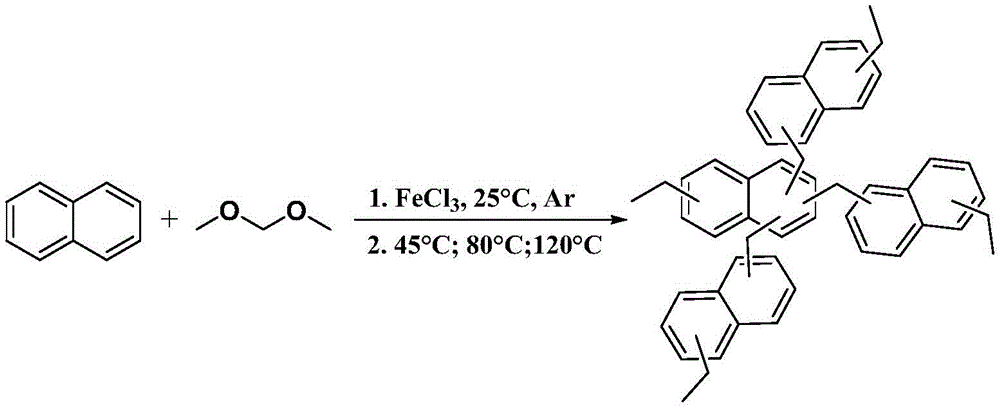
Naphthyl hypercrosslinked microporous polymer and preparation method and application thereof
Technical Field
The invention belongs to the technical field of gas adsorption and separation, and relates to solid CO2Preparation of the adsorbent, in particular to a naphthyl hypercrosslinked microporous polymer and a preparation method and application thereof.
Background
Excess CO emission2The living environment and the life health of human beings are seriously influenced, and the rapid development of effective CO is urgently needed2Trapping and storage technologies are used to address increasingly serious environmental concerns. Adsorption method, in particular using solid CO2Adsorbents are considered to be a very promising treatment technology because of their economic, environmental and low cost advantages. The porous organic polymer is an effective solid CO2The preparation process of the adsorbent is simple and efficient, and noble metals are not involved. Microporous polymers including covalent organic frameworks, conjugated microporous polymers, self-polymerized microporous polymers, and hypercrosslinked microporous polymers have the advantages of high specific surface area, high porosity, high stability, etc. and are widely applied to CO2And (4) trapping and storing. In particular, hypercrosslinked microporous polymers which can be prepared in a simple and efficient manner based on different kinds of organic monomers are of great interest to researchers.
CN107428917A discloses a method for CO2The captured and converted modified porous hypercrosslinked polymer is prepared by combining benzyl halide autopolymerization reaction of friedel-crafts reaction with subsequent reaction and modification, and the porous hypercrosslinked polymer with large specific surface area and stronger CO is displayed2Trapping capacity, but the reaction process uses more catalysts and involves halogen which is difficult to degrade, and environmental protection pressure is met; CN109201007A discloses a carbon dioxide adsorbent, a preparation method and an application thereof, and proposes the preparation of solid CO on biomass charcoal by loading organic amine and ionic liquid2Method of adsorbent, but soaking time is overLong time, ultra-low temperature freeze drying and high cost; CN113603828A discloses a carbon dioxide collector and a preparation method thereof, and proposes the preparation of CO with high selectivity by using N-vinyl pyrrolidone, tetraene silane, acrylamide monomers, alkynol and other medicines2The trapping agent still faces the trouble of higher cost. Therefore, high-performance solid CO can be prepared in a green manner by a low-cost and high-efficiency method2An adsorbent is necessary.
Disclosure of Invention
One of the purposes of the invention is to provide a preparation method of naphthyl hypercrosslinked microporous polymer, which has simple steps and low cost.
Another object of the present invention is to provide a naphthyl hypercrosslinked microporous polymer prepared by the above preparation method, which has a very large specific surface area and high stability, CO2The trapping capacity is stronger.
It is another object of the present invention to provide the above naphthyl hypercrosslinked microporous polymer in CO2The application in adsorption.
In order to achieve the purpose, the technical scheme adopted by the invention is as follows:
in a first aspect, the invention provides a method for preparing naphthyl solid CO by utilizing a hypercrosslinking technology2A method of adsorbing a substance comprising the steps of:
(1) naphthalene and dimethanol formal are mixed according to a molar ratio of 1: 2.9-3.1, fully dissolving in an organic solvent at room temperature, subsequently adding a catalyst into the reaction solution, and fully dispersing under the protection of inert atmosphere;
(2) carrying out the hypercrosslinking reaction in stages: firstly heating to 45-50 ℃ for reaction for 5-6h, then heating to 70-90 ℃ for reaction for 5-6h, and finally heating to 100-120 ℃ for reaction for 20-30 h;
(3) cooling to room temperature after the reaction is finished, filtering, washing the crude product with methanol and hydrochloric acid respectively, and finally washing with water until the filtrate is clear;
(4) and (3) extracting the washed crude product in methanol, and drying in vacuum to finally prepare the naphthyl hypercrosslinked microporous polymer.
