EP0294127A2 - Mit Harz laminierter Gummistopfen - Google Patents
Mit Harz laminierter Gummistopfen Download PDFInfo
- Publication number
- EP0294127A2 EP0294127A2 EP88304894A EP88304894A EP0294127A2 EP 0294127 A2 EP0294127 A2 EP 0294127A2 EP 88304894 A EP88304894 A EP 88304894A EP 88304894 A EP88304894 A EP 88304894A EP 0294127 A2 EP0294127 A2 EP 0294127A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- rubber stopper
- rubber
- laminated
- fluorine resin
- resin film
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65D—CONTAINERS FOR STORAGE OR TRANSPORT OF ARTICLES OR MATERIALS, e.g. BAGS, BARRELS, BOTTLES, BOXES, CANS, CARTONS, CRATES, DRUMS, JARS, TANKS, HOPPERS, FORWARDING CONTAINERS; ACCESSORIES, CLOSURES, OR FITTINGS THEREFOR; PACKAGING ELEMENTS; PACKAGES
- B65D51/00—Closures not otherwise provided for
- B65D51/002—Closures to be pierced by an extracting-device for the contents and fixed on the container by separate retaining means
Definitions
- This invention relates to rubber stoppers for bottles or vials and in particular those used for sealing bottles or vials containing vaccines, chemicals, and the like.
- rubber stoppers The qualities and properties required in Japan of a rubber stopper or closure for medical vials (hereinafter referred to as "rubber stoppers") should be according to the test of a rubber stopper for a liquor transfusion of 11th Revision, Japanese Pharmacopoeia.
- Such rubber stoppers are required to satisfy a number of requirements, such as resistance to gas permeability, non-elusive properties, highly clean properties, resistance to chemicals and water absorption, resistance to needle piercing, good self-sealing and sliding properties etc.
- the invention is characterised in that both surfaces of the rubber stopper are laminated, the upper surface of a head or cap portion of the rubber stopper being laminated with a fluorine resin film and some or all of a body portion adjacent a lower surface of the head or cap portion of the rubber stopper, which is inserted into the vial, is laminated with a fluorine resin film.
- the whole surface of the stopper beneath the head or cap portion, including the body portion may alternatively be laminated with a fluorine resin film.
- a rubber stopper comprises a head or cap portion at an upper end of the rubber stopper which covers the mouth of a vial, in use, and a body portion provided at a lower end of the rubber stopper beneath and adjacent the head portion, the body portion being inserted into the mouth of the vial,.
- the body portion and optionally the lower part of the head portion are laminated with a fluorine resin film as described in the prior art and additionally, the upper surface of the head portion is also laminated with a fluorine resin film instead of a silicone coating according to the prior art.
- a fluorine resin film as described in the prior art and additionally, the upper surface of the head portion is also laminated with a fluorine resin film instead of a silicone coating according to the prior art.
- Both the upper surface of a head portion of therubber stopper and a body under the head are laminated with a fluorine resin film which has a high lubricity to obtain a rubber stopper, in which the occurrence of piercing fragments is remarkably reduced.
- the body portion of the stopper is laminated with a fluorine resin film
- some or all of the body portion can be laminated with a fluorine resin film such that the sealing means, i.e. a lower surface of the head portion which seals against the vial mouth, comprises an exposed rubber surface and optionally the upper end of the body portion may also be exposed rubber.
- the body portion and sealing means can be wholly laminated with a fluorine resin film.
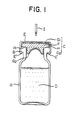
- Fig. 1 shows one embodiment of a rubber stopper which can be inserted into the mouth of a vial.
- a rubber stopper C an upper portion of the head portion of the stopper is laminated with a fluorine resin film G1 .
- the body portion, which is inserted into the mouth of a vial A containing a pharmaceutical chemical D is laminated with a fluorine resin film G2.
- a sealing lip H of the head portion contacts the lip of the mouth of the vial A.
- the lip H is not laminated such that a part F of the rubber is exposed.
- the vial A is closed by the rubber stopper C, which is covered by an aluminum cap E and fastened to the vial by a flap E′.
- I is an injection needle.
- Figs. 2 to Fig. 4 show another embodiment of the rubber stopper which is suitable for use in a freeze drying method for the preparation of a drug.
- Items C, G1, G2 and H have the same meanings as in the case of Fig. 1.
