EP1918974A2 - Ensemble de spectromètre de masse et procédé de spectrométrie de masse liquide ambiante - Google Patents
Ensemble de spectromètre de masse et procédé de spectrométrie de masse liquide ambiante Download PDFInfo
- Publication number
- EP1918974A2 EP1918974A2 EP07119671A EP07119671A EP1918974A2 EP 1918974 A2 EP1918974 A2 EP 1918974A2 EP 07119671 A EP07119671 A EP 07119671A EP 07119671 A EP07119671 A EP 07119671A EP 1918974 A2 EP1918974 A2 EP 1918974A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- analytes
- liquid
- liquid sample
- laser
- nozzle
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/02—Details
- H01J49/10—Ion sources; Ion guns
- H01J49/14—Ion sources; Ion guns using particle bombardment, e.g. ionisation chambers
- H01J49/145—Ion sources; Ion guns using particle bombardment, e.g. ionisation chambers using chemical ionisation
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/02—Details
- H01J49/04—Arrangements for introducing or extracting samples to be analysed, e.g. vacuum locks; Arrangements for external adjustment of electron- or ion-optical components
- H01J49/0431—Arrangements for introducing or extracting samples to be analysed, e.g. vacuum locks; Arrangements for external adjustment of electron- or ion-optical components for liquid samples
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/02—Details
- H01J49/04—Arrangements for introducing or extracting samples to be analysed, e.g. vacuum locks; Arrangements for external adjustment of electron- or ion-optical components
- H01J49/0459—Arrangements for introducing or extracting samples to be analysed, e.g. vacuum locks; Arrangements for external adjustment of electron- or ion-optical components for solid samples
- H01J49/0463—Desorption by laser or particle beam, followed by ionisation as a separate step
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/02—Details
- H01J49/10—Ion sources; Ion guns
- H01J49/16—Ion sources; Ion guns using surface ionisation, e.g. field-, thermionic- or photo-emission
- H01J49/165—Electrospray ionisation
Definitions
- the invention relates to a method for mass spectrometry, more particularly to a method for ambient liquid mass spectrometry that is capable of conducting direct analysis of mass spectrometry on a liquid sample under atmospheric pressure.
- the present invention also relates to a mass spectrometer assembly for conducting the method of ambient liquid mass spectrometry.
- electrospray ionization mass spectrometry A method for mass spectrometry is called electrospray ionization mass spectrometry (ESI-MS), which involves ionizing proteins contained in a liquid sample, followed by a protein analysis.
- ESI-MS electrospray ionization mass spectrometry
- an electrospray ionization mass spectrometer (ESI-MS) 1 includes an electrospray ionization device 11.
- the electrospray ionization device 11 performs an electrospray ionization procedure to ionize the proteins in the liquid sample.
- An electric field is established between an open end 111 of a capillary 112 and an entrance side 121 of a mass analyzer 12. Subsequently, the liquid sample is pushed through the capillary 112 toward the open end 111. As the electric field force overcomes the surface tension of the liquid sample at the open end 111 of the capillary 112, aerosol droplets containing multivalent electric charges and protein molecules are formed, and are pushed into the mass analyzer 12 through the entrance side 121.
- the multivalent electrons are attached to the protein molecules to form ionized protein molecules with relatively lower mass-to-charge ratio (m/z) values as the charged droplets dwindle in size when traveling through the air from the open end 111 of the capillary 112 toward the entrance side 121 of the mass analyzer 12.
- body fluids or other biochemical solutions normally contain a high concentration of various salts.
- a "desalination" pre-process such as dialysis, is required to prevent the protein molecules from being ionized by acquiring charges from the salts that are present in the body fluids/biochemical solutions, to thereby result in a simpler ion peak configuration in the mass spectrum obtained from ESI-MS.
- professional personnel are required to execute the "desalination" pre-process, which is a tedious, time consuming and inconvenient process.
- MALDI-MS matrix-assisted laser desorption ionization mass spectrometry
- a water soluble organic acidic matrix of highly laser light absorbing small organic molecules is mixed with a liquid sample containing protein molecules before the mixture is dehydrated to form a crystal.
- a laser beam is irradiated on the surface of the crystal, causing ionization and desorption of the protein molecules.
- the ionized protein molecules are introduced into a mass analyzer for mass spectrometric analysis.
- the desorption process of MALDI-MS needs to be conducted in vacuum.
- the matrix used in MALDI-MS is generally an organic acid, which affects the analyte (e.g., proteins) chemically, causing the structure of the analyte to change.
- SALDI-MS surface-assisted laser desorption/ionization mass spectrometry
- the object of the present invention is to provide a laser desorption device, a mass spectrometer assembly, and a method for mass spectrometry that is capable of conducting mass analysis directly on a liquid sample under atmospheric pressure.
- a method for mass spectrometry which is named “ambient liquid mass spectrometry” (ALMS), and which includes the steps of:
- a laser desorption device for use in a mass spectrometer assembly.
- the mass spectrometer assembly includes a receiving unit, an electrospray unit, and a voltage supplying member.
- the laser desorption device includes a sample stage and a laser transmission mechanism. The sample stage and the laser transmission mechanism are arranged with the receiving unit, the electrospray unit, and the voltage supplying member in a manner such that all the steps of the abovementioned method can be duly carried out.
- the laser transmission mechanism can be one of an ultraviolet (UV) laser, an infrared (IR) laser, a nitrogen laser, an argon ion laser, a helium-neon laser, a carbon dioxide (CO 2 ) laser, and a garnet (Nd:YAG) laser.
- a suitable matrix is added to a liquid sample prior to conducting a mass spectrometric analysis.
- a laser beam 821 is irradiated onto a liquid sample 4, which includes a solution 41 that contains analytes 412 and a material 413 serving as a matrix for absorbing laser energy, and which is disposed in the passage way of a receiving unit 6 adapted to admit therein ionized analytes 414 that are derived from the liquid sample 4 for mass spectrometric analysis.
- ALMS ambient liquid mass spectrometry
- an infrared laser beam is employed to irradiate directly on an aqueous solution to obtain accurate mass spectrometric analysis results.
- ALMS Abmbient Liquid Mass Spectrometry
- Operation procedure of ALMS is relatively simple and rapid, and resolution thereof is higher than that of ESI-MS.
- ALMS is capable of accurately detecting molecular weights of analytes, even when the analytes are macromolecules, such as proteins, thereby showing an outstanding ability in protein identification.
- the method of ALMS according to the present invention can be implemented by performing the following steps:
- a liquid sample 4 on a sample stage 81, that includes a solution 41 containing a plurality of analytes 412 and a matrix material 413 for absorbing laser energy so as to assist in desorption of the analytes 412.
- the solution 41 includes a solvent 411 that contains the analytes 412 and the matrix material 413 therein.
- an electrospray unit 5 that includes a reservoir 52 for accommodating a liquid electrospray medium 51, and a nozzle 53 which is disposed downstream of the reservoir 52, and which is configured to sequentially form liquid drops 511 of the electrospray medium 51 thereat.
- a receiving unit 6 that is spaced apart from the nozzle 53 for receiving and analyzing ionized analytes 414 derived from the liquid sample 4.
- a detector 7 for detecting signals generated as a result of analyzing the ionized analytes 414 by the receiving unit 6, and for generating a mass spectrum of the liquid sample 4 from the signals.
- the electrospray medium forming the liquid drops is a solution normally used in electrospray methods.
- An example of the electrospray medium is a solution containing a volatile liquid such that the liquid portion in the liquid drops can vaporize prior to the receipt of the ionized analytes by the mass analyzer.
- the volatile liquid is preferably one with a low polarity, such as isoacetonitrile, acetone, alcohol, etc.
- the electrospray medium is preferably a solution that contains a volatile liquid and that does not contain an acid, such as methanol aqueous solution.
- an aqueous solution containing methanol and acetic acid and a “methanol aqueous solution” are used as the electrospray medium in the embodiments of the present invention, respectively.
- the ion portion of the obtained analytes is multivalent with each charge being contributed by a proton (H + ) .
- One of the main objects that the method of ALMS aims at is the detection of analytes from a liquid sample including a solution that contains the analytes and a matrix material. Therefore, no limitation is imposed on the types of solutions and the kinds of analytes detectable for the implementations of the present invention. Whether the solution is an aqueous solution, contains an organic solvent, or is a body fluid secreted by an organism and having a complicated composition, and whether the analytes are macroscopic molecules such as proteins, or are microscopic molecules such as ordinary compounds, mass spectrometric analysis results can be obtained through implementing the method of ALMS according to the present invention.
- the liquid sample under study can include various solutions, including organism's body fluids, chemical solutions, environment sampling solutions, or various eluates from liquid chromatography, etc.
- the organism's body fluid can be selected from the group consisting of blood, tear, milk, perspiration, intestinal juice, brains fluid, spinal fluid, lymph, pus, blood serum, saliva, nasal mucus, urine, and excrement.
- the chemical solution can be an organic solution.
- the matrix material ismade fromamaterial that is non-transmissible by laser, such as gold, carbon, cobalt, iron, 2,5-dihydroxybenzoic acid (2,5-DHB), 3,5-dimethoxy-4-hydroxycinnamic: acid (sinapinic acid, (SA)), ⁇ -cyano-4-hydroxycinnamic acid ( ⁇ -CHC), and a combination thereof.
- amaterial that is non-transmissible by laser such as gold, carbon, cobalt, iron, 2,5-dihydroxybenzoic acid (2,5-DHB), 3,5-dimethoxy-4-hydroxycinnamic: acid (sinapinic acid, (SA)), ⁇ -cyano-4-hydroxycinnamic acid ( ⁇ -CHC), and a combination thereof.
- SA sinapinic acid
- ⁇ -CHC ⁇ -cyano-4-hydroxycinnamic acid
- the solution included in the liquid sample is an aqueous solution
- water molecules contained in the aqueous solution would be the material serving as the matrix, and an infrared laser beam is used for desorption.
- a material serving as a matrix is added into the solution to form the liquid sample under study prior to implementing the method of ALMS. Descriptions related to detailed operational practices and mechanisms for the method of ALMS will be described in subsequent embodiments.
- the first preferred embodiment of a mass spectrometer assembly implementing the method of ALMS is adapted to conduct mass spectrometric analysis on a liquid sample 4.
- the liquid sample 4 includes a solution 41 including a solvent that contains a plurality of analytes 412 and a material 413 serving as a matrix (also referred to as a matrix material 413) for assisting in desorption of at least one of the analytes 412.
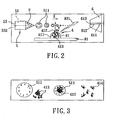
- the mass spectrometer assembly includes an electrospray unit 5, a receiving unit 6, a voltage supplying member 3, and a laser desorption device 8.
- the laser desorption device 8 includes a sample stage 81 on which the liquid sample 4 is placed, a laser transmission mechanism 82 that is capable of transmitting a laser beam 821 and that is disposed to irradiate the liquid sample 4, a lens 83 that is disposed to receive the laser beam 821 from the laser transmission mechanism 82 for focusing the energy carried by the laser beam 821, and a reflector 84 that is disposed to change the path of the laser beam 821.
- the laser transmission mechanism 82 is an ultraviolet laser transmission mechanism 82a that is capable of transmitting the laser beam 821.
- the laser desorption device 8 is designed as long as the laser desorption device 8 is capable of irradiating the liquid sample 4 such that, upon irradiation, at least one of the analytes 412 contained in the solution 41 of the liquid sample 4 is desorbed. Therefore, in practice, the lens 83 and the reflector 84 can be varied in position as required, or can even be completely eliminated according to other embodiments of the present invention.
- the sample stage 81 of the laser desorption device 8 includes a support member 811 that is made from a material non-transmissive by laser, and a hoister platform 812 that is provided for mounting of the support member 811 thereon, and that is movable.
- the support member 811 is provided for placement of the liquid sample 4, and has a support surface 813 for placement of the liquid sample 4 directly thereon. This way, an operator can begin performing the method of ALMS by dripping the liquid sample 4 on the support surface 813.
- the receiving unit 6 is disposed to admit therein ionized analytes 414 that are derived from the liquid sample 4, and that are to be analyzed for mass spectrometric analysis.
- the receiving unit 6 includes a mass analyzer 61 disposed for analyzing the ionized analytes 414.
