CN1756871A - Iridescent fabrics from polyamide yarns - Google Patents
Iridescent fabrics from polyamide yarns Download PDFInfo
- Publication number
- CN1756871A CN1756871A CNA038023032A CN03802303A CN1756871A CN 1756871 A CN1756871 A CN 1756871A CN A038023032 A CNA038023032 A CN A038023032A CN 03802303 A CN03802303 A CN 03802303A CN 1756871 A CN1756871 A CN 1756871A
- Authority
- CN
- China
- Prior art keywords
- yarn
- acid
- dyeable
- nylon
- polymer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/0096—Multicolour dyeing
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P3/00—Special processes of dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form, classified according to the material treated
- D06P3/82—Textiles which contain different kinds of fibres
- D06P3/8204—Textiles which contain different kinds of fibres fibres of different chemical nature
- D06P3/8209—Textiles which contain different kinds of fibres fibres of different chemical nature mixtures of fibres containing amide groups
-
- D—TEXTILES; PAPER
- D01—NATURAL OR MAN-MADE THREADS OR FIBRES; SPINNING
- D01D—MECHANICAL METHODS OR APPARATUS IN THE MANUFACTURE OF ARTIFICIAL FILAMENTS, THREADS, FIBRES, BRISTLES OR RIBBONS
- D01D5/00—Formation of filaments, threads, or the like
- D01D5/253—Formation of filaments, threads, or the like with a non-circular cross section; Spinnerette packs therefor
-
- D—TEXTILES; PAPER
- D03—WEAVING
- D03D—WOVEN FABRICS; METHODS OF WEAVING; LOOMS
- D03D15/00—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used
- D03D15/20—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used characterised by the material of the fibres or filaments constituting the yarns or threads
- D03D15/283—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used characterised by the material of the fibres or filaments constituting the yarns or threads synthetic polymer-based, e.g. polyamide or polyester fibres
-
- D—TEXTILES; PAPER
- D03—WEAVING
- D03D—WOVEN FABRICS; METHODS OF WEAVING; LOOMS
- D03D15/00—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used
- D03D15/50—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used characterised by the properties of the yarns or threads
- D03D15/54—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used characterised by the properties of the yarns or threads coloured
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/004—Dyeing with phototropic dyes; Obtaining camouflage effects
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P3/00—Special processes of dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form, classified according to the material treated
- D06P3/02—Material containing basic nitrogen
- D06P3/04—Material containing basic nitrogen containing amide groups
- D06P3/24—Polyamides; Polyurethanes
- D06P3/241—Polyamides; Polyurethanes using acid dyes
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P3/00—Special processes of dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form, classified according to the material treated
- D06P3/02—Material containing basic nitrogen
- D06P3/04—Material containing basic nitrogen containing amide groups
- D06P3/24—Polyamides; Polyurethanes
- D06P3/242—Polyamides; Polyurethanes using basic dyes
-
- D—TEXTILES; PAPER
- D10—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B2331/00—Fibres made from polymers obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polycondensation products
- D10B2331/02—Fibres made from polymers obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polycondensation products polyamides
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T442/00—Fabric [woven, knitted, or nonwoven textile or cloth, etc.]
- Y10T442/30—Woven fabric [i.e., woven strand or strip material]
Landscapes
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Mechanical Engineering (AREA)
- Coloring (AREA)
- Knitting Of Fabric (AREA)
- Woven Fabrics (AREA)
- Artificial Filaments (AREA)
Abstract
闪光织物包括可阳离子染色的尼龙聚合物纱和可酸染色的尼龙聚合物纱。可酸染色纱具有大于35胺端基(AEG酸)克当量每1000千克聚合物。
The shimmering fabrics include cationically dyeable nylon polymer yarns and acid-dyed nylon polymer yarns. The acid-dyed yarns have a g-equivalent weight of more than 35 amine end groups (AEG acid ) per 1000 kg of polymer.
Description
相关申请的交叉参考Cross References to Related Applications
本申请要求2002年1月23日提交的临时申请No.60/351,023的优先权。This application claims priority to Provisional Application No. 60/351,023, filed January 23,2002.
技术领域technical field
本发明涉及显示虹彩外观的织物。织物包括可阳离子染色的聚酰胺纱和可酸染色的聚酰胺纱。The present invention relates to fabrics exhibiting an iridescent appearance. Fabrics include cationically dyeable polyamide yarns and acid dyeable polyamide yarns.
背景技术Background technique
闪光织物是已知的。这些织物显示从双颜色效果到彩虹颜色的外观。一般情况下闪光织物的颜色由观察角确定。当衣服符合穿用者的身体时,用于服装应用的闪光织物是从多个角度观察的。结果是,对于衣服的观察者多于一种表观颜色占优势。Glitter fabrics are known. These fabrics display looks ranging from two-tone effects to rainbow colors. In general, the color of the glitter fabric is determined by the viewing angle. Glitter fabrics for apparel applications are viewed from multiple angles as the garment conforms to the wearer's body. The result is that more than one apparent color predominates to the observer of the garment.
Kobsa等人的美国专利5,741,590公开了一种闪光织物。Kobsa织物包括具有同心外皮(外套)和芯的单一双组分复丝纱,该复丝纱为圆形横截面。Kobsa双组分长丝的外皮和芯部分为可酸染色尼龙66和可碱染色尼龙66。或者,Kobsa双组分长丝的外皮和芯部分分别为可酸染色尼龙66和可碱染色聚酯(包含2wt%5-磺基间苯二甲酸二甲酯的聚对苯二甲酸乙二醇酯聚合物)。在Kobsa长丝的任一情况下,将外皮和芯部分一起在相同染浴中染色但单独接受不同的染料。此外,Kobsa依赖于双组分长丝纺丝,该纺丝包括复杂和昂贵的双组分喷丝组件。需要这些组件用于加入两种熔融聚合物流和相应地结合物流成适当的几何形状。US Patent 5,741,590 to Kobsa et al. discloses a glittering fabric. Kobsa fabrics comprise a single bicomponent multifilament yarn of circular cross-section with a concentric sheath (jacket) and core. The sheath and core of Kobsa bicomponent filaments are acid dyeable nylon 66 and alkali dyeable nylon 66. Alternatively, the sheath and core of Kobsa bicomponent filaments are respectively acid dyeable nylon 66 and alkali dyeable polyester (polyethylene terephthalate containing 2 wt% 5-dimethyl sulfoisophthalate). ester polymers). In either case of Kobsa filaments, the sheath and core portions are dyed together in the same dyebath but receive different dyes separately. In addition, Kobsa relies on bicomponent filament spinning, which includes complex and expensive bicomponent spin packs. These components are required for joining the two molten polymer streams and accordingly combining the streams into the proper geometry.
Takahashi等人的美国专利6,279,356B1公开了纱线的经编以由干扰获得织物颜色色调和“改进光泽和虹彩”的效果。Takahashi织物是经编针织物且都优选由黑色基础织造纱或颜色为嵌入织造纱颜色的互补色的基础织造纱组成。纱可以是天然纤维、合成纤维如聚酯或尼龙、或半合成纤维如人造丝或乙酸酯。用于达到这些颜色效果的Takahashi方法要求在纺丝之前进行多个聚合物着色步骤以及织物的常规染色步骤。染色的例子包括基础织造用聚酯长丝的阳离子染料的使用,及碱性染料和分散性染料的使用,两者共同用于分别染色嵌入织造用聚酯长丝。在任一情况下,使用阳离子染料。然而,由于可利用的阳离子染料调色板的有限范围和标准尼龙固有阳离子染色性的缺乏,阳离子染料很少用于尼龙纱服装应用。US Patent 6,279,356 B1 to Takahashi et al. discloses warp knitting of yarns to obtain fabric color shades and "improved luster and iridescence" effects from disturbance. Takahashi fabrics are warp knitted fabrics and are preferably composed of either a black base weave yarn or a base weave yarn of a color complementary to the color of the embedded weave yarn. Yarns may be natural fibers, synthetic fibers such as polyester or nylon, or semi-synthetic fibers such as rayon or acetate. The Takahashi method used to achieve these color effects requires multiple steps of polymer coloring as well as conventional dyeing of the fabric prior to spinning. Examples of dyeing include the use of cationic dyes for base weaving polyester filaments, and the use of basic dyes and disperse dyes, both of which are used in combination to dye polyester filaments for embedded weaving, respectively. In either case, cationic dyes are used. However, cationic dyes are rarely used in nylon yarn apparel applications due to the limited range of available cationic dye palettes and the lack of inherent cationic dyeability of standard nylons.