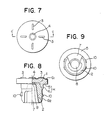
- the rubber stopper C comprises a head portion l, a body portion 2 projecting from a lower surface of the head portion 1, a convex portion 3 on the head portion 1, a concave portion 4 also on the head portion 1, wherein the needle of a syringe enters the stopper.
- the stopper further comprises sealing means H consisting of a flange surface 5 on the lower side of the head portion 1 and another sealing surface 6 at an upper end of the body portion 2 adjacent flange surface 5, both the sealing means having exposed rubber surfaces.
- the body portion 2 is cut or notched at two positions to form two legs 7 which are used in a vacuum drying process, the deepest part 8 of the cut portion 7 and protrusions 9 and 10 on the body portion 2 being used for maintaining a via
- Fig. 5 and Fig. 6 show a further embodiment of the rubber stopper suitable for use in a reduced pressure gas replacement method in which body portion 2 is completely laminated with a fluorine resin film G2 but a part of the rubber surface on the lower side of the head portion is exposed.
- Fig. 7 to Fig. 9 show a further embodiment of the rubber stopper suitable for use in a freeze drying method in which only one cut part 7 is provided.
- Fig. 10 and Fig. 11 show yet another embodiment of the rubber stopper corresponding to a modification of that shown in Fig. 5 and Fig. 6, in which all of the upper and lower surfaces of the head portion and the entire surface of the body portion are laminated with a fluorine resin film G1 and G2.
- the rubber stoppers of the types shown in Fig. 2 to Fig. 9 are suitable for use in preparation of drugs by a positive pressure method wherein the inner pressure of a vial is higher than the atmospheric pressure, or a negative pressure method wherein the inner pressure of a vial is lower than the atmospheric pressure, or a gas replacement method.
- the rubber stoppers of the types as shown in Fig. 10 and Fig. 11 are suitable for use in preparation of drugs by a normal pressure method wherein the inner pressure of a vial is substantially the same as the atmospheric pressure.
- examples of the fluorine resin film used for laminating the upper surface of the head portion of the rubber stopper, a part or all of the body portion or the entire whole lower surface under the head portion of the rubber stopper including the body portion are tetrafluoroethyleneethylene copolymer (ETFE), tetrafluoroethylene-hexafluoropropylene copolymer (FEP), tetrafluoroethylene-perfluoroalkyl vinyl ether copolymer (PFA), chlorotrifluoroethylene-ethylene copolymer (ECTPE), polyvinyl fluoride (PVF), polyvinylidene fluoride (PVDF), polychlorotrifluoroethylene (PCTFE), polytetrafluoroethylene (PTFE) and the like.
- the fluorine resin used in laminating the upper surface of the head portion and that used for the entire lower surface or for the body surface may be the same or different resins.
- the thickness of the fluorine resin film used for laminating the upper surface of the head portion or the surface of a part of all of the body portion is preferably between 0.01 and 0.2 mm. If the thickness of the laminate is less than 0.01 mm, the film tends to break during molding or processing so that the warranty of the quality of the product may be insufficient, whilst if its thickness is more than 0.2 mm, the rigidity of the laminate is too great to maintain proper self-sealing and needle piercing properties.
- a strong adhesive surface between a fluorine resin film and a rubber surface it is preferable to subject the surface of the film to a pretreatment in a conventional manner, for example, corona discharge, plasma discharge, glow discharge, arc discharge, sputter etching, etc.
- a strong adhesive surface can be obtained only by subjecting the surface of the film to a cleaning treatment or primer undercoating treatment, for example, using silicone compounds, epoxy resins, chloroprene rubbers, etc. In any method, it is important to adjust the adhesive strength between the film and the rubber surface to between 1 and 30 Kg/cm.
- the rubber composition used for a base body of the rubber stopper can be substantially prepared by a prior art technique, i.e. by adding a vulcanizing agent, vulcanization accelerator, vulcanization activator, processing aid, filler, reinforcing agent, etc to at least one member selected from the group consisting of isopreneisobutylene copolymer rubber (IIR), chlorinated or brominated IIR (CIIR, BIIR), acrylonitrile-butadiene copolymer rubber (NBR), acrylonitrile-butadiene-isoprene triple copolymer (NBIR), isoprene rubber (IR), butadiene rubber (BR), styrene-butadiene rubber (SBR), ethylene-propylene rubber (EPM), ethylene-propylene-diene rubber (EPDM), chlorosulfonated polystyrene (CSM), ethylene-vinyl acetate copolymer (EVA), sty
- Production of a laminated rubber stopper according to the present invention can be carried out by a process similar to that described in our Japanese Patent Publication No. 53184/1982 and our Japanese Patent Laid-Open Publication No. 272134/1986, which process comprises placing and combining a fluorine resin film, the upper surface of which is surface-treated, and a non-vulcanized rubber sheet, in order, on a lower metal mould which has a recess for forming the body portion of the stopper, pressing and heating them between the lower mould and an upper metal mould to vulcanize and form an intermediate product, cutting or trimming the intermediate product in line with the shape of the body portion, charging again the thus resulting intermediate product in a lower metal mould having a recess corresponding to the body portion, then placing thereon a non-vulcanized.
- a rubber stopper of the present invention can be manufactured, in which the upper surface of the head and a part or all of the body portion are laminated with a fluorine resin film, but maintaining an exposed rubber surface on the sealing portion of the head, as shown in Fig. 1 to 9.
- Another modified process comprises combining and charging a fluorine resin film, non-vulcanized compound rubber sheet and a fluorine resin film stacked in order between an upper metal mould having a recess corresponding to the head portion of the rubber stopper and a lower metal mould having a recess corresponding to the body portion, pressing and heating them to effect vulcanization, forming and then trimming along the outer diameter of the rubber stopper, thus obtaining another rubber stopper of the present invention, in which the upper and lower surface of the rubber stopper are wholly laminated with the fluorine resin film, as shown in Fig. 10 and Fig. 11.
- the above described rubber composition was kneaded using two rolls according to a method described in "The Society of Rubber Industry Japan Standard” (1979) and then used for the preparation of a rubber stopper as shown in Fig. 6 by a process comprising forming a rubber stopper simultaneously with laminating it with various fluorine resin films.
- the body portion was laminated with the following ETFE film 1) in every case, while the head portion was laminated with the following four fluorine resin films 1) to 4), these fluorine resin films being manufactured by Daikin Kogyo Co. and subjected to a sputter etching treatment on one side and having a thickness of 75 ⁇ m:
- the resuling intermediate product was cut or trimmed to a predetermined size and the cut or trimmed body portion was charged in the lower metal mould, on which were placed, in order, a non-vulcanized compound rubber 1, one of the foregoing resin films 1) to 4) for G1 and an upper metal mould having a recess for forming the head portion, and pressed with heating at a temperature of 150 ⁇ 1°C, thus obtaining a rubber stopper as shown in Fig. 6, followed by cutting a separate into individual stoppers and washing.
- a comparative sample 1 was prepared in an analogous manner to that described above, although the head portion was not laminated (Comparative Example 1), and surface-treating or laminating the head by the prior art methods to obtain rubber stoppers each having a shape as shown in Fig. 6 (Comparative Examples 2 to 4).
- the rubber surface of the head portion was coated twice with a silicone emulsion diluted 30 times with water, using a brush.
- the rubber surface of the head portion was coated twice with an isopropyl alcohol solution containing 1.2% by weight of a silicone oil, using a brush.
- the rubber surface of the head portion was coated with a polypropylene film (Mitsui Polypro -commercial name- manufactured by Mitsui Sekiyu Kagaku Co., 75 ⁇ m thickness).
- the water absorption ratio of the rubber stoppers in Examples 1 to 4 according to the present invention was very low, i.e. 1/10 to 1/120 times less than that of Comparative Examples 1 to 4.
- the laminated rubber stopper according to the present invention has the following advantages:
- the drug or pharmaceutical production speed can effectively be increased by the use of the rubber stopper of the present invention, from which a large decrease of the drug production cost can be expected.
- occurrence of fine particles can be remarkably reduced in the rubber stopper of the present invention, although a number of fine particles occur due to a silicone in the rubber stopper of the prior art, it is possible to achieve a pharmaceutical preparation with high quality. Moreover, occurrence of fragments due to needle penetration can be prevented.
- the laminated rubber stopper of the present invention is capable of exhibiting better properties at any stage of preparation of a drug using a medical vial, storage and application and thus largely serve to a medical vial preparation with a high quality.
Landscapes
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Medical Preparation Storing Or Oral Administration Devices (AREA)
- Closures For Containers (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP131297/87 | 1987-05-29 | ||
| JP62131297A JP2545540B2 (ja) | 1987-05-29 | 1987-05-29 | 両面ラミネ−トゴム栓 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0294127A2 true EP0294127A2 (de) | 1988-12-07 |
| EP0294127A3 EP0294127A3 (en) | 1990-05-30 |
| EP0294127B1 EP0294127B1 (de) | 1994-11-09 |
Family
ID=15054674
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP88304894A Expired - Lifetime EP0294127B1 (de) | 1987-05-29 | 1988-05-27 | Mit Harz laminierter Gummistopfen |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP0294127B1 (de) |
| JP (1) | JP2545540B2 (de) |
| DE (1) | DE3852061T2 (de) |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5064083A (en) * | 1990-03-08 | 1991-11-12 | The West Company, Incorporated | Closure device |
| EP0564037A1 (de) * | 1992-04-02 | 1993-10-06 | Nycomed Imaging As | Verschleissfester Stöpsel zur Vermeidung des Erzeugungs von Teilchen beim Durchbohren |
| EP0639114A4 (de) * | 1992-05-06 | 1995-04-19 | Mallinckrodt Medical Inc | Behälter und verschluss zur aufrechterhaltung der stabilität von kochsalzlösungen. |
| EP0814027A1 (de) * | 1995-04-05 | 1997-12-29 | Daikyo Seiko, Ltd. | Laminierter Gummipfropfen |
| WO2001022998A1 (en) * | 1999-09-29 | 2001-04-05 | Aaipharma, Inc. | Stable calcitriol solution for packaging in vials |
| EP1228973A1 (de) * | 2001-01-19 | 2002-08-07 | Daikyo Seiko, Ltd. | Laminierter Gummipfropfen für Medizinbehälter |
| EP1760002A1 (de) * | 2005-08-31 | 2007-03-07 | West Pharmaceutical Services Deutschland Gmbh & Co. KG | Medikamentenflasche oder dergleichen Behälter |
| EP1792657A1 (de) * | 2005-12-01 | 2007-06-06 | Eppendorf Ag | Deckelgefäss |
| US20090202759A1 (en) * | 2006-07-06 | 2009-08-13 | Arkema Inc. | Flexible multilayer vinylidene fluoride tubes |
| US20090297442A1 (en) * | 2006-06-21 | 2009-12-03 | Stig Hemstad | Radiopharmaceutical products |
| US20100206836A1 (en) * | 2007-10-18 | 2010-08-19 | Tsuyoshi Koshidaka | Vial rubber-stopper |
| WO2012052394A1 (en) * | 2010-10-18 | 2012-04-26 | Sanofi Pasteur | Method of storing a vaccine containing an aluminum adjuvant |
| RU2448733C2 (ru) * | 2006-06-21 | 2012-04-27 | Джи-И Хелткер Лимитед | Радиофармацевтические продукты |
| US8512691B2 (en) | 1996-12-24 | 2013-08-20 | Biogen Idec Ma Inc. | Stable liquid interferon-beta formulations |
| FR3082729A1 (fr) | 2018-06-26 | 2019-12-27 | Laboratoire Francais Du Fractionnement Et Des Biotechnologies | Conditions de stockage d'une composition de proteines comprenant du tensioactif et evolution de la teneur en tensioactif |
| CN113474083A (zh) * | 2019-01-04 | 2021-10-01 | 仪器实验室公司 | 用于高刺穿计数应用的容器塞子 |
| EP4253276A1 (de) * | 2022-03-30 | 2023-10-04 | Datwyler Pharma Packaging Belgium | Verschlussstopfen für medizinische oder pharmazeutische substanzen enthaltende behälter |
| USD1056701S1 (en) | 2022-03-30 | 2025-01-07 | Datwyler Pharma Packaging Belgium | Stopper |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2528535B2 (ja) * | 1990-03-20 | 1996-08-28 | 株式会社ニッショー | バイアル用ゴム栓の製造方法 |
| JPH0698176B2 (ja) * | 1991-05-29 | 1994-12-07 | 川澄化学工業株式会社 | 医療容器の口部及び医療容器 |
| JPH07323072A (ja) * | 1994-05-31 | 1995-12-12 | Daikyo Seiko:Kk | 医薬品用積層ゴム栓 |
| JPH08175554A (ja) * | 1994-12-22 | 1996-07-09 | Kazusa Koshitsu Chrome:Kk | ゴム栓及びゴム栓の製造方法 |
| JP3634944B2 (ja) * | 1997-06-18 | 2005-03-30 | 内外化成株式会社 | 医療用キャップ |
| JP3676909B2 (ja) * | 1997-07-08 | 2005-07-27 | 内外化成株式会社 | 医療用キャップ |
| JP2001245956A (ja) * | 2000-03-03 | 2001-09-11 | Ohtsu Tire & Rubber Co Ltd :The | 医療用ゴム栓 |
| JP2001301781A (ja) * | 2000-04-19 | 2001-10-31 | Daikyo Seiko Ltd | 医薬医療容器用ゴム栓 |
| JP2002345925A (ja) * | 2001-05-28 | 2002-12-03 | Ohtsu Tire & Rubber Co Ltd :The | 医療用の積層ゴム製品およびその製造方法 |
| DE60315198T2 (de) * | 2002-09-17 | 2008-04-17 | Glaxosmithkline Biologicals S.A. | Artikelhaltevorrichtung und fördersystem |
| AU2003900033A0 (en) * | 2003-01-07 | 2003-01-23 | Procork Pty Ltd | Container stopper |
| JP2004216753A (ja) * | 2003-01-16 | 2004-08-05 | Sumitomo Rubber Ind Ltd | 医薬用ゴム栓の製造方法 |
| US20090196798A1 (en) * | 2008-02-06 | 2009-08-06 | Robert Sassa | Barrier with Low Extractables and Resealing Properties |
| CN201427724Y (zh) * | 2009-03-27 | 2010-03-24 | 赵舜培 | 一种密封结构 |
| JP5750267B2 (ja) * | 2010-01-15 | 2015-07-15 | 住友化学株式会社 | 有機半導体素子用の液状組成物の保管方法 |
| JP2014079373A (ja) * | 2012-10-16 | 2014-05-08 | Sumitomo Rubber Ind Ltd | キャップでシールされる医療用ゴム栓 |
| CN103271822A (zh) * | 2013-05-22 | 2013-09-04 | 盛州橡塑胶(苏州)有限公司 | 无缝隙覆膜胶塞及其制备方法 |
| JP6288558B2 (ja) * | 2014-03-28 | 2018-03-07 | 大日本印刷株式会社 | プラズマ処理された表面平滑化フッ素系樹脂フィルムおよびその製造方法 |
| CN111148956B (zh) * | 2017-09-28 | 2021-07-02 | 豪夫迈·罗氏有限公司 | 用于冻干小瓶的小瓶塞子和用于封闭冻干小瓶的封闭方法 |
| CN110638801A (zh) * | 2019-08-23 | 2020-01-03 | 四川科瑞德制药股份有限公司 | 一种注射用药品及其制备方法 |
| CN111961372A (zh) * | 2020-08-25 | 2020-11-20 | 江苏润德医用材料有限公司 | 用于卤化丁基橡胶塞的涂膜液、涂膜胶塞及二者的制备方法和应用 |
| JP7610855B2 (ja) * | 2022-08-05 | 2025-01-09 | 有限会社米田精密金型製作所 | 容器の蓋体 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3628681A (en) * | 1969-10-06 | 1971-12-21 | Plastics Consulting And Mfg Co | Stopper |
| JPS5547851U (de) * | 1978-09-25 | 1980-03-28 | ||
| JPS5747637A (en) * | 1980-09-06 | 1982-03-18 | Daikyo Gomme Seikou:Kk | Manufacture of laminate rubber stopper |
| US4499148A (en) * | 1983-01-10 | 1985-02-12 | Canton Bio-Medical Products, Inc. | Composite materials of silicone elastomers and polyolefin films, and method of making |
| JPS59200649A (ja) * | 1983-04-26 | 1984-11-14 | ダイキン工業株式会社 | 医薬品用ゴム栓 |
| DE3346351A1 (de) * | 1983-12-22 | 1985-07-11 | Pharma-Gummi Wimmer West Gmbh, 5180 Eschweiler | Pharmazeutischer stopfen, kolben od. dgl. und verfahren zum herstellen von pharmazeutischen stopfen, kolben od. dgl. |
| JPS6131441U (ja) * | 1984-07-31 | 1986-02-25 | 株式会社 大協ゴム精工 | 樹脂ラミネ−ト医薬品用ゴム栓 |
-
1987
- 1987-05-29 JP JP62131297A patent/JP2545540B2/ja not_active Expired - Lifetime
-
1988
- 1988-05-27 DE DE19883852061 patent/DE3852061T2/de not_active Expired - Lifetime
- 1988-05-27 EP EP88304894A patent/EP0294127B1/de not_active Expired - Lifetime
Cited By (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5064083A (en) * | 1990-03-08 | 1991-11-12 | The West Company, Incorporated | Closure device |
| EP0564037A1 (de) * | 1992-04-02 | 1993-10-06 | Nycomed Imaging As | Verschleissfester Stöpsel zur Vermeidung des Erzeugungs von Teilchen beim Durchbohren |
| AU666910B2 (en) * | 1992-04-02 | 1996-02-29 | Nycomed Imaging As | Improved stopper for reduction of particulate matter |
| EP0639114A4 (de) * | 1992-05-06 | 1995-04-19 | Mallinckrodt Medical Inc | Behälter und verschluss zur aufrechterhaltung der stabilität von kochsalzlösungen. |
| EP0814027A1 (de) * | 1995-04-05 | 1997-12-29 | Daikyo Seiko, Ltd. | Laminierter Gummipfropfen |
| US6286699B1 (en) | 1995-04-05 | 2001-09-11 | Daikyo Seiko, Ltd. | Laminated rubber stopper |
| US8512691B2 (en) | 1996-12-24 | 2013-08-20 | Biogen Idec Ma Inc. | Stable liquid interferon-beta formulations |
| US9522174B2 (en) | 1996-12-24 | 2016-12-20 | Biogen Ma Inc. | Stable liquid interferon beta formulations |
| US8932574B2 (en) | 1996-12-24 | 2015-01-13 | Biogen Idec Ma Inc. | Stable liquid interferon beta formulations |
| US8512692B2 (en) | 1996-12-24 | 2013-08-20 | Biogen Idec Ma Inc. | Methods of treating multiple sclerosis with stable liquid interferon-beta formulations |
| WO2001022998A1 (en) * | 1999-09-29 | 2001-04-05 | Aaipharma, Inc. | Stable calcitriol solution for packaging in vials |
| US6645635B2 (en) | 2001-01-19 | 2003-11-11 | Daikyo Seiko, Ltd. | Laminated rubber stopper for a medicament vial |
| EP1228973A1 (de) * | 2001-01-19 | 2002-08-07 | Daikyo Seiko, Ltd. | Laminierter Gummipfropfen für Medizinbehälter |
| EP1760002A1 (de) * | 2005-08-31 | 2007-03-07 | West Pharmaceutical Services Deutschland Gmbh & Co. KG | Medikamentenflasche oder dergleichen Behälter |
| EP1792657A1 (de) * | 2005-12-01 | 2007-06-06 | Eppendorf Ag | Deckelgefäss |
| CN105106982A (zh) * | 2006-06-21 | 2015-12-02 | 通用电气健康护理有限公司 | 放射性药物产品 |
| US20090297442A1 (en) * | 2006-06-21 | 2009-12-03 | Stig Hemstad | Radiopharmaceutical products |
| RU2448733C2 (ru) * | 2006-06-21 | 2012-04-27 | Джи-И Хелткер Лимитед | Радиофармацевтические продукты |
| US20090202759A1 (en) * | 2006-07-06 | 2009-08-13 | Arkema Inc. | Flexible multilayer vinylidene fluoride tubes |
| US9056447B2 (en) * | 2006-07-06 | 2015-06-16 | Arkema Inc. | Flexible multilayer vinylidene fluoride tubes |
| US20100206836A1 (en) * | 2007-10-18 | 2010-08-19 | Tsuyoshi Koshidaka | Vial rubber-stopper |
| EA026876B1 (ru) * | 2010-10-18 | 2017-05-31 | Санофи Пастер | Контейнер для хранения вакцины, содержащей адъювант на основе алюминия, и его применение |
| US9066895B2 (en) | 2010-10-18 | 2015-06-30 | Sanofi Pasteur Sa | Method of storing a vaccine containing an aluminum adjuvant |
| WO2012052394A1 (en) * | 2010-10-18 | 2012-04-26 | Sanofi Pasteur | Method of storing a vaccine containing an aluminum adjuvant |
| AU2011317649B2 (en) * | 2010-10-18 | 2014-09-18 | Sanofi Pasteur | Method of storing a vaccine containing an aluminum adjuvant |
| US9938055B2 (en) | 2010-10-18 | 2018-04-10 | Sanofi Pasteur | Method of storing a vaccine containing an aluminum adjuvant |
| FR3082729A1 (fr) | 2018-06-26 | 2019-12-27 | Laboratoire Francais Du Fractionnement Et Des Biotechnologies | Conditions de stockage d'une composition de proteines comprenant du tensioactif et evolution de la teneur en tensioactif |
| WO2020002436A1 (fr) | 2018-06-26 | 2020-01-02 | Laboratoire Francais Du Fractionnement Et Des Biotechnologies | Conditions de stockage d'une composition de proteines comprenant du tensioactif et evolution de la teneur en tensioactif |
| CN113474083A (zh) * | 2019-01-04 | 2021-10-01 | 仪器实验室公司 | 用于高刺穿计数应用的容器塞子 |
| EP4253276A1 (de) * | 2022-03-30 | 2023-10-04 | Datwyler Pharma Packaging Belgium | Verschlussstopfen für medizinische oder pharmazeutische substanzen enthaltende behälter |
| WO2023186979A1 (de) | 2022-03-30 | 2023-10-05 | Datwyler Pharma Packaging Belgium | Verschlussstopfen für medizinische oder pharmazeutische substanzen enthaltende behälter |
| USD1056701S1 (en) | 2022-03-30 | 2025-01-07 | Datwyler Pharma Packaging Belgium | Stopper |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0294127B1 (de) | 1994-11-09 |
| DE3852061T2 (de) | 1995-03-16 |
| EP0294127A3 (en) | 1990-05-30 |
| JPS63296756A (ja) | 1988-12-02 |
| JP2545540B2 (ja) | 1996-10-23 |
| DE3852061D1 (de) | 1994-12-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0294127B1 (de) | Mit Harz laminierter Gummistopfen | |
| EP0264273B1 (de) | Beschichteter Kolben für Spritzen | |
| EP1258234B1 (de) | Kunststoffbehälter für flüssige medikamente | |
| EP0172613A2 (de) | Mit einer aus Kunststoffharz bestehender Schicht versehener Gummistopfen | |
| US6286699B1 (en) | Laminated rubber stopper | |
| US8092878B2 (en) | Cryogenic, elastomeric closure for cryogen containers | |
| EP1228973B1 (de) | Laminierter Gummipfropfen für Medizinbehälter | |
| US5078941A (en) | Process for the production of a resin-laminated rubber closure for a medical vial | |
| CN102123919B (zh) | 制造容器封闭体的方法 | |
| EP0607573B1 (de) | Barriereüberzug | |
| DK2223677T3 (en) | Molded rubber products | |
| US7056971B2 (en) | Essentially gas-impermeable thermoplastic elastomer | |
| JP2504915B2 (ja) | 採血管組立体 | |
| EP0789092A2 (de) | Blutentnahmeröhrchen | |
| EP4197518B1 (de) | Medizinischer gummistopfen | |
| US5219083A (en) | Stopper for reduction of particulate matter | |
| CN110385904A (zh) | 一种橡胶/氟塑料复合的再密封隔垫及其制备方法 | |
| SE434336B (sv) | Farmaceutisk behallare | |
| CA2091020C (en) | Stopper for reduction of particulate matter | |
| DK201670703A1 (en) | Food container | |
| CN102670400A (zh) | 一种注射用药品包装组合物及其制备方法 | |
| JPH0240491B2 (de) | ||
| EP0306820A1 (de) | Kunststoffkappe mit niedriger Sauerstoffdurchlässigkeit sowie Verfahren zu ihrer Herstellung | |
| JP2974883B2 (ja) | 医薬品用容器兼注射器及び滑栓 | |
| JP2647994B2 (ja) | 医薬品容器用ゴム栓 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): DE FR GB IT |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): DE FR GB IT |
|
| RHK1 | Main classification (correction) |
Ipc: B65D 51/00 |
|
| 17P | Request for examination filed |
Effective date: 19901122 |
|
| 17Q | First examination report despatched |
Effective date: 19920406 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| ITF | It: translation for a ep patent filed | ||
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE FR GB IT |
|
| REF | Corresponds to: |
Ref document number: 3852061 Country of ref document: DE Date of ref document: 19941215 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20070524 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20070523 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20070525 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20070510 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: PE20 Expiry date: 20080526 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20080526 |