CN110721663A - Modified cellulose particle and preparation method and application thereof - Google Patents
Modified cellulose particle and preparation method and application thereof Download PDFInfo
- Publication number
- CN110721663A CN110721663A CN201911077821.4A CN201911077821A CN110721663A CN 110721663 A CN110721663 A CN 110721663A CN 201911077821 A CN201911077821 A CN 201911077821A CN 110721663 A CN110721663 A CN 110721663A
- Authority
- CN
- China
- Prior art keywords
- modified cellulose
- cellulose particles
- particle
- reaction
- cellulose particle
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000002245 particle Substances 0.000 title claims abstract description 49
- 239000001913 cellulose Substances 0.000 title claims abstract description 48
- 229920002678 cellulose Polymers 0.000 title claims abstract description 48
- 238000002360 preparation method Methods 0.000 title abstract description 7
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 claims abstract description 28
- 238000006243 chemical reaction Methods 0.000 claims abstract description 17
- PFKFTWBEEFSNDU-UHFFFAOYSA-N carbonyldiimidazole Chemical compound C1=CN=CN1C(=O)N1C=CN=C1 PFKFTWBEEFSNDU-UHFFFAOYSA-N 0.000 claims abstract description 14
- 108091006003 carbonylated proteins Proteins 0.000 claims abstract description 10
- NWZSZGALRFJKBT-KNIFDHDWSA-N (2s)-2,6-diaminohexanoic acid;(2s)-2-hydroxybutanedioic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O.NCCCC[C@H](N)C(O)=O NWZSZGALRFJKBT-KNIFDHDWSA-N 0.000 claims abstract description 9
- IKDUDTNKRLTJSI-UHFFFAOYSA-N hydrazine monohydrate Substances O.NN IKDUDTNKRLTJSI-UHFFFAOYSA-N 0.000 claims abstract description 9
- 238000000502 dialysis Methods 0.000 claims abstract description 8
- 230000008081 blood perfusion Effects 0.000 claims abstract description 7
- 238000003756 stirring Methods 0.000 claims abstract description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 6
- 239000008367 deionised water Substances 0.000 claims abstract 2
- 229910021641 deionized water Inorganic materials 0.000 claims abstract 2
- 238000000034 method Methods 0.000 claims description 9
- 238000007865 diluting Methods 0.000 claims 1
- 238000001035 drying Methods 0.000 claims 1
- 230000008014 freezing Effects 0.000 claims 1
- 238000007710 freezing Methods 0.000 claims 1
- 201000010099 disease Diseases 0.000 abstract description 5
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract description 5
- 238000004108 freeze drying Methods 0.000 abstract description 5
- 238000009098 adjuvant therapy Methods 0.000 abstract description 2
- 102000004169 proteins and genes Human genes 0.000 description 15
- 108090000623 proteins and genes Proteins 0.000 description 15
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 12
- 230000006315 carbonylation Effects 0.000 description 7
- 238000005810 carbonylation reaction Methods 0.000 description 7
- OAKJQQAXSVQMHS-UHFFFAOYSA-N hydrazine group Chemical group NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 7
- 230000032683 aging Effects 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- 230000004048 modification Effects 0.000 description 5
- 238000012986 modification Methods 0.000 description 5
- 230000002000 scavenging effect Effects 0.000 description 5
- 125000003172 aldehyde group Chemical group 0.000 description 4
- 125000000468 ketone group Chemical group 0.000 description 4
- 230000035882 stress Effects 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 3
- 229940098773 bovine serum albumin Drugs 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 239000004005 microsphere Substances 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- HGINCPLSRVDWNT-UHFFFAOYSA-N Acrolein Chemical compound C=CC=O HGINCPLSRVDWNT-UHFFFAOYSA-N 0.000 description 2
- 102000015081 Blood Coagulation Factors Human genes 0.000 description 2
- 108010039209 Blood Coagulation Factors Proteins 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 241000700605 Viruses Species 0.000 description 2
- 239000003463 adsorbent Substances 0.000 description 2
- 239000003114 blood coagulation factor Substances 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 229920002521 macromolecule Polymers 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- 108090000765 processed proteins & peptides Proteins 0.000 description 2
- NHJVRSWLHSJWIN-UHFFFAOYSA-N 2,4,6-trinitrobenzenesulfonic acid Chemical compound OS(=O)(=O)C1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O NHJVRSWLHSJWIN-UHFFFAOYSA-N 0.000 description 1
- HWTDMFJYBAURQR-UHFFFAOYSA-N 80-82-0 Chemical compound OS(=O)(=O)C1=CC=CC=C1[N+]([O-])=O HWTDMFJYBAURQR-UHFFFAOYSA-N 0.000 description 1
- 208000024827 Alzheimer disease Diseases 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- 208000006545 Chronic Obstructive Pulmonary Disease Diseases 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 208000018737 Parkinson disease Diseases 0.000 description 1
- 208000001647 Renal Insufficiency Diseases 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000003851 biochemical process Effects 0.000 description 1
- 230000008049 biological aging Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 230000003915 cell function Effects 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 230000001951 hemoperfusion Effects 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 239000003547 immunosorbent Substances 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 238000005184 irreversible process Methods 0.000 description 1
- 201000006370 kidney failure Diseases 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000012567 medical material Substances 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000036285 pathological change Effects 0.000 description 1
- 231100000915 pathological change Toxicity 0.000 description 1
- 230000004796 pathophysiological change Effects 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 239000012460 protein solution Substances 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000009092 tissue dysfunction Effects 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 108700012359 toxins Proteins 0.000 description 1
- 238000000870 ultraviolet spectroscopy Methods 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
Landscapes
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Materials For Medical Uses (AREA)
Abstract
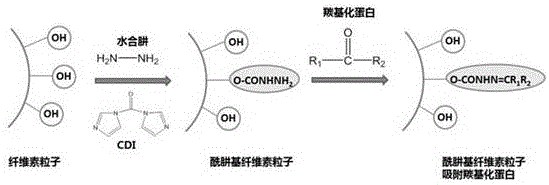
一种改性纤维素粒子,其特征在于,为酰肼基官能化纤维素粒子,制备方法包括如下步骤:将纤维素粒子悬浮于适量二甲基亚砜中,加入纤维素粒子质量0.8~1.5倍量的羰二咪唑,室温搅拌2~4小时后,逐滴加入过量水合肼溶液,继续反应12~24小时。终止反应后,用去离子水稀释反应体系并用透析袋透析2~4日,冷冻干燥后保存。该改性纤维素粒子应用于血液灌流装置,清除羰基化蛋白质,对于衰老性疾病的辅助治疗有较为广阔的应用前景。A modified cellulose particle is characterized in that it is a hydrazide group-functionalized cellulose particle, and the preparation method includes the following steps: suspending the cellulose particle in an appropriate amount of dimethyl sulfoxide, adding 0.8-1.5% of the mass of the cellulose particle Double the amount of carbonyldiimidazole, stir at room temperature for 2-4 hours, add excess hydrazine hydrate solution dropwise, and continue the reaction for 12-24 hours. After the reaction was terminated, the reaction system was diluted with deionized water and dialyzed with a dialysis bag for 2 to 4 days, and then stored after freeze-drying. The modified cellulose particles are used in blood perfusion devices to remove carbonylated proteins, and have broad application prospects for adjuvant therapy of senile diseases.
Description
技术领域technical field
本发明属于医用材料领域,特别是涉及一种改性纤维素粒子。The invention belongs to the field of medical materials, in particular to a modified cellulose particle.
