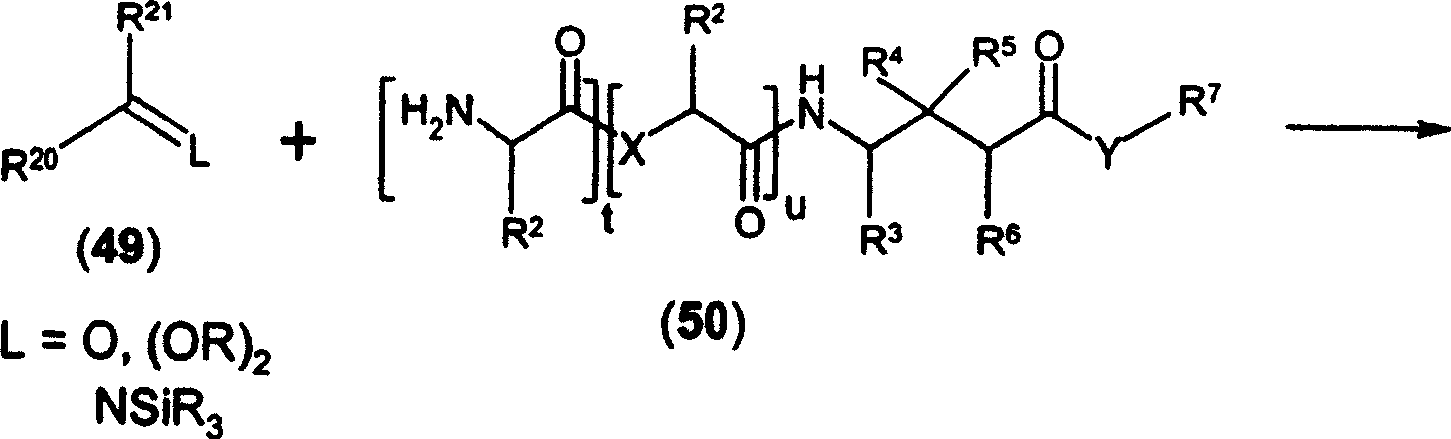
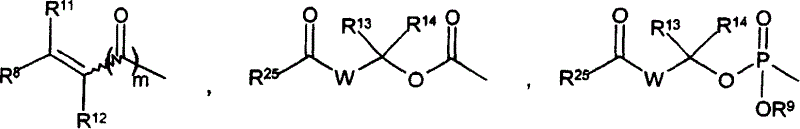
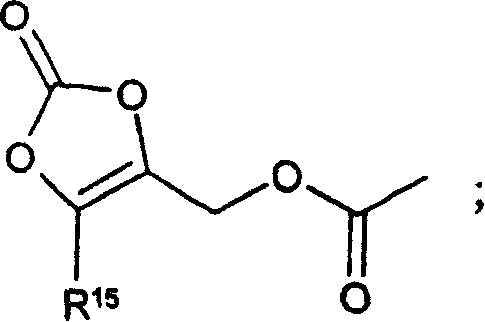
CN1753673A - Prodrugs of gaba analogs, compositions and uses thereof - Google Patents
Prodrugs of gaba analogs, compositions and uses thereof Download PDFInfo
- Publication number
- CN1753673A CN1753673A CN02814572.0A CN02814572A CN1753673A CN 1753673 A CN1753673 A CN 1753673A CN 02814572 A CN02814572 A CN 02814572A CN 1753673 A CN1753673 A CN 1753673A
- Authority
- CN
- China
- Prior art keywords
- substituted
- butyl
- dioxan
- benzyl
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Landscapes
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
本申请要求35 U.S.C.§119(e)要求2001年6月11日申请的美国临时申请60/297,521、2001年6月14日申请的美国临时申请60/298,514和2002年3月19日申请的美国临时申请60/366,090的利益,本文引用这些献作为参考。This application claims 35 U.S.C. §119(e) requirements of U.S. Provisional Application 60/297,521 filed June 11, 2001, U.S. Provisional Application 60/298,514 filed June 14, 2001, and U.S. Provisional Application 60/298,514 filed March 19, 2002 These documents are hereby incorporated by reference for the benefit of provisional application 60/366,090.
1. 发明领域 1. Field of invention
本发明一般涉及GABA类似物的前药,GABA类似物前药的药物组合物,GABA类似物的前药的制备方法,GABA类似物前药和GABA类似物前药的药物组合物的应用方法。更为具体地,本发明涉及加巴喷丁和pregabalin的前药,加巴喷丁和pregabalin的前药的药物组合物,加巴喷丁和pregabalin的前药的制备方法,加巴喷丁和pregabalin的前药和加巴喷丁和pregabalin的前药的药物组合物的应用方法。The present invention generally relates to prodrugs of GABA analogs, pharmaceutical compositions of GABA analog prodrugs, preparation methods of GABA analog prodrugs, application methods of GABA analog prodrugs and GABA analog prodrugs. More specifically, the present invention relates to the prodrug of gabapentin and pregabalin, the pharmaceutical composition of the prodrug of gabapentin and pregabalin, the preparation method of the prodrug of gabapentin and pregabalin, the prodrug of gabapentin and pregabalin and the prodrug of gabapentin and pregabalin Methods of application of the pharmaceutical composition.
2. 发明背景 2. Background of the invention
Gamma(″γ″)-氨基丁酸(″GABA″)是哺乳动物中枢神经系统中的一种主要抑制性递质。GABA没有从血流中被有效地转运到脑(即GABA没有有效地跨过血-脑屏障)。因此,脑细胞实质上提供了所有的在脑中发现的GABA(BABA是通过谷氨酸与磷酸吡哆醛的脱羧作用而生物合成的)。Gamma ("γ")-aminobutyric acid ("GABA") is a major inhibitory transmitter in the mammalian central nervous system. GABA is not efficiently transported from the bloodstream to the brain (ie GABA does not efficiently cross the blood-brain barrier). Thus, brain cells provide virtually all of the GABA found in the brain (BABA is biosynthesized by the decarboxylation of glutamate with pyridoxal phosphate).
GABA通过与特定的膜蛋白(即GABAA受体)结合而调节神经元的兴奋性,从而导致离子通道开放。氯化物离子进入并通过离子通道导致受体细胞的超极化,从而防止将神经脉冲传递到其它细胞。已在癫痫发作、运动疾病(如多发性硬化、动作性震颤、迟发性运动障碍)、恐慌、焦虑、抑郁、酒精中毒和躁狂行为)的个体中观察到低水平的GABA。GABA regulates neuronal excitability by binding to specific membrane proteins (i.e., GABAA receptors), resulting in ion channel opening. The entry of chloride ions into and through the ion channel causes hyperpolarization of the recipient cell, preventing the transmission of nerve impulses to other cells. Low levels of GABA have been observed in individuals with epileptic seizures, movement disorders (eg, multiple sclerosis, action tremor, tardive dyskinesia), panic, anxiety, depression, alcoholism, and manic behavior.
关于多种常见病症和/或常见医学疾病的低GABA水平的提示激发了对具有优于GABA的药学性能(例如透过血脑屏障的能力)的GABA类似物制备的浓厚兴趣。因此,已在本领域中合成了多种具有重要的药学性能的GABA类似物(例如参见Satzinger等人,美国专利4,024,175;Silverman等人,美国专利5,563,175;Horwell等人,美国专利6,020,370;Silverman等人,美国专利6,028,214;Horwell等人,美国专利6,103,932;Silverman等人,美国专利6,117,906;Silverman,国际公开WO 92/09560;Silverman等人,国际公开WO93/23383;Horwell等人,国际公开WO 97/29101,HORWELL等人,国际公开WO 97/33858;Horwell等人,国际公开WO 97/33859;Bryans等人,国际公开WO 98/17627;Guglietta等人,国际公开WO 99/08671;Bryans等人,国际公开WO 99/21824;Bryans等人,国际公开WO99/31057;Belliotti等人,国际公开WO 99/31074;Bryans等人,国际公开WO 99/31075;Bryans等人,国际公开WO 99/61424;Bryans等人,国际公开WO 00/15611;Bryans,国际公开WO 00/31020;Bryans等人,The suggestion of low GABA levels in many common conditions and/or common medical diseases has stimulated intense interest in the preparation of GABA analogs with superior pharmaceutical properties to GABA, such as the ability to cross the blood-brain barrier. Accordingly, a variety of GABA analogs with important pharmaceutical properties have been synthesized in the art (see, for example, Satzinger et al., U.S. Patent 4,024,175; Silverman et al., U.S. Patent 5,563,175; Horwell et al., U.S. Patent 6,020,370; Silverman et al. , U.S. Patent 6,028,214; Horwell et al., U.S. Patent 6,103,932; Silverman et al., U.S. Patent 6,117,906; Silverman, International Publication WO 92/09560; Silverman et al., International Publication WO 93/23383; Horwell et al., International Publication WO 97/29101 , HORWELL et al., International Publication WO 97/33858; Horwell et al., International Publication WO 97/33859; Bryans et al., International Publication WO 98/17627; Guglietta et al., International Publication WO 99/08671; Publication WO 99/21824; Bryans et al. International Publication WO 99/31057; Belliotti et al. International Publication WO 99/31074; Bryans et al. International Publication WO 99/31075; Bryans et al. International Publication WO 99/61424; et al., International Publication WO 00/15611; Bryans, International Publication WO 00/31020; Bryans et al.,
国际公开WO 00/50027;和Bryans等人,国际公开WO 02/00209)。International Publication WO 00/50027; and Bryans et al., International Publication WO 02/00209).
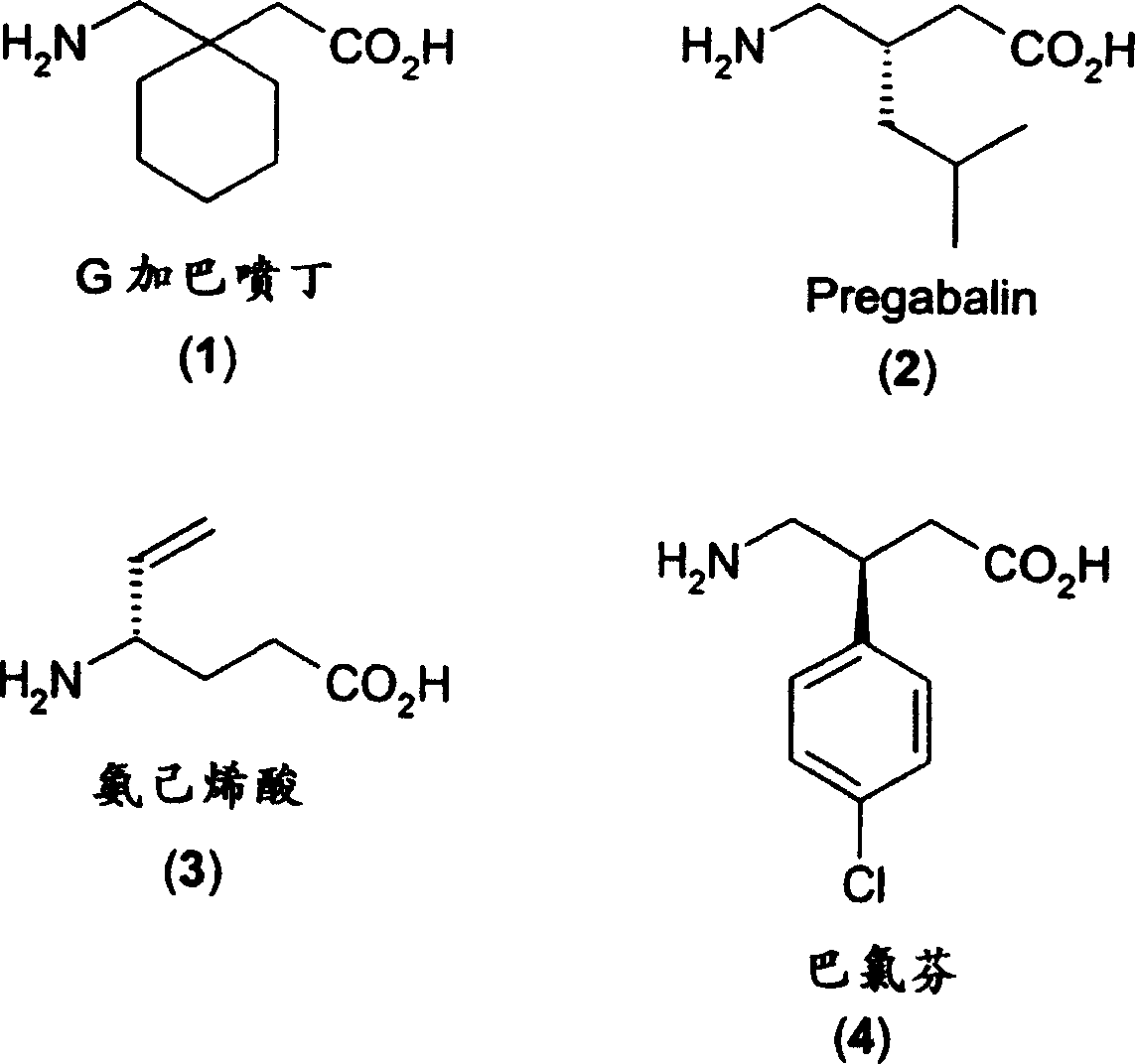
例如药学上重要的GABA类似物包括以上所示的加巴喷丁(1)、pregabalin(2)、氨己烯酸(3)和巴氯芬(4)。加巴喷丁是一种可以通过血脑屏障的亲脂性GABA类似物,自1994年以来它展用于临床治疗癫痫。加巴喷丁还可能具有对以下疾病的有用的治疗作用:慢性痛症(例如神经病性疼痛、肌肉和骨骼疼痛)、精神病(例如恐慌、焦虑、抑郁、酒精中毒和躁狂行为)、运动疾病(例如多发性硬化、动作性震颤、迟发性运动障碍)等(Magnus,Epilepsia,1999,40:S66-S72)。目前,加巴喷丁还用于临床控制神经病性疼痛。在疼痛和癫痫的临床模型中具有比加巴喷丁更大疗效的pregabalin现处于III期临床试验阶段。For example, pharmaceutically important GABA analogs include gabapentin (1), pregabalin (2), vigabatrin (3) and baclofen (4) shown above. Gabapentin, a lipophilic GABA analog that can cross the blood-brain barrier, has been clinically used in the treatment of epilepsy since 1994. Gabapentin may also have useful therapeutic effects in chronic pain disorders (such as neuropathic pain, muscular and skeletal pain), psychosis (such as panic, anxiety, depression, alcoholism, and manic behavior), motor sclerosis, action tremor, tardive dyskinesia), etc. (Magnus, Epilepsia, 1999, 40: S66-S72). Gabapentin is also currently used clinically for the management of neuropathic pain. Pregabalin, which has greater efficacy than gabapentin in clinical models of pain and epilepsy, is currently in phase III clinical trials.
许多GABA类似物的一个重要问题是γ-氨基与羧基官能团发生分子内反应形成γ-内酰胺,其实例如以下的加巴喷丁。由于γ-内酰胺(5)的毒性,它的形成给加巴喷丁制剂带来严重的困难。例如加巴喷丁的毒性(LD50,小鼠)大于8000mg/kg,而对应的内酰胺(5)的毒性(LD50,小鼠)为300mg/kg。因此,由于安全的原因,必须使在GABA类似物的合成和/或GABA类似物或GABA类似物的组合物的制剂和/或储存过程中副产物如内酰胺的形成最小化(特别是在An important problem with many GABA analogs is the intramolecular reaction of the γ-amino group with the carboxyl functional group to form a γ-lactam, such as gabapentin below. Due to the toxicity of the gamma-lactam (5), its formation poses serious difficulties for the formulation of gabapentin. For example, the toxicity (LD 50 , mouse) of gabapentin is greater than 8000 mg/kg, while the toxicity (LD 50 , mouse) of the corresponding lactam (5) is 300 mg/kg. Therefore, for safety reasons, the formation of by-products such as lactams must be minimized during the synthesis of GABA analogs and/or formulation and/or storage of GABA analogs or compositions of GABA analogs (especially in
加巴喷丁的情况下)。in the case of gabapentin).
已通过应用特定的附加纯化步骤、对药物组合物中的助剂材料的精确选择和谨慎的控制步骤(Augurt等人,美国专利6,054,482)部分克服了GABA类似物的内酰胺污染问题,特别是在加巴喷丁的情况下的内酰胺污染问题。但是,防止内酰胺污染的尝试在GABA类似物如加巴喷丁或其组合物的合成或储存方面并没有完全获得成功。The problem of lactam contamination of GABA analogs has been partially overcome, especially in the Lactam contamination issues in the case of gabapentin. However, attempts to prevent lactam contamination have not been entirely successful in the synthesis or storage of GABA analogs such as gabapentin or combinations thereof.
快速的系统清除率是许多GABA类似物、包括加巴喷丁的另一个重要的问题,因此需要频繁地服药以保持系统循环中的治疗和预防浓度(Bryans等人,Med.Res.REV.,1999,19,149-177)。每天施用三次300-600mg剂量的加巴喷丁的服药方案一般用于抗惊厥治疗。更高的剂量(1800-3600mg/天分开的剂量)一般用于治疗神经病性痛症。Rapid systemic clearance is another important issue for many GABA analogs, including gabapentin, so frequent dosing is required to maintain therapeutic and prophylactic concentrations in the systemic circulation (Bryans et al., Med. Res. REV., 1999, 19 , 149-177). A dosing regimen of gabapentin in doses of 300-600 mg three times daily is typically used for anticonvulsant therapy. Higher doses (1800-3600 mg/day divided doses) are generally used in the treatment of neuropathic pain.
持续释放制剂是快速的系统清除率的一个常规解决方案,它对于本领域技术人员来说是已知的(例如参见“Remington’sPharmaceutical Sciences,”Philadelphia College of Pharmacyand Science,17TH Edition,1985)。渗透递送系统也是已知的药物持续递送的方法(例如参见Verma等人,Drug Dev.Ind.Pharm.,2000,26:695-708)。许多GABA类似物、包括加巴喷丁和pregabalin不通过大肠吸收。相反,这些化合物一般在小肠中被大的中性氨基酸转运剂(″LNAA″)吸收(Jezyk等人,Pharm.RES.,1999,16,519-526)。常规剂型的通过胃肠道的近端吸收区域的快速通道阻碍了将持续释放技术成功地应用于许多GABA类似物。Sustained-release formulations are a conventional solution for rapid systemic clearance and are known to those skilled in the art (see, for example, "Remington's Pharmaceutical Sciences," Philadelphia College of Pharmacy and Science, 17TH Edition, 1985). Osmotic delivery systems are also known methods for sustained drug delivery (see eg Verma et al., Drug Dev. Ind. Pharm., 2000, 26:695-708). Many GABA analogs, including gabapentin and pregabalin, are not absorbed through the large intestine. Instead, these compounds are generally absorbed in the small intestine by the large neutral amino acid transporter ("LNAA") (Jezyk et al., Pharm. RES., 1999, 16, 519-526). The rapid passage of conventional dosage forms through the proximal absorption zone of the gastrointestinal tract has hindered the successful application of sustained release technology to many GABA analogs.
因此,非常需要有效的GABA类似物的持续释放方案,以将由于这些化合物的快速系统清除率而导致的服药频率的增加最小化。还需要基本上纯的并在制剂或储存过程中不自发地内酰胺化的纯GABA类似物,(特别是加巴喷丁和pregablin类似物)。Therefore, there is a great need for effective sustained release regimens of GABA analogs to minimize the increase in dosing frequency due to the rapid systemic clearance of these compounds. There is also a need for pure GABA analogs, (especially gabapentin and pregablin analogs) that are substantially pure and do not spontaneously lactamize during formulation or storage.
3.发明概述3. Summary of invention
本发明通过提供GABA类似物的前药、GABA类似物的前药的药物组合物和GABA类似物的前药的制备方法而致力于这些和其它需要。本发明还提供GABA类似物的前药的应用方法,和将GABA类似物的前药的药物组合物用于治疗或预防常见疾病和/或病症的方法。The present invention addresses these and other needs by providing prodrugs of GABA analogs, pharmaceutical compositions of prodrugs of GABA analogs, and methods of preparing prodrugs of GABA analogs. The present invention also provides the application method of the prodrug of the GABA analog, and the method of using the pharmaceutical composition of the prodrug of the GABA analog to treat or prevent common diseases and/or conditions.
重要地,本发明提供的前药可以具有显著的具体应用于医学的药学优点。首先,本发明提供的GABA类似物的前药的基元一般在体内是不稳定的(即在从患者上清除前药之前通过酶促或化学方法裂解产生大量的GABA类似物)。第二,通过从前药上裂解基元而提供的基元衍生物和它的任何代谢物在根据一般遵循GABA类似物的服药方案施用于哺乳动物时一般无毒。Importantly, the prodrugs provided by the present invention can have significant pharmaceutical advantages with particular application in medicine. First, the moieties of the prodrugs of the GABA analogs provided herein are generally unstable in vivo (ie, enzymatic or chemical cleavage yields large amounts of the GABA analog prior to clearance of the prodrug from the patient). Second, the derivatives of the motif provided by cleavage of the motif from the prodrug and any metabolites thereof are generally non-toxic when administered to mammals according to the dosing regimen generally followed by GABA analogs.
本发明的化合物具有一个与GABA类似物的γ氨基相连的基元。此基元可以与GABA类似物的γ氨基直接相连,或者任选可以与α-氨基酸基元的氨基或α-羟基酸基元的羟基相连,所述基元自身与GABA类似物的γ氨基相连。The compounds of the present invention have a moiety attached to the gamma amino group of the GABA analog. This motif may be directly attached to the gamma amino group of the GABA analog, or optionally may be attached to the amino group of an alpha-amino acid motif or the hydroxyl group of an alpha-hydroxy acid moiety which itself is attached to the gamma amino group of the GABA analog .
本发明的化合物还可以具有与GABA类似物的羧基相连的基元。羧基基元一般为酯或硫酯基。大量的酯或硫酯基可以用于形成羧基基元。The compounds of the invention may also have a moiety attached to the carboxyl group of the GABA analog. The carboxyl group is generally an ester or thioester group. A large number of ester or thioester groups can be used to form carboxyl moieties.
因此,本发明的化合物可以包括多至4个基元,包括依次与γ氨基相连的1个羧基基元和至多3个氨基基元(即,因此使各基元依次从GABA类似物的N-末端裂解)。本发明的化合物可以包含2个氨基基元和1个羧基基元、2个氨基基元、1个氨基基元和1个羧基基元或1个氨基基元。优选在包含氨基基元和羧基基元的本发明的化合物中,羧基基元在与氨基相连的基元的完全裂解之前被水解。Thus, the compounds of the present invention may comprise up to 4 motifs, including 1 carboxyl motif and up to 3 amino motifs sequentially attached to the gamma amino group (i.e., so that the motifs are sequentially sequenced from the N- end cleavage). The compounds of the invention may comprise 2 amino moieties and 1 carboxyl moiety, 2 amino moieties, 1 amino moiety and 1 carboxyl moiety or 1 amino moiety. Preferably in compounds of the invention comprising an amino moiety and a carboxyl moiety, the carboxyl moiety is hydrolyzed prior to complete cleavage of the moiety to which the amino group is attached.
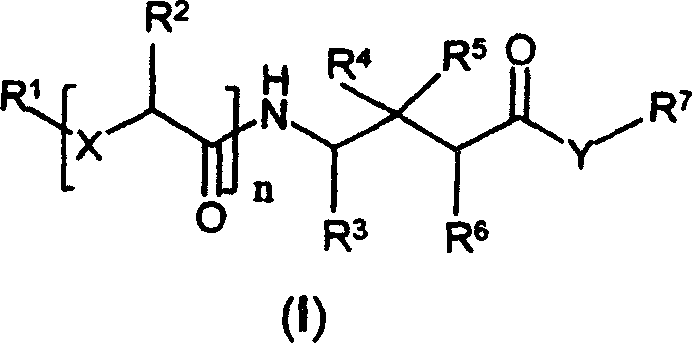
本发明第一方面提供式(I)、式(II)或式(III)的化合物:A first aspect of the present invention provides a compound of formula (I), formula (II) or formula (III):
或其药学上可接受的盐、水合物或溶剂化物,其中:or a pharmaceutically acceptable salt, hydrate or solvate thereof, wherein:
m、n、t和u独立地为0或1;m, n, t and u are independently 0 or 1;
X为O或NR16;X is 0 or NR 16 ;
W为O或NR17;W is O or NR 17 ;
Y为O或S;Y is O or S;
R1选自氢、R24C(O)-、R25OC(O)-、R24C(S)-、R25OC(S)-、R25SC(O)-、R25SC(S)-、(R9O)(R10O)P(O)-、R25S-、R 1 is selected from hydrogen, R 24 C(O)-, R 25 OC(O)-, R 24 C(S)-, R 25 OC(S)-, R 25 SC(O)-, R 25 SC( S)-, (R 9 O)(R 10 O)P(O)-, R 25 S-,
每个R2独立地选自氢、烷基、取代的烷基、烷氧基、取代的烷氧基、酰基、取代的酰基、酰基氨基、取代的酰基氨基、烷基氨基、取代的烷基氨基、烷基亚磺酰基、取代的烷基亚磺酰基、烷基磺酰基、取代的烷基磺酰基、烷硫基、取代的烷硫基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、芳氧基、取代的芳氧基、氨基甲酰基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、二烷基氨基、取代的二烷基氨基、卤素、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基、取代的杂芳基烷基、杂烷氧基、取代的杂烷氧基、杂芳氧基和取代的杂芳氧基,或者任选R2和R16与它们结合的原子一起形成环杂烷基或取代的环杂烷基环;Each R is independently selected from hydrogen, alkyl, substituted alkyl, alkoxy, substituted alkoxy, acyl, substituted acyl, acylamino, substituted acylamino, alkylamino, substituted alkyl Amino, alkylsulfinyl, substituted alkylsulfinyl, alkylsulfonyl, substituted alkylsulfonyl, alkylthio, substituted alkylthio, alkoxycarbonyl, substituted alkoxycarbonyl, Aryl, substituted aryl, arylalkyl, substituted arylalkyl, aryloxy, substituted aryloxy, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted Cycloheteroalkyl, dialkylamino, substituted dialkylamino, halogen, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, substituted heteroaryl alkyl, heteroalkoxy, substituted heteroalkoxy, heteroaryloxy, and substituted heteroaryloxy, or optionally R and R together with the atoms to which they are bound form a cycloheteroalkyl or substituted cycloheteroalkyl ring;
R3和R6独立地选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基;R and R are independently selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted cycloalkyl, ring Heteroalkyl, substituted cycloheteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, and substituted heteroarylalkyl;
R4和R5独立地选自氢、烷基、取代的烷基、酰基、取代的酰基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂芳基烷基和取代的杂芳基烷基,或者任选R4和R5与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基、取代的环杂烷基或桥连的环烷基环;R and R are independently selected from hydrogen, alkyl, substituted alkyl, acyl, substituted acyl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted cycloalkyl , cycloheteroalkane group, substituted cycloheteroalkyl, heteroarylalkyl and substituted heteroarylalkyl, or optionally R and R together with the carbon atoms to which they are bound form cycloalkyl, substituted cycloalkyl, ring Heteroalkyl, substituted cycloheteroalkyl, or bridged cycloalkyl rings;
R8和R12独立地选自氢、酰基、取代的酰基、烷氧基羰基、取代的烷氧基羰基、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基,或者任选R8和R12与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环;R and R are independently selected from hydrogen, acyl, substituted acyl, alkoxycarbonyl, substituted alkoxycarbonyl, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, Substituted arylalkyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, hetero Arylalkyl and substituted heteroarylalkyl, or optionally R and R taken together with the carbon atom to which they are bound form cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring;
R11选自氢、烷基、取代的烷基、酰基、取代的酰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、氰基、环烷基、取代的环烷基、杂环烷基、取代的杂环烷基、杂芳基、取代的杂芳基、杂芳基烷基、取代的杂芳基烷基、烷氧基羰基、取代的烷氧基羰基、环杂烷氧基羰基、取代的环杂烷氧基羰基、芳氧基羰基、取代的芳氧基羰基、杂芳氧基羰基、取代的杂芳氧基羰基和硝基; R is selected from hydrogen, alkyl, substituted alkyl, acyl, substituted acyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, carbamoyl, cyano, cycloalkyl , substituted cycloalkyl, heterocycloalkyl, substituted heterocycloalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, substituted heteroarylalkyl, alkoxycarbonyl, substituted Alkoxycarbonyl, cycloheteroalkoxycarbonyl, substituted cycloheteroalkoxycarbonyl, aryloxycarbonyl, substituted aryloxycarbonyl, heteroaryloxycarbonyl, substituted heteroaryloxycarbonyl, and nitro;
R7、R9、R10、R15、R16和R17独立地选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基;R 7 , R 9 , R 10 , R 15 , R 16 and R 17 are independently selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl , cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl and substituted heteroarylalkyl;
R13和R14独立地选自氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基,或者任选R13和R14与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环;R and R are independently selected from hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, substituted alkoxycarbonyl, aryl , substituted aryl, arylalkyl, substituted arylalkyl , carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, and substituted heteroaryl Alkyl, or optionally R and R together with the carbon atoms to which they are bound form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring;
R20和R21独立地选自氢、酰基、取代的酰基、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基,或者任选R20和R21与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环;R and R are independently selected from hydrogen, acyl, substituted acyl, alkyl , substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted Cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl and substituted heteroarylalkyl , or optionally R 20 and R 21 together with their bonded carbon atoms form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring;
R22和R23独立地选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基和取代的芳基烷基,或者任选R22和R23与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环; R22 and R23 are independently selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, and substituted arylalkyl, or optionally R22 and R23 are combined with them The carbon atoms of are taken together to form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring;
R24选自氢、酰基、取代的酰基、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基;并且 R is selected from hydrogen, acyl, substituted acyl, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, and substituted heteroarylalkyl; and
R25选自酰基、取代的酰基、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基。 R is selected from acyl, substituted acyl, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted cycloalkyl, cyclohetero Alkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, and substituted heteroarylalkyl.
在第二方面,本发明提供本发明的化合物的药物组合物。所述药物组合物一般包含一种或多种本发明的化合物和药学上可接受的赋形剂。In a second aspect, the invention provides pharmaceutical compositions of compounds of the invention. The pharmaceutical compositions generally comprise one or more compounds of the invention and a pharmaceutically acceptable excipient.
在第三方面,本发明提供治疗或预防以下疾病的方法:癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病或乙醇戒断综合征。所述方法一般包括给需要这种治疗或预防的患者施用治疗有效量的本发明的化合物。In a third aspect, the present invention provides a method of treating or preventing epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases, panic, pain (especially neuropathic pain and muscle and bone pain), inflammatory disease (ie, arthritis), insomnia, gastrointestinal disease, or alcohol withdrawal syndrome. The methods generally comprise administering to a patient in need of such treatment or prophylaxis a therapeutically effective amount of a compound of the invention.
在第四方面,本发明提供用于治疗或预防需要这种治疗或预防的患者的以下疾病的药物组合物:癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病或乙醇戒断综合征。所述方法一般包括给需要这种治疗或预防的患者施用治疗有效量的本发明的药物组合物。In a fourth aspect, the present invention provides a pharmaceutical composition for use in the treatment or prevention of epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases in a patient in need of such treatment or prevention , panic, pain (especially neuropathic pain and musculoskeletal pain), inflammatory disease (ie, arthritis), insomnia, gastrointestinal disease, or alcohol withdrawal syndrome. The methods generally comprise administering to a patient in need of such treatment or prophylaxis a therapeutically effective amount of a pharmaceutical composition of the invention.
在第五方面,本发明包括用于对需要治疗的患者进行给药的GABA类似物衍生物化合物,M-G,其中M为基元而G由GABA类似物,H-G(其中H为氢)衍生。基元M一旦从G上裂解,以及它的任何代谢产物对大鼠表现出大于0.2mmol/kg/天的致癌毒性剂量(TD50)。而且在对大鼠进行结肠给药时基元M在体内以充足的速率从G上裂解,以产生:In a fifth aspect, the invention encompasses a GABA analog derivative compound, MG, wherein M is a motif and G is derived from a GABA analog, HG, wherein H is hydrogen, for administration to a patient in need thereof. Once the motif M is cleaved from G, and any of its metabolites exhibit a carcinogenic toxic dose (TD 50 ) of greater than 0.2 mmol/kg/day to rats. Moreover, the motif M is cleaved from G in vivo at a sufficient rate upon colonic administration to rats to produce:
(i)通过结肠施用等摩尔剂量的H-G而得到至少为血浆H-G的Cmax的120%的血浆H-G的最大浓度(Cmax);和(i) colonically administered equimolar doses of HG to obtain a maximum concentration of plasma HG ( Cmax ) that is at least 120% of the Cmax of plasma HG; and
(ii)通过结肠施用等摩尔剂量的H-G而得到至少为AUC的120%的AUC。(ii) An AUC of at least 120% of the AUC is obtained by colonic administration of equimolar doses of H-G.
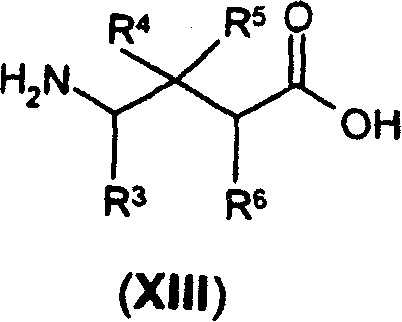
优选M-G为式(XIV)的衍生物:Preferably M-G is a derivative of formula (XIV):
或其药学上可接受的盐、水合物或溶剂化物,其中:or a pharmaceutically acceptable salt, hydrate or solvate thereof, wherein:
R为氢,或R和R6与它们结合的原子一起形成氮杂环丁烷、取代的氮杂环丁烷、吡咯烷或取代的吡咯烷环;和R is hydrogen, or R and R together with the atoms to which they are bonded form an azetidine, substituted azetidine, pyrrolidine or substituted pyrrolidine ring; and
Y、R3、R4、R5、R6和R7为如前面定义。Y, R 3 , R 4 , R 5 , R 6 and R 7 are as defined above.
最优选M为式(XV)的衍生物:Most preferably M is a derivative of formula (XV):
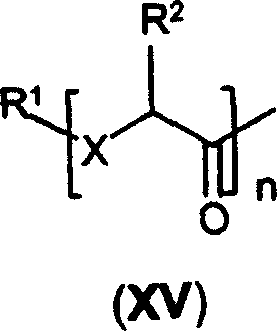
其中:n、X、R1和R2如前面定义。Wherein: n, X, R 1 and R 2 are as defined above.
4. 发明详述 4. Detailed Description of the Invention
4.1 定义 4.1 Definition
“ 活性转运或活性转运方法”指如下的跨过细胞膜的分子运动:" Active transport or active transport method " refers to the movement of molecules across a cell membrane as follows:
a)直接或间接地取决于能量介导的方法(即通过ATP水解、离子梯度等驱动);a) depend directly or indirectly on energy-mediated approaches (i.e. driven by ATP hydrolysis, ion gradients, etc.);
或or
b)发生通过与特定转运蛋白的相互作用介导的易化扩散b) Facilitated diffusion occurs through interactions with specific transporters
“ 烷基”指通过从母体烷烃、烯烃或炔烃的单一碳原子上除去一个氢原子而衍生的饱和或不饱和、直链、支链或环一价烃基。典型的烷基包括但不限于甲基;乙基如乙烷基、乙烯基、乙炔基;丙基如丙-1-基、丙-2-基、环丙-1-基、丙-1-烯-1-基、丙-1-烯-2-基、丙-2-烯-1-基(烯丙基)、环丙-1-烯-1-基;环丙-2-烯-1-基、丙-1-炔-1-基、丙-2-炔-1-基等;丁基如丁-1-基、丁-2-基、2-甲基-丙-1-基、2-甲基-丙-2-基、环丁-1-基、丁-1-烯-1-基、丁-1-烯-2-基、2-甲基-丙-1-烯-1-基、丁-2-烯-1-基、丁-2-烯-2-基、丁-1,3-二烯-1-基、丁-1,3-二烯-2-基、环丁-1-烯-1-基、环丁-1-烯-3-基、环丁-1,3-二烯-1-基、丁-1-炔-1-基、丁-1-炔-3-基、丁-3-炔-1-基等等。" Alkyl " means a saturated or unsaturated, straight-chain, branched-chain or cyclic monovalent hydrocarbon group derived by the removal of a hydrogen atom from a single carbon atom of a parent alkane, alkene or alkyne. Typical alkyl groups include, but are not limited to, methyl; ethyl such as ethyl, vinyl, ethynyl; propyl such as prop-1-yl, prop-2-yl, cycloprop-1-yl, prop-1- En-1-yl, prop-1-en-2-yl, prop-2-en-1-yl (allyl), cycloprop-1-en-1-yl; cycloprop-2-en-1 -yl, prop-1-yn-1-yl, prop-2-yn-1-yl, etc.; butyl such as but-1-yl, but-2-yl, 2-methyl-prop-1-yl, 2-Methyl-prop-2-yl, cyclobut-1-yl, but-1-en-1-yl, but-1-en-2-yl, 2-methyl-prop-1-en-1 -yl, but-2-en-1-yl, but-2-en-2-yl, but-1,3-dien-1-yl, but-1,3-dien-2-yl, ring But-1-en-1-yl, cyclobut-1-en-3-yl, cyclobut-1,3-dien-1-yl, but-1-yn-1-yl, but-1-yne -3-yl, but-3-yn-1-yl and the like.
术语“ 烷基”具体意指包括具有任何饱和程度或水平的基团,即具有独有的碳-碳单键的基团、具有一个或多个碳-碳双键的基团、具有一个或多个碳-碳叁键的基团和具有碳-碳单键、双键和叁键的混合物的基团。当意指特定的饱和水平时,使用表述“链烷基”、“链烯基”和“炔基”。优选烷基包含1-20个碳原子,更优选1-10个碳原子。The term " alkyl " is specifically meant to include groups having any degree or level of saturation, i.e., groups having exclusively carbon-carbon single bonds, groups having one or more carbon-carbon double bonds, groups having one or Groups with multiple carbon-carbon triple bonds and groups with mixtures of carbon-carbon single, double and triple bonds. The expressions "alkanyl", "alkenyl" and "alkynyl" are used when a particular level of saturation is intended. Preferably the alkyl group contains 1-20 carbon atoms, more preferably 1-10 carbon atoms.
“ 链烷基”指通过从母体烷烃的单一碳原子上除去一个氢原子而衍生的饱和的支链、直链或环烷基。典型的链烷基包括但不限于甲基、乙基、丙基如丙-1-基、丙-2-基(异丙基)、环丙-1-基等;丁基如丁-1-基、丁-2-基(仲丁基)、2-甲基-丙-1-基(异丁基)、2-甲基-丙-2-基(叔丁基)、环丁-1-基等。" Alkanyl " means a saturated branched, straight chain or cyclic alkyl group derived by the removal of a hydrogen atom from a single carbon atom of a parent alkane. Typical alkanyl groups include, but are not limited to, methyl, ethyl, propyl such as prop-1-yl, prop-2-yl (isopropyl), cycloprop-1-yl, etc.; butyl such as but-1- Base, but-2-yl (sec-butyl), 2-methyl-prop-1-yl (isobutyl), 2-methyl-prop-2-yl (tert-butyl), cyclobutan-1- Base etc.
“ 链烯基”指通过从母体烯烃的单一碳原上除去一个氢原子而衍生的具有至少一个碳-碳双键的不饱和支链、直链或环烷基。所述基团的双键可以顺式或反式构象。典型的链烯基包括但不限于乙烯基;丙烯基如丙-1-烯-1-基、丙-1-烯-2-基、丙-2-烯-1-基(烯丙基)、丙-2-烯-2-基、环丙-1-烯-1-基;环丙-2-烯-1-基;丁烯基如丁-1-烯-1-基、丁-1-烯-2-基、2-甲基-丙-1-烯-1-基、丁-2-烯-1-基、丁-2-烯-1-基、丁-2-烯-2-基、丁-1,3-二烯-1-基、丁-1,3-二烯-2-基、环丁-1-烯-1-基、环丁-1-烯-3-基、环丁-1,3-二烯-1-基等等。" Alkenyl " means an unsaturated branched, straight chain or cycloalkyl group having at least one carbon-carbon double bond derived by the removal of a hydrogen atom from a single carbon atom of a parent olefin. The double bond of the group can be in cis or trans configuration. Typical alkenyl groups include, but are not limited to, vinyl; propenyl such as prop-1-en-1-yl, prop-1-en-2-yl, prop-2-en-1-yl (allyl), Pro-2-en-2-yl, cycloprop-1-en-1-yl; cycloprop-2-en-1-yl; butenyl such as but-1-en-1-yl, but-1- En-2-yl, 2-methyl-prop-1-en-1-yl, but-2-en-1-yl, but-2-en-1-yl, but-2-en-2-yl , But-1,3-dien-1-yl, but-1,3-dien-2-yl, cyclobut-1-en-1-yl, cyclobut-1-en-3-yl, ring But-1,3-dien-1-yl and the like.
“ 炔基”指通过从母体炔基的单一碳原子上除去一个氢原子而衍生的具有至少一个碳-碳叁键的不饱和支链、直链或环烷基。典型的炔基包括但不限于乙炔基;丙炔基如丙-1-炔-1-基,丙-2-炔-1-基等;丁炔基如丁-1-炔-1-基,丁-1-炔-3-基,丁-3-炔-1-基等等。" Alkynyl " means an unsaturated branched, straight chain or cycloalkyl group having at least one carbon-carbon triple bond derived by the removal of a hydrogen atom from a single carbon atom of a parent alkynyl group. Typical alkynyl groups include but are not limited to ethynyl; propynyl such as prop-1-yn-1-yl, prop-2-yn-1-yl, etc.; butynyl such as but-1-yn-1-yl, but-1-yn-3-yl, but-3-yn-1-yl and the like.
“ 酰基”指基团-C(O)R,其中R为本文定义的氢、烷基、环烷基、环杂烷基、芳基、芳基烷基、杂烷基、杂芳基、杂芳基烷基。代表性的实例包括但不限于甲酰基、乙酰基、环己基羰基、环己基甲基羰基、苯甲酰基、苄基羰基等。" Acyl " means the group -C(O)R, where R is hydrogen, alkyl, cycloalkyl, cycloheteroalkyl, aryl, arylalkyl, heteroalkyl, heteroaryl, hetero Arylalkyl. Representative examples include, but are not limited to, formyl, acetyl, cyclohexylcarbonyl, cyclohexylmethylcarbonyl, benzoyl, benzylcarbonyl, and the like.
“ 酰基氨基”(或可选择的“酰基酰氨基”)指基团-NR’C(O)R,其中R’和R各自独立地为如本文定义的氢、烷基、环烷基、环杂烷基、芳基、芳基烷基、杂烷基、杂芳基、杂芳基烷基。代表性的实例包括但不限于甲酰基氨基、乙酰基氨基(即乙酰氨基),环己基羰基氨基,环己基甲基-羰基氨基,苯甲酰基氨基(即苯甲酰氨基)、苄基羰基氨基等。" Acylamino " (or alternatively "acylamido") refers to the group -NR'C(O)R, wherein R' and R are each independently hydrogen, alkyl, cycloalkyl, ring as defined herein Heteroalkyl, aryl, arylalkyl, heteroalkyl, heteroaryl, heteroarylalkyl. Representative examples include, but are not limited to, formylamino, acetylamino (i.e., acetylamino), cyclohexylcarbonylamino, cyclohexylmethyl-carbonylamino, benzoylamino (i.e., benzylamino), benzylcarbonylamino wait.
“ 酰氧基”指基团-OC(O)R,其中R为如本文定义的氢、烷基、环烷基、环杂烷基、芳基、芳基烷基、杂烷基、杂芳基或杂芳基烷基。代表性的实例包括但不限于乙酰氧基(或乙酸基)、丁氧基(butyloxy或butoxy)、苯甲酰氧基等。" Acyloxy " refers to the group -OC(O)R, where R is hydrogen, alkyl, cycloalkyl, cycloheteroalkyl, aryl, arylalkyl, heteroalkyl, heteroaryl, as defined herein radical or heteroarylalkyl. Representative examples include, but are not limited to, acetoxy (or acetate), butyloxy (or butoxy), benzoyloxy, and the like.
“ 烷基氨基”意指基团-NHR,其中R代表如本文定义的烷基或环烷基。代表性的实例包括但不限于甲基氨基、乙基氨基、1-甲基乙基氨基、环己基氨基等。" Alkylamino " means a group -NHR in which R represents alkyl or cycloalkyl as defined herein. Representative examples include, but are not limited to, methylamino, ethylamino, 1-methylethylamino, cyclohexylamino, and the like.
“ 烷氧基”指基团-OR,其中R代表本文定义的烷基或环烷基。代表性的实例包括但不限于甲氧基、乙氧基、丙氧基、丁氧基、环己氧基等。" Alkoxy " refers to the group -OR in which R represents alkyl or cycloalkyl as defined herein. Representative examples include, but are not limited to, methoxy, ethoxy, propoxy, butoxy, cyclohexyloxy, and the like.
“ 烷氧基羰基”指基团-C(O)-烷氧基,其中烷氢基如本文定义。" Alkoxycarbonyl " refers to the group -C(O)-alkoxy where alkylhydrogen is as defined herein.
“ 烷基磺酰基”指基团-S(O)2R,其中R为本文定义的烷基或环烷基。代表性的实例包括但不限于甲基磺酰基、乙基磺酰基、丙基磺酰基、丁基磺酰基等。" Alkylsulfonyl " refers to the group -S(O) 2R , where R is alkyl or cycloalkyl as defined herein. Representative examples include, but are not limited to, methylsulfonyl, ethylsulfonyl, propylsulfonyl, butylsulfonyl, and the like.
“ 烷基亚磺酰基”指基团-S(O)R,其中R为本文定义的烷基或环烷基。代表性的实例包括但不限于甲基亚磺酰基、乙基亚磺酰基、丙基亚磺酰基、丁基亚磺酰基等。" Alkylsulfinyl " refers to the group -S(O)R, where R is alkyl or cycloalkyl as defined herein. Representative examples include, but are not limited to, methylsulfinyl, ethylsulfinyl, propylsulfinyl, butylsulfinyl, and the like.
“ 烷硫基”指基团-SR,其中R为本文定义的可以任选如本文所述被取代的烷基或环烷基。代表性的实例包括但不限于甲硫基、乙硫基、丙硫基、丁硫基等。" Alkylthio " refers to a group -SR where R is alkyl or cycloalkyl as defined herein which may be optionally substituted as described herein. Representative examples include, but are not limited to, methylthio, ethylthio, propylthio, butylthio, and the like.
“ 氨基”指基团-NH2-。" Amino " refers to the group -NH2- .
“ 芳基”指通过除去母体芳环系统中的单一碳原上的一个氢原子而衍生的一价芳香烃基。典型的芳基包括但不限于由以下衍生的基团:醋蒽烯、苊、acephenanthrylene、蒽、薁、苯、、蒄、荧蒽、芴、并六苯、萘并四并苯、hexalene、反式苯并二茚、顺式苯并二茚、茚满、茚、萘、并八苯(octacene)、并八苯(octaphene)、octalene、卵苯、戊-2,4-二烯、并五苯、并环戊二烯、戊芬(phetaphene)、苝、并三苯(phenalene)、菲、苉、pleiadene、芘、皮蒽、玉红省、苯并菲、联三萘等。优选芳基含有6-20个碳原子,更优选6-12个碳原子。" Aryl " refers to a monovalent aromatic hydrocarbon radical derived by removal of a hydrogen atom from a single carbon atom in a parent aromatic ring system. Typical aryl groups include, but are not limited to, groups derived from the following: acethanene, acenaphthene, acephenanthrylene, anthracene, azulene, benzene, onium, perene, fluoranthene, fluorene, hexacene, naphthotetracene, hexalene, Trans-benzobiindene, cis-benzobiindene, indane, indene, naphthalene, octacene, octaphene, octacene, ovalene, penta-2,4-diene, and Pentacene, pentacyclopentadiene, phetaphene, perylene, phenalene, phenanthrene, perylene, pleiadene, pyrene, picanthracene, rubin, triphenylene, ternaphthalene, etc. Preferred aryl groups contain 6-20 carbon atoms, more preferably 6-12 carbon atoms.
“ 芳基烷基”指非环烷基,其中与碳原子,通常为末端或SP3碳原子结合的氢原子被芳基取代。典型的芳基烷基包括但不限于苄基、2-苯基乙-1-基、2-苯基乙烯-1-基、萘基甲基、2-萘基乙-1-基、2-萘基乙烯-1-基、萘并苄基、2-萘并苯基乙-1-基等。其中如果欲指定特定的烷基部分,使用术语芳基链烷基、芳基链烯基和/或芳基炔基。优选芳基烷基为(C6-C30)芳基烷基,例如芳基烷基的链烷基、链烯基或炔基部分为(C1-C10),而芳基部分为(C6-C20),更优选芳基烷基为(C6-C20)芳基烷基,例如芳基烷基的链烷基、链烯基或炔基部分为(C1-C8),而芳基部分为(C6-C12)。" Arylalkyl " means a non-cyclic alkyl group in which a hydrogen atom bonded to a carbon atom, usually a terminal or SP 3 carbon atom, is replaced by an aryl group. Typical arylalkyl groups include, but are not limited to, benzyl, 2-phenylethen-1-yl, 2-phenylethen-1-yl, naphthylmethyl, 2-naphthyleth-1-yl, 2- Naphthylvin-1-yl, naphthobenzyl, 2-naphthophenyleth-1-yl, etc. Where a specific alkyl moiety is intended, the terms arylalkanyl, arylalkenyl and/or arylalkynyl are used. Preferably the arylalkyl is (C 6 -C 30 )arylalkyl, for example the alkanyl, alkenyl or alkynyl portion of the arylalkyl is (C 1 -C 10 ) and the aryl portion is ( C 6 -C 20 ), more preferably arylalkyl is (C 6 -C 20 )arylalkyl, for example the alkanyl, alkenyl or alkynyl portion of arylalkyl is (C 1 -C 8 ), and the aryl moiety is (C 6 -C 12 ).
“ 芳基烷氧基”指其中的芳基烷基如本文定义的-O-芳基烷基。" Arylalkoxy " means an -O-arylalkyl group in which the arylalkyl group is as defined herein.
“ 芳氧基羰基”指基团-C(O)-O-芳基,其中芳基如本文定义。" Aryloxycarbonyl " means the group -C(O)-O-aryl wherein aryl is as defined herein.
“ AUC”是从零时间至无穷大外推的血浆药物浓度-对-时间曲线下的面积。" AUC " is the area under the plasma drug concentration-vs-time curve extrapolated from time zero to infinity.
“ 桥连的环烷基”指选自以下的基团:" Bridged cycloalkyl " refers to a group selected from the group consisting of:
其中:in:
A为(CR35R36)b;A is (CR 35 R 36 ) b ;
R35和R36独立地选自氢和甲基;R 35 and R 36 are independently selected from hydrogen and methyl;
R33和R34独立地选自氢和甲基;R 33 and R 34 are independently selected from hydrogen and methyl;
b为1-4的整数;和b is an integer from 1 to 4; and
c为0-2的整数。c is an integer of 0-2.
“ 氨基甲酰基”指基团-C(O)N(R)2,其中各R基团独立地为本文定义的氢、烷基、环烷基或芳基,所述基团可以任选被取代,如本文定义。" Carbamoyl " refers to the group -C(O)N(R) 2 , wherein each R group is independently hydrogen, alkyl, cycloalkyl, or aryl as defined herein, which may optionally be Replace, as defined herein.
“ 羧基”意指基团-C(O)OH。" Carboxy " means the group -C(O)OH.