Preferably, the catalyst in the step (1) is anhydrous ferric trichloride.
Preferably, the organic solvent in step (1) is nitrobenzene or dichloromethane.
Preferably, the extraction time in step (4) is 20-30 h.
Preferably, the temperature of the vacuum drying in the step (4) is 60-80 ℃, and the drying time is 20-30 h.
In a second aspect, the present invention provides a naphthyl hypercrosslinked microporous polymer prepared by the above preparation method.
The naphthyl hypercrosslinked microporous polymer prepared by the invention has rich microporous structure, average pore diameter less than 2nm and BET specific surface area up to 2870m2The specific surface area of Langmuir reaches 3055m2(ii) a total pore volume of 1.09cm-3The volume of the micro pores reaches 0.58cm-3The catalyst has good thermal stability and is very suitable for adsorbing gas.
In a third aspect, the present invention provides the above naphthyl hypercrosslinked microporous polymer in CO2The application in adsorption.
The naphthyl hypercrosslinked microporous polymer prepared by the invention has CO under the condition of 273K/1bar2The adsorption amount is up to 3.8mmol/g, CO2Strong trapping capacity and can be used as solid CO2An adsorbent.
Compared with the prior art, the invention has the following beneficial effects:
(1) the method provided by the invention has the advantages of simple operation, concise route and high yield, the used reagents are common reagents, the cost is low, and the product does not contain halogen;
(2) the naphthyl hypercrosslinked microporous polymer prepared by the invention has the advantages of great specific surface area, higher thermal stability and CO2Strong trapping capacity and can be used as solid CO2An adsorbent.
Drawings
FIG. 1 is a Fourier transform infrared spectrum of a hypercrosslinked microporous polymer;
FIG. 2 is a view of a hypercrosslinked microporous polymer13C CP/MAS nuclear magnetic resonance spectrogram;
FIG. 3 is a thermogravimetric plot of a hypercrosslinked microporous polymer;
FIG. 4 is a scanning electron micrograph of a hypercrosslinked microporous polymer;
FIG. 5 is N of a hypercrosslinked microporous polymer2Isothermal adsorption-desorption curve;
FIG. 6 is a pore size distribution curve of a hypercrosslinked microporous polymer;
FIG. 7 is CO of a hypercrosslinked microporous polymer2Adsorption profile.
Detailed Description
The invention is described in further detail below with reference to the figures and specific examples.
The raw materials and reagents used in the following examples are commercially available products unless otherwise specified, and the purity thereof was analytical purity or higher.
Example 1
Naphthalene (3.4mmol), dimethanol formal (10.0mmol) were dissolved well in nitrobenzene (50mL) at 25 ℃ followed by FeCl3(10.0mmol) was added to the reaction solution and dispersed well under protection of Ar atmosphere. Carrying out the hypercrosslinking reaction in stages: firstly heating to 45 ℃ for reaction for 5h, then heating to 80 ℃ for reaction for 5h, and finally heating to 120 ℃ for reaction for 24 h. After the reaction is finished and the temperature is cooled to room temperature, crude products obtained by filtration are washed for three times by using methanol and hydrochloric acid respectively, then washed by using deionized water until filtrate is clear, and then the crude products are extracted for 24 hours in the methanol by using a Soxhlet extractor and dried for 24 hours in a vacuum drying oven at the temperature of 80 ℃, so that the naphthyl hypercrosslinked microporous polymer is finally prepared, namely HCLP 1, and the yield is 97.7%.
The reaction equation is as follows:
comparative example 1
Anthracene (3.4mmol), dimethanol formal (10.0mmol) were dissolved well in nitrobenzene (50mL) at 25 ℃ followed by FeCl3(10.0mmol) was added to the reaction solution and dispersed well under protection of Ar atmosphere. Carrying out the hypercrosslinking reaction in stages: firstly heating to 45 ℃ for reaction for 5h, and then heating to 8Reacting at 0 ℃ for 5h, and finally heating to 120 ℃ for reacting for 24 h. After the reaction is finished and the temperature is cooled to room temperature, the crude product obtained by filtration is washed for three times by using methanol and hydrochloric acid respectively, then washed by using deionized water until the filtrate is clear, and then the crude product is extracted for 24 hours in the methanol by using a Soxhlet extractor and dried for 24 hours in a vacuum drying oven at the temperature of 80 ℃, so that the anthracene-based hypercrosslinked microporous polymer is finally prepared, which is named as HCLP 2, and the yield is 93.3%.
