EP2154258B1 - Auf Nickel basierende Superlegierung, zugehöriges Verfahren zur unidirektionalen Verfestigung und daraus gebildete Gussstücke - Google Patents
Auf Nickel basierende Superlegierung, zugehöriges Verfahren zur unidirektionalen Verfestigung und daraus gebildete Gussstücke Download PDFInfo
- Publication number
- EP2154258B1 EP2154258B1 EP09166869A EP09166869A EP2154258B1 EP 2154258 B1 EP2154258 B1 EP 2154258B1 EP 09166869 A EP09166869 A EP 09166869A EP 09166869 A EP09166869 A EP 09166869A EP 2154258 B1 EP2154258 B1 EP 2154258B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- nickel
- base superalloy
- casting
- alloy
- superalloy according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000005266 casting Methods 0.000 title claims abstract description 64
- 229910000601 superalloy Inorganic materials 0.000 title claims abstract description 34
- 238000000034 method Methods 0.000 title claims abstract description 28
- 230000008569 process Effects 0.000 title description 12
- 238000007711 solidification Methods 0.000 title description 8
- 239000013078 crystal Substances 0.000 claims abstract description 34
- 239000000203 mixture Substances 0.000 claims abstract description 34
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims abstract description 22
- 229910052735 hafnium Inorganic materials 0.000 claims abstract description 22
- VBJZVLUMGGDVMO-UHFFFAOYSA-N hafnium atom Chemical compound [Hf] VBJZVLUMGGDVMO-UHFFFAOYSA-N 0.000 claims abstract description 22
- 229910052782 aluminium Inorganic materials 0.000 claims abstract description 20
- 229910052715 tantalum Inorganic materials 0.000 claims abstract description 20
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 claims abstract description 20
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims abstract description 19
- 229910052702 rhenium Inorganic materials 0.000 claims abstract description 19
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 claims abstract description 19
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims abstract description 18
- 229910052721 tungsten Inorganic materials 0.000 claims abstract description 18
- 239000010937 tungsten Substances 0.000 claims abstract description 18
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 claims abstract description 16
- 229910052726 zirconium Inorganic materials 0.000 claims abstract description 16
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims abstract description 14
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 14
- 229910017052 cobalt Inorganic materials 0.000 claims abstract description 14
- 239000010941 cobalt Substances 0.000 claims abstract description 14
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 claims abstract description 14
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 claims abstract description 13
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 claims abstract description 13
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims abstract description 13
- 229910052804 chromium Inorganic materials 0.000 claims abstract description 13
- 239000011651 chromium Substances 0.000 claims abstract description 13
- 229910052750 molybdenum Inorganic materials 0.000 claims abstract description 13
- 239000011733 molybdenum Substances 0.000 claims abstract description 13
- 239000010936 titanium Substances 0.000 claims abstract description 13
- 229910052719 titanium Inorganic materials 0.000 claims abstract description 13
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims abstract description 9
- 229910052796 boron Inorganic materials 0.000 claims abstract description 9
- 239000012535 impurity Substances 0.000 claims abstract description 9
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 9
- 229910052727 yttrium Inorganic materials 0.000 claims abstract description 8
- VWQVUPCCIRVNHF-UHFFFAOYSA-N yttrium atom Chemical compound [Y] VWQVUPCCIRVNHF-UHFFFAOYSA-N 0.000 claims abstract description 8
- 208000003351 Melanosis Diseases 0.000 claims description 33
- 238000001816 cooling Methods 0.000 claims description 11
- 230000007547 defect Effects 0.000 claims description 6
- 238000004519 manufacturing process Methods 0.000 claims description 5
- 229910052751 metal Inorganic materials 0.000 claims description 3
- 239000002184 metal Substances 0.000 claims description 3
- 229910045601 alloy Inorganic materials 0.000 abstract description 60
- 239000000956 alloy Substances 0.000 abstract description 60
- 208000016311 Freckling Diseases 0.000 abstract description 29
- 239000000470 constituent Substances 0.000 description 18
- 230000015572 biosynthetic process Effects 0.000 description 10
- 238000005275 alloying Methods 0.000 description 9
- 230000000694 effects Effects 0.000 description 8
- 230000009467 reduction Effects 0.000 description 8
- 238000002474 experimental method Methods 0.000 description 7
- 230000008023 solidification Effects 0.000 description 7
- 206010014970 Ephelides Diseases 0.000 description 6
- 230000008901 benefit Effects 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 239000010955 niobium Substances 0.000 description 3