“致癌效力(TD50)”(参见Peto等人,Environmental HealthPerspectives 1984,58,1-8)定义为导致确定的动物种类在其标准寿命结束时有半数受试动物患肿瘤的以mg/kg体重/天为单位的慢性剂量-等级。由于相关的肿瘤经常发生在对照动物身上,TD50更明确地定义为:以mg/kg体重/天为单位的剂量-等级,这种剂量-等级如果在所述种类的标准寿命期间慢性施用的话,将可能在该期间保持无肿瘤。可以计算任何特定类型的瘤,任何特定组织或它们的任何组合的TD50。"Carcinogenic potency (TD 50 )" (see Peto et al., Environmental Health Perspectives 1984, 58, 1-8) is defined as the dose in mg/kg body weight that causes tumors in half of the animals tested at the end of their standard life span for a defined animal species Chronic dose-grades per day. Since the associated tumors frequently occur in control animals, the TD50 is more clearly defined as: the dose-level in mg/kg bw/day if administered chronically during the standard lifespan of the species in question , will likely remain tumor-free during this period. TD50 can be calculated for any particular type of tumor, for any particular tissue, or any combination thereof.
“ C max”是在施用血管外剂量的药物之后观察到的血浆最高药物浓度。" Cmax " is the highest observed plasma drug concentration following administration of an extravascular dose of drug.
“ 本发明的化合物”指由本文所述通式包含的化合物,它包括任何在本文公开其结构的通式范围之内的特定化合物。本发明的化合物可以由它们的化学结构和/或化学名称鉴定。当化学结构和化学名称冲突时,化学结构决定化合物的身份。本发明的化合物可以包含一个或多个手性中心和/或双键,因此可以作为立体异构体如双键异构体(即几何异构体)、旋光对映体或非对映异构体存在。因此,本文所述的化学结构包含所示化合物的所有可能的旋光对映体和立体异构体,包括立体异构体纯形式(例如几何学纯、旋光对映体纯或非对映异构体纯)和对映异构体和立体异构体混合物。可以使用本领域技术人员已知的分离技术或手性合成技术将对映异构体和立体异构体混合物拆分成它们的组成旋光对映体或立体异构体。本发明的化合物还可以多种互变异构体形式存在,包括烯醇形式、酮形式或它们的混合物。因此,本文所述的结构式包括所示化合物的所有可能的互变异构体形式。本发明的化合物还包括同位素标记的化合物,其中一个或多个原子的原子量不同于在自然中通常发现的原子量。可以包含在本发明的化合物中的同位素的实例包括但不限于2H、3H、13C、14C、15N、18O、17O、31P、32P、35S、18F和36Cl。而且,应该理解当例示本发明化合物的部分结构时,方括号表示该部分结构与分子的剩余部分的结合点。" Compounds of the invention " refers to compounds encompassed by the formulas described herein, which include any specific compound within the scope of a formula whose structure is disclosed herein. The compounds of the present invention can be identified by their chemical structures and/or chemical names. When chemical structure and chemical name conflict, the chemical structure determines the identity of the compound. The compounds of the present invention may contain one or more chiral centers and/or double bonds and may therefore be present as stereoisomers such as double bond isomers (i.e. geometric isomers), optical enantiomers or diastereoisomers body exists. Accordingly, the chemical structures depicted herein encompass all possible enantiomers and stereoisomers of the compounds shown, including stereoisomerically pure forms (e.g., geometrically pure, enantiomerically pure, or diastereomerically pure) stereopure) and mixtures of enantiomers and stereoisomers. Enantiomeric and stereoisomeric mixtures can be resolved into their constituent optical antipodes or stereoisomers using separation techniques or chiral synthesis techniques known to those skilled in the art. The compounds of the present invention may also exist in various tautomeric forms, including enol forms, keto forms or mixtures thereof. Accordingly, the structural formulas described herein include all possible tautomeric forms of the compounds shown. Compounds of the invention also include isotopically labeled compounds in which one or more atoms have an atomic mass other than that normally found in nature. Examples of isotopes that may be included in compounds of the invention include, but are not limited to , 2H , 3H , 13C , 14C, 15N , 18O, 17O, 31P, 32P , 35S , 18F , and 36 Cl. Furthermore, it should be understood that when illustrating a partial structure of a compound of the invention, square brackets indicate the point of attachment of that partial structure to the remainder of the molecule.
“ 本发明的组合物”指至少一种本发明的化合物和药学上可接受的赋形剂,通过它将所述化合物施用于人。本发明的化合物在施用于人时以分离的形式施用,它意味着从合成的有机反应混合物中分离。A " composition of the invention " refers to at least one compound of the invention and a pharmaceutically acceptable excipient by which the compound is administered to a human. When administered to humans, the compounds of the present invention are administered in isolated form, which means isolated from synthetic organic reaction mixtures.
“ 氰基”意指基团-CN。" Cyano " means the group -CN.
“ 环烷基”指饱和或不饱和环烷基。其中如果欲指定特定的饱和水平,使用术语“环烷基”或“环链烯基。典型的环烷基包括但不限于由环丙烷、环丁烷、环戊烷、环己烷等衍生的基团。优选环烷基为(C3-C10)环烷基,更优选(C3-C7)环烷基。" Cycloalkyl " means a saturated or unsaturated cycloalkyl group. Where a particular level of saturation is intended, the term "cycloalkyl" or "cycloalkenyl" is used. Typical cycloalkyl groups include, but are not limited to, those derived from cyclopropane, cyclobutane, cyclopentane, cyclohexane, etc. Group. Preferably the cycloalkyl group is (C 3 -C 10 )cycloalkyl group, more preferably (C 3 -C 7 )cycloalkyl group.
“ 环杂烷基”指饱和或不饱和烷基,其中一个或多个碳原子(和任何相连的氢原子)独立地被相同或不同的杂原子取代。典型的取代碳原子的杂原子包括但不限于N、P、O、S、Si等,其中如果欲指定特定的饱和水平,使用术语“环杂链烷基”或“环杂链烯基”。典型的环杂烷基包括但不限于环氧化物、咪唑烷、吗啉、哌嗪、哌啶、吡唑烷、吡咯烷、奎宁环等。" Cycloheteroalkyl " refers to a saturated or unsaturated alkyl group in which one or more carbon atoms (and any attached hydrogen atoms) are independently replaced by the same or different heteroatoms. Typical heteroatoms replacing carbon atoms include, but are not limited to, N, P, O, S, Si, etc., where the term "cycloheteroalkanyl" or "cycloheteroalkenyl" is used if a particular level of saturation is intended. Typical cycloheteroalkyl groups include, but are not limited to, epoxides, imidazolidines, morpholines, piperazines, piperidines, pyrazolidines, pyrrolidines, quinuclidines, and the like.
“ 环杂烷氧基羰基”指基闭-C(O)-OR,其中R为如以上定义的环杂烷基。" Cycloheteroalkoxycarbonyl " refers to the group -C(O)-OR, wherein R is cycloheteroalkyl as defined above.
“ 由胆汁酸衍生”指结构上与式(XVII)或(XVIII)的化合物有关的部分:" Derived from bile acids " refers to moieties structurally related to compounds of formula (XVII) or (XVIII):
其中每一个D、E和F独立地为H或OH。wherein each of D, E and F is independently H or OH.
所述部分的结构与上述化合物相同,除了1或2位以外。在这些位置,与羟基和/或羧基的羟基部分结合的氢原子已被用作另一部分的结合点的共价键取代,该另一部分优选为GABA类似物或GABA类似物衍生物。The moiety has the same structure as the above compound except for the 1 or 2 position. At these positions, the hydrogen atoms bound to the hydroxyl moiety of the hydroxyl and/or carboxyl groups have been replaced by a covalent bond serving as a binding point for another moiety, preferably a GABA analog or a GABA analog derivative.
“ 由GABA类似物衍生”指结构上与GABA类似物相美的部分。所述部分的结构与所述化合物相同,除了1或2位以外。在这些位置,与氨基和(任选的)羧基的羟基部分结合的氢原子已被用作另一部分的结合点的共价键取代。" Derived from a GABA analog " refers to a moiety that is structurally equivalent to a GABA analog. The moiety has the same structure as the compound, except for the 1 or 2 position. At these positions, the hydrogen atom bound to the hydroxyl moiety of the amino and (optional) carboxyl groups has been replaced by a covalent bond that serves as a point of attachment for the other moiety.
“ 二烷基氨基”意指基团-NRR’,其中R和R’独立地代表本文定义的烷基或环烷基。代表性的实例包括但不限于二甲基氨基、甲基乙基氨基、二(1-甲基乙基)氨基、(环己基)(甲基)氨基、(环己基)(乙基)氨基、(环己基)(丙基)氨基等。" Dialkylamino " means a group -NRR' wherein R and R' independently represent alkyl or cycloalkyl as defined herein. Representative examples include, but are not limited to, dimethylamino, methylethylamino, bis(1-methylethyl)amino, (cyclohexyl)(methyl)amino, (cyclohexyl)(ethyl)amino, (cyclohexyl)(propyl)amino, etc.
除非另外指出,“ GABA类似物”指具有以下结构的化合物:Unless otherwise indicated, " GABA analogs " refer to compounds having the following structure:
其中:in:
R为氢,或者R和R6和与它们结合的原子一起形成氮杂环丁烷、取代的氮杂环丁烷、吡咯烷或取代的吡咯烷环;R is hydrogen, or R and R together form an azetidine, a substituted azetidine, a pyrrolidine or a substituted pyrrolidine ring with the atoms bound to them;
R3和R6独立地选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基;和R and R are independently selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted cycloalkyl, ring heteroalkyl, substituted cycloheteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, and substituted heteroarylalkyl; and
R4和R5独立地选自氢、烷基、取代的烷基、酰基、取代的酰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基、或者任选R4和R5和与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基、取代的环杂烷基或桥连的环烷基环。R and R are independently selected from hydrogen, alkyl, substituted alkyl, acyl, substituted acyl, aryl, substituted aryl, arylalkyl , substituted arylalkyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl and substituted heteroarylalkyl, or optionally R and R and Together with the carbon atoms to which they are bound, form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl or bridged cycloalkyl ring.
“ 卤素”意指氟、氯、溴或碘。" Halogen " means fluorine, chlorine, bromine or iodine.
“ 杂烷氧基”意指-O-杂烷基,其中杂烷基如本文定义。" Heteroalkoxy " means -O-heteroalkyl, wherein heteroalkyl is as defined herein.
“ 杂烷基、杂链烷基、杂链烯基、杂炔基”分别指烷基、链烷基、链烯基和炔基,其中一个或多个碳原子(和任何相连的氢原子)各自独立地被相同或不同的杂原子取代。典型的杂原子基团包括但不限于:-O-、-S-、-O-O-、-S-S-、-O-S-、-NR’-、=N-N=、-N=N-、-N=N-NR’、-PH-、-P(O)2-、-O-P(O)2-、-S(O)-、-S(O)2-、-SnH2-等,其中R’为氢、烷基、取代的烷基、环烷基、取代的环烷基、芳基或取代的芳基。" Heteroalkyl, heteroalkanyl, heteroalkenyl, heteroalkynyl " means alkyl, alkanyl, alkenyl, and alkynyl, respectively, in which one or more carbon atoms (and any attached hydrogen atoms) are each independently substituted with the same or different heteroatoms. Typical heteroatom groups include, but are not limited to: -O-, -S-, -OO-, -SS-, -OS-, -NR'-, =NN=, -N=N-, -N=N -NR', -PH-, -P(O) 2 -, -OP(O) 2 -, -S(O)-, -S(O) 2 -, -S n H 2 -, etc., wherein R' is hydrogen, alkyl, substituted alkyl, cycloalkyl, substituted cycloalkyl, aryl, or substituted aryl.
“ 杂芳基”指通过除去杂芳香环系统的单一原子上的一个氢原子而衍生的一价杂芳香基团。典型的杂芳基包括但不限于由以下衍生的基团:吖啶、砷杂茚、咔唑、β-咔啉、苯并二氢吡喃、苯并吡喃、噌啉、呋喃、咪唑、吲唑、吲哚、二氢吲哚、中氮茚、异苯并呋喃、异苯并吡喃、异吲哚、异二氢吲哚、异喹啉、异噻唑、异噁唑、萘啶、噁二唑、噁唑、啶、菲啶、菲咯啉、吩嗪、酞嗪、蝶啶、嘌呤、吡喃、吡嗪、吡唑、哒嗪、吡啶、嘧啶、吡咯、吡咯嗪(pyrrolizine)、喹唑啉、喹啉、喹嗪、喹喔啉、四唑、噻二唑、噻唑、噻吩、三唑、咕吨等。优选杂芳基为5-20元杂芳基,更优选5-10元杂芳基。优选的杂芳基是由噻吩、吡咯、苯并噻吩、苯并呋喃、吲哚、吡啶、喹啉、咪唑、噁唑和吡嗪衍生的杂芳基。" Heteroaryl "refers to a monovalent heteroaromatic group derived by removal of a hydrogen atom from a single atom of a heteroaromatic ring system. Typical heteroaryl groups include, but are not limited to, groups derived from acridine, arsine, carbazole, β-carboline, chroman, chromene, cinnoline, furan, imidazole, Indazole, indole, indoline, indolizine, isobenzofuran, isobenzopyran, isoindole, isoindoline, isoquinoline, isothiazole, isoxazole, naphthyridine, Oxadiazole, oxazole, thidine, phenanthridine, phenanthroline, phenazine, phthalazine, pteridine, purine, pyran, pyrazine, pyrazole, pyridazine, pyridine, pyrimidine, pyrrole, pyrrolizine ), quinazoline, quinoline, quinazine, quinoxaline, tetrazole, thiadiazole, thiazole, thiophene, triazole, xanthene, etc. Preferably the heteroaryl is a 5-20 membered heteroaryl, more preferably a 5-10 membered heteroaryl. Preferred heteroaryl groups are those derived from thiophene, pyrrole, benzothiophene, benzofuran, indole, pyridine, quinoline, imidazole, oxazole and pyrazine.
“ 杂芳氧基羰基”指基团-C(O)-OR,其中R为本文定义的杂芳基。" Heteroaryloxycarbonyl " refers to the group -C(O)-OR where R is heteroaryl as defined herein.
“ 杂芳基烷基”指无环烷基,其中与碳原子,通常为末端或SP3碳原子结合的一个氢原子被杂芳基取代。其中如果欲指定特定的烷基部分,使用术语杂芳基链烷基、杂芳基链烯基和/或杂芳基炔基。在优选的实施方案中,杂芳基烷基为6-30元杂芳基烷基,例如杂芳基烷基的链烷基、链烯基或炔基部分为1-10元,而杂芳基部分为5-20元杂芳基,更优选6-20元杂芳基烷基,例如杂芳基烷基的链烷基、链烯基或炔基部分为1-8元,而杂芳基部分为5-12-元杂芳基。" Heteroarylalkyl " refers to an acyclic alkyl group in which one hydrogen atom bonded to a carbon atom, usually a terminal or SP 3 carbon atom, is replaced by a heteroaryl group. Where a specific alkyl moiety is intended, the terms heteroarylalkanyl, heteroarylalkenyl and/or heteroarylalkynyl are used. In preferred embodiments, heteroarylalkyl is 6-30 membered heteroarylalkyl, for example the alkanyl, alkenyl or alkynyl moiety of heteroarylalkyl is 1-10 membered, and heteroaryl The base part is 5-20 membered heteroaryl, more preferably 6-20 membered heteroarylalkyl, for example, the alkanyl, alkenyl or alkynyl part of heteroarylalkyl is 1-8 membered, and heteroaryl The base part is 5-12-membered heteroaryl.
“ 被动扩散”指不由特定的转运蛋白介导的试剂的摄入。基本上不能被动扩散的试剂具有一个在体外小于5×10-6cm/秒,通常小于1×10-6cm/秒(缺少流出机理)地跨过标准细胞单层(例如Caco-2)的渗透率。" Passive diffusion " refers to the uptake of an agent that is not mediated by a specific transporter. Reagents that are substantially non-passively diffusible have an in vitro velocity of less than 5 x 10 -6 cm/sec, usually less than 1 x 10 -6 cm/sec (lacking an efflux mechanism) across a standard cell monolayer (e.g. Caco-2). permeability.
“ 药学上可接受的”意指联邦或国家政府批准或可以批准的,或者在美国药典或其它公认的用于动物、更具体是人的药典中列出。" Pharmaceutically acceptable " means approved or may be approved by a federal or state government, or listed in the US Pharmacopoeia or other recognized pharmacopoeia for use in animals, more particularly humans.
“ 药学上可接受的盐”指本发明化合物的盐,它是药学上可接受的并具有期望的母体化合物的药理学活性。这些盐包括:(1)酸加成盐,它是与诸如以下的无机酸形成的:如盐酸、氢溴酸、硫酸、硝酸、磷酸等;或者与诸如以下的有机酸形成:乙酸、丙酸、、己酸、环戊烷丙酸、乙醇酸、丙酮酸、乳酸、丙二酸、琥珀酸、苹果酸、马来酸、富马酸、酒石酸、柠檬酸、苯甲酸、3-(4-羟基苯甲酰基)苯甲酸、肉桂酸、杏仁酸、甲磺酸、乙磺酸、1,2-乙二磺酸、2-羟基乙磺酸、苯磺酸、4-氯苯磺酸、2-萘磺酸、4-甲苯磺酸、樟脑磺酸、4-甲基双环2.2.2-辛-2-烯-1-甲酸、葡庚糖酸、3-苯基丙酸、三甲基乙酸、叔丁基乙酸、月桂基硫酸、葡糖酸、谷氨酸、羟基萘甲酸、水杨酸、硬脂酸、己二烯二酸等;或(2)当母体化合物中存在的酸性质子被金属离子如碱金属离子、碱土离子或铝离子取代时形成的盐;或者与有机碱如乙醇胺、二乙胺胺、三乙醇胺、N-甲基葡糖胺等形成的配合物。" Pharmaceutically acceptable salt " refers to a salt of a compound of the present invention that is pharmaceutically acceptable and possesses the desired pharmacological activity of the parent compound. These salts include: (1) acid addition salts, which are formed with inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, etc.; or with organic acids such as acetic acid, propionic acid ,, caproic acid, cyclopentanepropionic acid, glycolic acid, pyruvic acid, lactic acid, malonic acid, succinic acid, malic acid, maleic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, 3-(4- hydroxybenzoyl)benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, 1,2-ethanedisulfonic acid, 2-hydroxyethanesulfonic acid, benzenesulfonic acid, 4-chlorobenzenesulfonic acid, 2 -Naphthalenesulfonic acid, 4-toluenesulfonic acid, camphorsulfonic acid, 4-methylbicyclo 2.2.2-oct-2-ene-1-carboxylic acid, glucoheptonic acid, 3-phenylpropionic acid, trimethylacetic acid , tert-butylacetic acid, lauryl sulfate, gluconic acid, glutamic acid, hydroxynaphthoic acid, salicylic acid, stearic acid, adienedioic acid, etc.; or (2) when the acidic protons present in the parent compound are Salts formed when metal ions such as alkali metal ions, alkaline earth ions or aluminum ions are substituted; or complexes formed with organic bases such as ethanolamine, diethylamineamine, triethanolamine, N-methylglucamine, etc.
“ 药学上可接受的赋形剂”指与本发明的化合物一起施用的稀释剂、助剂、赋形剂或载体。" Pharmaceutically acceptable excipient " refers to a diluent, adjuvant, excipient or carrier with which a compound of the present invention is administered.
“ 患者”包括人。术语“人”和“患者”在本文中可以互换使用。“ 预防(Dreventing或prevention)”指得疾病或病症的风险降低(即使得至少一种临床疾病症状不在接触或易患疾病但尚未经历或表现疾病的症状的患者身上形成)。" Patient " includes humans. The terms "human" and "patient" are used interchangeably herein. " Dreventing or prevention" refers to reducing the risk of a disease or disorder (ie, such that at least one clinical symptom of disease does not develop in patients exposed to or susceptible to the disease but who have not yet experienced or exhibited symptoms of the disease).
“ 前药”指需要在体内转化释放活性药物的药物分子的衍生物。前药通常(虽然不是必需)在转化成母体药物之前无药理学活性。" Prodrug " refers to a derivative of a drug molecule that requires transformation in vivo to release the active drug. Prodrugs are usually, though not necessarily, pharmacologically inactive until converted to the parent drug.
“ 基元”指一种保护基形式,该保护基在用于屏蔽药物分子内的官能团时将药物转化为前药。通常基元通过键与药物结合,该键在体内被酶促或非酶促方式裂解。" Motive " refers to a form of protecting group that, when used to mask a functional group within a drug molecule, converts a drug into a prodrug. Typically the motif is bound to the drug by a bond that is cleaved in vivo either enzymatically or non-enzymatically.
“ 保护基”指一组原子,它在与分子屏蔽物中的反应性官能团结合时,降低或防止官能团的反应性。保护基的实例可以见于Green等人,″Protective Groups in Organic Chemistry(有机化学中的保护基)″,(Wiley,2nd ED.1991)和Harrison等人,″Compendium ofSynthetic Organic Methods(有机合成方法简述)″,第1-8卷(JohnWiley和Sons,1971-1996)。代表性的氨基保护基包括但不限于甲酰基、乙酰基、三氟乙酰基、苄基、苄氧基羰基(″CBZ″)、叔丁氧基羰基(″Boc″)、三甲基甲硅烷基(″TMS″)、2-三甲基甲硅烷基-乙磺酰基(″SES″)、三苯甲基和取代的三苯甲基、烯丙基氧羰基、9-芴基甲基氧羰基(″FMOC″)、硝基-藜芦基氧羰基(″NVOC″)等。代表性的羟基保护基包括但不限于其中的羟基被酰基化或烷基化的基团如苄基,和三苯甲基醚以及烷基醚、四氢吡喃基醚、三烷基甲硅烷基醚和烯丙基醚。" Protecting group " refers to a group of atoms which, when bound to a reactive functional group in a molecular shield, reduces or prevents the reactivity of the functional group. Examples of protecting groups can be found in Green et al., "Protective Groups in Organic Chemistry (Protective Groups in Organic Chemistry)", (Wiley, 2nd ED. 1991) and Harrison et al., "Compendium of Synthetic Organic Methods , 1-8 (John Wiley and Sons, 1971-1996). Representative amino protecting groups include, but are not limited to, formyl, acetyl, trifluoroacetyl, benzyl, benzyloxycarbonyl ("CBZ"), tert-butoxycarbonyl ("Boc"), trimethylsilyl ("TMS"), 2-trimethylsilyl-ethanesulfonyl ("SES"), trityl and substituted trityl, allyloxycarbonyl, 9-fluorenylmethyloxy carbonyl ("FMOC"), nitro-veratryloxycarbonyl ("NVOC"), and the like. Representative hydroxy protecting groups include, but are not limited to, groups in which the hydroxy group is acylated or alkylated such as benzyl, and trityl ethers as well as alkyl ethers, tetrahydropyranyl ethers, trialkylsilanes base ethers and allyl ethers.
“取代的”指其中的一个或多个氢原子各自独立地被相同或不同的取代基取代的基团。典型的取代基包括但不限于-X、-R29、=O-、-OR29、-SR29、-S-、=S、-NR29R30、=NR29、-CX3、-CF3、-CN、-OCN、-SCN、-NO、-NO2、=N2、-N3、-S(O)2O、-S(O)2OH、-S(O)2R29、-OS(O2)O-、-OS(O)2R29、-P(O)(O-)2、-P(O)(OR29)(O-)、-OP(O)(OR29)(OR30)、-C(O)R29、-C(S)R29、-C(O)OR29、-C(O)NR29R30、-C(O)O-、-C(S)OR29、-NR31C(O)NR29R30、-NR31C(S)NR29R30、-NR31C(NR29)NR29R30和-C(NR29)NR29R30,其中各个X独立地为卤素;各个R29和R30独立地为氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基、取代的杂芳基烷基、-NR31R32、-C(O)R31或-S(O)2R31,或者任选R29和R30与它们同时结合的原子一起形成环杂烷基或取代的环杂烷基环;和R31和R32独立地为氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基或取代的杂芳基烷基。"Substituted" refers to groups in which one or more hydrogen atoms are each independently replaced by the same or different substituents. Typical substituents include but are not limited to -X, -R 29 , =O - , -OR 29 , -SR 29 , -S - , =S, -NR 29 R 30 , =NR 29 , -CX 3 , -CF 3 , -CN, -OCN, -SCN, -NO, -NO 2 , =N 2 , -N 3 , -S(O) 2 O, -S(O) 2 OH, -S(O) 2 R 29 , -OS(O 2 )O - , -OS(O) 2 R 29 , -P(O)(O - ) 2 , -P(O)(OR 29 )(O - ), -OP(O)( OR 29 )(OR 30 ), -C(O)R 29 , -C(S)R 29 , -C(O)OR 29 , -C(O)NR 29 R 30 , -C(O)O-, -C(S)OR 29 , -NR 31 C(O)NR 29 R 30 , -NR 31 C(S)NR 29 R 30 , -NR 31 C(NR 29 )NR 29 R 30 and -C(NR 29 ) NR 29 R 30 , wherein each X is independently halogen; each R 29 and R 30 is independently hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted aryl Alkyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl , substituted heteroarylalkyl, -NR 31 R 32 , -C(O)R 31 or -S(O) 2 R 31 , or optionally R 29 and R 30 together with the atoms to which they are simultaneously bonded form a ring hetero Alkyl or substituted cycloheteroalkyl ring; and R and R are independently hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, Cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, or substituted Heteroarylalkyl.
“ 转运蛋白”指在将分子转运进入和/或通过细胞过程中起直接或间接作用的蛋白。例如,转运蛋白可以是但不限于溶质携带转运剂、共同转运剂、反向转运剂(counter transporter)、单一转运剂、协同转运剂、对向转运剂(antiporter)、泵、平衡转运剂、集中转运剂和其它蛋白,它们调节活性转运、依赖能量的转运、促进扩散、交换机制和特定的吸收机制。转运蛋白还可以是但不限于识别底物并通过载体调节的转运剂或受体调节的转运剂影响其进入和离开细胞的膜结合蛋白。转运蛋白还可以是但不限于参与通过将底物转运通过或离开细胞的细胞内表达的蛋白。转运蛋白还可以是但不限于不直接转运底物但与底物结合将其保持在受体附近的暴露在细胞表面的蛋白或糖蛋白,或者影响底物进入或通过细胞的转运蛋白。载体蛋白实例包括:肠和肝胆汁酸转运剂、二肽转运剂、寡肽转运剂、单糖转运剂(例如SGLT1)、磷酸盐转运剂、一羧酸转运剂、β-糖蛋白转运剂、有机阴离子转运剂(OAT)和有机阳离子转运剂。受体调节的转运蛋白的实例包括:病毒受体、免疫球蛋白受体、细菌毒素受体、植物凝集素受体、细菌粘附受体、维生素转运剂和细胞因子生长因子受体。" Transporter " refers to a protein that plays a direct or indirect role in the transport of molecules into and/or through a cell. For example, a transporter can be, but is not limited to, a solute-carrying transporter, a co-transporter, a counter transporter, a mono-transporter, a co-transporter, an antiporter, a pump, an equilibrium transporter, a concentrating Transport agents and other proteins that regulate active transport, energy-dependent transport, facilitated diffusion, exchange mechanisms, and specific uptake mechanisms. A transporter can also be, but is not limited to, a membrane-bound protein that recognizes a substrate and affects its entry and exit from a cell via a carrier-mediated transporter or a receptor-regulated transporter. A transport protein can also be, but is not limited to, an intracellularly expressed protein involved in transporting a substrate through or out of the cell. A transporter can also be, but is not limited to, a cell surface exposed protein or glycoprotein that does not directly transport a substrate but binds to the substrate to keep it in the vicinity of a receptor, or a transporter that affects the entry or passage of a substrate into or through a cell. Examples of carrier proteins include: intestinal and hepatic bile acid transporters, dipeptide transporters, oligopeptide transporters, monosaccharide transporters (eg SGLT1), phosphate transporters, monocarboxylic acid transporters, beta-glycoprotein transporters, Organic anion transporters (OATs) and organic cation transporters. Examples of receptor-regulated transporters include: viral receptors, immunoglobulin receptors, bacterial toxin receptors, plant lectin receptors, bacterial adhesion receptors, vitamin transporters, and cytokine growth factor receptors.
任何疾病或病症的“ 治疗(treating或treatment)”在一个实施方案中指改善疾病或病症(即停止或减少疾病或它的至少一种临床症状的发展)。在另一个实施方案中,“治疗(treating或treatment)”指改善至少一个患者可能不可辨别的身体参数。在进一步的另一个实施方案中,“治疗(treating或treatment)”指抑制在身体上(例如稳定可辨别的症状),生理上(例如稳定物理参数)或同时抑制疾病或病症。在进一步的另一个实施方案中,“治疗(treating或treatment)”指延迟疾病或病症的发生。" Treating or treatment " of any disease or condition refers, in one embodiment, to ameliorating the disease or condition (ie, halting or reducing the development of the disease or at least one of its clinical symptoms). In another embodiment, "treating" or "treatment" refers to improving at least one physical parameter that may not be discernible in a patient. In yet another embodiment, "treating or treatment" refers to inhibiting a disease or condition either physically (eg, stabilizing discernible symptoms), physiologically (eg, stabilizing a physical parameter), or simultaneously. In yet another embodiment, "treating" refers to delaying the onset of a disease or condition.
“ 治疗有效量”意指化合物在施用于患者以治疗疾病时足以进行这种疾病的治疗的数量。“治疗有效量”将随化合物、疾病和它的严重程度以及待治疗的患者的年龄、体重等而变。" Therapeutically effective amount " means an amount of a compound which, when administered to a patient to treat a disease, is sufficient to effect treatment of the disease. The "therapeutically effective amount" will vary with the compound, the disease and its severity, and the age, weight, etc. of the patient to be treated.
现在更详细地参照本发明的优选的实施方案。虽然结合所述优选的实施方案描述本发明,但可以理解这并不意味着将本发明限定于这些优选的实施方案。相反,本申请旨在覆盖可以包括在所附的权利要求定义的本发明的构思和范围之内的替换、修改和等同的方案。Reference will now be made in more detail to the preferred embodiments of the invention. While the invention has been described in conjunction with the preferred embodiments, it will be understood that it is not intended to limit the invention to these preferred embodiments. On the contrary, this application is intended to cover alternatives, modifications and equivalents, which may be included within the spirit and scope of the invention as defined by the appended claims.
4.2本发明的化合物 4.2 Compounds of the invention
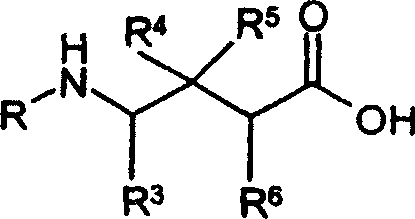
本领域技术人员将认识到式(I)、(II)和(III)的化合物在某些结构特征上具有共同点。这些化合物都为已结合基元的GABA类似物(即γ-氨基丁酸衍生物)。具体地,R2、R3、R4、R5、R6、X和Y是式(I)、(II)和(III)的化合物中发现的共同取代基。Those skilled in the art will recognize that compounds of formulas (I), (II) and (III) have certain structural features in common. These compounds are all GABA analogs (ie derivatives of gamma-aminobutyric acid) that have incorporated motifs. In particular, R 2 , R 3 , R 4 , R 5 , R 6 , X and Y are common substituents found in compounds of formulas (I), (II) and (III).
本发明的化合物包括式(I)、式(II)或式(III)的化合物Compounds of the present invention include compounds of formula (I), formula (II) or formula (III)
或其药学上可接受的盐、水合物或溶剂化物,其中:or a pharmaceutically acceptable salt, hydrate or solvate thereof, wherein:
n、t、u、X、Y、R1、R2、R3、R4、R5、R6、R7、R20、R21、R22和R23如前述定义。n, t, u, X, Y, R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 20 , R 21 , R 22 and R 23 are as defined above.
在一个优选的实施方案中,式(I)、(II)和(III)的化合物不包括以下化合物:In a preferred embodiment, the compounds of formula (I), (II) and (III) do not include the following compounds:
当R3和R6都为氢时,R4和R5不都为氢或不都为甲基;When R 3 and R 6 are both hydrogen, R 4 and R 5 are not both hydrogen or not all methyl;
在式(I)的化合物中,当n为O或当n为1,而X为NR16时,则R1不为氢;In the compound of formula (I), when n is O or when n is 1, and X is NR 16 , then R 1 is not hydrogen;
在式(I)的化合物中,R1、R7O-、R24C(O)-、R25C(O)-和R25O-都不为由胆汁酸衍生的部分;In compounds of formula (I), none of R 1 , R 7 O-, R 24 C(O)-, R 25 C(O)- and R 25 O- is a bile acid-derived moiety;
在式(I)的化合物中,当R1为R24C(O)-而n为0时,R24不为甲基、叔丁基、2-氨基乙基、3-氨基丙基、苄基、苯基或2-(苯甲酰氧基甲基)苯基;In the compound of formula (I), when R 1 is R 24 C(O)- and n is 0, R 24 is not methyl, tert-butyl, 2-aminoethyl, 3-aminopropyl, benzyl phenyl, phenyl or 2-(benzoyloxymethyl)phenyl;
在式(I)的化合物中,当R1为R25OC(O)-时,R25不为R26C(O)CR13R14-,其中R26选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基;In the compound of formula (I), when R 1 is R 25 OC(O)-, R 25 is not R 26 C(O)CR 13 R 14 -, wherein R 26 is selected from hydrogen, alkyl, substituted Alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl and substituted heteroarylalkyl;
在式(I)的化合物中,当R1为R25OC(O)-而n为0时,R25不为甲基、叔丁基或苄基;In the compound of formula (I), when R 1 is R 25 OC (O)- and n is 0, R 25 is not methyl, tert-butyl or benzyl;
在式(I)的化合物中,当n为0而R1为R25C(O)OCR13R14OC(O)-时,如果R13或R14之任一为氢、烷氧基羰基、取代的烷氧基羰基、氨基甲酰基、环烷氧基羰基或取代的环烷氧基羰基,则R13或R14中的另一个不为氢;In the compound of formula (I), when n is 0 and R 1 is R 25 C(O)OCR 13 R 14 OC(O)-, if either R 13 or R 14 is hydrogen, alkoxycarbonyl , substituted alkoxycarbonyl, carbamoyl, cycloalkoxycarbonyl or substituted cycloalkoxycarbonyl, then the other of R 13 or R 14 is not hydrogen;
在式(I)的化合物中,当n为1,X为NH,R3、R5和R6各自为氢,而R4为环己基时,则R2不为苄基;In the compound of formula (I), when n is 1, X is NH, R 3 , R 5 and R 6 are each hydrogen, and R 4 is cyclohexyl, then R 2 is not benzyl;
在式(II)的化合物中,当t为1,u为0时,R20或R21都不为2-羟基-3-甲基-5-氯苯基;和In a compound of formula (II), when t is 1 and u is 0, neither R nor R is 2 -hydroxyl-3- methyl -5-chlorophenyl; and
在式(II)的化合物中,当u为1而X为0时,t为1。In compounds of formula (II), t is 1 when u is 1 and X is 0.
在式(I)、(II)和(III)的化合物的一个实施方案中,当R3和R6各自为氢时,R4和R5不都为氢或不都为甲基。In one embodiment of the compounds of formula (I), (II) and (III), when R3 and R6 are each hydrogen, R4 and R5 are not both hydrogen or not both methyl.
在式(I)的化合物的一个实施方案中,当n为0或当n为1而X为NR16时,R1不为氢。在式(I)的化合物的另一个实施方案中,R1、R7O-、R24C(O)-、R25C(O)-或R25O-都不是由胆汁酸衍生的部分。在式(I)的化合物的另一个实施方案中,当R1为R24C(O)-而n为0时,R24不为烷基、取代的烷基、芳基烷基、芳基或取代的芳基。在式(I)的化合物的另一个实施方案中,当R1为R24C(O)-而n为0时,则R24不为C1-4链烷基、苄基、苯基或取代的苯基。在式(I)的化合物的另一个实施方案中,当R1为R24C(O)-而n为0时,R24不为甲基、叔丁基、2-氨基乙基、3-氨基丙基、苄基、苯基或2-(苯甲酰氧基甲基)-苯基。在式(I)的化合物的另一个实施方案中,当R1为R25OC(O)-时,R25不为R26C(O)CR13R14-。在式(I)的化合物的另一个实施方案中,当R1为R25OC(O)-而n为0时,R25不为烷基或芳基烷基。在式(I)的化合物的另一个实施方案中,当R1为R25OC(O)-而n为0时,R25不为C1-4链烷基或苄基。在式(I)的化合物的另一个实施方案,当R1为R25OC(O)-而n为0,R25不为甲基、叔丁基或苄基。在式(I)的化合物的另一个实施方案中,当n为0而R1为R25C(O)OCR13R14OC(O)-时,如果R13或R14之任一为氢、烷氧基羰基、取代的烷氧基羰基、氨基甲酰基、环烷氧基羰基或取代的环烷氧基羰基,则R13或R14中的另一个不为氢。在式(I)的化合物的另一个实施方案中,当R3、R5和R6各自为氢时,R4不为环己基。在式(I)的化合物的另一个实施方案中,当n为1,X为NH,R3、R5、R6各自为氢而R2为苄基时,R4不为环己基。In one embodiment of the compounds of formula (I), R 1 is other than hydrogen when n is 0 or when n is 1 and X is NR 16 . In another embodiment of the compounds of formula (I), none of R 1 , R 7 O-, R 24 C(O)-, R 25 C(O)- or R 25 O- is a moiety derived from a bile acid . In another embodiment of the compounds of formula (I), when R 1 is R 24 C(O)— and n is 0, R 24 is not alkyl, substituted alkyl, arylalkyl, aryl or substituted aryl. In another embodiment of the compound of formula (I), when R 1 is R 24 C(O)- and n is 0, then R 24 is not C 1-4 alkanyl, benzyl, phenyl or Substituted phenyl. In another embodiment of the compounds of formula (I), when R 1 is R 24 C(O)- and n is 0, R 24 is not methyl, tert-butyl, 2-aminoethyl, 3- Aminopropyl, benzyl, phenyl or 2-(benzoyloxymethyl)-phenyl. In another embodiment of the compounds of formula (I), when R 1 is R 25 OC(O)—, R 25 is other than R 26 C(O)CR 13 R 14 —. In another embodiment of the compounds of formula (I), when R 1 is R 25 OC(O)— and n is 0, R 25 is not alkyl or arylalkyl. In another embodiment of the compounds of formula (I), when R 1 is R 25 OC(O)— and n is 0, R 25 is other than C 1-4 alkanyl or benzyl. In another embodiment of the compounds of formula (I), when R 1 is R 25 OC(O)- and n is 0, R 25 is other than methyl, tert-butyl or benzyl. In another embodiment of the compounds of formula (I), when n is 0 and R 1 is R 25 C(O)OCR 13 R 14 OC(O)—, if either R 13 or R 14 is hydrogen , alkoxycarbonyl, substituted alkoxycarbonyl, carbamoyl, cycloalkoxycarbonyl or substituted cycloalkoxycarbonyl, then the other of R or R is not hydrogen. In another embodiment of the compounds of formula (I), when R3 , R5 and R6 are each hydrogen, R4 is not cyclohexyl. In another embodiment of the compounds of formula (I), when n is 1, X is NH, R3 , R5 , R6 are each hydrogen and R2 is benzyl, R4 is other than cyclohexyl.
在式(II)的化合物的一个实施方案中,R20和R21都不为2-羟基-3-甲基-5-氯苯基。在式(II)的化合物的一个实施方案中,当u为1而X为0时,t为1。In one embodiment of the compound of formula (II), neither R20 nor R21 is 2-hydroxy-3-methyl-5-chlorophenyl. In one embodiment of the compound of formula (II), t is 1 when u is 1 and X is 0.
在式(I)、(II)和(III)的化合物的一个实施方案中,n为0。在另一个实施方案中,n为1。当n为1,而X为NR6时,优选α-氨基酸为L-立体化学构型。In one embodiment of the compounds of formula (I), (II) and (III), n is 0. In another embodiment, n is 1. When n is 1 and X is NR6 , the alpha-amino acid is preferably in the L-stereochemical configuration.
在式(I)和(II)的化合物的另一个实施方案中,R7选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基链烷基、取代的芳基链烷基、环烷基、取代的环烷基、环杂链烷基和取代的环杂链烷基。在一个优选的实施方案中,Y为O而R7为氢。在另一个实施方案中,Y为O而R7为链烷基、取代的链烷基、链烯基、取代的链烯基、芳基或取代的芳基。优选R7为甲基、乙基、苄基、-C(CH3)=CH2、-CH2C(O)N(CH3)2、In another embodiment of the compounds of formula (I) and (II), R is selected from the group consisting of hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkanyl, substituted aryl Alkanyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyi and substituted cycloheteroalkyi. In a preferred embodiment, Y is O and R7 is hydrogen. In another embodiment, Y is O and R is alkanyl , substituted alkanyl, alkenyl, substituted alkenyl, aryl or substituted aryl. Preferably R 7 is methyl, ethyl, benzyl, -C(CH 3 )=CH 2 , -CH 2 C(O)N(CH 3 ) 2 ,
或 or
其中V为O或CH2。wherein V is O or CH 2 .
在式(I)、(II)和(III)的化合物的一个优选的实施方案中,R2选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基。优选R2选自氢、链烷基、取代的链烷基、芳基、取代的芳基、芳基链烷基、取代的芳基链烷基、环烷基、杂芳基烷基和取代的杂芳基链烷基。In a preferred embodiment of the compounds of formula (I), (II) and (III), R is selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, Substituted arylalkyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, hetero Arylalkyl and substituted heteroarylalkyl. Preferably R is selected from the group consisting of hydrogen, alkanyl, substituted alkanyl, aryl, substituted aryl, arylalkanyl, substituted arylalkanyl, cycloalkyl, heteroarylalkyl and substituted The heteroaryl chain alkyl.
在式(I)、(II)和(III)的化合物的另一个实施方案中,X为NH,而R2为氢、环烷基或链烷基。优选R2为氢、甲基、异丙基、异丁基、仲丁基、叔丁基、环戊基或环己基。在另一个实施方案中,X为NH,而R2为取代的链烷基。优选R2为-CH2OH,-CH(OH)CH3、-CH2CO2H、-CH2CH2CO2H、-CH2CONH2、-CH2CH2CONH2、-CH2CH2SCH3,CH2SH、-CH2(CH2)3NH2或-CH2CH2CH2NHC(NH)NH2。在另一个实施方案中,X为NH,而R2选自芳基、芳基链烷基、取代的芳基链烷基和杂芳基链烷基。In another embodiment of the compounds of Formulas (I), (II) and (III), X is NH and R is hydrogen, cycloalkyl or alkanyl. Preferably R2 is hydrogen, methyl, isopropyl, isobutyl, sec-butyl, tert-butyl, cyclopentyl or cyclohexyl. In another embodiment, X is NH and R is substituted alkanyl. Preferably R 2 is -CH 2 OH, -CH(OH)CH 3 , -CH 2 CO 2 H, -CH 2 CH 2 CO 2 H, -CH 2 CONH 2 , -CH 2 CH 2 CONH 2 , -CH 2 CH2SCH3 , CH2SH , -CH2 ( CH2 ) 3NH2 or -CH2CH2CH2NHC (NH) NH2 . In another embodiment, X is NH and R is selected from aryl, arylalkanyl, substituted arylalkanyl, and heteroarylalkanyl.
优选R2为苯基、苄基、4-羟基苄基、4-溴苄基、2-咪唑基或2-吲哚基。在另一个实施方案中,X为NR16,而R2和R16和与它们结合的原子一起形成环杂烷基或取代的环杂烷基环。Preferably R2 is phenyl, benzyl, 4-hydroxybenzyl, 4-bromobenzyl, 2-imidazolyl or 2-indolyl. In another embodiment, X is NR 16 and R 2 and R 16 and the atoms to which they are joined together form a cycloheteroalkyl or substituted cycloheteroalkyl ring.
优选R2和R16和与它们结合的原子一起形成氮杂环丁烷、吡咯烷或哌啶环。Preferably R2 and R16 together with the atoms to which they are bound form an azetidine, pyrrolidine or piperidine ring.
在式(I)、(II)和(III)的化合物的另一个实施方案中,R3为氢。在另一个实施方案中,R6为氢。在另一个实施方案中,R3和R6独立地选自氢、烷基、取代的烷基、芳基、取代的芳基、环烷基和取代的环烷基。优选R3和R6独立地选自氢和链烷基。更优选R3为氢或链烷基,而R6为氢。In another embodiment of the compounds of formula (I), (II) and (III), R3 is hydrogen. In another embodiment, R6 is hydrogen. In another embodiment, R and R are independently selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, cycloalkyl, and substituted cycloalkyl. Preferably R3 and R6 are independently selected from hydrogen and alkanyl. More preferably R3 is hydrogen or alkanyl and R6 is hydrogen.
在式(I)、(II)和(III)的化合物的另一优选的实施方案中,R4和R5独立地选自氢、烷基、取代的烷基、环烷基、取代的环烷基、环杂烷基和取代的环杂烷基。优选R4和R5独立地选自氢、链烷基和取代的链烷基。In another preferred embodiment of the compounds of formula (I), (II) and (III), R and R are independently selected from hydrogen, alkyl, substituted alkyl, cycloalkyl, substituted ring Alkyl, cycloheteroalkyl, and substituted cycloheteroalkyl. Preferably R4 and R5 are independently selected from hydrogen, alkanyl and substituted alkanyl.
在式(I)、(II)和(III)的化合物的另一个实施方案中,R4和R5与它们结合的碳原子一起形成环烷基或取代的环烷基环。优选R4和R5和与它们结合的碳原子一起形成环丁基、取代的环丁基、环戊基、取代的环戊基、环己基或取代的环己基环。在另一个实施方案中,R4和R5和与它们结合的碳原子一起形成环杂烷基或取代的环杂烷基环。在另一个实施方案中,R4和R5与它们结合的碳原子一起形成桥连的环烷基环。In another embodiment of the compounds of formula (I), (II) and (III), R 4 and R 5 together with the carbon atom to which they are bound form a cycloalkyl or substituted cycloalkyl ring. Preferably R4 and R5 together with the carbon atom to which they are bound form a cyclobutyl, substituted cyclobutyl, cyclopentyl, substituted cyclopentyl, cyclohexyl or substituted cyclohexyl ring. In another embodiment, R4 and R5 together with the carbon atom to which they are bound form a cycloheteroalkyl or substituted cycloheteroalkyl ring. In another embodiment, R4 and R5 together with the carbon atoms to which they are bound form a bridged cycloalkyl ring.
在式(I)的化合物的一个实施方案中,n为1,R1为R24C(O)-或R24C(S)-,而R24为烷基、取代的烷基、杂烷基、取代的杂烷基、芳基、取代的芳基、杂芳基或取代的杂芳基。优选R24为甲基、乙基、2-丙基、叔丁基、-CH2OCH(CH3)2、苯基或3-吡啶基。In one embodiment of the compound of formula (I), n is 1, R 1 is R 24 C(O)- or R 24 C(S)-, and R 24 is alkyl, substituted alkyl, heteroalkane radical, substituted heteroalkyl, aryl, substituted aryl, heteroaryl, or substituted heteroaryl. Preferably R 24 is methyl, ethyl, 2-propyl, tert-butyl, —CH 2 OCH(CH 3 ) 2 , phenyl or 3-pyridyl.
在式(I)的化合物的另一个实施方案中,n为1,R1为R25OC(O)-或R25SC(O)-,而R25为烷基、取代的烷基、杂烷基、芳基、取代的芳基、杂芳基或取代的杂芳基。优选R25为乙基、2-丙基、新戊基、-CH2OCH(CH3)2、苯基或2-吡啶基。In another embodiment of the compounds of formula (I), n is 1, R 1 is R 25 OC(O)- or R 25 SC(O)-, and R 25 is alkyl, substituted alkyl, hetero Alkyl, aryl, substituted aryl, heteroaryl, or substituted heteroaryl. Preferably R 25 is ethyl, 2-propyl, neopentyl, —CH 2 OCH(CH 3 ) 2 , phenyl or 2-pyridyl.
式(I)的化合物的一个优选的实施方案包括式(IV)的化合物:A preferred embodiment of the compound of formula (I) includes the compound of formula (IV):
或其药学上可接受的盐、水合物或溶剂化物,其中:or a pharmaceutically acceptable salt, hydrate or solvate thereof, wherein:
n、Y、R2、R3、R4、R5、R6、R7、R13、R14、R16和R25如前述定义。n, Y, R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 13 , R 14 , R 16 and R 25 are as defined above.
在一个优选的实施方案中,式(IV)的化合物不包括以下化合物:当R13或R14为氢、烷氧基羰基、取代的烷氧基羰基、氨基甲酰基、环烷氧基羰基或取代的环烷氧基羰基时,R13或R14中的另一个不为氢;而R25C(O)不为由胆汁酸衍生的部分。In a preferred embodiment, the compound of formula (IV) does not include the following compounds: when R 13 or R 14 is hydrogen, alkoxycarbonyl, substituted alkoxycarbonyl, carbamoyl, cycloalkoxycarbonyl or In the case of substituted cycloalkoxycarbonyl, the other of R 13 or R 14 is not hydrogen; and R 25 C(O) is not a moiety derived from bile acid.
在式(IV)的化合物的一个实施方案中,R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、芳基、芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基或杂芳基(优选当R13为烷氧基羰基、环烷氧基羰基或氨基甲酰基时,R14为甲基)。更优选R13和R14独立地为氢、甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、环戊基、环己基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、异丁氧基羰基、仲丁氧基羰基、叔丁氧基羰基、环己氧基羰基、苯基、苄基、苯乙基或3-吡啶基。In one embodiment of the compound of formula (IV), R and R are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, aryl, arylalkyl, carbamoyl, cycloalkane radical, substituted cycloalkyl, cycloalkoxycarbonyl or heteroaryl (preferably when R 13 is alkoxycarbonyl, cycloalkoxycarbonyl or carbamoyl, R 14 is methyl). More preferably R and R are independently hydrogen, methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, cyclopentyl, cyclohexyl, methoxy Carbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl, cyclohexyloxycarbonyl, phenyl, Benzyl, phenethyl or 3-pyridyl.
在式(IV)的化合物的另一个实施方案中,R13和R14独立地为氢、链烷基、取代的链烷基、环烷基或取代的环烷基。优选R13和R14为氢、链烷基或环烷基。更优选R13和R14独立地为氢、甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、环戊基或环己基。甚至更优选R13为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、环戊基或环己基而R14为氢,或者R13为甲基而R14为甲基。In another embodiment of the compounds of formula (IV), R13 and R14 are independently hydrogen, alkanyl, substituted alkanyl, cycloalkyl or substituted cycloalkyl. Preferably R13 and R14 are hydrogen, alkanyl or cycloalkyl. More preferably R13 and R14 are independently hydrogen, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, cyclopentyl or cyclohexyl. Even more preferably R is methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, cyclopentyl or cyclohexyl and R is hydrogen , or R is methyl and R14 is methyl.
在式(IV)的化合物另一个实施方案中,R13和R14独立地为氢、芳基、芳基烷基或杂芳基。更优选R13和R14独立地为氢、苯基、苄基、苯乙基或3-吡啶基。甚至更优选R13为苯基、苄基、苯乙基或3-吡啶基和R14为氢。In another embodiment of the compounds of formula (IV), R13 and R14 are independently hydrogen, aryl, arylalkyl or heteroaryl. More preferably R13 and R14 are independently hydrogen, phenyl, benzyl, phenethyl or 3-pyridyl. Even more preferably R13 is phenyl, benzyl, phenethyl or 3-pyridyl and R14 is hydrogen.
在式(IV)的化合物另一个实施方案中,R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、氨基甲酰基或环烷氧基羰基。优选当R13为烷氧基羰基、环烷氧基羰基或氨基甲酰基时,R14为甲基。更优选R13为甲氧基羰基、乙氧基羰基、异丙氧基羰基、丁氧基羰基、异丁氧基羰基、仲丁氧基羰基、叔丁氧基羰基或环己氧基羰基,而R14为甲基。In another embodiment of the compounds of formula (IV), R13 and R14 are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, carbamoyl or cycloalkoxycarbonyl. Preferably when R 13 is alkoxycarbonyl, cycloalkoxycarbonyl or carbamoyl, R 14 is methyl. More preferably R is methoxycarbonyl, ethoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl or cyclohexyloxycarbonyl, And R 14 is methyl.
