 | 2011-07-13 | Merz Pharma Gmbh & Co Kgaa | 1-amino-alkylcyclohexane derivatives for the treatment of mast cell mediated diseases. |
| TWI432188B (en) | 2008-12-19 | 2014-04-01 | Merz Pharma Gmbh & Co Kgaa | 1-amino-alkylcyclohexane derivatives for the treatment of inflammatory skin diseases |
| FR3038605B1 (en) | 2015-07-06 | 2018-08-24 | Universite Amiens Picardie Jules Verne | VICINAL PRIMARY DIAMINS ASSOCIATED WITH CHELATING MOTIFS OF METALS AND / OR FREE RADICALS, ACTIVE AGAINST CARBONYL AND OXIDIZING STRESSES AND THEIR USE |
| WO2018207950A1 (en) * | 2017-05-12 | 2018-11-15 | 横山 茂之 | Class a gpcr-binding compound modifier |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995032945A1 (en) * | 1994-06-01 | 1995-12-07 | Arris Pharmaceutical Corporation | Compositions and methods for treating mast-cell mediated conditions |
| WO1996009297A1 (en) * | 1994-09-23 | 1996-03-28 | Arris Pharmaceutical Corporation | Compositions and methods for treating mast-cell inflammatory condition |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4845242A (en) * | 1987-04-28 | 1989-07-04 | Georgia Tech Research Corporation | Isocoumarins with basic substituents as serine proteases inhibitors, anticoagulants and anti-inflammatory agents |
| US5525623A (en) * | 1993-03-12 | 1996-06-11 | Arris Pharmaceutical Corporation | Compositions and methods for the treatment of immunomediated inflammatory disorders |
| SK282537B6 (en) * | 1993-03-12 | 2002-10-08 | Axys Pharmaceuticals, Inc. | A tryptase inhibitor or a pharmaceutically acceptable salt thereof, a pharmaceutical composition thereof, and its use |
| JPH11503417A (en) * | 1995-03-24 | 1999-03-26 | アリス・ファーマシューティカル・コーポレイション | Reversible protease inhibitor |
| TW438591B (en) * | 1995-06-07 | 2001-06-07 | Arris Pharm Corp | Reversible cysteine protease inhibitors |
-
1997
- 1997-07-30 EP EP97937066A patent/EP0934293A1/en not_active Withdrawn
- 1997-07-30 PL PL97331465A patent/PL331465A1/en unknown
- 1997-07-30 JP JP50913698A patent/JP2001509787A/en active Pending
- 1997-07-30 EE EEP199900036A patent/EE9900036A/en unknown
- 1997-07-30 AU AU39670/97A patent/AU733621B2/en not_active Ceased
- 1997-07-30 CA CA002262542A patent/CA2262542A1/en not_active Abandoned
- 1997-07-30 SK SK85-99A patent/SK8599A3/en unknown
- 1997-07-30 HU HU0003267A patent/HUP0003267A3/en unknown
- 1997-07-30 SI SI9720047A patent/SI9720047A/en unknown
- 1997-07-30 NZ NZ333713A patent/NZ333713A/en unknown
- 1997-07-30 FI FI990171A patent/FI990171A7/en unknown
- 1997-07-30 CN CN97196877A patent/CN1073103C/en not_active Expired - Fee Related
- 1997-07-30 CZ CZ99297A patent/CZ29799A3/en unknown
- 1997-07-30 WO PCT/US1997/013422 patent/WO1998004537A1/en not_active Ceased
- 1997-07-30 KR KR1019997000757A patent/KR20000029679A/en not_active Withdrawn
-
1999
- 1999-01-29 NO NO990433A patent/NO990433L/en not_active Application Discontinuation
- 1999-02-18 LV LVP-99-27A patent/LV12291B/en unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995032945A1 (en) * | 1994-06-01 | 1995-12-07 | Arris Pharmaceutical Corporation | Compositions and methods for treating mast-cell mediated conditions |
| WO1996009297A1 (en) * | 1994-09-23 | 1996-03-28 | Arris Pharmaceutical Corporation | Compositions and methods for treating mast-cell inflammatory condition |
Also Published As
| Publication number | Publication date |
|---|---|
| FI990171L (en) | 1999-03-23 |
| LV12291A (en) | 1999-06-20 |
| CZ29799A3 (en) | 1999-06-16 |