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 description 3
- 238000005204 segregation Methods 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 230000001627 detrimental effect Effects 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 239000003999 initiator Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 239000000155 melt Substances 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 239000006104 solid solution Substances 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 238000004873 anchoring Methods 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229910001338 liquidmetal Inorganic materials 0.000 description 1
- 229910052758 niobium Inorganic materials 0.000 description 1
- 230000006911 nucleation Effects 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 238000010587 phase diagram Methods 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 238000003672 processing method Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- -1 tantalum (-3) Chemical compound 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C19/00—Alloys based on nickel or cobalt
- C22C19/03—Alloys based on nickel or cobalt based on nickel
- C22C19/05—Alloys based on nickel or cobalt based on nickel with chromium
- C22C19/051—Alloys based on nickel or cobalt based on nickel with chromium and Mo or W
- C22C19/057—Alloys based on nickel or cobalt based on nickel with chromium and Mo or W with the maximum Cr content being less 10%
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22D—CASTING OF METALS; CASTING OF OTHER SUBSTANCES BY THE SAME PROCESSES OR DEVICES
- B22D27/00—Treating the metal in the mould while it is molten or ductile ; Pressure or vacuum casting
- B22D27/04—Influencing the temperature of the metal, e.g. by heating or cooling the mould
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22D—CASTING OF METALS; CASTING OF OTHER SUBSTANCES BY THE SAME PROCESSES OR DEVICES
- B22D27/00—Treating the metal in the mould while it is molten or ductile ; Pressure or vacuum casting
- B22D27/04—Influencing the temperature of the metal, e.g. by heating or cooling the mould
- B22D27/045—Directionally solidified castings
Definitions
- the present invention generally relates to nickel-base superalloy compositions and processes for producing directionally-solidified castings therefrom. More particularly, the invention is directed to a nickel-base superalloy that is castable as single-crystal articles suitable for use as components of gas turbines and other high temperature applications.
- Components of gas turbines such as blades (buckets), vanes (nozzles) and combustor components, are typically formed of nickel, cobalt or iron-base superalloys characterized by desirable mechanical properties at turbine operating temperatures. Because the efficiency of a gas turbine is dependent on its operating temperatures, there is an ongoing effort to develop components, and particularly turbine buckets, nozzles, and combustor components, that are capable of withstanding higher temperatures. As the material requirements for gas turbine components have increased, various processing methods and alloying constituents have been used to enhance the mechanical, physical and environmental properties of components formed from superalloys.
- buckets, nozzles and other components employed in demanding applications are often cast by unidirectional casting techniques to have directionally-solidified (DS) or single-crystal (SX) microstructures, characterized by a crystal orientation or growth direction in a selected direction to produce columnar polycrystalline or single-crystal articles.
- DS directionally-solidified
- SX single-crystal
- the nickel-base superalloy commercially known as René N5, disclosed in commonly-assigned U.S. Patent No. 6,074,602 has a composition of, by weight percent, about 5-10 chromium, about 5-10 cobalt, about 0-2 molybdenum, about 3-10 tungsten, about 3-8 tantalum, about 0-2 titanium, about 5-7 aluminum, about 0-6 rhenium, about 0-0.50 hafnium,about 0-0.07 carbon, about 0-0.015 boron, and about 0-0.075 yttrium, the balance being nickel and incidental impurities.
- the composition of N5 is characterized by controlled concentrations of certain critical alloying elements to achieve a desired mix of properties.
- properties include high temperature creep strength, oxidation and corrosion resistance, resistance to low and high cycle fatigue (LCF and HCF), and single-crystal castability.
- N5 performs extremely well in applications within gas turbines, buckets and nozzles cast from N5 can exhibit external and internal freckle chains. Because excessive freckling can necessitate scrappage of a casting, there is a desire to reduce the incidence and level of freckling exhibited by the N5 alloy.
- the present invention provides an alloy composition and method by which the incidence of freckling can be reduced in a unidirectionally-solidified casting having properties similar to unidirectionally-solidified castings produced from the N5 nickel-base superalloy.
- a nickel-base superalloy that is particularly suitable for producing castings with unidirectional crystal structures.
