EP2575807B1 - Formulation comprenant du sulpiride et une composition homeopathique synergistique pour le traitement de la depression - Google Patents
Formulation comprenant du sulpiride et une composition homeopathique synergistique pour le traitement de la depression Download PDFInfo
- Publication number
- EP2575807B1 EP2575807B1 EP10729933.1A EP10729933A EP2575807B1 EP 2575807 B1 EP2575807 B1 EP 2575807B1 EP 10729933 A EP10729933 A EP 10729933A EP 2575807 B1 EP2575807 B1 EP 2575807B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- acid
- sodium
- polyoxyethylene
- excipient
- mixtures
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K41/00—Medicinal preparations obtained by treating materials with wave energy or particle radiation ; Therapies using these preparations
- A61K41/0004—Homeopathy; Vitalisation; Resonance; Dynamisation, e.g. esoteric applications; Oxygenation of blood
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0087—Galenical forms not covered by A61K9/02 - A61K9/7023
- A61K9/0095—Drinks; Beverages; Syrups; Compositions for reconstitution thereof, e.g. powders or tablets to be dispersed in a glass of water; Veterinary drenches
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
- A61K9/2018—Sugars, or sugar alcohols, e.g. lactose, mannitol; Derivatives thereof, e.g. polysorbates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4841—Filling excipients; Inactive ingredients
- A61K9/4866—Organic macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/20—Hypnotics; Sedatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
Definitions
- the present invention relates to a formulation based on sulpiride and a synergistic homeopathic composition that exhibits a profound antidepressant activity.
- This invention discloses an improved pharmaceutical product which is used as antidepressant, based on the formulation comprising:
- the homeopathic composition described under point (ii) can be considered as modified Krebs cycle solution (or homeopathic modified Krebs solution).
- the above cited formulation exhibits a profound antidepressant activity due to synergistic action of homeopathic composition of Krebs cycle-involving components and herbal extracts, providing:
- depression is one of the most important disorders that affects the modern people.
- the depressive disorder is a serious illness that affects energy, sleep, appetite, libido, and the ability to function (expecially major depression), and has to be treated with various drugs usually called antidepressants.
- antidepressants usually called antidepressants.
- the latter involve several classes of compounds:
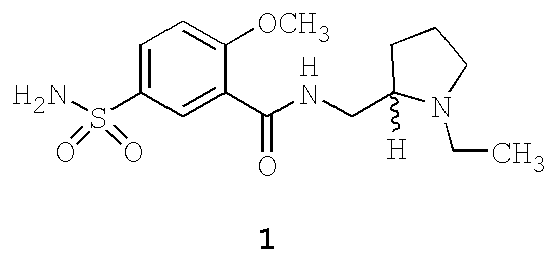
- Sulpiride ( 1 ) is a well-known active pharmaceutical ingredient (API) from the class of dopamine D 2 and D 3 receptor antagonists with antipsychotic and antidepressant activities.
- sulpiride Main pharmacological use of sulpiride involves treatment of psychoses such as schizophrenia at daily dosages of 400-1.600 mg [ G. Alfredsson, F. A. Wiesel: Relationships between clinical effects and monoamine metabolites and amino acids in sulpiride-treated schizophrenic patients, Psychopharmacology (Berl.) 101 (1990) 324-331 ; G. Alfredsson, L. Bjerkenstedt, G. Edman, C. Harnryd, G. Oxenstierna, G. Sedvall, F. A.
- Wiesel Relationships between drug concentrations in serum and CSF, clinical effects and monoaminergic variables in schizophrenic patients treated with sulpiride or chlorpromazine, Acta Psychiatr. Scand. Suppl. 311 (1984) 49-74 ].
- sulpiride is usually administered either in 2-3 divided oral doses, or by intravenous injection.
- Table 1 shows effects of sulpiride in combinations with various APIs: Table 1 Sulpiride combined with Publication No. Applicant Pharmacological activity 1. cannabinoids GB2456183 antipsychotic GW PHARMA LTD, ... 2. sertraline CN101138562 antidepressant YANFANG CHEN 3. venlafaxine CN1931153 antidepressant CHEN YANFANG 4. oxybutynin and tolterodine US2008207737 treatment of hyperhidrosis ZINGER MENNI 5. trazodone RU2334509 treatment of eczema GOU VPO VOENNO MED ... 6.
