CN108398406B - A biosensor for detecting uracil glycosylase (UDG) and its application - Google Patents
A biosensor for detecting uracil glycosylase (UDG) and its application Download PDFInfo
- Publication number
- CN108398406B CN108398406B CN201810029772.6A CN201810029772A CN108398406B CN 108398406 B CN108398406 B CN 108398406B CN 201810029772 A CN201810029772 A CN 201810029772A CN 108398406 B CN108398406 B CN 108398406B
- Authority
- CN
- China
- Prior art keywords
- hap1
- udg
- hap2
- solution
- biosensor
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N21/00—Investigating or analysing materials by the use of optical means, i.e. using sub-millimetre waves, infrared, visible or ultraviolet light
- G01N21/62—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light
- G01N21/63—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light optically excited
- G01N21/64—Fluorescence; Phosphorescence
Landscapes
- Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Physics & Mathematics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Investigating Or Analysing Materials By The Use Of Chemical Reactions (AREA)
Abstract
本发明提供了一种检测尿嘧啶糖基化酶的生物传感器,包括UDG模板、S‑HAP1杂交链,HAP2‑AgNCs和ExoIII酶,可采用荧光检测测定尿嘧啶糖基化酶的活性,荧光检测的激发波长为560nm,发射波长为625nm,检测波段为575‑750nm。本发明的生物传感器特异性好、灵敏度高,反应条件温和、反应速度快;采用银簇荧光检测,操作简便、检测周期短、易携带;工艺成本低,适用于产业化中价廉的要求;制备方法简单,性能稳定,重复性好,适用于医疗卫生领域对UDG的检测。
The present invention provides a biosensor for detecting uracil glycosylase, including UDG template, S-HAP1 hybrid chain, HAP2-AgNCs and ExoIII enzyme, the activity of uracil glycosylase can be determined by fluorescence detection, and fluorescence detection The excitation wavelength is 560nm, the emission wavelength is 625nm, and the detection band is 575‑750nm. The biosensor of the invention has good specificity, high sensitivity, mild reaction conditions and fast reaction speed; adopts silver cluster fluorescence detection, which is easy to operate, has a short detection period and is easy to carry; the process cost is low, and it is suitable for the requirement of low price in industrialization; The preparation method is simple, the performance is stable and the repeatability is good, and the invention is suitable for the detection of UDG in the medical and health field.
Description
技术领域technical field
本发明属于生物传感器技术领域,涉及一种基于ExoIII辅助的循环放大及银簇荧光强度变化检测尿嘧啶糖基化酶的生物传感器。The invention belongs to the technical field of biosensors, and relates to a biosensor for detecting uracil glycosylase based on ExoIII-assisted cyclic amplification and changes in the fluorescence intensity of silver clusters.
背景技术Background technique
UDG(尿嘧啶糖基化酶)是各种哺乳动物发生碱基错配时首要的DNA切除修复酶,负责尿嘧啶的移除。它主要是通过切断DNA中错误插入的尿嘧啶(U)与糖基之间的N糖苷键,移去U,产生无碱基位点(AP site),然后由AP内切核酸酶(AP endonucleases1, APE1)切断DNA单链,最后再由DNA聚合酶和DNA连接酶识别并修复该断裂位点,从而完成对错配DNA的修复。UDG (uracil glycosylase) is the primary DNA excision repair enzyme responsible for the removal of uracil when base mismatches occur in various mammals. It mainly cuts off the N-glycosidic bond between the wrongly inserted uracil (U) and the sugar group in DNA, removes U, and generates an abasic site (AP site), which is then processed by AP endonucleases (AP endonucleases1). , APE1) cuts the DNA single strand, and finally is recognized and repaired by DNA polymerase and DNA ligase to complete the repair of mismatched DNA.
目前报道的UDG的检测方法包括放射免疫分析法、化学免疫发光分析、化学发光酶免疫分析技术、电化学发光免疫分析技术等,这些方法往往存在仪器昂贵、操作复杂、价格昂贵、灵敏度低、对人体有放射性等问题。因此,目前急需建立一种快速,准确,灵敏且高特异性的检测方法来检测尿嘧啶糖基化酶。The currently reported detection methods for UDG include radioimmunoassay, chemiimmunoluminescence assay, chemiluminescence enzyme immunoassay technology, electrochemiluminescence immunoassay technology, etc. These methods often have the disadvantages of expensive instruments, complicated operations, high price, low sensitivity, and poor sensitivity. The human body has radioactivity and other problems. Therefore, there is an urgent need to establish a rapid, accurate, sensitive and highly specific detection method to detect uracil glycosylase.