在式(IV)的化合物另一个实施方案中,R13和R14和与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。优选R13和R14和与它们结合的碳原子一起形成环烷基环。更优选R13和R14和与它们结合的碳原子一起形成环丁基、环戊基或环己基环。In another embodiment of the compounds of formula (IV), R 13 and R 14 and the carbon atom to which they are bound together form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. Preferably R 13 and R 14 together with the carbon atom to which they are bound form a cycloalkyl ring. More preferably R 13 and R 14 together with the carbon atom to which they are bound form a cyclobutyl, cyclopentyl or cyclohexyl ring.
在式(IV)的化合物另一个实施方案中,R25为酰基、取代的酰基、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基或取代的杂芳基烷基。优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基。更优选R25为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基或3-吡啶基。In another embodiment of the compound of formula (IV), R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, Cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, or substituted Heteroarylalkyl. Preferably R 25 is acyl, substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl. More preferably R is methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1,1-dimethyl Oxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-dioxan-2-yl )-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propyl, 1-( 1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3-dioxolane- 2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxybenzyl, 1 -(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy-2-benzene Ethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl)-2-phenylethyl, 1-(1,3-diox Alk-2-yl)-2-phenethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styrene , cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or 3-pyridyl.
在式(IV)的化合物另一个实施方案中,R25为酰基或取代的酰基。更优选R25为乙酰基、丙酰基、丁酰基、苯甲酰基或苯乙酰基。In another embodiment of the compounds of formula (IV), R 25 is acyl or substituted acyl. More preferably R 25 is acetyl, propionyl, butyryl, benzoyl or phenylacetyl.
在式(IV)的化合物另一个实施方案中,R25为链烷基或取代的链烷基。优选R25为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基或1-(1,3-二噁烷-2-基)-2-苯乙基。更优选R25为甲基、乙基、丙基、异丙基、丁基、1,1-二甲氧基乙基或1,1-二乙氧基乙基。In another embodiment of the compounds of Formula (IV), R 25 is alkanyl or substituted alkanyl. Preferably R is methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1,1-dimethoxy Diethyl ethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-dioxan-2-yl) -Ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propyl, 1-(1 , 3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3-dioxolane-2 -yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxybenzyl, 1- (1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy-2-phenylethyl 1,1-diethoxy-2-phenethyl, 1-(1,3-dioxolan-2-yl)-2-phenethyl or 1-(1,3-dioxane -2-yl)-2-phenylethyl. More preferably R 25 is methyl, ethyl, propyl, isopropyl, butyl, 1,1-dimethoxyethyl or 1,1-diethoxyethyl.
在式(IV)的化合物另一个实施方案中,R25为芳基、芳基烷基或杂芳基。优选R25为苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基或3-吡啶基。In another embodiment of the compounds of formula (IV), R 25 is aryl, arylalkyl or heteroaryl. Preferably R 25 is phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl or 3-pyridyl.
在式(IV)的化合物另一个实施方案中,R25为环烷基或取代的环烷基。更优选R25为环丙基、环丁基、环戊基或环己基。In another embodiment of the compounds of Formula (IV), R 25 is cycloalkyl or substituted cycloalkyl. More preferably R 25 is cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
在式(IV)的化合物另一个实施方案中,R25为酰基、取代的酰基、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基或取代的杂芳基烷基;而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、取代的烷氧基羰基、环烷氧基羰基、取代的环烷氧基羰基或氨基甲酰基时,R14为甲基)。优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、芳基、芳基烷基、氨基甲酰基、环烷基、环烷氧基羰基或杂芳基(优选当R13为烷氧基羰基、取代的烷氧基羰基、环烷氧基羰基、取代的环烷氧基羰基或氨基甲酰基时,R14为甲基)。更优选R25为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基或3-吡啶基,而R13和R14独立地为氢、甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、环戊基、环己基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、异丁氧基羰基、仲丁氧基羰基、叔丁氧基羰基、环己氧基羰基、苯基、苄基、苯乙基或3-吡啶基。甚至更优选R25为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、环己基或3-吡啶基,而R13和R14独立地为氢、甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、环戊基、环己基、甲氧基羰基、乙氧基羰基、异丙氧基羰基、环己氧基羰基、苯基、苄基、苯乙基或3-吡啶基。In another embodiment of the compound of formula (IV), R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, Cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, or substituted Heteroarylalkyl; while R and R are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, substituted alkoxycarbonyl, aryl , substituted aryl, arylalkyl, Substituted arylalkyl, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl, heteroaryl or substituted heteroaryl (preferably when R is When alkoxycarbonyl, substituted alkoxycarbonyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl or carbamoyl, R 14 is methyl). Preferably R is acyl , substituted acyl, alkyl, substituted alkyl, aryl , arylalkyl, cycloalkyl or heteroaryl, and R and R are independently hydrogen, alkyl, substituted Alkyl, alkoxycarbonyl, aryl, arylalkyl, carbamoyl, cycloalkyl, cycloalkoxycarbonyl or heteroaryl (preferably when R is alkoxycarbonyl, substituted alkoxycarbonyl , cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl or carbamoyl, R 14 is methyl). More preferably R is methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1,1-dimethyl Oxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-dioxan-2-yl )-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propyl, 1-( 1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3-dioxolane- 2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxybenzyl, 1 -(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy-2-benzene Ethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl)-2-phenylethyl, 1-(1,3-diox Alk-2-yl)-2-phenethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styrene base, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or 3-pyridyl, and R 13 and R 14 are independently hydrogen, methyl, ethyl, propyl, isopropyl, butyl, iso Butyl, sec-butyl, tert-butyl, cyclopentyl, cyclohexyl, methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl, cyclohexyloxycarbonyl, phenyl, benzyl, phenethyl or 3-pyridyl. Even more preferably R is methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl base, 1,1-dimethoxybenzyl, 1,1-diethoxybenzyl, 1,1-dimethoxy-2-phenethyl, 1,1-diethoxy-2- Phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, cyclohexyl or 3-pyridyl, and R13 and R are independently hydrogen, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, cyclopentyl, cyclohexyl, methoxycarbonyl, ethoxy ylcarbonyl, isopropoxycarbonyl, cyclohexyloxycarbonyl, phenyl, benzyl, phenethyl or 3-pyridyl.
在式(IV)的化合物另一个实施方案中,R25为酰基、取代的酰基、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基或取代的杂芳基烷基,而R13和R14和与它们结合的原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14与它们结合的原子一起形成环烷基或取代的环烷基环。更优选R25为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基或3-吡啶基中,而R13和R14和与它们结合的原子一起形成环丁基、环戊基或环己基环。In another embodiment of the compound of formula (IV), R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, Cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, or substituted Heteroarylalkyl, while R and R together with the atoms to which they are bonded form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. Preferably R25 is acyl, substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, while R13 and R14 together with the atoms to which they are bound form cycloalkyl or a substituted cycloalkyl ring. More preferably R is methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1,1-dimethyl Oxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-dioxan-2-yl )-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propyl, 1-( 1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3-dioxolane- 2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxybenzyl, 1 -(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy-2-benzene Ethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl)-2-phenylethyl, 1-(1,3-diox Alk-2-yl)-2-phenethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styrene In base, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or 3-pyridyl, and R 13 and R 14 and the atoms they are bound together form a cyclobutyl, cyclopentyl or cyclohexyl ring.
在式(IV)的化合物另一个实施方案中,R25为酰基或取代的酰基,而R14和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、取代的烷氧基羰基、环烷氧基羰基、取代的环烷氧基羰基或氨基甲酰基时,R14为甲基)。优选R25为乙酰基、丙酰基、丁酰基、苯甲酰基或苯乙酰基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、环烷氧基羰基或氨基甲酰基时,R14为甲基)。In another embodiment of the compounds of formula (IV), R is acyl or substituted acyl, and R and R are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, substituted alkoxy arylcarbonyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl , heteroaryl or substituted heteroaryl (preferably when R 13 is alkoxycarbonyl, substituted alkoxycarbonyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl or carbamoyl, R 14 is methyl). Preferably R is acetyl, propionyl, butyryl, benzoyl or phenylacetyl, and R and R are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, substituted alkoxy Carbonyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl, Heteroaryl or substituted heteroaryl (preferably when R 13 is alkoxycarbonyl, cycloalkoxycarbonyl or carbamoyl, R 14 is methyl).
在式(IV)的化合物另一个实施方案中,R25为链烷基或取代的链烷基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、取代的烷氧基羰基、环烷氧基羰基、取代的环烷氧基羰基或氨基甲酰基时,R14为甲基)。优选R25为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基或1-(1,3-二噁烷-2-基)-2-苯乙基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、环烷氧基羰基或氨基甲酰基时,R14为甲基)。In another embodiment of the compounds of formula (IV), R is alkanyl or substituted alkanyl, and R and R are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, Substituted alkoxycarbonyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted ring Alkoxycarbonyl, heteroaryl or substituted heteroaryl (preferably when R is alkoxycarbonyl, substituted alkoxycarbonyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl or carbamoyl , R 14 is methyl). Preferably R is methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1,1-dimethoxy Diethyl ethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-dioxan-2-yl) -Ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propyl, 1-(1 , 3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3-dioxolane-2 -yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxybenzyl, 1- (1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy-2-phenylethyl 1,1-diethoxy-2-phenethyl, 1-(1,3-dioxolan-2-yl)-2-phenethyl or 1-(1,3-dioxane -2-yl)-2-phenethyl, while R and R are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, substituted alkoxycarbonyl, aryl, substituted aryl , arylalkyl, substituted arylalkyl, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl, heteroaryl, or substituted heteroaryl (Preferably when R 13 is alkoxycarbonyl, cycloalkoxycarbonyl or carbamoyl, R 14 is methyl).
在式(IV)的化合物另一个实施方案中,R25为芳基、取代的芳基、芳基烷基、取代的芳基烷基、杂芳基或取代的杂芳基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、取代的烷氧基羰基、环烷氧基羰基、取代的环烷氧基羰基或氨基甲酰基时,R14为甲基)。优选R25为苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基或3-吡啶基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、环烷氧基羰基或氨基甲酰基时,R14为甲基)。In another embodiment of the compound of formula (IV), R 25 is aryl, substituted aryl, arylalkyl, substituted arylalkyl, heteroaryl, or substituted heteroaryl, and R 13 and R is independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, substituted alkoxycarbonyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, carbamoyl , cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl, heteroaryl or substituted heteroaryl (preferably when R 13 is alkoxycarbonyl, substituted alkoxy When carbonyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl or carbamoyl, R 14 is methyl). Preferably R 25 is phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl or 3-pyridyl, and R 13 and R 14 are independently hydrogen, alkyl, substituted alkyl, Alkoxycarbonyl, substituted alkoxycarbonyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxy Carbonyl, substituted cycloalkoxycarbonyl, heteroaryl or substituted heteroaryl (preferably when R 13 is alkoxycarbonyl, cycloalkoxycarbonyl or carbamoyl, R 14 is methyl).
在式(IV)的化合物另一个实施方案中,R25为环烷基或取代的环烷基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、取代的烷氧基羰基、环烷氧基羰基、取代的环烷氧基羰基或氨基甲酰基时,R14为甲基)。优选R25为环丙基、环丁基、环戊基或环己基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、氨基甲酰基、环烷基、取代的环烷基、环烷氧基羰基、取代的环烷氧基羰基、杂芳基或取代的杂芳基(优选当R13为烷氧基羰基、环烷氧基羰基或氨基甲酰基时,R14为甲基)。In another embodiment of the compounds of formula (IV), R is cycloalkyl or substituted cycloalkyl, and R and R are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, Substituted alkoxycarbonyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted ring Alkoxycarbonyl, heteroaryl or substituted heteroaryl (preferably when R is alkoxycarbonyl, substituted alkoxycarbonyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl or carbamoyl , R 14 is methyl). Preferably R is cyclopropyl , cyclobutyl, cyclopentyl or cyclohexyl, and R and R are independently hydrogen, alkyl, substituted alkyl, alkoxycarbonyl, substituted alkoxycarbonyl , Aryl, substituted aryl, arylalkyl, substituted arylalkyl, carbamoyl, cycloalkyl, substituted cycloalkyl, cycloalkoxycarbonyl, substituted cycloalkoxycarbonyl, heteroaryl radical or substituted heteroaryl (preferably when R 13 is alkoxycarbonyl, cycloalkoxycarbonyl or carbamoyl, R 14 is methyl).
在式(IV)的化合物另一个实施方案中,R25为酰基、取代的酰基、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基或取代的杂芳基烷基,而R13和R14独立地为氢、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基。优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14独立地为氢、链烷基、取代的链烷基、环烷基或取代的环烷基。更优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14独立地为氢、甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、环戊基或环己基。在以上实施方案中,R25优选为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基或3-吡啶基。In another embodiment of the compound of formula (IV), R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, Cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, or substituted Heteroarylalkyl, and R 13 and R 14 are independently hydrogen, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl, or heteroaryl. Preferably R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, and R and R are independently hydrogen, alkanyl, substituted Alkanyl, cycloalkyl or substituted cycloalkyl. More preferably R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, and R and R are independently hydrogen, methyl, ethyl propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, cyclopentyl or cyclohexyl. In the above embodiments, R is preferably methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1 , 1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-diox Alk-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propane base, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3- Dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxy Benzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy Base-2-phenethyl, 1,1-diethoxy-2-phenethyl, 1-(1,3-dioxolan-2-yl)-2-phenethyl, 1-(1 , 3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, benzene Ethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or 3-pyridyl.
在式(IV)的化合物另一个实施方案中,R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14独立地为氢、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基。优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14独立地为氢、芳基、芳基烷基或杂芳基。更优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14独立地为氢、苯基、苄基、苯乙基或3-吡啶基。在以上实施方案中,R25优选为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基或3-吡啶基。In another embodiment of the compound of formula (IV), R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, and R and R 14 is independently hydrogen, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl, or heteroaryl. Preferably R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, and R and R are independently hydrogen, aryl, aryl Alkyl or heteroaryl. More preferably R is acyl , substituted acyl, alkyl , substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, and R and R are independently hydrogen, phenyl, benzyl base, phenethyl or 3-pyridyl. In the above embodiments, R is preferably methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1 , 1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-diox Alk-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propane base, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3- Dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxy Benzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy Base-2-phenethyl, 1,1-diethoxy-2-phenethyl, 1-(1,3-dioxolan-2-yl)-2-phenethyl, 1-(1 , 3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, benzene Ethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or 3-pyridyl.
在式(IV)的化合物另一个实施方案中,R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14独立地为氢、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基。优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14独立地为氢、烷基、取代的烷基、烷氧基羰基、取代的烷氧基羰基、氨基甲酰基、环烷氧基羰基或取代的环烷氧基羰基(优选当R13为烷氧基羰基、取代的烷氧基羰基、氨基甲酰基、环烷氧基羰基或取代的环烷氧基羰基时,R14为甲基;更优选R13为甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、异丁氧基羰基、仲丁氧基羰基、叔丁氧基羰基或环己氧基羰基,而R14为甲基)。在以上实施方案中、R25优选为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基或3-吡啶基。In another embodiment of the compound of formula (IV), R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, and R and R 14 is independently hydrogen, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl, or heteroaryl. Preferably R is acyl , substituted acyl, alkyl, substituted alkyl, aryl , arylalkyl, cycloalkyl or heteroaryl, and R and R are independently hydrogen, alkyl, substituted Alkyl, alkoxycarbonyl, substituted alkoxycarbonyl, carbamoyl, cycloalkoxycarbonyl or substituted cycloalkoxycarbonyl (preferably when R is alkoxycarbonyl, substituted alkoxycarbonyl, When carbamoyl, cycloalkoxycarbonyl or substituted cycloalkoxycarbonyl, R 14 is methyl; more preferably R 13 is methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl , butoxycarbonyl, isobutoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl or cyclohexyloxycarbonyl, and R 14 is methyl). In the above embodiments, R is preferably methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1 , 1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-diox Alk-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propane base, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3- Dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxy Benzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy Base-2-phenethyl, 1,1-diethoxy-2-phenethyl, 1-(1,3-dioxolan-2-yl)-2-phenethyl, 1-(1 , 3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, benzene Ethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or 3-pyridyl.
在式(IV)的化合物另一个实施方案中,R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14和与它们结合的原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14和与它们结合的原子一起形成环烷基或取代的环烷基环。更优选R25为酰基、取代的酰基、烷基、取代的烷基、芳基、芳基烷基、环烷基或杂芳基,而R13和R14和与它们结合的原子一起形成环丁基、环戊基或环己基环。在以上实施方案中、R25优选为甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基或3-吡啶基。In another embodiment of the compound of formula (IV), R is acyl , substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, and R and R 14 and the atoms to which they are bonded together form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. Preferably R is acyl, substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, while R and R together with the atoms to which they are bound form a cycloalkane radical or substituted cycloalkyl ring. More preferably R is acyl, substituted acyl, alkyl, substituted alkyl, aryl, arylalkyl, cycloalkyl or heteroaryl, and R and R together with the atoms to which they are bound form a ring Butyl, cyclopentyl or cyclohexyl rings. In the above embodiments, R is preferably methyl , ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, isopentyl, sec-pentyl, neopentyl, 1 , 1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)-ethyl, 1-(1,3-diox Alk-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3-dioxolan-2-yl)-propane base, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethoxybutyl, 1-(1,3- Dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethoxybenzyl, 1,1-diethoxy Benzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl)-benzyl, 1,1-dimethoxy Base-2-phenethyl, 1,1-diethoxy-2-phenethyl, 1-(1,3-dioxolan-2-yl)-2-phenethyl, 1-(1 , 3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, benzene Ethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or 3-pyridyl.
在式(I)和(III)的化合物的另一个实施方案中,R1为In another embodiment of the compounds of formula (I) and (III), R is
m为0,而R8、R11和R12如前面定义。m is 0, and R 8 , R 11 and R 12 are as defined above.
在式(I)和(III)的化合物的一个实施方案中,R11为酰基、烷氧基羰基、芳氧基羰基、环烷氧基羰基或氨基甲酰基,R8为氢、烷氧基羰基、烷基、芳基、芳基烷基或氰基,而R12为氢、烷氧基羰基、烷基、取代的烷基、芳基或芳基烷基。In one embodiment of the compounds of formula (I) and (III), R is acyl , alkoxycarbonyl, aryloxycarbonyl, cycloalkoxycarbonyl or carbamoyl, R is hydrogen, alkoxy carbonyl, alkyl, aryl, arylalkyl or cyano, and R is hydrogen, alkoxycarbonyl, alkyl, substituted alkyl, aryl or arylalkyl.
在式(I)和(III)的化合物的另一个实施方案中,R11选自乙酰基、丙酰基、丁酰基、异丁酰基、新戊酰基、环戊烷羰基、环己烷羰基、苯甲酰基、苯乙酰基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、异丁氧基羰基、仲丁氧基羰基、叔丁氧基羰基、环戊基氧羰基、环己氧基羰基、苯氧基羰基、苄氧基羰基、氨基甲酰基、N-甲基氨基甲酰基、N-乙基氨基甲酰基、N-丙基氨基甲酰基、N-异丙基氨基甲酰基、N-丁基氨基甲酰基、N-异丁基氨基甲酰基、N-仲丁基氨基甲酰基、N-叔丁基氨基甲酰基、N-环戊基氨基甲酰基、N-环己基氨基甲酰基、N-苯基氨基甲酰基、N-苄基氨基甲酰基、N,N-二甲基氨基甲酰基、N,N-二乙基氨基甲酰基、N,N-二丙基氨基甲酰基、N,N-二异丙基氨基甲酰基、N,N-二丁基氨基甲酰基、N,N-二苄基氨基甲酰基、N-吡咯烷基氨基甲酰基、N-哌啶基氨基甲酰基和N-吗啉基氨基甲酰基。更优选R11选自乙酰基、丙酰基、丁酰基、异丁酰基、环己烷羰基、苯甲酰基、苯乙酰基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、环己氧基羰基、苯氧基羰基、苄氧基羰基、氨基甲酰基、N-甲基氨基甲酰基、N-乙基氨基甲酰基、N-丙基氨基甲酰基、N-异丙基氨基甲酰基、N-苯基氨基甲酰基、N-苄基氨基甲酰基、N,N-二甲基氨基甲酰基、N,N-二乙基氨基甲酰基、N,N-二丙基氨基甲酰基、N-吡咯烷基氨基甲酰基、N-哌啶基氨基甲酰基和N-吗啉基氨基甲酰基。In another embodiment of the compounds of formula (I) and (III), R is selected from acetyl, propionyl, butyryl, isobutyryl, pivaloyl, cyclopentanecarbonyl, cyclohexanecarbonyl, benzene Formyl, phenylacetyl, methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl , cyclopentyloxycarbonyl, cyclohexyloxycarbonyl, phenoxycarbonyl, benzyloxycarbonyl, carbamoyl, N-methylcarbamoyl, N-ethylcarbamoyl, N-propylcarbamoyl , N-isopropylcarbamoyl, N-butylcarbamoyl, N-isobutylcarbamoyl, N-sec-butylcarbamoyl, N-tert-butylcarbamoyl, N-cyclopentyl Carbamoyl, N-cyclohexylcarbamoyl, N-phenylcarbamoyl, N-benzylcarbamoyl, N,N-dimethylcarbamoyl, N,N-diethylcarbamoyl, N,N-dipropylcarbamoyl, N,N-diisopropylcarbamoyl, N,N-dibutylcarbamoyl, N,N-dibenzylcarbamoyl, N-pyrrolidinyl Carbamoyl, N-piperidinylcarbamoyl and N-morpholinylcarbamoyl. More preferably R is selected from acetyl, propionyl, butyryl, isobutyryl, cyclohexanecarbonyl, benzoyl, phenylacetyl, methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxy ylcarbonyl, butoxycarbonyl, cyclohexyloxycarbonyl, phenoxycarbonyl, benzyloxycarbonyl, carbamoyl, N-methylcarbamoyl, N-ethylcarbamoyl, N-propylcarbamoyl Acyl, N-isopropylcarbamoyl, N-phenylcarbamoyl, N-benzylcarbamoyl, N,N-dimethylcarbamoyl, N,N-diethylcarbamoyl, N , N-dipropylcarbamoyl, N-pyrrolidinylcarbamoyl, N-piperidinylcarbamoyl and N-morpholinylcarbamoyl.
在式(I)和(III)的化合物的另一个实施方案中,R8选自氢、甲基、乙基、丙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、苯氧基羰基、苄氧基羰基和氰基。更优选R8选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基。In another embodiment of the compounds of formula (I) and (III), R is selected from hydrogen, methyl, ethyl, propyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxy ylcarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, phenoxycarbonyl, benzyloxycarbonyl and cyano. More preferably R is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl.
在式(I)和(III)的化合物的另一个实施方案中,R12选自氢、甲基、乙基、丙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、苯氧基羰基和苄氧基羰基。更优选R12选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基。In another embodiment of the compounds of formula (I) and (III), R is selected from hydrogen, methyl, ethyl, propyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxy oxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, phenoxycarbonyl and benzyloxycarbonyl. More preferably R12 is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl.
在式(I)和(III)的化合物的另一个实施方案中,R11选自氢、烷氧基羰基、烷基、取代的烷基、芳基、芳基烷基,而R8和R12与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。优选R11选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基,而R8和R12和与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。更优选R11为氢或甲基,而R8和R12和与它们结合的碳原子一起形成环戊-1-烯、环己-1-烯、2-环戊烯-1-酮、2-环己烯-1-酮、2-(5H)-呋喃酮或5,6-二氢-吡喃-2-酮环。In another embodiment of the compounds of formula (I) and (III), R is selected from hydrogen, alkoxycarbonyl, alkyl, substituted alkyl, aryl, arylalkyl, and R and R 12 together with the carbon atoms to which they are bound form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. Preferably R11 is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl, while R8 and R12 and the carbon to which they are bound The atoms are taken together to form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. More preferably R 11 is hydrogen or methyl, and R 8 and R 12 form cyclopent-1-ene, cyclohex-1-ene, 2-cyclopenten-1-one, 2 - cyclohexen-1-one, 2-(5H)-furanone or 5,6-dihydro-pyran-2-one ring.
在式(I)和(III)的化合物的另一个实施方案中,R12选自氢、烷氧基羰基、烷基、取代的烷基、芳基、芳基烷基,而R8和R11与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。优选R12选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基,而R8和R11与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。更优选R12选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基,而R8和R11与它们结合的碳原子一起形成γ-丁内酯、δ-戊内酯或2,2-二甲基-1,3-二噁烷-4,6-二酮环。In another embodiment of the compounds of formula (I) and (III), R is selected from hydrogen, alkoxycarbonyl, alkyl, substituted alkyl, aryl, arylalkyl, and R and R 11 together with the carbon atoms to which they are bound form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. Preferably R 12 is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl, and R 8 and R 11 are bound to their carbon atoms taken together to form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. More preferably R 12 is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl, and R 8 and R 11 are bound to their carbon The atoms together form a gamma-butyrolactone, delta-valerolactone or 2,2-dimethyl-1,3-dioxane-4,6-dione ring.
在式(I)和(III)的化合物的另一个实施方案中、R1为In another embodiment of the compounds of formula (I) and (III), R is
而R15选自烷基、取代的烷基、环烷基、取代的环烷基、芳基、取代的芳基、杂芳基和取代的杂芳基。优选R15为甲基、乙基、丙基、异丙基、环戊基、环己基、苯基、4-羟基苯基、苄基、4-羟基苄基或3-吡啶基。And R is selected from the group consisting of alkyl, substituted alkyl, cycloalkyl, substituted cycloalkyl, aryl, substituted aryl, heteroaryl and substituted heteroaryl. Preferably R 15 is methyl, ethyl, propyl, isopropyl, cyclopentyl, cyclohexyl, phenyl, 4-hydroxyphenyl, benzyl, 4-hydroxybenzyl or 3-pyridyl.
在式(I)和(III)的另一个实施方案中,R1为In another embodiment of formulas (I) and (III), R is
其中R37为氢、烷基、取代的烷基、酰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、杂环烷基、取代的环杂烷基、杂芳基、取代的杂芳基、杂芳基烷基或取代的杂芳基烷基;wherein R is hydrogen, alkyl, substituted alkyl, acyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, heterocycloalkyl, substituted cycloheteroalkane radical, heteroaryl, substituted heteroaryl, heteroarylalkyl or substituted heteroarylalkyl;
Z为O、N或S;和Z is O, N or S; and
Ar为芳基、取代的芳基、杂芳基或取代的杂芳基。Ar is aryl, substituted aryl, heteroaryl or substituted heteroaryl.
优选Z和CH2OC(O)-彼此共轭相连(例如与六元环系统的1,4或1,2相关)。Preferably Z and CH2OC (O)- are conjugated to each other (for example in relation to 1,4 or 1,2 of a six-membered ring system).
在式(I)和(III)的另一个实施方案中,R1为In another embodiment of formulas (I) and (III), R is
或 or
其中q为0或1;where q is 0 or 1;
R38和R39独立地为氢、烷基、取代的烷基、环烷基、取代的烷基、芳基、取代的芳基、杂芳基和取代的杂芳基;R and R are independently hydrogen, alkyl, substituted alkyl, cycloalkyl, substituted alkyl, aryl, substituted aryl, heteroaryl , and substituted heteroaryl;
R40和R41独立地为氢、烷基、取代的烷基、环烷基、取代的烷基、芳基、取代的芳基、杂芳基和取代的杂芳基;或和与它们结合的碳原子一起形成环烷基环;R and R are independently hydrogen, alkyl, substituted alkyl, cycloalkyl, substituted alkyl, aryl, substituted aryl, heteroaryl, and substituted heteroaryl; or and combinations thereof carbon atoms together form a cycloalkyl ring;
R42和R43独立地为烷基、取代的烷基、环烷基、取代的烷基、芳基、取代的芳基、杂芳基和取代的芳基或和与它们结合的碳原子一起形成芳基、取代的芳基、杂芳基或取代的芳基环;R 42 and R 43 are independently alkyl, substituted alkyl, cycloalkyl, substituted alkyl, aryl, substituted aryl, heteroaryl, and substituted aryl or together with the carbon atom to which they are bound forming an aryl, substituted aryl, heteroaryl, or substituted aryl ring;
和R37如前面定义。and R37 are as defined above.
在式(I)-(IV)的化合物的一个优选的实施方案中,Y为O,R3、R6和R7为氢,而R4和R5与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、桥连的环烷基或取代的桥连的环烷基环。In a preferred embodiment of the compounds of formulas (I)-(IV), Y is O, R 3 , R 6 and R 7 are hydrogen, and R 4 and R 5 together with the carbon atoms to which they are bound form cycloalkane radical, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, bridged cycloalkyl, or substituted bridged cycloalkyl ring.
在式(I)-(IV)的化合物的另一个优选的实施方案中,R4和R5和与它们结合的碳原子一起形成环烷基或取代的环烷基环。在一个实施方案中,n为0,t为0而u为0。在另一个实施方案中,n为1,而R2为氢、甲基、2-丙基、2-丁基、异丁基、叔丁基、环戊基、环己基、苯基、苄基、4-羟基苄基、4-溴苄基、2-咪唑基、2-吲哚基、-CH2OH、-CH(OH)CH3、-CH2CO2H、-CH2CH2CO2H、-CH2CONH2、-CH2CH2CONH2、-CH2SCH3、CH2SH、-CH2(CH2)3NH2或-CH2CH2CH2NHC(NH)NH2。在另一个实施方案中,n为1,而R2和R16与它们结合的原子一起形成吡咯烷环。In another preferred embodiment of the compounds of formulas (I)-(IV), R 4 and R 5 together with the carbon atom to which they are bound form a cycloalkyl or substituted cycloalkyl ring. In one embodiment, n is 0, t is 0 and u is 0. In another embodiment n is 1 and R is hydrogen, methyl, 2-propyl, 2 -butyl, isobutyl, tert-butyl, cyclopentyl, cyclohexyl, phenyl, benzyl , 4-hydroxybenzyl, 4-bromobenzyl, 2-imidazolyl, 2-indolyl, -CH 2 OH, -CH(OH)CH 3 , -CH 2 CO 2 H, -CH 2 CH 2 CO 2 H, -CH 2 CONH 2 , -CH 2 CH 2 CONH 2 , -CH 2 SCH 3 , CH 2 SH, -CH 2 (CH 2 ) 3 NH 2 or -CH 2 CH 2 CH 2 NHC(NH)NH 2 . In another embodiment, n is 1 and R and R together with the atoms to which they are joined form a pyrrolidine ring.
在式(I)-(IV)的化合物的另一个优选的实施方案中,R4和R5和与它们结合的碳原子一起形成环丁基或取代的环丁基环。优选所述取代的环丁基环被一或多个选自以下的取代基取代:链烷基、取代的链烷基、卤素、羟基、羧基和烷氧基羰基。In another preferred embodiment of the compounds of formulas (I)-(IV), R 4 and R 5 and the carbon atom to which they are bound together form a cyclobutyl or substituted cyclobutyl ring. Preferably the substituted cyclobutyl ring is substituted with one or more substituents selected from the group consisting of alkanyl, substituted alkanyl, halogen, hydroxy, carboxyl and alkoxycarbonyl.
在式(I)-(IV)的化合物的另一个优选的实施方案中,R4和R5和与它们结合的碳原子一起形成环戊基或取代的环戊基环。优选所述环戊基环被链烷基、取代的链烷基、卤素、羟基、羧基或烷氧基羰基取代。更优选所述环戊基环被链烷基取代。甚至更优选所述环戊基环选自In another preferred embodiment of the compounds of formulas (I)-(IV), R 4 and R 5 and the carbon atom to which they are bound together form a cyclopentyl or substituted cyclopentyl ring. Preferably the cyclopentyl ring is substituted with alkanyl, substituted alkanyl, halo, hydroxy, carboxy or alkoxycarbonyl. More preferably the cyclopentyl ring is substituted with an alkanyl group. Even more preferably said cyclopentyl ring is selected from
和 and
优选在以上实施方案的一个更具体的版本中,R7为氢。Preferably in a more specific version of the above embodiment, R7 is hydrogen.
在式(I)-(IV)的化合物的另一个优选的实施方案中,R4和R5和与它们结合的碳原子一起形成环己基或取代的环己基环。优选所述环己基环被链烷基、取代的链烷基、卤素、羟基、羧基或烷氧基羰基取代。更优选所述环己基环被链烷基取代。甚至更优选所述环己基环选自In another preferred embodiment of the compounds of formulas (I)-(IV), R 4 and R 5 together with the carbon atom to which they are bound form a cyclohexyl or substituted cyclohexyl ring. Preferably the cyclohexyl ring is substituted with alkanyl, substituted alkanyl, halo, hydroxy, carboxy or alkoxycarbonyl. More preferably the cyclohexyl ring is substituted with an alkanyl group. Even more preferably said cyclohexyl ring is selected from
和 and
优选在以上实施方案的一个更具体的版本中,R7为氢。Preferably in a more specific version of the above embodiment, R7 is hydrogen.
在式(I)-(IV)的化合物的另一个优选的实施方案中,R4和R5与它们结合的碳原子一起形成环杂烷基或取代的环杂烷基环。In another preferred embodiment of the compounds of formulas (I)-(IV), R 4 and R 5 together with the carbon atom to which they are bound form a cycloheteroalkyl or substituted cycloheteroalkyl ring.
在一个实施方案中,n为0。在另一个实施方案中,n为1,而R2为氢、甲基、2-丙基、2-丁基、异丁基、叔丁基、环戊基、环己基、苯基、苄基、4-羟基苄基、4-溴苄基、2-咪唑基、2-吲哚基、-CH2OH、-CH(OH)CH3、-CH2CO2H、-CH2CH2CO2H、-CH2CONH2、-CH2CH2CONH2、-CH2CH2SCH3、CH2SH、-CH2(CH2)3NH2或-CH2CH2CH2NHC(NH)NH2。在另一个实施方案中,n为1,而R2和R16和与它们结合的原子一起形成吡咯烷环。优选R4和R5和与它们结合的碳原子一起形成环杂链烷基环。更优选所述环杂链烷基环选自In one embodiment, n is zero. In another embodiment n is 1 and R is hydrogen, methyl, 2-propyl, 2 -butyl, isobutyl, tert-butyl, cyclopentyl, cyclohexyl, phenyl, benzyl , 4-hydroxybenzyl, 4-bromobenzyl, 2-imidazolyl, 2-indolyl, -CH 2 OH, -CH(OH)CH 3 , -CH 2 CO 2 H, -CH 2 CH 2 CO 2 H, -CH 2 CONH 2 , -CH 2 CH 2 CONH 2 , -CH 2 CH 2 SCH 3 , CH 2 SH, -CH 2 (CH 2 ) 3 NH 2 or -CH 2 CH 2 CH 2 NHC(NH )NH 2 . In another embodiment, n is 1 and R and R together with the atoms to which they are bound form a pyrrolidine ring. Preferably R4 and R5 together with the carbon atom to which they are bound form a cycloheteroalkanyl ring. More preferably the cycloheteroalkanyl ring is selected from
或 or
其中Z为O、S(O)p或NR18 where Z is O, S(O) p or NR 18
P为0、1或2;和P is 0, 1 or 2; and
R18选自氢、烷基、取代的烷基、酰基和烷氧基羰基。更优选所述环杂链烷基环选自R 18 is selected from hydrogen, alkyl, substituted alkyl, acyl and alkoxycarbonyl. More preferably the cycloheteroalkanyl ring is selected from
和 and
优选地,在以上实施方案的一个更具体的版本中,R7为氢。Preferably, in a more specific version of the above embodiment, R 7 is hydrogen.
在式(I)-(IV)的化合物的另一个实施方案中,R4和R5和与它们结合的碳原子一起形成桥连的环烷基环。在一个实施方案中,n为0。在另一个实施方案中,n为1,而R2为氢、甲基、2-丙基、2-丁基、异丁基、叔丁基、环戊基、环己基、苯基、苄基、4-羟基苄基、4-溴苄基、2-咪唑基、2-吲哚基、-CH2OH、-CH(OH)CH3、-CH2CO2H、-CH2CH2CO2H、-CH2CONH2、-CH2CH2CONH2、-CH2CH2SCH3、CH2SH、-CH2(CH2)3NH2或-CH2CH2CH2NHC(NH)NH2。在另一个实施方案中,n为1和R2和R16与它们结合的原子一起形成吡咯烷环。优选所述桥连的环烷基为In another embodiment of the compounds of formulas (I)-(IV), R4 and R5 and the carbon atom to which they are bound together form a bridged cycloalkyl ring. In one embodiment, n is zero. In another embodiment n is 1 and R is hydrogen, methyl, 2-propyl, 2 -butyl, isobutyl, tert-butyl, cyclopentyl, cyclohexyl, phenyl, benzyl , 4-hydroxybenzyl, 4-bromobenzyl, 2-imidazolyl, 2-indolyl, -CH 2 OH, -CH(OH)CH 3 , -CH 2 CO 2 H, -CH 2 CH 2 CO 2 H, -CH 2 CONH 2 , -CH 2 CH 2 CONH 2 , -CH 2 CH 2 SCH 3 , CH 2 SH, -CH 2 (CH 2 ) 3 NH 2 or -CH 2 CH 2 CH 2 NHC(NH )NH 2 . In another embodiment, n is 1 and R2 and R16 together with the atoms to which they are bound form a pyrrolidine ring. Preferably the bridged cycloalkyl group is
或 or
优选在以上实施方案的一个更具体的版本中,R7为氢。Preferably in a more specific version of the above embodiment, R7 is hydrogen.
在式(I)-(IV)的化合物的另一个实施方案中,Y为O,R6和R7为氢,R4为烷基或环烷基,R5为氢或烷基,而R3为氢或烷基。在一个实施方案中,n为0。在另一个实施方案中,n为1和R2为氢、甲基、2-丙基、2-丁基、异丁基、叔丁基、环戊基、环己基、苯基、苄基、4-羟基苄基、4-溴苄基、2-咪唑基、2-吲哚基、-CH2OH、-CH(OH)CH3、-CH2CO2H、-CH2CH2CO2H、-CH2CONH2、-CH2CH2CONH2、-CH2CH2SCH3、CH2SH、-CH2(CH2)3NH2或-CH2CH2CH2NHC(NH)NH2。在另一个实施方案中,n为1和R2和R16与它们结合的原子一起形成吡咯烷环。优选R4为环烷基,R5为氢或甲基,而R3为氢或甲基。优选R3为氢,R4为异丁基而R5为氢。In another embodiment of the compounds of formulas (I)-(IV), Y is O, R and R are hydrogen, R is alkyl or cycloalkyl, R is hydrogen or alkyl, and R 3 is hydrogen or alkyl. In one embodiment, n is zero. In another embodiment, n is 1 and R is hydrogen, methyl, 2-propyl, 2-butyl, isobutyl, t-butyl, cyclopentyl, cyclohexyl, phenyl, benzyl, 4-Hydroxybenzyl, 4-bromobenzyl, 2-imidazolyl, 2-indolyl, -CH 2 OH, -CH(OH)CH 3 , -CH 2 CO 2 H, -CH 2 CH 2 CO 2 H, -CH 2 CONH 2 , -CH 2 CH 2 CONH 2 , -CH 2 CH 2 SCH 3 , CH 2 SH, -CH 2 (CH 2 ) 3 NH 2 or -CH 2 CH 2 CH 2 NHC(NH) NH2 . In another embodiment, n is 1 and R2 and R16 together with the atoms to which they are bound form a pyrrolidine ring. Preferably R4 is cycloalkyl, R5 is hydrogen or methyl, and R3 is hydrogen or methyl. Preferably R3 is hydrogen, R4 is isobutyl and R5 is hydrogen.
在式(I)-(IV)的化合物的另一个实施方案中,Y为O,R5和R7为氢或链烷基,R3和R6为氢,而R4为取代的杂烷基。In another embodiment of the compounds of formulas (I)-(IV), Y is O, R and R are hydrogen or alkanyl, R and R are hydrogen, and R is a substituted heteroalkane base.
优选R4为Preferably R4 is
A为NR19、O或S;A is NR 19 , O or S;
B为烷基、取代的烷基、烷氧基、卤素、羟基、羧基、烷氧基羰基或氨基;B is alkyl, substituted alkyl, alkoxy, halogen, hydroxy, carboxyl, alkoxycarbonyl, or amino;
R19为氢、烷基、环烷基或芳基;R 19 is hydrogen, alkyl, cycloalkyl or aryl;
j为0-4的整数;j is an integer of 0-4;
k为1-4的整数;和k is an integer from 1 to 4; and
l为0-3的整数。l is an integer of 0-3.
更优选k为1。More preferably, k is 1.
在式(I)-(IV)的化合物的另一个实施方案中,Y为O,R5和R7为氢或链烷基,R3和R6为氢,而R4为取代的链烷基、环烷基或取代的环烷基。优选R4选自In another embodiment of the compounds of formulas (I)-(IV), Y is O, R and R are hydrogen or alkanyl, R and R are hydrogen, and R is substituted alkane radical, cycloalkyl or substituted cycloalkyl. Preferably R is selected from
优选R4为Preferably R4 is
和 and
h为1-6的整数;和h is an integer from 1 to 6; and
i为0-6的整数。i is an integer of 0-6.
更优选h为1、2、3或4,而i为0或1。甚至更优选R4选自More preferably h is 1, 2, 3 or 4 and i is 0 or 1 . Even more preferably R is selected from
和 and
优选式(I)-(IV)的化合物由式(XIII)的GABA类似物衍生:Preferably compounds of formulas (I)-(IV) are derived from GABA analogs of formula (XIII):
其中式(XIII)的GABA类似物选自:Wherein the GABA analog of formula (XIII) is selected from:
1-氨基甲基-1-环己烷乙酸;1-Aminomethyl-1-cyclohexaneacetic acid;
1-氨基甲基-1-(3-甲基环己烷)乙酸;1-Aminomethyl-1-(3-methylcyclohexane)acetic acid;
1-氨基甲基-1-(4-甲基环己烷)乙酸;1-Aminomethyl-1-(4-methylcyclohexane)acetic acid;
1-氨基甲基-1-(4-异丙基环己烷)乙酸;1-Aminomethyl-1-(4-isopropylcyclohexane)acetic acid;
1-氨基甲基-1-(4-叔丁基环己烷)乙酸;1-Aminomethyl-1-(4-tert-butylcyclohexane)acetic acid;
1-氨基甲基-1-(3,3-二甲基环己烷)乙酸;1-aminomethyl-1-(3,3-dimethylcyclohexane)acetic acid;
1-氨基甲基-1-(3,3,5,5-四甲基环己烷)乙酸;1-aminomethyl-1-(3,3,5,5-tetramethylcyclohexane)acetic acid;
1-氨基甲基-1-环戊烷乙酸;1-Aminomethyl-1-cyclopentaneacetic acid;
1-氨基甲基-1-(3-甲基环戊烷)乙酸;1-Aminomethyl-1-(3-methylcyclopentane)acetic acid;
1-氨基甲基-1-(3,4-二甲基环戊烷)乙酸;1-aminomethyl-1-(3,4-dimethylcyclopentane)acetic acid;
7-氨基甲基-二环[2.2.1]庚-7-基乙酸;7-Aminomethyl-bicyclo[2.2.1]hept-7-ylacetic acid;
9-氨基甲基-二环[3.3.1]壬-9-基乙酸;9-Aminomethyl-bicyclo[3.3.1]non-9-ylacetic acid;
4-氨基甲基-4-(四氢吡喃-4-基)乙酸;4-Aminomethyl-4-(tetrahydropyran-4-yl)acetic acid;
3-氨基甲基-3-(四氢吡喃-3-基)乙酸;3-aminomethyl-3-(tetrahydropyran-3-yl)acetic acid;
4-氨基甲基-4-(四氢硫代吡喃-4-基)乙酸;4-Aminomethyl-4-(tetrahydrothiopyran-4-yl)acetic acid;
3-氨基甲基-3-(四氢硫代吡喃-3-基)乙酸;3-Aminomethyl-3-(tetrahydrothiopyran-3-yl)acetic acid;
3-氨基甲基-5-甲基-己酸;3-Aminomethyl-5-methyl-hexanoic acid;
3-氨基甲基-5-甲基-庚酸;3-Aminomethyl-5-methyl-heptanoic acid;
3-氨基甲基-5-甲基-辛酸;3-Aminomethyl-5-methyl-octanoic acid;
3-氨基甲基-5-甲基-壬酸;3-Aminomethyl-5-methyl-nonanoic acid;
3-氨基甲基-5-甲基-癸酸;3-Aminomethyl-5-methyl-decanoic acid;
3-氨基甲基-5-环丙基-己酸;3-Aminomethyl-5-cyclopropyl-hexanoic acid;
3-氨基甲基-5-环丁基-己酸;3-Aminomethyl-5-cyclobutyl-hexanoic acid;
3-氨基甲基-5-环戊基-己酸;3-Aminomethyl-5-cyclopentyl-hexanoic acid;
3-氨基甲基-5-环己基-己酸;3-Aminomethyl-5-cyclohexyl-hexanoic acid;
3-氨基甲基-5-苯基-己酸;3-Aminomethyl-5-phenyl-hexanoic acid;
3-氨基甲基-5-苯基-戊酸;3-Aminomethyl-5-phenyl-pentanoic acid;
3-氨基甲基-4-环丁基-丁酸;3-Aminomethyl-4-cyclobutyl-butanoic acid;
3-氨基甲基-4-环戊基-丁酸;3-Aminomethyl-4-cyclopentyl-butanoic acid;
3-氨基甲基-4-环己基-丁酸;3-Aminomethyl-4-cyclohexyl-butanoic acid;
3-氨基甲基-4-苯氧基-丁酸;3-Aminomethyl-4-phenoxy-butyric acid;
3-氨基甲基-5-苯氧基-己酸;和3-Aminomethyl-5-phenoxy-hexanoic acid; and
3-氨基甲基-5-苄基硫烷基-戊酸。3-Aminomethyl-5-benzylsulfanyl-pentanoic acid.
特别优选的式(I)的实施方案包括式(V)和(VI)的化合物:Particularly preferred embodiments of formula (I) include compounds of formulas (V) and (VI):
其中R1、R2、R7和R16如前面定义。wherein R 1 , R 2 , R 7 and R 16 are as defined above.
在式(V)和(VI)的化合物的一个实施方案中,n为0。在另一个实施方案中,n为1,而R2为氢、甲基、2-丙基、2-丁基、异丁基、叔丁基、环戊基、环己基、苯基、苄基、4-羟基苄基、4-溴苄基、2-咪唑基、2-吲哚基、-CH2OH、-CH(OH)CH3、-CH2CO2H、-CH2CH2CO2H、-CH2CONH2、-CH2CH2CONH2、-CH2CH2SCH3、-CH2SH、-CH2(CH2)3NH2或-CH2CH2CH2NHC(NH)NH2。优选在以上实施方案中,R7为氢。In one embodiment of the compounds of formula (V) and (VI), n is 0. In another embodiment n is 1 and R is hydrogen, methyl, 2-propyl, 2 -butyl, isobutyl, tert-butyl, cyclopentyl, cyclohexyl, phenyl, benzyl , 4-hydroxybenzyl, 4-bromobenzyl, 2-imidazolyl, 2-indolyl, -CH 2 OH, -CH(OH)CH 3 , -CH 2 CO 2 H, -CH 2 CH 2 CO 2 H, -CH 2 CONH 2 , -CH 2 CH 2 CONH 2 , -CH 2 CH 2 SCH 3 , -CH 2 SH, -CH 2 (CH 2 ) 3 NH 2 or -CH 2 CH 2 CH 2 NHC( NH) NH2 . Preferably in the above embodiments R7 is hydrogen.
在式(V)和(VI)的化合物的另一个实施方案中,n为1,R1为R24C(O)-或R24C(S)-;而R24为烷基、取代的烷基、杂烷基、芳基、取代的芳基、杂芳基或取代的杂芳基。优选R24为甲基、乙基、2-丙基、叔丁基、-CH2OCH(CH3)2、苯基或3-吡啶基。优选在此实施方案中,R7为氢、链烷基、取代的链烷基、链烯基、取代的链烯基、芳基或取代的芳基。更优选R7为氢、甲基、乙基、苄基、-C(CH3)=CH2、-CH2C(O)N(CH3)2、In another embodiment of the compounds of formula (V) and (VI), n is 1, R 1 is R 24 C(O)- or R 24 C(S)-; and R 24 is alkyl, substituted Alkyl, heteroalkyl, aryl, substituted aryl, heteroaryl, or substituted heteroaryl. Preferably R 24 is methyl, ethyl, 2-propyl, tert-butyl, —CH 2 OCH(CH 3 ) 2 , phenyl or 3-pyridyl. Preferably in this embodiment R7 is hydrogen, alkanyl, substituted alkanyl, alkenyl, substituted alkenyl, aryl or substituted aryl. More preferably R 7 is hydrogen, methyl, ethyl, benzyl, -C(CH 3 )=CH 2 , -CH 2 C(O)N(CH 3 ) 2 ,
或 or
其中V为O或CH2。wherein V is O or CH 2 .
最优选R7为氢。Most preferably R7 is hydrogen.
在式(V)和(VI)的化合物的另一个实施方案中,n为1,R1为R25OC(O)-或R25SC(O)-;而R25为烷基、取代的烷基、杂烷基、芳基、取代的芳基、杂芳基或取代的杂芳基。优选R25为乙基、2-丙基、新戊基、-CH2OCH(CH3)2、苯基或2-吡啶基。优选在此实施方案中R7为氢、链烷基、取代的链烷基、链烯基、取代的链烯基、芳基或取代的芳基。更优选R7为氢、甲基、乙基、苄基、-C(CH3)=CH2、-CH2C(O)N(CH3)2、In another embodiment of the compounds of formula (V) and (VI), n is 1, R 1 is R 25 OC(O)- or R 25 SC(O)-; and R 25 is alkyl, substituted Alkyl, heteroalkyl, aryl, substituted aryl, heteroaryl, or substituted heteroaryl. Preferably R 25 is ethyl, 2-propyl, neopentyl, —CH 2 OCH(CH 3 ) 2 , phenyl or 2-pyridyl. Preferably in this embodiment R7 is hydrogen, alkanyl, substituted alkanyl, alkenyl, substituted alkenyl, aryl or substituted aryl. More preferably R 7 is hydrogen, methyl, ethyl, benzyl, -C(CH 3 )=CH 2 , -CH 2 C(O)N(CH 3 ) 2 ,
或 or
其中V为O或CH2。wherein V is O or CH 2 .
最优选R7为氢。Most preferably R7 is hydrogen.
在式(V)和(VI)的化合物的另一个实施方案中、R1为In another embodiment of the compounds of formula (V) and (VI), R is
而R15选自烷基、取代的烷基、环烷基、取代的环烷基、芳基、取代的芳基、杂芳基和取代的杂芳基。And R is selected from the group consisting of alkyl, substituted alkyl, cycloalkyl, substituted cycloalkyl, aryl, substituted aryl, heteroaryl and substituted heteroaryl.
优选R15为甲基、乙基、丙基、异丙基、环戊基、环己基、苯基、4-羟基苯基、苄基、4-羟基苄基或3-吡啶基。在此实施方案的一个更具体的版本中,R7为氢、链烷基、取代的链烷基、链烯基、取代的链烯基、芳基或取代的芳基。更优选R7为氢、甲基、乙基、苄基,-C(CH3)=CH2、-CH2C(O)N(CH3)2、Preferably R 15 is methyl, ethyl, propyl, isopropyl, cyclopentyl, cyclohexyl, phenyl, 4-hydroxyphenyl, benzyl, 4-hydroxybenzyl or 3-pyridyl. In a more specific version of this embodiment, R is hydrogen, alkanyl, substituted alkanyl, alkenyl, substituted alkenyl, aryl, or substituted aryl. More preferably R 7 is hydrogen, methyl, ethyl, benzyl, -C(CH 3 )=CH 2 , -CH 2 C(O)N(CH 3 ) 2 ,
或 or
其中V为O或CH2。wherein V is O or CH 2 .