- the modified Krebs solution like herein-mentioned, has not been recognized as sole homeopathic therapy, but has been used in combination with other homeopathic remedies. Also, so far, it has not been recognized as component in combined homeopathic-allopathic therapy.
- German based company produces and sales such homeopathic modified Krebs solution for the indication: "stimulation of blocked enzymatic systems in degenerative diseases, as well as in defective enzymatic functions", which gives a property of unspecific metabolic activator according to the opinion of the manufacturer [ A. Bier: Ordinatio Antihomotoxica et Materia Medica, Heel GmbH, Baden-Baden, Germany (1991 )].
- the present invention discloses an improved pharmaceutical product which is used as antidepressant, based on the formulation consisting of:
- the duration of administering was three months (90 days; also subcutaneously).
- Hamilton rating scale for depression also known as HAM-D or HAMD test, which is generally accepted as "golden standard” for quantification of severity of depression symptoms like: low mood, anxiety, agitation, insomnia, weight loss
- M. Hamilton A rating scale for depression, J. Neurol. Neurosurg. Psychiatr. 23 (1960) 56-62 ; J. L. Hedlung, B. W. Viewig: The Hamilton rating scale for depression: a comprehensive review, J. Oper. Psychiatr. 10 (1979) 149-165 ].
- Results are presented in Table 2. showing Hamilton rating scale for depression (HAMD) test results of controlled-study of the antidepressant activity of the composition from the invention in comparison to placebo.
- scores can range from 0 to 54.
- One interpretation suggests that scores between 0 and 6 indicate a normal person with regard to depression, scores between 7 and 17 indicate mild depression, scores between 18 and 24 indicate moderate depression, and scores over 24 indicate severe depression.
- Paired T-test is usually chosen to establish the difference between groups, i.e. their mean values during the Study.
- the formulation of the present invention allows effective antidepressant therapy without side-effects which are common at classical dosage rates of sulpiride administration such as: sedation, constipation, and dryness of mouth [ T. Tsukamoto, M. Asakura, T. Tsuneizumi, Y. Satoh, T. Shinozuka, K. Hasegawa: Therapeutic effects and side-effects in patients with major depression treated with sulpiride once a day, Prog. Neuropsychopharmacol. Biol. Psychiatry 18 (1994) 615-618 ].
- the formulation of this invention is consisting of:
- excipients are selected from the groups consisting of diluents, isotonic salts, emulsifiers (solubilizers), thickeners, fillers, binders, disintegrants, lubricants, preservatives, antioxidants, and stabilizers.
- the diluent is a liquid compound selected from the group consisting of purified water, glycerol, 1,2-propyleneglycol, liquid polyethyleneglycols, liquid polyglycerols, aqueous sorbitol, dimethylsulfoxide, or mixtures of these substances.
- oral solutions can contain as diluent also 1,3-propyleneglycol and 1,3-butanediol.
- the diluent is selected from the group comprising purified water, glycerol, 1,2-propyleneglycol, honey, aqueous sorbitol, or mixtures of these substances.
- isotonic salts that can be used in preparation of the formulation is selected from the group consisting of sodium chloride (NaCl), potassium chloride (KCl), magnesium chloride (MgCl 2 ) and its hydrates, calcium chloride (CaCl 2 ) and its hydrates, or mixtures of these substances.
- the emulsifier is selected from the group comprising: sodium laurylsulphate, sodium lauryl ethyleneglycolsulphate, sodium lauryl diethyleneglycolsulphate, potassium laurylsulphate, potassium lauryl ethyleneglycolsulphate, potassium lauryl diethyleneglycolsulphate; sodium or potassium cocoamphodipropionate; disodium or dipotassium cocoamphodiacetate; polyoxyethylene(10) laurylether, polyoxyethylene(23) laurylether, polyoxyethylene(10) stearylether; polyoxyethylene(23) stearylether; polyoxyethylene(10) oleylether; polyoxyethylene(23) oleylether, other ethoxylates of higher fatty alcohols with H.L.B.