用于在纺织品材料中达到虹彩的其它已知方法由Asano等人的US专利6,326,094B1公开。Asano公开了从不同有机聚合物的层压层制备的复合交替层压长丝结构,所述层压层具有不同的折射率并且提供可见光的反射和干扰或紫外和红外射线的反射。这些效果可用于纺织品以依赖于观察角产生色调和强度变化的颜色。Asano等人的方法要求昂贵和精密的多组分喷丝组件和多个聚合物流,以制备不同有机聚合物层的交替层压,得到此类型的层压合成长丝。Another known method for achieving iridescence in textile materials is disclosed by US Patent 6,326,094 B1 to Asano et al. Asano discloses composite alternating laminated filament structures prepared from laminated layers of different organic polymers having different refractive indices and providing reflection and interference of visible light or reflection of ultraviolet and infrared rays. These effects can be used on textiles to produce colors that vary in hue and intensity depending on the viewing angle. The method of Asano et al. requires expensive and sophisticated multicomponent spin packs and multiple polymer streams to produce alternating laminations of different organic polymer layers to obtain laminated synthetic filaments of this type.
从可酸染色和可阳离子染色的合成聚合物纤维制备已知的闪光织物。一般情况下,采用例如由可酸染色和可阳离子染色聚酯的圆形横截面长丝组成的复丝纱达到这类已知织物中的虹彩。此外,可以采用相同的方式使用可酸染色的尼龙与可阳离子染色的尼龙,其中一种纱线具有添纱到织物面上的特别亮光泽。然而,基本所有尼龙织物的这种现有技术闪光织物均采用标准酸性染料尼龙纱。这样的标准酸性染料尼龙纱不特别良好地保持酸性染料,它允许更多的酸性染料沾染可阳离子染色的纱。因此,在单一染浴中染色这些现有技术织物要求染色辅助化学品以防止两类染料之间的反应。Known iridescent fabrics are produced from acid-dyeable and cationically-dyeable synthetic polymer fibers. In general, the iridescence in known fabrics of this type is achieved using, for example, multifilament yarns composed of circular cross-section filaments of acid-dyeable and cationically-dyeable polyesters. Furthermore, acid-dyeable nylon can be used in the same way as cation-dyeable nylon, one of the yarns having a particularly bright sheen plated onto the fabric face. However, such prior art glitter fabrics for virtually all nylon fabrics use standard acid dyed nylon yarns. Such standard acid dye nylon yarns do not hold acid dye particularly well, which allows more acid dye to stain the cationically dyeable yarn. Therefore, dyeing these prior art fabrics in a single dyebath requires dyeing auxiliary chemicals to prevent reactions between the two types of dyes.
发明概述Summary of the invention
本发明的闪光织物包括可深度酸染色的尼龙纱,其中使用提高水平的胺端基获得深度染色性能。这使得能够在单一染浴中染色织物而不使用染色辅助化学品以防止酸性染料和阳离子染料之间的反应。在此使用的深度酸染色尼龙聚合物纱与酸性染料在染浴中快速且完全地反应,使酸性染料不可用于与存在的任何其它染料反应。结果是,如果不能消除的话,最小化与阳离子染色纱的交叉沾染并降低使用染料辅助化学品的需要。The iridescent fabrics of the present invention comprise deep acid dyeable nylon yarns in which deep dyeing performance is achieved using increased levels of amine end groups. This enables fabrics to be dyed in a single dyebath without the use of dyeing auxiliary chemicals to prevent reactions between acid and cationic dyes. The deeply acid-dyed nylon polymer yarn used here reacts with acid dyes quickly and completely in the dyebath, making the acid dyes unavailable to react with any other dyes present. The result is to minimize, if not eliminate, cross-contamination with cationic dyed yarns and reduce the need to use dye aux chemicals.
因此,本发明允许使用阳离子染料与尼龙,该使用如上所述是很少进行的。因此,本发明的闪光织物基本都是尼龙,选择性地具有一部分斯潘德克斯弹性体纤维。本发明的织物非常适于要求基本均为尼龙织物的纺织品应用,如连裤袜和许多新的无缝衣服。Thus, the present invention allows the use of cationic dyes with nylon, which use is rarely performed as described above. Thus, the glitter fabric of the present invention is essentially all nylon, optionally with a portion of spandex. The fabrics of the present invention are well suited for textile applications requiring substantially all nylon fabrics, such as pantyhose and many new seamless garments.
另外,由于深度酸染色尼龙聚合物纱的使用最小化与阳离子染色纱的交叉沾污,可以使用酸性染料和阳离子染料在单一浴中染色本发明的闪光织物。因此,本发明允许在单一步骤中染色织物,而不依赖多步骤纱染色。In addition, the iridescent fabrics of the present invention can be dyed in a single bath using acid dyes and cationic dyes due to the use of deeply acid-dyed nylon polymer yarns to minimize cross contamination with cationic dyed yarns. Thus, the present invention allows fabrics to be dyed in a single step rather than relying on multi-step yarn dyeing.
此外,制备本发明闪光织物的方法基于常规尼龙熔体纺丝技术,并不依赖复杂的多组分喷丝组件,或本体着色聚合物。Furthermore, the method of making the iridescent fabrics of the present invention is based on conventional nylon melt spinning technology and does not rely on complex multi-component spin packs, or mass colored polymers.
附图简述Brief description of the drawings
图1A是从用于制备本发明织物的纱中单一聚酰胺纤维长轴法向取的三叶形纤维的横截面视图。Figure 1A is a cross-sectional view of a trilobal fiber taken normal to the long axis of a single polyamide fiber in the yarn used to make the fabric of the present invention.
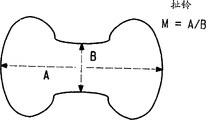
图1B是从用于制备本发明织物的纱中单一聚酰胺纤维长轴法向取的扯铃形纤维的横截面视图。Figure 1B is a cross-sectional view of a diabolo-shaped fiber taken normal to the long axis of a single polyamide fiber in the yarn used to make the fabric of the present invention.
图2是本发明织物一部分的透视图。Figure 2 is a perspective view of a portion of the fabric of the present invention.
优选实施方案的详细描述DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENT
本发明是闪光纺织品织物。优选的织物是针织袜和无缝针织物。一般在图2中10处显示本发明的织物。织物包括两种聚酰胺复丝纱。在织物构造之后结合纱线。本发明的闪光织物是两面的,即,单一组分纱在一个织物面上占优势。因此,构造闪光织物使得一种纱基本在织物的第一面20上占优势而第二种纱基本在织物的第二面30上占优势,如图2所示。由本发明织物显示的闪光外观可以是反差色或互补色的一种。The present invention is a glitter textile fabric. Preferred fabrics are hosiery and seamless knits. A fabric of the present invention is shown generally at 10 in FIG. 2 . The fabric consisted of two polyamide multifilament yarns. The yarns are combined after fabric construction. The glitter fabrics of the present invention are two-sided, that is, a single component yarn predominates on one side of the fabric. Thus, the iridescent fabric is constructed such that one yarn predominates substantially on the
复丝聚酰胺纱是可阳离子染色(也称为可碱染色)的尼龙聚合物纱和可酸染色的尼龙聚合物纱。纱线的染色性和对通常用于尼龙的不同类别染料的接受性不同。已知尼龙或脂族聚酰胺纱可强烈地被所谓的酸性染料染色,其中在重复酰胺基团之间的至少约85%聚合物链连接是脂族基团,在此通常的尼龙聚合物称为可酸染色的。可以通过在聚合物中引入磺酸酯基团而使尼龙变成耐酸性染料的,如在US专利5,164,261中所述。在US 5,164,261中,教导聚酰胺与少量,约1-4wt%5-磺基间苯二甲酸钠盐的共聚。由带有二羧酸的磺酸酯如此改性的尼龙聚合物用于制备在此称为可碱染色、可阳离子染色、或简单cat-染料纱的纱。此名称是由于耐酸性染料性的化学基团,该耐酸性染料性在纱上带来可阳离子染色性能。阳离子染料已知用于在某些服装尼龙纱中的应用,但与酸性染料相比更少使用。Multifilament polyamide yarns are cationically dyeable (also known as alkali dyeable) nylon polymer yarns and acid dyeable nylon polymer yarns. Yarns differ in their dyeability and receptivity to different classes of dyes commonly used in nylon. It is known that nylon or aliphatic polyamide yarns can be dyed intensively with so-called acid dyes, in which at least about 85% of the polymer chain linkages between repeating amide groups are aliphatic groups, where the usual nylon polymers are called It is acid-stainable. Nylon can be made resistant to acid dyes by introducing sulfonate groups into the polymer, as described in US Patent 5,164,261. In US 5,164,261, the copolymerization of polyamides with small amounts, about 1-4 wt%, of 5-sulfoisophthalic acid sodium salt is taught. Nylon polymers so modified with sulfonate esters of dicarboxylic acids are used to make yarns referred to herein as base-dyeable, cation-dyeable, or simply cat-dye yarns. The name is due to the chemical group for the acid dye resistance that imparts cationic dyeability on the yarn. Cationic dyes are known for use in some apparel nylon yarn applications, but are less commonly used than acid dyes.