发明内容SUMMARY OF THE INVENTION
为了解决以上现有技术中检测尿嘧啶糖基化酶的方法特异性和灵敏度都比较低、成本高、操作复杂的问题,本发明提供了一种特异性和灵敏度高、成本低、检测速度快的基于Exo III辅助的循环放大及银簇荧光强度变化检测尿嘧啶糖基化酶的生物传感器。In order to solve the problems of low specificity and sensitivity, high cost and complicated operation in the method for detecting uracil glycosylase in the prior art, the present invention provides a method with high specificity and sensitivity, low cost and fast detection speed. A biosensor for the detection of uracil glycosylase based on Exo III-assisted cyclic amplification and changes in silver cluster fluorescence intensity.
为实现上述目的,本发明采用如下技术方案。In order to achieve the above objects, the present invention adopts the following technical solutions.
一种检测尿嘧啶糖基化酶(UDG)的生物传感器,包括UDG模板、S-HAP1杂交双链,HAP2-AgNCs(含发卡探针2的银簇)和ExoIII酶(核酸外切酶III);A biosensor for the detection of uracil glycosylase (UDG), including UDG template, S-HAP1 hybrid duplex, HAP2-AgNCs (silver clusters containing hairpin probe 2) and ExoIII enzyme (exonuclease III) ;
所述UDG模板的序列如SEQ No. 1所示;The sequence of the UDG template is shown in SEQ No. 1;
所述S链的序列如SEQ No. 4所示;The sequence of the S chain is shown in SEQ No. 4;
所述HAP1的序列如SEQ No. 2所示;The sequence of the HAP1 is shown in SEQ No. 2;
所述HAP2的序列如SEQ No. 3所示。The sequence of the HAP2 is shown in SEQ No. 3.
所述S-HAP杂交双链采用以下制备方法获得:将缓冲液、S链溶液、HAP1溶液混合均匀,37℃恒温反应2h。The S-HAP hybrid duplex is obtained by the following preparation method: mixing the buffer solution, the S chain solution and the HAP1 solution uniformly, and reacting at a constant temperature of 37° C. for 2 hours.
所述HAP2-AgNCs采用以下制备方法获得:将缓冲液、HAP2的溶液、AgNO3混合后置于4℃下15-30min;然后加入冷NaHBO4溶液,4℃静置4h以上,即得。The HAP2-AgNCs are obtained by the following preparation method: mixing buffer, HAP2 solution, and AgNO3, and placing at 4 °C for 15-30 min; then adding cold NaHBO4 solution and standing at 4 °C for more than 4 hours.
所述HAP2、AgNO3与NaHBO4的摩尔比为1:6:6。The molar ratio of the HAP2, AgNO 3 and NaHBO 4 is 1:6:6.
一种利用上述生物传感器检测UDG的方法,包括以下步骤:A method for detecting UDG using the above-mentioned biosensor, comprising the following steps:
(1)将S链和HAP1杂交成S-HAP1杂交双链;(1) Hybridize S-chain and HAP1 into S-HAP1 hybrid duplex;
(2)将UDG模板、ExoIII、S-HAP1杂交双链、HAP2-AgNCs在缓冲液中混匀,测定荧光强度,然后加入系列浓度UDG标准溶液或待测液,在37℃下反应2h,检测荧光强度;(2) Mix the UDG template, ExoIII, S-HAP1 hybrid double-stranded, and HAP2-AgNCs in the buffer to measure the fluorescence intensity, then add a series of concentrations of UDG standard solution or test solution, react at 37 °C for 2 hours, and detect The fluorescence intensity;
(3)根据系列浓度UDG标准溶液的荧光强度做标准曲线,计算回归方程,根据待测物的荧光强度,计算得所含UDG的含量。(3) Make a standard curve according to the fluorescence intensity of the UDG standard solution with a series of concentrations, calculate the regression equation, and calculate the content of UDG contained according to the fluorescence intensity of the analyte.
