CN1177832C - Novel sulfonamide derivatives as bone resorption inhibitors and as cell adhesion inhibitors - Google Patents
Novel sulfonamide derivatives as bone resorption inhibitors and as cell adhesion inhibitors Download PDFInfo
- Publication number
- CN1177832C CN1177832C CNB998040894A CN99804089A CN1177832C CN 1177832 C CN1177832 C CN 1177832C CN B998040894 A CNB998040894 A CN B998040894A CN 99804089 A CN99804089 A CN 99804089A CN 1177832 C CN1177832 C CN 1177832C
- Authority
- CN
- China
- Prior art keywords
- alkyl
- aryl
- formula
- heteroaryl
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/15—Sulfonamides having sulfur atoms of sulfonamide groups bound to carbon atoms of six-membered aromatic rings
- C07C311/16—Sulfonamides having sulfur atoms of sulfonamide groups bound to carbon atoms of six-membered aromatic rings having the nitrogen atom of at least one of the sulfonamide groups bound to hydrogen atoms or to an acyclic carbon atom
- C07C311/19—Sulfonamides having sulfur atoms of sulfonamide groups bound to carbon atoms of six-membered aromatic rings having the nitrogen atom of at least one of the sulfonamide groups bound to hydrogen atoms or to an acyclic carbon atom to an acyclic carbon atom of a hydrocarbon radical substituted by carboxyl groups
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/18—Sulfonamides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/16—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D215/36—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/06—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D239/08—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with hetero atoms directly attached in position 2
- C07D239/12—Nitrogen atoms not forming part of a nitro radical
- C07D239/16—Nitrogen atoms not forming part of a nitro radical acylated on said nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/26—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D333/30—Hetero atoms other than halogen
- C07D333/34—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Rheumatology (AREA)
- Epidemiology (AREA)
- Physical Education & Sports Medicine (AREA)
- Urology & Nephrology (AREA)
- Cardiology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Heart & Thoracic Surgery (AREA)
- Pain & Pain Management (AREA)
- Ophthalmology & Optometry (AREA)
- Vascular Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pyridine Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
本发明涉及式I磺酰胺衍生物、它们生理学上可耐受的盐和它们的前药,The present invention relates to formula I sulfonamide derivatives, their physiologically tolerable salts and their prodrugs,
其中R1、R2、R4、R5和R6具有以下指明含义。所述式I化合物为有价值的药学活性的化合物。它们是玻连蛋白受体拮抗剂和细胞粘附抑制剂并且抑制通过破骨细胞的骨重吸收。例如它们适宜于治疗和预防至少部分由不合乎需要程度的例如骨质疏性的骨重吸收引起的疾病。本发明还涉及制备所述式I化合物的方法、特别是它们作为药用活性成分的用途和含有它们的药用制剂。wherein R 1 , R 2 , R 4 , R 5 and R 6 have the meanings indicated below. The compounds of formula I are valuable pharmaceutically active compounds. They are vitronectin receptor antagonists and inhibitors of cell adhesion and inhibit bone resorption by osteoclasts. For example, they are suitable for the treatment and prevention of diseases caused at least in part by an undesirable degree of bone resorption, eg osteoporosis. The invention also relates to processes for the preparation of said compounds of formula I, in particular their use as pharmaceutically active ingredients and pharmaceutical preparations containing them.
人骨经受不断的动态的恢复过程,包括骨重吸收和骨形成。这些过程由特别用于这些目的的多种类型细胞所控制。骨重吸收建立在通过破骨细胞的骨基质的破坏的基础上。大多数骨障碍基于骨形成和骨重吸收之间的平衡紊乱。骨质疏松症是以骨质降低和增强的导致增加骨折风险的骨脆性为其特征的疾病。这是由于在所述正在进行重新塑造的过程中,对骨重吸收而言在新骨形成中的不足引起的。常规骨质疏松症治疗包括例如给予双膦酸盐、雌激素、雌激素/孕激素(激素替代疗法或HRT)、雌激素激动剂/拮抗剂(选择性雌激素受体调节剂或SERMs)、降钙素、维生素D类似物、甲状旁腺激素、生长激素促分泌素或氟化钠(Jardine等,Annual Reports in MedicinalChemistry 1996,31,211)。Human bone undergoes a constant dynamic process of restoration, including bone resorption and bone formation. These processes are controlled by various types of cells that are specialized for these purposes. Bone resorption is based on the destruction of the bone matrix by osteoclasts. Most bone disorders are based on a disturbance in the balance between bone formation and bone resorption. Osteoporosis is a disease characterized by decreased bone mass and increased bone fragility leading to increased fracture risk. This is due to a deficit in new bone formation for bone resorption during the ongoing remodeling process. Conventional osteoporosis treatment includes, for example, administration of bisphosphonates, estrogens, estrogen/progestogens (hormone replacement therapy or HRT), estrogen agonists/antagonists (selective estrogen receptor modulators or SERMs), Calcitonin, vitamin D analogues, parathyroid hormone, growth hormone secretagogue, or sodium fluoride (Jardine et al., Annual Reports in Medicinal Chemistry 1996, 31, 211).
活化的破骨细胞为具有直径高达400μm的除去骨基质的多核细胞。活化的破骨细胞附着于所述骨基质表面并分泌蛋白水解酶和酸进入所谓“密封区”,该密封区为它们的细胞膜和骨基质之间的区域。酸性环境和蛋白酶引起骨破坏。式I化合物抑制经过破骨细胞的骨重吸收。Activated osteoclasts are bone matrix depleted multinucleated cells with diameters up to 400 μm. Activated osteoclasts attach to the bone matrix surface and secrete proteolytic enzymes and acids into the so-called "seal zone", which is the area between their cell membrane and the bone matrix. An acidic environment and proteases cause bone destruction. Compounds of formula I inhibit bone resorption by osteoclasts.
多种研究已显示破骨细胞附着于所述骨受到在所述破骨细胞表面的整联蛋白受体的控制。整联蛋白为受体的超家族,其特别包括血小板上的血纤蛋白原受体αIIbβ3和玻连蛋白受体αvβ3。所述玻连蛋白受体αvβ3为膜糖蛋白,其在多种细胞例如内皮细胞、血管平滑肌细胞、破骨细胞和肿瘤细胞的细胞表面表达。在破骨细胞膜上表达的玻连蛋白受体αvβ3控制附着于骨和骨重吸收的过程并因此有助于骨质疏松症。在这种情况下,αvβ3粘合于骨基质蛋白例如骨桥蛋白、骨涎蛋白和血小板反应蛋白(thrombospontin),其含有三肽基序Arg-Gly-Asp(或者RGD)。Various studies have shown that attachment of osteoclasts to the bone is controlled by integrin receptors on the surface of the osteoclasts. Integrins are a superfamily of receptors which include notably the fibrinogen receptor α lib β 3 and the vitronectin receptor α v β 3 on platelets. The vitronectin receptor αvβ3 is a membrane glycoprotein expressed on the cell surface of various cells such as endothelial cells, vascular smooth muscle cells, osteoclasts and tumor cells. The vitronectin receptor αvβ3 expressed on the osteoclast membrane controls the process of attachment to bone and bone resorption and thus contributes to osteoporosis. In this case, αvβ3 binds to bone matrix proteins such as osteopontin, bone sialoprotein and thrombospontin, which contain the tripeptide motif Arg-Gly-Asp (or RGD).
Horton和同事描述RGD肽和抗玻连蛋白受体抗体(23C6),该抗体抑制通过破骨细胞和破骨细胞游走的牙齿破坏(Horton等,Exp.Cell.Res.1991,195,368)。在J.Cell Biol.1990,111,1713中,Sato等描述了锯鳞血抑肽(echistatin)(一种来自蛇毒的RGD肽)作为在组织培养基中有效的骨重吸收抑制剂和作为破骨细胞粘附于骨的抑制剂。Fisher等(Endocrinology 1993,132,1411)能够在大鼠体内显示锯鳞血抑肽也抑制骨重吸收。Horton and co-workers describe RGD peptide and anti-vitronectin receptor antibody (23C6) that inhibit tooth destruction by osteoclasts and osteoclast migration (Horton et al., Exp. Cell. Res. 1991, 195, 368) . In J. Cell Biol. 1990, 111, 1713, Sato et al. describe echistatin (an RGD peptide from snake venom) as a potent inhibitor of bone resorption in tissue Inhibitor of osteocyte adhesion to bone. Fisher et al. (Endocrinology 1993, 132, 1411) were able to show in rats that serastatin also inhibits bone resorption.
这进一步显示人的主动脉血管平滑肌细胞上的玻连蛋白αvβ3刺激这些细胞游走进入新血管内膜,最后导致动脉硬化和血管成形术后再狭窄(Brown等,Cardiovascular Res.1994,28,1815)。This further shows that vitronectin α v β 3 on human aortic vascular smooth muscle cells stimulates the migration of these cells into neovascular intima, eventually leading to atherosclerosis and restenosis after angioplasty (Brown et al., Cardiovascular Res.1994, 28, 1815).
Brooks等(Cell 1994,79,1157)显示针对αvβ3或αvβ3拮抗剂的抗体通过在血管成形术期间介导血管细胞的编程性细胞死亡能够引起肿瘤皱缩。所述玻连蛋白受体αvβ3也参与多种其它类型癌症的发展并且在恶性黑素瘤细胞过度表达(Engleman等,Annual Reports inMedicinal Chemistry 1996,31,191)。所述黑素瘤细胞侵袭与这种过度表达相关(Stracke等,Encylopedia of Cancer,第III卷,1855,AcademicPress,1997;Hillis等,Clinical Science 1996,91,639)。Carron等(CancerRes.1998,58,1930)描述使用αvβ3拮抗剂抑制所述肿瘤生长和抑制恶性肿瘤的血钙过多。Brooks et al. (Cell 1994, 79, 1157 ) showed that antibodies against αvβ3 or αvβ3 antagonists can cause tumor shrinkage by mediating apoptosis of vascular cells during angioplasty. The vitronectin receptor αvβ3 is also involved in the development of various other types of cancer and is overexpressed in malignant melanoma cells (Engleman et al., Annual Reports in Medicinal Chemistry 1996, 31, 191). The melanoma cell invasion is associated with this overexpression (Sracke et al., Encylopedia of Cancer, Vol. III, 1855, Academic Press, 1997; Hillis et al., Clinical Science 1996, 91, 639). Carron et al. (Cancer Res. 1998, 58, 1930 ) describe the use of αvβ3 antagonists to inhibit the growth of such tumors and to inhibit hypercalcemia of malignancies.
Cherest等(Science 1995,270,1500)描述其抑制所述bFGF诱导的大鼠眼中血管成形术过程的抗αvβ3抗体或αvβ3拮抗剂,这是其能够被用来治疗在治疗视网膜病的性质。所述玻连蛋白受体的影响或其中它所涉及的相互作用的影响因此提供影响不同疾病状态的可能性,对此其治疗和预防继续需要适宜的药物活性成分。Cherest et al. (Science 1995, 270, 1500) describe an anti - αvβ3 antibody or an αvβ3 antagonist that inhibits the bFGF-induced angioplasty process in the rat eye, which can be used therapeutically in the treatment of Nature of retinopathy. The influence of the vitronectin receptor or of the interactions in which it is involved thus offers the possibility of influencing different disease states, the treatment and prevention of which continue to require suitable pharmaceutical active ingredients.
WO-A-94/12181描述取代的芳香或非芳香环系统,并且WO-A-94/08577描述作为血纤蛋白原受体拮抗剂和血小板聚集抑制剂的取代杂环。EP-A-528586和EP-A-528587公开了氨基烷基-取代的或杂环基-取代的苯丙氨酸衍生物,并且WO-A-95/32710公开了作为通过破骨细胞的骨重吸收抑制剂的芳基衍生物。WO-A-96/00574描述了苯并二氮杂类并且WO-A-96/00730描述了血纤蛋白原受体拮抗剂模板,尤其是其连接于含氮5-元环的作为玻连蛋白受体拮抗剂的苯并二氮杂类。WO-A-98/00395(DE-A-19654483)描述了衍生于酪氨酸骨架的玻连蛋白受体拮抗剂。EP-A-820991(德国专利申请19629816.4)描述了环烷基衍生物和欧洲专利申请97122520.6描述了其为玻连蛋白受体拮抗剂的氨基甲酸酯衍生物。进一步的研究已显示式I磺酰胺衍生物为特别强的玻连蛋白受体和由破骨细胞的骨重吸收的抑制剂。WO-A-94/12181 describes substituted aromatic or non-aromatic ring systems and WO-A-94/08577 describes substituted heterocycles as fibrinogen receptor antagonists and platelet aggregation inhibitors. EP-A-528586 and EP-A-528587 disclose aminoalkyl-substituted or heterocyclyl-substituted phenylalanine derivatives, and WO-A-95/32710 discloses bone Aryl Derivatives of Reuptake Inhibitors. WO-A-96/00574 describes benzodiazepines and WO-A-96/00730 describes fibrinogen receptor antagonist templates, especially those attached to nitrogen-containing 5-membered rings as glass The benzodiazepine class of zonulin receptor antagonists. WO-A-98/00395 (DE-A-19654483) describes vitronectin receptor antagonists derived from a tyrosine backbone. EP-A-820991 (German patent application 19629816.4) describes cycloalkyl derivatives and European patent application 97122520.6 describes carbamate derivatives which are vitronectin receptor antagonists. Further studies have shown that the sulfonamide derivatives of formula I are particularly potent vitronectin receptors and inhibitors of bone resorption by osteoclasts.
本发明涉及式I化合物,其所有的立体异构体形式和任何比例的混合物形式和它们生理学上可耐受的盐和它们的前药,The present invention relates to compounds of formula I, all stereoisomeric forms thereof and mixtures thereof in any proportion and their physiologically tolerable salts and their prodrugs,
其中in
R1和R2彼此独立为氢或为未取代的或由R3取代的(C1-C6)-烷基,或者其中基团R1-和R2-一起为饱和的或不饱和的二价(C2-C9)-亚烷基,如其中p为2、3、4、5、6、7、8或9的基团-(CH2)p-。其为未取代的或者由一个或多个选自卤素、(C1-C6)-烷基、(C1-C6)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C6)-烷基-、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C6)-烷基-、(C3-C12)-环烷基、(C3-C12)-环烷基-(C1-C6)-烷基-和氧代的基团取代,其中由为未取代的或由R3特别是由一个或两个基团R3取代的5-元至7-元饱和的或未饱和的环并且为碳环或含有一个或两个环氮原子的杂环能够稠合于(C2-C9)-亚烷基中的碳-碳键上;R3为(C1-C10)-烷基、(C3-C20)-单环烷基、(C5-C20)-双环烷基、(C5-C20)-三环烷基、(C1-C8)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C4)-烷基-、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C4)-烷基-、卤素、三氟甲基、氰基、羟基、氧代、硝基、氨基、-NH-(C1-C4)-烷基、-N((C1-C4)-烷基)2、-NH-CO-(C1-C4)-烷基、-CO-(C1-C4)-烷基;R4为氢、(C1-C6)-烷基-CO-O-(C1-C4)-烷基-或者为未取代的或由选自羟基、(C1-C4)-烷氧基、(C1-C4)-烷基-S(O)2-、-NR7R7’和-N+R7R7’R7”Q-的基团取代的(C1-C6)-烷基,其中R7、R7’和R7”彼此独立为氢、(C1-C6)-烷基、(C5-C14)-芳基或(C5-C14)-芳基-(C1-C6)-烷基-并且Q-为生理学上可耐受的阴离子,或者其中R4为下面基团之一R 1 and R 2 are independently of each other hydrogen or unsubstituted or substituted by R 3 (C 1 -C 6 )-alkyl, or wherein the radicals R 1 - and R 2 - together are saturated or unsaturated Divalent (C 2 -C 9 )-alkylene groups, such as the group -(CH 2 ) p - in which p is 2, 3, 4, 5, 6, 7, 8 or 9. It is unsubstituted or consists of one or more selected from halogen, (C 1 -C 6 )-alkyl, (C 1 -C 6 )-alkoxy, (C 6 -C 14 )-aryl, ( C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl-, (C 5 -C 14 )-heteroaryl, (C 5 -C 14 )-heteroaryl-(C 1 - C 6 )-alkyl-, (C 3 -C 12 )-cycloalkyl, (C 3 -C 12 )-cycloalkyl-(C 1 -C 6 )-alkyl- and oxo group substitution , wherein 5-membered to 7-membered saturated or unsaturated ring is unsubstituted or substituted by R 3 especially by one or two groups R 3 and is carbocyclic or contains one or two ring nitrogens A heterocyclic ring of atoms can be fused to a carbon-carbon bond in (C 2 -C 9 )-alkylene; R 3 is (C 1 -C 10 )-alkyl, (C 3 -C 20 )-mono Cycloalkyl, (C 5 -C 20 )-bicycloalkyl, (C 5 -C 20 )-tricycloalkyl, (C 1 -C 8 )-alkoxy, (C 6 -C 14 )-aryl Base, (C 6 -C 14 )-aryl-(C 1 -C 4 )-alkyl-, (C 5 -C 14 )-heteroaryl, (C 5 -C 14 )-heteroaryl-( C 1 -C 4 )-alkyl-, halogen, trifluoromethyl, cyano, hydroxyl, oxo, nitro, amino, -NH-(C 1 -C 4 )-alkyl, -N((C 1 -C 4 )-alkyl) 2 , -NH-CO-(C 1 -C 4 )-alkyl, -CO-(C 1 -C 4 )-alkyl; R 4 is hydrogen, (C 1 - C 6 )-Alkyl-CO-O-(C 1 -C 4 )-Alkyl- is either unsubstituted or composed of hydroxyl, (C 1 -C 4 )-alkoxy, (C 1 -C 4 )-Alkyl (C 1 -C 6 )-alkyl groups substituted by groups of -S(O) 2 -, -NR 7 R 7' and -N + R 7 R 7' R 7” Q- , wherein R 7 , R 7' and R 7" are independently hydrogen, (C 1 -C 6 )-alkyl, (C 5 -C 14 )-aryl or (C 5 -C 14 )-aryl-(C 1 -C 6 )-alkyl- and Q - is a physiologically tolerable anion, or wherein R 4 is one of the following groups
其中所述基团借以连接的键通过虚线指明;wherein the bond by which the groups are attached is indicated by a dashed line;
R5为(C1-C20)-烷基、(C3-C20)-单环烷基、(C5-C20)-双环烷基、(C5-C20)-三环烷基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C6)-烷基-或(C5-C14)-杂芳基-(C1-C6)-烷基-,其中芳基、杂芳基、烷基、单环烷基、双环烷基和三环烷基中的每一个为未取代的或者由一个、两个或三个基团R3取代,并且其中在所述烷基、所述单环烷基、所述双环烷基和所述三环烷基中,一个或多个碳原子,特别是一个、两个、三个或四个碳原子可被相同的或不同的选自氮、氧和硫的原子替代;R 5 is (C 1 -C 20 )-alkyl, (C 3 -C 20 )-monocycloalkyl, (C 5 -C 20 )-bicycloalkyl, (C 5 -C 20 )-tricycloalkane radical, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl- or (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, wherein each of aryl, heteroaryl, alkyl, monocycloalkyl, bicycloalkyl and tricycloalkyl is unsubstituted or substituted by one, two or three groups R 3 , and wherein among said alkyl, said monocycloalkyl, said bicycloalkyl and said tricycloalkyl, one or Multiple carbon atoms, in particular one, two, three or four carbon atoms, may be replaced by identical or different atoms selected from nitrogen, oxygen and sulfur;
R6为氢、(C1-C6)-烷基-O-CO-、羟基、(C1-C6)-烷基-O-CO-O-或硝基。R 6 is hydrogen, (C 1 -C 6 )-alkyl-O-CO-, hydroxy, (C 1 -C 6 )-alkyl-O-CO-O- or nitro.
能够在式I化合物中出现几次的所有基团如基团R3能够彼此独立具有所指明的含义,并且在每种情况下可以是相同的或不同的。在每种情况下,彼此独立具有指明含义的基团可以是相同的或不同的。All radicals which can occur several times in the compounds of the formula I, such as the radical R 3 , can independently of one another have the indicated meanings and can be identical or different in each case. The radicals which independently of one another have the indicated meanings may be identical or different in each case.
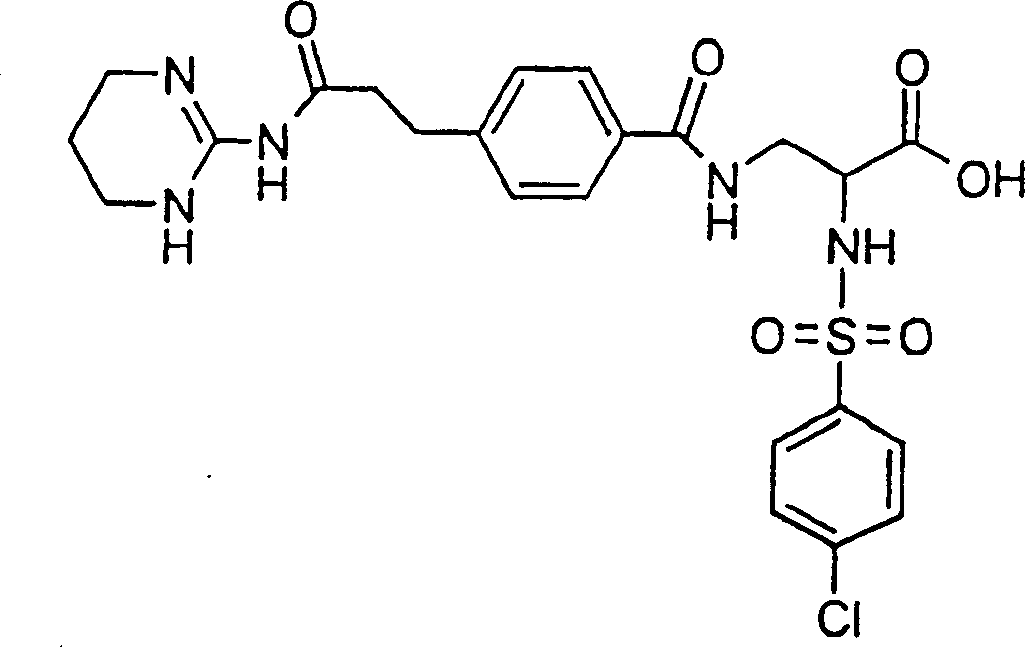
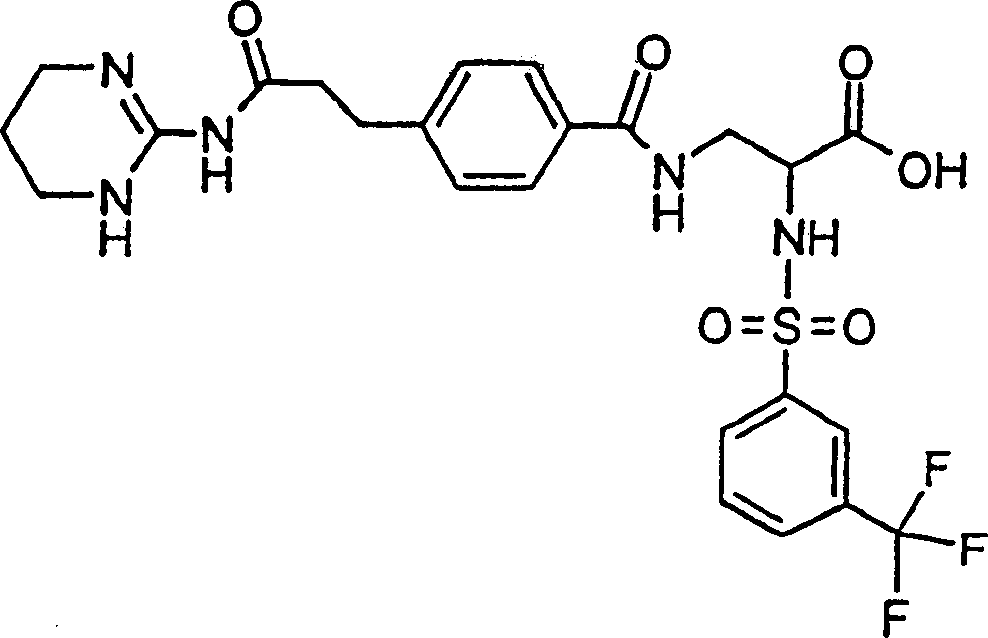
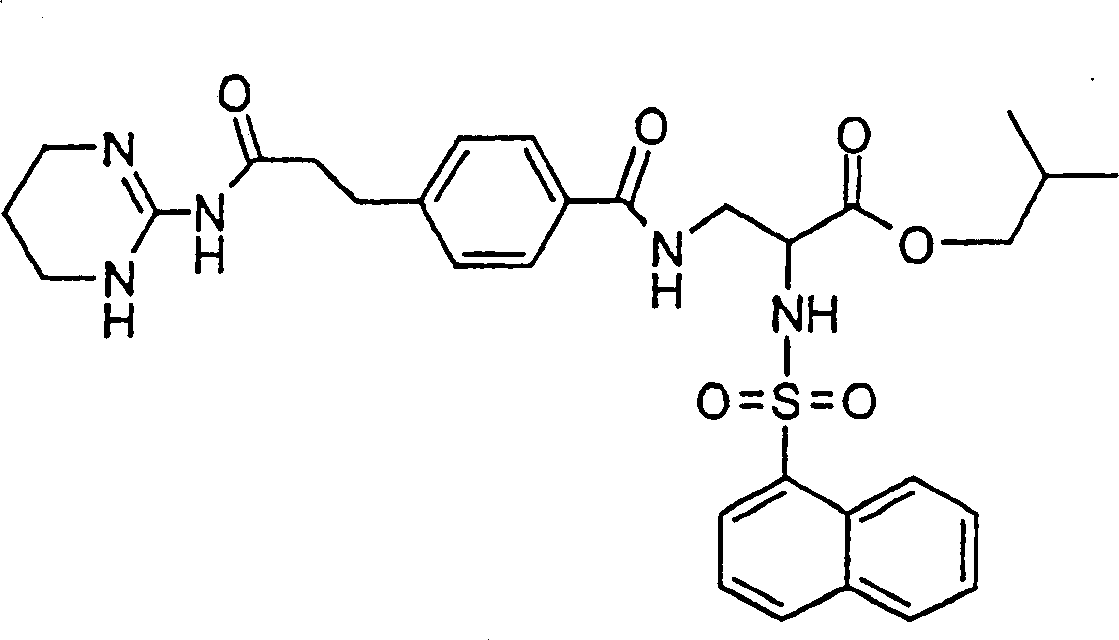
优选本发明的式I化合物为(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)丙酸或它的(C1-C4)-烷基酯和它的生理学上可耐受的盐。Preferably, the compound of formula I of the present invention is (2S)-2-(naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylaminomethyl) Acyl)-ethyl)-benzoylamino)propionic acid or its (C 1 -C 4 )-alkyl esters and its physiologically tolerable salts.