可以加工成可酸染色和可碱染色聚酰胺并且也可采用本发明横截面长丝形状熔体纺丝的合适合成聚合物纤维包括:尼龙66(聚己二酰己二胺)、尼龙6(聚己酰胺(polycaproamide))、尼龙7(聚庚酰胺(polyenanthamide))、尼龙612(聚十二烷二酰己二胺)、尼龙11、尼龙12及尼龙66和尼龙6的共聚酰胺,如来自1,6-己二胺、ε-己内酰胺和己二酸的聚合物,和从己二酸、1,6-己二胺和间苯二甲酸制备的聚合物,或从己二酸、1,6-己二胺和2-甲基-1,5-戊二胺或2-乙基-1,4-丁二胺制备的聚合物;包含至多15wt%,如0.5-15wt%聚间苯二甲酰1,6-己二胺或聚己二酰(2-甲基)1,5-戊二胺或聚己二酰(2-乙基)1,4-丁二胺的尼龙66的共聚酰胺和包含至多15wt%,如0.5-15wt%尼龙66的尼龙6的共聚酰胺。Suitable synthetic polymer fibers that can be processed into acid- and alkali-dyeable polyamides and that can also be melt spun in the cross-sectional filament shape of the present invention include: Nylon 66 (polyhexamethylene adipamide), Nylon 6 ( Polycaproamide), nylon 7 (polyenanthamide), nylon 612 (polyhexamethylene dodecyl diamide), nylon 11, nylon 12 and copolyamides of nylon 66 and nylon 6, such as from Polymers of 1,6-hexanediamine, ε-caprolactam and adipic acid, and polymers prepared from adipic acid, 1,6-hexanediamine and isophthalic acid, or from adipic acid, 1,6 - polymers prepared from hexamethylenediamine and 2-methyl-1,5-pentanediamine or 2-ethyl-1,4-butylenediamine; containing up to 15 wt%, such as 0.5-15 wt% polymphthalylene Copolyamide of Nylon 66 with 1,6-hexamethylenediamine or poly(2-methyl)pentanediamine or poly(2-ethyl)butanediamine adipylamide and copolyamides of nylon 6 comprising up to 15 wt%, such as 0.5-15 wt% nylon 66.
典型地,在上述段落中描述的尼龙聚合物和共聚酰胺是固有可酸染色的。这些聚合物中游离胺端基(AEG)的数目是至少25克当量每1000千克尼龙聚合物。为使聚合物更深度酸染色,需要提高水平的游离胺端基。可酸染色纱的优选和提高的AEG水平大于35克当量每1000千克尼龙聚合物。优选是60-130克当量每1000千克尼龙聚合物。至多约130克当量每1000千克聚合物的胺端基浓度可用于可酸染色的聚合物组分,以达到织物外观性能。更特别地,对于可酸染色的纱聚合物,最优选约70克当量每1000千克尼龙聚合物的AEG水平。Typically, the nylon polymers and copolyamides described in the preceding paragraphs are inherently acid dyeable. The number of free amine end groups (AEG) in these polymers is at least 25 gram equivalents per 1000 kg of nylon polymer. For more intense acid staining of the polymer, increased levels of free amine end groups are required. Preferred and enhanced AEG levels for acid dyeable yarns are greater than 35 gram equivalents per 1000 kilograms of nylon polymer. Preferably it is 60-130 gram equivalent per 1000 kg of nylon polymer. Amine end group concentrations of up to about 130 gram equivalents per 1000 kilograms of polymer may be used in the acid dyeable polymer component to achieve fabric appearance properties. More particularly, for acid dyeable yarn polymers, an AEG level of about 70 gram equivalents per 1000 kilograms of nylon polymer is most preferred.
采用在聚合物中共聚的阳离子染料改性剂制备碱染色纱聚合物。Windley的美国专利5,164,261描述了这种阳离子染料改性的聚酰胺的制备。在本发明中,优选在聚合期间采用0.5-4%优选的阳离子染料改性剂5-磺基间苯二甲酸的二甲酯改性聚合物。典型地,在高压釜中使用本领域已知的标准聚合程序,将称重数量的5-磺基间苯二甲酸二甲酯钠盐与已知数量的聚酰胺前体盐结合。聚合物中存在的阳离子染料改性剂的更优选数量是约0.75-约3wt%,由聚合物的总硫分析确定。将阳离子染料改性剂的数量报导为当量磺酰酯基团。优选的磺酰酯基团浓度是至少15克当量每1000千克聚合物到约150克当量每1000千克聚合物,优选大于30克当量每1000千克聚合物。优选闪光织物的可阳离子染色尼龙纱的胺端基浓度不大于40克当量每1000千克聚合物。优选在磺酸酯端基(Scat)和胺端基(AEGcat)之间的浓度差(ρ)大于或等于零。Base-dyed yarn polymers were prepared using cationic dye modifiers copolymerized in the polymer. US Patent 5,164,261 to Windley describes the preparation of such cationic dye-modified polyamides. In the present invention, it is preferred to modify the polymer with 0.5-4% of the preferred cationic dye modifier, dimethyl ester of 5-sulfoisophthalic acid, during polymerization. Typically, a weighed amount of dimethyl 5-sulfoisophthalate sodium salt is combined with a known amount of polyamide precursor salt in an autoclave using standard polymerization procedures known in the art. A more preferred amount of cationic dye modifier present in the polymer is from about 0.75 to about 3 weight percent, as determined by total sulfur analysis of the polymer. The amount of cationic dye modifier is reported as equivalent sulfonyl ester groups. A preferred concentration of sulfonyl ester groups is at least 15 gram equivalents per 1000 kg polymer to about 150 gram equivalents per 1000 kg polymer, preferably greater than 30 gram equivalents per 1000 kg polymer. It is preferred that the cationically dyeable nylon yarn of the iridescent fabric has an amine end group concentration of not greater than 40 gram equivalents per 1000 kilograms of polymer. Preferably the concentration difference (ρ) between the sulfonate end groups (S cat ) and the amine end groups (AEG cat ) is greater than or equal to zero.
ρ=Scat-AEGcat≥0 公式1ρ=S cat -AEG cat ≥0
每种纱包括横截面形状为圆形、非圆形、三叶或扯铃的长丝。在图1A中显示三叶形状,在图1B中显示扯铃形状。三叶形状的进一步特征在于其改性比M为1.5-4。M比例定义为接触横截面极端的最大旁切圆半径A除以最小内切圆半径B,如图1A所示。扯铃横截面的M比例是1.5-6并定义为横截面最长长度A除以最小尺寸B,如图1B所示。Each yarn comprises filaments with circular, non-circular, trilobal, or diabolo cross-sectional shapes. A trilobal shape is shown in FIG. 1A and a diabolo shape is shown in FIG. 1B . The trilobal shape is further characterized in that its modification ratio M is 1.5-4. The M ratio is defined as the largest circumscribed circle radius A divided by the smallest inscribed circle radius B in contact with the extremities of the cross section, as shown in Figure 1A. The M ratio of a diabolo cross-section is 1.5-6 and is defined as the longest length A of the cross-section divided by the smallest dimension B, as shown in Figure 1B.