背景技术Background technique
随着老龄化社会的来临,与年龄、衰老相关的疾病发病率日益增加。现有研究表明,活性羰基类物质参与了多种与年龄相关的疾病(如关节炎、肾功能衰竭、心血管疾病、帕金森氏症、阿尔茨海默症、癌症、糖尿病、慢性阻塞性肺病等)和应激的启动以及发展过程,甚至直接参与和启动了衰老过程。因此,羰基应激衰老理论认为羰基应激是生物衰老的核心生化过程之一。羰基应激是指生物体系中活性羰基类物质的产生超过了清除能力,从而导致蛋白质等生物大分子的羰基化修饰,使生物大分子发生结构改变和功能丧失,导致细胞和组织功能紊乱,最终出现机体病理生理改变和加速衰老过程。With the advent of an aging society, the incidence of age- and aging-related diseases is increasing. Existing research shows that active carbonyls are involved in a variety of age-related diseases (eg, arthritis, renal failure, cardiovascular disease, Parkinson's disease, Alzheimer's disease, cancer, diabetes, chronic obstructive pulmonary disease) etc.) and stress initiation and development process, and even directly participate in and initiate the aging process. Therefore, the carbonyl stress aging theory believes that carbonyl stress is one of the core biochemical processes of biological aging. Carbonyl stress refers to the fact that the production of active carbonyl substances in the biological system exceeds the scavenging ability, which leads to the carbonylation modification of biological macromolecules such as proteins, resulting in structural changes and functional loss of biological macromolecules, resulting in cell and tissue dysfunction. There are pathophysiological changes in the body and accelerated aging process.
目前解决这个问题的策略,大多数为清除体内活性羰基类物质或者阻断蛋白质羰基化进程,较少涉及如何解决已经形成的羰基化蛋白。蛋白羰基化是一个不可逆的过程,当羰基化蛋白在体内堆积超过一定量时,将会导致一系列生理、病理改变。At present, most of the strategies to solve this problem are to remove active carbonyl substances in the body or block the protein carbonylation process, and less involve how to solve the already formed carbonylation proteins. Protein carbonylation is an irreversible process. When the carbonylation protein accumulates in the body in excess of a certain amount, it will lead to a series of physiological and pathological changes.
纤维素以其优良的特性与安全性,被广泛用于医药领域,可用于药物分离和纯化、蛋白质、肽、凝血因子、酶、病毒和其他生物活性介质以及用作免疫吸附材料,在血液灌流装置填料中也有应用。血液灌流的主要机制是填料的吸附作用,可直接清除患者血液中的病原体或毒素,调节人体的微环境的稳定性,从而达到缓解症状和治疗疾病目的,效果比较明显。Due to its excellent properties and safety, cellulose is widely used in the field of medicine, and can be used in the separation and purification of drugs, proteins, peptides, coagulation factors, enzymes, viruses and other biologically active media, as well as as immunosorbent materials, in blood perfusion. It is also used in device packing. The main mechanism of hemoperfusion is the adsorption of fillers, which can directly remove pathogens or toxins in the patient's blood and regulate the stability of the human body's microenvironment, thereby achieving the purpose of relieving symptoms and treating diseases, and the effect is relatively obvious.
申请号为201210334217.7的中国专利公开了一种一种血液灌流用多孔纤维素微球吸附剂及其制备方法,所制备的多孔纤维素微球的粒径在0.1~2mm,纤维素微球内部孔尺寸主要分布在5nm~40nm,比表面积为400~1000m2/g,可耐操作压力7~15bar,其制备的特征在于利用反相悬浮体系得到多孔球形微球,再经过交联得到具备可耐操作压力7~15bar的多孔纤维素微球,可用作生物医用材料的载体使用或者用作血液灌流用吸附剂材料,但是该方法较为复杂。Chinese Patent Application No. 201210334217.7 discloses a porous cellulose microsphere adsorbent for blood perfusion and a preparation method thereof. The size is mainly distributed in 5nm~40nm, the specific surface area is 400~1000m2/g, and the operating pressure is 7~15bar. Its preparation is characterized by using a reversed-phase suspension system to obtain porous spherical microspheres. Porous cellulose microspheres with a pressure of 7-15 bar can be used as a carrier for biomedical materials or as an adsorbent material for blood perfusion, but the method is complicated.
发明内容SUMMARY OF THE INVENTION
本发明要解决的技术问题是提供含有一种清除羰基化蛋白质的纤维素粒子及其制备方法。The technical problem to be solved by the present invention is to provide cellulose particles containing a scavenging carbonylation protein and a preparation method thereof.
为解决上述技术问题,本发明的技术方案是:For solving the above-mentioned technical problems, the technical scheme of the present invention is:
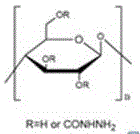
一种改性纤维素粒子,为酰肼基官能化纤维素粒子,其分子示意式为:A modified cellulose particle is a hydrazide group-functionalized cellulose particle, and its molecular formula is:
一种改性纤维素粒子的合成路线为:A synthetic route of modified cellulose particles is:
一种改性纤维素粒子改性以及与羰基化蛋白的作用示意如下:The modification of a modified cellulose particle and the interaction with carbonylated protein are shown as follows:
其制备方法包括如下步骤:Its preparation method comprises the following steps:
取纤维素粒子悬浮于适量二甲基亚砜(DMSO)中,加入纤维素粒子质量的0.8~1.5倍量的羰二咪唑(CDI),室温搅拌2~4小时后,逐滴加入过量水合肼溶液,继续反应12~24小时。终止反应后,用水稀释反应体系并用透析袋透析2~4日,冷冻干燥后保存。Take cellulose particles and suspend them in an appropriate amount of dimethyl sulfoxide (DMSO), add carbonyldiimidazole (CDI) in an amount of 0.8 to 1.5 times the mass of the cellulose particles, stir at room temperature for 2 to 4 hours, and add excess hydrazine hydrate dropwise. solution, and continue to react for 12 to 24 hours. After the reaction was terminated, the reaction system was diluted with water and dialyzed with a dialysis bag for 2 to 4 days, and then stored after freeze-drying.
进一步地,所述水合肼溶液的浓度为78-82%。Further, the concentration of the hydrazine hydrate solution is 78-82%.
优选地,所述透析袋的分子量为2000-4000kD。Preferably, the molecular weight of the dialysis bag is 2000-4000 kD.
进一步地,所述二甲基亚砜的纯度≥99.9%,羰二咪唑的纯度≥97.0%。Further, the purity of the dimethyl sulfoxide is greater than or equal to 99.9%, and the purity of carbonyldiimidazole is greater than or equal to 97.0%.
一种改性纤维素粒子应用于血液灌流装置,清除羰基化蛋白质。A modified cellulose particle used in blood perfusion devices to remove carbonylated proteins.
与现有技术相比,本发明具有以下有益效果:Compared with the prior art, the present invention has the following beneficial effects:
1、本发明利用水合肼与纤维素粒子上的羟基反应,将酰肼基接到纤维素粒子上,得到酰肼基官能化的纤维素粒子,粒子直径为50~70μm,圆形,表面粗糙,有均匀孔径,340℃左右分解,产物性能较未官能化的纤维素粒子相比,酰肼基官能化纤维素粒子的粒径以及形状均没有明显变化,热稳定性亦没有明显变化。1. The present invention utilizes hydrazine hydrate to react with hydroxyl groups on cellulose particles to connect hydrazide groups to cellulose particles to obtain hydrazide group-functionalized cellulose particles, with a particle diameter of 50-70 μm, round, and rough surface. , has a uniform pore size, and decomposes at about 340 °C. Compared with the unfunctionalized cellulose particles, the product properties of the hydrazide group-functionalized cellulose particles have no obvious changes in particle size and shape, and no significant changes in thermal stability.
2、纤维素粒子的优良特性得以保留,基于酰肼基可以结合醛基或酮基,而羰基化蛋白质上有醛基或酮基,并且增加可吸附羰基化蛋白的功能。2. The excellent properties of cellulose particles are retained. Based on the hydrazide group, the aldehyde group or ketone group can be combined, and the carbonylated protein has an aldehyde group or a ketone group, and the function of adsorbing the carbonylated protein is increased.
3、传统纤维素粒子主要用于分离纯化蛋白质、肽、凝血因子、酶、病毒等物质,本发明在传统的纤维素粒子引入酰肼基,获得酰肼基纤维素粒子,并证明了其对羰基化蛋白质具有清除作用,对于衰老性疾病的辅助治疗有较为广阔的应用前景。3. Traditional cellulose particles are mainly used to separate and purify proteins, peptides, coagulation factors, enzymes, viruses and other substances. In the present invention, hydrazide groups are introduced into traditional cellulose particles to obtain hydrazide-based cellulose particles. Carbonylated proteins have a scavenging effect and have broad application prospects for adjuvant therapy of aging diseases.
4、该改性纤维素粒子的制备方法简单,粒径可控。4. The preparation method of the modified cellulose particles is simple and the particle size is controllable.
具体实施方式Detailed ways
下面将结合具体实施例来详细说明本发明,在此本发明的示意性实施例以及说明用来解释本发明,但并不作为对本发明的限定。The present invention will be described in detail below with reference to specific embodiments. The exemplary embodiments and descriptions of the present invention are used to explain the present invention, but are not intended to limit the present invention.
实施例1Example 1
将2g 纤维素粒子(CB)悬浮于20ml二甲基亚砜(DMSO)中,加入1.6g羰二咪唑(CDI),室温搅拌2~4小时后,逐滴加入8 ml水合肼溶液,继续反应12小时。终止反应后,用水稀释反应体系并用透析袋(2000-4000kD)透析2~4日。冷冻干燥后保存,得到改性纤维素粒子。Suspend 2g of cellulose particles (CB) in 20ml of dimethyl sulfoxide (DMSO), add 1.6g of carbonyldiimidazole (CDI), stir at room temperature for 2 to 4 hours, add 8 ml of hydrazine hydrate solution dropwise, and continue the reaction 12 hours. After the reaction was terminated, the reaction system was diluted with water and dialyzed with a dialysis bag (2000-4000kD) for 2 to 4 days. After freeze-drying, the modified cellulose particles were obtained.
实施例2Example 2
将4g 纤维素粒子(CB)悬浮于40 ml二甲基亚砜(DMSO)中,加入6g羰二咪唑(CDI),室温搅拌3小时后,逐滴加入15 ml水合肼溶液,继续反应18小时。终止反应后,用水稀释反应体系并用透析袋(2000-4000kD)透析2~4日。冷冻干燥后保存。4g of cellulose particles (CB) were suspended in 40 ml of dimethyl sulfoxide (DMSO), 6g of carbonyldiimidazole (CDI) was added, and after stirring at room temperature for 3 hours, 15 ml of hydrazine hydrate solution was added dropwise, and the reaction was continued for 18 hours. . After the reaction was terminated, the reaction system was diluted with water and dialyzed with a dialysis bag (2000-4000kD) for 2 to 4 days. Store after freeze drying.
实施例3Example 3
将5g 纤维素粒子(CB)悬浮于50 ml二甲基亚砜(DMSO)中,加入5g羰二咪唑(CDI),室温搅拌4小时后,逐滴加入18 ml水合肼溶液,继续反应24小时。终止反应后,用水稀释反应体系并用透析袋(2000-4000kD)透析2~4日。冷冻干燥后保存。Suspend 5g of cellulose particles (CB) in 50 ml of dimethyl sulfoxide (DMSO), add 5g of carbonyldiimidazole (CDI), stir at room temperature for 4 hours, add 18 ml of hydrazine hydrate solution dropwise, and continue the reaction for 24 hours . After the reaction was terminated, the reaction system was diluted with water and dialyzed with a dialysis bag (2000-4000kD) for 2 to 4 days. Store after freeze drying.