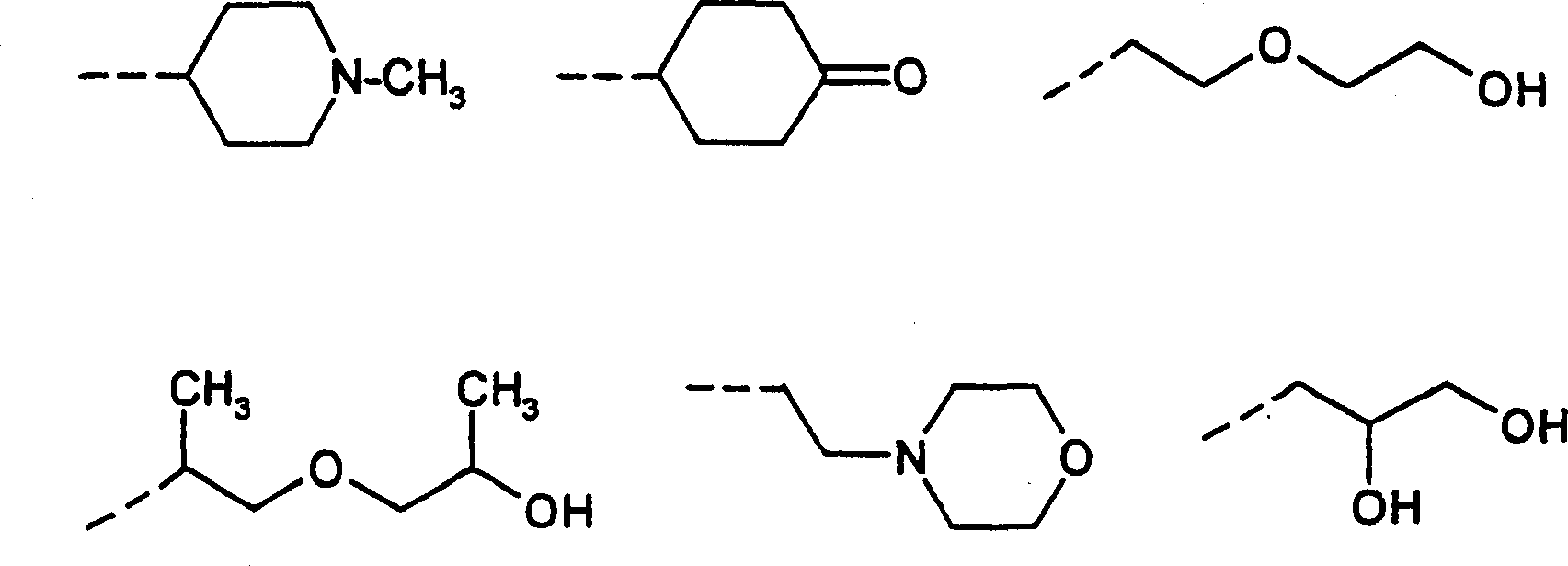
烷基可以是直链的或分枝的并且可以是饱和的或单不饱和的或多不饱和的。如果它们具有取代基或者作为其它基团的取代基出现,这也应用于例如烷氧基、烷氧基羰基或芳基烷基。这同样适用于亚烷基(=二价烷基=饱和的或不饱和的链烷二基)。适宜的含有1至20个碳原子的烷基的实例为甲基、乙基、丙基、丁基、戊基、己基、庚基、辛基、壬基、癸基、十一烷基、十二烷基、十四烷基、十六烷基、十八烷基和二十烷基,所有这些基团的正异构体、异丙基、异丁基、异戊基、新戊基、异己基、异癸基、3-甲基戊基、2,3,4-三甲基己基、仲丁基、叔丁基、叔戊基。一组优选烷基由甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基和叔丁基形成。相应于上述一价基团的二价基团例如亚甲基、1,1-亚乙基(=甲基亚甲基)、1,2-亚乙基、1,3-亚丙基、1,2-亚丙基(=1-甲基亚乙基和2-甲基亚乙基)、2,3-亚丁基(=1,2-二甲基-1,2-亚乙基)、1,4-亚丁基、1,6-亚己基为亚烷基的实例。Alkyl groups may be straight-chain or branched and may be saturated or mono- or polyunsaturated. This also applies, for example, to alkoxy, alkoxycarbonyl or arylalkyl if they have substituents or occur as substituents of other groups. The same applies to alkylene groups (=divalent alkyl groups=saturated or unsaturated alkanediyl groups). Examples of suitable alkyl groups having 1 to 20 carbon atoms are methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, undecyl, deca Dialkyl, tetradecyl, hexadecyl, octadecyl and eicosyl, all normal isomers of these groups, isopropyl, isobutyl, isopentyl, neopentyl, Isohexyl, isodecyl, 3-methylpentyl, 2,3,4-trimethylhexyl, sec-butyl, tert-butyl, tert-amyl. One preferred group of alkyl groups is formed by methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl and tert-butyl. Divalent groups corresponding to the above-mentioned monovalent groups such as methylene, 1,1-ethylene (=methylmethylene), 1,2-ethylene, 1,3-propylene, 1 , 2-propylene (=1-methylethylene and 2-methylethylene), 2,3-butylene (=1,2-dimethyl-1,2-ethylene), 1,4-butylene, 1,6-hexylene are examples of alkylene.
不饱和烷基为例如链烯基如乙烯基、1-丙烯基、烯丙基、丁烯基、3-甲基-2-丁烯基,或者为炔基如乙炔基、1-丙炔基或炔丙基。不饱和亚烷基即亚链烯基和亚炔基(=烯二基和炔二基)能够一样为直链的或分枝的。亚链烯基的实例为1,2-亚乙烯基或亚丙烯基,亚炔基的实例为亚乙炔基或亚丙炔基。当它们被取代时,烷基也能够为不饱和的。在所述烷基部分,不饱和的芳基烷基的实例为苯乙烯基(=2-苯基乙烯基)。Unsaturated alkyl is, for example, alkenyl such as vinyl, 1-propenyl, allyl, butenyl, 3-methyl-2-butenyl, or alkynyl such as ethynyl, 1-propynyl or propargyl. Unsaturated alkylene groups, ie alkenylene and alkynylene groups (=alkenediyl and alkynediyl) can likewise be straight-chain or branched. Examples of alkenylene are 1,2-ethenylene or propenylene, examples of alkynylene are ethynylene or propynylene. Alkyl groups can also be unsaturated when they are substituted. In the alkyl moiety, an example of unsaturated arylalkyl is styryl (=2-phenylethenyl).
除非另外特别指出,环烷基能够为单环、双环或三环,即它们能够为单环烷基、双环烷基和三环烷基,条件是它们具有适宜数目的碳原子。单环烷基为例如环丙基、环丁基、环戊基、环己基、环庚基、环辛基、环壬基、环癸基、环十一烷基、环十二烷基、环十四烷基、环十八烷基,它们也能够被例如(C1-C4)-烷基取代。可以提及的取代的环烷基的实例为4-甲基环己基和2,3-二甲基环戊基。Unless specifically stated otherwise, cycloalkyl groups can be monocyclic, bicyclic or tricyclic, ie they can be monocycloalkyl, bicycloalkyl and tricycloalkyl provided they have the appropriate number of carbon atoms. Monocycloalkyl is, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, cycloundecyl, cyclododecyl, cyclododecyl, cyclo Tetradecyl, cyclooctadecyl, which can also be substituted eg by (C 1 -C 4 )-alkyl. Examples of substituted cycloalkyl groups that may be mentioned are 4-methylcyclohexyl and 2,3-dimethylcyclopentyl.
双环烷基和三环烷基能够为未取代的或在任何所需适宜的位置例如通过一个或多个氧代基团和/或一个或多个相同的或不同的(C1-C4)-烷基例如甲基或异丙基,优选为甲基取代。所述双环或三环基团借以连接的游离键能够定位在分子中的任何要求的位置上;所述基团因此能够借助桥端原子或在桥上的原子来连接。所述游离键也能够定位在任何要求的立体化学位置上,例如在外-位置或内-位置上。双环烷基和三环烷基的实例为莰基(camphanyl)、龙脑基、金刚烷基例如1-金刚烷基和2-金刚烷基、蒈基、表异龙脑基(epiisobornyl)、表龙脑基、降龙脑基(norbornyl)和降蒎基。Bicycloalkyl and tricycloalkyl groups can be unsubstituted or in any desired suitable position, for example via one or more oxo groups and/or one or more identical or different (C 1 -C 4 ) -Alkyl such as methyl or isopropyl, preferably methyl substituted. The free bond by which the bicyclic or tricyclic groups are attached can be positioned at any desired position in the molecule; the groups can thus be attached via a bridge terminal atom or an atom on a bridge. The free bond can also be positioned in any desired stereochemical position, for example in the exo-position or in the endo-position. Examples of bicycloalkyl and tricycloalkyl groups are camphanyl, bornyl, adamantyl such as 1-adamantyl and 2-adamantyl, caronyl, epiisobornyl, epiisobornyl, Borneoyl, norbornyl (norbornyl) and norpineyl.
卤素例如为氟、氯、溴或碘。Halogen is, for example, fluorine, chlorine, bromine or iodine.
(C5-C14)-芳基包括杂环(C5-C14)-芳基(=(C5-C14)-杂芳基),其中所述5至14个环碳原子中的一个或多个由杂原子如氮、氧或硫和碳环(C6-C14)-芳基替代。碳环(C6-C14)-芳基的实例为苯基、萘基、联苯基、蒽基或芴基,其中1-萘基、2-萘基和苯基为优选。如果不另外陈述,芳基特别是苯基可以是未取代的或由一个或多个基团,优选为一个、两个或三个相同的或不同的基团取代。特别是芳基能够由相同的或不同的选自(C1-C8)-烷基(特别是(C1-C4)-烷基)、(C1-C8)-烷氧基(特别是(C1-C4)-烷氧基)、卤素例如氟、氯和溴、硝基、氨基、三氟甲基、羟基、亚甲二氧基、氰基、羟基羰基、氨基羰基、(C1-C4)-烷氧基羰基、苯基、苯氧基、苄基和苄氧基的基团取代。在本发明的式I化合物中一般只有多达两个硝基能够作为取代基发生。(C 5 -C 14 )-aryl includes heterocyclic (C 5 -C 14 )-aryl (=(C 5 -C 14 )-heteroaryl), wherein of the 5 to 14 ring carbon atoms One or more are replaced by heteroatoms such as nitrogen, oxygen or sulfur and carbocyclic (C 6 -C 14 )-aryl groups. Examples of carbocyclic (C 6 -C 14 )-aryl groups are phenyl, naphthyl, biphenyl, anthracenyl or fluorenyl, with 1-naphthyl, 2-naphthyl and phenyl being preferred. If not stated otherwise, aryl groups, especially phenyl groups, may be unsubstituted or substituted by one or more groups, preferably one, two or three identical or different groups. In particular aryl can consist of identical or different groups selected from (C 1 -C 8 )-alkyl (in particular (C 1 -C 4 )-alkyl), (C 1 -C 8 )-alkoxy ( In particular (C 1 -C 4 )-alkoxy), halogens such as fluorine, chlorine and bromine, nitro, amino, trifluoromethyl, hydroxy, methylenedioxy, cyano, hydroxycarbonyl, aminocarbonyl, (C 1 -C 4 )-Alkoxycarbonyl, phenyl, phenoxy, benzyl and benzyloxy radical substitution. In the compounds of the formula I according to the invention generally only up to two nitro groups can occur as substituents.
在单取代的苯基中,所述取代基能够定位在2-位,3-位或4-位上,优选3-和4-位。如果苯基被二取代,取代基能够处于2,3-位,2,4-位、2,5-位、2,6-位、3,4-位或3,5-位。在二取代苯基中,所述两个取代基优选排列在相对于所述连接位置的3,4-位。在三取代苯基中,所述取代基能够处于2,3,4-位、2,3,5-位、2,3,6-位、2,4,5-位、2,4,6-位或3,4,5-位。相似地,萘基和其它芳基能够在任何要求的位置上被取代,例如1-萘基在2-、3-、4-、5-、6-、7-和8-位上,2-萘基在1-、3-、4-、5-、6-、7-和8-位上被取代。In monosubstituted phenyl groups, the substituents can be positioned in the 2-position, 3-position or 4-position, preferably the 3- and 4-position. If the phenyl group is disubstituted, the substituent can be in the 2,3-position, 2,4-position, 2,5-position, 2,6-position, 3,4-position or 3,5-position. In a disubstituted phenyl group, the two substituents are preferably arranged at the 3,4-position relative to the attachment position. In trisubstituted phenyl, the substituents can be at 2,3,4-position, 2,3,5-position, 2,3,6-position, 2,4,5-position, 2,4,6 -bit or 3, 4, 5-bit. Similarly, naphthyl and other aryl groups can be substituted at any desired position, for example 1-naphthyl at the 2-, 3-, 4-, 5-, 6-, 7- and 8-positions, 2- Naphthyl is substituted at the 1-, 3-, 4-, 5-, 6-, 7- and 8-positions.
除了碳环体系以外,(C5-C14)-芳基也能够为单环或多环例如双环或三环,其中1、2、3、4或5个环碳原子由杂原子特别是由相同的或不同的选自氮、氧和硫的杂原子取代的芳香环体系。杂环(C5-C14)-芳基和(C5-C14)-杂芳基的实例为吡啶基像2-吡啶基、3-吡啶基和4-吡啶基、吡咯基像2-吡咯基和3-吡咯基、呋喃基像2-呋喃基和3-呋喃基、噻吩基像2-噻吩基和3-噻吩基、咪唑基、吡唑基、噁唑基、异噁唑基、噻唑基、异噻唑基、四唑基、哒嗪基、吡嗪基、嘧啶基、吲哚基、异吲哚基、吲唑基、2,3-二氮杂萘基、喹啉基、异喹啉基、喹喔啉基、喹唑啉基、噌啉基、β-咔啉基或苯并稠和的、环戊烷稠和的、环己烷稠和的或环庚烷稠和的这些基团的衍生物。所述杂环体系能够在所有适宜的位置由如上提到的碳环芳基体系的相同的取代基取代。In addition to carbocyclic ring systems, (C 5 -C 14 )-aryl can also be monocyclic or polycyclic, for example bicyclic or tricyclic, in which 1, 2, 3, 4 or 5 ring carbon atoms are replaced by heteroatoms, especially by The same or different heteroatom substituted aromatic ring systems selected from nitrogen, oxygen and sulfur. Examples of heterocyclic (C 5 -C 14 )-aryl and (C 5 -C 14 )-heteroaryl groups are pyridyl like 2-pyridyl, 3-pyridyl and 4-pyridyl, pyrrolyl like 2- Pyrrolyl and 3-pyrrolyl, furyl like 2-furyl and 3-furyl, thienyl like 2-thienyl and 3-thienyl, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, Thiazolyl, isothiazolyl, tetrazolyl, pyridazinyl, pyrazinyl, pyrimidinyl, indolyl, isoindolyl, indazolyl, 2,3-diazinyl, quinolinyl, iso Quinolinyl, quinoxalinyl, quinazolinyl, cinnolinyl, β-carbolinyl or benzo-fused, cyclopentane-fused, cyclohexane-fused or cycloheptane-fused Derivatives of these groups. The heterocyclic ring systems can be substituted in all suitable positions by the same substituents as mentioned above for the carbocyclic aryl systems.
在所述系列的这些杂芳基中,优选具有选自N、O和S的1、2或3个杂原子特别是1或2个杂原子的单环或双环芳香环体系,其可以是未取代的或者由1、2或3个选自(C1-C6)-烷基、(C1-C6)-烷氧基、氟、氯、硝基、氨基、三氟甲基、羟基、(C1-C4)-烷氧基羰基、苯基、苯氧基、苄氧基和苄基的取代基取代。在此特别优选为具有选自N、O和S的1至3个杂原子特别为具有1或2个杂原子的单环或双环芳香5-元至10-元环体系,其能够被1至2个选自(C1-C4)-烷基、(C1-C4)-烷氧基、苯基、苯氧基、苄基和苄氧基的取代基取代。更特别优选为含有1或2个特别是1个选自N、O和S的杂原子的5-元或6-元单环杂芳基和9-或10-元双环杂芳基,其为未取代的或如先前描述的那样被取代。Among these heteroaryl groups in the series, preference is given to monocyclic or bicyclic aromatic ring systems having 1, 2 or 3 heteroatoms, in particular 1 or 2 heteroatoms, selected from N, O and S, which may be Substituted or by 1, 2 or 3 selected from (C 1 -C 6 )-alkyl, (C 1 -C 6 )-alkoxy, fluorine, chlorine, nitro, amino, trifluoromethyl, hydroxyl , (C 1 -C 4 )-alkoxycarbonyl, phenyl, phenoxy, benzyloxy and benzyl substituents. Particular preference is given here to monocyclic or bicyclic aromatic 5- to 10-membered ring systems having 1 to 3 heteroatoms selected from N, O and S, in particular 1 or 2 heteroatoms, which can be replaced by 1 to Substitution with 2 substituents selected from (C 1 -C 4 )-alkyl, (C 1 -C 4 )-alkoxy, phenyl, phenoxy, benzyl and benzyloxy. More particularly preferred are 5- or 6-membered monocyclic heteroaryls and 9- or 10-membered bicyclic heteroaryls containing 1 or 2, in particular 1, heteroatom selected from N, O and S, which are Unsubstituted or substituted as previously described.
如果所述两个基团R1-和R2-一起表示二价饱和的或未饱和的(C2-C9)-亚烷基时,这两个基团与它们连接的两个氮原子和这两个氮原子连接的胍基的所述中心碳原子一起形成单环的1,3-二氮杂杂环,所述杂环在基团(CH2)2-CO-NH的2-位连接于所述氮原子。可如同所指明的那样在(C2-C9)-亚烷基上和也在胍基的氮原子上被取代的所述1,3-二氮杂杂环基团的实例为2-咪唑基、4,5-二氢-2-咪唑基、1,4,5,6-四氢-2-嘧啶基或所述4,5,6,7-四氢-1H-1,3-二氮杂-2-基。如果5-元至7-元环稠和于(C2-C9)-亚烷基上的碳-碳键时,那么两个基团R1和R2与它们连接的两个氮原子和这两个氮原子连接的胍基的述中心碳原子一起形成双环杂环,其连接于基团(CH2)2-CO-NH上的氮原子上并且其能够如同所指明的那样被取代。所述稠和(或缩合)的5-元至7-元环可为饱和的、单不饱和的或双不饱和的或芳香的。因此,例如,环戊烷环、环己烷环、环己烯环、环己二烯环、环庚烷环或苯环能够被缩合。能够连接于基团(CH2)2-CO-NH的氮原子上的这样的双环杂环的基团的实例为1,3a,4,5,6,6a-六氢-1,3-二氮杂戊烯-2-基、1H-苯并咪唑-2-基、3a,4,5,6,7,7a-六氢-1H-苯并咪唑-2-基、4,5,6,7-四氢-1H-苯并咪唑-2-基、4,7-二氢-1H-苯并咪唑-2-基或1H-咪唑并[4,5-b]吡啶-2-基。如果缩合的环被取代和/或如果(C2-C9)-亚烷基被取代,它们优选彼此独立由相同或不同的基团R3单取代或双取代。如果表示R1和/或R2的烷基被取代,它们优选彼此独立由相同或不同的基团R3单取代或双取代,特别为单取代。If the two groups R 1 - and R 2 - together represent a divalent saturated or unsaturated (C 2 -C 9 )-alkylene group, these two groups and the two nitrogen atoms to which they are attached The central carbon atom of the guanidine group connected to these two nitrogen atoms forms a monocyclic 1,3-diazaheterocyclic ring, and the heterocyclic ring is in the 2- position is attached to the nitrogen atom. An example of said 1,3-diazaheterocyclic group which may be substituted as indicated on the (C 2 -C 9 )-alkylene group and also on the nitrogen atom of the guanidino group is 2-imidazole base, 4,5-dihydro-2-imidazolyl, 1,4,5,6-tetrahydro-2-pyrimidinyl or the 4,5,6,7-tetrahydro-1H-1,3-di Azepine-2-yl. If the 5- to 7-membered ring is fused to a carbon-carbon bond on a (C 2 -C 9 )-alkylene group, then the two groups R 1 and R 2 with the two nitrogen atoms to which they are attached and The central carbon atoms of the guanidino group to which these two nitrogen atoms are bonded together form a bicyclic heterocycle which is bonded to the nitrogen atom of the group ( CH2 ) 2 -CO-NH and which can be substituted as indicated. The fused (or condensed) 5- to 7-membered ring may be saturated, mono- or di-unsaturated or aromatic. Thus, for example, a cyclopentane ring, cyclohexane ring, cyclohexene ring, cyclohexadiene ring, cycloheptane ring or benzene ring can be condensed. An example of such a bicyclic heterocyclic group capable of being attached to the nitrogen atom of the group (CH 2 ) 2 —CO—NH is 1,3a,4,5,6,6a-hexahydro-1,3-di Azapenten-2-yl, 1H-benzimidazol-2-yl, 3a, 4, 5, 6, 7, 7a-hexahydro-1H-benzimidazol-2-yl, 4, 5, 6, 7-tetrahydro-1H-benzimidazol-2-yl, 4,7-dihydro-1H-benzimidazol-2-yl or 1H-imidazo[4,5-b]pyridin-2-yl. If the condensed rings are substituted and/or if the (C 2 -C 9 )-alkylene groups are substituted, they are preferably mono- or disubstituted independently of one another by identical or different radicals R 3 . If the alkyl groups representing R 1 and/or R 2 are substituted, they are preferably mono- or di-substituted, in particular mono-substituted independently of each other, by identical or different radicals R 3 .