Comparative example 2
Pyrene (3.4mmol), dimethanol formal (10.0mmol) were dissolved well in nitrobenzene (50mL) at 25 deg.C, followed by FeCl3(10.0mmol) was added to the reaction solution and dispersed well under protection of Ar atmosphere. Carrying out the hypercrosslinking reaction in stages: firstly heating to 45 ℃ for reaction for 5h, then heating to 80 ℃ for reaction for 5h, and finally heating to 120 ℃ for reaction for 24 h. After the reaction is finished and the temperature is cooled to room temperature, crude products obtained by filtration are washed for three times by using methanol and hydrochloric acid respectively, then washed by using deionized water until filtrate is clear, then a Soxhlet extractor is used for extracting the crude products in the methanol for 24 hours and drying the crude products in a vacuum drying oven at 80 ℃ for 24 hours, and finally pyrenyl hypercrosslinked microporous polymers are prepared, named as HCLP 3, and the yield is 81.1%.
(1) Structural characterization
Molecular Structure of the hypercrosslinked microporous polymers prepared in examples 1-3 by Fourier transform Infrared Spectroscopy and13and C CP/MAS nuclear magnetic resonance spectrum characterization. 1550 and 1590cm in FIG. 1-1The absorption peak in the range is derived from stretching vibration of C ═ C on the aromatic ring, 2900cm-1The left and right peaks are from C-H stretching oscillations at the methylene bridge, indicating that the dimethanol formal successfully weaves the fused ring aromatic. In FIG. 2, the peak at 130ppm corresponds to the unsubstituted aromatic carbon, the peak near 41ppm corresponds to the carbon peak of dimethanol formal, and the solid content13The occurrence of the crosslinking reaction was further confirmed by C CP/MAS NMR.
(2) Performance testing
As shown in FIG. 3, the ultra-crosslinked microporous polymer has only 5% mass loss from 30-200 ℃ by thermogravimetric analysis, and the loss is caused by gas or H adsorbed in the pore channel2O and the like. The second partThe weight loss on heating is in the temperature range of 250-600 ℃, and the weight loss is mainly due to the evaporation and combustion of the volatile compounds obtained by the thermal decomposition of the polymer chains. In general, the thermal weight loss of the hypercrosslinked microporous polymer within 900 ℃ is less than 40 percent in total, which indicates that the adsorbent generally has better thermal stability.
As shown in FIG. 4, it can be seen from SEM that the hypercrosslinked microporous polymers are all formed by stacking randomly aggregated irregular spherical small particles, and have a large amount of pore structures, and the whole body has an amorphous porous morphological structure.
As shown in FIG. 5, it can be seen from the nitrogen desorption curve of BET that the hypercrosslinked microporous polymers are all similar type I N2Isothermal adsorption curve type, i.e. at a relatively low pressure (P/P)0<0.001) with a larger N2The adsorption amount indicates that a large amount of microporous structures exist in the system; n is a radical of2Isothermal adsorption curves at higher relative pressures (P/P)0>0.9) exhibits a steeply rising trend, which indicates the presence of mesopores or macropores in the system, which result from the interparticle porosity or voids. The branch of the desorption curve of the hypercrosslinked microporous polymer shows a significant hysteresis, which is consistent with the results caused by the phenomenon of elastic deformation or swelling due to gas adsorption.
As shown in FIG. 6, it can be seen from the pore size distribution curve that the pore size of the hypercrosslinked microporous polymer is mostly less than 2nm, which is the region where the micropores are located; some pore sizes are even smaller than 0.7nm, which are the regions where the micropores are located.
See table 1 for specific pore structures of the hypercrosslinked microporous polymers. It can be seen from Table 1 that HCLP 1 has the largest BET and Langmuir specific surface areas, up to 2870m, respectively2(ii)/g and 3055m2In terms of a/g and exhibits a maximum total pore volume (1.09 cm)-3In terms of/g) and micropore volume (0.58 cm)-3In terms of/g). The outstanding pore structure characteristics ensure that the HCLP 1 is produced to be applied to CO2The advantage of trapping.
TABLE 1 pore Structure characteristics of hypercrosslinked microporous polymers
a)Specific surface area calculated using BET equation;
b)specific surface area calculated using Langmuir equation;
c)using non-local density functional theory model in P/P0Total pore volume determined at 0.995;
d)using t-Plot model at P/P00.995 determined micropore volume.
(3) Capturing CO2Application of
The ability of the hypercrosslinked microporous polymer to capture CO2 was explored. As shown in FIG. 7, HCLP 1 exhibited the largest CO at 273K/1bar2The adsorption capacity is as high as 3.8mmol/g, which is far superior to HCLP 2(2.7mmol/g) and HCLP 3(1.7mmol/g) prepared by using anthracene and pyrene as raw materials.
In conclusion, the use of the external crosslinking agent dimethanol formal can convert naphthalene into solid CO with high specific surface area and strong thermal stability under simpler conditions2Adsorbent having high CO content2Trapping capacity.