根据本发明的优选实施方案,可阳离子染色或可酸染色尼龙纱具有特别亮的光泽,采用不大于0.1wt%的聚合物去光剂水平即可达到该光泽。此光亮、更有光泽的纱在织物的第一面上占优势。优选让光亮、高光泽尼龙聚合物纱包括具有非圆形横截面形状的长丝。更优选是三叶和扯铃横截面形状。其它的更无光泽的纱可具有任何横截面形状。具有暗光泽的聚酰胺纱是有用的,但不是必须改性以达到本发明的闪光织物效果。无光聚合物纱的去光剂水平等于或大于约0.8wt%,优选为约1.5%。二氧化钛是用于在此公开的任何聚酰胺聚合物的更优选去光剂。聚酰胺聚合物的其它去光剂,如硫化锌也是合适的。According to a preferred embodiment of the present invention, the cationically dyeable or acid dyeable nylon yarns have an exceptionally bright luster, which can be achieved with a polymeric delustering agent level of no greater than 0.1 wt%. This brighter, more lustrous yarn predominates on the first side of the fabric. Preferably, the bright, high gloss nylon polymer yarn comprises filaments having a non-circular cross-sectional shape. More preferred are trilobal and diabolo cross-sectional shapes. Other more matte yarns can have any cross-sectional shape. Polyamide yarns with a dull sheen are useful, but not necessarily modified, to achieve the shiny fabric effect of the present invention. The matte polymer yarn has a matting agent level equal to or greater than about 0.8 wt%, preferably about 1.5%. Titanium dioxide is a more preferred matting agent for any of the polyamide polymers disclosed herein. Other matting agents for polyamide polymers, such as zinc sulfide, are also suitable.
如本领域已知的那样,由添纱多纱的能力达到织物的双侧面或分层效果。提供添纱多纱的能力的市售无缝针织机由Lonati S.p.a.(意大利)制造,该公司生产无缝针织机的SANTONI单和双针织版本。无缝针织机也可购自San Giacomo和Monarch,如技术人员已知的那样。除双侧面无缝针织构造以外,可以使用常规两侧面袜针织构造技术或任何其它双侧面织物编织、经编、圆筒形针织或平底板针织工艺是制备本发明织物的有用方式。The double sided or layered effect of the fabric is achieved from the ability to plate multiple yarns, as is known in the art. A commercially available seamless knitting machine offering the ability to plate multiple yarns is manufactured by Lonati S.p.a. (Italy), which manufactures SANTONI single and double knit versions of the seamless knitting machine. Seamless knitting machines are also commercially available from San Giacomo and Monarch, as known to the skilled person. In addition to double sided seamless knit construction, conventional two sided hosiery knit construction techniques or any other double sided fabric knitting, warp knitting, circular knitting or flat knitting process can be useful ways of making the fabrics of the present invention.
可以由制备FDY(全拉伸纱)、POY(部分取向纱)、和LOY(低取向纱)的已知方法制备本发明的纱。在FDY(全拉伸纱)的情况下,纺丝机上的内联加工(in-line processing)由如下操作组成:在成套导丝辊(进料辊)周围进行几次往复,往复的数目足以防止这些辊上的滑移,然后将纱通过另-套辊(拉伸辊),该拉伸辊在足够速度下旋转以拉伸预定数量(拉伸比)的纱,和最后热定形并在4800m/min速度下卷绕之前采用蒸汽箱松弛纱。选择性地可以使用另外的热定形方法,如受热辊,和可以在拉伸辊和卷绕机之间引入另外成套导丝辊以控制张力,同时定形或松弛纱。任选在最后卷绕步骤之前也可以施加纺丝整理的第二次应用,和/或另外的交织。The yarns of the present invention can be prepared by known methods of making FDY (fully drawn yarn), POY (partially oriented yarn), and LOY (lowly oriented yarn). In the case of FDY (fully drawn yarn), the in-line processing on the spinning frame consists of several reciprocations around the set of godet rolls (feed rolls), the number of reciprocations being sufficient Preventing slippage on these rolls, the yarn is then passed through another set of rolls (draw rolls), which rotate at sufficient speed to draw a predetermined amount (draw ratio) of the yarn, and finally heat-set and placed on A steam box was used to relax the yarn before winding at a speed of 4800 m/min. Optionally, additional heat setting methods may be used, such as heated rolls, and additional sets of godet rolls may be introduced between the draw rolls and winder to control tension while setting or relaxing the yarn. Optionally, a second application of spin finish, and/or additional interlacing may also be applied before the final winding step.
在POY的情况下,另外的内联加工仅由如下操作组成:在两个以相同速度旋转的导丝辊上制备S-吊线,然后将纱送到高速卷绕机,在此情况下该卷绕机在4800m/min下运转。S-吊线的使用有益于控制张力,但不是必须的。这种POY可以直接用作编织或针织的扁丝,或用作变形的原料。In the case of POY, the additional inline processing consists only of the preparation of S-hangers on two godet rolls rotating at the same The winding machine runs at 4800m/min. The use of S-hanging wires is beneficial for tension control, but is not required. This POY can be directly used as flat yarn for weaving or knitting, or as a raw material for deformation.
在LOY的情况下,纺丝程序非常相似于POY,区别在于使用1000m/min或以下的卷绕速度。这些纱要求通过第二阶段进一步加工,如在常规拉伸加捻或拉伸卷绕机上。In the case of LOY, the spinning procedure is very similar to POY, with the difference that a winding speed of 1000 m/min or less is used. These yarns require further processing through a second stage, such as on a conventional draw twist or draw winder.
使用上述的已知方法,任一聚合物类型的可酸染色和可碱染色的本发明纱的生产可遵循相同的纺丝程序。首先,可以将合适类型的聚酰胺颗粒加入到计量设备中,并可以由计量泵将熔融聚合物送到过滤器组件,通过包含选择形状的毛细孔的喷丝头板挤出,以在纺丝温度下得到所需的长丝横截面。这些横截面形状可包括圆形、非圆形、三叶和扯铃。纺丝温度可以为270℃-300℃。可以将从喷丝头板出现的长丝束由调节的骤冷空气冷却,采用纺丝整理剂(油/水乳液)处理,任选进行交织,送入在两个以相同速度旋转的导丝辊上的“S-吊线”张力控制结构,然后送到纱线高速卷绕机,该卷绕机在4800米每分钟下卷绕。这样制备的POY可任选直接用作编织或针织的扁丝,或用作拉伸变形的原料。The production of acid-dyeable and alkali-dyeable yarns of the invention of either polymer type may follow the same spinning procedure using the known methods described above. First, a suitable type of polyamide pellets can be fed into a metering device, and a metering pump can send the molten polymer to a filter assembly to be extruded through a spinneret plate containing capillary holes of a chosen shape to temperature to obtain the desired filament cross-section. These cross-sectional shapes may include circular, non-circular, trilobal, and diabolo. The spinning temperature can be 270°C-300°C. The bundle of filaments emerging from the spinneret plate can be cooled by conditioned quench air, treated with a spin finish (oil/water emulsion), optionally interlaced, fed into two guide wires rotating at the same speed The "S-hanging wire" tension control structure on the roll is then sent to the yarn high speed winder, which winds at 4800 meters per minute. The POY thus produced can optionally be used directly as woven or knitted flat yarn, or as a raw material for stretch texturing.
此外根据本发明,提供制备闪光纺织品织物的方法。该方法包括在单一染浴中染色包括如下物质的织物:可阳离子染色的尼龙聚合物纱和可酸染色的尼龙聚合物纱。在染浴中存在至少一种酸性染料和至少一种阳离子染料。如以上对于织物所述,用于制备本发明长丝的酸染色聚合物的优选和提高的AEG水平大于35克当量每1000千克尼龙聚合物。优选的是60-130克当量每1000千克尼龙聚合物。至多约130克当量每1000千克聚合物的胺端基浓度可用于可酸染色的聚合物组分,以达到织物外观性能。更特别地,对于可酸染色的纱聚合物,最优选约70克当量每1000千克尼龙聚合物的AEG水平。此提高的胺端基水平使得能够在单一染浴中染色织物而不使用染色辅助化学品,以防止酸性染料和阳离子染料之间的反应。Further according to the present invention, there is provided a method of preparing a iridescent textile fabric. The method includes dyeing a fabric comprising: cationically dyeable nylon polymer yarn and acid dyeable nylon polymer yarn in a single dyebath. At least one acid dye and at least one cationic dye are present in the dyebath. As noted above for fabrics, the preferred and enhanced AEG levels of the acid dyed polymers used to make the filaments of the present invention are greater than 35 gram equivalents per 1000 kilograms of nylon polymer. Preferred is 60-130 gram equivalents per 1000 kg of nylon polymer. Amine end group concentrations of up to about 130 gram equivalents per 1000 kilograms of polymer may be used in the acid dyeable polymer component to achieve fabric appearance properties. More particularly, for acid dyeable yarn polymers, an AEG level of about 70 gram equivalents per 1000 kilograms of nylon polymer is most preferred. This increased level of amine end groups enables the dyeing of fabrics in a single dyebath without the use of dyeing auxiliary chemicals to prevent reactions between acid and cationic dyes.