出现在式I化合物中的旋光性碳原子能够彼此独立具有R构型或S构型。式I化合物能够以纯的对映体或纯的非对映体的形式呈现或者以对映体混合物的形式呈现,例如以外消旋体的形式或非对映体混合物的形式呈现。本发明涉及纯的对映体和对映体混合物以及涉及纯的非对映体和非对映体混合物。本发明包括式I的两种或多于两种立体异构体和混合物中所有比例的立体异构体。式I化合物能够任选作为E异构体或Z异构体呈现。本发明涉及纯的E异构体和纯的Z异构体和以所有比例存在的E/Z混合物。本发明也包括所有互变异构体形式的式I化合物,例如,除了所述式I中显示的形式以外,也包括其中酰基胍单位作为-CO-N=C(NHR1)-NR2R6基团呈现的形式,并且包括其通过不同位置的流动氢原子区分的所有其它的形式。例如通过层析法能够把非对映体包括E/Z异构体分离成为单个的异构体。通过常规方法例如通过手性相上的层析法或通过拆分,能够将外消旋体分离成为两个对映体。通过使用立体化学均一的起始原料或者通过使用立体选择性反应也能够得到立体化学均一的式I化合物。The optically active carbon atoms present in the compounds of formula I can independently of each other have the R-configuration or the S-configuration. The compounds of formula I can be present in the form of pure enantiomers or pure diastereomers or in the form of mixtures of enantiomers, for example in the form of racemates or in the form of mixtures of diastereomers. The present invention relates to the pure enantiomers and enantiomeric mixtures as well as to the pure diastereomers and diastereomeric mixtures. The present invention includes two or more than two stereoisomers of formula I and all ratios of stereoisomers in mixtures. Compounds of formula I can optionally be present as E isomer or Z isomer. The present invention relates to pure E isomers and pure Z isomers and E/Z mixtures in all ratios. The invention also includes compounds of formula I in all tautomeric forms, for example, in addition to the forms shown in said formula I, also in which the acylguanidine unit is represented as -CO-N=C(NHR 1 )-NR 2 R 6 forms, and include all other forms that are distinguished by mobile hydrogen atoms in different positions. Diastereomers, including E/Z isomers, can be separated into individual isomers, for example, by chromatography. Racemates can be separated into the two enantiomers by conventional methods, for example by chromatography on chiral phases or by resolution. Stereochemically uniform compounds of formula I can also be obtained by using stereochemically uniform starting materials or by using stereoselective reactions.
式I化合物的生理学上可耐受的盐为生理学上可接受的,特别是药学上可利用盐的非毒性的盐。含有酸性基团例如羧基的式I化合物的此类盐例如为碱金属盐或碱土金属盐,例如钠盐、钾盐、镁盐和钙盐,还有与生理学上可耐受的季胺离子的盐和与氨和生理学上可耐受的有机胺,例如三乙胺、乙醇胺或三-(2-羟乙基)胺形成的酸加成盐。含有碱性基团的式I化合物例如与无机酸如盐酸、硫酸或磷酸或者与有机羧酸和磺酸,例如乙酸、枸橼酸、苯甲酸、马来酸、富马酸、酒石酸、甲磺酸或对甲苯磺酸形成的酸加成盐。含有碱性基团和酸性基团例如胍基和羧基的式I化合物能够作为两性离子(内铵盐)呈现,其同样包括在本发明中。Physiologically tolerable salts of the compounds of the formula I are the non-toxic salts of the physiologically acceptable, especially pharmaceutically usable salts. Such salts of compounds of the formula I containing acidic groups such as carboxyl groups are, for example, alkali metal salts or alkaline earth metal salts, such as sodium, potassium, magnesium and calcium salts, also in combination with physiologically tolerable quaternary ammonium ions. Salts and acid addition salts with ammonia and physiologically tolerable organic amines, for example triethylamine, ethanolamine or tris-(2-hydroxyethyl)amine. Compounds of the formula I containing basic groups, for example, with mineral acids such as hydrochloric acid, sulfuric acid or phosphoric acid or with organic carboxylic and sulfonic acids, such as acetic acid, citric acid, benzoic acid, maleic acid, fumaric acid, tartaric acid, methanesulfonic acid Acid addition salts formed with acid or p-toluenesulfonic acid. Compounds of the formula I which contain basic groups and acidic groups such as guanidine and carboxyl groups can be present as zwitterions (betaines) and are likewise included in the present invention.
当R4为由正电荷铵基团取代的烷基时,包含在式I化合物中的生理学上可耐受的阴离子Q-特别为一价阴离子或等价的多价阴离子的非毒性的、生理学上可利用的,特别也为药学上可利用的无机酸或有机酸,例如适用于形成酸加成盐的以上提到的酸之一的阴离子或阴离子等价物。因此,Q-可为例如选自氯化物、硫酸盐、磷酸盐、乙酸盐、枸橼酸盐、苯甲酸盐、马来酸盐、富马酸盐、酒石酸盐、甲磺酸盐和对甲苯磺酸盐的所述阴离子(或者为阴离子等价物)之一。When R is an alkyl group substituted with a positively charged ammonium group, the physiologically tolerable anion Q contained in the compound of formula I is a non-toxic, physiologically acceptable monovalent anion or an equivalent polyvalent anion. Commercially available, especially also pharmaceutically available inorganic or organic acids, for example anions or anion equivalents of one of the above-mentioned acids which are suitable for the formation of acid addition salts. Thus, Q - may be selected from, for example, chlorides, sulfates, phosphates, acetates, citrates, benzoates, maleates, fumarates, tartrates, methanesulfonates and One of said anions (or anion equivalents) of p-toluenesulfonate.
通过本领域技术人员已知的常规方法例如通过在溶剂或分散物中使式I化合物与无机酸或有机酸或碱结合,或者通过从其它的盐进行阳离子交换或阴离子交换,能够制备式I化合物的盐。本发明也包括式I化合物的所有盐,其由于低的生理学上可耐受性并不直接适宜于在药剂中使用,但是例如适宜于用作进行式I化合物的其它化学修饰的中间体或者作为用于制备生理学上可耐受的盐的起始原料。Compounds of formula I can be prepared by conventional methods known to those skilled in the art, for example by combining compounds of formula I with inorganic or organic acids or bases in solvents or dispersions, or by cation exchange or anion exchange from other salts of salt. The invention also includes all salts of the compounds of the formula I which are not directly suitable for use in medicaments due to their low physiological tolerability, but which are suitable, for example, as intermediates for carrying out other chemical modifications of the compounds of the formula I or as Starting material for the preparation of physiologically tolerated salts.
此外,本发明包括式I化合物的所有溶剂合物,例如水合物或与醇类的加合物,并且也包括式I化合物的衍生物,例如酯类、前药和其它的生理学上可耐受的衍生物以及式I化合物的活性代谢物。本发明特别涉及式I化合物的前药,它们在生理条件下能够转化为式I化合物。适宜的式I化合物的前药即具有以所要求的方式改善了性质的式I化合物的化学修饰衍生物对于本领域技术人员是已知的。涉及前药的更详细的信息发现在例如Fleisher等,Advanced Drug DeliveryReviews 19(1996)115-130; 前药设计,H.Bundgaard,Ed.,Elsevier,1985;H.Bundgaard, 未来的药物16(1991)443;Saulnier等,Bioorg.Med.Chem.Lett.4(1994)1985;Safadi等,Pharmaceutical Res.10(1993)1350中,其通过引用结合到本文中。式I化合物的适宜的前药特别为羧酸基团的酯前药,特别是当基团COOR4中的R4为氢时,存在COOH基团,例如该基团的烷基酯像(C1-C6)-烷基酯或(C1-C4)-烷基酯,并且也可为可酰基化的含氮基团的酰基前药和氨基甲酸酯前药例如氨基并且特别为胍基。在所述酰基前药或氨基甲酸酯前药中,在这些基团中位于氮原子上的氢原子被酰基基团或氨基甲酸酯基团取代一次或多次,例如两次。用于所述酰基前药和氨基甲酸酯前药的适宜的酰基基团和氨基甲酸酯基团有例如基团R10-CO和R11O-CO,其中R10为氢、(C1-C18)-烷基、(C3-C14)-环烷基、(C3-C14)-环烷基-(C1-C8)-烷基-、(C5-C14)-芳基,其中1至5个碳原子能够由杂原子例如N、O或S替代,或者为(C5-C14)-芳基-(C1-C8)-烷基-,其中在所述芳基部分中的1至5个碳原子能够由杂原子例如N、O或S替代,并且其中R11具有除了氢以外对R10所指明的含义。Furthermore, the invention includes all solvates of the compounds of the formula I, such as hydrates or adducts with alcohols, and also derivatives of the compounds of the formula I, such as esters, prodrugs and other physiologically tolerable Derivatives of and active metabolites of compounds of formula I. The present invention relates in particular to prodrugs of compounds of formula I which are capable of being converted into compounds of formula I under physiological conditions. Suitable prodrugs of the compounds of the formula I, ie chemically modified derivatives of the compounds of the formula I having properties improved in the desired manner, are known to the person skilled in the art. More detailed information concerning prodrugs is found in, for example, Fleisher et al., Advanced Drug Delivery Reviews 19 (1996) 115-130; Prodrug Design , H. Bundgaard, Ed., Elsevier, 1985; H. Bundgaard, Future Drugs 16 (1991 ) 443; Saulnier et al., Bioorg. Med. Chem. Lett. 4 (1994) 1985; Safadi et al., Pharmaceutical Res. 10 (1993) 1350, which are incorporated herein by reference. Suitable prodrugs of compounds of formula I are in particular ester prodrugs of carboxylic acid groups, especially when R in the group COOR 4 is hydrogen, a COOH group is present, for example an alkyl ester of this group like (C 1 -C 6 )-Alkyl esters or (C 1 -C 4 )-Alkyl esters, and also acyl prodrugs and carbamate prodrugs of acylated nitrogen-containing groups such as amino groups and in particular Guanidino. In the acyl or carbamate prodrugs, the hydrogen atoms located on the nitrogen atoms in these groups are substituted one or more times, for example twice, by an acyl group or a carbamate group. Suitable acyl and carbamate groups for said acyl prodrugs and carbamate prodrugs are, for example, the groups R 10 —CO and R 11 O—CO, where R 10 is hydrogen, (C 1 -C 18 )-Alkyl, (C 3 -C 14 )-Cycloalkyl, (C 3 -C 14 )-Cycloalkyl-(C 1 -C 8 )-Alkyl-, (C 5 -C 14 )-aryl, wherein 1 to 5 carbon atoms can be replaced by heteroatoms such as N, O or S, or (C 5 -C 14 )-aryl-(C 1 -C 8 )-alkyl-, wherein 1 to 5 carbon atoms in said aryl moiety can be replaced by heteroatoms such as N, O or S, and wherein R 11 has the meaning indicated for R 10 except hydrogen.
在式I化合物中,基团R1和R2优选为氢或一起为饱和的或不饱和的,特别是饱和的二价(C2-C5)-亚烷基、特别为(C2-C4)-亚烷基、尤其为(C2-C3)-亚烷基,其为未取代的或由一个或两个相同的或不同的选自卤素、(C1-C6)-烷基、(C1-C6)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C6)-烷基-、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C6)-烷基-、(C3-C12)-环烷基、(C3-C12)-环烷基-(C1-C6)-烷基-和氧代的基团取代,这里为未取代的或由R3取代,特别是由一个或两个基团R3取代,并且其为碳环或含有一个或两个环氮原子的杂环的5-元至7-元饱和的或不饱和的环能够稠和于所述亚烷基中的碳-碳键上。在式I化合物中,基团R1和R2特别优选为氢或为其中p为数目2、3、4或5,优选为2、3或4,特别优选为2或3的基团-(CH2)P-,并且其为未取代的或者由一个或两个相同的或不同的选自卤素、(C1-C6)-烷基、(C1-C6)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C6)-烷基-、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C6)-烷基-、(C3-C12)-环烷基、(C3-C12)-环烷基-(C1-C6)-烷基-和氧代的基团取代,这里为未取代的或由R3特别是由一个或两个基团R3取代,并且其为碳环或含有一个或两个环氮原子的杂环的5-元至7-元饱和的或不饱和的环能够稠和于基团-(CH2)P-中的碳-碳键上。所述基团R1和R2更特别优选一起为基团-(CH2)P-,其中p为数目2、3、4或5,优选为2、3或4,特别优选为2或3,其优选为未取代的。所述基团R1-和R2-特别优选一起为所述二价基团-CH2-CH2-CH2-,即R1和R2与它们连接的氮原子和与这两个氮原子连接的胍基中心碳原子一起形成1,4,5,6-四氢-2-嘧啶基;In compounds of formula I, the radicals R 1 and R 2 are preferably hydrogen or together are saturated or unsaturated, in particular saturated divalent (C 2 -C 5 )-alkylene, in particular (C 2 - C 4 )-Alkylene, especially (C 2 -C 3 )-Alkylene, which is unsubstituted or consists of one or two identical or different halogens, (C 1 -C 6 )- Alkyl, (C 1 -C 6 )-alkoxy, (C 6 -C 14 )-aryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl-, ( C 5 -C 14 )-heteroaryl, (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, (C 3 -C 12 )-cycloalkyl, (C 3 -C 12 )-cycloalkyl-(C 1 -C 6 )-alkyl- and oxo-substituted, here unsubstituted or substituted by R 3 , in particular by one or two radicals R 3 5-membered to 7-membered saturated or unsaturated rings which are carbocyclic or heterocyclic containing one or two ring nitrogen atoms can be fused to a carbon-carbon bond in the alkylene group . In the compounds of formula I, the radicals R and R are particularly preferably hydrogen or a radical in which p is the number 2, 3, 4 or 5, preferably 2, 3 or 4, particularly preferably 2 or 3, -( CH 2 ) P -, and it is unsubstituted or consists of one or two identical or different selected from halogen, (C 1 -C 6 )-alkyl, (C 1 -C 6 )-alkoxy, (C 6 -C 14 )-aryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl-, (C 5 -C 14 )-heteroaryl, (C 5 - C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, (C 3 -C 12 )-cycloalkyl, (C 3 -C 12 )-cycloalkyl-(C 1 -C 6 )-alkyl- and oxo-substituted groups, here unsubstituted or substituted by R 3 especially by one or two groups R 3 , and which are carbocyclic or contain one or two ring nitrogen atoms The 5- to 7-membered saturated or unsaturated ring of the heterocycle can be fused to a carbon-carbon bond in the group -(CH 2 ) P -. The radicals R 1 and R 2 are more particularly preferably together a radical -(CH 2 ) P -, where p is the number 2, 3, 4 or 5, preferably 2, 3 or 4, particularly preferably 2 or 3 , which is preferably unsubstituted. The groups R 1 - and R 2 - are particularly preferably together the divalent group -CH 2 -CH 2 -CH 2 -, ie R 1 and R 2 are connected to the nitrogen atom to which they are attached and to the two nitrogens The central carbon atoms of the guanidine group connected by the atoms together form 1,4,5,6-tetrahydro-2-pyrimidinyl;
R3优选为(C1-C10)-烷基、(C3-C20)-单环烷基、(C5-C20)-双环烷基、(C5-C20)-三环烷基、(C1-C8)-烷氧基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C4)-烷基-、(C5-C14)-杂芳基-(C1-C4)-烷基-、卤素、三氟甲基、氰基、氧代、-N((C1-C4)-烷基)2或-NH-CO-(C1-C4)-烷基。R3更优选为(C1-C4)-烷基、(C3-C10)-单环烷基、(C5-C12)-双环烷基、(C5-C12)-三环烷基、(C1-C4)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C4)-烷基-、卤素、三氟甲基、氰基、氧代、-N((C1-C4)-烷基)2或-NH-CO-(C1-C4)-烷基。R3特别优选为(C1-C4)-烷基、(C3-C10)-单环烷基、(C5-C12)-双环烷基、(C5-C12)-三环烷基、(C1-C4)-烷氧基、(C6-C14)-芳基、卤素、三氟甲基、氰基、氧代、-N((C1-C4)-烷基)2或-NH-CO-(C1-C4)-烷基。R 3 is preferably (C 1 -C 10 )-alkyl, (C 3 -C 20 )-monocycloalkyl, (C 5 -C 20 )-bicycloalkyl, (C 5 -C 20 )-tricyclic Alkyl, (C 1 -C 8 )-alkoxy, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-( C 1 -C 4 )-alkyl-, (C 5 -C 14 )-heteroaryl-(C 1 -C 4 )-alkyl-, halogen, trifluoromethyl, cyano, oxo, -N ((C 1 -C 4 )-Alkyl) 2 or -NH-CO-(C 1 -C 4 )-Alkyl. R 3 is more preferably (C 1 -C 4 )-alkyl, (C 3 -C 10 )-monocycloalkyl, (C 5 -C 12 )-bicycloalkyl, (C 5 -C 12 )-tricycloalkyl Cycloalkyl, (C 1 -C 4 )-alkoxy, (C 6 -C 14 )-aryl, (C 6 -C 14 )-aryl-(C 1 -C 4 )-alkyl-, Halogen, trifluoromethyl, cyano, oxo, -N((C 1 -C 4 )-alkyl) 2 or -NH-CO-(C 1 -C 4 )-alkyl. R 3 is particularly preferably (C 1 -C 4 )-alkyl, (C 3 -C 10 )-monocycloalkyl, (C 5 -C 12 )-bicycloalkyl, (C 5 -C 12 )-tricycloalkyl Cycloalkyl, (C 1 -C 4 )-alkoxy, (C 6 -C 14 )-aryl, halogen, trifluoromethyl, cyano, oxo, -N((C 1 -C 4 ) -alkyl) 2 or -NH-CO-(C 1 -C 4 )-alkyl.
R4优选为氢或未取代的或取代的(C1-C6)-烷基,特别优选为氢或由选自(C1-C4)-烷氧基、(C1-C4)-烷基-S(O)2-和-NR7R7’的基团取代的(C1-C6)-烷基,其中R7和R7’彼此独立为氢或(C1-C4)-烷基。R4非常特别优选为氢或未取代的或取代的(C1-C4)-烷基,此外优选为氢或如前指明的未取代的或取代的(C1-C4)-烷基。R 4 is preferably hydrogen or unsubstituted or substituted (C 1 -C 6 )-alkyl, particularly preferably hydrogen or a group selected from (C 1 -C 4 )-alkoxy, (C 1 -C 4 ) -Alkyl-S(O) 2 - and -NR 7 R 7' group substituted (C 1 -C 6 )-alkyl, wherein R 7 and R 7' are independently hydrogen or (C 1 -C 4 )-Alkyl. R 4 is very particularly preferably hydrogen or unsubstituted or substituted (C 1 -C 4 )-alkyl, furthermore preferably hydrogen or unsubstituted or substituted (C 1 -C 4 )-alkyl as indicated above .
R5优选为(C1-C20)-烷基、(C3-C20)-单环烷基、(C5-C20)-双环烷基、(C5-C20)-三环烷基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C6)-烷基-或(C5-C14)-杂芳基-(C1-C6)-烷基-,其中所述芳基、杂芳基、烷基、单环烷基、双环烷基和三环烷基中的每一个为未取代的或者由一个、两个或三个相同或不同的基团R3取代。R5更优选为(C1-C10)-烷基、(C3-C15)-单环烷基、(C5-C15)-双环烷基、(C5-C15)-三环烷基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C6)-烷基-或(C5-C14)-杂芳基-(C1-C6)-烷基-,其中所述芳基、所述杂芳基、烷基、单环烷基、双环烷基和三环烷基中的每一个为未取代的或者由一个、两个或三个相同或不同的基团R3取代。除了这些优选基团以外,一类优选的基团R5由其中能被取代的或者否则如上指明的那样被修饰的基团(C3-C20)-单环烷基、(C5-C20)-双环烷基和(C5-C20)-三环烷基形成,并且更优选由(C5-C15)-单环烷基、(C5-C15)-双环烷基、(C5-C15)-三环烷基形成。另一类优选的基团R5由基团(C1-C20)-烷基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C6)-烷基-或(C5-C14)-杂芳基-(C1-C6)-烷基-形成,其中(C6-C14)-芳基和(C5-C14)-杂芳基为优选,其能够被取代或者否则如同以上指明的那样被修饰。一类特别优选的基团R5由基团苯基和萘基形成,即可以由未取代的或如同以上指明那样取代的苯基、1-萘基和2-萘基形成。R 5 is preferably (C 1 -C 20 )-alkyl, (C 3 -C 20 )-monocycloalkyl, (C 5 -C 20 )-bicycloalkyl, (C 5 -C 20 )-tricyclic Alkyl, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl- or ( C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, wherein the aryl, heteroaryl, alkyl, monocycloalkyl, bicycloalkyl and tricycloalkyl Each of is unsubstituted or substituted by one, two or three identical or different groups R 3 . R 5 is more preferably (C 1 -C 10 )-alkyl, (C 3 -C 15 )-monocycloalkyl, (C 5 -C 15 )-bicycloalkyl, (C 5 -C 15 )-tricycloalkyl Cycloalkyl, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl- or (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, wherein said aryl, said heteroaryl, alkyl, monocycloalkyl, bicycloalkyl and tricyclic Each of the alkyl groups is unsubstituted or substituted by one, two or three identical or different groups R 3 . In addition to these preferred groups, one class of preferred groups R 5 consists of groups (C 3 -C 20 )-monocycloalkyl, (C 5 -C 20 )-bicycloalkyl and (C 5 -C 20 )-tricycloalkyl are formed, and are more preferably formed from (C 5 -C 15 )-monocycloalkyl, (C 5 -C 15 )-bicycloalkyl, (C 5 -C 15 )-Tricycloalkyl formation. Another preferred group R 5 consists of the groups (C 1 -C 20 )-alkyl, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 - C 14 )-aryl-(C 1 -C 6 )-alkyl- or (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl- forms, wherein (C 6 -C 14 )-Aryl and (C 5 -C 14 )-heteroaryl are preferred, which can be substituted or otherwise modified as indicated above. A particularly preferred class of radicals R5 is formed from the radicals phenyl and naphthyl, ie from phenyl, 1-naphthyl and 2-naphthyl which may be unsubstituted or substituted as specified above.
R6优选为氢或(C1-C6)-烷基-O-CO-,特别优选为氢或(C1-C4)-烷基-O-CO-,特别为氢。R 6 is preferably hydrogen or (C 1 -C 6 )-alkyl-O-CO—, particularly preferably hydrogen or (C 1 -C 4 )-alkyl-O—CO—, especially hydrogen.
优选的式I化合物为那些其中一个或多个基团具有优选的含义或者具有一个特别的或一些特别的它们的分别含义的化合物,这样优选的含义或特殊含义的所有结合为本发明的目的。特别优选的式I化合物为那些其中R1和R2为氢或一起为饱和的或不饱和的二价(C2-C5)-亚烷基,特别为氢或一起为其中p为数目2、3、4或5的基团-(CH2)p-的化合物,这里(C2-C5)-亚烷基和基团-(CH2)p-为未取代的或者由选自卤素、(C1-C6)-烷基、(C1-C6)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C6)-烷基-、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C6)-烷基-、(C3-C12)-环烷基、(C3-C12)-环烷基-(C1-C6)-烷基-和氧代的基团取代,并且这里为未取代的或由R3特别由一个或两个基团R3取代,并且为碳环或含有一个或两个环氮原子的杂环的5-元至7-元饱和的或不饱和的环能够稠和于(C2-C5)-亚烷基和基团-(CH2)P-中的碳-碳键上;Preferred compounds of formula I are those in which one or more radicals have a preferred meaning or have one particular or some particulars of their respective meanings, all combinations of such preferred meanings or special meanings being the object of the present invention. Particularly preferred compounds of formula I are those in which R and R are hydrogen or together a saturated or unsaturated divalent (C 2 -C 5 )-alkylene group, in particular hydrogen or together in which p is the number 2 , 3, 4 or 5 of the group -(CH 2 ) p -compounds, where (C 2 -C 5 )-alkylene and the group -(CH 2 ) p - are unsubstituted or are selected from halogen , (C 1 -C 6 )-alkyl, (C 1 -C 6 )-alkoxy, (C 6 -C 14 )-aryl, (C 6 -C 14 )-aryl-(C 1 - C 6 )-Alkyl-, (C 5 -C 14 )-Heteroaryl, (C 5 -C 14 )-Heteroaryl-(C 1 -C 6 )-Alkyl-, (C 3 -C 12 )-cycloalkyl, (C 3 -C 12 )-cycloalkyl-(C 1 -C 6 )-alkyl- and oxo-substituted, and here unsubstituted or by R 3 in particular by one or two groups R 3 substituted, and a carbocyclic or heterocyclic 5-membered to 7-membered saturated or unsaturated ring containing one or two ring nitrogen atoms can be fused to (C 2 -C 5 )-alkylene and the group -(CH 2 ) P -on the carbon-carbon bond;
R3为(C1-C10)-烷基、(C3-C20)-单环烷基、(C5-C20)-双环烷基、(C5-C20)-三环烷基、(C1-C8)-烷氧基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C4)-烷基-、(C5-C14)-杂芳基-(C1-C4)-烷基-、卤素、三氟甲基、氰基、氧代、-N((C1-C4)-烷基)2或-NH-CO-(C1-C4)-烷基;R 3 is (C 1 -C 10 )-alkyl, (C 3 -C 20 )-monocycloalkyl, (C 5 -C 20 )-bicycloalkyl, (C 5 -C 20 )-tricycloalkane radical, (C 1 -C 8 )-alkoxy, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-(C 1 -C 4 )-alkyl-, (C 5 -C 14 )-heteroaryl-(C 1 -C 4 )-alkyl-, halogen, trifluoromethyl, cyano, oxo, -N( (C 1 -C 4 )-alkyl) 2 or -NH-CO-(C 1 -C 4 )-alkyl;
R4为氢或其为未取代的或由选自(C1-C4)-烷氧基、(C1-C4)-烷基-S(O)2-和NR7R7’的基团取代的(C1-C6)-烷基,其中R7和R7’彼此独立为氢或(C1-C4)-烷基;R 4 is hydrogen or it is unsubstituted or consists of (C 1 -C 4 )-alkoxy, (C 1 -C 4 )-alkyl-S(O) 2 - and NR 7 R 7' (C 1 -C 6 )-alkyl group substituted, wherein R 7 and R 7' are independently hydrogen or (C 1 -C 4 )-alkyl;
R5为(C1-C20)-烷基、(C3-C20)-单环烷基、(C5-C20)-双环烷基、(C5-C20)-三环烷基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C6)-烷基-或(C5-C14)-杂芳基-(C1-C6)-烷基-,其中所述芳基、杂芳基、烷基、单环烷基、双环烷基和三环烷基中每一个为未取代的或者由一个、两个或三个基团R3取代;R 5 is (C 1 -C 20 )-alkyl, (C 3 -C 20 )-monocycloalkyl, (C 5 -C 20 )-bicycloalkyl, (C 5 -C 20 )-tricycloalkane radical, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl- or (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, wherein each of the aryl, heteroaryl, alkyl, monocycloalkyl, bicycloalkyl and tricycloalkyl one is unsubstituted or substituted by one, two or three groups R3 ;
R6为氢或(C1-C6)-烷基-O-CO-;R 6 is hydrogen or (C 1 -C 6 )-alkyl-O-CO-;
其所有的立体异构体形式及其所有比例的混合物的形式和它们生理学上可耐受的盐和它们的前药。All stereoisomeric forms and mixtures thereof in all proportions and their physiologically tolerable salts and their prodrugs.