以上实施例中二甲基亚砜的纯度≥99.9%,羰二咪唑的纯度≥97.0%。In the above embodiment, the purity of dimethyl sulfoxide is greater than or equal to 99.9%, and the purity of carbonyldiimidazole is greater than or equal to 97.0%.
实验例Experimental example
将实施例1~3中得到的改性纤维素粒子以及未进行改性的纤维素粒子(对照组)进行,The modified cellulose particles obtained in Examples 1 to 3 and the unmodified cellulose particles (control group) were subjected to
取代率是指对纤维素粒子进行肼基改性中肼基的含量。The substitution rate refers to the content of hydrazine groups in the hydrazine group modification of the cellulose particles.
(1)取代率测定:(1) Determination of substitution rate:
通过TNBS(硝基苯磺酸)法测肼基的量,结果见表一,其中取代率是指对纤维素粒子进行肼基改性中肼基的含量。The amount of hydrazine groups was measured by TNBS (nitrobenzenesulfonic acid) method, and the results are shown in Table 1, where the substitution rate refers to the content of hydrazine groups in the hydrazine group modification of cellulose particles.
表一、肼基的含量:
(2)羰基蛋白清除率的测定:(2) Determination of carbonyl protein clearance rate:
将牛血清白蛋白(BSA)与丙烯醛混合过夜,得到羰基化的牛血清白蛋白;将对照组以及
实施例1~3中的纤维素粒子浸入羰基化蛋白溶液24小时,利用紫外-可见光谱检测羰基化蛋
白溶液蛋白质含量,计算纤维素粒子吸附羰基化蛋白质的量;
由表1看出,实施例1~3均能有效地接上肼基,而基于酰肼基可以结合醛基或酮基,而羰基化蛋白质上有醛基或酮基,从而让增加了增加可吸附羰基化蛋白的功能,表2为羰基蛋白清除率的实验,本发明的技术方案能对羰基蛋白具有一定的清除作用,从而,本改性纤维素粒子可以应用于血液灌流装置,清除羰基化蛋白质。It can be seen from Table 1 that Examples 1 to 3 can be effectively connected to hydrazine groups, and based on hydrazide groups, aldehyde groups or ketone groups can be combined, and carbonylation proteins have aldehyde groups or ketone groups, thereby increasing the increase in The function of adsorbing carbonyl protein, Table 2 is the experiment of carbonyl protein scavenging rate, the technical scheme of the present invention can have a certain scavenging effect on carbonyl protein, so the modified cellulose particles can be used in blood perfusion devices to remove carbonyl protein.
上述实施例仅例示性说明本发明的原理及其功效,而非用于限制本发明。任何熟悉此技术的人士皆可在不违背本发明的精神及范畴下,对上述实施例进行修饰或改变。因此,举凡所属技术领域中具有通常知识者在未脱离本发明所揭示的精神与技术思想下所完成的一切等效修饰或改变,仍应由本发明的权利要求所涵盖。The above-mentioned embodiments merely illustrate the principles and effects of the present invention, but are not intended to limit the present invention. Anyone skilled in the art can modify or change the above embodiments without departing from the spirit and scope of the present invention. Therefore, all equivalent modifications or changes made by those with ordinary knowledge in the technical field without departing from the spirit and technical idea disclosed in the present invention should still be covered by the claims of the present invention.
Claims (7)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911077821.4A CN110721663A (en) | 2019-11-06 | 2019-11-06 | Modified cellulose particle and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911077821.4A CN110721663A (en) | 2019-11-06 | 2019-11-06 | Modified cellulose particle and preparation method and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN110721663A true CN110721663A (en) | 2020-01-24 |
Family
ID=69224898
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201911077821.4A Pending CN110721663A (en) | 2019-11-06 | 2019-11-06 | Modified cellulose particle and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110721663A (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008061427A1 (en) * | 2006-11-24 | 2008-05-29 | Bioregen Biomedical (Shanghai) Co., Ltd. | Dihydrazide compounds, preparation and uses thereof |
| US20090269407A1 (en) * | 2008-04-28 | 2009-10-29 | Surmodics, Inc. | Poly-alpha(1-4)glucopyranose-based matrices with hydrazide crosslinking |
| CN103442735A (en) * | 2011-01-31 | 2013-12-11 | 蒂姆·鲍登 | Active elements for alleviating undesired medical conditions in the technical field |
-
2019
- 2019-11-06 CN CN201911077821.4A patent/CN110721663A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008061427A1 (en) * | 2006-11-24 | 2008-05-29 | Bioregen Biomedical (Shanghai) Co., Ltd. | Dihydrazide compounds, preparation and uses thereof |
| US20090269407A1 (en) * | 2008-04-28 | 2009-10-29 | Surmodics, Inc. | Poly-alpha(1-4)glucopyranose-based matrices with hydrazide crosslinking |
| CN103442735A (en) * | 2011-01-31 | 2013-12-11 | 蒂姆·鲍登 | Active elements for alleviating undesired medical conditions in the technical field |
Non-Patent Citations (1)
| Title |
|---|
| VENUKUMAR VEMULA ET AL: "Fluorescence labeling of carbonylated lipids and proteins in cells using coumarin-hydrazide", 《REDOXBIOLOGY》 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112791712B (en) | Adsorbent for removing protein-bound uremic toxin through blood perfusion and preparation method thereof | |
| TWI404539B (en) | Absorbing agent for medical use | |
| JPS6171061A (en) | Specific adsorbing method and apparatus of heparin | |
| CN113509919B (en) | An adsorbent that can be used to remove endotoxin and inflammatory factors in the blood of patients with sepsis and its preparation method | |
| CN108636374B (en) | A kind of dopamine-grafted sulfonated sodium alginate double-crosslinked microsphere and its preparation method and use | |
| CN113426423B (en) | Adsorbent for removing LDL in extracorporeal circulation of blood and its preparation method and perfusion device | |
| CN105126784A (en) | Adsorbent, preparation method thereof, and adsorption device for blood perfusion | |
| CN101716494A (en) | Magnetic compatible microsphere for purifying thrombin and preparation method and application thereof | |
| Liu et al. | Metal–organic framework (UiO-66 and UiO-66-NH 2)-based adsorbents for bilirubin removal used in hemoperfusion | |
| JPS639869B2 (en) | ||
| JP2001316420A (en) | Production method of absorbent for reducing fibrinogen and/or fibrin level, absorbent, and use thereof for manufacturing absorption device | |
| CN110721663A (en) | Modified cellulose particle and preparation method and application thereof | |
| CN102489266A (en) | Medium and method for separating and purifying polyethylene glycol modifier | |
| JP5202305B2 (en) | Albumin solution to remove stabilizer molecules | |
| Chen et al. | Functionalized carbon nanotube-embedded poly (vinyl alcohol) microspheres for efficient removal of tumor necrosis factor-α | |
| CN103301446B (en) | Affinity adsorption material for treating hyperbilirubinemia and preparation method thereof | |
| CN117986664B (en) | Adsorption material for removing endotoxin and preparation method and application thereof | |
| CN1169612C (en) | A kind of blood purification adsorbent based on montmorillonite and preparation method thereof | |
| CN114225919B (en) | Endotoxin adsorbent and preparation method and application thereof | |
| CN116618024A (en) | A kind of blood purification adsorbent and preparation method thereof | |
| JPS5836624B2 (en) | Adsorbent for blood processing | |
| CN101745116B (en) | Preparation method of copper ion chelation adsorption medium for treating Wilson's disease | |
| CN111743976A (en) | Application of a kind of silica aerogel in the preparation of medicine for treating osteoarthritis | |
| CN119771358B (en) | A magnetic composite adsorbent for hemoperfusion, its preparation method and application | |
| Quirion et al. | Reduction of the in vitro hemolytic activity of soybean lecithin liposomes by treatment with a block copolymer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20200124 |
|
| WD01 | Invention patent application deemed withdrawn after publication |