测试方法Test Methods
尼龙聚合物的相对粘度(RV)可以根据ASTM D789-86测量并定义为8.4wt%聚合物在90%甲酸和10%水的溶液中于25℃下的粘度与以相同粘度单位在25℃下测量的甲酸水溶液本身的粘度的比例。The relative viscosity (RV) of nylon polymers can be measured according to ASTM D789-86 and is defined as the viscosity of 8.4 wt% polymer in a solution of 90% formic acid and 10% water at 25°C compared to the viscosity at 25°C in the same viscosity units Measure the viscosity of the formic acid aqueous solution itself.
尼龙聚合物胺端基(AEG)浓度和聚合物羰基端基浓度由滴定方法测量,如在“工业化学分析百科全书(Encyclopedia of IndustrialChemical Analysis)”17卷293-294页(John Wiley & Sons Inc.,1973)上所述。一般情况下,测定AEG浓度的方法包括取称重的聚合物、纱或织物样品;根据预计的胺端基水平为1-2克并在50ml苯酚和甲醇混合物(8∶2体积比)中溶解此样品。过滤此溶液以除去去光剂和不溶物。将此上清液补充到100ml总体积并采用由基本标准碱标准化的高氯酸滴定。对电位滴定或酸指示剂终点记录滴定中消耗的标准化高氯酸的数量,如本领域已知的那样。高氯酸的此体积与所取样品的胺端基重量浓度有关。以克当量每106克聚酰胺报导AEG水平。Nylon polymer amine end group (AEG) concentration and polymer carbonyl end group concentration were measured by titration methods as described in "Encyclopedia of Industrial Chemical Analysis" vol. 17 pp. 293-294 (John Wiley & Sons Inc. , 1973) above. In general, the method for determining the concentration of AEG involves taking a weighed sample of polymer, yarn or fabric; 1-2 grams based on the expected amine end group level and dissolving it in 50 ml of a mixture of phenol and methanol (8:2 by volume) This sample. This solution was filtered to remove the deluster and insolubles. This supernatant was made up to a total volume of 100 ml and titrated with perchloric acid normalized to basic standard base. The amount of normalized perchloric acid consumed in the titration is recorded for potentiometric or acid indicator endpoints, as known in the art. This volume of perchloric acid is related to the amine end group weight concentration of the sample taken. AEG levels are reported in gram equivalents per 106 grams of polyamide.
按照相似于AEG测定的规定,由采用标准化碱氢氧化物的滴定测定游离羧酸基团。与聚酰胺样品重量一起,消耗的标准化碱氢氧化物数量与聚合物中的羧基端基数目有关。以克当量每106克聚酰胺报导羧端基。Free carboxylic acid groups were determined by titration with standardized alkali hydroxides following a protocol similar to the AEG determination. Along with the polyamide sample weight, the normalized amount of alkali hydroxide consumed is related to the number of carboxyl end groups in the polymer. Carboxyl end groups are reported in gram equivalents per 106 grams of polyamide.
通过本领域技术人员已知的分析方法,测定用于本发明的可碱染色聚合物中磺酸酯基团的浓度。一般情况下,将称重的样品取得并溶于聚酰胺的合适溶剂体系中。由本领域已知方法从在氧化之后的总硫酸盐浓度确定总硫含量。或者,使用校准标准物的x射线荧光方法适用于总硫分析。假定总硫完全源于聚合期间加入的磺酸酯基团。The concentration of sulfonate groups in the alkali-dyeable polymers used in the invention is determined by analytical methods known to those skilled in the art. Typically, weighed samples are taken and dissolved in a suitable solvent system for the polyamide. The total sulfur content is determined from the total sulfate concentration after oxidation by methods known in the art. Alternatively, an x-ray fluorescence method using calibration standards is suitable for total sulfur analysis. The total sulfur was assumed to be entirely derived from the sulfonate groups added during polymerization.
实施例Example
实施例1-部分AExample 1 - Part A
从改性以提供阳离子染色性的聚合物制备具有亮光泽、三叶横截面形状的第一尼龙复丝平(未变形)纱。总纱分特是22并且每个纱有9个长丝。从包含1.5wt%作为阳离子染料改性剂的5-磺基间苯二甲酸的尼龙66聚合物(聚己二酰己二胺)纺丝此纱线。此聚合物的甲酸RV为31.5,二氧化钛去光剂含量为0.02wt%,胺端基浓度为42克当量每100Okg聚合物,并且磺酸酯基团浓度为55克当量每1000kg聚合物。在此对于可阳离子染色纱线的磺酸酯端基和胺端基之间的差值由公式1给出并且等于下式:A first nylon multifilament flat (untextured) yarn having a glossy, trilobal cross-sectional shape is prepared from a polymer modified to provide cationic dyeability. The total yarn decitex is 22 and each yarn has 9 filaments. This yarn was spun from a nylon 66 polymer (polyhexamethylene adipamide) containing 1.5 wt% of 5-sulfoisophthalic acid as a cationic dye modifier. This polymer had a formic acid RV of 31.5, a titanium dioxide delustering agent content of 0.02 wt%, an amine end group concentration of 42 gram equivalents per 1000 kg polymer, and a sulfonate group concentration of 55 gram equivalents per 1000 kg polymer. Here the difference between the sulfonate end groups and the amine end groups for cationically dyeable yarns is given by
ρ=Scat-AEGcat=55-42=13。ρ= Scat - AEGcat =55-42=13.
将此聚合物熔融并通过保持在279.5℃具有多个三叶毛细管的喷丝头板挤出。将挤出的长丝在空气流中冷却,收敛成纱线并采用以已知方式施加的油/水乳液整理。使此纱首先与进料辊接触,然后送到拉伸辊装置。拉伸纱线1.5倍。在4800米每分钟的速度下卷绕拉伸的纱。容易由阳离子染料染色以此方式制备的此尼龙纱。相同的纱基本不受通常酸性染料影响。此纱的韧度是48cN/特,断裂伸长率是36.5%,沸水收缩率是7.1%。三叶横截面的改性比(M)是1.56,是以旁切圆和内切圆直径的比例测量的,如图1A所示。This polymer was melted and extruded through a spinneret plate with multiple three-lobe capillaries maintained at 279.5°C. The extruded filaments are cooled in a stream of air, converged into yarns and finished with an oil/water emulsion applied in a known manner. The yarn is first brought into contact with feed rolls and then sent to a draw roll arrangement. Stretch the yarn 1.5 times. The stretched yarn was wound at a speed of 4800 meters per minute. The nylon yarn prepared in this way is easily dyed by cationic dyes. The same yarn is largely unaffected by usual acid dyes. The tenacity of this yarn was 48 cN/tex, the elongation at break was 36.5%, and the shrinkage in boiling water was 7.1%. The modification ratio (M) of the trefoil cross-section was 1.56, measured as the ratio of the diameters of the circumscribed and inscribed circles, as shown in Figure 1A.
第二纱线是尼龙覆盖的DuPont LYCRA牌斯潘德克斯。在每米1600捻数下以标准工业方法单覆盖此22分特LYCRA斯潘德克斯。尼龙组分是26分特14长丝深度酸染色POY,在单独的步骤中拉伸它以得到22分特平(未变形)纱。单个长丝横截面是圆形的。此深酸染料POY是来自40RV(甲酸)的尼龙66聚合物。由已知聚合方法将胺端基浓度(AEG酸)提高到70克当量每1000千克聚合物并且二氧化钛去光剂含量是1.5wt%。覆盖的纱具有由LYCRA斯潘德克斯赋予的拉伸性能和由表面上可见的尼龙组分确定的外观。此尼龙纱容易由标准酸性染料染色并且不被阳离子染料沾污。The second yarn was DuPont LYCRA(R) brand spandex covered in nylon. The 22 dtex LYCRA(R) spandex was single covered by standard industry methods at 1600 twists per meter. The nylon component was 26 decitex 14 filament deep acid dyed POY which was drawn in a separate step to give 22 decitex (untextured) yarn. Individual filaments are circular in cross-section. This deep acid dye POY is a nylon 66 polymer from 40RV (formic acid). The amine end group concentration (AEG acid) was increased to 70 gram equivalents per 1000 kg polymer by known polymerization methods and the titanium dioxide delustering agent content was 1.5 wt%. The covered yarn has stretch properties imparted by LYCRA(R) spandex and an appearance defined by the apparent nylon component. This nylon yarn is easily dyed by standard acid dyes and is not stained by cationic dyes.
在交替构造过程中,在400针LANATI404针织机上选择1×1针织第一尼龙纱和第二(覆盖)纱,以形成连裤袜。结构是一个交替过程,22f14覆盖的扁平长丝纱在一个进料中而亮22f9扁平可阳离子染色纱在另一个进料中。设计是在LYCRA斯潘德克斯覆盖纱上的1×1编织集圈选择。在此构造中,亮22f9纱添纱到衣服的前部。In an alternate construction process, the first nylon yarn and the second (cover) yarn were selected 1 x 1 knitted on a 400 gauge LANATI 404 knitting machine to form the pantyhose. The construction was an alternating process with the 22f14 covered flat filament yarn in one feed and the bright 22f9 flat cationic dyeable yarn in the other feed. The design is a 1 x 1 braid tuck option on LYCRA(R) spandex cover yarn. In this construction, bright 22f9 yarn is plated to the front of the garment.
然后在单一染浴中,使用两种单独的染料,酸性染料和选择用于对比颜色的其它阳离子染料染色衣服。阳离子染料是亮红,购自CIBA的Maxilon Red 3GLN,相对针织衣服中可阳离子染色尼龙组分纱为0.4wt%。酸性染料是亮绿松石蓝酸性染料,购自BASF的Acidolbr.Blue M-5G,相对针织衣服中可酸染色尼龙组分纱为0.4wt%。染色机是使用1升不锈钢密闭烧杯的Roaches Pyrotec 2002(RoachesInternational LTD)。在开始时,染浴和织物在30℃,阳离子染料与相对织物总重量为5.0%的硫酸钠(芒硝)和0.1%Tinegal MR(购自CIBA的碱性染料的阳离子染料阻滞剂)一起加入。在28∶1的染料液对织物物品比例下,将水浴调节到pH6.0。将温度以1.0℃每分钟升高到60℃和保持10分钟。将返回35℃的冷却速率设定在2.5℃每分钟。这是为防止阳离子染料耗尽到可阳离子染色纱上。向此相同染浴中,加入阴离子染料,与在物品总重量基础上的1.0%Sandogen NH(购自Clariant的阴离子染料的染料阻滞剂)混合。将染浴温度以1.0℃每分钟升高到98℃并在此温度下保持45分钟。以7.0℃每分钟将染浴冷却至50℃。将此针织物样品取出,在冷水中清洗并在无翻滚下干燥。The garment is then dyed in a single dyebath using two separate dyes, an acid dye and another cationic dye selected for contrasting colour. The cationic dye is bright red, Maxilon Red 3GLN purchased from CIBA, 0.4wt% relative to the cationic dyeable nylon component yarn in knitted clothes. The acid dye is bright turquoise blue acid dye, available from BASF's Acidolbr.Blue M-5G, which is 0.4wt% relative to the acid dyeable nylon component yarn in the knitted garment. The stainer was a Roaches Pyrotec 2002 (Roaches International LTD) using a 1 liter stainless steel airtight beaker. At the start, with the dyebath and fabric at 30°C, the cationic dye was added with 5.0% sodium sulphate (glauber's salt) and 0.1% Tinegal MR (a cationic dye blocker for basic dyes from CIBA) relative to the total weight of the fabric . At a dye liquor to fabric item ratio of 28:1, the water bath was adjusted to pH 6.0. The temperature was increased at 1.0°C per minute to 60°C and held for 10 minutes. The cooling rate back to 35°C was set at 2.5°C per minute. This is to prevent the cationic dye from being used up on the cationically dyeable yarn. To this same dyebath, the anionic dye was added, mixed with 1.0% Sandogen NH (a dye blocker for anionic dyes available from Clariant) based on the total weight of the article. The dyebath temperature was increased at 1.0°C per minute to 98°C and held at this temperature for 45 minutes. The dyebath was cooled to 50°C at 7.0°C per minute. The knit samples were removed, washed in cold water and dried without tumble.
由染色和整理织物专家小组检验此染色衣服。同意此衣服在前侧看,具有非常不寻常且有吸引力的闪光外观,其中在前侧高光泽纱是主要的。织物颜色是基本暗的淡紫色,它强烈地随观察角变化,特别是在织物的合股中。织物合股的深度显示灰蓝色,而升高的部分具有亮粉红色调的光泽。组分染料颜色,即亮红和刺眼绿松石色,不在此衣服中出现。衣服的背侧显示一些蓝色阴影而没有闪光效果。The dyed garment was inspected by a panel of experts in dyeing and finishing fabrics. Agreed that this garment has a very unusual and attractive shimmery look when viewed from the front where the high sheen yarn is dominant on the front. The fabric color is an essentially dark lavender that varies strongly with viewing angle, especially in the plies of the fabric. The depth of the fabric plies shows a gray-blue color, while the raised parts have a bright pink-toned sheen. The component dye colors, bright red and harsh turquoise, do not appear in this garment. The back side of the garment shows some shades of blue without the glitter effect.
实施例1-部分BExample 1 - Part B
在部分B中,纱线、衣服构造和染色方法与部分A中的那些相同,区别在于使用的染料。染料由如下物质组成:亮黄阳离子染料,购自CIBA的Maxilon Yellow GL 200%,以占样品中可阳离子染色尼龙重量0.1wt%施加;和强烈紫色染料,NYLOSAN Violet F-BL 180%,以占可酸染色尼龙组分重量0.9wt%施加。染色过程与部分A相同。In Part B, the yarn, garment construction, and dyeing method are the same as those in Part A, with the difference being the dye used. The dyes consisted of the following: Brilliant yellow cationic dye, Maxilon Yellow GL from CIBA 200%, applied at 0.1 wt% of the weight of cationically dyeable nylon in the sample; and intense violet dye, NYLOSAN Violet F-BL 180%, applied at 0.1% by weight of The acid dyeable nylon component was applied at 0.9 wt%. The dyeing process is the same as in Part A.
在此实施例中的视觉评定方法与部分A相同。整理的染色衣服具有总体丁香色,伴随有从紫色到暖银色的短暂闪光区域。没观察到原始的亮黄色并且衣服的背侧不显示虹彩。The visual assessment method in this example is the same as in Part A. Finished dyed garments have an overall lilac color with brief shimmering areas ranging from purple to warm silver. The original bright yellow color was not observed and the backside of the garment did not show iridescence.
实施例1-部分CExample 1 - Part C
部分C的纱线、衣服、染色方法和视觉评定与部分A中的那些相同。在部分C中使用了不同的染料,其由如下物质组成:鼠尾草绿阳离子染料,Sevron Yellow 3RL和Sevron Blue CAN的混合物,两者购自YORKSHIRE CHEMICALS。以占样品中可阳离子染色尼龙组分重量0.2wt%施用这些染料。以占可酸染色尼龙组分重量0.2wt%的更低浓度施用与部分B中所用相同的强烈紫色酸性染料,NYLOSAN Violet F-BL 180%。Yarns, garments, dyeing methods, and visual assessments for Part C are the same as those for Part A. A different dye was used in part C, consisting of the following: Sage Green cationic dye, a mixture of Sevron Yellow 3RL and Sevron Blue CAN, both purchased from YORKSHIRE CHEMICALS. These dyes were applied at 0.2% by weight of the cationically dyeable nylon component of the samples. The same intense violet acid dye used in Part B, NYLOSAN Violet F-BL 180%, was applied at a lower concentration of 0.2% by weight of the acid dyeable nylon component.
整理的染色衣服具有总体暗灰色,伴随有从淡紫色到银色的生动闪光区域。没观察到原始的鼠尾草绿色并且衣服的背侧不显示虹彩。Finished dyed garments have an overall dark gray color with areas of vivid shimmer ranging from lavender to silver. The original sage green was not observed and the backside of the garment did not show iridescence.