- Thickeners in the liquid final dosage forms of the formulation are selected from the group consisting of: polyacrylic acid, its co-polymers, or their sodium, or potassium salts; methylcellulose; sodium carboxymethylcellulose; 2-hydroxyethylcellulose; 2-hydroxypropylcellulose; starch; modified starches; polyglycerols; polyethyleneglycols; gelatin; pectin; agar agar; carrageenans; gum arabic; alginic acid; sodium alginate; or mixtures of these substances.
- Filler is selected from the group consisting of microcrystalline cellulose, lactose monohydrate, calcium hydrogenphosphate, calcium sulfate dihydrate, calcium carbonate, colloidal silicic acid, sorbitol, inulin, starch, modified starches, dextrin, glucose, fructose, basic magnesium carbonate, calcium silicate, saccharose, or mixtures of these substances.
- binders are selected from the group comprising gelatin, lactose monohydrate, sorbitol, saccharose, xylitol, maltitol, starch, modified starches, methylcellulose, 2-hydroxyethylcellulose, 2-hydroxypropylcellulose, 2-hydroxypropyl methylcellulose, sodium carboxymethylcellulose, polyethyleneglycols, polyglycerols, polyvinylpyrrolidone, polyvinylpyrrolidone co-polymers, carrageenans, maltodextrin, or mixtures of these substances.
- Disintegrants in solid dosage forms are selected from the group consisting of starch, modified starches, sodium starch glycolate, methylcellulose, sodium carboxymethylcellulose, 2-hydroxyethylcellulose, 2-hydroxypropylcellulose, 2-hydroxypropyl methylcellulose, polyvinylpyrrolidone, polyvinylpyrrolidone co-polymers, or mixtures of these substances.
- Lubricants in solid dosage forms are selected from the group consisting of magnesium stearate, calcium stearate, zinc stearate, stearic acid, talc, silicon dioxide, or mixtures of these substances.
- Preservatives are selected from the group comprising methyl 4-hydroxybenzoate, ethyl 4-hydroxybenzoate, propyl 4-hydroxybenzoate, butyl 4-hydroxybenzoate, sorbic acid, potassium sorbate, benzoic acid, sodium benzoate, 2-phenoxyethanol, 4-chloro-m-cresol, thymol, eugenol, or mixtures of these substances.
- Antioxidants are selected from the group consisting of 2,6-di- terc- butyl-4-hydroxytoluene (BHT), terc -butylhydroxyanisole (BHA), tocopherol, tocopheryl acetate, ascorbic acid, or mixtures of these substances.
- Stabilizers are selected from the group consisting of disodium ethylenediamine tetraacetate (Na 2 EDTA ⁇ 2H 2 O), disodium N-(2-hydroxyethyl)ethylenediamine triacetate [Na 2 H(HEDTA)], disodium diethylenetriamine pentaacetate [Na 2 H 3 (DTPA)], disodium citrate [Na 2 C(OH) (COOH) (CH 2 COO) 2 ], or mixtures of these substances.
- the formulation of this invention involves the final dosage forms of injections, tablets, capsules, oral solutions, and syrups.
- the formulation is produced by common procedures known to those skilled in the art of pharmaceutical technology [ J. Swarbrick (Ed.): Encyclopedia of Pharmaceutical Technology, 3rd Ed. (2007) Informa Healthcare Inc., USA ].
- Injections are produced by dissolution of homeopathic Krebs cycle components and herbal extracts in purified water, followed by addition of sulpiride ( 1 ) and sodium chloride (or other physiologically compatible inorganic salt up to isotonic level).
- sulpiride ( 1 ) and sodium chloride or other physiologically compatible inorganic salt up to isotonic level.
- Oral solutions are obtained by dissolution of sulpiride in purified water or its mixtures with ethanol, 1,2-propyleneglycol and similar diluents (solvents) with or without addition of pharmaceutically acceptable acids such as sulfuric, hydrochloric, phosphoric, citric, malic, fumaric, tartaric, benzenesulfonic acid, etc.
- pharmaceutically acceptable acids such as sulfuric, hydrochloric, phosphoric, citric, malic, fumaric, tartaric, benzenesulfonic acid, etc.