非常特别优选的式I化合物为那些化合物,其中Very particularly preferred compounds of formula I are those in which
R1和R2为氢或一起为饱和的或不饱和的二价(C2-C4)-亚烷基,特别为氢或一起为其中p为数目2、3或4的基团-(CH2)p-,这里(C2-C4)-亚烷基和基团-(CH2)p-为未取代的或者由选自卤素、(C1-C6)-烷基、(C1-C6)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C6)-烷基-、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C6)-烷基-、(C3-C12)-环烷基、(C3-C12)-环烷基-(C1-C6)-烷基-和氧代的基团取代,并且这里其为未取代的或由R3特别由一个或两个基团R3取代,并且为碳环或含有一个或两个环氮原子的杂环的5-元至7-元饱和的或不饱和的环能够稠和于(C2-C4)-亚烷基和基团-(CH2)P-中的碳-碳键上;R 1 and R 2 are hydrogen or together a saturated or unsaturated divalent (C 2 -C 4 )-alkylene group, in particular hydrogen or together a group in which p is the number 2, 3 or 4 -( CH 2 ) p -, where (C 2 -C 4 )-alkylene and the group -(CH 2 ) p - are unsubstituted or are formed from the group consisting of halogen, (C 1 -C 6 )-alkyl, ( C 1 -C 6 )-alkoxy, (C 6 -C 14 )-aryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl-, (C 5 -C 14 )-heteroaryl, (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, (C 3 -C 12 )-cycloalkyl, (C 3 -C 12 ) -Cycloalkyl-(C 1 -C 6 )-alkyl- and oxo-substituted groups, and here they are unsubstituted or substituted by R 3 in particular by one or two groups R 3 , and are carbon 5- to 7-membered saturated or unsaturated rings of rings or heterocycles containing one or two ring nitrogen atoms can be fused to (C 2 -C 4 )-alkylene and the group -(CH 2 ) on the carbon-carbon bond in P- ;
R3为(C1-C4)-烷基、(C3-C10)-单环烷基、(C5-C12)-双环烷基、(C5-C12)-三环烷基、(C1-C4)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C4)-烷基-、卤素、三氟甲基、氰基、氧代、-N((C1-C4)-烷基)2或-NH-CO-(C1-C4)-烷基;R 3 is (C 1 -C 4 )-alkyl, (C 3 -C 10 )-monocycloalkyl, (C 5 -C 12 )-bicycloalkyl, (C 5 -C 12 )-tricycloalkane radical, (C 1 -C 4 )-alkoxy, (C 6 -C 14 )-aryl, (C 6 -C 14 )-aryl-(C 1 -C 4 )-alkyl-, halogen, Trifluoromethyl, cyano, oxo, -N((C 1 -C 4 )-alkyl) 2 or -NH-CO-(C 1 -C 4 )-alkyl;
R4为氢或(C1-C6)-烷基;R 4 is hydrogen or (C 1 -C 6 )-alkyl;
R5为(C1-C10)-烷基、(C3-C15)-单环烷基、(C5-C15)-双环烷基、(C5-C15)-三环烷基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C6)-烷基-或(C5-C14)-杂芳基-(C1-C6)-烷基-,其中所述芳基、杂芳基、烷基、单环烷基、双环烷基和三环烷基中每一个为未取代的或者由一个、两个或三个R3基团取代;R 5 is (C 1 -C 10 )-alkyl, (C 3 -C 15 )-monocycloalkyl, (C 5 -C 15 )-bicycloalkyl, (C 5 -C 15 )-tricycloalkane radical, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl- or (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, wherein each of the aryl, heteroaryl, alkyl, monocycloalkyl, bicycloalkyl and tricycloalkyl one is unsubstituted or substituted by one, two or three R groups;
R6优选为氢或(C1-C4)-烷基-O-CO-;R 6 is preferably hydrogen or (C 1 -C 4 )-alkyl-O-CO-;
其所有的立体异构体形式及其所有比例的混合物的形式和它们生理学上可耐受的盐和它们的前药。All stereoisomeric forms and mixtures thereof in all proportions and their physiologically tolerable salts and their prodrugs.
特别优选的式I化合物为这样的化合物,其中:Particularly preferred compounds of formula I are those wherein:
R1和R2为氢或一起为饱和的或不饱和的二价(C2-C3)-亚烷基,特别为氢或一起为其中p为数目2或3的基团-(CH2)p-,这里(C2-C3)-亚烷基和基团-(CH2)p-为未取代的或者由选自卤素、(C1-C6)-烷基、(C1-C6)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C6)-烷基-、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C6)-烷基-、(C3-C12)-环烷基、(C3-C12)-环烷基-(C1-C6)-烷基-和氧代的基团取代,并且这里为未取代的或由R3特别由一个或两个R3基团取代并且为碳环或含有一个或两个环氮原子的杂环的5-元至7-元饱和的或不饱和的环能够稠和于(C2-C3)-亚烷基和基团-(CH2)P-中的碳-碳键上;R 1 and R 2 are hydrogen or together a saturated or unsaturated divalent (C 2 -C 3 )-alkylene group, in particular hydrogen or together a group in which p is the number 2 or 3 -(CH 2 ) p -, where (C 2 -C 3 )-alkylene and the group -(CH 2 ) p - are unsubstituted or are formed from the group consisting of halogen, (C 1 -C 6 )-alkyl, (C 1 -C 6 )-alkoxy, (C 6 -C 14 )-aryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl-, (C 5 -C 14 ) -heteroaryl, (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, (C 3 -C 12 )-cycloalkyl, (C 3 -C 12 )-cyclo Alkyl-(C 1 -C 6 )-alkyl- and oxo groups are substituted and here unsubstituted or substituted by R 3 especially by one or two R 3 groups and are carbocyclic or contain one A 5-membered to 7-membered saturated or unsaturated ring of a heterocyclic ring with two ring nitrogen atoms can be fused in a (C 2 -C 3 )-alkylene group and a group -(CH 2 ) P - on the carbon-carbon bond;
R3为(C1-C4)-烷基、(C3-C10)-单环烷基、(C5-C12)-双环烷基、(C5-C12)-三环烷基、(C1-C4)-烷氧基、(C6-C14)-芳基、卤素、三氟甲基、氰基、氧代、-N((C1-C4)-烷基)2或-NH-CO-(C1-C4)-烷基;R 3 is (C 1 -C 4 )-alkyl, (C 3 -C 10 )-monocycloalkyl, (C 5 -C 12 )-bicycloalkyl, (C 5 -C 12 )-tricycloalkane radical, (C 1 -C 4 )-alkoxy, (C 6 -C 14 )-aryl, halogen, trifluoromethyl, cyano, oxo, -N((C 1 -C 4 )-alk base) 2 or -NH-CO-(C 1 -C 4 )-alkyl;
R4为氢或(C1-C6)-烷基;R 4 is hydrogen or (C 1 -C 6 )-alkyl;
R5为(C1-C10)-烷基、(C3-C15)-单环烷基、(C5-C15)-双环烷基、(C5-C15)-三环烷基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C6)-烷基-或(C5-C14)-杂芳基-(C1-C6)-烷基-,其中所述芳基、杂芳基、烷基、单环烷基、双环烷基和三环烷基中每一个为未取代的或者由一个、两个或三个R3基团取代;R 5 is (C 1 -C 10 )-alkyl, (C 3 -C 15 )-monocycloalkyl, (C 5 -C 15 )-bicycloalkyl, (C 5 -C 15 )-tricycloalkane radical, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl- or (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl-, wherein each of the aryl, heteroaryl, alkyl, monocycloalkyl, bicycloalkyl and tricycloalkyl one is unsubstituted or substituted by one, two or three R groups;
R6为氢或(C1-C6)-烷基-O-CO-;R 6 is hydrogen or (C 1 -C 6 )-alkyl-O-CO-;
其所有的立体异构体形式及其所有比例的混合物的形式和它们生理学上可耐受的盐和它们的前药。All stereoisomeric forms and mixtures thereof in all proportions and their physiologically tolerable salts and their prodrugs.
此外优选的式I化合物为那些以所有它们的立体异构体形式和以所有比例的它们的混合物的形式存在的和它们生理学上可耐受的盐和它们的前药存在的化合物,其中R5为(C6-C14)-芳基或(C5-C14)-杂芳基,优选为(C6-C14)-芳基,其中芳基和杂芳基每一个为未取代的或者由一个、两个或三个相同的或不同的R3基团取代,并且优选为未取代的或者由一个或两个相同的或不同的R3基团取代。甚至更优选式I化合物为那些以所有它们的立体异构体形式和以所有比例的它们的混合物的形式存在和它们生理学上可耐受的盐和它们的前药存在的化合物,其中R5为萘基例如1-萘基或2-萘基,其为未取代的或者由一个、两个或三个R3基团取代,并且其优选为未取代的例如未取代的1-萘基或未取代的2-萘基。Further preferred compounds of the formula I are those present in all their stereoisomeric forms and in the form of their mixtures in all proportions and their physiologically tolerable salts and their prodrugs, wherein R 5 is (C 6 -C 14 )-aryl or (C 5 -C 14 )-heteroaryl, preferably (C 6 -C 14 )-aryl, wherein each of aryl and heteroaryl is unsubstituted or substituted by one, two or three identical or different R3 groups, and preferably unsubstituted or substituted by one or two identical or different R3 groups. Even more preferred compounds of formula I are those in all their stereoisomeric forms and in the form of their mixtures in all proportions and their physiologically tolerable salts and their prodrugs, wherein R is Naphthyl such as 1-naphthyl or 2-naphthyl, which is unsubstituted or substituted by one, two or three R groups, and which is preferably unsubstituted such as unsubstituted 1-naphthyl or unsubstituted Substituted 2-naphthyl.
优选的式I化合物还为那些其中所述两个基团R4O-CO-和R5-SO2-NH-连接的碳原子具有S构型的化合物,其以所有它们的立体异构体形式和以所有比例的它们的混合物形式和它们生理学上可耐受的盐和它们的前药存在的化合物。Preferred compounds of formula I are also those in which the carbon atom to which the two radicals R 4 O—CO— and R 5 —SO 2 —NH— are attached has the S configuration, in all their stereoisomers Forms and their mixtures in all proportions and their physiologically tolerable salts and their prodrugs.
一组特殊的式I化合物通过其中R1和R2彼此独立为氢或为未取代或由R3取代的(C1-C6)-烷基的化合物形成,A special group of compounds of formula I is formed by compounds in which R and R independently of each other are hydrogen or unsubstituted or substituted by R 3 ( C 1 -C 6 )-alkyl,
或者其中基团R1-和R2-一起为饱和的或不饱和的二价(C2-C9)-亚烷基,例如其中p为2、3、4、5、6、7、8或9的基团-(CH2)p-,其为未取代的或者由一个或多个选自卤素、(C1-C6)-烷基、(C1-C6)-烷氧基、(C6-C14)-芳基、(C6-C14)-芳基-(C1-C6)-烷基、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C6)-烷基、(C3-C12)-环烷基和(C3-C12)-环烷基-(C1-C6)-烷基-和氧代的基团取代,这里为未取代的或由R3特别由一个或两个R3基团取代,并且为碳环或含有一个或两个环氮原子的杂环的5-元至7-元饱和的或不饱和的环能够稠和于(C2-C9)-亚烷基中的碳-碳键上;or in which the radicals R 1 - and R 2 - together are saturated or unsaturated divalent (C 2 -C 9 )-alkylene groups, for example in which p is 2, 3, 4, 5, 6, 7, 8 or 9 groups -(CH 2 ) p -, which are unsubstituted or composed of one or more selected from halogen, (C 1 -C 6 )-alkyl, (C 1 -C 6 )-alkoxy , (C 6 -C 14 )-aryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl, (C 5 -C 14 )-heteroaryl, (C 5 - C 14 )-heteroaryl-(C 1 -C 6 )-alkyl, (C 3 -C 12 )-cycloalkyl and (C 3 -C 12 )-cycloalkyl-(C 1 -C 6 ) -Alkyl- and oxo-substituted groups, here unsubstituted or substituted by R3 in particular by one or two R3 groups, and carbocyclic or heterocyclic containing one or two ring nitrogen atoms A 5-membered to 7-membered saturated or unsaturated ring can be fused to a carbon-carbon bond in (C 2 -C 9 )-alkylene;
R3为(C1-C8)-烷基、(C1-C8)-烷氧基、(C5-C14)-芳基、(C5-C14)-芳基-(C1-C4)-烷基、(C5-C14)-杂芳基、(C5-C14)-杂芳基-(C1-C4)-烷基、卤素、三氟甲基、羟基、氧代、硝基、氨基、NH-(C1-C4)-烷基、N-((C1-C4)-烷基)2、NH-CO-(C1-C4)-烷基、CO-(C1-C4)-烷基;R 3 is (C 1 -C 8 )-alkyl, (C 1 -C 8 )-alkoxy, (C 5 -C 14 )-aryl, (C 5 -C 14 )-aryl-(C 1 -C 4 )-Alkyl, (C 5 -C 14 )-Heteroaryl, (C 5 -C 14 )-Heteroaryl-(C 1 -C 4 )-Alkyl, Halogen, Trifluoromethyl , hydroxy, oxo, nitro, amino, NH-(C 1 -C 4 )-alkyl, N-((C 1 -C 4 )-alkyl) 2 , NH-CO-(C 1 -C 4 )-alkyl, CO-(C 1 -C 4 )-alkyl;
R4为氢、未取代的或由选自羟基、(C1-C4)-烷氧基、(C1-C4)-烷基-SO2-、-NR7R7’和-N+R7R7’R7”Q-的基团取代的(C1-C6)-烷基-CO-O-(C1-C4)-烷基或者(C1-C6)-烷基,其中R7、R7’和R7”彼此独立为氢、(C1-C6)-烷基、(C5-C14)-芳基或(C5-C14)-芳基-(C1-C6)-烷基并且Q-为生理学上可耐受的阴离子,或者其中R4为所述基团之一;R 4 is hydrogen, unsubstituted or composed of hydroxyl, (C 1 -C 4 )-alkoxy, (C 1 -C 4 )-alkyl-SO 2 -, -NR 7 R 7' and -N + R 7 R 7' R 7” Q - substituted (C 1 -C 6 )-alkyl-CO-O-(C 1 -C 4 )-alkyl or (C 1 -C 6 )- Alkyl, wherein R 7 , R 7' and R 7" are independently hydrogen, (C 1 -C 6 )-alkyl, (C 5 -C 14 )-aryl or (C 5 -C 14 )-aryl Group-(C 1 -C 6 )-alkyl and Q - is a physiologically tolerable anion, or wherein R 4 is one of said groups;
其中所述基团借以连接的游离键通过虚线表明;wherein the free bond through which the group is attached is indicated by a dashed line;
R5为(C1-C20)-烷基、(C5-C20)-单环烷基、(C5-C20)-双环烷基、(C5-C20)-三环烷基、(C6-C14)-芳基、(C5-C14)-杂芳基、(C6-C14)-芳基-(C1-C6)-烷基-或(C5-C14)-杂芳基-(C1-C6)-烷基,其中所述芳基、杂芳基、烷基、单环烷基、双环烷基和三环烷基中每一个为未取代的或者由一个、两个或三个R3基团取代,并且其中在所述烷基、单环烷基、双环烷基和三环烷基中,一个或多个碳原子,特别是一个、两个、三个或四个碳原子;可被相同的或不同的选自氮、氧和硫的原子替代;R6为氢、(C1-C6)-烷基-O-CO、羟基、(C1-C6)-烷基-O-CO-O或者硝基;其所有的立体异构体形式及其所有比例的混合物的形式和它们生理学上可耐受的盐和它们的前药。R 5 is (C 1 -C 20 )-alkyl, (C 5 -C 20 )-monocycloalkyl, (C 5 -C 20 )-bicycloalkyl, (C 5 -C 20 )-tricycloalkane radical, (C 6 -C 14 )-aryl, (C 5 -C 14 )-heteroaryl, (C 6 -C 14 )-aryl-(C 1 -C 6 )-alkyl- or (C 5 -C 14 )-heteroaryl-(C 1 -C 6 )-alkyl, wherein each of the aryl, heteroaryl, alkyl, monocycloalkyl, bicycloalkyl and tricycloalkyl is unsubstituted or substituted by one, two or three R groups, and wherein in said alkyl, monocycloalkyl, bicycloalkyl and tricycloalkyl, one or more carbon atoms, especially is one, two, three or four carbon atoms; can be replaced by the same or different atoms selected from nitrogen, oxygen and sulfur; R 6 is hydrogen, (C 1 -C 6 )-alkyl-O- CO, hydroxy, (C 1 -C 6 )-alkyl-O-CO-O or nitro; all stereoisomeric forms and mixtures thereof in all proportions and their physiologically tolerated salts and their prodrugs.
本发明也涉及用于制备式I化合物的方法。所述化合物一般能够例如在会聚合成过程中通过连接能够从式I逆合成(retrosynthetically)衍化的两种或多种片段来制备。在式I化合物的制备中,在引入能在分别的合成步骤中导致不合乎需要的反应或副反应的官能团时,它以之后转化为合乎需要的官能团的前体形式存在或者通过适合于所述合成问题的保护基团策略暂时阻断官能团的所述合成过程中,这通常是有利的或必要的。这样的策略对本领域技术人员是熟知的(参见例如Greene和Wuts, 有机合成中的保护基团,Wiley,1991)。作为前体基团的实例,硝基和氰基可被提及,它们之后能够通过还原例如通过催化氢化分别转化为氨基和氨基甲基。The present invention also relates to processes for the preparation of compounds of formula I. Said compounds can generally be prepared, for example, by linking two or more fragments capable of retrosynthetically derivatizing from formula I during a convergent synthesis. In the preparation of compounds of formula I, when introducing a functional group which can lead to undesirable reactions or side reactions in the respective synthetic steps, it is present in the form of a precursor which is then converted into the desired functional group or by means of a suitable Protecting Group Strategies for Synthetic Issues It is often beneficial or necessary to temporarily block functional groups during the synthesis described. Such strategies are well known to those skilled in the art (see eg Greene and Wuts, Protecting Groups in Organic Synthesis , Wiley, 1991). As examples of precursor groups, nitro and cyano groups may be mentioned, which can then be converted by reduction, for example by catalytic hydrogenation, into amino and aminomethyl groups, respectively.
式I化合物能够例如通过在本来已知的方法中连接式II的羧酸或羧酸衍生物来制备Compounds of formula I can be prepared, for example, by linking carboxylic acids or carboxylic acid derivatives of formula II in methods known per se
其中R4和R5如上对式I所指明的那样定义,或者其中另外的官能团以后来转化为存在于式I化合物中的所述基团的前体形式存在,或者其中官能团以保护的形式存在,并且其中X为具有式III的胍或胍的衍生物的亲核取代的离去基团,wherein R and R are as defined above for formula I, or wherein additional functional groups are present in the form of precursors which are later converted to said groups present in compounds of formula I, or wherein the functional groups are present in protected form , and wherein X is a nucleophilically substituted leaving group of guanidine or a derivative of guanidine having formula III,
其中R1、R2和R6如上对所述式I所指明的那样定义,或者另外的官能团以其后来转化为存在于所述式I化合物中的基团的前体形式存在,或者官能团以保护的形式存在。wherein R 1 , R 2 and R 6 are as defined above for said formula I, or additional functional groups are present in the form of precursors for their subsequent conversion into groups present in said formula I compounds, or functional groups are present in the form of The form of protection exists.
在式II中,基团COX优选为羧酸基团COOH或活化的羧酸衍生物。X例如为羟基或卤素尤其为氯或溴、烷氧基、优选为甲氧基或乙氧基、芳氧基例如苯氧基或五氟苯氧基、苯硫基、甲硫基、2-吡啶硫代或借助氮原子连接的氮杂环基,特别为吡咯,例如1-咪唑基。X能够另外为例如((C1-C4)-烷基)-O-CO-O-或甲苯磺酰氧基,因此活化的酸衍生物能够为混合酸酐。In formula II, the group COX is preferably a carboxylic acid group COOH or an activated carboxylic acid derivative. X is for example hydroxy or halogen especially chlorine or bromine, alkoxy, preferably methoxy or ethoxy, aryloxy such as phenoxy or pentafluorophenoxy, phenylthio, methylthio, 2- Pyridinethio or nitrogen heterocyclyl attached via a nitrogen atom, especially pyrrole, eg 1-imidazolyl. X can additionally be eg ((C 1 -C 4 )-alkyl)-O-CO-O- or tosyloxy, so the activated acid derivative can be a mixed anhydride.
如果X为羟基,即如果式III的胍与羧酸反应,然后为了方便所述羧酸首先活化。例如,用二环己基碳二亚胺(DCCI)或与O-((氰基(乙氧基羰基)-亚甲基)氨基)-1,1,3,3-四甲基脲鎓(uronium)四氟硼酸盐(TOTU;Konig等,Proc.第21届Europ.Peptide Symp.1990(Eds.Giralt,Andreu),Escom,Leiden 1991,第143页)或其它在肽化学中常见的活化试剂能够进行所述活化。If X is a hydroxyl group, ie if the guanidine of formula III is reacted with a carboxylic acid, then the carboxylic acid is first activated for convenience. For example, with dicyclohexylcarbodiimide (DCCI) or with O-((cyano(ethoxycarbonyl)-methylene)amino)-1,1,3,3-tetramethyluronium (uronium ) Tetrafluoroborate (TOTU; Konig et al., Proc. 21st Europ. Peptide Symp. 1990 (Eds. Giralt, Andreu), Escom, Leiden 1991, p. 143) or other activating reagents common in peptide chemistry enabling the activation.