所述ExoIII浓度为1-20U/μL,优选为1-10 U/μL;The ExoIII concentration is 1-20 U/μL, preferably 1-10 U/μL;
所述S-HAP1杂交链浓度为0.01-5 μM;The concentration of the S-HAP1 hybrid chain is 0.01-5 μM;
所述UDG模板浓度为50-100 nM。The UDG template concentration was 50-100 nM.
所述荧光检测条件为:激发波长为560nm,发射波长为625nm,检测波段为575-750nm。The fluorescence detection conditions are as follows: the excitation wavelength is 560 nm, the emission wavelength is 625 nm, and the detection band is 575-750 nm.
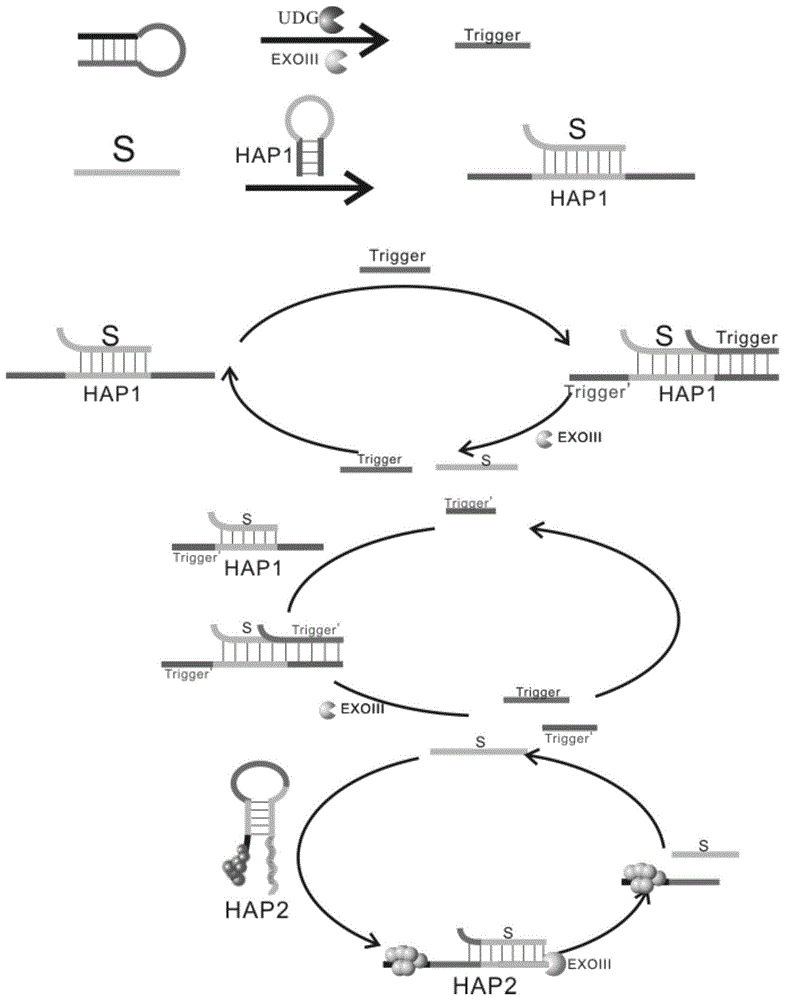
本生物传感器的工作原理如下:The working principle of this biosensor is as follows:
S链与HAP1部分碱基互补配对的,可杂交成双链S-HAP1,UDG模板在目标物UDG和ExoIII存在的条件下在“U”碱基处被切割成2段,产生Trigger序列(5’-GCAAGAGTGACATCATAGAC AAAAA-3’)。此时Trigger的5’端会与事先杂交好的S-HAP1中HAP1的3’端杂交使得HAP1的3’端为平末端,在ExoIII存在的情况下,ExoIII会从3’端切割HAP1链,最终使得S链和Trigger链均从双链体系中释放出来,另外会留下HAP1的5’端部分碱基,该部分中有9个碱基和Trigger链中5’端的9个碱基是相同的,因此该部分相当于是一个次级Trigger,从而实现了Trigger链的循环放大。HAP2的5’端12个碱基为合银簇的序列,3’端包括富含G的序列。当HAP2为发夹结构时,事先将HAP2合成好银簇,由于HAP2的5’端为银簇,5'端银簇和3’端富含G的序列靠近,从而产生强烈的荧光,由于该体系中产生的S链的5’端会与HAP2的3’端结合从而将HAP2打开,S链把HAP2打开后,就会使其荧光强度骤降,同时由于S链和HAP2的互补配对使得HAP2的3’端为平末端,在ExoIII存在的情况下,ExoIII会从发夹2的3’末端富含G的序列开始切割,使得S链重新游离出来,释放到体系中,进行新的和HAP2杂交的反应,实现了信号的放大。通过测定检测加入待测物前后的荧光强度变化,测定所含UDG的含量。The S chain is complementary to the HAP1 part, which can be hybridized into a double-stranded S-HAP1. The UDG template is cut into two segments at the "U" base in the presence of the target UDG and ExoIII, resulting in a Trigger sequence (5 '-GCAAGAGTGACATCATAGAC AAAAA-3'). At this time, the 5' end of the Trigger will hybridize with the 3' end of HAP1 in the pre-hybridized S-HAP1, so that the 3' end of HAP1 is a blunt end. In the presence of ExoIII, ExoIII will cut the HAP1 chain from the 3' end, Finally, both the S chain and the Trigger chain are released from the double-stranded system, and the bases at the 5' end of HAP1 are left, and the 9 bases in this part are the same as the 9 bases at the 5' end of the Trigger chain. , so this part is equivalent to a secondary Trigger, thus realizing the circular amplification of the Trigger chain. The 12 bases at the 5' end of HAP2 are the sequence of the silver cluster, and the 3' end includes the G-rich sequence. When HAP2 is a hairpin structure, HAP2 is synthesized into a silver cluster in advance. Since the 5' end of HAP2 is a silver cluster, the silver cluster at the 5' end and the G-rich sequence at the 3' end are close to each other, resulting in strong fluorescence. The 5' end of the S chain generated in the system will combine with the 3' end of HAP2 to open HAP2. After the S chain opens HAP2, its fluorescence intensity will drop sharply. At the same time, due to the complementary pairing of the S chain and HAP2, HAP2 The 3' end of the hairpin is blunt-ended. In the presence of ExoIII, ExoIII will cut from the G-rich sequence at the 3' end of
本发明具有以下优点:The present invention has the following advantages:
本发明的生物传感器特异性好、灵敏度高,反应条件温和、反应速度快;采用银簇荧光检测,操作简便、检测周期短、易携带;工艺成本低,适用于产业化中价廉的要求;制备方法简单,性能稳定,重复性好,适用于医疗卫生领域对UDG的检测。The biosensor of the invention has good specificity, high sensitivity, mild reaction conditions and fast reaction speed; adopts silver cluster fluorescence detection, which is easy to operate, has a short detection period and is easy to carry; the process cost is low, and it is suitable for the requirement of low price in industrialization; The preparation method is simple, the performance is stable, and the repeatability is good, and the invention is suitable for the detection of UDG in the medical and health field.
附图说明Description of drawings
图1为本生物传感器的工作原理图;Figure 1 is a schematic diagram of the working principle of the biosensor;
图2为荧光强度比随ExoIII浓度的变化图;Fig. 2 is a graph showing the change of fluorescence intensity ratio with ExoIII concentration;
图3为荧光强度随S-HAP1杂交双链浓度的变化;Figure 3 is the change of fluorescence intensity with the concentration of S-HAP1 hybridized duplex;
图4为荧光强度随UDG模板浓度的变化;Fig. 4 is the change of fluorescence intensity with the concentration of UDG template;
图5为荧光强度随UDG浓度的变化;Fig. 5 is the change of fluorescence intensity with the concentration of UDG;
图6为检测UDG的标准曲线。Figure 6 is a standard curve for detecting UDG.
具体实施方式Detailed ways
下面结合实施例和附图对本发明做进一步说明,但本发明不受下述实施例的限制。The present invention will be further described below with reference to the embodiments and the accompanying drawings, but the present invention is not limited by the following embodiments.
实施例1 HAP2-AgNCs的制备。Example 1 Preparation of HAP2-AgNCs.
配置PB缓冲溶液(浓度为20mM),PB缓冲溶液是由磷酸氢二钠与磷酸二氢钠组成,分别称取0.7163g的磷酸氢二钠与0.3120g的磷酸二氢钠,各配成100ml溶液,然后取一部分磷酸氢二钠与一部分磷酸二氢钠混合,将其混合溶液的pH值调至6.5然后备用。Configure PB buffer solution (concentration is 20mM). PB buffer solution is composed of disodium hydrogen phosphate and sodium dihydrogen phosphate. Weigh 0.7163g of disodium hydrogen phosphate and 0.3120g of sodium dihydrogen phosphate, respectively, to make 100ml of solution. , and then mix a part of disodium hydrogen phosphate with a part of sodium dihydrogen phosphate, adjust the pH value of the mixed solution to 6.5 and then use it for later use.
配制AgNO3浓度为2mM,体积为1mL,AgNO3现用现配,避光存放。Prepare AgNO 3 with a concentration of 2mM and a volume of 1mL. AgNO 3 is prepared for immediate use and stored in the dark.
配制NaHBO4浓度为2mM,体积为1mL,NaHBO4现用现配,用0℃冰水配制。The concentration of NaHBO 4 is 2mM and the volume is 1mL. The NaHBO 4 is prepared as it is, and prepared with ice water at 0°C.
取1mL的离心管,加入76μL的PB(20mM),加入15μL HAP2(100μM),加入4.5μL的AgNO3(2mM),震荡1min,放于4℃冰箱30min;再加入4.5μL NaHBO4(2mM)于反应体系中,震荡1min,放在4℃冰箱4h以上,获得HAP2-AgNCs溶液。Take a 1 mL centrifuge tube, add 76 μL PB (20 mM), add 15 μL HAP2 (100 μM), add 4.5 μL AgNO 3 (2 mM), shake for 1 min, and place in a 4°C refrigerator for 30 min; then add 4.5 μL NaHBO 4 (2 mM) In the reaction system, shake for 1 min, and place in a refrigerator at 4 °C for more than 4 h to obtain a HAP2-AgNCs solution.
取30μL制备好的HAP2-AgNCs于离心管中,加入120μL超纯水混匀,用移液枪取150μL溶液于微量比色皿中,使用荧光分析仪对其进行扫描,激发光560nm,测得其发射峰在625nm,说明有溶液中存在HAP2-AgNCs。Take 30 μL of the prepared HAP2-AgNCs in a centrifuge tube, add 120 μL of ultrapure water and mix well, use a pipette to take 150 μL of the solution into a microcuvette, scan it with a fluorescence analyzer, and measure the excitation light at 560 nm. Its emission peak is at 625 nm, indicating that there are HAP2-AgNCs in solution.
实施例2 荧光强度随ExoIII浓度的变化。Example 2 Variation of fluorescence intensity with ExoIII concentration.
将2μL的S链(100μM)和2μL的HAP1(100μM)、2μL的NEBuffer2.1混合均匀,使其在37℃下反应2h,得到S-HAP1杂交双链备用;
将2μL的UDG模板(1μM)、3μL的ExoIII(1U/μL、5U/μL、10U/μL、15U/μL、20U/μL)、4μL的NEBuffer 2.1、2μL的S-HAP1杂交双链(5μM)、8μL HAP2-AgNCs(15μM)以及超纯水(21μL)混匀,测定荧光强度,然后加入1μL的UDG(50U/mL),在37℃下反应2h,检测荧光强度。2 μL of UDG template (1 μM), 3 μL of ExoIII (1U/μL, 5U/μL, 10U/μL, 15U/μL, 20U/μL), 4 μL of NEBuffer 2.1, 2 μL of S-HAP1 hybridization duplex (5 μM) , 8 μL of HAP2-AgNCs (15 μM) and ultrapure water (21 μL) were mixed to measure the fluorescence intensity, then 1 μL of UDG (50 U/mL) was added, and the reaction was carried out at 37 °C for 2 h to detect the fluorescence intensity.