- the latter facilitate dissolution of sulpiride in water by forming its salts which are smoothly soluble in water and water-based mixtures.
- the homeopathic modified Krebs solution according to the invention is added, and the product is homogenized by short mixing.
- the oral solution can be stabilized by addition of preservatives like methyl- and propyl-4-hydroxybenzoate, antioxidant (e.g. BHT), and stabilizer (e.g. Na 2
- Syrup is produced by homogenization of sulpiride and homeopathic modified Krebs solution according to the invention in viscous aqueous solution of saccharose, honey, glucose, fructose, sorbitol, or their mixtures.
- intermediate solution is thickened by addition of edible thickeners like gelatin, pectin, starch, modified starches, sodium carboxymethylcellulose, or mixtures of these substances.
- Sweetener is selected from the group consisting of sodium saccharin, acesulfame potassium, sucralose, sodium or calcium cyclamate, xylitol, sorbitol, glycyrrhizin, extract of Liquorice root, or mixtures of these substances.
- a liquid homeopathic part of the formulation is converted into a powder by adsorption onto an inert inorganic carrier.
- the latter can be a filler or a mixture of filler and binder as defined above, e.g. microcrystalline cellulose, and/or lactose monohydrate. This operation is performed in a vacuum evaporator (at laboratory scale), or in a spray-drier (at industrial scale).
- powderous homeopathic part of the formulation (adsorbed on the filler) is homogenized with sulpiride, and subjected to either:
- Capsules are produced by filling of gelatin or vegetable capsules either with a homogeneous mixture of powderous homeopathic part of the formulation with sulpiride, or with afore-mentioned granulate (produced by wet granulation technique).
- Composition 1000 mL of injection solution: (a) sulpiride (1; 8.33 g; 0.83% w/w), (b) homeopathic modified Krebs solution (216.67 g; 21.67% w/w), (c) sodium chloride (8.90 g; 0.89% w/w), (d) purified water (766.10 g; 76.61% w/w).
- each ampoule (2.4 mL) of thus prepared oral solution contains: 20 mg of sulpiride ( 1 ) and 260 mg of homeopathic modified Krebs solution (20 mg of each of its 26 components).
- Composition 1000 g-scale: (a) citric acid (D8; 38.46 g; 3.85% w/w), (b) cis -aconitic acid (D8; 38.46 g; 3.85% w/w), (c) ⁇ -ketoglutaric acid (D8; 38.46 g; 3.85% w/w), (d) succinic acid (D10; 38.46 g; 3.85% w/w), (e) fumaric acid (D8; 38.46 g; 3.85% w/w), (f) DL-malic acid (D8; 38.46 g; 3.85% w/w), (g) sodium diethyloxaloacetate (D6; 38.46 g; 3.85% w/w), (h) sodium pyruvate (D8; 38.46 g; 3.85% w/w), (i) barium oxalosuccinate (D10; 38.46 g; 3.85% w/w), (j) coen
- Composition 1000 g of oral solution: (a) sulpiride ( 1 ; 20.00 g; 2% w/w), (b) homeopathic modified Krebs solution (260.00 g; 26% w/w), (c) ethanol (300.00 g; 30% w/w), (d) purified water (420.00 g; 42% w/w).
- Each mL of thus prepared oral solution contains: 20 mg of sulpiride ( 1 ) and 260 mg of homeopathic modified Krebs solution (10 mg of each of its 26 components).
- Composition 1000 g of syrup: (a) sulpiride ( 1 ; 4.00 g; 0.4% w/w), (b) homeopathic modified Krebs solution (104.00 g; 10.4% w/w), (c) saccharose (600.00 g; 60% w/w), (d) methyl 4-hydroxybenzoate (2.00 g; 0.2% w/w), (e) propyl 4-hydroxybenzoate (1.00 g; 0.1% w/w), (f) ethanol (96%; 10.00 g; 1% w/w), (g) BHT (0.01 g; 0.001% w/w), (h) Na 2 EDTA (0.1 g; 0.01% w/w), (i) purified water (278.89 g; 27.9% w/w) .
- Each 5 mL-dosing spoon of thus prepared syrup contains: 20 mg of sulpiride (1) and 520 mg of homeopathic modified Krebs solution (20 mg of each of its 26 components).