除了式III游离的胍以外,胍盐也能够被用于与式II化合物的反应中,由此能够就地制备或在分离步骤中借助碱制备游离的胍。以本身已知的方法,式II活化的羧酸衍生物与式III的胍(衍生物)的反应优选在质子或者非质子极性但为惰性的有机溶剂中进行。在这种情况下,在从0℃至这些溶剂的沸点温度下,例如在甲基酯(X=甲氧基)或者在乙基酯(X=乙氧基)与胍的反应中,已证实溶剂像甲醇、异丙醇、叔丁醇、二甲基甲酰胺或四氢呋喃为适宜的。所述类型COX化合物与无盐的胍的反应有利地在非质子惰性溶剂例如二甲基甲酰胺、四氢呋喃、二甲氧基乙烷或二氧六环中进行,如果适当,可加入碱例如叔丁醇钾或甲醇钠。然而,在式II化合物与胍的反应中,例如当使用碱如氢氧化钠时水也能用作溶剂。如果X为例如氯,伴随加入酸清除剂例如另外的碱或者在用于结合生成的盐酸的过量的胍(衍生物)存在下,所述反应有利地进行。提取所述反应混合物,如果要求,然后将反应产物经本领域技术人员熟悉的常规方法纯化。In addition to the free guanidine of the formula III, guanidine salts can also be used in the reaction with compounds of the formula II, whereby the free guanidine can be prepared in situ or in a separate step with the aid of a base. In a manner known per se, the reaction of the activated carboxylic acid derivative of the formula II with the guanidine (derivative) of the formula III is preferably carried out in a protic or aprotic polar but inert organic solvent. In this case, at temperatures from 0° C. to the boiling point of these solvents, for example in the reaction of methyl esters (X = methoxy) or ethyl esters (X = ethoxy) with guanidine, it has been demonstrated that Solvents like methanol, isopropanol, tert-butanol, dimethylformamide or tetrahydrofuran are suitable. The reaction of COX compounds of said type with salt-free guanidine is advantageously carried out in an aprotic solvent such as dimethylformamide, tetrahydrofuran, dimethoxyethane or dioxane, if appropriate with the addition of a base such as t- potassium butoxide or sodium methoxide. However, water can also be used as solvent in the reaction of the compound of formula II with guanidine, eg when using a base such as sodium hydroxide. If X is eg chlorine, the reaction advantageously proceeds with the addition of an acid scavenger such as an additional base or in the presence of excess guanidine (derivative) for binding the hydrochloric acid formed. The reaction mixture is extracted and, if desired, the reaction product is then purified by conventional methods familiar to those skilled in the art.
保护基团任选仍存在于从式II和III化合物中得到的产物并然后通过标准方法除去。例如,通过以三氟乙酸处理,将叔丁酯基团转化为所述羧酸基团,通过氢化除去苄基或者通过仲胺除去芴基甲氧基羰基。如果要求,通过标准方法然后进行其它的反应,例如酰化反应或酯化反应。另外,能够通过已知方法然后进行转化为生理学上可耐受的盐或前药。The protecting group is optionally still present on the products obtained from compounds of formula II and III and is then removed by standard methods. For example, the tert-butyl ester group is converted to the carboxylic acid group by treatment with trifluoroacetic acid, the benzyl group is removed by hydrogenation or the fluorenylmethoxycarbonyl group is removed by a secondary amine. Further reactions, such as acylation or esterification, are then carried out by standard methods, if desired. In addition, conversion to physiologically tolerable salts or prodrugs can then be carried out by known methods.
与给出式I化合物有关的式II和III的原料为市售的或者能够根据或类似于在所述文献中描述的方法进行制备。式II起始成分的制备借助以下流程1中的实例阐明,本发明并不局限于这种合成或它们的起始成分。对本领域技术人员而言,进行所示合成的改进不引起任何问题,这对本发明化合物的制备是必要的。The starting materials of the formulas II and III which are relevant to give the compounds of the formula I are commercially available or can be prepared according to or analogously to methods described in said literature. The preparation of the starting components of formula II is illustrated by means of the examples in Scheme 1 below, the invention not being limited to this synthesis or their starting components. Modifications of the shown syntheses, which are necessary for the preparation of the compounds of the invention, do not pose any problem to those skilled in the art.
因此,例如在吡啶和哌啶的存在下,式IV的羧基苯甲醛能够例如与式V的丙二酸酯盐反应,得到式VI的肉桂酸衍生物,其例如在披钯炭存在下氢化后,得到式VII化合物。在所述羧酸基团活化后,式VII化合物能够与式VIII的2,3-二氨基丙酸衍生物缩合,得到式IX化合物(流程1)。所述缩合能够在例如TOTU或者另一种用于活化羧酸的常规试剂存在下进行。Thus, for example in the presence of pyridine and piperidine, carboxybenzaldehydes of the formula IV can be reacted, for example, with malonate salts of the formula V to give cinnamic acid derivatives of the formula VI, for example after hydrogenation in the presence of palladium on carbon , to obtain the compound of formula VII. After activation of the carboxylic acid group, compounds of formula VII can be condensed with 2,3-diaminopropionic acid derivatives of formula VIII to give compounds of formula IX (Scheme 1). The condensation can be carried out in the presence of eg TOTU or another conventional reagent for activating carboxylic acids.
在式VIII中,Y能够为基团R5-SO2-,其存在于本发明所述最终式I化合物中并且其然后能够保留在所述分子中,或者Y能够为暂时保护2-位氨基的基团并且在之后的步骤中被除去以得到游离的氨基的基团,该氨基然后能够通过用于制备磺酰胺的标准方法例如通过使所述游离胺与式R5-SO2-Cl的磺酰氯反应转化为R5-SO2-NH基团。表示Y的保护基团的一个实例为能够通过催化氢化除去的所述苄氧基羰基(Z基团)。适宜用于引入基团R5-SO2的所述式R5-SO2-Cl的磺酰氯和其它的磺酸衍生物为市售的或者能够按照或类似于在所述文献中描述的方法进行制备。代替叔丁酯出现在式VIII和IX化合物中的其它的酯可以存在,其仅暂时保护所述酸基团或者其也能够出现在本发明式I的最终化合物中并且能够保留在所述分子中。类似于VI化合物的化合物也能够通过其它转化羰基成为烯烃例如通过Wittig反应的方法得到。In formula VIII, Y can be the group R 5 -SO 2 - which is present in the final compound of formula I of the invention and which can then remain in the molecule, or Y can be the temporarily protected amino group in the 2-position and is removed in a later step to give a free amino group which can then be prepared by standard methods for the preparation of sulfonamides, for example by combining said free amine with the formula R 5 —SO 2 —Cl The sulfonyl chloride reaction converts to the R 5 -SO 2 -NH group. An example of a protecting group representing Y is said benzyloxycarbonyl group (Z group) which can be removed by catalytic hydrogenation. Sulfonyl chlorides and other sulfonic acid derivatives of the formula R 5 -SO 2 -Cl suitable for introducing the group R 5 -SO 2 are commercially available or can be followed or analogously to the procedures described in the literature Prepare. Instead of the tert-butyl ester present in the compounds of the formulas VIII and IX other esters may be present which only temporarily protect the acid group or which can also be present in the final compounds of the formula I according to the invention and can remain in the molecule . Compounds similar to compounds of VI can also be obtained by other methods of converting carbonyls to alkenes, for example by the Wittig reaction.
流程1Process 1
式IX化合物为其中X为甲氧基的式II化合物的实例。这些由上述的合成中得到的和含有活化的羧酸衍生物基团的化合物和类似物能够直接与式III化合物反应。然而,在标准条件下,在以上合成中得到的化合物也能够首先通过裂解所述甲基酯基团或另一个存在于式IX化合物有关的位置上的酯基团转化成为相应的羧酸,其在就地活化后,例如与TOTU或DCCI活化后,或者在转化成为活化的羧酸衍生物之后与所述式III的胍反应。如果打算制备例如作为活化的酸衍生物的羧酰氯衍生物(式II,X=Cl),这种转化能够例如通过使用亚硫酰氯进行。如果打算制备例如来自所述羧酸的所述甲基酯(式II,X=甲氧基),这能够通过用在甲醇中的氯化氢气体处理进行。以本身已知的方法,其它活化的酸衍生物能够从所述羧酰氯或者直接从其中它们被碱化的所述羧酸(式II,X=OH)来制备。实例为其通过以羰基二咪唑(参见Staab,Angew.Chem.Int.Ed.Engl.1,351-367(1962))处理所述酸得到的咪唑(式II,X=1-咪唑基),或者在惰性溶剂中,在胺例如三乙胺存在下,通过与氯甲酸酯例如氯甲酸乙酯反应或与甲苯磺酰氯反应得到的所述混合酸酐。多种适宜于制备活化的羧酸衍生物的方法在J.March, 高等有机化学,第三版,John Wiley & Sons,1985,第350页中的文献中详细指明。Compounds of formula IX are examples of compounds of formula II wherein X is methoxy. These compounds and analogs derived from the above syntheses and containing activated carboxylic acid derivative groups can be reacted directly with compounds of formula III. However, under standard conditions, the compounds obtained in the above syntheses can also be converted firstly to the corresponding carboxylic acids by cleaving the methyl ester group or another ester group present in the relevant position of the compound of formula IX, which The guanidine of formula III is reacted after in situ activation, eg with TOTU or DCCI, or after conversion to an activated carboxylic acid derivative. If one intends to prepare, for example, carboxylic acid chloride derivatives (formula II, X=Cl) as activated acid derivatives, this transformation can be carried out, for example, by using thionyl chloride. If it is intended to prepare, for example, the methyl ester from the carboxylic acid (formula II, X = methoxy), this can be done by treatment with hydrogen chloride gas in methanol. In a manner known per se, other activated acid derivatives can be prepared from the carboxylic acid chlorides or directly from the carboxylic acids (formula II, X=OH) in which they are basified. Examples are their imidazoles (formula II, X = 1-imidazolyl) obtained by treating the acid with carbonyldiimidazole (cf. Staab, Angew. Chem. Int. Ed. Engl. 1, 351-367 (1962)), Or said mixed anhydride obtained by reaction with a chloroformate such as ethyl chloroformate or with toluenesulfonyl chloride in the presence of an amine such as triethylamine in an inert solvent. A number of suitable methods for the preparation of activated carboxylic acid derivatives are specified in J. March, Advanced Organic Chemistry , Third Edition, John Wiley & Sons, 1985, p. 350.
式I化合物为有价值的药用活性成分,其适合例如用于治疗和预防骨障碍、肿瘤疾病或心血管疾病。式I化合物和它们生理学上可耐受的盐和它们的前药作为用于治疗或预防的药剂能够给予动物,优选为哺乳动物并且特别是人。它们能够以它们自身或者与另一种药物的混合物或者以药用制剂的形式给予,该药剂允许肠道或者非肠道给药并且除了常规药用无毒的载体和/或添加剂以外,其含有作为活性成分的有效量的至少一种式I化合物和/或它的生理学上可耐受的盐和/或它的前药。The compounds of the formula I are valuable pharmaceutically active ingredients which are suitable, for example, for the treatment and prophylaxis of bone disorders, neoplastic diseases or cardiovascular diseases. The compounds of the formula I and their physiologically tolerable salts and their prodrugs can be administered to animals, preferably mammals and especially humans, as therapeutic or prophylactic agents. They can be administered by themselves or in admixture with another drug or in the form of pharmaceutical preparations which allow enteral or parenteral administration and which contain, in addition to conventional pharmaceutically acceptable non-toxic carriers and/or additives, An effective amount of at least one compound of the formula I and/or its physiologically tolerable salts and/or its prodrugs as active ingredient.
因此,本发明也涉及用作药剂的式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药,涉及式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药在治疗和预防以上或以下提及的疾病的药剂生产中的用途,例如用于治疗和预防骨疾病,并且也涉及的式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药在治疗和预防这些疾病中的用途。本发明另外涉及除了常规药用无害载体以外,还含有有效量的至少一种式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药的药用制剂或者药用组合物。Accordingly, the present invention also relates to compounds of formula I and/or their physiologically tolerable salts and/or their prodrugs for use as medicaments, to compounds of formula I and/or their physiologically tolerable salts and /or the use of their prodrugs in the production of medicaments for the treatment and prevention of the diseases mentioned above or below, for example for the treatment and prevention of bone diseases, and also to compounds of the formula I and/or their physiological tolerability The use of salts and/or their prodrugs in the treatment and prevention of these diseases. The present invention further relates to pharmaceutical preparations or pharmaceutical preparations containing, in addition to conventional pharmaceutically acceptable carriers, an effective amount of at least one compound of formula I and/or their physiologically tolerable salts and/or their prodrugs combination.
所述药剂能够例如以丸剂、片剂、薄膜片剂、包衣片剂、颗粒剂、硬和软明胶胶囊剂、溶液剂、糖浆剂、乳剂、混悬剂或气溶胶混合物的形式口服给药。然而,给药也能够例如以栓剂的形式直肠给药或者例如以注射溶液剂或输注溶液剂、微囊、植入剂或棒剂的形式进行静脉、肌内或皮下非肠道给药,或者例如以软膏剂、溶液剂或酊剂的形式进行经皮或局部给药,或者例如以气溶胶或鼻喷雾剂的形式以其它方式给药。The medicament can be administered orally, for example, in the form of pills, tablets, film tablets, coated tablets, granules, hard and soft gelatin capsules, solutions, syrups, emulsions, suspensions or aerosol mixtures . However, the administration can also be administered rectally, for example in the form of suppositories or parenterally, for example in the form of injection or infusion solutions, microcapsules, implants or sticks, intravenous, intramuscular or subcutaneous, Alternatively, transdermally or topically, eg, in the form of an ointment, solution or tincture, or otherwise, eg, in the form of an aerosol or nasal spray.
本发明所述药用制剂以本身已知的并且对本领域技术人员是熟悉的方法制备,除了式I化合物和/或它的(它们的)生理学上可耐受的盐和/或它的(它们的)前药以外,使用药用惰性无机载体或有机载体。对丸剂、片剂、包衣片剂和硬明胶胶囊剂的生产而言,使用例如乳糖、玉米淀粉或它们的衍生物、滑石粉、硬脂酸或其盐等是可能的。用于软明胶胶囊剂和栓剂的载体为例如脂肪、蜡、半固体和液体多元醇、天然或硬化油类等。用于生产溶液剂例如注射液或者乳剂或糖浆剂的适宜的载体为例如水、醇类、甘油、多醇类、蔗糖、转化糖、葡萄糖、植物油等。用于微囊剂、植入剂或棒剂的适宜的载体为例如葡糖酸和乳酸的共聚物。所述药用制剂正常含有大约0.5至90%(重量)的式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药。在所述药用制剂中所述式I活性成分和/或它的生理学上可耐受的盐和/或它的前药的量正常为0.2至500mg,优选为1至200mg。The pharmaceutical formulations of the present invention are prepared by methods known per se and familiar to those skilled in the art, except that the compound of formula I and/or its (their) physiologically tolerable salts and/or its (their) In addition to prodrugs, pharmaceutical inert inorganic carriers or organic carriers are used. For the production of pills, tablets, coated tablets and hard gelatine capsules it is possible to use, for example, lactose, corn starch or their derivatives, talc, stearic acid or its salts and the like. Carriers for soft gelatine capsules and suppositories are, for example, fats, waxes, semi-solid and liquid polyols, natural or hardened oils and the like. Suitable carriers for producing solutions such as injections or emulsions or syrups are, for example, water, alcohols, glycerin, polyols, sucrose, invert sugar, glucose, vegetable oils and the like. Suitable carriers for microcapsules, implants or sticks are, for example, copolymers of gluconic and lactic acids. Said pharmaceutical preparations normally contain about 0.5 to 90% by weight of compounds of the formula I and/or their physiologically tolerable salts and/or their prodrugs. The amount of the active ingredient of formula I and/or its physiologically tolerable salt and/or its prodrug in the pharmaceutical preparation is normally 0.2 to 500 mg, preferably 1 to 200 mg.
除了所述活性成分和载体以外,所述药用制剂可另外含有添加剂,例如填充剂、崩解剂、粘合剂、润滑剂、湿润剂、稳定剂、乳化剂、防腐剂、甜味剂、着色剂、矫味剂或芳香剂、增稠剂、稀释剂、缓冲中物质,并且也含有用于得到贮存作用的溶剂或者助溶剂,并且也含有用于改变等渗压力的盐、包衣剂或抗氧化剂。它们也能含有两种或多种式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药。另外,除了至少一种式I化合物和/或它的生理学上可耐受的盐和/或它的前药以外,它们也能含有一种或多种其它的治疗或预防性的活性成分。In addition to the active ingredients and carriers, the pharmaceutical preparations may additionally contain additives such as fillers, disintegrants, binders, lubricants, wetting agents, stabilizers, emulsifiers, preservatives, sweeteners, Coloring agents, flavoring or aromatic agents, thickeners, diluents, buffering substances, and also contain solvents or co-solvents for obtaining storage effects, and also contain salts, coating agents for changing the isotonic pressure or antioxidants. They can also contain two or more compounds of the formula I and/or their physiologically tolerable salts and/or their prodrugs. In addition, besides at least one compound of the formula I and/or its physiologically tolerable salts and/or its prodrugs, they can also contain one or more further therapeutically or prophylactically active ingredients.
式I化合物为玻连蛋白受体拮抗剂和细胞粘附抑制剂。它们具有例如抑制破骨细胞结合于骨表面并且由此通过破骨细胞抑制骨重吸收的能力。例如,式I化合物的作用能够在一种试验中得到证实,在该试验中测定了玻连蛋白结合至含有所述玻连蛋白受体的细胞的抑制作用。以下给出这样试验的细节。作为玻连蛋白受体拮抗剂,式I化合物和它们的生理学上可耐受的盐和它们的前药一般适合于治疗和预防基于细胞-细胞相互作用过程或细胞-基质相互作用过程中玻连蛋白受体与它们的配体之间相互作用的疾病,或者其能够通过抑制这种类型的相互作用,或者用于它们的预防、缓解或治愈所要求的这种类型的相互作用的抑制作用。正如本文开始解释的那样,这样的相互作用例如在骨重吸收、在血管生成或在血管平滑肌细胞增殖中起部分作用。因此,式I化合物和它们的生理学上可耐受的盐和它们的前药适用于例如缓解或治愈至少部分由不合乎要求程度的骨重吸收、血管生成或血管平滑肌细胞增殖引起的疾病。Compounds of formula I are vitronectin receptor antagonists and cell adhesion inhibitors. They have, for example, the ability to inhibit the binding of osteoclasts to the bone surface and thereby inhibit bone resorption by osteoclasts. For example, the action of compounds of the formula I can be demonstrated in an assay in which the inhibition of vitronectin binding to cells containing said vitronectin receptor is determined. Details of such tests are given below. As vitronectin receptor antagonists, the compounds of formula I and their physiologically tolerable salts and their prodrugs are generally suitable for the treatment and prophylaxis of vitronectin-based Diseases in which protein receptors interact with their ligands, or can be inhibited by inhibiting this type of interaction, or the inhibition of this type of interaction is required for their prevention, alleviation or cure. As explained at the outset, such interactions play a part, for example, in bone resorption, in angiogenesis or in proliferation of vascular smooth muscle cells. The compounds of the formula I and their physiologically tolerable salts and their prodrugs are therefore suitable, for example, for alleviating or curing diseases which are caused at least in part by an undesirable degree of bone resorption, angiogenesis or proliferation of vascular smooth muscle cells.
能够使用本发明式I化合物治疗和预防的骨疾病特别为骨质疏松症、高钙血症、例如由转移引起的骨质减少、牙齿疾病、甲状旁腺机能亢进、在类风湿性关节炎和培吉特氏疾病中的关节周的侵蚀。另外,式I化合物能够用于缓解、避免或治疗由糖皮质激素、甾体或肾上腺皮质激素治疗或者由性激素缺乏引起的骨病。所有这些疾病以基于骨形成与骨破坏之间平衡的骨丢失为其特征,并且其能够有利地受抑制经破骨细胞的骨重吸收的影响。在例如治疗或预防骨质疏松症中,在与常规骨质疏松症治疗联合、例如与双膦酸盐、雌激素、雌激素/孕激素、雌激素激动剂/拮抗剂、降钙素、维生素D类似物、甲状旁腺激素、生长激素促分泌素或氟化钠的联合治疗中,式I化合物和/或它的生理学上可耐受的盐和/或它的前药也能够有利地用作骨重吸收抑制剂。式I化合物和/或它的生理学上可耐受的盐和/或它的前药能够和如上列出的在治疗或预防骨质疏松症样疾病中有效的其它的活性成分同时以任何顺序先后地,联合地或分开地连续给予。对在这样的联合治疗或预防中的用途,式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药和一种或多种像那些先前列出的药物那样的其它活性成分能够一起存在于单一药用制剂中,例如片剂和颗粒剂,或者其能够存在于包含在单一包装中或两种或多种分开的包装中的两种或多种分开的药用制剂中。式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药在这样的联合治疗或预防中的用途和它们在生产用于这样联合治疗或预防的药剂中的用途也是本发明的目的。本发明另外涉及包括有效量的至少一种式I化合物和/或它的生理学上可耐受的盐和/或它的前药与至少一种如上列出的药物在治疗或预防骨质疏松症中或者在抑制骨重吸收中有效的其它活性成分和常规药用无害载体的药用制剂。所述以上有关药用制剂的解释相应应用于这样的药用联合制剂中。Bone diseases which can be treated and prevented using the compounds of the formula I according to the invention are in particular osteoporosis, hypercalcemia, e.g. osteopenia caused by metastases, dental diseases, hyperparathyroidism, in rheumatoid arthritis and Periarticular erosions in Paget's disease. In addition, the compounds of formula I can be used to alleviate, avoid or treat bone diseases caused by glucocorticoid, steroid or corticosteroid therapy or by sex hormone deficiency. All of these diseases are characterized by bone loss based on a balance between bone formation and bone destruction, and which can be favorably influenced by the inhibition of bone resorption via osteoclasts. In combination with conventional osteoporosis therapy, e.g. with bisphosphonates, estrogens, estrogens/progestins, estrogen agonists/antagonists, calcitonin, vitamin In the combination therapy of D analogs, parathyroid hormone, growth hormone secretagogue or sodium fluoride, the compound of formula I and/or its physiologically tolerable salt and/or its prodrug can also be advantageously used As a bone resorption inhibitor. The compound of formula I and/or its physiologically tolerable salt and/or its prodrug can be used in any order simultaneously with the other active ingredients listed above that are effective in the treatment or prevention of osteoporosis-like diseases Continuously, jointly or separately. For use in such combined therapy or prophylaxis, the compounds of formula I and/or their physiologically tolerable salts and/or their prodrugs and one or more other drugs like those previously listed The active ingredients can be presented together in a single pharmaceutical formulation, such as tablets and granules, or they can be presented in two or more separate pharmaceutical formulations contained in a single package or in two or more separate packages middle. The use of the compounds of formula I and/or their physiologically tolerable salts and/or their prodrugs in such combination therapy or prevention and their use in the production of medicaments for such combination therapy or prevention is also subject to the present invention. purpose of the invention. The present invention additionally relates to the treatment or prevention of osteoporosis comprising an effective amount of at least one compound of formula I and/or its physiologically tolerable salt and/or its prodrug and at least one of the drugs listed above Pharmaceutical formulations of other active ingredients effective in inhibiting bone resorption and conventional pharmaceutically acceptable carriers. The above explanations regarding pharmaceutical preparations apply correspondingly to such pharmaceutical combination preparations.
除了用作经破骨细胞的骨重吸收抑制剂外,式I化合物和它们的生理学上可耐受的盐和它们的前药能够用作肿瘤生长和肿瘤转移的抑制剂,用作抗炎药物、用于治疗或预防心血管疾病例如动脉硬化或再狭窄,或者用于治疗或预防肾病变或视网膜病例如糖尿病视网膜病。作为肿瘤生长或肿瘤转移的抑制剂,式I化合物和/或它们的生理学上可耐受的盐和/或它们的前药也能够有利地与常规癌症疗法联合用药。在Bertino(编者),Encyclopedia of Cancer,Academic Press,1997中给出常规癌症疗法的实例,其通过引用结合到本文中。所有以上提及的与使用式I化合物与常规骨质疏松症联合治疗的陈述,例如可能的给药模式和药用组合制剂,可相应地应用于式I化合物与常规癌症的联合治疗。In addition to their use as inhibitors of bone resorption by osteoclasts, the compounds of formula I and their physiologically tolerable salts and their prodrugs can be used as inhibitors of tumor growth and tumor metastasis, as anti-inflammatory drugs , for the treatment or prevention of cardiovascular diseases such as arteriosclerosis or restenosis, or for the treatment or prevention of nephropathy or retinopathy such as diabetic retinopathy. As inhibitors of tumor growth or tumor metastasis, the compounds of the formula I and/or their physiologically tolerable salts and/or their prodrugs can also advantageously be administered in combination with conventional cancer therapies. Examples of conventional cancer therapies are given in Bertino (Ed.), Encyclopedia of Cancer, Academic Press, 1997, which is incorporated herein by reference. All statements made above in connection with the use of the compounds of the formula I in combination therapy with conventional osteoporosis, such as possible modes of administration and pharmaceutical combination preparations, apply correspondingly to the combination therapy of the compounds of formula I with conventional cancers.
