JP2004123735A - 光学活性(s)−ヒダントイン誘導体 - Google Patents
光学活性(s)−ヒダントイン誘導体 Download PDFInfo
- Publication number
- JP2004123735A JP2004123735A JP2003316597A JP2003316597A JP2004123735A JP 2004123735 A JP2004123735 A JP 2004123735A JP 2003316597 A JP2003316597 A JP 2003316597A JP 2003316597 A JP2003316597 A JP 2003316597A JP 2004123735 A JP2004123735 A JP 2004123735A
- Authority
- JP
- Japan
- Prior art keywords
- hydroxy
- methylhydantoin
- renal failure
- optically active
- agent
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- UPSFNXFLBMJUQW-VKHMYHEASA-N (5s)-5-hydroxy-1-methylimidazolidine-2,4-dione Chemical compound CN1[C@@H](O)C(=O)NC1=O UPSFNXFLBMJUQW-VKHMYHEASA-N 0.000 claims abstract description 19
- 229940124423 agent for renal failure Drugs 0.000 claims description 8
- 150000003839 salts Chemical class 0.000 claims description 8
- 239000004480 active ingredient Substances 0.000 claims description 4
- UPSFNXFLBMJUQW-UHFFFAOYSA-N 5-hydroxy-1-methylimidazolidine-2,4-dione Chemical compound CN1C(O)C(=O)NC1=O UPSFNXFLBMJUQW-UHFFFAOYSA-N 0.000 abstract description 41
- 239000003814 drug Substances 0.000 abstract description 15
- 208000001647 Renal Insufficiency Diseases 0.000 abstract description 13
- 201000006370 kidney failure Diseases 0.000 abstract description 13
- 239000002207 metabolite Substances 0.000 abstract description 13
- 230000003287 optical effect Effects 0.000 abstract description 13
- 239000000126 substance Substances 0.000 abstract description 8
- 239000013543 active substance Substances 0.000 abstract description 6
- 208000020832 chronic kidney disease Diseases 0.000 abstract description 4
- 208000022831 chronic renal failure syndrome Diseases 0.000 abstract description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract description 2
- 231100000086 high toxicity Toxicity 0.000 abstract 1
- 230000001473 noxious effect Effects 0.000 abstract 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 36
- WJRBRSLFGCUECM-UHFFFAOYSA-N hydantoin Chemical group O=C1CNC(=O)N1 WJRBRSLFGCUECM-UHFFFAOYSA-N 0.000 description 22
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 21
- 239000013078 crystal Substances 0.000 description 19
- 229910052739 hydrogen Inorganic materials 0.000 description 16
- -1 sodium and potassium Chemical class 0.000 description 16
- 150000001875 compounds Chemical class 0.000 description 14
- 238000000034 method Methods 0.000 description 14
- 229940091173 hydantoin Drugs 0.000 description 13
- UPSFNXFLBMJUQW-GSVOUGTGSA-N (5r)-5-hydroxy-1-methylimidazolidine-2,4-dione Chemical compound CN1[C@H](O)C(=O)NC1=O UPSFNXFLBMJUQW-GSVOUGTGSA-N 0.000 description 12
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- DDRJAANPRJIHGJ-UHFFFAOYSA-N creatinine Chemical compound CN1CC(=O)NC1=N DDRJAANPRJIHGJ-UHFFFAOYSA-N 0.000 description 12
- 239000000203 mixture Substances 0.000 description 12
- 238000005160 1H NMR spectroscopy Methods 0.000 description 11
- 238000002844 melting Methods 0.000 description 10
- 230000008018 melting Effects 0.000 description 10
- 239000003795 chemical substances by application Substances 0.000 description 9