- Composition 1000 g of tablet mixture: (a) Microcrystalline cellulose (600.00 g; 60% w/w), (b) lactose monohydrate (305.89 g; 30.6% w/w), (c) sodium starch glycolate (25.00 g; 2.5% w/w), (d) polyvinylpyrrolidone (20.00 g; 2% w/w), (e) sulpiride ( 1 ; 40.00 g; 4% w/w), (f) homeopathic modified Krebs solution (300.00 g; corresponds to 150 mg per each 500 mg-tablet), (g) BHT (0.01 g; 0.001% w/w), (h) Na 2 EDTA (0.1 g; 0.01% w/w), (i) magnesium stearate (9.00 g; 0.9% w/w).
- Each tablet contains: 20 mg of sulpiride ( 1 ) and 150 mg of homeopathic modified Krebs solution (5.8 mg of each of its 26 components).
- Composition 1000 g of mixture for capsules: (a) Microcrystalline cellulose (945.50 g; 94-55% w/w), (b) sulpiride ( 1 ; 44.50 g; 4.45% w/w), (c) homeopathic modified Krebs solution (577.50 g; corresponds to 260 mg per each 450 mg net-weight capsule content), (d) magnesium stearate (10.00 g; 1% w/w).
- Each capsule contains: 20 mg of sulpiride ( 1 ) and 260 mg of homeopathic modified Krebs solution (10 mg of each of its 26 components).
- Literature data show that sulpiride exhibits similar range of efficacy at dosages of at least 150 mg/day. In contrast, our results prove that this dosage can be decreased to only 20 mg/day with preserved high antidepressant activity.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Biomedical Technology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Hematology (AREA)
- Alternative & Traditional Medicine (AREA)
- Dermatology (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Anesthesiology (AREA)
- Pain & Pain Management (AREA)
- Psychiatry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Cosmetics (AREA)
Claims (15)
- Une formulation thérapeutique, comprenant:(ii) une composition homéopathique comprenant l'acide citrique (D8), l'acide cis-aconitique (D8), l'acide α-cétoglutarique (D8), l'acide succinique (D10), l'acide fumarique (D8), l'acide DL-malique (D8), le diéthylomaloacétate de sodium (D6), le pyruvate de sodium (D8), l'oxalosuccinate de baryum (D10), le coenzyme A (D8), le nicotinamide-adénine-dinucléotide (NAD; D8), l'adénosine-triphosphate (ATP ; D10), l'acide ascorbique (D6), l'hydrochlorure de thiamine (D6), le phosphate sodique de riboflavine (D6), l'hydrochlorure de pyridoxine (D6), la nicotinamide (D6), la cystéine (D6), l'acide DL-α-lipoïque (D6), le soufre (D10), le foie du soufre (D10), l'orotate de magnésium (D6), l'oxalate de cérium (D8), le phosphate de manganèse (D6), l'extrait de fleur de Pulsatille des prés (Pulsatilla pratensis L.; D6), l'extrait de Betterave rouge (Beta vulgaris ssp. vulgaris var. conditiva, D4), tous dans la même quantité relative; et(iii) un ou plusieurs excipients qui contribuent à la forme posologique finale désirée: injection, comprimés, capsules, solution orale, et sirop.
- Une formulation selon la revendication 1, caractérisée en ce que l'excipient est sélectionné parmi un ou plusieurs groupes consistant des diluants, des sels isotoniques, des émulsifiants, des épaississants, des agents de charge, des liants, des désintégrants, des lubrifiants, des conservateurs, des antioxydants, et des agents stabilisants.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est diluant sélectionné parmi le groupe consistant de l'eau purifiée, l'éthanol, le glycérol, le 1,2-propylène-glycol, le 1,3-propylène-glycol, le butane-1,3-diol, les polyéthylène-glycols liquides, les polyglycérols liquides, le sorbitol aqueux, le diméthylsulfoxide, le miel, ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est sel isotonique sélectionné parmi le groupe consistant du chlorure de sodium (NaCl), le chlorure de potassium (KCl), le chlorure de magnésium (MgCl2) et ses hydrates, le chlorure de calcium (CaCl2) et ses hydrates, ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est émulsifiant sélectionné parmi le groupe consistant du laurylsulfate de sodium, le lauryl-éthylène-glycol-sulfate de sodium, le lauryl-diéthylène-glycol-sulfate de sodium, le laurylsulfate de potassium, le lauryl-éthylène-glycol-sulfate de potassium, le lauryl-diéthylène-glycol-sulfate de potassium; le cocoamphodipropionate de sodium ou potassium; le cocoamphodiacétate de disodium ou dipotassium; le polyomyéthylène(10)-lauryléther, le polyoxyéthylène(23)-lauryléther, le polyomyéthylène(10)-stéaryléther; le polyomyéthylène(23)-stéaryléther; le polyoxyéthylène(10)-oleyléther; le polyomyéthylène(23)-oleyléther, autres éthoxylates des alcools gras supérieurs avec une valeur de HLB ≥10; le polyoxyéthylène(10)-laurate, le polyoxyéthylène(23)-laurate, le polyomyéthylène(10)-stéarate, le polyoxyéthylène(23)-stéarate, le polyoxyéthylène(10)-oléate, le polyoxyéthylène(23)-oléate, autres éthoxylates des acides gras supérieurs avec une valeur de HLB >10; dérivés polyglycériques des alcools gras supérieurs avec une valeur de HLB ≥10; dérivés polyglycériques des acides gras supérieurs avec une valeur de HLB ≥10; le monolaurate de polyoxyéthylène-sorbitane, le monostéarate de polyoxyéthylène-sorbitane, le monooléate de polyoxyéthylène-sorbitane, autres dérivés de sorbitane avec une valeur de HLB ≥10; ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est épaississant sélectionné parmi le groupe consistant de l'acide polyacrylique, ces copolymères, ou leurs sels sodiques ou potassiques; la méthylcellulose; la carboxyméthylcellulose sodique; la 2-hydroxyéthylcellulose; la 2-hydroxypropylcellulose; l'amidon; les amidons modifiés; les polyglycérols; les polyéthylèneglycols; la gélatine; la pectine; l'agar-agar; le carraghénane; la gomme arabique; l'acide alginique; l'alginate de sodium; ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est agent de charge sélectionné parmi le groupe consistant de la cellulose microcristalline, le lactose monohydraté, l'hydrogénophosphate de calcium, le sulfate de calcium dihydraté, le carbonate de calcium, l'acide silicique colloïdal, le sorbitol, l'inuline, l'amidon, les amidons modifiés, la dextrine, le glucose, le fructose, le carbonate de magnésium basique, le silicate de calcium, le saccharose, ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est liant sélectionné parmi le groupe consistant de la gélatine, le lactose monohydraté, le sorbitol, le saccharose, le xylitol, le maltitol, l'amidon, les amidons modifiés, la méthylcellulose, la 2-hydroxyéthylcellulose, la 2-hydroxypropylcellulose, la 2-hydroxypropylméthylcellulose, la carboxyméthylcellulose sodique, les polyéthylèneglycols, les polyglycérols, la polyvinylpyrrolidone, les copolymères de polyvinylpyrrolidone, le carraghénane, la maltodextrine, ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est désintégrant sélectionné parmi le groupe consistant de l'amidon, les amidons modifiés, le glycolate sodique d'amidon, la méthylcellulose, la carboxyméthylcellulose sodique, la 2-hydroxyéthylcellulose, la 2-hydroxypropylcellulose, la 2-hydroxypropylméthylcellulose, la polyvinylpyrrolidone, les copolymères de polyvinylpyrrolidone, ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est lubrifiant sélectionné parmi le groupe consistant du stéarate de magnésium, le stéarate de calcium, le stéarate de zinc, l'acide stéarique, le talc, le dioxyde de silicium, ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est conservateur sélectionné parmi le groupe consistant du méthyl-4-hydroxybenzoate, l'éthyl-4-hydroxybenzoate, le propyl-4-hydroxybenzoate, le butyl-4-hydroxybenzoate, l'acide sorbique, le sorbate de potassium, l'acide benzoïque, le benzoate de sodium, le 2-phénoxyéthanol, le 4-chloro-M-cresol, le thymol, l'eugénol, ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est antioxydant sélectionné parmi le groupe consistant du 2,6-di-tert-butyl-4-hydroxytoluène (BHT), le tert-butylhydroxyanisole (BHA), le tocophérol, l'acétate de tocophéryl, l'acide ascorbique, ou les mélanges desdites substances.
- Une formulation selon la revendication 2, caractérisée en ce que l'excipient est agent stabilisant sélectionné parmi le groupe consistant de l'éthylène-diamine-tétraacétate de disodium (Na2EDTA•2H2O), le N-(2-hydroxyéthyl)-éthylène-diamine-triacétate de disodium [Na2H(HEDTA)], le diéthylène-triamine-pentaacétate de disodium [Na2H3(DTPA)], le citrate de disodium [Na2C(OH)(COOH) (CH2COO)2], ou les mélanges desdites substances.
- Une formulation selon les revendications 1-2 avec l'excipient sélectionné selon une ou plusieurs revendications 3-13 où:(i) le sulpiride est présent dans la quantité de 0,1-25% masse/masse;(ii) une composition homéopathique dans la quantité de 10-75% masse/masse; et(iii) un ou plusieurs excipients qui contribuent à atteindre les 100% masse/masse de la formulation.
- Une formulation selon une quelconque des revendications précédentes pour l'usage comme agent thérapeutique pour le traitement du syndrome dépressif, de l'anxiété, et comme agent thérapeutique hypnotique.
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/HR2010/000015 WO2011148203A1 (fr) | 2010-05-28 | 2010-05-28 | Formulation à base de sulpiride et d'une composition homéopathique synergique destinée au traitement d'un syndrome dépressif |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2575807A1 EP2575807A1 (fr) | 2013-04-10 |
| EP2575807B1 true EP2575807B1 (fr) | 2014-02-19 |
Family
ID=42697602
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP10729933.1A Active EP2575807B1 (fr) | 2010-05-28 | 2010-05-28 | Formulation comprenant du sulpiride et une composition homeopathique synergistique pour le traitement de la depression |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP2575807B1 (fr) |
| HR (1) | HRP20140321T1 (fr) |
| WO (1) | WO2011148203A1 (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3544603A2 (fr) * | 2016-11-23 | 2019-10-02 | Bohne Askøy AS | Prévention et/ou traitement du syndrome de fatigue |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2516454A (en) | 2013-07-22 | 2015-01-28 | Epiplex Ltd | Reduction of epileptic seizures |
| EP3090738A1 (fr) * | 2015-05-04 | 2016-11-09 | Universidade de Santiago de Compostela | Riboflavine pour le traitement d'un accident ischémique et/ou d'autres maladies associés à l'excitotoxicité du glutamate |
| CN109481459B (zh) * | 2018-11-23 | 2021-07-20 | 济南康和医药科技有限公司 | 一种复方电解质葡萄糖注射液及其制备方法 |
| BR102019008505A2 (pt) * | 2019-04-26 | 2020-11-03 | Mônica Filomena Assis De Souza | Composição homeopática para tratamento de animais domésticos, silvestres, peixes, anfíbios e outros |
| CN119384278A (zh) * | 2022-06-10 | 2025-01-28 | 上海日馨医药科技股份有限公司 | 治疗精神类疾病的方法 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100493556C (zh) * | 2006-06-01 | 2009-06-03 | 张宝山 | 一种治疗抑郁情绪障碍性疾病的药物 |
-
2010
- 2010-05-28 WO PCT/HR2010/000015 patent/WO2011148203A1/fr not_active Ceased
- 2010-05-28 HR HRP20140321AT patent/HRP20140321T1/hr unknown
- 2010-05-28 EP EP10729933.1A patent/EP2575807B1/fr active Active
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3544603A2 (fr) * | 2016-11-23 | 2019-10-02 | Bohne Askøy AS | Prévention et/ou traitement du syndrome de fatigue |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2575807A1 (fr) | 2013-04-10 |
| WO2011148203A1 (fr) | 2011-12-01 |
| HRP20140321T1 (hr) | 2014-05-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| ES2646816T3 (es) | Composiciones de dosificaciones orales de liberación retardada que contienen CDDO-Me amorfo | |
| EP2575807B1 (fr) | Formulation comprenant du sulpiride et une composition homeopathique synergistique pour le traitement de la depression | |
| CZ282679B6 (cs) | Použití 2-amino-6-n-propylamino-4,5,6,7-tetrahydrobenzothiazolu pro výrobu farmaceutického prostředku | |
| WO2021201238A1 (fr) | Procédés d'administration d'une préparation injectable d'aripiprazole | |
| CN104758269A (zh) | 一种乙酰半胱氨酸泡腾片 | |
| CN104013638B (zh) | 当药黄素及其衍生物的用途 | |
| WO2014010656A1 (fr) | Agent d'accélération de la baisse de concentration d'alcool supérieur dans le sang | |
| KR100709528B1 (ko) | 혈당콘트롤용 의약조성물 | |
| JP5961034B2 (ja) | 安定化方法 | |