当使用式I化合物时,剂量在较宽的限制中能够变化并且为常规的,其适宜于在每一个体病例中的个体条件。例如,它依所使用的化合物、依所治疗的疾病的性质和严重性以及依是否治疗或是否进行预防急性或慢性疾病而定。在口服给药情况下,日剂量一般为0.01至100mg/kg,优选为0.1至50mg/kg,特别为0.1至5mg/kg,例如0.3至0.5mg/kg(在每种情况中以每kg体重mg计)在体重大约75kg的成人中取得有效结果。在静脉给药情况中,日剂量一般也为大约0.01至100mg/kg,优选为0.05至10mg/kg(在每种情况中每kg体重)。特别是在相对大量给药的情况中,所述日剂量能够分成几份给药,例如2、3或4份给药。总而言之,依个体行为,对所指明的日剂量向上或向下浮动是必要的。When using compounds of the formula I, the dosage can vary within wide limits and is customary, being adapted to the individual conditions in each individual case. It depends, for example, on the compound used, on the nature and severity of the disease being treated, and on whether acute or chronic disease is being treated or prevented. In the case of oral administration, the daily dosage is generally 0.01 to 100 mg/kg, preferably 0.1 to 50 mg/kg, especially 0.1 to 5 mg/kg, for example 0.3 to 0.5 mg/kg (in each case expressed as per kg body weight mg) effective results were obtained in adults weighing approximately 75 kg. In the case of intravenous administration, the daily dosage is also generally about 0.01 to 100 mg/kg, preferably 0.05 to 10 mg/kg (per kg body weight in each case). Especially in the case of relatively large doses, the daily dose can be divided into several doses, for example 2, 3 or 4 doses. In general, depending on individual behaviour, it may be necessary to vary upwards or downwards from the indicated daily dose.
除了用作药用活性成分以外,式I化合物也能够用作活性成分的媒介或载体,以转运所述活性成分特异性到达所述作用部位(=药物靶向;参见例如 靶向药物传递,R.C.Juliano, 实验药理学手册,第100卷,Ed.Born,G.V.R.等,Springer Verlag,其通过引用结合到本文中)。所转运的活性成分特别为那些能够用于治疗上述疾病的成分。In addition to being used as pharmaceutically active ingredients, the compounds of formula I can also be used as vehicles or carriers for active ingredients to transport them specifically to the site of action (=drug targeting; see e.g. Targeted drug delivery , RCJuliano , Handbook of Experimental Pharmacology , Vol. 100, Ed. Born, GVR et al., Springer Verlag, which is incorporated herein by reference). The active ingredients to be delivered are in particular those which can be used in the treatment of the diseases mentioned above.
式I化合物和它们的盐能够另外用于诊断目的,例如用在体外诊断和作为生物化学研究的辅助工具,其中阻断玻连蛋白受体或者影响细胞-细胞或细胞-基质相互作用是合乎要求的。它们能够另外用作制备其它化合物,特别是其它药用活性成分的合成中间体,这能够例如通过修饰或引入基团或者官能团从式I化合物中得到。The compounds of formula I and their salts can additionally be used for diagnostic purposes, for example in vitro diagnostics and as an aid in biochemical research, where blocking the vitronectin receptor or influencing cell-cell or cell-matrix interactions is desirable of. They can additionally be used as synthesis intermediates for the preparation of other compounds, in particular other pharmaceutically active ingredients, which can be obtained from compounds of the formula I, for example by modifying or introducing groups or functional groups.
实施例Example
所述产物借助质谱(MS)和/或NMR谱鉴定。依最后合成步骤和/或处理方法(例如进行冷冻干燥)而定,在一些情况中,化合物部分或者完全以盐的形式得到,例如以乙酸盐、三氟乙酸盐或者盐酸盐的形式得到。The products are identified by means of mass spectroscopy (MS) and/or NMR spectroscopy. Depending on the last synthetic step and/or the method of processing (e.g. freeze-drying), in some cases the compounds are obtained partly or completely in the form of salts, e.g. as acetates, trifluoroacetates or hydrochlorides get.
实施例1Example 1
(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(Naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzene Formylamino)-propionic acid
a)4-(2-甲氧基羰基-乙烯基)苯甲酸a) 4-(2-methoxycarbonyl-vinyl)benzoic acid
将18.74g(0.12mol)丙二酸一甲酯钾盐悬浮于18ml吡啶中,在室温下及伴随搅拌下加入15.01g(0.1mol)的4-羧基苯甲醛和0.85g(0.01mol)哌啶。将所述混合物回流直到CO2完全逸出(大约2小时),然后加入另外60ml吡啶并在回流下把所述混合物搅拌另外1小时。伴随搅拌下,以500ml冰和110ml浓盐酸处理该反应混合物。加入完毕后,将反应混合物搅拌另外20分钟,并把该产物抽吸过滤,用水洗涤,并从异丙醇中重结晶。产量:12.85g。Suspend 18.74g (0.12mol) potassium monomethyl malonate in 18ml of pyridine, add 15.01g (0.1mol) of 4-carboxybenzaldehyde and 0.85g (0.01mol) of piperidine at room temperature with stirring . The mixture was refluxed until complete CO2 evolution (approximately 2 hours), then another 60 ml of pyridine was added and the mixture was stirred at reflux for another 1 hour. With stirring, the reaction mixture was treated with 500 ml of ice and 110 ml of concentrated hydrochloric acid. After the addition was complete, the reaction mixture was stirred for a further 20 minutes, and the product was filtered off with suction, washed with water and recrystallized from isopropanol. Yield: 12.85g.
1H-NMR(200MHz,d6-DMSO):δ=3.75(s,3H,OCH3),6.76(d,J=15Hz,1H,CHCOOCH3),7.73(d,J=15Hz,1H,Ar-CH),7.84(d,J=9Hz,2H,Ar-H),7.98(d,J=9Hz,2H,Ar-H),13.11(s,宽,1H,COOH)。MS(Cl+):m/e=207.2(M+H+)。 1 H-NMR (200MHz, d 6 -DMSO): δ=3.75 (s, 3H, OCH 3 ), 6.76 (d, J=15Hz, 1H, CHCOOCH 3 ), 7.73 (d, J=15Hz, 1H, Ar -CH), 7.84 (d, J=9Hz, 2H, Ar-H), 7.98 (d, J=9Hz, 2H, Ar-H), 13.11 (s, broad, 1H, COOH). MS (Cl + ): m/e = 207.2 (M+H + ).
HPLC:RP18,Nucleosil 300-5-C18,250×4mm;缓冲液A:H2O,0.1%三氟乙酸(TFA);缓冲液B:乙腈(80%v/v)/H2O(20%v/v),0.1%TFA;梯度:第一个5分钟90%缓冲液A/10%缓冲液B,然后在20分钟期间至90%缓冲液B,然后5分钟90%缓冲液B;流速1ml/分钟;Rt=18.05分钟。HPLC: RP18, Nucleosil 300-5-C18, 250×4mm; Buffer A: H 2 O, 0.1% trifluoroacetic acid (TFA); Buffer B: Acetonitrile (80% v/v)/H 2 O (20 % v/v), 0.1% TFA; Gradient: 90% Buffer A/10% Buffer B for the first 5 minutes, then to 90% Buffer B during 20 minutes, then 90% Buffer B for 5 minutes; Flow rate 1 ml/min; Rt = 18.05 min.
b)4-(2-甲氧基羰基-乙基)苯甲酸b) 4-(2-Methoxycarbonyl-ethyl)benzoic acid
将8g(38.8mmol)的4-(2-甲氧基羰基-乙烯基)苯甲酸悬浮于250ml二氧六环中,并在室温下,在1巴氢气压下经Pd/C(10%浓度)氢化7小时。将所述混合物过滤,并在真空下除去溶剂。产量:8.05g。8 g (38.8 mmol) of 4-(2-methoxycarbonyl-vinyl)benzoic acid were suspended in 250 ml of dioxane and heated over Pd/C (10% conc. ) hydrogenated for 7 hours. The mixture was filtered and the solvent was removed under vacuum. Yield: 8.05 g.
1H-NMR(200MHz,d6-DMSO):δ=2.67(t,J=8Hz,2H,CH2-COOCH3),2.93(t,J=8Hz,2H,Ar-CH2),3.59(s,3H,OCH3),7.35(d,2H,Ar-H),7.86(d,J=9Hz,2H,Ar-H),12.80(s,宽,1H,COOH)。 1 H-NMR (200MHz, d 6 -DMSO): δ = 2.67 (t, J = 8Hz, 2H, CH 2 -COOCH 3 ), 2.93 (t, J = 8Hz, 2H, Ar-CH 2 ), 3.59 ( s, 3H, OCH3 ), 7.35 (d, 2H, Ar-H), 7.86 (d, J=9Hz, 2H, Ar-H), 12.80 (s, broad, 1H, COOH).
MS(Cl+):m/e=209.2(M+H+)。MS (Cl + ): m/e = 209.2 (M+H + ).
HPLC:RP18,Nucleosil 300-5-C18,250×4mm;缓冲液A:H2O,0.1%TFA;缓冲液B:乙腈(80%v/v)/H2O(20%v/v),0.1%TFA;梯度:第一个5分钟90%缓冲液A,10%缓冲液B,然后在20分钟期间至90%缓冲液B,然后5分钟90%缓冲液B;流速1ml/分钟;Rt=17.03分钟。HPLC: RP18, Nucleosil 300-5-C18, 250×4mm; Buffer A: H 2 O, 0.1% TFA; Buffer B: Acetonitrile (80% v/v)/H 2 O (20% v/v) , 0.1% TFA; gradient: first 5 minutes 90% buffer A, 10% buffer B, then to 90% buffer B during 20 minutes, then 90% buffer B for 5 minutes; flow rate 1ml/min; Rt = 17.03 minutes.
c)(2S)-2-苄氧基羰基氨基-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)丙酸叔丁酯c) (2S)-tert-butyl 2-benzyloxycarbonylamino-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)propionate
将354mg(1.7mmol)的4-(2-甲氧基羰基-乙基)苯甲酸和500mg(1.7mmol)的(2S)-3-氨基-2-苄氧基羰基氨基丙酸叔丁酯溶于3ml二甲基甲酰胺中并以557mg(1.7mmol)的O-((氰基-(乙氧基羰基)-亚甲基)氨基)-1,1,3,3-四甲基脲鎓四氟硼酸盐(TOTU)和204mg(1.7mmol)二异丙基乙胺处理,并且在室温下于pH7-8将所述混合物搅拌7小时。真空除去溶剂,将该残余物溶于乙酸乙酯中并将该溶液每次以KHSO4溶液和NaHCO3溶液洗涤3次直至中性。分离有机相并干燥并真空蒸馏除去溶剂。产量:770mg.MS(ES+):m/e=485.2(M+H+)。Dissolve 354mg (1.7mmol) of 4-(2-methoxycarbonyl-ethyl)benzoic acid and 500mg (1.7mmol) of (2S)-3-amino-2-benzyloxycarbonylaminopropionic acid tert-butyl ester In 3ml of dimethylformamide and with 557mg (1.7mmol) of O-((cyano-(ethoxycarbonyl)-methylene)amino)-1,1,3,3-tetramethyluronium Tetrafluoroborate (TOTU) was treated with 204 mg (1.7 mmol) diisopropylethylamine and the mixture was stirred at pH 7-8 at room temperature for 7 hours. The solvent was removed in vacuo, the residue was dissolved in ethyl acetate and the solution was washed 3 times each with KHSO 4 solution and NaHCO 3 solution until neutral. The organic phase is separated off and dried and the solvent is distilled off in vacuo. Yield: 770 mg. MS (ES + ): m/e = 485.2 (M + H + ).
d)(2S)-2-氨基-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯d) (2S)-2-amino-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid tert-butyl ester
将6.88g(14.2mmol)的(2S)-2-苄基氧羰基氨基-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于200ml甲醇中并加入852mg(14.2mmol)乙酸。在室温下,于1巴氢气压下经Pd/C(5%浓度)将所述混合物氢化7小时。将所述混合物过滤并真空除去溶剂。以正庚烷洗涤所述残余物并真空干燥。产量:6.9g。MS(ES+):m/e=351.2(M+H+)。Dissolve 6.88g (14.2mmol) of (2S)-2-benzyloxycarbonylamino-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid tert-butyl ester in 200ml methanol and add 852mg (14.2mmol) acetic acid. The mixture was hydrogenated over Pd/C (5% strength) under 1 bar of hydrogen at room temperature for 7 hours. The mixture was filtered and the solvent was removed in vacuo. The residue was washed with n-heptane and dried in vacuo. Yield: 6.9 g. MS (ES + ): m/e = 351.2 (M+H + ).
e)(2S)-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-2-(萘-1-磺酰基氨基)-丙酸叔丁酯e) (2S)-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-2-(naphthalene-1-sulfonylamino)-propionic acid tert-butyl ester
将0.123g(0.35mmol)的(2S)-2-氨基-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二甲基甲酰胺中,并以0.196ml(1.4mmol)三乙胺和0.159g(0.7mmol)的1-萘磺酰氯处理。在室温下将所述溶液搅拌4小时。真空除去溶剂,使残余物溶于二氯甲烷中并以水将所述溶液洗涤三次。以硫酸钠干燥所述有机相,过滤并且真空除去溶剂。在硅胶柱上将所述残余物层析,以正庚烷/乙酸乙酯(1/1)洗脱。产量:47mg。MS(ES+):m/e=541.3(M+H+)。Dissolve 0.123 g (0.35 mmol) of (2S)-2-amino-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid tert-butyl ester in 2 ml of dimethyl formamide and treated with 0.196 ml (1.4 mmol) of triethylamine and 0.159 g (0.7 mmol) of 1-naphthalenesulfonyl chloride. The solution was stirred at room temperature for 4 hours. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and the solution was washed three times with water. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue was chromatographed on a silica gel column eluting with n-heptane/ethyl acetate (1/1). Yield: 47 mg. MS (ES + ): m/e = 541.3 (M+H + ).
f)(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸叔丁酯f) (2S)-2-(naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl) -Benzoylamino)-tert-butyl propionate
将47mg(0.087mmol)的(2S)-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-2-(萘-1-磺酰基氨基)-丙酸叔丁酯溶于2ml二甲基甲酰胺中并加入86mg(0.87mmol)的2-氨基-1,4,5,6-四氢嘧啶。在室温下将所述反应物搅拌20小时。真空除去溶剂,在硅胶柱上将所述残余物层析,以二氯甲烷/甲醇(1/1)洗脱,随后以二氯甲烷/甲醇/乙酸/水(85/15/1.5/1.5)洗脱。产量:46mg。MS(ES+):m/e=608.3(M+H+)。47 mg (0.087 mmol) of (2S)-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-2-(naphthalene-1-sulfonylamino)-propionic acid tert-butyl The ester was dissolved in 2 ml of dimethylformamide and 86 mg (0.87 mmol) of 2-amino-1,4,5,6-tetrahydropyrimidine was added. The reaction was stirred at room temperature for 20 hours. The solvent was removed in vacuo and the residue was chromatographed on a silica gel column eluting with dichloromethane/methanol (1/1) followed by dichloromethane/methanol/acetic acid/water (85/15/1.5/1.5) elute. Yield: 46 mg. MS (ES + ): m/e = 608.3 (M+H + ).
g)(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸g) (2S)-2-(naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl) -benzoylamino)-propionic acid
将45mg(0.074mmol)的(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二氯甲烷中并加入2ml三氟乙酸。在室温下将所述溶液搅拌3小时。真空除去溶剂并将甲苯加入到所述残余物中并然后真空除去。将所述残余物溶于乙腈/水(1/1)中并且冷冻干燥。产量:50mg。MS(ES+):m/e=552(M+H+)。45 mg (0.074 mmol) of (2S)-2-(naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl )-Ethyl)-benzoylamino)-tert-butyl propionate was dissolved in 2 ml of dichloromethane and 2 ml of trifluoroacetic acid was added. The solution was stirred at room temperature for 3 hours. The solvent was removed in vacuo and toluene was added to the residue and then removed in vacuo. The residue was dissolved in acetonitrile/water (1/1) and lyophilized. Yield: 50 mg. MS (ES + ): m/e = 552 (M+H + ).
实施例2Example 2
(2S)-2-(喹啉-8-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(quinoline-8-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)- Benzoylamino)-propionic acid
a)(2S)-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-2-(喹啉-8-磺酰基氨基)-丙酸叔丁酯a) (2S)-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-2-(quinoline-8-sulfonylamino)-propionic acid tert-butyl ester
将0.123g(0.35mmol)的(2S)-2-氨基-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二甲基甲酰胺中,并以0.196ml(1.4mmol)三乙胺和0.159g(0.7mmol)的8-喹啉磺酰氯处理。在室温下,将所述溶液搅拌4小时。真空除去溶剂,把所述残余物溶于二氯甲烷中并以水把所述溶液洗涤三次。以硫酸钠干燥所述有机相,过滤并且真空除去溶剂。在硅胶柱上将所述残余物层析,以正庚烷/乙酸乙酯(1/1)洗脱,产量:67mg。MS(ES+):m/e=542.2(M+H+)。Dissolve 0.123 g (0.35 mmol) of (2S)-2-amino-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid tert-butyl ester in 2 ml of dimethyl formamide and treated with 0.196 ml (1.4 mmol) of triethylamine and 0.159 g (0.7 mmol) of 8-quinolinesulfonyl chloride. The solution was stirred at room temperature for 4 hours. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and the solution was washed three times with water. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue was chromatographed on a silica gel column, eluting with n-heptane/ethyl acetate (1/1), yield: 67 mg. MS (ES + ): m/e = 542.2 (M+H + ).
b)(2S)-2-(喹啉-8-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸叔丁酯b) (2S)-2-(quinoline-8-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-tert-butyl propionate
将65mg(0.12mmol)的(2S)-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-2-(喹啉-8-磺酰基氨基)-丙酸叔丁酯溶于1ml二甲基甲酰胺中并加入59mg(0.6mmol)的2-氨基-1,4,5,6-四氢嘧啶。在室温下将所述反应物搅拌20小时。真空除去溶剂,在硅胶柱上将所述残余物层析,以二氯甲烷/甲醇(1/1)洗脱,随后以二氯甲烷/甲醇/乙酸/水(85/15/1.5/1.5)洗脱。产量:72mg。MS(ES+):m/e=609.3(M+H+)。65 mg (0.12 mmol) of (2S)-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-2-(quinoline-8-sulfonylamino)-propionic acid tert The butyl ester was dissolved in 1 ml of dimethylformamide and 59 mg (0.6 mmol) of 2-amino-1,4,5,6-tetrahydropyrimidine was added. The reaction was stirred at room temperature for 20 hours. The solvent was removed in vacuo and the residue was chromatographed on a silica gel column eluting with dichloromethane/methanol (1/1) followed by dichloromethane/methanol/acetic acid/water (85/15/1.5/1.5) elute. Yield: 72 mg. MS (ES + ): m/e = 609.3 (M+H + ).
c)(2S)-2-(喹啉-8-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸c) (2S)-2-(quinoline-8-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-propionic acid
将72mg(0.11mmol)的(2S)-2-(喹啉-8-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二氯甲烷中并加入2ml三氟乙酸。在室温下将所述溶液搅拌3小时。真空除去溶剂并将甲苯加入到所述残余物中并然后真空除去。将所述残余物溶于乙腈/水(1/1)中并且冷冻干燥。产量:54mg。MS(ES+):m/e=553(M+H+)。72 mg (0.11 mmol) of (2S)-2-(quinoline-8-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylaminomethyl) Acyl)-ethyl)-benzoylamino)-propionic acid tert-butyl ester was dissolved in 2 ml of dichloromethane and 2 ml of trifluoroacetic acid was added. The solution was stirred at room temperature for 3 hours. The solvent was removed in vacuo and toluene was added to the residue and then removed in vacuo. The residue was dissolved in acetonitrile/water (1/1) and lyophilized. Yield: 54 mg. MS (ES + ): m/e = 553 (M+H + ).
实施例3Example 3
(2S)-2-(4-乙酰氨基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(4-Acetamido-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl) -benzoylamino)-propionic acid
a)(2S)-2-(4-乙酰氨基-苯磺酰基氨基)-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯a) (2S)-2-(4-acetylamino-benzenesulfonylamino)-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid tert-butyl ester
将0.123g(0.35mmol)的(2S)-2-氨基-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二甲基甲酰胺中,并以0.196ml(1.4mmol)三乙胺和0.164g(0.7mmol)的4-乙酰氨基-苯磺酰氯处理。在室温下,将所述溶液搅拌4小时。真空除去溶剂,把所述残余物溶于二氯甲烷中并以水将所述溶液洗涤三次。以硫酸钠干燥所述有机相,过滤并且真空除去溶剂。在硅胶柱上将所述残余物层析,以正庚烷/乙酸乙酯(1/2)洗脱,产量:90mg。MS(ES+):m/e=548(M+H+)。Dissolve 0.123 g (0.35 mmol) of (2S)-2-amino-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid tert-butyl ester in 2 ml of dimethyl formamide and treated with 0.196 ml (1.4 mmol) of triethylamine and 0.164 g (0.7 mmol) of 4-acetamido-benzenesulfonyl chloride. The solution was stirred at room temperature for 4 hours. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and the solution was washed three times with water. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue was chromatographed on a silica gel column, eluting with n-heptane/ethyl acetate (1/2), yield: 90 mg. MS (ES + ): m/e = 548 (M+H + ).
b)(2S)-2-(4-乙酰氨基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸叔丁酯b) (2S)-2-(4-acetylamino-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl base)-benzoylamino)-tert-butyl propionate
将87mg(0.16mmol)的(2S)-2-(4-乙酰氨基-苯磺酰基氨基)-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于1ml二甲基甲酰胺中并加入79mg(0.8mmol)的2-氨基-1,4,5,6-四氢嘧啶。在室温下将所述反应物搅拌20小时。真空除去溶剂,在硅胶柱上将所述残余物层析,以二氯甲烷/甲醇(1/1)洗脱,随后以二氯甲烷/甲醇/乙酸/水(85/15/1.5/1.5)洗脱。产量:57mg。MS(ES+):m/e=615.3(M+H+)。87 mg (0.16 mmol) of (2S)-2-(4-acetylamino-benzenesulfonylamino)-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid The tert-butyl ester was dissolved in 1 ml of dimethylformamide and 79 mg (0.8 mmol) of 2-amino-1,4,5,6-tetrahydropyrimidine was added. The reaction was stirred at room temperature for 20 hours. The solvent was removed in vacuo and the residue was chromatographed on a silica gel column eluting with dichloromethane/methanol (1/1) followed by dichloromethane/methanol/acetic acid/water (85/15/1.5/1.5) elute. Yield: 57 mg. MS (ES + ): m/e = 615.3 (M+H + ).
c)(2S)-2-(4-乙酰氨基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸c) (2S)-2-(4-acetylamino-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl base)-benzoylamino)-propionic acid
将57mg(0.09mmol)的(2S)-2-(4-乙酰氨基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二氯甲烷中并加入2ml三氟乙酸。在室温下将所述溶液搅拌3小时。真空除去溶剂并将甲苯加入到所述残余物中并然后真空除去。将所述残余物溶于乙腈/水(1/1)中并且冷冻干燥。产量:42mg。MS(ES+):m/e=559.2(M+H+)。57mg (0.09mmol) of (2S)-2-(4-acetylamino-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylamino Formyl)-ethyl)-benzoylamino)-propionic acid tert-butyl ester was dissolved in 2 ml of dichloromethane and 2 ml of trifluoroacetic acid was added. The solution was stirred at room temperature for 3 hours. The solvent was removed in vacuo and toluene was added to the residue and then removed in vacuo. The residue was dissolved in acetonitrile/water (1/1) and lyophilized. Yield: 42 mg. MS (ES + ): m/e = 559.2 (M+H + ).