优选R7为氢。Preferably R7 is hydrogen.
式(V)和(VI)的化合物的特别优选的实施方案为选自以下的化合物:1-{[((5-甲基-2-氧代-1,3-二氧环戊-4-烯-4-基)甲氧基)羰基]氨基甲基}-1-环己烷乙酸和3-{[((5-甲基-2-氧代-1,3-二氧环戊-4-烯-4-基)甲氧基)羰基]氨基甲基}-5-甲基-己酸。A particularly preferred embodiment of the compounds of formulas (V) and (VI) are compounds selected from the group consisting of: 1-{[((5-methyl-2-oxo-1,3-dioxolane-4- En-4-yl)methoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid and 3-{[((5-methyl-2-oxo-1,3-dioxolane-4 -en-4-yl)methoxy)carbonyl]aminomethyl}-5-methyl-hexanoic acid.
在式(V)和(VI)的化合物的另一个实施方案中,R1为In another embodiment of the compounds of formula (V) and (VI), R is
m为0,而R8、R11和R12如前面定义。在式(V)和(VI)的化合物的一个实施方案中,R11为酰基、烷氧基羰基、芳氧基羰基、环烷氧基羰基、氨基甲酰基或取代的氨基甲酰基,R8为氢、烷氧基羰基、烷基、芳基、芳基烷基或氰基,而R12为氢、烷氧基羰基、烷基、取代的烷基、芳基或芳基烷基。在式(V)和(VI)的化合物的另一个实施方案中,R11选自乙酰基、丙酰基、丁酰基、异丁酰基、新戊酰基、环戊烷羰基、环己烷羰基、苯甲酰基、苯乙酰基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、异丁氧基羰基、仲丁氧基羰基、叔丁氧基羰基、环戊基氧羰基、环己氧基羰基、苯氧基羰基、苄氧基羰基、氨基甲酰基、N-甲基氨基甲酰基、N-乙基氨基甲酰基、N-丙基氨基甲酰基、N-异丙基氨基甲酰基、N-丁基氨基甲酰基、N-异丁基氨基甲酰基、N-仲丁基氨基甲酰基、N-叔丁基氨基甲酰基、N-环戊基氨基甲酰基、N-环己基氨基甲酰基、N-苯基氨基甲酰基、N-苄基氨基甲酰基、N,N-二甲基氨基甲酰基、N,N-二乙基氨基甲酰基、N,N-二丙基氨基甲酰基、N,N-二异丙基氨基甲酰基、N,N-二丁基氨基甲酰基、N,N-二苄基氨基甲酰基、N-吡咯烷基氨基甲酰基、N-哌啶基氨基甲酰基和N-吗啉基氨基甲酰基。在式(V)和(VI)的化合物的另一个实施方案中、R11选自乙酰基、丙酰基、丁酰基、异丁酰基、环己烷羰基、苯甲酰基、苯乙酰基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、环己氧基羰基、苯氧基羰基、苄氧基羰基、氨基甲酰基、N-甲基氨基甲酰基、N-乙基氨基甲酰基、N-丙基氨基甲酰基、N-异丙基氨基甲酰基、N-苯基氨基甲酰基、N-苄基氨基甲酰基、N,N-二甲基氨基甲酰基、N,N-二乙基氨基甲酰基、N,N-二丙基氨基甲酰基、N-吡咯烷基氨基甲酰基、N-哌啶基氨基甲酰基和N-吗啉基氨基甲酰基。m is 0, and R 8 , R 11 and R 12 are as defined above. In one embodiment of the compounds of formula (V) and (VI), R 11 is acyl, alkoxycarbonyl, aryloxycarbonyl, cycloalkoxycarbonyl, carbamoyl or substituted carbamoyl, R 8 is hydrogen, alkoxycarbonyl, alkyl, aryl, arylalkyl or cyano, and R is hydrogen, alkoxycarbonyl, alkyl, substituted alkyl, aryl or arylalkyl. In another embodiment of the compounds of formula (V) and (VI), R is selected from acetyl, propionyl, butyryl, isobutyryl, pivaloyl, cyclopentanecarbonyl, cyclohexanecarbonyl, benzene Formyl, phenylacetyl, methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl , cyclopentyloxycarbonyl, cyclohexyloxycarbonyl, phenoxycarbonyl, benzyloxycarbonyl, carbamoyl, N-methylcarbamoyl, N-ethylcarbamoyl, N-propylcarbamoyl , N-isopropylcarbamoyl, N-butylcarbamoyl, N-isobutylcarbamoyl, N-sec-butylcarbamoyl, N-tert-butylcarbamoyl, N-cyclopentyl Carbamoyl, N-cyclohexylcarbamoyl, N-phenylcarbamoyl, N-benzylcarbamoyl, N,N-dimethylcarbamoyl, N,N-diethylcarbamoyl, N,N-dipropylcarbamoyl, N,N-diisopropylcarbamoyl, N,N-dibutylcarbamoyl, N,N-dibenzylcarbamoyl, N-pyrrolidinyl Carbamoyl, N-piperidinylcarbamoyl and N-morpholinylcarbamoyl. In another embodiment of the compounds of formula (V) and (VI), R is selected from acetyl, propionyl, butyryl, isobutyryl, cyclohexanecarbonyl, benzoyl, phenylacetyl, methoxy ylcarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, cyclohexyloxycarbonyl, phenoxycarbonyl, benzyloxycarbonyl, carbamoyl, N-methylaminomethyl Acyl, N-ethylcarbamoyl, N-propylcarbamoyl, N-isopropylcarbamoyl, N-phenylcarbamoyl, N-benzylcarbamoyl, N,N-dimethyl Carbamoyl, N,N-diethylcarbamoyl, N,N-dipropylcarbamoyl, N-pyrrolidinylcarbamoyl, N-piperidinylcarbamoyl, and N-morpholinylamino formyl.
在式(V)和(VI)的化合物的一个实施方案中,R8选自氢、甲基、乙基、丙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、苯氧基羰基、苄氧基羰基和氰基。优选R8选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基。In one embodiment of the compounds of formula (V) and (VI), R is selected from hydrogen, methyl, ethyl, propyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxy Carbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, phenoxycarbonyl, benzyloxycarbonyl and cyano. Preferably R is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl.
在式(V)和(VI)的化合物的另一个实施方案中,R12选自氢、甲基、乙基、丙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、苯氧基羰基和苄氧基羰基。优选R12选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基。In another embodiment of the compounds of formula (V) and (VI), R is selected from hydrogen, methyl, ethyl, propyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxy oxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, phenoxycarbonyl and benzyloxycarbonyl. Preferably R12 is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl.
在式(V)和(VI)的化合物的另一个实施方案中,R11选自氢、烷氧基羰基、烷基、取代的烷基、芳基、芳基烷基,而R8和R12与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。优选R11选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基,而R8和R12与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。更优选R11为氢或甲基,而R8和R12和与它们结合的碳原子一起形成2-环戊烯-1-酮、2-环己烯-1-酮、2-(5H)-呋喃酮或5,6-二氢-吡喃-2-酮环。In another embodiment of the compounds of formula (V) and (VI), R is selected from hydrogen, alkoxycarbonyl, alkyl, substituted alkyl, aryl, arylalkyl, and R and R 12 together with the carbon atoms to which they are bound form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. Preferably R 11 is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl, and R 8 and R 12 are bound to their carbon atoms taken together to form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. More preferably R 11 is hydrogen or methyl, and R 8 and R 12 together with the carbon atom to which they are bound form 2-cyclopenten-1-one, 2-cyclohexen-1-one, 2-(5H) -furanone or 5,6-dihydro-pyran-2-one ring.
在式(V)和(VI)的化合物的另一个实施方案中,R12选自氢、烷氧基羰基、烷基、取代的烷基、芳基、芳基烷基,而R8和R11和与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。优选R12选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基,而R8和R11与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基或取代的环杂烷基环。更优选R12选自氢、甲基、乙基、异丙基、苯基、苄基、甲氧基羰基、乙氧基羰基和丁氧基羰基,而R8和R11和与它们结合的碳原子一起形成γ-丁内酯、δ-戊内酯或2,2-二甲基-1,3-二噁烷-4,6-二酮环。In another embodiment of the compounds of formula (V) and (VI), R is selected from hydrogen, alkoxycarbonyl, alkyl, substituted alkyl, aryl, arylalkyl, and R and R 11 and the carbon atoms to which they are bound together form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. Preferably R 12 is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl, and R 8 and R 11 are bound to their carbon atoms taken together to form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl or substituted cycloheteroalkyl ring. More preferably R12 is selected from hydrogen, methyl, ethyl, isopropyl, phenyl, benzyl, methoxycarbonyl, ethoxycarbonyl and butoxycarbonyl, and R8 and R11 and their combination The carbon atoms together form a gamma-butyrolactone, delta-valerolactone or 2,2-dimethyl-1,3-dioxane-4,6-dione ring.
在以上式(V)和(VI)的化合物的实施方案的一个更具体的版本中,R7为氢、链烷基、取代的链烷基、链烯基、取代的链烯基、芳基或取代的芳基。更优选R7为氢、甲基、乙基、苄基、-C(CH3)=CH2、-CH2C(O)N(CH3)2、In a more specific version of the embodiments of the compounds of formula (V) and (VI) above, R is hydrogen, alkanyl, substituted alkenyl, alkenyl, substituted alkenyl, aryl or substituted aryl. More preferably R 7 is hydrogen, methyl, ethyl, benzyl, -C(CH 3 )=CH 2 , -CH 2 C(O)N(CH 3 ) 2 ,
或 or
其中V为O或CH2。wherein V is O or CH 2 .
最优选R7为氢。Most preferably R7 is hydrogen.
式(V)和(VI)的化合物的特别优选的实施方案是选自以下的化合物:A particularly preferred embodiment of the compounds of formulas (V) and (VI) are compounds selected from the group consisting of:
1-{(1-甲基-3-氧代-丁-1-烯基)氨基甲基}-1-环己烷乙酸哌啶鎓;1-{(1-Methyl-3-oxo-but-1-enyl)aminomethyl}-1-cyclohexanepiperidinium acetate;
1-{1-[(2-氧代-四氢呋喃-3-基亚基)乙基氨基甲基}-1-环己烷乙酸哌啶鎓;1-{1-[(2-Oxo-tetrahydrofuran-3-ylidene)ethylaminomethyl}-1-cyclohexanepiperidinium acetate;
1-{(2-碳甲氧基(carbomethoxy)-环戊-1-烯基)氨基甲基}-1-环己烷乙酸哌啶鎓;和piperidinium 1-{(2-carbomethoxy-cyclopent-1-enyl)aminomethyl}-1-cyclohexaneacetate; and
1-{(1-甲基-2-(乙氧基羰基)-3-乙氧基-3-氧代丙-1-烯基)氨基甲基}-1-环己烷乙酸哌啶鎓。piperidinium 1-{(1-methyl-2-(ethoxycarbonyl)-3-ethoxy-3-oxoprop-1-enyl)aminomethyl}-1-cyclohexaneacetate.
在一个特别优选的实施方案中,式(IV)的化合物具有式(VII)或(VIII)的结构:In a particularly preferred embodiment, the compound of formula (IV) has the structure of formula (VII) or (VIII):
或其药学上可接受的盐、水合物或溶剂化物,其中:or a pharmaceutically acceptable salt, hydrate or solvate thereof, wherein:
n、R2、R7、R13、R14、R16和R25如前面定义。n, R 2 , R 7 , R 13 , R 14 , R 16 and R 25 are as defined above.
在一个优选的实施方案中,式(VII)和(VIII)的化合物不包括以下化合物:In a preferred embodiment, the compounds of formula (VII) and (VIII) do not include the following compounds:
如果R13或R14之任一为氢、烷氧基羰基、取代的烷氧基羰基、氨基甲酰基、环烷氧基羰基或取代的环烷氧基羰基,则R13或R14中的另一个不为氢;和If either R 13 or R 14 is hydrogen, alkoxycarbonyl, substituted alkoxycarbonyl, carbamoyl, cycloalkoxycarbonyl or substituted cycloalkoxycarbonyl, then R 13 or R 14 the other is not hydrogen; and
R25C(O)不为由胆汁酸衍生的部分。R 25 C(O) is not a bile acid derived moiety.
在式(VII)和(VIII)的化合物的一个实施方案中,n为0。在另一个实施方案中,n为1。当n为1时,优选α-氨基酸为L-立体化学构型。In one embodiment of the compounds of formula (VII) and (VIII), n is 0. In another embodiment, n is 1. When n is 1, the α-amino acid is preferably in the L-stereochemical configuration.
在式(VII)和(VIII)的化合物的另一个实施方案中,R7为氢、链烷基、取代的链烷基、链烯基、取代的链烯基、芳基或取代的芳基。优选R7为氢、甲基、乙基、苄基、-C(CH3)=CH2、-CH2C(O)N(CH3)2、In another embodiment of the compounds of formula (VII) and (VIII), R is hydrogen, alkanyl, substituted alkanyl, alkenyl, substituted alkenyl, aryl, or substituted aryl . Preferably R 7 is hydrogen, methyl, ethyl, benzyl, -C(CH 3 )=CH 2 , -CH 2 C(O)N(CH 3 ) 2 ,
或 or
其中V为O或CH2。wherein V is O or CH 2 .
最优选R7为氢。Most preferably R7 is hydrogen.
在式(VII)和(VIII)的化合物的另一个实施方案中,n为0。在式(VII)和(VIII)的化合物的另一个实施方案中,n为1,R16为氢而R2为氢、甲基、2-丙基、2-丁基、异丁基、叔丁基、环戊基、环己基、苯基、苄基、4-羟基苄基、4-溴苄基、2-咪唑基、2-吲哚基、-CH2OH、-CH(OH)CH3、-CH2CO2H、-CH2CH2CO2H、-CH2CONH2、-CH2CH2CONH2、-CH2CH2SCH3、-CH2SH、-CH2(CH2)3NH2或-CH2CH2CH2NHC(NH)NH2。优选R16为氢,而R2为氢、甲基、2-丙基、2-丁基、异丁基、叔丁基、环己基、苯基或苄基。在另一个实施方案中,n为1,而R2和R16和与它们结合的原子一起形成吡咯烷环。In another embodiment of the compounds of formula (VII) and (VIII), n is 0. In another embodiment of the compounds of formula (VII) and (VIII), n is 1, R is hydrogen and R is hydrogen , methyl, 2 -propyl, 2-butyl, isobutyl, tert Butyl, cyclopentyl, cyclohexyl, phenyl, benzyl, 4-hydroxybenzyl, 4-bromobenzyl, 2-imidazolyl, 2-indolyl, -CH2OH , -CH(OH)CH 3. -CH 2 CO 2 H, -CH 2 CH 2 CO 2 H, -CH 2 CONH 2 , -CH 2 CH 2 CONH 2 , -CH 2 CH 2 SCH 3 , -CH 2 SH, -CH 2 (CH 2 ) 3 NH 2 or —CH 2 CH 2 CH 2 NHC(NH)NH 2 . Preferably R16 is hydrogen and R2 is hydrogen, methyl, 2-propyl, 2-butyl, isobutyl, tert-butyl, cyclohexyl, phenyl or benzyl. In another embodiment, n is 1 and R and R together with the atoms to which they are bound form a pyrrolidine ring.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为甲基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is methyl and R14 is hydrogen.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为乙基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is ethyl and R14 is hydrogen.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为丙基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is propyl and R14 is hydrogen.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为异丙基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is isopropyl and R14 is hydrogen .
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为丁基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is butyl and R14 is hydrogen.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为异丁基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is isobutyl and R14 is hydrogen .
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为仲丁基和R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is sec-butyl and R14 is hydrogen .
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为叔丁基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is tert-butyl and R14 is hydrogen .
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为环戊基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is cyclopentyl and R14 is hydrogen .
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为环己基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is cyclohexyl and R14 is hydrogen.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为甲基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is methyl and R14 is methyl .
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为甲氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is methoxycarbonyl and R 14 is methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为乙氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is ethoxycarbonyl and R 14 is methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为丙氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is propoxycarbonyl and R 14 is methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为异丙氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is isopropoxycarbonyl and R 14 For methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为丁氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is butoxycarbonyl and R 14 is methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为异丁氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is isobutoxycarbonyl and R 14 For methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为仲丁氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is sec-butoxycarbonyl and R 14 For methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为叔丁氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is tert-butoxycarbonyl and R 14 For methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为环己氧基羰基而R14为甲基。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is cyclohexyloxycarbonyl and R 14 For methyl.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25为选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为苯基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from the group consisting of methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, Isopentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl) -Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1, 3-dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-di Ethoxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-di Methoxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxane-2- base)-benzyl, 1,1-dimethoxy-2-phenethyl, 1,1-diethoxy-2-phenethyl, 1-(1,3-dioxolane-2- Base)-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl , 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is phenyl and R 14 is hydrogen .
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为苄基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is benzyl and R14 is hydrogen.
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为苯乙基而R14为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R13 is phenethyl and R14 is hydrogen .
在式(VII)和(VIII)的化合物的另一个实施方案中,R25选自甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、戊基、异戊基、仲戊基、新戊基、1,1-二甲氧基乙基、1,1-二乙氧基乙基、1-(1,3-二氧戊环-2-基)-乙基、1-(1,3-二噁烷-2-基)-乙基、1,1-二甲氧基丙基、1,1-二乙氧基丙基、1-(1,3-二氧戊环-2-基)-丙基、1-(1,3-二噁烷-2-基)-丙基、1,1-二甲氧基丁基、1,1-二乙氧基丁基、1-(1,3-二氧戊环-2-基)-丁基、1-(1,3-二噁烷-2-基)-丁基、1,1-二甲氧基苄基、1,1-二乙氧基苄基、1-(1,3-二氧戊环-2-基)-苄基、1-(1,3-二噁烷-2-基)-苄基、1,1-二甲氧基-2-苯乙基、1,1-二乙氧基-2-苯乙基、1-(1,3-二氧戊环-2-基)-2-苯乙基、1-(1,3-二噁烷-2-基)-2-苯乙基、乙酰基、丙酰基、丁酰基、苯甲酰基、苯乙酰基、苯基、4-甲氧基苯基、苄基、苯乙基、苯乙烯基、环丙基、环丁基、环戊基、环己基和3-吡啶基,R13为3-吡啶基而R4为氢。In another embodiment of the compounds of formula (VII) and (VIII), R is selected from methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, pentyl, iso Pentyl, sec-pentyl, neopentyl, 1,1-dimethoxyethyl, 1,1-diethoxyethyl, 1-(1,3-dioxolan-2-yl)- Ethyl, 1-(1,3-dioxan-2-yl)-ethyl, 1,1-dimethoxypropyl, 1,1-diethoxypropyl, 1-(1,3 -Dioxolan-2-yl)-propyl, 1-(1,3-dioxan-2-yl)-propyl, 1,1-dimethoxybutyl, 1,1-diethyl Oxybutyl, 1-(1,3-dioxolan-2-yl)-butyl, 1-(1,3-dioxan-2-yl)-butyl, 1,1-dimethyl Oxybenzyl, 1,1-diethoxybenzyl, 1-(1,3-dioxolan-2-yl)-benzyl, 1-(1,3-dioxan-2-yl )-benzyl, 1,1-dimethoxy-2-phenylethyl, 1,1-diethoxy-2-phenylethyl, 1-(1,3-dioxolan-2-yl )-2-phenylethyl, 1-(1,3-dioxan-2-yl)-2-phenylethyl, acetyl, propionyl, butyryl, benzoyl, phenylacetyl, phenyl, 4-methoxyphenyl, benzyl, phenethyl, styryl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and 3-pyridyl, R 13 is 3-pyridyl and R 4 is hydrogen.
式(VII)和(VIII)的化合物的特别优选的实施方案包括选自以下的化合物:Particularly preferred embodiments of the compounds of formula (VII) and (VIII) include compounds selected from the group consisting of:
1-{[(α-乙酸基乙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-acetoxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-丙酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-propionyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-丁酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-butyryloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-异丁酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-isobutyryloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-新戊酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-pivaloyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-苯甲酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-Benzoyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-乙酸基丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-acetoxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-丁酰氧基丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-butyryloxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-异丁酰氧基丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-isobutyryloxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-苯甲酰氧基丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-benzoyloxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-乙酸基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-acetoxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-丙酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-propionyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-丁酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-butyryloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-异丁酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-isobutyryloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-新戊酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-pivaloyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-2,2-二乙氧基丙酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-2,2-diethoxypropionyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-2-(1,3-二氧戊环-2-基)丙酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-2-(1,3-dioxolan-2-yl)propionyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-(2-氨基-2-甲基丙酰基)氧异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-(2-Amino-2-methylpropionyl)oxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-苯甲酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-Benzoyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-烟酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-nicotinoyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-乙酸基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-acetoxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-丁酰氧基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-butyryloxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-异丁酰氧基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-isobutyryloxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-苯甲酰氧基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-Benzoyloxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-乙酸基苄氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-acetoxybenzyloxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(α-苯甲酰氧基苄氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(α-benzoyloxybenzyloxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(1-(3-甲基丁酰氧基)-2-苯基乙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(1-(3-methylbutyryloxy)-2-phenylethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[(1-苯甲酰氧基-2-苯基乙氧基)羰基]氨基甲基}-1-环己烷乙酸;1-{[(1-Benzoyloxy-2-phenylethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid;
1-{[N-(α-异丁酰氧基乙氧基)羰基]-4-溴苯基丙氨酰基(ananinyl)-氨基甲基}-1-环己烷乙酸;1-{[N-(α-isobutyryloxyethoxy)carbonyl]-4-bromophenylalanyl (ananinyl)-aminomethyl}-1-cyclohexaneacetic acid;
3-{[(α-异丁酰氧基乙氧基)羰基]氨基甲基}-5-甲基己酸;3-{[(α-isobutyryloxyethoxy)carbonyl]aminomethyl}-5-methylhexanoic acid;
3-{[(α-异丁酰氧基异丁氧基)羰基]氨基甲基}-5-甲基己酸;和3-{[(α-isobutyryloxyisobutoxy)carbonyl]aminomethyl}-5-methylhexanoic acid; and
3-{[(α-苯甲酰氧基异丁氧基)羰基]氨基甲基}-5-甲基己酸。3-{[(α-Benzoyloxyisobutoxy)carbonyl]aminomethyl}-5-methylhexanoic acid.
在一个实施方案中,本发明的化合物具有式(II)的结构:In one embodiment, compounds of the invention have the structure of formula (II):
在式(II)的化合物的一个实施方案中,当R3、R5和R6为氢,R4不为苯基或取代的苯基。更优选R4不为4-氯苯基。In one embodiment of the compound of formula (II), when R3 , R5 and R6 are hydrogen, R4 is not phenyl or substituted phenyl. More preferably R4 is not 4-chlorophenyl.
在一个优选的实施方案中,式(II)的化合物具有式(IX)和(X)的结构:In a preferred embodiment, the compound of formula (II) has the structures of formulas (IX) and (X):
在式(IX)和(X)的化合物的一个实施方案中,t为0。在另一个实施方案中,t为1而R2为氢、甲基、2-丙基、2-丁基、异丁基、叔丁基、环戊基、环己基、苯基、苄基、4-羟基苄基、4-溴苄基、2-咪唑基、2-吲哚基、-CH2OH、-CH(OH)CH3、-CH2CO2H、-CH2CH2CO2H、-CH2CONH2、-CH2CH2CONH2、-CH2CH2SCH3、-CH2SH、-CH2(CH2)3NH2或-CH2CH2CH2NHC(NH)NH2。In one embodiment of the compounds of formula (IX) and (X), t is zero. In another embodiment, t is 1 and R is hydrogen, methyl, 2-propyl, 2-butyl, isobutyl, t-butyl, cyclopentyl, cyclohexyl, phenyl, benzyl, 4-Hydroxybenzyl, 4-bromobenzyl, 2-imidazolyl, 2-indolyl, -CH 2 OH, -CH(OH)CH 3 , -CH 2 CO 2 H, -CH 2 CH 2 CO 2 H, -CH 2 CONH 2 , -CH 2 CH 2 CONH 2 , -CH 2 CH 2 SCH 3 , -CH 2 SH, -CH 2 (CH 2 ) 3 NH 2 or -CH 2 CH 2 CH 2 NHC(NH )NH 2 .
在式(IX)和(X)的化合物的另一个实施方案中,R20和R21独立地选自烷基、取代的烷基、芳基、取代的芳基、杂芳基和取代的杂芳基。优选R20和R21独立地选自烷基、取代的芳基和杂芳基。在一个实施方案中,R20为甲基而R21为甲基。优选在此最后的实施方案中,R7为氢、甲基、乙基、苄基、-C(CH3)=CH2、-CH2C(O)N(CH3)2、In another embodiment of the compounds of formula (IX) and (X), R and R are independently selected from alkyl, substituted alkyl, aryl, substituted aryl, heteroaryl and substituted hetero Aryl. Preferably R20 and R21 are independently selected from alkyl, substituted aryl and heteroaryl. In one embodiment, R 20 is methyl and R 21 is methyl. Preferably in this last embodiment R7 is hydrogen, methyl, ethyl, benzyl, -C( CH3 )= CH2 , -CH2C (O)N( CH3 ) 2 ,
或 or
其中V为O或CH2。wherein V is O or CH 2 .
最优选R7为氢。Most preferably R7 is hydrogen.
在式(IX)和(X)的化合物的另一个实施方案中,R20和R21和与它们结合的碳原子一起形成环烷基或取代的环烷基环。在一个实施方案中,R20和R21和与它们结合的碳原子一起形成环己基环。优选在此最后的实施方案中,R7为氢、甲基、乙基、苄基、-C(CH3)=CH2、-CH2C(O)N(CH3)2、In another embodiment of the compounds of formula (IX) and (X), R 20 and R 21 and the carbon atom to which they are bound together form a cycloalkyl or substituted cycloalkyl ring. In one embodiment, R 20 and R 21 and the carbon atom to which they are bound together form a cyclohexyl ring. Preferably in this last embodiment R7 is hydrogen, methyl, ethyl, benzyl, -C( CH3 )= CH2 , -CH2C (O)N( CH3 ) 2 ,
或 or
其中V为O或CH2。wherein V is O or CH 2 .
最优选R7为氢。Most preferably R7 is hydrogen.
在一个实施方案中,本发明的化合物具有式(III)的结构:In one embodiment, compounds of the invention have the structure of formula (III):
在式(III)的化合物的一个实施方案中,n为1,R1为氢而R2为芳基烷基。优选R2为苄基。在式(III)的化合物的另一个实施方案中,n为0,而R1为R25OC(O)-。优选R25为烷基或取代的烷基。更优选R25为乙基。在式(III)的化合物的另一个实施方案中,R22和R23为氢。在另一个实施方案中,R22和R23为烷基或取代的烷基。优选R22和R23为甲基。In one embodiment of the compound of formula (III), n is 1, R 1 is hydrogen and R 2 is arylalkyl. Preferably R2 is benzyl. In another embodiment of the compounds of formula (III), n is 0 and R 1 is R 25 OC(O)—. Preferably R 25 is alkyl or substituted alkyl. More preferably R 25 is ethyl. In another embodiment of the compounds of formula (III), R22 and R23 are hydrogen. In another embodiment, R22 and R23 are alkyl or substituted alkyl. Preferably R22 and R23 are methyl.
在一个优选的实施方案中,式(III)的化合物具有式(XI)的结构:In a preferred embodiment, the compound of formula (III) has the structure of formula (XI):
在式(XI)的化合物的一个实施方案中,n为1,X为NH,Y为0,R1为氢,R2为苄基,R22为甲基而R23为甲基。在另一个实施方案中,n为0,Y为0,R1为R25OC(O)-,R25为乙基,R22为氢而R23为氢。In one embodiment of the compound of formula (XI), n is 1, X is NH, Y is 0, R1 is hydrogen, R2 is benzyl, R22 is methyl and R23 is methyl. In another embodiment, n is 0, Y is 0, R1 is R25OC (O)-, R25 is ethyl, R22 is hydrogen and R23 is hydrogen.
在另一个实施方案中,式(III)的化合物具有式(XII)的结构:In another embodiment, the compound of formula (III) has the structure of formula (XII):
在式(XII)的化合物的一个实施方案中,n为1,X为NH,Y为0,R1为氢,R2为苄基,R22为甲基和R23为甲基。在另一个实施方案中,n为0,Y为0,R1为R25OC(O)-,R25为乙基,R22为氢而R23为氢。In one embodiment of the compound of formula (XII), n is 1, X is NH, Y is 0, R1 is hydrogen, R2 is benzyl, R22 is methyl and R23 is methyl. In another embodiment, n is 0, Y is 0, R1 is R25OC (O)-, R25 is ethyl, R22 is hydrogen and R23 is hydrogen.
本发明还包括用于对需要治疗的患者行给药的GABA类似物衍生物,M-G,其中M为基元,而G是GABA类似物,H-G(其中H为氢)的衍生物。基元M一旦从G和其任何代谢产物上裂解,表现出对大鼠的致癌毒性剂量(TD50)大于0.2mmol/kg/天。而且,在对大鼠进行结肠给药时,基元M在体内以充足的速率从G上裂解产生:The invention also includes derivatives of GABA analogs, MG, wherein M is a motif and G is a derivative of the GABA analog, HG, wherein H is hydrogen, for administration to a patient in need thereof. Once the motif M is cleaved from G and any of its metabolites, it exhibits a carcinogenic toxic dose (TD 50 ) to rats greater than 0.2 mmol/kg/day. Moreover, upon colonic administration to rats, the motif M is cleaved from G at a sufficient rate in vivo:
(i)至少为通过结肠施用等摩尔剂量的H-G获得的血浆H-G的最大浓度(Cmax)的120%的血浆H-G的Cmax;和 ( i) a Cmax of plasma HG that is at least 120% of the maximum concentration ( Cmax ) of plasma HG obtained by colonic administration of equimolar doses of HG; and
(ii)至少为通过结肠施用等摩尔剂量的H-G获得的AUC的120%的AUC。(ii) An AUC of at least 120% of the AUC obtained by colonic administration of equimolar doses of H-G.
优选M-G具有式(XIV)的结构:Preferably M-G has the structure of formula (XIV):
或其药学上可接受的盐、水合物或溶剂化物,其中:or a pharmaceutically acceptable salt, hydrate or solvate thereof, wherein:
M为基元;M is the primitive;
Y为O或S;Y is O or S;
R为氢,或者R和R6和与它们结合的原子一起形成氮杂环丁烷、取代的氮杂环丁烷、吡咯烷或取代的吡咯烷环;R is hydrogen, or R and R together form an azetidine, a substituted azetidine, a pyrrolidine or a substituted pyrrolidine ring with the atoms bound to them;
R3和R6独立地选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基;R and R are independently selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted cycloalkyl, ring Heteroalkyl, substituted cycloheteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, and substituted heteroarylalkyl;
R4和R5独立地选自氢、烷基、取代的烷基、酰基、取代的酰基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基,或者任选R4和R5和与它们结合的碳原子一起形成环烷基、取代的环烷基、环杂烷基、取代的环杂烷基或桥连的环烷基环;和R and R are independently selected from hydrogen, alkyl, substituted alkyl, acyl, substituted acyl, aryl, substituted aryl, arylalkyl , substituted arylalkyl, cycloalkyl, substituted Cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl and substituted heteroarylalkyl, or optionally R 4 and R 5 and together with the carbon atoms to which they are bound form a cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted cycloheteroalkyl, or bridged cycloalkyl ring; and
R7选自氢、烷基、取代的烷基、芳基、取代的芳基、芳基烷基、取代的芳基烷基、环烷基、取代的环烷基、环杂烷基、取代的环杂烷基、杂烷基、取代的杂烷基、杂芳基、取代的杂芳基、杂芳基烷基和取代的杂芳基烷基。 R is selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, arylalkyl, substituted arylalkyl, cycloalkyl, substituted cycloalkyl, cycloheteroalkyl, substituted Cycloheteroalkyl, heteroalkyl, substituted heteroalkyl, heteroaryl, substituted heteroaryl, heteroarylalkyl, and substituted heteroarylalkyl.
在一个优选的实施方案中,M具有式(XV)的结构:In a preferred embodiment, M has the structure of formula (XV):
其中:in:
n、X、R1和R2如前面定义。n, X, R1 and R2 are as defined above.
在一个实施方案中,M-G包括这样的化合物:其中H-G一旦从M上裂解,基本上不存在任何具有式(XVI)的结构的内酰胺:In one embodiment, M-G includes compounds wherein H-G, once cleaved from M, is substantially free of any lactam having the structure of formula (XVI):
其中R为氢,而R3、R4、R5和R6如前面定义。wherein R is hydrogen and R 3 , R 4 , R 5 and R 6 are as defined above.
优选基元M或任何由M形成的代谢产物在G-M裂解时不形成甲醛或新戊酸。在一个实施方案中,在对大鼠进行结肠给药时,基元M在体内以充足的速率从G上裂解产生至少为通过结肠施用等摩尔剂量的H-G获得的血浆H-G的Cmax的200%,最优选至少1000%的血浆H-G的Cmax。优选在对大鼠进行结肠给药时,基元M在体内以充足的速率从G上裂解产生至少为通过结肠施用等摩尔剂量的H-G获得的血浆的AUC的200%,最优选至少500%的血浆H-G的AUC。在另一个实施方案中,在给狗口服(例如使用渗透性迷你型泵设备)大约60μmol当量的H-G/kg的剂量之后,基元M在体内以充足的速率从H-G上裂解产生至少为在进行相同方式给药之后由等摩尔剂量的H-G获得的血浆H-G浓度的200%的服药后12小时的血浆浓度。Preferably the motif M or any metabolite formed from M does not form formaldehyde or pivalic acid upon cleavage of GM. In one embodiment, upon colonic administration to rats, the motif M is cleaved from G in vivo at a sufficient rate to yield at least 200% of the Cmax of plasma HG obtained by colonically administered equimolar doses of HG , most preferably at least 1000% of the Cmax of plasma HG. Preferably, upon colonic administration to rats, the motif M is cleaved from G in vivo at a sufficient rate to produce an AUC of at least 200%, most preferably at least 500%, of the AUC of plasma obtained by colonically administered equimolar doses of HG. AUC of plasma HG. In another embodiment, following oral administration (e.g., using an osmotic mini-pump device) to a dog at a dose of about 60 μmol equivalents of HG/kg, motif M is cleaved from HG at a sufficient rate in vivo to produce at least Plasma concentration 12 hours after administration that is 200% of the plasma HG concentration obtained with an equimolar dose of HG after administration in the same manner.
4.3本发明化合物的合成 4.3 Synthesis of compounds of the present invention
本发明的化合物可以通过路线1-17所示的合成方法获得。本领域技术人员将认识到本发明的化合物的优选的合成途径是将基元与GABA类似物结合。已在GABA类似物的合成技术中描述了多种方法(例如参见Satzinger等人,美国专利4,024,175;Silverman等人,美国专利5,563,175;Horwell等人,美国专利6,020,370;Silverman等人,美国专利6,028,214;Horwell等人,美国专利6,103,932;Silverman等人,美国专利6,117,906;Silverman,国际公开WO92/09560;Silverman等人,国际公开WO 93/23383;Horwell等人,国际公开WO 97/29101,Horwell等人,国际公开WO 97/33858;Horwell等人,国际公开WO 97/33859;Bryans等人,国际公开WO98/17627;Guglietta等人,国际公开WO 99/08671;Bryans等人,国际公开WO 99/21824;Bryans等人,国际公开WO 99/31057;Belliotti等人,国际公开WO 99/31074;Bryans等人,国际公开WO 99/31075;Bryans等人,国际公开WO 99/61424;Bryans等人,国际公开WO00/15611;Bryans,国际公开WO 00/31020;和Bryans等人,国际公开WO 00/50027)。其它方法在GABA类似物合成技术中是已知的,且本领域技术人员可以容易地获得。本文所述的基元在本领域中是已知的,并可以由确定的方法制备并结合到GABA类似物是(例如参见Green等人,″Protective Groups in Organic Chemistry(有机化学中的保护基)″,(Wiley,第二版,1991);Harrison等人,″Compendium ofSynthetic Organic Methods(有机合成方法简述)″,第1-8卷(JohnWiley和Sons,1971-1996);″Beilstein Handbook of OrganicChemistry(Beilstein有机化学手册),″Beilstein Institute ofOrganic Chemistry(Beilstein有机化学研究所),法兰克福,德国;Feiser等人,″Reactions for Organic Synthesis(有机合成反应),″第1-17卷,Wiley Interscience;Trost等人,″ComprehensiveOrganic Synthesis(综合有机合成)″,Pergamon Press,1991;″Theilheimer’s Synthetic Methods of OrganicChemistry(Theilheimer有机化学合成方法),″第1-45卷,Karger,1991;March,″Advanced Organic Chemistry(高等有机化学),″Wiley Interscience,1991;Larock″Comprehensive OrganicTransformations(综合有机转化),″VCH发行人,1989;Paquette,″Encyclopedia of Reagents for Organic Synthesis(有机合成试剂百科全书),″John Wiley & Sons,1995,Bodanzsky,″Principlesof Peptide Synthesis(肽合成原理),″Springer Verlag,1984;Bodanzsky,″Practice of Peptide Synthesis(肽合成实践)″Springer Verlag,1984)。The compounds of the present invention can be obtained by the synthetic methods shown in schemes 1-17. Those skilled in the art will recognize that a preferred synthetic route to the compounds of the invention is to combine the motif with a GABA analog. Various methods have been described in the art of synthesis of GABA analogs (see, for example, Satzinger et al., U.S. Patent 4,024,175; Silverman et al., U.S. Patent 5,563,175; Horwell et al., U.S. Patent 6,020,370; Silverman et al., U.S. Patent 6,028,214; Horwell et al. et al., U.S. Patent 6,103,932; Silverman et al., U.S. Patent 6,117,906; Silverman, International Publication WO 92/09560; Silverman et al., International Publication WO 93/23383; Horwell et al., International Publication WO 97/29101; Publication WO 97/33858; Horwell et al. International Publication WO 97/33859; Bryans et al. International Publication WO 98/17627; Guglietta et al. International Publication WO 99/08671; Bryans et al. International Publication WO 99/21824; People such as Belliotti, International Publication WO 99/31074; Bryans et al., International Publication WO 99/31075; Bryans et al., International Publication WO 99/61424; Bryans et al., International Publication WO 99/61424; Bryans et al., International Publication WO 00 /15611; Bryans, International Publication WO 00/31020; and Bryans et al., International Publication WO 00/50027). Other methods are known in the art of GABA analog synthesis and are readily available to those skilled in the art. The motifs described herein are known in the art and can be prepared by established methods and incorporated into GABA analogs (see, for example, Green et al., "Protective Groups in Organic Chemistry (protective groups in organic chemistry) ", (Wiley, Second Edition, 1991); Harrison et al., "Compendium of Synthetic Organic Methods (Brief Description of Organic Synthetic Methods)", Volumes 1-8 (John Wiley and Sons, 1971-1996); "Beilstein Handbook of Organic Chemistry (Beilstein Handbook of Organic Chemistry), "Beilstein Institute of Organic Chemistry, Frankfurt, Germany; Feiser et al., "Reactions for Organic Synthesis," vol. 1-17, Wiley Interscience; Trost et al., "Comprehensive Organic Synthesis (Comprehensive Organic Synthesis)", Pergamon Press, 1991; "Theilheimer's Synthetic Methods of Organic Chemistry (Theilheimer's Synthetic Methods of Organic Chemistry), "vol. Advanced Organic Chemistry), "Wiley Interscience, 1991; Larock" Comprehensive Organic Transformations, "VCH Publisher, 1989; Paquette, "Encyclopedia of Reagents for Organic Synthesis (Encyclopedia of Organic Synthetic Reagents), "John Wiley & Sons , 1995, Bodanzsky, "Principles of Peptide Synthesis (peptide synthesis principle), "Springer Verlag, 1984; Bodanzsky, "Practice of Peptide Synthesis (peptide synthesis practice)" Springer Verlag, 1984).
因此,用于制备本发明的化合物及其中间产物的原料可以商购得到或可以由已知的合成方法制备。其它用于合成本文所述的前药的方法如本领域中所述,或者对于考虑以上提供的参考资料的本领域技术人员来说是非常显而易见的,并可以用于合成本发明的化合物。因此,本文路线中所示的方法是进行例示而不是进行综合。Accordingly, starting materials for the preparation of the compounds of the present invention and their intermediates are commercially available or may be prepared by known synthetic methods. Other methods for synthesizing the prodrugs described herein are described in the art, or will be readily apparent to one of skill in the art considering the references provided above, and can be used to synthesize the compounds of the invention. Therefore, the methods shown in the routes in this article are illustrative rather than comprehensive.
在以下的任一路线中,在用基元或其它保护基将GABA类似物的氨基官能化以后,可以通过许多本领域技术人员已知的合成方法将羧酸基团转化为酯或硫酯。在一个优选的实施方案中,可以将GABA类似物与醇或硫醇在偶合剂(例如碳化二亚胺和二甲基氨基吡啶)存在下反应得到酯。在另一个优选的实施方案中,可以将GABA类似物与烷基卤在碱存在下反应得到酯。其它将GABA类似物转化成为酯或硫酯的方法在本领域技术人员所熟悉的本文提供的参考资料范围内。In either of the following routes, after functionalization of the amino group of the GABA analog with a radical or other protecting group, the carboxylic acid group can be converted to an ester or thioester by a number of synthetic methods known to those skilled in the art. In a preferred embodiment, esters can be obtained by reacting GABA analogs with alcohols or thiols in the presence of coupling agents such as carbodiimide and dimethylaminopyridine. In another preferred embodiment, esters can be obtained by reacting GABA analogs with alkyl halides in the presence of a base. Other methods of converting GABA analogs to esters or thioesters are within the scope of the references provided herein, which are familiar to those skilled in the art.
如以上路线1所示,可以将羧酸直接与GABA类似物衍生物(6)的末端氨基(或羟基)基团结合得到加合物(7)。进行这种反应的试剂对本领域技术人员来说是已知的,并包括但不限于碳化二亚胺、铵盐、鏻盐等。可选择地,羧酸衍生物如酰氯、对称酐或混合酐与GABA类似物(6)在碱(例如氢氧化物、叔胺等)存在下的反应可以用于合成(7)。As shown in Scheme 1 above, the adduct (7) can be obtained by directly combining the carboxylic acid with the terminal amino (or hydroxyl) group of the GABA analog derivative (6). Reagents for such reactions are known to those skilled in the art and include, but are not limited to, carbodiimides, ammonium salts, phosphonium salts, and the like. Alternatively, the reaction of carboxylic acid derivatives such as acid chlorides, symmetrical anhydrides or mixed anhydrides with GABA analogs (6) in the presence of bases (eg hydroxides, tertiary amines, etc.) can be used for the synthesis of (7).
路线2Route 2
如图2所示,可以通过在碱(例如氢氧化物、叔胺等)存在下用多种碳酸衍生物进行处理而将GABA类似物衍生物(6)转化为氨基甲酸酯(8)。可选择地,可以将已知的对异氰酸酯(9)或(10)的醇加成用于合成(8)。As shown in Figure 2, GABA analog derivatives (6) can be converted to carbamates (8) by treatment with various carbonic acid derivatives in the presence of a base (eg, hydroxide, tertiary amine, etc.). Alternatively, known alcohol additions to isocyanates (9) or (10) can be used for the synthesis of (8).
如路线3所示,可以通过在偶合剂存在下用硫代酸进行处理而将GABA类似物衍生物(6)转化为硫代酰胺(11)。用于进行这种反应的试剂对本领域技术人员来说是已知的,且包括但不限于碳化二亚胺、铵盐、鏻盐等。可选择地,硫代酸衍生物如硫代酰氯、对称酐或混合酐与(6)在碱(如氢氧化物、叔胺等)存在下的反应可以用于合成硫代酰胺(11)。在另一个方法中,可以通过在五硫化磷(当n=0时)存在下进行加热而将酰胺(7)转化为硫代酰胺(11)。As shown in Scheme 3, GABA analog derivatives (6) can be converted to thioamides (11) by treatment with thioacids in the presence of couplers. Reagents for carrying out such reactions are known to those skilled in the art and include, but are not limited to, carbodiimides, ammonium salts, phosphonium salts, and the like. Alternatively, the reaction of thioacid derivatives such as thioacid chlorides, symmetrical anhydrides or mixed anhydrides with (6) in the presence of bases (eg hydroxides, tertiary amines, etc.) can be used to synthesize thioamides (11). In another method, amides (7) can be converted to thioamides (11) by heating in the presence of phosphorus pentasulfide (when n=0).
可以由对应的硫代碳酸酯衍生物(即分别为P=O,Q=S和P=SQ=O)(其中W为氯化物、咪唑基或4-硝基苯氧基)与GABA类似物衍生物(6)在碱存在下进行反应合成硫代氨基甲酸酯(12)和(13)。还可以通过硫醇与异氰酸酯(9)或(10)反应形成硫代氨基甲酸酯(13)。可以通过GABA类似物衍生物(6)与二硫代碳酸酯衍生物(即P和Q=S)(其中W为氯化物、咪唑基或4-硝基苯氧基)在碱存在下进行反应而制备二硫代氨基甲酸酯(14)(P=S,Q=S)(参见路线4)。The corresponding thiocarbonate derivatives (i.e., P=O, Q=S and P=SQ=O, respectively) (wherein W is chloride, imidazolyl or 4-nitrophenoxy) and GABA analogs Derivative (6) reacts in the presence of base to synthesize thiocarbamate (12) and (13). Thiocarbamates (13) can also be formed by reacting thiols with isocyanates (9) or (10). Can be reacted by GABA analog derivatives (6) with dithiocarbonate derivatives (i.e. P and Q=S) (wherein W is chloride, imidazolyl or 4-nitrophenoxy) in the presence of a base Instead, dithiocarbamate (14) (P=S, Q=S) is prepared (see Scheme 4).
路线4Route 4
一种用于合成式(IV)的化合物的方法如路线5例示。One method for the synthesis of compounds of formula (IV) is exemplified in scheme 5.
路线5Route 5
在碱存在下用芳香离去基团如对硝基苯酚处理氯甲酸酯(15)得到对硝基苯基碳酸酯(16)。卤化物的相互交换得到碘化物(17),将此碘化物与金属或羧酸的四烷基铵盐反应得到化合物(18)。任选在三甲基甲硅烷基氯化物存在下用GABA类似物衍生物(19)处理(18)得到式(IV)的化合物。制备相关的酰氧基烷基氨基甲酸酯化合物的方法已在本领域中描述(Alexander,美国专利4,760,057;Alexander,美国专利4,916,230;Alexander,美国专利5,466,811;Alexander,美国专利5,684,018)。Treatment of chloroformate (15) with an aromatic leaving group such as p-nitrophenol in the presence of a base affords p-nitrophenyl carbonate (16). Interchange of halides affords iodide (17), which is reacted with a metal or a tetraalkylammonium salt of a carboxylic acid to yield compound (18). Treatment of (18) with GABA analog derivatives (19), optionally in the presence of trimethylsilyl chloride, affords compounds of formula (IV). Methods for preparing related acyloxyalkylcarbamate compounds have been described in the art (Alexander, US Patent 4,760,057; Alexander, US Patent 4,916,230; Alexander, US Patent 5,466,811; Alexander, US Patent 5,684,018).
可选择地,式(IV)的化合物可以如路线6所示的分步方式由碳酸酯(18)制备。这里(18)与任选作为酯保护的α-氨基酸(20)的反应得到中间产物(21),所述中间产物在脱保护时(如果需要的话)提供化合物(22),然后用本领域中已知的标准肽偶合剂将其与GABA类似物(23)偶合。Alternatively, compounds of formula (IV) can be prepared from carbonates (18) in a stepwise manner as shown in Scheme 6. Here the reaction of (18) with an optionally protected α-amino acid (20) as an ester affords an intermediate (21) which upon deprotection (if desired) affords compound (22), which can then be modified using methods known in the art It was coupled to the GABA analog (23) by known standard peptide coupling reagents.
路线6Route 6
另一种合成式(IV)的化合物的方法通过将GABA类似物衍生物(19)羰基化为中间产物氨基甲酸类而进行的,它是通过在本领域中公开的方法的改型中进行就地烷基化反应而收集的(Butcher,Synlett,1994,825-6;Ferres等人,U.S.Patent 4,036,829)。使二氧化碳气体冒泡进入在溶剂如DMF或NMP中的含有(19)和碱(例如CS2CO3,Ag2CO3或AgO)的溶液。任选在作为催化剂的碘化物离子存在下加入活化的卤化物,并继续羰基化直到反应完成。此方法如路线7关于由卤化物(24)制备式(IV)的化合物所示。Another method for the synthesis of compounds of formula (IV) is carried out by carbonylation of GABA analogue derivatives (19) to intermediate carbamates by modification of methods disclosed in the art. collected from the alkylation reaction (Butcher, Synlett, 1994, 825-6; Ferres et al., US Patent 4,036,829). Carbon dioxide gas is bubbled into a solution containing (19) and a base (eg CS2CO3 , Ag2CO3 or AgO) in a solvent such as DMF or NMP. An activated halide is added, optionally in the presence of iodide ion as a catalyst, and the carbonylation is continued until the reaction is complete. This method is shown in Scheme 7 for the preparation of compounds of formula (IV) from halides (24).
路线7Route 7
可选择地,式(IV)的化合物可以如路线8所示的分步的方式制备。羧基保护的α-氨基酸(20)的羰基化和烷基化提供中间产物(21),它在脱保护时与GABA类似物(23)偶合,如前面路线6所示。Alternatively, compounds of formula (IV) can be prepared in a stepwise manner as shown in Scheme 8. Carbonylation and alkylation of the carboxy-protected α-amino acid (20) affords an intermediate (21), which upon deprotection couples with a GABA analog (23), as previously shown in Scheme 6.
路线8Route 8
但另外一个合成式(IV)的化合物的方法依赖GABA类似物的酮氨基甲酸酯衍生物的氧化(Gallop等人,标题为“Methods forSynthesis of 前药from 1-Acyl-Alkyl Derivatives andCompositions Thereof(由1-酰基-烷基衍生物及其衍生物合成前药的方法)”的共同未决的美国专利申请)。如路线9所示,酮氨基甲酸酯(25)的氧化得到式(IV)的化合物。优选的溶剂包括但不限于叔丁醇、二乙基醚、乙酸、己烷、二氯乙烷、二氯甲烷、乙酸乙酯、乙腈、甲醇、氯仿和水。一般地,氧化剂可以是生物体(例如酵母或细菌),或者化学试剂(例如酶或过氧化物)。优选的氧化剂包成功地用于将酮Baeyer-Villager氧化为酯或内酯的氧化剂(Strukul,Angnew.Chem.Int.ED.,1998,37,1198;Renz等人,Eur.J.Org.Chem.1999,737;Beller等人,in“Transition Metals in Organic Synthesis”Chapter 2,Wiley VCH;Stewart,Current Organic Chemistry,1998,2,195;Kayser等人,Synlett,1999,1,153)。But another method for the synthesis of compounds of formula (IV) relies on the oxidation of ketocarbamate derivatives of GABA analogs (Gallop et al., titled "Methods for Synthesis of prodrugs from 1-Acyl-Alkyl Derivatives and Compositions Thereof (by 1-acyl-alkyl derivatives and methods for the synthesis of prodrugs of their derivatives)" co-pending US patent application). As shown in Scheme 9, oxidation of ketocarbamate (25) affords compounds of formula (IV). Preferred solvents include, but are not limited to, t-butanol, diethyl ether, acetic acid, hexane, dichloroethane, dichloromethane, ethyl acetate, acetonitrile, methanol, chloroform, and water. Generally, an oxidizing agent can be an organism (such as yeast or bacteria), or a chemical agent (such as an enzyme or a peroxide). Preferred oxidizing agents include those successfully used to oxidize Baeyer-Villager ketones to esters or lactones (Strukul, Angnew. Chem. Int. ED., 1998, 37, 1198; Renz et al., Eur. J. Org. Chem .1999, 737; Beller et al., in "Transition Metals in Organic Synthesis" Chapter 2, Wiley VCH; Stewart, Current Organic Chemistry, 1998, 2, 195; Kayser et al., Synlett, 1999, 1, 153).
路线9Route 9
本发明的其它化合物可以由适宜的酮氨基甲酸酯衍生物通过Baeyer-Villager类型氧化合成,条件是它们不含有在反应条件下容易分解或转化的化学官能团。Other compounds of the present invention can be synthesized from suitable ketocarbamate derivatives by Baeyer-Villager type oxidations, provided they do not contain chemical functionalities that are readily decomposed or transformed under the reaction conditions.
酮氨基甲酸酯(25)可以由对应的α-羟基酮化合物(26)通过以下方法制备:直接通过与异氰酸酯(9)反应,或者首先将α-羟基酮化合物转化为卤代氯甲酸酯或活化的碳酸酯中间产物(27),然后与化合物(19)反应,如路线10例示。Ketocarbamate (25) can be prepared from the corresponding α-hydroxyketone compound (26) either directly by reaction with isocyanate (9) or by first converting the α-hydroxyketone compound to a halochloroformate or the activated carbonate intermediate (27), which is then reacted with compound (19), as exemplified in Scheme 10.
路线10Route 10
可选择地,可以根据上述的偶合方法,通过如路线11所示的α-氨基酸氨基甲酸酯(28)以分步方式制备酮氨基甲酸酯(25)。Alternatively, keto carbamates (25) can be prepared in a stepwise manner via α-amino acid carbamates (28) as shown in Scheme 11 according to the coupling method described above.
路线11Route 11
注意到以上路线10所用的GABA类似物的异氰酸酯衍生物(即化合物(9))的一种制备方法是从如路线12所示的适宜的六元酐(29)开始的。通过与醇或硫醇亲核试剂反应打开酐环,得到羧酸(30)。以2步顺序(即首先将羧基活化成混合酐、酰基卤或合成等同物,然后用叠氮化物取代)或直接(例如通过用PH2P(O)N3处理)将此化合物转化为中间产物酰基叠氮化物。通过在适宜的溶剂(例如甲苯)中在0℃至120℃的温度下热分解而将酰基叠氮化物中间产物进行库尔提斯重排,得到异氰酸酯(9)。任选不将异氰酸酯分离,而是就地形成,并通过与α-羟基酮(26)反应终止其形成,得到目标产物(25)。Note that one preparation of the isocyanate derivative of the GABA analog used in Scheme 10 above (ie compound (9)) starts from the appropriate hexaanhydride (29) as shown in Scheme 12. Opening of the anhydride ring by reaction with an alcohol or thiol nucleophile affords carboxylic acid (30). Conversion of this compound to intermediate The product acyl azide. Curtis rearrangement of the acyl azide intermediate by thermal decomposition in a suitable solvent such as toluene at temperatures ranging from 0°C to 120°C affords the isocyanate (9). The isocyanate is optionally not isolated, but formed in situ, and its formation terminated by reaction with a-hydroxyketone (26) to give the desired product (25).
路线12Route 12
一种用于合成氧代间二氧杂环戊烯基(oxodioxolenyl)甲基氨基甲酸酯前药(36)的方法公开在路线13中。在碱存在下用光气或羰基二咪唑处理羟基酮(31)得到环碳酸酯(32)。用N-溴琥珀酰亚胺和偶氮异丁腈将自由基溴化得到溴化物(33),将其转化为醇(34)。通过与4-硝基苯基氯甲酸酯反应将醇(34)转化成二碳酸酯(dicarbonate)(35),然后使其与GABA类似物衍生物(19)反应得到前药(36)。可选择地,化合物(34)与异氰酸酯(9)反应得到化合物(36),其中n为0。A method for the synthesis of oxodioxolenyl methyl carbamate prodrug (36) is disclosed in Scheme 13. Treatment of hydroxyketones (31) with phosgene or carbonyldiimidazole in the presence of base affords cyclic carbonates (32). Radical bromination with N-bromosuccinimide and azoisobutyronitrile affords the bromide (33), which is converted to the alcohol (34). Alcohol (34) was converted to dicarbonate (35) by reaction with 4-nitrophenyl chloroformate, which was then reacted with GABA analog derivative (19) to give prodrug (36). Alternatively, compound (34) is reacted with isocyanate (9) to give compound (36), wherein n is 0.
路线13Route 13
可以通过路线14所公开的方法合成前药(41)。将羧酸(37)与醇(38)(例如二环己基碳化二亚胺和吡啶)偶合得到酯(39)。通过与4-硝基苯基氯甲酸酯反应将酯(39)转化为活性碳酸酯(40),然后与GABA类似物衍生物(19)反应得到前药(41)。The prodrug (41) can be synthesized by the method disclosed in Scheme 14. Coupling of carboxylic acid (37) with alcohol (38) such as dicyclohexylcarbodiimide and pyridine affords ester (39). Conversion of ester (39) to activated carbonate (40) by reaction with 4-nitrophenyl chloroformate followed by reaction with GABA analog derivative (19) afforded prodrug (41).
路线14Route 14
可以通过任选在作为催化剂的仲胺存在下,在路线15所示的脱水条件下使活性羰基化合物(42)与GABA类似物衍生物(19)(其中R16=H)反应而简单地合成烯胺前药如(43)。It can be synthesized simply by reacting a reactive carbonyl compound (42) with a GABA analog derivative (19) (where R 16 =H) under the dehydration conditions shown in Scheme 15, optionally in the presence of a secondary amine as a catalyst Enamine prodrugs such as (43).
路线15Route 15
可以通过路线16所示的途径合成化合物(III)。Compound (III) can be synthesized by the route shown in Scheme 16.
GABA类似物(23)与α-活化的酯衍生物(44)反应得到氨基酯(45)。Reaction of GABA analogs (23) with α-activated ester derivatives (44) affords amino esters (45).
通过酰基化将(45)的氨基封端得到(46)(例如使用上述的方法),并在标准条件下将游离酸酯化得到二酯(47)。进行Dieckman缩合,然后进行脱羧得到酮(48)。然后进行过氧酸氧化得到内酯(III)。Amino endcapping of (45) by acylation affords (46) (eg, using methods described above), and esterification of the free under standard conditions affords diesters (47). Dieckman condensation followed by decarboxylation affords ketone (48). This is followed by peroxyacid oxidation to afford the lactone (III).
路线16Route 16
可以如路线17所述,通过在脱水条件下用GABA类似物衍生物(50)处理酮或酮衍生物(49)而合成亚胺前药(II)。Imine prodrugs (II) can be synthesized as described in Scheme 17 by treating ketones or ketone derivatives (49) with GABA analog derivatives (50) under dehydrating conditions.
路线17Route 17
可以通过本领域中已知的常规方法合成磷前药。类似地,可以通过使用本领域中所述的方法合成具有S-N键的前药。Phosphorous prodrugs can be synthesized by conventional methods known in the art. Similarly, prodrugs with S-N bonds can be synthesized by using methods described in the art.
4.4本发明化合物的治疗应用 4.4 Therapeutic Applications of the Compounds of the Invention
根据本发明,将本发明的化合物和/或组合物施用于患有以下疾病的患者,优选人:癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病或乙醇戒断综合征。而且在某些实施方案中,可以给患者,优选人施用本发明的化合物和/或组合物作为对多种疾病或病症的预防措施。因此,本发明的化合物和/或组合物可以作为一种预防措施施用于易患以下疾病的患者:癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病和乙醇戒断综合征。因此,本发明的化合物和/或组合物可以用于预防一种疾病或病症,同时治疗另一种疾病和病症(例如预防精神病并治疗胃肠疾病;预防神经病性疼痛并治疗乙醇戒断综合征)。According to the invention, the compounds and/or compositions of the invention are administered to patients, preferably humans, suffering from epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases, panic, Pain (especially neuropathic pain and musculoskeletal pain), inflammatory disease (ie, arthritis), insomnia, gastrointestinal disease, or alcohol withdrawal syndrome. Also in certain embodiments, the compounds and/or compositions of the present invention may be administered to a patient, preferably a human, as a prophylactic against various diseases or conditions. Accordingly, the compounds and/or compositions of the present invention may be administered as a prophylactic measure to patients predisposed to epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases, panic, Pain (especially neuropathic pain and musculoskeletal pain), inflammatory disease (ie, arthritis), insomnia, gastrointestinal disorders, and alcohol withdrawal syndrome. Thus, the compounds and/or compositions of the present invention can be used to prevent one disease or condition while treating another (e.g., preventing psychosis and treating gastrointestinal disorders; preventing neuropathic pain and treating alcohol withdrawal syndrome ).
可以由本领域中已知的方法确定本发明的化合物和/或组合物治疗癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病和乙醇戒断综合征的合适性(例如参见Satzinger等人,美国专利4,024,175;Satzinger等人,美国专利4,087,544;Woodruff,美国专利5,084,169;Silverman等人,美国专利5,563,175;Singh,美国专利6,001,876;Horwell等人,美国专利6,020,370;Silverman等人,美国专利6,028,214;Horwell等人,美国专利6,103,932;Silverman等人,美国专利6,117,906;Silverman,国际公开WO 92/09560;Silverman等人,国际公开WO 93/23383;Horwell等人,国际公开WO 97/29101,Horwell等人,国际公开WO 97/33858;Horwell等人,国际公开WO 97/33859;Bryans等人,国际公开WO 98/17627;Guglietta等人,国际公开WO99/08671;Bryans等人,国际公开WO 99/21824;Bryans等人,国际公开WO 99/31057;Magnus-Miller等人,国际公开WO 99/37296;Bryans等人,国际公开WO 99/31075;Bryans等人,国际公开WO99/61424;Pande,国际公开WO 00/23067;Bryans,国际公开WO00/31020;Bryans等人,国际公开WO 00/50027;和Bryans等人,国际公开WO 02/00209)。本发明的化合物和/或组合物可以用于通过本领域中所述的方法(参见上述)治疗或预防癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病和乙醇戒断综合征。因此,本领域技术人员能够分析和使用本发明的化合物和/或组合物以治疗或预防癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病和乙醇戒断综合征。Treatment of epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases, panic, pain (especially neuropathic pain) with the compounds and/or compositions of the present invention can be determined by methods known in the art. and muscle and bone pain), inflammatory diseases (i.e., arthritis), insomnia, gastrointestinal disorders, and ethanol withdrawal syndrome (see, for example, Satzinger et al., U.S. Patent 4,024,175; Satzinger et al., U.S. Patent 4,087,544; Woodruff , US Patent 5,084,169; Silverman et al, US Patent 5,563,175; Singh, US Patent 6,001,876; Horwell et al, US Patent 6,020,370; Silverman et al, US Patent 6,028,214; Horwell et al, US Patent 6,103,932; Silverman et al, US Patent 6,117,906 ; Silverman, International Publication WO 92/09560; Silverman et al., International Publication WO 93/23383; Horwell et al., International Publication WO 97/29101, Horwell et al., International Publication WO 97/33858; Horwell et al., International Publication WO 97/33858; 97/33859; Bryans et al. International Publication WO 98/17627; Guglietta et al. International Publication WO 99/08671; Bryans et al. International Publication WO 99/21824; Bryans et al. International Publication WO 99/31057; Magnus-Miller Bryans et al., International Publication WO 99/31075; Bryans et al., International Publication WO 99/61424; Pande, International Publication WO 00/23067; Bryans, International Publication WO 00/31020; Bryans et al. people, International Publication WO 00/50027; and Bryans et al., International Publication WO 02/00209). The compounds and/or compositions of the present invention may be used for the treatment or prevention of epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases, panic by methods described in the art (see above) , pain (especially neuropathic pain and musculoskeletal pain), inflammatory disease (ie, arthritis), insomnia, gastrointestinal disorders, and alcohol withdrawal syndrome. Thus, those skilled in the art will be able to analyze and use the compounds and/or compositions of the present invention for the treatment or prevention of epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases, panic, pain (especially neuropathic pain and muscular and skeletal pain), inflammatory diseases (ie, arthritis), insomnia, gastrointestinal disorders, and alcohol withdrawal syndrome.
4.5治疗/预防给药 4.5 Therapeutic/Prophylactic Dosing
本发明的化合物和/或组合物可以有利地用于人医学。如以上部分4.4所述,本发明的化合物或组合物用于治疗或预防癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病或乙醇戒断综合征。The compounds and/or compositions of the invention can advantageously be used in human medicine. As described in section 4.4 above, the compounds or compositions of the present invention are useful in the treatment or prevention of epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases, panic, pain (especially neuropathic pain) and muscle and bone pain), inflammatory disease (ie, arthritis), insomnia, gastrointestinal disease, or alcohol withdrawal syndrome.
本发明的化合物或组合物在用于治疗或预防上述疾病或病症时,可以单一、与其它试剂联合施用或应用。本发明的化合物和/或组合物还可以单一、与其它药学活性试剂,包括本发明的其它化合物联合施用或应用。When the compound or composition of the present invention is used to treat or prevent the above-mentioned diseases or conditions, it can be administered or applied singly or in combination with other agents. The compounds and/or compositions of the present invention may also be administered or used singly, in combination with other pharmaceutically active agents, including other compounds of the present invention.
本发明提供通过给患者施用治疗有效量的本发明的组合物或化合物而进行治疗和预防的方法。所述患者可以是动物,更优选为哺乳动物,最优选为人。The invention provides methods of treatment and prevention by administering to a patient a therapeutically effective amount of a composition or compound of the invention. The patient may be an animal, more preferably a mammal, most preferably a human.
本发明的化合物或组合物,包含一种或多种本发明的化合物,优选进行口服。本发明的化合物和/或组合物还可以通过其它常规途径给药,例如通过灌输或丸剂注射,通过上皮或粘膜与皮肤衬层(例如口粘膜、直肠和肠粘膜等)进行给药。给药可以是系统给药或局部给药。多种递送系统是已知的可以用于施用本发明的化合物和或组合物的递送系统(例如胶囊包束于脂质体、微粒、微胶囊、胶囊等)。给药方法包括但不限于皮内、肌内、腹膜内、静脉内、皮下、鼻内、硬膜外、口、舌下、鼻内、脑内、阴道内、透皮、直肠、吸入或局部给药,特别对耳、鼻、眼或皮肤进行给药。A compound or composition of the invention, comprising one or more compounds of the invention, is preferably administered orally. The compounds and/or compositions of the present invention may also be administered by other conventional routes, such as by infusion or bolus injection, through epithelial or mucosal and skin linings (eg, oral mucosa, rectal and intestinal mucosa, etc.). Administration can be systemic or local. A variety of delivery systems are known that can be used to administer the compounds and or compositions of the invention (eg, encapsulation in liposomes, microparticles, microcapsules, capsules, etc.). Methods of administration include, but are not limited to, intradermal, intramuscular, intraperitoneal, intravenous, subcutaneous, intranasal, epidural, oral, sublingual, intranasal, intracerebral, intravaginal, transdermal, rectal, inhalation, or topical Administration, in particular to the ear, nose, eye or skin.
在特别优选的实施方案中,本发明的化合物和/或组合物可以通过持续释放系统,优选口服持续释放系统递送。在一个实施方案中,可以使用泵(参见Langer,supra;Sefton,1987,CRC Crit Ref BiomedEng.14:201;Saudek等人,1989,N.Engl.J Med.321:574)。In a particularly preferred embodiment, the compounds and/or compositions of the invention may be delivered by a sustained release system, preferably an oral sustained release system. In one embodiment, a pump can be used (see Langer, supra; Sefton, 1987, CRC Crit Ref Biomed Eng. 14:201; Saudek et al., 1989, N. Engl. J Med. 321:574).
在另一个实施方案中,可以使用聚合材料(参见“MedicalApplications of Controlled Release(控释医药应用),”Langer和Wise(eds.),CRC Pres.,Boca Raton,Florida(1974);“Controlled Drug Bioavailability(受控的药物生物利用度),”Drug Product Design和Performance,Smolen和Ball(eds.),Wiley,New York(1984);Ranger和Peppas,1983,J Macromol.Sci.Rev.Macromol Chem.23:61;还参见Levy等人,1985,Science 228:190;During等人,1989,Ann.Neurol.25:351;Howard等人,1989,J.Neurosurg.71:105)。在一个优选的实施方案中,将聚合材料用于口服持续释放递送。优选的聚合物包括羧甲基纤维素钠、羟丙基纤维素、羟丙基甲基纤维素和羟乙基纤维素(最优选羟基丙基甲基纤维素)。其它优选的纤维素醚已被描述(Alderman,Int.J.Pharm.Tech.& Prod.Mfr.,1984,5(3)1-9)。影响药物释放的因素对于本领域技术人员来说是已知的,并描述在本领域中(Bamba等人,Int.J.Pharm.,1979,2,307)。In another embodiment, polymeric materials can be used (see "Medical Applications of Controlled Release (controlled release pharmaceutical applications)," Langer and Wise (eds.), CRC Press., Boca Raton, Florida (1974); "Controlled Drug Bioavailability (Controlled Drug Bioavailability), "Drug Product Design and Performance, Smolen and Ball (eds.), Wiley, New York (1984); Ranger and Peppas, 1983, J Macromol. Sci. Rev. Macromol Chem.23 : 61; see also Levy et al., 1985, Science 228: 190; During et al., 1989, Ann. Neurol. 25: 351; Howard et al., 1989, J. Neurosurg. 71: 105). In a preferred embodiment, polymeric materials are used for oral sustained release delivery. Preferred polymers include sodium carboxymethylcellulose, hydroxypropylcellulose, hydroxypropylmethylcellulose and hydroxyethylcellulose (most preferably hydroxypropylmethylcellulose). Other preferred cellulose ethers have been described (Alderman, Int. J. Pharm. Tech. & Prod. Mfr., 1984, 5(3) 1-9). Factors affecting drug release are known to those skilled in the art and described in the art (Bamba et al., Int. J. Pharm., 1979, 2, 307).
在另一个实施方案中,可以将肠衣制剂用于口服持续释放给药。优选的包衣材料包括具有依赖于pH的稳定性(即pH控制的释放)的聚合物、具有缓慢或依赖于pH的膨胀、溶解或侵蚀速率(即时间控制的释放)的聚合物,通过酶降解(即酶控释放)的聚合物和形成通过增压而被破坏薄膜层(即压力控制释放)的聚合物。In another embodiment, enteric coated formulations can be used for oral sustained release administration. Preferred coating materials include polymers with pH-dependent stability (i.e., pH-controlled release), polymers with slow or pH-dependent swelling, dissolution or erosion rates (i.e., time-controlled release), by enzymatic Polymers that degrade (ie, enzyme-controlled release) and polymers that form thin film layers that are disrupted by pressurization (ie, pressure-controlled release).
在另一个实施方案中,将渗透递送系统用于口服释放给药(Verma等人,Drug Dev.Ind.Pharm.,2000,26:695-708)。在一个优选的实施方案中,将OROSTM渗透装置用于口服持续释放递送装置(Theeuwes等人,美国专利3,845,770;Theeuwes等人,美国专利3,916,899)。In another embodiment, an osmotic delivery system is used for oral release administration (Verma et al., Drug Dev. Ind. Pharm., 2000, 26:695-708). In a preferred embodiment, OROS ™ osmotic devices are used in oral sustained release delivery devices (Theeuwes et al., US Patent 3,845,770; Theeuwes et al., US Patent 3,916,899).
在另一个实施方案中,可以将控释系统放置在本发明的化合物和/或组合物的靶体附近,从而仅需要部分系统剂量(例如参见Goodson,in“Medical Applications of Controlled Release(控释的医学应用),”supra,vol.2,pp.115-138(1984))。还可以应用在Langer,1990,Science 249:1527-1533中所述的其它控释系统。In another embodiment, a controlled release system can be placed near the target of the compounds and/or compositions of the invention so that only a fraction of the systemic dose is required (see, e.g., Goodson, in "Medical Applications of Controlled Release") Medical applications), "supra, vol.2, pp.115-138 (1984)). Other controlled release systems as described in Langer, 1990, Science 249: 1527-1533 may also be used.
本发明的化合物和/或组合物优选在对患者进行体内给药时提供GABA类似物(例如加巴喷丁和pregablin)。虽然不意在受理论限制,本发明的化合物和/或组合物的基元可以经化学和/或酶促裂解。在哺乳动物的胃、肠腔、肠组织、血液、肝、脑或任何适宜的组织中存在的一种或多种酶可以酶裂解本发明的化合物或组合物的基元。裂解机理对于本发明而言不是重要的。优选通过从本发明的化合物裂解前药而形成的GABA类似物不含大量的内酰胺污染物(优选小于0.5wt%,更优选小于0.2wt%,最优选小于0.1wt%)。由本发明的前药释放内酰胺污染物的程度可以使用标准体外分析方法评价。The compounds and/or compositions of the invention preferably provide a GABA analog (eg gabapentin and pregablin) when administered in vivo to a patient. While not intending to be bound by theory, the moieties of the compounds and/or compositions of the invention may be chemically and/or enzymatically cleaved. One or more enzymes present in the stomach, lumen, intestinal tissue, blood, liver, brain, or any suitable tissue of a mammal can enzymatically cleave a moiety of a compound or composition of the invention. The cleavage mechanism is not critical to the invention. Preferably, GABA analogs formed by cleavage of prodrugs from compounds of the invention do not contain significant lactam contamination (preferably less than 0.5 wt%, more preferably less than 0.2 wt%, most preferably less than 0.1 wt%). The extent to which lactam contaminants are released by the prodrugs of the invention can be assessed using standard in vitro assays.
虽然不意在受理论限制,本发明的化合物和/或组合物的基元可以在被胃肠道吸收前(例如在胃或肠腔内)和/或在被胃肠道吸收后(例如在肠组织、血液、肝或其它适宜的哺乳动物的组织)被裂解。如果本发明的化合物的基元在被肠道吸收前被裂解,则所得的GABA类似物可以常规地吸收进入系统循环(例如通过位于小肠的大的中性氨基酸转运剂)。如果本发明的化合物的基元在被胃肠道吸收后裂解,则这些GABA类似物前药可以有机会通过被动扩散、活性转运或者同时进行被动扩散和活性转运而被吸收进入系统循环。While not intending to be bound by theory, the compounds and/or compositions of the invention may be moieties prior to absorption from the gastrointestinal tract (e.g., in the stomach or intestinal lumen) and/or after absorption from the gastrointestinal tract (e.g., in the intestinal tract). tissue, blood, liver or other suitable mammalian tissue) is lysed. If the moiety of the compound of the invention is cleaved prior to intestinal absorption, the resulting GABA analog can be routinely absorbed into the systemic circulation (eg, via large neutral amino acid transporters located in the small intestine). If the moiety of the compounds of the invention is cleaved after absorption from the gastrointestinal tract, these GABA analog prodrugs may have the opportunity to be absorbed into the systemic circulation by passive diffusion, active transport, or both passive diffusion and active transport.
如果本发明的化合物的基元在被胃肠道吸收后裂解,则这些GABA类似物前药可以有机会从大肠吸收进入系统循环。在这种情况下,本发明的化合物或组合物优选作为持续释放系统给药。在一个优选的实施方案中,本发明的化合物或组合物通过口服持续释放给药而递送。优选在此实施方案中,本发明的化合物或组合物每天二次给药(更优选每天一次)。If the moieties of the compounds of the invention are cleaved after absorption from the gastrointestinal tract, these GABA analog prodrugs may have the opportunity to be absorbed from the large intestine into the systemic circulation. In this case, the compounds or compositions according to the invention are preferably administered as a sustained release system. In a preferred embodiment, the compound or composition of the invention is delivered by oral sustained release administration. Preferably in this embodiment, the compound or composition of the invention is administered twice a day (more preferably once a day).
4.6本发明的组合物 4.6 Compositions of the invention
本发明的组合物包含治疗有效量的一种或多种优选为纯化形式的本发明的化合物和适量的药学上可接受的赋形剂,以给患者提供适宜的给药形式。当对患者进行给药时,本发明的化合物和药学上可接受的赋形剂优选为无菌。当静脉内施用本发明的化合物时,水是一种优选的赋形剂。还可以将盐水溶液和右旋糖与甘油水溶液用作液体赋形剂,特别是用于注射液。适宜的药学赋形剂还包括诸如以下的赋形剂:淀粉、葡萄糖、乳糖、蔗糖、明胶、麦芽、大米、面粉、白垩、硅胶、硬脂酸钠、单硬脂酸甘油酯、滑石、氯化钠、无水脱脂乳、甘油、丙二醇、水、乙醇等。如果需要的话,本发明的组合物还可以包含少量的润湿剂和乳化剂或者pH缓冲剂。此外,可以使用助剂、稳定剂、增稠剂、润滑剂和着色剂。The compositions of the invention comprise a therapeutically effective amount of one or more compounds of the invention, preferably in purified form, and a suitable amount of a pharmaceutically acceptable excipient to provide a suitable dosage form for the patient. When administered to a patient, the compounds of the invention and pharmaceutically acceptable excipients are preferably sterile. Water is a preferred vehicle when administering the compounds of the invention intravenously. Saline solutions and aqueous dextrose and glycerol solutions can also be employed as liquid excipients, particularly for injectable solutions. Suitable pharmaceutical excipients also include excipients such as starch, glucose, lactose, sucrose, gelatin, malt, rice, flour, chalk, silica gel, sodium stearate, glyceryl monostearate, talc, chlorine Sodium chloride, anhydrous skimmed milk, glycerin, propylene glycol, water, ethanol, etc. The compositions of the present invention, if desired, can also contain minor amounts of wetting and emulsifying agents, or pH buffering agents. In addition, auxiliaries, stabilizers, thickeners, lubricants and colorants may be used.
在一个实施方案中,本发明的组合物不含有通过分子内环化形成的内酰胺副产物。在一个优选的实施方案中,本发明的组合物还对于长期储存(优选大于1年)是稳定的,并不会形成大量内酰胺(优选少于0.5wt%内酰胺,更优选少于0.2wt%内酰胺,最优选少于0.1wt%内酰胺)。In one embodiment, the compositions of the invention are free of lactam by-products formed by intramolecular cyclization. In a preferred embodiment, the compositions of the present invention are also stable for long-term storage (preferably greater than 1 year) and do not form significant amounts of lactams (preferably less than 0.5 wt. % lactams, more preferably less than 0.2 wt. % lactam, most preferably less than 0.1 wt% lactam).
含有本发明的化合物的药物组合物可以由常规的混合、溶解、制粒、制糖衣丸、研磨、乳化、胶囊化、截留(entrapping)或冻干方法制备。所述药物组合物可以常规的方式,使用一种或多种生理学上可接受的载体、稀释剂、赋形剂或助剂制备,从而有利于将本发明的化合物制成可以药用的制剂。适宜的制剂取决于所选择的给药途径。Pharmaceutical compositions containing the compounds of this invention can be prepared by conventional methods of mixing, dissolving, granulating, dragee-making, levigating, emulsifying, encapsulating, entrapping or lyophilizing. The pharmaceutical compositions can be prepared in a conventional manner using one or more physiologically acceptable carriers, diluents, excipients or auxiliaries, which facilitate the preparation of the compounds of the present invention into pharmaceutically acceptable preparations. Proper formulation is dependent upon the route of administration chosen.
本发明的组合物的剂型可以是溶液、悬浮液、乳液、片剂、丸剂、药丸、胶囊、含有液体的胶囊、粉末、持续释放制剂、栓剂、乳液、气溶胶、喷雾剂、悬浮液或任何其它适于应用的形式。在一个实施方案中,药学上可接受的赋形剂是胶囊(例如参见Grosswald等人,美国专利5,698,155)。其它适宜的药物赋形剂的实例已描述在本领域中(参见Remington’s Pharmaceutical Sciences(Remington药物科学),Philadelphia College of Pharmacy and Science(费城药物学和科学学院),第17版,1985)。将优选的本发明的组合物制备用于口服递送,特别是用于口服持续释放给药。Dosage forms of the compositions of the present invention may be solutions, suspensions, emulsions, tablets, pills, pills, capsules, capsules containing liquids, powders, sustained release formulations, suppositories, emulsions, aerosols, sprays, suspensions or any other forms suitable for the application. In one embodiment, the pharmaceutically acceptable excipient is a capsule (see eg Grosswald et al., US Patent 5,698,155). Examples of other suitable pharmaceutical excipients have been described in the art (see Remington's Pharmaceutical Sciences (Remington Pharmaceutical Sciences), Philadelphia College of Pharmacy and Science (Philadelphia College of Pharmacy and Science), 17th Edition, 1985). Preferred compositions of the invention are prepared for oral delivery, especially for oral sustained release administration.
例如,口服递送的组合物可以是片剂、锭剂、含水或含油悬浮物、颗粒剂、散剂、乳液、胶囊、糖浆或酏剂。口服组合物可以包含一种或多种任选的试剂,例如甜味剂如果糖、天冬甜素或糖精、调味剂如薄荷、冬青油或樱桃着色剂和防腐剂,以提供药学上可口的制剂。而且在片剂或丸剂的情况下,可以将所述组合物包衣以延迟在胃肠道降解和吸收,从而提供长期的持续作用。包围渗透活性驱动化合物的选择性渗透性膜也适用于口服给药的本发明的化合物和组合物。在这些后来的平台中,来自胶囊周围环境的液体被驱动化合物吸收,所述化合物通过孔径膨胀以取代试剂或试剂组合物。这些递送平台与即时速率制剂的锥形图案相比可以提供基本上零级递送曲线。还可以使用延时材料如单硬脂酸甘油酯或硬脂酸甘油酯。口服组合物可以包括标准赋形剂如甘露醇、乳糖、淀粉、硬脂酸镁、糖精钠、纤维素、碳酸镁等。这些赋形剂优选为药用等级。For example, compositions for oral delivery may be tablets, lozenges, aqueous or oily suspensions, granules, powders, emulsions, capsules, syrups or elixirs. Oral compositions may contain one or more optional agents, such as sweetening agents such as fructose, aspartame or saccharin, flavoring agents such as peppermint, oil of wintergreen or cherry coloring agents and preservatives, to provide a pharmaceutically palatable formulation. preparation. Also in the case of tablets or pills, the composition may be coated to delay degradation and absorption in the gastrointestinal tract, thereby providing long lasting action. Selectively permeable membranes surrounding osmotically active driving compounds are also suitable for orally administered compounds and compositions of the invention. In these later platforms, fluid from the surrounding environment of the capsule is absorbed by the drive compound, which expands through the pores to displace the reagent or reagent composition. These delivery platforms can provide an essentially zero order delivery profile compared to the tapered pattern of an immediate rate formulation. A time delay material such as glyceryl monostearate or glyceryl stearate may also be employed. Oral compositions can include standard excipients such as mannitol, lactose, starch, magnesium stearate, sodium saccharine, cellulose, magnesium carbonate, and the like. These excipients are preferably of pharmaceutical grade.
对于口服液体制剂,例如悬浮液、酏剂和溶液,适宜的载体、赋形剂或稀释剂包括水、盐水、烷撑二醇(例如丙二醇)、聚烷撑二醇(例如聚乙二醇)油、醇、pH4和pH6之间的微酸性缓冲剂(例如大约5mM至大约50mM的乙酸盐、柠檬酸盐、抗坏血酸盐)等。此外,可以加入调味剂、防腐剂、着色剂、胆盐、酰基内毒碱等。For oral liquid preparations such as suspensions, elixirs and solutions, suitable carriers, excipients or diluents include water, saline, alkylene glycols (such as propylene glycol), polyalkylene glycols (such as polyethylene glycol) Oils, alcohols, slightly acidic buffers between pH 4 and pH 6 (eg, about 5 mM to about 50 mM acetate, citrate, ascorbate), and the like. In addition, flavoring agents, preservatives, coloring agents, bile salts, acyl endoxaloids, etc. may be added.
还可以考虑通过其它途径对组合物进行给药。对于颊给药,组合物的剂型可以是以常规方法制备的片剂、锭剂等。适于与喷雾器和液体喷雾装置和EHD气溶胶装置一起应用的液体药物制剂一般包括本发明的化合物和药学上可接受的赋形剂。优选药学上可接受的赋形剂是液体如醇、水、聚乙二醇或全氟化碳。任选加入另一种物质改变本发明的化合物的溶液或悬浮液的气溶胶性能。优选此物质为液体如醇、乙二醇、聚乙二醇或脂肪酸。其它适用于气溶胶装置的液体药物溶液或悬浮液的制备方法对本领域技术人员来说是已知的(例如参见Biesalski,美国专利5,112,598;Biesalski,美国专利5,556,611)。本发明的化合物还可以制成直肠或阴道组合物如栓剂或保持灌肠剂,例如它包含常规栓剂基质如可可油或或其它甘油酯。除了前述的制剂之外,还可以将本发明的化合物制成沉淀制剂。这种长期作用制剂可以通过植入(例如皮下或肌内)或通过肌内注射进行给药。因此,例如本发明的化合物可以与适宜的聚合或疏水材料(例如在可接受的油中的乳液)或离子交换树脂一起制备,或者制成少量溶解的衍生物,例如少量溶解的盐。Administration of the compositions by other routes is also contemplated. For buccal administration, the dosage form of the composition may be tablets, lozenges and the like prepared in a conventional manner. Liquid pharmaceutical formulations suitable for use with nebulizers and liquid spray devices and EHD aerosol devices generally comprise a compound of the invention and a pharmaceutically acceptable excipient. Preferred pharmaceutically acceptable excipients are liquids such as alcohols, water, polyethylene glycol or perfluorocarbons. Optionally adding another substance modifies the aerosol properties of the solution or suspension of the compound of the invention. Preferably the substance is a liquid such as alcohol, glycol, polyethylene glycol or fatty acid. Other methods of preparing liquid drug solutions or suspensions suitable for aerosol devices are known to those skilled in the art (see, eg, Biesalski, US Patent 5,112,598; Biesalski, US Patent 5,556,611). The compounds of the invention may also be formulated in rectal or vaginal compositions such as suppositories or retention enemas, eg containing conventional suppository bases such as cocoa butter or other glycerides. In addition to the aforementioned formulations, the compounds of the present invention may also be formulated as a precipitation formulation. Such long acting formulations may be administered by implantation (eg subcutaneously or intramuscularly) or by intramuscular injection. Thus, for example, the compounds of the invention may be prepared with suitable polymeric or hydrophobic materials (eg, emulsions in acceptable oils) or ion exchange resins, or as slightly soluble derivatives, eg, as slightly soluble salts.
当本发明的化合物为酸性时,它可以作为游离酸、药学上可接受的盐、溶剂化物或水合物包含在任何上述的制剂中。药学上可接受的盐基酊上保留了游离酸的活性,可以通过与碱反应制备,并趋于比对应的游离酸形式更溶于水和其它质子溶剂。When the compound of the present invention is acidic, it may be included in any of the above formulations as the free acid, pharmaceutically acceptable salt, solvate or hydrate. Pharmaceutically acceptable base tinctures retain the activity of the free acids, can be prepared by reaction with bases, and tend to be more soluble in water and other protic solvents than the corresponding free acid forms.
4.7应用方法和剂量 4.7 Application method and dosage
本发明的化合物或其组合物一般以有效量应用以实现期望的目的。用于治疗或预防以下诸如以下疾病或病症的应用:癫痫、抑郁、焦虑、精神病、昏厥发作、运动机能减退、颅异常、神经变性疾病、恐慌、疼痛(特别是神经病性疼痛和肌肉和骨骼疼痛)、炎性疾病(即关节炎)、失眠、胃肠疾病或乙醇戒断综合征,本发明的化合物或其组合物以治疗有效量施用或应用。The compounds of the present invention, or compositions thereof, are generally employed in an effective amount to achieve the desired purpose. Use for the treatment or prevention of diseases or conditions such as epilepsy, depression, anxiety, psychosis, syncope, hypokinesia, cranial abnormalities, neurodegenerative diseases, panic, pain (especially neuropathic pain and muscular and skeletal pain) ), inflammatory disease (ie arthritis), insomnia, gastrointestinal disease or alcohol withdrawal syndrome, the compounds of the present invention or compositions thereof are administered or applied in a therapeutically effective amount.
本文公开的有效地治疗特定疾病或病症的本发明的化合物的数量取决于疾病或病症的性质,并可以由前述的本领域中已知的标准临床技术确定。此外,体外或体内试验可以任选用于帮助鉴定最佳剂量范围。本发明的化合物的给药数量当然取决于治疗的受试者、受试者的重量、病痛的严重程度、给药方式和处方医师的判断。The amount of a compound of the invention disclosed herein that is effective in treating a particular disease or condition depends on the nature of the disease or condition and can be determined by standard clinical techniques known in the art as previously described. In addition, in vitro or in vivo assays can optionally be used to help identify optimal dosage ranges. The amount of a compound of the invention administered will of course depend on the subject being treated, the subject's weight, the severity of the affliction, the mode of administration and the judgment of the prescribing physician.
例如,可以通过单一给药、多次应用或控释而在药物组合物中递送剂量。在一个优选的实施方案中,本发明的化合物通过口服持续释放给药递送。优选在此实施方案中,本发明的化合物每天施用二次(更优选每天一次)。剂量可以间断地重复,可以单独或与其它药物联合提供,并可以持续有效地治疗疾病或病症所需的时间长度。For example, dosages can be delivered in pharmaceutical compositions by single administration, multiple applications, or controlled release. In a preferred embodiment, the compounds of the invention are delivered by oral sustained release administration. Preferably in this embodiment, the compound of the invention is administered twice a day (more preferably once a day). Doses can be repeated intermittently, given alone or in combination with other agents, and can be continued for the length of time necessary to effectively treat the disease or condition.
适宜的口服剂量范围取决于母体GABA类似物药物的效力,但一般为大约0.001mg至大约200mg本发明的化合物/千克体重。当GABA类似物为加巴喷丁时,典型的成人的母体药物的日剂量为900mg/天至3600mg/天,而加巴喷丁前药的剂量可以调节至提供等摩尔量的加巴喷丁。其它GABA类似物可以比加巴喷丁(例如pregabalin)更为有效,而且较低剂量对母体药物和任何前药是适宜的(在等摩尔的基础上测定)。可以由本领域技术人员已知的方法容易地确定剂量范围。A suitable oral dosage range will depend on the potency of the parent GABA analog drug, but will generally be from about 0.001 mg to about 200 mg of the compound of the invention/kg body weight. When the GABA analog is gabapentin, typical adult daily doses of the parent drug are 900 mg/day to 3600 mg/day, while the dose of the gabapentin prodrug can be adjusted to provide equimolar amounts of gabapentin. Other GABA analogs may be more potent than gabapentin (eg pregabalin), and lower doses are appropriate (measured on an equimolar basis) for the parent drug and any prodrugs. Dosage ranges can be readily determined by methods known to those skilled in the art.
本发明的化合物优选在用于人之前进行在体外和体内目标治疗或预防活性的试验。例如,体外试验可以用于确定本发明的特定化合物或本发明的化合物的组合物的给药是否优选用于减少惊厥。使用动物模型系统还可以证明本发明的化合物是有效和安全的。The compounds of the invention are preferably tested for in vitro and in vivo therapeutic or prophylactic activity of interest prior to use in humans. For example, in vitro assays can be used to determine whether administration of a particular compound of the invention or a combination of compounds of the invention is preferred for reducing convulsions. Compounds of the invention can also be demonstrated to be effective and safe using animal model systems.
优选治疗有效剂量的本文所述的本发明的化合物将提供治疗益处同时基本上不导致毒性。本发明的化合物的毒性可以使用标准药学方法测定,并可以容易地由本领域技术人员确定。毒性与治疗作用的剂量比是治疗指数。本发明的化合物优选表现出特别高的治疗疾病和病症的治疗指数。Preferably, a therapeutically effective dose of a compound of the invention described herein will provide therapeutic benefit while causing substantially no toxicity. The toxicity of the compounds of the present invention can be determined using standard pharmaceutical methods and can be readily determined by one skilled in the art. The dose ratio of toxic to therapeutic effects is the therapeutic index. The compounds of the invention preferably exhibit particularly high therapeutic indices for the treatment of diseases and disorders.
本文所述的本发明的化合物的剂量在包括具有少量毒性或没有毒性的有效剂量的循环浓度范围之内。The dosage of the compounds of the invention described herein lies within a range of circulating concentrations that include the effective dose with little or no toxicity.
4.8.联合治疗 4.8. Combination therapy
在本发明的某些实施方案中,本发明的化合物可以与至少一种其它治疗剂用于联合治疗。本发明的化合物和治疗剂可以起加合,更优选协同作用。在一个优选的实施方案中,含有本发明的化合物的组合物与其它治疗剂同时给药,所述治疗剂可以与本发明的化合物一样作为组合物的一部分或作为不同的组合物。在另一个实施方案中,包含本发明的化合物的组合物可以在另一种治疗给药之前或之后给药。In certain embodiments of the invention, compounds of the invention may be used in combination therapy with at least one other therapeutic agent. Compounds of the invention and therapeutic agents may act additively, more preferably synergistically. In a preferred embodiment, compositions containing compounds of the invention are administered concurrently with other therapeutic agents, which may be part of the same composition as the compounds of the invention or as a different composition. In another embodiment, a composition comprising a compound of the invention may be administered before or after another therapeutic administration.
5.实施例 5. Example
参照以下实施例进一步定义本发明,所述实施例详述描述本发明的化合物和组合物的制备以及本发明的化合物和组合物的应用试验。本领域技术人员可以明显看出在不背离本发明的保护范围的情况下可以对原料和方法实施多种改型。The invention is further defined with reference to the following examples which describe in detail the preparation of the compounds and compositions of the invention and the testing of the compounds and compositions of the invention. It will be apparent to those skilled in the art that various modifications can be made in the materials and methods without departing from the scope of the invention.
在以下的实施例中,以下的缩写具有以下含义。如果没有定义缩写,则它具有其通常接受的含义。In the following examples, the following abbreviations have the following meanings. If an abbreviation is not defined, it has its generally accepted meaning.
AIBN = 2,2’-偶氮双(异丁腈)AIBN = 2,2'-Azobis(isobutyronitrile)
Atm = 大气压Atm = atmospheric pressure
Boc = 叔丁氧基羰Boc = tert-butoxycarbonyl
Cbz = 苄氧基(carbobenzyloxy)Cbz = Benzyloxy (carbobenzyloxy)
CPM = 每分钟计数CPM = counts per minute
DCC = 二环己基碳化二亚胺DCC = Dicyclohexylcarbodiimide
DMAP = 4-N,N-二甲基氨基吡啶DMAP = 4-N, N-dimethylaminopyridine
DMEM = 极限eagl e培养基DMEM = extreme eagle medium
DMF = N,N-二甲基甲酰胺DMF = N,N-Dimethylformamide
DMSO = 二甲亚砜DMSO = dimethyl sulfoxide
Fmoc = 9-芴基甲氧羰基Fmoc = 9-fluorenylmethoxycarbonyl
g = 克g = gram
h = 小时h = hours
HBSS = Hanks缓冲盐溶液HBSS = Hanks buffered saline solution
L = 升L = liter
LC/MS = 液相色谱/质谱LC/MS = liquid chromatography/mass spectrometry
M = 摩尔M = mole
min = 分min = points
ml = 毫升ml = milliliter
mmol = 毫摩尔mmol = millimole
NBS = N-溴琥珀酰亚胺NBS = N-Bromosuccinimide
NHS = N-羟基琥珀酰亚胺NHS = N-hydroxysuccinimide
PBS = 磷酸盐缓冲盐水PBS = Phosphate Buffered Saline
THF = 四氢呋喃THF = Tetrahydrofuran
TFA = 三氟乙酸TFA = trifluoroacetic acid
TMS = 三甲基甲硅烷基TMS = Trimethylsilyl
μL = 微升μL = microliter
μM = 微摩尔μM = micromole
v/v = 体积比体积v/v = volume to volume
实施例1Example 1
1-{[(α-新戊酰氧基甲氧基)羰基]氨基甲基}-1-环己烷乙酸(51)1-{[(α-pivaloyloxymethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (51)
步骤A: 对硝基苯基碳酸氯甲酯(52) Step A: p-nitrophenyl chloromethyl carbonate (52)
将对硝基苯酚(100g,0.72moles)溶于无水四氢呋喃(3L)并剧烈搅拌。室温下往此溶液加入氯甲酸氯甲酯(70mL,0.79moles),然后加入三乙基胺(110mL)。搅拌1h后,将反应混合物过滤并将滤液浓缩,然后用乙酸乙酯(1L)稀释。用10%碳酸钾(3×500mL)和1NHCl(2×300mL)、盐水(2×300mL)洗涤有机溶液,并用无水硫酸钠干燥。除去溶剂得到157g(95%)的标题化合物(52),为一种固体。所述化合物对LC-MS不稳定。1H NMR(CDCl3,400MHz):5.86(s,2H),7.44(d,J=9Hz,2H),8.33(d,J=9Hz,2H)。p-Nitrophenol (100 g, 0.72 moles) was dissolved in anhydrous THF (3 L) and stirred vigorously. To this solution was added chloromethyl chloroformate (70 mL, 0.79 moles) followed by triethylamine (110 mL) at room temperature. After stirring for 1 h, the reaction mixture was filtered and the filtrate was concentrated, then diluted with ethyl acetate (1 L). The organic solution was washed with 10% potassium carbonate (3 x 500 mL) and 1N HCl (2 x 300 mL), brine (2 x 300 mL), and dried over anhydrous sodium sulfate. Removal of the solvent gave 157 g (95%) of the title compound (52) as a solid. The compound is unstable to LC-MS. 1 H NMR (CDCl 3 , 400 MHz): 5.86 (s, 2H), 7.44 (d, J=9 Hz, 2H), 8.33 (d, J=9 Hz, 2H).
步骤B: 对硝基苯基碳酸碘甲酯(53) Step B: iodomethyl p-nitrophenyl carbonate (53)
在氮气氛下将对硝基苯基碳酸氯甲酯(52)(100g,0.43moles)、碘化钠(228g,1.30moles)和50g无水分子筛(4)加到2L丙酮,同时进行机械搅拌。在40℃下将所得的混合物搅拌5h(通过1H NMR监测)。搅拌完成时,过滤除去固体物质,并在减压下除去溶剂。将残余物再溶于二氯甲烷(1L)并用饱和碳酸钠水溶液(300mL)洗涤二次,然后用水(300mL)洗涤。分离有机层并用无水硫酸钠干燥。除去溶剂得到123.6g(89%)的标题化合物(53),在静置时为一种固体。发现所述化合物对LC-MS不稳定。1H NMR(CDCl3,400MHz):6.06(s,2H),7.42(d,J=9Hz,2H),8.30(d,J=9Hz,2H).13C NMR(CDCl3,100MHz):155.1,151.0,146.0,125.8,125.7,121.9,33.5。Under a nitrogen atmosphere, p-nitrophenyl chloromethyl carbonate (52) (100 g, 0.43 moles), sodium iodide (228 g, 1.30 moles) and 50 g of anhydrous molecular sieves (4 Å) were added to 2 L of acetone while mechanically Stir. The resulting mixture was stirred at 40° C. for 5 h (monitored by 1 H NMR). When stirring was complete, the solid material was removed by filtration, and the solvent was removed under reduced pressure. The residue was redissolved in dichloromethane (1 L) and washed twice with saturated aqueous sodium carbonate (300 mL) and then with water (300 mL). The organic layer was separated and dried over anhydrous sodium sulfate. Removal of the solvent gave 123.6 g (89%) of the title compound (53) as a solid on standing. The compound was found to be unstable to LC-MS. 1 H NMR (CDCl 3 , 400MHz): 6.06 (s, 2H), 7.42 (d, J=9Hz, 2H), 8.30 (d, J=9Hz, 2H). 13 C NMR (CDCl 3 , 100MHz): 155.1 , 151.0, 146.0, 125.8, 125.7, 121.9, 33.5.
步骤C: 三甲基乙酸银(54) Step C: Silver trimethyl acetate (54)
将新戊酸(50g,0.49moles)溶于乙腈(1.3L),然后加入氧化银(70g,0.29moles),并剧烈搅拌。然后在氮气氛下加入660mL水。将所得的悬浮液于70℃下在暗处搅拌1h。用C盐滤垫过滤后,除去溶剂得到86g(82%)的标题化合物(54),为一种灰白色固体,将其不经进一步纯化用于下一步反应。根据类似的方法制备本申请中所述的其它银盐。Pivalic acid (50 g, 0.49 moles) was dissolved in acetonitrile (1.3 L), then silver oxide (70 g, 0.29 moles) was added with vigorous stirring. Then 660 mL of water was added under nitrogen atmosphere. The resulting suspension was stirred at 70 °C in the dark for 1 h. After filtration through a pad of celite, the solvent was removed to afford 86 g (82%) of the title compound (54) as an off-white solid, which was used in the next reaction without further purification. The other silver salts described in this application were prepared according to similar methods.
步骤D: 新戊酰氧基甲基碳酸对硝基苯基酯(55) Step D: p-nitrophenyl pivaloyloxymethyl carbonate (55)
向在无水甲苯(1L)中的对硝基苯基碳酸碘甲酯(53)(62g,0.19moles)的溶液加入三甲基乙酸银(80g,0.38moles)。在55℃和氮气氛下搅拌3h后,将反应混合物冷却至室温,并用C盐滤垫过滤。用10%碳酸钾(500mL)洗涤滤液。除去溶剂得到43g(75%)的标题化合物(55),为一种黄色的油。1H NMR(CDCl3,400MHz):1.25(s,9H),5.88(s,2H),7.40(d,J=9Hz,2H),8.29(d,J=9HZ,2H).13CNMR(CDCl3,100MHz):177.0,155.3,151.6,145.8,125.6,121.9,83.1,39.1,27.0。To a solution of iodomethyl p-nitrophenyl carbonate (53) (62 g, 0.19 moles) in dry toluene (1 L) was added silver trimethyl acetate (80 g, 0.38 moles). After stirring at 55 °C under nitrogen atmosphere for 3 h, the reaction mixture was cooled to room temperature and filtered through a pad of celite. The filtrate was washed with 10% potassium carbonate (500 mL). Removal of the solvent gave 43 g (75%) of the title compound (55) as a yellow oil. 1 H NMR (CDCl 3 , 400MHz): 1.25(s, 9H), 5.88(s, 2H), 7.40(d, J=9Hz, 2H), 8.29(d, J=9HZ, 2H). 13 CNMR(CDCl 3 , 100MHz): 177.0, 155.3, 151.6, 145.8, 125.6, 121.9, 83.1, 39.1, 27.0.
步骤E: 1-{[(α-新戊酰氧基甲氧基)羰基]氨基甲基}-1-环己烷乙酸 Step E: 1-{[(α-pivaloyloxymethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(51) (51)
在无水二氯甲烷(100mL)中将加巴喷丁游离碱(24g,0.14moles)制浆,然后分别用氯三甲基硅烷(18.6mL,0.28moles)和三乙基胺(10mL,0.15moles)处理。将所得的悬浮液加热,并搅拌至实现任何固体的完全溶解。通过均衡(equalizing)加入漏斗将以上的加巴喷丁溶液加到缓慢回流,并在氮气氛下机械搅拌在二氯甲烷(100mL)中的新戊酰氧基甲基碳酸对硝基苯基酯(55)(20g,67mmol)和三乙基胺(10mL,0.15moles)的溶液。将所得的黄色溶液搅拌1.5h。搅拌完成时(通过茚三酮染色监测),将混合物过滤,并将滤液浓缩。将残余物溶于乙酸乙酯(500mL),用1N HCl(3×100mL)、盐水(2×100mL)洗涤,并用无水硫酸钠干燥。除去溶剂后,将粗产物溶于乙醇(300mL),然后加入1g 5%Pd/C。将所得的混合物在50psi氢大气下搅拌15分钟,然后用C盐滤垫过滤。浓缩后,将残余物溶于乙酸乙酯,用5%H2SO4洗涤,并用无水硫酸钠干燥。在减压下除去溶剂后,用硅胶对残余物进行色谱纯化(4∶1己烷∶乙酸乙酯)得到15g(68%)的标题化合物(51),为一种固体。M.p.:79-81℃;1HNMR(CDCl3,400MHz):1.21(s,9H),1.3-1.5(m,10H),2.32(s,2H),3.26(s,2H),5.33(M,1H),5.73(s,2H).13C NMR(CDCl3,400MHz):21.7,26.2,27.3,34.3,38.2,39.2,80.6,155.9,176.8,178.0.MS(ESI)m/z 328.36(M-H)-,330.32(M+H)+,352.33(M+Na)+。Gabapentin free base (24 g, 0.14 moles) was slurried in anhydrous dichloromethane (100 mL) and treated with chlorotrimethylsilane (18.6 mL, 0.28 moles) and triethylamine (10 mL, 0.15 moles) respectively . The resulting suspension was heated and stirred until complete dissolution of any solids was achieved. Add the above gabapentin solution to slow reflux via an equalizing addition funnel, and mechanically stir p-nitrophenyl pivaloyloxymethylcarbonate (55) in dichloromethane (100 mL) under a nitrogen atmosphere. (20 g, 67 mmol) and triethylamine (10 mL, 0.15 moles). The resulting yellow solution was stirred for 1.5 h. When stirring was complete (monitored by ninhydrin staining), the mixture was filtered and the filtrate was concentrated. The residue was dissolved in ethyl acetate (500 mL), washed with 1N HCl (3 x 100 mL), brine (2 x 100 mL), and dried over anhydrous sodium sulfate. After removing the solvent, the crude product was dissolved in ethanol (300 mL), and then 1 g of 5% Pd/C was added. The resulting mixture was stirred under a 50 psi hydrogen atmosphere for 15 minutes, then filtered through a pad of celite. After concentration, the residue was dissolved in ethyl acetate, washed with 5% H2SO4 , and dried over anhydrous sodium sulfate . After removal of the solvent under reduced pressure, the residue was chromatographed on silica gel (4:1 hexane:ethyl acetate) to afford 15 g (68%) of the title compound (51) as a solid. Mp: 79-81°C; 1 HNMR (CDCl 3 , 400MHz): 1.21 (s, 9H), 1.3-1.5 (m, 10H), 2.32 (s, 2H), 3.26 (s, 2H), 5.33 (M, 1H), 5.73(s, 2H). 13 C NMR (CDCl 3 , 400MHz): 21.7, 26.2, 27.3, 34.3, 38.2, 39.2, 80.6, 155.9, 176.8, 178.0. MS (ESI) m/z 328.36 (MH ) - , 330.32(M+H) + , 352.33(M+Na) + .
实施例2Example 2
1-{[(α-乙酸基乙氧基)羰基]氨基甲基}-1-环己烷乙酸(56)1-{[(α-acetoxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (56)
步骤A: 1-氯乙基-对硝基苯基碳酸酯(57) Step A: 1-Chloroethyl-p-nitrophenyl carbonate (57)
向冰冷的含有在二氯甲烷(60mL)中的对硝基苯酚(1.39g,10mmol)和吡啶(0.81g,10mmol)的反应混合物加入氯甲酸1-氯乙酯(1.2mL,11mmol)。将混合物于0℃下搅拌30分钟,然后在室温下搅拌1h。在减压下除去溶剂后,将残余物溶于醚,用水、10%柠檬酸和水洗涤。用Na2SO4干燥醚层,并在减压下蒸发得到2.4g(97%)的标题化合物(57),为一种米色固体。1H NMR(CDCl3):1.93(d,3H),6.55(q,1H),7.42(d,2H),8.28(d,2H)。To an ice-cold reaction mixture containing p-nitrophenol (1.39 g, 10 mmol) and pyridine (0.81 g, 10 mmol) in dichloromethane (60 mL) was added 1-chloroethyl chloroformate (1.2 mL, 11 mmol). The mixture was stirred at 0 °C for 30 min, then at room temperature for 1 h. After removing the solvent under reduced pressure, the residue was dissolved in ether, washed with water, 10% citric acid and water. The ether layer was dried over Na2SO4 and evaporated under reduced pressure to give 2.4 g (97%) of the title compound (57) as a beige solid. 1 H NMR (CDCl 3 ): 1.93 (d, 3H), 6.55 (q, 1H), 7.42 (d, 2H), 8.28 (d, 2H).
步骤B: α-乙酸基乙基-对硝基苯基碳酸酯(58) Step B: α-Acetoxyethyl-p-nitrophenyl carbonate (58)
将在乙酸(15mL)中的1-氯乙基-对硝基苯基碳酸酯(57)(0.5g,2mmol)和乙酸汞(1.5g,4.4mmol)的混合物于室温下搅拌24h。在减压下除去乙酸后,将残余物溶于醚,并用水、0.5%(v/v)NaHCO3水溶液和水洗涤。用Na2SO4干燥醚层,并浓缩至干。用硅胶色谱处理所得的残余物(己烷∶乙酸乙酯(95∶5))得到0.45g(84%)的标题化合物(58)。1H NMR(CDCl3,400MHz):1.55(d,J=5.6Hz,3H),2.07(s,3H),6.78(q,J=5.6Hz,1H),7.36(d,J=9.6Hz,2H),8.22(d,J=9.6Hz,2H)。A mixture of 1-chloroethyl-p-nitrophenyl carbonate (57) (0.5 g, 2 mmol) and mercury acetate (1.5 g, 4.4 mmol) in acetic acid (15 mL) was stirred at room temperature for 24 h. After removal of acetic acid under reduced pressure, the residue was dissolved in ether and washed with water, 0.5% (v/v) aqueous NaHCO 3 and water. The ether layer was dried over Na2SO4 and concentrated to dryness. The resulting residue was chromatographed on silica gel (hexane:ethyl acetate (95:5)) to give 0.45 g (84%) of the title compound (58). 1 H NMR (CDCl 3 , 400MHz): 1.55(d, J=5.6Hz, 3H), 2.07(s, 3H), 6.78(q, J=5.6Hz, 1H), 7.36(d, J=9.6Hz, 2H), 8.22 (d, J = 9.6 Hz, 2H).
步骤C: 1-{[(α-乙酸基乙氧基)羰基]氨基甲基}-1-环己烷乙酸(56) Step C: 1-{[(α-Acetoxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (56)
向含有在二氯甲烷(20mL)中的加巴喷丁(633mg,3.7mmol)和三乙基胺(1.03mL,7.4mmol)的混合物加入三甲基氯硅烷(0.93mL,7.4mmol),并将混合物搅拌直至形成澄清的溶液。加入含有在二氯甲烷(10mL)中的α-乙酸基乙基-对硝基苯基碳酸酯(58)(1g,3.7mmol)的溶液,并将所得的混合物搅拌30分钟。用10%柠檬酸(20mL)洗涤反应混合物,并分离有机层。进一步用醚(3×10mL)萃取含水层,并用MgSO4干燥合并的有机萃取物。过滤后,在减压下除去有机溶剂。用硅胶色谱处理所得的残余物(己烷∶乙酸乙酯(4∶1))得到700mg(63%)的标题化合物(56)。1H NMR(CDCl3,400MHz):1.27-1.60(m,10H),1.55(d,3H),2.08(s,3H),2.38(s,2H),3.25(m,2H),5.31(t,1H),6.81(q,1H).MS(ESI)m/z 302.22(M+H)+。通过溶于水(5mL),加入等摩尔量的0.5N NaHCO3,然后冻干而将酸形式定量转化为对应的钠盐。To a mixture containing gabapentin (633 mg, 3.7 mmol) and triethylamine (1.03 mL, 7.4 mmol) in dichloromethane (20 mL) was added trimethylchlorosilane (0.93 mL, 7.4 mmol), and the mixture was stirred until a clear solution is formed. A solution containing α-acetoxyethyl-p-nitrophenyl carbonate (58) (1 g, 3.7 mmol) in dichloromethane (10 mL) was added, and the resulting mixture was stirred for 30 minutes. The reaction mixture was washed with 10% citric acid (20 mL), and the organic layer was separated. The aqueous layer was further extracted with ether (3 x 10 mL), and the combined organic extracts were dried over MgSO 4 . After filtration, the organic solvent was removed under reduced pressure. The resulting residue was chromatographed on silica gel (hexane:ethyl acetate (4:1)) to give 700 mg (63%) of the title compound (56). 1 H NMR (CDCl 3 , 400MHz): 1.27-1.60(m, 10H), 1.55(d, 3H), 2.08(s, 3H), 2.38(s, 2H), 3.25(m, 2H), 5.31(t , 1H), 6.81 (q, 1H). MS (ESI) m/z 302.22 (M+H) + . The acid form was quantitatively converted to the corresponding sodium salt by dissolving in water (5 mL), adding an equimolar amount of 0.5N NaHCO3 , and then lyophilizing.
实施例3Example 3
1-{[(α苯甲酰氧基苄氧基)羰基]氨基甲基}-1-环己烷乙酸(59)1-{[(α-benzoyloxybenzyloxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (59)
步骤A: α-苯甲酰基苄基碳酸对硝基苯基酯(60) Step A: α-Benzoylbenzyl p-nitrophenyl carbonate (60)
室温下向在60mL CH2Cl2中的安息香(2.0g,9.4mmol)的溶液分别加入DMAP(1.21g,9.9mmol)和对硝基苯基-氯甲酸酯(1.99g,9.9mmol)。室温下搅拌3h后,用水终止反应,并用乙酸乙酯/己烷(2×100mL)萃取。用无水硫酸钠干燥合并的有机萃取物。在减压下除去溶剂得到标题化合物(60),将其不经纯化而用于下一步反应。To a solution of benzoin (2.0 g, 9.4 mmol) in 60 mL CH2Cl2 was added DMAP (1.21 g, 9.9 mmol) and p-nitrophenyl-chloroformate (1.99 g, 9.9 mmol) respectively at room temperature. After stirring at room temperature for 3 h, the reaction was quenched with water and extracted with ethyl acetate/hexane (2 x 100 mL). The combined organic extracts were dried over anhydrous sodium sulfate. The solvent was removed under reduced pressure to give the title compound (60), which was used in the next reaction without purification.
步骤B:1-{[(α-苯甲酰基苄氧基)羰基]氨基甲基}-1-环己烷乙酸 Step B: 1-{[(α-Benzoylbenzyloxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(61) (61)
0℃下向在CH2Cl2中的加巴喷丁(1.70g,9.9mmol)的悬浮液加入三乙基胺(2.76mL,19.8mmol)和TMSCl(2.51mL,19.8mmol)。室温下将反应物搅拌30分钟。向此混合物中加入在CH2Cl2中的化合物(60)(在以上步骤A中制备),并将所得的混合物于室温下搅拌5h。用二氯甲烷稀释反应混合物,用盐水洗涤,并用Na2SO4干燥有机层。在减压下除去溶剂后,用硅胶色谱法纯化残余物,用在CH2Cl2中的5%甲醇洗脱得到3.78g(90%,经过两步)的标题化合物(61)。1H NMR(CDCl3,400MHz):δ1.48-1.35(m,10H),2.30(s,2H),3.24(d,J=7.2Hz,2H),5.58(t,J=6.8Hz,1H),6.85(s,1H),7.50-7.33(m,8H),7.93(d,J=7.2Hz,2H)。To a suspension of gabapentin (1.70 g, 9.9 mmol) in CH2Cl2 was added triethylamine (2.76 mL, 19.8 mmol) and TMSCl (2.51 mL, 19.8 mmol) at 0 °C. The reaction was stirred at room temperature for 30 minutes. To this mixture was added compound (60) in CH2Cl2 (prepared in Step A above), and the resulting mixture was stirred at room temperature for 5 h. The reaction mixture was diluted with dichloromethane, washed with brine, and the organic layer was dried over Na2SO4 . After removal of the solvent under reduced pressure, the residue was purified by silica gel chromatography eluting with 5% methanol in CH2Cl2 to afford 3.78 g (90% over two steps) of the title compound (61). 1 H NMR (CDCl 3 , 400MHz): δ1.48-1.35(m, 10H), 2.30(s, 2H), 3.24(d, J=7.2Hz, 2H), 5.58(t, J=6.8Hz, 1H ), 6.85 (s, 1H), 7.50-7.33 (m, 8H), 7.93 (d, J=7.2Hz, 2H).
步骤C: 1-{[(α-苯甲酰氧基苄氧基)羰基]氨基甲基}-1-环己烷乙酸 Step C: 1-{[(α-benzoyloxybenzyloxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(59) (59)
室温下向在40mLCH2Cl2中的1-{[(α-苯甲酰基苄氧基)羰基]氨基甲基}-1-环己烷乙酸(61)(1.89g,4.6mmol)的溶液分别加入77%mCPBA(2.07g,9.2mmol)和NaHCO3(O.78g,9.2mmol),并将所得的混合物于室温下搅拌过夜。用10%柠檬酸酸化反应混合物,并用CH2Cl2萃取。用盐水洗涤有机萃取物,并用Na2SO4干燥。减压下除去溶剂后,通过反相制备HPLC(乙腈-水,0.1%甲酸)纯化残余物,得到960mg(49%)的标题化合物(59)。1H NMR(CDCl3,400MHz):δ1.58-1.35(m,LOH),2.34(s,2H),3.26(dd,J=6.8,0.8Hz,2H),5.38(t,J=6.8Hz,1H),7.46-7.26(M,5H),7.63-7.55(M,3H),7.89(s,1H),8.08(dd,J=8.8,1.2Hz,2H)。To a solution of 1-{[(α-benzoylbenzyloxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (61) (1.89 g, 4.6 mmol) in 40 mL CH 2 Cl 2 at room temperature 77% mCPBA (2.07 g, 9.2 mmol) and NaHCO 3 (0.78 g, 9.2 mmol) were added, and the resulting mixture was stirred at room temperature overnight. The reaction mixture was acidified with 10% citric acid and extracted with CH2Cl2 . The organic extracts were washed with brine and dried over Na2SO4 . After removing the solvent under reduced pressure, the residue was purified by reverse phase preparative HPLC (acetonitrile-water, 0.1% formic acid) to afford 960 mg (49%) of the title compound (59). 1 H NMR (CDCl 3 , 400MHz): δ1.58-1.35(m, LOH), 2.34(s, 2H), 3.26(dd, J=6.8, 0.8Hz, 2H), 5.38(t, J=6.8Hz , 1H), 7.46-7.26 (M, 5H), 7.63-7.55 (M, 3H), 7.89 (s, 1H), 8.08 (dd, J=8.8, 1.2Hz, 2H).
实施例4Example 4
1-{[(α-乙酸基苄氧基)羰基]氨基甲基}-1-环己烷乙酸(62)1-{[(α-Acetoxybenzyloxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (62)
根据实施例3的方法,并用1-羟基-1-苯基-丙-2-酮代替安息香,得到300mg的标题化合物(62)。1H NMR(CDCl3,400MHz):δ1.41(m,10H),2.19(s,3H),2.33(s,2H),3.27(dd,J=6.6,1.6Hz,2H),5.36(t,J=6.6Hz,1H),7.40(M,3H),7.52(M,2H),7.63(s,1H)。According to the method of Example 3, and substituting benzoin with 1-hydroxy-1-phenyl-propan-2-one, 300 mg of the title compound (62) was obtained. 1 H NMR (CDCl 3 , 400MHz): δ1.41(m, 10H), 2.19(s, 3H), 2.33(s, 2H), 3.27(dd, J=6.6, 1.6Hz, 2H), 5.36(t , J=6.6Hz, 1H), 7.40(M, 3H), 7.52(M, 2H), 7.63(s, 1H).
实施例5Example 5
1-{[(α-苯甲酰氧基乙氧基)羰基氨基甲基}-1-环己烷乙酸(63)1-{[(α-Benzoyloxyethoxy)carbonylaminomethyl}-1-cyclohexaneacetic acid (63)
根据实施例3的方法,并用2-羟基-1-苯基-1-丙酮代替安息香,得以5mg的标题化合物(63)。1H NMR(CDCl3,400MHz):δ1.44-1.36(m,10H),1.62(d,J=5.6Hz,3H),2.34(s,2H),3.24(d,J=6.8Hz,2H),5.28(t,J=6.8Hz,1H),7.06(q.J=5.6Hz,1H),7.44(M,2H),7.56(M,1H),8.03(dd,J=8.4,1.6Hz,2H)。According to the method of Example 3, substituting 2-hydroxy-1-phenyl-1-propanone for benzoin, 5 mg of the title compound (63) was obtained. 1 H NMR (CDCl 3 , 400MHz): δ1.44-1.36(m, 10H), 1.62(d, J=5.6Hz, 3H), 2.34(s, 2H), 3.24(d, J=6.8Hz, 2H ), 5.28(t, J=6.8Hz, 1H), 7.06(qJ=5.6Hz, 1H), 7.44(M, 2H), 7.56(M, 1H), 8.03(dd, J=8.4, 1.6Hz, 2H ).
实施例6Example 6
1-{[(1-苯甲酰氧基-2-苯基乙氧基)羰基]氨基甲基}-1-环己烷乙酸1-{[(1-Benzoyloxy-2-phenylethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(64) (64)
步骤A: 2-苯基-1,3-二噻烷(65) Step A: 2-Phenyl-1,3-dithiane (65)
室温下向在CH2Cl2(150mL)中的苯甲醛(10.6g,100mmol)和1,3-丙烷二硫醇的溶液滴加BF3。将Et2O(6.3mL,50mmol)和所得的混合物于室温下搅拌2h。然后用CH2Cl2稀释反应混合物,过滤并用盐水、饱和NaHCO3、盐水洗涤滤液,并用Na2SO4干燥。在减压下除去溶剂得到-种白色固体,用1∶1的醚和己烷的混合物将其重结晶得到17.0g(87%)的标题化合物(65),为白色结晶针。1H NMR(CDCl3,400MHz):δ1.91(M,1H),2.14(M,1H),2.89(M,2H),3.04(M,2H),5.16(s,1H),7.35-7.28(M,3H),7.46(M,2H)。To a solution of benzaldehyde (10.6 g, 100 mmol) and 1,3 - propaneedithiol in CH2Cl2 (150 mL) was added BF3 dropwise at room temperature. Et2O (6.3 mL, 50 mmol) and the resulting mixture were stirred at room temperature for 2 h. The reaction mixture was then diluted with CH2Cl2 , filtered and the filtrate was washed with brine, saturated NaHCO3 , brine and dried over Na2SO4 . The solvent was removed under reduced pressure to give a white solid, which was recrystallized from a 1:1 mixture of ether and hexanes to give 17.0 g (87%) of the title compound (65) as white crystalline needles. 1 H NMR (CDCl 3 , 400MHz): δ1.91(M, 1H), 2.14(M, 1H), 2.89(M, 2H), 3.04(M, 2H), 5.16(s, 1H), 7.35-7.28 (M, 3H), 7.46 (M, 2H).
步骤B: 2-苯基-1-(2-苯基-[1,3]-二噻烷-2-基)-乙醇(66) Step B: 2-Phenyl-1-(2-phenyl-[1,3]-dithian-2-yl)-ethanol (66)
-30℃下向在THF中的2-苯基-1,3-二噻烷(65)(4.0g,20.4mmol)的溶液加入在THF(15.3mL,24.4mmol)中的1.6M正丁基锂的溶液。在-30℃下搅拌30分钟后,在-30℃下滴加在四氢呋喃中的苯基乙酰基醛(2.45g,20.4mmol)的溶液。将所得的反应混合物于0℃下再搅拌1h。用饱和NH4Cl溶液终止反应,并用乙酸乙酯萃取。用饱和NH4Cl溶液、盐水洗涤合并的有机萃取物,并用Na2SO4干燥。过滤和浓缩后,通过硅胶快速色谱法纯化粗产物(在己烷中的25%乙酸乙酯)得到2.63g(71%)的标题化合物(66)。1H NMR(CDCl3,400MHz):δ1.97(m,2H),2.23(dd,J=4.0,1.2Hz,1H),2.43(dd,J=13.6,10.2Hz,1H),2.77(M,4H),3.02(d,J=13.6Hz,1H),4.07(M,1H),7.44-7.13(M,8H),8.02(dd,J=8.4,1.4Hz,2H)。To a solution of 2-phenyl-1,3-dithiane (65) (4.0 g, 20.4 mmol) in THF was added 1.6 M n-butyl in THF (15.3 mL, 24.4 mmol) at -30°C Lithium solution. After stirring at -30°C for 30 minutes, a solution of phenylacetylaldehyde (2.45 g, 20.4 mmol) in tetrahydrofuran was added dropwise at -30°C. The resulting reaction mixture was stirred at 0 °C for an additional 1 h. The reaction was quenched with saturated NH4Cl solution and extracted with ethyl acetate. The combined organic extracts were washed with saturated NH4Cl solution, brine and dried over Na2SO4 . After filtration and concentration, the crude product was purified by silica gel flash chromatography (25% ethyl acetate in hexanes) to afford 2.63 g (71%) of the title compound (66). 1 H NMR (CDCl 3 , 400MHz): δ1.97(m, 2H), 2.23(dd, J=4.0, 1.2Hz, 1H), 2.43(dd, J=13.6, 10.2Hz, 1H), 2.77(M , 4H), 3.02 (d, J = 13.6Hz, 1H), 4.07 (M, 1H), 7.44-7.13 (M, 8H), 8.02 (dd, J = 8.4, 1.4Hz, 2H).
步骤C: 2-羟基-1,3-二苯基-丙-1-酮(67) Step C: 2-Hydroxy-1,3-diphenyl-propan-1-one (67)
向在100mL 9∶1的乙腈和水的混合物中的2-苯基-1-(2-苯基-[1,3]-二噻烷-2-基)-乙醇(66)(2.50g,7.9mmol)的溶液加入高氯酸汞水合物(4.1g,10.3mmol)。将所得的混合物于室温下搅拌5分钟,而薄层色谱处理显示反应完成。用乙酸乙酯稀释混合物,用C盐滤垫过滤,用饱和NaHCO3、盐水洗涤滤液,并用Na2SO4干燥。在减压下除去溶剂,并通过硅胶快速色谱法纯化粗产物(在己烷中的20%乙酸乙酯)得到1.32g(74%)的标题化合物(67)。1H NMR(CDCl3,400MHz):δ2.90(dd,J=14.4,7.0Hz,1H),3.20(dd,J=14.4,4.0Hz,1H),3.70(d,J=6.8Hz,1H),5.35(M,1H),7.28-7.11(M,5H),7.53(m,2H),7.65(M,1H),7.93(d,J=7.2HZ,2H)。To 2-phenyl-1-(2-phenyl-[1,3]-dithian-2-yl)-ethanol (66) in 100 mL of a 9:1 mixture of acetonitrile and water (2.50 g, 7.9 mmol) was added to a solution of mercury perchlorate hydrate (4.1 g, 10.3 mmol). The resulting mixture was stirred at room temperature for 5 minutes, and TLC showed the reaction to be complete. The mixture was diluted with ethyl acetate, filtered through a pad of celite, the filtrate was washed with saturated NaHCO 3 , brine, and dried over Na 2 SO 4 . The solvent was removed under reduced pressure and the crude product was purified by silica gel flash chromatography (20% ethyl acetate in hexanes) to afford 1.32 g (74%) of the title compound (67). 1 H NMR (CDCl 3 , 400MHz): δ2.90(dd, J=14.4, 7.0Hz, 1H), 3.20(dd, J=14.4, 4.0Hz, 1H), 3.70(d, J=6.8Hz, 1H ), 5.35 (M, 1H), 7.28-7.11 (M, 5H), 7.53 (m, 2H), 7.65 (M, 1H), 7.93 (d, J=7.2HZ, 2H).
步骤D: 1-{[(1-苯甲酰氧基-2-苯基乙氧基)羰基]氨基甲基}-1-环己 Step D: 1-{[(1-Benzoyloxy-2-phenylethoxy)carbonyl]aminomethyl}-1-cyclohexyl
烷乙酸(64) Alkaneacetic acid (64)
根据实施例3的方法,并用2-羟基-1,3-二苯基-丙-1-酮代替安息香,得到181mg的标题化合物(64)。1H NMR(CDCl3,400MHz):δ1.45-1.29(m,10H),2.24(d,J=13.6Hz,1H),2.28(d,J=13.6Hz,1H),3.22(M,4H),5.26(t,J=6.6Hz,1H),7.16(t,J=5.6Hz,1H),7.33-7.25(M,5H),7.40(M,2H),7.57(m,1H),8.02(M,2H)。According to the method of Example 3, substituting 2-hydroxy-1,3-diphenyl-propan-1-one for benzoin, 181 mg of the title compound (64) was obtained. 1 H NMR (CDCl 3 , 400MHz): δ1.45-1.29(m, 10H), 2.24(d, J=13.6Hz, 1H), 2.28(d, J=13.6Hz, 1H), 3.22(M, 4H ), 5.26(t, J=6.6Hz, 1H), 7.16(t, J=5.6Hz, 1H), 7.33-7.25(M, 5H), 7.40(M, 2H), 7.57(m, 1H), 8.02 (M, 2H).
实施例7 1-{[(1-(3-甲基丁酰氧基)-2-苯基乙氧基)羰基]氨基甲 Example 7 1-{[(1-(3-methylbutyryloxy)-2-phenylethoxy)carbonyl]aminomethyl
基}-1-环己烷乙酸(68) Base}-1-cyclohexaneacetic acid (68)
根据实施例6的方法,并用3-甲基丁醛代替步骤A中的苯甲醛,得到95mg的标题化合物(68)。1H NMR(CDCl3,400MHz):δ0.88-0.90(M,6H),1.16-1.29(m,10H),2.06(M,1H),2.16(M,2H),2.26(M,2H),3.08(d,J=6.8Hz,2H),3.19(M,2H),5.22(t,J=6.8Hz,1H),6.93(t,J=6Hz,1H),7.31-7.23(M,5H)。According to the method of Example 6, and substituting 3-methylbutyraldehyde for benzaldehyde in step A, 95 mg of the title compound (68) were obtained. 1 H NMR (CDCl 3 , 400MHz): δ0.88-0.90(M, 6H), 1.16-1.29(m, 10H), 2.06(M, 1H), 2.16(M, 2H), 2.26(M, 2H) , 3.08(d, J=6.8Hz, 2H), 3.19(M, 2H), 5.22(t, J=6.8Hz, 1H), 6.93(t, J=6Hz, 1H), 7.31-7.23(M, 5H ).
实施例8Example 8
1-{[(α-苯甲酰氧基丁氧基)羰基]氨基甲基}-1-环己烷乙酸(69)1-{[(α-Benzoyloxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (69)
根据实施例6的方法,并用丁醛代替步骤B中的苯基乙醛,得到240mg的标题化合物(69)。1H NMR(CDCl3,400MHz):δ0.99(t,J=7.6Hz,3H),1.52-1.38(M,12H),1.89(M,2H),2.31(s,2H),3.24(M,2H),5.34(t,J=6.6Hz,1H),6.70(t,J=5.6Hz,1H),7.42(M,2H),7.56(M,1H),8.04(M,2H)。According to the method of Example 6, butyraldehyde was used instead of phenylacetaldehyde in Step B, 240 mg of the title compound (69) were obtained. 1 H NMR (CDCl 3 , 400MHz): δ0.99(t, J=7.6Hz, 3H), 1.52-1.38(M, 12H), 1.89(M, 2H), 2.31(s, 2H), 3.24(M , 2H), 5.34(t, J=6.6Hz, 1H), 6.70(t, J=5.6Hz, 1H), 7.42(M, 2H), 7.56(M, 1H), 8.04(M, 2H).
实施例9Example 9
1-{[(α-乙酸基丁氧基)羰基]氨基甲基}-1-环己烷乙酸(70)1-{[(α-acetoxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (70)
根据实施例6的方法,并分别用乙醛代替步骤A中的苯甲醛和用丁醛代替步骤B中的苯基乙醛,得到42mg的标题化合物(70)。1HNMR(CD3OD,400MHz):δ0.95(M,3H),1.52-1.31(M,12H),1.72(M,2H),2.02(s,3H),2.27(s,2H),3.20(s,2H),6.67(t,J=5.6Hz,1H)。According to the method of Example 6, and replacing the benzaldehyde in step A with acetaldehyde and the phenylacetaldehyde in step B with butyraldehyde, 42 mg of the title compound (70) were obtained. 1 HNMR (CD 3 OD, 400MHz): δ0.95(M, 3H), 1.52-1.31(M, 12H), 1.72(M, 2H), 2.02(s, 3H), 2.27(s, 2H), 3.20 (s, 2H), 6.67 (t, J=5.6Hz, 1H).
实施例10Example 10
1-{[(α-丁酰氧基丁氧基)羰基]氨基甲基}-1-环己烷乙酸(71)1-{[(α-butyryloxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (71)
根据实施例3的方法,并用丁偶姻代替安息香,得到210mg的标题化合物(71)。1H NMR(CDCl3,400MHz);80.93(M,6H),1.37-1.76(M,16H),2.30(M,4H),3.23(M,2H),5.25(broad triplet,1H),6.73(M,1H).MS(ESI)m/z 356.45(M-H)+。According to the method of Example 3, but using butyroin instead of benzoin, 210 mg of the title compound (71) was obtained. 1 H NMR (CDCl 3 , 400MHz); 80.93 (M, 6H), 1.37-1.76 (M, 16H), 2.30 (M, 4H), 3.23 (M, 2H), 5.25 (broad triplet, 1H), 6.73 ( M, 1H). MS (ESI) m/z 356.45 (MH) + .
实施例11Example 11
1-{[(α-丙酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸(72)1-{[(α-propionyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (72)
步骤A: 1-碘乙基-对硝基苯基碳酸酯(73) Step A: 1-iodoethyl-p-nitrophenyl carbonate (73)
将在无水丙酮中的1-氯乙基-对硝基苯基碳酸酯(0.5g,2mmol)和NaI(0.6g,4mmol)的混合物于40℃下搅拌3h。过滤后,在减压下将滤液浓缩,得以480mg(72%)的标题化合物(73),将其不经进一步纯化用于下一步反应。A mixture of 1-chloroethyl-p-nitrophenyl carbonate (0.5 g, 2 mmol) and NaI (0.6 g, 4 mmol) in anhydrous acetone was stirred at 40 °C for 3 h. After filtration, the filtrate was concentrated under reduced pressure to obtain 480 mg (72%) of the title compound (73), which was used in the next reaction without further purification.
步骤B: α-丙酰氧基乙基-对硝基苯基碳酸酯(74) Step B: α-propionyloxyethyl-p-nitrophenyl carbonate (74)
将在甲苯(20mL)中的1-碘乙基-对硝基苯基碳酸酯(73)(0.51g,1.5mmol)和丙酸银(0.54g,3mmol)的混合物于50℃下搅拌24h。将反应混合物过滤除去固体,并在减压下浓缩滤液。将所得的残余物进行硅胶色谱处理(20%CH2Cl2/己烷,然后用40%CH2Cl2/己烷),得到0.39g(92%)的标题化合物(74)。1H NMR(CDCl3,400MHz):1.16(t,J=7.6Hz,3H),1.61(d,J=5.6Hz,3H),2.41(q,J=7.6Hz,2H),6.84(q,1H,J=5.6Hz),7.39(d,J=9.2Hz,2H),8.28(d,J=9.2Hz,2H)。A mixture of 1-iodoethyl-p-nitrophenyl carbonate (73) (0.51 g, 1.5 mmol) and silver propionate (0.54 g, 3 mmol) in toluene (20 mL) was stirred at 50 °C for 24 h. The reaction mixture was filtered to remove solids, and the filtrate was concentrated under reduced pressure. The resulting residue was chromatographed on silica gel ( 20 % CH2Cl2 /hexanes, then 40% CH2Cl2 /hexanes) to afford 0.39 g (92%) of the title compound (74). 1 H NMR (CDCl 3 , 400MHz): 1.16(t, J=7.6Hz, 3H), 1.61(d, J=5.6Hz, 3H), 2.41(q, J=7.6Hz, 2H), 6.84(q, 1H, J=5.6Hz), 7.39(d, J=9.2Hz, 2H), 8.28(d, J=9.2Hz, 2H).
步骤C: 1-{[(α-丙酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸 Step C: 1-{[(α-propionyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(72) (72)
向在二氯甲烷(30mL)中的加巴喷丁(160mg,2.76mmol)和三乙基胺(0.77mL,5.5mmol)的混合物加入三甲基氯硅烷(0.71mL,5.5mmol),并将所得的混合物搅拌直至形成澄清溶液。向以上溶液加入在二氯甲烷(10mL)中的α-丙酰氧基乙基-对硝基苯基碳酸酯(74)(0.39g,1.4mmol)的溶液。搅拌30分钟后用10%柠檬酸(20mL)洗涤反应混合物,并分离有机层。进一步用醚(3×10mL)萃取含水层,并用MgSO4干燥合并的有机萃取物。减压下除去溶剂后,用反相制备HPLC(乙腈,水,1%甲酸)将残余物纯化得到190mg(44%)的标题化合物(72)。1HNMR(CD3OD,400MHz):1.09(t,J=7.6Hz,3H),1.36-1.54(m,10H),1.44(d,J=5.6Hz,3H),2.28(s,2H),2.31(q,J=7.6Hz,2H),3.22(s,2H),6.67(q,J=5.6Hz,1H).MS(ESI)m/z 316.25(M+H)+。To a mixture of gabapentin (160 mg, 2.76 mmol) and triethylamine (0.77 mL, 5.5 mmol) in dichloromethane (30 mL) was added trimethylchlorosilane (0.71 mL, 5.5 mmol), and the resulting mixture Stir until a clear solution forms. To the above solution was added a solution of α-propionyloxyethyl-p-nitrophenyl carbonate (74) (0.39 g, 1.4 mmol) in dichloromethane (10 mL). After stirring for 30 minutes the reaction mixture was washed with 10% citric acid (20 mL), and the organic layer was separated. The aqueous layer was further extracted with ether (3 x 10 mL), and the combined organic extracts were dried over MgSO 4 . After removing the solvent under reduced pressure, the residue was purified by reverse phase preparative HPLC (acetonitrile, water, 1% formic acid) to afford 190 mg (44%) of the title compound (72). 1 HNMR (CD 3 OD, 400MHz): 1.09(t, J=7.6Hz, 3H), 1.36-1.54(m, 10H), 1.44(d, J=5.6Hz, 3H), 2.28(s, 2H), 2.31 (q, J = 7.6 Hz, 2H), 3.22 (s, 2H), 6.67 (q, J = 5.6 Hz, 1H). MS (ESI) m/z 316.25 (M+H) + .
实施例12Example 12
1-{[(α-丁酰氧基乙氧基)羰基]氨基甲基}环己烷乙酸(75)1-{[(α-butyryloxyethoxy)carbonyl]aminomethyl}cyclohexaneacetic acid (75)
步骤A: α-丁酰氧基乙基-对硝基苯基碳酸酯(76) Step A: α-Butyryloxyethyl-p-nitrophenyl carbonate (76)
将在甲苯(40mL)中的1-碘乙基-对硝基苯基碳酸酯(73)(1.5g,4.5mmol)和丁酸银(1.3g,6.7mmol)的混合物于90℃下在油浴中搅拌24h。在减压下将反应混合物过滤并将滤液浓缩。将所得的残余物进行硅胶色谱处理(20%CH2Cl2/己烷,然后用40%CH2Cl2/己烷),得到0.46g(36%)的标题化合物(76)。1H NMR(CDCl3,400MHz):0.95(t,J=7.6Hz,3H),1.61(d,J=5.6Hz,3H),1.67(m.2H),2.41(t,J=7.6Hz,2H),6.84(q,1H,J=5.6Hz),7.39(d,J=9.2Hz,2H),8.28(d,J=9.2Hz,2H).MS(ESI)m/z 298.28(M+H)+。A mixture of 1-iodoethyl-p-nitrophenyl carbonate (73) (1.5 g, 4.5 mmol) and silver butyrate (1.3 g, 6.7 mmol) in toluene (40 mL) was dissolved in oil at 90 °C Stir in the bath for 24h. The reaction mixture was filtered under reduced pressure and the filtrate was concentrated. The resulting residue was chromatographed on silica gel (20% CH2Cl2 /hexanes, then 40% CH2Cl2 /hexanes) to afford 0.46 g (36%) of the title compound ( 76 ). 1 H NMR (CDCl 3 , 400MHz): 0.95(t, J=7.6Hz, 3H), 1.61(d, J=5.6Hz, 3H), 1.67(m.2H), 2.41(t, J=7.6Hz, 2H), 6.84(q, 1H, J=5.6Hz), 7.39(d, J=9.2Hz, 2H), 8.28(d, J=9.2Hz, 2H). MS(ESI) m/z 298.28(M+ H) + .
步骤B: 1-{[(α-丁酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸 Step B: 1-{[(α-butyryloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(75) (75)
向含有在二氯甲烷(30mL)中的加巴喷丁(530mg,3.1mmol)和三乙基胺(0.89mL,6.4mmol)的混合物加入三甲基氯硅烷(0.83mL,6.4mmol),并将所得的混合物搅拌直至形成澄清溶液。往此溶液加入在二氯甲烷(10mL)中的α-丁酰氧基乙基-对硝基苯基碳酸酯(76)(0.46g,1.6mmol)的溶液,并将所得的混合物搅拌30分钟。用10%柠檬酸(20mL)洗涤反应混合物,并分离有机相。进一步用醚(3×10mL)萃取含水相,并用MgSO4干燥合并的有机相,然后真空浓缩。通过反相制备HPLC(乙腈,水,1%甲酸)将所得的残余物纯化得到70mg(21%)的标题化合物(75)。1H NMR(CD3OD,400MHz):0.95(t,J=7.6Hz,3H),1.32-1.58(m,10H),1.42(d,J=5.6Hz,3H),1.67(M,2H),2.24(s,2H),2.30(t,J=7.6Hz,2H),3.24(s,2H),6.74(q,J=5.6Hz,1H).MS(ESI)m/z 330.28(M+H)+。To a mixture containing gabapentin (530 mg, 3.1 mmol) and triethylamine (0.89 mL, 6.4 mmol) in dichloromethane (30 mL) was added trimethylchlorosilane (0.83 mL, 6.4 mmol), and the resulting The mixture was stirred until a clear solution formed. To this solution was added a solution of α-butyryloxyethyl-p-nitrophenyl carbonate (76) (0.46 g, 1.6 mmol) in dichloromethane (10 mL), and the resulting mixture was stirred for 30 minutes . The reaction mixture was washed with 10% citric acid (20 mL), and the organic phase was separated. The aqueous phase was further extracted with ether (3 x 10 mL), and the combined organic phases were dried over MgSO 4 and concentrated in vacuo. The resulting residue was purified by reverse phase preparative HPLC (acetonitrile, water, 1% formic acid) to afford 70 mg (21%) of the title compound (75). 1 H NMR (CD 3 OD, 400MHz): 0.95(t, J=7.6Hz, 3H), 1.32-1.58(m, 10H), 1.42(d, J=5.6Hz, 3H), 1.67(M, 2H) , 2.24(s, 2H), 2.30(t, J=7.6Hz, 2H), 3.24(s, 2H), 6.74(q, J=5.6Hz, 1H).MS(ESI) m/z 330.28(M+ H) + .
通过以下方法将酸形式定量转化为对应的钠盐:溶于水(5mL),加入等摩尔量的0.5N NaHCO3,然后冻干。The acid form was quantitatively converted to the corresponding sodium salt by dissolving in water (5 mL), adding an equimolar amount of 0.5N NaHCO3 , and then lyophilizing.
实施例13Example 13
1-{[(α-异丁酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸(77)1-{[(α-isobutyryloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (77)
根据实施例2的方法,并用异丁酸银代替丁酸银,得到70mg(21%)的标题化合物(77)。1H NMR(CD3OD,400MHz):1.12(d,J=7.2Hz,3H),1.14(d,J=7.2Hz,3H),1.32-1.58(m,10H),1.44(d,J=5.6Hz,3H),2.28(s,2H),2.56(M,1H),3.25(m,2H),6.73(q,J=5.6Hz,1H).MS(ESI)m/z 330.30(M+H)+。According to the method of Example 2, and using silver isobutyrate instead of silver butyrate, 70 mg (21%) of the title compound (77) were obtained. 1 H NMR (CD 3 OD, 400MHz): 1.12(d, J=7.2Hz, 3H), 1.14(d, J=7.2Hz, 3H), 1.32-1.58(m, 10H), 1.44(d, J= 5.6Hz, 3H), 2.28(s, 2H), 2.56(M, 1H), 3.25(m, 2H), 6.73(q, J=5.6Hz, 1H).MS(ESI) m/z 330.30(M+ H) + .
通过以下方法将酸形式定量转化为对应的钠盐:溶于水(5mL),加入等摩尔量的0.5N NaHCO3,然后冻干。The acid form was quantitatively converted to the corresponding sodium salt by dissolving in water (5 mL), adding an equimolar amount of 0.5N NaHCO3 , and then lyophilizing.
实施例14Example 14
1-{[(α-新戊酰氧基乙氧基)羰基]氨基甲基}-1-环己烷乙酸(78)1-{[(α-pivaloyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (78)
根据实施例12的方法,并用三甲基乙酸银代替丁酸盐,得到80mg(36%)的标题化合物(78)。1H NMR(CDCl3,400MHz):1.13(s,9H),1.32-1.58(m,10H),1.41(d,J=5.6Hz,3H),2.27(s,2H),3.25(M,2H),5.41(t,1H),6.73(q,J=5.6Hz,1H).MS(ESI)m/z344.20(M+H)+。Following the procedure of Example 12, and substituting trimethylsilver acetate for butyrate, 80 mg (36%) of the title compound (78) were obtained. 1 H NMR (CDCl 3 , 400MHz): 1.13(s, 9H), 1.32-1.58(m, 10H), 1.41(d, J=5.6Hz, 3H), 2.27(s, 2H), 3.25(M, 2H ), 5.41 (t, 1H), 6.73 (q, J = 5.6 Hz, 1H). MS (ESI) m/z 344.20 (M+H) + .
通过以下方法将酸形式定量转化为对应的钠盐:溶于水(5mL),加入等摩尔量的0.5N NaHCO3,然后冻干。The acid form was quantitatively converted to the corresponding sodium salt by dissolving in water (5 mL), adding an equimolar amount of 0.5N NaHCO3 , and then lyophilizing.
实施例15Example 15
1-{(α-乙酸基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸(79)1-{(α-Acetoxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (79)
根据实施例2的方法,并用氯甲酸1-氯-2-甲基丙基酯代替氯甲酸1-氯乙酯,得到212mg(38%)的标题化合物(79)。1H NMR(CD3OD,400MHz):0.99(M,6H),1.32-1.58(m,10H),1.88(M,1H),2.08(s,3H),2.38(s,2H),3.25(s,2H),6.52(d,J=4.4Hz,1H);MS(ESI)mlz330.30(M+H)+。Following the procedure of Example 2, substituting 1-chloro-2-methylpropyl chloroformate for 1-chloroethyl chloroformate, 212 mg (38%) of the title compound (79) were obtained. 1 H NMR (CD 3 OD, 400MHz): 0.99 (M, 6H), 1.32-1.58 (m, 10H), 1.88 (M, 1H), 2.08 (s, 3H), 2.38 (s, 2H), 3.25 ( s, 2H), 6.52 (d, J=4.4Hz, 1H); MS (ESI) mlz 330.30 (M+H) + .
实施例16Example 16
1-{[(α-丙酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸(80)1-{[(α-propionyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (80)
根据实施例11的方法,并用1-氯-2-甲基丙基-对硝基苯基碳酸酯代替1-氯乙基-对硝基苯基碳酸酯,得到190mg(44%)的标题化合物(80)。1H NMR(CD3OD,400MHz):0.90(d,J=6.6Hz,3H),0.91(d,J=6.6Hz,3H),0.98(T,J=7.6Hz,3H),1.32-1.58(m,10H),1.83(m,1H),2.18(s,2H),2.28(q,J=7.6Hz,2H),3.25(s,2H),6.52(d,J=4.4Hz,1H).MS(ESI)mlz 344.34(M+H)+。According to the method of Example 11, and substituting 1-chloro-2-methylpropyl-p-nitrophenyl carbonate for 1-chloroethyl-p-nitrophenyl carbonate, 190 mg (44%) of the title compound were obtained (80). 1 H NMR (CD 3 OD, 400MHz): 0.90 (d, J=6.6Hz, 3H), 0.91 (d, J=6.6Hz, 3H), 0.98 (T, J=7.6Hz, 3H), 1.32-1.58 (m, 10H), 1.83(m, 1H), 2.18(s, 2H), 2.28(q, J=7.6Hz, 2H), 3.25(s, 2H), 6.52(d, J=4.4Hz, 1H) .MS (ESI) mlz 344.34 (M+H) + .
实施例17Example 17
1-{[(α-丁酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸(81)1-{[(α-butyryloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (81)
根据实施例2的方法,并分别用氯甲酸1-氯-2-甲基丙基酯和丁酸汞代替氯甲酸1-氯乙基酯和乙酸汞,得到95mg(36%)的标题化合物(81)。1H NMR(CD30D,400MHz):1.12(t,J=7.6Hz,3H),1.13(d,J=6.6Hz,3H),1.14(d,J=6.6Hz,3H),1.32-1.58(m,10H),1.87(m,2H),2.22(m,1H),2.42(s,2H),2.46(t,J=7.6Hz,2H),3.44(m,2H),6.78(d,J=4.8Hz,1H).MS(ESI)m/z 358.30(M+H)+。According to the method of Example 2, and replace 1-chloroethyl chloroformate and mercury acetate with 1-chloro-2-methylpropyl chloroformate and mercury butyrate respectively, obtain 95 mg (36%) of the title compound ( 81). 1 H NMR (CD 3 OD, 400MHz): 1.12 (t, J=7.6Hz, 3H), 1.13 (d, J=6.6Hz, 3H), 1.14 (d, J=6.6Hz, 3H), 1.32-1.58 (m, 10H), 1.87(m, 2H), 2.22(m, 1H), 2.42(s, 2H), 2.46(t, J=7.6Hz, 2H), 3.44(m, 2H), 6.78(d, J = 4.8 Hz, 1H). MS (ESI) m/z 358.30 (M+H) + .
实施例18Example 18
1-{[(α-异丁酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸(82)1-{[(α-isobutyryloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (82)
根据实施例2的方法,并分别用氯甲酸1-氯-2-甲基丙基酯和异丁酸汞代替氯甲酸1-氯乙酯和乙酸汞,得到95mg(36%)的标题化合物(82)。1H NMR(CD3OD,400MHz):0.95(d,J=7.2Hz,3H),0.97(d,J=7.2Hz,3H),1.05(d,J=6.6Hz,3H),1.06(d,J=6.6Hz,3H),1.32-1.58(m,10H),1.98(m,1H),2.24(s,2H),2.45(m,1H),3.24(m,2H),6.42(d,J=4.8Hz,1H).MS(ESI)m/z 358.27(M+H)+。According to the method of Example 2, and replace 1-chloroethyl chloroformate and mercury acetate with 1-chloro-2-methylpropyl chloroformate and mercury isobutyrate respectively, obtain 95 mg (36%) of the title compound ( 82). 1 H NMR (CD 3 OD, 400MHz): 0.95(d, J=7.2Hz, 3H), 0.97(d, J=7.2Hz, 3H), 1.05(d, J=6.6Hz, 3H), 1.06(d , J=6.6Hz, 3H), 1.32-1.58(m, 10H), 1.98(m, 1H), 2.24(s, 2H), 2.45(m, 1H), 3.24(m, 2H), 6.42(d, J = 4.8 Hz, 1H). MS (ESI) m/z 358.27 (M+H) + .
实施例19Example 19
1-{[(α-新戊酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸(83)1-{[(α-pivaloyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (83)
根据实施例12的方法,并分别用1-氯-2-甲基丙基-对硝基苯基碳酸酯和三甲基乙酸银代替1-氯乙基-对硝基苯基碳酸酯和丁酸银,得到10mg(9%)的标题化合物(83)。1H NMR(CD3OD,400MHz):0.98(d,J=6.6Hz,3H),0.99(d,J=6.6Hz,3H),1.19(s,9H),1.32-1.58(m,10H),2.08(m,1H),2.28(s,2H),3.21(m,2H),6.49(d,1H);MS(ESI)m/z 372.31(M+H)+。According to the method of embodiment 12, and replace 1-chloroethyl-p-nitrophenyl carbonate and butyl with 1-chloro-2-methylpropyl-p-nitrophenyl carbonate and trimethyl silver acetate respectively silver acid to afford 10 mg (9%) of the title compound (83). 1 H NMR (CD 3 OD, 400MHz): 0.98(d, J=6.6Hz, 3H), 0.99(d, J=6.6Hz, 3H), 1.19(s, 9H), 1.32-1.58(m, 10H) , 2.08 (m, 1H), 2.28 (s, 2H), 3.21 (m, 2H), 6.49 (d, 1H); MS (ESI) m/z 372.31 (M+H) + .
实施例20Example 20
1-{[(α-苯甲酰氧基异丁氧基)羰基]氨基甲基-1-环己烷乙酸(84)1-{[(α-Benzoyloxyisobutoxy)carbonyl]aminomethyl-1-cyclohexaneacetic acid (84)
根据实施例11的方法,并分别用1-氯-2-甲基丙基-对硝基苯基碳酸酯和苯甲酸银代替1-对硝基苯基碳酸1-氯乙基酯和丙酸银,得到109mg(40%)的标题化合物(84)。1H NMR(CD3OD,400MHz):1.18(d,J=7.2Hz,6H),1.32-1.58(m,10H),2.42(M,1H),2.28(s,2H),3.45(s,2H),6.99(d,J=4.8Hz,1H),7.76(M,2H),7.92(M,1H),8.26(M,2H)。MS(ESI)m/z 392.22(M+H)+。According to the method of Example 11, and replace 1-chloroethyl 1-p-nitrophenylcarbonate and propionic acid with 1-chloro-2-methylpropyl-p-nitrophenyl carbonate and silver benzoate respectively Silver, yielded 109 mg (40%) of the title compound (84). 1 H NMR (CD 3 OD, 400MHz): 1.18(d, J=7.2Hz, 6H), 1.32-1.58(m, 10H), 2.42(M, 1H), 2.28(s, 2H), 3.45(s, 2H), 6.99 (d, J = 4.8 Hz, 1H), 7.76 (M, 2H), 7.92 (M, 1H), 8.26 (M, 2H). MS (ESI) m/z 392.22 (M+H) + .
实施例21Example 21
1-{[(α-乙酸基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸(85)1-{[(α-Acetoxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (85)
步骤A: 异丙烯基-对硝基苯基碳酸酯(86) Step A: Isopropenyl-p-nitrophenyl carbonate (86)
0℃下向在二氯甲烷(200mL)中的对硝基苯酚(5.76g,41.5mmol)和氯甲酸异丙烯基酯(5g,41.5mmol)的混合物加入在二氯甲烷(50mL)中的吡啶(3.4mL,42mmol)的溶液。将所得的混合物于0℃下搅拌30分钟,然后在室温下搅拌1h。减压下除去溶剂后,将残余物溶于醚,并用水、10%柠檬酸和再次用水洗涤。用Na2SO4干燥醚层,并在减压下蒸发得到8.7g(94%)的标题化合物(86),这一种米色固体。1H NMR(CDCl3,400MHz):2.05(s,3H),4.81(M,1H),4.95(d,J=2Hz,1H),7.42(d,J=9.2Hz,2H),8.28(d,J=9.2Hz,2H)。To a mixture of p-nitrophenol (5.76 g, 41.5 mmol) and isopropenyl chloroformate (5 g, 41.5 mmol) in dichloromethane (200 mL) was added pyridine in dichloromethane (50 mL) at 0 °C (3.4 mL, 42 mmol). The resulting mixture was stirred at 0 °C for 30 min, then at room temperature for 1 h. After removing the solvent under reduced pressure, the residue was dissolved in ether and washed with water, 10% citric acid and again with water. The ether layer was dried over Na2SO4 and evaporated under reduced pressure to afford 8.7 g (94%) of the title compound (86) as a beige solid. 1 H NMR (CDCl 3 , 400MHz): 2.05(s, 3H), 4.81(M, 1H), 4.95(d, J=2Hz, 1H), 7.42(d, J=9.2Hz, 2H), 8.28(d , J=9.2Hz, 2H).