| KR101697773B1 (ko) | 독소필린을 포함하는 변형 방출 조성물 | |
| KR102095536B1 (ko) | 천연물 추출물의 용출률 및 붕해성이 개선된 경구용 제제 | |
| US20110117070A1 (en) | Compositions and methods for treating headache | |
| CN104645334B (zh) | N‑乙酰半胱氨酸活性炭组合物及其制备方法和应用 | |
| WO2010087358A1 (fr) | Nouvelle composition | |
| JP4463206B2 (ja) | グリチルリチン高濃度製剤 | |
| KR20250013306A (ko) | 페오놀 및 아포시닌을 포함하는 액체 제형 | |
| JP7114227B2 (ja) | 清肺湯エキス末を含有する錠剤 | |
| CN101766605B (zh) | 包含普拉克索的可在口中分散的药物组合物 | |
| KR100709531B1 (ko) | 당뇨병성 합병증의 예방 또는 진전저지용 의약조성물 | |
| CN114053282B (zh) | 委陵菜酸和山莨菪碱胶囊在制备治疗糖尿病药物中的应用 | |
| KR102375872B1 (ko) | 옥수수불검화 정량 추출물 및 후박 추출물을 유효성분으로 포함하며 iLet(innovative Low excipient tablet) 기술을 이용한 정제사이즈 축소를 통해 복용 편의성이 향상된 치주질환 예방 또는 치료용 약학적 조성물 | |
| WO2025085034A1 (fr) | Formulation de clobazam sous forme de capsule | |
| CN1562345A (zh) | 包含脑代谢激活剂和自由基清除剂的联合用药物 | |
| US20240091173A1 (en) | Common cold symptom relieving agent | |
| CN112716945A (zh) | 药物组合物及其应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20121025 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA RS |
|
| RAX | Requested extension states of the european patent have changed |
Extension state: RS Payment date: 20121025 Extension state: BA Payment date: 20121025 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20131002 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA RS |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 652678 Country of ref document: AT Kind code of ref document: T Effective date: 20140315 |
|
| REG | Reference to a national code |
Ref country code: HR Ref legal event code: TUEP Ref document number: P20140321 Country of ref document: HR Ref country code: DE Ref legal event code: R096 Ref document number: 602010013636 Country of ref document: DE Effective date: 20140403 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: HR Ref legal event code: T1PR Ref document number: P20140321 Country of ref document: HR |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20140219 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 652678 Country of ref document: AT Kind code of ref document: T Effective date: 20140219 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140619 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140519 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: RIEDERER HASLER AND PARTNER PATENTANWAELTE AG, LI |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140619 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602010013636 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140528 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| 26N | No opposition filed |
Effective date: 20141120 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20140528 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140531 Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140531 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602010013636 Country of ref document: DE Effective date: 20141120 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20150130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140528 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140602 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140528 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140520 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20100528 Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| REG | Reference to a national code |
Ref country code: HR Ref legal event code: ODRP Ref document number: P20140321 Country of ref document: HR Payment date: 20180517 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: HR Payment date: 20180517 Year of fee payment: 9 Ref country code: DE Payment date: 20180518 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140219 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602010013636 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: HR Ref legal event code: PBON Ref document number: P20140321 Country of ref document: HR Effective date: 20190528 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20191203 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20190528 |