实施例4Example 4
(2S)-2-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-benzenesulfonylamino)-3-(4-(2-(1,4,5 , 6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-propionic acid
a)2-((1S)-1-叔丁氧基羰基-2-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-乙基氨磺酰基)-苯甲酸甲酯a) 2-((1S)-1-tert-butoxycarbonyl-2-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-ethylsulfamoyl)-benzoic acid methyl ester
将0.123g(0.35mmol)的(2S)-2-氨基-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二甲基甲酰胺中,并以0.196ml(1.4mmol)三乙胺和0.164g(0.7mmol)的2-(甲氧基羰基)-苯磺酰氯处理。在室温下,将所述溶液搅拌4小时。真空除去溶剂,把所述残余物溶于二氯甲烷中并以水将所述溶液洗涤三次。以硫酸钠干燥所述有机相,过滤并且真空除去溶剂。在硅胶柱上将所述残余物层析,以正庚烷/乙酸乙酯(1/1)洗脱,产量:75mg。MS(ES+):m/e=549(M+H+)。Dissolve 0.123 g (0.35 mmol) of (2S)-2-amino-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid tert-butyl ester in 2 ml of dimethyl formamide and treated with 0.196 ml (1.4 mmol) of triethylamine and 0.164 g (0.7 mmol) of 2-(methoxycarbonyl)-benzenesulfonyl chloride. The solution was stirred at room temperature for 4 hours. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and the solution was washed three times with water. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue was chromatographed on a silica gel column, eluting with n-heptane/ethyl acetate (1/1), yield: 75 mg. MS (ES + ): m/e = 549 (M+H + ).
b)(2S)-2-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸叔丁酯b) (2S)-2-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-benzenesulfonylamino)-3-(4-(2-(1,4 , 5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-propionic acid tert-butyl ester
将75mg(0.13mmol)的2-((1S)-1-叔丁氧基羰基-2-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-乙基氨磺酰基)-苯甲酸甲酯溶于1ml二甲基甲酰胺中并加入68mg(0.69mmol)的2-氨基-1,4,5,6-四氢嘧啶。在室温下将所述反应物搅拌20小时。真空除去溶剂,在硅胶柱上将所述残余物层析,以二氯甲烷/甲醇(1/1)洗脱,随后以二氯甲烷/甲醇/乙酸/水(85/15/1.5/1.5)洗脱。产量:54mg。MS(ES+):m/e=683.3(M+H+)。75 mg (0.13 mmol) of 2-((1S)-1-tert-butoxycarbonyl-2-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-ethylsulfamoyl )-methyl benzoate was dissolved in 1 ml dimethylformamide and 68 mg (0.69 mmol) of 2-amino-1,4,5,6-tetrahydropyrimidine was added. The reaction was stirred at room temperature for 20 hours. The solvent was removed in vacuo and the residue was chromatographed on a silica gel column eluting with dichloromethane/methanol (1/1) followed by dichloromethane/methanol/acetic acid/water (85/15/1.5/1.5) elute. Yield: 54 mg. MS (ES + ): m/e = 683.3 (M+H + ).
c)(2S)-2-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸c) (2S)-2-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-benzenesulfonylamino)-3-(4-(2-(1,4 , 5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-propionic acid
将54mg(0.08mmol)的(2S)-2-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二氯甲烷中并加入2ml三氟乙酸。在室温下将所述溶液搅拌3小时。真空除去溶剂并将甲苯加入到所述残余物中并然后真空除去。将所述残余物溶于乙腈/水(1/1)中并且冷冻干燥。产量:34mg。MS(ES+):m/e=627(M+H+)。54 mg (0.08 mmol) of (2S)-2-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-benzenesulfonylamino)-3-(4-(2 -(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-tert-butyl propionate was dissolved in 2ml of dichloromethane and added 2ml of trifluoroacetic acid . The solution was stirred at room temperature for 3 hours. The solvent was removed in vacuo and toluene was added to the residue and then removed in vacuo. The residue was dissolved in acetonitrile/water (1/1) and lyophilized. Yield: 34 mg. MS (ES + ): m/e = 627 (M+H + ).
通法1(实施例5至24的化合物的合成)General Method 1 (Synthesis of Compounds of Examples 5 to 24)
步骤a:与磺酰氯的反应Step a: Reaction with Sulfonyl Chloride
将0.2g的(2S)-2-氨基-3-(4-(2-甲氧基羰基-乙基)-苯甲酰基氨基)-丙酸叔丁酯溶于2ml二甲基甲酰胺中,并以4摩尔当量三乙胺和2摩尔当量适当的磺酰氯处理。在室温下,将所述溶液搅拌4小时。真空除去溶剂,把所述残余物溶于二氯甲烷中并以水将所述溶液洗涤三次。以硫酸钠干燥所述有机相,过滤并且真空除去溶剂。在硅胶上将所述残余物层析,以正庚烷/乙酸乙酯(1/1)洗脱。Dissolve 0.2 g of (2S)-2-amino-3-(4-(2-methoxycarbonyl-ethyl)-benzoylamino)-propionic acid tert-butyl ester in 2 ml of dimethylformamide, and treated with 4 molar equivalents of triethylamine and 2 molar equivalents of the appropriate sulfonyl chloride. The solution was stirred at room temperature for 4 hours. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and the solution was washed three times with water. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue is chromatographed on silica gel eluting with n-heptane/ethyl acetate (1/1).
步骤b:酰基胍的形成Step b: Formation of acylguanidine
将步骤a的产物溶于1ml二甲基甲酰胺中并加入5摩尔当量2-氨基-1,4,5,6-四氢嘧啶。在室温下将所述反应混合物搅拌20小时。真空除去溶剂,在硅胶上将所述残余物层析,以二氯甲烷/甲醇(1/1)洗脱,随后以二氯甲烷/甲醇/乙酸/水(85/15/1.5/1.5)洗脱。The product of step a was dissolved in 1 ml of dimethylformamide and 5 molar equivalents of 2-amino-1,4,5,6-tetrahydropyrimidine were added. The reaction mixture was stirred at room temperature for 20 hours. The solvent was removed in vacuo and the residue was chromatographed on silica gel eluting with dichloromethane/methanol (1/1) followed by dichloromethane/methanol/acetic acid/water (85/15/1.5/1.5) take off.
步骤c:叔丁酯的裂解Step c: Cleavage of tert-butyl ester
将步骤b的产物溶于2ml二氯甲烷中并加入2ml三氟乙酸。在室温下将所述溶液搅拌3小时。真空除去溶剂并将甲苯加入到所述残余物中并然后真空除去。将所述残余物溶于乙腈/水(1/1)中并且冷冻干燥。The product of step b was dissolved in 2 ml of dichloromethane and 2 ml of trifluoroacetic acid was added. The solution was stirred at room temperature for 3 hours. The solvent was removed in vacuo and toluene was added to the residue and then removed in vacuo. The residue was dissolved in acetonitrile/water (1/1) and lyophilized.
实施例5Example 5
(2S)-2-(4-叔丁基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(4-tert-butyl-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-propionic acid
使用步骤a中的4-叔丁基苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 4-tert-butylbenzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 83mg 547.2(M+H)+ Product of step a 83mg 547.2(M+H) +
步骤b的产物 85mg 614.3(M+H)+ Product of step b 85mg 614.3(M+H) +
步骤c的产物(标题化合物) 75mg 558.3(M+H)+ Product of step c (title compound) 75mg 558.3(M+H) +
实施例6Example 6
(2S)-2-(丙烷-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(Propane-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzene Formylamino)-propionic acid
使用步骤a中的丙烷-1-磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using propane-1-sulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 54mg 457.3(M+H)+ Product of step a 54mg 457.3(M+H) +
步骤b的产物 55mg 524.3(M+H)+ Product of step b 55mg 524.3(M+H) +
步骤c的产物(标题化合物) 45mg 468.3(M+H)+ Product of step c (title compound) 45 mg 468.3 (M+H) +
实施例7Example 7
(2S)-2-(2-苯基-乙烷磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(2-Phenyl-ethanesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-propionic acid
使用步骤a中的反式-β-苯乙烯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using trans-β-styrenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 49mg 517.1(M+H)+ Product of step a 49mg 517.1(M+H) +
步骤b的产物 45mg 584.3(M+H)+ Product of step b 45mg 584.3(M+H) +
步骤c的产物(标题化合物) 37mg 528.3(M+H)+ Product of step c (title compound) 37mg 528.3(M+H) +
实施例8Example 8
(2S)-2-(4-丙基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(4-Propyl-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl) -benzoylamino)-propionic acid
使用步骤a中的4-(正丙基)-苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 4-(n-propyl)-benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 78mg 533.2(M+H)+ Product of step a 78mg 533.2(M+H) +
步骤b的产物 73mg 600.4(M+H)+ Product of step b 73mg 600.4(M+H) +
步骤c的产物(标题化合物) 55mg 544.3(M+H)+ Product of step c (title compound) 55 mg 544.3 (M+H) +
实施例9Example 9
(2S)-2-(2-甲基-丙烷-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(2-Methyl-propane-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)- Ethyl)-benzoylamino)-propionic acid
使用步骤a中的2-甲基-丙烷-1-磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 2-methyl-propane-1-sulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 21mg 471.3(M+H)+ Product of step a 21mg 471.3(M+H) +
步骤b的产物 10mg 538.4(M+H)+ Product of step b 10mg 538.4(M+H) +
步骤c的产物(标题化合物) 13mg 482.3(M+H)+ Product of step c (title compound) 13 mg 482.3 (M+H) +
实施例10Example 10
(2S)-2-(4-丁氧基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(4-Butoxy-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-propionic acid
使用步骤a中的4-(正丁氧基)-苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Method 1 using 4-(n-butoxy)-benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 83mg 563.2(M+H)+ Product of step a 83mg 563.2(M+H) +
步骤b的产物 83mg 630.4(M+H)+ Product of step b 83mg 630.4(M+H) +
步骤c的产物(标题化合物) 63mg 574.3(M+H)+ Product of step c (title compound) 63mg 574.3(M+H) +
实施例11Example 11
(2S)-2-(2-氰基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(2-cyano-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl) -benzoylamino)-propionic acid
使用步骤a中的2-氰基-苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 2-cyano-benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 91mg 516.2(M+H)+ Product of step a 91mg 516.2(M+H) +
步骤b的产物 71mg 583.3(M+H)+ Product of step b 71mg 583.3(M+H) +
步骤c的产物(标题化合物) 73mg 527.3(M+H)+ Product of step c (title compound) 73mg 527.3(M+H) +
实施例12Example 12
(2S)-2-(7,7-二甲基-2-氧代-二环[2.2.1]庚-1-基甲磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(7,7-Dimethyl-2-oxo-bicyclo[2.2.1]hept-1-ylmethylsulfonylamino)-3-(4-(2-(1,4 , 5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-propionic acid
使用步骤a中的10-樟脑磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Method 1 using 10-camphorsulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 85mg 565.3(M+H)+ Product of step a 85mg 565.3(M+H) +
步骤b的产物 88mg 632.4(M+H)+ Product of step b 88mg 632.4(M+H) +
步骤c的产物(标题化合物) 71mg 576.4(M+H)+ Product of step c (title compound) 71 mg 576.4 (M+H) +
实施例13Example 13
(2S)-2-(4-氯-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(4-Chloro-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)- Benzoylamino)-propionic acid
使用步骤a中的4-氯-苯磺酰氯,按照通法1,合成所述标题化合物。The title compound was synthesized following General Method 1 using 4-chloro-benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 91mg 525.1(M+H)+ Product of step a 91mg 525.1(M+H) +
步骤b的产物 82mg 592.3(M+H)+ Product of step b 82mg 592.3(M+H) +
步骤c的产物(标题化合物) 64mg 536.3(M+H)+ Product of step c (title compound) 64mg 536.3(M+H) +
实施例14Example 14
(2S)-2-(3-氯-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(3-Chloro-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)- Benzoylamino)-propionic acid
使用步骤a中的3-氯-苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 3-chloro-benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 65mg 525.2(M+H)+ Product of step a 65mg 525.2(M+H) +
步骤b的产物 58mg 592.3(M+H)+ Product of step b 58mg 592.3(M+H) +
步骤c的产物(标题化合物) 52mg 536.3(M+H)+ Product of step c (title compound) 52mg 536.3(M+H) +
实施例15Example 15
(2S)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-2-(3-三氟甲基-苯磺酰基氨基)-丙酸(2S)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-2-(3-trifluoro Methyl-benzenesulfonylamino)-propionic acid
使用步骤a中的3-三氟甲基-苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 3-trifluoromethyl-benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 86mg 559.2(M+H)+ Product of step a 86mg 559.2(M+H) +
步骤b的产物 94mg 626.3(M+H)+ Product of step b 94mg 626.3(M+H) +
步骤c的产物(标题化合物) 84mg 570.3(M+H)+ Product of step c (title compound) 84mg 570.3(M+H) +
实施例16Example 16
(2S)-2-(4-甲氧基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(4-methoxy-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-propionic acid
使用步骤a中的4-甲氧基-苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 4-methoxy-benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 66mg 521.1(M+H)+ Product of step a 66mg 521.1(M+H) +
步骤b的产物 71mg 588.3(M+H)+ Product of step b 71mg 588.3(M+H) +
步骤c的产物(标题化合物) 49mg 532.3(M+H)+ Product of step c (title compound) 49 mg 532.3 (M+H) +
实施例17Example 17
(2S)-2-苯磺酰基氨基-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-Benzenesulfonylamino-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)- propionic acid
使用步骤a中的苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 76mg 491.2(M+H)+ Product of step a 76mg 491.2(M+H) +
步骤b的产物 77mg 558.3(M+H)+ Product of step b 77mg 558.3(M+H) +
步骤c的产物(标题化合物) 64mg 502.3(M+H)+ Product of step c (title compound) 64mg 502.3(M+H) +
实施例18Example 18
(2S)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-2-(噻吩-2-磺酰基氨基)-丙酸(2S)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-2-(thiophene-2- Sulfonylamino)-propionic acid
使用步骤a中的2-噻吩磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Method 1 using 2-thiophenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 87mg 497.1(M+H)+ Product of step a 87mg 497.1(M+H) +
步骤b的产物 74mg 564.2(M+H)+ Product of step b 74mg 564.2(M+H) +
步骤c的产物(标题化合物) 64mg 508.2(M+H)+ Product of step c (title compound) 64mg 508.2(M+H) +
实施例19(2S)-2-(联苯基-4-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸Example 19 (2S)-2-(biphenyl-4-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)- Ethyl)-benzoylamino)-propionic acid
使用步骤a中的4-联苯基磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Method 1 using 4-biphenylsulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 39mg 567.1(M+H)+ Product of step a 39mg 567.1(M+H) +
步骤b的产物 40mg 634.4(M+H)+ Product of step b 40mg 634.4(M+H) +
步骤c的产物(标题化合物) 33mg 578.3(M+H)+ Product of step c (title compound) 33mg 578.3(M+H) +
实施例20Example 20
(2S)-2-(萘-2-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(Naphthalene-2-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzene Formylamino)-propionic acid
使用步骤a中的2-萘磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 2-naphthalenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 76mg 541.2(M+H)+ Product of step a 76mg 541.2(M+H) +
步骤b的产物 74mg 608.3(M+H)+ Product of step b 74mg 608.3(M+H) +
步骤c的产物(标题化合物) 62mg 552.3(M+H)+ Product of step c (title compound) 62mg 552.3(M+H) +
实施例21Example 21
(2S)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-2-(2,4,6-三甲基-苯磺酰基氨基)-丙酸(2S)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-2-(2,4, 6-Trimethyl-benzenesulfonylamino)-propionic acid
使用步骤a中的2,4,6-三甲基苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 2,4,6-trimethylbenzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 68mg 533.2(M+H)+ Product of step a 68mg 533.2(M+H) +
步骤b的产物 71mg 600.4(M+H)+ Product of step b 71mg 600.4(M+H) +
步骤c的产物(标题化合物) 54mg 544.3(M+H)+ Product of step c (title compound) 54mg 544.3(M+H) +
实施例22Example 22
(2S)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-2-(4-三氟甲基-苯磺酰基氨基)-丙酸(2S)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-2-(4-trifluoro Methyl-benzenesulfonylamino)-propionic acid
使用步骤a中的4-三氟甲基-苯磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using 4-trifluoromethyl-benzenesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 105mg 559.3(M+H)+ Product of step a 105mg 559.3(M+H) +
步骤b的产物 93mg 626.4(M+H)+ Product of step b 93mg 626.4(M+H) +
步骤c的产物(标题化合物) 70mg 570.3(M+H)+ Product of step c (title compound) 70mg 570.3(M+H) +
实施例23Example 23
(2S)-2-(丁烷-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(Butane-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)- Benzoylamino)-propionic acid
使用步骤a中的丁烷-1-磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using butane-1-sulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 87mg 471.4(M+H)+ Product of step a 87mg 471.4(M+H) +
步骤b的产物 60mg 538.4(M+H)+ Product of step b 60mg 538.4(M+H) +
步骤c的产物(标题化合物) 57mg 482.3(M+H)+ Product of step c (title compound) 57mg 482.3(M+H) +
实施例24Example 24
(2S)-2-甲磺酰基氨基-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-Methanesulfonylamino-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)- propionic acid
使用步骤a中的甲磺酰氯,按照通法1,合成标题化合物。The title compound was synthesized following General Procedure 1 using methanesulfonyl chloride in step a.
产量 MS(ES+):m/eYield MS(ES + ): m/e
步骤a的产物 87.8mg 429.3(M+H)+ Product of step a 87.8mg 429.3(M+H) +
步骤b的产物 98mg 496.4(M+H)+ Product of step b 98mg 496.4(M+H) +
步骤c的产物(标题化合物) 74mg 440.3(M+H)+ Product of step c (title compound) 74mg 440.3(M+H) +
通法2(实施例25至27的化合物的合成)General Method 2 (Synthesis of Compounds of Examples 25 to 27)
步骤a:与磺酰氯的反应Step a: Reaction with Sulfonyl Chloride
将0.1g的(2S)-2-氨基-3-(4-(2-羧基-乙基)-苯甲酰基氨基)-丙酸叔丁酯盐酸盐溶于3ml二甲基甲酰胺中,并在0℃下以3摩尔当量二异丙基乙基胺和2摩尔当量适当的磺酰氯处理。在0℃下,将所述溶液搅拌3小时。通过加入乙酸乙酯稀释反应混合物,溶液以硫酸氢钾水溶液洗涤两次和以饱和氯化钠水溶液洗涤一次。以硫酸镁干燥所述有机相,过滤并真空除去溶剂。在硅胶上将所述残余物层析,以二氯甲烷/甲醇/乙酸/水(97.5/2.5/0.25/0.25)洗脱。0.1 g of (2S)-2-amino-3-(4-(2-carboxy-ethyl)-benzoylamino)-propionic acid tert-butyl ester hydrochloride was dissolved in 3 ml of dimethylformamide, and treated with 3 molar equivalents of diisopropylethylamine and 2 molar equivalents of the appropriate sulfonyl chloride at 0°C. The solution was stirred at 0°C for 3 hours. The reaction mixture was diluted by adding ethyl acetate, and the solution was washed twice with aqueous potassium hydrogensulfate and once with saturated aqueous sodium chloride. The organic phase was dried over magnesium sulfate, filtered and the solvent was removed in vacuo. The residue was chromatographed on silica gel eluting with dichloromethane/methanol/acetic acid/water (97.5/2.5/0.25/0.25).
步骤b:酰基胍的形成Step b: Formation of acylguanidine
将步骤a的产物溶于2ml四氢呋喃中并加入1.2摩尔当量2-氨基-1,4,5,6-四氢嘧啶、4摩尔当量二异丙基乙基胺和1.1摩尔当量O-(7-氮杂苯并三唑-1-基)-1,1,3,3-四甲基脲鎓六氟磷酸盐。在室温下将所述反应物搅拌20小时。真空除去溶剂并将所述残余物溶于乙酸乙酯中,并且所述溶液以饱和碳酸氢钠水溶液洗涤两次和以饱和氯化钠水溶液洗涤一次。以硫酸镁干燥所述有机相,过滤并且真空除去溶剂。在硅胶上将所述残余物层析,以二氯甲烷/甲醇/乙酸/水(85/15/1.5/1.5)洗脱,随后以二氯甲烷/甲醇/乙酸/水(9/1/0.1/0.1)洗脱。The product of step a was dissolved in 2 ml tetrahydrofuran and 1.2 molar equivalents of 2-amino-1,4,5,6-tetrahydropyrimidine, 4 molar equivalents of diisopropylethylamine and 1.1 molar equivalents of O-(7- Azabenzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate. The reaction was stirred at room temperature for 20 hours. The solvent was removed in vacuo and the residue was dissolved in ethyl acetate, and the solution was washed twice with saturated aqueous sodium bicarbonate and once with saturated aqueous sodium chloride. The organic phase was dried over magnesium sulfate, filtered and the solvent was removed in vacuo. The residue was chromatographed on silica gel eluting with dichloromethane/methanol/acetic acid/water (85/15/1.5/1.5) followed by dichloromethane/methanol/acetic acid/water (9/1/0.1 /0.1) elution.
步骤c:叔丁酯酯的裂解Step c: Cleavage of tert-butyl ester
将所述步骤b的产物溶于1.5ml三氟乙酸/水(95/5)中。在室温下将所述溶液搅拌1小时。真空除去溶剂并将甲苯加入到所述残余物中并然后真空除去。将所述残余物溶于乙腈/水(1/1)中并且冷冻干燥。The product of step b was dissolved in 1.5 ml trifluoroacetic acid/water (95/5). The solution was stirred at room temperature for 1 hour. The solvent was removed in vacuo and toluene was added to the residue and then removed in vacuo. The residue was dissolved in acetonitrile/water (1/1) and lyophilized.
实施例25Example 25
(2S)-2-(丙烷-2-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-(propane-2-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzene Formylamino)-propionic acid
使用步骤a中的丙烷-2-磺酰氯,按照通法2,合成标题化合物。The title compound was synthesized following General Method 2 using propane-2-sulfonyl chloride in step a.
产量 MS:m/e MS: m/e
步骤a的产物 17mg 443.2(M+H)+(FAB+)Product of step a 17mg 443.2(M+H) + (FAB + )
步骤b的产物 7mg 524.2(M+H)+(ES+)Product of step b 7mg 524.2(M+H) + (ES + )
步骤c的产物(标题化合物) 7.2mg 468.2(M+H)+(ES+)Product of step c (title compound) 7.2 mg 468.2 (M+H) + (ES + )
实施例26Example 26
(2S)-2-氯甲磺酰基氨基-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸(2S)-2-Chloromethylsulfonylamino-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino) -propionic acid
使用步骤a中的氯甲磺酰氯,按照通法2,合成标题化合物。The title compound was synthesized following General Method 2 using chloromethanesulfonyl chloride in step a.
产量 MS:m/e MS: m/e
步骤a的产物 94mg 449.1(M+H)+(FAB+)Product of step a 94mg 449.1(M+H) + (FAB + )
步骤b的产物 28mg 530.2(M)+(ES+)Product of step b 28mg 530.2(M) + (ES + )
步骤c的产物(标题化合物) 34mg 474.2(M+H)+(ES+)Product of step c (title compound) 34 mg 474.2 (M+H) + (ES + )
实施例27Example 27
(2S)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-2-(2,2,2-三氟-乙磺酰基氨基)-丙酸(2S)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzoylamino)-2-(2,2, 2-Trifluoro-ethanesulfonylamino)-propionic acid
使用步骤a中的2,2,2-三氟乙磺酰氯,按照通法2,合成标题化合物。The title compound was synthesized following General Method 2 using 2,2,2-trifluoroethanesulfonyl chloride in step a.
产量 MS:m/e MS: m/e
步骤a的产物 43mg 483.2(M+H)+(FAB+)Product of step a 43mg 483.2(M+H) + (FAB + )
步骤b的产物 24mg 564.2(M+H)+(ES+)Product of step b 24mg 564.2(M+H) + (ES + )
步骤c的产物(标题化合物) 22mg 508.2(M+H)+(ES+)Product of step c (title compound) 22 mg 508.2 (M+H) + (ES + )
实施例28Example 28
(2S)-2-(4-叔丁基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸乙酯(2S)-2-(4-tert-butyl-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-propionic acid ethyl ester
将700mg(2S)-2-(4-叔丁基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸溶于100ml乙醇中并加入15滴浓硫酸。将所述反应溶液沸腾3.5小时。真空除去溶剂,把所述残余物溶于二氯甲烷中并以饱和碳酸氢钠水溶液洗涤三次。以二氯甲烷将所述水相提取一次并以饱和氯化钠水溶液洗涤所述合并的有机相两次。以硫酸钠干燥所述有机相,过滤并真空除去溶剂。将所述残余物溶于2N盐酸水溶液中。真空除去盐酸,把所述残余物溶于乙腈中并加入水。将该混合物冷冻干燥。产量:480mg。MS(ES+):m/e=586.4(M+H)+。700mg of (2S)-2-(4-tert-butyl-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)- Ethyl)-benzoylamino)-propionic acid was dissolved in 100 ml ethanol and 15 drops of concentrated sulfuric acid were added. The reaction solution was boiled for 3.5 hours. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and washed three times with saturated aqueous sodium bicarbonate. The aqueous phase was extracted once with dichloromethane and the combined organic phases were washed twice with saturated aqueous sodium chloride. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue was dissolved in 2N aqueous hydrochloric acid. The hydrochloric acid was removed in vacuo, the residue was dissolved in acetonitrile and water was added. The mixture was lyophilized. Yield: 480mg. MS (ES + ): m/e = 586.4 (M+H) + .