步骤B: 2-氯异丙基-对硝基苯基碳酸酯(87) Step B: 2-Chloroisopropyl-p-nitrophenyl carbonate (87)
将异丙烯基-对硝基苯基碳酸酯(86)(8.7g,39mmol)溶于在密封容器中的4M氯化氢/二噁烷。将混合物于室温下搅拌16h。在减压下除去溶剂得到10g(100%)的标题化合物(87),将其不经进一步纯化用于下一步反应。1H NMR(CDCl3,400MHz):2.10(s,6H),7.42(d,2H,J=9.2Hz),8.28(d,J=9.2Hz,2H)。Isopropenyl-p-nitrophenyl carbonate (86) (8.7 g, 39 mmol) was dissolved in 4M hydrogen chloride/dioxane in a sealed container. The mixture was stirred at room temperature for 16 h. The solvent was removed under reduced pressure to afford 10 g (100%) of the title compound (87), which was used in the next reaction without further purification. 1 H NMR (CDCl 3 , 400 MHz): 2.10 (s, 6H), 7.42 (d, 2H, J = 9.2 Hz), 8.28 (d, J = 9.2 Hz, 2H).
步骤C: α-乙酸基异丙基-对硝基苯基碳酸酯(88) Step C: α-Acetoxyisopropyl-p-nitrophenyl carbonate (88)
将在二氯甲烷(20mL)中的2-氯异丙基-对硝基苯基碳酸酯(87)(0.5g,1.93mmol)和乙酸汞(1.0g,3.13mmol)的混合物于室温下搅拌24h。将反应混合物过滤除去固体,并在减压下浓缩滤液。用硅胶色谱处理所得的残余物(20%CH2Cl2/己烷,然后用40%CH2Cl2/己烷),得到227mg(50%)的标题化合物(88)。1H NMR(CDCl3,400MHz):1.90(s,6H),2.07(s,3H),7.28(d,2H,J=9.2Hz),8.28(d,J=9.2Hz,2H)。A mixture of 2-chloroisopropyl-p-nitrophenyl carbonate (87) (0.5 g, 1.93 mmol) and mercury acetate (1.0 g, 3.13 mmol) in dichloromethane (20 mL) was stirred at room temperature 24h. The reaction mixture was filtered to remove solids, and the filtrate was concentrated under reduced pressure. The resulting residue was chromatographed on silica gel ( 20 % CH2Cl2 /hexanes, then 40% CH2Cl2 /hexanes) to afford 227 mg (50%) of the title compound (88). 1 H NMR (CDCl 3 , 400 MHz): 1.90 (s, 6H), 2.07 (s, 3H), 7.28 (d, 2H, J=9.2Hz), 8.28 (d, J=9.2Hz, 2H).
步骤D: 1-{(α-乙酸基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸(85) Step D: 1-{(α-Acetoxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (85)
向包含在二氯甲烷(30mL)中的加巴喷丁(257mg,1.5mmol)和三乙基胺(0.46mL,3.3mmol)的混合物加入三甲基氯硅烷(0.38mL,3mmol),并将搅拌混合物至澄清。加入含有在二氯甲烷(10mL)中的α-乙酸基异丙基-对硝基苯基碳酸酯(88)(0.23g,0.8mmol)的溶液,并搅拌30分钟。用盐水(10mL)洗涤反应混合物,并分离有机层。进一步用醚(3×10mL)萃取含水层,并用MgSO4干燥合并的有机萃取物,真空浓缩。用硅胶对所得的残余物色谱处理(己烷∶乙酸乙酯(4∶1))得到40mg(16%)的标题化合物(85)。1H NMR(CD3OD,400MHz):1.32-1.58(m,10H),1.80(s,6H),2.02(s,3H),2.27(s,2H),3.30(s,2H).MS(ESI)m/z 316.21(M+H)+。To a mixture containing gabapentin (257 mg, 1.5 mmol) and triethylamine (0.46 mL, 3.3 mmol) in dichloromethane (30 mL) was added trimethylchlorosilane (0.38 mL, 3 mmol), and the mixture was stirred to clarify. Add a solution containing α-acetoxyisopropyl-p-nitrophenyl carbonate (88) (0.23 g, 0.8 mmol) in dichloromethane (10 mL) and stir for 30 minutes. The reaction mixture was washed with brine (10 mL), and the organic layer was separated. The aqueous layer was further extracted with ether (3 x 10 mL), and the combined organic extracts were dried over MgSO 4 and concentrated in vacuo. The resulting residue was chromatographed on silica gel (hexane:ethyl acetate (4:1)) to afford 40 mg (16%) of the title compound (85). 1 H NMR (CD 3 OD, 400MHz): 1.32-1.58(m, 10H), 1.80(s, 6H), 2.02(s, 3H), 2.27(s, 2H), 3.30(s, 2H).MS( ESI) m/z 316.21 (M+H) + .
实施例22Example 22
1-{[(α-丁酰氧基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸(89)1-{[(α-butyryloxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (89)
根据实施例21的方法,并用丁酸汞代替乙酸汞,得到5mg(5%)的标题化合物(89)。1H NMR(CD3OD,400MHz):0.99(t,J=7.6Hz,3H),1.32-1.58(m,10H),1.60(M,2H),1.85(s,6H),2.22(t,J=7.6,2H),2.27(s,2H),3.20(s,2H).MS(ESI)m/z 344.24(M+H)+,366.30(M+Na)+。Following the procedure of Example 21, and substituting mercury butyrate for mercury acetate, 5 mg (5%) of the title compound (89) was obtained. 1 H NMR (CD 3 OD, 400MHz): 0.99(t, J=7.6Hz, 3H), 1.32-1.58(m, 10H), 1.60(M, 2H), 1.85(s, 6H), 2.22(t, J=7.6, 2H), 2.27 (s, 2H), 3.20 (s, 2H). MS (ESI) m/z 344.24 (M+H) + , 366.30 (M+Na) + .
实施例23Example 23
1-{[(α-异丁酰氧基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸(90)1-{[(α-isobutyryloxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (90)
根据实施例21的方法,并用异丁酸汞代替乙酸汞,得到109mg(43%)的标题化合物(90)。1H NMR(CD3OD,400MHz):1.19(d.J=7.2Hz,6H),1.32-1.58(m,10H),1.82(s,6H),2.38(s,2H),3.25(s,2H).MS(ESI)344.22(M+H)+,366.24(M+Na)+。According to the method of Example 21, and using mercury isobutyrate instead of mercury acetate, 109 mg (43%) of the title compound (90) were obtained. 1 H NMR (CD 3 OD, 400MHz): 1.19(dJ=7.2Hz, 6H), 1.32-1.58(m, 10H), 1.82(s, 6H), 2.38(s, 2H), 3.25(s, 2H) .MS (ESI) 344.22 (M+H) + , 366.24 (M+Na) + .
实施例24Example 24
1-{[(α-苯甲酰氧基异丙氧基)羰基]氨基甲基}-1-环己烷乙酸(91)1-{[(α-Benzoyloxyisopropoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (91)
根据实施例21的方法,并用苯甲酸汞代替乙酸汞,得到170mg(58%)的标题化合物(91)。1H NMR(CDCl3,400MHz):1.32-1.58(m,10H),1.95(s,6H),2.30(s,2H),3.20(d,J=6.8,2H),5.41(t,J=6.8Hz,1H),7.40(M,2H),7.52(m,1H),7.98(m,2H).MS(ESI)m/z400.29(M+Na)+。According to the method of Example 21, and using mercury benzoate instead of mercury acetate, 170 mg (58%) of the title compound (91) were obtained. 1 H NMR (CDCl 3 , 400MHz): 1.32-1.58(m, 10H), 1.95(s, 6H), 2.30(s, 2H), 3.20(d, J=6.8, 2H), 5.41(t, J= 6.8 Hz, 1H), 7.40 (M, 2H), 7.52 (m, 1H), 7.98 (m, 2H). MS (ESI) m/z 400.29 (M+Na) + .
实施例25Example 25
1-{[(α-烟酰氧基异丁氧基)羰基]氧基甲基}-1-环己烷乙酸(92)1-{[(α-nicotinoyloxyisobutoxy)carbonyl]oxymethyl}-1-cyclohexaneacetic acid (92)
步骤A: 1-{[(α-氯异丁氧基)羰基氨基甲基-1-环己烷乙酸(93) Step A: 1-{[(α-Chloroisobutoxy)carbonylaminomethyl-1-cyclohexaneacetic acid (93)
向含有在二氯甲烷(150mL)中的加巴喷丁(1.71g,10mmol)和三乙基胺(3.06mL,22mmol)的混合物加入三甲基氯硅烷(1.4mL,11mmol),并将所得的混合物搅拌至澄清(大约20分钟)。然后在0℃下加入含有在二氯甲烷(10mL)中的1-氯-2-甲基丙基氯甲酸酯(1.27mL,11mmol)的溶液,并在室温下搅拌60分钟。用10%柠檬酸(30mL)洗涤反应混合物,并分离有机层。进一步用醚(3×20mL)萃取含水层,并用MgSO4干燥合并的有机相,然后真空浓缩。用硅胶色谱处理残余物,用己烷∶乙酸乙酯(1∶4)洗脱得到2.37g(77%)的标题化合物。1HNMR(CDCl3,400MHz):δ1.04(d,J=6.4Hz,3H),1.06(d,J=6.4Hz,3H),1.36-1.53(m,10H),2.15(M,1H),2.34(s,2H),3.24(M,2H),5.39(t,1H),6.32(d,J=5.6Hz),1H)。MS(ESI)m/z306.34(M+H+)。To a mixture containing gabapentin (1.71 g, 10 mmol) and triethylamine (3.06 mL, 22 mmol) in dichloromethane (150 mL) was added trimethylchlorosilane (1.4 mL, 11 mmol), and the resulting mixture was stirred until clear (approximately 20 minutes). Then a solution containing 1-chloro-2-methylpropyl chloroformate (1.27 mL, 11 mmol) in dichloromethane (10 mL) was added at 0°C and stirred at room temperature for 60 minutes. The reaction mixture was washed with 10% citric acid (30 mL), and the organic layer was separated. The aqueous layer was further extracted with ether (3 x 20 mL), and the combined organic phases were dried over MgSO 4 and concentrated in vacuo. Chromatography of the residue on silica gel eluting with hexane:ethyl acetate (1:4) afforded 2.37 g (77%) of the title compound. 1 HNMR (CDCl 3 , 400MHz): δ1.04(d, J=6.4Hz, 3H), 1.06(d, J=6.4Hz, 3H), 1.36-1.53(m, 10H), 2.15(M, 1H) , 2.34 (s, 2H), 3.24 (M, 2H), 5.39 (t, 1H), 6.32 (d, J=5.6Hz), 1H). MS (ESI) m/z 306.34 (M+H + ).
步骤B: 1-{[(α-烟酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸 Step B: 1-{[(α-nicotinoyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(92) (92)
室温下将在丙酮中的(93)(268mg,0.88mmol)、1,8-二氮杂双环[5.4.0]十一碳-7-烯(DBU)(158μL,1.01mmol)和烟酸(637mg,5.2mmol)的混合物搅拌48h。过滤后,将滤液真空浓缩,并通过反相制备HPLC法纯化所得的残余物,得到50mg(14%)的标题化合物。1HNMR(CD3OD,400MHz):δ1.07(d,J=6.8Hz,3H),1.09(d,J=6.8Hz,3H),1.32-1.58(m,10H),2.19(M,1H),2.26(s,2H),3.23(M,2H),6.78(d,J=4.8Hz,1H),7.58(m,1H),8.39(d,J=6.4Hz,1H),8.76(d,J=4.4Hz,1H),9.10(s,1H).MS(ESI)m/z393.42(M+H+)。(93) (268 mg, 0.88 mmol), 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) (158 μL, 1.01 mmol) and nicotinic acid ( 637mg, 5.2mmol) the mixture was stirred for 48h. After filtration, the filtrate was concentrated in vacuo and the resulting residue was purified by reverse phase preparative HPLC to afford 50 mg (14%) of the title compound. 1 HNMR (CD 3 OD, 400MHz): δ1.07(d, J=6.8Hz, 3H), 1.09(d, J=6.8Hz, 3H), 1.32-1.58(m, 10H), 2.19(M, 1H ), 2.26(s, 2H), 3.23(M, 2H), 6.78(d, J=4.8Hz, 1H), 7.58(m, 1H), 8.39(d, J=6.4Hz, 1H), 8.76(d , J = 4.4 Hz, 1H), 9.10 (s, 1H). MS (ESI) m/z 393.42 (M+H + ).
实施例26Example 26
1-{[(α-2,2-二乙氧基丙酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷1-{[(α-2,2-diethoxypropionyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexane
乙酸(94) Acetic acid (94)
步骤A: 1-{[(α-氯异丁氧基)羰基]氨基甲基}-1-环己烷乙酸苄基酯 Step A: Benzyl 1-{[(α-chloroisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetate
(95) (95)
向在二氯甲烷中的(93)(1.02g,3.34mmol)的溶液加入1,3-二环己基碳化二亚胺(758mg,3.67mmol)。室温下搅拌30分钟后,加入苄醇(380μL,3.67mmol)和4-(二甲基氨基)吡啶(催化量)。将所得的混合物于室温下搅拌16h。过滤后,用10%柠檬酸洗涤滤液,用Na2SO4干燥并浓缩。用硅胶色谱处理残余物,用10%乙酸乙酯/己烷洗脱得到820mg(62%)的标题化合物。1H NMR(CDCl3,400MHz):δ1.03(d,J=6.4Hz,3H),1.05(d,J=6.4Hz,3H),1.36-1.53(m,10H),2.13(M,1H),2.35(s,2H),3.22(M,2H),5.11(s,2H),5.49(t,1H),6.32(d,J=4.8Hz),1H),7.34(m,5H).MS(ESI)m/z396.24(M+H+)。To a solution of (93) (1.02 g, 3.34 mmol) in dichloromethane was added 1,3-dicyclohexylcarbodiimide (758 mg, 3.67 mmol). After stirring at room temperature for 30 minutes, benzyl alcohol (380 μL, 3.67 mmol) and 4-(dimethylamino)pyridine (catalytic amount) were added. The resulting mixture was stirred at room temperature for 16 h. After filtration, the filtrate was washed with 10% citric acid, dried over Na2SO4 and concentrated. Chromatography of the residue on silica gel eluting with 10% ethyl acetate/hexanes afforded 820 mg (62%) of the title compound. 1 H NMR (CDCl 3 , 400MHz): δ1.03(d, J=6.4Hz, 3H), 1.05(d, J=6.4Hz, 3H), 1.36-1.53(m, 10H), 2.13(M, 1H ), 2.35(s, 2H), 3.22(M, 2H), 5.11(s, 2H), 5.49(t, 1H), 6.32(d, J=4.8Hz), 1H), 7.34(m, 5H). MS (ESI) m/z 396.24 (M+H + ).
步骤B: 2,2-二乙氧基丙酸铯(96) Step B: Cesium 2,2-diethoxypropionate (96)
10℃下向搅拌的14mL(0.2mol)丙酮酸和80ml原甲酸三乙酯的溶液中加入1mL浓硫酸。将所得的混合物于5-10℃下搅拌1h,然后用200mL二氯甲烷稀释。连续地用水(3×80mL)和饱和氯化钠溶液(80mL)洗涤有机溶液,然后用无水硫酸钠干燥。将混合物过滤,浓缩得到定量产率的2,2-二乙氧基丙酸,为一种油。1H NMR(CDCl3,400MHz):δ1.30(t,6H),1.61(s,3H),3.57(q,4H),8.62(s,1H)。通过以下方法将酸形式定量转化成它的铯盐:将酸溶于水(25mL),然后用等摩尔量的碳酸铯处理,随后冻干。1H NMR(D2O,400MHz):δ0.98(t,6H),1.28(s,3H),3.22(q,2H),3.47(q,2H)。1 mL of concentrated sulfuric acid was added to a stirred solution of 14 mL (0.2 mol) of pyruvic acid and 80 mL of triethyl orthoformate at 10°C. The resulting mixture was stirred at 5-10 °C for 1 h, then diluted with 200 mL of dichloromethane. The organic solution was washed successively with water (3 x 80 mL) and saturated sodium chloride solution (80 mL), then dried over anhydrous sodium sulfate. The mixture was filtered and concentrated to give 2,2-diethoxypropanoic acid in quantitative yield as an oil. 1 H NMR (CDCl 3 , 400 MHz): δ 1.30 (t, 6H), 1.61 (s, 3H), 3.57 (q, 4H), 8.62 (s, 1H). The acid form was quantitatively converted to its cesium salt by dissolving the acid in water (25 mL) and treating with an equimolar amount of cesium carbonate followed by lyophilization. 1 H NMR (D 2 O, 400 MHz): δ 0.98 (t, 6H), 1.28 (s, 3H), 3.22 (q, 2H), 3.47 (q, 2H).
步骤C: 1-{[(α-2,2-二乙氧基丙酰氧基异丁氧基)羰基]氨基甲 Step C: 1-{[(α-2,2-diethoxypropionyloxyisobutoxy)carbonyl]aminomethyl
基}-1-环己烷乙酸苄基酯(97) Benzyl}-1-cyclohexaneacetate (97)
室温下将在丙酮中的(95)(200mg,0.51mmol)和碘化钠(114mg,0.76mmol)的混合物搅拌1h。加入2,2-二乙氧基丙酸铯(96)(300mg,1.02mmol)和DMF(20mL),并将所得的混合物于40℃下搅拌18h。过滤后,将滤液浓缩并用硅胶快速柱色谱法纯化所得的残余物,用10%乙酸乙酯/己烷洗脱得到100mg(37%)的标题化合物。MS(ESI)m/z522.34(M+H+)。A mixture of (95) (200 mg, 0.51 mmol) and sodium iodide (114 mg, 0.76 mmol) in acetone was stirred at room temperature for 1 h. Cesium (96) 2,2-diethoxypropionate (300 mg, 1.02 mmol) and DMF (20 mL) were added, and the resulting mixture was stirred at 40° C. for 18 h. After filtration, the filtrate was concentrated and the resulting residue was purified by flash column chromatography on silica gel eluting with 10% ethyl acetate/hexanes to afford 100 mg (37%) of the title compound. MS (ESI) m/z 522.34 (M+H + ).
步骤D: 1-{[(α-2,2二乙氧基丙酰氧基异丁氧基)羰基]氨基甲 Step D: 1-{[(α-2,2diethoxypropionyloxyisobutoxy)carbonyl]aminomethyl
基}-1-环己烷乙酸(94) Base}-1-cyclohexaneacetic acid (94)
氢气氛和室温下将(97)(200mg,0.38mmol)和5%Pd-C(催化量)的混合物搅拌16h。过滤后,将滤液浓缩,并通过反相制备HPLC法纯化所得的残余物得到98mg(60%)的标题化合物。1H NMR(CDCl3,400MHz):δ0.97(D,J=6.8Hz,6H),1.19(t,J=6.4Hz,3H),1.21(t,J=6.4Hz,3H),1.32-1.58(m,10H,),1.51(s,3H),2.06(M,1H),2.30(s,2H),3.23(M,2H),3.46(M,2H),3.56(M,2H),5.30(t,1H,NH),6.59(d,J=4.8Hz,1H).MS(ESI)m/z 432.24(M+H+)。A mixture of (97) (200 mg, 0.38 mmol) and 5% Pd—C (catalytic amount) was stirred at room temperature under hydrogen atmosphere for 16 h. After filtration, the filtrate was concentrated and the resulting residue was purified by reverse phase preparative HPLC to afford 98 mg (60%) of the title compound. 1 H NMR (CDCl 3 , 400MHz): δ0.97(D, J=6.8Hz, 6H), 1.19(t, J=6.4Hz, 3H), 1.21(t, J=6.4Hz, 3H), 1.32- 1.58(m, 10H), 1.51(s, 3H), 2.06(M, 1H), 2.30(s, 2H), 3.23(M, 2H), 3.46(M, 2H), 3.56(M, 2H), 5.30 (t, 1H, NH), 6.59 (d, J = 4.8 Hz, 1H). MS (ESI) m/z 432.24 (M+H + ).
实施例27Example 27
1-{[(α-(2-氨基-2-甲基丙酰基)氧异丁氧基)羰基]氨基甲基}-1-环1-{[(α-(2-amino-2-methylpropionyl)oxyisobutoxy)carbonyl]aminomethyl}-1-ring
己烷乙酸(98) Hexaneacetic acid (98)
根据实施例26的方法,并用2-氨基-2-甲基丙酸代替2,2-二乙氧基丙酸,得到标题化合物。1H NMR(CDCl3,400MHz):δ0.97(d,J=6.8Hz,6H),1.44(s,3H),1.45(s 3H),1.32-1.58(m,10H,),2.05(M,1H),2.30(s,2H),3.23(m,2H),5.50(t,1H,NH),6.58(d,J=4.8Hz,1H).MS(ESI)m/z 373.48(M+H+)。Following the procedure of Example 26, substituting 2-amino-2-methylpropionic acid for 2,2-diethoxypropionic acid, the title compound was obtained. 1 H NMR (CDCl 3 , 400MHz): δ0.97(d, J=6.8Hz, 6H), 1.44(s, 3H), 1.45(s 3H), 1.32-1.58(m, 10H,), 2.05(M , 1H), 2.30(s, 2H), 3.23(m, 2H), 5.50(t, 1H, NH), 6.58(d, J=4.8Hz, 1H). MS(ESI) m/z 373.48(M+ H + ).
实施例28Example 28
1-{[(α-异丁酰氧基丁氧基)羰基]氨基甲基}-1-环己烷乙酸(99)1-{[(α-isobutyryloxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (99)
步骤A: 2-异丙基-1,3-二噻烷(100) Step A: 2-Isopropyl-1,3-dithiane (100)
0℃下向在二氯甲烷中的异丁醛(9.1mL,100mmol)和1,3-丙烷二醇(10mL,100mmol)的混合物中加入三氟化硼二乙基醚合物(6.4mL,50mmol)。将所得的混合物于0℃下搅拌30分钟,并于室温下搅拌30分钟。用盐水、5%NaHCO3和再次用盐水洗涤反应混合物。分离有机相,并用Na2SO4干燥,然后浓缩得到16g(100%)的标题化合物,为一种黄色液体。将其不经进一步纯化而带入下一步。1H NMR(CDCl3,400MHz):δ1.057(D,J=7.2Hz,3H),1.059(d,J=7.2Hz,3H),1.80(M,1H),1.97-2.08(m,2H),2.82(M,4H),4.00(d,J=5.2Hz,1H)。To a mixture of isobutyraldehyde (9.1 mL, 100 mmol) and 1,3-propanediol (10 mL, 100 mmol) in dichloromethane was added boron trifluoride diethyl etherate (6.4 mL, 50mmol). The resulting mixture was stirred at 0°C for 30 minutes and at room temperature for 30 minutes. The reaction mixture was washed with brine, 5% NaHCO 3 and brine again. The organic phase was separated and dried over Na2SO4 , then concentrated to give 16 g (100%) of the title compound as a yellow liquid. It was carried on to the next step without further purification. 1 H NMR (CDCl 3 , 400MHz): δ1.057(D, J=7.2Hz, 3H), 1.059(d, J=7.2Hz, 3H), 1.80(M, 1H), 1.97-2.08(m, 2H ), 2.82 (M, 4H), 4.00 (d, J=5.2Hz, 1H).
步骤B: 2-异丙基-2-(α-羟基丁基)-1,3-二噻烷(101) Step B: 2-Isopropyl-2-(α-hydroxybutyl)-1,3-dithiane (101)
-20℃下向在无水四氢呋喃(50mL)中的(100)(4g,24.7mmol)的溶液滴加正丁基锂(1.6M在己烷中,18.5mL,29.6mmol)。将搅拌的混合物在室温下加热4h,然后再次冷却至-20℃。往此溶液缓慢加入在无水四氢呋喃(10mL)中的正丁醛溶液(2.7mL,29.6mmol)。将所得的混合物在-20℃和室温之间的温度下搅拌16h,用饱和氯化铵溶液终止反应,并用乙酸乙酯萃取混合物。分离有机层并用Na2SO4干燥。减压下除去溶剂后,用硅胶对残余物进行快速柱色谱处理,用5%乙酸乙酯/己烷洗脱得到5g(85%)的标题化合物,为一种黄色的油。1HNMR(CDCl3,400MHz):δ0.96(t,J=7.2Hz,3H),1.11(d,J=6.8,Hz,3H),1.17(d,J=6.8Hz,3H),1.42-1.52(M,2H),1.76(M,1H),1.87-1.95(M,2H),2.04(M,2H),2.62(M,4H),2.94(M,2H),4.03(d,J=5.2HZ,1H)。To a solution of (100) (4 g, 24.7 mmol) in anhydrous tetrahydrofuran (50 mL) was added n-butyllithium (1.6M in hexane, 18.5 mL, 29.6 mmol) dropwise at -20°C. The stirred mixture was heated at room temperature for 4 h, then cooled to -20 °C again. To this solution was slowly added a solution of n-butyraldehyde (2.7 mL, 29.6 mmol) in anhydrous THF (10 mL). The resulting mixture was stirred at a temperature between -20 °C and room temperature for 16 h, quenched with saturated ammonium chloride solution, and the mixture was extracted with ethyl acetate. The organic layer was separated and dried over Na2SO4 . After removal of the solvent under reduced pressure, the residue was flash column chromatographed on silica gel with 5% ethyl acetate/hexane to afford 5 g (85%) of the title compound as a yellow oil. 1 HNMR (CDCl 3 , 400MHz): δ0.96(t, J=7.2Hz, 3H), 1.11(d, J=6.8, Hz, 3H), 1.17(d, J=6.8Hz, 3H), 1.42- 1.52(M, 2H), 1.76(M, 1H), 1.87-1.95(M, 2H), 2.04(M, 2H), 2.62(M, 4H), 2.94(M, 2H), 4.03(d, J= 5.2HZ, 1H).
步骤C: 4-羟基-2-甲基庚烷-3-酮(102) Step C: 4-Hydroxy-2-methylheptan-3-one (102)
在剧烈搅拌下向在乙腈(270mL)中的(101)(5.0g,21.4mmol)的溶液加入在甲醇(30mL)中的Hg(ClO4)2的溶液。室温下将所得的混合物的搅拌2h。过滤后,在不加热的情况下将滤液减压浓缩。用硅胶快速柱色谱法(10%乙酸乙酯/己烷)纯化残余物得到2.8g(91%)的标题化合物,为一种无色液体。1H NMR(CDCl3,400 MHz):S 0.91(t,J=7.2Hz,3H),1.09(d,J=7.2Hz,3H),1.10(d,J=7.2Hz,3H),1.35-1.46(M,4H),1.75(M,1H),2.80(M,1H),3.45(d,J=5.2Hz,1H),4.29(M,1H)。To a solution of (101 ) (5.0 g, 21.4 mmol) in acetonitrile (270 mL) was added a solution of Hg(ClO 4 ) 2 in methanol (30 mL) with vigorous stirring. The resulting mixture was stirred at room temperature for 2 h. After filtration, the filtrate was concentrated under reduced pressure without heating. The residue was purified by silica gel flash column chromatography (10% ethyl acetate/hexane) to give 2.8 g (91%) of the title compound as a colorless liquid. 1 H NMR (CDCl 3 , 400 MHz): S 0.91(t, J=7.2Hz, 3H), 1.09(d, J=7.2Hz, 3H), 1.10(d, J=7.2Hz, 3H), 1.35- 1.46 (M, 4H), 1.75 (M, 1H), 2.80 (M, 1H), 3.45 (d, J=5.2Hz, 1H), 4.29 (M, 1H).
步骤D: 2-甲基庚烷-3-酮-4-对硝基苯基碳酸酯(103) Step D: 2-Methylheptan-3-one-4-p-nitrophenyl carbonate (103)
0℃下向在无水二氯甲烷中的(102)(1.1g,7.6mmol)、氯甲酸对硝基苯基酯(1.84g,9.2mmol)的混合物中缓慢加入在二氯甲烷中的4-二甲基氨基吡啶(1.12g,9.2mmol)的溶液。0℃下搅拌1h和在室温下搅拌4h以后,用10%柠檬酸终止反应。分离有机相,用Na2SO4干燥,并真空浓缩。快速柱色谱处理残余物,用30%二氯甲烷/己烷洗脱,得到2g(85%)的标题化合物,为一种米色固体。1H NMR(CDCl3,400MHz):δ0.99(t,J=7.6Hz,3H),1.12(d,J=6.8Hz,3H),1.18(d,J=6.8Hz,3H),1.51(M,2H),1.84(M,2H),2.82(M,1H),5.17(M,1H),7.42(d,J=6.8Hz,2H),8.25(d,J=6.8Hz,2H)。To a mixture of (102) (1.1 g, 7.6 mmol), p-nitrophenyl chloroformate (1.84 g, 9.2 mmol) in anhydrous dichloromethane was slowly added 4 in dichloromethane at 0 °C. - A solution of dimethylaminopyridine (1.12 g, 9.2 mmol). After stirring for 1 h at 0°C and 4 h at room temperature, the reaction was quenched with 10% citric acid. The organic phase was separated , dried over Na2SO4 , and concentrated in vacuo. Flash column chromatography of the residue eluting with 30% dichloromethane/hexanes afforded 2 g (85%) of the title compound as a beige solid. 1 H NMR (CDCl 3 , 400MHz): δ0.99(t, J=7.6Hz, 3H), 1.12(d, J=6.8Hz, 3H), 1.18(d, J=6.8Hz, 3H), 1.51( M, 2H), 1.84 (M, 2H), 2.82 (M, 1H), 5.17 (M, 1H), 7.42 (d, J=6.8Hz, 2H), 8.25 (d, J=6.8Hz, 2H).
步骤E: 1-{[(α-异丁酰基丁氧基)羰基]氨基甲基}-1-环己烷乙酸 Step E: 1-{[(α-isobutyrylbutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(104) (104)
向含有在二氯甲烷(20mL)中的加巴喷丁(820mg,4.8mmol)和三乙基胺(1.35mL,9.6mmol)的混合物加入三甲基氯硅烷(1.22mL,9.6mmol),并将所得的混合物搅拌20分钟。往此溶液中加入在二氯甲烷(10mL)中的(103)(1g,3.2mmol),并将所得的混合物搅拌60分钟。用10%柠檬酸(20mL)洗涤反应混合物,并分离有机层。用醚(3×10mL)进一步萃取含水层,并用MgSO4干燥合并的有机萃取物,然后在真空下浓缩。用硅胶色谱处理残余物,用己烷∶乙酸乙酯(4∶1)洗脱除去对硝基苯酚,然后再用己烷∶乙酸乙酯(1∶4)洗脱得到(72%)的标题化合物。1H NMR(CDCl3,400MHz):δ0.91(t,J=7.2Hz,3H),1.04(d,J=6.8Hz,3H),1.12(d,J=6.8HZ,3H),1.36-1.53(m,12H),1.74(M,2H),2.33(s,2H),2.78(M,1H),3.22(M,2H),5.11(M,1H),5.48(t,1H,NH).MS(ESI)m/z 342.24(M+H+)。To a mixture containing gabapentin (820 mg, 4.8 mmol) and triethylamine (1.35 mL, 9.6 mmol) in dichloromethane (20 mL) was added trimethylchlorosilane (1.22 mL, 9.6 mmol), and the resulting The mixture was stirred for 20 minutes. To this solution was added (103) (1 g, 3.2 mmol) in dichloromethane (10 mL), and the resulting mixture was stirred for 60 minutes. The reaction mixture was washed with 10% citric acid (20 mL), and the organic layer was separated. The aqueous layer was further extracted with ether (3 x 10 mL), and the combined organic extracts were dried over MgSO 4 and concentrated in vacuo. Chromatography of the residue on silica gel eluting with hexane:ethyl acetate (4:1) to remove p-nitrophenol followed by hexane:ethyl acetate (1:4) gave (72%) the title compound. 1 H NMR (CDCl 3 , 400MHz): δ0.91(t, J=7.2Hz, 3H), 1.04(d, J=6.8Hz, 3H), 1.12(d, J=6.8HZ, 3H), 1.36- 1.53(m, 12H), 1.74(M, 2H), 2.33(s, 2H), 2.78(M, 1H), 3.22(M, 2H), 5.11(M, 1H), 5.48(t, 1H, NH) .MS (ESI) m/z 342.24 (M+H + ).
步骤F: 1-{[(α-异丁酰氧基丁氧基)羰基]氨基甲基}-1-环己烷乙酸 Step F: 1-{[(α-isobutyryloxybutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid
(99) (99)
向在二氯甲烷(20mL)中的(104)(780mg,2.3mmol)的溶液加入间氯过氧苯甲酸(1.03g,4.6mmol)和NaHCO3(386mg,4.6mmol)。室温下搅拌16h后,再加入一批间氯过氧苯甲酸(791mg,4.6mmol)和NaHCO3(386mg,4.6mmol)。将所得的混合物再搅拌8h,然后用10%柠檬酸处理。过滤后,分离有机层,用Na2SO4干燥并浓缩。通过反相制备HPLC法纯化残余物得到79mg(11%)的标题化合物。1HNMR(CDCl3,400MHz):δ0.94(t,J=7.2Hz,3H),1.153(d,J=7.2Hz,3H),1.150(d,J=7.2Hz,3H),1.32-1.58(M,12H),1.74(M,2H),2.28(s,2H),2.56(m,1H),3.23(m,2H),5.27(t,J=6.8Hz,1H,NH),6.71(t,J=5.6Hz,1H).MS(ESI)MLZ 358.30(M+H+)。To a solution of (104) (780 mg, 2.3 mmol) in dichloromethane (20 mL) was added m-chloroperoxybenzoic acid (1.03 g, 4.6 mmol) and NaHCO 3 (386 mg, 4.6 mmol). After stirring at room temperature for 16 h, another batch of m-chloroperoxybenzoic acid (791 mg, 4.6 mmol) and NaHCO 3 (386 mg, 4.6 mmol) were added. The resulting mixture was stirred for a further 8 h, then treated with 10% citric acid. After filtration, the organic layer was separated, dried over Na2SO4 and concentrated. The residue was purified by reverse phase preparative HPLC to afford 79 mg (11%) of the title compound. 1 HNMR (CDCl 3 , 400MHz): δ0.94(t, J=7.2Hz, 3H), 1.153(d, J=7.2Hz, 3H), 1.150(d, J=7.2Hz, 3H), 1.32-1.58 (M, 12H), 1.74(M, 2H), 2.28(s, 2H), 2.56(m, 1H), 3.23(m, 2H), 5.27(t, J=6.8Hz, 1H, NH), 6.71( t, J = 5.6 Hz, 1H). MS (ESI) MLZ 358.30 (M+H + ).
通过以下方法将以上的酸定量转化为对应的钠盐:将酸溶于水(5mL),然后加入等摩尔量的0.5N NaHCO3并冻干。The above acids were quantitatively converted to the corresponding sodium salts by dissolving the acids in water (5 mL), followed by the addition of an equimolar amount of 0.5 N NaHCO 3 and lyophilization.
实施例29Example 29
1-{[(α-异丁酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸甲酯Methyl 1-{[(α-isobutyryloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetate
(105) (105)
步骤A: 1-{[(α-氯异丁氧基)羰基]氨基甲基}-1-环己烷乙酸甲酯 Step A: Methyl 1-{[(α-chloroisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetate
(106) (106)
将(93)(1.0g,3.3mmol)、苯(90mL)和甲醇(10mL)的混合物冷却至0℃。在0℃下缓慢加入三甲基甲硅烷基二偶氮甲烷直至保持黄颜色。将混合物于0℃下搅拌30分钟直至反应完全(用TLC监测)。减压下除去溶剂后,用硅胶色谱处理所得的残余物,用10%乙酸乙酯/己烷洗脱得到760mg(72%)的标题化合物。MS(ESI)m/z 320.24(M+H+)。A mixture of (93) (1.0 g, 3.3 mmol), benzene (90 mL) and methanol (10 mL) was cooled to 0 °C. Trimethylsilyldiazomethane was added slowly at 0°C until the yellow color remained. The mixture was stirred at 0 °C for 30 minutes until the reaction was complete (monitored by TLC). After removal of the solvent under reduced pressure, the resulting residue was chromatographed on silica gel eluting with 10% ethyl acetate/hexanes to afford 760 mg (72%) of the title compound. MS (ESI) m/z 320.24 (M+H + ).
步骤B: 1-{{[(α-异丁酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙 Step B: 1-{{[(α-isobutyryloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexane
酸甲酯(105) Acid methyl ester (105)
将在氯仿中的(106)(760mg,2.38mmol)、碳酸银(394mg,1.4mmol)和异丁酸(442μl,4.76mmol)的混合物在室温下搅拌24h。加入另一批碳酸银(394mg,1.4mmol)和异丁酸(442μL,4.76mmol),并将所得的混合物再搅拌24小时。过滤后,将滤液浓缩并用硅胶快速柱色谱法纯化所得的残余物,用10%乙酸乙酯/己烷洗脱得到560mg(63%)的标题化合物。1H NMR(CDCl3,400MHz):δ0.94(d,J=6.8Hz,3H),0.96(D,J=6.8Hz,3H),1.15(d,J=7.2HZ,3H),1.17(d,J=7.2HZ,3H),1.32-1.58(m,10H),2.01(m,1H),2.19(s,2H),2.55(m,1H),3.18(m,2H),3.67(s,3H),5.33(t,1H),6.56(d,J=4.8Hz,1H)。MS(ESI)m/z 372.38(M+H+)。A mixture of (106) (760 mg, 2.38 mmol), silver carbonate (394 mg, 1.4 mmol) and isobutyric acid (442 μl, 4.76 mmol) in chloroform was stirred at room temperature for 24 h. Another batch of silver carbonate (394 mg, 1.4 mmol) and isobutyric acid (442 μL, 4.76 mmol) was added, and the resulting mixture was stirred for an additional 24 hours. After filtration, the filtrate was concentrated and the resulting residue was purified by flash column chromatography on silica gel, eluting with 10% ethyl acetate/hexanes to afford 560 mg (63%) of the title compound. 1 H NMR (CDCl 3 , 400MHz): δ0.94(d, J=6.8Hz, 3H), 0.96(D, J=6.8Hz, 3H), 1.15(d, J=7.2HZ, 3H), 1.17( d, J=7.2HZ, 3H), 1.32-1.58(m, 10H), 2.01(m, 1H), 2.19(s, 2H), 2.55(m, 1H), 3.18(m, 2H), 3.67(s , 3H), 5.33 (t, 1H), 6.56 (d, J=4.8Hz, 1H). MS (ESI) m/z 372.38 (M+H + ).
实施例30Example 30
1-{[(α-苯甲酰氧基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸甲酯Methyl 1-{[(α-benzoyloxyisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetate
(107) (107)
将1-{[(α-苯甲酸基异丁氧基)羰基]氨基甲基}-1-环己烷乙酸(84)(150mg,0.38mmol)、苯(18mL)和甲醇(2mL)的混合物冷却至0℃。在0℃下缓慢加入三甲基甲硅烷基二偶氮甲烷直至保持黄颜色。将混合物在0℃下搅拌30分钟直至反应完全(通过TLC监测)。减压下除去溶剂后,用硅胶色谱处理残余物,用5%乙酸乙酯/己烷洗脱得到98mg(64%)的标题化合物。1H NMR(CDCl3,400MHz):δ1.02(d,J=6.4Hz,3H),1.03(d,J=6.4Hz,3H),1.32-1.52(m,10H),2.14(M,1H),2.27(s,2H),3.17(M,2H),3.62(s,3H),5.40(t,1H),6.81(d,J=4.8Hz,1H),7.40(M,2H),7.54(M,1H),8.12(m,2H).MS(ESI)m/z 406.29(M+H+)。A mixture of 1-{[(α-benzoylisobutoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid (84) (150 mg, 0.38 mmol), benzene (18 mL) and methanol (2 mL) Cool to 0 °C. Trimethylsilyldiazomethane was added slowly at 0°C until the yellow color remained. The mixture was stirred at 0 °C for 30 minutes until the reaction was complete (monitored by TLC). After removal of the solvent under reduced pressure, the residue was chromatographed on silica gel eluting with 5% ethyl acetate/hexanes to afford 98 mg (64%) of the title compound. 1 H NMR (CDCl 3 , 400MHz): δ1.02(d, J=6.4Hz, 3H), 1.03(d, J=6.4Hz, 3H), 1.32-1.52(m, 10H), 2.14(M, 1H ), 2.27(s, 2H), 3.17(M, 2H), 3.62(s, 3H), 5.40(t, 1H), 6.81(d, J=4.8Hz, 1H), 7.40(M, 2H), 7.54 (M, 1H), 8.12 (m, 2H). MS (ESI) m/z 406.29 (M+H + ).
实施例31Example 31
1-{(N-[(α-异丁酰氧基乙氧基)羰基]-4-溴苯基丙氨酰基]氨基甲基1-{(N-[(α-isobutyryloxyethoxy)carbonyl]-4-bromophenylalanyl]aminomethyl
-1-环己烷乙酸(108) -1-Cyclohexaneacetic acid (108)
步骤A: 1-{(4-溴苯基丙氨酰基)氨基甲基}-1-环己烷乙酸酯(109) Step A: 1-{(4-Bromophenylalanyl)aminomethyl}-1-cyclohexaneacetate (109)
向40mL小瓶中加入N-Boc-4-溴苯基丙氨酸(1.72g,5mmol)、二环己基碳化二亚胺(1.24g,6mmol)、N-羟基琥珀酰亚胺(0.7g,6mmol)和乙腈(20mL)。将反应混合物在25℃下振荡4h。过滤除去沉淀的的二环己基脲。往滤液中加入盐酸加巴喷丁(1.04g,6mmol)和氢氧化钠(0.4g,10mmol)的水溶液(30mL)。将反应物于22-25℃下搅拌16h。用乙酸乙酯(100mL)稀释反应混合物,并用0.5M柠檬酸水溶液(2×100mL)和水(2×100mL)洗涤。分离有机相,干燥(MgSO4),过滤并减压浓缩。将残余物溶于三氟乙酸(40mL),并在22-25℃下静置2h。减压下除去溶剂。将残余物溶于水(4mL),并用0.25μM尼龙膜过滤器过滤,然后通过HPLC(Phenomenex 250×21.2mm,用5μm LUNAC18柱,100%水洗脱5分钟,然后用在水中的0-60%乙腈和0.05%TFA以20mL/min的速率洗脱20分钟)纯化。合并纯馏分,并在减压下除去溶剂得到1.7g(70%)的标题化合物(109),为一种白色固体。MS(ESI)m/z 397.02,399.01(M+H+)。Add N-Boc-4-bromophenylalanine (1.72g, 5mmol), dicyclohexylcarbodiimide (1.24g, 6mmol), N-hydroxysuccinimide (0.7g, 6mmol) into a 40mL vial ) and acetonitrile (20 mL). The reaction mixture was shaken at 25 °C for 4 h. Precipitated dicyclohexylurea was removed by filtration. To the filtrate was added an aqueous solution (30 mL) of gabapentin hydrochloride (1.04 g, 6 mmol) and sodium hydroxide (0.4 g, 10 mmol). The reaction was stirred at 22-25 °C for 16 h. The reaction mixture was diluted with ethyl acetate (100 mL) and washed with 0.5M aqueous citric acid (2 x 100 mL) and water (2 x 100 mL). The organic phase was separated, dried ( MgSO4 ), filtered and concentrated under reduced pressure. The residue was dissolved in trifluoroacetic acid (40 mL) and left standing at 22-25 °C for 2 h. The solvent was removed under reduced pressure. The residue was dissolved in water (4 mL), filtered with a 0.25 μM nylon membrane filter, then passed through HPLC (Phenomenex 250×21.2 mm, with a 5 μm LUNAC18 column, 100% water for 5 minutes, and then 0-60 % acetonitrile and 0.05% TFA at a rate of 20 mL/min for 20 minutes). The pure fractions were combined and the solvent was removed under reduced pressure to give 1.7 g (70%) of the title compound (109) as a white solid. MS (ESI) m/z 397.02, 399.01 (M+H + ).
步骤B: 1-{[N-[(α-异丁酰氧基乙氧基)羰基]-4-溴苯基丙氨酸氨基 Step B: 1-{[N-[(α-isobutyryloxyethoxy)carbonyl]-4-bromophenylalanine amino
甲基}-1-环己烷乙酸(108) Methyl}-1-cyclohexaneacetic acid (108)
0℃下向在二氯甲烷中的(109)(200mg,0.51mmol)的搅拌的悬浮液中加入三乙基胺(141μL,1.01mmol)和三甲基氯硅烷(129mL,1.01mmol)。将所得的混合物于0℃下搅拌15分钟,加入在二氯甲烷中的α-异丁酰氧基乙基-对硝基苯基碳酸酯(111)(144mg,0.51mmol)的溶液。将混合物于室温下搅拌7h(通过LC/MS监测),然后用二氯甲烷稀释反应混合物,用柠檬酸酸化。分离有机层,用盐水洗涤,并用Na2SO4干燥。过滤和浓缩后,用制备LC/MS法纯化粗产物得到92mg的标题化合物。1H-NMR(CD3OD,400MHz):δ1.10(M,6H),1.46-1.25(m,13H),2.20(m,2H),2.48(M,1H),2.84(m,1H),3.06(M,1H),3.17(M,1H),4.36(m,1H),6.67(q,J=5.6Hz,1H),7.17(d,J=2.0,8.0Hz,2H),7.42(DD,J=2.0,8.0Hz,2H)。To a stirred suspension of (109) (200 mg, 0.51 mmol) in dichloromethane at 0 °C was added triethylamine (141 μL, 1.01 mmol) and trimethylchlorosilane (129 mL, 1.01 mmol). The resulting mixture was stirred at 0°C for 15 minutes and a solution of α-isobutyryloxyethyl-p-nitrophenyl carbonate (111) (144 mg, 0.51 mmol) in dichloromethane was added. The mixture was stirred at room temperature for 7 h (monitored by LC/MS), then the reaction mixture was diluted with dichloromethane and acidified with citric acid. The organic layer was separated, washed with brine, and dried over Na2SO4 . After filtration and concentration, the crude product was purified by preparative LC/MS to afford 92 mg of the title compound. 1 H-NMR (CD 3 OD, 400MHz): δ1.10(M, 6H), 1.46-1.25(m, 13H), 2.20(m, 2H), 2.48(M, 1H), 2.84(m, 1H) , 3.06(M, 1H), 3.17(M, 1H), 4.36(m, 1H), 6.67(q, J=5.6Hz, 1H), 7.17(d, J=2.0, 8.0Hz, 2H), 7.42( DD, J = 2.0, 8.0 Hz, 2H).
实施例32Example 32
3-{[(α-异丁酰氧基乙氧基)羰基]氨基甲基}-5-甲基己酸(110)3-{[(α-isobutyryloxyethoxy)carbonyl]aminomethyl}-5-methylhexanoic acid (110)
步骤A:α-异丁酰氧基乙基-对硝基苯基碳酸酯(111) Step A: α-isobutyryloxyethyl-p-nitrophenyl carbonate (111)
将在二氯甲烷(10mL)中的1-氯乙基-对硝基苯基碳酸酯(57)(2.0g,8.14mmol)和异丁酸汞(6.13g,16.29mmol)的溶液于45℃下搅拌24h。然后将反应物冷却至室温,并用己烷稀释以沉淀汞盐。用C盐滤垫过滤沉淀物,并在真空下并将滤液浓缩得到2.5g粗产物。用硅胶色谱处理残余物,用10%二氯甲烷/己烷至20%二氯甲烷/己烷进行梯度洗脱得到1.2g(52%)的标题化合物。1H-NMR(CDCl3,400MHz):δ1.21-1.99(M,6H),1.62(d,J=5.6Hz,3H),2.61(M,1H),6.84(q,J=5.6Hz,1H),7.41(dt,J=6.8,2.4Hz,2H),8.29(dt,J=6.8,2.4Hz,2H)。A solution of 1-chloroethyl-p-nitrophenyl carbonate (57) (2.0 g, 8.14 mmol) and mercuric isobutyrate (6.13 g, 16.29 mmol) in dichloromethane (10 mL) was dissolved at 45° C. Under stirring for 24h. The reaction was then cooled to room temperature and diluted with hexanes to precipitate mercury salts. The precipitate was filtered through a pad of celite, and the filtrate was concentrated under vacuum to give 2.5 g of crude product. The residue was chromatographed on silica gel eluting with a gradient of 10% dichloromethane/hexane to 20% dichloromethane/hexane to afford 1.2 g (52%) of the title compound. 1 H-NMR (CDCl 3 , 400MHz): δ1.21-1.99 (M, 6H), 1.62 (d, J=5.6Hz, 3H), 2.61 (M, 1H), 6.84 (q, J=5.6Hz, 1H), 7.41 (dt, J=6.8, 2.4Hz, 2H), 8.29 (dt, J=6.8, 2.4Hz, 2H).
步骤B: 3-{[(α-异丁酰氧基乙氧基)羰基]氨基甲基-5-甲基己酸 Step B: 3-{[(α-isobutyryloxyethoxy)carbonyl]aminomethyl-5-methylhexanoic acid
(110) (110)
0℃下向在无水二氯甲烷(10mL)中的pregabalin(2)(150mg,0.94mmol)的搅拌的悬浮液加入三乙基胺(0.26mL,1.88mmol)和三甲基氯硅烷(0.24mL,1.88mmol)。在0℃下搅拌15分钟后,加入在二氯甲烷(3mL)中的α-异丁酰氧基乙基-对硝基苯基碳酸酯(111)(267mg,0.94mmol)的溶液。将所得的混合物于室温下搅拌1.5h。用柠檬酸将反应混合物酸化,并用二氯甲烷萃取。用盐水洗涤合并的有机萃取物,并用Na2SO4干燥。过滤和蒸发后,用硅胶色谱法纯化粗产物,首先用二氯甲烷洗脱除去硝基苯酚,然后用在二氯甲烷中的30%乙酸乙酯洗脱得到130mg(48%)的标题化合物,为两种非对映异构体的混合物。1H-NMR(CDCI3,400MHz):δ0.90(m,6H),1.70(M,8H),1.46(d,J=5.6HZ,3H),1.66(1H,M),2.15(M,1H),2.33(M,2H),2.53(m,1H),3.12(m,1H),3.29(M,1H),5.08(t,J=6.0Hz,1H),6.79(M,1H)。To a stirred suspension of pregabalin (2) (150 mg, 0.94 mmol) in anhydrous dichloromethane (10 mL) was added triethylamine (0.26 mL, 1.88 mmol) and trimethylchlorosilane (0.24 mL, 1.88 mmol). After stirring at 0 °C for 15 minutes, a solution of α-isobutyryloxyethyl-p-nitrophenyl carbonate (111) (267 mg, 0.94 mmol) in dichloromethane (3 mL) was added. The resulting mixture was stirred at room temperature for 1.5 h. The reaction mixture was acidified with citric acid and extracted with dichloromethane. The combined organic extracts were washed with brine and dried over Na2SO4 . After filtration and evaporation, the crude product was purified by silica gel chromatography, eluting first with dichloromethane to remove the nitrophenol, then with 30% ethyl acetate in dichloromethane to give 130 mg (48%) of the title compound, It is a mixture of two diastereoisomers. 1 H-NMR (CDCI 3 , 400MHz): δ0.90 (m, 6H), 1.70 (M, 8H), 1.46 (d, J=5.6HZ, 3H), 1.66 (1H, M), 2.15 (M, 1H), 2.33(M, 2H), 2.53(m, 1H), 3.12(m, 1H), 3.29(M, 1H), 5.08(t, J=6.0Hz, 1H), 6.79(M, 1H).