结果如图2所示,其中,“-S”代表的是体系中没有游离的S链时的荧光强度,即未加入UDG时的荧光强度;“+S”代表体系中存在游离的S链时的荧光强度,即加入UDG后的荧光强度,柱形图中数字为加入后荧光值/加入前荧光值。由图可知,检测到的荧光信号强度随着ExoIII的浓度在1-20U/μL区间内逐渐降低,当反应体系中时ExoIII的浓度在10U/μL,荧光强度比最小。The results are shown in Figure 2, where "-S" represents the fluorescence intensity when there is no free S chain in the system, that is, the fluorescence intensity when UDG is not added; "+S" represents when there is free S chain in the system The fluorescence intensity of , that is, the fluorescence intensity after adding UDG, the number in the bar graph is the fluorescence value after adding/the fluorescence value before adding. It can be seen from the figure that the detected fluorescence signal intensity gradually decreased with the concentration of ExoIII in the range of 1-20U/μL. When the concentration of ExoIII in the reaction system was 10U/μL, the fluorescence intensity ratio was the smallest.
实施例3 荧光强度随S-HAP1杂交双链浓度的变化。Example 3 Variation of fluorescence intensity with the concentration of S-HAP1 hybridized duplex.
将2μL的S链(100μM)和2μL的HAP1(100μM)、2μL的NEBuffer2.1混合均匀,使其在37℃下反应2h,得到S-HAP1杂交双链备用;
将2μL的UDG模板(1μM)、3μL的ExoIII(10U/μL)、4μL的NEBuffer 2.1、2μL的S-HAP1杂交双链(0.01μM、0.1μM、0.5μM、1μM、5μM)、8μL HAP2-AgNCs(15μM)以及超纯水(21μL)混匀,测定荧光强度,然后加入1μL的UDG(50U/mL),在37℃下反应2h,检测荧光强度。2 μL of UDG template (1 μM), 3 μL of ExoIII (10 U/μL), 4 μL of NEBuffer 2.1, 2 μL of S-HAP1 hybridization duplex (0.01 μM, 0.1 μM, 0.5 μM, 1 μM, 5 μM), 8 μL of HAP2-AgNCs (15 μM) and ultrapure water (21 μL) and mixed well to measure the fluorescence intensity, then add 1 μL of UDG (50 U/mL), react at 37°C for 2 h, and measure the fluorescence intensity.
结果如图3,检测到的荧光信号强度随着S-HAP1杂交双链的浓度在0.01-5μM 区间内逐渐下降,当反应体系中S-HAP1杂交双链的浓度为5μM时,荧光强度值大。The results are shown in Figure 3. The detected fluorescence signal intensity gradually decreased with the concentration of S-HAP1 hybrid duplex in the range of 0.01-5 μM. When the concentration of S-HAP1 hybrid duplex in the reaction system was 5 μM, the fluorescence intensity value was large. .
实施例4 荧光强度随UDG模板浓度的变化。Example 4 Variation of fluorescence intensity with UDG template concentration.
将2μL的S链(100μM)和2μL的HAP1(100μM)、2μL的NEBuffer2.1混合均匀,使其在37℃下反应2h,得到S-HAP1杂交双链备用;
将2μL的UDG模板(50nM、100nM、500nM、1μM)、3μL的ExoIII(10U/μL)、4μL的NEBuffer 2.1、2μL的S-HAP1杂交双链(5μM)、8μL HAP2-AgNCs(15μM)以及超纯水(21μL)混匀,测定荧光强度,然后加入1μL的UDG(50U/mL),在37℃下反应2h,检测荧光强度。2 μL of UDG template (50 nM, 100 nM, 500 nM, 1 μM), 3 μL of ExoIII (10 U/μL), 4 μL of NEBuffer 2.1, 2 μL of S-HAP1 hybridization duplex (5 μM), 8 μL of HAP2-AgNCs (15 μM) and ultra Pure water (21 μL) was mixed, and the fluorescence intensity was measured, then 1 μL of UDG (50 U/mL) was added, and the reaction was carried out at 37 °C for 2 h to measure the fluorescence intensity.
结果如图4所示,检测到的荧光信号强度随着UDG模板的浓度在50nM-1μM区间内逐渐下降,当反应体系中UDG模板的浓度为1μM,荧光强度值最大。The results are shown in Figure 4. The detected fluorescence signal intensity gradually decreased with the concentration of UDG template in the range of 50nM-1μM. When the concentration of UDG template in the reaction system was 1μM, the fluorescence intensity value was the largest.
实施例5 UDG的检测。Example 5 Detection of UDG.