- 238000000921 elemental analysis Methods 0.000 description 9
- WAPNOHKVXSQRPX-SSDOTTSWSA-N (R)-1-phenylethanol Chemical compound C[C@@H](O)C1=CC=CC=C1 WAPNOHKVXSQRPX-SSDOTTSWSA-N 0.000 description 7
- WAPNOHKVXSQRPX-ZETCQYMHSA-N (S)-1-phenylethanol Chemical compound C[C@H](O)C1=CC=CC=C1 WAPNOHKVXSQRPX-ZETCQYMHSA-N 0.000 description 7
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 7
- RHYBFKMFHLPQPH-UHFFFAOYSA-N N-methylhydantoin Chemical compound CN1CC(=O)NC1=O RHYBFKMFHLPQPH-UHFFFAOYSA-N 0.000 description 7
- 229940079593 drug Drugs 0.000 description 7
- 238000004128 high performance liquid chromatography Methods 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- SYTBZMRGLBWNTM-SNVBAGLBSA-N (R)-flurbiprofen Chemical compound FC1=CC([C@H](C(O)=O)C)=CC=C1C1=CC=CC=C1 SYTBZMRGLBWNTM-SNVBAGLBSA-N 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 6
- 229940109239 creatinine Drugs 0.000 description 6
- 230000000704 physical effect Effects 0.000 description 6
- 230000002829 reductive effect Effects 0.000 description 6
- 239000000741 silica gel Substances 0.000 description 6
- 229910002027 silica gel Inorganic materials 0.000 description 6
- 229930024421 Adenine Natural products 0.000 description 5
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 5
- 241000700159 Rattus Species 0.000 description 5
- 229960000643 adenine Drugs 0.000 description 5
- 239000008280 blood Substances 0.000 description 5
- 210000004369 blood Anatomy 0.000 description 5
- 238000001914 filtration Methods 0.000 description 5
- 150000007975 iminium salts Chemical class 0.000 description 5
- 238000002360 preparation method Methods 0.000 description 5
- 231100000331 toxic Toxicity 0.000 description 5
- 230000002588 toxic effect Effects 0.000 description 5
- 125000005976 1-phenylethyloxy group Chemical group 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical class CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 4
- 238000003820 Medium-pressure liquid chromatography Methods 0.000 description 4
- 238000010521 absorption reaction Methods 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- 238000003818 flash chromatography Methods 0.000 description 4
- 239000002808 molecular sieve Substances 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 3
- 229920002261 Corn starch Polymers 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000008120 corn starch Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 3
- 229940090044 injection Drugs 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 230000003907 kidney function Effects 0.000 description 3
- 239000003921 oil Substances 0.000 description 3
- 235000019198 oils Nutrition 0.000 description 3
- 230000000144 pharmacologic effect Effects 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 239000003579 shift reagent Substances 0.000 description 3
- 238000010898 silica gel chromatography Methods 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 125000001424 substituent group Chemical group 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- 229920000856 Amylose Polymers 0.000 description 2
