EP0300737A2 - Dérivés de gènes peptides de calcitonine apparentés - Google Patents
Dérivés de gènes peptides de calcitonine apparentés Download PDFInfo
- Publication number
- EP0300737A2 EP0300737A2 EP88306600A EP88306600A EP0300737A2 EP 0300737 A2 EP0300737 A2 EP 0300737A2 EP 88306600 A EP88306600 A EP 88306600A EP 88306600 A EP88306600 A EP 88306600A EP 0300737 A2 EP0300737 A2 EP 0300737A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- peptide
- group
- asn
- phe
- gly
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 108090000932 Calcitonin Gene-Related Peptide Proteins 0.000 title description 7
- 102000004414 Calcitonin Gene-Related Peptide Human genes 0.000 title description 2
- 150000003839 salts Chemical class 0.000 claims abstract description 8
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 4
- 150000001875 compounds Chemical class 0.000 claims abstract 5
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 47
- 238000000034 method Methods 0.000 claims description 41
- 230000008569 process Effects 0.000 claims description 19
- 125000006239 protecting group Chemical group 0.000 claims description 18
- 125000003396 thiol group Chemical group [H]S* 0.000 claims description 14
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 11
- 238000010532 solid phase synthesis reaction Methods 0.000 claims description 7
- 230000015572 biosynthetic process Effects 0.000 claims description 6
- 238000003786 synthesis reaction Methods 0.000 claims description 6
- 125000000524 functional group Chemical group 0.000 claims description 4
- 125000003368 amide group Chemical group 0.000 claims 2
- 239000003085 diluting agent Substances 0.000 claims 1
- 239000003937 drug carrier Substances 0.000 claims 1
- 239000001257 hydrogen Substances 0.000 claims 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims 1
- 239000008194 pharmaceutical composition Substances 0.000 claims 1
- 239000012071 phase Substances 0.000 claims 1
- 238000002360 preparation method Methods 0.000 claims 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 89
- 150000001413 amino acids Chemical group 0.000 description 45
- 239000000843 powder Substances 0.000 description 45
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 42
- 239000011347 resin Substances 0.000 description 41
- 229920005989 resin Polymers 0.000 description 41
- 229940024606 amino acid Drugs 0.000 description 40
- 235000001014 amino acid Nutrition 0.000 description 38
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 36
- 229960000583 acetic acid Drugs 0.000 description 30
- 235000011054 acetic acid Nutrition 0.000 description 29
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 24
- -1 p-methoxybenzyloxy carbonyl Chemical group 0.000 description 22
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical compound C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 description 20
- 239000000243 solution Substances 0.000 description 18
- QMMFVYPAHWMCMS-UHFFFAOYSA-N Dimethyl sulfide Chemical compound CSC QMMFVYPAHWMCMS-UHFFFAOYSA-N 0.000 description 15
- 239000000872 buffer Substances 0.000 description 15
- 238000010828 elution Methods 0.000 description 14
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 13
- 229910000040 hydrogen fluoride Inorganic materials 0.000 description 13
- 239000000203 mixture Substances 0.000 description 13
- 210000002966 serum Anatomy 0.000 description 12
- 238000006243 chemical reaction Methods 0.000 description 11
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 10
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 10
- BNIILDVGGAEEIG-UHFFFAOYSA-L disodium hydrogen phosphate Chemical compound [Na+].[Na+].OP([O-])([O-])=O BNIILDVGGAEEIG-UHFFFAOYSA-L 0.000 description 10
- 229910000397 disodium phosphate Inorganic materials 0.000 description 10
- 235000019800 disodium phosphate Nutrition 0.000 description 10
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 9
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 9
- 238000004458 analytical method Methods 0.000 description 9
- 239000011575 calcium Substances 0.000 description 9
- 229910052791 calcium Inorganic materials 0.000 description 9
- 229920005654 Sephadex Polymers 0.000 description 8
- 239000012507 Sephadex™ Substances 0.000 description 8
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 8
- 230000001603 reducing effect Effects 0.000 description 8
- BDAGIHXWWSANSR-UHFFFAOYSA-M Formate Chemical compound [O-]C=O BDAGIHXWWSANSR-UHFFFAOYSA-M 0.000 description 7
- ODKSFYDXXFIFQN-BYPYZUCNSA-N L-arginine Chemical compound OC(=O)[C@@H](N)CCCN=C(N)N ODKSFYDXXFIFQN-BYPYZUCNSA-N 0.000 description 7
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical group OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 7
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 7
- 102100038518 Calcitonin Human genes 0.000 description 6
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 6
- 229910019142 PO4 Inorganic materials 0.000 description 6
- 238000006482 condensation reaction Methods 0.000 description 6
- 230000007062 hydrolysis Effects 0.000 description 6
- 238000006460 hydrolysis reaction Methods 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 6
- 239000010452 phosphate Substances 0.000 description 6
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 5
- DHBXNPKRAUYBTH-UHFFFAOYSA-N 1,1-ethanedithiol Chemical compound CC(S)S DHBXNPKRAUYBTH-UHFFFAOYSA-N 0.000 description 5
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Chemical group OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 5
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 5
- PMZXXNPJQYDFJX-UHFFFAOYSA-N acetonitrile;2,2,2-trifluoroacetic acid Chemical compound CC#N.OC(=O)C(F)(F)F PMZXXNPJQYDFJX-UHFFFAOYSA-N 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 238000005859 coupling reaction Methods 0.000 description 5
- 230000014759 maintenance of location Effects 0.000 description 5
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 5
- 230000000704 physical effect Effects 0.000 description 5
- 229920001467 poly(styrenesulfonates) Polymers 0.000 description 5
- 238000004007 reversed phase HPLC Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- DKIDEFUBRARXTE-UHFFFAOYSA-N 3-mercaptopropanoic acid Chemical compound OC(=O)CCS DKIDEFUBRARXTE-UHFFFAOYSA-N 0.000 description 4
- USFZMSVCRYTOJT-UHFFFAOYSA-N Ammonium acetate Chemical compound N.CC(O)=O USFZMSVCRYTOJT-UHFFFAOYSA-N 0.000 description 4
- 239000005695 Ammonium acetate Substances 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 4
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 4
- RHGKLRLOHDJJDR-BYPYZUCNSA-N L-citrulline Chemical compound NC(=O)NCCC[C@H]([NH3+])C([O-])=O RHGKLRLOHDJJDR-BYPYZUCNSA-N 0.000 description 4
- 125000000415 L-cysteinyl group Chemical group O=C([*])[C@@](N([H])[H])([H])C([H])([H])S[H] 0.000 description 4
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 4
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 4
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 4
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 4
- RHGKLRLOHDJJDR-UHFFFAOYSA-N Ndelta-carbamoyl-DL-ornithine Natural products OC(=O)C(N)CCCNC(N)=O RHGKLRLOHDJJDR-UHFFFAOYSA-N 0.000 description 4
- 108010033276 Peptide Fragments Proteins 0.000 description 4
- 102000007079 Peptide Fragments Human genes 0.000 description 4
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Chemical compound CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 125000003277 amino group Chemical group 0.000 description 4
- 235000019257 ammonium acetate Nutrition 0.000 description 4
- 229940043376 ammonium acetate Drugs 0.000 description 4
- 239000004202 carbamide Substances 0.000 description 4
- 235000013477 citrulline Nutrition 0.000 description 4
- 229960002173 citrulline Drugs 0.000 description 4
- 238000009833 condensation Methods 0.000 description 4
- 230000005494 condensation Effects 0.000 description 4
- 229910001873 dinitrogen Inorganic materials 0.000 description 4
- VHJLVAABSRFDPM-QWWZWVQMSA-N dithiothreitol Chemical compound SC[C@@H](O)[C@H](O)CS VHJLVAABSRFDPM-QWWZWVQMSA-N 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 230000007071 enzymatic hydrolysis Effects 0.000 description 4
- 238000006047 enzymatic hydrolysis reaction Methods 0.000 description 4
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 4
- 238000010647 peptide synthesis reaction Methods 0.000 description 4
- 239000007790 solid phase Substances 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 241000700159 Rattus Species 0.000 description 3
- 238000005903 acid hydrolysis reaction Methods 0.000 description 3
- 150000001540 azides Chemical class 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 239000012153 distilled water Substances 0.000 description 3
- 150000002148 esters Chemical class 0.000 description 3
- 125000002795 guanidino group Chemical group C(N)(=N)N* 0.000 description 3
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 3
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 3
- UHPQFNXOFFPHJW-UHFFFAOYSA-N (4-methylphenyl)-phenylmethanamine Chemical compound C1=CC(C)=CC=C1C(N)C1=CC=CC=C1 UHPQFNXOFFPHJW-UHFFFAOYSA-N 0.000 description 2
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 2
- 125000001433 C-terminal amino-acid group Chemical group 0.000 description 2
- 208000020446 Cardiac disease Diseases 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 2
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 2
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 2
- 239000004472 Lysine Substances 0.000 description 2
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 2
- 239000004473 Threonine Substances 0.000 description 2
- 208000025865 Ulcer Diseases 0.000 description 2
- 150000008065 acid anhydrides Chemical class 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 229960005261 aspartic acid Drugs 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 2
- 210000004899 c-terminal region Anatomy 0.000 description 2
- 150000001718 carbodiimides Chemical class 0.000 description 2
- PFKFTWBEEFSNDU-UHFFFAOYSA-N carbonyldiimidazole Chemical compound C1=CN=CN1C(=O)N1C=CN=C1 PFKFTWBEEFSNDU-UHFFFAOYSA-N 0.000 description 2
- 230000002490 cerebral effect Effects 0.000 description 2
- 238000010168 coupling process Methods 0.000 description 2
- 235000019253 formic acid Nutrition 0.000 description 2
- 229960002989 glutamic acid Drugs 0.000 description 2
- 208000019622 heart disease Diseases 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 239000007791 liquid phase Substances 0.000 description 2
- 208000030159 metabolic disease Diseases 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- UYWQUFXKFGHYNT-UHFFFAOYSA-N phenylmethyl ester of formic acid Natural products O=COCC1=CC=CC=C1 UYWQUFXKFGHYNT-UHFFFAOYSA-N 0.000 description 2
- 230000002035 prolonged effect Effects 0.000 description 2
- 229960001153 serine Drugs 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 229960002898 threonine Drugs 0.000 description 2
- 125000002088 tosyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])[H])S(*)(=O)=O 0.000 description 2
- ITMCEJHCFYSIIV-UHFFFAOYSA-N triflic acid Chemical compound OS(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-N 0.000 description 2
- 231100000397 ulcer Toxicity 0.000 description 2
- IMVJCTWESJIARS-QWWZWVQMSA-N (2r,3r)-2,3-bis(sulfanyl)butane-1,4-diol Chemical compound OC[C@@H](S)[C@H](S)CO IMVJCTWESJIARS-QWWZWVQMSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- JNVXRQOSRUDXDY-UHFFFAOYSA-N 1,1-diiodoethane Chemical compound CC(I)I JNVXRQOSRUDXDY-UHFFFAOYSA-N 0.000 description 1
- ADFXKUOMJKEIND-UHFFFAOYSA-N 1,3-dicyclohexylurea Chemical compound C1CCCCC1NC(=O)NC1CCCCC1 ADFXKUOMJKEIND-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- CKLJMWTZIZZHCS-UHFFFAOYSA-N D-OH-Asp Natural products OC(=O)C(N)CC(O)=O CKLJMWTZIZZHCS-UHFFFAOYSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-UHFFFAOYSA-N D-alpha-Ala Natural products CC([NH3+])C([O-])=O QNAYBMKLOCPYGJ-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 101000741445 Homo sapiens Calcitonin Proteins 0.000 description 1
- QNAYBMKLOCPYGJ-UWTATZPHSA-N L-Alanine Natural products C[C@@H](N)C(O)=O QNAYBMKLOCPYGJ-UWTATZPHSA-N 0.000 description 1
- CKLJMWTZIZZHCS-UWTATZPHSA-N L-Aspartic acid Natural products OC(=O)[C@H](N)CC(O)=O CKLJMWTZIZZHCS-UWTATZPHSA-N 0.000 description 1
- 235000019766 L-Lysine Nutrition 0.000 description 1
- FFEARJCKVFRZRR-UHFFFAOYSA-N L-Methionine Natural products CSCCC(N)C(O)=O FFEARJCKVFRZRR-UHFFFAOYSA-N 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 1
- 125000003412 L-alanyl group Chemical group [H]N([H])[C@@](C([H])([H])[H])(C(=O)[*])[H] 0.000 description 1
- 229930064664 L-arginine Natural products 0.000 description 1
- 235000014852 L-arginine Nutrition 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- 239000004201 L-cysteine Substances 0.000 description 1
- 235000013878 L-cysteine Nutrition 0.000 description 1
- 239000004395 L-leucine Substances 0.000 description 1
- 235000019454 L-leucine Nutrition 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- 229930195722 L-methionine Natural products 0.000 description 1
- 229930182821 L-proline Natural products 0.000 description 1
- 108090000189 Neuropeptides Proteins 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 230000003187 abdominal effect Effects 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 229960003767 alanine Drugs 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 238000007098 aminolysis reaction Methods 0.000 description 1
- 150000008064 anhydrides Chemical class 0.000 description 1
- 210000002376 aorta thoracic Anatomy 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 235000009697 arginine Nutrition 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- 125000004744 butyloxycarbonyl group Chemical group 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 229940105329 carboxymethylcellulose Drugs 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000007979 citrate buffer Substances 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- MGHPNCMVUAKAIE-UHFFFAOYSA-N diphenylmethanamine Chemical compound C=1C=CC=CC=1C(N)C1=CC=CC=C1 MGHPNCMVUAKAIE-UHFFFAOYSA-N 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 238000002523 gelfiltration Methods 0.000 description 1
- 239000012362 glacial acetic acid Substances 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 229960002885 histidine Drugs 0.000 description 1
- 229940045644 human calcitonin Drugs 0.000 description 1
- 238000006698 hydrazinolysis reaction Methods 0.000 description 1
- JBFYUZGYRGXSFL-UHFFFAOYSA-N imidazolide Chemical compound C1=C[N-]C=N1 JBFYUZGYRGXSFL-UHFFFAOYSA-N 0.000 description 1
- 229910052816 inorganic phosphate Inorganic materials 0.000 description 1
- 239000000543 intermediate Substances 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 229960003136 leucine Drugs 0.000 description 1
- 235000018977 lysine Nutrition 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229960004452 methionine Drugs 0.000 description 1
- XELZGAJCZANUQH-UHFFFAOYSA-N methyl 1-acetylthieno[3,2-c]pyrazole-5-carboxylate Chemical compound CC(=O)N1N=CC2=C1C=C(C(=O)OC)S2 XELZGAJCZANUQH-UHFFFAOYSA-N 0.000 description 1
- 150000004702 methyl esters Chemical class 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 125000000636 p-nitrophenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)[N+]([O-])=O 0.000 description 1
- 229960005190 phenylalanine Drugs 0.000 description 1
- 125000000405 phenylalanyl group Chemical group 0.000 description 1
- NMHMNPHRMNGLLB-UHFFFAOYSA-N phloretic acid Chemical compound OC(=O)CCC1=CC=C(O)C=C1 NMHMNPHRMNGLLB-UHFFFAOYSA-N 0.000 description 1
- 229960002429 proline Drugs 0.000 description 1
- 235000018102 proteins Nutrition 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 230000006340 racemization Effects 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000006722 reduction reaction Methods 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 238000003746 solid phase reaction Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000002798 spectrophotometry method Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 229960004295 valine Drugs 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/57527—Calcitonin gene related peptide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/12—Drugs for disorders of the metabolism for electrolyte homeostasis
- A61P3/14—Drugs for disorders of the metabolism for electrolyte homeostasis for calcium homeostasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/55—Design of synthesis routes, e.g. reducing the use of auxiliary or protecting groups
Definitions
- This invention relates to novel human calcitonin gene related peptide (hereinafter designated as h-CGRP) derivatives, useful for the treatment of calcium metabolic disorders, cardiac disease and ulcers, and for the improvement of cerebral circulation.
- h-CGRP human calcitonin gene related peptide
- the h-CGRP has an amino acid sequence of the formula wherein A3 is Asp or Asn, A22 is Val or Met, and A25 is Asn or Ser, and is known to have a various biological proterties [Nature, 308( 19): 746-748 (1984), FEBS Letters, 183 (2): 403 (1985), Neuropeptides, 4 : 425-434 (1984) and Nature, 313 (3): 5456 (1984) ].
- An object of the present invention is to provide novel h-CGRP derivatives.
- the present invention provides novel h-CGRP derivatives of the formula wherein R is H or H-Ala-NH- and when A is Asp, B is Asp or Glu, C is Leu and D is Val; when A is Asn and B is Gly, C is Phe and D is Gly; when A is Asn, B is Asp or Glu and C is Leu, D is Gly;and when A is Asn, B is Asp or Glu, and C is Phe, D is Val, or pharmaceutically acceptable salt thereof.
- a peptice (1) of the present invention can be synthesized by a known conventional process for peptide synthesis.
- a peptide (1) is synthesized by converting a carboxyl in phenylalanyl in the C-terminal to an amide, condensing successively a protected amino acid and/or a protected lower peptide in the order of amino acid sequence as shown in the formula (1), when R is H, removing the protective group for L-cysteinyl and mercapto in ⁇ -mercaptropropionyl group and the protective group for the functional group in the other side chain by acid hydrolysis, and when R is H-Ala-NH-, removing the protective group for mercapto in L-cysteinyl and theprotective group for the functional group in the other side chain by acid hydrolysis, and oxidizing L-cysteinyl ( or ⁇ -mercaptopropionylgroup ) and mercapto in L-cysteinyl to form a disulfide bridge at a final satge of the condensation reaction.
- the condensation reaction can be effected by repeating the removal and addition of a protective group and the condensation reaction according to the process for conventional peptide synthesis.
- a protective group for a starting material and intermediates used in the process for the production of a peptide (1) of the present invention is a known protective group in peptide chemistry, for example a protective group which can be easily removed by a known procedure such as hydrolysis, acid decomposition, reduction, aminolysis or hydrazinolysis.
- the protective groups are well known in the literature of peptide chemistry.
- Examples of preferred protective groups are butyloxy carbonyl, benzyloxy carbonyl or p-methoxybenzyloxy carbonyl for a ⁇ -amino group, benzyloxy carbonyl or p-chlorobenzyloxycarbonyl for a side chain amino group such as ⁇ -amino group in lysine, a methyl ester or benzyl ester for an ⁇ -carboxyl group, benzyl ester for a side chain carboxyl group such as the side chain carboxyl in aspartic acid or glutamic acid, benzyl for a hydroxyl group in serine or threonine, mesitylene-2-sulfonyl or tosyl for the amino group of guanidino in arginine and p-methoxylbenzyl or acetamidomethyl for the mercapto group of cysteine or ⁇ -mercaptopropionic acid.
- each amino acid and/or lower peptide can be effected by reacting an amino acid or a lower peptide having protcted ⁇ -amino and activated terminal ⁇ -carboxyl, with an amino acid or a lower peptide having free ⁇ -amino and a protected terminal carboxyl group, or alternatively by reacting an amino acid or a lower peptide having activated ⁇ -amino and protected terminal carboxyl with an amino acid or a lower peptide having a protected ⁇ -amino and free terminal carboxyl group.
- a carboxyl group can be activated by conversion, for example to acid azide, acid anhydride, acid imidazolide or an activated ester such as cyanomethyl ester, p-nitrophenyl esteror N-hydroxysuccinimide ester. Furthermore, it can be activated by using a condensation reagent, for example a carbodiimide such as N, N′-dicyclohexyl-carbodiimide (DCC), N-ethyl-N′-3-dimethylaminopropyl-carbodiimide or N,N′-carbonyldiimidazole.
- a condensation reagent for example a carbodiimide such as N, N′-dicyclohexyl-carbodiimide (DCC), N-ethyl-N′-3-dimethylaminopropyl-carbodiimide or N,N′-carbonyldiimidazole.
- Examples of prefrred condensation reactions used in the present invention are the azide method, the activated ester method, the mixed anhydride method and the carbodiimide method.
- the condensation reaction it is preferred to avoid or at least to minimize the racemization reaction; and preferred such processes are the azide method, the activated ester method, the Wunsch Method [Z. Naturforsch., 21b, 426 (1966)] or the Geiger method [Chem. Ber., 103, 788 (1970)].
- a peptide (1) can be synthesized in any procedure : however, it is preferable to construct an amino acid sequence by connecting amino acid and/or lower peptide in order from the C-terminal.
- a peptide (1) wherein R is H can be obtaianed by removing the protective groups in the protected peptide chain, namely ⁇ -mercaptopropionyl pentatriaconta-peptideamide having protected ⁇ -amino, side chain carboxyl, hydroxyl, guanidino and mercapto groups.
- These protective groups are preferably removed by onestep removal with acid hydrolysis using for example trifluoro-methane sulfonic acid or anhydrous hydrogen fluoride to obtain ⁇ -mercaptopropionyl pentatriaconta-peptideamide having free mercapto group.
- a peptide (1) wherein R is H-Ala-NH- can be obtained by removing the protective groups in the protected peptide chain, namely heptatriaconta-peptideamide having protected ⁇ -amino, side chain carboxyl, hydroxyl, guanidino and mercapto groups. These protective groups are preferably removed by the same way as above to obtain heptatriaconta-peptideamide having free mercapto group.
- an inner molecular disulfide linkage is formed by oxidation to obtain the peptide (1).
- Disulfide linkage can be effected generally by oxidation with oxygen in water, diiodo-ethane in an organic solvent, iodine in glacial acetic acid or potassium ferricyanide in water.
- a solid phase peptide synthesis method can be used in part or in total as the process of peptide (1) synthesis.
- peptide fragment (3-37) [hereinafter peptide of amino acid sequence constituted of amino acids from Nos. 3 to 37 is abbreviated as peptide fragment (3-37) or peptide (3.37)] is synthesized by a solid phase method, and the ⁇ -amino group in the said peptide is acylatd with ⁇ -mercaptopropionic acid to obtain a protected pentatriaconta-peptide-bound resin or a protected heptatriaconta-peptide-bound resin.
- a peptide (1) can be obtained by constructing an inner molecule disulfide bond as set forth in the process by liquid phase synthesis hereinabove.
- resin used in the solid phase method are conventional resins such as benzhydrylamine resin or p-methyl-benzhydrylamine resin.
- a resin with the desired functional equivalent or cross-linkage can be synthesized but is also commercially available.
- an amino acid is condensed in the resin, in the order of amino acid sequence of formula (1), from C-terminal amino acid to the third number of amino acid (amino acid No. 3) or from C-terminal amino acid to the 1st number of amino acid (amino acid No.1), and in the case of a peptide wherein R is H, it is acylated with ⁇ -mercaptopropionic acid at the condensation to the third amino acid.
- a functional group in the amino acid isprotected by a known method. Examples of protective groups are set forth hereinbefore.
- a resin in a reaction vessel is swelled by adding dichloromethane, chloroform, dimethylformamide, benzene or a solvent for swelling the resin, in a ratio of 2-20 ml solvent per 1 g resin.
- 1-6 equivalents of t-butyloxycarbonyl (hereinafter designated Boc-) amino acid per 1 equivalent of amino group in the resin are first reacted with DCC, and the obtained acid anhydride, which is separated from a by-product dicyclohexylurea (hereinafter designatd DCU), is added to the resin hereinabove.
- the amount of condensation agent (DCC) is 0.5-3 equivalents per 1 equivalent of Boc-amino acid. The reaction proceeds generally for 5-60 minutes.
- the coupling amount of amino acid or peptide can be determined according to a conventional method [T. Fairwell et al., Biochemistry, 22 : 2691 (1983) ] by checking the amount of Boc-amino acid upon sampling the Boc-amino acid-resin or Boc-peptide-resin obtained in each process.
- the protective group for the ⁇ -amino group, Boc is removed by an acid such as trifluoroacetic acid and the condensation reaction is performed.
- An automatic solid phase synthesizer can be used; however, a manual procedure can also be used.
- the entire operation is preferably performed under a nitrogen gas atmosphere.
- a peptide fragment (3-37) bound to resin is acylated with ⁇ -mercaptopropionic acid in the final step to obtain a ⁇ -mercaptopropionyl-protected pentatriaconta peptide-amide-bound resin.
- the thus-obtained protected pentatriaconta peptideamide-bound resin or protected heptatriaconta peptide amide-bound resin is, as previously described, treated with anhydrous hydrogen fluoride to remove the protective group and resin in one-step, whereby ⁇ -mercaptopropionyl pentatriaconta peptide-amide having a free mercapto group or heptatriaconta peptide-amide having a free mercapto group can be obtained.
- a peptide (1) can be obtained by forming intra molecular disulfide bonds in the above peptide-amide. Isolation and purification:
- the thus-obtained peptide (1) can be purified by aurification methods which are well known in peptide or protein chemistry.
- aurification methods which are well known in peptide or protein chemistry.
- a gel-filtration method can be carried out using Sephadex G-25, Sephadex G-50 or Sephadex LH-20 (trade names), or ion-exchange chromatography using carboxy methyl cellulose, or another resin, or HPLC can be used.
- a peptide (1) of the present invention can be obtained according to the process in the form of free base or salt.
- a salt with a known organic acid such as acetic acid, citric acid, malic acid or tartaric acid can be prepared.
- Serum calcium and serum phosphate reducing activities Serum calcium and serum phosphate reducing activities:
- the peptide (1) of the present invention and known h-CGRP (each 80 ⁇ g) dissolved in citrate buffer, pH.6.5, containing 0.1% bovine serum albumin (hereinafter designated dissolving medium) (1 ml) were administered intravenously into the tail vein of Wistar rats, body weight 80-90 g, 5-6 rats in one group, at 80 ⁇ g /kg. After 30 and 60 mins. of administration, blood samples were collected from the abdominal descending aorta. The serum calcium concentration was measusred by atomic adsorption spectrophotometry. Serum phosphate was measured by a method according to Goldenberg et al. [Clin. Chem., 12: 872-882 (1966)].
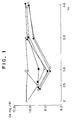
- Figure 1 is a graph showing the rat serum calcium reducing effect of h-CGRP and peptide (1) of the present invention
- Figure 2 is a graph showing the rat serum inorganic phosphate reducing effect of h-CGRP and peptide (1) of the present invention.
- the concentrations of serum calcium and phosphate were reduced by more than 20-30% in desalanyl-deamino- [Asp14 ] h-CGRP(2-37), 80 ⁇ g /kg, administered group [ - ⁇ - ] , [Asp14] h-CGRP(1-37) [ - ⁇ - ] , [Asn3,Phe15,Gly23] h-CGRP(1-37) [ - ⁇ - ] , [Asn3,Asp14, Gly23 ] h-CGRP(1-37) [ - ⁇ - ] , and [Asn3,Asp14,Phe15 ] h-CGRP(1-37) [ - ⁇ - ] as compared with control group (dissolving medium administered group) [ - X - ] and these activities were observed to continue after 2 hours. These activities were stronger and those of h-CGRP [ - ⁇ - ]
- the peptide (1) of the present invention has stronger serum calcium reducing activity and serum phosphate reducing activity as comparaed with known h-CGRP. Furhtermore, it has longer-lasting activity as compared with known h-CGRP. Moreover, it is useful for treatment of calcium metabolic disorders, cardiac disease and ulcers, or for the improvement of cerebral circulation.
- This powder (200 mg) was dissolved in 50 mM Na2HPO4 buffer (pH 7.5, 15 ml) containing 8 Murea and 5mM dithiothreitol, and stirred for 1 hour, then diluted with 50 mM Na2HPO4 buffer (pH 7.5, 1300 ml), thereafter 20 mM K3Fe(CN)6 aqueous solution (9 ml) was added thereto.
- Residual powder 200 mg was treated by the same way to obtain furhter white poentrée (60 mg).
- Combined powder was charged on a column (2.5 x 25 cm) of Cm-Toyopearl 650 M (trademark), and eluted by linear gradient elution with 0.1 N aq. acetic acid (700 ml) ⁇ 0.4 M aq. ammonium acetate (pH 5, 700 ml).
- Fractions (each 10 ml) Nos. 105-123 were collected and freeze dried to obtain white powder.
- Buffer 0.1% TFA-acetonitrile (a gradient elution with acetonitrile concentration from 28 to 32% for 20 minutes)
- Solid phase peptide synthesizer 430-A peptide synthesizer Applied Biosystems Inc.
- MBHA-resin (Applied Biosystems Inc., amino group: 0.48mM/g) (1.04 g) in a raction vessel for solid phase peptide synthesis was treated with DCM (8 ml) (4 times, each 1 min.), DCM solution (8 ml) containing 60% TFA (20 min.), DCM (4 ml) (3 times, each 15 sec.), DMF solution (3 ml) containing DIEA (1 ml) (2 times, each 1 min.) and DMF (8 ml) (6 times, each 40 sec.),in this order, under a nitrogen gas atmosphere with stirring. The material was filtered after each treatment.
- Boc-Phe-MBHA-resin hereinabove was washed 4 times with DCM (8 ml, each 1 min.) in the reaction vessel and filtered. 40% DCM solution (8 ml) conataining 60% TFA was added thereto and stirred for 20 min. to remove Boc. The thus-obtained resin was washed 3 times with DCM (4 ml, each 15 sec.), 2 times with DMF solution (3 ml) containing DIEA (1 ml) (each 1 min.) and 6 times with DMF (8 ml, each 40 sec.), in this order, and filtered.
- amino acids (sequence from Nos. 1 to 35) were subjected to a coupling reaction and at a final step of the raction Boc was removed by DCM solution containing 60% TFA, then washed with DCM, DIEA and DMF to obtain protected [Asn3, Phe15,Gly23 ] h-CGRP(1-37)-MBHA-resin.
- the protected amino acids used in the process are as follows:
- the powder (152 mg) was dissolved in 50 mM Na2HPO4 buffer (pH 7.5, 10 ml) containing 8 M urea and 5 mM dithiothreitol, and stirred for 1 hour, then diluted with 50 mM Na2HPO4 buffer (pH 7.5, 1125 ml), thereafter 20 mM K3Fe(CN)6 aqueous solution (8 ml) was added thereto.
- Residual powder (154 mg) was treated by the same way to obtain furhter white powder (40 mg). Combined powder was charged on a column (2.5 x 20 cm) of CM-Toyopearl 650 M (trademark), and eluted by linear gradient elution with distilled water (700 ml) ⁇ 0.4 M aq. ammonium acetate (pH 5,700 ml). Fractions (each 10 ml) Nos. 108-116 were collected and freeze dried to obtain a white powder. The thus obtained white powder dissolved in 0.1 M acetic acid was charged on a column of Sephadex G-25 Fine (trade name) (2.6x 90 cm) and eluted with 0.1 M acetic acid.
- Sephadex G-25 Fine trade name
- Buffer 0.1% TFA-acetonitrile (a gradient elution with acetonitrile concentration from 28 to 32% for 25 minutes)
- Arg is analysed by citrulline.
- the protected amino acids used in the process are as follows:
- This powder (150 mg) was dissolved in 50 mM Na2HPO4 buffer (pH 7.5, 10 ml) containing 8 M urea and 5 mM dithiothreitol, and stirred for 1 hour at room temperature, then diluted with 50 mM Na2HPO4 buffer (pH 7.5, 1125 ml), there-after 20 mM K3Fe(CN)6 aqueous solution (8 ml) was added thereto.
- Buffer 0.1% TFA-acetonitrile (a gradient elution with acetonitrile concentration from 30 to 33% for 20 minutes)
- Arg is analysed by citrulline.
- the protected amino acids used in the process are as follows:
- This powder was dissolved in 50 mM Na2HPO4 buffer (pH 7.5, 20 ml) containing 8 M urea and 5mM dithiothreitol, and stirred for 1 hour, then diluted with 50 mM Na2HPO4 buffer (pH 7.5, 2000 ml), thereafter 20 mM K3Fe(CN)6 aqueous solution (10 ml) was added thereto.
- Buffer 0.1% TFA-acetonitrile (a gradient elution with acetonitrile concentration from 29 to 34% for 30 minutes).
- Arg is analysed by citrulline.
- the protected amino acids used in the process are as follows:
- This powder was dissolved in 50 mM Na2HPO4 buffer (pH 7.5, 20 ml) containing 8 M urea and 5 mM dithiothreitol, and stirred at room temperature for 1 hour, then diluted with 50 mM Na2HPO4 buffer (pH 7.5, 2000 ml), thereafter 20 mM K3Fe(CN)6 aqueous solution (12 ml) was added thereto.
- Buffer 0.1% TFA-acetonitrile (a gradient elution with acetonitrile concentration from 31.5 to 32.5% for 30 minutes
- Amino acids (sequence from Nos. 3 to 37) were subjected to a coupling reaction by the solid phase synthesis as same as of in Example 1 and the peptide was acylated with MBzl- ⁇ -mercaptopropionic acid at the final stage of reaction to obtain protected-desalanyl-deamino [Asp14] h-CGRP (2-37)-MBHA-resin.
- the protected amino acids used in the process are as follows:
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Endocrinology (AREA)
- Animal Behavior & Ethology (AREA)
- Hematology (AREA)
- Biochemistry (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Zoology (AREA)
- Toxicology (AREA)
- Gastroenterology & Hepatology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Diabetes (AREA)
- Biophysics (AREA)
- Obesity (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Rheumatology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP62182891A JPS6426598A (en) | 1987-07-22 | 1987-07-22 | Calcitonin gene related peptide derivative |
| JP182891/87 | 1987-07-22 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0300737A2 true EP0300737A2 (fr) | 1989-01-25 |
| EP0300737A3 EP0300737A3 (en) | 1990-02-28 |
| EP0300737B1 EP0300737B1 (fr) | 1993-04-14 |
Family
ID=16126205
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP88306600A Expired - Lifetime EP0300737B1 (fr) | 1987-07-22 | 1988-07-19 | Dérivés de gènes peptides de calcitonine apparentés |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US4992530A (fr) |
| EP (1) | EP0300737B1 (fr) |
| JP (1) | JPS6426598A (fr) |
| DE (1) | DE3880226T2 (fr) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0385712A3 (fr) * | 1989-02-28 | 1991-02-06 | Asahi Kasei Kogyo Kabushiki Kaisha | Peptide utilisable dans la préparation d'une composition en vue de la prévention ou du traitement d'états susceptibles d'être traités par un inhibiteur de l'agrégation plaquettaire |
| EP0672419A4 (fr) * | 1991-12-12 | 1995-06-22 | Asahi Chemical Ind | Regulateur de la pression sanguine. |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1992015317A1 (fr) * | 1991-03-08 | 1992-09-17 | Amylin Pharmaceuticals, Inc. | Preparation synthetique d'amyline et d'analogues d'amyline |
| HU222249B1 (hu) * | 1991-03-08 | 2003-05-28 | Amylin Pharmaceuticals Inc. | Eljárás amilin agonista peptidszármazékok és ezeket tartalmazó gyógyszerkészítmények előállítására |
| US5567679A (en) * | 1993-12-13 | 1996-10-22 | Daly; Theodore J. | Use of CGRP in treating alopecia |
| CA2292902C (fr) * | 1999-12-24 | 2012-12-18 | Alain Cadieux | Utilisation de peptide calcitonine lie aux genes dans la prevention et le soulagement de l'asthme et d'affections bronchospasmodiques connexes |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS62129297A (ja) * | 1985-08-09 | 1987-06-11 | Toyo Jozo Co Ltd | カルシトニン遺伝子関連ペプチド誘導体 |
-
1987
- 1987-07-22 JP JP62182891A patent/JPS6426598A/ja active Pending
-
1988
- 1988-07-19 EP EP88306600A patent/EP0300737B1/fr not_active Expired - Lifetime
- 1988-07-19 DE DE8888306600T patent/DE3880226T2/de not_active Expired - Fee Related
- 1988-07-21 US US07/222,761 patent/US4992530A/en not_active Expired - Fee Related
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0385712A3 (fr) * | 1989-02-28 | 1991-02-06 | Asahi Kasei Kogyo Kabushiki Kaisha | Peptide utilisable dans la préparation d'une composition en vue de la prévention ou du traitement d'états susceptibles d'être traités par un inhibiteur de l'agrégation plaquettaire |
| EP0672419A4 (fr) * | 1991-12-12 | 1995-06-22 | Asahi Chemical Ind | Regulateur de la pression sanguine. |
Also Published As
| Publication number | Publication date |
|---|---|
| US4992530A (en) | 1991-02-12 |
| DE3880226T2 (de) | 1993-07-29 |
| EP0300737B1 (fr) | 1993-04-14 |
| JPS6426598A (en) | 1989-01-27 |
| DE3880226D1 (de) | 1993-05-19 |
| EP0300737A3 (en) | 1990-02-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5049654A (en) | Calcitonin gene related peptide derivatives | |
| US4607023A (en) | Natriuretic | |
| EP0212432B1 (fr) | Dérivés peptide apparentés au gène de la calcitonine | |
| US5175146A (en) | Synthetic calcitonin peptides | |
| JPH0532696A (ja) | 副甲状腺ホルモン誘導体 | |
| HU191263B (en) | Process for producing factor for promoting letting out growth hormone of human pancreatic origine | |
| JP2001527507A (ja) | 改良された環状crf拮抗剤 | |
| HU206126B (en) | Process for producing growth hormone releasing factor derivatives | |
| EP0136475B1 (fr) | Peptides libérant de l'hormone de croissance et méthode pour le traitement de mammifères | |
| JPH085916B2 (ja) | 新規カルシトニン誘導体 | |
| US4606856A (en) | [Des-1-amino, 8-glycine]calcitonin | |
| US4537716A (en) | Des-serine2 or des-glycine2 -leucine22 calcitonin | |
| EP0188214A2 (fr) | Peptides libérant l'hormone de croissance et méthodes de traitement de mammifères avec ces peptides | |
| US4824937A (en) | Synthetic natriuretic peptides | |
| EP0300737B1 (fr) | Dérivés de gènes peptides de calcitonine apparentés | |
| EP0315687B1 (fr) | (n-alpha-acyl, 8-glycine, des-19-leucine)-calcitonine | |
| IE53488B1 (en) | Crf and analogues | |
| JPH051798B2 (fr) | ||
| JP2678993B2 (ja) | 新規なアルキル化された成長ホルモン放出ペプチド及びそれにより哺乳動物を処理する方法 | |
| US4820804A (en) | Analogs of [1,7-di-alanine, des-19-leucine]calcitonin | |
| US6127519A (en) | Calcitonin derivatives | |
| US4639509A (en) | [16,19-Di-alanine] calcitonin | |
| EP0370165B1 (fr) | Dérivés de calcitonine et leurs sels | |
| JPH0421699A (ja) | カルシトニン遺伝子関連ペプチド誘導体 | |
| JP3294695B2 (ja) | エルカトニン水溶液剤の振とう安定化剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): DE FR GB IT |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): DE FR GB IT |
|
| 17P | Request for examination filed |
Effective date: 19900529 |
|
| 17Q | First examination report despatched |
Effective date: 19920527 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: ASAHI KASEI KOGYO KABUSHIKI KAISHA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE FR GB IT |
|
| ET | Fr: translation filed | ||
| ITF | It: translation for a ep patent filed | ||
| REF | Corresponds to: |
Ref document number: 3880226 Country of ref document: DE Date of ref document: 19930519 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19960709 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19960710 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19960726 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19970719 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19970719 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19980331 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19980401 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050719 |