- the mass analyzer 61 is formed with a conduit 611 that is in air communication with the environment.
- the detector 7 is disposed to receive signals generated by the mass analyzer 61 as a result of analyzing the ionized analytes 414 so as to generate a mass spectrometric analysis result, i.e., a mass spectrum.
- the electrospray unit 5 includes a reservoir 52 for accommodating a liquid electrospray medium 51, a nozzle 53 (in the embodiments of the present invention, the nozzle 53 is a capillary 53a) which is disposed downstream of the reservoir 52, and which is configured to sequentially form liquid drops 511 of the electrospraymedium 51 thereat, and a pump 54 disposed downstream of the reservoir 52 and upstream of the nozzle 53 for drawing the electrospray medium 51 into the nozzle 53.
- the nozzle 53 is spaced apart from the mass analyzer 61 of the receiving unit 6 in a longitudinal direction so as to define a traveling path (X).
- the voltage supplying member 3 is disposed to establish between the nozzle 53 of the electrospray unit 5 and the mass analyzer 61 of the receiving unit 6 a potential difference which is of an intensity such that the liquid drops 511 are laden with a plurality of charges, and such that the liquid drops 511 are forced to leave the nozzle 53 as multiple-charged ones for heading toward the mass analyzer 61 along the traveling path (X).
- the nozzle 53 is made from a metal material, and a first central axis 532 of the nozzle 53 and a second central axis 612 of the conduit 611 in the mass analyzer 61 are substantially parallel to each other.
- the support member 811 of the sample stage 81 extends in the longitudinal direction such that the support surface 813 thereof defines a leveled plane in the longitudinal direction.
- the distance between projections of an outlet 531 of the nozzle 53 and an entrance 613 into the conduit 611 of the mass analyzer 61 on the leveled plane is approximately 8 mm.
- the shortest distance between the liquid sample 4 and the outlet 531 of the nozzle 53 is 1.5 mm.
- the laser transmission mechanism 82 of the laser desorption device 8 transmits the laser beam 821 to irradiate the liquid sample 4
- at least one of the analytes 412 contained in the solution 41 of the liquid sample 4 is desorbed to fly along a flying path (Y) which intersects the traveling path (X) so as to enable said at least one of the analytes 412 to be occluded in the multiple-charged liquid drops 511.
- the second preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the first preferred embodiment.
- the electrospray unit 5' of the second preferred embodiment further includes an airstream supplying mechanism 55' for accelerating vaporization of the multiple-charged liquid drops 511 (refer to FIGS.2 to 4) to result in dwindling in size thereof when approaching the mass analyzer 61 (refer to FIG.5) along thetravelingpath (X).
- the airstream supplying mechanism 55' surrounds the nozzle 53, and supplies a nitrogen airstream 5.51'.
- the temperature of the nitrogen airstream 551' can be controlled by the user between the room temperature and 325°C as is required.
- the third preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the first preferred embodiment.
- the difference between the first and third preferred embodiments is that the nozzle 53" of the electrospray unit 5" of the third preferred embodiment is made from a non-metal material, and the electrospray unit 5" further includes a micro-tube 56".
- the micro-tube 56" includes a tubular body 561" connected between and disposed in fluid communication with the pump 54 and the nozzle 53", and a center portion 562" connected to the tubular body 561" and coupled to the voltage supplying member 3 (refer to FIG. 4) such that the potential difference is established between the micro-tube 56" and the mass analyzer 61 of the receiving unit 6.
- the fourth preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the first preferred embodiment.
- the difference between the first and fourth preferred embodiments is that the sample stage 81'" of the laser desorption device 8"' includes a movable track 814"', and a support member set 815'" including a plurality of support members 816"' (only one is visible in FIG. 7) connected in sequence and mounted movably on the track 814"'.
- a plurality of liquid samples 4 are first contained in containers 10 (e.g., test tubes or centrifuge tubes) (only one is visible in FIG. 7), respectively. Subsequently, each of the containers 10 is disposed on a corresponding one of the support members 816"'.
- the support members 816"' move along the track 814"', carrying the liquid samples 4 thereon, such that the liquid samples 4 are sequentially disposed at a predefined location set by the operator.
- the liquid sample 4 is irradiated by the laser beam 821 transmitted by the laser transmission mechanism 82 of the laser desorption device 8, and subsequent mass spectrometric analysis is conducted.
- the fifth preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the first preferred embodiment.
- the laser transmission mechanism of the fifth preferred embodiment is an infrared (IR) laser 82c instead of the ultraviolet laser 82a as in the first preferred embodiment.
- the sixth preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the fourth preferred embodiment.
- the laser transmission mechanism of the sixth preferred embodiment is the infrared (IR) laser 82c (as shown in FIG.4) instead of the ultraviolet laser 82a.
- each of the components of the mass spectrometer assembly according to the present invention can be designed to be movable so as to permit adjustments of the positions thereof by the user as are required, such that relative positions or distances among the various components of the mass spectrometer assembly can be determined.
- parameters such as the energy, frequency, incident angle of the laser beam irradiated by the laser transmission mechanism, and the composition and flow rate of the electrospray medium, etc., can be adjusted according to the objectives aimed, so as to obtain optimal detection results.
- the exemplary methods and the comparative example are conducted according to the third preferred embodiment of the present invention.
- the mass analyzer conducts the scans with a 2s/scan scanning rate. For each liquid sample presented, the molecular weight of the solvent is excluded from a scanning range of the mass analyzer.
- the electrospray medium used was a 20 vol% methanol aqueous solution
- the matrix material for the liquid sample was in the form of carbon powders with varying concentrations, respectively.
- the composition of the liquid sample used, and the figure number of corresponding mass spectrum for each of exemplary methods 1 and 2 and comparative example 1 are tabulated in Table 1 below.
- Table 1 Liquid Sample Mass spectrum Analytes Carbon powder Concentration Comparative example 1 myoglobin (10 -5 M), cytochrome c (10 -5 M), lysozyme (10 -5 M) 0 mg/ ⁇ L
- FIG. 8(b) Exemplary Method 2 0.8 mg/ ⁇ L
- the applicant predicted that the mass spectra obtained should present the formation of "un-denatured proteins".
- the molecular weight of myoglobin resulted from exemplary methods 1 and 2, where ALMS analysis was used, should be 17567 Da, instead of 16951 Da, which is the molecular weight of a denatured protein short of one heme molecular (molecular weight of 616 Da).
- FIGS. 8(b) and 8(c) there are three ion peaks, which are respectively denoted by " ⁇ ", " ⁇ ", "•”, and whose molecular weights are calculated by a computer software to be 12232 Da, 14306 Da, and 17567 Da, respectively.
- the calculated molecular weights almost completely correspond to the molecular weights of myoglobin, cytochrome c, and lysozyme as provided by the manufacturer.
- the detected myoglobin is in an un-denatured state.
- the results confirm that the method of ALMS works effectively, and is capable of conducting direct detection on a liquid sample including a protein so as to obtain accurate and satisfactory quantitative results.
- the reason for this success is that, upon irradiation, laser energy of the ultraviolet laser beam is passed on to at least one of the analytes (proteins) contained in the solution of the liquid sample via the matrix material (carbon powders) so that the analyte is successfully desorbed.
- the liquid sample used in comparative example 1 does not contain carbon powders or any other materials to serve as a matrix, the analytes could not be effectively desorbed from the liquid sample (or the volume of desorbed analytes was too small). Since no or a minimal number of analytes reached and was detected by the mass analyzer for mass spectrometric analysis, corresponding signals for the analytes could not be generated.
- the peak shown in FIG. 8 (a) is an interference signal, and is relatively enlarged due to the absence of analyte signals.
- FIG. 9(d) Exemplary Method 7 Tolene Cinnamic acid benzyl ester (2*10 -2 M)
- FIG.9(e) Exemplary Method 8 n-hexane Cetylpyridinium chloride (1*10 -4 M)
- FIGS.9(a) to 9(f) corresponding ion peaks formed by analytes of the liquid samples used in exemplary methods 3 to 8.
- the molecular weights obtained after calculation match with the known facts, confirming the operability of the method of ALMS on liquid samples provided with organic solutions and organic compounds.
- a linear equation obtained through linear regression analysis demonstrates that glycosylated hemoglobin/ hemoglobin (HbA1/Hb) values of in blood of a diabetes patient obtained using ALMS analysis has a specific relationship with those obtained using ionic chromatography, which is a currently common method used in the medical field for obtaining the quantities of Hb and HbA1. Therefore, the (HbA1/Hb) values obtained using ALMS analysis should have a certain degree of credibility and reference value. In particular, it is reported that it takes approximately an hour, including preparation work on the samples, to conduct analysis using ionic chromatography. On the other hand, instantaneous detection and result can be obtained using ALMS analysis. Therefore, the method of ALMS should have the potential of replacing the method of ionic chromatography in providing the basis for diagnoses of diseases.
- the present invention is in deed capable of performing rapid and accurate mass spectrometric analysis directly on a liquid sample.
- the sample to be analyzed i.e., whether it is a body fluid with a complicated composition, or an organic solution, a protein solution, etc.
- qualitative information about the contents therein can be obtained through the method of ALMS according to the present invention.
- relative quantitative information on various analytes in a liquid sample such as compositional proportions of the analytes in the liquid sample, can also be reflected through the use of ALMS analysis. It is of special importance that when a liquid sample includes an aqueous solution, by irradiating the liquid sample with infrared laser, satisfactory detection results can be obtained through ALMS analysis.
- a mass spectrometer assembly implementing the method of ALMS should be capable of being connected in series to other analytic instruments.
- a high performance liquid chromatograph (HPLC) is taken as an example hereinbelow for illustration.
- HPLC high performance liquid chromatograph
- ALMS analysis can be conducted by irradiating laser on the eluted sample when it is disposed between the electrospray unit and the mass analyzer of the mass spectrometer assembly implementing the method of ALMS.
- the method of ALMS according to the present invention is conducted directly under atmospheric pressure, instead of vacuum, and since operation time needed is extremely short, the cost of instrumentation for implementing the present invention, the technical requirements for manufacturing such instrumentation and for operation of such method have all greatly reduced as compared to matrix-assisted laser desorption ionization mass spectrometry (MALDI-MS) of the prior art.
- MALDI-MS matrix-assisted laser desorption ionization mass spectrometry
- the method of ALMS according to the present invention can be used to analyze various kinds of liquid samples, including protein aqueous solutions, body fluids, and organic solutions containing organic compounds, etc., can all be analyzed directly (with minimal sample preparation), as opposed to making the originally liquid samples into solid samples.
- satisfactory results can be obtained both for qualitative analysis (i.e., the determination of the identity of the analytes detected) and relative quantitative analysis (i.e., the quantity of various kinds of analytes contained in the liquid sample).
- the present invention is advantageous in related fields, where qualitative analysis of analytes in a large quantity of liquid samples or determination of relative concentrations of analytes in liquid samples is required, such as in medical fields, environmental examination, criminal judgment, academic research, etc.
- the method of ALMS according to the present invention can also be applied to the analysis of a body fluid secreted by an organism. Through identities and relative concentrations of substances in an organism' s body fluid, the biological condition of the organism can be determined.
- the mass spectrometer assembly implementing the method of ALMS according to the present invention can be connected in series to other analytic instruments, such as a high performance liquid chromatograph (HPLC), so that an operator can conduct ALMS analysis so as to obtain information on the substances contained in the sample in sequence with conducting sample purification.
- HPLC high performance liquid chromatograph
Landscapes
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- Engineering & Computer Science (AREA)
- Plasma & Fusion (AREA)
- Optics & Photonics (AREA)
- Other Investigation Or Analysis Of Materials By Electrical Means (AREA)
- Electron Tubes For Measurement (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNA200610142595XA CN101173914A (zh) | 2006-10-30 | 2006-10-30 | 大气压力液相质谱分析方法及大气压力液相质谱仪 |
| US11/746,282 US20080116366A1 (en) | 2006-11-17 | 2007-05-09 | Laser desorption device, mass spectrometer assembly, and method for ambient liquid mass spectrometry |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1918974A2 true EP1918974A2 (fr) | 2008-05-07 |
| EP1918974A3 EP1918974A3 (fr) | 2010-03-24 |
Family
ID=39060250
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP07119671A Withdrawn EP1918974A3 (fr) | 2006-10-30 | 2007-10-30 | Ensemble de spectromètre de masse et procédé de spectrométrie de masse liquide ambiante |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP1918974A3 (fr) |
| JP (2) | JP2008147165A (fr) |
| RU (1) | RU2007140232A (fr) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2296791A4 (fr) * | 2008-05-12 | 2014-01-08 | Shimadzu Corp | « source d ions à capture de gouttelettes » couplée à un appareil analyseur de mobilité et procédé associé |
| EP2287600A4 (fr) * | 2008-05-20 | 2014-01-08 | Shimadzu Corp | Spectromètre de masse à ionisation à la pression atmosphérique |
| US8835179B2 (en) | 2010-10-29 | 2014-09-16 | Scinopharm Taiwan, Ltd. | Real-time monitor solid phase peptide synthesis by mass spectrometry |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| HU226837B1 (hu) | 2006-05-31 | 2009-12-28 | Semmelweis Egyetem | Folyadéksugárral mûködõ deszorpciós ionizációs eljárás és eszköz |
| CN102483369B (zh) | 2009-05-27 | 2015-11-25 | 英国质谱有限公司 | 用于鉴定生物组织的系统和方法 |
| EP2798344B1 (fr) | 2011-12-28 | 2023-12-13 | Micromass UK Limited | Système et procédé d'ionisation à évaporation d'échantillons en phase liquide |
| CN104254901B (zh) | 2011-12-28 | 2018-05-04 | 英国质谱有限公司 | 碰撞离子发生器和分离器 |
| WO2016063327A1 (fr) * | 2014-10-20 | 2016-04-28 | 株式会社島津製作所 | Dispositif d'ionisation de pression atmosphérique |
| JP6593689B2 (ja) * | 2015-08-27 | 2019-10-23 | 高知県公立大学法人 | Maldi質量分析用マトリックス及びその製法並びにそれを用いた質量分析法 |
| JP7045990B2 (ja) * | 2016-06-24 | 2022-04-01 | 株式会社住化分析センター | 物理構造の評価方法 |
| KR102062447B1 (ko) * | 2018-03-13 | 2020-01-03 | 가천대학교 산학협력단 | 그래핀을 이용한 maldi 질량 분석 플레이트 및 이를 이용하는 질량 분석 방법 |
| US11532472B2 (en) * | 2020-07-30 | 2022-12-20 | Ricoh Company, Ltd. | Mass spectrometry |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6849847B1 (en) * | 1998-06-12 | 2005-02-01 | Agilent Technologies, Inc. | Ambient pressure matrix-assisted laser desorption ionization (MALDI) apparatus and method of analysis |
| EP1113850A4 (fr) * | 1998-09-17 | 2006-02-01 | Advion Biosciences Inc | Systeme monolithique integre microfabrique d'electronebulisation et de chromatographie en phase liquide et procede associe |
| US7087898B2 (en) * | 2000-06-09 | 2006-08-08 | Willoughby Ross C | Laser desorption ion source |
| DE10144250A1 (de) * | 2001-08-31 | 2003-04-03 | Fraunhofer Ges Forschung | Verbesserte massenspektrometrische Analyse unter Verwendung von Nanopartikeln |
| GB0128586D0 (en) * | 2001-11-29 | 2002-01-23 | Amersham Biosciences Ab | Graphite anchor targets |
| DE102004002729B4 (de) * | 2004-01-20 | 2008-11-27 | Bruker Daltonik Gmbh | Ionisierung desorbierter Analytmoleküle bei Atmosphärendruck |
| JP2005243466A (ja) * | 2004-02-26 | 2005-09-08 | Japan Science & Technology Agency | レーザイオン化装置及びその利用 |
| US7465920B2 (en) * | 2004-03-30 | 2008-12-16 | University Of Yamanashi | Ionization method and apparatus for mass analysis |
| TWI271771B (en) * | 2006-01-27 | 2007-01-21 | Univ Nat Sun Yat Sen | Electrospray-assisted laser desorption ionization devices, mass spectrometers, and methods for mass spectrometry |
-
2007
- 2007-09-28 JP JP2007253798A patent/JP2008147165A/ja active Pending
- 2007-10-30 EP EP07119671A patent/EP1918974A3/fr not_active Withdrawn
- 2007-10-30 RU RU2007140232/28A patent/RU2007140232A/ru not_active Application Discontinuation
-
2013
- 2013-07-11 JP JP2013145875A patent/JP5707630B2/ja active Active
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2296791A4 (fr) * | 2008-05-12 | 2014-01-08 | Shimadzu Corp | « source d ions à capture de gouttelettes » couplée à un appareil analyseur de mobilité et procédé associé |
| EP2287600A4 (fr) * | 2008-05-20 | 2014-01-08 | Shimadzu Corp | Spectromètre de masse à ionisation à la pression atmosphérique |
| US8835179B2 (en) | 2010-10-29 | 2014-09-16 | Scinopharm Taiwan, Ltd. | Real-time monitor solid phase peptide synthesis by mass spectrometry |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2013257332A (ja) | 2013-12-26 |
| JP2008147165A (ja) | 2008-06-26 |
| EP1918974A3 (fr) | 2010-03-24 |
| RU2007140232A (ru) | 2009-05-10 |
| JP5707630B2 (ja) | 2015-04-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1918974A2 (fr) | Ensemble de spectromètre de masse et procédé de spectrométrie de masse liquide ambiante | |
| US7696475B2 (en) | Electrospray-assisted laser desorption ionization device, mass spectrometer, and method for mass spectrometry | |
| US20080116366A1 (en) | Laser desorption device, mass spectrometer assembly, and method for ambient liquid mass spectrometry | |
| US11867684B2 (en) | Sample dispenser including an internal standard and methods of use thereof | |
| US11430648B2 (en) | System and methods for ionizing compounds using matrix-assistance for mass spectrometry and ion mobility spectrometry | |
| US11830716B2 (en) | Mass spectrometry analysis of microorganisms in samples | |
| EP3633710B1 (fr) | Génération d'ions utilisant un matériau poreux mouillé | |
| Takats et al. | Ambient mass spectrometry using desorption electrospray ionization (DESI): instrumentation, mechanisms and applications in forensics, chemistry, and biology | |
| Liu et al. | Development, characterization, and application of paper spray ionization | |
| US7718958B2 (en) | Mass spectroscopic reaction-monitoring method | |
| US7968842B2 (en) | Apparatus and systems for processing samples for analysis via ion mobility spectrometry | |
| EP1741120B1 (fr) | Procede et systeme de desorption-ionisation par electronebulisation | |
| US8445842B2 (en) | Mechanical holder for surface analysis | |
| US7687772B2 (en) | Mass spectrometric imaging method under ambient conditions using electrospray-assisted laser desorption ionization mass spectrometry | |
| EP2122661A1 (fr) | Désorption-ionisation par électrospray intégré | |
| Zhai et al. | Direct biological sample analyses by laserspray ionization miniature mass spectrometry | |
| US20210302288A1 (en) | Systems and methods for analyzing an analyte extracted from a sample using an adsorbent material | |
| Walles et al. | Approaches for coupling solid-phase microextraction to nanospray | |
| CN101173914A (zh) | 大气压力液相质谱分析方法及大气压力液相质谱仪 | |
| Zhu et al. | High-throughput screening using a synchronized pulsed self-aspiration vacuum electrospray ionization miniature mass spectrometer | |
| CN117413339A (zh) | 分析仪系统 | |
| Abu‐Rabie | Direct analysis of dried blood spot samples | |
| EP1193730A1 (fr) | Dispositif d'analyse à ionisation à pression atmosphérique et méthode d'analyse d'échantillons associée | |
| US20240194469A1 (en) | Mass spectrometry analysis of microorganisms in samples | |
| Walworth | Liquid Extraction Based Surface Sampling: Liquid Microjunction Surface Sampling Probes Coupled with Mass Spectrometry |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: H01J 49/14 20060101ALI20100215BHEP Ipc: H01J 49/16 20060101ALI20100215BHEP Ipc: H01J 49/04 20060101AFI20100215BHEP |
|
| AKY | No designation fees paid | ||
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: 8566 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20100925 |