- 230000005526 G1 to G0 transition Effects 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 238000005481 NMR spectroscopy Methods 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- GRFUMPFWJKGLQC-ZETCQYMHSA-N [(1s)-1-phenylethyl]carbamic acid Chemical group OC(=O)N[C@@H](C)C1=CC=CC=C1 GRFUMPFWJKGLQC-ZETCQYMHSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 150000001340 alkali metals Chemical class 0.000 description 2
- 239000003125 aqueous solvent Substances 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 238000010531 catalytic reduction reaction Methods 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 235000014113 dietary fatty acids Nutrition 0.000 description 2
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- YXJPWWGLCOPUGI-NCJHBDPTSA-N europium;(2z)-4,7,7-trimethyl-2-(2,2,2-trifluoro-1-hydroxyethylidene)bicyclo[2.2.1]heptan-3-one Chemical compound [Eu].C1CC2(C)C(=O)\C(=C(/O)C(F)(F)F)C1C2(C)C.C1CC2(C)C(=O)\C(=C(/O)C(F)(F)F)C1C2(C)C.C1CC2(C)C(=O)\C(=C(/O)C(F)(F)F)C1C2(C)C YXJPWWGLCOPUGI-NCJHBDPTSA-N 0.000 description 2
- 239000000194 fatty acid Substances 0.000 description 2
- 229930195729 fatty acid Natural products 0.000 description 2
- 229910000042 hydrogen bromide Inorganic materials 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 231100000636 lethal dose Toxicity 0.000 description 2
- 231100000053 low toxicity Toxicity 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 150000002739 metals Chemical class 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 229920001592 potato starch Polymers 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 2
- 230000006340 racemization Effects 0.000 description 2
- 239000002516 radical scavenger Substances 0.000 description 2
- 230000008085 renal dysfunction Effects 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- 238000004809 thin layer chromatography Methods 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- 231100000820 toxicity test Toxicity 0.000 description 2
- FANCTJAFZSYTIS-IQUVVAJASA-N (1r,3s,5z)-5-[(2e)-2-[(1r,3as,7ar)-7a-methyl-1-[(2r)-4-(phenylsulfonimidoyl)butan-2-yl]-2,3,3a,5,6,7-hexahydro-1h-inden-4-ylidene]ethylidene]-4-methylidenecyclohexane-1,3-diol Chemical compound C([C@@H](C)[C@@H]1[C@]2(CCCC(/[C@@H]2CC1)=C\C=C\1C([C@@H](O)C[C@H](O)C/1)=C)C)CS(=N)(=O)C1=CC=CC=C1 FANCTJAFZSYTIS-IQUVVAJASA-N 0.000 description 1
- JXGVXCZADZNAMJ-NSHDSACASA-N (2s)-1-phenylmethoxycarbonylpyrrolidine-2-carboxylic acid Chemical class OC(=O)[C@@H]1CCCN1C(=O)OCC1=CC=CC=C1 JXGVXCZADZNAMJ-NSHDSACASA-N 0.000 description 1
- PGNXGEOSTMOHNC-UHFFFAOYSA-N 5-hydroxy-3-methylimidazolidine-2,4-dione Chemical compound CN1C(=O)NC(O)C1=O PGNXGEOSTMOHNC-UHFFFAOYSA-N 0.000 description 1
- GDJCNIGBOGZFCS-UHFFFAOYSA-N 5-imino-1-methylimidazolidine-2,4-dione Chemical class CN1C(=O)NC(=O)C1=N GDJCNIGBOGZFCS-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 208000009304 Acute Kidney Injury Diseases 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 1
- 229940123457 Free radical scavenger Drugs 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 206010060891 General symptom Diseases 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 208000003623 Hypoalbuminemia Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 229940123973 Oxygen scavenger Drugs 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 208000033626 Renal failure acute Diseases 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 206010047115 Vasculitis Diseases 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 238000000862 absorption spectrum Methods 0.000 description 1
- YLEIFZAVNWDOBM-ZTNXSLBXSA-N ac1l9hc7 Chemical compound C([C@H]12)C[C@@H](C([C@@H](O)CC3)(C)C)[C@@]43C[C@@]14CC[C@@]1(C)[C@@]2(C)C[C@@H]2O[C@]3(O)[C@H](O)C(C)(C)O[C@@H]3[C@@H](C)[C@H]12 YLEIFZAVNWDOBM-ZTNXSLBXSA-N 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 239000003377 acid catalyst Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 231100000215 acute (single dose) toxicity testing Toxicity 0.000 description 1
- 201000011040 acute kidney failure Diseases 0.000 description 1
- 208000012998 acute renal failure Diseases 0.000 description 1
- 238000011047 acute toxicity test Methods 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- SRVFFFJZQVENJC-IHRRRGAJSA-N aloxistatin Chemical compound CCOC(=O)[C@H]1O[C@@H]1C(=O)N[C@@H](CC(C)C)C(=O)NCCC(C)C SRVFFFJZQVENJC-IHRRRGAJSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000003472 antidiabetic agent Substances 0.000 description 1
- 239000003524 antilipemic agent Substances 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 239000004067 bulking agent Substances 0.000 description 1
- OSVHLUXLWQLPIY-KBAYOESNSA-N butyl 2-[(6aR,9R,10aR)-1-hydroxy-9-(hydroxymethyl)-6,6-dimethyl-6a,7,8,9,10,10a-hexahydrobenzo[c]chromen-3-yl]-2-methylpropanoate Chemical compound C(CCC)OC(C(C)(C)C1=CC(=C2[C@H]3[C@H](C(OC2=C1)(C)C)CC[C@H](C3)CO)O)=O OSVHLUXLWQLPIY-KBAYOESNSA-N 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- FNAQSUUGMSOBHW-UHFFFAOYSA-H calcium citrate Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O.[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O FNAQSUUGMSOBHW-UHFFFAOYSA-H 0.000 description 1
- 239000001354 calcium citrate Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 229940084030 carboxymethylcellulose calcium Drugs 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 231100000517 death Toxicity 0.000 description 1
- CSCPPACGZOOCGX-WFGJKAKNSA-N deuterated acetone Substances [2H]C([2H])([2H])C(=O)C([2H])([2H])[2H] CSCPPACGZOOCGX-WFGJKAKNSA-N 0.000 description 1
- 238000000502 dialysis Methods 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 239000002934 diuretic Substances 0.000 description 1
- 230000001882 diuretic effect Effects 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 125000000219 ethylidene group Chemical group [H]C(=[*])C([H])([H])[H] 0.000 description 1
- LNBHUCHAFZUEGJ-UHFFFAOYSA-N europium(3+) Chemical compound [Eu+3] LNBHUCHAFZUEGJ-UHFFFAOYSA-N 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 239000003889 eye drop Substances 0.000 description 1
- 229940012356 eye drops Drugs 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 229940093181 glucose injection Drugs 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 229940126904 hypoglycaemic agent Drugs 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- UKVIEHSSVKSQBA-UHFFFAOYSA-N methane;palladium Chemical compound C.[Pd] UKVIEHSSVKSQBA-UHFFFAOYSA-N 0.000 description 1
- 125000001434 methanylylidene group Chemical group [H]C#[*] 0.000 description 1
- MJJCZWPYKMGQHU-UHFFFAOYSA-N methylparabanic acid Chemical compound CN1C(=O)NC(=O)C1=O MJJCZWPYKMGQHU-UHFFFAOYSA-N 0.000 description 1
- 210000000885 nephron Anatomy 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 239000008024 pharmaceutical diluent Substances 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000005648 plant growth regulator Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 239000012929 tonicity agent Substances 0.000 description 1
- 235000013337 tricalcium citrate Nutrition 0.000 description 1
- 239000002441 uremic toxin Substances 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Images
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
【解決手段】本発明(S)−(−)−5−ヒドロキシ−1−メチルヒダントインは、薬物の体内動態に優れ安全性の高い光学活性体である。本発明S体化合物はその対掌体であるR体と比べて、毒性の高い代謝物へはほとんど変化しないため、特に腎機能障害によって有害物質が排出されずに体内に蓄積される慢性腎不全の患者に投与する腎不全用剤として使用する場合に非常に好ましいものである。
Description
さらに疾患の種類や患者に応じて、その治療に最適な上記以外の剤型、シロップ剤、坐剤、吸入剤、エアゾール剤、点眼剤、外用剤(軟膏剤、ゲル剤、貼付剤など)等に製剤化することができる。
(1)(S)−(−)−5−ヒドロキシ−1−メチルヒダントインおよびその薬学的に許容される塩。
(2)上記(1)記載の化合物またはその薬学的に許容される塩を有効成分として含有する腎不全用剤。
(3)腎不全の治療又は予防剤である上記(2)記載の腎不全用剤。
(4)急性腎不全の治療又は予防剤である上記(3)記載の腎不全用剤。
(5)慢性腎不全の治療又は予防剤である上記(3)記載の腎不全用剤。
(6)腎不全進展抑制剤である上記(2)記載の腎不全用剤。
(7)経口剤である上記(2)記載の腎不全用剤。
(8)錠剤である上記(2)記載の腎不全用剤。
以下に実施例を挙げて本発明を更に具体的に説明するが、本発明はこれらによって何ら限定されるものではない。
融点はMP−21型融点測定器(ヤマト)で測定し、補正はしていない。旋光度(〔α〕D)は10cmのセルを用い、日本分光のDIP−140型旋光計で測定した。1H−NMRは、Bruker社のAM−400Wb型核磁気共鳴装置で測定し、テトラメチルシラン(TMS)を内部標準に用いた。1H−NMRによるエナンチオマーの分析は5−ヒドロキシ−1−メチルヒダントイン(5mg)を含む重アセトン(6mL)溶液にシフト試薬[Eu(tfc)3、30mg]を加えることによって行った。赤外吸収スペクトル(IR)はFT−200型フーリエ変換赤外分光光度計(堀場)を使用し、KBr法で測定した。元素分折はCHN Corder MT−5型元素分析装置(ヤナコ)で測定した。
Dean−Starkの水分離装置を付けたフラスコ中で(±)−5−ヒドロキシ−1−メチルヒダントイン(106.8g,0.82mol)、(S)−1−フェニルエタノール(25.0g,0.21mol)及びp−トルエンスルホン酸(1.0g)の混合物をベンゼン(2.5L)に懸濁し、水分が完全に出なくなるまで48時間加熱還流した。不溶物を濾去した後、ベンゼン溶液を希炭酸水素ナトリウム水溶液で洗浄した。無水硫酸ナトリウム上で乾燥した後、減圧濃縮すると二つのジアステレオマー(5R,1’S)−1−メチル−5−(1’−フェニルエトキシ)ヒダントイン(3a)及び(5S,1’S)−1−メチル−5−(1’−フェニルエトキシ)ヒダントイン(3b)の2.5:1混合物(40.0g)が無色の油状物として得られた。この混合物をシリカゲル(6L)を用い、ベンゼン/酢酸エチル(1:1)を溶出液としたカラムクロマトグラフィーで分離するとジアステレオマー3a(17.0g)及び3b(4.0g)の粗結晶が得られた。各粗結晶を酢酸エチル/石油エーテルから再結晶して、ジアステレオマー3a(15.5g,32%)及び3b(2.5g,5%)を白色結晶として得た。
(1)1−メチルヒダントイン(114g,1.0mol)を1,2−ジクロルエタン(600mL)に懸濁し、油浴上95乃至100℃に加熱した。攪拌しながら臭素(57mL,1.1mol)を6時間かけて滴下し、臭素の色が消えて(約1時間)からさらに1時間加熱攪拌した。減圧濃縮して得られる固形物を1,2−ジクロルエタンから再結晶し、第3結晶まで合わせてイミニウム塩(1−メチルヒダントイン−5−エニウム ブロミド;166.8g,86%)が白色結晶として得られた。
融点:73−74℃、〔α〕24 D:+159.9°(c1,CHCl3)、Rf:0.48、1H−NMR(CDCl3):δ 1.54(d,3H,J=6.5Hz,CH3),2.74(s,3H,CH3−N),4.68(s,1H,N−CH−C=O),5.10(q,1H,J=6.5Hz,CH7),7.31−7.41(m,5H,arom.H),8.68(s,1H,NH)、IR:νmax KBr(cm-1)3250,2966,1794,1778,1724,1705,1446,1350,1315,1111,1097,1066,756,694,559、元素分析(C12H14N2O3):計算値;C,61.53%、H,6.02%、N,11.96%;実測値;C,61.42%、H,6.15%、N,11.86%
融点:106−107.5℃、〔α〕24 D:+148.9°(c1,CHCl3)、Rf:0.42、1H−NMR(CDCl3):δ 1.55(d,3H,J=6.5Hz,CH3),2.68(s,3H,CH3−N),4.78(q,1H,J=6.5Hz,―CH),5.04(s,1H,N−CH−C=O),7.28−7.38(m,5H,arom.H),8.07(s,1H,NH)、IR:νmax KBr(cm-1)3265,2977,1797,1776,1728,1705,1444,1321,1306,1095,1036,762,698,530、元素分析(C12H14N2O3):計算値;C,61.53%、H,6.02%、N,11.96%;実測値;C,61.34%、H,6.09%、N,11.90%
融点:73−74℃、〔α〕24 D:−155.2°(c1,CHCl3)、Rf:0.48、1H−NMR(CDCl3):δ 1.54(d,3H,J=6.5Hz,CH3),2.74(s,3H,CH3−N),4.68(s,1H,N−CH−C=O),5.10(q,1H,J=6.5Hz,CH),7.31−7.41(m,5H,arom.H),8.15(s,1H,NH)、IR:νmax KBr(cm-1)3251,2966,1794,1778,1724,1705,1446,1350,1315,1111,1097,1066,756,694,559、元素分析(C12H14N2O3):計算値;C,61.53%、H,6.02%、N,11.96%;実測値;C,61.36%、H,6.12%、N,11.94%
融点:101−102℃、〔α〕24 D:−148.7°(c1,CHCl3)、Rf:0.42、1H−NMR(CDCl3):δ 1.55(d,3H,J=6.5Hz,CH3),2.67(s,3H,CH3−N),4.77(q,1H,J=6.5Hz,CH),5.04(s,1H,N−CH−C=O),7.27−7.37(m,5H,arom.H),8.63(s,1H,NH)、IR:νmax KBr(cm-1)3261,2977,1797,1776,1730,1705,1444,1321,1306,1095,1036,762,700,530、元素分析(C12H14N2O3):計算値;C,61.53%、H,6.02%、N,11.96%;実測値;C,61.45%、H,6.05%、N,12.00%
融点:146.5−149.5℃、〔α〕24 D:+8.9°(c1,MeOH)
1H−NMR(DMSO−d6):δ 2.74(s,3H,CH3),4.96(d,1H,J=8.8Hz,CH),6.86(d,1H,J=8.8Hz,OH),10.74(s,1H,NH)、IR:νmax KBr(cm-1)3396,3174,3082,2750,1790,1741,1452,1109、元素分析(C4H6N2O3):計算値;C,36.93%、H,4.65%、N,21.53%;実測値;C,37.27%、H,4.72%、N,21.43%
融点:147.0−149.5℃、〔α〕24 D:−9.0°(c1,MeOH)、1H−NMR(DMSO−d6):δ 2.74(s,3H,CH3),4.96(d,1H,J=8.8Hz,CH),6.86(d,1H,J=8.8Hz,OH),10.74(s,1H,NH)、IR:νmax KBr(cm-1)3396,3182,3076,2750,1738,1454,1109、元素分析(C4H6N2O3):計算値;C,36.93%、H,4.65%、N,21.53%;実測値;C,36.95%、H,4.63%、N,21.37%
融点:147.0−149.0℃、〔α〕24 D:―8.9°(c1,MeOH)、1H−NMR(DMSO−d6):δ 2.74(s,3H,CH3),4.96(d,1H,J=8.8Hz,CH),6.86(d,1H,J=8.8Hz,OH),10.75(s,1H,NH)、IR:νmax KBr(cm-1)3402,3182,3076,2750,1734,1450,1107、元素分析(C4H6N2O3):計算値;C,36.93%、H,4.65%、N,21.53%;実測値;C,36.99%、H,4.55%、N,21.59%
以上得られた5−ヒドロキシ−1−メチルヒダントイン光学活性体の光学純度は以下の2種の方法で検定した。
(1)シフト試薬を用いるプロトン核磁気共鳴(1H−NMR)法
(±)−5−ヒドロキシ−1−メチルヒダントインをシフト試薬トリス[3−(2,2,2−トリフルオロ−1−ヒドロキシエチリデン)−d−カンホラト]ユーロピウム(III)[Eu(tfc)3](1:6重量比)と共に重アセトン中で1H−NMRスペクトルを測定すると、5位のメチン基のシグナル(ダブレット)は互いに2本づつに分離した。また、5位の水酸基のシグナル(ダブレット)は一部重なり、見かけ上トリプレットを示した。しかし、同じ条件下での(S)−(−)−及び(R)−(+)−5−ヒドロキシ−1−メチルヒダントインの1H−NMRスペクトルは対応する位置にタブレットを示すのみであった。このことは上記の(S)−(−)−及び(R)−(+)−5−ヒドロキシ−1−メチルヒダントインがラセミ化することなく単離されたことを示している。また、これらのシグナル強度比から(S)−(−)−及び(R)−(+)−5−ヒドロキシ−1−メチルヒダントインの光学純度(enantiomeric excess,e.e.)は99.5%e.e.以上と結論した。
(S)−1−フェニルエチルカルバメートを化学結合させたアミロースでシリカゲルを被覆した固定相を充填したキラルカラム(キラルパックAS、ダイセル化学工業)を用いて(±)−5−ヒドロキシ−1−メチルヒダントインの直接光学分割に成功した。即ち、キラルパックASカラムを用い、エタノールを添加したヘキサンを移動相として(±)−5−ヒドロキシ−1−メチルヒダントインを分析した結果、エナンチオマーの良好な分離が認められた。さらに、0.1%の酢酸を添加しカラム温度を室温から0℃に下げることによりほぼ完全な分離が達成できた。
(1)薬理作用
8週齢のウイスター系雄性ラットにアデニンを経口投与して腎不全モデル動物を作製した(腎と透析、1991年、臨時増刊号、440〜445頁及びNephron、44巻、1986年、230〜234頁等を参照)。アデニン(200mg/kg)を連日投与することによって、腎機能の指標となる血中クレアチニン値は徐々に上昇し、投与前の0.44±0.02mg/dLから約3週間後には4.12±0.53mg/dLに上昇した。また、クレアチニン・クリアランス(mL/kg/hr)の値は、315.3±13.8から25.7±4.4に低下し、腎機能は10分の1程度に低下した。
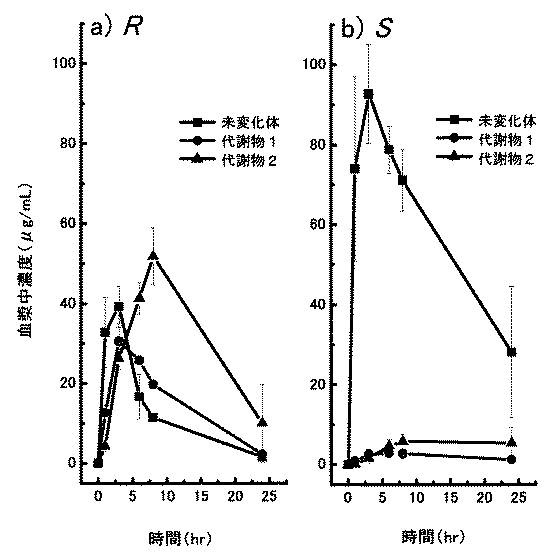
上記の腎不全モデルラット(アデニン投与23日目)に、(S)−(−)−及び(R)−(+)−5−ヒドロキシ−1−メチルヒダントインを各々100mg/kgで単回経口投与した後、血漿中の未変化体及び代謝物(代謝物1:1−メチルイミダゾリジントリオン及び代謝物2:5−メチルオキサルル酸)の濃度推移を調べた。その結果を図1に示すが、腎不全モデルラットにおいては、本発明S体はその対掌体であるR体と比べてほとんど代謝物に変換されないことが明らかになった。
SD系ラット(6週齢)に(±)−5−ヒドロキシ−1−メチルヒダントインを静脈内に単回した急性毒性試験において、投与量2000mg/kgでも死亡例はなく、本未変化体の致死量は、2000mg/kg以上と推定された。しかし、同じ毒性試験において、代謝物1の概略の致死量は150乃至300mg/kgであった。このように代謝物1の毒性は比較的高いと考えられ、図1に示した光学異性体の血中動態の結果より、S体はR体よりも代謝物1に代謝されにくいため、S体の方がR体より安全性が高いことが示唆された。また、各光学活性体をマウスに単回経口投与して一般症状等の発現の最小用量を調べた結果、R体はS体の5分の1の用量にて症状発現が認められ、S体の方が副作用的にも優れていることが認められた。
Claims (2)
- (S)−(−)−5−ヒドロキシ−1−メチルイミダゾリジン−2,4−ジオンおよびその薬学的に許容される塩。
- (S)−(−)−5−ヒドロキシ−1−メチルイミダゾリジン−2,4−ジオン又はその薬学的に許容される塩を有効成分として含有する腎不全用剤。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003316597A JP2004123735A (ja) | 2002-09-11 | 2003-09-09 | 光学活性(s)−ヒダントイン誘導体 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002265958 | 2002-09-11 | ||
| JP2003316597A JP2004123735A (ja) | 2002-09-11 | 2003-09-09 | 光学活性(s)−ヒダントイン誘導体 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2004123735A true JP2004123735A (ja) | 2004-04-22 |
Family
ID=32301581
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003316597A Pending JP2004123735A (ja) | 2002-09-11 | 2003-09-09 | 光学活性(s)−ヒダントイン誘導体 |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP2004123735A (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7569701B2 (en) | 2002-04-10 | 2009-08-04 | Nippon Zoki Pharmaceutical Co., Ltd. | Crystal form of 5-hydroxy-1-methylimidazolidin-2,4-dione |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0372463A (ja) * | 1989-08-09 | 1991-03-27 | Nippon Zoki Pharmaceut Co Ltd | 腎機能改善剤 |
| JPH04273835A (ja) * | 1991-02-28 | 1992-09-30 | Tokyo Kasei Kogyo Kk | 光学活性アルコールの製造法 |
| JP2001163806A (ja) * | 1999-09-29 | 2001-06-19 | Daicel Chem Ind Ltd | 光学異性体分離剤 |
| JP2002241316A (ja) * | 2001-02-19 | 2002-08-28 | Sumitomo Chem Co Ltd | 菊酸アルキルエステルの光学分割法 |
-
2003
- 2003-09-09 JP JP2003316597A patent/JP2004123735A/ja active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0372463A (ja) * | 1989-08-09 | 1991-03-27 | Nippon Zoki Pharmaceut Co Ltd | 腎機能改善剤 |
| JPH04273835A (ja) * | 1991-02-28 | 1992-09-30 | Tokyo Kasei Kogyo Kk | 光学活性アルコールの製造法 |
| JP2001163806A (ja) * | 1999-09-29 | 2001-06-19 | Daicel Chem Ind Ltd | 光学異性体分離剤 |
| JP2002241316A (ja) * | 2001-02-19 | 2002-08-28 | Sumitomo Chem Co Ltd | 菊酸アルキルエステルの光学分割法 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7569701B2 (en) | 2002-04-10 | 2009-08-04 | Nippon Zoki Pharmaceutical Co., Ltd. | Crystal form of 5-hydroxy-1-methylimidazolidin-2,4-dione |
| US7858806B2 (en) | 2002-04-10 | 2010-12-28 | Nippon Zoki Pharmaceutical Co., Ltd. | Crystal form of 5-hydroxy-1-methylhydantoin |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2332416C2 (ru) | Производные имидазола, способ их получения, фармацевтическая композиция на их основе, их применение в качестве периферически-селективных ингибиторов дофамин-бета-гидролазы и промежуточные соединения | |
| US10308604B2 (en) | Method for producing pyrrole derivative, and intermediate thereof | |
| FI102752B (fi) | Menetelmä verenpainetta alentavien 4-alkyyli-imidatsolijohdannaisten v almistamiseksi | |
| EP0040591A1 (fr) | Dérivés de pyridoxine, leur procédé de préparation ainsi que leur utilisation en thérapeutique | |
| FR2853650A1 (fr) | Procede de dedoublement d'amines utiles pour le traitement de desordres associes au syndrome d'insulino-resistance | |
| EP0167459B1 (fr) | 2-amino oxazolines et leur procédé d'obtention | |
| US4134991A (en) | Derivatives of 2-(3-phenyl-2-aminopropionyloxy)-acetic acid | |
| JP2004123735A (ja) | 光学活性(s)−ヒダントイン誘導体 | |
| CA2128119C (fr) | Nouveaux derives de benzospiroalcene, leur procede de preparation et les compositions pharmaceutiques qui les contiennent | |
| JP2004123736A (ja) | 光学活性(r)−ヒダントイン誘導体 | |
| US20060241163A1 (en) | Optically active (S)-hydantoin derivative | |
| EP0559571A1 (fr) | Nouveaux dérivés de thiazolidine-2,4-dione, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent | |
| EP0143016B1 (fr) | Dérivés de 4-(3-alkynyloxy-2-hydroxy-propyl)-piperazin-1-yl-N-phényl acétamide, leur préparation et leur application en thérapeutique | |
| US20060241162A1 (en) | Optically active (R)-hydantoin derivative | |
| CA1227197A (fr) | Derives de l'amino-5 dithiole-1,2 one-3 leur preparation et les compositions medicinales qui les contiennent | |
| CN1771240A (zh) | 3-苯基呋喃-2-酮衍生物作为环加氧酶的抑制剂 | |
| EP2486019A1 (fr) | Procede de synthese de la 2-thiohistidine et analogues | |
| EP0723974A1 (fr) | Dérivés de mercaptoalcanoyldipeptides, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent | |
| RU2824994C1 (ru) | Способ получения дексмедетомидина и его фармацевтически приемлемых солей | |
| WO2025136150A1 (ru) | Способ получения дексмедетомидина и его фармацевтически приемлемых солей | |
| EP0151052B1 (fr) | Aminoéthylimidazole, composition pharmaceutique en contenant et procédé de préparation | |
| EP0501870A1 (fr) | Nouvelles compositions pharmaceutiques, notamment pour le traitement des colopathies fonctionnelles et leur procédé d'obtention | |
| EP0275221B1 (fr) | Nouveaux dérivés du N-(1H-indol 4-yl) benzamide ainsi que leurs sels, leur application à titre de médicaments et les compositions les renfermant | |
| RU2339631C1 (ru) | Способ получения эзомепразола | |
| EP0430800B1 (fr) | Nouvelles benzothiazolinones substituées, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20060907 |
|
| A131 | Notification of reasons for refusal |
Effective date: 20100122 Free format text: JAPANESE INTERMEDIATE CODE: A131 |
|
| RD02 | Notification of acceptance of power of attorney |
Effective date: 20100303 Free format text: JAPANESE INTERMEDIATE CODE: A7422 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20100303 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7422 Effective date: 20100512 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20100512 |
|
| A072 | Dismissal of procedure |
Effective date: 20100615 Free format text: JAPANESE INTERMEDIATE CODE: A073 |
|
| A131 | Notification of reasons for refusal |
Effective date: 20101130 Free format text: JAPANESE INTERMEDIATE CODE: A131 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110128 |
|
| A02 | Decision of refusal |
Effective date: 20110222 Free format text: JAPANESE INTERMEDIATE CODE: A02 |