将2μL的S链(100μM)和2μL的HAP1(100μM)、2μL的NEBuffer2.1混合均匀,使其在37℃下反应2h,得到S-HAP1杂交双链备用;
将2μL的UDG模板(1μM)、3μL的ExoIII(10U/μL)、4μL的NEBuffer 2.1、2μL的S-HAP1杂交双链(5μM)、8μL HAP2-AgNCs(15μM)以及超纯水(21μL)混匀,测定荧光强度,然后加入1μL的UDG(0.0005U/mL、0.005U/mL、0.05U/mL、0.5U/mL、5U/mL、50U/mL)或待测液,在37℃下反应2h,检测荧光强度。
检测结果如图5所示,荧光信号强度随着UDG浓度在0.0005U/ml- 50U/ml区间内逐渐下降;根据系列浓度UDG标准溶液的荧光强度做标准曲线,如图6所示;计算得回归方程为Y= -161.4logC+436.92,相关系数为0.998。The detection results are shown in Figure 5. The fluorescence signal intensity gradually decreases with the concentration of UDG in the range of 0.0005U/ml- 50U/ml; the standard curve is made according to the fluorescence intensity of the UDG standard solution of a series of concentrations, as shown in Figure 6; The regression equation is Y= -161.4logC+436.92, and the correlation coefficient is 0.998.
<110> 济南大学<110> Jinan University
<120> 一种检测尿嘧啶糖基化酶(UDG)的生物传感器及其应用<120> A biosensor for detecting uracil glycosylase (UDG) and its application
<130> 20180112<130> 20180112
<160> 4<160> 4
<170> PatentIn version 3.5<170> PatentIn version 3.5
<210> 1<210> 1
<211> 31<211> 31
<212> DNA<212> DNA
<213> Artificial Sequence<213> Artificial Sequence
<220><220>
<223> UDG T<223> UDG T
<400> 1<400> 1
gtctaugcaa gagtgacatc atagacaaaa a 31gtctaugcaa gagtgacatc atagacaaaa a 31
<210> 2<210> 2
<211> 54<211> 54
<212> DNA<212> DNA
<213> Artificial Sequence<213> Artificial Sequence
<220><220>
<223> HAP1<223> HAP1
<400> 2<400> 2
gcaagagtga tataagtctg aatgagcggg tggggtgggg tggggcactc ttgc 54gcaagagtga tataagtctg aatgagcggg tggggtgggg tggggcactc ttgc 54
<210> 3<210> 3
<211> 53<211> 53
<212> DNA<212> DNA
<213> Artificial Sequence<213> Artificial Sequence
<220><220>
<223> HAP2<223> HAP2
<400> 3<400> 3
cccttaatcc ccgctcatat gcgtactgaa tgagcgggtg gggtggggtg ggg 53cccttaatcc ccgctcatat gcgtactgaa tgagcgggtg gggtggggtg ggg 53
<210> 4<210> 4
<211> 37<211> 37
<212> DNA<212> DNA
<213> Artificial Sequence<213> Artificial Sequence
<220><220>
<223> S<223>S
<400> 4<400> 4
ccccacccca ccccacccgc tcattcagac taaaaaa 37ccccacccca ccccacccgc tcattcagac taaaaaa 37
Claims (6)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810029772.6A CN108398406B (en) | 2018-01-12 | 2018-01-12 | A biosensor for detecting uracil glycosylase (UDG) and its application |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810029772.6A CN108398406B (en) | 2018-01-12 | 2018-01-12 | A biosensor for detecting uracil glycosylase (UDG) and its application |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN108398406A CN108398406A (en) | 2018-08-14 |
| CN108398406B true CN108398406B (en) | 2020-11-24 |
Family
ID=63094729
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201810029772.6A Active CN108398406B (en) | 2018-01-12 | 2018-01-12 | A biosensor for detecting uracil glycosylase (UDG) and its application |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN108398406B (en) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109459423B (en) * | 2018-10-22 | 2021-06-15 | 济南大学 | A kind of biosensor for detecting uracil glycosidase (UDG) activity and preparation method thereof |
| CN109444105B (en) * | 2018-12-28 | 2021-03-30 | 济南大学 | Fluorescent biosensor for detecting DNA glycosylase UDG and preparation method thereof |
| CN109752362B (en) * | 2019-01-10 | 2021-06-15 | 济南大学 | A kind of biosensor for detecting uracil-DNA glycosylase and preparation method thereof |
| CN110734961B (en) * | 2019-11-29 | 2021-10-29 | 福州大学 | An enzyme-free biosensor for detection of uracil-DNA glycosylase activity |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104293927A (en) * | 2014-09-28 | 2015-01-21 | 南京诺唯赞生物科技有限公司 | Uracil-DNA glycosylase activity measurement method |
| CN105506078B (en) * | 2015-12-18 | 2019-07-09 | 山东大学 | One kind is for being measured in parallel uracil-DNA glycosylase and the active method and its application of restriction endonuclease IV and kit |