| AU733621B2 (en) | 2001-05-17 |
| FI990171A7 (en) | 1999-03-23 |
| SK8599A3 (en) | 2000-03-13 |
| CA2262542A1 (en) | 1998-02-05 |
| FI990171A0 (en) | 1999-01-29 |
| HUP0003267A3 (en) | 2002-02-28 |
| CN1226892A (en) | 1999-08-25 |
| EE9900036A (en) | 1999-08-16 |
| WO1998004537A1 (en) | 1998-02-05 |
| SI9720047A (en) | 1999-08-31 |
| JP2001509787A (en) | 2001-07-24 |
| NO990433L (en) | 1999-03-25 |
| NO990433D0 (en) | 1999-01-29 |
| KR20000029679A (en) | 2000-05-25 |
| NZ333713A (en) | 2000-12-22 |
| EP0934293A1 (en) | 1999-08-11 |
| PL331465A1 (en) | 1999-07-19 |
| HUP0003267A2 (en) | 2001-06-28 |
| AU3967097A (en) | 1998-02-20 |
| LV12291B (en) | 2000-04-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1073103C (en) | Compounds and compositions for treating diseases associated with tryptase activity | |
| CN1110305C (en) | Naphthyl-substituted benzimidazole derivatives as anticoagulants | |
| CN1243751C (en) | Piperidine compounds used as antiallergic drugs | |
| CN100337626C (en) | Acylated, heteroaryl-condensed cycloalkenylamines and their use as pharmaceuticals | |
| CN1298715C (en) | Non-imidazole compounds as histamine H3 antagonists | |
| CN1207289C (en) | Metalloprotease inhibitors, pharmaceutical compositions containing them and their pharmaceutical use, preparation methods and intermediates thereof | |
| CN1117082C (en) | Substituted imidazoles having anti-cancer and cytokine inhibitory activity | |
| CN1230431C (en) | Arylmethylamine derivatives as tryptase inhibitors | |
| CN1365281A (en) | Metalloprotease inhibitors | |
| CN1658875A (en) | 1-(4-piperidinyl)benzimidazolones as histamine H3 antagonists | |
| CN1642938A (en) | new compound | |
| CN1662524A (en) | Indole derivatives useful as histamine h3 antagonists | |
| CN1211572A (en) | Cyclic amine derivatives-CCR-3 receptor antagonists | |
| CN1684952A (en) | Novel piperidine derivatives for use in the treatment of chemokine mediated disease states | |
| CN1812980A (en) | Piperidinyl- and piperazinyl-alkylcarbamate derivatives, their preparation and therapeutic use | |
| CN1491208A (en) | Acylated 6,7,8,9-tetrahydro-5H-benzocycloheptenylamines and their use as pharmaceuticals | |
| CN1658874A (en) | (1-4-piperidinyl)benzimidazole derivatives useful as histamine H3 antagonists | |
| CN1032438A (en) | Novel substituted N-(1-alkyl-3-hydroxy-4-piperidinyl)benzamides | |
| HK1044337A1 (en) | Amide compounds | |
| CN1642913A (en) | Piperidine or 8-aza-bicyclo[3.2.1]oct-3-yl derivatives useful as modulators of chemokine receptor activity (especially CCR5) | |
| CN1921858A (en) | Piperidinyl alkyl carbamate derivatives, their preparation and their use as FAAH enzyme inhibitors | |
| CN1458922A (en) | Urea derivative and adhesive-molecule inhibitor containing same as ctive ingredient | |
| CN1114591C (en) | Cyclic amide compound | |
| CN1993358A (en) | Thiazole derivatives as chemokine receptor antagonists | |
| CN1173957C (en) | Substituted piperazinones and their therapeutic use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C56 | Change in the name or address of the patentee |
Owner name: AXYS PHARMACEUTICAL CORPORATION Free format text: FORMER NAME OR ADDRESS: ARRIS PHARMACEUTICAL CORPORATION |
|
| CP01 | Change in the name or title of a patent holder |
Patentee after: AXYS Pharmaceuticals, Inc. Patentee before: Arris Pharmaceutical Corp. |
|
| C19 | Lapse of patent right due to non-payment of the annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |