EP0433220B1 - Naphtalocyanine substituée et son utilisation - Google Patents
Naphtalocyanine substituée et son utilisation Download PDFInfo
- Publication number
- EP0433220B1 EP0433220B1 EP90810875A EP90810875A EP0433220B1 EP 0433220 B1 EP0433220 B1 EP 0433220B1 EP 90810875 A EP90810875 A EP 90810875A EP 90810875 A EP90810875 A EP 90810875A EP 0433220 B1 EP0433220 B1 EP 0433220B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- compound
- formula
- compound according
- integer
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 C*[N+]([N-][N-]1)N=N[N+]1N=O Chemical compound C*[N+]([N-][N-]1)N=N[N+]1N=O 0.000 description 1
Classifications
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/242—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers
- G11B7/244—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only
- G11B7/246—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only containing dyes
- G11B7/248—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only containing dyes porphines; azaporphines, e.g. phthalocyanines
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B47/00—Porphines; Azaporphines
- C09B47/04—Phthalocyanines abbreviation: Pc
- C09B47/06—Preparation from carboxylic acids or derivatives thereof, e.g. anhydrides, amides, mononitriles, phthalimide, o-cyanobenzamide
- C09B47/067—Preparation from carboxylic acids or derivatives thereof, e.g. anhydrides, amides, mononitriles, phthalimide, o-cyanobenzamide from phthalodinitriles naphthalenedinitriles, aromatic dinitriles prepared in situ, hydrogenated phthalodinitrile
- C09B47/0675—Preparation from carboxylic acids or derivatives thereof, e.g. anhydrides, amides, mononitriles, phthalimide, o-cyanobenzamide from phthalodinitriles naphthalenedinitriles, aromatic dinitriles prepared in situ, hydrogenated phthalodinitrile having oxygen or sulfur linked directly to the skeleton
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S430/00—Radiation imagery chemistry: process, composition, or product thereof
- Y10S430/146—Laser beam
Definitions
- the invention relates to tetra- (2,3-naphtho) -tetraazaporphins (polyoxaalkylene naphthalocyanines) substituted in the 1,4-position with polyoxyalkylene groups, a process for their preparation, a composition from a substrate and a compound according to the invention, and the use of the composition as an optical Information recording material.
- GB-A-2 168 372 describes octaalkoxyphthalocyanines and naphthalocyanines and in GB-A-2 200 650 corresponding metal salts.
- the disclosed compounds can be dissolved in liquid crystals and used in electro-optical display systems.
- J. Chem. Soc. Perkin Trans. I, pp. 2453-2458 (1988) describe MJ Cook et al. Octaalkoxynaphthalocyanine and corresponding copper salts.
- the dyes absorb radiation in the near infrared range and can be used for laser marking or for image projections with IR lasers.
- the present invention relates to compounds of the formula I. wherein R the group represents and M represents two H or Cu or Ni, R1-OH, -Cl, -Br, C1-C30-alkoxy or -OC (O) R2 means m for an integer from 2 to 6 and n for one Are from 1 to 20, and R2 is C1-C20-alkyl, C2-C20-alkenyl, cyclopentyl, cyclohexyl, unsubstituted or substituted by C1-C18-alkyl, C6-C10-aryl or C6-C10-arylmethyl.
- M preferably represents Ni.
- n preferably represents 1 to 10, particularly 1 to 5.
- the group -C m H 2m - can be, for example, ethylene, 1,2- or 1,4-propylene, 1,2-, 1,3-, 1,4- or 2,3-butylene, 1, 2-, 1,3-, 1,4-, 1,5- or 2,3-pentylene and 1,2-, 1,3-, 1,4-, 1,5-, 2,3- or 3 , Trade 4-hexylene. 1,2- or 1,3-propylene and especially ethylene are preferred.
- a preferred embodiment are compounds of formula I, wherein R is the group means where m is an integer from 1 to 20 and R1 has the meanings given above.
- R 1 preferably contains 1 to 18, in particular 1 to 12 and in particular 1 to 6, carbon atoms. Examples are methyloxy, ethyloxy and the isomers of propyloxy, butyloxy, pentyloxy, hexyloxy, heptyloxy, octyloxy, nonyloxy, decyloxy, undecyloxy, dodecyloxy, tridecyloxy, tetradecyloxy, pentadecyloxy, hexadecyloxy, heptadecyloxy, octadecyloxy and nonadecyloxy, nonadecyloxy R1 can be linear or branched as alkoxy.
- R2 in the meaning of alkyl can be linear or branched and preferably contain 1 to 18, particularly 1 to 12 and in particular 1 to 6 carbon atoms.
- Suitable alkyl groups are e.g. Methyl, ethyl and the isomers of propyl, butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, undecyl, dodecyl, tridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl, octadecyl, nonadecyl and eicosyl.
- R2 in the meaning of alkenyl can be linear or branched and preferably contain 2 to 18, particularly 2 to 12 and in particular 2 to 6 carbon atoms. Examples are allyl, Butenyl, pentenyl, hexenyl, heptenyl, octenyl, decenyl, dodecenyl, tetradecenyl, hexadecenyl and octadecenyl.
- R2 in the meaning of aryl is preferably naphthyl and especially phenyl, and preferably benzyl as arylmethyl.
- the alkyl can be linear or branched and preferably contain 1 to 12, especially 1 to 6, carbon atoms. Some examples are methyl, ethyl, dimethyl, n- or i-propyl, n- or t-butyl, hexyl, octyl, decyl or dodecylphenyl or benzyl.
- a preferred subgroup are those compounds of the formula I in which R1 is -OH, C1-C18-alkoxy or -OC (O) -R2, and R2 is C1-C18-alkyl, phenyl, benzyl, C1-C12-alkylphenyl or C1- C12 alkylbenzyl. R1-OH or C1-C12-alkoxy and R2 C1-C12-alkyl are particularly preferred.
- R is the group means wherein n is a number from 1 to 5 and R1 is C1-C12 alkoxy. Particularly preferably, n is a number from 1 to 3 and R1 is C1-C4-alkoxy.
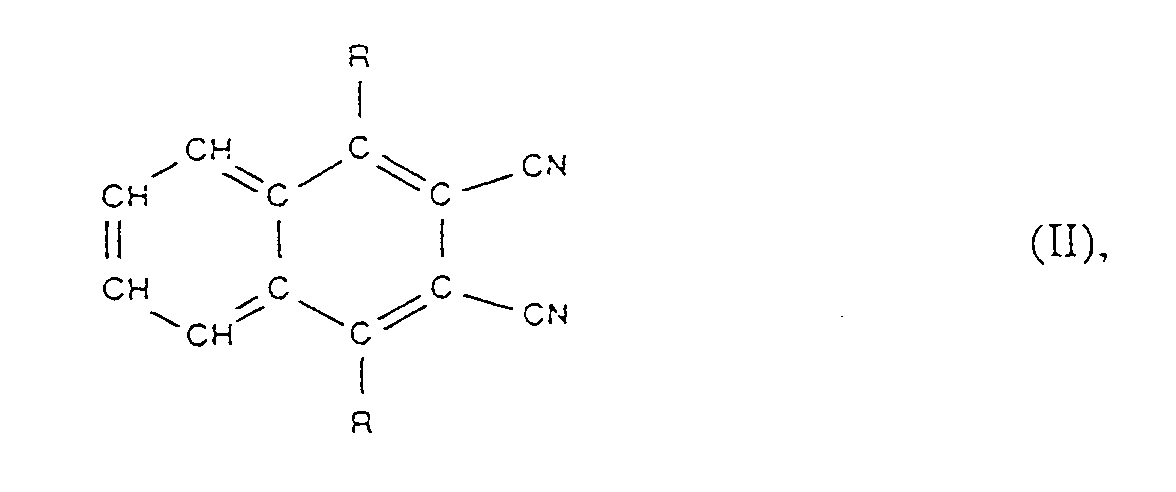
- the compounds of formula II are new and also an object of the invention. You can easily by reacting 1 equivalent of 1,4-dihydroxy-2,3-dicyanonaphthalene with 2 equivalents of p-toluenesulfonic acid ester of the formula at temperatures of appropriately 40 to 150 ° C in the presence of a solvent (for example dimethylformamide) and an alkali metal carbonate (for example K2CO3) are obtained, where R has the meanings given for formula I.
- a solvent for example dimethylformamide
- an alkali metal carbonate for example K2CO3
- the process according to the invention is expediently carried out at a temperature of at least 100 ° C., preferably 100-200 ° C.
- those of the formula RH are used as solvents and those of the formula LiR as lithium salts.
- the lithium salts are e.g. obtained by adding RH lithium to an excess of solvent and then allowing it to react. An excess of lithium salts LiR is advantageously used.
- the compound of formula II is then added to the solution of the lithium salts and the mixture is heated.
- the reaction mixture is then allowed to cool and is suitably hydrolyzed with an acid, e.g. Acetic acid.
- the compounds according to the invention, in which M in formula I M stands for two H can then be isolated in a known manner by means of extraction, crystallization and chromatographic methods.
- the compounds obtained after process step a) are expediently dissolved in a polar solvent, for example alkanols (for example methanol, ethanol, n- or i-propanol, n- or t-butanol, pentanol, hexanol, heptanol) , Octanol), halogenated hydrocarbons (e.g. CH2Cl2, CHCl3, 1,1,1-trichloroethane, 1,1,2,2-tetrachloroethane) or ethers (e.g. diethyl ether, di-n-butyl ether, tetrahydrofuran, dioxane).
- alkanols for example methanol, ethanol, n- or i-propanol, n- or t-butanol, pentanol, hexanol, heptanol
- Octanol halogenated hydrocarbons
- the reaction with a metal salt MX2 can take place, for example, at 10 to 150 ° C, preferably 20 to 80 ° C.
- X can preferably be Cl, Br, acetate or acetylacetonate.
- the metal salts of formula I formed can be purified in the usual way by crystallization or chromatographic methods.
- the compounds according to the invention are brown solid substances which have an absorption maximum in the near IR range (hereinafter abbreviated to NIR) at approximately 850 nm and an absorption minimum (window) at approximately 780 nm. They are thermally stable and soluble, especially in polar and non-polar substrates such as Solvents, liquid crystals and plastics.
- the solubility can be a multiple of the solubility of previously known compounds with alkoxy groups.
- This property is particularly advantageous for opto-electronic applications such as Information storage, because the compounds according to the invention can easily be incorporated into plastics over a long period of time because of their high solubility and good light and temperature resistance.
- the compounds according to the invention can also be used as NIR absorbers for both non-erasable and erasable information storage systems using the inexpensive and handy NIR laser diodes.
- a non-erasable system is based on the formation of ablation by marking with laser radiation. This technique is described in the literature (Angewandte Chemie, Vol. 101, page 1475-1502 (1989)).
- a recording polymer layer is applied, in which one of the NIR dyes according to the invention is dissolved.
- Information is written point by point using a laser diode of e.g. 830 nm and reads the stored information based on the change in transmission or reflection.
- the NIR dyes according to the invention can also be used for another non-erasable system based on the viscoelastic properties of polymers.
- the dye layer is applied directly to a plastic base (e.g. polycarbonate), the glass transition temperature of which is around 100-160 ° C.
- a plastic base e.g. polycarbonate
- a reflective metal layer e.g. Aluminum or gold.
- Those which contain the NIR dye according to the invention dissolved in a polymer whose glass transition temperature is about 100-160 ° C. can also be used as the glass or plastic base.
- Such systems are e.g. in SPIE Vol. 1078 "Optical Data Storage Topic Meeting" (1989), pages 80-87.
- marking e.g. with a laser diode of 830 nm there is a local expansion of the exposed point, which results in a change in transmission or reflection.
- the compounds according to the invention can also be used as NIR absorbers for an erasable system based on the viscoelastic properties of polymers.
- This system uses a double layer consisting of an expansion layer and a plastically deformable retention layer.
- An elastomer of a crosslinked polymer such as, for example, polyurethanes or epoxy resins, whose glass transition temperature is about 25 ° C., can be used in the expansion layer.
- This layer contains a dye such as phthalocyanine derivatives, which have a strong absorption, for example at about 690 nm.
- thermoplastic-based polymers such as polyesters, epoxy resins or polycarbonates, which have a glass transition temperature of approximately 100-160 ° C.
- This layer contains at least one of the NIR dyes according to the invention.
- Plastics such as polycarbonates or polymethyl methacrylate or glasses can be used as a base.
- the dye concentration in the two layers is between approximately 0.001 and 10% by weight, preferably between approximately 0.01 and 3% by weight.
- the recording double layer can, for example, be applied to the substrate using the spin coating technique, each layer thickness preferably being less than 1 »m. If necessary, a reflective metal layer can also be vapor-deposited.
- Information is written point by point using a dye laser of, for example, 690 nm, which first causes a local expansion in the expansion layer and finally leads to a viscoelastic deformation in the retention layer.
- a dye laser of, for example, 690 nm
- the transmission or reflection of the points described changes, which can be seen with a laser diode of, for example, 780 nm.
- the written information can be erased either point by point or integrally with a flash.
- the compounds of the formula I are preferably present in an amount of 0.001 to 10% by weight, in particular 0.01 to 5% by weight, based on the substrate.
- Substrates into which the compounds of the formula I are incorporated are preferably transparent.
- the substrate is preferably a plastic.
- the plastic can e.g. a thermoset, a thermoplastic or a structurally cross-linked polymer.
- a thermoset e.g. a thermoset, a thermoplastic or a structurally cross-linked polymer.
- Such polymers are familiar to the person skilled in the art.
- Another object of the invention is the use of the composition according to the invention as an optical information recording material by marking with lasers.
- Example B4 The compound according to Example B4 is dissolved in dimethylformamide. This dye solution is then poured into a two-component epoxy resin (65 parts by weight of glycidylated phenol novolak and 35 parts by weight of 1,4-butanediol diglycidyl ether, hardener: 55 parts by weight of bis (3-methyl-4-aminocyclohex-1-yl) methane, 40 parts by weight of isophoronediamine, 3 , 5 parts by weight of 1,3,5-tris (dimethylaminomethyl) phenol, 1.5 parts by weight of salicylic acid) (glass transition temperature range: 94-110 ° C) using an ultrasonic bath mixed and dissolved for 5 minutes.
- epoxy resin 65 parts by weight of glycidylated phenol novolak and 35 parts by weight of 1,4-butanediol diglycidyl ether, hardener: 55 parts by weight of bis (3-methyl-4-aminocyclohex-1-y
- the solution is applied to a glass substrate by means of a spin coater and then dried at 40 ° C. under vacuum for two hours. This layer is then heated and crosslinked at 80 ° C. for 8 hours. The absorption maximum is 845 nm. The dye concentration is 0.3% by weight. The layer thickness is approximately 1 »m. Aluminum is evaporated onto this layer with a layer thickness of approximately 1500 ⁇ . The recording layer is exposed point by point with a laser diode of 830 nm. The resulting change in reflection is measured at the same wavelength using a microscope spectrophotometer (USMP 80 from Carl Zeiss). The reflection decreases from 70% to 35% due to laser marking.
- Aluminum is evaporated on a glass substrate under vacuum (layer thickness of about 3000 ⁇ ).
- a two-component epoxy resin 55 parts by weight of epoxidized bisphenol-A, 45 parts by weight of polypropylene glycol diglycidyl ether, hardener: 70 parts by weight of the reaction product of 7 parts by weight of 1, 4-butanediol diglycidyl ether with 58 parts by weight of dimerized C1 Hex-hexahydric acid and 35 parts by weight of 30 wt.
- Dodecylsuccinic anhydride with the addition of catalytic amounts of dimethylbenzylamine (glass temperature range at -19 to 0 ° C) and a copper phthalocyanine (substituted with 2,5-SO3H and 1,5-SO2NHC6H4-CONHCH2CH2SO2CH2CH2OSO3H) applied.
- the layer production is carried out as in Example C1, except for the conditions of preheating (at 60 ° C. for one hour) and crosslinking (at 120 ° C. for one hour).
- the absorption maximum is 676 nm.
- the layer thickness is approximately 1.2 »m.
- the retention layer is then applied, which consists of the NIR dye according to Example B5 and the same epoxy resin as in Example C1.
- the layer production and crosslinking conditions are as in Example C14.
- the absorption maximum of the NIR dye in the polymer is 858 nm.
- the concentration of the dye in the polymer is 0.25% by weight.
- the layer thickness is approximately 0.8 »m.
- the recording layer is exposed point by point with a dye laser (dye: Cresyl violet (670 nm)).
- the resulting change in reflection is measured at 780 nm using a microscope spectrometer (see example C1). A decrease in reflection from 55% to 30% due to the laser marking is observed.
- the stored information is then erased integrally using a flash.
- the writing, reading and erasing procedure is repeated 30 times, with no appreciable reduction in the decrease in reflection being observed.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Optical Record Carriers And Manufacture Thereof (AREA)
- Thermal Transfer Or Thermal Recording In General (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Non-Silver Salt Photosensitive Materials And Non-Silver Salt Photography (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Claims (16)
- Composés de formule I
R est le groupe -(OCmH2m)n-R₁, et M représente deux hydrogènes ou encore Cu ou Ni,
R₁ est -OH, -Cl, -Br, ou un radical alcoxy en C₁-C₃₀ ou -OC(O)R₂,
m est un nombre entier de 2 à 6, et n est un nombre de 1 à 20, et
R₂ est un radical alkyle en C₁-C₂₀, alcényle en C₂-C₂₀, cyclopentyle, cyclohexyle, aryle en C₆-C₁₀ ou arylméthyle en C₆-C₁₀ non substitué, ou substitué par des substituants alkyle en C₁-C₁₈. - Composés selon la revendication 1, dans lesquels, dans la formule I, M est Ni.
- Composés selon la revendication 1, dans lesquels m est un nombre entier de 2 à 4.
- Composés selon la revendication 3, dans lesquels m vaut 2 ou 3.
- Composés selon la revendication 1, dans lesquels n est un nombre de 1 à 10.
- Composés selon la revendication 1, dans lesquels R, dans la formule I, est le groupe -(OCH₂-CH₂)m-R₁, où m est un entier de 1 à 10 et R₁ a les significations données dans la revendication 1.
- Composés selon la revendication 1, dans lesquels R₁ est -OH ou un radical alcoxy en C₁-C₁₈ ou -OC(O)-R₂, et R₂ est un radical alkyle en C₁-C₁₈, phényle, benzyle, (alkyle en C₁-C₁₂)phényle ou (alkyle en C₁-C₁₂)benzyle.
- Composés selon la revendication 7, dans lesquels R₁ est -OH ou un radical alcoxy en C₁-C₁₂, et R₂ est un radical alkyle en C₁-C₁₂.
- Composés selon la revendication 1, dans lesquels R, dans la formule I, est le groupe -(OCH₂-CH₂)n-R₁, où n est nombre de 1 à 5 et R₁ est un radical alcoxy en C₁-C₁₂.
- Composés selon la revendication 9, dans lesquels n est un nombre de 1 à 3 et R₁ est un radical alcoxy en C₁-C₄.
- Procédé pour préparer des composés de formule I selon la revendication 1, dans lesquels :a) on fait réagir un composé de formule II
- Composition contenant :a) un substrat, etb) au moins un composé de formule I selon la revendication 1, selon une répartition uniforme ou sur au moins une surface.
- Composition selon la revendication 12, dans laquelle le composé de formule I est présent, par rapport au substrat, en une quantité de 0,001 à 10 % en poids.
- Composition selon la revendication 12, dans laquelle le substrat est un matériau plastique thermoplastique, à réticulation thermique ou à réticulation structurale.
- Composés de formule II
R₁ est -OH, -Cl, -Br ou un radical alcoxy en C₁-C₃₀ ou encore -OC(O)R₂,
m est un nombre entier de 2 à 6 et n est un nombre de 1 à 20, et
R₂ est un radical alkyle en C₁-C₂₀, alcényle en C₂-C₂₀, cyclopentyle, cyclohexyle, aryle en C₆-C₁₀ ou arylméthyle en C₆-C₁₀ non substitué ou substitué par des substituants alkyle en C₁-C₁₈. - Utilisation d'une composition selon la revendication 12 comme matériau d'enregistrement optique d'informations par marquage avec des lasers.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH418489 | 1989-11-22 | ||
| CH4184/89 | 1989-11-22 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0433220A2 EP0433220A2 (fr) | 1991-06-19 |
| EP0433220A3 EP0433220A3 (fr) | 1991-07-10 |
| EP0433220B1 true EP0433220B1 (fr) | 1995-01-25 |
Family
ID=4271358
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP90810875A Expired - Lifetime EP0433220B1 (fr) | 1989-11-22 | 1990-11-13 | Naphtalocyanine substituée et son utilisation |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US5229507A (fr) |
| EP (1) | EP0433220B1 (fr) |
| JP (1) | JPH03176492A (fr) |
| CA (1) | CA2030392A1 (fr) |
| DE (1) | DE59008355D1 (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006015412A1 (fr) * | 2004-08-09 | 2006-02-16 | Silverbrook Research Pty Ltd | Colorant de cyanine ayant des groupes peripheriques multifonctionnels |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5260435A (en) * | 1990-07-23 | 1993-11-09 | Nippon Oil And Fats Co., Ltd. | Derivative of naphthalocyanine containing perfluoroalkyl group, process for preparing the same and optical recording medium |
| DE4310371A1 (de) * | 1993-03-30 | 1994-10-06 | Basf Ag | Verfahren zur Herstellung von Naphthalocyaninen |
| GB9405970D0 (en) * | 1994-03-25 | 1994-05-11 | Secr Defence | Substituted phthalocyanines |
| US7201963B2 (en) * | 2002-01-15 | 2007-04-10 | Gentex Corporation | Pre-processed workpiece having a surface deposition of absorber dye rendering the workpiece weld-enabled |
| JP4558542B2 (ja) * | 2005-03-10 | 2010-10-06 | 独立行政法人科学技術振興機構 | フォトクロミック材料 |
| JP2007169477A (ja) * | 2005-12-22 | 2007-07-05 | Fujifilm Corp | ナフタロシアニン化合物およびその製造方法 |
| JP4950770B2 (ja) * | 2006-09-21 | 2012-06-13 | 日東電工株式会社 | 光半導体封止用エポキシ樹脂組成物およびその硬化体ならびにそれを用いた光半導体装置 |
| JP5408821B2 (ja) * | 2008-11-28 | 2014-02-05 | 国立大学法人長岡技術科学大学 | ナフタロシアニン化合物及びその製造方法 |
| JP2010197305A (ja) * | 2009-02-26 | 2010-09-09 | Fujifilm Corp | ナフタロシアニン化合物を用いた製品の真偽を証明する方法、シグナル変換方法及び印刷用インク又はトナー、及びナフタロシアニン化合物の製造方法 |
| JP5408824B2 (ja) * | 2009-03-30 | 2014-02-05 | 国立大学法人長岡技術科学大学 | ナフタロシアニン化合物及びその製造方法 |
| KR101577693B1 (ko) | 2013-12-27 | 2015-12-15 | 나노씨엠에스(주) | 폴리 옥소가교형 프탈로시아닌 화합물, 이의 제조방법 및 이를 이용한 근적외선 흡수 및 반사 조성물 |
| US20160272822A1 (en) * | 2015-03-20 | 2016-09-22 | GM Global Technology Operations LLC | Material composition and method for laser ablation |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB8431446D0 (en) * | 1984-12-13 | 1985-01-23 | Secr Defence | Alkoxyphthalocyanines |
| US4725525A (en) * | 1985-02-04 | 1988-02-16 | Hoebbst Celanese Corporation | Recording information media comprising chromophores |
| DE3622590A1 (de) * | 1985-07-04 | 1987-01-08 | Olympus Optical Co | Aufzeichnungsmaterial fuer optische informationen |
| US4904567A (en) * | 1985-08-13 | 1990-02-27 | Mitsubishi Kasei Corporation | Optical recording member |
| JPH089271B2 (ja) * | 1987-02-19 | 1996-01-31 | 三菱化学株式会社 | 光学記録体 |
| GB8614673D0 (en) * | 1986-06-17 | 1986-07-23 | Secr Defence | Alkoxyphthalocyanines |
| US4780867A (en) * | 1986-10-02 | 1988-10-25 | Optical Data, Inc. | Method for erasably recording data by viscoelastic shear deformation |
| EP0313943B1 (fr) * | 1987-10-20 | 1993-08-04 | MITSUI TOATSU CHEMICALS, Inc. | 1,2-Naphtalocyanine absorbant dans l'infrarouge, et matériaux d'enregistrement l'utilisant |
| US4943681A (en) * | 1989-12-22 | 1990-07-24 | Toyo Ink Manufacturing Co., Ltd. | Optical recording medium and process for the production thereof |
-
1990
- 1990-11-13 EP EP90810875A patent/EP0433220B1/fr not_active Expired - Lifetime
- 1990-11-13 DE DE59008355T patent/DE59008355D1/de not_active Expired - Fee Related
- 1990-11-16 US US07/614,815 patent/US5229507A/en not_active Expired - Fee Related
- 1990-11-20 JP JP2312987A patent/JPH03176492A/ja active Pending
- 1990-11-20 CA CA002030392A patent/CA2030392A1/fr not_active Abandoned
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006015412A1 (fr) * | 2004-08-09 | 2006-02-16 | Silverbrook Research Pty Ltd | Colorant de cyanine ayant des groupes peripheriques multifonctionnels |
| US7153956B2 (en) | 2004-08-09 | 2006-12-26 | Silverbrook Research Pty Ltd | Cyanine dye having multifunctional peripheral groups |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2030392A1 (fr) | 1991-05-23 |
| EP0433220A3 (fr) | 1991-07-10 |
| EP0433220A2 (fr) | 1991-06-19 |
| US5229507A (en) | 1993-07-20 |
| JPH03176492A (ja) | 1991-07-31 |
| DE59008355D1 (de) | 1995-03-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0433220B1 (fr) | Naphtalocyanine substituée et son utilisation | |
| DE3882894T2 (de) | 1,2-Naphtalocyanine, Infrarotabsorber und sie verwendende Aufzeichnungsmaterialien. | |
| DE68922557T2 (de) | Polyurethane mit nichtlinearen optischen Eigenschaften und diese enthaltende Materialien, diese enthaltende optische Vorrichtung und Verfahren zur Herstellung dieser Verbindungen und Materialien. | |
| DE68909697T2 (de) | Polyurethane, hergestellt aus nichtlinearen, optisch aktiven Diolen. | |
| DE3873914T2 (de) | Substituierte phthalocyanine. | |
| DE3686044T2 (de) | Optisches speichermedium. | |
| EP0272565B1 (fr) | Phtalocyanines mono- et disubstituées | |
| DE3521915A1 (de) | Aufzeichnungsmaterial fuer optische informationen | |
| EP0192215B1 (fr) | Complexes de tétraphényldithiolène, benzoines substituées asymétriquement ainsi que matériaux d'enregistrement optiques contenant ces complexes | |
| DE69520068T2 (de) | Optische Aufzeichungsmedium mit Formazanmetallkomplexfarbstoff | |
| DE3927872A1 (de) | Dithiolat-metallkomplexverbindungen, verfahren zu ihrer herstellung und sie enthaltendes optisches informationsaufzeichnungsmaterial | |
| DE69812259T2 (de) | Photostabilisierte cyaninfarbstoffe und optischer aufzeichnungsträger | |
| DE69130054T2 (de) | Perfluoralkyliertes naphthalocyaninderivat, seine herstellung sowie ein optisches aufzeichnungsmedium | |
| DE69614777T2 (de) | Formazan-Cyanin-Copolymere für optische Aufzeichnungsschichten und Elemente | |
| DE60004970T2 (de) | Squarylium-verbindung und diese enthaltendes optisches aufzeichungsmedium | |
| DE3914151A1 (de) | Squarylium-verbindungen und deren verwendung in optischen aufzeichnungsmaterialien | |
| DE69415652T2 (de) | Optisches informationsaufzeichnungsmedium, und ein darin enthaltendes nahes infrarotabsorbierendes material | |
| DE69420169T2 (de) | Phthalocyaninverbindung und optisches Aufzeichnungsmedium das diese enthält | |
| DE68918536T2 (de) | Optisches Aufzeichnungsmedium. | |
| DE69331772T2 (de) | Squaryliumverbindungen und diese benutzende optische Informationsaufzeichnungsmittel | |
| DE3854820T2 (de) | Naphthalocyaninderivate und Verfahren zu ihrer Herstellung sowie optische Aufzeichnungsträger unter Verwendung dieser Derivate und Herstellungsverfahren hierfür | |
| EP0335236A2 (fr) | Colorants méthiniques et milieu d'enregistrement contenant ces colorants | |
| DE69733643T2 (de) | Phthalocyaninverbindungen, deren herstellungsverfahren und optisches aufzeichnungsmedium das diese enthält | |
| DE69532096T2 (de) | Optisches Aufzeichnungsmedium | |
| EP0464543A1 (fr) | Colorants polyméthine |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| 17P | Request for examination filed |
Effective date: 19901116 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): BE CH DE FR GB IT LI NL SE |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): BE CH DE FR GB IT LI NL SE |
|
| 17Q | First examination report despatched |
Effective date: 19930805 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| ITF | It: translation for a ep patent filed | ||
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE CH DE FR GB IT LI NL SE |
|
| REF | Corresponds to: |
Ref document number: 59008355 Country of ref document: DE Date of ref document: 19950309 |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19950313 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19960926 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19960930 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19961004 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19961024 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 19961107 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 19961125 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 19961230 Year of fee payment: 7 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PFA Free format text: CIBA-GEIGY AG TRANSFER- NOVARTIS AG |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971113 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971114 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971130 Ref country code: FR Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 19971130 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971130 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19971130 |
|
| BERE | Be: lapsed |
Owner name: CIBA-GEIGY A.G. Effective date: 19971130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19980601 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19971113 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19980801 |
|
| EUG | Se: european patent has lapsed |
Ref document number: 90810875.6 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 19980601 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20051113 |