实施例33Example 33
3-{[(α-异丁酰氧基异丁氧基)羰基]氨基甲基}-5-甲基-己酸(112)3-{[(α-isobutyryloxyisobutoxy)carbonyl]aminomethyl}-5-methyl-hexanoic acid (112)
步骤A: 1-氯-2-甲基丙基-对硝基苯基碳酸酯(113) Step A: 1-Chloro-2-methylpropyl-p-nitrophenyl carbonate (113)
向含有在二氯甲烷(200mL)中的对硝基苯酚(4.06g,29mmol)和1-氯-2-甲基丙基氯甲酸酯(5.0g,29mmol)的冰冷的反应混合物中加入在二氯甲烷(50mL)中的吡啶(2.78mL,32mmol)的溶液。将混合物于0℃下搅拌30min,然后在室温下搅拌1h。在减压下蒸发溶剂后,将残余物溶于醚,并用水、10%柠檬酸和再次用水洗涤。分离醚层,用Na2SO4干燥,并减压蒸发得到7.9g(100%)的标题化合物,为一种米色固体。1H NMR(CDCl3,400MHz):δ1.12(d,J=6.6Hz,3H),1.13(d,J=6.6Hz,3H),2.29(M,1H),6.24(d,J=4.8Hz,1H),7.42(d,J=9.2Hz,2H),8.28(d,J=9.2Hz,2H)。To an ice-cold reaction mixture containing p-nitrophenol (4.06 g, 29 mmol) and 1-chloro-2-methylpropyl chloroformate (5.0 g, 29 mmol) in dichloromethane (200 mL) was added in Solution of pyridine (2.78 mL, 32 mmol) in dichloromethane (50 mL). The mixture was stirred at 0 °C for 30 min, then at room temperature for 1 h. After evaporation of the solvent under reduced pressure, the residue was dissolved in ether and washed with water, 10% citric acid and again with water. The ether layer was separated, dried over Na2SO4 , and evaporated under reduced pressure to give 7.9 g (100%) of the title compound as a beige solid. 1 H NMR (CDCl 3 , 400MHz): δ1.12(d, J=6.6Hz, 3H), 1.13(d, J=6.6Hz, 3H), 2.29(M, 1H), 6.24(d, J=4.8 Hz, 1H), 7.42 (d, J=9.2Hz, 2H), 8.28 (d, J=9.2Hz, 2H).
步骤B: α-异丁酰氧基异丁基-对硝基苯基碳酸酯(114) Step B: α-isobutyryloxyisobutyl-p-nitrophenyl carbonate (114)
根据(111)的制备方法,并用(113)代替(57),得到产率为15%的标题化合物,且其原料的回收率为70%。1H-NMR(CDCl3,400MHz):δ1.07(d,J=6.8Hz),1.21(M,6H),2.18(M,1H),2.26(M,1H),6.60(d,J=5.2Hz,1H),7.42(M,2H),8.28(M,2H)。Following the preparation of (111), substituting (113) for (57), the title compound was obtained in 15% yield with 70% recovery of starting material. 1 H-NMR (CDCl 3 , 400MHz): δ1.07(d, J=6.8Hz), 1.21(M, 6H), 2.18(M, 1H), 2.26(M, 1H), 6.60(d, J= 5.2Hz, 1H), 7.42(M, 2H), 8.28(M, 2H).
步骤C: 3-{[(α-异丁酰氧基异丁氧基)羰基]氧基甲基}-5-甲基-己酸 Step C: 3-{[(α-isobutyryloxyisobutoxy)carbonyl]oxymethyl}-5-methyl-hexanoic acid
(112) (112)
根据(110)的制备方法,并用(114)代替(111),得到标题化合物,为两种非对映异构体的混合物,产率为51%。1H-NMR(CDCl3,400MHz):δ0.89(m,12H),1.17(m,8H),1.65(m,1H),2.02(m,1H),2.16(m,1H),2.33(m,2H),2.56(m,1H),3.13(m,1H),3.30(m,1H),5.00(m,1H),6.57-6.56(m,1H)。Following the preparation of (110) and substituting (114) for (111), the title compound was obtained as a mixture of two diastereoisomers in 51% yield. 1 H-NMR (CDCl 3 , 400MHz): δ0.89(m, 12H), 1.17(m, 8H), 1.65(m, 1H), 2.02(m, 1H), 2.16(m, 1H), 2.33( m, 2H), 2.56(m, 1H), 3.13(m, 1H), 3.30(m, 1H), 5.00(m, 1H), 6.57-6.56(m, 1H).
实施例34Example 34
3-{[(α-苯甲酰氧基异丁氧基)羰基]氨基甲基-5-甲基-己酸(115)3-{[(α-Benzoyloxyisobutoxy)carbonyl]aminomethyl-5-methyl-hexanoic acid (115)
步骤A: α-苯甲酰氧基异丁基-对硝基苯基碳酸酯(116) Step A: α-Benzoyloxyisobutyl-p-nitrophenyl carbonate (116)
根据(111)的制备方法,用(113)代替(57)并用苯甲酸汞代替异丁酸汞,得到标题化合物,产率为11%,而原料的回收率为50%。1H-NMR(CDCl3,400MHz):δ1.15(d,J=3.2Hz,3H),1.16(d,J=3.2Hz,3H),2.30(m,1H),6.87(d,J=4.4Hz,1H),7.42(dd,J=7.2,2.0Hz,2H),7.48(t,J=7.6Hz,2H),7.62(t,J=7.6Hz,1H),8.09(dd,J=8.0,1.0Hz,2H),8.27(dd,J=7.2,2.0Hz,2H)。Following the preparation of (111), substituting (113) for (57) and mercury benzoate for mercury isobutyrate, the title compound was obtained in 11% yield and 50% recovery of starting material. 1 H-NMR (CDCl 3 , 400MHz): δ1.15(d, J=3.2Hz, 3H), 1.16(d, J=3.2Hz, 3H), 2.30(m, 1H), 6.87(d, J= 4.4Hz, 1H), 7.42(dd, J=7.2, 2.0Hz, 2H), 7.48(t, J=7.6Hz, 2H), 7.62(t, J=7.6Hz, 1H), 8.09(dd, J= 8.0, 1.0 Hz, 2H), 8.27 (dd, J=7.2, 2.0 Hz, 2H).
步骤B: 3-{[(α-苯甲酰氧基异丁氧基)羰基]氨基甲基}-5-甲基-己酸 Step B: 3-{[(α-Benzoyloxyisobutoxy)carbonyl]aminomethyl}-5-methyl-hexanoic acid
(115) (115)
根据(110)的制备方法,并用(116)代替(111),得到标题化合物,为两种非对映异构体的混合物,产率为58%。1H-NMR(CDCl3,400MHz):δ0.87(m,6H),1.05(m,6H),1.16(m,2H),1.64(m,1H),2.17(m,2H),2.32(m,2H),3.12(m,1H),3.29(m,1H),5.01(br s,1H),6.82(m,1H),7.44(m,2H),7.57(m,1H),8.05(m,2H)。Following the preparation of (110) and substituting (116) for (111), the title compound was obtained as a mixture of two diastereoisomers in 58% yield. 1 H-NMR (CDCl 3 , 400MHz): δ0.87(m, 6H), 1.05(m, 6H), 1.16(m, 2H), 1.64(m, 1H), 2.17(m, 2H), 2.32( m, 2H), 3.12(m, 1H), 3.29(m, 1H), 5.01(br s, 1H), 6.82(m, 1H), 7.44(m, 2H), 7.57(m, 1H), 8.05( m, 2H).
实施例35Example 35
1-{[((5-甲基-2-氧代-1,3-二氧环戊-4-烯-4-基)甲氧基)羰基]氨1-{[((5-methyl-2-oxo-1,3-dioxol-4-en-4-yl)methoxy)carbonyl]ammonia
基甲基}-1-环己烷乙酸(117) Methyl}-1-cyclohexaneacetic acid (117)
步骤A: 苄基2-二偶氮-3-氧代-丁酸(118) Step A: Benzyl 2-diazo-3-oxo-butanoic acid (118)
0℃下向在乙腈(200mL)中的乙酰乙酸苄基酯(5.0g,26.01mmol)和4-乙酰氨基-苯磺酰基叠氮化物(6.25g,26.01mmol)的溶液中滴加三乙基胺(10.9mL,78.03mmol)。将所得的混合物于0℃下搅拌30min,并在室温下搅拌4h。减压浓缩后,将残余物与2∶1乙醚/石油醚(3×100mL)一起研磨。用以硅胶截头的C盐滤垫过滤合并的有机萃取物。在减压除去溶剂得到4.74g的标题化合物,为一种米色结晶。1H-NMR(CDCl3,400MHz):δ2.49(s,3H),5.27(s,2H),7.38(M,5H)。To a solution of benzyl acetoacetate (5.0 g, 26.01 mmol) and 4-acetamido-benzenesulfonyl azide (6.25 g, 26.01 mmol) in acetonitrile (200 mL) was added dropwise triethyl Amine (10.9 mL, 78.03 mmol). The resulting mixture was stirred at 0 °C for 30 min and at room temperature for 4 h. After concentration under reduced pressure, the residue was triturated with 2:1 diethyl ether/petroleum ether (3 x 100 mL). The combined organic extracts were filtered through a pad of Celite truncated with silica gel. The solvent was removed under reduced pressure to give 4.74 g of the title compound as beige crystals. 1 H-NMR (CDCl 3 , 400 MHz): δ 2.49 (s, 3H), 5.27 (s, 2H), 7.38 (M, 5H).
步骤B: 苄基2-羟基-3-氧代-丁酸(119) Step B: Benzyl 2-hydroxy-3-oxo-butanoic acid (119)
将在THF(110mL)和H2O(50mL)中的二偶氮化合物(118)(4.74g,21.74mmol)的溶液与Rh2(OAc)2(77mg,0.17mmol)一起回流加热4小时,并冷却至室温。将混合物在真空下浓缩,并用乙酸乙酯萃取残余物水溶液。用盐水洗涤合并的有机萃取物,用Na2SO4干燥,过滤,并真空浓缩得到4.5g粗产物。1H-NMR(CDCl3,400MHz):δ2.28(s,3H),3.90(s,1H),4.82(s,1H),5.26(m,2H),7.37(m.5H)。A solution of diazo compound (118) (4.74 g, 21.74 mmol) in THF (110 mL) and H 2 O (50 mL) was heated at reflux with Rh 2 (OAc) 2 (77 mg, 0.17 mmol) for 4 hours, and cool to room temperature. The mixture was concentrated under vacuum, and the aqueous residue was extracted with ethyl acetate. The combined organic extracts were washed with brine, dried over Na2SO4 , filtered, and concentrated in vacuo to afford 4.5 g of crude product. 1 H-NMR (CDCl 3 , 400 MHz): δ 2.28 (s, 3H), 3.90 (s, 1H), 4.82 (s, 1H), 5.26 (m, 2H), 7.37 (m.5H).
步骤C: 4-苄氧基羰基5-甲基-2-氧代-1.3-二氧环戊-4-烯(120) Step C: 4-Benzyloxycarbonyl 5-methyl-2-oxo-1.3-dioxol-4-ene (120)
0℃下向在THF(50mL)中的羰基二咪唑(6.88g,42.45mmol)的悬浮液加入在无水THF(50mL)中的醇(119)(4.50g,21.22mmol)的溶液。将所得的混合物在0℃下搅拌5小时,在室温下搅拌过夜。将混合物真空浓缩,并用水和乙酸乙酯/己烷分配残余物。分离有机层,用饱和NH4Cl、盐水洗涤,并用Na2SO4干燥。过滤和浓缩后,用硅胶快速色谱法纯化粗产物,用在己烷中的20%乙酸乙酯洗脱得到2.6g的标题化合物。1H-NMR(CDCl3,400MHz):δ2.48(s,3H),5.27(s,2H),7.37(br.s,5H)。To a suspension of carbonyldiimidazole (6.88 g, 42.45 mmol) in THF (50 mL) was added a solution of alcohol (119) (4.50 g, 21.22 mmol) in anhydrous THF (50 mL) at 0°C. The resulting mixture was stirred at 0°C for 5 hours and at room temperature overnight. The mixture was concentrated in vacuo and the residue was partitioned with water and ethyl acetate/hexanes. The organic layer was separated, washed with saturated NH4Cl , brine, and dried over Na2SO4 . After filtration and concentration, the crude product was purified by flash chromatography on silica gel eluting with 20% ethyl acetate in hexanes to afford 2.6 g of the title compound. 1 H-NMR (CDCl 3 , 400 MHz): δ 2.48 (s, 3H), 5.27 (s, 2H), 7.37 (br.s, 5H).
步骤D: 5-甲基-2-氧代-1.3-二氧环戊-4-烯基-4-甲酸(121) Step D: 5-Methyl-2-oxo-1.3-dioxol-4-enyl-4-carboxylic acid (121)
向在50mL乙醇中的化合物(120)(2.6g,10.92mmol)的溶液加入260mg PD/C(5%),并将所得的混合物在氢大气下搅拌1h。过滤和在减压下除去溶剂得到1.62g的标题化合物。1H-NMR(CD3OD,400MHz):δ2.41(s,3H)。To a solution of compound (120) (2.6 g, 10.92 mmol) in 50 mL of ethanol was added 260 mg of PD/C (5%), and the resulting mixture was stirred under a hydrogen atmosphere for 1 h. Filtration and removal of solvent under reduced pressure gave 1.62 g of the title compound. 1 H-NMR (CD 3 OD, 400 MHz): δ2.41 (s, 3H).
步骤E:4-羟基甲基-5-甲基-2-氧代-1.3-二氧环戊-4-烯(122) Step E: 4-Hydroxymethyl-5-methyl-2-oxo-1.3-dioxol-4-ene (122)
0℃下向在无水二氯甲烷(50mL)中的酸(121)(1.62g,11.10mmol)和无水DMF(112μL)的溶液滴加草酰氯(6.1mL,2M溶液,12.2mmol)。在0℃下搅拌30分钟和在室温下搅拌1h后,在减压下除去溶剂。将残余物溶于无水二氯甲烷(65mL),并冷却至-78℃。10分钟内往此溶液滴加Bu4NBH4(3.14g,12.2mmol,在20mL二氯甲烷中)的溶液。在-78℃下搅拌1h后,小心地用0.1N HCl(30mL)终止混合物反应,并加热至室温。分离含水层,用EtOAc(3×50mL)萃取,用盐水洗涤合并的有机萃取物,并用Na2SO4干燥。减压下除去溶剂后,用硅胶进行柱色谱处理,用在二氯甲烷中的50%EtOAc洗脱得到767mg的标题化合物。1H-NMR(CD3OD,400MHz):δ2.09(s,3H),4.34(s,2H)。To a solution of acid (121) (1.62 g, 11.10 mmol) and dry DMF (112 μL) in dry dichloromethane (50 mL) was added oxalyl chloride (6.1 mL, 2M solution, 12.2 mmol) dropwise at 0°C. After stirring at 0 °C for 30 minutes and at room temperature for 1 h, the solvent was removed under reduced pressure. The residue was dissolved in anhydrous dichloromethane (65 mL), and cooled to -78°C. To this solution was added a solution of Bu4NBH4 (3.14 g , 12.2 mmol in 20 mL of dichloromethane) dropwise over 10 min. After stirring at -78 °C for 1 h, the mixture was carefully quenched with 0.1 N HCl (30 mL) and allowed to warm to room temperature. The aqueous layer was separated, extracted with EtOAc (3 x 50 mL ), the combined organic extracts were washed with brine, and dried over Na2SO4 . After removal of the solvent under reduced pressure, column chromatography on silica gel eluting with 50% EtOAc in dichloromethane afforded 767 mg of the title compound. 1 H-NMR (CD 3 OD, 400 MHz): δ 2.09 (s, 3H), 4.34 (s, 2H).
步骤F:1-{[((5-甲基-2-氧代-1,3-二氧环戊-4-烯-4-基)甲氧基)Step F: 1-{[((5-Methyl-2-oxo-1,3-dioxol-4-en-4-yl)methoxy)
羰基]-氨基甲基}-1-环己烷乙酸苄基酯(123) Carbonyl]-aminomethyl}-1-cyclohexane benzyl acetate (123)
将在甲苯中的醇(122)(767mg,5.9mmol)和1-异氰酸基甲基-1-环己烷乙酸苄基酯(5.9mmol)的悬浮液回流过夜。减压下除去溶剂后,用快速柱色谱法纯化残余物,用在己烷中的30%EtOAc的洗脱得到510mg的标题化合物。1H-NMR(CD3OD,400MHz):δ1.58-1.30(m,10H),2.18(s,3H),2.35(s,2H),3.17(d,J=6.8Hz,2H),4.80(s,2H),5.11(s,2H),5.44(t,J=6.8Hz,1H),7.36(m,5H)。A suspension of alcohol (122) (767 mg, 5.9 mmol) and benzyl 1-isocyanatomethyl-1-cyclohexaneacetate (5.9 mmol) in toluene was refluxed overnight. After removal of the solvent under reduced pressure, the residue was purified by flash column chromatography eluting with 30% EtOAc in hexanes to afford 510 mg of the title compound. 1 H-NMR (CD 3 OD, 400MHz): δ1.58-1.30 (m, 10H), 2.18 (s, 3H), 2.35 (s, 2H), 3.17 (d, J=6.8Hz, 2H), 4.80 (s, 2H), 5.11 (s, 2H), 5.44 (t, J=6.8Hz, 1H), 7.36 (m, 5H).
步骤G: 1-{[((5-甲基-2-氧代-1.3-二氧环戊-4-烯-4-基)甲氧基)羰 Step G: 1-{[((5-Methyl-2-oxo-1.3-dioxol-4-en-4-yl)methoxy)carbonyl
基]-氨基甲基}-1-环己烷乙酸(117) Base]-aminomethyl}-1-cyclohexaneacetic acid (117)
向在乙醇(20mL)中的化合物(123)(510mg,1.41mmol)的溶液加入59mg PD/C(5%),并将所得的混合物在氢大气下搅拌1h。减压下过滤和除去挥发物得到粗产物,通过制备LC/MS法纯化粗产物得到105mg的标题化合物。1H-NMR(CD3OD,400MHz):δ1.52-1.36(m,10H),2.16(s,3H),2.27(s,2H),3.22(s,2H),4.86(s,2H)。To a solution of compound (123) (510 mg, 1.41 mmol) in ethanol (20 mL) was added 59 mg of PD/C (5%), and the resulting mixture was stirred under a hydrogen atmosphere for 1 h. Filtration and removal of volatiles under reduced pressure gave the crude product, which was purified by preparative LC/MS to give 105 mg of the title compound. 1 H-NMR (CD 3 OD, 400MHz): δ1.52-1.36(m, 10H), 2.16(s, 3H), 2.27(s, 2H), 3.22(s, 2H), 4.86(s, 2H) .
实施例36Example 36
1-{(1-甲基-3-氧代-丁-1-羰基)氨基甲基}-1-环己烷乙酸哌啶鎓1-{(1-Methyl-3-oxo-butan-1-carbonyl)aminomethyl}-1-cyclohexanepiperidinium acetate
(124) (124)
将2,4-戊二酮(103μL,1mmol)、加巴喷丁(171mg,1mmol)和哌啶(99μL,1mmol)混入无水甲醇(10mL)。将所得的混合物加热回流4h。减压下除去溶剂得到纯度大于90%的标题化合物1H NMR(CDCl3,400MHz):δ1.34-1.62(M,12H),1.71(M,4H),1.94(s,3H),1.96(s,3H),2.26(s,2H),2.98(M,4H),3.38(d,J=6Hz,2H),4.90(s,1H),5.20(s,br,2H),8.64(t,J=6Hz,1H).MS(ESI)m/z252.35(M-H-)。2,4-Pentanedione (103 μL, 1 mmol), gabapentin (171 mg, 1 mmol) and piperidine (99 μL, 1 mmol) were mixed into anhydrous methanol (10 mL). The resulting mixture was heated to reflux for 4h. The solvent was removed under reduced pressure to give the title compound with a purity greater than 90% 1 H NMR (CDCl 3 , 400 MHz): δ 1.34-1.62 (M, 12H), 1.71 (M, 4H), 1.94 (s, 3H), 1.96 ( s, 3H), 2.26(s, 2H), 2.98(M, 4H), 3.38(d, J=6Hz, 2H), 4.90(s, 1H), 5.20(s, br, 2H), 8.64(t, J = 6 Hz, 1H). MS (ESI) m/z 252.35 (MH - ).
实施例37Example 37
1-1-{[(2-氧代-四氢呋喃-3-基亚基)乙基]氨基甲基}-1-环己烷乙酸1-1-{[(2-oxo-tetrahydrofuran-3-ylidene)ethyl]aminomethyl}-1-cyclohexaneacetic acid
哌啶鎓(125) Piperidinium (125)
将2-乙酰基丁内酯(108μL,1mmol)、加巴喷丁(171mg,1mmol)和哌啶(99μL,1mmol)混入无水甲醇(10mL)。回流加热6h后,在减压下除去溶剂得到纯度大于90%的标题化合物。1H NMR(CDCl3,400MHz):δ1.34-1.62(m,12H),1.71(m,4H),1.94(s,3H),2.24(s,2H),2.81(T,J=7.6Hz,2H),2.99(M,4H),3.31(d,J=6.4Hz,2H),4.23(t,J=7.6Hz,2H),5.17(s,br,2H),8.64(t,J=6.4Hz,1H).MS(ESI)m/z 280.34(M-H-)。2-Acetylbutyrolactone (108 μL, 1 mmol), gabapentin (171 mg, 1 mmol) and piperidine (99 μL, 1 mmol) were mixed into anhydrous methanol (10 mL). After heating at reflux for 6 h, the solvent was removed under reduced pressure to afford the title compound with a purity greater than 90%. 1 H NMR (CDCl 3 , 400MHz): δ1.34-1.62(m, 12H), 1.71(m, 4H), 1.94(s, 3H), 2.24(s, 2H), 2.81(T, J=7.6Hz , 2H), 2.99(M, 4H), 3.31(d, J=6.4Hz, 2H), 4.23(t, J=7.6Hz, 2H), 5.17(s, br, 2H), 8.64(t, J= 6.4 Hz, 1H). MS (ESI) m/z 280.34 (MH-).
实施例38Example 38
1-{(2-碳甲氧基-环戊-1-烯基)氨基甲基}-1-环己烷乙酸哌啶鎓1-{(2-Carbonmethoxy-cyclopent-1-enyl)aminomethyl}-1-cyclohexanepiperidinium acetate
(126) (126)
将2-氧代环戊烷甲酸甲酯(124μL,1mmol)、加巴喷丁(171mg,1mmol)和哌啶(99μL,1mmol)混入无水甲醇(10mL)。加热回流16h后,在减压下除去溶剂得到纯度大于90%的标题化合物。1H NMR(CDCl3,400MHz):δ1.29-1.60(M,12H),1.72(M,4H),1.79(M,J=7.6Hz,2H),2.24(s,2H),2.49(t,J=7.6Hz,2H),2.55(t,J=7.6Hz,2H),2.99(M,4H),3.24(d,J=6.8Hz,2H),3.63(s,3H),5.06(s,br,2H),7.93(s,br,1H).MS(ESI)m/z 294.36(M-H-)。Methyl 2-oxocyclopentanecarboxylate (124 μL, 1 mmol), gabapentin (171 mg, 1 mmol) and piperidine (99 μL, 1 mmol) were mixed into anhydrous methanol (10 mL). After heating to reflux for 16 h, the solvent was removed under reduced pressure to obtain the title compound with a purity greater than 90%. 1 H NMR (CDCl 3 , 400MHz): δ1.29-1.60(M, 12H), 1.72(M, 4H), 1.79(M, J=7.6Hz, 2H), 2.24(s, 2H), 2.49(t , J=7.6Hz, 2H), 2.55(t, J=7.6Hz, 2H), 2.99(M, 4H), 3.24(d, J=6.8Hz, 2H), 3.63(s, 3H), 5.06(s , br, 2H), 7.93 (s, br, 1H). MS (ESI) m/z 294.36 (MH-).
实施例39Example 39
1-{(1-甲基-2-(乙氧基羰基)-3-乙氧基-3-氧代丙-1-烯基)氨基甲1-{(1-methyl-2-(ethoxycarbonyl)-3-ethoxy-3-oxoprop-1-enyl)aminomethyl
基}-1-环己烷乙酸哌啶鎓(127) Base}-1-cyclohexane piperidinium acetate (127)
将乙酰基丙二酸二乙酯(202mg,1mmol)、加巴喷丁(171mg,1mmol)和哌啶(99μL,1mmol)混入无水乙醇(10mL)。加热回流16h后,在减压下除去溶剂得到纯度大于90%的标题化合物。1H NMR(CDCl3,400MHz):δ1.28(t,J=7.2Hz,6H),1.38-1.64(m,12H),1.75(m,4H),1.96(s,3H),2.23(s,2H),2.99(m,4H),3.24(d,J=5.2Hz,2H),4.20(q,J=7.2Hz,4H),4.35(s,br,2H),7.79(t,J=5.2Hz,1H).MS(ESI)m/z 354.38(M-H-)。Diethyl acetylmalonate (202 mg, 1 mmol), gabapentin (171 mg, 1 mmol) and piperidine (99 μL, 1 mmol) were mixed into absolute ethanol (10 mL). After heating to reflux for 16 h, the solvent was removed under reduced pressure to obtain the title compound with a purity greater than 90%. 1 H NMR (CDCl 3 , 400MHz): δ1.28(t, J=7.2Hz, 6H), 1.38-1.64(m, 12H), 1.75(m, 4H), 1.96(s, 3H), 2.23(s , 2H), 2.99(m, 4H), 3.24(d, J=5.2Hz, 2H), 4.20(q, J=7.2Hz, 4H), 4.35(s, br, 2H), 7.79(t, J= 5.2 Hz, 1H). MS (ESI) m/z 354.38 (MH - ).
实施例40Example 40
1-{[(α-(2-(2-甲基-1,3-二氧戊环-2-基)羧基异丁氧基)羰基]-氨基1-{[(α-(2-(2-methyl-1,3-dioxolan-2-yl)carboxyisobutoxy)carbonyl]-amino
甲基}-1-环己烷乙酸(128) Methyl}-1-cyclohexaneacetic acid (128)
步骤A: 2-甲基-1,3-二氧戊环-2-甲酸(129) Step A: 2-Methyl-1,3-dioxolane-2-carboxylic acid (129)
0℃下向含有在无水二氯甲烷(100mL)中的丙酮酸乙酯(11.1mL,0.1mol)和乙二醇(5.6mL,0.1mol)的搅拌的混合物中加入三氟化硼二醚合物(6.4mL,0.05mol)和催化量的乙酸。将所得的混合物在40℃搅拌16h,然后用100mL二氯甲烷稀释。连续地用饱和氯化钠溶液(2×80mL)洗涤有机溶液。分离有机层,并浓缩合并的有机萃取物。室温下用1N氢氧化钠处理残余物。室温下搅拌3h(由TLC监测)后,加入柠檬酸将pH调至4。用二氯甲烷萃取产物,用Na2SO4干燥,并浓缩得到5.1g(38%)的标题化合物(129),为一种澄清的液体。将此物质不经进一步纯化用于下一步反应。1HNMR(CDCl3,400MHz):51.55(s,3H),4.03(m,4H)。To a stirred mixture containing ethyl pyruvate (11.1 mL, 0.1 mol) and ethylene glycol (5.6 mL, 0.1 mol) in anhydrous dichloromethane (100 mL) was added boron trifluoride diether at 0 °C mixture (6.4 mL, 0.05 mol) and a catalytic amount of acetic acid. The resulting mixture was stirred at 40 °C for 16 h, then diluted with 100 mL of dichloromethane. The organic solution was washed successively with saturated sodium chloride solution (2 x 80 mL). The organic layer was separated, and the combined organic extracts were concentrated. The residue was treated with 1N sodium hydroxide at room temperature. After stirring at room temperature for 3 h (monitored by TLC), the pH was adjusted to 4 by adding citric acid. The product was extracted with dichloromethane , dried over Na2SO4 , and concentrated to give 5.1 g (38%) of the title compound (129) as a clear liquid. This material was used in the next reaction without further purification. 1 H NMR (CDCl 3 , 400 MHz): 51.55 (s, 3H), 4.03 (m, 4H).
步骤B: 苄基1-{[(α-(2-(2-甲基-1,3-二氧戊环-2-基)羧基异丁氧 Step B: Benzyl 1-{[(α-(2-(2-methyl-1,3-dioxolan-2-yl)carboxyisobutoxy
基)羰基]氨基甲基}-1-环己烷乙酸苄基酯(130) Base) carbonyl] aminomethyl}-1-cyclohexane acetic acid benzyl ester (130)
室温下将含有在氯仿中的1-{[(α-氯异丁氧基)羰基氨基甲基}-1-环己烷乙酸苄基酯(95)(1g,2.53mmol)、(129)(673mg,5.1mmol)、碳酸银(557mg,2.53mmol)和三乙基胺(709μl,5.1mmol)的混合物搅拌16h。过滤后,将滤液浓缩。用硅胶色谱法纯化所得的残余物,用15%乙酸乙酯/己烷洗脱得到510mg(41%)的标题化合物(130)。MS(ESI)m/z 492.40(M+H+)。Benzyl 1-{[(α-chloroisobutoxy)carbonylaminomethyl}-1-cyclohexaneacetate (95) (1 g, 2.53 mmol), (129) ( A mixture of 673 mg, 5.1 mmol), silver carbonate (557 mg, 2.53 mmol) and triethylamine (709 μl, 5.1 mmol) was stirred for 16 h. After filtration, the filtrate was concentrated. The resulting residue was purified by silica gel chromatography eluting with 15% ethyl acetate/hexane to afford 510 mg (41%) of the title compound (130). MS (ESI) m/z 492.40 (M+H + ).
步骤C: 1-{[(α-(2-(2-甲基-1,3-二氧戊环-2-基)羧基异丁氧基)羰 Step C: 1-{[(α-(2-(2-methyl-1,3-dioxolan-2-yl)carboxyisobutoxy)carbonyl
基]-氨基甲基}-1-环己烷乙酸(128) Base]-aminomethyl}-1-cyclohexaneacetic acid (128)
在氢气氛和室温下将在乙醇中的(130)(470mg,0.96mmol)和5%Pd-C(催化量)的混合物搅拌16h。过滤和浓缩得到382mg(100%)的标题化合物(128)。1H NMR(CDCl3,400MHz):δ0.96(d,J=6.8Hz,3H),0.97(d,J=6.8Hz,3H),1.32-1.58(m,10H),1.59(s,3H),2.06(M,1H),2.32(s,2H),3.26(m,2H),4.08(M,4H),5.29(t,1H,NH),6.55(d,J=4.8Hz,1H).MS(ESI)m/z 402.32(M+H+)。通过以下方法将酸形式定量转化为对应的钠盐:溶于水(5mL),加入等摩尔量的0.5N NaHCO3,然后冻干。A mixture of (130) (470 mg, 0.96 mmol) and 5% Pd—C (catalytic amount) in ethanol was stirred at room temperature under hydrogen atmosphere for 16 h. Filtration and concentration afforded 382 mg (100%) of the title compound (128). 1 H NMR (CDCl 3 , 400MHz): δ0.96(d, J=6.8Hz, 3H), 0.97(d, J=6.8Hz, 3H), 1.32-1.58(m, 10H), 1.59(s, 3H ), 2.06(M, 1H), 2.32(s, 2H), 3.26(m, 2H), 4.08(M, 4H), 5.29(t, 1H, NH), 6.55(d, J=4.8Hz, 1H) .MS (ESI) m/z 402.32 (M+H + ). The acid form was quantitatively converted to the corresponding sodium salt by dissolving in water (5 mL), adding an equimolar amount of 0.5N NaHCO3 , and then lyophilizing.
实施例41Example 41
体外测定前药的Caco-2细胞渗透性 In vitro assay of Caco-2 cell permeability of prodrugs
可以使用本领域中已知的方法对本发明的前药的被动渗透性进行体外评价(例如参见Stewart等人,PHARM.RES.,1995,12,693)。例如,可以通过检查前药流过培养的极化细胞单层(例如Caco-2细胞)来评价被渗透性。将得自连续培养物的Caco-2细胞(传代少于28)高密度地接种到Transwell聚碳酸酯过滤器。用DMEM/10%牛胎儿血清+0.1mM非必需氨基酸+2mM L-GLn、5% CO2/95%,37℃供养细胞直至实验当天。在溢出泵抑制剂(250μM MK-571,250μM维拉帕米,1mM氧氟沙星)存在下,在pH6.5下和在顶部(在含有1mM CaCl2、1MM MgCl2、150mM NaCl、3mM KCl、1mM NaH2PO4、5mM葡萄糖的50mM MES缓冲液中)以及在pH7.4下在底侧(在含有10mM HEPES的Hanks平均盐溶液中)进行渗透性研究。将插入物置于含有缓冲液的12孔或24孔平板,并在37℃下培养30min。将前药(200μM)加到顶部和底侧隔室(供者),并使用LC/MS/MS以1小时的间隔测定在相对的隔室(接受者)中的前药和/或释放的母体药物的浓度。用以下方程计算表观渗透值(Papp):The passive permeability of the prodrugs of the invention can be assessed in vitro using methods known in the art (see eg Stewart et al., PHARM. RES., 1995, 12, 693). For example, permeabilization can be assessed by examining the flow of the prodrug through a cultured monolayer of polarized cells (eg, Caco-2 cells). Caco-2 cells (passage less than 28) from continuous culture were seeded at high density onto Transwell polycarbonate filters. The cells were fed with DMEM/10% fetal calf serum + 0.1 mM non-essential amino acids + 2 mM L-GLn, 5% CO 2 /95%, 37°C until the day of the experiment. In the presence of overflow pump inhibitors (250 μM MK-571, 250 μM verapamil, 1 mM ofloxacin), at pH 6.5 and on top (in the presence of 1 mM CaCl 2 , 1MM MgCl 2 , 150 mM NaCl, 3 mM KCl , 1 mM NaH 2 PO 4 , 5 mM glucose in 50 mM MES buffer) and on the bottom side (in Hanks mean salt solution containing 10 mM HEPES) at pH 7.4. Inserts were placed in 12- or 24-well plates containing buffer and incubated at 37°C for 30 min. The prodrug (200 μM) was added to the top and bottom side compartments (donor), and the prodrug and/or released prodrug in the opposite compartment (acceptor) was measured at 1 hour intervals using LC/MS/MS. Concentration of parent drug. Calculate the apparent permeability value (Papp) using the following equation:
Papp=Vr(dC/dt)//(AC0)P app =V r (dC/dt)//(AC 0 )
这里的Vr为以mL为单位的接受隔室的体积;dC/dt为前药和母体药物的流通量(μM/S),它根据接受隔室内的浓度对时间的曲线的斜率测定;C0为以μM为单位的前药的最初浓度;A为以cm2为单位的膜表面积。优选具有显著的穿过细胞的渗透性的前药表现出的Papp的值≥1×10-6cm/s,更优选Papp的值≥1×10-65cm/s,进一步更优选Papp的值≥5×10-5cm/s。关于GABA类似物的前药所得的典型的Papp值如以下表所示:
此表中的数据表明本文公开的前药具有高细胞渗透性,并应该良好地被肠吸收。除了化合物(83),前药顶部至底侧渗透性超过它们的底侧至顶部渗透性。这表明这些化合物可以是在Caco细胞顶膜中存在的活性转运机制的底物(虽然这种穿过细胞的渗透性的某些组成是通过被动扩散调节的)。(83)的底侧至顶部的的渗透性越大表明尽管存在流出泵抑制剂MK-571,维拉帕米和氧氟沙星,这种化合物可以流动通过底侧膜。The data in this table indicate that the prodrugs disclosed herein are highly cell permeable and should be well absorbed by the intestine. With the exception of compound (83), the top-to-bottom side permeability of the prodrugs exceeded their bottom-side to top permeability. This suggests that these compounds may be substrates for an active transport mechanism present in the apical membrane of Caco cells (although some components of this permeability across the cell are regulated by passive diffusion). The greater bottom-to-top permeability of (83) indicated that this compound could flow through the bottom-side membrane despite the presence of the efflux pump inhibitors MK-571, verapamil and ofloxacin.
实施例42Example 42
在对大鼠进行加巴喷丁或加巴喷丁前药的结肠内给药之后的加巴喷丁Gabapentin following intracolonic administration of gabapentin or a gabapentin prodrug to rats
的吸收 Absorption
在6-24小时内缓慢地释放药物的持续释放的口服剂型一般在结肠内释放大量的剂量。因此适用于这种剂型的药物优选表现出良好的结肠吸收。进行本实验以评价加巴喷丁前药在口服持续释放剂型中的应用。Sustained-release oral dosage forms, which release the drug slowly over a period of 6-24 hours, generally deliver a substantial dose in the colon. Drugs suitable for use in such dosage forms therefore preferably exhibit good colonic absorption. This experiment was performed to evaluate the use of gabapentin prodrugs in oral sustained release dosage forms.
步骤A:给药方案 Step A: Dosing regimen
商购得到大鼠并在升结肠和颈静脉预插套管。动物在实验时是清醒的。所有的动物禁食过夜直至服药后4小时。通过插管以相当于25mg加巴喷丁/kg的剂量将加巴喷丁或加巴喷丁前药(59)、(63)、(69)、(72)、(77)、(79)、(85)、(117)和(126)的溶液(在水或PEG 400中)直接施用于结肠。在8小时内间隔地从颈静脉获得血样(0.5mL),并通过加入乙腈/甲醇立即终止反应,以防止前药前一步转化。如下述分析血样。Rats were obtained commercially and precannulated in the ascending colon and jugular vein. Animals were awake during the experiment. All animals were fasted overnight until 4 hours after dosing. Gabapentin or gabapentin prodrugs (59), (63), (69), (72), (77), (79), (85), (117) and A solution of (126) (in water or PEG 400) was applied directly to the colon. Blood samples (0.5 mL) were obtained from the jugular vein at intervals over 8 hours, and the reaction was stopped immediately by the addition of acetonitrile/methanol to prevent the previous step conversion of the prodrug. Blood samples were analyzed as described below.
步骤B:结肠吸收药物的样品制备 Step B: Sample Preparation for Colonically Absorbed Drugs
1.在空白1.5mL eppendorf管中,加入300μL 50/50乙腈/甲醇和20μL对氯苯基丙氨酸作为内标。1. In a blank 1.5mL eppendorf tube, add 300μL 50/50 acetonitrile/methanol and 20μL p-chlorophenylalanine as internal standard.
2.在不同的时间点收集大鼠血液,立即将100μL血液加到eppendorf管,并搅拌混合。2. Rat blood was collected at different time points, and 100 μL of blood was immediately added to an eppendorf tube and stirred to mix.
3.将10μL加巴喷丁标准溶液(0.04,0.2,1,5,25,100μg/ml)加到90μL空白大鼠血液以组成最终的校准标准(0.004,0.02,0.1,0.5,2.5,10μg/ml),然后将300μL 50/50乙腈/甲醇加到每个管中,随后加入20μL对氯苯基丙氨酸。3. Add 10 μL of gabapentin standard solution (0.04, 0.2, 1, 5, 25, 100 μg/ml) to 90 μL of blank rat blood to form the final calibration standard (0.004, 0.02, 0.1, 0.5, 2.5, 10 μg/ml) , then add 300 µL of 50/50 acetonitrile/methanol to each tube, followed by 20 µL of p-chlorophenylalanine.
4.将样品搅拌并以14,000rpm速率离心10分钟。4. The sample was stirred and centrifuged at 14,000 rpm for 10 minutes.
5.取上清液用于LC/MS/MS分析。5. Take the supernatant for LC/MS/MS analysis.
步骤C: LC/MS/MS分析 Step C: LC/MS/MS Analysis
将配备Shidmadzu 10ADVp二元泵和CTC HTS-PAL自动取样器的API 2000 LC/MS/MS分光计用于分析。在分析期间将Zorbax XDB C84.6×150mm柱加热到45℃。流动相为0.1%甲酸(A)和乙腈与0.1%甲酸(B)。梯度条件是:5%B流脱1分钟,然后98%B洗脱3分钟,然后保持用98%B洗脱2.5分钟。将流动相复原到5%B洗脱2分钟。将TurboIonSpray来源用于API 2000。以正离子方式完成分析,并将MRM转变172/137用于分析加巴喷丁(所用的MRM转变对于(59)为426/198,对于(63)为364/198,对于(69)为392/198,对于(72)为316/198,对于(77)为330/198,对于(79)为330/198,对于(85)为316/198和对于(117)为327.7/153.8)。注射20μL样品。使用Analyst1.1定量软件将峰积分。在对这些前药中的每一种进行结肠给药后,加巴喷丁最大血浆浓度(Cmax)以及加巴喷丁血浆浓度地时间曲线下的面积(AUC)明显大于(>2倍)加巴喷丁本身结肠给药产生的曲线下的面积。例如,前药(77)提供的加巴喷丁Cmax和AUC值比加巴喷丁本身大10倍。此数据表明本发明的化合物可以制成适于促进吸收和/或有效地持续释放GABA类似物的组合物,从而由于GABA类似物的快速系统清除率而将服药频率最小化。An API 2000 LC/MS/MS spectrometer equipped with a Shidmadzu 10ADVp binary pump and a CTC HTS-PAL autosampler was used for the analysis. A Zorbax XDB C8 4.6 x 150mm column was heated to 45°C during the analysis. The mobile phases were 0.1% formic acid (A) and acetonitrile with 0.1% formic acid (B). Gradient conditions were: 5% B for 1 minute, then 98% B for 3 minutes, then hold 98% B for 2.5 minutes. Return the mobile phase to 5% B and elute for 2 min. Use TurboIonSpray source for API 2000. The analysis was done in positive ion mode and MRM transitions 172/137 were used for the analysis of gabapentin (MRM transitions used were 426/198 for (59), 364/198 for (63), 392/198 for (69), 316/198 for (72), 330/198 for (77), 330/198 for (79), 316/198 for (85) and 327.7/153.8 for (117)). Inject 20 μL of sample. The peaks were integrated using Analyst 1.1 quantitative software. Following colonic administration of each of these prodrugs, the maximum plasma concentration (C max ) of gabapentin and the area under the time curve (AUC) of the plasma concentration of gabapentin were significantly greater (>2-fold) than that produced by colonic administration of gabapentin itself. The area under the curve of . For example, the prodrug (77) provided gabapentin Cmax and AUC values 10-fold greater than gabapentin itself. This data suggests that the compounds of the invention can be formulated in compositions suitable for enhanced absorption and/or effective sustained release of the GABA analog, thereby minimizing dosing frequency due to the rapid systemic clearance of the GABA analog.
实施例43Example 43
在使用迷你型泵给猎狗施用前药后的加巴喷丁的持续释放 Sustained release of gabapentin following prodrug administration to hunting dogs using a mini pump
将加巴喷丁或加巴喷丁前药(77)和(82)(以等于10mg加巴喷丁/kg的剂量)溶于适宜的溶剂(例如水、PEG 400等),并将其填充至预称重的Alzet迷你渗透泵装置(模型2001D)(Durect Corp.,Cupertino,CA)。通过在37℃下将填充的Alzet在渗透盐水中浸泡3小时并在4℃下在密封容器中储存过夜而进行预平衡。然后让四只禁食的雄性猎狗口服(大约6.5kg)。动物在每次服药后4小时进食。在48小时内间隔地取血样(1.0mL),并立即对血浆进行处理。将血浆样品冻干,并在-80℃下储存直至使用上述的方法进行分析。两种前药所提供的服药后12小时的加巴喷丁的血浆浓度比在Alzet装置中施用加巴喷丁本身后所见的加巴喷丁的浓度大2倍。此数据进一步证明本发明的化合物可以制成适于有效地持续释放GABA类似物的组合物。Dissolve gabapentin or gabapentin prodrugs (77) and (82) (at a dose equal to 10 mg gabapentin/kg) in a suitable solvent (e.g. water, PEG 400, etc.) and fill it into pre-weighed Alzet® mini osmotic Pump unit (Model 2001D) (Durect Corp., Cupertino, CA). Pre-equilibration was performed by soaking the filled Alzet(R) in permeate saline for 3 hours at 37°C and stored overnight at 4°C in a sealed container. Four fasting male hounds were then administered orally (approximately 6.5 kg). Animals were fed 4 hours after each dose. Blood samples (1.0 mL) were taken at intervals over 48 hours, and plasma was processed immediately. Plasma samples were lyophilized and stored at -80°C until analysis using the method described above. Both prodrugs provided plasma concentrations of gabapentin at 12 hours post-dose that were 2-fold greater than those seen after administration of gabapentin itself in the Alzet(R) device. This data further demonstrates that the compounds of the invention can be formulated into compositions suitable for effective sustained release of GABA analogs.
实施例44Example 44
大鼠结肠内pregabalin或pregabalin前药给药后的pregabalin的Intracolonic pregabalin or pregabalin prodrug administration in rats
吸收 absorb
用pregabalin和pregabalin前药(110)和(112)重复实施例41的方案。在这些前药的每一种进行结肠给药之后,pregabalin的最大血浆浓度(Cmax)以及pregabalin血浆浓度对时间曲线下的面积(AUC)明显大于pregabalin本身结肠给药产生的曲线下面积(>2倍)。The protocol of Example 41 was repeated with pregabalin and pregabalin prodrugs (110) and (112). Following colonic administration of each of these prodrugs, the maximum plasma concentration ( Cmax ) of pregabalin and the area under the pregabalin plasma concentration versus time curve (AUC) were significantly greater than the area under the curve produced by colonic administration of pregabalin itself (> 2 times).
最后,应该注意到存在实施本发明的可选择的方式。因此,本实施方案被认为是例示而不是限定,且本发明不受本文提供的细节限制,而是可在所附的权利要求的范围和等同物之内作修改。Finally, it should be noted that there are alternative ways of implementing the invention. Accordingly, the present embodiments are to be considered as illustrative rather than limiting, and the invention is not to be limited by the details presented herein but may be modified within the scope and equivalents of the appended claims.
全部引入本文引述的所有公开和专利作为参考。All publications and patents cited herein are incorporated by reference in their entirety.
Claims (117)
Applications Claiming Priority (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US29752101P | 2001-06-11 | 2001-06-11 | |
| US60/297,521 | 2001-06-11 | ||
| US29851401P | 2001-06-14 | 2001-06-14 | |
| US60/298,514 | 2001-06-14 | ||
| US36609002P | 2002-03-19 | 2002-03-19 | |
| US60/366,090 | 2002-03-19 | ||
| PCT/US2002/018689 WO2002100347A2 (en) | 2001-06-11 | 2002-06-11 | Prodrugs of gaba analogs, compositions and uses thereof |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2008102144219A Division CN101381339B (en) | 2001-06-11 | 2002-06-11 | Prodrugs of gaba analogs, compositions and uses thereof |
| CN 200810169355 Division CN101434572B (en) | 2001-06-11 | 2002-06-11 | Prodrugs of GABA analogs, compositions and uses thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1753673A true CN1753673A (en) | 2006-03-29 |
| CN1753673B CN1753673B (en) | 2010-09-29 |
Family
ID=34589891
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 200810169355 Expired - Lifetime CN101434572B (en) | 2001-06-11 | 2002-06-11 | Prodrugs of GABA analogs, compositions and uses thereof |
| CN02814572.0A Expired - Fee Related CN1753673B (en) | 2001-06-11 | 2002-06-11 | Prodrugs of GABA analogues, compositions and applications thereof |
| CN2008102144219A Expired - Fee Related CN101381339B (en) | 2001-06-11 | 2002-06-11 | Prodrugs of gaba analogs, compositions and uses thereof |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 200810169355 Expired - Lifetime CN101434572B (en) | 2001-06-11 | 2002-06-11 | Prodrugs of GABA analogs, compositions and uses thereof |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2008102144219A Expired - Fee Related CN101381339B (en) | 2001-06-11 | 2002-06-11 | Prodrugs of gaba analogs, compositions and uses thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (3) | CN101434572B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104271549A (en) * | 2012-05-03 | 2015-01-07 | 帝斯曼知识产权资产管理有限公司 | A new intermediate compound for preparing vitamin B6 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105777586A (en) * | 2016-04-14 | 2016-07-20 | 安徽省逸欣铭医药科技有限公司 | S(+)vigabatrin ester derivative and preparation method and application thereof |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5051448A (en) * | 1984-07-24 | 1991-09-24 | The Mclean Hospital Corporation | GABA esters and GABA analog esters |
| US5827819A (en) * | 1990-11-01 | 1998-10-27 | Oregon Health Sciences University | Covalent polar lipid conjugates with neurologically active compounds for targeting |
| ZA953078B (en) * | 1994-04-28 | 1996-01-05 | Alza Corp | Effective therapy for epilepsies |
| JP2002508361A (en) * | 1997-12-16 | 2002-03-19 | ワーナー−ランバート・カンパニー | ((Cyclo) alkyl-substituted) -γ-aminobutyric acid derivatives (= GABA analogs), their preparation and use in the treatment of neurological diseases |
| GB2365425A (en) * | 2000-08-01 | 2002-02-20 | Parke Davis & Co Ltd | Alkyl amino acid derivatives useful as pharmaceutical agents |
-
2002
- 2002-06-11 CN CN 200810169355 patent/CN101434572B/en not_active Expired - Lifetime
- 2002-06-11 CN CN02814572.0A patent/CN1753673B/en not_active Expired - Fee Related
- 2002-06-11 CN CN2008102144219A patent/CN101381339B/en not_active Expired - Fee Related
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104271549A (en) * | 2012-05-03 | 2015-01-07 | 帝斯曼知识产权资产管理有限公司 | A new intermediate compound for preparing vitamin B6 |
| CN104271549B (en) * | 2012-05-03 | 2017-07-04 | 帝斯曼知识产权资产管理有限公司 | It is a kind of new to prepare vitamin B6Midbody compound |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1753673B (en) | 2010-09-29 |
| CN101381339B (en) | 2012-03-07 |
| CN101434572A (en) | 2009-05-20 |
| CN101381339A (en) | 2009-03-11 |
| CN101434572B (en) | 2013-03-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1305839C (en) | Phenylglycine derivatives | |
| CN1111520C (en) | Matrix metallo-proteinase inhibitor | |
| CN1075062C (en) | Acetic acid derivatives | |
| CN1044249C (en) | Matrix metalloprotease inhibitors | |
| CN1161331C (en) | Hydrazine derivative | |
| CN87107371A (en) | The glutaramide diuretic agents of spiral shell-replacement | |
| CN1196671C (en) | Mono- and di-substituted 3-propyl-γ-aminobutyric acid | |
| CN1316995A (en) | Hydroxy 2-pipecolate hydroxamide acid derivatives as MMP inhititors | |
| CN1741986A (en) | Amide derivatives and their use as 11-beta hydroxysteroid dehydrogenase inhibitors | |
| CN1087904A (en) | Carboxylic acid derivatives, pharmaceutical compositions containing these compounds and processes for their preparation | |
| CN1242014A (en) | Novel heterocyclic amide compounds and medicinal uses thereof | |
| CN1732161A (en) | 1,3-diamino-2-hydroxypropane pro-drug derivatives | |
| CN1688548A (en) | Process for preparing quinolin antibiotic intermediates | |
| CN1040986A (en) | Preparation of cycloalkyl substituted glutaramide derivatives | |
| CN1175953A (en) | Novel dipeptide amidines as thrombin inhibitors | |
| CN1276778A (en) | Biphenyl-5-alkanoic acid derivs. and use thereof | |
| CN1265647A (en) | Acyclic Metalloproteinase Inhibitors | |
| CN1418193A (en) | heterocyclic side chain containing metalloprotease inbibitors | |
| CN86105870A (en) | Renin inhibitors with 5-amino-2,5-disubstituted-4-hydroxyvaleric acid | |
| CN1305454A (en) | Anthranilic acid derivatives as inhibitors of CGMP phosphodiesterase | |
| CN1906164A (en) | Cathepsin cysteine protease inhibitors | |
| CN1217932C (en) | Antibacterial agents | |
| HK1044933B (en) | Branched chain amino acid-dependent aminotranserase inhibitors and their use in the treatment of diabetic retinopathy | |
| CN1080634A (en) | Pentamethylene-and cyclopentene-' beta '-amino acids | |
| CN1291197A (en) | Novel acetamide derivative and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1087628 Country of ref document: HK |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: GR Ref document number: 1087628 Country of ref document: HK |
|
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20100929 Termination date: 20170611 |