实施例1-部分DExample 1 - Part D
部分D的纱线、衣服、染色方法和视觉评定与部分A中的那些相同。在部分D中使用不同的染料,其由如下物质组成:亮黄阳离子染料,购自CIBA的Maxilon Yellow GL200%,以占样品中可阳离子染色尼龙组分重量0.2wt%施加;和亮蓝色染料,NYLOSAN Blue F-2RFL 160%以占可酸染色尼龙组分重量1.4wt%的浓度施加。Yarns, garments, dyeing methods, and visual assessments for Part D are the same as those for Part A. Different dyes were used in Part D, consisting of: Brilliant Yellow Cationic Dye, Maxilon Yellow GL 200% from CIBA, applied at 0.2 wt% of the weight of the cationically dyeable nylon component in the sample; and Brilliant Blue Dye , NYLOSAN Blue F-2RFL 160% was applied at a concentration of 1.4% by weight of the acid dyeable nylon component.
整理的染色衣服具有总体丁香色,伴随有从紫色到暖银色的强烈短暂的闪光区域。没观察到原始的亮黄色并且衣服的背侧不显示虹彩。The finished dyed garment has an overall lilac color with intense, fleeting areas of shimmer ranging from purple to warm silver. The original bright yellow color was not observed and the backside of the garment did not show iridescence.
实施例1-部分EExample 1 - Part E
此实施例的纱线、衣服、染色方法和视觉评定与部分A中的那些相同。在部分E中使用不同的染料,其由如下物质组成:亮黄阳离子染料,购自CIBA的Maxilon Yellow GL200%,以占样品中可阳离子染色尼龙组分重量0.2wt%施加;和强烈紫色酸性染料,NYLOSAN VioletF-BL 180%,以占可酸染色尼龙组分重量0.9wt%施加。The yarn, garment, dyeing method, and visual evaluation for this example were the same as those in Part A. Different dyes were used in Part E, consisting of: Brilliant Yellow Cationic Dye, Maxilon Yellow GL 200% from CIBA, applied at 0.2% by weight of the cationically dyeable nylon component of the sample; and Intense Violet Acid Dye , NYLOSAN VioletF-BL 180%, applied in an amount of 0.9wt% by weight of the acid dyeable nylon component.
整理的染色衣服具有更斑点外观而不是部分A-D中其它衣服的总体基本色。相信此观察是由于用于部分E的在较浅黄色和暗蓝组分之间的强烈对比。部分E衣服显示强烈虹彩和几乎金属光辉。衣服的背侧不显示虹彩。The finished dyed garment has a more speckled appearance rather than the overall base color of the other garments in sections A-D. It is believed that this observation is due to the strong contrast between the lighter yellow and dark blue components for Part E. Part E garments show intense iridescence and an almost metallic sheen. The back side of the garment does not show iridescence.
实施例1-部分FExample 1 - Part F
对比Compared
在此对比例中,制备基本与用于制备部分A第二纱基本相同的平(未变形)POY组分。从其制备此纱的聚合物包含35克当量的胺端基每1000千克聚合物(AEG酸)。所有其它试验详细情况,包括染色和视觉评定方法,均与本发明部分E相同。此对比例说明使用标准酸性染料尼龙纱代替高胺端基浓度更深染色纱的效果。此更低胺端基浓度可酸染色纱不如深染料纱那样强烈地保持酸性染料,允许更多的酸性染料沾污可阳离子染色的纱。In this comparative example, substantially the same flat (untextured) POY component as was used to prepare the Part A second yarn was prepared. The polymer from which this yarn was made contained 35 gram equivalents of amine end groups per 1000 kg of polymer (AEG acid ). All other test details, including staining and visual assessment methods, are the same as in Part E of the present invention. This comparative example illustrates the effect of using a standard acid dyed nylon yarn in place of a more deeply dyed yarn with a high amine end group concentration. This lower amine end group concentration acid dyeable yarn does not retain acid dye as strongly as dark dyed yarn, allowing more acid dye to stain cationically dyeable yarn.
在染色此衣服之后,由蓝色酸性染料的相当更多可阳离子染色纱的沾污导致灰绿色和闪光效果的基本损失。After dyeing this garment, the staining of the considerably more cationically dyeable yarn by the blue acid dye resulted in a substantial loss of sage green and sparkle effects.
实施例2Example 2
在本发明的此实施例中,制备“无缝”圆形针织物衣服并根据实施例1部分A的染色程序染色。制备的衣服由具有亮光泽的平(未变形)POY的第一纱和具有三叶横截面形状的复丝组成。此平POY具有56分特总重量并包含20个长丝。此纱从可阳离子染色尼龙66聚合物纺成,该聚合物的RV(甲酸)为31.5,二氧化钛含量为0.02wt%,胺端基浓度为42克当量每1000kg聚合物,并且磺酸酯基团浓度为55克当量每1000kg聚合物。将此纱采用已知方式通过具有多个有型毛细管的喷丝头板纺丝,得到每个纱线有20个三叶横截面长丝的POY。In this example of the invention, a "seamless" circular knit garment was prepared and dyed according to the dyeing procedure of Example 1 Part A. The prepared garment consisted of a first yarn of flat (untextured) POY having a glossy sheen and a multifilament having a trilobal cross-sectional shape. This flat POY has a total weight of 56 decitex and contains 20 filaments. The yarn is spun from a cationically dyeable nylon 66 polymer having an RV (formic acid) of 31.5, a titanium dioxide content of 0.02 wt%, an amine end group concentration of 42 gram equivalents per 1000 kg of polymer, and sulfonate groups The concentration was 55 gram equivalents per 1000 kg of polymer. The yarn was spun in a known manner through a spinneret plate having shaped capillaries to obtain a POY having 20 filaments of trilobal cross-section per yarn.
第二纱线是深度可酸染色、完全无光泽、圆形横截面尼龙66变形纱的市售样品,该变形纱覆盖17分特DuPont LYCRA斯潘德克斯T175。如在实施例1的部分A-F中,由于被染色的尼龙纱覆盖,LYCRA斯潘德克斯不必达到本发明的闪光视觉效果。从其制备此纱的深染料尼龙聚合物与用于制备实施例1部分A的第二纱的相同。聚合物是40RV,胺端基浓度是70克当量每1000千克聚合物并且TiO2含量是1.5wt%。将此纱由常规POY途径纺丝并通过摩擦假捻变形而变形,然后用于采用已知方式覆盖LYCRA斯潘德克斯。The second yarn was a commercially available sample of a deeply acid-dyeable, completely matte, circular cross-section nylon 66 textured yarn covered with 17 dtex DuPont LYCRA® Spandex T175. As in Example 1, Parts A-F, the LYCRA(R) spandex does not have to achieve the sparkle visual effect of the present invention due to being covered by the dyed nylon yarn. The deep dyed nylon polymer from which this yarn was made was the same as used to make the second yarn of Example 1 Part A. The polymer was 40 RV, the amine end group concentration was 70 gram equivalents per 1000 kg polymer and the TiO2 content was 1.5 wt%. This yarn was spun by the conventional POY route and texturized by frictional false twist texturing and then used to cover LYCRA(R) spandex in known manner.
在13英寸28隔距的SANTONI SM8-83 TOP无缝机上,使用单一运动衫构造,添纱到背侧将第一纱和第二纱编织在一起。由实施例1的方法使用占可阳离子染色尼龙组分重量0.15wt%的作为阳离子染料的Maxilon Blue TL,和分别占酸染色尼龙组分纱重量0.2wt%和0.4wt%的酸性染料NYLOSAN Bordeaux NBL和NYLOSAN Yellow N7GI染色获得的无缝圆形针织衣服。On a 13" 28 gauge SANTONI SM8-83 TOP seamless machine, using a single jersey construction, plating to the back side to weave the first and second yarns together. The method of Example 1 uses Maxilon Blue TL as cationic dyestuff accounting for 0.15wt% by weight of cationic dyeable nylon component, and acid dye NYLOSAN Bordeaux NBL accounting for 0.2wt% and 0.4wt% of acid dyed nylon component yarn weight respectively Seamless circular knit garment dyed with NYLOSAN Yellow N7GI.
主观地由专家组评定衣服的外观。衣服织物的前面是相当均匀的微红棕色。添纱了亮光泽纱的衣服织物的背侧总体具有李子色,具有粉红阴影和虹彩蓝的有光泽闪光。The appearance of the garments was subjectively assessed by a panel of experts. The front of the garment fabric is a fairly uniform reddish brown. The backside of the garment fabric plated with bright luster yarn had an overall plum color with a shade of pink and a lustrous sparkle of iridescent blue.