实施例29Example 29
(2S)-2-(4-叔丁基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸异丙酯(2S)-2-(4-tert-butyl-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-isopropyl propionate
将700mg(2S)-2-(4-叔丁基-苯磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸溶于100ml异丙醇中并加入15滴浓硫酸。将所述反应溶液沸腾2.5天。真空除去溶剂,把所述残余物溶于二氯甲烷中并以饱和碳酸氢钠水溶液洗涤三次。以二氯甲烷将所述水相提取一次并以饱和氯化钠水溶液洗涤所述合并的有机相两次。以硫酸钠干燥所述有机相,过滤并真空除去溶剂。将所述残余物溶于2N盐酸水溶液中。真空除去盐酸,把所述残余物溶于乙腈中并加入水。将该混合物冷冻干燥。产量:444mg。MS(ES+):m/e=600.4(M+H)+。700mg of (2S)-2-(4-tert-butyl-benzenesulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)- Ethyl)-benzoylamino)-propionic acid was dissolved in 100 ml of isopropanol and 15 drops of concentrated sulfuric acid were added. The reaction solution was boiled for 2.5 days. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and washed three times with saturated aqueous sodium bicarbonate. The aqueous phase was extracted once with dichloromethane and the combined organic phases were washed twice with saturated aqueous sodium chloride. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue was dissolved in 2N aqueous hydrochloric acid. The hydrochloric acid was removed in vacuo, the residue was dissolved in acetonitrile and water was added. The mixture was lyophilized. Yield: 444mg. MS (ES + ): m/e = 600.4 (M+H) + .
实施例30Example 30
(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸乙酯(2S)-2-(Naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzene Formylamino)-ethyl propionate
将590mg(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸溶于80ml乙醇中并加入12滴浓硫酸。将所述反应溶液沸腾3小时。真空除去溶剂,把所述残余物溶于二氯甲烷中并以饱和碳酸氢钠水溶液洗涤三次。以二氯甲烷将所述水相提取一次并以饱和氯化钠水溶液洗涤所述合并的有机相两次。以硫酸钠干燥所述有机相,过滤并真空除去溶剂。将所述残余物溶于2N盐酸水溶液中。真空除去盐酸,把所述残余物溶于乙腈中并加入水。将该混合物冷冻干燥。产量:381mg。MS(ES+):m/e=580.3(M+H)+。590 mg of (2S)-2-(naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl) -Benzoylamino)-propionic acid was dissolved in 80 ml of ethanol and 12 drops of concentrated sulfuric acid were added. The reaction solution was boiled for 3 hours. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and washed three times with saturated aqueous sodium bicarbonate. The aqueous phase was extracted once with dichloromethane and the combined organic phases were washed twice with saturated aqueous sodium chloride. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue was dissolved in 2N aqueous hydrochloric acid. The hydrochloric acid was removed in vacuo, the residue was dissolved in acetonitrile and water was added. The mixture was lyophilized. Yield: 381 mg. MS (ES + ): m/e = 580.3 (M+H) + .
实施例31Example 31
(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸异丙酯(2S)-2-(Naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzene Formylamino)-isopropyl propionate
将1.5g(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸溶于250ml异丙醇中并加入1ml浓硫酸。将所述反应溶液沸腾3天。真空除去溶剂,把所述残余物溶于二氯甲烷中并以饱和碳酸氢钠水溶液洗涤三次。以二氯甲烷将所述水相提取一次并以饱和氯化钠水溶液洗涤所述合并的有机相两次。以硫酸钠干燥所述有机相,过滤并真空除去溶剂。将所述残余物溶于2N盐酸水溶液中。真空除去盐酸,把所述残余物溶于乙腈中并加入水。将该混合物冷冻干燥。产量:950mg。MS(ES+):m/e=594.4(M+H)+。1.5g (2S)-2-(naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl )-benzoylamino)-propionic acid was dissolved in 250 ml of isopropanol and 1 ml of concentrated sulfuric acid was added. The reaction solution was boiled for 3 days. The solvent was removed in vacuo, the residue was dissolved in dichloromethane and washed three times with saturated aqueous sodium bicarbonate. The aqueous phase was extracted once with dichloromethane and the combined organic phases were washed twice with saturated aqueous sodium chloride. The organic phase was dried over sodium sulfate, filtered and the solvent was removed in vacuo. The residue was dissolved in 2N aqueous hydrochloric acid. The hydrochloric acid was removed in vacuo, the residue was dissolved in acetonitrile and water was added. The mixture was lyophilized. Yield: 950mg. MS (ES + ): m/e = 594.4 (M+H) + .
实施例32Example 32
(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸异丁酯(2S)-2-(Naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl)-benzene Formylamino)-isobutyl propionate
将600mg(2S)-2-(萘-1-磺酰基氨基)-3-(4-(2-(1,4,5,6-四氢嘧啶-2-基氨基甲酰基)-乙基)-苯甲酰基氨基)-丙酸溶于12ml异丁醇中并加入0.1ml浓硫酸。将所述反应溶液沸腾24小时。真空除去溶剂,并在硅胶上将所述残余物层析,以二氯甲烷/甲醇/乙酸/水(9/1/0.1/0.1)洗脱。将所述产物溶于乙酸/水中并冷冻干燥。产量:250mg。MS(ES+):m/e=608.5(M+H)+。600mg of (2S)-2-(naphthalene-1-sulfonylamino)-3-(4-(2-(1,4,5,6-tetrahydropyrimidin-2-ylcarbamoyl)-ethyl) -Benzoylamino)-propionic acid was dissolved in 12 ml of isobutanol and 0.1 ml of concentrated sulfuric acid was added. The reaction solution was boiled for 24 hours. The solvent was removed in vacuo and the residue was chromatographed on silica gel eluting with dichloromethane/methanol/acetic acid/water (9/1/0.1/0.1). The product was dissolved in acetic acid/water and lyophilized. Yield: 250 mg. MS (ES + ): m/e = 608.5 (M+H) + .
药理试验pharmacological test
例如,借助例如类似于WO-A-95/32710的破骨细胞重吸收试验(“PIT ASSAY”),能够测定本发明化合物对骨重吸收的抑制作用,其通过引用结合到本文中。For example, the inhibitory effect of compounds of the invention on bone resorption can be determined by means of, for example, an osteoclast resorption assay ("PIT ASSAY") similar to WO-A-95/32710, which is incorporated herein by reference.
例如,如同以下所描述的那样,能够测定本发明化合物抑制玻连蛋白受体αvβ3的抑制作用。For example, compounds of the invention can be assayed for inhibition of vitronectin receptor [alpha] v [beta] 3 as described below.
用于测量结合于人玻连蛋白的293细胞的抑制作用试验(Vn/293细胞试验)293 Cell Inhibition Assay for Measuring Binding to Human Vitronectin (Vn/293 Cell Assay)
1.人玻连蛋白的纯化1. Purification of Human Vitronectin
从人的血浆中分离人玻连蛋白并按照Yatohyo等的方法中的亲合层析法纯化( 细胞结构与功能,1988,23,281-292)。Human vitronectin was isolated from human plasma and purified by affinity chromatography according to the method of Yatohyo et al. ( Cell Structure and Function , 1988, 23, 281-292).
2.细胞试验2. Cell Assay
按照所述FACS方法,选择以DNA序列共转染用于玻连蛋白受体αvβ3的αv和β3亚单位的293细胞(人胚肾细胞系)的用于高比例表达(>500,000αvβ3受体/细胞)。培养所选择的细胞并借助FACS再次分选,以得到具有表达比例为每细胞>1,000,000模板的αvβ3的稳定的细胞系(15D)。Following the FACS method, 293 cells (human embryonic kidney cell line) co-transfected with DNA sequences for the αv and β3 subunits of the vitronectin receptor αvβ3 were selected for high ratio expression (> 500,000 α v β 3 receptors/cell). Selected cells were cultured and re-sorted by FACS to obtain a stable cell line (15D) with expression of αvβ3 at a ratio of >1,000,000 template per cell.
在4℃下,以在磷酸盐缓冲的盐水溶液(PBS)中的人玻连蛋白(0.01mg/ml,0.05ml/孔)将具有平底的Linbro 96孔组织培养板包被过夜,然后以0.5%浓度的BSA(小牛血清白蛋白)阻断。制备10-10mol/l至2×10-3mol/l的所述受试物质在含有葡萄糖的DMEM培养液中的溶液,并且将0.05ml/孔溶液加入到在每一种情况下的板上。将表达为高水平的αvβ3(例如15D)的细胞悬浮于含有葡萄糖的DMEM培养液中,并将所述悬浮液调整到内容物为25,000个细胞/0.05ml培养液。将该细胞悬浮液0.05ml加入到每孔中,并在37℃下将所述板孵育90分钟。以温热的PBS将所述板洗涤三次,以除去未结合的细胞。在含有0.25%Triton X-100的枸橼酸缓冲液(25mM,pH5.0)中冷冻所述结合的细胞。然后加入己糖酰胺酶底物对硝基苯基-N-乙酰基-β-D-氨基葡糖苷,并在37℃下将所述板孵育90分钟。以甘氨酸(50mM)/EDTA(5mM)缓冲液(pH10.4)终止所述反应,并且在405-650nm下测量每孔的吸收。按照标准方法分析所述数据。Linbro 96-well tissue culture plates with flat bottoms were coated overnight at 4°C with human vitronectin (0.01 mg/ml, 0.05 ml/well) in phosphate-buffered saline (PBS), and then incubated with 0.5 % concentration of BSA (bovine serum albumin) blocking. 10 -10 mol/l to 2 x 10 -3 mol/l solutions of the test substance in DMEM culture fluid containing glucose were prepared, and 0.05 ml/well of the solution was added to the plate in each case superior. Cells expressing high levels of α v β 3 (eg, 15D) were suspended in DMEM culture fluid containing glucose, and the suspension was adjusted to a content of 25,000 cells/0.05 ml of culture fluid. 0.05 ml of this cell suspension was added to each well, and the plate was incubated at 37°C for 90 minutes. The plates were washed three times with warm PBS to remove unbound cells. The bound cells were frozen in citrate buffer (25 mM, pH 5.0) containing 0.25% Triton X-100. The hexosamidase substrate p-nitrophenyl-N-acetyl-β-D-glucosaminidase was then added and the plate was incubated at 37°C for 90 minutes. The reaction was stopped with glycine (50 mM)/EDTA (5 mM) buffer (pH 10.4) and the absorbance of each well was measured at 405-650 nm. The data were analyzed according to standard methods.
得到以下所述试验结果(抑制浓度IC50)。The test results (inhibitory concentration IC 50 ) described below were obtained.
实施例编号 IC50Vn/293细胞试验Example number IC 50 Vn/293 cell test
1 6nM1 6nM
2 52nM2 52nM
3 27nM3 27nM
5 18nM5 18nM
6 32nM6 32nM
7 28nM7 28nM
8 20nM8 20nM
9 69nM9 69nM
10 29nM10 29nM
11 24nM11 24nM
12 27nM12 27nM
13 27nM13 27nM
14 10nM14 10nM
15 6nM15 6nM
16 26nM16 26nM
17 9nM17 9nM
18 14nM18 14nM
19 38nM19 38nM
20 5nM20 5nM
21 21nM21 21nM
22 10nM22 10nM
23 30nM23 30nM
24 100nM24 100nM
Claims (16)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US1248998A | 1998-01-23 | 1998-01-23 | |
| US09/012,489 | 1998-01-23 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1293662A CN1293662A (en) | 2001-05-02 |
| CN1177832C true CN1177832C (en) | 2004-12-01 |
Family
ID=21755208
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB998040894A Expired - Fee Related CN1177832C (en) | 1998-01-23 | 1999-01-16 | Novel sulfonamide derivatives as bone resorption inhibitors and as cell adhesion inhibitors |
Country Status (25)
| Country | Link |
|---|---|
| EP (1) | EP1049677A1 (en) |
| JP (1) | JP2002501054A (en) |
| KR (1) | KR20010034319A (en) |
| CN (1) | CN1177832C (en) |
| AP (1) | AP1269A (en) |
| AR (1) | AR014456A1 (en) |
| AU (1) | AU752882B2 (en) |
| BG (1) | BG104630A (en) |
| BR (1) | BR9907735A (en) |
| CA (1) | CA2318221A1 (en) |
| EA (1) | EA003102B1 (en) |
| HR (1) | HRP20000493A2 (en) |
| HU (1) | HUP0100520A3 (en) |
| ID (1) | ID26219A (en) |
| IL (1) | IL137423A0 (en) |
| NO (1) | NO318795B1 (en) |
| NZ (1) | NZ505613A (en) |
| PL (1) | PL341871A1 (en) |
| SK (1) | SK10632000A3 (en) |
| TR (1) | TR200002160T2 (en) |
| TW (1) | TWI247742B (en) |
| UA (1) | UA63990C2 (en) |
| WO (1) | WO1999037621A1 (en) |
| YU (1) | YU47200A (en) |
| ZA (1) | ZA99476B (en) |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000006169A1 (en) * | 1998-07-29 | 2000-02-10 | Merck & Co., Inc. | Integrin receptor antagonists |
| EP1028114A1 (en) | 1999-02-13 | 2000-08-16 | Aventis Pharma Deutschland GmbH | Novel guanidine derivatives as inhibitors of cell adhesion |
| EP1065207A1 (en) | 1999-07-02 | 2001-01-03 | Aventis Pharma Deutschland GmbH | Naphthyridine derivatives, processes for their preparation, their use, and pharmaceutical compositions comprising them |
| EP1065208A1 (en) * | 1999-07-02 | 2001-01-03 | Aventis Pharma Deutschland GmbH | Substituted purine derivatives as inhibitors of cell adhesion |
| EP1070707A1 (en) | 1999-07-21 | 2001-01-24 | Aventis Pharma Deutschland GmbH | 1,4,5,6-tetrahydropyrimidine derivative as a vitronectin inhibitor |
| US6849639B2 (en) | 1999-12-14 | 2005-02-01 | Amgen Inc. | Integrin inhibitors and their methods of use |
| EP1108721A1 (en) | 1999-12-15 | 2001-06-20 | Aventis Pharma Deutschland GmbH | Thienylalanine derivatives as inhibitors of cell adhesion |
| US20030199571A1 (en) * | 1999-12-24 | 2003-10-23 | Gordon Bruton | (Hetero) Bicyclymethanesulfonylamino-substituted hydroxamic acid derivatives |
| FR2808798A1 (en) | 2000-05-09 | 2001-11-16 | Hoechst Marion Roussel Inc | New N-heterocyclyl-aminoacid derivatives and analogs, are vitronectin analogs useful e.g. for treating osteoporosis, tumor growth or metastasis, inflammation or cardiovascular disease |
| FR2847254B1 (en) | 2002-11-19 | 2005-01-28 | Aventis Pharma Sa | NOVEL VITRONECTIN RECEPTOR ANTAGONIST DERIVATIVES, PROCESS FOR PREPARING THEM, THEIR APPLICATION AS MEDICAMENTS AND THE PHARMACEUTICAL COMPOSITIONS REFLECTING THEM |
| EP1727811B1 (en) | 2004-03-24 | 2014-11-12 | Shire Orphan Therapies GmbH | New compounds for the inhibition of angiogenesis and use of thereof |
| FR2870541B1 (en) | 2004-05-18 | 2006-07-14 | Proskelia Sas | ANTIGONISTIC PYRIMIDINE DERIVATIVES OF VITRONECTIN RECEPTOR |
| GB0412553D0 (en) * | 2004-06-04 | 2004-07-07 | Univ Aberdeen | Therapeutic agents for the treatment of bone conditions |
| UA87854C2 (en) | 2004-06-07 | 2009-08-25 | Мерк Энд Ко., Инк. | N-(2-benzyl)-2-phenylbutanamides as androgen receptor modulators |
| FR2873585B1 (en) * | 2004-07-27 | 2006-11-17 | Aventis Pharma Sa | NEW GALENIC FORMULATIONS OF ACTIVE PRINCIPLES |
| GB0705400D0 (en) | 2007-03-21 | 2007-05-02 | Univ Aberdeen | Therapeutic compounds andm their use |
| GB0817207D0 (en) | 2008-09-19 | 2008-10-29 | Pimco 2664 Ltd | therapeutic apsac compounds and their use |
| GB0817208D0 (en) | 2008-09-19 | 2008-10-29 | Pimco 2664 Ltd | Therapeutic apsap compounds and their use |
| GB201311361D0 (en) | 2013-06-26 | 2013-08-14 | Pimco 2664 Ltd | Compounds and their therapeutic use |
| ES2864079T3 (en) | 2014-05-30 | 2021-10-13 | Pfizer | Carbonitrile derivatives as selective androgen receptor modulators |
| CA2970578C (en) | 2014-12-17 | 2024-01-02 | Pimco 2664 Limited | N-(4-hydroxy-4-methyl-cyclohexyl)-4-phenyl-benzenesulfonamide and n-(4-hydroxy-4-methyl-cyclohexyl)-4-(2-pyridyl)-benzenesulfonamide compounds and their therapeutic use |
| WO2023275715A1 (en) | 2021-06-30 | 2023-01-05 | Pfizer Inc. | Metabolites of selective androgen receptor modulators |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10501222A (en) * | 1994-05-27 | 1998-02-03 | メルク エンド カンパニー インコーポレーテッド | Compounds for inhibiting osteoclast-mediated bone resorption |
| JP3895792B2 (en) * | 1995-12-08 | 2007-03-22 | プロスケリア・エス・ア・エス | Bone formation promoter |
-
1999
- 1999-01-16 IL IL13742399A patent/IL137423A0/en unknown
- 1999-01-16 AU AU25181/99A patent/AU752882B2/en not_active Ceased
- 1999-01-16 SK SK1063-2000A patent/SK10632000A3/en unknown
- 1999-01-16 KR KR1020007008041A patent/KR20010034319A/en not_active Abandoned
- 1999-01-16 ID IDW20001407A patent/ID26219A/en unknown
- 1999-01-16 CA CA002318221A patent/CA2318221A1/en not_active Abandoned
- 1999-01-16 CN CNB998040894A patent/CN1177832C/en not_active Expired - Fee Related
- 1999-01-16 EA EA200000785A patent/EA003102B1/en not_active IP Right Cessation
- 1999-01-16 HR HR20000493A patent/HRP20000493A2/en not_active Application Discontinuation
- 1999-01-16 AP APAP/P/2000/001863A patent/AP1269A/en active
- 1999-01-16 TR TR2000/02160T patent/TR200002160T2/en unknown
- 1999-01-16 NZ NZ505613A patent/NZ505613A/en unknown
- 1999-01-16 JP JP2000528545A patent/JP2002501054A/en not_active Abandoned
- 1999-01-16 BR BR9907735-3A patent/BR9907735A/en not_active IP Right Cessation
- 1999-01-16 WO PCT/EP1999/000242 patent/WO1999037621A1/en not_active Ceased
- 1999-01-16 YU YU47200A patent/YU47200A/en unknown
- 1999-01-16 EP EP99904789A patent/EP1049677A1/en not_active Withdrawn
- 1999-01-16 PL PL99341871A patent/PL341871A1/en unknown
- 1999-01-16 UA UA2000084983A patent/UA63990C2/en unknown
- 1999-01-16 HU HU0100520A patent/HUP0100520A3/en unknown
- 1999-01-22 ZA ZA9900476A patent/ZA99476B/en unknown
- 1999-01-29 AR ARP990100244A patent/AR014456A1/en not_active Application Discontinuation
- 1999-04-30 TW TW088100862A patent/TWI247742B/en active
-
2000
- 2000-07-21 NO NO20003765A patent/NO318795B1/en unknown
- 2000-07-21 BG BG104630A patent/BG104630A/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| ID26219A (en) | 2000-12-07 |
| SK10632000A3 (en) | 2001-02-12 |
| PL341871A1 (en) | 2001-05-07 |
| ZA99476B (en) | 1999-08-05 |
| EP1049677A1 (en) | 2000-11-08 |
| YU47200A (en) | 2002-11-15 |
| IL137423A0 (en) | 2001-07-24 |
| UA63990C2 (en) | 2004-02-16 |
| AP1269A (en) | 2004-04-03 |
| AU752882B2 (en) | 2002-10-03 |
| AP2000001863A0 (en) | 2000-09-30 |
| AR014456A1 (en) | 2001-02-28 |
| BG104630A (en) | 2001-04-30 |
| TWI247742B (en) | 2006-01-21 |
| WO1999037621A1 (en) | 1999-07-29 |
| CA2318221A1 (en) | 1999-07-29 |
| CN1293662A (en) | 2001-05-02 |
| KR20010034319A (en) | 2001-04-25 |
| HUP0100520A1 (en) | 2001-07-30 |
| HUP0100520A3 (en) | 2002-11-28 |
| NO20003765D0 (en) | 2000-07-21 |
| BR9907735A (en) | 2000-10-17 |
| EA003102B1 (en) | 2002-12-26 |
| JP2002501054A (en) | 2002-01-15 |
| HRP20000493A2 (en) | 2001-06-30 |
| EA200000785A1 (en) | 2001-02-26 |
| AU2518199A (en) | 1999-08-09 |
| NO318795B1 (en) | 2005-05-09 |
| TR200002160T2 (en) | 2001-07-23 |
| NZ505613A (en) | 2002-11-26 |
| NO20003765L (en) | 2000-09-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1177832C (en) | Novel sulfonamide derivatives as bone resorption inhibitors and as cell adhesion inhibitors | |
| CN1205191C (en) | Novel acylguanidine derivatives as bone resorption inhibitors and vitronectin receptor antagonists | |
| CN1158279C (en) | 1-(p-thienylbenzyl)imidazoles as angiotensin (1-7) receptor agonists, processes for their preparation, uses thereof and pharmaceutical preparations containing them | |
| CN1057292C (en) | Bicyclic compounds, pharmaceutical compositions containing them and uses thereof | |
| CN1296346C (en) | Acylated 6,7,8,9-tetrahydro-5H-benzocycloheptenyl amines and their use as pharmaceutical | |
| CN1214026C (en) | Indol-3-yl derivatives | |
| CN1306509A (en) | Substituted benzamides, their prodn. and use as cysteine protease inhibitors | |
| CN1390196A (en) | Novel derivatives of dicarboxylic acid having pharmaceutical properties | |
| CN1107467A (en) | 1-Benzenesulfonyl-1,3-dihydro-indol-2-one derivatives, their preparation and pharmaceutical compositions containing them | |
| CN1807426A (en) | Heteroaryl urea neuropeptide y y5 receptor antagonists | |
| CN1688553A (en) | Chemical process | |
| CN1694879A (en) | Isoquinoline derivatives having kinasae inhibitory activity and drugs containing the same | |
| CN1688577A (en) | Triazaspiro [5.5] undecane derivatives and drugs comprising the same as the active ingredient | |
| CN1788001A (en) | 2,4-Bis(anilino)pyrimidines useful in the treatment of neoplastic diseases, inflammatory and immune system disorders | |
| CN1291978A (en) | Meta-azacyclic amino benzoic acid compound and derivatives thereof being integrin antagonists | |
| CN1193623A (en) | Substituted purine derivatives, their preparation methods, applications and compositions containing them | |
| CN1036566A (en) | 1,3,4,5-Tetrahydrobenzo[c,d]ole compounds | |
| CN1215055C (en) | Arylsulfonylamino substituted hydroxamic acid derivatives | |
| CN1138322A (en) | 2,3-Diaminopropionic acid derivatives | |
| CN1300281A (en) | Compounds with growth hormone releasing properties | |
| CN1298705C (en) | Novel piperidine-4-sulfamide compound, its preparing method and medicinal composition containing them | |
| CN1171888C (en) | Substituted purine derivatives, preparation method and application | |
| CN1053063A (en) | Quinazoline derivatives and preparation methods thereof | |
| CN1571781A (en) | 4-imidazolin-2-one compounds | |
| CN1218467A (en) | Sulphur derivatives comprising amide bond, method for preparing same, use thereof as drugs, and pharmaceutical compositions containing such derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C19 | Lapse of patent right due to non-payment of the annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |