JP2010077286A - Silicone resin composition and adhesive film - Google Patents
Silicone resin composition and adhesive film Download PDFInfo
- Publication number
- JP2010077286A JP2010077286A JP2008247382A JP2008247382A JP2010077286A JP 2010077286 A JP2010077286 A JP 2010077286A JP 2008247382 A JP2008247382 A JP 2008247382A JP 2008247382 A JP2008247382 A JP 2008247382A JP 2010077286 A JP2010077286 A JP 2010077286A
- Authority
- JP
- Japan
- Prior art keywords
- silicone resin
- resin composition
- adhesive film
- film
- acrylate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 229920002050 silicone resin Polymers 0.000 title claims abstract description 29
- 239000011342 resin composition Substances 0.000 title claims abstract description 24
- 239000002313 adhesive film Substances 0.000 title claims abstract description 18
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 claims abstract description 13
- 239000003054 catalyst Substances 0.000 claims abstract description 11
- 239000003505 polymerization initiator Substances 0.000 claims abstract description 8
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 3
- 239000000758 substrate Substances 0.000 abstract description 8
- 239000003795 chemical substances by application Substances 0.000 abstract description 4
- SCPYDCQAZCOKTP-UHFFFAOYSA-N silanol Chemical compound [SiH3]O SCPYDCQAZCOKTP-UHFFFAOYSA-N 0.000 abstract description 3
- BFXIKLCIZHOAAZ-UHFFFAOYSA-N methyltrimethoxysilane Chemical compound CO[Si](C)(OC)OC BFXIKLCIZHOAAZ-UHFFFAOYSA-N 0.000 abstract description 2
- 239000012790 adhesive layer Substances 0.000 description 10
- 239000000126 substance Substances 0.000 description 7
- 238000001035 drying Methods 0.000 description 6
- 229920002799 BoPET Polymers 0.000 description 5
- 239000004820 Pressure-sensitive adhesive Substances 0.000 description 5
- 230000000052 comparative effect Effects 0.000 description 5
- -1 polysiloxane structure Polymers 0.000 description 5
- 229920005989 resin Polymers 0.000 description 5
- 239000011347 resin Substances 0.000 description 5
- 239000012298 atmosphere Substances 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 3
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 3
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- CQQXCSFSYHAZOO-UHFFFAOYSA-L [acetyloxy(dioctyl)stannyl] acetate Chemical compound CCCCCCCC[Sn](OC(C)=O)(OC(C)=O)CCCCCCCC CQQXCSFSYHAZOO-UHFFFAOYSA-L 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 125000004386 diacrylate group Chemical group 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 3
- 229910052753 mercury Inorganic materials 0.000 description 3
- YDKNBNOOCSNPNS-UHFFFAOYSA-N methyl 1,3-benzoxazole-2-carboxylate Chemical compound C1=CC=C2OC(C(=O)OC)=NC2=C1 YDKNBNOOCSNPNS-UHFFFAOYSA-N 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 150000002978 peroxides Chemical class 0.000 description 3
- 229920000139 polyethylene terephthalate Polymers 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- 229910001868 water Inorganic materials 0.000 description 3
- QNODIIQQMGDSEF-UHFFFAOYSA-N (1-hydroxycyclohexyl)-phenylmethanone Chemical compound C=1C=CC=CC=1C(=O)C1(O)CCCCC1 QNODIIQQMGDSEF-UHFFFAOYSA-N 0.000 description 2
- TXBCBTDQIULDIA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC(CO)(CO)CO TXBCBTDQIULDIA-UHFFFAOYSA-N 0.000 description 2
- FDSUVTROAWLVJA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol;prop-2-enoic acid Chemical compound OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OCC(CO)(CO)COCC(CO)(CO)CO FDSUVTROAWLVJA-UHFFFAOYSA-N 0.000 description 2
- LWRBVKNFOYUCNP-UHFFFAOYSA-N 2-methyl-1-(4-methylsulfanylphenyl)-2-morpholin-4-ylpropan-1-one Chemical compound C1=CC(SC)=CC=C1C(=O)C(C)(C)N1CCOCC1 LWRBVKNFOYUCNP-UHFFFAOYSA-N 0.000 description 2
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 2
- 229920000178 Acrylic resin Polymers 0.000 description 2
- 239000004925 Acrylic resin Substances 0.000 description 2
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 2
- 229910000831 Steel Inorganic materials 0.000 description 2
- MPIAGWXWVAHQBB-UHFFFAOYSA-N [3-prop-2-enoyloxy-2-[[3-prop-2-enoyloxy-2,2-bis(prop-2-enoyloxymethyl)propoxy]methyl]-2-(prop-2-enoyloxymethyl)propyl] prop-2-enoate Chemical compound C=CC(=O)OCC(COC(=O)C=C)(COC(=O)C=C)COCC(COC(=O)C=C)(COC(=O)C=C)COC(=O)C=C MPIAGWXWVAHQBB-UHFFFAOYSA-N 0.000 description 2
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 2
- 238000007259 addition reaction Methods 0.000 description 2
- 238000006482 condensation reaction Methods 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- 239000012024 dehydrating agents Substances 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 239000003999 initiator Substances 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical compound CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 229920001296 polysiloxane Polymers 0.000 description 2
- 239000010959 steel Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 229920002803 thermoplastic polyurethane Polymers 0.000 description 2
- VXUYXOFXAQZZMF-UHFFFAOYSA-N titanium(IV) isopropoxide Chemical compound CC(C)O[Ti](OC(C)C)(OC(C)C)OC(C)C VXUYXOFXAQZZMF-UHFFFAOYSA-N 0.000 description 2
- 239000002023 wood Substances 0.000 description 2
- PRBBFHSSJFGXJS-UHFFFAOYSA-N (2,2-dimethyl-3-prop-2-enoyloxypropyl) prop-2-enoate;3-hydroxy-2,2-dimethylpropanoic acid Chemical compound OCC(C)(C)C(O)=O.C=CC(=O)OCC(C)(C)COC(=O)C=C PRBBFHSSJFGXJS-UHFFFAOYSA-N 0.000 description 1
- MFWFDRBPQDXFRC-LNTINUHCSA-N (z)-4-hydroxypent-3-en-2-one;vanadium Chemical class [V].C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O MFWFDRBPQDXFRC-LNTINUHCSA-N 0.000 description 1
- POILWHVDKZOXJZ-ARJAWSKDSA-M (z)-4-oxopent-2-en-2-olate Chemical class C\C([O-])=C\C(C)=O POILWHVDKZOXJZ-ARJAWSKDSA-M 0.000 description 1
- LRZPQLZONWIQOJ-UHFFFAOYSA-N 10-(2-methylprop-2-enoyloxy)decyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCOC(=O)C(C)=C LRZPQLZONWIQOJ-UHFFFAOYSA-N 0.000 description 1
- KWVGIHKZDCUPEU-UHFFFAOYSA-N 2,2-dimethoxy-2-phenylacetophenone Chemical compound C=1C=CC=CC=1C(OC)(OC)C(=O)C1=CC=CC=C1 KWVGIHKZDCUPEU-UHFFFAOYSA-N 0.000 description 1
- HWSSEYVMGDIFMH-UHFFFAOYSA-N 2-[2-[2-(2-methylprop-2-enoyloxy)ethoxy]ethoxy]ethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCOCCOCCOC(=O)C(C)=C HWSSEYVMGDIFMH-UHFFFAOYSA-N 0.000 description 1
- IHEDBVUTTQXGSJ-UHFFFAOYSA-M 2-[bis(2-oxidoethyl)amino]ethanolate;titanium(4+);hydroxide Chemical compound [OH-].[Ti+4].[O-]CCN(CC[O-])CC[O-] IHEDBVUTTQXGSJ-UHFFFAOYSA-M 0.000 description 1
- JMWGZSWSTCGVLX-UHFFFAOYSA-N 2-ethyl-2-(hydroxymethyl)propane-1,3-diol;2-methylprop-2-enoic acid Chemical class CC(=C)C(O)=O.CC(=C)C(O)=O.CC(=C)C(O)=O.CCC(CO)(CO)CO JMWGZSWSTCGVLX-UHFFFAOYSA-N 0.000 description 1
- RIWRBSMFKVOJMN-UHFFFAOYSA-N 2-methyl-1-phenylpropan-2-ol Chemical compound CC(C)(O)CC1=CC=CC=C1 RIWRBSMFKVOJMN-UHFFFAOYSA-N 0.000 description 1
- NJBCRXCAPCODGX-UHFFFAOYSA-N 2-methyl-n-(2-methylpropyl)propan-1-amine Chemical compound CC(C)CNCC(C)C NJBCRXCAPCODGX-UHFFFAOYSA-N 0.000 description 1
- UPTHZKIDNHJFKQ-UHFFFAOYSA-N 2-methylprop-2-enoic acid;propane-1,2,3-triol Chemical compound CC(=C)C(O)=O.CC(=C)C(O)=O.OCC(O)CO UPTHZKIDNHJFKQ-UHFFFAOYSA-N 0.000 description 1
- VYVFQBFOMKEKBG-UHFFFAOYSA-L 3,3-dibutyl-2,4,3-benzodioxastannepine-1,5-dione Chemical compound O=C1O[Sn](CCCC)(CCCC)OC(=O)C2=CC=CC=C21 VYVFQBFOMKEKBG-UHFFFAOYSA-L 0.000 description 1
- XOJWAAUYNWGQAU-UHFFFAOYSA-N 4-(2-methylprop-2-enoyloxy)butyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCOC(=O)C(C)=C XOJWAAUYNWGQAU-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- DBCAQXHNJOFNGC-UHFFFAOYSA-N 4-bromo-1,1,1-trifluorobutane Chemical compound FC(F)(F)CCCBr DBCAQXHNJOFNGC-UHFFFAOYSA-N 0.000 description 1
- JHWGFJBTMHEZME-UHFFFAOYSA-N 4-prop-2-enoyloxybutyl prop-2-enoate Chemical compound C=CC(=O)OCCCCOC(=O)C=C JHWGFJBTMHEZME-UHFFFAOYSA-N 0.000 description 1
- SAPGBCWOQLHKKZ-UHFFFAOYSA-N 6-(2-methylprop-2-enoyloxy)hexyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCOC(=O)C(C)=C SAPGBCWOQLHKKZ-UHFFFAOYSA-N 0.000 description 1
- FIHBHSQYSYVZQE-UHFFFAOYSA-N 6-prop-2-enoyloxyhexyl prop-2-enoate Chemical compound C=CC(=O)OCCCCCCOC(=O)C=C FIHBHSQYSYVZQE-UHFFFAOYSA-N 0.000 description 1
- YJVIKVWFGPLAFS-UHFFFAOYSA-N 9-(2-methylprop-2-enoyloxy)nonyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCOC(=O)C(C)=C YJVIKVWFGPLAFS-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- BOTDANWDWHJENH-UHFFFAOYSA-N Tetraethyl orthosilicate Chemical compound CCO[Si](OCC)(OCC)OCC BOTDANWDWHJENH-UHFFFAOYSA-N 0.000 description 1
- DAKWPKUUDNSNPN-UHFFFAOYSA-N Trimethylolpropane triacrylate Chemical compound C=CC(=O)OCC(CC)(COC(=O)C=C)COC(=O)C=C DAKWPKUUDNSNPN-UHFFFAOYSA-N 0.000 description 1
- OKKRPWIIYQTPQF-UHFFFAOYSA-N Trimethylolpropane trimethacrylate Chemical compound CC(=C)C(=O)OCC(CC)(COC(=O)C(C)=C)COC(=O)C(C)=C OKKRPWIIYQTPQF-UHFFFAOYSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- ULQMPOIOSDXIGC-UHFFFAOYSA-N [2,2-dimethyl-3-(2-methylprop-2-enoyloxy)propyl] 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(C)(C)COC(=O)C(C)=C ULQMPOIOSDXIGC-UHFFFAOYSA-N 0.000 description 1
- KNSXNCFKSZZHEA-UHFFFAOYSA-N [3-prop-2-enoyloxy-2,2-bis(prop-2-enoyloxymethyl)propyl] prop-2-enoate Chemical compound C=CC(=O)OCC(COC(=O)C=C)(COC(=O)C=C)COC(=O)C=C KNSXNCFKSZZHEA-UHFFFAOYSA-N 0.000 description 1
- ISKQADXMHQSTHK-UHFFFAOYSA-N [4-(aminomethyl)phenyl]methanamine Chemical compound NCC1=CC=C(CN)C=C1 ISKQADXMHQSTHK-UHFFFAOYSA-N 0.000 description 1
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 description 1
- 150000004075 acetic anhydrides Chemical class 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 125000005370 alkoxysilyl group Chemical group 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- YHWCPXVTRSHPNY-UHFFFAOYSA-N butan-1-olate;titanium(4+) Chemical compound [Ti+4].CCCC[O-].CCCC[O-].CCCC[O-].CCCC[O-] YHWCPXVTRSHPNY-UHFFFAOYSA-N 0.000 description 1
- BPOZNMOEPOHHSC-UHFFFAOYSA-N butyl prop-2-enoate;prop-2-enoic acid Chemical compound OC(=O)C=C.CCCCOC(=O)C=C BPOZNMOEPOHHSC-UHFFFAOYSA-N 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 239000007809 chemical reaction catalyst Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 150000004696 coordination complex Chemical class 0.000 description 1
- 239000012975 dibutyltin dilaurate Substances 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- WNEDOMBPJDUQPS-BFIADXHOSA-L dioctyltin(2+);(z)-4-hydroxy-4-oxobut-2-enoate Chemical compound CCCCCCCC[Sn](OC(=O)\C=C/C(O)=O)(OC(=O)\C=C/C(O)=O)CCCCCCCC WNEDOMBPJDUQPS-BFIADXHOSA-L 0.000 description 1
- 150000002009 diols Chemical class 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- SBRXLTRZCJVAPH-UHFFFAOYSA-N ethyl(trimethoxy)silane Chemical compound CC[Si](OC)(OC)OC SBRXLTRZCJVAPH-UHFFFAOYSA-N 0.000 description 1
- STVZJERGLQHEKB-UHFFFAOYSA-N ethylene glycol dimethacrylate Substances CC(=C)C(=O)OCCOC(=O)C(C)=C STVZJERGLQHEKB-UHFFFAOYSA-N 0.000 description 1
- 239000003925 fat Substances 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 238000001879 gelation Methods 0.000 description 1
- 230000004313 glare Effects 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- GIWKOZXJDKMGQC-UHFFFAOYSA-L lead(2+);naphthalene-2-carboxylate Chemical compound [Pb+2].C1=CC=CC2=CC(C(=O)[O-])=CC=C21.C1=CC=CC2=CC(C(=O)[O-])=CC=C21 GIWKOZXJDKMGQC-UHFFFAOYSA-L 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 150000005309 metal halides Chemical class 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- GEMHFKXPOCTAIP-UHFFFAOYSA-N n,n-dimethyl-n'-phenylcarbamimidoyl chloride Chemical compound CN(C)C(Cl)=NC1=CC=CC=C1 GEMHFKXPOCTAIP-UHFFFAOYSA-N 0.000 description 1
- UIEKYBOPAVTZKW-UHFFFAOYSA-L naphthalene-2-carboxylate;nickel(2+) Chemical compound [Ni+2].C1=CC=CC2=CC(C(=O)[O-])=CC=C21.C1=CC=CC2=CC(C(=O)[O-])=CC=C21 UIEKYBOPAVTZKW-UHFFFAOYSA-L 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 229910017464 nitrogen compound Inorganic materials 0.000 description 1
- 150000002830 nitrogen compounds Chemical class 0.000 description 1
- 125000005461 organic phosphorous group Chemical group 0.000 description 1
- 125000002524 organometallic group Chemical group 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 239000002985 plastic film Substances 0.000 description 1
- 229920006255 plastic film Polymers 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 238000010526 radical polymerization reaction Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- LFQCEHFDDXELDD-UHFFFAOYSA-N tetramethyl orthosilicate Chemical compound CO[Si](OC)(OC)OC LFQCEHFDDXELDD-UHFFFAOYSA-N 0.000 description 1
- 238000005979 thermal decomposition reaction Methods 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- CPUDPFPXCZDNGI-UHFFFAOYSA-N triethoxy(methyl)silane Chemical compound CCO[Si](C)(OCC)OCC CPUDPFPXCZDNGI-UHFFFAOYSA-N 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
Landscapes
- Compositions Of Macromolecular Compounds (AREA)
- Adhesive Tapes (AREA)
- Adhesives Or Adhesive Processes (AREA)
Abstract
Description
本発明は基材適性および密着性に優れ、かつ再はく離性にも優れるシリコーン樹脂組成物および粘着フィルムに関する。 The present invention relates to a silicone resin composition and an adhesive film that are excellent in substrate suitability and adhesion, and also excellent in releasability.
近年、液晶ディスプレイや有機ELディスプレイなどの薄型表示装置の実用化、低コスト化、高機能化などによって、これらの表示装置はテレビやコンピューターのディスプレイといった据置き型の機器だけでなく、携帯電話、携帯情報端末、携帯音楽プレーヤー、携帯ゲーム機、デジタルカメラ、デジタルビデオなどの携帯機器にも採用されるようになっている。こうした表示装置の表面には、傷付き防止、汚れ防止、帯電防止、防眩、反射防止などを目的とした機能性フィルムが用いられている。また、看板や広告用として、施工や取替えが容易な再はく離性ディスプレイフィルムが使用されている。 In recent years, due to the practical use, low cost, high functionality, etc. of thin display devices such as liquid crystal displays and organic EL displays, these display devices are not only stationary devices such as televisions and computer displays, but also mobile phones, It is also adopted in portable devices such as portable information terminals, portable music players, portable game machines, digital cameras, and digital videos. On the surface of such a display device, a functional film for the purpose of preventing scratches, preventing dirt, preventing charging, preventing glare, preventing reflection, and the like is used. In addition, releasable display films that are easy to construct and replace are used for signs and advertisements.
従来、このようなフィルムの粘着剤としてアクリル系樹脂、ウレタン系樹脂、シリコーン系樹脂などが用いられてきたが、アクリル系樹脂やウレタン系樹脂は貼り付け時に巻き込まれた泡が抜けにくいという問題があった。機能性フィルムはアフターマーケットで消費者が購入して自ら貼り付けを行うことも多く、貼り付け面とフィルムとの間に気泡が入ってしまった場合、泡抜け性が悪いと何度も剥がして貼り直す必要があるため好ましくなく、粘着層と基材フィルムとの密着性に劣るフィルムは、貼り付けと剥離を繰り返すことで貼付け面に粘着層が残ってしまうなど、再はく離性に問題のあるケースがあった。 Conventionally, acrylic resins, urethane resins, silicone resins, and the like have been used as adhesives for such films. However, acrylic resins and urethane resins have a problem that bubbles entrained during pasting are difficult to escape. there were. Functional films are often purchased and pasted by consumers at the aftermarket, and if air bubbles enter between the pasting surface and the film, they will be peeled off many times if the bubble removal is poor. It is not desirable because it needs to be re-attached. Films with poor adhesion between the adhesive layer and the base film have problems with re-peelability, such as the adhesive layer remaining on the application surface by repeated application and peeling. There was a case.
シリコーン系樹脂としては付加反応型および過酸化物硬化型が用いられているが(例えば特許文献1、2)、付加反応型は反応時に水、イオウ、チッソ化合物、有機金属塩などが存在すると硬化阻害が生じるおそれがあるため、これらを含有する基材や雰囲気下では使用できなかった。また、過酸化物硬化型は概ね150℃以上の高温条件でなければ硬化しないため、プラスチックフィルムのような耐熱性の低い基材には使用できなかった。
本発明の課題は基材適性および密着性に優れ、かつ再はく離性にも優れるシリコーン樹脂組成物および粘着フィルムを提供することである。 The subject of this invention is providing the silicone resin composition and adhesive film which are excellent in base-material suitability and adhesiveness, and are excellent also in releasability.
本発明者らが鋭意検討を行った結果、硬化触媒を含有するアルコール脱離型シリコーン樹脂において、さらに多官能(メタ)アクリレート、重合開始剤を配合することによって前記課題を解決できることを見出した。 As a result of intensive studies by the present inventors, it has been found that the above problem can be solved by further blending a polyfunctional (meth) acrylate and a polymerization initiator in an alcohol detachable silicone resin containing a curing catalyst.
本発明からなるシリコーン樹脂組成物はアルコール脱離型シリコーン樹脂を主成分とするため、特定の基材に対する硬化阻害や高温条件にする必要がなく、幅広い基材に対して使用できる。また、密着性と再はく離性を両立できているため、再はく離型粘着剤として有用である。さらに、本発明のシリコーン樹脂組成物を塗布したフィルムは優れた再はく離型粘着フィルムとなるため、目的に応じた加工を行うことにより傷付き防止、汚れ防止、帯電防止、防眩、反射防止などを目的とした表示装置用機能性粘着フィルムとして有用である。また、看板や広告として用いられる再はく離性ディスプレイ粘着フィルムとしても有用である。 Since the silicone resin composition according to the present invention contains an alcohol-detachable silicone resin as a main component, it is not necessary to inhibit the curing of a specific substrate or to use a high temperature condition, and can be used for a wide range of substrates. Moreover, since both adhesiveness and releasability can be achieved, it is useful as a releasable pressure-sensitive adhesive. Furthermore, since the film coated with the silicone resin composition of the present invention becomes an excellent re-peeling adhesive film, it is possible to prevent scratches, dirt, antistatic, antiglare, antireflection, etc. by processing according to the purpose. It is useful as a functional pressure-sensitive adhesive film for a display device for the purpose. Moreover, it is useful also as a releasable display adhesive film used as a signboard or an advertisement.
本発明に用いるアルコール脱離型シリコーン樹脂は、ポリシロキサン構造を骨格として末端にアルコキシシリル基を有し、縮合反応により硬化可能な樹脂である。アルコール脱離型シリコーン樹脂は市販品を用いても良いし、例えば特開平1−113429号公報に開示されるように、シラノールで末端が停止したポリジオルガノシロキサン(シリコーンジオール)をメチルトリメトキシシラン、メチルトリエトキシシラン、エチルトリメトキシシラン、テトラエトキシシランなどの末端キャッピング剤と反応させることにより合成してもよい。 The alcohol-leaving silicone resin used in the present invention is a resin that has a polysiloxane structure as a skeleton and has an alkoxysilyl group at the terminal and can be cured by a condensation reaction. Commercially available alcohol-leaving silicone resins may be used. For example, as disclosed in JP-A-1-113429, polydiorganosiloxane terminated with silanol (silicone diol) is converted to methyltrimethoxysilane, You may synthesize | combine by making it react with terminal capping agents, such as methyltriethoxysilane, ethyltrimethoxysilane, and tetraethoxysilane.
硬化触媒はアルコール脱離型シリコーン樹脂の縮合反応を促進するため添加される。具体的には、有機錫、無機錫、チタン触媒、ビスマス触媒、金属錯体、白金触媒、塩基性物質及び有機燐酸化物等が使用できる。有機錫の具体例としては、ジブチル錫ジラウリレート、ジオクチル錫ジマレート、ジブチル錫フタレート、オクチル酸第一錫、ジブチル錫ジアセテート等が挙げられる。金属錯体としては、テトラブチルチタネート、テトライソプロピルチタネート、トリエタノールアミンチタネート等のチタネート化合物類、オクチル酸鉛、ナフテン酸鉛、ナフテン酸ニッケル、ナフテン酸コバルト等のカルボン酸金属塩、アルミニウムアセチルアセテート錯体等の金属アセチルアセテート錯体、バナジウムアセチルアセトナート錯体等の金属アセチルアセトナート錯体などが挙げられる。通常、シリコーン樹脂組成物固形分に対して1〜2%程度の添加で十分な効果を発現し、用途によって適宜添加量を調整する。 The curing catalyst is added to accelerate the condensation reaction of the alcohol detachable silicone resin. Specifically, organic tin, inorganic tin, titanium catalyst, bismuth catalyst, metal complex, platinum catalyst, basic substance, organic phosphorous oxide, and the like can be used. Specific examples of the organic tin include dibutyltin dilaurate, dioctyltin dimaleate, dibutyltin phthalate, stannous octylate, dibutyltin diacetate and the like. Examples of metal complexes include titanate compounds such as tetrabutyl titanate, tetraisopropyl titanate, triethanolamine titanate, carboxylic acid metal salts such as lead octylate, lead naphthenate, nickel naphthenate, cobalt naphthenate, aluminum acetyl acetate complex, etc. And metal acetylacetonate complexes such as metal acetylacetate complexes and vanadium acetylacetonate complexes. Usually, the addition of about 1 to 2% with respect to the solid content of the silicone resin composition produces a sufficient effect, and the addition amount is appropriately adjusted depending on the application.
多官能(メタ)アクリレートは2以上の官能基を有するアクリレートまたはメタクリレートである。多官能アクリレートとして、1,4−ブタンジオールジアクリレート、1,6−ヘキサンジオールジアクリレート、ネオペンチルグリコールジアクリレート、ポリエチレングリコールジアクリレート、ヒドロキシピバリン酸ネオペンチルグリコールジアクリレート、ジシクロペンタニルジアクリレート、カプロラクトン変性ジシクロペンテニルジアクリレート、エチレンオキシド変性リン酸ジアクリレート、アリル化シクロヘキシルジアクリレート、イソシアヌレートジアクリレート、トリメチロールプロパントリアクリレート、ジペンタエリスリトールトリアクリレート、プロピオン酸変性ジペンタエリスリトールトリアクリレート、ペンタエリスリトールトリアクリレート、プロピレンオキシド変性トリメチロールプロパントリアクリレート、トリス(アクリロキシエチル)イソシアヌレート、ジペンタエリスリトールペンタアクリレート、プロピオン酸変性ジペンタエリスリトールペンタアクリレート、ジペンタエリスリトールヘキサアクリレート、カプロラクトン変性ジペンタエリスリトールヘキサアクリレートなどが挙げられる。 The polyfunctional (meth) acrylate is an acrylate or methacrylate having two or more functional groups. As polyfunctional acrylates, 1,4-butanediol diacrylate, 1,6-hexanediol diacrylate, neopentyl glycol diacrylate, polyethylene glycol diacrylate, hydroxypivalate neopentyl glycol diacrylate, dicyclopentanyl diacrylate, Caprolactone-modified dicyclopentenyl diacrylate, ethylene oxide-modified phosphate diacrylate, allylated cyclohexyl diacrylate, isocyanurate diacrylate, trimethylolpropane triacrylate, dipentaerythritol triacrylate, propionic acid-modified dipentaerythritol triacrylate, pentaerythritol triacrylate Acrylate, propylene oxide modified trimethylolpropane triacryle , Tris (acryloxyethyl) isocyanurate, dipentaerythritol pentaacrylate, propionic acid-modified dipentaerythritol pentaacrylate, dipentaerythritol hexaacrylate, caprolactone-modified dipentaerythritol hexaacrylate.
多官能メタクリレートとして、エチレングリコールジメタクリレート、トリエチレングリコールジメタクリレート、1,4−ブタンジオールジメタクリレート、ネオペンチルグリコールジメタクリレート、1,6−ヘキサンジオールジメタクレート、1,9−ノナンジオールジメタクリレート、1,10−デカンジオールジメタクリレート、グリセリンジメタクリレート、ジメチロールートリシクロデカンジメタクリレート、トリメチロールプロパントリメタクリレートやエトキシ化トリメチロールプロパントリメタクリレートなどが挙げられる。これらの多官能(メタ)アクリレートは単独で用いても良いし、2種以上を併用しても良い。また、多官能(メタ)アクリレートとともに単官能アクリレートを用いても良い。 As polyfunctional methacrylate, ethylene glycol dimethacrylate, triethylene glycol dimethacrylate, 1,4-butanediol dimethacrylate, neopentyl glycol dimethacrylate, 1,6-hexanediol dimethacrylate, 1,9-nonanediol dimethacrylate, Examples include 1,10-decanediol dimethacrylate, glycerin dimethacrylate, dimethylol-tricyclodecane dimethacrylate, trimethylolpropane trimethacrylate, ethoxylated trimethylolpropane trimethacrylate, and the like. These polyfunctional (meth) acrylates may be used alone or in combination of two or more. Moreover, you may use monofunctional acrylate with polyfunctional (meth) acrylate.
重合開始剤は多官能(メタ)アクリレートを硬化させるために使用される。ラジカル重合可能な重合開始剤であれば特に限定されないが、過酸化物などの熱分解タイプのラジカル発生剤を用いる場合は低温で硬化可能という本発明のシリコーン樹脂組成物の特長を損なわないため、1分間半減期温度が140℃以下のものを使用することが好ましい。また、高温にする必要がないため、ベンゾフェノンやアセトフェノンなどの紫外線分解タイプのラジカル発生剤を開始剤として使用しても良い。市販品としては、イルガキュア184、イルガキュア651、イルガキュア907(チバ・スペシャルティ・ケミカルズ社製)や、ダロキュア1173(メルク社製)が挙げられる。上記重合開始剤はいずれもモノマーに対して1〜20部、好ましくは3〜10部添加する。量が少なすぎると硬化不良となるおそれがあり、量が多すぎるとラジカル発生時の分解物が硬化樹脂の物性に影響を及ぼすおそれがある。 The polymerization initiator is used to cure the polyfunctional (meth) acrylate. It is not particularly limited as long as it is a polymerization initiator capable of radical polymerization, but when using a thermal decomposition type radical generator such as peroxide, the characteristics of the silicone resin composition of the present invention that can be cured at a low temperature are not impaired. It is preferable to use a one-minute half-life temperature of 140 ° C. or lower. Moreover, since it is not necessary to raise the temperature, an ultraviolet decomposition type radical generator such as benzophenone or acetophenone may be used as an initiator. Examples of commercially available products include Irgacure 184, Irgacure 651, Irgacure 907 (manufactured by Ciba Specialty Chemicals) and Darocur 1173 (manufactured by Merck). The polymerization initiator is added in an amount of 1 to 20 parts, preferably 3 to 10 parts, based on the monomer. If the amount is too small, curing may be poor, while if the amount is too large, decomposition products at the time of radical generation may affect the physical properties of the cured resin.
紫外線の照射条件は、超高圧水銀灯、高圧水銀灯、低圧水銀灯、カーボンアーク、メタルハライドランプ等を用い、100〜400nm、好ましくは200〜400nmの波長領域で100〜1000mJ/cm2、好ましくは200〜400mJ/cm2のエネルギーを有する紫外線を照射する。硬化阻害を防止するため、窒素ガス等の不活性ガス下で照射を行っても良い。 Ultraviolet irradiation conditions are ultra-high pressure mercury lamp, high pressure mercury lamp, low pressure mercury lamp, carbon arc, metal halide lamp, etc., and 100 to 400 nm, preferably 200 to 400 nm, preferably 100 to 1000 mJ / cm 2 , preferably 200 to 400 mJ. Irradiate ultraviolet rays having energy of / cm 2 . In order to prevent curing inhibition, irradiation may be performed under an inert gas such as nitrogen gas.
本発明のシリコーン樹脂組成物は前記4成分を必須成分とする。また、本発明のシリコーン樹脂組成物は前記4成分以外に公知の添加剤を用いることができる。例えば脱水剤の添加によって組成物の保存性を向上することができ、離型剤の添加によって密着性と再剥離性を調整することができ、溶剤の使用によってフィルムへの塗工性を向上することができる。なお、親水性の溶剤を用いると含有水分の影響により保存性が低下するおそれがあるため、疎水性の溶剤を用いることが好ましい。 The silicone resin composition of the present invention comprises the four components as essential components. Moreover, the silicone resin composition of this invention can use a well-known additive other than the said 4 component. For example, the storage stability of the composition can be improved by adding a dehydrating agent, the adhesion and removability can be adjusted by adding a release agent, and the coating property to the film is improved by using a solvent. be able to. In addition, since there exists a possibility that preservability may fall under the influence of moisture content when a hydrophilic solvent is used, it is preferable to use a hydrophobic solvent.
本発明のシリコーン樹脂組成物を用いて粘着フィルムを作成するには、PETなどの基材に硬化後の膜厚が50〜150μm程度となるように塗布し、100〜140℃程度、より好ましくは120〜140℃で2〜3分間程度乾燥、硬化させ、離型フィルムを貼り合わせるなどの方法が用いられる。光重合開始剤を用いる場合には、乾燥、硬化が必要な照射強度で光を照射すれば良い。本発明のシリコーン樹脂組成物は100℃程度から硬化可能であるため、PETなどの耐熱性の低い基材に対しても問題なく使用することができる。 In order to create an adhesive film using the silicone resin composition of the present invention, it is applied to a substrate such as PET so that the film thickness after curing is about 50 to 150 μm, and is preferably about 100 to 140 ° C., more preferably A method of drying and curing at 120 to 140 ° C. for about 2 to 3 minutes and bonding a release film is used. In the case of using a photopolymerization initiator, light may be irradiated at an irradiation intensity that requires drying and curing. Since the silicone resin composition of the present invention can be cured from about 100 ° C., it can be used without problems even on substrates having low heat resistance such as PET.
以下、実施例に基づいて本発明をより具体的に説明する。当然、本発明は実施例に何ら制約されるものではない。 Hereinafter, based on an Example, this invention is demonstrated more concretely. Of course, the present invention is not limited to the embodiments.
アルコール脱離型シリコーン樹脂の製造
反応装置に末端にシラノール変成を導入したジメチルポリシロキサン(粘度20000mPa・s/25℃)600重量部を添加し、水分量が100ppm未満となるよう減圧下において100℃で2時間攪拌した。反応装置に窒素を充填し、常圧とした上で末端キャッピング剤としてテトラメトキシシラン40重量部と、末端キャッピング反応触媒としてギ酸(88%水溶液)0.31重量部およびジイソブチルアミン0.78重量部を混合したものを添加して、80℃雰囲気下で2時間反応させた。内容物をサンプリングし、テトラアルチルチタネートを添加してゲル化が起こらないことを確認し、反応完了を確認した。その後、減圧下において110℃で3時間攪拌することによって触媒を分解および除去を行い、室温に冷却後にトルエンを添加して不揮発分を60%に調整した。さらに脱水剤(東レ・ダウコーニング社製、商品名Z−6300)12重量部を添加して15分間以上攪拌を行うことにより系内を脱水し、樹脂組成物1を得た。
Production of alcohol-leaving silicone resin 600 parts by weight of dimethylpolysiloxane (viscosity 20000 mPa · s / 25 ° C.) introduced with silanol modification at the end is added to the reaction apparatus, and the water content is reduced to 100 ° C. under reduced pressure so that the water content is less than 100 ppm. For 2 hours. The reactor was filled with nitrogen and brought to normal pressure, and then 40 parts by weight of tetramethoxysilane as an end capping agent, 0.31 part by weight of formic acid (88% aqueous solution) and 0.78 part by weight of diisobutylamine as an end capping reaction catalyst. The mixture was added and reacted for 2 hours at 80 ° C. atmosphere. The contents were sampled, tetraartyl titanate was added and it was confirmed that gelation did not occur, and the completion of the reaction was confirmed. Thereafter, the catalyst was decomposed and removed by stirring at 110 ° C. for 3 hours under reduced pressure. After cooling to room temperature, toluene was added to adjust the nonvolatile content to 60%. Further, 12 parts by weight of a dehydrating agent (trade name Z-6300, manufactured by Toray Dow Corning Co., Ltd.) was added, and the system was dehydrated by stirring for 15 minutes or more to obtain a resin composition 1.
実施例1
樹脂組成物1 100重量部に対して、硬化触媒としてジオクチル錫ジアセテート(日東化成製、商品名ネオスタンU−820)0.9重量部、多官能(メタ)アクリレートとしてネオペンチルグリコールジアクリレート(共栄社化学工業社製、商品名ライトアクリレートNP−A)10重量部、重合開始剤としてパーキュアO(日本油脂社製、商品名)0.5重量部を添加することにより、実施例1のシリコーン樹脂組成物を得た。厚み100μm、両面易接着コーティング済みのPETフィルムを基材として、マイクロメーター付フィルムアプリケーターを使用し、乾燥後に樹脂厚みが約30μmとなるように実施例1のシリコーン樹脂組成物を塗工した。フィルムを120℃で3分間乾燥し、離型フィルムを貼り合わせることにより、実施例1の粘着フィルムを得た。
Example 1
Resin composition 1 100 parts by weight of dioctyltin diacetate (product name: Neostan U-820, manufactured by Nitto Kasei) as a curing catalyst, 0.9 part by weight of polyfunctional (meth) acrylate, neopentyl glycol diacrylate (Kyoeisha) The silicone resin composition of Example 1 was added by adding 10 parts by weight of Chemical Industry Co., Ltd., trade name Light Acrylate NP-A) and 0.5 parts by weight of Percure O (made by Nippon Oil & Fats, trade name) as a polymerization initiator. I got a thing. Using a PET film having a thickness of 100 μm and already coated on both sides with easy adhesion, a film applicator with a micrometer was used, and the silicone resin composition of Example 1 was applied so that the resin thickness after drying was about 30 μm. The film was dried at 120 ° C. for 3 minutes, and a release film was bonded to obtain an adhesive film of Example 1.
実施例2
樹脂組成物1 100重量部に対して、硬化触媒としてジオクチル錫ジアセテート(日東化成製、商品名ネオスタンU−820)0.9重量部、多官能(メタ)アクリレートとしてネオペンチルグリコールジアクリレート(共栄社化学工業社製、商品名ライトアクリレートNP−A)5重量部、単官能アクリレートとしてブチルアクリレート(共栄社化学工業社製、商品名ライトアクリレートBA)5重量部、重合開始剤としてイルガキュア184(チバ・スペシャルティ・ケミカルズ社製、商品名)0.25重量部、イルガキュア907(チバ・スペシャルティ・ケミカルズ社製、商品名)0.25重量部を添加することにより、実施例2のシリコーン樹脂組成物を得た。厚み100μm、両面易接着コーティング済みのPETフィルムを基材として、マイクロメーター付フィルムアプリケーターを使用し、乾燥後に樹脂厚みが約30μmとなるように実施例1のシリコーン樹脂組成物を塗工した。フィルムを120℃で3分間乾燥後、300mJ/cm2のエネルギーを有する紫外線を照射した後に離型フィルムを貼り合わせることにより、実施例2の粘着フィルムを得た。
Example 2
Resin composition 1 100 parts by weight of dioctyltin diacetate (product name: Neostan U-820, manufactured by Nitto Kasei) as a curing catalyst, 0.9 part by weight of polyfunctional (meth) acrylate, neopentyl glycol diacrylate (Kyoeisha) Chemical Industry Co., Ltd., trade name Light Acrylate NP-A) 5 parts by weight, monofunctional acrylate butyl acrylate (Kyoeisha Chemical Industry Co., Ltd., trade name Light Acrylate BA) 5 parts by weight, polymerization initiator Irgacure 184 (Ciba Specialty) -Silicone composition of Example 2 was obtained by adding 0.25 weight part and Irgacure 907 (made by Ciba Specialty Chemicals, brand name) 0.25 weight part by the Chemicals company make, brand name). . Using a PET film with a thickness of 100 μm and a double-sided easy-adhesion coating as a base material, a film applicator with a micrometer was used, and the silicone resin composition of Example 1 was applied so that the resin thickness after drying was about 30 μm. The film was dried at 120 ° C. for 3 minutes, irradiated with ultraviolet rays having an energy of 300 mJ / cm 2 , and then the release film was bonded to obtain an adhesive film of Example 2.