- the superalloy has a composition consisting of, by weight, 6% to 8% chromium, 6% to 9% cobalt, 0% to 2% molybdenum, 4% to 6% tungsten, 6.4% to 6.9% tantalum, 0% to 2% titanium, 5% to 7% aluminum, 2.7% to 3.0% rhenium, 0.3% to 0.7% hafnium, 0.04% to 0.08% carbon, 0.002% to 0.006% boron, 0% to 0.075% yttrium, 0.002% to 0.004% zirconium, the balance being nickel and incidental impurities.
- a method for producing the nickel-base superalloy as a unidirectionally-solidified casting.
- the method generally entails pouring a molten quantity of the composition into a heated zone of a preheated mold comprising a main cavity having the shape of the cast article, withdrawing the mold from the heated zone into a cooling tank to directionally solidify the molten metal, and then cooling the mold to produce the casting and a unidirectional crystal structure.
- the unidirectional crystal structure of the casting is substantially free of freckle defects.
- the characterization of a casting being substantially free of freckles is meant to indicate a total combined length of all freckles of not more than 0.5 inch (about 12 mm).
- a significant advantage of this invention is that, though the composition of the casting contains the same alloying constituents as N5 and the alloying levels of these constituents are within or overlap the broadest ranges disclosed for N5, it has been determined that the employment of particular levels and relative proportions of these constituents are able to produce unexpected superior results as compared to the N5 alloy processed under identical conditions. In particular, the reduction in the incidence of freckling is greater than was expected for composition modifications alone, and the result is the absence of freckling that would otherwise be an expected property for N5 under the same processing conditions.
- the unidirectional crystal structure of the casting is a single crystal with a preferred single crystal direction of ⁇ 001>, though crystalline structures having orientations other than ⁇ 001> are also within the scope of the invention.
- the alloy composition and castings that can be produced in accordance with the invention are well suited for components of a gas turbine, such as buckets, nozzles, and other components of gas turbines.
- FIG. 1 depicts a first stage bucket 10 for a land-based gas turbine, such as the H and FB class gas turbines used in the power-generating industry and manufactured by the assignee of the invention.
- the bucket 10 has an airfoil 12 and shank 14, with a dovetail 16 formed on the shank 14 for anchoring the bucket 10 to a turbine disk (not shown).
- the bucket 10 is preferably unidirectionally cast to have a columnar single crystal (SX) or columnar polycrystalline (DS) microstructure. While the advantages of this invention will be described with reference to components of a gas turbine, such as the bucket 10 shown in FIG. 1 , the teachings of this invention are generally applicable to other components that benefit from being cast from a high-temperature material such as N5.
- freckles form in part as a result of molten metal convection in the casting mold which disrupts unidirectional solidification processes, producing irregularities seen on SX and DS casting surfaces as little chains of equiaxed, crystals.
- Freckles can act as grain initiators during the solidification process that cause unacceptable off-axial grains, and may reduce fatigue life of the casting.
- external and internal freckling can be inhibited and even eliminated in an alloy whose constituent levels fall within the ranges disclosed for N5 levels in US6,074,602 , the contents of which relating to composition of the N5 alloy are incorporated herein by reference.
- the alloy can be used to produce large castings that are typically more prone to freckling, including the large turbine buckets of land-based gas turbines such as that of FIG. 1 , whose overall lengths are often in the range of about fifteen to about forty inches (about forty to about one hundred centimeters).
- compositional modifications to the nickel-base N5 superalloy that achieve the reduction in freckling were initially based on data generated from a modeling technique, through which correlations were identified and analyzed regarding the overall effect of each element of the N5 alloy on freckle formation. Broad ranges disclosed in US6,074,602 for N5 are summarized in Table I below.
- R c Freckle Susceptibility Index
- the convection potency factor is a factor developed for use in the modeling technique, and takes into account several variables, including the slope of the liquidus line in the phase diagram, the segregation coefficient, solutal volume expansion coefficient, and thermal volume expansion coefficient. From the equation, it can be seen that elements with a negative convection potency factor (P i ) reduce the value of the Freckle Susceptibility Index, R e , and therefore are expected to reduce the risk of freckling, whereas elements with a positive P i value increase R c and are therefore expected to increase the risk of freckling.
- P i convection potency factor
- hafnium should have the strongest influence on freckle formation, and also the greatest beneficial effect on freckle prevention if present at sufficiently high levels. Tantalum and carbon also have beneficial effects, though not as pronounced as hafnium.
- aluminum, tungsten, and rhenium are expected to have a moderate detrimental effect due to their moderate P i values in combination with their significant alloying levels in the N5 alloy.
- the influence that any one element might have on the Freckle Susceptibility Index, R e can be predicted based on the product of its convection potency factor, P i , and its concentration tolerance ( ⁇ C i ) in the production specification for the N5 alloy.
- P i convection potency factor
- ⁇ C i concentration tolerance
- tantalum (-3) and carbon (-5) should have moderate positive affects
- rhenium (8), tungsten (6), and aluminum (2) should have moderate negative affects
- cobalt, chromium, and molybdenum should have negligible affects (approximately 0), as will titanium, columbium, and zirconium if present as impurities.
- the nickel-base alloy of the present invention nominally contains more hafnium, tantalum and carbon and less rhenium, tungsten, aluminum, and zirconium than the nominal N5 composition.
- tantalum and aluminum are both gamma prime (Ni 3 X) formers, a lower level of aluminum can be offset by the higher level of tantalum to maintain the gamma prime content desired for N5.
- rhenium and tungsten are both solid solution strengtheners, and as such attempts to offset reductions in both of these elements must be by increasing another solid solution strengthener in order to maintain the desirable properties of N5.
- experiments conducted to verify the predictions of the modeling technique suggested that the influence of hafnium may be sufficiently strong to eliminate freckling if hafnium levels exceed the hafnium upper limit for N5 (0.50 weight percent).
- the nickel-base alloy has the following broad, preferred, and nominal compositions, by weight.
- TABLE III Constituent Broad Range Preferred Range Nominal Chromium 6 to 8 % 6.75 to 7.25 % 7 % Cobalt 6 to 9 % 7 to 8 % 7.5 % Molybdenum 0 to 2 % 1.3 to 1.7 % 1.5 % Tungsten 4 to 6 % 4.5 to 5.0 % 4.75 % Tantalum 6.4 to 6.9 % 6.5 to 6.8 % 6.7 % Titanium 0 to 2 % 0 to 0.05 % 0 to 0.02 % Aluminum 5 to 7 % 6.0 to 6.2 % 6.1 % Rhenium 2.7 to 3.0 % 2.75 to 2.9 % 2.8 % Carbon 0.04 to 0.08 % 0.05 to 0.07 % 0.06 % Hafnium 0.3 to 0.7 % 0.4 to 0.6 % 0.5 % Boron 0.002 to 0.006 % 0.003 to 0.005 %
- the balance of the alloy is nickel and incidental impurities. Aside from freckling reduction, the alloy of this invention preferably has essentially the same properties as, and potentially superior properties to, the N5 alloy, and therefore provides an excellent alternative to N5.
- Components of the alloy having SX or DS microstructures can be produced by generally conventional casting unidirectional casting techniques.
- single-crystal castings are produced from a melt of the alloy, for example, prepared by known vacuum induction melting techniques.
- heat transfer conditions during the solidification of the casting are controlled so that one section of the casting article does not cool faster than the rest of the casting to avoid the nucleation and formation of secondary grains from the melt in competition with the primary columnar single crystal.
- Secondary and multi-grains are further controlled by adjusting the heat transfer conditions during the withdrawal of the mold into a cooling bath or radiation cooling zone to ensure that all parts of the casting cool at approximately the same rate.
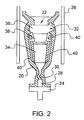
- FIG. 2 represents a shell mold 20 of a type suitable for producing a single-crystal casting of the alloy of this invention.
- the mold 20 is preferably formed of a material such as alumina or silica, and has an internal cavity 22 corresponding to the desired shape of a casting 32, represented as a turbine bucket similar to that of FIG. 1 .
- the cavity 22 is configured to produce the casting 32 with an airfoil portion 34, shank 36, and dovetail 38.
- the mold 20 is shown secured to a chill plate 24 and placed in a heating zone 26 to heat the mold 20 to a temperature equal to or above the melting temperature of the alloy, and more particularly above the liquidus temperature of the alloy (about 1397°C).
- the casting process is preferably carried out in a vacuum or an inert atmosphere.
- the molten alloy is poured into the preheated mold 20, after which unidirectional solidification is initiated by withdrawing the base of the mold 20 and chill plate 24 downwardly at a fixed rate to a cooling zone (not shown), which may contain a liquid metal cooling bath or a vacuum or ambient or cooled air for radiation cooling.
- the casting 32 grows epitaxially (for example, with the ⁇ 100> orientation) based on the crystalline structure and orientation of a small block of single-crystal seed material 28 at the base of the mold 20, from which a single crystal forms from a crystal selector 30.
- the columnar single crystal becomes larger in the enlarged section of the cavity 22.
- a bridge 40 connects protruding sections of the casting 32 with lower sections of the casting 32 so that a unidirectional columnar single crystal forms substantially throughout the casting 32.
- the casting 32 is deemed to be a substantially columnar single crystal if more than 50% of the casting is single crystal.
- a DS casting can be produced in a similar manner, though with modifications to the mold 20, such a growth zone at the base of the mold 20 that is open to the chill plate 24, and omission of the crystal selector 30.
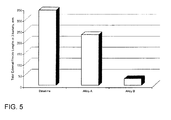
- Alloys A and B were substantially identical to the Baseline Alloy except for their levels of tungsten (which differed by only 0.15% from the Baseline), tantalum (which differed by only 0.24% or 0.25% from the Baseline), aluminum (which differed by only 0.15% from the Baseline), rhenium (which differed by only 0.11 % and 0.13% from the Baseline), hafnium (which differed by only 0.06% or 0.36% from the Baseline), and zirconium (which differed by only 0.004% from the Baseline). Nonetheless, it will be seen that these small changes in chemistry had a significant impact on the incidence of freckling in their respective alloys.
- FIGS. 3 and 4 are scanned images showing external freckle chains in buckets formed of the Baseline Alloy and Alloy B, respectively.
- FIG. 3 more than twenty freckle chains can be seen on the bucket root of the Baseline Alloy casting.
- FIG. 5 is a graph plotting freckle length measurement data obtained from the buckets cast from all three alloys.
Landscapes
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Chemical & Material Sciences (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Turbine Rotor Nozzle Sealing (AREA)
- Crystals, And After-Treatments Of Crystals (AREA)
Claims (15)
- Superlegierung auf Nickelbasis, die eine Zusammensetzung aufweist, die, bezogen auf das Gewicht, besteht aus:6% bis 8% Chrom,6% bis 9% Kobalt,0% bis 2% Molybdän;4% bis 6% Wolfram,6,4% bis 6,9% Tantal,0% bis 2% Titan,5% bis 7% Aluminium,2,7% bis 3,0% Rhenium,0,3% bis 0,7% Hafnium,0,04% bis 0,08% Kohlenstoff,0,002% bis 0,006% Bor,0% bis 0,075% Yttrium,0,002% bis 0,004% Zirkonium,Rest Nickel und üblichen Verunreinigungen.
- Superlegierung auf Nickelbasis nach Anspruch 1, worin die Superlegierung auf Nickelbasis in der Form eines Gussstückes vorliegt.
- Superlegierung auf Nickelbasis nach Anspruch 2, worin das Gussstück im wesentlichen frei von Seigerungsfehlern ist.
- Superlegierung auf Nickelbasis nach Anspruch 2, worin das Gussstück eine unidirektionale Kristallstruktur aufweist.
- Superlegierung auf Nickelbasis nach Anspruch 4, worin die unidirektionale Kristallstruktur ein Einkristall ist.
- Superlegierung auf Nickelbasis nach Anspruch 4, worin das Gussstück eine Komponente für eine Gasturbine ist.
- Superlegierung auf Nickelbasis nach irgendeinem vorhergehenden Anspruch, worin die Zusammensetzung 6,5 bis 6,8 Gew.-% Tantal enthält.
- Superlegierung auf Nickelbasis nach irgendeinem vorhergehenden Anspruch, worin die Zusammensetzung 6 bis 6,2 Gew.-% Aluminium enthält.
- Superlegierung auf Nickelbasis nach irgendeinem vorhergehenden Anspruch, worin die Zusammensetzung 2,75 bis 2,9 Gew.-% Rhenium enthält.
- Superlegierung auf Nickelbasis nach irgendeinem vorhergehenden Anspruch, worin die Zusammensetzung 0,4 bis 0,6 Gew.-% Hafnium enthält.
- Superlegierung auf Nickelbasis nach irgendeinem vorhergehenden Anspruch, worin die Zusammensetzung 0,002 bis 0,003 Gew.-% Zirkonium enthält.
- Superlegierung auf Nickelbasis nach irgendeinem der Ansprüche 1 bis 6, worin die Zusammensetzung, bezogen auf das Gewicht, weniger als 4,90% Wolfram, mehr als 6,44% Tantal, weniger als 6,20% Aluminium und weniger als 2,91% Rhenium enthält.
- Verfahren zum Herstellen der Superlegierung auf Nickelbasis nach irgendeinem vorhergehenden Anspruch, wobei das Verfahren umfasst:Giessen einer geschmolzenen Menge der Zusammensetzung in eine erhitzte Zone einer vererhitzten Form, die einen Haupthohlraum umfasst,Herausziehen der Form aus der erhitzten Zone in eine Kühlzone zum gerichteten Erstarren des geschmolzenen Metalles und dannAbkühlen der Form zum Herstellen eines Gussstückes, das eine unidirektionale Kristallstruktur aufweist, die im wesentlichen frei von Seigerungsdefekten ist.
- Superlegierung auf Nickelbasis nach Anspruch 1, in der Form eines Gussstückes, das eine unidirektionale Kristallstruktur aufweist, die im wesentlichen frei von Seigerungsdefekten ist, wobei die Superlegierung eine Zusammensetzung aufweist, die, bezogen auf das Gewicht, besteht aus:6,75% bis 7,25% Chrom,7% bis 8% Kobalt,1,3% bis 1,7% Molybdän;4,5% bis 5,0% Wolfram,6,5% bis 6,8% Tantal,0% bis 0,05% Titan,6,0% bis 6,2% Aluminium,2,75% bis 2,90% Rhenium,0,4% bis 0,6% Hafnium,0,04% bis 0,08% Kohlenstoff,0,003% bis 0,005% Bor,0% bis 0,075% Yttrium,0,002% bis 0,003% Zirkonium,Rest Nickel und üblichen Verunreinigungen.
- Superlegierung auf Nickelbasis nach Anspruch 14, wobei die Zusammensetzung, bezogen auf das Gewicht, besteht aus:etwa 7% Chrom,etwa 7,5% Kobalt,etwa 1,5% Molybdän;etwa 4,75% Wolfram,etwa 6,7% Tantal,etwa 0% bis 0,02% Titan,etwa 6,1% Aluminium,etwa 2,8% Rhenium,etwa 0,5% Hafnium,etwa 0,06% Kohlenstoff,etwa 0,004% Bor,0% bis 0,03% Yttrium,etwa 0,0025% Zirkonium,Rest Nickel und üblichen Verunreinigungen.
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/187,049 US20100034692A1 (en) | 2008-08-06 | 2008-08-06 | Nickel-base superalloy, unidirectional-solidification process therefor, and castings formed therefrom |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2154258A1 EP2154258A1 (de) | 2010-02-17 |
| EP2154258B1 true EP2154258B1 (de) | 2011-05-18 |
Family
ID=41402441
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP09166869A Active EP2154258B1 (de) | 2008-08-06 | 2009-07-30 | Auf Nickel basierende Superlegierung, zugehöriges Verfahren zur unidirektionalen Verfestigung und daraus gebildete Gussstücke |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20100034692A1 (de) |
| EP (1) | EP2154258B1 (de) |
| JP (1) | JP2010037658A (de) |
| AT (1) | ATE510035T1 (de) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8974865B2 (en) | 2011-02-23 | 2015-03-10 | General Electric Company | Component and a method of processing a component |
| US9555471B2 (en) | 2014-01-28 | 2017-01-31 | General Electric Company | Casting method and cast article |
| US10265764B2 (en) | 2014-01-28 | 2019-04-23 | General Electric Company | Casting method and cast article |
| WO2015183955A2 (en) * | 2014-05-27 | 2015-12-03 | Questek Innovations Llc | Highly processable single crystal nickel alloys |
| US11181002B2 (en) | 2016-09-29 | 2021-11-23 | General Electric Company | Turbine systems with sealing components |
| EP3335817A1 (de) * | 2016-12-19 | 2018-06-20 | General Electric Company | Giessverfahren und gussartikel |
| CA3068408C (en) * | 2017-06-30 | 2023-01-17 | Siemens Aktiengesellschaft | An additive manufacturing technique for precipitation-hardened superalloy powdered material |
Family Cites Families (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1562082A (en) * | 1977-10-17 | 1980-03-05 | Gen Electric | Nickel-base olloys |
| US5100484A (en) | 1985-10-15 | 1992-03-31 | General Electric Company | Heat treatment for nickel-base superalloys |
| US6074602A (en) | 1985-10-15 | 2000-06-13 | General Electric Company | Property-balanced nickel-base superalloys for producing single crystal articles |
| US4895201A (en) * | 1987-07-07 | 1990-01-23 | United Technologies Corporation | Oxidation resistant superalloys containing low sulfur levels |
| US5151249A (en) * | 1989-12-29 | 1992-09-29 | General Electric Company | Nickel-based single crystal superalloy and method of making |
| US5489194A (en) * | 1990-09-14 | 1996-02-06 | Hitachi, Ltd. | Gas turbine, gas turbine blade used therefor and manufacturing method for gas turbine blade |
| EP0560296B1 (de) * | 1992-03-09 | 1998-01-14 | Hitachi Metals, Ltd. | Hochgradig heisskorrosionsbeständige und hochfeste Superlegierung, hochgradig heisskorrosionsbeständiges und hochfestes Gussstück mit Einkristallgefüge, Gasturbine und kombiniertes Kreislaufenergieerzeugungssystem |
| JPH09157777A (ja) * | 1995-12-12 | 1997-06-17 | Mitsubishi Materials Corp | 耐熱疲労特性、高温クリープおよび高温耐食性に優れたNi基合金 |
| JP3146345B2 (ja) | 1996-03-11 | 2001-03-12 | アムコー テクノロジー コリア インコーポレーティド | バンプチップスケール半導体パッケージのバンプ形成方法 |
| DE19624056A1 (de) * | 1996-06-17 | 1997-12-18 | Abb Research Ltd | Nickel-Basis-Superlegierung |
| DE19624055A1 (de) * | 1996-06-17 | 1997-12-18 | Abb Research Ltd | Nickel-Basis-Superlegierung |
| US5925198A (en) * | 1997-03-07 | 1999-07-20 | The Chief Controller, Research And Developement Organization Ministry Of Defence, Technical Coordination | Nickel-based superalloy |
| US6217286B1 (en) * | 1998-06-26 | 2001-04-17 | General Electric Company | Unidirectionally solidified cast article and method of making |
| US6096141A (en) * | 1998-08-03 | 2000-08-01 | General Electric Co. | Nickel-based superalloys exhibiting minimal grain defects |
| JP4222540B2 (ja) * | 2000-08-30 | 2009-02-12 | 独立行政法人物質・材料研究機構 | ニッケル基単結晶超合金、その製造方法およびガスタービン高温部品 |
| JP4184648B2 (ja) * | 2001-10-19 | 2008-11-19 | 株式会社日立製作所 | 強度及び耐食性に優れたNi基単結晶合金とその製造法 |
| US6789315B2 (en) * | 2002-03-21 | 2004-09-14 | General Electric Company | Establishing a throat area of a gas turbine nozzle, and a technique for modifying the nozzle vanes |
| US6909988B2 (en) * | 2002-10-15 | 2005-06-21 | General Electric Company | Method for positioning defects in metal billets |
| CA2440573C (en) * | 2002-12-16 | 2013-06-18 | Howmet Research Corporation | Nickel base superalloy |
| WO2005064027A1 (ja) * | 2003-12-26 | 2005-07-14 | Kawasaki Jukogyo Kabushiki Kaisha | Ni基超耐熱合金及びそれを用いたガスタービン部品 |
| KR20060045542A (ko) * | 2004-04-07 | 2006-05-17 | 유나이티드 테크놀로지스 코포레이션 | 내산화성 초합금 및 제품 |
| JP4885530B2 (ja) * | 2005-12-09 | 2012-02-29 | 株式会社日立製作所 | 高強度高延性Ni基超合金と、それを用いた部材及び製造方法 |
| JP4719583B2 (ja) * | 2006-02-08 | 2011-07-06 | 株式会社日立製作所 | 強度、耐食性及び耐酸化特性に優れた一方向凝固用ニッケル基超合金及び一方向凝固ニッケル基超合金の製造方法 |
| JP5252348B2 (ja) * | 2006-03-20 | 2013-07-31 | 独立行政法人物質・材料研究機構 | Ni基超合金とその製造方法およびタービンブレードまたはタービンベーン部品 |
| FR2899240B1 (fr) * | 2006-03-31 | 2008-06-27 | Snecma Sa | Alliage a base de nickel |
| WO2007122931A1 (ja) * | 2006-03-31 | 2007-11-01 | National Institute For Materials Science | Ni基超合金とその製造方法 |
| JP4773303B2 (ja) * | 2006-08-22 | 2011-09-14 | 株式会社日立製作所 | 強度、耐食性、耐酸化特性に優れたニッケル基単結晶超合金及びその製造方法 |
| RU2415190C2 (ru) * | 2006-09-13 | 2011-03-27 | Нэшнл Инститьют Фор Матириалз Сайенс | МОНОКРИСТАЛЛИЧЕСКИЙ СУПЕРСПЛАВ НА ОСНОВЕ Ni |
| EP1914327A1 (de) * | 2006-10-17 | 2008-04-23 | Siemens Aktiengesellschaft | Nickel-Basis-Superlegierung |
| US7704332B2 (en) * | 2006-12-13 | 2010-04-27 | United Technologies Corporation | Moderate density, low density, and extremely low density single crystal alloys for high AN2 applications |
-
2008
- 2008-08-06 US US12/187,049 patent/US20100034692A1/en not_active Abandoned
-
2009
- 2009-07-30 AT AT09166869T patent/ATE510035T1/de not_active IP Right Cessation
- 2009-07-30 EP EP09166869A patent/EP2154258B1/de active Active
- 2009-08-05 JP JP2009181999A patent/JP2010037658A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| ATE510035T1 (de) | 2011-06-15 |
| US20100034692A1 (en) | 2010-02-11 |
| JP2010037658A (ja) | 2010-02-18 |
| EP2154258A1 (de) | 2010-02-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2154258B1 (de) | Auf Nickel basierende Superlegierung, zugehöriges Verfahren zur unidirektionalen Verfestigung und daraus gebildete Gussstücke | |
| EP0789087B1 (de) | Hochfeste Superlegierung auf Nickelbasis für gerichtet erstarrte Giesteilen | |
| JP4659164B2 (ja) | 一方向凝固鋳造品並びにその製造方法 | |
| EP0577316B1 (de) | Monokristalline Superlegierung auf Nickelbasis | |
| JP6767155B2 (ja) | 物品及び物品の形成方法 | |
| EP2204475A2 (de) | Unidirektionales Erstarrungsverfahren und daraus geformte Gussformen | |
| US20130022803A1 (en) | Unidirectionally-solidification process and castings formed thereby | |
| CA1206398A (en) | Superalloy single crystal articles | |
| US8186418B2 (en) | Unidirectional solidification process and apparatus therefor | |
| JP4885530B2 (ja) | 高強度高延性Ni基超合金と、それを用いた部材及び製造方法 | |
| US9144842B2 (en) | Unidirectional solidification process and apparatus and single-crystal seed therefor | |
| Erickson | Polycrystalline cast superalloys | |
| JP4266196B2 (ja) | 強度、耐食性及び耐酸化特性に優れたニッケル基超合金 | |
| JP4222540B2 (ja) | ニッケル基単結晶超合金、その製造方法およびガスタービン高温部品 | |
| JP4719583B2 (ja) | 強度、耐食性及び耐酸化特性に優れた一方向凝固用ニッケル基超合金及び一方向凝固ニッケル基超合金の製造方法 | |
| Clemens et al. | Advanced solidification processing of an industrial gas turbine engine component | |
| JP4607490B2 (ja) | ニッケル基超合金及び単結晶鋳造品 | |
| JP7187864B2 (ja) | 合金の製造方法 | |
| JP2005139548A (ja) | ニッケル基超合金及び単結晶鋳造品 | |
| KR20180081313A (ko) | 방향성 응고 Ni기 초내열 합금 및 이의 제조 방법 | |
| JP4773303B2 (ja) | 強度、耐食性、耐酸化特性に優れたニッケル基単結晶超合金及びその製造方法 | |
| CA2503326C (en) | Heat treatment of alloys having elements for improving grain boundary strength | |
| TWI663263B (zh) | 高抗潛變等軸晶鎳基超合金 | |
| TW201522656A (zh) | 高應力等軸晶鎳基合金 | |
| JP7160305B2 (ja) | TiAl鋳造合金およびその製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA RS |
|
| 17P | Request for examination filed |
Effective date: 20100817 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: B22D 27/04 20060101ALI20101001BHEP Ipc: C22C 19/05 20060101AFI20101001BHEP |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602009001290 Country of ref document: DE Effective date: 20110630 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20110518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110818 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110919 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110918 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110819 Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110829 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110731 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20120330 |
|
| 26N | No opposition filed |
Effective date: 20120221 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110801 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602009001290 Country of ref document: DE Effective date: 20120221 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110730 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20120727 Year of fee payment: 4 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110730 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110518 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20130730 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602009001290 Country of ref document: DE Effective date: 20140201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140201 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130730 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130731 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130731 |