Claims (9)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US35102302P | 2002-01-23 | 2002-01-23 | |
| US60/351,023 | 2002-01-23 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN1756871A true CN1756871A (en) | 2006-04-05 |
Family
ID=27613450
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNA038023032A Pending CN1756871A (en) | 2002-01-23 | 2003-01-21 | Iridescent fabrics from polyamide yarns |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20030220037A1 (en) |
| EP (1) | EP1468138A1 (en) |
| JP (1) | JP2005515318A (en) |
| KR (1) | KR20040079939A (en) |
| CN (1) | CN1756871A (en) |
| WO (1) | WO2003062522A1 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101906727A (en) * | 2010-06-25 | 2010-12-08 | 南通通远鑫纺织品有限公司 | Short-process low-carbon dyeing and finishing process of dual-polyester chameleon crumpled decorative cloth |
| CN103215671A (en) * | 2013-04-17 | 2013-07-24 | 江苏好易纺织科技有限公司 | Polytrimethylene-tereph-thalate (PTT) fiber of carpet and preparation method for PTT fiber |
| CN112111828A (en) * | 2019-06-19 | 2020-12-22 | 吴江海角工业用布有限公司 | Production and preparation method of high-strength heat-resistant nylon spring wire |
| CN112663172A (en) * | 2020-12-23 | 2021-04-16 | 常州灵达特种纤维有限公司 | High-performance polyester-polyamide blend fiber and preparation method thereof |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IL154571A0 (en) * | 2003-02-20 | 2003-09-17 | Nilit Ltd | Process for making polyamide textile articles bearing designs in different colors |
| US20090136704A1 (en) * | 2007-11-27 | 2009-05-28 | Invista North America S. A R. I. | Dual acid/cationic dyeable polyamide polymer fibers and yarns, methods of making the same, and textile articles including dual acid/cationic dyeable polyamide polymer fibers |
| US20110203057A1 (en) * | 2008-10-13 | 2011-08-25 | Hietpas Geoffrey D | Fabric including polyurethane elastic yarn |
| US10179970B2 (en) * | 2013-03-15 | 2019-01-15 | Columbia Insurance Company | Process for dyeing fabric |
| MX2021004939A (en) * | 2018-10-29 | 2021-11-12 | Dror Selivansky | Concrete curing blankets and anti-bacterial textile fibers. |
| US11566353B2 (en) | 2019-04-05 | 2023-01-31 | Nike, Inc. | Knit component with differing visual effects |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1161411A (en) * | 1965-11-01 | 1969-08-13 | Ici Ltd | A process for Dyeing Textile Materials and the Textile Materials so obtained |
| DE1919746A1 (en) * | 1969-04-18 | 1970-11-05 | Basf Ag | Differentiated dyeable textiles |
| US3987022A (en) * | 1973-09-19 | 1976-10-19 | E. I. Du Pont De Nemours And Company | Yellow to orange disazo cationic dyes containing two heterocyclic quaternary ammonium groups |
| US4075378A (en) * | 1975-09-12 | 1978-02-21 | E. I. Du Pont De Nemours And Company | Polyamide filaments with a basic-dyeable sheath and an acid-dyeable core and dyeing process therefor |
| US4295329A (en) * | 1980-06-10 | 1981-10-20 | E. I. Du Pont De Nemours And Company | Cobulked continuous filament heather yarn method and product |
| US5340886A (en) * | 1989-07-17 | 1994-08-23 | Basf Corporation | Acid-dye resistant polyamide products and process for preparation |
| EP0721522B1 (en) * | 1993-09-28 | 1997-05-28 | E.I. Du Pont De Nemours And Company | Iridescent fabrics |
| GB9405546D0 (en) * | 1994-03-21 | 1994-05-04 | Collier Campbell Ltd | Textile fabrics |
| WO1999018268A1 (en) * | 1997-10-02 | 1999-04-15 | Nissan Motor Co., Ltd. | Fiber structure and textile using same |
| JP3416038B2 (en) * | 1997-10-13 | 2003-06-16 | 帝人ファイバー株式会社 | Warp knitted fabric with interference color tone |
| BR0012348A (en) * | 1999-07-08 | 2002-03-26 | Du Pont | Method of treating the textile surface of an article and article |
| GB2373256B (en) * | 2001-03-14 | 2005-03-30 | Du Pont | Fabrics comprising melt spun yarns having high lustre |
-
2003
- 2003-01-21 WO PCT/US2003/001697 patent/WO2003062522A1/en not_active Ceased
- 2003-01-21 EP EP20030705835 patent/EP1468138A1/en not_active Withdrawn
- 2003-01-21 KR KR10-2004-7011250A patent/KR20040079939A/en not_active Withdrawn
- 2003-01-21 JP JP2003562379A patent/JP2005515318A/en not_active Withdrawn
- 2003-01-21 CN CNA038023032A patent/CN1756871A/en active Pending
- 2003-01-22 US US10/349,230 patent/US20030220037A1/en not_active Abandoned
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101906727A (en) * | 2010-06-25 | 2010-12-08 | 南通通远鑫纺织品有限公司 | Short-process low-carbon dyeing and finishing process of dual-polyester chameleon crumpled decorative cloth |
| CN101906727B (en) * | 2010-06-25 | 2013-04-17 | 南通通远鑫纺织品有限公司 | Short-process low-carbon dyeing and finishing process of dual-polyester chameleon crumpled decorative cloth |
| CN103215671A (en) * | 2013-04-17 | 2013-07-24 | 江苏好易纺织科技有限公司 | Polytrimethylene-tereph-thalate (PTT) fiber of carpet and preparation method for PTT fiber |
| CN112111828A (en) * | 2019-06-19 | 2020-12-22 | 吴江海角工业用布有限公司 | Production and preparation method of high-strength heat-resistant nylon spring wire |
| CN112663172A (en) * | 2020-12-23 | 2021-04-16 | 常州灵达特种纤维有限公司 | High-performance polyester-polyamide blend fiber and preparation method thereof |
| CN112663172B (en) * | 2020-12-23 | 2023-01-10 | 常州灵达特种纤维有限公司 | High-performance polyester-polyamide blend fiber and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20040079939A (en) | 2004-09-16 |
| JP2005515318A (en) | 2005-05-26 |
| WO2003062522A1 (en) | 2003-07-31 |
| US20030220037A1 (en) | 2003-11-27 |
| EP1468138A1 (en) | 2004-10-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US3593513A (en) | Dyeing of mixed synthetic polymeric yarns | |
| US3681910A (en) | Composite yarn product | |
| CN1756871A (en) | Iridescent fabrics from polyamide yarns | |
| EP1373608B1 (en) | Melt spun yarns having high lustre | |
| AU2002245681A1 (en) | Melt spun yarns having high lustre | |
| US6524503B2 (en) | Process for the manufacture of polyamide yarns dyeable in melange shades | |
| JP4955463B2 (en) | Composite fiber | |
| WO2000065135A1 (en) | Polyparaphenylene terephthalamide fiber and method for producing the same | |
| JPH0441738A (en) | Dyed fabric comprising polyester fiber and polyamide fiber cord and its preparation | |
| JP2005325481A (en) | Polyamide sheath-core conjugated fiber having excellent opacity, uv light-shielding property, heat-shielding property, moisture-absorbing or releasing property, and color-developing property and covering elastic yarn and bulky processed yarn using the same | |
| HK1090103A (en) | Iridescent fabrics from polyamide yarns | |
| KR100785237B1 (en) | Polyester Sheath Core Composite Yarn and Manufacturing Method Thereof | |
| JP5543748B2 (en) | Woven knitted fabric using nylon 11 yarn and dyeing method thereof | |
| KR880000293B1 (en) | Textured nylon-6 filament | |
| JP4207607B2 (en) | Polyester sewing thread for embroidery | |
| JP2663604B2 (en) | Normal pressure dyeable anti-pill fiber | |
| JPH0860439A (en) | Polymer blend fiber and mixed yarn thereof | |
| JPH09228181A (en) | Fluff | |
| JP2015067923A (en) | Cation-dyeable polyamide fiber, woven and knitted fabric, and sewn product | |
| WO2017091159A1 (en) | Combined nylon filament yarn | |
| KR980009545A (en) | Process for producing pleochroism polyester yarn | |
| JP2008163470A (en) | Interknitted pantyhose excellent in luster | |
| JP2022030198A (en) | Stretchable base fabric for vehicle | |
| KR20100116830A (en) | Method for manufacturing cation dyeable polyamide yarn | |
| JP2019014987A (en) | Low-temperature dyeable polyester latency crimpable composite thread |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1090103 Country of ref document: HK |
|
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: WD Ref document number: 1090103 Country of ref document: HK |