比較例1
樹脂組成物1 100重量部に対して、硬化触媒としてジオクチル錫ジアセテート(日東化成製、商品名ネオスタンU−820)0.9重量部を添加することにより、比較例1のシリコーン樹脂組成物を得た。厚み100μm、両面易接着コーティング済みのPETフィルムを基材として、マイクロメーター付フィルムアプリケーターを使用し、乾燥後に樹脂厚みが約30μmとなるように比較例1のシリコーン樹脂組成物を塗工した。フィルムを120℃で3分間乾燥し、離型フィルムを貼り合わせることにより、比較例1の粘着フィルムを得た。
Comparative Example 1
By adding 0.9 part by weight of dioctyltin diacetate (manufactured by Nitto Kasei, trade name Neostan U-820) as a curing catalyst to 100 parts by weight of the resin composition 1, the silicone resin composition of Comparative Example 1 was obtained. Obtained. Using a PET film having a thickness of 100 μm and a double-sided easy-adhesion coating as a substrate, a film applicator with a micrometer was used, and the silicone resin composition of Comparative Example 1 was applied so that the resin thickness after drying was about 30 μm. The pressure-sensitive adhesive film of Comparative Example 1 was obtained by drying the film at 120 ° C. for 3 minutes and bonding the release film.
試験評価方法
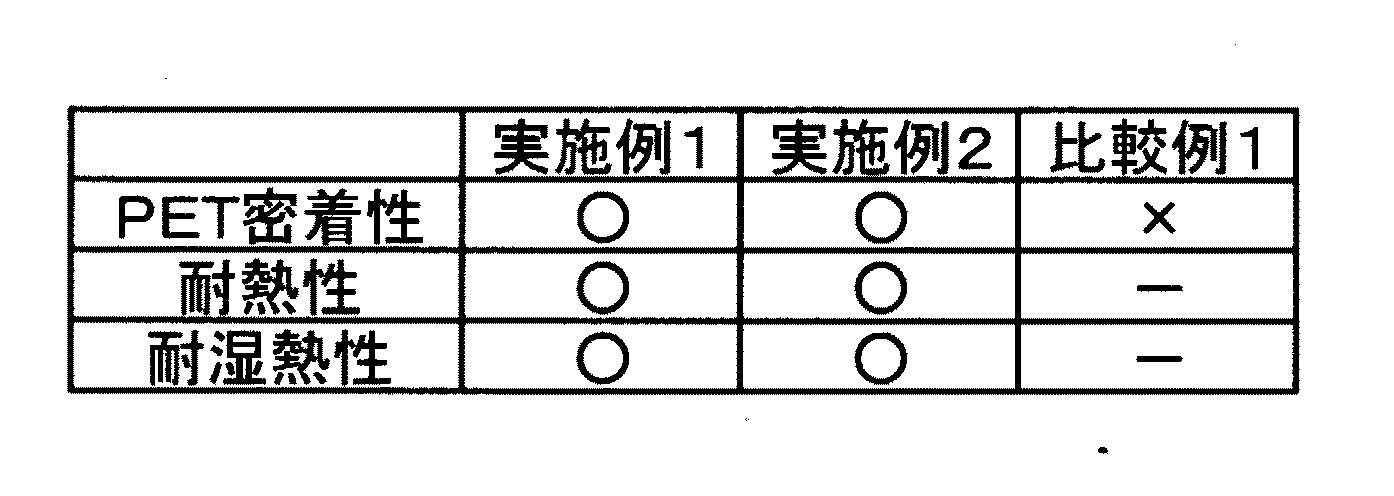
各粘着フィルムについて以下の方法で試験評価を行い、結果を表1にまとめた。
PET密着性
粘着フィルムから離型フィルムを剥がした際の粘着層の状態を確認した。
○:離型フィルムへの粘着層の付着なし、 ×:離型フィルムへの粘着層の付着あり
耐熱性
粘着フィルムを被着体として木材または鋼板(詳細)に貼り合わせ、80℃雰囲気下で7日間放置後に粘着フィルムを剥がした際の粘着層の状態を確認した。
○:被着体への粘着層の付着なし、 ×:被着体への粘着層の付着あり
耐湿熱性
粘着フィルムを木材および鋼板に貼り合わせ、60℃、90%RH雰囲気下で7日間放置後に粘着フィルムを剥がした際の粘着層の状態を確認した。
○:被着体への粘着層の付着なし、 ×:被着体への粘着層の付着あり
Test Evaluation Method Each adhesive film was subjected to test evaluation by the following method, and the results are summarized in Table 1.
PET adhesiveness The state of the adhesive layer when the release film was peeled off from the adhesive film was confirmed.
○: Adhesive layer does not adhere to release film, ×: Adhesive layer adheres to release film, heat resistance Adhesive film is bonded to wood or steel plate (details) as adherend, 7 in 80 ° C atmosphere The state of the pressure-sensitive adhesive layer when the pressure-sensitive adhesive film was peeled off after standing for a day was confirmed.
○: Adhesive layer does not adhere to adherend, ×: Adhesive layer adheres to adherend Moisture and heat resistance Adhesive film is bonded to wood and steel plate, and left to stand in an atmosphere of 60 ° C. and 90% RH for 7 days The state of the adhesive layer when peeling the adhesive film was confirmed.
○: Adhesive layer does not adhere to adherend, ×: Adhesive layer adheres to adherend
実施例の各粘着フィルムはPETフィルムへの密着性が良好であり、耐熱性、耐湿熱性とも良好であった。比較例1の粘着フィルムはPETフィルムへの密着性が劣っていた。 Each adhesive film of the examples had good adhesion to the PET film, and both heat resistance and heat and humidity resistance were good. The adhesive film of Comparative Example 1 had poor adhesion to the PET film.
Claims (2)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008247382A JP2010077286A (en) | 2008-09-26 | 2008-09-26 | Silicone resin composition and adhesive film |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008247382A JP2010077286A (en) | 2008-09-26 | 2008-09-26 | Silicone resin composition and adhesive film |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2010077286A true JP2010077286A (en) | 2010-04-08 |
Family
ID=42208090
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2008247382A Pending JP2010077286A (en) | 2008-09-26 | 2008-09-26 | Silicone resin composition and adhesive film |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP2010077286A (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9334244B2 (en) | 2010-05-12 | 2016-05-10 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9365557B2 (en) | 2008-12-19 | 2016-06-14 | Vertex Pharmaceuticals Incorporated | Substituted pyrazin-2-amines as inhibitors of ATR kinase |
| US9630956B2 (en) | 2010-05-12 | 2017-04-25 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9791456B2 (en) | 2012-10-04 | 2017-10-17 | Vertex Pharmaceuticals Incorporated | Method for measuring ATR inhibition mediated increases in DNA damage |
| US9862709B2 (en) | 2011-09-30 | 2018-01-09 | Vertex Pharmaceuticals Incorporated | Processes for making compounds useful as inhibitors of ATR kinase |
| US10813929B2 (en) | 2011-09-30 | 2020-10-27 | Vertex Pharmaceuticals Incorporated | Treating cancer with ATR inhibitors |
| CN113736087A (en) * | 2021-09-13 | 2021-12-03 | 北京天山新材料技术有限公司 | Organic silicon resin and preparation method thereof, organic silicon conductive adhesive composition and organic silicon conductive adhesive |
| US11464774B2 (en) | 2015-09-30 | 2022-10-11 | Vertex Pharmaceuticals Incorporated | Method for treating cancer using a combination of DNA damaging agents and ATR inhibitors |
-
2008
- 2008-09-26 JP JP2008247382A patent/JP2010077286A/en active Pending
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9365557B2 (en) | 2008-12-19 | 2016-06-14 | Vertex Pharmaceuticals Incorporated | Substituted pyrazin-2-amines as inhibitors of ATR kinase |
| US9701674B2 (en) | 2008-12-19 | 2017-07-11 | Vertex Pharmaceuticals Incorporated | Substituted pyrazines as ATR kinase inhibitors |
| US10961232B2 (en) | 2008-12-19 | 2021-03-30 | Vertex Pharmaceuticals Incorporated | Substituted pyrazines as ATR kinase inhibitors |
| US10479784B2 (en) | 2008-12-19 | 2019-11-19 | Vertex Pharmaceuticals Incorporated | Substituted pyrazin-2-amines as inhibitors of ATR kinase |
| US9334244B2 (en) | 2010-05-12 | 2016-05-10 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9630956B2 (en) | 2010-05-12 | 2017-04-25 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10208027B2 (en) | 2011-09-30 | 2019-02-19 | Vertex Pharmaceuticals Incorporated | Processes for preparing ATR inhibitors |
| US9862709B2 (en) | 2011-09-30 | 2018-01-09 | Vertex Pharmaceuticals Incorporated | Processes for making compounds useful as inhibitors of ATR kinase |
| US10813929B2 (en) | 2011-09-30 | 2020-10-27 | Vertex Pharmaceuticals Incorporated | Treating cancer with ATR inhibitors |
| US10822331B2 (en) | 2011-09-30 | 2020-11-03 | Vertex Pharmaceuticals Incorporated | Processes for preparing ATR inhibitors |
| US9791456B2 (en) | 2012-10-04 | 2017-10-17 | Vertex Pharmaceuticals Incorporated | Method for measuring ATR inhibition mediated increases in DNA damage |
| US11464774B2 (en) | 2015-09-30 | 2022-10-11 | Vertex Pharmaceuticals Incorporated | Method for treating cancer using a combination of DNA damaging agents and ATR inhibitors |
| CN113736087A (en) * | 2021-09-13 | 2021-12-03 | 北京天山新材料技术有限公司 | Organic silicon resin and preparation method thereof, organic silicon conductive adhesive composition and organic silicon conductive adhesive |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2010077286A (en) | Silicone resin composition and adhesive film | |
| JP5322968B2 (en) | Pressure-sensitive adhesive composition, pressure-sensitive adhesive film, method for producing pressure-sensitive adhesive composition, and method for producing pressure-sensitive adhesive film | |
| JP6479856B2 (en) | Pressure-sensitive adhesive sheet, method for producing the same, and method for producing an optical member using the same | |
| JP5841536B2 (en) | Transparent adhesive tape, film laminate with metal thin film, cover panel-touch panel module laminate, cover panel-display panel module laminate, touch panel module-display panel module laminate, and image display device | |
| CN102171305B (en) | Photocurable adhesive composition | |
| JP5715955B2 (en) | Radiation curable adhesive composition for optical member and adhesive optical member | |
| JP5859942B2 (en) | Manufacturing method of laminate | |
| TWI625340B (en) | Sclerosing composition | |
| CN109423231A (en) | Adhesive sheet and display body | |
| WO2019139055A1 (en) | Photocurable adhesive composition and adhesive sheet | |
| CN105143388A (en) | Ultraviolet curable adhesive composition and adhesive | |
| JP7387400B2 (en) | UV thermosetting adhesive composition | |
| JP7076448B2 (en) | Liquid Adhesive Compositions, Adhesive Sheets and Adhesive Methods | |
| JP6363797B2 (en) | Optical pressure-sensitive adhesive composition and optical pressure-sensitive adhesive film | |
| JP2011063701A (en) | Method for producing self-adhesive composition, method for producing self-adhesive film, raw material composition for self-adhesive, and self-adhesive film | |
| JP2015221878A (en) | Pattern electrode sheet | |
| TW202003748A (en) | Adhesive tape and method for manufacturing electronic component | |
| JP7688850B2 (en) | Pressure-sensitive adhesive composition | |
| JP2017179230A (en) | Transparent adhesive sheet for optics | |
| JP5886886B2 (en) | Adhesive film | |
| TW202007747A (en) | Adhesive composition, adhesive and adhesive sheet especially reducing a haze value under a low temperature environment and improving optical transparency | |
| TW201541188A (en) | Resin composition hardened by active energy ray, hardened object, adhesive and laminated film | |
| CN112739533B (en) | Adhesive tape and method for manufacturing electronic component | |
| JP2020196883A (en) | Double-sided adhesive sheet and laminate | |
| JP2010095699A (en) | Silicone resin composition and pressure-sensitive adhesive film |