| CN106929563B (en) * | 2017-02-24 | 2018-10-12 | 山东师范大学 | The two active methods of step series signals amplification detection UDG mediated by enzyme are repaired based on excision |
| CN106995840B (en) * | 2017-03-20 | 2020-05-05 | 山东师范大学 | Method for detecting activity of thymine DNA glycosylase based on double-signal amplification strategy mediated by cyclic enzyme repair |
-
2018
- 2018-01-12 CN CN201810029772.6A patent/CN108398406B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN108398406A (en) | 2018-08-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Chen et al. | A dual-readout chemiluminescent-gold lateral flow test for multiplex and ultrasensitive detection of disease biomarkers in real samples | |
| CN108398406B (en) | A biosensor for detecting uracil glycosylase (UDG) and its application | |
| CN108107028A (en) | A kind of detection atriphos(ATP)Biosensor | |
| CN104374765B (en) | A kind of electrochemiluminescence aptamer sensor, Its Preparation Method And Use | |
| CN114295694B (en) | Electrochemiluminescence aptamer sensor for detecting breast cancer HER-2 and detection method thereof | |
| CN105158320B (en) | Electrochemical sensor based on aptamer detection kanamycins and preparation method thereof | |
| CN104328192B (en) | Ribozyme amplified high-sensitivity electrochemical immunoassay method | |
| CN107024471B (en) | A kind of sulfurated hydrogen detection method based on chemical luminous system | |
| CN108802142B (en) | Electrochemical biosensor based on magnetic biocomposites and lead ion-dependent DNase to detect thrombin | |
| CN111426834A (en) | Biosensor based on dual aptamer for detection of exosomes and its preparation method and application | |
| CN107449759A (en) | A kind of mercury ion efficient detection method and probe molecule and kit | |
| CN108359714A (en) | A kind of biosensor of detection mercury ion | |
| CN103630517B (en) | A thrombin detection method based on split aptamer and water-soluble conjugated polymer | |
| CN110455756A (en) | A method for simultaneously detecting divalent lead ions and divalent copper ions | |
| CN106841335A (en) | A kind of preparation method of antibiotic self energizing aptamer sensor | |
| CN104165999A (en) | Homogeneous chemiluminescence immune assay method based on adjacent position striking effect | |
| CN106770100A (en) | A kind of method that hemoglobin is detected based on graphene quantum dot | |
| CN104897742A (en) | Biosensor used for detecting tetracycline based on nucleic acid aptamer and preparation method thereof | |
| CN116103374B (en) | Fluorescent biosensor for detecting exosomes based on CRISPR-Cas system | |
| CN103969436A (en) | Novel ultra-sensitive detection method of alkaline phosphatase | |
| CN104155289B (en) | A kind of solid-state electrochemistry illumination sensor detecting mercury ion and its preparation method and application | |
| Song et al. | A dual-switch electrochemical aptasensor for label-free detection of thrombin and ATP based on split aptamers | |
| CN104049090B (en) | A kind of test kit detecting alanylamino peptase | |
| CN120214036A (en) | An electrochemiluminescent aptasensor based on CoOOH-cDNA/Apt/AgNWs/Tb@A-COF/GCE working electrode | |
| CN108956991B (en) | Fluorescence resonance energy transfer biosensor and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20250429 Address after: Building 7, 3rd Floor, North of Jinxing Tonghuang Road, Daxing District, Beijing 102600 Patentee after: Beijing Ruidi Information Technology Co.,Ltd. Country or region after: China Address before: 250022 No. 336, South Xin Zhuang West Road, Shizhong District, Ji'nan, Shandong Patentee before: University of Jinan Country or region before: China |
|
| TR01 | Transfer of patent right |